Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synergistic interactions in LaMnO3/CuO composites with enhanced supercapacitive performance
Alisha
Dhakal
 *a,
Felio R.
Perez
b,
Shawn David
Pollard
a and
Sanjay R.
Mishra
*a
*a,
Felio R.
Perez
b,
Shawn David
Pollard
a and
Sanjay R.
Mishra
*a
aDepartment of Physics and Materials Science, The University of Memphis, Memphis, Tennessee 38152, USA. E-mail: srmishra@memphis.edu; adhakal@memphis.edu
bIntegrated Microscopy Center, The University of Memphis, Memphis, Tennessee 38152, USA
First published on 6th February 2026
Abstract
Composites of La-based perovskites with metal oxides have attracted significant attention in electrochemistry owing to their synergistic effects. In this work, LaMnO3 (LMO)/CuO composites with varying ratios of (100–x)% LMO![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x% CuO (where wt% x = 0, 10, 30, 50, and 100) were successfully synthesized via autocombustion technique followed by annealing at 1000 °C. The morphology and phase purity of the composites were confirmed by X-ray diffraction (XRD) and field-emission scanning electron microscopy (FESEM), respectively. Among all compositions, LMO/CuO(9
x% CuO (where wt% x = 0, 10, 30, 50, and 100) were successfully synthesized via autocombustion technique followed by annealing at 1000 °C. The morphology and phase purity of the composites were confirmed by X-ray diffraction (XRD) and field-emission scanning electron microscopy (FESEM), respectively. Among all compositions, LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) exhibited the highest BET specific surface area of 14 m2 g−1 with an average pore size of 2.11 nm. Compared to single-phase LaMnO3 or CuO, the composites demonstrated increased oxygen vacancies, as confirmed by XPS O 1s spectra. These structural features directly contributed to superior electrochemical performance, with LMO/CuO(9
1) exhibited the highest BET specific surface area of 14 m2 g−1 with an average pore size of 2.11 nm. Compared to single-phase LaMnO3 or CuO, the composites demonstrated increased oxygen vacancies, as confirmed by XPS O 1s spectra. These structural features directly contributed to superior electrochemical performance, with LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) achieving a specific capacitance (CSP) of 646.8 F g−1 at 1 mV s−1 and 517.1 F g−1 at 1 A g−1. Furthermore, it delivered an energy density (Ed) of 25.8 Wh kg−1 at a power density (Pd) of 397.6 W kg−1, along with excellent cyclic stability, retaining 99.8% capacitance after 5000 charge/discharge cycles. These findings underscore the crucial role of synergistic interactions between LaMnO3 and CuO in enhancing electrochemical performance, thereby positioning LMO/CuO composites as promising candidates for next-generation energy storage materials.
1) achieving a specific capacitance (CSP) of 646.8 F g−1 at 1 mV s−1 and 517.1 F g−1 at 1 A g−1. Furthermore, it delivered an energy density (Ed) of 25.8 Wh kg−1 at a power density (Pd) of 397.6 W kg−1, along with excellent cyclic stability, retaining 99.8% capacitance after 5000 charge/discharge cycles. These findings underscore the crucial role of synergistic interactions between LaMnO3 and CuO in enhancing electrochemical performance, thereby positioning LMO/CuO composites as promising candidates for next-generation energy storage materials.
Introduction
The global population is increasing, while fossil fuel resources are depleting rapidly. To address the energy crisis, there is an increasing demand for sustainable energy storage/conversion devices that must possess high energy density (Ed)/power density (Pd), long cyclic stability, and be environmentally friendly.1–4 Supercapacitors represent one class of energy-storage devices capable of meeting these requirements and are generally categorized into three types: (i) electric double-layer capacitors (EDLCs), (ii) pseudocapacitors, and (iii) hybrid capacitors.5 In EDLCs, charges are deposited on the surface without any chemical changes, making it a non-faradaic system. On the other hand, faradaic reactions, in which oxidation and reduction occur at the anode and cathode, are also involved and are referred to as pseudocapacitors.6 A hybrid supercapacitor combines EDLCs and pseudocapacitors. Carbon-based materials operate via the EDLC mechanism to store energy, with higher charge/discharge rates, higher power density, and longer lifetimes.7,8 However, carbon-based materials have a lower energy density than batteries.9The charge-storage mechanism in transition-metal oxides, including Mn2O3, RuO2, NiCo2O4, Co3O4, and V2O5, and mixed oxides such as perovskites, plays a critical role in pseudocapacitive energy-storage applications, owing to their ability to adopt multiple oxidation states and their stable morphology and high structural integrity.10 They offer high specific capacitance (CSP)6 due to fast, reversible redox reactions and higher Ed. Among oxides, RuO2 exhibits a high CSP ∼ 1580 F g−1 at 1 mV s−1.11,12 However, the high cost makes RuO2 less attractive for commercialization. Overcoming these limitations is crucial to meet the benchmark requirements for widespread commercial adoption.
Perovskite-type oxides have attracted considerable interest for supercapacitor applications due to their favorable physical and chemical properties, oxygen storage (inherent to oxygen vacancies), high electrical conductivity, thermal stability, and low cost.13,14 The perovskites are in the form of ABO3 (A is a lanthanide, B is a transition metal).15 Among perovskite materials studied for supercapacitors, LaMnO3 has attracted considerable attention owing to its unique combination of La and Mn atoms. In the presence of oxygen vacancies, LaMnO3 perovskites (LaMnO3−δ) lead to a partial reduction of Mn3+ ions to Mn2+ ions. These vacancies facilitate anionic intercalation, and Mn ions return to their original ionic states (Mn2+ and Mn3+), thereby enhancing the material's supercapacitance,16 Conversely, excess oxygen (LaMnO3+δ) promotes further oxidation of Mn3+ to Mn4+.12 However, its low specific surface area and tendency to agglomerate decrease its electrochemical performance. To overcome this problem, perovskites are often mixed with other metal oxides or carbon-based compounds. Among metal oxides, CuO is a promising candidate owing to its natural abundance, cost-effectiveness, eco-friendliness, ease of synthesis, and high theoretical capacitance (1783 F g−1).17
For example, A. Arya et al. synthesized LaMnO3–NiO composites via a sol–gel-assisted hydrothermal method and obtained a CSP of 170 F g−1 at 10 mV s−1. This improvement was attributed to the synergistic interaction between LaMnO3 and NiO, which enhanced both ion transport efficiency and faradaic redox reactions.18 A. Dhakal et al. followed the autocombustion method to synthesize LaMnO3–Mn3O4 composites and reported a CSP of 660 F g−1 at 1 A g−1 in 6 M KOH.19 P. M. Shafi et al. synthesized LaMnO3/rGO/PANI through a situ polymerization route with a CSP of 802 F g−1 at 1 A g−1 in 3 M KOH.20 The improved CSP was attributed to the higher specific surface area and a greater number of electroactive sites. B. Patil et al. prepared LaMnO3–rGO composites by mixing the components and performing electrochemical measurements in 1 M Na2SO4. The CSP of LaMnO3/rGO75% displayed a high value of 641 F g−1 at 1 A g−1 due to the larger surface area and high electrical conductivity of rGO and oxygen vacancy in LaMnO3.21 LaMnO3–CeO2 composites were synthesized via a hydrothermal method by S. Nagamuthu et al., achieving a CSP of 262 F g−1 at 1 A g−1 in 1 M Na2SO4.22 The enhanced CSP was due to the synergistic redox activity of Ce/Mn ions and improved conductivity. R. Packiaraj et al. synthesized mixed metal oxides composed of ZnO–NiO–CuO through the sol–gel method and reported a high CSP of 1831 F g−1 at 1 A g−1 in 2 M KOH, which was higher than that of single or binary metal oxides.23 This performance improvement was attributed to the synergistic interactions among metal oxides, in which nickel and copper enhanced active-site density and electrical conductivity, while zinc contributed to good chemical and thermal stability. MnO2–CuO was prepared by K. M. Racik et al. using a hydrothermal method and obtained the CSP of 279.12 F g−1 at 0.5 A g−1 in 1 KOH.24 The higher capacitance compared to the individual oxides was attributed to the combined effects of MnO2's structural stability and CuO's high electrical conductivity.
Building on the argument above, in this study, we synthesized LMO/CuO composites via a facile autocombustion technique and systematically investigated their electrochemical performance in an aqueous electrolyte. The incorporation of CuO into LaMnO3 is expected to enhance charge storage through synergistic effects arising from both structural and interfacial contributions. Lattice mismatch and strain at the disordered interface between oxide particles promote the formation of oxygen vacancies, thereby enabling polaron hopping.25 The O2−-deficient interface leaves behind localized electrons that stabilize mixed-valence states (Mn3+/Mn4+ ↔ Cu2+/Cu+), broadening pseudocapacitive processes. The composite thus has a broader spectrum of accessible valence states with more charge storage per unit mass. Furthermore, the addition of secondary oxides prevents LaMnO3 particles from agglomerating, thereby increasing the number of redox-active sites accessible to the electrolyte. Thus, the addition of CuO to LaMnO3 forms heterojunctions that may reduce charge-transfer resistance, thereby enhancing electron mobility within the composite. Collectively, these effects could improve the CSP, rate capability, and cyclic stability of the LMO/CuO composites.
Experimental
Synthesis of LMO composites
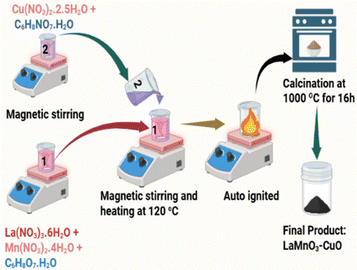
LMO/CuO composites were synthesized via an autocombustion method. In the first step, a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar mixture of lanthanum(III) nitrate hexahydrate and manganese(II) nitrate tetrahydrate, with a 1
1 molar mixture of lanthanum(III) nitrate hexahydrate and manganese(II) nitrate tetrahydrate, with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 metal nitrate-to-citric acid monohydrate ratio, was dissolved in 30 mL of deionized (DI) water to prepare solution 1. Similarly, copper(II) nitrate hemipentahydrate and citric acid monohydrate were dissolved in 30 mL of DI water to form solution 2. Both solutions were magnetically stirred separately for 30 min. Solution 2 was added to solution 1 under continuous magnetic stirring until a homogeneous mixture was obtained. After 15 minutes, the mixture was heated to 120 °C until auto-ignition occurred. The resulting powder was transferred to a furnace and annealed at 1000 °C for 16 hours. The final product was obtained as LMO/CuO(100–x)%: x%, (with Wt% x = 0, 100, 10, 30, and 50) composites named as LMO/CuO(1
1 metal nitrate-to-citric acid monohydrate ratio, was dissolved in 30 mL of deionized (DI) water to prepare solution 1. Similarly, copper(II) nitrate hemipentahydrate and citric acid monohydrate were dissolved in 30 mL of DI water to form solution 2. Both solutions were magnetically stirred separately for 30 min. Solution 2 was added to solution 1 under continuous magnetic stirring until a homogeneous mixture was obtained. After 15 minutes, the mixture was heated to 120 °C until auto-ignition occurred. The resulting powder was transferred to a furnace and annealed at 1000 °C for 16 hours. The final product was obtained as LMO/CuO(100–x)%: x%, (with Wt% x = 0, 100, 10, 30, and 50) composites named as LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9
1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), respectively. The chemical reaction for the formation of LMO/CuO composites is presented in Scheme 1 and eqn (1).
1), respectively. The chemical reaction for the formation of LMO/CuO composites is presented in Scheme 1 and eqn (1).| La(NO3)3·6H2O + Mn(NO3)2·4H2O + Cu(NO3)2·2.5H2O + C6H8O7·H2O → LaMnO3 + CuO + 7/2 N2 + 6CO2 + 27/2 H2O + 6O2 | (1) |
Preparation of the electrode
To clean the substrate (Ni-foam) and the electrode, the preparation method described in our previous work is followed.26 Briefly, the Ni-foam was cleaned by sequential ultrasonication for 10 minutes each in 16 mL of 37 wt% HCl, deionized (DI) water, and ethanol, followed by drying at 110 °C for 2 hours. For electrode fabrication, an electrode solution was prepared by dispersing 6 mg of LMO/CuO composite and 1 mg of polyvinylidene fluoride (PVDF) in 1 mL of N-methyl-2-pyrrolidone (NMP) under magnetic stirring. A volume of 0.6 mL of this homogeneous mixture was uniformly coated onto the pre-cleaned Ni-foam and then dried at 100 °C for 20 hours in a vacuum furnace. The active material loading was determined by subtracting the bare Ni-foam's original mass from that of the coated, dried electrode.Characterization
The prepared LMO/CuO composites were characterized using various analytical tools. The crystal structure and phase studies were conducted using XRD (Bruker D8 with Cu Kα1 radiation) and FESEM (Hitachi S-470). The elemental and chemical compositions were investigated using energy-dispersive X-ray spectroscopy (EDX; Bruker Esprit Spectrum 2.3) and X-ray photoelectron spectroscopy (XPS; Thermo Scientific K-Alpha). The surface area and porosity of the composites were measured using a NOVAtouch surface area analyzer (Quantachrome Instruments).Electrochemical analysis
Electrochemical measurements were performed in a three-electrode configuration in a 1 M KOH electrolyte at ambient temperature using a Gamry instrument (IFC1000-07045). The Ag/AgCl electrode served as the reference electrode, a platinum plate as the counter electrode, and LMO/CuO composites coated on nickel foam as the working electrode. Cyclic voltammetry (CV) was performed at scan rates from 1 to 200 mV s−1 over a potential window of 0 to 0.6 V. Chronopotentiometry (charge–discharge) tests were conducted at various current densities within a voltage range of 0 to 0.45 V. Electrochemical impedance spectroscopy (EIS) was recorded in the frequency range from 10 mHz to 100 kHz. The CSP, (F g−1), Ed (Wh kg−1), and Pd (W kg−1) were calculated from the CV and charge–discharge (GCD) data using eqn (2)–(5).27–30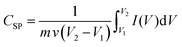 | (2) |
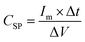 | (3) |
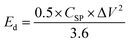 | (4) |
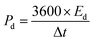 | (5) |
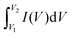 gives the area enclosed in the CV curves, m implies mass of the active material in the electrode, v (mV s−1) represents scan rate, V2–V1 (ΔV) is a potential window, I(V) corresponds to the response current, Im denotes the current density, and Δt denotes the discharge time.
gives the area enclosed in the CV curves, m implies mass of the active material in the electrode, v (mV s−1) represents scan rate, V2–V1 (ΔV) is a potential window, I(V) corresponds to the response current, Im denotes the current density, and Δt denotes the discharge time.
Results and discussion
Structure and morphology
Fig. 1(a) presents the crystalline phases of LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, 0
0, 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 9
1, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 7
1, 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and 1
3, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composites as determined by powder XRD at room temperature. The composites LMO/CuO(1
1) composites as determined by powder XRD at room temperature. The composites LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0) only exhibit a single phase of LaMnO3 with prominent diffraction peaks at 22.83, 32.34, 32.68, 39.95, 46.64, 52.31, 57.68, and 67.69°, corresponding to (012), (110), (104), (202), (024), (122), (300), and (220) crystal planes of LaMnO3 with a rhombohedral crystal structure and space group of R
0) only exhibit a single phase of LaMnO3 with prominent diffraction peaks at 22.83, 32.34, 32.68, 39.95, 46.64, 52.31, 57.68, and 67.69°, corresponding to (012), (110), (104), (202), (024), (122), (300), and (220) crystal planes of LaMnO3 with a rhombohedral crystal structure and space group of R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c (167) (PDF 01-076-9017). Similarly, for the molar ratio of LMO/CuO(0
c (167) (PDF 01-076-9017). Similarly, for the molar ratio of LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the main diffraction peaks at 32.54, 35.59, 38.72, 48.76, 53.59, 58.19, 61.60, 66.24, and 68.17 correspond to the (110), (
1), the main diffraction peaks at 32.54, 35.59, 38.72, 48.76, 53.59, 58.19, 61.60, 66.24, and 68.17 correspond to the (110), (![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 11), (111), (
11), (111), (![[2 with combining macron]](https://www.rsc.org/images/entities/char_0032_0304.gif) 02), (020), (202), (
02), (020), (202), (![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) 13), (
13), (![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) 11), and (220), crystal planes of CuO with a monoclinic crystal structure and space group C2/c15 (PDF 01-085-7183). However, the remaining three composites exhibit both distinguishable LaMnO3 and CuO phases, with no other impurities present. Fig. 1(b) presents the Rietveld-refined XRD patterns of the LMO/CuO composites obtained using GSAS-II. The estimated composition of composites extracted from the Rietveld refinement are; LMO/CuO(100%
11), and (220), crystal planes of CuO with a monoclinic crystal structure and space group C2/c15 (PDF 01-085-7183). However, the remaining three composites exhibit both distinguishable LaMnO3 and CuO phases, with no other impurities present. Fig. 1(b) presents the Rietveld-refined XRD patterns of the LMO/CuO composites obtained using GSAS-II. The estimated composition of composites extracted from the Rietveld refinement are; LMO/CuO(100%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0%), LMO/CuO(0%
0%), LMO/CuO(0%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100%), LMO/CuO(84%
100%), LMO/CuO(84%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16%), LMO/CuO(75%
16%), LMO/CuO(75%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25%), and LMO/CuO(65%
25%), and LMO/CuO(65%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35%) for samples LMO/CuO(1
35%) for samples LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9
1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), respectively. The crystallite size was calculated using the Scherrer equation
1), respectively. The crystallite size was calculated using the Scherrer equation 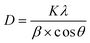 ,31 where D is the crystallite size (nm), K is the Scherrer constant (0.9), λ is the X-ray wavelength (1.5406 Å), β is the full width at half maximum (FWHM) of the diffraction peak, and θ is the Bragg diffraction angle. The characteristic diffraction peaks (2θ) of LaMnO3 were observed at 22.83, 32.34, 32.68, 39.95, and 46.64°, while those of CuO appeared at 35.59, 38.72, 48.76, and 61.60°. The calculated crystallite sizes of LaMnO3 and CuO are summarized in Table S1.
,31 where D is the crystallite size (nm), K is the Scherrer constant (0.9), λ is the X-ray wavelength (1.5406 Å), β is the full width at half maximum (FWHM) of the diffraction peak, and θ is the Bragg diffraction angle. The characteristic diffraction peaks (2θ) of LaMnO3 were observed at 22.83, 32.34, 32.68, 39.95, and 46.64°, while those of CuO appeared at 35.59, 38.72, 48.76, and 61.60°. The calculated crystallite sizes of LaMnO3 and CuO are summarized in Table S1.
 | ||
| Fig. 1 (a) XRD pattern of LMO/CuO composites. (b) Rietveld refinement of XRD data for LMO/CuO composites was performed using GSAS-II. | ||
The FESEM images in Fig. 2(a–e) show the surface morphology of pure LMO/CuO composites with varying ratios (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, 0
0, 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 9
1, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 7
1, 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and 1
3, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)). Fig. 2(a) shows the LMO/CuO(1
1)). Fig. 2(a) shows the LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0) composites, which are spongier and more fragile with a chain-like structure. LMO/CuO(0
0) composites, which are spongier and more fragile with a chain-like structure. LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) sample (Fig. 2(b)) displays a relatively dense and well-faceted crystalline structure, composed of tightly packed grains with smooth surfaces. The LMO/CuO composites exhibit distinct morphological transformations with varying CuO content. The LMO/CuO(9
1) sample (Fig. 2(b)) displays a relatively dense and well-faceted crystalline structure, composed of tightly packed grains with smooth surfaces. The LMO/CuO composites exhibit distinct morphological transformations with varying CuO content. The LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) sample (Fig. 2(c)) shows a porous and loosely aggregated structure, likely due to the dominant LaMnO3 phase. As the CuO content increases in LaMnO3 at a 7
1) sample (Fig. 2(c)) shows a porous and loosely aggregated structure, likely due to the dominant LaMnO3 phase. As the CuO content increases in LaMnO3 at a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio (Fig. 2(d)), the morphology resembles granular particles connected in a chain, with the presence of voids and open channels, indicating loosely packed particles and enhanced interparticle connectivity. At a 1
3 ratio (Fig. 2(d)), the morphology resembles granular particles connected in a chain, with the presence of voids and open channels, indicating loosely packed particles and enhanced interparticle connectivity. At a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (Fig. 2(e)), the microstructure appears highly uniform, with fine particles distributed densely across the surface between LaMnO3 and CuO phases. The elementary mapping of O (red), La (blue), Mn (green), and Cu (magenta) is shown in Fig. S1. The corresponding EDX spectra (Fig. 2(a′–e′)) confirm the expected elemental composition of each sample. These EDX results validate the presence of La, Mn, Cu, and O in LMO/CuO composites.32 Minor peaks for Au and C are also present, which are attributed to the gold coating and carbon tape used during FESEM sample preparation.
1 ratio (Fig. 2(e)), the microstructure appears highly uniform, with fine particles distributed densely across the surface between LaMnO3 and CuO phases. The elementary mapping of O (red), La (blue), Mn (green), and Cu (magenta) is shown in Fig. S1. The corresponding EDX spectra (Fig. 2(a′–e′)) confirm the expected elemental composition of each sample. These EDX results validate the presence of La, Mn, Cu, and O in LMO/CuO composites.32 Minor peaks for Au and C are also present, which are attributed to the gold coating and carbon tape used during FESEM sample preparation.
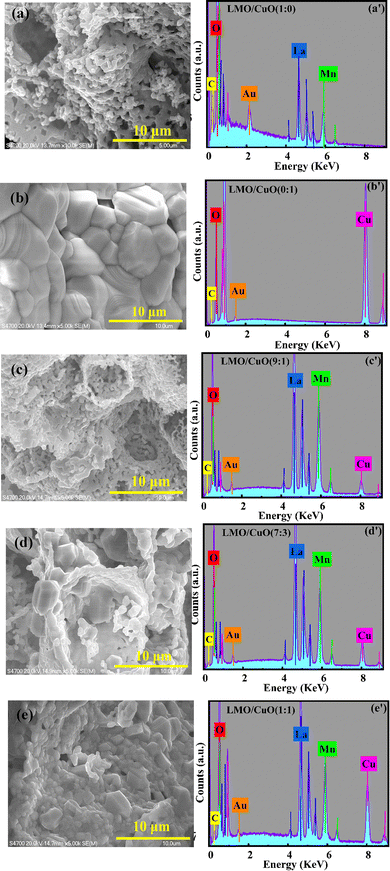 | ||
Fig. 2 (a–e) FESEM images and (a′–e′) elemental mapping of the LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0 0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9 1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7 1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1 3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composites, respectively. 1) composites, respectively. | ||
The surface area and pore size distribution of the LMO/CuO composites were evaluated using N2 adsorption–desorption isotherms, as shown in Fig. 3(a–e). All curves show a gradual increase at low P/P0, followed by a sharp rise near P/P0 ∼ 0.9–1.0, characteristic of type IV isotherms (IUPAC classification), typical of mesoporous materials. LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), with a moderate slope and a rapid rise at high P/P0, exhibits mesoporous behavior with a moderate surface area, whereas LMO/CuO(0
0), with a moderate slope and a rapid rise at high P/P0, exhibits mesoporous behavior with a moderate surface area, whereas LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), with much lower adsorption volume, may be due to a low surface area and few pores in CuO. Among the samples, LMO/CuO(9
1), with much lower adsorption volume, may be due to a low surface area and few pores in CuO. Among the samples, LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) exhibited the highest Brunauer–Emmett–Teller (BET) surface area of 13.82 m2 g−1, while the BET values for all composites are summarized in Table S1. The pore volume and pore radius distribution, derived from Barrett–Joyner–Halenda (BJH) analysis of the desorption branch, are displayed in the inset Fig. 3(a–e), confirming the mesoporous nature of the composites. In all insets, the peak is sharply located below ∼10 nm, averaging 2–5 nm, proving that the pores are mesoporous. Beyond 15 nm, dV/dr nearly drops to zero, indicating the absence of macropores in the compounds. Well-ordered porosity, as evidenced by a narrow pore-size distribution, could enhance ion diffusion in the electrode material.33 Such mesoporosity, combined with a high specific surface area, facilitates efficient ion diffusion and charge transport, which are critical for enhanced electrochemical performance. Notably, CuO shows a very low specific surface area (Fig. 2(b)).
1) exhibited the highest Brunauer–Emmett–Teller (BET) surface area of 13.82 m2 g−1, while the BET values for all composites are summarized in Table S1. The pore volume and pore radius distribution, derived from Barrett–Joyner–Halenda (BJH) analysis of the desorption branch, are displayed in the inset Fig. 3(a–e), confirming the mesoporous nature of the composites. In all insets, the peak is sharply located below ∼10 nm, averaging 2–5 nm, proving that the pores are mesoporous. Beyond 15 nm, dV/dr nearly drops to zero, indicating the absence of macropores in the compounds. Well-ordered porosity, as evidenced by a narrow pore-size distribution, could enhance ion diffusion in the electrode material.33 Such mesoporosity, combined with a high specific surface area, facilitates efficient ion diffusion and charge transport, which are critical for enhanced electrochemical performance. Notably, CuO shows a very low specific surface area (Fig. 2(b)).
 | ||
| Fig. 3 N2 adsorption–desorption isotherms of LMO/CuO composites (a–e) with corresponding BJH pore size distribution curves (insets). | ||
The chemical composition of LMO/CuO composites was determined by XPS analysis and presented in Fig. 4(a–e). Fig. 4(a) shows the La 3d XPS spectra, which exhibit distinct spin–orbit peaks at 834.4 and 838.0 eV, corresponding to La 3d5/2, along with additional peaks at 850.8 and 855.4 eV associated with La 3d3/2. These findings confirm the presence of La3+ in the LMO/CuO composites, which aligns with the results reported by.22,34 Meanwhile, Fig. 4(b) presents the Mn 2p spectra, which reveal broad line widths and complex peak structures, indicating the coexistence of multiple Mn oxidation states. The major peaks observed at 639.98 eV and 651.78 eV correspond to Mn 2p3/2 and Mn 2p1/2, respectively,35 with a consistent spin–orbit separation of approximately 11.8 eV. Each peak is further resolved into components attributed to Mn2+, Mn3+, and Mn4+ oxidation states (Fig. 4(d)). Specifically, peaks at 641.2 eV and 652.3 eV correspond to Mn2+, those at 642.3 eV and 653.9 eV represent Mn3+, and peaks at 644.0 eV and 655.5 eV relate to Mn4+.5,36,37
 | ||
| Fig. 4 XPS spectra of high-resolution of (a) La 3d and (b) Mn 2p and deconvoluted spectra of (c) Cu 2p, (d) Mn 2p, and (e) O 1s of LMO/CuO composites. | ||
Similarly, the deconvoluted Cu 2p XPS spectrum (Fig. 4(c)) reveals characteristic Cu2+ peaks at around 935.41 and 954.89 eV, along with Cu+ peaks located at 933.81 and 953.34 eV, respectively. In addition, two distinct satellite peaks are observed at 941.18 and 943.77 eV, further confirming the presence of multiple copper oxidation states.38 The O 1s XPS spectra of transition metal oxides are deconvoluted into three Gaussian peaks, generally appearing in the ranges of 529–530 eV, 531–532 eV, and 532–535 eV, corresponding to lattice oxygen (O1), oxygen vacancies (O2), and chemisorbed oxygen species associated with surface hydroxyl groups (O3), respectively.39–41 As illustrated in Fig. 4(e), all three oxygen species O1, O2, and O3 are observed in the spectra of single-phase LaMnO3, CuO, and the LMO/CuO composites. The corresponding O2/O1 ratios are 0.66, 0.46, 0.95, 0.66, and 0.74 for LMO/CuO with ratios 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0, 0
0, 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 9
1, 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 7
1, 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, and 1
3, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively. The nearly 1
1, respectively. The nearly 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 O2/O1 ratio in LMO/CuO(9
1 O2/O1 ratio in LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composites indicates the presence of oxygen vacancies, providing further evidence of lattice-defect formation. The calculated Mn3+/Mn2+ ratios were 1.68, 1.29, 1.31, and 1.33, while the Mn3+/Mn4+ ratios were 1.69, 1.42, 1.36, and 1.23 for LMO/CuO(1
1) composites indicates the presence of oxygen vacancies, providing further evidence of lattice-defect formation. The calculated Mn3+/Mn2+ ratios were 1.68, 1.29, 1.31, and 1.33, while the Mn3+/Mn4+ ratios were 1.69, 1.42, 1.36, and 1.23 for LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(9
0), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), respectively. Similarly, the Cu2+/Cu1+ ratios were determined to be 2.12, 1.11, 0.86, and 0.50 for LMO/CuO(0
1), respectively. Similarly, the Cu2+/Cu1+ ratios were determined to be 2.12, 1.11, 0.86, and 0.50 for LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9
1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), respectively. Notably, the LMO/CuO(9
1), respectively. Notably, the LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composite exhibits a high Mn3+/Mn4+ and Cu2+/Cu1+ ratios, indicating that these oxidation states (Cu2+, Mn3+) can readily undergo reversible redox transitions, which is highly favorable for enhancing electrochemical activity.
1) composite exhibits a high Mn3+/Mn4+ and Cu2+/Cu1+ ratios, indicating that these oxidation states (Cu2+, Mn3+) can readily undergo reversible redox transitions, which is highly favorable for enhancing electrochemical activity.
Electrochemical analysis
Fig. 5(a) shows the CV curves of LMO/CuO composites at 1 mV s−1 in the potential range 0 to 0.6 V. Among them, LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) CV curves enclosed the largest integral area, which indicates having a higher CSP. Furthermore, Fig. 5(b) displays the chronopotentiometry charge/discharge curves of the LMO/CuO composite electrode at 1 A g−1. The discharge time of the LMO/CuO(9
1) CV curves enclosed the largest integral area, which indicates having a higher CSP. Furthermore, Fig. 5(b) displays the chronopotentiometry charge/discharge curves of the LMO/CuO composite electrode at 1 A g−1. The discharge time of the LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) has the largest value compared to other composites, further highlighting its better performance in electrochemical properties. Based on XPS data and faradaic reaction, the possible oxygen intercalation/deintercalation reactions during the energy storage of LaMnO3 and CuO are,42–44
1) has the largest value compared to other composites, further highlighting its better performance in electrochemical properties. Based on XPS data and faradaic reaction, the possible oxygen intercalation/deintercalation reactions during the energy storage of LaMnO3 and CuO are,42–44| La[Mn2+2δ,Mn3+1−2δ]O3−δ + 2δOH− ↔ LaMn3+O3 + δH2O + 2δe− | (6) |
| LaMn3+O3 + 2δOH− ↔ La[Mn4+2δ,Mn3+1−2δ]O3+δ + δH2O + 2δe− | (7) |
| 2CuO + H2O + 2e− ↔ Cu2O + 2OH− | (8) |
| Cu2O + H2O + 2OH− ↔ 2Cu(OH)2 + 2e− | (9) |
The CV curves of the LMO/CuO composite electrode exhibit distinct pairs of redox peaks, indicative of strong faradaic behavior (Fig. 5(c)). Although the redox activity indicates effective charge-transfer processes, the individual peaks corresponding to LaMnO3 and CuO are not clearly distinguishable due to their overlapping potential regions. As the scan rate increases from 1 to 200 mV s−1, the anodic peaks shift to more positive potentials, while the cathodic peaks shift to more negative potentials. This behavior is attributed to enhanced electrical polarization and partially irreversible redox processes, suggesting that redox reactions occur more rapidly at the interface between the active electrode material and the electrolyte at higher scan rates.
Electrochemical energy storage processes can be classified as capacitive-controlled (capacitor-like) or diffusion-controlled (battery-like). To distinguish these kinetic contributions and understand the charge-storage behavior of electrode materials, electrochemical techniques such as cyclic voltammetry (CV) and electrochemical impedance spectroscopy (EIS) are commonly employed. In CV, the current response (i) as a function of the scan rate (ν) provides valuable insight into the underlying charge storage mechanism. This relationship is typically described by a power-law eqn (10):45
| i = avb | (10) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9
1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), the b-values are 0.59, 0.61, 0.63, 0.64, and 0.66, respectively. A more detailed picture is given in Fig. S2(a). The obtained b-values, ranging from 0.5 to 1.0, reflect a combined charge-storage mechanism involving both diffusion and surface (capacitively controlled) effects. To understand the diffusion coefficient of LMO/CuO composites, let us consider b = 0.5, then the power-law eqn (10) can be expressed in terms of the Randles–Sevcik equation (eqn (11)):46
1), the b-values are 0.59, 0.61, 0.63, 0.64, and 0.66, respectively. A more detailed picture is given in Fig. S2(a). The obtained b-values, ranging from 0.5 to 1.0, reflect a combined charge-storage mechanism involving both diffusion and surface (capacitively controlled) effects. To understand the diffusion coefficient of LMO/CuO composites, let us consider b = 0.5, then the power-law eqn (10) can be expressed in terms of the Randles–Sevcik equation (eqn (11)):46| ip = 2.69 × 105AC0(n3Dv/RT) | (11) |
| i(V) = k1v + k2v1/2 | (12) |
For analytical purposes, eqn (12) can be rearranged slightly to
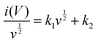 | (13) |
In this equation, parameters k1 and k2 are constant. The current response for diffusion-controlled processes varies v1/2, whereas for capacitive-controlled processes it varies directly with v. In detail, the relation between the i(V)/v1/2 and v1/2 gives a straight line with slope (k1) and y-intercept (k2) (eqn (13)). Solving for k1 and k2 at each potential allows us to qualitatively estimate the diffusion (k2v1/2) and capacitive contributions (k1v). The diffusion percentage, even at 200 mV s−1, is 76, 71, 64, 66, and 60% for LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9
1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), respectively, and shown in Fig. S2(c). The diffusion-controlled process that dominates the energy storage mechanism indicates a faradaic redox reaction.
1), respectively, and shown in Fig. S2(c). The diffusion-controlled process that dominates the energy storage mechanism indicates a faradaic redox reaction.
The charge/discharge curves of LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at different current densities from 1 to 15 A g−1 are shown in Fig. 5(d). Distinct sloping plateau regions are observed across various current densities, consistent with typical pseudocapacitive behavior and consistent with the CV results. The CV profiles recorded at different scan rates and the corresponding charge–discharge curves measured at multiple current densities for LMO/CuO(1
1) at different current densities from 1 to 15 A g−1 are shown in Fig. 5(d). Distinct sloping plateau regions are observed across various current densities, consistent with typical pseudocapacitive behavior and consistent with the CV results. The CV profiles recorded at different scan rates and the corresponding charge–discharge curves measured at multiple current densities for LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) are shown in Fig. S3(a–h).
1) are shown in Fig. S3(a–h).
The CSP was calculated from the CV curves by using eqn (2) and is 369, 284, 647, 430, and 334 F g−1 at 1 mV s−1 of LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9
1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), respectively, and shown in Fig. 5(e). The plot reveals that the CSP decreases with increasing scan rate, which can be attributed to the electrochemical kinetics at the electrode–electrolyte interface. At lower scan rates, electrolyte ions have sufficient time to penetrate into the bulk of the electrode material, thereby increasing capacitance. In contrast, at higher scan rates, ion diffusion is limited, and ions primarily interact with the electrode surface, thereby reducing capacitance. From the charge/discharge curves (eqn (3)), the CSP at 1 A g−1 are 338, 203, 517, 465, and 129 F g−1, as presented in Table S2. The calculation of the CSP of LMO/CuO composites at different current densities is presented in Fig. 4(f). According to our previous work, the CSP (10 F g−1 at 1 mV s−1) from bare Ni-form could be ignored.48 Table S2 presents the CSP relationship with scan rate and current density for the LMO/CuO composites, including the Ed and Pd values. Here, the CSP of LMO/CuO(9
1), respectively, and shown in Fig. 5(e). The plot reveals that the CSP decreases with increasing scan rate, which can be attributed to the electrochemical kinetics at the electrode–electrolyte interface. At lower scan rates, electrolyte ions have sufficient time to penetrate into the bulk of the electrode material, thereby increasing capacitance. In contrast, at higher scan rates, ion diffusion is limited, and ions primarily interact with the electrode surface, thereby reducing capacitance. From the charge/discharge curves (eqn (3)), the CSP at 1 A g−1 are 338, 203, 517, 465, and 129 F g−1, as presented in Table S2. The calculation of the CSP of LMO/CuO composites at different current densities is presented in Fig. 4(f). According to our previous work, the CSP (10 F g−1 at 1 mV s−1) from bare Ni-form could be ignored.48 Table S2 presents the CSP relationship with scan rate and current density for the LMO/CuO composites, including the Ed and Pd values. Here, the CSP of LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composites is greater than others, maybe due to (i) the synergetic effect between the LaMnO3 and CuO, and (ii) loose packing morphology. Further increasing the CuO ratio decreases the CSP, likely due to particle agglomeration, thereby reducing the availability of redox-active sites.
1) composites is greater than others, maybe due to (i) the synergetic effect between the LaMnO3 and CuO, and (ii) loose packing morphology. Further increasing the CuO ratio decreases the CSP, likely due to particle agglomeration, thereby reducing the availability of redox-active sites.
Fig. 6(a) shows the electrochemical impedance spectroscopy (EIS) of LMO/CuO composites, with a well-fitted equivalent circuit that accounts for all impedance components. The frequency range is divided into three parts. In the low-frequency region, the slope of the curve corresponds to the Warburg impedance (W), which indicates electrolyte diffusion at the electrode.49 In the low-frequency region, the Nyquist plot of LMO/CuO composites shows a straight line with a phase angle of approximately 70° (a vertical line for an ideal capacitor), indicating good capacitive behavior.50 Such non-ideal behavior is typically represented in the equivalent circuit by incorporating a constant-phase element (CPE):51
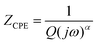 | (14) |
In eqn (14), ZCPE represents the impedance of the constant phase element (CPE), j is the imaginary unit (j2 = −1), and ω denotes the angular frequency defined as ω = 2πf, where f is the frequency in Hertz (Hz). The parameters Q and α are frequency-independent constants, with Q. Corresponding to the interfacial differential capacitance. The dimensionless exponent α (0 < α ≤ 1) characterizes the deviation from ideal capacitive behavior. ZCPE becomes a pure capacitor when α = 1, a resistor when α = 0, and a Warburg impedance when α = 0.5. In the high-frequency region, the intersection of the Nyquist plot with the x-axis corresponds to the solution resistance (R1), which includes the combined contributions from the intrinsic resistance of the substrate, the ionic resistance of the electrolyte, and the contact resistance at the interface between the active material and the current collector.52 The small semicircle in the high frequency region indicates (inset Fig. 6(a)) the charge transfer resistance (R2), which is the lowest value of 6.35 Ω for LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The fitted parameters obtained from the equivalent circuit model from Gamry Echem Analyst are summarized in Table S3. The lower R2 value indicates faster charge-transfer kinetics at the electrode–electrolyte interface. The slope of the low-frequency region indicates the diffusion coefficient of the electrode materials.49
1). The fitted parameters obtained from the equivalent circuit model from Gamry Echem Analyst are summarized in Table S3. The lower R2 value indicates faster charge-transfer kinetics at the electrode–electrolyte interface. The slope of the low-frequency region indicates the diffusion coefficient of the electrode materials.49
Long cyclic stability is the desired property of any efficient supercapacitor device. The cyclic stability test was performed by repeating the charge–discharge measurements of the LMO/CuO composites at 10 A g−1 in the same potential window (0–0.45 V) over 5000 cycles, as shown in Fig. 6(b) (solid symbols). We observed some capacitance fluctuations that may be attributed to temperature changes during continuous charging and discharging.53 After 5000 cycles, the CSP remains 175, 148, 234, 190, and 121 F g−1 for LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9
1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) with retention percentages of 97.2, 98.8, 99.8, 97.6, and 97.2%, respectively.
1) with retention percentages of 97.2, 98.8, 99.8, 97.6, and 97.2%, respectively.
The Coulombic efficiency (η) was calculated by using the following eqn (15):54
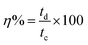 | (15) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composite has the highest Ed of 26 Wh kg−1 with respective Pd of 398 W kg−1. Similarly, LMO/CuO(1
1) composite has the highest Ed of 26 Wh kg−1 with respective Pd of 398 W kg−1. Similarly, LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) have Ed of 17, 10, 23, and 6 Wh kg−1 with corresponding Pd of 399, 399, 398, and 401 W kg−1, respectively, and presented in Table S2. The LMO/CuO composites exhibit respectable electrochemical performance in comparison with previously reported values, as summarized in Table S4. Fig. 6(d) represents the EIS measurements recorded after 5000 charge–discharge cycles (stability test). The R2 values were found to be 9.65, 13.90, 7.25, 9.16, and 10.61 Ω for LMO/CuO(1
1) have Ed of 17, 10, 23, and 6 Wh kg−1 with corresponding Pd of 399, 399, 398, and 401 W kg−1, respectively, and presented in Table S2. The LMO/CuO composites exhibit respectable electrochemical performance in comparison with previously reported values, as summarized in Table S4. Fig. 6(d) represents the EIS measurements recorded after 5000 charge–discharge cycles (stability test). The R2 values were found to be 9.65, 13.90, 7.25, 9.16, and 10.61 Ω for LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0), LMO/CuO(0
0), LMO/CuO(0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(9
1), LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), LMO/CuO(7
1), LMO/CuO(7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and LMO/CuO(1
3), and LMO/CuO(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), respectively, listed in Table S5. R2 values increased relative to those observed prior to cyclic stability, which can be attributed to partial electrode surface degradation and the formation of resistive layers during prolonged electrochemical cycling, as evidenced by FESEM images (Fig. 6(f)). Repeated ion intercalation/deintercalation and continuous exposure to the electrolyte may lead to structural distortion, surface passivation, and the accumulation of OH− and K+ ions at the active surface, thereby increasing charge-transfer and solution resistances. Fig. 6(e) presents the FESEM image of the LMO/CuO(9
1), respectively, listed in Table S5. R2 values increased relative to those observed prior to cyclic stability, which can be attributed to partial electrode surface degradation and the formation of resistive layers during prolonged electrochemical cycling, as evidenced by FESEM images (Fig. 6(f)). Repeated ion intercalation/deintercalation and continuous exposure to the electrolyte may lead to structural distortion, surface passivation, and the accumulation of OH− and K+ ions at the active surface, thereby increasing charge-transfer and solution resistances. Fig. 6(e) presents the FESEM image of the LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) electrode before the stability test, which closely resembles Fig. 2(c), indicating that the coating of the active material on the nickel foam is uniform and well-adhered. However, in the FESEM image (Fig. 6(f)), the particle surfaces of the LMO/CuO composites show reduced asperities after the 5000-cycle stability test, compared with their original morphology. This transformation can be attributed to several factors, including prolonged exposure to the alkaline electrolyte, which may lead to surface etching or the formation of passivation layers that can fill surface irregularities. The FESEM images of other composites before and after the stability test are presented in Fig. S4. The surfaces appear smoother after cyclic stability, although the overall morphology of the composites does not differ significantly from that of the source material.
1) electrode before the stability test, which closely resembles Fig. 2(c), indicating that the coating of the active material on the nickel foam is uniform and well-adhered. However, in the FESEM image (Fig. 6(f)), the particle surfaces of the LMO/CuO composites show reduced asperities after the 5000-cycle stability test, compared with their original morphology. This transformation can be attributed to several factors, including prolonged exposure to the alkaline electrolyte, which may lead to surface etching or the formation of passivation layers that can fill surface irregularities. The FESEM images of other composites before and after the stability test are presented in Fig. S4. The surfaces appear smoother after cyclic stability, although the overall morphology of the composites does not differ significantly from that of the source material.
In the LaMnO3–CuO composite, the CSP exhibits a pronounced enhancement at the 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 wt% ratio, attributable to an optimal synergistic interplay that maximizes heterogeneous interfaces and redox-active sites while mitigating agglomeration-induced drawbacks at higher CuO loadings. This composition fosters superior electronic conductivity, accelerated ion diffusion via oxygen vacancies, and balanced faradaic contributions from LaMnO3's anion intercalation and Mn3+/Mn4+ redox processes, positioning it as the ideal “sweet spot” for pseudocapacitive performance. At the atomic level, CuO introduces Cu2+ ↔ Cu1+ redox centers that complement Mn states through efficient d-orbital overlap, engineers lattice defects to enhance O2− mobility and electrolyte adsorption, and tunes the electronic structure via p-type hole injection, thereby lowering charge-transfer barriers and boosting overall capacitance retention. The observed reduction in electrochemical performance at higher CuO content may arise from increased heterogeneity, which leads to CuO clusters and higher interparticle resistance within the composite. Additionally, these clusters block redox-active sites on the LaMnO3 surface. Thus, at ∼10% CuO, the composite achieves an optimal balance of Cu2+/Cu1+ redox couples, and enhanced conductivity synergizes with Mn3+/Mn4+ hopping without disrupting the perovskite framework.
10 wt% ratio, attributable to an optimal synergistic interplay that maximizes heterogeneous interfaces and redox-active sites while mitigating agglomeration-induced drawbacks at higher CuO loadings. This composition fosters superior electronic conductivity, accelerated ion diffusion via oxygen vacancies, and balanced faradaic contributions from LaMnO3's anion intercalation and Mn3+/Mn4+ redox processes, positioning it as the ideal “sweet spot” for pseudocapacitive performance. At the atomic level, CuO introduces Cu2+ ↔ Cu1+ redox centers that complement Mn states through efficient d-orbital overlap, engineers lattice defects to enhance O2− mobility and electrolyte adsorption, and tunes the electronic structure via p-type hole injection, thereby lowering charge-transfer barriers and boosting overall capacitance retention. The observed reduction in electrochemical performance at higher CuO content may arise from increased heterogeneity, which leads to CuO clusters and higher interparticle resistance within the composite. Additionally, these clusters block redox-active sites on the LaMnO3 surface. Thus, at ∼10% CuO, the composite achieves an optimal balance of Cu2+/Cu1+ redox couples, and enhanced conductivity synergizes with Mn3+/Mn4+ hopping without disrupting the perovskite framework.
Conclusion
LaMnO3–CuO composites were synthesized via a facile autocombustion route and thoroughly characterized to establish the correlation between their structure and electrochemical performance. Rietveld refinement confirmed the compositional ratios, while XRD verified the coexistence of distinct phases. XPS analysis revealed an increased concentration of oxygen vacancies, indicative of enhanced defect-assisted conductivity and redox activity. In contrast, BET measurements indicated a higher surface area for the LMO/CuO (9 : 1) composite, thereby facilitating efficient ion access. These structural and surface features collectively provide a favorable framework for charge storage. Electrochemical studies demonstrated that the LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) composite achieved the best performance, delivering CSP of 647 F g−1 at 1 mV s−1 and 517 F g−1 at 1 A g−1, with excellent cyclic stability (99.8% retention after 5000 cycles). Overall, a 75% enhancement in specific capacitance is observed for LMO/CuO(9
1) composite achieved the best performance, delivering CSP of 647 F g−1 at 1 mV s−1 and 517 F g−1 at 1 A g−1, with excellent cyclic stability (99.8% retention after 5000 cycles). Overall, a 75% enhancement in specific capacitance is observed for LMO/CuO(9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) compared to pure LMO. It also exhibited a high energy density of 26 Wh kg−1 at a power density of 398 W kg−1. The superior performance is attributed to the synergistic interaction between LaMnO3 and CuO, which enhances redox activity, electron transport, and defect chemistry. These results demonstrate that controlled incorporation of CuO (∼10%) into LaMnO3 provides an optimal balance of conductivity, stability, and capacitance, underscoring the potential of perovskite–oxide composites for next-generation supercapacitors.
1) compared to pure LMO. It also exhibited a high energy density of 26 Wh kg−1 at a power density of 398 W kg−1. The superior performance is attributed to the synergistic interaction between LaMnO3 and CuO, which enhances redox activity, electron transport, and defect chemistry. These results demonstrate that controlled incorporation of CuO (∼10%) into LaMnO3 provides an optimal balance of conductivity, stability, and capacitance, underscoring the potential of perovskite–oxide composites for next-generation supercapacitors.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data are available in following link: https://zenodo.org/uploads/17614003.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ya00339c.
Acknowledgements
NSF-CBET, Award #: 2434668. The authors would like to thank the Department of Physics and Materials Science and the Integrated Microscopic Center (IMC) at the University of Memphis for their support of this research.References
- P. Simon and Y. Gogotsi, Materials for electrochemical capacitors, Nat. Mater., 2008, 7, 845–854 CrossRef.
- K. S. Kumar, N. Choudhary, Y. Jung and J. Thomas, Recent advances in two-dimensional nanomaterials for supercapacitor electrode applications, ACS Energy Lett., 2018, 3, 482–495 CrossRef.
- A. W. Anwar, A. Majeed, N. Iqbal, W. Ullah, A. Shuaib and U. Ilyas, et al., Specific capacitance and cyclic stability of graphene based metal/metal oxide nanocomposites: a review, J. Mater. Sci. Technol., 2015, 31(7), 699–707 CrossRef.
- Q. Wei, F. Xiong, S. Tan, L. Huang, E. H. Lan and B. Dunn, et al., Porous one-dimensional nanomaterials: design, fabrication and applications in electrochemical energy storage, Adv. Mater., 2017, 29, 1602300 CrossRef PubMed.
- P. Ma, N. Lei, B. Yu, Y. Liu, G. Jiang and J. Dai, et al., Flexible supercapacitor electrodes based on carbon cloth-supported LaMnO3/MnO nano-arrays by one-step electrodeposition, Nanomaterials, 2019, 9, 1676 CrossRef.
- Z. Yu, B. Duong, D. Abbitt and J. Thomas, Highly ordered MnO2 nanopillars for enhanced supercapacitor performance, Adv. Mater., 2013, 25, 3302–3306 CrossRef PubMed.
- B. Song, C. Sizemore, L. Li, X. Huang, Z. Lin and K. S. Moon, et al., Triethanolamine functionalized graphene-based composites for high performance supercapacitors, J. Mater. Chem. A, 2013, 3, 21789–21796 Search PubMed.
- J. Tripathi, V. Salve, Z. Ansari, S. Patange, P. Thakur and V. Chavan, et al., Tailored MIL-101, Ionics, 2025, 907–922 Search PubMed.
- G. Kothandam, G. Singh, X. Guan, J. M. Lee, K. Ramadass and S. Joseph, et al., Recent advances in carbon-based electrodes for energy storage and conversion, Adv. Sci., 2023, 10, 2301045 CrossRef PubMed.
- R. R. Salunkhe, Y. V. Kaneti and Y. Yamauchi, Metal–organic framework-derived nanoporous metal oxides toward supercapacitor applications: progress and prospects, ACS Nano, 2017, 11, 5293–5308 CrossRef PubMed.
- C. D. Lokhande, D. P. Dubal and O. S. Joo, Metal oxide thin film based supercapacitors, Curr. Appl. Phys., 2011, 11, 255–270 CrossRef.
- C. C. Hu and W. C. Chen, Effects of substrates on the capacitive performance of RuOx nH2O and activated carbon–RuOx electrodes for supercapacitors, Electrochim. Acta, 2004, 49, 3469–3477 CrossRef.
- J. T. Mefford, W. G. Hardin, S. Dai, K. P. Johnston and K. J. Stevenson, Anion charge storage through oxygen intercalation in LaMnO3 perovskite pseudocapacitor electrodes, Nat. Mater., 2014, 13, 726–732 CrossRef PubMed.
- X. Xu, S. Li, H. Zhang, Y. Shen, S. M. Zakeeruddin and M. Graetzel, et al., A power pack based on organometallic perovskite solar cell and supercapacitor, ACS Nano, 2015, 9, 1782–1787 CrossRef PubMed.
- P. Gao, M. Grätzel and M. K. Nazeeruddin, Organohalide lead perovskites for photovoltaic applications, Energy Environ. Sci., 2014, 7, 2448–2463 RSC.
- P. M. Shafi, N. Joseph, A. Thirumurugan and A. C. Bose, Enhanced electrochemical performances of agglomeration-free LaMnO3 perovskite nanoparticles and achieving high energy and power densities with symmetric supercapacitor design, Chem. Eng. J., 2018, 338, 147–156 CrossRef.
- V. Senthilkumar, Y. S. Kim, S. Chandrasekaran, B. Rajagopalan, E. J. Kim and J. S. Chung, Comparative supercapacitance performance of CuO nanostructures for energy storage device applications, RSC Adv., 2015, 5, 20545–20553 Search PubMed.
- A. Arya, S. Tanwar, M. Iqbal, A. Sharma and A. L. Sharma, Synergetic effect driven LaMnO3@ NiO composite based high energy semi-solid supercapacitor, J Energy Storage, 2025, 105, 114778 CrossRef.
- A. Dhakal, F. A. Perez, S. Karna and S. R. Mishra, LaMnO3-Mn3O4 nanocomposite: Synergetic effect towards high electrochemical performance, J. Alloys Compd., 2024, 1008, 176262 CrossRef.
- P. M. Shafi, V. Ganesh and A. C. Bose, LaMnO3/RGO/PANI ternary nanocomposites for supercapacitor electrode application and their outstanding performance in all-solid-state asymmetrical device design, ACS Appl. Energy Mater., 2018, 1, 2802–2812 Search PubMed.
- B. Patil, R. Jagtap, D. Narkhede and S. Pardeshi, Design of LaMnO3/rGO composite electrode materials for high-performance energy storage devices. Discover, Electrochemistry, 2025, 2, 1 Search PubMed.
- S. Nagamuthu, S. Vijayakumar and K. S. Ryu, Cerium oxide mixed LaMnO3 nanoparticles as the negative electrode for aqueous asymmetric supercapacitor devices, Mater. Chem. Phys., 2017, 199, 543–551 CrossRef.
- R. Packiaraj, K. Mahendraprabhu, P. Devendran, N. Nallamuthu, B. Palanivel and K. S. Venkatesh, et al., Electrochemical performances of ZnO–NiO–CuO mixed metal oxides as smart electrode material for solid-state asymmetric device fabrication, Energy Fuels, 2021, 361, 603–617 Search PubMed.
- K. M. Racik, A. Manikandan, M. Mahendiran, P. Prabakaran, J. Madhavan and M. V. A. Raj, Fabrication of manganese oxide decorated copper oxide (MnO2/CuO) nanocomposite electrodes for energy storage supercapacitor devices, Phys. E, 2020, 119, 114033 CrossRef.
- J. Gazquez, S. Bose, M. Sharma, M. A. Torija, S. J. Pennycook and C. Leighton, et al., Lattice mismatch accommodation via oxygen vacancy ordering in epitaxial La0.5Sr0.5CoO3-δ thin films, APL Mater., 2013, 1(1), 012105 CrossRef.
- A. Dhakal, F. A. Perez and S. R. Mishra, Electrochemical assessment of tailored Mn2O3 cuboidal hierarchical particles prepared using urea and Piperazine, Electrochim. Acta, 2024, 507, 145169 CrossRef.
- M. Sethi, U. S. Shenoy and D. K. Bhat, A porous graphene–NiFe2O4 nanocomposite with high electrochemical performance and high cycling stability for energy storage applications, Nanoscale Adv., 2020, 2, 4229–4241 Search PubMed.
- Y. Wang, Y. Song and Y. Xia, Electrochemical capacitors: mechanism, materials, systems, characterization and applications, Chem. Soc. Rev., 2016, 45, 5925–5950 RSC.
- J. Yan, T. Wei, B. Shao, Z. Fan, W. Qian and M. Zhang, et al., Preparation of a graphene nanosheet/polyaniline composite with high specific capacitance, Carbon, 2010, 48, 487–493 CrossRef.
- A. J. Bard, L. R. Faulkner and H. S. White, Electrochemical methods: fundamentals and applications, John Wiley and Sons, 2022 Search PubMed.
- U. Vladimir and I. Popov, Metrological characterization of X-ray diffraction methods for determination of crystallite size in nano-scale materials, Mater. Charact., 2013, 58, 883–891 Search PubMed.
- V. Salve, P. Agale, S. Balgude, S. Mardikar, S. Dhotre and P. More, Enhanced photocatalytic activity of SnO2@ gC3N4 heterojunctions for methylene blue and bisphenol-A degradation: effect of interface structure and porous nature, RSC Adv., 2025, 15, 15651–15669 RSC.
- M. Thommes, K. Kaneko, A. V. Neimark, J. P. Olivier, F. Rodriguez-Reinoso, J. Rouquerol, et al., Physisorption of gases, 2015 Search PubMed.
- A. Dhakal, F. Perez and S. R. Mishra, Unveiling the Synergistic Potential of LaMnO3-CeO2 Composites in Supercapacitor Applications, RSC Sustainability, 2025, 3, 5556–5570 Search PubMed.
- P. P. Ma, Q. L. Lu, N. Lei, Y. K. Liu, B. Yu and J. M. Dai, et al., Effect of A-site substitution by Ca or Sr on the structure and electrochemical performance of LaMnO3 perovskite, Electrochim. Acta, 2020, 332, 135489 CrossRef.
- H. Mo, H. Nan, X. Lang, S. Liu, L. Qiao and X. Hu, et al., Influence of calcium doping on performance of LaMnO3 supercapacitors, Ceram. Int., 2018, 44, 9733–9741 CrossRef.
- H. W. Nesbitt and D. J. A. M. Banerjee, Interpretation of XPS Mn (2p) spectra of Mn oxyhydroxides and constraints on the mechanism of MnO2 precipitation, Am. Mineral., 1998, 83, 305–315 CrossRef.
- J. Balamurugan, C. Li, T. D. Thanh, O. K. Park, N. H. Kim and J. H. Lee, Hierarchical design of Cu1−xNixS nanosheets for high-performance asymmetric solid-state supercapacitors, J. Mater. Chem. A, 2017, 5, 19760–19772 RSC.
- Z. Luo, R. Miao, T. D. Huan, I. M. Mosa, A. S. Poyraz and W. Zhong, et al., Mesoporous MoO3–x material as an efficient electrocatalyst for hydrogen evolution reactions, Adv. Energy Mater., 2016, 6, 1600528 CrossRef.
- X. Lu, Y. Zeng, M. Yu, T. Zhai, C. Liang and S. Xie, et al., Oxygen-deficient hematite nanorods as high-performance and novel negative electrodes for flexible asymmetric supercapacitors, Adv. Mater., 2014, 26, 3148–3155 CrossRef PubMed.
- M. Kandasamy, S. Sahoo, S. K. Nayak, B. Chakraborty and C. S. Rout, Recent advances in engineered metal oxide nanostructures for supercapacitor applications: experimental and theoretical aspects, J. Mater. Chem. A, 2021, 9, 17643–17700 RSC.
- H. Zhao, Q. Zhu, X. Ye, L. Wang and S. Dong, Electrochemical properties of LaMO3 (M= Cr, Mn, and Co) perovskite materials, Coatings, 2024, 14, 147 CrossRef.
- X. Ye, S. Dong, X. Jin, J. Wei, L. Wang and Y. Zhang, Enhancement in the electrochemical performance of strontium (Sr)-doped LaMnO3 as supercapacitor materials, Coatings, 2022, 12, 1739 CrossRef.
- A. Ajith, E. Thomas, C. H. Suresh and R. R. Philip, Advances and strategies in nanoarchitectured TiO2-based electrodes for supercapacitors, J. Energy Storage, 2025, 111, 115289 CrossRef.
- H. Lindström, S. Södergren, A. Solbrand, H. Rensmo, J. Hjelm and A. Hagfeldt, et al., Li+ ion insertion in TiO2 (anatase). 2. Voltammetry on nanoporous films, J. Phys. Chem. B, 1997, 101, 7717–7722 CrossRef.
- J. E. Randles, A cathode ray polarograph. Part II. The current-voltage curves, Trans. Faraday Soc., 1948, 44, 327–338 RSC.
- T. C. Liu, W. G. Pell, B. E. Conway and S. L. Roberson, Behavior of molybdenum nitrides as materials for electrochemical capacitors: comparison with ruthenium oxide, J. Electrochem. Soc., 1998, 145, 1882 CrossRef.
- A. Dhakal, S. R. Mishra and F. Perez, Urea-Driven Hydrothermal Synthesis of Mn2O3: Electrochemical Performance Across Various Electrolytes for Supercapacitor Applications, Energy Adv., 2025, 4, 878–895 RSC.
- C. Guan, J. Liu, C. Cheng, H. Li, X. Li and W. Zhou, et al., Hybrid structure of cobalt monoxide nanowire@ nickel hydroxidenitrate nanoflake aligned on nickel foam for high-rate supercapacitor, Energy Environ. Sci., 2011, 4, 4496–4499 RSC.
- Y. Gao, S. Chen, D. Cao, G. Wang and J. Yin, Electrochemical capacitance of Co3O4 nanowire arrays supported on nickel foam, J. Power Sources, 2010, 195, 1757–1760 CrossRef CAS.
- J. Wang, J. Polleux, J. Lim and B. Dunn, Pseudocapacitive contributions to electrochemical energy storage in TiO2 (anatase) nanoparticles, J. Phys. Chem. C, 2007, 111, 14925–14931 Search PubMed.
- C. Peng, L. Yanyue, J. Yuanyuan, W. Haiyan and L. Zijiong, Synthesis of LaMnO3-reduced graphene oxide or Sr composite and their application in electrochemical properties, Mater. Res. Express, 2020, 7, 25023 Search PubMed.
- H. M. El Sharkawy, A. S. Dhmees, A. R. Tamman, S. M. El Sabagh, R. M. Aboushahba and N. K. Allam, N-doped carbon quantum dots boost the electrochemical supercapacitive performance and cyclic stability of MoS2, J. Energy Storage, 2020, 27, 101078 CrossRef.
- A. Noori, M. F. El-Kady, M. S. Rahmanifar, R. B. Kaner and M. F. Mousavi, Towards establishing standard performance metrics for batteries, supercapacitors and beyond, Chem. Soc. Rev., 2019, 48, 1272–1341 RSC.
| This journal is © The Royal Society of Chemistry 2026 |