Intramolecular carbolithiation reactions for the preparation of 3-alkenylpyrrolidines
Abstract
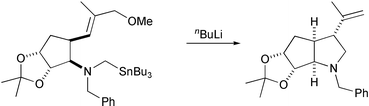
Tin–lithium exchange allows the formation of α-amino-organolithium species that undergo anionic cyclization onto allylic ethers to give 3-alkenylpyrrolidines. The methodology has been applied to the synthesis of an advanced intermediate related to the natural product (–)-α-kainic acid.

 Please wait while we load your content...
Please wait while we load your content...