The rate of the Pd/C catalyzed Heck coupling of Ar-I with CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-R is accelerated tenfold by the presence of Aliquat 336 (A336), a well known phase transfer catalyst, and an ionic liquid. Both when conducted in A336 as solvent, and in an isooctane/A336/aqueous triphasic mixture, the Heck reaction of aryl iodides with electron deficient olefins, catalyzed by Pd/C, proceeds with high yields and selectivity. When KOH is used instead of Et3N, selective formation of the biphenyl rather than the Heck product, is observed. Aryl bromides react more sluggishly, and only the more activated ones undergo the Heck reaction. In the absence of the olefin, aryl halides possessing an electron withdrawing group are reduced to the corresponding Ar-H.
CH-R is accelerated tenfold by the presence of Aliquat 336 (A336), a well known phase transfer catalyst, and an ionic liquid. Both when conducted in A336 as solvent, and in an isooctane/A336/aqueous triphasic mixture, the Heck reaction of aryl iodides with electron deficient olefins, catalyzed by Pd/C, proceeds with high yields and selectivity. When KOH is used instead of Et3N, selective formation of the biphenyl rather than the Heck product, is observed. Aryl bromides react more sluggishly, and only the more activated ones undergo the Heck reaction. In the absence of the olefin, aryl halides possessing an electron withdrawing group are reduced to the corresponding Ar-H.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH-R is accelerated tenfold by the presence of Aliquat 336 (A336), a well known phase transfer
CH-R is accelerated tenfold by the presence of Aliquat 336 (A336), a well known phase transfer 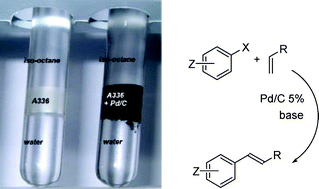

 Please wait while we load your content...
Please wait while we load your content...