Efficient synthesis of (−)- and (+)-tricyclic compounds with enone functionalities in rings A and C. A novel class of orally active anti-inflammatory and cancer chemopreventive agents
Abstract
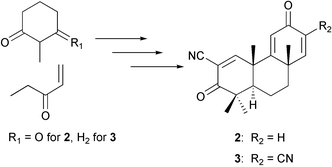
Novel tricyclic compounds with enone functionalities in rings A and C [tricyclic-bis-enone (TBE) compounds] were designed on the basis of the structure of a synthetic triterpenoid, 2-cyano-3,12-dioxooleana-1,9(11)-dien-28-oic acid (CDDO) (1), which is a promising drug candidate for prevention and/or treatment of cancer and inflammatory diseases whose pathogenesis may involve excessive production of nitric oxide (NO) and/or prostaglandins. A series of TBE compounds in racemic form shows high inhibitory activity against production of NO induced by interferon-γ (IFN-γ) in mouse macrophages. One of these compounds, (±)-(4aβ,8aβ,10aα)-1,2,4a,6,8a,9,10,10a-octahydro-1,1,4a,8a-tetramethyl-2,6-dioxophenanthrene-3,7-dicarbonitrile ((±)-3), is orally active at 15 mg kg−1 (single administration) in a preliminary in vivo study using mouse peritoneal inflammation induced by thioglycollate and IFN-γ. Therefore, we desired to synthesize optically active TBE compounds for a comparison of the biological potency of both enantiomers. We now describe the synthesis of both enantiomers of (4aβ,8aβ,10aα)-1,2,4a,6,8a,9,10,10a-octahydro-1,1,4a,8a-tetramethyl-2,6-dioxophenanthrene-3-carbonitrile (2) and 3 from commercially available simple compounds. Interestingly, (+)-3 having the same configuration as the CDDO antipode shows about 10 times higher inhibitory activity than (−)-3 on NO production in mouse macrophages. In contrast, (−)-3 inhibits proliferation of MCF-7 breast cancer cells, whereas (+)-3 does not.

 Please wait while we load your content...
Please wait while we load your content...