 Open Access Article
Open Access ArticlePerovskites beyond efficiency: stability challenges from space to Earth†
André F. V. Fonseca a,
Lyubov A. Frolova
a,
Lyubov A. Frolova b,
Pavel A. Troshin
b,
Pavel A. Troshin bc,
Jianyu Yuan
bc,
Jianyu Yuan d and
Ana F. Nogueira
d and
Ana F. Nogueira *a
*a
aLaboratório de Nanotecnologia e Energia Solar, Instituto de Química, Universidade Estadual de Campinas (UNICAMP), Campinas, Brazil. E-mail: anafla@unicamp.br
bFederal Research Center for Problems of Chemical Physics and Medicinal Chemistry RAS, Chernogolovka, Moscow Region, Russia
cZhengzhou Research Institute of HIT, 26 Longyuan East 7th, Jinshui District, Zhengzhou, Henan Province 450000, China
dState Key Laboratory of Bioinspired Interfacial Materials Science, Institute of Functional Nano & Soft Materials (FUNSOM), Soochow University, Suzhou, Jiangsu, China
First published on 12th March 2026
Abstract
To advance metal halide perovskite (MHP) optoelectronics beyond efficiency, international collaboration is essential, with an emphasis on stability, cost, and scalability. MHPs demonstrate remarkable stability in extreme space-level radiation; nevertheless, their stability under everyday conditions, particularly when exposed to humidity and thermal cycling, remains a significant challenge. Strategies such as low-dimensional interface passivation, quantum-dot integration, and in situ synchrotron studies are among the methods emphasized in this perspective to mitigate these problems. We propose pathways of collaborative research aimed at accelerating the development of durable, high-performance perovskite devices by synthesizing insights from the broader literature in conjunction with findings from a BRICS joint initiative project.
Introduction
A delicate balance exists between economic growth and sustainable development, shaping the current global energy landscape. The expansion of economic activity, coupled with increasing energy consumption and the effects of globalization, exerts increasing pressure on the environment.1 As a result, addressing environmental degradation and advancing sustainability have become global priorities.2The BRICS initiative was launched in 2006 to promote political and economic cooperation among the world's leading emerging economies: Brazil, Russia, India, China, and South Africa. The gross domestic product (GDP) of the BRICS countries in 2024, as reported by the International Monetary Fund (IMF), is approximately US$28 trillion, representing around 24.8% of global GDP.3 As significant producers and also consumers of energy, these countries are undergoing a substantial transition toward renewable energy adoption and environmental sustainability.4 Recently, it was reported that in 2024 the BRICS coalition accounted for 51% of global solar electricity generation, marking a significant increase from the 15% recorded a decade earlier.5 China was the principal leader of this expansion, accounting for 39% of global solar electricity generation in 2024, up from 12% in 2014. Significant growth was also observed in India and Brazil, which accounted for 6.3% (up from 2.5% in 2014) and 3.5% (from 0.01% in 2014), respectively.5
Given the BRICS nations’ accelerating solar deployment, there is growing interest in emerging photovoltaic technologies (e-PV) that could combine efficiency, scalability, and low production costs.6 Among the most promising e-PV technologies, metal halide perovskite (MHP)-based technologies have achieved certified efficiencies of 27% for single-junction and 30.1% for all-perovskite tandem configurations.7
In this perspective, we summarize the main findings and discussions from the I Workshop on Perovskite Technologies, which emerged from a joint project involving researchers from MHP-PV technology from Brazil, Russia, and China.
The core perspective: divergent pathways for stability
A critical, yet often overlooked, perspective in perovskite photovoltaics is the fundamental divergence in stability requirements between different application environments. Research reveals a striking paradox: MHPs can exhibit exceptional tolerance to extreme stressors encountered in space—such as high-energy particle radiation and severe thermal cycling—while degrading rapidly under moderate terrestrial conditions like ambient humidity and mild thermal fluctuations. This dichotomy is not a mere contradiction but a crucial insight. It indicates that the dominant degradation mechanisms are application-specific. Consequently, the future research and development pathway must evolve from a singular pursuit of a universally “stable” perovskite towards the targeted design of materials, interfaces, and devices optimized for distinct operational regimes. This perspective argues that recognizing and embracing this divergence is key to transitioning perovskite solar cells from high-performance lab prototypes to durable technologies for both terrestrial and space-based power generation.Efficiency alone isn’t enough anymore
Recent non-certified PV research, highlighted in the 5th annual Emerging PV Report, underscores the growing importance of operational stability as a key metric for assessing long-term device durability.6 While perovskite solar cells (PSCs) have achieved impressive power conversion efficiency (PCE) values, their stability under continuous operation remains a significant challenge. A common issue found in the literature is the lack of detailed stability reporting, especially under outdoor conditions, which hinders accurate comparisons. Therefore, including both test conditions and initial PCE values is crucial for estimating energy generation and degradation rates, enabling reliable benchmarking of device longevity across different studies.6,8Liu et al. recently evaluated the commercialization potential of PSCs, focusing on efficiency, stability, and cost-effectiveness. Based on current industrial processes, they estimated a manufacturing cost of $0.57 per W and a levelized cost of electricity (LCOE) of $0.22 per kWh, with materials accounting for ∼70% of the total cost (see Fig. 1a).9 Their analysis highlights that PSCs could surpass silicon solar cells if efficiencies exceed 25% and lifetimes reach at least 25 years. The first milestone has already been achieved, as certified efficiencies of above 25% were reported in 2019,7 underscoring the realistic potential of PSCs to become a commercially available technology with further advances in stability and fabrication.9
 | ||
| Fig. 1 (a) Reported results for the LCOE of PSCs at different stages.9 Reproduced from ref. 9 under the Creative Commons CC BY license. © 2025 The Author(s). Published by Springer Nature. (b) Distribution of publications* reporting high-efficiency PSCs and those explicitly addressing device stability. *Data included herein are derived from Clarivate™ (Web of Science™). © Clarivate 2025. All rights reserved. | ||
Thus, while reducing the fabrication costs of perovskite solar modules, particularly through improvements in the electron transport layer (ETL) and transparent conductive oxide (TCO), remains essential,9 enhancing the device stability is more critical than ever. This likely explains the growing research focus on stability over the efficiency record race since 2017, as illustrated in Fig. 1b. At the workshop on perovskite technologies, organized by the BRICS researchers involved in the joint project, one of the most frequently mentioned terms was indeed ‘stability’. It was also the focus of several discussions on film- and device-level characterizations, as well as on the application of low-dimensional materials, such as 2D and 0D nanomaterials, in this context. The following sections will provide a detailed discussion of these subjects.
Promising for space, still challenging on Earth
To illustrate the stability issues in practical applications, we focus on extremely-high-altitude applications (aerospace). PSCs are very attractive for these applications due to their light weight and flexible design. The principal metric for space solar arrays, in addition to high efficiency, is the power-to-weight ratio, which significantly affects payload weight and space launch costs. Flexibility can also improve the system's adaptability, thereby simplifying spacecraft design. These metrics are vital for the growing market of high-altitude unmanned aerial vehicles (UAVs), where lower power input requirements are permissible owing to shorter operational lifetimes. In this context, the characteristics of MHPs enable PSCs to surpass the actual competitors, such as silicon and A3B5 (also known as III–V) solar cells.10,11Additionally, the ability of perovskite-based PV technology to integrate scalable printing deposition techniques, such as roll-to-roll or inkjet printing, can enable the complete in situ production of photovoltaic modules during space missions.12,13
A satellite in low-Earth orbit (LEO) typically has a mission life of 3 to 5 years, which is significantly shorter than the 25-year minimum working life required for photovoltaic panels, as discussed above.14 However, a concern about perovskites and this kind of application is that the harsh conditions of space, including thermal cycling, high vacuum, and cosmic radiation, can lead to rapid degradation in device performance (Fig. 2a).15 As also discussed above, although metal-halide PSCs have achieved very high PCEs, their durability under real-world conditions remains the critical limitation. Outdoor perovskite devices are commonly exposed to temperature cycles of varying intensity, driven by diurnal fluctuations or geographic conditions, which can induce crystal lattice changes, interfacial reactions, and photocatalyzed thermal degradation.16 It is necessary to develop strategies to enhance thermal resilience, including improving perovskite crystallinity, relieving interlayer stress with buffer layers, and emphasizing standardized temperature-cycling protocols for meaningful cross-study comparisons.8,14,17 Interestingly, MHPs can withstand extremely high γ-ray doses of up to 10 MGy. Using infrared near-field optical microscopy (IR s-SNOM, Fig. 2b), Troshin et al. observed that in MAPbI3, organic cations were depleted from the grains and accumulated at grain boundaries after 50 kGy. In contrast, in FAPbI3, FA cations were primarily lost from grain boundaries. The triple-cation (CsMAFA)PbI3 composition exhibited radiation-induced phase segregation into MA- and FA-rich regions, revealing a previously unrecognized degradation pathway. Among all these compositions, FAPbI3 was reported to exhibit exceptional radiation hardness (Fig. 2c), remaining free of metallic lead even under harsh conditions.18 This is a promising finding, as, among the many studies on the composition of MHPs, the FAPbI3 composition has recently been highlighted as the optimal absorber material not only for single-junction PSCs but also for the narrow-bandgap absorber in tandem perovskites.19
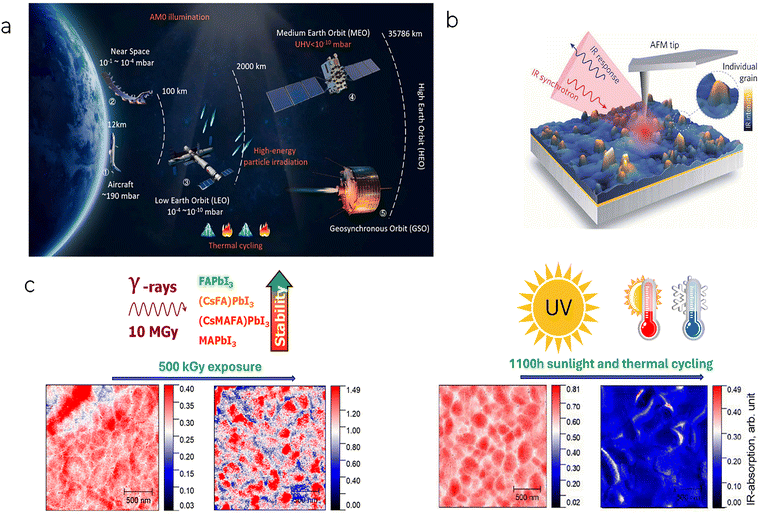 | ||
| Fig. 2 (a) Space environment of various satellite orbits.11 Reproduced with permission. Copyright © 2020 Elsevier Ltd. (b) Broadband IR radiation from a synchrotron is focused onto a metallic tip, which tightly confines the fields at its apex to interact with the sample surface in standard tapping-mode AFM.104 Reproduced under the Creative Commons CC BY-NC license. © 2019 The Authors. Published by the American Association for the Advancement of Science. (c) IR s-SNOM of the MHP films after exposure to gamma-rays and under thermal-cycling and photostability tests.18,105 Reproduced with permission. Copyright © 2025 American Chemical Society. | ||
These findings highlight IR s-SNOM as a powerful method for spatially resolved analysis of degradation pathways and aging mechanisms in perovskite materials, enabling the assessment of ion segregation via nanoscale mapping.20–22
A less explored strategy involving the relatively easy partial replacement of Pb2+ with other elements23 was investigated by Troshin et al., who demonstrated that europium (Eu) or dysprosium (Dy) incorporation suppresses the degradation, showing remarkable radiation resistance under extreme gamma-ray (5.5 MGy) and 8.5 MeV electron exposure (up to 3 × 1016 e− cm−2).10,24 Further extending this strategy, the group systematically investigated partial substitution of Pb2+ in double-cation Cs0.12FA0.88PbI3 perovskites.25 They found that incorporating specific rare-earth cations, such as Nd3+, Er3+, and Tb3+, could dramatically suppress light-induced recrystallization and photodegradation, with the best formulations maintaining their optical properties and phase purity after an impressive 9000 hours of simulated sunlight exposure. Optimized loadings of Er3+ and Zn2+ enabled solar cells that retained 67–70% of their initial efficiency after 5000 hours of continuous operation, underscoring the potential of this approach for enhancing device longevity. The primary effect of the electron beam on perovskites is a reduction in the charge-carrier diffusion length. However, the lowest value is still greater than the thickness of the films used in solar cells, resulting in no significant impact on PSC performance.26 It was also observed that PSCs based on triple-cation perovskites are tolerant to high-energy proton (14 MeV) and electron (>1 MeV) radiation in both glass-based and flexible substrates.14 Tests under 68 MeV proton irradiation demonstrate very low degradation (<6%) at a dosage of 1013 p+ cm−2, where commercially available space PV degrades >22%.27 Another study reports that PSCs are also resilient “fast neutrons” (i.e., with an energy > 10 MeV), which represent one of the most drastic forms of radiation in space environments.12,27 CsFAPbI3 mainly degrades through δ-phase segregation of FAPbI3, forming a Cs-rich phase; Cu+-doped films instead produce an FA-rich phase, demonstrating remarkable resistance to γ-rays and high-energy electrons (8.5 MeV).28,29
Taking advantage of this tolerance, perovskite-based high-energy sensors are increasingly recognized as superior alternatives to conventional chalcogenide-based detectors for gamma- and X-ray detection. They provide exceptional energy resolution, cost efficiency, scalable manufacturing, and enhanced spectral quality.30–32
However, upon exposure to AM1.5 solar-simulated light, MAPbI3 films exhibited a gradual loss of MA+ cations, starting at the grain boundaries and progressing inward toward the grain cores and the bulk material.20–22 Troshin et al. investigated the tunability of the optoelectronic properties of lead halide perovskites through halide mixing. The mixed-halide perovskites tend to be unstable under illumination due to segregation into Br-rich and I-rich domains, leading to performance loss and reduced operational stability. Using IR s-SNOM, they directly visualized the real-time dynamics of segregation, showing that iodoplumbate species are expelled from the grains and form a separate I-rich phase along the grain boundaries.33 Addressing this issue is a fundamental challenge for tandem solar cells. Frolova et al. demonstrated that light-induced halide phase segregation in wide-bandgap CsPbI2Br can be suppressed entirely through partial Pb2+ substitution with Sr2+, Sn2+, In3+, or Bi3+.34 Troshin and colleagues also examined the intrinsic photostability of lead halides and showed that PbI2 undergoes substantial photolysis under prolonged illumination, leading to the formation of metallic lead, whereas PbBr2 remains notably stable under similar conditions. This fundamental difference was linked to the higher vacancy formation energy of PbBr2.35,36
Frolova et al. reduced the photoinduced degradation of complex lead halides through a systematic screening of more than 30 modifying cations introduced as 5% Pb2+ substitutes in the PbI2 lattice. The most effective stabilizing cations preserved the optical absorbance of films and completely prevented Pb0 formation even after 400 hours of continuous illumination. Incorporating optimized ions such as Bi3+, Eu2+/3+, and Ag+ effectively reduces metallic lead formation, thereby supporting the development of highly efficient, durable perovskite solar cells with extended operational lifespans.37,38
In terms of thermal-cycling stability, space cells undergo more rapid daily cycles (light and temperature) and experience greater temperature fluctuations: from −120 to +120 °C every 90 minutes in LEO.39 Mechanical residual strains in multilayer thin-film device stacks become problematic during thermal cycling due to differing thermal expansion coefficients.40 Troshin et al. investigated the stability of PSCs under space-like conditions using an in situ characterization system capable of operating under ultrahigh vacuum (10−8 Pa) and a wide temperature range, thereby excluding the effects of moisture and oxygen. They observed that under repeated thermal cycling (150–430 K), the perovskite absorber irreversibly decomposed. Elevated temperatures also triggered phase transitions and enhanced ion migration across interfaces. These structural and compositional changes resulted in a decline in device PCE primarily due to a reduction in short-circuit current.15
To examine how temperature affects the light-induced degradation of MAPbI3 and PbI2 thin films under anoxic (i.e., inert-atmosphere) conditions, Troshin et al. investigated the effects of temperature on the degradation of these films. The study revealed that both MAPbI3 and PbI2 photolysis exhibit relatively high activation energies, approximately 85 and 106 kJ mol−1, respectively. As a result, lowering the operating temperature from 55 to 30 °C would increase the material's estimated lifetime by more than one to two orders of magnitude.41
Therefore, it is noted that exposing perovskite films to heat and light causes significant degradation, even in the absence of oxygen and moisture, with fully inorganic perovskites being the most stable absorber type.42 Therefore, despite PSCs achieving high efficiency nowadays, they still exhibit intrinsic instability, mainly under light stress and thermal cycling.
The disparities in stability challenges between terrestrial and space applications are dependent on the specific stressors associated with each context. On Earth, the primary concerns include protection against humidity, oxygen, and daily photothermal degradation. Conversely, devices deployed in space are subjected to high-energy particle radiation, ultra-high vacuum conditions, and severe thermal cycling. As a result, the optimal perovskite solar cell may vary depending on the application. For terrestrial use, a wide array of compositional options are available, with interfacial passivation using 2D or quasi-2D structures as a predominant strategy. In space applications, FAPbI3 is considered the most suitable absorber due to its exceptional radiation tolerance, particularly when doped with alternative cations.
However, the MHPs are more stable even under harsh conditions, making them promising for high-altitude applications, such as space PV; they still require improvements in fundamental terrestrial stability issues (Fig. 2c). Some strategies to address these problems are presented in the following sections.
Low-dimensional structures for improving stability
As mentioned above, although PSCs have reached efficiencies of 27%,7 their stability remains a significant barrier to adoption in the photovoltaic market. External factors, including oxygen, moisture, light, and heat, primarily drive PSC degradation.35,36A scalable strategy to address this stability problem is the use of long-chain organic molecules to passivate perovskite grain boundaries, thereby preventing exposure to oxygen and moisture,43–46 a well-known Achilles’ heel of PSCs. When using bulky cation molecules for passivation, their large size prevents them from fitting into the cuboctahedral spaces of a 3D perovskite without disrupting its regular lattice. Once the 3D structure is sliced along different crystallographic planes, it can form lower-dimensional structures,47 such as two-dimensional (2D, n = 1) and quasi-2D (when n > 1) perovskites, as shown in Fig. 3a. In general, there is a reported trend of the refractive index increasing with the number of inorganic layers, along with enhanced thermodynamic stability and charge transfer.48,49 Conversely, due to quantum confinement, the bandgap and work function increase as n decreases.47,50
 | ||
| Fig. 3 (a) Schematic representation of 2D and quasi-2D perovskites and the corresponding trends in their properties.48 Reproduced under the CC-BY 4.0 license. © 2025 The Authors. Published by American Chemical Society. (b) Quantum-well behavior of excitons in 2D and quasi-2D perovskites. (c) Facet-dependent 2D passivation in PSCs employing different organic cations.63 Reproduced with permission. Copyright © 2023 American Chemical Society. (d) General observed trend of 2D phase formation as a function of the organic cation structure. | ||
Since the formation energy of the quasi-2D phase is typically lower than that of the pure 2D phase, it is common to observe a mixture of 2D phases under different confinement regimes, which can sometimes be detrimental to PSC efficiency due to irregularities in the energy landscape.51 Thus, controlling the orientation of 2D layers within 3D–2D heterostructures remains a considerable challenge.52
This difficulty is particularly relevant because 2D halide perovskites exhibit strong anisotropy between in-plane and out-of-plane directions, which directly impacts electronic transport.51,53 The exciton experiences quantum confinement due to reduced dimensionality, as well as dielectric confinement arising from the significant difference in dielectric constants between the inorganic framework and the organic cation layers (see Fig. 3b).53 For this reason, they are also referred to as quantum wells. Although the Förster resonance energy transfer (FRET) mechanism is a plausible mechanism of energy transfer between well-packed films,54 electrically extracting photogenerated charges from these low-dimensional phases is not straightforward.55
A recent study used π-conjugated organic spacers to extract these excitons. The extended cation chain enables π–π stacking and improved orbital alignment between layers, promoting hybridization and facilitating efficient exciton movement throughout the structure.50 There are limited publications on this subject; however, numerous studies focus on controlling 2D formation and stability to prevent charge blockage.55,56
Molecular additives can also be designed to stabilize the 3D perovskite lattice. For instance, Dremova et al. demonstrated that modifying MHP films with isonipecotic acid effectively stabilizes the photoactive phase, leading to remarkable long-term operational stability in solar cells.57 This highlights the potential of targeted molecular engineering as a versatile tool for enhancing phase stability without necessarily inducing low-dimensional structuring. The growth of the 2D phase is reported to be governed by the concentration of the bulky cation, the pKa of the ammonium group, and the subsequent thermal annealing.47,58,59 However, the formation and stability of 2D and quasi-2D perovskite layers are also strongly influenced by the chemical structure of the organic spacer cation.48 Cations with extended alkyl chains promote the formation of the 2D perovskite phase and augment steric hindrance, thereby improving thermal stability. They impede the transition to higher-dimensional phases during thermal aging, which are advantageous for the overall performance.60,61 Choghaei et al. recently investigated the influence of alkylammonium spacer cation length on the electronic and optical properties of 2D perovskites. They observed notable odd–even effects, in which even-numbered chains exhibit efficient packing, whereas odd-numbered chains cause distortions in the Pb–I–Pb bond angles, thereby affecting ionization energy, optical bandgap, and charge-carrier mobility.62 Park et al. observed that passivation occurs selectively on specific facets of the MHP (Fig. 3c). The atomic configuration of each facet influences the orientation of the passivation layer. This facet dependence facilitated the identification of 2-amidinopyridine hydroiodide as an effective passivation agent, capable of providing uniform coverage on both the (100) and (111) facets.63 Recent studies by Nogueira and colleagues indicate that chain flexibility, rather than length alone, critically influences phase purity and crystallization pathways, with flexible cations favoring the formation of mixed-n phases.58 Investigations have been conducted into the structural implications of substituting the phenethylammonium (PEA) spacer with a bromine atom at different positions on the phenyl ring in two-dimensional hybrid perovskites (2D MHPs). 2D MHPs with a ‘gauche’ conformation exhibit slower dynamics and stronger hydrogen-bonding interactions with inorganic frameworks, leading to lower band gaps, lower defect densities, and superior thermal stability.64
Therefore, both the formation and stability of 2D layers are complex and dynamic. To address these complexities in 2D/3D compositions, advanced in situ techniques such as photoluminescence (PL) and grazing-incidence wide-angle X-ray scattering (GIWAXS) are increasingly employed, enabling real-time monitoring of phase formation and transformation and providing insights into crystallization and 2D/3D interfacial dynamics.59,65 Also, due to the low concentration of the salts commonly used to passivate MHPs, it is not always possible to confirm or rule out the formation of 2D phases using traditional methods such as XRD or PL techniques.47 In this context, synchrotron-based techniques can aid in elucidating the passivation mechanism of low-dimensional materials.20,66
For instance, Nogueira et al. reported that 2D/3D perovskite interfaces undergo dynamic structural transformations under thermal stress, in which the 2D layer partially converts to a mixed 2D/3D phase, while the 3D bulk remains intact. In situ synchrotron X-ray scattering revealed that this process suppresses lead iodide formation and maintains device performance, underscoring the importance of 2D passivation in enhancing thermal stability and extending device lifetime.67 A recent study by Tan et al. demonstrated that the octylammonium-based 2D/3D perovskite heterostructure undergoes dynamic evolution during device degradation. Initially, phase-pure 2D interlayers evolve along distinct pathways, leading to varied device stabilities. By employing mixed solvents to modulate crystallinity and phase purity, they achieved robust 2D interlayer formation. The optimized 2D/3D devices reached a power conversion efficiency of 25.9% and maintained 91% of their initial performance after more than 1000 hours at 85 °C under maximum power point tracking.16,68
Recent studies by Nogueira et al. using in situ PL also demonstrated that a higher ratio of quasi-2D phases improves device performance. In contrast, pure 2D layers can hinder current extraction due to the quantum-well behavior described above. In this context, highly concentrated 2-thiophenemethylammonium (TMA) provides more effective surface passivation than PEA due to a more balanced formation energy among 2D and quasi-2D phases.69 In this context, the use of small secondary ammonium cations, such as dipropylammonium (DPA), has been reported to yield materials with n > 10 that are not only efficient and thermodynamically stable but also robust under heat stress.70
In the context of bottom passivation (buried interface), a recent study introduces interfacial guanidine-based molecular layers onto NiOx. The synergistic effect of functional groups that interact with Ni species and surface hydroxyls enhances hole extraction. Concurrently, the guanidine groups interact with residual PbI2 at the buried perovskite interface, promoting the formation of a two-dimensional perovskite that passivates defects associated with Pb2+ and iodine vacancies, thereby minimizing ion migration.71
It is also important to point out that some bulky organic ions are reported to be effective passivating agents but do not form 2D perovskites (Fig. 3d).72–75 These compounds can passivate the grain boundaries of the MHP film without creating a crystalline phase, a process referred to as ‘amorphous passivation’.66 A recent study compared two fluorinated benzamidine salts, both containing the same perovskite anchoring group, benzamidine, but differing in the degree of fluorination at the opposite extremity: a monofluoro and a trifluoro derivative. The less fluorinated cation formed a 3D/2D heterojunction, whereas the more fluorinated salt produced an amorphous passivation layer on the perovskite films. Nevertheless, the more fluorinated salt achieved an efficiency of 24%. Interestingly, the 2D passivated perovskite devices demonstrated greater longevity under various storage conditions.66
Even lower-dimensional systems, such as 1D and 0D structures, tend to form in molecules with highly sterically hindered ammonium groups or in bulkier A′ cations. 1D and 0D perovskites are promising candidates; however, research on them is still in its early stages.47 A possible passivation strategy is to form the 0D phase, such as perovskite quantum dots (PQDs), and then deposit them onto the MHP film to create the heterojunction. Recently, Fonseca et al. reported the deposition of CsPbBr3 PQDs onto a triple-cation MHP for n–i–p configuration PSCs.76 Passivation of interfacial defects and grain boundaries enhanced charge injection, thereby improving efficiency. A key aspect was the 3D/0D halide exchange, which, in this case, did not have the adverse effects often reported in the literature. The hysteresis remained unaffected, and the halide movement helped to maintain halide equilibrium under stress, likely acting as an ion reservoir.76
The literature remains controversial about whether low-dimensional (LD) structures improve or hinder PSC stability. We believe this stems from sensitive processing parameters that determine the phase distribution, n-value, and crystallographic orientation. These factors are often under-reported in the literature. Studies reveal that 2D/quasi-2D passivation improves stability when spacer cations remain confined to surfaces in low-n well-oriented phases that block moisture and/or ion ingress while allowing charge extraction. However, the same interfaces can destabilize devices when spacers intercalate into the bulk, form high-n phases, or adopt random orientations that introduce energetic barriers and trap states. Furthermore, post-fabrication migration progressively increases n-values, causing initially beneficial passivation layers to evolve into less stable phases under thermal stress, explaining why efficiency improvements are reproduced, but stability gains are not. Reproducibility failures may arise because variations in precursor stoichiometry, ligand purity, solvent timing, and annealing protocols determine whether spacers remain at the surface or diffuse into the bulk of the MHP, yet these processing details are not always well documented with sufficient precision for replication.
In general, some open questions remain regarding the use of LD materials for PSC passivation, which can undergo (or not) the formation of a heterostructure, including:
• Spatial distribution: the arrangement of 2D layers, particularly at grain boundaries, and their evolution under thermal stress, is still not fully understood.
• Engineered heteroatom ligands: fluorination and mixed-cation treatments could thermodynamically favor stable low-n phases and prevent ion migration.
• n-value control: limiting interdiffusion enables more precise regulation of the n-value, potentially leading to higher-purity structures and expanding the range of possible device designs.
• Band alignment: adjusting the n-value, combined with A′-site engineering, enables fine-tuning of band offsets between 3D and 2D domains and the regulation of the quantum wells.
• Functional bulky cations: designing bulky cations that simultaneously passivate the perovskite interface and introduce complementary functionalities, such as enhanced hydrophobicity, dipole-induced field effects, or energy-level alignment, could improve both stability and efficiency.
• 2D/3D interface dynamics: the 2D/3D interface is highly dynamic and changes after thermal stress, with the type of organic cation playing a significant role. Can such interfaces withstand current accelerated heating tests and/or outdoor conditions?
Quantum dots – tiny building blocks for stable solar cells?
Beyond layered 2D/3D architectures, zero-dimensional PQDs offer an alternative and complementary approach to advancing perovskite photovoltaic technology. One of the most attractive features of PQDs is their solution crystallinity. They are pre-crystallized materials, ready for deposition, suitable for printing techniques, and offer additional opportunities through room-temperature processing, serving as building blocks for the deposition of perovskite-absorbing layers.77 QD inks could, for example, enable space missions by providing pre-crystallized materials for further deposition in new PSCs or even for repairing damaged devices.Usually, the synthesis of these PQDs is carried out via the hot injection (HI) method, in which the long-chain ligand-passivated FA precursor is injected into a solution of Pb and I salts at high temperature.78,79 However, the HI synthesis typically requires high temperatures, an inert gas atmosphere, and long-chain insulating ligands, which limit scalability and practical applications. To overcome these drawbacks, Yuan et al. reported a scalable and straightforward method for preparing PQDs under ambient conditions, thereby eliminating the need for a polar solvent and yielding a 40-fold increase in production. The resulting short-ligand-passivated PNC inks are compatible with various solution-deposition techniques, such as spray coating, allowing for the fabrication of large-area films.80
Once the PQDs are prepared, a suitable method for altering their composition is to utilize the fast ionic exchange enabled by their ionic character.16,81 Yuan et al. explored fast cross-cation exchange in PQDs based on MA+ and FA+. The mixed-cation PQDs demonstrate enhanced photostability and can be used directly in one-step-coated photovoltaic devices, achieving a PCE of 15.05% with MA0.5FA0.5PbI3 PQDs.82 The halide composition can also be tuned through simple ion-exchange post-treatments.83
Among the PQD compositions, FAPbI3 is distinguished by its narrow band gap, high ambient stability, and extended carrier lifetimes, making it an attractive candidate for high-performance QD photovoltaics. However, the efficiency and stability of PQD photovoltaic technologies are significantly limited by a trade-off between carrier transport blockage caused by protective ligands and nonradiative recombination losses due to defects or trap states when researchers attempt to remove these ligands in order to improve dot-to-dot charge coupling during film formation.84–86 A common strategy employed by many researchers is to first synthesize high-quality PQDs and then perform ligand exchange, which can be carried out either in the liquid phase or in the solid state after film formation.87,88
Kong et al. recently developed an organic–inorganic hybrid active layer combining a conjugated polymer (PCDOT-T) with FAPbI3 QDs, providing complementary absorption and a favorable interface/morphology for efficient charge separation and transport. The champion device achieves a record efficiency of 13.11% using a one-step-coated organic/QD hybrid bulk heterojunction blend.89 Yuan et al. recently explored dual-site molecular ligands based on thiophene amines utilizing a dual-phase synergistic ligand exchange (DSLE) method in both the solution and solid states. The native insulating ligands (OA/OAm) of FAPbI3 PQDs were successfully exchanged, resulting in efficient electronic coupling and charge transport, yielding a PCE of 18.21%. The devices also exhibited enhanced stability, with a T80 of ∼1400 h (20–30% humidity, 25 °C).84
Recently, Yuan et al. employed a passivation strategy based on exchange with a fluorinated pseudo-halide anion ligand, hexafluorophosphate (PF6−), for FAPbI3 PQDs. The pseudo-halide simultaneously passivates iodide vacancies, enhances electronic coupling by reducing the interdot distance, suppresses ion migration, and provides a hydrophobic barrier. An unprecedented 19.01% efficiency is achieved with good operational stability.90
It is well known that buried interface engineering in PSCs actively directs the crystallization of bulk-MHP films, controlling grain orientation, suppressing phase segregation under stress, and enhancing charge extraction; this also applies to PQDs.91–93 Yuan et al. reported the use of a cyclic passivator (CyP) for modifying buried interfaces. CyP's ability to bind chemically to defect sites results from its isotropic nature, which enhances defect passivation and facilitates effective interfacial charge extraction and transport, leading to a PCE of 17.50% for CsPbI3 PQD solar cells. Furthermore, the CyP-assisted buried-interface modulation enables the reuse of costly TiO2/FTO electrodes, thereby supporting the refurbishment of QD solar cells, maintaining more than 90% of their initial efficiency after four cycles of device recycling.94
Nevertheless, recent progress in PQD inks helps address the limitations of the traditional, labor-intensive layer-by-layer fabrication process, which is a significant barrier to the development of scalable, printable QD solar cells, as reported by Yuan et al. They prepared conductive PQD inks by introducing a sequential acylation–coordination technique that combines Lewis base-mediated surface restoration with amine-assisted ligand removal. Without the need for solid-state ligand exchange, this method enables direct one-step film deposition, producing films with a homogeneous energy landscape, improved electronic coupling, uniform morphology, and structural order. Consequently, FAPbI3 QD solar cells outperformed devices prepared with traditional QD inks and layer-by-layer techniques, achieving a champion efficiency of 16.61% (certified: 16.20%). Additionally, the conductive inks demonstrated compatibility with scalable blade coating, enabling the high-speed fabrication of large-area devices (up to 50 mm s−1 and 9 × 9 cm2).95
The use of artificial intelligence (AI), especially machine learning (ML) models, in the context of QDs is currently being widely explored.96–98 Some researchers are applying these concepts in real time to optimize the synthesis of PQDs in microfluidic synthesis.99–101 ML-assisted synthesis not only provides insight into the nucleation, growth, and ripening processes but also enables the precise preparation of PQDs with targeted emission wavelength and full width at half maximum (FWHM) under optimized, time- and energy-efficient, and environmentally friendly conditions (Fig. 4).102 With the rapid advancements in AI, deep learning, reinforcement learning, and large language models, research in PSCs is increasingly shifting from a predominantly data-driven to an intelligence-driven approach, automating the production of materials up to device assembly using automated platforms.98,103
 | ||
| Fig. 4 Summary of the state-of-the-art applications of PQDs in photovoltaics.106 Reproduced with permission from the Royal Society of Chemistry. Copyright © 2025. | ||
Conclusions
The BRICS nations are assuming an increasingly strategic role in the global transition to renewable energy, accounting for over half of the world's solar electricity generation in 2024. In this context, halide perovskite photovoltaics emerge as transformative technologies capable of democratizing access to clean energy owing to their low fabrication costs, tunable optoelectronic properties, and rapidly advancing efficiency metrics. Nevertheless, despite significant performance gains, the intrinsic and extrinsic stability of perovskite materials continues to pose limitations to their widespread adoption, particularly under operational and environmental stress.Discussions at a BRICS joint project workshop on perovskite technologies underscored the consensus that achieving commercial viability requires shifting the research emphasis from record-breaking efficiencies to durability, reproducibility, and scalability.
Notably, PSCs exhibit an intriguing paradox: while they demonstrate exceptional stability under extreme space conditions (radiation doses up to 10 MGy and thermal cycling from −120 °C to 120 °C), they degrade rapidly under moderate terrestrial conditions (humidity and modest thermal cycles). This dichotomy suggests that moisture- and oxygen-induced degradation mechanisms are more critical than thermal stress or high-energy radiation. Understanding this apparent contradiction is essential for designing more effective encapsulation and stabilization strategies.
Innovative methods for analyzing stability now could be based on characterization techniques, including IR s-SNOM, which facilitates nanoscale spatial mapping of degradation mechanisms, and in situ synchrotron radiation analyses for real-time monitoring of structural transformations. Furthermore, enhanced durability can be achieved by integrating the recent advances in AI to accelerate the discovery of stable materials.
The integration of advanced characterization techniques, especially in situ and synchrotron-based techniques, offers a pathway to expedite the discovery of stable compositions and interfaces. Low-dimensional engineering, employing 2D, quasi-2D, and 0D nanostructures, has demonstrated the ability to suppress ion migration, passivate defects, and enhance thermal and photochemical stability. Furthermore, recent advances in ligand chemistry and quantum dot inks are enabling scalable, low-temperature processes compatible with industrial coating methodologies. By synergistically integrating these approaches, BRICS nations have a unique opportunity to help the transition from “efficiency to resilience,” which represents not merely a scientific challenge but also a strategic imperative for energy security and environmental sustainability.
Conflicts of interest
There are no conflicts to declare.Data availability
No new experimental data were generated or analysed in this Perspective. All information discussed is based on previously published studies, which are cited in the article.Acknowledgements
The authors thank all participants in the I BRICS Workshop on Perovskite Technologies for their valuable discussions, which contributed to this work. A. F. V. F. acknowledges support from the São Paulo Research Foundation (FAPESP) under grants 2023/10395-4 and 2024/05914-5. A. F. N. acknowledges the support from FAPESP (grant 2017/11986-5), CNPq (grant 440177/2022-7), Shell, and the strategic importance of the support provided by ANP (Brazil's National Oil, Natural Gas, and Biofuels Agency). J. Yuan acknowledges the support from the Natural Science Foundation of China (No. 52261145696). P. A. T. and L. A. F. acknowledge the support from the Russian Science Foundation (project no. 19-73-30020P). Grammarly software was used to assist with grammar and spelling checks during the preparation of this work.References
- A. F. Nogueira, G. Doubek and H. Zanin, Mater. Adv., 2025, 6, 3368–3370 RSC.
- Q. Sun, R. Ma, Z. Xi, H. Wang, C. Jiang and H. Chen, J. Clean. Prod., 2023, 396, 136488 CrossRef.
- International Monetary Fund (IMF) Database, https://data.imf.org/ (accessed 1 September 2025).
- S. Ali, N. S. Al-Nassar, M. I. Sindhu and M. Naveed, Res. Int. Bus. Finance, 2025, 74, 102698 CrossRef.
- N. Fulghum, C. Bruce-Lockhart and L. Orso, Solar BRICS: Emerging economies now lead the world's clean energy race, Ember, 3 July 2025, https://ember-energy.org/latest-insights/solar-brics-emerging-economies-now-lead-the-worlds-clean-energy-race/ (accessed 3 March 2026).
- O. Almora, G. C. Bazan, C. I. Cabrera, L. A. Castriotta, S. Erten-Ela, K. Forberich, K. Fukuda, F. Guo, J. Hauch, A. W. Y. Ho-Baillie, T. J. Jacobsson, R. A. J. Janssen, T. Kirchartz, R. R. Lunt, X. Mathew, D. B. Mitzi, M. K. Nazeeruddin, J. Nelson, A. F. Nogueira, U. W. Paetzold, B. P. Rand, U. Rau, T. Someya, C. Sprau, L. Vaillant-Roca and C. J. Brabec, Adv. Energy Mater., 2024, 15, 2404386 CrossRef.
- NREL, Chart of Best Research-Cell Efficiencies Provided by NREL, https://www.nrel.gov/pv/cell-efficiency.html (accessed August 2025) Search PubMed.
- M. V. Khenkin, E. A. Katz, A. Abate, G. Bardizza, J. J. Berry, C. Brabec, F. Brunetti, V. Bulović, Q. Burlingame, A. Di Carlo, R. Cheacharoen, Y. B. Cheng, A. Colsmann, S. Cros, K. Domanski, M. Dusza, C. J. Fell, S. R. Forrest, Y. Galagan, D. Di Girolamo, M. Grätzel, A. Hagfeldt, E. von Hauff, H. Hoppe, J. Kettle, H. Köbler, M. S. Leite, S. (Frank) Liu, Y. L. Loo, J. M. Luther, C. Q. Ma, M. Madsen, M. Manceau, M. Matheron, M. McGehee, R. Meitzner, M. K. Nazeeruddin, A. F. Nogueira, Ç. Odabaşı, A. Osherov, N. G. Park, M. O. Reese, F. De Rossi, M. Saliba, U. S. Schubert, H. J. Snaith, S. D. Stranks, W. Tress, P. A. Troshin, V. Turkovic, S. Veenstra, I. Visoly-Fisher, A. Walsh, T. Watson, H. Xie, R. Yıldırım, S. M. Zakeeruddin, K. Zhu and M. Lira-Cantu, Nat. Energy, 2020, 5, 35–49 CrossRef.
- Y. Liu, Z. Zhang, T. Wu, W. Xiang, Z. Qin, X. Shen, Y. Peng, W. Shen, Y. Li and L. Han, Nanomicro Lett., 2025, 17, 1–11 CAS.
- M. I. Ustinova, L. A. Frolova, A. V. Rasmetyeva, N. A. Emelianov, M. N. Sarychev, G. V. Shilov, P. P. Kushch, N. N. Dremova, G. A. Kichigina, A. I. Kukharenko, D. P. Kiryukhin, E. Z. Kurmaev, I. S. Zhidkov and P. A. Troshin, J. Mater. Chem. A: Mater., 2024, 12, 13219–13230 RSC.
- J. Yang, Q. Bao, L. Shen and L. Ding, Nano Energy, 2020, 76, 105019 CrossRef CAS.
- G. M. Paternò, V. Robbiano, L. Santarelli, A. Zampetti, C. Cazzaniga, V. Garcìa Sakai and F. Cacialli, Sustainable Energy Fuels, 2019, 3, 2561–2566 RSC.
- L. McMillon-Brown, J. M. Luther and T. J. Peshek, ACS Energy Lett., 2022, 7, 1040–1042 CrossRef CAS.
- D. Angmo, S. Yan, D. Liang, A. D. Scully, A. S. R. Chesman, M. Kellam, N. W. Duffy, N. Carter, R. Chantler, C. Chen and M. Gao, ACS Appl. Energy Mater., 2024, 7, 1777–1791 CrossRef CAS.
- X. Lu, Z. Wang, Q. Lin, J. Sheng, A. F. Akbulatov, S. M. Aldoshin, P. Troshin, Y. Shi and L. Li, Authorea, 2025 DOI:10.22541/au.175547361.10502563/v1.
- J. Y. Kim, J. W. Lee, H. S. Jung, H. Shin and N. G. Park, Chem. Rev., 2020, 120, 7867–7918 CrossRef CAS PubMed.
- L. Wu, S. Hu, F. Yang, G. Li, J. Wang, W. Zuo, J. J. Jerónimo-Rendon, S.-H. Turren-Cruz, M. Saba, M. Saliba, M. K. Nazeeruddin, J. Pascual, M. Li and A. Abate, Nat. Rev. Mater., 2025, 10, 536–549 CrossRef.
- V. V. Ozerova, N. A. Emelianov, D. P. Kiryukhin, P. P. Kushch, G. V. Shilov, G. A. Kichigina, S. M. Aldoshin, L. A. Frolova and P. A. Troshin, J. Phys. Chem. Lett., 2023, 14, 743–749 CrossRef CAS PubMed.
- Y. Ahmed, X. Qi, P. Moazzezi and M. I. Saidaminov, ACS Energy Lett., 2026, 11(1), 79–89, DOI:10.1021/acsenergylett.5c03275.
- M. G. D. Guaita, R. Szostak, F. M. C. da Silva, A. de Morais, R. F. Moral, T. Kodalle, V. C. Teixeira, C. M. Sutter-Fella, H. C. N. Tolentino and A. F. Nogueira, Adv. Funct. Mater., 2024, 34, 1–11 CrossRef.
- N. A. Emelianov, V. V. Ozerova, Y. S. Fedotov, M. V. Zhidkov, R. R. Saifutyarov, M. S. Malozovskaya, M. S. Leshchev, E. V. Golosov, L. A. Frolova and P. A. Troshin, Materials, 2023, 16, 1–10 CrossRef PubMed.
- M. Lee, L. Wang, D. Zhang, J. Li, J. Kim, J. S. Yun and J. Seidel, Adv. Mater., 2024, 36, 2407291, DOI:10.1002/adma.202407291.
- T. A. de Souza Carvalho, L. F. Magalhaes, C. I. do Livramento Santos, T. A. Z. de Freitas, B. R. Carvalho Vale, A. F. Vale da Fonseca and M. A. Schiavon, Chem. – Eur. J., 2023, 29, e202202518 CrossRef CAS PubMed.
- M. I. Ustinova, L. A. Frolova, A. V. Rasmetyeva, N. A. Emelianov, M. N. Sarychev, P. P. Kushch, N. N. Dremova, G. A. Kichigina, A. I. Kukharenko, D. P. Kiryukhin, E. Z. Kurmaev, I. S. Zhidkov and P. A. Troshin, Chem. Eng. J., 2024, 493, 152522 CrossRef CAS.
- M. I. Ustinova, G. V. Shilov, D. V. Korchagin, N. N. Dremova, P. A. Troshin, S. M. Aldoshin and L. A. Frolova, Mendeleev Commun., 2025, 35, 569–572 CrossRef CAS.
- D. Pérez-del-Rey, C. Dreessen, A. M. Igual-Muñoz, L. van den Hengel, M. C. Gélvez-Rueda, T. J. Savenije, F. C. Grozema, C. Zimmermann and H. J. Bolink, Sol. RRL, 2020, 4, 1–6 Search PubMed.
- F. Lang, G. E. Eperon, K. Frohna, E. M. Tennyson, A. Al-Ashouri, G. Kourkafas, J. Bundesmann, A. Denker, K. G. West, L. C. Hirst, H. C. Neitzert and S. D. Stranks, Adv. Energy Mater., 2021, 11, 2102246, DOI:10.1002/aenm.202102246.
- M. I. Ustinova, M. N. Sarychev, N. A. Emelianov, Y. Li, Y. Zhuo, T. Zheng, S. D. Babenko, E. D. Tarasov, P. P. Kushch, N. N. Dremova, G. A. Kichigina, A. V. Rasmetyeva, A. I. Kukharenko, D. P. Kiryukhin, E. Z. Kurmaev, X. Xu, P. A. Troshin, L. A. Frolova and I. S. Zhidkov, EcoMat, 2025, 7, e12512 CrossRef CAS.
- M. I. Ustinova, A. V. Rasmetyeva, A. I. Kukharenko, M. V. Lobanov, P. P. Kushch, N. A. Emelianov, D. V. Korchagin, G. A. Kichigina, M. N. Sarychev, D. P. Kiryukhin, E. Z. Kurmaev, P. A. Troshin, L. A. Frolova and I. S. Zhidkov, Mater. Today Energy, 2024, 45, 101687 CrossRef CAS.
- G. Kakavelakis, M. Gedda, A. Panagiotopoulos, E. Kymakis, T. D. Anthopoulos and K. Petridis, Adv. Sci., 2020, 7, 2002098, DOI:10.1002/advs.202002098.
- N. Shen, X. He, T. Gao, B. Xiao, Y. Wang, R. Ren, H. Qin, K. S. Bayikadi, Z. Liu, J. A. Peters, B. W. Wessels, L. Wang, X. Ouyang, S. Wei, Q. Sun, X. Liu, Y. Lai, X. Ouyang, Z. Chai, M. G. Kanatzidis and Y. He, Nat. Commun., 2025, 16, 8113, DOI:10.1038/s41467-025-63400-7.
- M. H. Miah, M. U. Khandaker, P. Saengkaew, A. Numan and M. A. Islam, RSC Adv., 2025, 15, 43446–43486 RSC.
- L. A. Frolova, S. Y. Luchkin, Y. Lekina, L. G. Gutsev, S. A. Tsarev, I. S. Zhidkov, E. Z. Kurmaev, Z. X. Shen, K. J. Stevenson, S. M. Aldoshin and P. A. Troshin, Adv. Energy Mater., 2021, 11, 2002934 CrossRef CAS.
- M. I. Ustinova, G. V. Shilov, D. V. Korchagin, P. A. Troshin, S. M. Aldoshin and L. A. Frolova, Mendeleev Commun., 2025, 35, 573–576 CrossRef CAS.
- I. S. Zhidkov, A. F. Akbulatov, A. I. Poteryaev, A. I. Kukharenko, A. V. Rasmetyeva, L. A. Frolova, P. A. Troshin and E. Z. Kurmaev, Coatings, 2023, 13, 2–9 Search PubMed.
- A. F. Akbulatov, M. I. Ustinova, L. Gutsev, S. A. Tsarev, N. N. Dremova, I. Zhidkov, S. Y. Luchkin, B. R. Ramachandran, L. Frolova, E. Z. Kurmaev, K. J. Stevenson, S. M. Aldoshin and P. A. Troshin, Nano Energy, 2021, 86, 106082 CrossRef CAS.
- M. I. Ustinova, G. V. Shilov, P. A. Troshin, S. M. Aldoshin and L. A. Frolova, Inorganics, 2025, 13(1), 13, DOI:10.3390/inorganics13010013.
- M. I. Ustinova, M. V. Lobanov, G. V. Shilov, N. N. Dremova, A. F. Akbulatov, L. G. Gutsev, I. S. Zhidkov, E. Z. Kurmaev, F. A. Prudnov, A. V. Ivanov, L. A. Frolova, S. M. Aldoshin and P. A. Troshin, Adv. Funct. Mater., 2025, 35, 2407571 CrossRef CAS.
- A. W. Y. Ho-Baillie, H. G. J. Sullivan, T. A. Bannerman, H. P. Talathi, J. Bing, S. Tang, A. Xu, D. Bhattacharyya, I. H. Cairns and D. R. McKenzie, Adv. Mater. Technol., 2022, 7, 2101059, DOI:10.1002/admt.202101059.
- M. Chen, Y. Dong, Y. Zhang, X. Zheng, G. R. McAndrews, Z. Dai, Q. Jiang, S. You, T. Liu, S. P. Harvey, K. Zhu, V. Oliveto, A. Jackson, R. Witteck, L. M. Wheeler, N. P. Padture, P. J. Dyson, M. D. McGehee, M. K. Nazeeruddin, M. C. Beard and J. M. Luther, ACS Energy Lett., 2024, 9, 2582–2589 CrossRef CAS.
- A. F. Akbulatov, M. I. Ustinova, G. V. Shilov, N. N. Dremova, I. S. Zhidkov, E. Z. Kurmaev, L. A. Frolova, A. F. Shestakov, S. M. Aldoshin and P. A. Troshin, J. Phys. Chem. Lett., 2021, 12, 4362–4367 CrossRef CAS.
- A. F. Akbulatov, L. A. Frolova, N. N. Dremova, I. Zhidkov, V. M. Martynenko, S. A. Tsarev, S. Y. Luchkin, E. Z. Kurmaev, S. M. Aldoshin, K. J. Stevenson and P. A. Troshin, J. Phys. Chem. Lett., 2020, 11, 333–339 CrossRef CAS PubMed.
- J. S. Yun, J. Kim, T. Young, R. J. Patterson, D. Kim, J. Seidel, S. Lim, M. A. Green, S. Huang and A. Ho-Baillie, Adv. Funct. Mater., 2018, 28, 1705363 CrossRef.
- Q. An, F. Paulus, D. Becker-Koch, C. Cho, Q. Sun, A. Weu, S. Bitton, N. Tessler and Y. Vaynzof, Matter, 2021, 4, 1683–1701 CrossRef CAS.
- D. W. DeQuilettes, S. M. Vorpahl, S. D. Stranks, H. Nagaoka, G. E. Eperon, M. E. Ziffer, H. J. Snaith and D. S. Ginger, Science, 2015, 348, 683–686, DOI:10.1126/science.aaa5333.
- F. L. de Araujo, M. Stefanelli, A. Agresti, S. Pescetelli, A. Di Vito, M. A. Der Maur, L. Vesce, A. F. Nogueira and A. Di Carlo, Nano Energy, 2025, 142, 111279, DOI:10.1016/j.nanoen.2025.111279.
- L. Scalon and Y. Vaynzof, Adv. Energy Mater., 2025, 2502686, DOI:10.1002/aenm.202502686.
- I. C. Ribeiro, F. D. Picoli, P. I. R. Moraes, A. F. V. Fonseca, L. N. Oliveira, A. F. Nogueira and J. L. F. Da Silva, ACS Appl. Energy Mater., 2025, 8, 3346–3359 CrossRef CAS.
- J. Ryeom, D. Y. Park, Y. M. Bahk, G. Choi and M. S. Jeong, J. Alloys Compd., 2025, 1030, 180811 CrossRef CAS.
- Y. Boeije, F. Lie, M. Dubajić, E. Garip, A. Maufort, R. I. Biega, S. Lenaers, M. Sauty, P. Ghosh, A. Radić, A. Loher, P. La Magna, H. Salway, A. Ashoka, X. W. Chua, Q. Gu, K. Van Hecke, L. Lutsen, D. Vanderzande, A. Rao, W. T. M. Van Gompel, L. Leppert and S. D. Stranks, J. Am. Chem. Soc., 2025, 147, 31541–31557 CrossRef CAS PubMed.
- Z. Shi, S. Liu, R. Luo, J. Ma, H. Tian, X. Wang, Z. Dong, X. Guo, J. Chen, J. Feng, C. Xiao, Y. Wu, W. Hu and Y. Hou, J. Am. Chem. Soc., 2025, 147(1), 1055–1062, DOI:10.1021/jacs.4c14473.
- M. G. D. Guaita, R. Szostak, F. M. C. da Silva, Z. Feng, L. Scalon, V. C. Teixeira, T. Kodalle, C. M. Sutter-Fella, S. S. Jang, H. C. N. Tolentino and A. F. Nogueira, Sol. RRL, 2025, 9, 1–16, DOI:10.1002/solr.202500404.
- I. Metcalf, S. Sidhik, H. Zhang, A. Agrawal, J. Persaud, J. Hou, J. Even and A. D. Mohite, Chem. Rev., 2023, 123, 9565–9652 CrossRef CAS PubMed.
- R. F. Moral, A. A. Malfatti-Gasperini, L. G. Bonato, B. R. C. Vale, A. F. V. Fonseca, L. A. Padilha, C. L. P. Oliveira and A. F. Nogueira, Mater. Horiz., 2023, 10, 5822–5834 RSC.
- R. Wang, X. Dong, Q. Ling, Z. Hu, Y. Gao, Y. Chen and Y. Liu, Angew. Chem., Int. Ed., 2023, 62, e202314690, DOI:10.1002/ange.202314690.
- Y. Tang, X. Dong, X. Li, Y. Zhou, C. Ran and Z. Wu, Chem. Eng. J., 2024, 499, 156503 CrossRef CAS.
- N. N. Dremova, G. V. Shilov, P. A. Troshin and L. A. Frolova, Mendeleev Commun., 2024, 34, 481–483 CrossRef CAS.
- L. Scalon, C. A. Nogueira, A. F. V. Fonseca, P. E. Marchezi, R. F. Moral, G. Grancini, T. Kodalle, C. M. Sutter-Fella, C. C. Oliveira, L. F. Zagonel and A. F. Nogueira, ACS Appl. Mater. Interfaces, 2024, 16, 51727–51737 CrossRef CAS PubMed.
- T. Kodalle, M. M. Byranvand, M. Goudreau, C. Das, R. Roy, M. Kot, S. Briesenick, M. Zohdi, M. Rai, N. Tamura, J. I. Flege, W. Hempel, C. M. Sutter-Fella and M. Saliba, Adv. Mater., 2024, 36, 1–12 CrossRef PubMed.
- J. Zhang, J. Wu, S. Langner, B. Zhao, Z. Xie, J. A. Hauch, H. A. Afify, A. Barabash, J. Luo, M. Sytnyk, W. Meng, K. Zhang, C. Liu, A. Osvet, N. Li, M. Halik, W. Heiss, Y. Zhao and C. J. Brabec, Adv. Funct. Mater., 2022, 32, 1–9 Search PubMed.
- B. P. Kore, W. Zhang, B. W. Hoogendoorn, M. Safdari and J. M. Gardner, Commun. Mater., 2021, 2, 1–10 Search PubMed.
- M. Choghaei, M. Schiffer, V. Tyagi, M. Righetto, J. Du, M. Buchmüller, K. O. Brinkmann, G. Brocks, P. Görrn, L. M. Herz, S. Tao, T. Riedl and S. Olthof, J. Mater. Chem. A, 2025, 13, 18935–18947 RSC.
- C. Ma, M. C. Kang, S. H. Lee, Y. Zhang, D. H. Kang, W. Yang, P. Zhao, S. W. Kim, S. J. Kwon, C. W. Yang, Y. Yang and N. G. Park, J. Am. Chem. Soc., 2023, 145, 24349–24357 CrossRef CAS PubMed.
- B. Toon, S. Ong, B. Febriansyah, T. J. N. Hooper, A. Kurkin, Y. Lekina, Y. Li, Z. Tan, N. Mathews and Z. Shen, Chem. Mater., 2025, 37(18), 7178–7192, DOI:10.1021/acs.chemmater.5c01245.
- R. Szostak, A. de Souza Gonçalves, J. N. de Freitas, P. E. Marchezi, F. L. de Araújo, H. C. N. Tolentino, M. F. Toney, F. das Chagas Marques and A. F. Nogueira, Chem. Rev., 2023, 123, 3160–3236 CrossRef CAS PubMed.
- X. Zheng, S. Ahmed, Y. Zhang, G. Xu, J. Wang, D. Lu, T. Shi, J. Tang, L. Yan, W. Chen, P. Han, Z. Liu, D. Khan, X. Wang and Z. Tang, Nano-Micro Lett., 2026, 18, 62, DOI:10.1007/s40820-025-01913-y.
- A. A. Sutanto, R. Szostak, N. Drigo, V. I. E. Queloz, P. E. Marchezi, J. C. Germino, H. C. N. Tolentino, M. K. Nazeeruddin, A. F. Nogueira and G. Grancini, Nano Lett., 2020, 20, 3992–3998 CrossRef CAS PubMed.
- S. Tan, M. C. Shih, Y. Lu, S. G. Choi, Y. Dong, J. H. Lee, I. Yavuz, B. W. Larson, S. Y. Park, T. Kodalle, R. Zhang, M. J. Grotevent, Y. K. Lin, H. Zhu, V. Bulović, C. M. Sutter-Fella, N. G. Park, M. C. Beard, J. W. Lee, K. Zhu and M. G. Bawendi, Science, 2025, 388, 639–645 CrossRef CAS PubMed.
- T. Kodalle, R. F. Moral, L. Scalon, R. Szostak, M. Abdelsamie, P. E. Marchezi, A. F. Nogueira and C. M. Sutter-Fella, Adv. Energy Mater., 2023, 13, 1–14 Search PubMed.
- J. Rodríguez-Romero, J. Sanchez-Diaz, C. Echeverría-Arrondo, S. Masi, D. Esparza, E. M. Barea and I. Mora-Seró, ACS Energy Lett., 2020, 5, 1013–1021 CrossRef.
- H. Baishya, R. Das Adhikari, M. J. Patel, S. Laha, D. Yadav, K. Bhattacharyya and P. K. Iyer, Small, 2025, 07345, 1–14 Search PubMed.
- J. C. Germino, R. Szostak, S. G. Motti, R. F. Moral, P. E. Marchezi, H. S. Seleghini, L. G. Bonato, F. L. De Araújo, T. D. Z. Atvars, L. M. Herz, D. Fenning, A. Hagfeldt and A. F. Nogueira, ACS Photonics, 2020, 7, 2282–2291 CrossRef CAS.
- L. Scalon, R. Szostak, F. L. Araújo, K. F. Adriani, J. F. R. V. Silveira, W. X. C. Oliveira, J. L. F. Da Silva, C. C. Oliveira and A. F. Nogueira, JACS Au, 2022, 2, 1306–1312 CrossRef CAS PubMed.
- R. J. E. Westbrook, T. J. Macdonald, W. Xu, L. Lanzetta, J. M. Marin-Beloqui, T. M. Clarke and S. A. Haque, J. Am. Chem. Soc., 2021, 143, 12230–12243 CrossRef CAS PubMed.
- Z. Min, B. Wang, Y. Kong, J. Guo, X. Ling, W. Ma and J. Yuan, ChemSusChem, 2025, 18(4), e202401852, DOI:10.1002/cssc.202401852.
- A. F. V. Fonseca, L. Scalon, B. R. C. Vale, M. G. D. Guaita, J. Bettini, Z. C. Brandão, L. F. Zagonel, L. A. Padilha and A. F. Nogueira, ACS Energy Lett., 2024, 9, 3177–3186 CrossRef CAS.
- J. Yuan, A. Hazarika, Q. Zhao, X. Ling, T. Moot, W. Ma and J. M. Luther, Joule, 2020, 4, 1160–1185 CrossRef CAS.
- C. de Mayrinck, A. F. V. da Fonseca and M. A. Schiavon, Quim. Nova, 2020, 43, 1264–1276 CAS.
- L. Protesescu, S. Yakunin, M. I. Bodnarchuk, F. Krieg, R. Caputo, C. H. Hendon, R. X. Yang, A. Walsh and M. V. Kovalenko, Nano Lett., 2015, 15, 3692–3696 CrossRef CAS PubMed.
- X. Zhao, D. Li, X. Zhang, H. Huang, C. Zhao, W. Ma and J. Yuan, J. Energy Chem., 2024, 92, 87–94 CrossRef CAS.
- S. Y. Park, G. Seo and T. Kim, et al., Adv. Opt. Mater., 2025, 13, e00968, DOI:10.1002/adom.202500968.
- C. Zhao, C. Cazorla, X. Zhang, H. Huang, X. Zhao, D. Li, J. Shi, Q. Zhao, W. Ma and J. Yuan, J. Am. Chem. Soc., 2024, 146, 4913–4921 CrossRef CAS PubMed.
- Q. A. Akkerman, V. D’Innocenzo, S. Accornero, A. Scarpellini, A. Petrozza, M. Prato and L. Manna, J. Am. Chem. Soc., 2015, 137, 10276–10281 CrossRef CAS PubMed.
- D. Li, C. Zhao, X. Zhang, X. Zhao, H. Huang, H. Li, F. Li and J. Yuan, Adv. Mater., 2025, 37, 2417346, DOI:10.1002/adma.202417346.
- A. F. V. da Fonseca, B. R. C. Vale, T. A. de S. Carvalho, J. Bettini, A. C. Pereira and M. A. Schiavon, J. Phys. Chem. C, 2021, 125, 27363–27371 CrossRef.
- J. M. Burrell, B. Adhikari, S. L. Abiodun and A. B. Greytak, ACS Energy Lett., 2025, 10(9), 4158–4183 CrossRef CAS.
- Y. Xu, X. Zhang, C. Zhu and Q. Yu, J. Mater. Chem. A, 2025, 13, 29688–29705 RSC.
- L. M. Wheeler, E. M. Sanehira, A. R. Marshall, P. Schulz, M. Suri, N. C. Anderson, J. A. Christians, D. Nordlund, D. Sokaras, T. Kroll, S. P. Harvey, J. J. Berry, L. Y. Lin and J. M. Luther, J. Am. Chem. Soc., 2018, 140, 10504–10513 CrossRef CAS PubMed.
- Y. Kong, H. Li, B. Li, D. Li, J. Guo, Q. Zhou, Z. Wang, W. Ma and J. Yuan, J. Energy Chem., 2025, 109, 1–7 CrossRef CAS.
- C. Zhao, D. Li, X. Zhang, H. Huang, C. Cazorla, X. Zhao, H. Li, Y. Chen, W. Zhu, T. Wu and J. Yuan, Adv. Mater., 2025, 37, e12201, DOI:10.1002/adma.202512201.
- M. H. de M. Rodrigues, D. M. Guilhermitti Neto, I. D. Barcelos, C. Labre, C. A. R. Costa, J. B. de Souza, J. A. Sobrinho and A. F. Nogueira, J. Mater. Chem. A, 2025, 13, 17799–17809 RSC.
- C. Zhang, Y. Son, H. Kim, S. H. Lee, X. Liang, G. Fu, S. U. Lee, D. A. Park, Q. Jiang, K. Zhu and N. G. Park, Joule, 2024, 8, 1394–1411 CrossRef CAS.
- A. F. V. Fonseca, G. M. Germano, L. Scalon, C. A. N. de Almeida, A. C. C. Barra, D. S. Ribeiro, Z. C. Brandão, F. C. Marques, I. Mora-Seró and A. F. Nogueira, Mater. Today Chem., 2025, 50, 103238 CrossRef CAS.
- H. Li, H. Huang, D. Li, X. Zhang, C. Zhao, X. Zhao, W. Ma and J. Yuan, Energy Environ. Sci., 2024, 18, 972–981 RSC.
- X. Zhang, H. Huang, C. Zhao, L. Jin, C. Lee, Y. Li, D.-H. Ko, W. Ma, T. Wu and J. Yuan, Nat. Energy, 2024, 9, 1378–1387 CrossRef CAS.
- A. F. V. Fonseca, C. E. Giarola, T. A. de S. Carvalho, F. S. Hojo de Souza and M. A. Schiavon, J. Chem. Phys., 2023, 159, 184705 CrossRef CAS PubMed.
- Y. Han, B. Tang, L. Wang, H. Bao, Y. Lu, C. Guan, L. Zhang, M. Le, Z. Liu and M. Wu, ACS Nano, 2020, 14, 14761–14768 CrossRef CAS PubMed.
- S. Diao, Q. Wu, S. Li, G. Xu, X. Ren, L. Tan, G. Jiang, P. Song and X. Meng, Mater. Horiz., 2025, 12, 4133–4164 RSC.
- Y. Deng, Y. Yuan, J. Ni, L. Zheng, J. Bi, J. Guo, R. Wang, H. Li, S. Zhang, H. Cai and J. Zhang, ACS Appl. Energy Mater., 2025, 8, 4701–4710 CrossRef CAS.
- Y. Ning, S. Guan, C. Cheng, B. Zhang, B. Qin and B. Huang, J. Mater. Chem. C, 2024, 13, 758–765 RSC.
- R. M. MacEiczyk, K. Dümbgen, I. Lignos, L. Protesescu, M. V. Kovalenko and A. J. Demello, Chem. Mater., 2017, 29, 8433–8439 CrossRef CAS.
- G. Chen, X. Zhu, C. Xing, Y. Wang, X. Xu, J. Bao, J. Huang, Y. Zhao, X. Wang, X. Zhou, X. Du and X. Wang, Adv. Photonics Res., 2023, 4, 2200230, DOI:10.1002/adpr.202200230.
- A. Li and C. Yi, Sol. RRL, 2025, 9, e202500681, DOI:10.1002/solr.202500681.
- R. Szostak, J. C. Silva, S. H. Turren-Cruz, M. M. Soares, R. O. Freitas, A. Hagfeldt, H. C. N. Tolentino and A. F. Nogueira, Sci. Adv., 2019, 5, 2–9 Search PubMed.
- N. A. Emelianov, V. V. Ozerova, I. S. Zhidkov, D. V. Korchagin, G. V. Shilov, A. L. Litvinov, E. Z. Kurmaev, L. A. Frolova, S. M. Aldoshin and P. A. Troshin, J. Phys. Chem. Lett., 2022, 13, 2744–2749 CrossRef CAS PubMed.
- X. Zhang, H. Huang, C. Zhao and J. Yuan, Chem. Soc. Rev., 2025, 54, 3017–3060 RSC.
Footnote |
| † This perspective is dedicated to the achievements of Professor Nam-Gyu Park, whose extensive and remarkable work on MHPs has inspired this BRICS group to collaborate in addressing the challenges of these fascinating materials. |
| This journal is © The Royal Society of Chemistry 2026 |
