 Open Access Article
Open Access ArticleTransformative progress in anion-exchange membranes for fuel-cell technology
Parul
Sharma
 and
Anurag Prakash
Sunda
and
Anurag Prakash
Sunda
 *
*
Department of Chemistry, J. C. Bose University of Science and Technology, YMCA, Faridabad – 121006, India. E-mail: anurag.sunda@gmail.com; anurag@jcboseust.ac.in
First published on 10th March 2026
Abstract
Anion-exchange-membrane fuel cells (AEMFCs) have gained significant attention in recent years due to their utilization of inexpensive metals. However, the conductivity of anion-exchange membranes in fuel cells still falls short when compared to the extensively researched proton-exchange-membrane fuel cells (PEMFC). Researchers at large are actively working towards the advancement of anion-exchange membranes that possess superior ionic conductivity, alkaline resistance, and mechanical stability. This work attempts to comprehensively review the recent transformative progress in attaining efficient anion-exchange membranes through both experimentation and computational approaches. The review delves into different aspects of anion-exchange-membrane performance, such as polymer structure, pendant group modifications, morphological traits, water management, etc., to ensure high ionic conductivity and alkaline stability. Additionally, the review explores diverse strategies to protect the cationic functional groups to address the concern of deterioration. To understand the role of pivotal factors, insights are provided into the design of anion-exchange membranes with emphasis on high conductivity, enriched by summarizing recent advancements, including molecular studies. The alkaline stability and hydration control of anion-exchange membranes are key parameters to focus on for exceptional performance potential in forthcoming applications.
1 Introduction
One of the biggest challenges in today's world is to find a balance between energy production and environmental pollution. The use of fossil fuels for generating energy or electricity is one of the leading causes of increasing environmental pollution. Fossil fuels liberate huge amounts of greenhouse gases, which adversely affect the environment and human health. Therefore, the use of renewable sources for the generation of energy or electricity has grown tremendously over the years, with improvement in the efficiency of solar cells, fuel cells, and super-capacitors. The best alternative to fossil fuels for generating energy is fuel cells.1–3 Fuel cells convert chemical energy into electrical energy with minimal emission of pollutants.4 Fuel cells generate zero noise pollution and possess high efficiency.5,6 There are several types of fuel cells that operate under different conditions and use different types of materials, for example the proton-exchange-membrane fuel cell (PEMFC),7,8 alkaline fuel cell (AFC),9 anion-exchange-membrane fuel cell (AEMFC),10 direct alcohol fuel cell (DAFC),11 molten carbonate fuel cell (MCFC),12 solid oxide fuel cell (SOFC),13 and phosphoric acid fuel cell (PAFC).14 All fuel cells consist of a cathode, anode, electrolyte, and fuel. At the anode, fuel is oxidized and releases electrons. At the cathode, air (oxidant) reacts with electrons and produces reduced species. Different fuel cells utilize different types of electrolytes and fuels.15–18Among polymer-electrolyte-based fuel cells, the use of ion-exchange membranes as electrolytes in fuel cells has attracted tremendous attention. Ion-exchange membranes as electrolytes in fuel cells possess several advantages, such as they can operate at low temperatures, possess low corrosion risks, and are safe and easy to handle compared to liquid electrolytes. Ion-exchange membranes as electrolytes in fuel cells are classified as either proton-exchange membranes or AEMs, which result in the generation of either acidic or alkaline environments, respectively. Proton-exchange membranes allow protons to pass through selectively while preventing the passage of anions, whereas anion-exchange membranes perform the reverse action.20 A schematic representation of various components of an AEM fuel cell is shown in Fig. 1. The comprehensive multiscale review by Lei and co-workers21 primarily centred on electrochemical mechanisms in alkaline media, with particular emphasis on the hydrogen oxidation reaction (HOR), oxygen reduction reaction (ORR), catalyst design, and electrode-level processes. Its major strength lies in correlating reaction kinetics with electrocatalyst behavior across multiple scales.
The review of Yassin and Dekel22 provided a broad, cross-cutting analysis of AEM-based technologies, including fuel cells, water electrolyzers, redox flow batteries, CO2 electrolysis systems, and gas separation platforms. While such wide-ranging scope was valuable for identifying common challenges across different electrochemical devices, it does not offer an in-depth, fuel-cell-specific membrane analysis. The recent review on membranes by Hsiao and co-workers23 mainly focuses on comparative analyses of PEM and AEM water electrolyzers, alkaline-stability enhancement strategies, and patent-related developments.
In contrast to these prior works, our review is specifically centred on AEMs for fuel-cell applications, with a strong emphasis on membrane molecular design, polymer architecture engineering, and transport mechanisms. We provide a detailed mechanistic discussion of hydroxide ion transport, clarifying the interplay between vehicular and Grotthuss-type mechanisms under varying hydration conditions. Importantly, we include a separate and structured section dedicated to AEM synthesis methodologies, rational membrane design and fabrication. Furthermore, we present a systematic evaluation of recent strategies to enhance AEM performance, highlighting multiple emerging and innovative directions reported in the literature. A distinctive strength of our review is the dedicated integration of computational modelling approaches alongside experimental advancements, thereby bridging theory-guided design with practical material development. This review mainly puts emphasis on AEMs for alkaline anion-exchange-membrane fuel cells. The present work discusses the essential design aspects of anion-exchange membranes (AEMs) in relation to applications, specifically tailoring them to meet the demands of future energy in terms of stability, performance and durability.
2 Anion-exchange-membrane fuel cells
NASA used an alkaline fuel cell in the Apollo series missions and on the space shuttle. The world's first fuel-cell ship, ‘The Hydra', used an alkaline fuel-cell system.24 In these alkaline fuel-cell systems, liquid KOH electrolyte was used, which corrodes the electrode by forming K2CO3 on reaction with CO2 present in the air.10 To address this challenge, researchers opted for an anion-exchange membrane in place of a liquid electrolyte. In an AEM, the carbonate content is very low compared to a liquid electrolyte; hence, corrosion of the electrode does not occur. An AEMFC consists of a membrane electrode assembly (MEA) where the polymer electrolyte AEM connects a cathode and an anode via an active catalyst layer. Oxidation of hydrogen gas occurs at the anode and reduction of oxygen gas occurs at the cathode. The following reaction occurs at the cathode and anode in the fuel cell for electricity generation.19| At the anode: H2 + 2OH− → 2H2O + 2e− |
| At the cathode: 1/2O2 + H2O + 2e− → 2OH− |
| Overall cell reaction: H2 + 1/2O2 → H2O |
AEMFCs have numerous advantages over PEMFCs, as summarized in Table 1. An AEMFC operates in an alkaline medium; therefore, the oxygen reduction reaction happens more effectively and easily compared to in a PEMFC.25 In a PEMFC, highly expensive metal catalyst electrodes (e.g., platinum) are used, which limits their usage at the industrial level, whereas an AEMFC can work with a low-cost non-platinum metal catalyst electrode (e.g., silver, transition metal oxides). In the past few years, alkaline AEMFCs have gained popularity and become a hot research topic26,27 because an increase in ionic conductivity up to 200 mS cm−1 has been recorded by several researchers.28
| Advantages | Disadvantages |
|---|---|
| (1) Tolerance to CO poisoning | (1) Membrane degradation |
| (2) Non-precious metal catalysts | (2) Limited commercial availability |
| (3) Broader fuel flexibility | (3) Challenging to attain high hydroxide conductivity |
| (4) Environment friendly | (4) Limited operating temperature range |
3 Anion-exchange membranes: role and performance
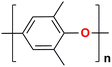
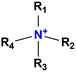
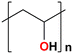
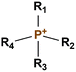
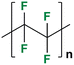
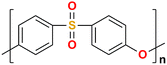
The AEM is a key component in an AEMFC because it is responsible for the transportation of hydroxide ions from the cathode to the anode.29 An anion-exchange membrane is made up of a hydrophobic polymer backbone attached to a hydrophilic cationic head, in which the cationic head is responsible for the transportation of hydroxide ions from the cathode to the anode.30 We can use poly(arylene ether), poly(2,6-dimethyl-1,4-phenylene oxide), poly(vinyl alcohol), poly(tetrafluoroethylene), etc. as the polymer backbone and quaternary ammonium, imidazolium and guanidinium ions, etc. as the cationic head groups for the AEM (see Table 2). Fumasep® FAA3, Tokuyama A201, AemionTM, Sustainion® 37–50, Durion TM1, and PiperION are some of the commercially available AEMs.31,32 Commercially available AEMs face several limitations that impede their widespread adoption in various applications, for example limited alkaline stability, lower conductivity, water management (which presents another challenge as AEMs require careful hydration control), mechanical instability, etc. These obstacles have driven researchers to develop membranes capable of surpassing current constraints to enhance the performance of electrochemical devices such as fuel cells and water electrolysers.33 Wang et al.34 showed ion transport enhancement with the help of bromide ions, using poly(bromo butylnorbornene) homopolymer AEMs. The authors found that water percolation greatly relied on the local hydrated structure of the alkaline membrane. Du et al.35 presented emerging concepts related to anion-exchange membranes as water electrolysers and highlighted the consideration of cross-linking or use of composites in AEMs to enhance alkaline stability.| Polymer backbone units | Name of polymer | Ref. | Cationic head groups | Group name | Ref. |
|---|---|---|---|---|---|
|
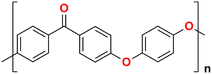
Polyphenyleneoxide | 36 |
|
Quaternary ammonium | 37 |
|
Polyvinylalcohol | 38 |
|
Phosphonium | 39 |
|
Polytetrafloroethylene | 40 |
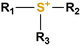
|
Sulfonium | 41 |
|
Polyethersulfone | 42 |
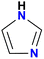
|
Imidazole | 43 |
|
Polyetheretherketone | 44 |

|
Pyrrolidine | 45 |
|
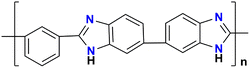
Polybenzimidazole | 46 |
|
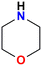
Morpholine | 47 |

|
Piperidine | 48 |
3.1 Application of anion-exchange membranes in fuel cells
PEMFCs have been successfully adopted in fuel-cell electric vehicles, with commercial models introduced by Toyota Motor Corporation and Hyundai Motor Company, and General Motors confirming the practical viability of fuel cells in transportation. Despite these advancements, large-scale deployment of PEMFC technology remains restricted due to the reliance on costly platinum metal catalysts, durability concerns associated with acidic membranes, and the need for highly corrosion-resistant components.3,49,50 In response to these limitations, AEMFCs have emerged as a promising alternative. Their operation in an alkaline medium allows the utilization of non-precious metal catalysts and more economically viable materials, thereby significantly reducing system cost.Accordingly, AEMFCs are gaining considerable research interest as a viable alternative for next-generation energy conversion systems. To evaluate their practical feasibility and performance potential, extensive research efforts have focused on the fabrication and single-cell testing of AEMFCs under optimized operating conditions. For instance, a series of polystyrene-block-polybutadiene-block-polystyrene (SBS) triblock copolymers functionalized with pyridinium groups (SBS-QA+py) were synthesized and evaluated in alkaline single-cell fuel cells by Anley et al.51 Within this series, SBS-Qdpy1 and SBS-Qdpy2 emerged as the most promising candidates due to their optimized balance between ion exchange capacity (IEC) and mechanical strength. Single-cell testing under fully humidified H2/O2 conditions (100% RH) at 80 °C and ambient pressure revealed peak power densities of 362.2 mW cm−2 for SBS-Qdpy1 and 398.1 mW cm−2 for SBS-Qdpy2. These values exceeded the performance of the commercial Sustainion X37-50T (347 mW cm−2 under identical conditions) and were higher than that of PiperION-A20 (355–377 mW cm−2 under similar conditions with minor differences in backpressure).
A series of novel poly(fluorenyl aryl piperidinium) (PFAP) copolymers were designed by Chen and group52 for both AEMs and anion-exchange ionomers (AEIs) to enhance AEMFC performance. The materials were systematically evaluated through single-cell H2/O2 fuel-cell testing under controlled temperature, humidity, and backpressure conditions to assess their practical applicability. Using PFBP-14 as the AEI and PFTP-13 as the AEM at 80 °C and 75%/100% RH, the cell delivered a peak power density of 1.67 W cm−2 without backpressure, which further increased to 2.34 W cm−1 under 1.3/1.3 bar backpressure. In contrast, commercial Fumatech-based AEMFCs typically achieve peak power densities of only 0.5 W cm−2, highlighting the significant advancement offered by the PFAP-based system. In order to evaluate the effect of different amounts of hydrophobic fluorinated groups, PTFx-QAPTP AEMs were synthesized by Yu et al.53 To assess their practical performance, single-cell fuel-cell tests were conducted. The PTF6-QAPTP membrane achieved a peak power density of 849 mW cm−2 at a current density of 1600 mA cm−2, while PTF3-QAPTP delivered 571 mW cm−2. The better performance of PTF6-QAPTP is likely due to its more distinct microphase separation, which leads to the formation of continuous and interconnected hydrophilic pathways.
3.2 Mechanism of hydroxide ion transport
Transportation of hydroxide ions occurs via AEMs involving different mechanisms, e.g., the Grotthuss mechanism, surface site hopping, convection, and diffusion. An illustration of these mechanism around the AEM is presented in Fig. 2. In the Grotthuss mechanism, hydroxide ion transportation occurs by simultaneous breakage and formation of hydrogen bonds with water molecules. In surface site hopping, the surface site refers to the cationic group attached to the polymer backbone, and hydroxide ions hop from one cationic group to another present in the anion-exchange membrane.54 Diffusion occurs due to electroosmotic drag, where OH− ions interact with water molecules and migrate across the membrane. The pressure gradient drives the convection transport of OH− anions via the AEM membrane. As the OH− ions move they carry water molecules with them through the membrane.10,55 Enormous efforts have been made to get insights into ion transport in polymer electrolytes using different techniques. For example, Foglia et al.56 used quasi-elastic neutron scattering to unveil characteristics of the ion dynamics in polymeric membranes, using the commercially available Fumatech FAD-55 membrane.3.3 Performance of anion-exchange membranes
A good AEM should possess high hydroxide conductivity, and excellent thermal, chemical, and mechanical stability. In an anion-exchange membrane, the polymer backbone and the attached cation both contribute significantly to hydroxide conductivity and the stability of the membrane.57 The performance of an AEM in an alkaline AEMFC depends on certain factors, such as ion exchange capacity, water uptake, alkaline stability, and the morphology of the membrane.58 All these factors contribute significantly to hydroxide conductivity and membrane stability. In the last decade, a lot of research has been carried out on AEMs to attain hydroxide conductivity comparable to that of PEMFCs.For water uptake measurements, the process involves several steps. First, the membrane samples are converted to the hydroxide form. Following that, they are dried until a consistent mass is achieved and then weighed, while the length of the membrane is measured. Next, these dried membrane samples are placed in a sealed container filled with water, typically for a period of 24 hours. After the immersion period, any excess surface water on the membranes is quickly removed by wiping with tissue paper. Immediately after this, the membranes are weighed and their dimensions are measured. The collected data from these steps are then used to calculate two important parameters: water uptake (WU) and swelling ratio (SR). In essence, this process assesses how much water the membrane can absorb and how much it swells when submerged, providing valuable information about its water-handling characteristics.10 Zheng et al.72 investigated fluorinated and hydrocarbon-based AEMs to explore role of water uptake and its kinetics. The authors found a characteristic time for water uptake for AEMs that varies with the polymer backbone, membrane structure, temperature and humidity level. Xu et al.73 demonstrated the importance of anisotropic swelling with hydration of the AEM, which facilitates ion transport effectively by filling the gap between the interface of the catalyst layer and the membrane.
The Dekel and Diesendruck group have put immense efforts into systemically examining the alkaline stability of AEMs, specifically targeting quaternary ammonium cation heads with various polymer backbones.83–87 The kinetic stability of quaternary ammonium cations is greatly influenced by membrane hydration and low hydration is less favourable for prolonged stability of the membrane. The authors suggested that the isoindolinium group can be a very good alternative for replacement of quaternary ammonium cation heads at low hydration or under dried conditions.88
A well-planned molecular design to enhance the alkaline durability of AEMs was introduced by Wang and team.89 Rather than altering the polymer backbone or introducing a new cationic headgroup, the authors showed that a simple positional isomerization of the piperidinium linkage can significantly improve membrane durability. Notably, both conventional PAP and the isomeric i-PAP share the same ether-free arylene backbone and identical piperidinium cations; the only structural difference lies in the attachment position of the nitrogen atom. This subtle yet important modification alters the local molecular geometry around the cationic center. As a result, the dominant Hofmann elimination pathway is significantly suppressed, leading to enhanced alkaline stability without compromising the original backbone or polymer structure. After immersion in 10 M aqueous NaOH at 80 °C for 360 h, the decomposition of piperidinium groups in the isomeric i-PAP membrane was approximately half of that observed in the conventional PAP membrane.
Another approach by Liu and coworkers90 reported the development of poly(diphenyl carbazole)-based AEMs incorporating multi-cationic alkoxy side chains. By precisely tuning the alkoxy chain in terms of carbazole content, the authors reported ordered ion transport channels through multi-cation dipole interactions, achieving high hydroxide conductivity of 154.6 mS cm−1 at 80 °C. Importantly, the introduction of an alkoxy chain (QPDPC-O-20) rather than only an alkyl chain (QPDPC-C-20) enhanced alkaline stability by promoting stronger intermolecular interactions and better water regulation, leading to 98.3% conductivity retention after 2000 h in 1 M KOH. However, after 600 hours in an alkaline solution of 5 M KOH, the QPDPC-O-20 membrane was able to maintain about 71.4% of its original conductivity, while the QPDPC-C-20 membrane retained 63.2%, indicating better stability of the alkoxy-modified membrane.
Wang et al.91 investigated poly(biphenylene alkylene)-based AEMs, PBPAnQA (n = 4, 5, 6), where n represents the alkylene spacer length between the backbone and the quaternary ammonium groups. The main objective was to systematically investigate how spacer length influences alkaline stability. The membranes were immersed in 5 M NaOH at 80 °C for 1000 h. Changes in chloride conductivity at 80 °C and structural variations from 1H NMR spectra were analyzed before and after alkaline treatment. After 1000 h aging, PBPA6QA (with a hexylene spacer) showed a conductivity decrease of only 21%, from 42.5 to 33.4 mS cm−1. In contrast, PBPA5QA and PBPA4QA exhibited higher conductivity losses of 35% and 46%, respectively. This trend was consistent with the extent of QA cation degradation: 18.3% for PBPA6QA, 22.7% for PBPA5QA, and 35.2% for PBPA4QA. The results clearly demonstrate that increasing the alkylene spacer length enhances alkaline stability.
Researchers have been actively conducting extensive studies at both experimental and theoretical levels to preserve specific properties and develop a suitable AEM. Substituted imidazolium groups generally exhibit higher alkaline stability than unsubstituted imidazolium due to reduced proton abstraction and improved steric protection. In piperidinium-based AEMs, N-methyl substitution at the 3-position results in superior alkaline stability compared to substitution at the 4-position, as reported in the work of Peng et al.96 These efforts aim to enhance hydroxide conductivity while ensuring the membrane's chemical and mechanical stability, along with balancing the IEC and water uptake. Motealleh and co-workers97 evaluated the durability of an imidazolium-functionalized AEM based on styrene (Sustainion) polymer and found a stable performance of 1 A cm−2 at 1.85 V for potential commercial application.
4 Synthesis methods of anion-exchange membranes
4.1 Solution-casting method
The most promising conventional method for production of AEMs is the solution-casting method (see Fig. 3). This process has four steps: dissolving the polymer, adding a functional group by chloromethylation, quaternizing it and casting it as a film. This method applies to soluble polymers, copolymers and their blends. The procedure involves dissolving the chosen polymer in a compatible solvent to form a uniform solution. Then the cation functional group is introduced via chloromethylation. This solution is subsequently spread or cast evenly onto an appropriate surface. As the solvent gradually evaporates, the polymer material solidifies, leading to the formation of a thin membrane.98 Samsudin et al.99 designed a composite AEM applying the solution-casting method using poly(vinyl alcohol) (PVA), poly(diallyldimethylammonium chloride) (PDDA), and nano-zirconia (NZ). The designed membrane showed OH− conductivity of 31.6 mS cm−1 at ambient temperature. The incorporation of NZ results in a low water uptake and swelling ratio. Wang et al.100 used the solution-casting method in their synthesis process for membranes. The authors used a triple block copolymer, poly(styrene-b-(ethylene-co-butylene)-b-styrene), and quaternary ammonium functional groups. The designed membrane showed an OH− conductivity of 72.13 mS cm−1, swelling ratio of 9.3% and current density of 350 mA cm−1 at 80 °C.4.2 Radiation grafting
Radiation grafting involves grafting of the functional groups onto the polymer membrane using radiation such as gamma rays or electron beams. This process starts with a pre-existing polymer substrate. The polymer substrate is then exposed to ionizing radiation, which generates radicals within the polymer matrix. The generated radicals initiate a grafting reaction between the polymer matrix and the selected monomer functional groups, leading to the integration of functional groups.3,98 Wang et al.101 reported hydroxide ion conductivity of 200 mS cm−1 at 80 °C using a 30 µm thick LDPE (low-density polyethylene)-based radiation-grafted anion-exchange membrane (RG-AEM). Biancolli et al.102 designed a radiation-grafted ETFE-based AEM and analysed the effect of irradiation conditions on the membrane. It was observed that subjecting the membrane to ionizing radiation doses below 40 kGy, while maintaining an inert N2 atmosphere apart from air, resulted in improved stability for the polymer backbone and enhanced alkaline resistance of the membrane. Biancolli and coworkers103 illustrated the use of radiation and tailored thickness of the polymer film to improve the ion-exchange capacity of AEMs.4.3 Polymerization
Polymerization processes are divided into two main categories: direct polymerization (DP) and post-polymerization functionalization (PPF). In direct polymerization, cationic monomers are created and then subjected to the polymerization process. Conversely, in PPF, cationic functional groups are introduced onto pre-formed polymer backbones.104 There are several types of DP methods, which include ring-opening-metathesis polymerization (ROMP) and radical polymerization. The ROMP method105 is used to polymerize cyclic alkenes. It involves the opening of the ring structure to form linear polymer chains, catalyzed by Ru complexes known as Grubbs' catalysts.106,107 Radical polymerization involves generation of radicals to initiate polymerization reactions. Peroxides or azo compounds are used to generate free radicals. Singh and coworkers108 developed an innovative di-quaternized cross-linked AEM using 1,4-bis(4-vinylbenzyl)-1,4-diazabicyclo[2,2,2]octane-1,4-diium (BVDOD) as a cross-linking agent and poly(vinylidene fluoride-co-hexafluoro propylene) (DHPVDF-co-HFP) as the polymer backbone through the radical polymerization method. Azobisisobutyronitrile (AIBN) was employed by the researchers as an initiator for free radicals. The newly created cross-linked AEM demonstrated an ion exchange capacity of 1.02 × 10−3 equivalents per gram and a hydroxide ion conductivity of 57 mS cm−1 at a temperature of 30 °C. All the membrane preparation methods are illustrated through pictorial representations in Fig. 3.5 Recent progress in anion-exchange membranes
Researchers have implemented various strategies to address issues such as the deterioration of the cationic head and the instability of the polymer's backbone. They have focused on enhancing the conductivity of hydroxide ions and achieving a well-defined morphology. These efforts have involved the creation of polymers with distinct structures, including block, comb-shaped, graft, and cross-linked polymers that incorporate cations (see Fig. 4). These structural modifications have led to the formation of micro-phase separation structures, resulting in improved conductivity, reduced swelling effects, and enhanced stability in an alkaline environment. The key performance-enhancement strategies can be identified as follows:• Molecular structure engineering of cationic groups
• Polymer backbone optimization
• Crosslinking and network design
• Hydrogen-bonding network construction
• Branched and hyperbranched polymer architectures
• Grafting and side-chain engineering approaches
 | ||
| Fig. 4 A schematic representation of different types of polymers from AEMs (recreated from Xu, Su and Lin,64 copyright 2020). | ||
These strategies place emphasis on controlled phase separation, ion-channel formation, mechanical reinforcement, and multiple hydrogen-bonding networks to improve dimensional stability, water retention, and ion transport efficiency. The emerging design dimensions in AEM development demonstrate performance competitiveness with proton-exchange membranes,109 supporting a rational approach to membrane engineering.
Recent research has focused on creating multiple hydrogen-bonding sites within the polymer structure to facilitate efficient OH ion transport and enhance the overall performance of the AEM. In this context, Wang and group110 developed an AEM based on a poly(oxindole benzofuran) backbone incorporating quaternary ammonium functional groups for efficient OH ion transport. In addition to the oxindole moieties, hydroxyl (–OH) groups were introduced into the side chains to promote multiple hydrogen-bonding interactions. The P(O-F50%-C50%)-GTA membrane demonstrated a high hydroxide ionic conductivity of 103 mS cm−1 at 80 °C. The membrane maintained acceptable mechanical strength (8.6 MPa). The ex situ alkaline stability tests revealed that the membrane retained more than 80% of its initial conductivity after 600 h.
Peng et al.111 studied a poly(terphenyl)-based AEM functionalized with quaternary ammonium groups along with incorporation of heteroaromatic units (e.g., thiophene/furan units). The results indicated that introduction of thiophene units into the polymer backbone significantly enhanced membrane performance with QP(TP30-co-DBT70) showing a hydroxide conductivity of 120 mS cm−1 at 80 °C, mechanical strength of 26.6 MPa, and a relatively low volume swelling ratio of 34.6%.
In the study conducted by Li and co-workers,114 an AEM was developed using polyphenylene oxide (PPO) that featured a comb-like structure. The cationic heads consisted of quaternary ammonium and were linked to a lengthy alkyl chain. This substantially decreased the swelling ratio and decreased the probability of nucleophilic attack of hydroxide ions on the cationic head. Zhang et al.115 developed a bent twisted block copolymer with 1,2-dimethyl imidazolium cations and attained a hydroxide conductivity of 35 mS cm−1 at room temperature. The fuel cell employed with this membrane showed a peak power density of 262 mW cm−2 at 50 °C. Chen et al.116 created a graft polymer using perfluorinated poly(diphenylene) with the alkoxy side chain of a piperidinium quaternary ammonium salt. A well-defined microphase-separated structure was found that improved the ionic conductivity. Their group analyzed the membrane by taking different lengths of side chains and witnessed an ionic conductivity of 80 mS cm−1 at 80 °C; the swelling ratio was also low at approximately 17.1%, and the power density was about 1.008 W cm−2 for the long-side-chain polymer. Wang and team117 crafted a branched poly(p-terphenyl triphenylmethane 1-methyl piperidine) anion-exchange membrane and reported a high hydroxide conductivity and alkaline stability. To boost conductivity, the branched membrane was crosslinked using multiple cations. The cross-linked version showed a tremendous increase in hydroxide conductivity of about 155.3 mS cm−1 at 80 °C. Wu et al.118 created an anion-exchange membrane composed of branched poly(aryl piperidinium). The synthesized membrane showed minimal water absorption and swelling. The research team discovered that this branched membrane displayed outstanding resistance to alkaline conditions and demonstrated a commendable hydroxide conductivity of approximately 145 mS cm−1 at 80 °C. Lee et al.119 produced an anion-exchange membrane utilizing Nafion and Aquivion-based perfluorinated precursors with trimethyl ammonium (TMA) cations. The authors conducted a comparison of properties like conductivity, morphology, and alkaline stability between Nafion and Aquivion-based AEMs and a commercial hydrocarbon-based Fumatech ionomer. The results indicated that all three membranes exhibited a phase-separated structure. The perfluorinated membrane displayed more pronounced phase separation due to its exceptionally high hydrophobic characteristics. Furthermore, the impact of relative humidity on membrane conductivity showed that perfluorinated membranes consistently exhibited superior conductivity at all humidity levels when compared with commercial Fumatech membranes. Lai et al.112 synthesized an AEM using block polymer poly(arylene ether nitrile ketone) (QFPENK-m-n) as a main chain, combining it with comb-shaped alkyl side chains. The morphology of QFPENK-m-n with varying m and n values is shown in the Fig. 5a–f. The AEM showed a well-defined phase-separated microphase morphology and hydroxide conductivity up to 34.3 mS cm−1 at 30 °C and 102.1 mS cm−1 at 80 °C. It was also observed that an increase in the hydrophobic segment resulted in a reduction in both water uptake and the percentage SR (see Fig. 5g).
 | ||
| Fig. 5 (a) Digital photo of the membrane, and (b) membrane cross-section and (c) surface SEM images of QFPESK-10-10; AFM phase images of (d)–(f) QFPESK-5-10, QFPESK-10-10 and QFPESK-15-10, respectively; (g) water uptake and swelling ratio of the QFPENK-m-n AEMs at different temperatures. This figure has been adapted/reproduced from Lai et al.112 with permission from John Wiley & Sons, Inc., copyright 2024. | ||
Huang et al.113 designed an AEM using poly(p-terphenyl-co-1,2-diphenyl ethane piperidinium) (PDTP). To produce a high-performance AEM named ‘PDTP-BMP-xC', their procedure included the crosslinking of the PDTP backbone, wherein the variable ‘x' denotes the extent of the crosslinker's length. It was observed that PDTP-BMP-6C exhibited a conductivity of 104.61 mS cm−1 when measured at 80 °C. Additionally, its power density at the same temperature reached 214 mW cm−2. The micro-phase morphology structure for the crosslinked membrane PDTP-BMP-xC can be seen in the Fig. 6.
 | ||
| Fig. 6 (a)–(c) Digital photos of the PDTP, PDTP-BMP-3C and PDTP-BMP-6C membranes immersed in DMSO; (d)–(f) TEM images of the membranes. This figure has been adapted/reproduced from Huang et al.113 with permission from Elsevier, copyright 2023. | ||
Chen et al.121 designed a poly(crown ether) (PCE)-based AEM-QAPCE-xC, where x represents the length of the alkyl chain. A quaternary ammonium cationic side chain was employed, and the PCE backbone was crosslinked using various alkyl chain lengths. Reportedly, QAPCE-16C exhibited an impressive ionic conductivity of 125 mS cm−1 at 80 °C. Furthermore, QAPCE-16C demonstrated favorable dimensional stability, as well as robust mechanical and thermal durability. Mandal et al.122 investigated the role of homopolymer, random and block polymerization in poly(norbornene) AEMs. The authors found that block-polymer performance had an edge over the random polymer structure, with a power density of 3.21 W cm−2. Caielli and team123 achieved a remarkable result in hydroxide conductivity, reaching 185 mS cm−1 at a temperature of 80 °C. This was accomplished through the creation of a membrane utilizing poly(aryl piperidinium) (PAP). Tao Wang and coworkers124 documented an extraordinarily elevated hydroxide conductivity of 227.8 mS cm−1 at 80 °C, employing an AEM based on poly(arylene alkylene). Their research group created a membrane composed of a poly(q-tetraphenylene-co-p-terphenylene alkylene) rigid backbone, integrating the quaternary ammonium cationic functional group. Wu and co-workers125 pursued a method for creating a notably effective AEM by applying a fluorination technique to the structure of poly(aryl piperidinium) (PAP). As a result, the fluorinated anion-exchange membrane (FPAP) displayed exceptional dimensional stability with a SR below 20 percent. The membrane exhibited remarkable mechanical characteristics, and the researchers observed tremendously high conductivity exceeding 150 mS cm−1 at 80 °C.
Zhang and their research team126 utilized covalent organic frameworks (COFs) to create an AEM. They synthesized a composite AEM by combining COF-DhaTGCl with PPO. Initially, they prepared a few-layered nanosheet structure using COF-DhaTGCl, and then introduced quaternary ammonium-functionalized PPO into these nanosheets. Their findings showed that at a doping level of 20 wt%, the conductivity of OH− ions reached 148.65 mS cm−1 at 80 °C, while at a doping level of 30%, the conductivity was measured at 136.52 mS cm−1 at the same temperature. The reason for this decrease in conductivity can be attributed to the aggregation of COF-DhaTGCl nanosheets within the membrane, which hinders the internal conduction pathways. Ju et al.127 conducted synthesis of an AEM based on poly(arylene ether ketone). They achieved this by employing Friedel–Crafts acylation, followed by the introduction of imidazole cationic groups through a bromination reaction. Their work yielded impressive results, with the AEM displaying its highest hydroxide conductivity of 126.6 mS cm−1 at an operating temperature of 80 °C. Additionally, they observed a peak power density of 389.71 mW cm−2. A partially fluorinated membrane (PFBE-co-VBC) with quaternary ammonium functional groups was successfully created by Hosimin and co-workers.120 The researchers also created a blended AEM by combining PFBE-co-VBC with PVDF. Analysis using FE-SEM revealed an interconnected network structure in the AEM (see Fig. 7). The blended membrane, with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio, demonstrated an ion conductivity of 1.0 × 10−2 S cm−1 and an ion-exchange capacity of 1.7 mmol g−1.
1 ratio, demonstrated an ion conductivity of 1.0 × 10−2 S cm−1 and an ion-exchange capacity of 1.7 mmol g−1.
 | ||
Fig. 7 FE-SEM cross-sectional images of a blended anion-exchange membrane (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.33). This figure has been adapted/reproduced from Hosimin et al.120 with permission from the Royal Society of Chemistry, copyright 2023. 0.33). This figure has been adapted/reproduced from Hosimin et al.120 with permission from the Royal Society of Chemistry, copyright 2023. | ||
Arunkumar and team128 devised a method to enhance the properties of the commercially available FAA3 AEM. They achieved this by incorporating carboxylic-acid-functionalized graphene nanofibers into the FAA3 matrix. Introducing these graphene nanofibers significantly improved the electrochemical performance and chemical stability of the FAA3 membrane. The original FAA3 membrane had a hydroxide conductivity of 28.7 mS cm−1, whereas the nanocomposite membrane with 1.70 wt% of nanofibers showed an enhanced conductivity of 58.8 mS cm−1 at 90 °C. Lee et al.129 created various PPO-based AEMs with quaternary ammonium groups having different side-chain lengths. Their observations revealed that when the lengths were similar, the crosslinked membranes demonstrated superior alkaline stability in comparison to their non-crosslinked counterparts. Specifically, the crosslinked cQPH membrane with a hexyl acyl chain exhibited an anion conductivity of 105 mS cm−1 at 80 °C due to effective phase separation. The various attempts in the recent development of novel AEMs and their performance are provided in Table 3.
| Polymer backbone with pendant subgroups | OH− conductivity (mS cm−1) | IEC (mmol g−1 | Water uptake (%) | Tensile strength (MPa) | λ | Ref. |
|---|---|---|---|---|---|---|
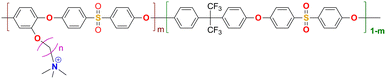
|
62.7 at 80 °C | 1.48 | 77.5 | 9.2 ± 1.9 | 29.1 | Lin et al.132 |
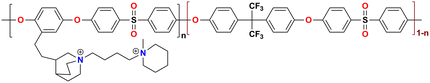
|
88 at 80 °C | 1.35 | 53.83 | 47 | 23 | Patil et al.133 |
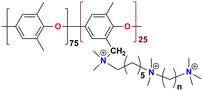
|
99 at 80 °C | 2.87 | 189 ± 5 | — | 34 | Zhu et al.134 |
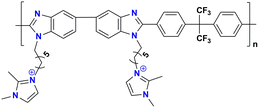
|
63.4 at 80 °C | 2.09 | 36.9 ± 3.7 | 13 ± 1.4 | — | Lin et al.135 |
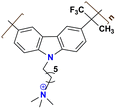
|
125 at 70 °C | 2.31 | 76 | — | — | Cha et al.136 |
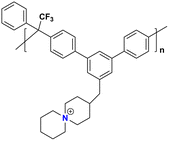
|
51 at 80 °C | 1.57 | 54 | — | 17 | Pham et al.137 |
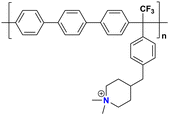
|
146 at 80 °C | 1.73 | 103 | — | 30 | Pham et al.137 |
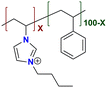
|
89.7 at 60 °C | 1.65 | 101 ± 5.2 | 15.5 | 34 | Park et al.138 |
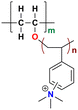
|
145 at 80 °C | 2.87 | 104 ± 9 | 30 | 20 ± 2 | Wang et al.139 |
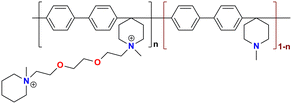
|
106 at 80 °C | 1.93 | 46.1 | 7.4 | — | Zhang et al.140 |
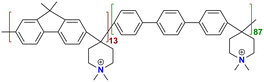
|
163 at 80 °C | 2.82 | 73 | 71 | — | Chen et al.141 |
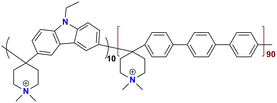
|
204.8 at 90 °C | 2.76 | 54.9 | 52.2 ± 1.6 | — | Yuan et al.142 |
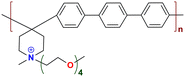
|
104.2 at 80 °C | 1.47 | 101 | 60 | — | Liu et al.143 |
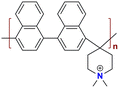
|
135.25 at 80 °C | 2.29–2.60 | 38.5 | 35 | 3.48–8.25 | Gao et al.144 |
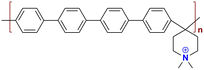
|
109.12 at 80 °C | 2.29–2.60 | 15 | 60 | 3.48–8.25 | Gao et al.144 |
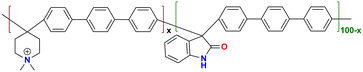
|
128.9 at 80 °C | 2.52 | 86.9 | 29.2 | 8.7 | Hu et al.145 |
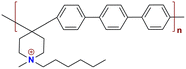
|
86.6 at 80 °C | 2.34 | 20 | 57 | — | Liu et al.143 |
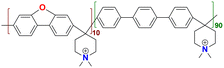
|
144.7 at 80 °C | 2.82 | 169.9 | 42 | — | Jeong et al.146 |
Hu et al.130 focused on backbone structure modification by introducing alkyl spacers of varying lengths (n = 0, 1, 2, 6, and 10) into poly(aryl-co-terphenyl piperidinium) (PDnTP-x) polymers. Short spacer membranes (n = 0–2) exhibited efficient ionic aggregation and continuous hydroxide transport pathways, achieving conductivities exceeding 150 mS cm−1 at 80 °C. Notably, the PD0TP-x membrane delivered a record peak power density of 2.67 W cm−2. Conversely, Kim et al.131 employed a side-chain modification strategy, keeping the same polymer backbone and cationic head as in the work of Hu and group, in which a hexyl alkyl chain spacer was introduced between the rigid polymer backbone and the cationic head group. This architecture promoted distinct microphase separation and optimized water management, leading to hydroxide conductivities exceeding 120 mS cm−1 and peak power densities up to 1.47 W cm−2. Overall, these studies demonstrate that precise molecular-level modification, either through backbone or side-chain modifications, critically enhances ion transport, stability, and fuel-cell performance, underscoring the strong structure-modification–performance relationship in AEM design.
6 Computational approach to anion-exchange membranes
Computational techniques provide a deep understanding of molecular interactions, enabling researchers to adapt properties for specific applications. Through simulations, researchers can predict the performance of membranes before their actual synthesis. The requirement for extensive experimental trials is considerably reduced by computational simulations, resulting in faster and more affordable membrane design.147,148 Gu et al.149 analysed different N3-substituted imidazolium-based AEMs, with 1,2-dimethyl-3-isopropylimidazolium [DMIIm]+, 1,2-dimethyl-3-butyl imidazolium [DMBIm]+, and 1,2,3-trimethylimidazolium [TMIm]+ ions, in aqueous solution through density functional theory. They reported that [TMIm]+ exhibited the lowest LUMO energy; hence, the probability of getting degraded by the nucleophilic attack of hydroxide ions is greater. Thus, [TMIm]+ is least stable in an alkaline environment compared to [DMIIm]+ and [DMBIm]+. [DMIIm]+ exhibited the highest LUMO energy; therefore, it is considered the stable cationic head in an alkaline environment. Park and team150 conducted molecular dynamics simulations to analyze the impact of functional groups on hydrocarbon anion-exchange membranes. The investigation focused on ethyl imidazolium (EI) and quaternary ammonium (QA) functional groups within the poly(arylene ether sulfone) (PES) backbone. The study involved calculating the mean-square displacement of OH− ions in both hydrated EI-PES and QA-PES systems at two distinct temperatures, 293 K and 353 K. Notably, the mean square displacement was observed to be highest in hydrated EI-PES at 353 K, signifying that EI-PES exhibited the highest conductivity among the examined groups. Moreover, through their analysis from radial distribution functions, it was found that EI-PES has greater alkaline stability compared to QA-PES. Takaba et al.151 carried out modelling of poly(arylene ether sulfone ketone)-based membranes containing fluorenyl groups and showed that OH− ion transport is mainly governed via surface diffusion of H3O2−. Zhang et al.152 analyzed three different types of AEMs based on poly(ether sulfone) (QAPIPPES), poly(phenylene oxide) (PI-PPO), and poly(aryl piperidinium) (PAP-BP). Through MD simulation, it was found PAP-BP showed a lower SR and efficient transportation of hydroxide ions (see Fig. 8). The obtained outcomes were also closely aligned with the experimental findings. Sepehr and research group153 explored the hydrated morphology and microscopic arrangement of a triblock copolymer, polystyrene-b-poly(ethylene-co-butylene)-b-polystyrene (SEBS), with alkyl-substituted quaternary ammonium cationic functional groups. This investigation employed Dissipative Particle Dynamics (DPD) simulation to gain insights into the molecular morphology. The conclusion drawn was that the membrane's morphology was influenced by the degree of hydration. Zadok et al.154 demonstrated the dominance of the hydroxide-paired structure over the conventional hydrated structure of hydroxide ions around the ammonium cations of AEMs and found a stable two-hydroxide ion-paired structure as a complex with water/ammonium groups. | ||
| Fig. 8 (a) Polymer structures; snapshots of systems (b) QAPIPPES, (c) PI-PPO, and (d) PAP-BP respectively at λ = 18; diffusion coefficients of (e) water molecules and (f) hydroxide ions in AEMs; MD snapshots of backbone segments with neighbouring water molecules and hydroxides in (g) QAPIPPES, (h) PI-PPO, and (i) PAP-BP. Panels (b)–(i) have been adapted/reproduced from Zhang et al.152 with permission from Elsevier, copyright 2022. | ||
Zelovich et al.155 investigated the effect of temperature on hydroxide ion diffusion, using quaternary trimethylammonium ions as a functional group, computationally using ab initio molecular dynamics (AIMD) methods as well as experimentally using 1H pulsed field gradient (PFG) nuclear magnetic resonance (NMR). For their theoretical investigation, a graphene bilayer served as the polymer backbone, and to validate their theoretical predictions, polysulfone (PSU)-based AEMs with TMA were utilized in their experimental analysis. In their examination, a notable increase in the diffusion of hydroxide ions at temperatures exceeding 400 K was observed. Furthermore, an unusual and distinct pattern in hydroxide ion diffusivity was detected. A kink around 350–400 K was observed, indicating that it is possible to achieve enhanced hydroxide ion diffusion at temperatures above the local minimum of the kink. Pan and co-workers156 analyzed an olefinic-based anion-exchange membrane with different types of mono and di-cationic functional groups, both experimentally and theoretically. From their investigation, it was concluded that di-cationic functional groups exhibit better stability and conductivity, a smaller swelling ratio and better water uptake. Lee et al.157 employed DPD mesoscale simulations to examine PPO with TMA cations under varying hydration levels and IEC. The simulation results indicated that higher hydration levels led to increased water and anion diffusivity, aligning with experimental findings. Additionally, the research revealed that elongating the hydrophobic alkyl chain augmented the enhanced phase-separation between hydrophilic and hydrophobic domains. Luo and co-workers158 examined the water-induced structure of SEBS with quaternary ammonium through mesoscale DPD calculations. Their study revealed that elevating the hydration level changed the structure from perforated and interconnected lamellae to a perfect lamellae.
Li et al.159 performed molecular dynamics simulations to understand the effect of side-chain position on hydroxide transport. Their research groups developed three types of long-side-chain poly(arylene indole piperidinium) anion-exchange membranes (AEMs): tadpole-chain configuration (t-PITPC10), pendant-chain arrangement (p-PITPC10), and a configuration where side chains are connected to the backbone but separated from the cationic groups (a-PITPC10). The simulation outcomes revealed that among the three, a-PITPC10 exhibited greater efficacy in improving the transport of hydroxide ions (see Fig. 9). Kim and their research team160 conducted molecular dynamics simulations to investigate how different functional groups affect the behavior of hydroxide ions in hydrated poly(ether ether ketone) (PEEK) with ammonium cationic heads. During the simulations, they systematically varied the concentration of functional groups, ranging from 0 to 40 mol%. Their findings indicated that as the amount of functional groups and water increased, the diffusion of ions also increased. However, they observed that when a large quantity of water was introduced into the simulation environment, it led to a lower polymer density and weakened connections between ionic sites. A combined study of DFT-based calculations and molecular dynamics simulations was conducted by Di Salvo and co-workers161 to analyse the effect of IEC on diffusion of hydroxide ions in AEMs. The ab initio calculations accurately projected the water absorption levels at various IECs, aligning well with the experimental observations. The study identified a critical IEC range of 1.2–1.7 meq g−1, within which the AEM experience excessive swelling.
 | ||
| Fig. 9 (a) Chemical structures of poly(arylene indole piperidinium) AEMs: without a side chain (PITP), with a tadpole chain (t-PITPC10), with a pendant chain (p-PITPC10), and with side chains connected to the backbone separated from the cationic groups (a-PITPC10); (b) snapshots of ion transport channels in AEMs at λ = 13; (c) distances between the adjacent cationic groups in AEMs at different hydration numbers; and (d) schematic diagram of adjacent hydration shells in a-PITPC10 at λ = 13. Panels (b)–(d) have been adapted/reproduced from Li et al.159 with permission from Elsevier, copyright 2023. | ||
Recently, Zelovich et al.162 analyzed the impact of functional groups on the diffusion of hydroxide ions and water in anion-exchange membranes using ab initio molecular dynamics (AIMD) techniques. Two distinct functional groups, trimethyl alkyl ammonium (TMA) and imidazolium (IMI), were employed in the investigation. A graphene bilayer served as the polymer framework. Their findings revealed that hydroxide ion diffusion was similar for both functional groups when temperatures exceeded 400 K. However, when temperatures dropped below 350 K, the TMA functional group exhibited greater hydroxide ion diffusivity. Conversely, in the case of IMI, higher water diffusivity was observed across all temperature ranges. Enhanced water diffusivity reduces the risk of cathode dehydration. Sharma et al.163 employed a molecular dynamics study using a quaternary ammonium functonalized Nafion membrane at varying hydration. The authors found a significant role of hydration in distinct inter-chain and intra-chain pendant group interactions with hydroxide ions. A coarse-grained molecular dynamics simulation was carried out by Chen and co-workers164 to explore two PEEK-based AEMs containing different types of side chains: one containing single quaternary (SQ) and the other containing gemini quaternary (GQ) ammonium groups. The simulation results demonstrated enhanced ionic conductivity in the case of GQ. The improvement was attributed to an increase in IEC rather than the self-diffusion coefficient, as both SQ and GQ exhibited similar self-diffusion coefficients in the simulations. Additionally, the study revealed that more water molecules surrounded the OH− groups for GQ, potentially contributing to its superior alkaline stability compared to SQ.
Ma and his research team165 examined the impact of incorporating a polar cyano-group side chain in poly(biphenyl piperidine-trifluoroacetophenone)-based AEMs, both theoretically and experimentally. Their findings revealed that AEMs containing the polar cyano group exhibited favorable ionic conductivity, improved mechanical properties, clearer microphase separation structures, and lower SR and WU. Through molecular dynamics simulations and experimental analysis, they demonstrated that AEMs with cyano-group side chains managed to strike a balance between high ionic conductivity and dimensional stability, addressing the inherent trade-off challenge in AEMs.
Chen and team166 investigated the effect of block size on hydroxide solvation and transportation in a diblock copolymer (PPO-b-PVBTMA). During their simulation, they kept the number of PVBTMA ions, hydroxide ions, and water molecules constant while increasing the PPO block count. Their findings revealed that an increase in the PPO block resulted in the constriction of water channels within the system. This constriction was supported by the reduction in the coordination number of water molecules around cationic groups and hydroxide ions, leading to a decrease in the diffusion constants of both hydroxide ions and water molecules. A study analyzing the impact of high-temperature operation on AEMFCs was conducted by Yassin and team.167 Using molecular studies, an increase in the AEMFC performance stability was found as the temperature increased from 45 °C up to 120 °C. Additionally, their findings indicated that when operating an AEMFC at a current density of 0.6 A cm2, the degradation rate constant decreases as the temperature increases. In a separate research endeavor, Yassin and group168 investigated the impact of membrane thickness on AEMFC fuel cells. They conducted a comprehensive study, combining theoretical analysis with experiments, focusing on commercially available FAA-3 AEMs with varying thicknesses, ranging from 5 to 50 micrometers (µm). The results revealed that reducing the thickness from 30 to 20 µm led to a moderate improvement in cell performance. However, a further reduction to 5 µm resulted in a significant enhancement in performance. Additionally, the researchers explored the influence of membrane thickness using two different cathode catalysts, Pt/C and Fe–N–C. In both cases, a decrease in membrane thickness led to an improvement in cell performance.
Sharma et al.169 investigated poly(ether ether ketone) (PEEK)-based membranes, primarily focusing on PEEK functionalized with trimethylammonium. In addition to PEEK-TMA, variations in alkyl chains and different cationic functional groups were examined to understand their influence on membrane behavior. From a computational perspective, the work employed all-atom molecular dynamics simulations using the GAFF force field, with the TIP3P water model, implemented in GROMACS. The systems were equilibrated under the NPT ensemble, followed by annealing cycles and extended production runs. Different hydration levels (λ = 5–25) were considered to systematically study the effect of water content on membrane morphology and ion transport. Importantly, the computational predictions were supported by experimental validation. The simulated dry density (1220 ± 4.2 kg m−3) showed less than 10% deviation from the reported experimental range (1260–1320 kg m−3), indicating accurate structural packing in the model. Similarly, the predicted glass-transition temperature (Tg) of 440–448 K was in close agreement with the experimental range of 418–423 K, supporting the reliability of the simulation approach. Furthermore, Pu and Sun170 investigated two AEMs containing piperidinium cationic head groups but different polymer backbones, a triphenyl-based backbone (QAPPT) and a quaterphenyl-based backbone (QAQPP) system. All-atom molecular dynamics simulations were performed using the OPLS/AA force field for the polymer chains, while the SPC/E water model was employed to describe water molecules.
To validate the reliability of their simulations, the authors first examined the dry-state membrane structure by calculating the IEC; the simulated IEC values were 2.80 mmol g−1 for QAPPT and 2.31 mmol g−1 for QAQPP, which showed close agreement with the corresponding experimental values of 2.6516 and 2.2922 mmol g−1, respectively. Beyond the dry state, the fully hydrated membrane structures were also assessed through swelling behavior. The calculated volume expansion from dry to fully hydrated conditions was 18.1% for QAPPT and 0.6% for QAQPP. These trends matched experimental swelling degree (SD) values of 9.5% for QAPPT and 2.3% for QAQPP.
7 Future perspectives and development trends of AEMs
Recent progress in the AEM field underscores that structural modification of the polymer and cationic head, as well as the arrangement of its hydrophilic and hydrophobic domain, plays a decisive role in enhancing ion transport, dimensional stability, and alkaline durability. Significant improvements have been achieved through the development of diverse polymer backbones and pendant cationic groups, along with innovative architectures such as block, comb-shaped, grafted, branched, and cross-linked systems. These approaches not only protect the integrity of cationic head-groups under highly alkaline conditions but also promote the formation of well-organized ionic domains that facilitate efficient hydroxide transport while limiting excessive swelling. Despite these advancements, several challenges remain before AEM-based technologies can be fully commercialized, such as:• Polymer and cation design: further improvement is still needed in the polymer backbone and the attached cationic groups so that the membrane can survive longer under strong alkaline conditions.171 By carefully modifying the main polymer structure and the side cationic groups, the overall alkaline durability can be increased. One important approach is to design cations that are sterically protected. In addition, removing or avoiding β-hydrogen atoms in the cation structure can help prevent common degradation reactions such as elimination and nucleophilic substitution. Researchers are also studying multi-cationic and dicationic functional groups, where more than one positive charge is present, because these groups can provide better chemical stability and ionic conductivity.
• Backbone rigidity: using an ether-free, fully aromatic backbone can significantly improve stability under strong alkaline conditions.172,173 A rigid backbone helps reduce segmental motion of the polymer chains, which minimizes water-induced swelling. However, if the backbone becomes too rigid, ion movement inside the membrane can slow down, which lowers conductivity. Therefore, it is important to maintain a proper balance between mechanical strength and chain flexibility to allow efficient ion transport. Moreover, poly(aryl piperidinium)-type backbones consistently provide improved alkaline durability due to elimination of benzylic sites, particularly when paired with sterically protected cyclic ammonium cations.
• Water management and hydration structure: hydroxide ion transport inside the membrane mainly depends on how water molecules surround the ions within the membrane structure.174 If the membrane absorbs too much water, it causes swelling. On the other hand, if there is not enough water, the continuous pathways needed for OH− movement get disturbed, and ion transport becomes difficult. Therefore, future membrane designs should focus on creating well-controlled and confined water channels that maintain a continuous pathway while avoiding excessive swelling.
• Role of Free Volume and chain dynamics: the free volume present inside a polymer plays an important role in controlling how water molecules move and how easily hydroxide ions can travel through the membrane.175 When the free volume is properly controlled, it can reduce the energy required for ion transport and improve conductivity. However, if the free volume becomes too high, it weakens the structure and reduces stability. The density of chain entanglement in the polymer also matters, as it influences mechanical strength. Side-chain engineering enhances conductivity through phase separation but often increases swelling unless constrained by crosslinking or hydrogen-bonding networks. For developing next-generation membranes, it will be very important to carefully evaluate these parameters.
• Linking AEM to electrochemical performance: to clearly understand how molecular design affects real device performance, membrane properties such as ionic conductivity, swelling behaviour, mechanical strength, and alkaline stability must be tested under practical operating conditions, including high current density, peak power density, elevated temperature, and varying humidity.176 Therefore, future studies may correlate molecular degradation mechanisms with voltage decay and performance loss. The stability rankings are shown to depend strongly on testing temperature, hydroxide concentration, and hydration state and need to be accommodated in rational design.
8 Conclusions
This review systematically highlighted the notable progress achieved in the domain of AEMs, particularly polymer backbone and pendant group developments in AEMFCs. A thorough approach was adopted in accentuating a range of efforts encompassing experimental and molecular studies. To counter the adverse impact of the highly alkaline environment on the cationic functional group's stability, researchers have innovatively devised various membrane structures, including block, comb, and graft polymers. The innovative strategy of random or block polymers serves dual purpose: (a) safeguarding the integrity of the cationic head, and (b) promoting the formation of a phase-separated membrane morphology. The optimization of structural characteristics through various cationic heads has made a significant impact on improvement of hydroxide ion conductivity and durability. Other strategies encompass crosslinking, polymer chain branching, and the integration of multi-cationic functional groups as pendant moieties. This review covers diverse membrane synthesis methodologies, encompassing distinct polymer backbones and cationic functional groups. AEM success for the goal of future decarbanization177 largely depends on integration of ionomer performance, durability and effective electro-catalysts at commercially feasible cost.178,179 It is noteworthy that increasing the side-chain length enhances conductivity but can also lead to greater swelling. In contrast, cross-linking enhances dimensional stability, though it may decrease molecular mobility.In addition, the assessment addresses the capability of computational methodologies in scrutinizing AEM properties. DFT methods have demonstrated their utility in evaluating the stability of the cationic head connected to the polymer backbone. Molecular dynamics simulations have yielded insights into the diffusion of hydroxide ions within the membrane. Computational techniques have further enabled the analysis of hydroxide ion conductivity, WU, SR, and various other membrane properties. Mesoscale calculations using DPD have been used in investigating the membrane morphology. Despite the remarkable advancements witnessed in recent years within the AEM field, certain challenges remain. Tackling aspects such as efficient water management, membrane stability under elevated temperatures, and the identification of optimal operating conditions is pivotal for unlocking the full potential of AEMFCs. With improved technology and machine-learning methods, the roles of free volume180 in the polymer matrix and chain entanglements175,181 need to be explored in detail for future generation AEMs. In summary, a decisive path through AEMs may certainly come true and we may get closer to a fossil-fuel-independent future with green and renewable energy.
Author contributions
PS and APS contributed to writing and review-editing. APS administrated the project and helped in the funding acquisition, analysis and discussion.Conflicts of interest
There are no conflicts to declare.Abbreviations
| AEMFC | Anion-exchange-membrane fuel cell |
| PEMFC | Proton-exchange-membrane fuel cell |
| AEM | Anion-exchange membrane |
| PPO | Polyphenyleneoxide |
| TMA | Trimethyl ammonium |
| WU | Water uptake |
| SR | Swelling ratio |
| QAPIPPES | Quaternary-ammonium-based piperidinium poly ether sulfone |
| PAP-BP | Biphenyl poly(aryl piperidinium) |
| PI-PPO | Piperidinium polyphenyleneoxide |
| BMP | 1-(6-Bromohexyl)-1-methylpiperidinium bromide |
| DFT | Density functional theory |
| DPD | Dissipative particle dynamics |
| Dha | Dihydroxyterephthalaldehyde |
| TGCl | Triaminoguanidinium chloride |
| IEC | Ion exchange capacity |
Data availability
No new data were generated or analyzed during this review. All data discussed in this manuscript are derived from previously published studies, which are cited in the reference list.Acknowledgements
PS thanks the Council of Scientific and Industrial Research, New Delhi for a Junior Research Fellowship (09/1256(19767)/2024-EMR-I). APS acknowledges the ‘Anusandhan National Research Foundation (ANRF)', Govt. of India for SERB – CRG/2022/001938 Research Grant.Notes and references
- B. G. Pollet, S. S. Kocha and I. Staffell, Curr. Opin. Electrochem., 2019, 16, 90–95 CrossRef CAS
.
- D. Li, A. R. Motz, C. Bae, C. Fujimoto, G. Yang, F.-Y. Zhang, K. E. Ayers and Y. S. Kim, Energy Environ. Sci., 2021, 14, 3393–3419 RSC
.
- G. Das, J.-H. Choi, P. K. T. Nguyen, D.-J. Kim and Y. S. Yoon, Polymers, 2022, 14, 1197 CrossRef CAS PubMed
.
- A. Arshad, H. M. Ali, A. Habib, M. A. Bashir, M. Jabbal and Y. Yan, Therm. Sci. Eng. Prog., 2019, 9, 308–321 CrossRef
.
- M. A. Abdelkareem, K. Elsaid, T. Wilberforce, M. Kamil, E. T. Sayed and A. Olabi, Sci. Total Environ, 2021, 752, 141803 CrossRef CAS PubMed
.
- N. A. A. Qasem and G. A. Q. Abdulrahman, Int. J. Energy Res., 2024, 2024, 7271748 CrossRef
.
- C. Kalyvas, A. Kucernak, D. Brett, G. Hinds, S. Atkins and N. Brandon, WENE, 2014, 3, 254–275 CrossRef CAS
.
- E. Qu, X. Hao, M. Xiao, D. Han, S. Huang, Z. Huang, S. Wang and Y. Meng, J. Power Sources, 2022, 533, 231386 CrossRef CAS
.
- A. T. Hamada, M. F. Orhan and A. M. Kannan, Energy Rep., 2023, 9, 6396–6418 CrossRef
.
- M. Hren, M. Božic, D. Fakin, K. S. Kleinschek and S. Gorgieva, Sustainable Energy Fuels, 2021, 5, 604–637 RSC
.
- N. Shaari, S. K. Kamarudin, R. Bahru, S. H. Osman and N. A. I. Md Ishak, Int. J. Energy Res., 2021, 45, 6644–6688 CrossRef CAS
.
- J. Rosen, T. Geary, A. Hilmi, R. Blanco-Gutierrez, C.-Y. Yuh, C. S. Pereira, L. Han, R. A. Johnson, C. A. Willman and H. Ghezel-Ayagh,
et al.
, J. Electrochem. Soc., 2020, 167, 064505 CrossRef CAS
.
- S. Z. Golkhatmi, M. I. Asghar and P. D. Lund, Renewable Sustainable Energy Rev., 2022, 161, 112339 CrossRef
.
-
S. P. Jiang, Q. Li, S. P. Jiang and Q. Li, Introduction to Fuel Cells: Electrochemistry and Materials, 2022, pp. 649–671 Search PubMed
.
- F. M. L. Figueiredo and F. M. B. Marques, WENE, 2013, 2, 52–72 CrossRef CAS
.
- L. Dubau, L. Castanheira, F. Maillard, M. Chatenet, O. Lottin, G. Maranzana, J. Dillet, A. Lamibrac, J.-C. Perrin, E. Moukheiber, A. ElKaddouri, G. De Moor, C. Bas, L. Flandin and N. Caqu, WIREs Energy Environ., 2014, 3, 540–560 CrossRef CAS
.
- N. Sazali, W. N. Wan Salleh, A. S. Jamaludin and M. N. Mhd Razali, Membranes, 2020, 10, 99 CrossRef CAS PubMed
.
- A. P. Sunda, S. Singh, S. Yadav and R. K. Singh, Chem. Phys. Chem., 2023, 24, e202300104 CrossRef CAS PubMed
.
- R. Vedarajan, R. Balaji and K. Ramya, Wiley Interdiscip. Rev.:Energy Environ., 2023, e466 CAS
.
-
N. Alonso-Vante and V. Di Noto, Electrocatalysis for Membrane Fuel Cells: Methods, Modeling,and
Applications, John Wiley & Sons, 2023 Search PubMed
.
- H. Lei, X. Yang, Z. Chen, D. Rawach, L. Du, Z. Liang, D.-S. Li, G. Zhang, A. C. Tavares and S. Sun, Adv. Mater., 2025, 37, 2410106 CrossRef CAS PubMed
.
- K. Yassin and D. R. Dekel, Front. Membr. Sci. Technol., 2025, 4, 1691096 CrossRef
.
- L. Liu, H. Ma, M. Khan and B. S. Hsiao, Membranes, 2024, 14, 85 CrossRef CAS PubMed
.
- M. Warshay and P. R. Prokopius, The Fuel Cell in Space: Yesterday, Today and Tomorrow, NASA Technical Memorandum NASA-TM-102366, 1989, https://ntrs.nasa.gov/citations/19900002488.
- Y.-J. Wang, J. Qiao, R. Baker and J. Zhang, Chem. Soc. Rev., 2013, 42, 5768–5787 RSC
.
- M. Muhyuddin, C. Santoro, L. Osmieri, V. C. A. Ficca, A. Friedman, K. Yassin, G. Pagot, E. Negro, A. Konovalova, G. Lindquist, L. Twight, M. Kwak, E. Berretti, V. D. Noto, F. Jaouen, L. Elbaz, D. R. Dekel, P. Mustarelli, S. W. Boettcher, A. Lavacchi and P. Atanassov, Chem. Rev., 2025, 125, 6906–6976 CrossRef CAS PubMed
.
- E. Tomasino, P. Scardi and N. Ataollahi, MRS Energy Sustain., 2025, 12, 203–222 CrossRef
.
- Z. Li, X. He, Z. Jiang, Y. Yin, B. Zhang, G. He, Z. Tong, H. Wu and K. Jiao, Electrochim. Acta, 2017, 240, 486–494 CrossRef CAS
.
- E. J. Park, P. Jannasch, K. Miyatake, C. Bae, K. Noonan, C. Fujimoto, S. Holdcroft, J. R. Varcoe, D. Henkensmeier, M. D. Guiver and Y. S. Kim, Chem. Soc. Rev., 2024, 53, 5704–5780 RSC
.
- J. Miyake and K. Miyatake, Sustainable Energy Fuels, 2019, 3, 1916–1928 RSC
.
- R. Vinodh, S. S. Kalanur, S. K. Natarajan and B. G. Pollet, Polymers, 2023, 15, 2144 CrossRef CAS PubMed
.
- D. Henkensmeier, M. Najibah, C. Harms, J. Žitka, J. Hnát and K. Bouzek, J. Electrochem. Energy Con. Stor., 2021, 18, 024001 CrossRef CAS
.
- A. P. Sunda and J. C. Bose, J. Eng. Technol. Sci., 2025, 1, 1–14 Search PubMed
.
- Z. Wang, G. Sun, N. H. C. Lewis, M. Mandal, A. Sharma, M. Kim, J. M. Montes de Oca, K. Wang, A. Taggart, A. B. Martinson, P. A. Kohl, A. Tokmakoff, S. N. Patel, P. F. Nealey and J. J. de Pablo, Nat. Commun., 2025, 16, 1099 CrossRef CAS PubMed
.
- N. Du, C. Roy, R. Peach, M. Turnbull, S. Thiele and C. Bock, Chem. Rev., 2022, 122, 11830–11895 CrossRef CAS PubMed
.
- J. Pan, J. Han, L. Zhu and M. A. Hickner, Chem. Mater., 2017, 29, 5321–5330 CrossRef CAS
.
- T. Nemeth, T. Nauser and L. Gubler, ChemSusChem, 2022, 15, e202201571 CrossRef CAS PubMed
.
- A. M. Samsudin, M. Bodner and V. Hacker, Polymers, 2022, 14, 3565 CrossRef CAS PubMed
.
- A. M. Barnes, Y. Du, W. Zhang, S. Seifert, S. K. Buratto and E. B. Coughlin, Macromolecules, 2019, 52, 6097–6106 CrossRef CAS
.
- J. Ye, S. Yu, C. Zheng, T. Sun, J. Liu and H. Li, Chem. Eng. J., 2022, 427, 131413 CrossRef CAS
.
- M. A. Hossain, H. Jang, S. C. Sutradhar, J. Ha, J. Yoo, C. Lee, S. Lee and W. Kim, Int. J. Hydrogen Energy, 2016, 41, 10458–10465 CrossRef CAS
.
- K. Wang, Z. Zhang, S. Li, H. Zhang, N. Yue, J. Pang and Z. Jiang, ACS Appl. Mater. Interfaces, 2021, 13, 23547–23557 CrossRef CAS PubMed
.
- U. Salma and N. Shalahin, Results Mater., 2023, 100366 CrossRef CAS
.
- J. Ren, J. Xu, M. Ju, X. Chen, P. Zhao, L. Meng, J. Lei and Z. Wang, Adv. Powder Mater., 2022, 1, 100017 CrossRef
.
- Z. Xu, S. Delgado, V. Atanasov, T. Morawietz, A. S. Gago and K. A. Friedrich, Membranes, 2023, 13, 328 CrossRef CAS PubMed
.
- W. Tang, Y. Yang, X. Liu, J. Dong, H. Li and J. Yang, Electrochim. Acta, 2021, 391, 138919 CrossRef CAS
.
- S. Bonizzoni, D. Stucchi, T. Caielli, E. Sediva, M. Mauri and P. Mustarelli, ChemElectroChem, 2023, 10, e202201077 CrossRef CAS
.
- D. Pan, P. M. Bakvand, T. H. Pham and P. Jannasch, J. Mater. Chem. A, 2022, 10, 16478–16489 RSC
.
- E. Cabukoglu, G. Georges, L. Kung, G. Pareschi and K. Boulouchos, Transp. Res. D: Transp. Environ., 2019, 70, 35–48 CrossRef
.
- B. Pivovar, Nat. Catal., 2019, 2, 562–565 CrossRef CAS
.
- B. E. Anley, Y. W. Ahmed, A. Yusuf, A. Candra, S. L. Kitaw, T.-Y. Wu, C.-C. Huang, J.-S. Wang, D. Thankachan, M. H. Khan, Y.-T. Cheng, C.-H. Wang and H.-C. Tsai, ACS Appl. Mater. Interfaces, 2026, 18, 6687–6700 CrossRef CAS PubMed
.
- N. Chen, H. H. Wang, S. P. Kim, H. M. Kim, W. H. Lee, C. Hu, J. Y. Bae, E. S. Sim, Y.-C. Chung, J.-H. Jang, S. J. Yoo, Y. Zhuang and Y. M. Lee, Nat. Commun., 2021, 12, 2367 CrossRef CAS PubMed
.
- Z. Yu, W. T. Gao, Y. J. Liu, Q. G. Zhang, A. M. Zhu and Q. L. Liu, J. Colloid Interface Sci., 2023, 651, 404–414 CrossRef CAS PubMed
.
-
S. C. Ramarez and R. R. Paz, New Trends in Ion Exchange Studies, IntechOpen, London, 2018, ch. 4 Search PubMed
.
-
B. Cermenek, J. Ranninger and V. Hacker, Ethanol, Elsevier, 2019, pp. 383–405 Search PubMed
.
- F. Foglia, Q. Berrod, A. J. Clancy, K. Smith, G. Gebel, V. G. Sakai, M. Appel, J.-M. Zanotti, M. Tyagi, N. Mahmoudi, T. S. Miller, J. R. Varcoe, A. P. Periasamy, D. J. L. Brett, P. R. Shearing, S. Lyonnard and P. F. McMillan, Nat. Mater., 2022, 21, 555–563 CrossRef CAS PubMed
.
- Z. Sun, B. Lin and F. Yan, ChemSusChem, 2018, 11, 58–70 CrossRef CAS PubMed
.
- F. Li, S. H. Chan and Z. Tu, Chem. Rec., 2023, e202300067 Search PubMed
.
- S. Gottesfeld, D. R. Dekel, M. Page, C. Bae, Y. Yan, P. Zelenay and Y. S. Kim, J. Power Sources, 2018, 375, 170–184 CrossRef CAS
.
- R. Kingsbury, K. Bruning, S. Zhu, S. Flotron, C. Miller and O. Coronell, Ind. Eng. Chem. Res., 2019, 58, 18663–18674 CrossRef CAS
.
-
E. Drioli and L. Giorno, Encyclopedia of membranes, Springer, 2018 Search PubMed
.
- F. Karas, J. Hnát, M. Paidar, J. Schauer and K. Bouzek, Int. J. Hydrogen Energy, 2014, 39, 5054–5062 CrossRef CAS
.
- B. Yang and Z. Cunman, Chem. Eng. J., 2023, 457, 141094 CrossRef CAS
.
- F. Xu, Y. Su and B. Lin, Front. Mater., 2020, 7, 4 CrossRef
.
- A. L. Frischknecht, P. J. int Veld, I. V. Kolesnichenko, D. J. Arnot and T. N. Lambert, ACS Appl. Polym. Mater., 2022, 4, 2470–2480 CrossRef CAS
.
- J. M. Clary, L. Wang, Y. Yan, A. L. Frischknecht and D. Vigil-Fowler, J. Membr. Sci., 2025, 717, 123517 CrossRef CAS
.
- A. Barnett, J. Lu and V. Molinero, J. Phys. Chem. C, 2021, 125, 27703–27713 CrossRef CAS
.
- W. F. Drayer, E. M. Duan, J. C. Johnson, K. I. Winey and A. L. Frischknecht, Macromolecules, 2025, 58, 10017–10025 CrossRef CAS
.
- F. Razmjooei, A. Farooqui, R. Reissner, A. S. Gago, S. A. Ansar and K. A. Friedrich, ChemElectroChem, 2020, 7, 3951–3960 CrossRef CAS
.
- R. Gutru, Z. Turtayeva, F. Xu, G. Maranzana, B. Vigolo and A. Desforges, Int. J. Hydrogen Energy, 2020, 45, 19642–19663 CrossRef CAS
.
- K. Otsuji, Y. Shirase, T. Asakawa, N. Yokota, K. Nagase, W. Xu, P. Song, S. Wang, D. A. Tryk and K. Kakinuma,
et al.
, J. Power Sources, 2022, 522, 230997 CrossRef CAS
.
- Y. Zheng, U. Ash, R. P. Pandey, A. G. Ozioko, J. Ponce-Gonzalez, M. Handl, T. Weissbach, J. R. Varcoe, S. Holdcroft, M. W. Liberatore, R. Hiesgen and D. R. Dekel, Macromolecules, 2018, 51, 3264–3278 CrossRef CAS
.
- Z. Xu, L. Wan, Y. Liao, P. Wang, K. Liu and B. Wang, J. Mater. Chem. A, 2021, 9, 23485–23496 RSC
.
- C. G. Arges, V. K. Ramani and P. N. Pintauro, Electrochem. Soc. Interface, 2010, 19, 31 CrossRef CAS
.
- W. E. Mustain, M. Chatenet, M. Page and Y. S. Kim, Energy Environ. Sci., 2020, 13, 2805–2838 RSC
.
- B. Lee, D. Yun, J.-S. Lee, C. H. Park and T.-H. Kim, J. Phys. Chem. C, 2019, 123, 13508–13518 CrossRef CAS
.
-
P. Sgarbossa, G. Crivellaro, F. Lanero, G. Pagot, A. R. Alvi, E. Negro, K. Vezzù and V. Di Noto, Electrocatalysis for Membrane Fuel Cells: Methods, Modeling and Applications, 2023, pp. 227–285 Search PubMed
.
- D. Li, E. J. Park, W. Zhu, Q. Shi, Y. Zhou, H. Tian, Y. Lin, A. Serov, B. Zulevi, E. D. Baca, C. Fujimoto, H. T. Chung and Y. S. Kim, Nat. Energy, 2020, 5, 378–385 CrossRef CAS
.
- R. Yu, H. Yang, X. Yu, J. Cheng, Y. Tan and X. Wang, Int. J. Hydrogen Energy, 2024, 50, 582–604 CrossRef CAS
.
- X. Su, L. Gao, L. Hu, N. A. Qaisrani, X. Yan, W. Zhang, X. Jiang, X. Ruan and G. He, J. Membr. Sci., 2019, 581, 283–292 CrossRef CAS
.
- K. Min, J. E. Chae, Y. Lee, H.-J. Kim and T.-H. Kim, J. Membr. Sci., 2022, 653, 120487 CrossRef CAS
.
- Y. Yuan, X. Du, H. Zhang, H. Wang and Z. Wang, J. Membr. Sci., 2022, 642, 119986 CrossRef CAS
.
- D. R. Dekel, M. Amar, S. Willdorf, M. Kosa, S. Dhara and C. E. Diesendruck, Chem. Mater., 2017, 29, 4425–4431 CrossRef CAS
.
- N. Gjineci, S. Aharonovich, D. R. Dekel and C. E. Diesendruck, ACS Appl. Mater. Interfaces, 2020, 12, 49617–49625 CrossRef CAS PubMed
.
- J. Muller, A. Zhegur, U. Krewer, J. R. Varcoe and D. R. Dekel, ACS Mater. Lett., 2020, 2, 168–173 CrossRef CAS PubMed
.
- I. Agami, N. Gjineci, S. Li, S. Srebnik, D. R. Dekel and C. E. Diesendruck, ACS Appl. Polym. Mater., 2022, 4, 9250–9256 CrossRef CAS
.
- S. Willdorf-Cohen, A. Zhegur-Khais, J. Ponce-Gonzalez, S. Bsoul-Haj, J. R. Varcoe, C. E. Diesendruck and D. R. Dekel, ACS Appl. Energy Mater., 2023, 6, 1085–1092 CrossRef CAS PubMed
.
- K. Aggarwal, N. Gjineci, A. Kaushansky, S. Bsoul, J. C. Douglin, S. Li, I. Salam, S. Aharonovich, J. R. Varcoe, D. R. Dekel and C. E. Diesendruck, ACS Mater. Au, 2022, 2, 367–373 CrossRef CAS PubMed
.
- T. Wang, D. Chen, C. Wang, H. Wei and Y. Ding, Adv. Funct. Mater., 2025, 35, 2422504 CrossRef CAS
.
- Q. Liu, J. Han, B. Liu, Y. Wang, Y. Pang, T. Mu, K. Dong and C. Zhao, J. Membr. Sci., 2025, 734, 124480 CrossRef CAS
.
- T. Wang, Y. Zhao, S. Wang, S. Cheng, S. Yang, H. Wei and Y. Ding, J. Power Sources, 2023, 557, 232590 CrossRef CAS
.
- J. Li, C. Liu, J. Ge, W. Xing and J. Zhu, Chem. – Eur. J., 2023, 29, e202203173 CrossRef CAS PubMed
.
- N. Chen, J. H. Park, C. Hu, H. H. Wang, H. M. Kim, N. Y. Kang and Y. M. Lee, J. Mater. Chem. A, 2022, 10, 3678–3687 RSC
.
- M. Guo, T. Ban, Y. Wang, X. Wang and X. Zhu, J. Colloid Interface Sci., 2023, 638, 349–362 CrossRef CAS PubMed
.
- A. Das, B. Sana, R. Bhattacharyya, P. Chandra Ghosh and T. Jana, ACS Appl. Polym. Mater., 2022, 4, 1523–1534 CrossRef CAS
.
- Z. Peng, Z. Ren, S. Chen, Y. Zhao, P. Jannasch and J. Yang, Int. J. Hydrogen Energy, 2025, 188, 151884 CrossRef CAS
.
- B. Motealleh, Z. Liu, R. I. Masel, J. P. Sculley, Z. Richard Ni and L. Meroueh, Int. J. Hydrogen Energy, 2021, 46, 3379–3386 CrossRef CAS
.
- S. Maurya, S.-H. Shin, Y. Kim and S.-H. Moon, RSC Adv., 2015, 5, 37206–37230 RSC
.
- A. M. Samsudin and V. Hacker, Polymers, 2019, 11, 1399 CrossRef CAS PubMed
.
- F. Wang, C. Li, J. Sang, Y. Cui and H. Zhu, Int. J. Hydrogen Energy, 2021, 46, 36301–36313 CrossRef CAS
.
- L. Wang, M. Bellini, H. A. Miller and J. R. Varcoe, J. Mater. Chem. A, 2018, 6, 15404–15412 RSC
.
- A. L. G. Biancolli, S. Bsoul-Haj, J. C. Douglin, A. S. Barbosa, R. R. de Sousa Jr, O. Rodrigues Jr, A. J. Lanfredi, D. R. Dekel and E. I. Santiago, J. Membr. Sci., 2022, 641, 119879 CrossRef CAS
.
- A. L. G. Biancolli, B. Chen, A. S. Menandro, F. C. Fonseca, E. I. Santiago and S. Holdcroft, J. Mater. Chem. A, 2024, 12, 21442–21454 RSC
.
- W. You, K. J. Noonan and G. W. Coates, Prog. Polym. Sci., 2020, 100, 101177 CrossRef CAS
.
- W. You, K. M. Hugar and G. W. Coates, Macromolecules, 2018, 51, 3212–3218 CrossRef CAS
.
- Y. Chen, M. M. Abdellatif and K. Nomura, Tetrahedron, 2018, 74, 619–643 CrossRef CAS
.
- Z. He, G. Wang, C. Wang, L. Guo, R. Wei, G. Song, D. Pan, R. Das, N. Naik and Z. Hu,
et al.
, Polym. Rev., 2021, 61, 689–713 CrossRef CAS
.
- A. K. Singh, S. Kumar, M. Bhushan and V. K. Shahi, Sep. Purif. Technol., 2020, 234, 116078 CrossRef
.
- R. Lv, Z. Peng, Y. Wu, M. M. Gomaa and J. Yang, J. Membr. Sci., 2026, 742, 125120 CrossRef CAS
.
- Q. Wang, T. Wei, Z. Peng, Y. Zhao, P. Jannasch and J. Yang, J. Colloid Interface Sci., 2025, 686, 304–317 CrossRef CAS PubMed
.
- Z. Peng, T. Wei, Q. Wang, Y. Zhao and J. Yang, J. Power Sources, 2025, 642, 236918 CrossRef CAS
.
- A. N. Lai, S. C. Li, P. Z. Zheng, S. F. Zhou and S. J. Wu, J. Appl. Polym. Sci., 2024, 141, e55047 CrossRef CAS
.
- J. Huang, Z. Yu, J. Tang, P. Wang, X. Zhang, J. Wang and X. Lei, Colloids Surf., A, 2023, 674, 131890 CrossRef CAS
.
- N. Li, T. Yan, Z. Li, T. Thurn-Albrecht and W. H. Binder, Energy Environ. Sci., 2012, 5, 7888–7892 RSC
.
- K. Zhang, S. Gong, B. Zhao, Y. Liu, N. A. Qaisrani, L. Li, F. Zhang and G. He, J. Membr. Sci., 2018, 550, 59–71 CrossRef CAS
.
- J. Chen, C. Shen and S. Gao, Desalination, 2023, 557, 116600 CrossRef CAS
.
- J. J. Wang, W. T. Gao, Y. S. L. Choo, Z. H. Cai, Q. G. Zhang, A. M. Zhu and Q. L. Liu, J. Colloid Interface Sci., 2023, 629, 377–387 CrossRef CAS PubMed
.
- X. Wu, N. Chen, H.-A. Klok, Y. M. Lee and X. Hu, Angew. Chem., 2022, 134, e202114892 CrossRef
.
- S. Lee, H. Lee, T.-H. Yang, B. Bae, N. A. T. Tran, Y. Cho, N. Jung and D. Shin, Membranes, 2020, 10, 306 CrossRef CAS PubMed
.
- S. Hosimin, V. Varshini, A. Kalaiyarasi, S. Vengatesan, S. Ravichandran, C. Djadocks and S. Vasudevan, New J. Chem., 2023, 47, 15987–15997 RSC
.
- J. H. Chen, X. B. Yue, Y. S. L. Choo, Z. Yu, X. H. Wang, X. L. Gao, W. T. Gao, Q. G. Zhang, A. M. Zhu and Q. L. Liu, J. Power Sources, 2023, 570, 233014 CrossRef CAS
.
- M. Mandal, G. Huang, N. U. Hassan, W. E. Mustain and P. A. Kohl, J. Mater. Chem. A, 2020, 8, 17568–17578 RSC
.
- T. Caielli, A. R. Ferrari, S. Bonizzoni, E. Sediva, A. Capr, M. Santoro, I. Gatto, V. Baglio and P. Mustarelli, J. Power Sources, 2023, 557, 232532 CrossRef CAS
.
- T. Wang, Y. Zhao, R. Ma, L. Li, Q. Ling, X. Li, Y. Ding and H. Wei, Synth. Met., 2023, 296, 117385 CrossRef CAS
.
- X. Wu, N. Chen, C. Hu, H.-A. Klok, Y. M. Lee and X. Hu, Adv. Mater., 2023, 35, 2210432 CrossRef CAS PubMed
.
- N. Zhang, X. Li, P. Li and S. Tang, Int. J. Hydrogen Energy, 2023, 25972–25983 CrossRef CAS
.
- M. Ju, Q. Ren, J. Xu, X. Chen, L. Meng, J. Lei, P. Zhao and Z. Wang, Chem. Eng. J., 2023, 466, 143023 CrossRef CAS
.
- I. Arunkumar, R. Gokulapriyan, V. Sakthivel, A. R. Kim, M. S. Oh, J. Y. Lee, S. Kim, S. Lee and D. J. Yoo, ACS Appl. Energy Mater., 2023, 6, 7702–7713 CrossRef CAS
.
- S.-B. Lee, C.-M. Min, J. Jang and J.-S. Lee, Polymer, 2020, 192, 122331 CrossRef
.
- C. Hu, J. H. Park, H. M. Kim, H. H. Wang, J. Y. Bae, N. Y. Kang, N. Chen and Y. M. Lee, J. Membr. Sci., 2022, 647, 120341 CrossRef CAS
.
- H. M. Kim, C. Hu, H. H. Wang, J. H. Park, N. Chen and Y. M. Lee, J. Membr. Sci., 2022, 644, 120109 CrossRef CAS
.
- C. X. Lin, X. L. Huang, D. Guo, Q. G. Zhang, A. M. Zhu, M. L. Ye and Q. L. Liu, J. Mater. Chem. A, 2016, 4, 13938–13948 RSC
.
- S. S. Patil, V. M. adhura, I. Kammakakam, M. H. Swamy, K. S. Patil, Z. Lai and A. R. HN, Electrochim. Acta, 2022, 426, 140826 CrossRef CAS
.
- L. Zhu, X. Yu and M. A. Hickner, J. Power Sources, 2018, 375, 433–441 CrossRef CAS
.
- B. Lin, F. Xu, Y. Su, J. Han, Z. Zhu, F. Chu, Y. Ren, L. Zhu and J. Ding, ACS Appl. Energy Mater., 2019, 3, 1089–1098 CrossRef
.
- M. S. Cha, J. E. Park, S. Kim, S.-H. Han, S.-H. Shin, S. H. Yang, T.-H. Kim, D. M. Yu, S. So and Y. T. Hong,
et al.
, Energy Environ. Sci., 2020, 13, 3633–3645 RSC
.
- T. H. Pham, J. S. Olsson and P. Jannasch, J. Mater. Chem. A, 2019, 7, 15895–15906 RSC
.
- H. J. Park, S. Y. Lee, T. K. Lee, H.-J. Kim and Y. M. Lee, J. Membr. Sci., 2020, 611, 118355 CrossRef CAS
.
- L. Wang, J. J. Brink, Y. Liu, A. M. Herring, J. Ponce-González, D. K. Whelligan and J. R. Varcoe, Energy Environ. Sci., 2017, 10, 2154–2167 RSC
.
- J. Zhang, K. Zhang, X. Liang, W. Yu, X. Ge, M. A. Shehzad, Z. Ge, Z. Yang, L. Wu and T. Xu, J. Mater. Chem. A, 2021, 9, 327–337 RSC
.
- N. Chen, S. Y. Paek, J. Y. Lee, J. H. Park, S. Y. Lee and Y. M. Lee, Energy Environ. Sci., 2021, 14, 6338–6348 RSC
.
- W. Yuan, L. Zeng, S. Jiang, C. Yuan, Q. He, J. Wang, Q. Liao and Z. Wei, J. Membr. Sci., 2022, 657, 120676 CrossRef CAS
.
- L. Liu, L. Bai, Z. Liu, S. Miao, J. Pan, L. Shen, Y. Shi and N. Li, J. Membr. Sci., 2023, 665, 121135 CrossRef CAS
.
- W. T. Gao, X. L. Gao, W. W. Gou, J. J. Wang, Z. H. Cai, Q. G. Zhang, A. M. Zhu and Q. L. Liu, J. Membr. Sci., 2022, 655, 120578 CrossRef CAS
.
- X. Hu, Y. Huang, L. Liu, Q. Ju, X. Zhou, X. Qiao, Z. Zheng and N. Li, J. Membr. Sci., 2021, 621, 118964 CrossRef CAS
.
- I. Jeong, K. Min, H. Kim, S. Y. Nam and T.-H. Kim, J. Membr. Sci., 2023, 122079 CrossRef CAS
.
- M. Karibayev, S. Kalybekkyzy, Y. Wang and A. Mentbayeva, Molecules, 2022, 27, 3574 CrossRef CAS PubMed
.
- C. N. M. Ouma, K. O. Obodo and D. Bessarabov, Membranes, 2022, 12, 1051 CrossRef CAS PubMed
.
- F. Gu, H. Dong, Y. Li, Z. Si and F. Yan, Macromolecules, 2014, 47, 208–216 CrossRef CAS
.
- C. H. Park, T.-H. Kim, D. J. Kim and S. Y. Nam, Int. J. Hydrogen Energy, 2017, 42, 20895–20903 CrossRef CAS
.
- H. Takaba, T. Hisabe, T. Shimizu and M. K. Alam, J. Membr. Sci., 2017, 522, 237–244 CrossRef CAS
.
- K. Zhang, W. Yu, X. Ge, L. Wu and T. Xu, J. Membr. Sci., 2022, 661, 120922 CrossRef CAS
.
- F. Sepehr, H. Liu, X. Luo, C. Bae, M. E. Tuckerman, M. A. Hickner and S. J. Paddison, Macromolecules, 2017, 50, 4397–4405 CrossRef CAS
.
- I. Zadok, D. R. Dekel and S. Srebnik, J. Phys. Chem. C, 2019, 123, 27355–27362 CrossRef CAS
.
- T. Zelovich, L. Vogt-Maranto, C. Simari, I. Nicotera, M. A. Hickner, S. J. Paddison, C. Bae, D. R. Dekel and M. E. Tuckerman, Chem. Mater., 2022, 34, 2133–2145 CrossRef CAS
.
- J. Pan, H. Zhu, H. Cao, B. Wang, J. Zhao, Z. Sun and F. Yan, J. Membr. Sci., 2021, 620, 118794 CrossRef CAS
.
- M.-T. Lee, J. Phys. Chem. C, 2019, 123, 10802–10815 CrossRef CAS
.
- X. Luo and S. J. Paddison, Solid State Ionics, 2019, 339, 115012 CrossRef
.
- R. Li, X. Chen, X. Zhou, Y. Shen and Y. Fu, Sep. Purif. Technol., 2023, 314, 123577 CrossRef CAS
.
- D. J. Kim, C. H. Park and S. Y. Nam, Int. J. Hydrogen Energy, 2016, 41, 7641–7648 CrossRef CAS
.
- J. L. Di Salvo, G. De Luca, A. Cipollina and G. Micale, J. Membr. Sci., 2020, 599, 117837 CrossRef
.
- T. Zelovich, D. R. Dekel and M. E. Tuckerman, J. Membr. Sci., 2023, 678, 121638 Search PubMed
.
- P. Sharma and A. P. Sunda, ACS Appl. Polym. Mater., 2025, 7, 1886–1895 CrossRef CAS
.
- S. Chen, H. Wang, J. Zhang, S. Lu and Y. Xiang, J. Membr. Sci., 2020, 605, 118105 CrossRef CAS
.
- W. Ma, Q. Liu, J. Li, L. Tian and H. Zhu, Int. J. Hydrogen Energy, 2023, 48, 15258–15268 CrossRef
.
- C. Chen, C. Arntsen and Y.-L. S. Tse, J. Chem. Phys., 2020, 152, 094903 Search PubMed
.
- K. Yassin, I. G. Rasin, S. Willdorf-Cohen, C. E. Diesendruck, S. Brandon and D. R. Dekel, J. Power Sources Adv., 2021, 11, 100066 Search PubMed
.
- K. Yassin, J. C. Douglin, I. G. Rasin, P. G. Santori, B. Eriksson, N. Bibent, F. Jaouen, S. Brandon and D. R. Dekel, Energy Convers. Manage., 2022, 270, 116203 CrossRef CAS
.
- R. Sharma, T. Chakraborti and J. K. Singh, J. Memb. Sci., 2025, 736, 124598 CrossRef CAS
.
- W. Pu and Z. Sun, Phys. Chem. Chem. Phys., 2025, 27, 4634–4642 RSC
.
- Y. Yang, H. Huang, Z. Li, C. Xiao, M. U. Haq and L. Zeng, Future Batteries, 2025, 5, 100016 CrossRef
.
- A. Sahul Hameed, S. Munusamy, R. Gokulapriyan and D. J. Yoo, ACS Appl. Polym. Mater., 2024, 6, 12341–12361 CrossRef CAS
.
- N. Chen, C. Hu and Y. M. Lee, Acc. Chem. Res., 2025, 58, 688–702 CrossRef CAS PubMed
.
- J. Xue, J. C. Douglin, K. Yassin, T. Huang, H. Jiang, J. Zhang, Y. Yin, D. R. Dekel and M. D. Guiver, Joule, 2024, 8, 1457–1477 CrossRef CAS
.
- J. Xue, J. C. Douglin, T. Huang, H. Jiang, J. Zhang, Y. Yin, D. R. Dekel and M. D. Guiver, J. Membr. Sci., 2025, 717, 123519 CrossRef CAS
.
- W. K. Ng, W. Y. Wong, N. A. Rosli and K. S. Loh, Separations, 2023, 10, 424 CrossRef CAS
.
- C. Santoro, A. Lavacchi, P. Mustarelli, V. Di Noto, L. Elbaz, D. R. Dekel and F. Jaouen, ChemSusChem, 2022, 15, e202200027 CrossRef CAS PubMed
.
- G. A. Lindquist, S. Z. Oener, R. Krivina, A. R. Motz, A. Keane, C. Capuano, K. E. Ayers and S. W. Boettcher, ACS Appl. Mater. Interfaces, 2021, 13, 51917–51924 CrossRef CAS
.
- J. Lim, J. M. Klein, S. G. Lee, E. J. Park, S. Y. Kang, S. Maurya, W. E. Mustain, S. Boettcher and Y. S. Kim, ACS Energy Lett., 2024, 9, 3074–3083 CrossRef CAS
.
- S. Dagar and A. P. Sunda, Mol. Syst. Des. Eng., 2026, 11, 255–268 RSC
.
- K. Yassin, I. G. Rasin, S. Brandon and D. R. Dekel, J. Membr. Sci., 2024, 690, 122164 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |





