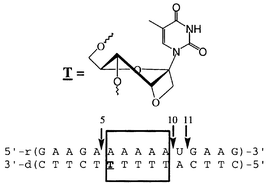
The North-form (3′-endo) constrained 1-(1′,3′-O-anhydro-β-D-psicofuranosyl)thymine block, ![[T with combining low line]](https://www.rsc.org/images/entities/b_char_0054_0332.gif) , was systematically incorporated at various sites, one at a time, into a set of four antisense oligonucleotides (AONs). The hybrids of these AONs with a matched 15mer RNA target were subjected to the RNase H cleavage reaction, and compared with that of the native counterpart, in order to probe how far the local influence of a single North-locked sugar is transmitted in steering conformational changes in the neighbouring nucleotides. It was found that the introduction of a single North-sugar locked
, was systematically incorporated at various sites, one at a time, into a set of four antisense oligonucleotides (AONs). The hybrids of these AONs with a matched 15mer RNA target were subjected to the RNase H cleavage reaction, and compared with that of the native counterpart, in order to probe how far the local influence of a single North-locked sugar is transmitted in steering conformational changes in the neighbouring nucleotides. It was found that the introduction of a single North-sugar locked ![[T with combining low line]](https://www.rsc.org/images/entities/b_char_0054_0332.gif) nucleotide in the AONs makes up to four of the neighbouring nucleotides at the 5′-end of the modification site resistant to the RNase H cleavage reaction. This suggests that a stretch of 5-nucleotides, including the
nucleotide in the AONs makes up to four of the neighbouring nucleotides at the 5′-end of the modification site resistant to the RNase H cleavage reaction. This suggests that a stretch of 5-nucleotides, including the ![[T with combining low line]](https://www.rsc.org/images/entities/b_char_0054_0332.gif) nucleotide, in the AON strand adopts a North-type conformation, giving a local RNA/RNA type hybrid structure instead of a regular DNA/RNA type duplex structure. Although these 5-nucleotide regions were completely resistant to RNase H promoted hydrolysis, they could serve as the binding site for the enzyme. Interestingly, none of these local adaptations of the RNA/RNA type structure were observable by CD spectroscopy, showing it to be an unsuitable means of monitoring any subtle alteration of the local structure. This work, therefore, constitutes an example of how the engineered conformation of a substrate can be used to exploit the stereochemical sensitivity of an enzyme to map local microscopic conformational changes. The other implication of this work is that it provides a new tool to gather local structural information, which may help to optimize the number of constrained residues which need to be incorporated to induce the antisense strand to adopt either A- or B-type geometry in the hybrid duplex, with or without the loss of RNase H recognition and/or cleavage properties.
nucleotide, in the AON strand adopts a North-type conformation, giving a local RNA/RNA type hybrid structure instead of a regular DNA/RNA type duplex structure. Although these 5-nucleotide regions were completely resistant to RNase H promoted hydrolysis, they could serve as the binding site for the enzyme. Interestingly, none of these local adaptations of the RNA/RNA type structure were observable by CD spectroscopy, showing it to be an unsuitable means of monitoring any subtle alteration of the local structure. This work, therefore, constitutes an example of how the engineered conformation of a substrate can be used to exploit the stereochemical sensitivity of an enzyme to map local microscopic conformational changes. The other implication of this work is that it provides a new tool to gather local structural information, which may help to optimize the number of constrained residues which need to be incorporated to induce the antisense strand to adopt either A- or B-type geometry in the hybrid duplex, with or without the loss of RNase H recognition and/or cleavage properties.
![[T with combining low line]](https://www.rsc.org/images/entities/b_char_0054_0332.gif) , was systematically incorporated at various sites, one at a time, into a set of four antisense
, was systematically incorporated at various sites, one at a time, into a set of four antisense ![[T with combining low line]](https://www.rsc.org/images/entities/b_char_0054_0332.gif)
![[T with combining low line]](https://www.rsc.org/images/entities/b_char_0054_0332.gif)

 Please wait while we load your content...
Please wait while we load your content...