 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Bio-based composites of alginate, cellulose, and Moringa oleifera for heavy metal removal in water purification: a comprehensive and critical review of mechanisms, fabrication, and performance
Abimbola Oluwatayo
Orisawayi
 *ab,
Krzysztof K.
Koziol
a and
Sameer S.
Rahatekar
a
*ab,
Krzysztof K.
Koziol
a and
Sameer S.
Rahatekar
a
aComposites and Advanced Materials Centre, Faculty of Engineering and Applied Sciences, Cranfield University, Bedfordshire MK43 0AL, UK. E-mail: abimbola.orisawayi@cranfield.ac.uk; bimboris_t@yahoo.com
bDepartment of Mechanical Engineering, School of Engineering and Engineering Technology, Olusegun Agagu University of Science and Technology (OAUSTECH), Okitipupa, Nigeria. E-mail: ao.orisawayi@oaustech.edu.ng
First published on 23rd December 2025
Abstract
The escalating prevalence of heavy metal contamination in aquatic ecosystems, driven by industrialisation, urbanisation, and population growth, has necessitated the development of sustainable and efficient water purification technologies. This review critically evaluates recent advances in developing and applying bio-based composites comprising sodium alginate, cellulose, and Moringa oleifera (M. oleifera) to remove heavy metals from aqueous systems. The review examines the physicochemical characteristics, adsorption mechanisms, and synergistic properties of these biopolymers, emphasising the role of the active compounds in each. The deduction from the comparative study of this review reveals cellulose-based composites demonstrating the highest overall adsorption performance, with several systems exceeding 1000 mg g−1 across different heavy metals. Although alginate composites achieve the highest single reported capacity, 1742 mg g−1 for Pb2+, their performance is more dependent on chemical or nanoparticle functionalisation. M. oleifera biosorbents show moderate adsorption capacities, with improvements mainly observed after chemical modification. Overall, cellulose composites exhibit the most consistent and versatile adsorption behaviour among the three materials. This review identifies potential applications, highlights key research gaps, and outlines future directions for advancing bio-based composite materials as viable solutions for sustainable water treatment.
Environmental significanceThis review highlights the potential of bio-based composites comprising alginate, cellulose, and Moringa oleifera as sustainable alternatives to conventional water treatment materials. These biopolymers, derived from renewable resources, offer low toxicity, biodegradability, and effective adsorption of toxic heavy metals from aqueous environments. Their application not only mitigates environmental pollution but also reduces dependency on synthetic, non-biodegradable materials that contribute to secondary waste generation. By valorising agricultural by-products and natural resources, such composites support circular economy principles and promote greener technologies for water purification, aligning with global efforts to address environmental sustainability and resource conservation. |
1 Introduction
The rapid growth of the global population has significantly increased industrial activities, reducing the availability of clean water.1–3 Water is one of the most pressing environmental concerns, making obtaining safe and affordable clean water increasingly challenging. Heavy metal contamination in water is also a major concern, arising from both natural and human activities.4–6 Both industrial processes and natural phenomena, such as the weathering of metal-rich rocks and geothermal activities, contribute to heavy metal pollution in water bodies.7–9 Drinking water is an invaluable resource for life, and ensuring access to water and sanitation by 2030 is a key objective outlined by the United Nations Sustainable Development Goals (UNSDGs).10Water is an essential resource, and numerous statistics have been collected to assess various aspects of its usage, availability, and quality. Several international organisations actively gather and analyse water-related data, including the United Nations Educational, Scientific and Cultural Organisation (UNESCO),11 the United Nations Children's Fund (UNICEF), and many others.12 According to data from 57 countries in 2015, approximately 84 Litres of wastewater per capita were generated, yet only 29 Litres underwent treatment. By 2021, global household wastewater production had reached 271 billion cubic meters, with treatment rates improving to 55.5% based on data collected from 234 countries.13,14 Furthermore, studies indicate that approximately 70% of the Earth's surface is covered by water, of which only 2.5% consists of freshwater. A mere 1% of this freshwater is readily accessible for human use.15 The current global population of 7.6 billion people must share this limited resource. The United Nations projects that by 2050, the global population will reach 9.8 billion, with approximately 4 billion people expected to experience water scarcity. This will exacerbate the existing crisis, as over two billion individuals already lack access to clean water.16 To address this challenge, efficient water treatment strategies must be developed to treat wastewater and natural water sources while ensuring sustainability through renewable energy sources.17–19
Various methods and materials have been proposed for tackling water contamination, with a growing emphasis on biopolymer-based solutions. Biopolymers, derived from natural sources such as cellulose, alginate (from brown algae), and chitosan (from crustacean shells), have gained considerable attention for water purification due to their biodegradability, eco-friendliness, and high adsorption capacity for heavy metals.20–22 Their properties, such as high adsorption capacity and eco-friendliness, make them suitable materials for water purification applications and have been widely explored in recent studies.23
Among these, cellulose is recognised as one of the most abundant polysaccharides on earth, characterised by its high mechanical strength, hydrophilicity, and ability to form stable composites.24,25 Alginate, extracted primarily from brown algae, is also rapidly gaining traction as a versatile biopolymer in different fields due to its unique gel-forming capabilities and non-toxic nature.26,27 The growing market for alginate reflects its increasing utilisation in water treatment, where it serves as an efficient medium for adsorbing heavy metal ions. Combining cellulose and alginate in composite forms presents a promising approach for enhancing adsorption efficiency and mechanical properties, making these biopolymers valuable for sustainable water treatment applications.21,24,28
In addition to biopolymers, M. oleifera has been extensively investigated as a cost-effective, eco-friendly biosorbent for removing heavy metal ions from water.29–32 These seeds contain natural cationic proteins and bioactive compounds that facilitate ion exchange and metal binding, improving water purification efficiency.33 Studies have also demonstrated that M. oleifera seed pods can effectively remove mixtures of metals in wastewater, achieving optimal removal efficiency under specific conditions, such as a 60-minute contact time and a 1.0 gram sorbent dose.34 The ability of M. oleifera to function as both a coagulant and an adsorbent positions it as a dual-function material for water treatment, enhancing its potential for integration with biopolymers like cellulose and alginate to develop advanced composite materials aimed at heavy metal ion contamination.27,35
Although biopolymers and M. oleifera seed powders benefit water treatment, few studies have examined their hybrid composites. This presents a significant research gap in developing and characterising hybrid composites.36 Few studies have been reported. Development of hybrid electrospun alginate-pulverised M. oleifera composites was done by Orisawayi et al.37 In their studies, pulverised M. oleifera at a minimum dose suspension was incorporated into sodium alginate fibre using the electrospinning techniques.
Another study reported the development of effective biosorbents made from combining M. oleifera and alginate beads for uranium removal from aqueous solutions. Orisawayi et al.32 further developed sodium alginate fibres through wet-spinning. In contrast, more recent investigations have combined sodium alginate with polyethyleneimine and M. oleifera leaves–seed beads for uranium adsorption, including isotherm and kinetic analyses.38 These composite systems have demonstrated improved adsorption capacity and favourable structural characteristics.
The selection of alginate, cellulose, and M. oleifera in this study stems from their complementary physicochemical and functional properties relevant to heavy-metal removal. Alginate offers a biocompatible, carboxyl-rich matrix with strong ion-binding capacity and efficient gel-forming behaviour, making it highly suitable for capturing multivalent metal ions.39–41 Cellulose, the most abundant natural polysaccharide, provides mechanical stability, a high surface area, and additional hydroxyl groups that boost adsorption.39,42–47 In contrast, M. oleifera seeds supply bioactive, cationic proteins and coagulant molecules capable of binding and aggregating dissolved metal ions.34,48,49 Although other biopolymers such as pectin, starch, and chitosan have been widely studied, they do not collectively offer this combination of mechanical robustness, adsorption efficiency, natural coagulation activity, and environmental sustainability.50–52
Therefore, the novelty of this review arises from its focus on evaluating alginate, cellulose, and M. oleifera as distinct materials for heavy-metal removal, combined with an assessment of how their complementary traits could be strategically melded to improve adsorptive performance. While many studies and reviews have examined these materials separately or with other biopolymers, none have explored their combined potential within a single analytical framework, offering a new perspective for designing more effective and sustainable adsorbent systems.
The study first outlines heavy metal contamination as a significant environmental concern, summarising key pollutants and regulatory limits set by the United States Environmental Protection Agency (EPA), World Health Organisation (WHO) and European Union (EU), including the origin or sources of the heavy metals. It then evaluates the limitations of conventional treatment methods, such as chemical precipitation, ion exchange, and membrane filtration, emphasising the need for sustainable alternatives. The focus then shifts to biopolymers, particularly sodium alginate and cellulose, exploring their adsorption mechanisms, composite formulations, and integration with M. oleifera to enhance performance. Fabrication techniques such as electrospinning and wet spinning are also reviewed for their role in optimising material properties. Having established the urgency of water pollution and the potential of biopolymer-based solutions, it is crucial first to understand the nature, sources, and health implications of the primary contaminants and heavy metals that threaten aquatic systems.
2 Background on heavy metals
Heavy metal ions such as lead (Pb2+), cadmium (Cd2+), cobalt (Co2+), nickel (Ni2+), barium (Ba2+), copper (Cu2+), chromium in both trivalent and hexavalent states (Cr3+/Cr6+), zinc (Zn2+), mercury (Hg2+), and arsenic in trivalent and pentavalent forms (As3+, As5+) constitute major contaminants in aquatic ecosystems. Their elevated toxicity and persistence in natural watersmake them a significant environmental concern.53,54 There are several primary sources of heavy metal ions. Fig. 1 illustrates the different sources of environmental pollution caused by heavy metals and the adverse effects of the metals on pollution by heavy metal ions.50,55,56Fig. 1a shows the primary industrial sources, such as the mining industries,57 textile industries,58,59 thermal and nuclear plants associated with the cement industry,60 the manufacturing and conservation of wood, dye production,61 metal plating and those associated with the steel manufacturing industries,62 energy and water cooling processes,30 the production of photographic materials,63 the manufacturing of various corrosive paints,64 and other industrial activities in the global oil and gas industries.65–67 | ||
| Fig. 1 (a and b) are the different sources of environmental pollution caused by heavy metals, and (c) the adverse effects of commonly encountered heavy metals on other human organs (all figures are adapted and (c) was modified with permission, Licensed under Elsevier's terms).50,55,56 | ||
However, heavy metal contamination is not limited to industrial activities alone. Fig. 1b shows a broader perspective, incorporating additional sources such as urban solid waste, wastewater effluents, e-waste, biosolids, fertilisers, pesticides, corrosion, pharmaceutical products, and natural occurrences, including volcanic eruptions. These diverse contamination sources contribute significantly to environmental pollution, making the development of sustainable remediation strategies imperative. Heavy metal ions are often described as metallic forms of elements that are mostly denser than water and have a large atomic radius.68 Heavy metal ions are dangerous and more prevalent, resulting from the persistent half-life.69,70 The common organic compounds found in most water bodies can be degraded over time. Still, when polluted into water bodies, the heavy metals remain an environmental issue as most of them are difficult to decompose in the water.
The United States Environmental Protection Agency (EPA) with Maximum Contaminant Levels (MCLs),71,72 The World Health Organisation (WHO), and the European Union (EU), with the maximum permissible level,73,74 have established regulatory limits for these contaminants to protect water quality and public health.68,75 Lead has been extensively studied as one of the metals causing environmental pollution, resulting from its high level of toxicity and often widespread presence.76,77 Contamination from lead is common and is primarily due to its use in many plumbing infrastructures, resulting from the erosion of natural deposits and its presence in most automobile batteries.78 The presence of lead, even at low blood concentrations of 1–2 µg dL−1, lead exposure may lead to severe health effects, including neurodevelopmental, cardiovascular, renal, and reproductive issues, and in children, could show slight deficits in attention span.79 The EPA MCL is 0.01 mg L−1, with WHO and EU also maintaining a 0.01 mg L−1 limit.
In addition, metal ions such as cadmium are another frequently encountered heavy metal pollutant because they are primarily released in most industrial processes. The EPA MCL is 0.005 mg L−1, while WHO and EU enforce limits of 0.003 mg (ref. 80) chromium is well-documented as an environmental contaminant and primarily originates from most industrial activities and processes, such as electroplating, textile manufacturing, and the stainless steel industry. In most research, this metal has been highlighted as it's toxic and carcinogenic, therefore causing concern. The EPA sets an MCL of 0.1 mg L−1, whereas the WHO and the EU impose stricter limits of 0.05 mg L−1.81
Mercury is also a highly toxic heavy metal introduced into the environment through various industrial activities, and processed are often contain mercury and waste in water bodies and can cause challenges for aquatic ecosystems; reports show that mercury can transform into methylmercury, known as a bioavailable form, that is accumulated in marine bodies and therefore affecting the aquatics organisms. This poses a serious Neurotoxin, as kidney damage bioaccumulates in aquatic organisms and is a health threat to humans consuming contaminated seafood. The EPA enforces an MCL of 0.002 mg L−1, the WHO sets 0.006 mg L−1, while the EU has a more stringent limit of 0.001 mg L−1.82,83
Arsenic, a naturally occurring metalloid, poses serious health risks. Arsenic can cause severe health conditions in the skin, causing skin damage or problems with the circulatory system, cancer, and cardiovascular diseases.7,84 It is a significant contaminant in the groundwater; due to its high toxicity, the EPA, WHO, and EU all impose a maximum limit of 0.01 mg L−1, particularly in regions where mainly agricultural activities have historically involved arsenic-based pesticides.82,84
Other metals, such as nickel, barium, copper, and zinc, pose significant environmental and health risks due to their persistence in water bodies. These metals also enter aquatic ecosystems through industrial discharge, mining, and improper waste disposal, contaminating drinking water sources and affecting marine life. Nickel exposure can lead to allergic reactions, respiratory issues, and carcinogenic effects, disrupting aquatic microbial activity. Nickel is commonly found in metal alloys, including mining waste and industrial effluents. Barium contamination originates from the oil drilling, glass, and paint industries. Soluble barium compounds pose health risks, causing hypertension, muscle weakness, and neurological disorders.84–86
Copper and zinc are essential metals but become toxic in excess, leaching from plumbing, mining, and fertilisers. Copper bioaccumulates in fish and amphibians, disrupting metabolism and causing liver, kidney, and neurological issues in humans. Zinc pollution can lead to immune suppression, developmental problems, and metabolic disorders, ultimately affecting fish growth and disrupting the balance of phytoplankton. The presence of these metals in water demands effective pollution control, water treatment, and stricter regulations to mitigate their toxic effects on human health and ecosystems.87–89
Table 1 provides a comparative overview of major heavy metal contaminants, their potential health risks, and their regulatory limits established by the EPA, WHO, and EU. Fig. 1c shows a retrieved study from a previous study retrieved from the literature, which shows the adverse effects of commonly encountered heavy metals on different human organs.56 Furthermore, these standards are crucial for maintaining water safety, and exposure to heavy metals can have severe biological consequences, affecting multiple human organs. This illustration complements the regulatory data presented in Table 1 by emphasising the physiological risks associated with prolonged exposure to heavy metals.
| Heavy metals68,71–74 | Potential health effects from long-term exposure above the MCL (unless specified as short-term)68,71–74 | EPA (drinking water MCL)71,72 (mg L−1) | WHO guideline value (MPL)74 (mg L−1) | EU drinking water standard (MPL)73 (mg L−1) | Sources of contaminants in drinking water68,71–74 | Industrial origins for the heavy metals |
|---|---|---|---|---|---|---|
| Lead (Pb) | Neurodevelopmental effects, cardiovascular, renal, and reproductive issues, children could show slight deficits in attention span | Action level = 0.010 | 0.01 | 0.01 | Corrosion of household plumbing systems; erosion of natural deposits | Battery manufacturing, metal smelting and refining, paint and pigment industries, ammunition production, plumbing and soldering waste |
| Cadmium (Cd) | Kidney damage, gastrointestinal toxicity, carcinogenic effects | 0.005 | 0.003 | 0.005 | Corrosion of galvanised pipes; erosion of natural deposits; discharge from metal refineries; runoff from waste batteries and paints | Electroplating industries, Ni–Cd battery production, PVC and plastic stabilisers, mining and smelting operations, pigment manufacturing |
| Cobalt (Co) | Allergic reactions, cardiovascular effects, and potential carcinogenicity | Not established | Not established | Not established | Industrial sources, mining activities, alloy production | Battery manufacturing (especially Li-ion and rechargeable batteries), superalloy and aerospace component production, mining and ore smelting of copper and nickel, chemical catalyst manufacturing, pigments, ceramics, and glass colouring industries, electroplating and metal finishing, cement and steel production waste |
| Nickel (Ni) | Respiratory issues, carcinogenic effects, microbial toxicity | Not established | 0.07 | 0.02 | Industrial discharges; erosion of natural deposits; nickel plating and battery industries | Electroplating and surface finishing, stainless steel manufacturing, mining and refinery effluents, catalyst production |
| Barium (Ba) | Hypertension or increase in blood pressure, muscle weakness, neurological disorders | 2 | 0.7 | Not established | Discharge of drilling wastes; discharge from metal refineries; erosion of natural deposits | Oil and gas drilling operations (example: barite-based drilling muds), paints, pigments, and ceramics manufacturing, glass and electronic component production, metal refining and alloy processing, rubber and plastic additives industries, fireworks and pyrotechnics, waste from chemical manufacturing processes |
| Copper (Cu) | Liver, kidney, neurological damage, bioaccumulation in aquatic species | Action level = 1.3 | 2.0 | 2.0 | Corrosion of household plumbing systems; erosion of natural deposits | Mining and smelting, electrical and electronics industries, metal pipe corrosion, pesticide formulation |
| Chromium (Cr) | Carcinogenic, severe respiratory effects, industrial exposure risk | 0.1 | 0.05 | 0.05 | Discharge from steel and pulp mills; erosion of natural deposits | Leather tanning, stainless steel and alloy production, electroplating, dye and pigment industries, wood preservation processes |
| Zinc (Zn) | Immune suppression, developmental issues, metabolic disorders | 5 | Not established | Not established | Corrosion of galvanised pipes; industrial discharges; erosion of natural deposits | Galvanisation industries, rubber and tyre manufacturing, pigment and paint industries, brass alloy production |
| Mercury (Hg) | Neurotoxin, kidney damage, bioaccumulates in marine organisms | 0.002 | 0.006 | 0.001 | Erosion of natural deposits; discharge from refineries and factories; runoff from landfills and croplands | Chlor-alkali plants, thermometer and lamp manufacturing, gold and silver mining, dental amalgam waste, chemical production processes |
| Arsenic (As) | Skin damage or problems with the circulatory system may have increased the risk of getting cancer | 0.010 as of 01/23/06 | 0.01 | 0.01 | Erosion of natural deposits; runoff from orchards, runoff from glass and electronics production wastes | Mining and ore processing, pesticide and herbicide manufacturing, semiconductor and microelectronics industries, and coal combustion effluents |
The legally enforceable Maximum Contaminant Levels (MCLs) ensure the safety of drinking water. The World Health Organisation (WHO) provides guidelines, values, and Maximum Permissible Levels (MPLs) based on health risk assessments. The European Union (EU) sets strict regulatory limits on drinking water quality that are mandatory for all EU member states.69–72
Several treatment technologies have been developed to address the environmental challenge posed by heavy metal contamination due to their hazardous effects and regulatory significance in water systems. The following section critically examines these technologies for the removal of heavy metals from water.
3 Water treatment technologies for removing heavy metals
3.1 Chemical precipitation
Chemical precipitation has been used and described as an effective method for removing heavy metals, primarily from wastewater. Chemical precipitation is widely used in industrial wastewater treatment due to its simplicity, cost-effectiveness, and established technology.90,91 This method uses chemical reagents that react with most metal ions to form an insoluble precipitate.92 Studies show that the primary precipitation mechanisms include hydroxide and sulphide precipitation, which facilitate the removal of these metal ions during the process. However, the main limitations of this method involve difficulties in removing mixed metals due to pH levels that may be difficult to control when hydroxide precipitation is used.93,94 In addition, there is also a risk of secondary contamination, particularly from sulphide precipitation, which can sometimes lead to the formation of toxic hydrogen sulphide (H2S) gas, as reported.953.2 Ion exchange
Another widely used method is ion exchange, which is often applied in water purification technologies, as this method relies on ion exchange to remove metal ions. During the process, ion exchange media include zeolite resins and synthetic organic polymers.96 These methods have been proven effective for eliminating cations and anions from freshwater, ensuring high removal efficiency. However, this method has several drawbacks, including the requirement for pretreatment and chemical regeneration, which can lead to secondary pollution due to the materials used. Therefore, ion exchange leaves some secondary pollution after water treatment.97 Studies suggest that this method is less effective for highly concentrated mixed-metal wastewater, making it more suitable for applications involving mixed heavy-metal solutions from aqueous solutions.97,983.3 Membrane separation
Membrane separation technologies are emerging methods employed for pressure-driven processes such as ultrafiltration, nanofiltration, reverse osmosis, concentration, and removing some heavy metal ions.99,100 This method is advantageous due to its simple operation, low energy consumption, and absence of significant phase changes, making it an environmentally friendly alternative. However, challenges associated with this method include the high cost of membrane materials and their susceptibility to fouling and degradation, which may reduce the long-term efficiency of the process.101 Despite these limitations, membrane separation remains a valuable technology for water treatment, particularly in removing low-concentration contaminants or pollutants from water.101,1023.4 Electrochemical technologies
Electrochemical methods, including electrocoagulation, electrodeposition, electrooxidation, and electrolocation, have been extensively explored for heavy metal removal. These techniques involve the application of electrical currents to induce coagulation, charge neutralisation, and precipitation of heavy metal ions.103 Electrochemical processes are known for their high removal efficiency, ease of operation, and minimal sludge production, reducing the need for additional conditioning treatments. However, their applicability is often limited by energy consumption, electrode material degradation, and the potential formation of secondary contaminants.104,1053.5 Bioremediation
The bioremediation technique is another method that utilises biological processes for water treatment. This approach includes microbial remediation and phytoremediation, which involve using microorganisms or plants to degrade, immobilise, or remove heavy metals from water bodies.106 Studies have shown that this method has been proven to be an environmentally sustainable method. It is also cost-effective and has been successfully applied for the restoration of the most polluted sites. However, bioremediation has significant limitations, including overdependence on low metal concentrations and long remediation cycles, making it challenging to scale up the process for industrial applications.107–1093.6 Adsorption
Over the decades, adsorption has emerged as one of the most efficient and widely used methods for removing heavy metals from contaminated water.55 The process has emerged as a promising alternative for water treatment. Adsorption is the process in which ions, atoms, or molecules adhere to the surface of a solid material. It differs from absorption, which involves the penetration of molecules into the interior of a solid.110,111 Based on the forces governing this phenomenon, adsorption is categorised as physisorption or chemisorption.112 This method relies on interfacial interactions between metal ions (adsorbate) and the materials used for their removal (adsorbent). Various media can be utilised to facilitate contaminant removal through mechanisms such as pore filling, surface binding, and chemical interactions.113 Some of the materials reported for use include activated carbon, carbon nanotubes, wood sawdust, alginate, cellulose, M. oleifera, chitosan, polymeric hydrogels, ion-exchange resins, and their composites. As illustrated in Fig. 2a, wastewater treatment methods are categorised into electrochemical treatments, physicochemical processes, and adsorption-based processes, highlighting their applications in contaminant removal.90,91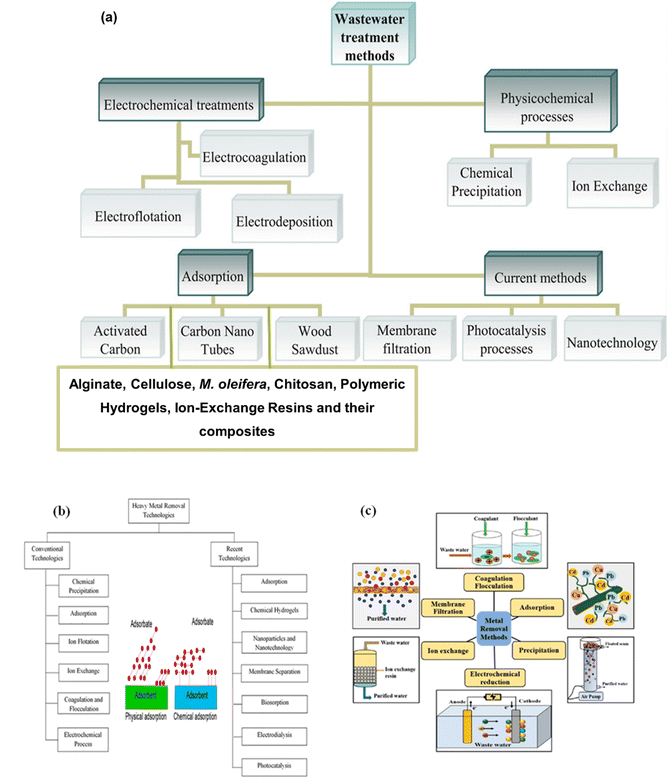 | ||
| Fig. 2 (a) Water treatment of remediation methods, including electrochemical, physicochemical, and adsorption-based processes (modified with permission, Licensed under Wiley's terms) (b), (modified with permission, Licensed under Elsevier's terms) and (c) heavy metal removal technologies, comparing conventional and advanced techniques, (adapted with permission, Licensed under ACS publication's terms).90,91,114 | ||
Fig. 2(b and c) provide a comparative overview of heavy metal removal technologies, distinguishing between conventional methods, such as chemical precipitation, ion exchange, and electrochemical processes, and advanced techniques, including nanotechnology, membrane filtration, and photocatalysis. The inset in Fig. 2a further illustrates the physical and chemical adsorption mechanisms of different adsorbate–adsorbent interactions, demonstrating their effectiveness in pollutant removal.91 Unfortunately, most reported methods or techniques are associated with high costs, operational complications, low efficiency, excessive chemical use, and secondary pollutants, which restrict their applications. With the development of highly flexible, easy-to-operate, and efficient adsorbent designs, adsorption has emerged as a promising alternative for water treatment. Adsorption is highly advantageous due to its simplicity, cost-effectiveness, high selectivity, and ability to treat dilute wastewater. The ability to recycle adsorbents has been reported to minimise secondary pollution, making adsorption a preferred choice for water treatment applications.
While conventional technologies demonstrate varying degrees of effectiveness, many are limited by high costs, secondary pollution, or low selectivity. These limitations have spurred the exploration of sustainable alternatives, particularly those derived from bio-based materials, as discussed in the next section.
4 Bio-based biodegradable composites and blends for water purification
4.1 Overview of sustainable biopolymer composites
Biopolymers are naturally occurring polymers produced by living organisms. Biological resources, including plants, animals, agricultural residues, and microorganisms, are viable feedstocks for synthesising biopolymers. Fig. 3 shows a typical classification and characteristics of biopolymers that have been reported.29 Among the primary sources derived from agriculture and plants are corn stalks, maize, wheat, potatoes, and barley. | ||
| Fig. 3 Classification and properties of biopolymers (adapted with permission, Licensed under Elsevier's terms).29 | ||
Biopolymers consist of monomeric units such as nucleotides, saccharides, or amino acids that form nucleic acids, carbohydrates, and proteins.112–114 Biopolymers are known to be renewable and eco-friendly alternatives to most synthetic polymers derived from fossil fuels.115–120 Biopolymers have gained significant attention due to their biodegradability and potential to address environmental challenges.121–124 The projections indicate that global plastic production is expected to surpass 1800 million metric tons annually by 2050. The focus on biopolymers, primarily cellulose and alginate, for water treatment is well-justified due to their abundant functional groups, which facilitate the efficient adsorption of heavy metal ions and other pollutants. Over the decades, several studies have highlighted the environmental issues associated with synthetic polymers, emphasising the need for biodegradable alternatives. Kogje et al.125 found that biopolymers derived from natural sources minimise plastic waste and have higher biodegradability than standard plastics. Similarly, Emre et al.126 also demonstrated the potential of polysaccharide-based biopolymers to reduce environmental pollution through improved adsorption. Researchers have documented the efficiency of biopolymers such as cellulose, alginate, and chitosan in adsorbing heavy metals from aqueous solutions.82,83,127,128
Moreover, alginate has emerged as a promising biopolymer, which also contains carboxyl groups that play a crucial role in the ion exchange process, making it an effective adsorbent for heavy metals like cadmium, chromium, and other metal ions.129,130 Several studies have also demonstrated the effectiveness of cellulose–alginate hydrogels in contaminant removal. In particular, the hydrogels have been shown to substantially enhance the adsorption efficiency of both dyes and heavy metal ions, achieving up to approximately 85% removal of methylene blue, which is associated with metal ions.131,132 The tensile strength and durability of cellulose, combined with the gel-forming ability of alginate, ensure the formation of stable and effective adsorbent composites. The environmental sustainability and cost-effectiveness of cellulose and alginate instead of synthetic polymers align with the increasing demand for eco-friendly water treatment materials. The interaction between the hydroxyl groups in cellulose and the carboxyl groups in alginate enhances the ion-exchange interactions and adsorption capacity of these hydrogels, making them practical for water purification.133,134Fig. 4 presents a detailed schematic representation of the sources and structures of cellulose and alginate, along with their physical and chemical modification methods to enhance their performance in water purification applications. Additionally, it categorises the significant approaches for modifying these biopolymers to improve their functionality. These modifications, categorised into physical and chemical, encompass blending, ultrasonic treatment, cross-linking, focusing on the use of crosslinking agents such as Ethylenediaminetetraacetic Acid (EDTA), Gamma-Linolenic Acid (GLA), Ethylene Glycol Monobutyl Ether (EGBE), Epichlorohydrin (ECH), and Polyethylene Glycol (PEG), including grafting to enhance the material's adsorption efficiency, mechanical stability, and chemical resistance in water remediation applications. These modification techniques are essential in tailoring cellulose–alginate composites for optimised performance in environmental applications.
 | ||
| Fig. 4 Schematic representation of the sources and structures of cellulose and alginate, along with their physical and chemical modification methods aimed at enhancing their performance in water purification applications (modified with permission, Licensed under Elsevier's terms).21 | ||
The adsorption and regeneration mechanisms of biopolymeric composites are very crucial. These have been extensively studied for their effectiveness in removing heavy metals. Understanding these mechanisms is essential for optimising their performance in water treatment applications. Fig. 5(a) presents a reported adsorption mechanism illustrating the interaction of metal ions (M+) with active functional groups in biopolymeric composites. The process involves electrostatic attraction, ion exchange, and surface complexation, facilitated by hydroxyl (–OH), carboxyl (–COO−), and amine (–NH2) groups.135 Adsorption efficiency is influenced by pH, where ion exchange dominates at lower pH levels. At the same time, electrostatic and surface complexation mechanisms become more prominent at higher pH values, as observed in several studies.136,137 The adsorption performance of cellulose–alginate composites has been well-documented, with removal efficiencies varying depending on the composite structure, porosity, and availability of functional groups. Furthermore, Fig. 5(b) highlights various regeneration strategies for restoring adsorption capacity. These include chemical regeneration using eluents such as Sodium hydroxide (NaOH), Hydrochloric acid HCl, Ethylenediaminetetraacetic acid (EDTA), and Sulfuric acid (H2SO4), as well as physical and biological treatments like oxidation, ultrasound, and thermal degradation. Integrating effective regeneration methods ensures the long-term usability of biopolymeric adsorbents, making them viable options for sustainable water purification.
 | ||
| Fig. 5 (a) Proposed adsorption mechanism of biopolymeric composites for heavy metal removal, and (b) regeneration methods and chemicals used for biopolymeric composites during the adsorption–desorption process (adapted with permission, Licensed under Elsevier's terms).21 | ||
4.2 Sodium alginate and its composites
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 tons, representing less than 10% of biosynthesised alginate. Therefore, there is considerable potential for alginate to be utilised in the design of sustainable composite materials. Primarily, alginate is extracted from brown seaweed algae such as (Ascophyllum spp., Laminaria spp., Macrocystis pyrifera, Sargassum spp, Alario, Ecklonia, Eisenia, Nercocystis, Sargassum, Cystoseira, Fucus, and several others).139 Studies show that seaweed-derived alginate is the most commercially utilised form, as bacterial alginate presents an alternative source with distinct advantages for several applications.140,141 Typically, alginate extracted from brown algae is treated with various chemicals at different synthesis stages. Briefly, the production process of sodium alginate begins with the harvesting and drying of seaweed, after which it undergoes mechanical processing to be converted into algal powder.142 This powder will be treated with hydrochloric acid (HCl) to extract the alginic acid, which serves as the precursor for sodium alginate including sodium carbonate (Na2CO3) as part of the extraction process. The extracted alginic acid will be washed, filtered, and treated with sodium hydroxide (NaOH) to form a sodium alginate (SA) solution.143 The solution is then further treated with HCl to enhance the purity and produce an alginic acid gel. The samples will then be neutralised with alkali agents such as sodium hydroxide and/or sodium carbonate, converting them into sodium alginate, a water-soluble polymer widely used across various industries.144 The purification of the extracted alginate was conducted through a chemically assisted process before filtration and drying. Specifically, the crude alginate was subjected to sequential treatments using calcium chloride (CaCl2), sodium chloride (NaCl), or further treated with ethanol to remove residual impurities, enhance polymer purity, and improve the physicochemical characteristics of the final biopolymer. The extraction process of alginate is illustrated in Fig. 6a, showing the key steps involved in alginate preparation from raw seaweed sources and its subsequent transformation into sodium alginate and its applications in the adsorption process, retrieved from the literature. In contrast, other literature shown in Fig. 6b explains the Industry process of sodium alginate extraction via calcium precipitation.
000 tons, representing less than 10% of biosynthesised alginate. Therefore, there is considerable potential for alginate to be utilised in the design of sustainable composite materials. Primarily, alginate is extracted from brown seaweed algae such as (Ascophyllum spp., Laminaria spp., Macrocystis pyrifera, Sargassum spp, Alario, Ecklonia, Eisenia, Nercocystis, Sargassum, Cystoseira, Fucus, and several others).139 Studies show that seaweed-derived alginate is the most commercially utilised form, as bacterial alginate presents an alternative source with distinct advantages for several applications.140,141 Typically, alginate extracted from brown algae is treated with various chemicals at different synthesis stages. Briefly, the production process of sodium alginate begins with the harvesting and drying of seaweed, after which it undergoes mechanical processing to be converted into algal powder.142 This powder will be treated with hydrochloric acid (HCl) to extract the alginic acid, which serves as the precursor for sodium alginate including sodium carbonate (Na2CO3) as part of the extraction process. The extracted alginic acid will be washed, filtered, and treated with sodium hydroxide (NaOH) to form a sodium alginate (SA) solution.143 The solution is then further treated with HCl to enhance the purity and produce an alginic acid gel. The samples will then be neutralised with alkali agents such as sodium hydroxide and/or sodium carbonate, converting them into sodium alginate, a water-soluble polymer widely used across various industries.144 The purification of the extracted alginate was conducted through a chemically assisted process before filtration and drying. Specifically, the crude alginate was subjected to sequential treatments using calcium chloride (CaCl2), sodium chloride (NaCl), or further treated with ethanol to remove residual impurities, enhance polymer purity, and improve the physicochemical characteristics of the final biopolymer. The extraction process of alginate is illustrated in Fig. 6a, showing the key steps involved in alginate preparation from raw seaweed sources and its subsequent transformation into sodium alginate and its applications in the adsorption process, retrieved from the literature. In contrast, other literature shown in Fig. 6b explains the Industry process of sodium alginate extraction via calcium precipitation.
 | ||
| Fig. 6 (a) Extraction and preparation of alginate from the raw sources and their application toward heavy metal removal, (adapted with permission, Licensed under Elsevier's terms) and (b) typical industrial process of sodium alginate extraction via calcium precipitation (Creative Commons Attribution (CC BY 4.0) from MDPI).21,142 | ||
Alginate has been invaluable because it is helpful in water purification applications due to the presence of hydroxyl (–OH) and carboxyl (–COO−) functional groups present in its polymer backbone.145 SA can effectively interact with heavy metal ions and other pollutants in aqueous environments. SA can also undergo an adsorption mechanism that allows contaminants to bind to the polymer surface, facilitating the removal of impurities and contributing to environmental remediation efforts.146 This property has positioned alginate as a promising material in sustainable water treatment technologies.
Structurally, alginates are linear block copolymer polysaccharides composed of two fundamental monomeric units: β-D-mannuronic acid (M-block) and α-L-guluronic acid (G-block), the latter being the C-5 epimer of the former.147 C-5 epimer of the former.147 These monomers are linked through β-(1–4) glycosidic bonds, forming an unbranched, water-soluble polymer chain. Additionally, alginate polymers can exhibit various sequential forms or arrangements of these monomeric units, including homopolymer M- or G-blocks, alternating MG-blocks, and more complex configurations such as GM-blocks and interspersed MG/GM sequences of varying lengths, with different interchangeable possibilities as shown in Fig. 7a–c, allowing for structural versatility and structurally modified model describing the interactions between alginate G-blocks and divalent cations, primarily Ca2+ illustrates their strong affinity for metal ions and other pollutants through ionic-displacement mechanisms. These interactions facilitate efficient regeneration via simple filtration and contribute to the formation of stable ionic gels, thereby making alginate-based systems excellent candidates for water-pollution remediation.147 A distinctive property of alginates is their ability to undergo reversible sol–gel transitions upon interaction with divalent and trivalent metal ions. Calcium chloride (CaCl2) is commonly used to induce gelation, particularly through interactions with the GG-block regions, facilitating the formation of a rigid, three-dimensional network often described using the “egg-box” model.148 This structural transformation occurs as calcium ions (Ca2+) form ionic cross-links between the G-block residues, forming a hydrogel. The schematic representation of this process, as depicted in Fig. 1c, is adapted from work.149 illustrates how calcium ions mediate the cross-linking of alginate chains, resulting in a stable gel network. The binding capacity with divalent metal cations reported is Pb2+ > Cu2+ > Cd2+ > Ba2+ > Sr2+ > Ca2+ > Co2+, Ni2+, Zn2+ > Mn2+.150 Beyond their gelation properties, alginates are extensively studied for their capacity to adsorb heavy metal ions from aqueous environments. The presence of abundant hydroxyl and carboxyl functional groups in the polymer backbone enables strong interactions with metal ions, making alginates a promising material for water purification and environmental remediation applications.38,151
 | ||
| Fig. 7 Schematic of alginic acid structure: structure of alginate showing (a) chain conformation (b) block distribution, and (adapted with permission, Licensed under Elsevier's terms) (c) a structurally modified model describing the interactions between alginate G-blocks and divalent cations (Ca2+) (Creative Commons Attribution (CC BY 4.0) from MDPI).147,149 | ||
 | ||
| Fig. 8 Examples of (a) fabrication of alginate-based composites, (b) adsorption mechanisms of alginate composites (adapted with permission, Licensed under Elsevier's terms), and (c) preparation of PANI@SA-SNM gel adsorbent using calcium alginate to encapsulate nZVI-rice straw composite (CANRC) for Pb2+, Zn2+, and Cd2+ removal (adapted with permission, Licensed under Elsevier's terms).41,153,159 | ||
4.2.2.1 Polymeric blends and graft copolymers. The formation of alginate-based polymeric blends and graft copolymers has been extensively employed to enhance adsorption selectivity and reusability. Studies have shown that surface grafting with functional groups such as thiol (–SH) and amine (–NH2) enhances the selective affinity for metal ions. Thiolates alginates exhibit strong binding to metal ions, while aminated variants demonstrate high adsorption of Cd2+.39,152 Cross-linking alginate, particularly with calcium ions, yields mechanically robust hydrogel beads that resist dissolution in aqueous media and maintain stable adsorption capacities across multiple use cycles.152,154 For instance, hydrogels are often based on ionic or covalent crosslinking without specific fillers or advanced frameworks. Calcium-cross-linked sodium alginate beads have demonstrated capacities ranging from 54.9 to 82.8 and 135.5 mg g−1 for Cu2+, Ag+, Fe2+, and Fe3+, respectively.154 The polyaniline–sodium alginate-MXene nanomaterial composite (PANI@SA-SNM) integrates MXene nanosheets and polyaniline within a sodium alginate hydrogel matrix, significantly enhancing the adsorption of Cu2+ and Hg2+ ions when used for their removal from aqueous solution. The interaction between polyaniline's redox-active nitrogen sites, MXene's layered surface functionalities, and alginate's carboxyl groups facilitates high metal uptake (up to 352.76 mg g−1), confirming the efficacy of multifunctional polymeric blends in adsorptive remediation.41 Modified alginate-based biocomposite hydrogel microsphere, effectively adsorbing Pb2+and Cu2 ions, has 369.6 mg g−1 and 124.1 mg g−1, and some studies also reported the potential cellulose–alginate sponges that exhibit high water permeability and excellent reusability properties. Studies have further demonstrated the effectiveness of alginate-based hybrid materials. Notably, mesoporous alginate/β-cyclodextrin beads exhibit remarkable adsorption capacities for Pb2+, Cu2+, Cd2+, and Ni2+ 21.09, 15.54, 2.47, and 2.68 mg g−1, respectively, highlighting the enhanced performance of alginate–polymer composites for heavy-metal removal.46,142 Moreover, sodium alginate-based carboxymethyl cellulose (CMC) hydrogel beads Pb2+ uptake (>600 mg g−1), demonstrating the benefits of combining carboxyl-rich alginate matrices with amine-rich copolymers. Similarly, the sodium alginate-g-poly(acrylic acid-co-acrylamide) nanocomposite hydrogel absorbed Pb2+, Cd2+, Ni2+, and Cu2+ at concentrations of 231.88, 235.62, 67.52, and 76.35 mg g−1, respectively.
4.2.2.2 Inorganic fillers and nanomaterials. Incorporating inorganic fillers, such as metal oxides and salts, into alginate matrices enhances ion exchange capabilities and structural rigidity while increasing the surface area. These additives interact physically or chemically with alginate to form functional hybrid structures. For instance, alginate-caged magnesium sulphate nanoparticle microbeads demonstrated an adsorption capacity of 84.7 mg g−1 for Pb2+.142 The inclusion of magnesium sulfate (MgSO4) likely provides ionic sites for selective lead interaction while boosting the mechanical robustness of the hydrogel structure. The carbonised composite manganese-crosslinked sodium alginate showed excellent removal of As3+ (189.29 mg g−1), As5+ (193.29 mg g−1), and Cr6+ (104.5 mg g−1).143 Manganese enhances redox activity, reducing toxic ions and subsequent immobilisation. This composite benefits from electrostatic and surface complexation mechanisms enabled by the manganese–carbon interface. The calcium alginate-nanoscale zero-valent iron (nZVI)-biochar composite reportedly adsorbs Pb2+, Zn2+, and Cd2+ with capacities of 47.99, 71.77, and 47.27 mg g−1, respectively,145 combining the adsorptive nature of biochar with the magnetic and reductive properties of nanoscale zero-valent iron (nZVI). The cross-linked alginate-rice husk ash-graphene oxide-chitosan nanoparticles (CL-ARCG-CNP) composite combines alginate with silica-rich rice husk ash, reduced graphene oxide, and chitosan nanoparticles, forming a cross-linked hybrid with a high Pb2+ adsorption capacity of 242.5 mg g−1. This multifunctional system leverages the high surface area of GO, the amine-rich functionality of chitosan, and the reactive silanol groups from rice husk ash, collectively enhancing Pb2+ chelation and stability in aqueous environments.40 The calcium carbonate on alginate/chitosan biocomposite (CSAX_Ca) was also reported to have an affinity for the pollutants Cu2+and Pb2+ at the adsorption capacities 429, 1742 mg g−1. This performance is attributed to the ionic exchange properties of CaCO3, combined with the carboxyl groups of alginates and the amine groups of chitosan, respectively.155 Such materials serve dual functions: adsorbing metal ions and reducing them to less toxic or immobilised forms while being easily recoverable due to their magnetic properties. These composites demonstrate the effectiveness of hybrid materials that combine inorganic fillers with alginate to produce multifunctional adsorbents. Their efficacy is further enhanced by the synergistic role of metal oxides in charge exchange, redox transformations, and maintaining structural integrity.156,157
4.2.2.3 Magnetic nanocomposites. Magnetic nanocomposites offer the dual benefits of effective heavy metal removal and straightforward post-treatment separation utilising external magnetic fields. These materials are essential in scalable water treatment technologies.158 The calcium alginate-nZVI-biochar composite for removing Pb, Zn, and Cd from water: insights into governing mechanisms and performance. This category is exemplified by calcium alginate-nZVI-biochar, as nZVI provides magnetic properties and facilitates the reductive precipitation of metal ions. The removal capacities for Pb2+, Zn2+, and Cd2 with absorption capacities of 47.99, 71.77, and 47.27.159 Demonstrate the synergistic role of nZVI with alginate's ion exchange capability. While no other strictly magnetic composites are explicitly mentioned in the dataset, this entry emphasises a growing research interest in merging magnetic responsiveness with adsorption functionalities. The advancement of magnetically recoverable alginate-based adsorbents represents a practical approach for real-time and reusable water purification applications.
4.2.2.4 Metal–organic frameworks (MOFS) and graphene-based composites (GBC). Advanced nanostructures such as reduced graphene oxide (rGO), thiacalixarene derivatives, and metal–organic frameworks (MOFs) significantly enhance alginate performance due to their high surface areas, π–π interactions, and diverse coordination environments.160–163 The alginate/reduced graphene double-network hydrogel beads and their single-network counterparts exhibited 169.5 and 72.5 mg g−1 capacities for Cu2+ and Cr2O72, respectively.63,163 The double-network structure offers improved mechanical stability and a higher density of adsorption sites. Meanwhile, rGO sheets promote π-electron-rich regions, facilitating cation-π interactions and electrostatic attractions. The tetrasodium thiacalixarene tetra sulfonate–sodium alginate nanocomposite hydrogel achieved broad-spectrum metal ion adsorption: Pb2+ (99.8 mg g−1), Ni2+ (67.4 mg g−1), Cu2+ (90.56 mg g−1), Cd2+ (94.5 mg g−1), Co2+ (74.9 mg g−1), and Cr3+ (79.2 mg g−1).144 As macrocyclic ligands, thiacalixarene derivatives provide tailored cavities that selectively complex metal ions. Their integration into alginate matrices substantially enhances binding specificity and capacity through host–guest chemistry. These advanced composites demonstrate the potential of incorporating MOFs, graphene derivatives, and supramolecular chemistries into alginate-based platforms to create highly selective and high-capacity adsorbents. Their tunable architectures and multifunctional binding sites facilitate the simultaneous removal of various metal ions from aqueous environments.
4.2.2.5 Bio-based/biowaste-derived alginate composites. Bio-based and biowaste materials are gaining popularity, as studies have shown that alginate composites derived from orange and nectarine peels (OAF and NAF) exhibit high adsorption capacities for Cr5+ ions. These agro-waste materials provide additional hydroxyl and phenolic functionalities that enhance hexavalent chromium's chelation and electrostatic attraction. Table 2 shows the adsorption capacities of alginate-based composites for heavy metal removal. Integrating alginate improves structural integrity and water dispersibility, demonstrating a green valorisation strategy for effective Cr5+ removal, with adsorption capacities of 224.3 and 256.5 mg g−1.64 A multi-metal adsorption study using M. oleifera extract encapsulated in sodium alginate matrices reported modest adsorption capacities for Co2+, Ni2+, Cu2+, Zn2+, manganese ion (Mn2+) and sometimes uranyl ion (UO22+). Although the uptake values (1.02–5.8 mg g−1) are relatively low, the system uses plant-derived bioactives to introduce additional binding functionalities into alginate networks. The biosorption mechanism is likely driven by phytochemical interactions combined with the carboxyl groups of alginates. Pollutants: Co2+, Ni2+, Cu2+, Zn2+, Mn2; adsorption capacities: 5.8, 4.78, 4.6, 1.3, 1.02 mg g−1.48 In another study, Sodium alginate was functionalised with M. oleifera seed powder and fabricated via wet spinning to explore its use for removing heavy metals, particularly Cu2+, Cd2+, and Ni2+, as investigated by Orisawayi et al.27 Although the adsorption capacity was not reported, future work was discussed to investigate this further. The study primarily aimed to investigate the natural bioactive compounds in M. oleifera that enhance metal binding. At the same time, the alginate matrix provides ionic carboxyl for additional sorption. This combination illustrates a sustainable approach for producing biodegradable, fibre-based adsorbents with a selective affinity for the metal. The electrospinning process fabricates a hybrid of pulverised M. oleifera seed powder embedded within a sodium alginate matrix, with polyethene oxide (PEO) as a co-spinning agent.37 The process was successful, as investigated, aiming to explore the feasibility of producing fibrous biosorbents that harness the natural adsorptive capacity of M. oleifera, the ion-exchange potential of alginate, and the fibre-forming capability of PEO. While the complete adsorption properties of these composites have not yet been evaluated, the conceptual integration of these materials through electrospinning could serve as a baseline for a potential method for generating nanostructured materials with improved surface area, porosity, and enhanced alginate mechanical properties, thereby facilitating improved interaction with heavy metal ions in aqueous solutions.
| S. no. | Alginate-based adsorbents | Pollution/target heavy metal ion(s) | Adsorption capacity (mg g−1) | References |
|---|---|---|---|---|
| 1 | Modified alginate-based biocomposite hydrogel microsphere | Pb2+ and Cu2+ | 369.6 (Pb2+) and 124.1 (Cu2+) | 164 |
| 2 | Mesoporous alginate/β-cyclodextrin polymeric beads | Pb2+, Cu2+ and Cd2+, Ni2+ | 21.09 (Pb2+), 15.54 (Cu2+), 2.47 (Cd2+) and 2.68 (Ni2+) | 46 |
| 3 | Alginate-caged magnesium sulfate nanoparticle microbeads | Pb2+ | 84.7 for Pb2+ | 165 |
| 4 | Carbonised composite manganese crosslinked sodium alginate | As3+, As5+ and Cr6+ | 189.29 (As3+), 193.29 (As5+) and 104.50 (Cr6+) | 166 |
| 5 | Amino-functionalised sodium alginate aerogel | Cr6+ and Cd2+ | 678.67 (Cr6+) and 464.23 (Cd2+) | 167 |
| 6 | Calcium alginate-nZVI-biochar | Pb2+, Zn2+ and Cd2+ | 47.99 (Pb2+), 71.77 (Zn2+) and 47.27 (Cd2+) | 159 |
| 7 | Sodium alginate-based carboxymethyl cellulose hydrogel beads | Pb2+ | — | 168 |
| 8 | Sodium alginate-g-poly(acrylic acid-co-acrylamide) nanocomposite hydrogel | Pb2+, Cd2+, Ni2+, Cu2+ | 231.88 (Pb2+), 235.62 (Cd2+), 67.52 (Ni2+) and 76.35 (Cu2+) | 169 |
| 9 | Alginate/reduced graphene double-network and single-network hydrogel beads | Cu2+, Cr2O72− | 169.5 (Cu2+) and 72.5 (Cr2O72−) | 154 |
| 10 | Tetrasodium thiacalixarenetetrasulfonate–sodium alginate nanocomposite hydrogel | Pb2+, Ni2+, Cu2+, Cd2+, Co2+ and Cr3+ | 99.8 (Pb2+), 67.4 (Ni2+), 90.56 (Cu2+), 94.5, 74.9 (Co2+) and 79.2 (Cr3+) | 170 |
| 11 | Sodium alginate hydrogel beads by post-crosslinking | Cu2+, Ag+ and Fe3+ | 54.9 (Cu2+), 82.8 (Ag+) and 135.5 (Fe3+) | 154 |
| 12 | Sodium alginate-functionalised M. oleifera seed wet-spinning | Cu2+, Cd2+ and Ni | — | 171 |
| 13 | MXene/polyaniline/sodium alginate (PANI@SA-SNM) gel | Cu2+, Hg2+ | 255.81 (Cu2+) and 352.76 (Hg2+) | 152 |
| 14 | Orange peels/alginate (OAF) nectarine peels/alginate (NAF) | Cr5+ | About 224.3 (Cr5+) – OAF and 256.5 (Cr5+) – NAF | 172 |
| 15 | Cross-linked alginate-rice husk ash-graphene oxide-chitosan nanoparticles (CL-ARCG-CNP) | Pb2+ | 242.5 (Pb2+) | 40 |
| 16 | Calcium carbonate on alginate/chitosan biocomposite (CSAX_Ca) | Cu2+ and Pb2+ | 429 (Cu2+) and 1742 (Pb2+) | 155 |
| 17 | Alginate + encapsulated M. oleifera | Co2+, Ni2+, Cu2+, Zn2+, and Mn2+ | 5.8 (Co2+), 4.78 (Ni2+), 4.6 (Cu2+), 1.3 (Zn2+), and 1.02 (Mn2+) | 48 |
In addition to alginate, cellulose, another abundant, renewable, and functional biopolymer, has demonstrated considerable promise in heavy metal ion adsorption, as detailed in other sections.
4.3 Cellulose-based adsorbents
Cellulose is the most abundant natural biopolymer on Earth, consisting of a long-chain polysaccharide composed of β-D-glucose units, which are often covalently linked by acetal functionalities between the equatorial (OH) groups on the carbon atoms, known as (C4) and (C1), via β-1,4-glycosidic bonds.173,174 Its unique molecular structure contributes to its exceptional physicochemical stability, particularly its insolubility in water, which arises from the extensive hydrogen bonding and crystallinity imparted by its glycosidic linkages.175–177 The long polymer chains are organised into two distinct regions: highly ordered crystalline domains confer mechanical strength and stability, and amorphous regions enhance chemical reactivity and biological interactions.178–180 Cellulose is predominantly obtained from plant cell walls, although microbial sources produce bacterial cellulose with unique nanostructures.178,179,181 Increasingly, agricultural residues are being explored as low-cost, renewable sources of cellulose for developing sustainable materials. Due to its intrinsic properties, renewability, biodegradability, chemical stability, non-toxicity, and the abundance of reactive hydroxyl groups, cellulose is an excellent platform for fabricating advanced functional materials. Among various cellulose-based materials, cellulose hydrogels and their regenerated counterparts have emerged as a prominent class of water purification media.45,176,182| S. no. | Cellulose-based composite | Pollution/target heavy metal ion(s) | Adsorption capacity (mg g−1) | References |
|---|---|---|---|---|
| 1 | Carboxymethyl cellulose/gelatin composite hydrogel | Cd2+, Hg2+ and Pb2+ | 147.7 (Cd2+), 88.62 (Hg2+) and 163.89 (Pb2+) | 198 |
| 2 | CuMOF on sodium alginate/chitosan/cellulose nanofibril composite hydrogel | Pb2+ | 531.38 for Pb2+ | 187 |
| 3 | Sodium alginate/cellulose nanofibre composite hydrogel | Pb2+ | 544.66 for Pb2+ | 188 |
| 4 | Porous kappa-carrageenan/cellulose hydrogels | Pb2+ | 486 ± 28.5 for Pb2+ | 199 |
| 5 | Cellulose hydrogels (G50) | UO22+ | 572.3 for UO22+ | 193 |
| 6 | Carboxymethyl cellulose/chitosan/alginic acid hydrogels | Cr6+, Ni2+ and Cu2+ | >750 for (Cr6+, Ni2+ and Cu2+) | 133 |
| 7 | Chitosan/cellulose phosphonate composite hydrogel | Pb2+ and Cu2+ | 211.42 (Pb2+) and 74.29 (Cu2+) | 51 |
| 8 | Cellulose (37%)–chitosan (63%) | Cu2+ | 94.3 for Cu2+ | 52 |
| 9 | Cellulose/chitosan/PVA/nano-Fe2O3 | Cu2+ | 15.95 for Cu2+ | 200 |
| 10 | Oxidised carboxymethyl cellulose hydrogel | Ag+, Pb2+, Cu2+ | 407 (Ag+), 1250 (Pb2+) and 1111 (Cu2+) | 201 |
| 11 | Wheat straw cellulose-g-poly(acrylic acid)/poly(vinyl alcohol) | Cu2+ | 142.7 for Cu2+ | 201 |
| 12 | Carboxymethyl cellulose-based cryogels | Pb2+, Ni2+, Co2+ | 550 (Pb2+), 620 (Ni2+) and 760 (Co2+) | 202 |
| 13 | Cellulose grafted with acrylonitrile (CelEnEs) | Cr5+ | — | 203 |
| 14 | Collagen/cellulose hydrogel beads (M-CS/PVA/CCNFs) | Cu2+ | 67.36 mg g−1 for (Cu2+) | 204 |
| 15 | Mercerized cellulose | Cu2+, Cd2+ and Pb2+ | 30.4 (Cu2+), 86.0 (Cd2+) and 205.9 (Pb2+) | 205 |
| 16 | Cellulose/ZrO2 | Ni2+ | 79.0 for Ni2+ | 206 |
| 17 | Cellulosic graft polymerisation of glycidyl methacrylate-co-methacrylic | Co2+ | 11.5 and 11 for Co2+ | 207 |
| 18 | Poly(ethylene imine)-modified cellulose | Cu2+ | 102 | 208 |
| 19 | Welan gum-modified cellulose | Cd2+, Pb2+ and Cu2+ | 83.6 (Cd2+), 77.0 (Pb2+) and 67.4 (Cu2+) | 43 |
| 20 | Cellulose acetate (CA)/silica composite | Cr5+ | 19.46 for (Cr5+) | 45 |
| 21 | Oxidised cellulose-based materials | Hg2+ | 258.75 for (Hg2+) | 182 |
| 22 | Cellulose-based composite hydrogel microsphere | Co2+ and Ni2+ | 358 (Co2+) and 373 (Ni2+) | 180 |
| 23 | Cellulose-based hydrogel-modified kaolin | Pb2+ and Cu2+ | 879.84 (Pb2+) and 543.50 (Cu2+) | 209 |
| 24 | Carboxymethyl cellulose hydrogel-pectin-based | Cd2+, Pb2+ and Cu2+ | 84.4 (Cd2+), 159.4 (Pb2+), and 125.6 (Cu2+) | 210 |
Recently, advancements in functional materials science have positioned cellulose, particularly in its nanoform known as nanocellulose.114,211 These materials are emerging sustainable biopolymers for various water treatment applications. Fig. 9 illustrates the functionalisation of cellulose through chemical modification.114 The abundant hydroxyl groups enable the introduction of various reactive moieties, such as carboxyl, amine, thiol, and sulfonate groups, as reported.212,213 This has been studied to significantly enhance the material's affinity for heavy metal ions in aqueous solutions, with the functional group transformations altering the surface charge, coordination capacity, and hydrophilicity.91,194 These nanocellulose-based systems exhibit a high surface area, increased porosity, enhanced mechanical strength, and aqueous stability, all of which are desirable characteristics for adsorbents specifically targeting the removal of divalent heavy metal contaminants such as Pb2+, Cd2+, Cr6+, and Cu2+ from wastewater.114,214 Furthermore, nanocellulose's high aspect ratio and tunable functional surfaces facilitate efficient diffusion, rapid ion exchange, and chelation processes, improving adsorption kinetics and capacity.214,215 Consequently, modified cellulose and its nanostructured derivatives serve as renewable, biodegradable, and highly effective materials for the adsorption and removal of toxic metal ions in water purification systems.216,217
 | ||
| Fig. 9 Emerging nanocellulose-based modifications of cellulose for enhanced removal of heavy metal ions from water (modified with permission, Licensed under ACS Publication's terms).114 | ||
While alginate, cellulose derivatives, and their composites have shown considerable promise as eco-friendly adsorbents in water purification, their performance can be significantly enhanced through hybridisation with plant-derived materials that offer active biosorption properties. One such material, M. oleifera seed powder, has garnered attention for its rich bioactive compounds and ability to adsorb heavy metal ions effectively. The following section explores the potential of M. oleifera as a natural biosorbent in sustainable water treatment. Beyond structural biopolymers like alginate and cellulose, plant-based biosorbents such as M. oleifera offer complementary adsorption mechanisms and bioactive functionalities, enriching the development of multifunctional composite systems for water purification.
5 M. oleifera-based adsorbents
The M. oleifera tree thrives in tropical and subtropical regions worldwide. It is often called the “miracle tree” or “drumstick” in English. Nowadays, M. oleifera has naturalised throughout the tropics, including regions in Africa, Central and South America, and Southeast Asia. M. oleifera has been introduced and cultivated across Europe for research purposes, enhancing its accessibility.218–2205.1 Biosorption mechanisms and functional components of M. oleifera
Research has shown that M. oleifera seeds are primarily protein-rich and exhibit active functions known for binding with pollutants. The tree is also reported to have been a preferred source of nutrition and second-generation biodiesel, and its components can be used as drugs. They have reportedly demonstrated an affinity for absorbing carbon dioxide from the atmosphere.219,221Fig. 10 displays the various M. oleifera biomass samples used in this study, including M. oleifera unpeeled seeds (MOU), M. oleifera shelled seeds (MOS), M. oleifera seed powder (MoP), M. oleifera husk (MOH), M. oleifera husk powder (MOHP), M. oleifera dried leaves (MODL), M. oleifera dried Leaves powder (MODLP), M. oleifera bark pieces (MOB), M. oleifera and bark powder (MOBP). These components represent the diverse functional fractions of M. oleifera investigated for coagulant and adsorbent applications in water purification. Studies suggest that each part contains a protein that can be used as an antimicrobial flocculant to remove wastewater impurities through electrostatic interactions between the cationic protein and colloids.222 Some studies have also shown that M. oleifera, known for its high content of bioactive compounds, shows promise in various water treatment applications due to its availability, biodegradability, and non-toxicity. Therefore, the coagulating properties make them a potential additive for alginate in water purification applications, presenting a promising alternative to alginate, as it has been previously used in the manufacture and functionalisation of alginate.223 However, only a few studies have explored the combination of M. oleifera with most biopolymers, such as alginate and cellulose. In the case of heavy metal ions, M. oleifera has been reported to remove heavy metals such as copper, cadmium, chromium, and lead at rates of 95%, 76%, 70%, and 93%, respectively.224,225 In a study on using M. oleifera seed for water treatment, the final concentration of copper was below the desirable limit for drinking water (less than 1 mg L−1).226,227 However, the removal of cadmium, chromium, and lead after coagulation with M. oleifera seed cake coagulant did not meet the limits of drinking water standards. | ||
| Fig. 10 Photographs and modified images of the (a) unpeeled seeds (MOU), (b) shelled seeds (MOS), (c) seed powder (MoP), (d) husk (MOH), (e) husk powder (MOHP), (f) dried leaves (MODL), (g) leaves powder (MODLP), (h) bark pieces (MP),(i) back powder (BPD).236 | ||
This inconsistency is closely related to the underlying mechanisms governing its removal efficiency was also observed in studies carried out by Orisawayi et al.,25 and study on the purification of river water using M. oleifera seed for copper removal for point-of-use household application discussed that the cationic proteins and bioactive compounds present in M. oleifera could function primarily as a natural coagulant.228
Several recent studies also discussed that the mechanisms are highly effective for metal ions such as Cu2+, which exhibit favourable interactions with the functional groups in the extract.229–231 However, ions such as Cd2+, Pb2+, and Cr3+ and Cr5+ possess lower charge densities, weaker binding affinities, or distinct hydrolysis behaviours, which could result in less efficient coagulation and adsorption. This possibly suggests reason M. oleifera is an excellent coagulant; its capacity as a high-affinity adsorbent is limited for specific metal species, and therefore, its performance may require enhancement through composite formulation or integration with other biopolymers.232,233 Therefore, additional treatments may be required to meet the standards of the EPA, WHO, EU, and some indigenous bureau standards, such as those of the indigenous peoples. The study's findings indicate that M. oleifera seed cake is suitable as a coagulant and is effective for pre-treatment applications for removing heavy metals from water systems.225
Fig. 11 presents an example of MOS biosorption comparison before and after 24 hours of brilliant green (BG) and biosorption of crystal violet (CV) of typical M. oleifera seed obtained from literature as when used, it was reported that adsorb heavy metal ions, these functions provide selective and effective absorption for various metal ions which belong to Class B, including Hg2+, Ag3+, Pd2+, Pt5+, Pt3+, Au3+, and Cs+. For instance, Benettayeb et al. observed an enhancement in sorption for the ions Pb2+, Cd2+, and Cu2+.226 Nwagbara et al.225 also demonstrated that adsorbents with amine groups possess unique properties, enabling them to adsorb compounds with cationic or anionic charges at different pH values, which are present in the M. oleifera seed and capable of removing these heavy metals from an aqueous solution.234 By using composite coagulants, drinking water standards can be met, and in many cases, heavy metals are not detected in the treated water. Polymers possess numerous functional groups, including carboxylic, amine, hydroxyl, and sulfonic. They can be used as complexing agents for the adsorptive removal of metal ions from aqueous solutions.40,235
 | ||
| Fig. 11 Illustration of the nature of M. oleifera seeds after 24 hours of the sorption process for heavy metal ions from an aqueous solution: (a) Brilliant Green (BG) and (b) Crystal Violet (CV) sorption.237 | ||
 | ||
| Fig. 12 A typical illustration step of processing M. oleifera parts for water treatment application. | ||
The initial processing step involves mechanical disintegration using grinders, blenders, or a traditional mortar and pestle. This process reduces particle size, increases surface area, and facilitates further downstream applications. A sieving stage follows to ensure particle homogeneity for consistent application. The protein-rich shelled seeds and gum exudates undergo aqueous or solvent-based extraction. The cationic proteins from the kernel interact with negatively charged colloids in water, promoting coagulation and flocculation. M. oleifera gum, due to its polysaccharide backbone and high molecular weight, enhances coagulation through bridging mechanisms and aids in viscosity control during composite synthesis.243,244 This process is particularly relevant in systems where organic turbidity or microbial contamination is a concern. The fibrous seed husks, bark, and other lignocellulosic fractions are subjected to pyrolysis or chemical activation to produce biochar or activated carbon. These materials exhibit a high surface area and porosity, essential for effective adsorption of heavy metal ions.245,246
Surface functional groups such as hydroxyl, carboxyl, and phenolic moieties facilitate metal binding through ion exchange, surface complexation, and electrostatic attraction. The performance of bio-based composites derived from M. oleifera, alginate, and cellulose is subsequently enhanced through systematic material modification techniques to improve structural integrity, processability, and adsorption efficiency in water purification systems.246–248 These modifications typically begin by mixing the primary biopolymers with binders or cross-linking agents, such as poly(vinyl alcohol) (PVA), starch, or modified cellulose derivatives, and the process seeks to strengthen the network structure, enhance the dispersion of M. oleifera components, and improve compatibility within the matrix materials.242,249,250
The modified blends can be fabricated into functional forms, such as beads, films, fibres, or pellets, each offering distinct surface area and porosity advantages for water treatment.249 Depending on the desired morphology and end-use application, various fabrication techniques, including casting, extrusion, wet spinning, electrospinning, and freeze-drying, are utilised.37,242,251
5.2 Heavy metal biosorption mechanism of M. oleifera in aqueous systems
Fig. 13. Schematic illustration of the various mechanisms involved in the biosorption of M. oleifera for removing toxic contaminants, such as Cr5+, V5+, and Pb2+; this was explained by Benettayeb et al.236 In a critical review of the emphasis, recent pieces of evidence study M. oleifera as a biosorbent for water and wastewater treatment. The primary biosorption mechanisms by which M. oleifera interacts with toxic heavy metal ions. The ion-exchange mechanism is central, whereby native ions (e.g., Na+, H+, Ca2+) present on the biosorbent surface are replaced by heavy metal ions (M2+), such as Pb2+, Cr5+, and V5+. The functional groups that facilitate this process reported that are peculiar to M. oleifera include hydroxyl (–OH), carboxyl (–COOH), carbonyl (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), and amine (–NH2) that are present in M. oleifera.151,252,253 The adsorption mechanisms encompass electrostatic attraction between negatively charged functional groups and metal cations, surface complexation, chemisorption, and intraparticle diffusion within the porous matrix. The overall biosorption performance is further influenced by the solution pH, the surface charge of the adsorbent, and the specific interaction modes governing metal–ligand binding. These interactive mechanisms collectively highlight M. oleifera's efficiency as a multifunctional biosorbent for remediating metal-contaminated water.254,255Table 4 also presents the biosorbents for heavy metal biosorption of various toxic heavy metal pollutants (main M. oleifera parts used for heavy metal adsorption).
O), and amine (–NH2) that are present in M. oleifera.151,252,253 The adsorption mechanisms encompass electrostatic attraction between negatively charged functional groups and metal cations, surface complexation, chemisorption, and intraparticle diffusion within the porous matrix. The overall biosorption performance is further influenced by the solution pH, the surface charge of the adsorbent, and the specific interaction modes governing metal–ligand binding. These interactive mechanisms collectively highlight M. oleifera's efficiency as a multifunctional biosorbent for remediating metal-contaminated water.254,255Table 4 also presents the biosorbents for heavy metal biosorption of various toxic heavy metal pollutants (main M. oleifera parts used for heavy metal adsorption).
 | ||
| Fig. 13 Schematic illustration of the various mechanisms involved in M. oleifera biosorption for removing toxic contaminants from aqueous solutions, such as Cr5+, V5+, and Pb2+. Probable mechanism ion-exchange mechanism between M. oleifera and metal ions (M2+) (Open access).237 | ||
| S. no. | Biosorbent part of M. oleifera with or without modification | Pollution/target heavy metal ion(s) | Adsorption capacity (mg g−1) | References | |
|---|---|---|---|---|---|
| 1 | Pure seed/leaves | Pb2+, Cd2+, Co2+ and Ni2+ | Seed 13.29 (Pb2+), 4.97 (Cd2+), 5.80 (Co2+) and 3.61(Ni2+) | Leaves 49.50 (Pb2+), 16.13(Cd2+), 10.94 (Co2+) and 10.16 (Ni2+) | 30 |
| 3 | Pods modified HNO3 0.3 M | Pb2+ and Cd2+ | 35.97 (Pb2+) and 18.24 (Cd2+) | 256 | |
| 4 | Pure seed | Pb2+ | For 5.6 (Pb2+) | 257 | |
| 5 | Leaves modified diethylamine functionalisation | Cr5+ | 60.6061 for (Cr5+) | 258 | |
| 6 | Pure leaves | Pb2+ | 45.83 for (Pb2+) | 222 | |
| 7 | Pure seed | Cd2+ | 357.14 for (Cd2+) | 259 | |
| 9 | Gum-modified acryloylation reaction | Hg2+ | 840.34 for (Hg2+) | 260 | |
| 10 | Pure seed pods | Cr5+ | 119.02 for (Cr5+) | 261 | |
| 11 | Pure seed and pure husk | Cu2+ and Cd2+ | 13.089 (Cu2+) and 13.123 (Cd2+) | 262 | |
| 12 | Seed modified with oil extraction to obtain M. oleifera cake (byproduct) | Pb2+ | 12.24 (Pb2+) | 263 | |
| 13 | Leaves modified with activated carbon | As5+ | 6.23 (As5+) | 264 | |
| 14 | Pure seed-modified oil extraction to obtain M. oleifera cake (byproduct) | Cd2+ | 7.864 (Cd2+) | 265 | |
| 15 | Seed oil extraction to obtain M. oleifera cake (byproduct) | Cr3+ | 3.191 (Cr3+) | 266 | |
| 16 | Leaves esterification with NaOH followed by citric acid treatment | Cd2+, Cu2+ and Ni2+ | 171.37 (Cd2+), 167.90 (Cu2+) and 163.88 (Ni2+) | 267 | |
| 17 | M. oleifera bark (MOB) | Cd2+ and Cu2+ on to MOB | 39.41 (Cd2+) and 36.59 (Cu2+) | 236 | |
| 18 | M. oleifera bark (MOB) | Ni2+ | 30.38 for (Ni2+) | 268 | |
| 19 | Wood | Cu2+, Ni 2+ and Zn2+ | 11.53 (Cu2+), 19.08 (Ni2+) and 17.67(Zn2+) | 269 | |
| 20 | Leaves citric acid treatment | Pb2+ | 209.54 for (Pb2+) | 269 | |
| 21 | Bark | Pb2+ | 34.6 for (Pb2+) | 270 | |
An evaluation of the biosorption capacities reported from the table reveals clear differences in performance among various M. oleifera plant parts. The gum-derived materials, particularly those modified via acryloylation, exhibit exceptionally high adsorption capacities, reaching 840.34 mg g−1 for Hg2+, indicating a high density of reactive functional groups. Modified leaves consistently show superior performance, achieving values above 150 mg g−1 for Cd2+, Cu2+, and Ni2+, especially when treated with NaOH–citric acid or activated carbon, suggesting that surface functionalisation significantly enhances metal-binding affinity. Seed-based materials, including seed cake by-products, also demonstrate promising performance, with adsorption capacities up to 357.14 mg g−1 for Cd2+, reflecting their favourable protein and lipid composition. By contrast, bark, wood, and unmodified seed or pod materials tend to exhibit lower uptake values. Based on these findings, there is a clear indication that leaves, gums, and chemically modified seed-derived materials are the most promising biosorbent components for heavy-metal remediation.
Various fabrication techniques have been employed to enhance bio-based composites' adsorption efficiency and stability, including electrospinning, wet spinning, hydrogel formation, and hybrid processing. These methods enable the formation of fibres or gels with high surface area, tunable porosity, and enhanced stability, all of which are critical for water treatment applications. While numerous studies have demonstrated the promising capabilities of M. oleifera, alginate, and cellulose, significant research gaps remain in integrating these materials effectively for real-world applications. The following section identifies these gaps and proposes future research pathways.
6 Comparative evaluation of sodium alginate, cellulose composited and the M. oleifera parts biosorbent
A systematic comparison of sodium alginate, cellulose, and M. oleifera composites is essential to establish their relative adsorption performance and identify the most efficient bio-based materials for heavy metal removal.114,271 Although each of these biopolymers exhibits distinctive structural features and functional groups that support metal ion binding, their adsorption efficiencies differ considerably depending on the degree of chemical modification, composite formulation, and the physicochemical characteristics of the target ions.185 This systematic comparison is based on the data retrieved from Tables 2–4 of this study. Fig. 14 shows the comparison of the adsorption capacities of alginate-based composites. Alginate composites show very high adsorption efficiencies, particularly when hybridised with metal oxides, nano-additives, or functional groups. Notable peak capacities include all metal ions Pb2+ at 1742 mg g−1 for CaCO3-alginate/chitosan composite,155 Cr6+ with 678.67 mg per g amino-functionalised alginate aerogel and Cd2+ with 464.23 mg g−1.272 The deduction from these findings shows an extraordinary adsorption capacity after chemical/nano-based functionalisation.In addition, Fig. 15 presents the comparison of adsorption capacities of cellulose-based composites extracted the study shows the peak values of capacities retrieved from the cellulose-based composites for metal ions with the highest adsorption are Pb2+ at 1250 mg g−1, with an oxidised CMC hydrogel with affinity with for Cu2+ at 1111 mg g−1, Co2+ with 760 mg g−1 and the CMC cryogel Ni2+ at 620 mg g−1.201,202 The findings show the strength of sustained high adsorption across multiple metal ions, broad selectivity, and stability in aqueous environments.
Furthermore, Fig. 14 also shows the comparison of the adsorption capacities of parts of M. oleifera with or without modification. The M. oleifera-based adsorbent study was limited to pure M. oleifera parts and modified treatment. Our findings show high adsorption capacities, particularly when chemically modified. Peak capacities include Hg2+ 840.34 mg g−1 for acryloylated M. oleifera gum,260 Cd2+ with 357.14 mg g−1 of the pure seed, and Pb2+ with capacities of 209.54 mg g−1 for citric-acid-modified leaves.259,269 Overall, the key findings from the comparative evaluation indicate a clear performance hierarchy among the three biopolymer systems. Cellulose-based composites show the highest overall efficiency, with several materials achieving capacities above 1000 mg g−1 for metals such as Pb2+ and Cu2+. Alginate composites display very high peak capacities, including the highest value reported (1742 mg g−1 for Pb2+), but this performance is strongly dependent on functionalisation. In contrast, M. oleifera biosorbents generally exhibit moderate adsorption, with higher capacities achieved only after chemical modification. Overall, cellulose demonstrates the most stable and versatile adsorption behaviour (Fig. 16).
 | ||
| Fig. 16 Comparison of the adsorption capacities of parts of M. oleifera with or without modifications. | ||
7 Consolidated comparative discussion of electrospinning and wet spinning
Building upon the comparative evaluation presented in the preceding section, it is essential to examine how the choice of fabrication technique further shapes the structural and functional attributes of these biopolymer-based adsorbents using the specific biopolymers and the biosorbent M. oleifera composites. The following discussion, therefore, consolidates the key features of electrospinning and wet spinning, highlighting how each method distinctly influences fibre morphology, active-site accessibility, and overall adsorption performance.Electrospinning and wet spinning have been identified as the key fabrication techniques for biopolymer-based and absorbent materials. However, consolidated information on the comparison of these techniques for these specific biopolymers on how these methods distinctly influence the final adsorbent's properties of the materials, such as general processing parameters, mechanical properties, microstructure and porosity, water interaction and adsorption properties and industrial suitability and economic perspectives. This section combines the findings of this study with relevant literature on biopolymer-based fibres for wastewater purification. The results align with previous studies on electrospinning wet-spinning alginate, cellulose and M. oleifera. The development of bio-based fibre materials for water treatment addressed in this research is very crucial in addressing the increasing contamination of both domestic and industrial wastewater, such as oil and gas, mining, chemical processing, and textile wastewater, among others, with heavy metal ions such as Cu2+, Ni2+, Pb2+, Cr6+, and Cd2+. These contaminants, common in effluents from the oil and gas, mining, chemical processing, and textile sectors, pose critical risks to human health and ecological integrity and several metals which could pose serious risks to human health, aquatic ecosystems, and environmental sustainability.273
This comparative analysis could serve as a guideline for researchers and industries currently working in water treatment, particularly those related to wastewater treatment and management.274Table 5 shows several comparisons, like the selected fabrication technique, which might be tailored to specific treatment goals, whether for heavy metal adsorption, mechanical durability, or large-scale industrial filtration.
| S. no. | Parameter | Electrospinning (alginate-based) | Wet-spinning (alginate-based) | Wet-spinning (cellulose-based) | References |
|---|---|---|---|---|---|
| (1) Processing comparison | |||||
| 1 | Solubility | Water-soluble, requires blending with polyethene oxide (PEO) for electrospinning and crosslinking for stability | Water-soluble, requires ionic crosslinking for stability | Water-insoluble retains stability in water | 36 |
| 2 | Processing method | Electrospinning via high voltage application onto a collector plate | Water-soluble, requires ionic crosslinking for stability | Wet-spinning using ionic liquid (EMIM DEP), then water coagulation | 32 and 276 |
| 3 | Crosslinking mechanism | Post-processing electrospinning crosslinking using CaCl2 | Ionic crosslinking via divalent Ca2+ (egg-box model) | Hydrogen bonding-based structural regeneration | 27 and 36 |
| 4 | Fibre morphology | Nanofibrous structure with a high surface area after spinning | Soft polymeric network fibres | Dense, well-packed fibres with strong interchain interactions | 27 and 36 |
| 5 | Spinnability | Requires precise control of viscosity and voltage | Easier to spin but prone to swelling | Challenging to spin due to high viscosity | 27, 32 and 36 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (2) Mechanical properties comparison | |||||
| 1 | Tensile strength | Not reported | With different concentrations of M. oleifera seed, but best at 1% MoP (lower) | With different concentrations of M. oleifera seed, but best at 2% MoP (higher) | 27 and 32 |
| 2 | Young's modulus | Not reported | Lower dependent on hydration state | Higher improved stiffness due to dense hydrogen bonding | 27 and 32 |
| 3 | Elongation at break | Not reported | Moderate, decreases with M. oleifera seed due to embrittlement | Higher retains flexibility at higher M. oleifera content | 27, 32 and 36 |
| 4 | Structural rigidity | Not reported | Soft and flexible, but weaker than cellulose-based fibres | Rigid and mechanically stable | 277 |
| 5 | Fracture behavior | Not reported | Soft and flexible, but weaker than cellulose-based fibres | Ductile failure can elongate before breaking | 27, 32 and 36 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (3) Microstructure and porosity | |||||
| 1 | Microstructure morphology | Highly porous nanostructure | Open pore structure, good for ion diffusion | Dense, compact structure | 27, 32 and 36 |
| 2 | Pore interconnectivity | Excellent, ideal for diffusion-based applications | Moderate, interconnected pores improve diffusion | Lower interconnectivity reduces diffusion efficiency | 27, 32 and 36 |
| Water interaction & adsorption properties | |||||
| 3 | Water interaction | Moderate hydrophilicity, tunable via crosslinking | Highly hydrophilic, swells in aqueous conditions | Water-stable, resistant to degradation | 27, 32 and 36 |
| 4 | Possible resistance to ion leaching | Less prone to ion leaching with possible Ca2+ ion compared to wet-spun alginate | Prone to Ca2+ ion leaching, impacting stability | Highly resistant to leaching | 27, 32 and 36 |
| 5 | Adsorption efficiency | Potential is higher due to nanofibre morphology, but adsorption was not conducted for these studies | Potential high, suitable for multiple metal ions | Moderate, selective for Cu2+ | 27, 32 and 36 |
| 6 | Heavy metal selectivity (based on the SEM-EDX characterisation) | Expected more selective adsorption due to surface functionalisation (recommended for future studies) | Broad-spectrum adsorption (Cu2+, Ni2+, Cd2+) | Selective adsorption, primarily Cu2+ | 27, 32 and 36 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (4) Industrial suitability and economic perspectives | |||||
| 1 | Industrial suitability | Best for high surface area applications (e.g., nanofiltration) | Best for filtration membranes requiring mechanical strength | Best for water treatment systems | 27, 32 and 36 |
| 2 | Recyclability potential | Moderate recyclability: crosslinking affects reusability | Possible limited recyclability due to ionic crosslinking | Possibility of more recyclable materials due to hydrogen bonding regeneration | 27, 32 and 36 |
| 3 | Cost-effectiveness | Higher cost due to high-voltage equipment and polymer additives | Low-cost, simple processing, widely available materials | Moderate cost, ionic liquid processing is expensive | 27, 32 and 36 |
| 4 | Sustainability factor | Sustainable but requires additional processing for stability due to the addition of PEO | Highly sustainable, from seaweed and plant-based sources | Sustainable, but depends on ionic liquid recycling | 278 and 279 |
| 5 | Processing challenges | Requires strict control (voltage, viscosity, humidity) | Crosslinking control is essential for stability | Complex ionic liquid handling limits the feasibility during the dissolution and wet-spinning process | 280 and 281 |
| 6 | Scalability for mass production | Scalable but requires advanced electrospinning setups | Scalable but requires precise crosslinking control | Scalable but ionic liquid recycling is a challenge | 32, 36 and 282 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||
| (5) Industrial suitability and economic perspectives | |||||
| 1 | Industrial suitability | Best for high surface area applications (e.g., nanofiltration) | Best for filtration membranes requiring mechanical strength | Best for water treatment systems | 27, 32, 36 and 283 |
| 2 | Recyclability potential | Moderate recyclability: crosslinking affects reusability | Possible limited recyclability due to ionic crosslinking | Possibility of more recyclable materials due to hydrogen bonding regeneration | 27, 32, 36 and 283 |
| 3 | Cost-effectiveness | Higher cost due to high-voltage equipment and polymer additives | Low-cost, simple processing, widely available materials | Moderate cost, ionic liquid processing is expensive | 27, 32, 36 and 284 |
| 4 | Sustainability factor | Sustainable but requires additional processing for stability due to the addition of PEO | Highly sustainable, from seaweed and plant-based sources | Sustainable, but depends on ionic liquid recycling | 27, 32 and 36 |
| 5 | Processing challenges | Requires strict control (voltage, viscosity, humidity) | Crosslinking control is essential for stability | Complex ionic liquid handling limits the feasibility during the dissolution and wet-spinning process | 27, 32 and 36 |
| 6 | Scalability for mass production | Scalable but requires advanced electrospinning setups | Scalable but requires precise crosslinking control | Scalable but ionic liquid recycling is a challenge | 27, 32, 36 and 280–282 |
The comparative assessment demonstrates that each fibre system offers distinct strengths relevant to water purification. Electrospun alginate fibres offer the highest porosity and surface area, making them theoretically ideal for adsorption-based applications; however, experimental data on their mechanical and adsorption performance remain limited. Wet-spun fibre, specifically those fabricated from alginate fibres, is the most cost-effective and sustainable; yet, their tendency to swell and leach Ca2+ compromises long-term structural stability. Wet-spun cellulose fibres deliver superior mechanical strength and water stability due to dense hydrogen bonding when ionic liquid was used,285 but their compact microstructure restricts diffusion and adsorption efficiency.
Overall, no single fabrication method is universally optimal. Instead, the results suggest that hybrid structures integrating the high surface area of electrospun alginate with the mechanical robustness of cellulose wet-spun fibres may offer the most balanced performance for advanced heavy-metal removal in water purification. Future studies can build on this analysis by modifying fibre compositions used in our study or related literature by integrating nanomaterials for enhanced selectivity to scale up the fibre for production in real-world applications.
To bridge the gap between scientific research and industrial adoption, a study has ensured that the development of bio-based water purification materials, including those incorporating M. oleifera, aligns with sustainability goals, regulatory compliance, and practical feasibility.275 Furthermore, we believe that countries such as developing nations, where low-cost and locally sourced materials are essential for clean water access, can use M. oleifera-based fibre composites to provide a viable, sustainable, and highly effective solution for addressing heavy metal contamination in drinking water and industrial wastewater.
8 Research gaps and future directions
Despite significant progress in developing biopolymer-based adsorbents for heavy metal remediation, key research gaps persist in the current literature. Many existing systems depend heavily on synthetic or chemically modified materials, raising concerns about sustainability, cost, and potential secondary pollution. Natural biopolymers like sodium alginate and cellulose are gaining increased attention due to their abundance, biodegradability, and functional groups suitable for metal ion binding. However, their full potential is yet to be realised, particularly in hybrid forms incorporating low-cost biosorbents such as M. oleifera. While seed extracts have been extensively studied as biosorbents because of their cationic proteins and bioactive components, limited research has been conducted on their integration with alginate and cellulose using advanced fabrication techniques like wet spinning and electrospinning. Despite the rising demand for biodegradable and renewable alternatives, several studies have focused on synthetic polymers and unsustainable materials. Integrating alginate, cellulose, and M. oleifera within engineered fibres marks an emerging research frontier. These materials can be utilised to develop adsorbents with tunable adsorption capacities based on optimised parameters such as pH, dosage, and contact time. However, the experimental frameworks for selecting and fine-tuning these parameters have not been fully developed. Furthermore, although the seed has been the most studied part of M. oleifera, other parts of the plant, such as the bark, husk, and leaves, contain functional bioactive compounds and should be comparatively assessed for their adsorption efficacy.Therefore, future studies should aim to:
(I) Explore underutilised parts of M. oleifera in combination with alginate, cellulose or their combinations.
(II) Optimise electrospinning and wet spinning methods to fabricate advanced biopolymeric adsorbents.
(III) Establish application-relevant parameters for enhanced adsorption capacities.
(IV) Validate composite performance in real water matrices and assess their regeneration, reusability potential and detailed assessment of adsorption performance.
These gaps highlight the need for systematic investigations that bridge materials science and environmental engineering. The insights gained from this review provide a foundational basis for selecting suitable material combinations, fabrication strategies, and operational parameters for improved heavy metal adsorption.
9 Conclusion
This review critically evaluates the potential of alginate, cellulose, and M. oleifera-based composites for heavy metal removal from aqueous systems. These bio-based materials offer environmentally friendly, low-cost alternatives to conventional synthetic adsorbents and align with the goals of sustainable water treatment. Sodium alginate and cellulose provide the necessary functional groups for efficient adsorption, while M. oleifera contributes additional bioactive compounds that enhance adsorption performance. Although significant progress has been made in their utilisation, a lack of integrated systems developed using advanced fabrication techniques such as electrospinning and wet spinning remains. The novelty of this study lies in its emphasis on the potential interactions among these bio-based components and the emerging fabrication strategies that can enhance their adsorption properties. Based on the systematic comparison of sodium alginate, cellulose, and M. oleifera composites, alginate-based systems consistently show that cellulose-based composites offer the most consistent and broadly effective adsorption performance. Alginate-based systems can reach exceptionally high capacities, though largely when modified. M. oleifera adsorbents remain effective and sustainable but generally show lower capacities unless chemically enhanced. Taken together, cellulose emerges as the most reliable high-performance bio-adsorbent, followed by alginate and M. oleifera. Notably, the review highlights how adsorption performance can be tuned through parameter optimisation rather than solely relying on mechanical strength or structural modifications. The major gaps remain in developing sustainable, high-performance bio-based adsorbents. The combined use of alginate, cellulose, and M. oleifera, especially within engineered fibres, remains underexplored, and optimisation frameworks for adsorption parameters are still limited. Furthermore, most work focuses only on the seed, leaving other functional plant parts insufficiently investigated. The findings herein contribute to the body of knowledge by outlining the suitability of these biopolymers as viable adsorbents for water purification and by identifying clear directions for material selection, design, and implementation. Ultimately, this review provides a basis for designing future studies to improve adsorption capacities through the development of tailored composites using sustainable materials and processes.Author contributions
Orisawayi Abimbola: conceptualisation, literature search, literature funding acquisition, analysis, visualisation, software, project administration, methodology, investigation, formal analysis, conceptualisation, writing – review & editing, writing – original draft. Krzysztof K. Koziol & Sameer S. Rahatekar: supervision.Permissions
All required permissions for the use of copyrighted or previously published materials have been obtained, and the permission documents are provided in the supplementary files submitted with this manuscript.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The author gratefully acknowledges Nigeria's Petroleum Technology Development Fund (PTDF) for its support under Grant Number PTDF/ED/OSS/PHD/AOO/1844/2020PHD152, which has been instrumental in facilitating doctoral research. Based on the findings of this study, the author proposes that the Federal Government of Nigeria, through its relevant ministries, investigate and promote the utilisation of abundant natural resources such as M. oleifera, seaweeds (for alginate extraction), and cellulose-rich ago-residues. Furthermore, establishing accessible electrospinning and wet-spinning facilities for researchers working in this area would significantly enhance national research capacity and foster innovation in sustainable water treatment technologies. Harnessing these bioresources for sustainable water treatment technologies could enhance national environmental strategies and contribute to achieving the UN Sustainable Development Goals (SDGs).References
- H. Chawla, S. K. Singh and A. K. Haritash, Reversing the Damage: Ecological Restoration of Polluted Water Bodies Affected by Pollutants Due to Anthropogenic Activities, Environ. Sci. Pollut. Res., 2023, 31(1), 127–143, DOI:10.1007/s11356-023-31295-w.
- M. S. Arias-Paić and J. A. Korak, Forward Osmosis for Ion Exchange Waste Brine Management, Environ. Sci. Technol. Lett., 2020, 7(2), 111–117, DOI:10.1021/acs.estlett.9b00733.
- T. F. Ediagbonya, O. I. Olaniyan, A. O. Orisawayi, F. E. Oziegbe and H. T. Wareromork, Risk Assessment of Polychlorinated Biphenyls (PCBs) in Surface and Groundwater in Three States in Nigeria, Water Pract. Technol., 2025, 20(7), 1557–1574, DOI:10.2166/wpt.2025.088.
- B. J. M. Chaúque, F. L. de Amorim Nascimento, K. J. S. Silva, R. B. Hoff, J. R. Goldim, M. B. Rott, R. A. Zanette and S. Verruck, Solar-Based Technologies for Removing Potentially Toxic Metals from Water Sources: A Review, Environ. Sci. Pollut. Res., 2025, 32(7), 3503–3530, DOI:10.1007/s11356-025-35897-4.
- A. Das, Applying the Water Quality Indices, Geographical Information System, and Advanced Decision-Making Techniques to Assess the Suitability of Surface Water for Drinking Purposes in Brahmani River Basin (BRB), Odisha, Environ. Sci. Pollut. Res., 2025, 1–36, DOI:10.1007/s11356-025-36329-z.
- S. Meftah, K. Meftah, M. Drissi, I. Radah, K. Malous, A. Amahrous, A. Chahid, T. Tamri, A. Rayyad, B. Darkaoui, S. Hanine, O. El-Hassan and L. Bouyazza, Heavy Metal Polluted Water: Effects and Sustainable Treatment Solutions Using Bio-Adsorbents Aligned with the SDGs, Discov. Sustain., 2025, 6(1), 137, DOI:10.1007/s43621-025-00895-6.
- M. K. Abd Elnabi, N. E. Elkaliny, M. M. Elyazied, S. H. Azab, S. A. Elkhalifa, S. Elmasry, M. S. Mouhamed, E. M. Shalamesh, N. A. Alhorieny, A. E. Abd Elaty, I. M. Elgendy, A. E. Etman, K. E. Saad, K. Tsigkou, S. S. Ali, M. Kornaros and Y. A.-G. Mahmoud, Toxicity of Heavy Metals and Recent Advances in Their Removal: A Review, Toxics, 2023, 11(7), 1–29, DOI:10.3390/toxics11070580.
- K. H. Hama Aziz, F. S. Mustafa, K. M. Omer, S. Hama, R. F. Hamarawf and K. O. Rahman, Heavy Metal Pollution in the Aquatic Environment: Efficient and Low-Cost Removal Approaches to Eliminate Their Toxicity: A Review, RSC Adv., 2023, 13(26), 17595–17610, 10.1039/D3RA00723E.
- S. Rajendran, V. Rathinam, A. Sharma, S. Vallinayagam and M. Muthusamy, Arsenic and Environment: A Systematic Review on Arsenic Sources, Uptake Mechanism in Plants, Health Hazards and Remediation Strategies, Top. Catal., 2024, 67(1–4), 325–341, DOI:10.1007/s11244-023-01901-9.
- X. Shi, D. Mao, K. Song, H. Xiang, S. Li and Z. Wang, Effects of Landscape Changes on Water Quality: A Global Meta-Analysis, Water Res., 2024, 260, 1–14, DOI:10.1016/j.watres.2024.121946.
- A. Nayak, P. Chaudhary, B. Bhushan, K. Ghai, S. Singh and M. Sillanpää, Removal of Emergent Pollutants: A Review on Recent Updates and Future Perspectives on Polysaccharide-Based Composites Vis-à-Vis Traditional Adsorbents, Int. J. Biol. Macromol., 2024, 258, 129092, DOI:10.1016/j.ijbiomac.2023.129092.
- United Nations, Progress on Change in Water-Use Efficiency Global Status and Acceleration Needs for SDG Indicator 6.4.1, 2021 Search PubMed.
- J. Song, Bridging Universities and the World: A Cross-National Analysis of Countries' Participation in the UNITWIN/UNESCO Chairs Programme, 1992–2020, Compare, 2024, 1–19, DOI:10.1080/03057925.2024.2352830.
- G. M. Geise, H. Lee, D. J. Miller, B. D. Freeman, J. E. McGrath and D. R. Paul, Water Purification by Membranes: The Role of Polymer Science, J. Polym. Sci., Part B: Polym. Phys., 2010, 48(15), 1685–1718, DOI:10.1002/polb.22037.
- A. Jiménez, L. Á. Prado and P. Saikia, Unleashing Capacity in the Water Sector: A Framework for Public Entities, Water Policy, 2024, 577–599, DOI:10.2166/wp.2024.038.
- P. Cookey, P. E. Cookey, M. Abiodun Peter-Cookey, Regenerative Sanitation: A Conceptual Framework towards Transforming Sanitation Service Improvement and Access Expansion, 2024, vol. 1, https://www.researchgate.net/publication/382184095 Search PubMed.
- S.-Y. Pan, S. W. Snyder, A. I. Packman, Y. J. Lin and P.-C. Chiang, Cooling Water Use in Thermoelectric Power Generation and Its Associated Challenges for Addressing Water-Energy Nexus, Water-Energy Nexus, 2018, 1(1), 26–41, DOI:10.1016/j.wen.2018.04.002.
- I. O. Oladele, S. O. Adelani, A. S. Taiwo, I. M. Akinbamiyorin, O. F. Olanrewaju and A. O. Orisawayi, Polymer-Based Nanocomposites for Supercapacitor Applications: A Review on Principles, Production and Products, RSC Adv., 2025, 15(10), 7509–7534, 10.1039/D4RA08601E.
- I. O. Oladele, T. B. Origbemisoye, A. S. Taiwo, S. Oyegunna, S. O. Adelani, O. F. Olanrewaju and A. O. Orisawayi, Alkaline Modified Coir and Unmodified Hemp Fiber Reinforced Epoxy Based Composite for Automotive Application, Adv. Mat. Sustain. Manuf., 2024, 1(2), 1–12, DOI:10.70322/amsm.2024.10010.
- Z. Zia, A. Hartland and M. R. Mucalo, Use of Low-Cost Biopolymers and Biopolymeric Composite Systems for Heavy Metal Removal from Water, Int. J. Environ. Sci. Technol., 2020, 17(10), 4389–4406, DOI:10.1007/s13762-020-02764-3.
- A. N. Doyo, R. Kumar and M. A. Barakat, Recent Advances in Cellulose, Chitosan, and Alginate Based Biopolymeric Composites for Adsorption of Heavy Metals from Wastewater, J. Taiwan Inst. Chem. Eng., 2023, 151, 1–18, DOI:10.1016/j.jtice.2023.105095.
- A. O. Orisawayi, H. Lu, I. J. Badruddin, P. D. Venkatraman, N. S. Britten, J. A. Butler, K. Koziol and S. S. Rahatekar, Deposition of Alginate-Oregano Nanofibres on Cotton Gauze for Potential Antimicrobial Applications, Int. J. Biol. Macromol., 2025, 319, 1–16, DOI:10.1016/j.ijbiomac.2025.145372.
- G. P. Udayakumar, S. Muthusamy, B. Selvaganesh, N. Sivarajasekar, K. Rambabu, S. Sivamani, N. Sivakumar, J. P. Maran and A. Hosseini-Bandegharaei, Ecofriendly Biopolymers and Composites: Preparation and Their Applications in Water-Treatment, Biotechnol. Adv., 2021, 52, 1–20, DOI:10.1016/j.biotechadv.2021.107815.
- A. K. Rana, V. K. Gupta, P. Hart and V. K. Thakur, Cellulose-Alginate Hydrogels and Their Nanocomposites for Water Remediation and Biomedical Applications, Environ. Res., 2024, 243, 1–25, DOI:10.1016/j.envres.2023.117889.
- A. O. Orisawayi, P. Boylla, K. K. Koziol and S. S. Rahatekar, Sustainable Wet-Spun Cellulose-Moringa Oleifera Composite Fibres for Potential Water Purification, RSC Adv., 2025, 15(22), 17730–17745, 10.1039/D5RA02386F.
- I. I. Kabir, C. C. Sorrell, S. S. Mofarah, W. Yang, A. C. Y. Yuen, M. T. Nazir and G. H. Yeoh, Alginate/Polymer-Based Materials for Fire Retardancy: Synthesis, Structure, Properties, and Applications, Polym. Rev., 2021, 61(2), 357–414, DOI:10.1080/15583724.2020.1801726.
- A. O. Orisawayi, K. K. Koziol and S. S. Rahatekar, Development and Characterisation of Integrated Wet-Spun Alginate-Moringa Oleifera Composite Fibers for Potential Water Purification, Carbohydr. Polym. Technol. Appl., 2025, 9, 1–15, DOI:10.1016/j.carpta.2024.100620.
- T. Russo, P. Fucile, R. Giacometti and F. Sannino, Sustainable Removal of Contaminants by Biopolymers: A Novel Approach for Wastewater Treatment. Current State and Future Perspectives, Processes, 2021, 9(4), 1–20, DOI:10.3390/pr9040719.
- P. R. Yaashikaa, P. Senthil Kumar and S. Karishma, Review on Biopolymers and Composites – Evolving Material as Adsorbents in Removal of Environmental Pollutants, Environ. Res., 2022, 212, 1–13, DOI:10.1016/j.envres.2022.113114.
- M. Abatal, M. Olguin, I. Anastopoulos, D. Giannakoudakis, E. Lima, J. Vargas and C. Aguilar, Comparison of Heavy Metals Removal from Aqueous Solution by Moringa Oleifera Leaves and Seeds, Coatings, 2021, 11(5), 1–14, DOI:10.3390/coatings11050508.
- N. Ueda Yamaguchi, F. Cusioli, H. B. Quesada, M. Eliana, C. Ferreira, R. Fagundes-Klen, A. Marquetotti, S. Vieira, R. G. Gomes, M. Fernandes Vieira and R. Bergamasco, A Review of Moringa Oleifera Seeds in Water Treatment: Trends and Future Challenges, Process Saf. Environ. Prot., 2021, 147, 405–420, DOI:10.1016/j.psep.2020.09.044.
- A. O. Orisawayi, K. K. Koziol and S. S. Rahatekar, Development and Characterisation of Integrated Wet-Spun Alginate-Moringa Oleifera Composite Fibers for Potential Water Purification, Carbohydr. Polym. Technol. Appl., 2025, 14, 8502–8512, DOI:10.1016/j.carpta.2024.100620.
- G. D. Worku and S. N. Abate, Efficiency Comparison of Natural Coagulants (Cactus Pads and Moringa Seeds) for Treating Textile Wastewater (in the Case of Kombolcha Textile Industry), Heliyon, 2025, 11(4), 1–19, DOI:10.1016/j.heliyon.2025.e42379.
- I. W. Maina, V. Obuseng and F. Nareetsile, Use of Moringa Oleifera (Moringa) Seed Pods and Sclerocarya Birrea (Morula) Nut Shells for Removal OfHeavy Metals from Wastewater and Borehole Water, J. Chem., 2016, 2016, 1–13, DOI:10.1155/2016/9312952.
- S. Mahfuz and X. S. Piao, Application of Moringa(Moringa Oleifera) as Natural Feed Supplement in Poultry Diets, Animals, 2019, 9(7), 1–19, DOI:10.3390/ani9070431.
- A. O. Orisawayi, K. Koziol, S. Hao, S. Tiwari and S. S. Rahatekar, Development of Hybrid Electrospun Alginate-Pulverized Moringa Composites, RSC Adv., 2024, 14(12), 8502–8512, 10.1039/D4RA00162A.
- A. O. Orisawayi, K. Koziol, S. Hao, S. Tiwari and S. S. Rahatekar, Development of Hybrid Electrospun Alginate-Pulverized Moringa Composites, RSC Adv., 2024, 14(12), 8502–8512, 10.1039/D4RA00162A.
- M. Hadj Brahim, A. Benettayeb, B. Haddou, M. Belkacem, N. E. H. Aouedj, R. Ould Adda, D. Moudir and A. Hosseini-Bandegharaei, Sodium Alginate-Polyethyleneimine-Moringa Oleifera (Leaves and Seeds) Beads for the Adsorption of Uranium: Isotherm and Kinetic Studies, J. Radioanal. Nucl. Chem., 2025, 1–17, DOI:10.1007/s10967-025-10026-0.
- V. U. Siddiqui, R. A. Ilyas, S. M. Sapuan, N. H. A. Hamid, P. S. Khoo, A. Chowdhury, M. S. N. Atikah, M. S. A. Rani and M. R. M. Asyraf, Alginate-Based Materials as Adsorbent for Sustainable Water Treatment, Int. J. Biol. Macromol., 2025, 298, 1–20, DOI:10.1016/j.ijbiomac.2025.139946.
- A. A. Nassar, M. F. Mubarak, A. K. El-Sawaf, M. A. Zayed and M. Hemdan, Efficient Lead Ion Removal from Aqueous Solutions for Wastewater Treatment Using a Novel Cross-Linked Alginate-Rice Husk Ash-Graphene Oxide-Chitosan Nanocomposite, Int. J. Biol. Macromol., 2025, 284, 1–13, DOI:10.1016/j.ijbiomac.2024.137983.
- J. Liang, X. Li, M. Wu, C. Chen, Z. Hu, M. Zhao and Y. Xue, MXene/Polyaniline/Sodium Alginate Composite Gel: Adsorption and Regeneration Studies and Application in Cu(II) and Hg(II) Removal, Sep. Purif. Technol., 2025, 353, 1–12, DOI:10.1016/j.seppur.2024.128298.
- X. Pei, L. Gan, Z. Tong, H. Gao, S. Meng, W. Zhang, P. Wang and Y. Chen, Robust Cellulose-Based Composite Adsorption Membrane for Heavy Metal Removal, J. Hazard. Mater., 2021, 406, 1–11, DOI:10.1016/j.jhazmat.2020.124746.
- J. Liu, T.-H. Xie, C. Deng, K.-F. Du, N. Zhang, J.-J. Yu, Y.-L. Zou and Y.-K. Zhang, Welan Gum-Modified Cellulose Bead as an Effective Adsorbentof Heavy Metal Ions (Pb 2+, Cu 2+, and Cd 2+) in Aqueous Solution, Sep. Sci. Technol., 2014, 49(7), 1096–1103, DOI:10.1080/01496395.2013.872658.
- M. K. Yoo, M. S. Reza, I. M. Kim and K. J. Kim, Physical Properties and Fibrillation Tendency of Regenerated Cellulose Fiber Dry Jet-Wet Spun from High-Molecular Weight Cotton Linter Pulp/NMMO Solution, Fibers Polym., 2015, 16(8), 1618–1628, DOI:10.1007/s12221-015-5313-y.
- A. A. Taha, Y. Wu, H. Wang and F. Li, Preparation and Application of Functionalized Cellulose Acetate/Silica Composite Nanofibrous Membrane via Electrospinning for Cr(VI) Ion Removal from Aqueous Solution, J. Environ. Manage., 2012, 112, 10–16, DOI:10.1016/j.jenvman.2012.05.031.
- M. Hassan, R. Naidu, J. Du, F. Qi, M. A. Ahsan and Y. Liu, Magnetic Responsive Mesoporous Alginate/β-Cyclodextrin Polymer Beads Enhance Selectivity and Adsorption of Heavy Metal Ions, Int. J. Biol. Macromol., 2022, 207, 826–840, DOI:10.1016/j.ijbiomac.2022.03.159.
- D. Xiang, F. Fang, X. Shi, C. Rao, S. Bao, B. Xian, F. Chu and T. Fang, Analysis and Comparative Study of Preparation, Mechanisms, and Application of Sodium Alginate-Based Composite Materials for Highly Efficient Removal of Cadmium Cations, J. Clean. Prod., 2025, 499, 145234, DOI:10.1016/j.jclepro.2025.145234.
- K. Radhakrishnan, L. Sethuraman, R. Panjanathan, A. Natarajan, V. Solaiappan and W. R. Thilagaraj, Biosorption of Heavy Metals from Actual Electroplating Wastewater Using Encapsulated Moringa Oleifera Beads in Fixed Bed Column, Desalination Water Treat., 2016, 57(8), 3572–3587, DOI:10.1080/19443994.2014.985725.
- A. G. Hassabo, S. Shaarawy, A. L. Mohamed and A. Hebiesh, Multifarious Cellulosic through Innovation of Highly Sustainable Composites Based on Moringa and Other Natural Precursors, Int. J. Biol. Macromol., 2020, 165, 141–155, DOI:10.1016/j.ijbiomac.2020.09.125.
- W. A. Al-Amrani and S. A. Onaizi, Adsorptive Removal of Heavy Metals from Wastewater Using Emerging Nanostructured Materials: A State-of-the-Art Review, Sep. Purif. Technol., 2024, 343, 1–60, DOI:10.1016/j.seppur.2024.127018.
- J. Sun, R. Hu, X. Zhao, T. Liu and Z. Bai, A Novel Chitosan/Cellulose Phosphonate Composite Hydrogel for Ultrafast and Efficient Removal of Pb(II) and Cu(II) from Wastewater, Carbohydr. Polym., 2024, 336, 1–11, DOI:10.1016/j.carbpol.2024.122104.
- S.-C. Yang, Y. Liao, K. G. Karthikeyan and X. J. Pan, Mesoporous Cellulose-Chitosan Composite Hydrogel Fabricated via the Co-Dissolution-Regeneration Process as Biosorbent of Heavy Metals, Environ. Pollut., 2021, 286, 1–10, DOI:10.1016/j.envpol.2021.117324.
- S. Eslamizad and M. Alehashem, Metal Contaminants in Rice Imported to Iran: A Comprehensive Assessment of Carcinogenic and Non-Carcinogenic Health Risks, J. Trace Elem. Med. Biol., 2025, 87, 1–10, DOI:10.1016/j.jtemb.2024.127568.
- M. Balali-Mood, K. Naseri, Z. Tahergorabi, M. R. Khazdair and M. Sadeghi, Toxic Mechanisms of Five Heavy Metals: Mercury, Lead, Chromium, Cadmium, and Arsenic, Front. Pharmacol, 2021, 12, 1–19, DOI:10.3389/fphar.2021.643972.
- A. Saravanan, P. S. Kumar, R. V. Hemavathy, S. Jeevanantham, P. Harikumar, G. Priyanka and D. R. A. Devakirubai, A Comprehensive Review on Sources, Analysis and Toxicity of Environmental Pollutants and Its Removal Methods from Water Environment, Sci. Total Environ., 2022, 812, 1–20, DOI:10.1016/j.scitotenv.2021.152456.
- N. Nirmala, V. Shriniti, K. Aasresha, J. Arun, K. P. Gopinath, S. S. Dawn, A. Sheeladevi, P. Priyadharsini, K. Birindhadevi, N. T. L. Chi and A. Pugazhendhi, Removal of Toxic Metals from Wastewater Environment by Graphene-Based Composites: A Review on Isotherm and Kinetic Models, Recent Trends, Challenges and Future Directions, Sci. Total Environ., 2022, 840, 1–10, DOI:10.1016/j.scitotenv.2022.156564.
- F. Yalcinkaya, E. Boyraz, J. Maryska and K. Kucerova, A Review on Membrane Technology and Chemical Surface Modification for the Oily Wastewater Treatment, Materials, 2020, 13(2), 1–14, DOI:10.3390/ma13020493.
- F. E. Ahmed, B. S. Lalia and R. Hashaikeh, A Review on Electrospinning for Membrane Fabrication: Challenges and Applications, Desalination, 2015, 15–30, DOI:10.1016/j.desal.2014.09.033.
- S. S. Ray, S.-S. Chen, C.-W. Li, N. C. Nguyen and H. T. Nguyen, A Comprehensive Review: Electrospinning Technique for Fabrication and Surface Modification of Membranes for Water Treatment Application, RSC Adv., 2016, 6(88), 85495–85514, 10.1039/C6RA14952A.
- P. Bazaanah and R. A. Mothapo, Sustainability of Drinking Water and Sanitation Delivery Systems in Rural Communities of the Lepelle Nkumpi Local Municipality, South Africa, Environ. Dev. Sustain., 2023, 26(6), 14223–14255, DOI:10.1007/s10668-023-03190-4.
- X. Zhang, H. Yang, R. Sun, M. Cui, N. Sun and S. Zhang, Evaluation and Analysis of Heavy Metals in Iron and Steel Industrial Area, Environ. Dev. Sustain., 2022, 24(9), 10997–11010, DOI:10.1007/s10668-021-01893-0.
- N. K. Soliman and A. F. Moustafa, Industrial Solid Waste for Heavy Metals Adsorption Features and Challenges; a Review, J. Mater. Res. Technol., 2020, 9(5), 10235–10253, DOI:10.1016/j.jmrt.2020.07.045.
- Z. Rahman and V. P. Singh, The Relative Impact of Toxic Heavy Metals (THMs) (Arsenic (As), Cadmium (Cd), Chromium (Cr)(VI), Mercury (Hg), and Lead (Pb)) on the Total Environment: An Overview, Environ. Monit. Assess., 2019, 191(7), 1–21, DOI:10.1007/s10661-019-7528-7.
- N. I. H. Hazril, A. A. Jalil, F. F. A. Aziz, N. S. Hassan, A. A. Fauzi, N. F. Khusnun, N. M. Izzudin, N. W. C. Jusoh, L. P. Teh, N. F. Jaafar and S. Rajendran, Selective Simultaneous Photo-Fenton Removal of Cr (VI) and Methyl Orange Dye over Critical Raw Material-Free Fibrous-Silica Irons Catalyst, Sustain. Mater. Technol., 2024, 41, 1–10, DOI:10.1016/j.susmat.2024.e00994.
- G. Yaqub, A. Khan, M. Zishan Ahmad and U. Irshad, Determination of Concentration of Heavy Metals in Fruits, Vegetables, Groundwater, and Soil Samples of the Cement Industry and Nearby Communities and Assessment of Associated Health Risks, J. Food Qual., 2021, 2021, 1–9, DOI:10.1155/2021/3354867.
- K. Zhang, Y. Shi, Z. Zhang, L. Li, N. Peng, G. Dai, F. Xia and X. Zhang, Magnetic High-Swelling Cyclodextrin Polymer Adsorbent for Rapid Removal of Pollutants and Efficient Recovery from Water, Sep. Purif. Technol., 2025, 366, 1–10, DOI:10.1016/j.seppur.2025.132741.
- Z. Arshad, A. Tanimu, K. Alhooshani and S. A. Ali, A Dual-Purpose β-Cyclodextrin-Derived Sorbent Decorated with Chelating Residues of Aminomethylphosphonate and Aspartic Acid for Removal of Polyaromatic Hydrocarbons and Toxic Metal Ions, J. Mol. Liq., 2025, 426, 1–15, DOI:10.1016/j.molliq.2025.127500.
- M. De Silva, G. Cao and K. C. Tam, Nanomaterials for the Removal and Detection of Heavy Metals: A Review, Environ. Sci. Nano, 2025, 1–23, 10.1039/D4EN01041H.
- A. Kaplan, M. N. Khan, S. Wahab, N. Assad, M. Adnan, Hafsa and M. Iqbal, Thallium in Soil Environments and Its Biological Availability, in Beneficial Elements for Remediation of Heavy Metals in Polluted Soil, Elsevier, 2025, pp. 373–398, DOI:10.1016/B978-0-443-26522-8.00013-1.
- K. Rehman, F. Fatima, I. Waheed and M. S. H. Akash, Prevalence of Exposure of Heavy Metals and Their Impact on Health Consequences, J. Cell. Biochem., 2018, 119(1), 157–184, DOI:10.1002/jcb.26234.
- U.S. Environmental Protection Agency, EPA Secondary Drinking Water Standards, U.S. Environmental Protection Agency, https://www.epa.gov/sdwa/secondary-drinking-water-standards-guidance-nuisance-chemicals (accessed 2025-03-16) Search PubMed.
- U.S. Environmental Protection Agency, National Primary Drinking Water Regulations, United States Environmental Protection Agency (EPA), https://www.epa.gov/ground-water-and-drinking-water/national-primary-drinking-water-regulations (accessed 2025-03-16) Search PubMed.
- European Commission, European Commission, 2023, https://eur-lex.europa.eu/eli/dir/1998/83/2015-10-27 (accessed 2025-03-16).
- World Health Organization, Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First and Second, Geneva, 2022 Search PubMed.
- M. Qadir, P. Drechsel, B. Jiménez Cisneros, Y. Kim, A. Pramanik, P. Mehta and O. Olaniyan, Global and Regional Potential of Wastewater as a Water, Nutrient and Energy Source, Nat. Resour. Forum, 2020, 44(1), 40–51, DOI:10.1111/1477-8947.12187.
- X. Wu, S. J. Cobbina, G. Mao, H. Xu, Z. Zhang and L. Yang, A Review of Toxicity and Mechanisms of Individual and Mixtures of Heavy Metals in the Environment, Environ. Sci. Pollut. Res., 2016, 23(9), 8244–8259, DOI:10.1007/s11356-016-6333-x.
- P. C. Nagajyoti, K. D. Lee and T. V. M. Sreekanth, Heavy Metals, Occurrence and Toxicity for Plants: A Review, Environ. Chem. Lett., 2010, 8(3), 199–216, DOI:10.1007/s10311-010-0297-8.
- R. Levin, C. L. Zilli Vieira, M. H. Rosenbaum, K. Bischoff, D. C. Mordarski and M. J. Brown, The Urban Lead (Pb) Burden in Humans, Animals and the Natural Environment, Environ. Res., 2021, 193, 1–20, DOI:10.1016/j.envres.2020.110377.
- G. J. Udom, B. Turyahabwe, A. Aturamu, O. M. Aziakpono, R. D. Agbana, O. G. Joseph, N. G. Udom, N. Mugide, O. P. Odey, H. Olot and O. E. Orisakwe, Heavy Metal and Metalloid Pollution: A Systematic Review of Health Implications for Pregnant Women, Children, and Geriatrics in the East African Region, Environ. Adv., 2025, 19, 1–14, DOI:10.1016/j.envadv.2025.100620.
- N. A. Suciu, R. De Vivo, N. Rizzati and E. Capri, Cd Content in Phosphate Fertilizer: Which Potential Risk for the Environment and Human Health?, Curr. Opin. Environ. Sci. Health, 2022, 30, 1–6, DOI:10.1016/j.coesh.2022.100392.
- M. Tumolo, V. Ancona, D. De Paola, D. Losacco, C. Campanale, C. Massarelli and V. F. Uricchio, Chromium Pollution in European Water, Sources, Health Risk, and Remediation Strategies: An Overview, Int. J. Environ. Res. Publ. Health, 2020, 17(15), 1–24, DOI:10.3390/ijerph17155438.
- C. Dwivedi, M. A. S. Salem, A. M. Khan, M. Ahmed, A. S. Bhat, H. Hamid and W. A. Khan, Fluorescence Detection of Mercury and Nitroaromatic Chemicals Based on Metal Oxide Derived Multiwall Carbon Nanotube Nanocomposite, Mater. Sci. Eng., B, 2025, 314, 1–11, DOI:10.1016/j.mseb.2025.118055.
- S. Agarwal, S. Kaushik, H. Saha, D. Paramanick, M. Mazhar, P. Basist, R. Khan and A. Alhalmi, Therapeutic Potential of Traditional Herbal Plants and Their Polyphenols in Alleviation of Mercury Toxicity, Naunyn-Schmiedebergs Arch. Pharmacol., 2025, 1–27, DOI:10.1007/s00210-025-03807-7.
- M. K. Yadav, D. Saidulu, A. K. Gupta, P. S. Ghosal and A. Mukherjee, Status and Management of Arsenic Pollution in Groundwater: A Comprehensive Appraisal of Recent Global Scenario, Human Health Impacts, Sustainable Field-Scale Treatment Technologies, J. Environ. Chem. Eng., 2021, 9(3), 1–22, DOI:10.1016/j.jece.2021.105203.
- T. K. Roy, S. K. Nag, U. B. Antu, S. A. Hossain, A. Al Bakky, M. T. Anjum, B. C. Sarker, M. R. Ullah, F. Farzana, N. A. Mahiddin, A. Biswas, P. Singha, M. S. Islam, Z. Ismail and A. M. Idris, A Comprehensive Assessment of Health Risks Associated with Heavy Metal Through Ingestion of Two Predominant Fish Species in a Developing Country, Biol. Trace Elem. Res., 2025, 1–11, DOI:10.1007/s12011-025-04540-1.
- D. Bachirou, N. B. B. L. Marie and N. Paul-Désiré, Assessment of Pollution and Ecological Risk Associated with Heavy Metals in Sediments from the Rivers of Batouri Gold Mining Area (East Cameroon): Geochemical and Statistical Approaches, J. Sediment. Environ., 2025, 1–12, DOI:10.1007/s43217-025-00216-x.
- K. Jomova, M. Makova, S. Y. Alomar, S. H. Alwasel, E. Nepovimova, K. Kuca, C. J. Rhodes and M. Valko, Essential Metals in Health and Disease, Chem. Biol. Interact., 2022, 367, 1–28, DOI:10.1016/j.cbi.2022.110173.
- M. A. Zoroddu, J. Aaseth, G. Crisponi, S. Medici, M. Peana and V. M. Nurchi, The Essential Metals for Humans: A Brief Overview, J. Inorg. Biochem., 2019, 195, 120–129, DOI:10.1016/j.jinorgbio.2019.03.013.
- R. Gaj, D. Kayzer, K. Głuchowska, K. Wielgusz and A. Wolna-Maruwka, A Case Study on the Effect of Foliar Nitrogen Fertilization on the Microbiological and Biochemical State of the Soil and the Uptake of Macro- and Microelements by Winter Triticale (Triticosecale), Agronomy, 2025, 15(2), 1–25, DOI:10.3390/agronomy15020467.
- A. Azimi, A. Azari, M. Rezakazemi and M. Ansarpour, Removal of Heavy Metals from Industrial Wastewaters: A Review, ChemBioEng Rev., 2017, 4(1), 37–59, DOI:10.1002/cben.201600010.
- R. Shrestha, S. Ban, S. Devkota, S. Sharma, R. Joshi, A. P. Tiwari, H. Y. Kim and M. K. Joshi, Technological Trends in Heavy Metals Removal from Industrial Wastewater: A Review, J. Environ. Chem. Eng., 2021, 9(4), 1–18, DOI:10.1016/j.jece.2021.105688.
- Y. Zhu, W. Fan, T. Zhou and X. Li, Removal of Chelated Heavy Metals from Aqueous Solution: A Review of Current Methods and Mechanisms, Sci. Total Environ., 2019, 678, 253–266, DOI:10.1016/j.scitotenv.2019.04.416.
- Q. Chen, Y. Yao, X. Li, J. Lu, J. Zhou and Z. Huang, Comparison of Heavy Metal Removals from Aqueous Solutions by Chemical Precipitation and Characteristics of Precipitates, J. Water Proc. Eng., 2018, 26, 289–300, DOI:10.1016/j.jwpe.2018.11.003.
- A. E. Lewis, Review of Metal Sulphide Precipitation, Hydrometallurgy, 2010, 104(2), 222–234, DOI:10.1016/j.hydromet.2010.06.010.
- H. P. Vu, L. N. Nguyen, Q. Wang, H. H. Ngo, Q. Liu, X. Zhang and L. D. Nghiem, Hydrogen Sulphide Management in Anaerobic Digestion: A Critical Review on Input Control, Process Regulation, and Post-Treatment, Bioresour. Technol., 2022, 346, 1–10, DOI:10.1016/j.biortech.2021.126634.
- A. V. Zatirakha, A. D. Smolenkov and O. A. Shpigun, Preparation and Chromatographic Performance of Polymer-Based Anion Exchangers for Ion Chromatography: A Review, Anal. Chim. Acta, 2016, 904, 33–50, DOI:10.1016/j.aca.2015.11.012.
- A. T. Moh Rabti, A. E. Gok, B. Yuzer and H. Selcuk, Evaluation of a Sustainable Dye-Exhausted Resin Regeneration Method for Cost-Effective Decolorization and Detoxification of Textile Wastewater, Eng. Sci. Technol. Int. J., 2025, 63, 1–24, DOI:10.1016/j.jestch.2025.101973.
- A. Singh, S. S. Shah, C. Sharma, V. Gupta, A. K. Sundramoorthy, P. Kumar and S. Arya, An Approach towards Different Techniques for Detection of Heavy Metal Ions and Their Removal from Waste Water, J. Environ. Chem. Eng., 2024, 12(3), 1–24, DOI:10.1016/j.jece.2024.113032.
- H. A. Abdulgader, V. Kochkodan and N. Hilal, Hybrid Ion Exchange – Pressure Driven Membrane Processes in Water Treatment: A Review, Sep. Purif. Technol., 2013, 116, 253–264, DOI:10.1016/j.seppur.2013.05.052.
- E. Kavitha, E. Poonguzhali, D. Nanditha, A. Kapoor, G. Arthanareeswaran and S. Prabhakar, Current Status and Future Prospects of Membrane Separation Processes for Value Recovery from Wastewater, Chemosphere, 2022, 291, 1–16, DOI:10.1016/j.chemosphere.2021.132690.
- A. Lee, J. W. Elam and S. B. Darling, Membrane Materials for Water Purification: Design, Development, and Application, Environ. Sci., 2016, 2(1), 17–42, 10.1039/C5EW00159E.
- S. J. Judd, Membrane Technology Costs and Me, Water Res., 2017, 122, 1–9, DOI:10.1016/j.watres.2017.05.027.
- J. Lu, S. Lv, H. S. Park and Q. Chen, Electrocatalytically Active and Charged Natural Chalcopyrite for Nitrate-Contaminated Wastewater Purification Extended to Energy Storage Zn-NO3− Battery, J. Hazard. Mater., 2024, 477, 1–13, DOI:10.1016/j.jhazmat.2024.135287.
- M. Seifi, A. Kamran-Pirzaman, A. Dehghani Kiadehi and M. Rahimnejad, A Comprehensive Comparison of Various Methods and Hybrid Systems in Leachate Treatment: A Review, Int. J. Environ. Sci. Technol., 2025, 1–96, DOI:10.1007/s13762-025-06367-8.
- C. Un, Assessing Biogas from Wastewater Treatment Plants for Sustainable Transportation Fuel: A Detailed Analysis of Energy Potential and Emission Reductions, Gases, 2025, 5(1), 1–18, DOI:10.3390/gases5010006.
- M. U. Saeed, N. Hussain, A. Sumrin, A. Shahbaz, S. Noor, M. Bilal, L. Aleya and H. M. N. Iqbal, Microbial Bioremediation Strategies with Wastewater Treatment Potentialities – A Review, Sci. Total Environ., 2022, 818, 1–11, DOI:10.1016/j.scitotenv.2021.151754.
- T. Gebregiorgis Ambaye, M. Vaccari, A. Franzetti, S. Prasad, F. Formicola, A. Rosatelli, A. Hassani, T. M. Aminabhavi and S. Rtimi, Microbial Electrochemical Bioremediation of Petroleum Hydrocarbons (PHCs) Pollution: Recent Advances and Outlook, Chem. Eng. J., 2023, 452, 1–22, DOI:10.1016/j.cej.2022.139372.
- C. S. Ezeonu, R. Tagbo, E. N. Anike, O. A. Oje and I. N. E. Onwurah, Biotechnological Tools for Environmental Sustainability: Prospects and Challenges for Environments in Nigeria—A Standard Review, Biotechnol. Res. Int., 2012, 2012, 1–26, DOI:10.1155/2012/450802.
- Y. Wang, Y. Pan, W. Han, C. S. Rossi, Q. Hui, Y. Guo, M. C. Owoseni, E. McAdam, Y.-C. Yong, B. Wang and Z. Yang, CRISPR-Enabled Sensors for Rapid Monitoring of Environmental Contaminants, TrAC, Trends Anal. Chem., 2025, 184, 1–17, DOI:10.1016/j.trac.2024.118128.
- P. Atkins, Shriver and Atkins' Inorganic Chemistry, Oxford University Press, USA, 9th edn, 2010 Search PubMed.
- S. J. Zarrouk and K. Mclean, Advanced Analytical Pressure-Transient Analysis Relevant to Geothermal Wells, Geothermal Well Test Analysis, 2019, vol. 89, pp. 89–111 Search PubMed.
- G. Li, Y. Tao, Y. Gao, P. Shen, X. Qian, B. Yin, R. J.-M. Pellenq and C. S. Poon, Water's Grip on CO2 Intake in Mesopores of Dicalcium Silicate, Cem. Concr. Res., 2025, 192, 1–11, DOI:10.1016/j.cemconres.2025.107842.
- S. M. Abegunde, K. S. Idowu, O. M. Adejuwon and T. Adeyemi-Adejolu, A Review on the Influence of Chemical Modification on the Performance of Adsorbents, Resour. Environ. Sustain., 2020, 1, 1–9, DOI:10.1016/j.resenv.2020.100001.
- J. Kaur, P. Sengupta and S. Mukhopadhyay, Critical Review of Bioadsorption on Modified Cellulose and Removal of Divalent Heavy Metals (Cd, Pb, and Cu), Ind. Eng. Chem. Res., 2022, 61(5), 1921–1954, DOI:10.1021/acs.iecr.1c04583.
- A. K. Dhara and A. K. Nayak, Biological Macromolecules: Sources, Properties, and Functions, in Biological Macromolecules, ed. A. Kumar Nayak, A. Kumar Dhara and D. Pal, Elsevier, 2022, pp. 3–22, DOI:10.1016/B978-0-323-85759-8.00005-1.
- M. Hussain, A. Riaz, H. Zeb, A. Ali, R. Mujahid, F. Ahmad and M. S. Zafar, Paving the Path to Water Security: The Role of Advanced Adsorbents in Wastewater Treatment, J. Water Proc. Eng., 2025, 71, 1–12, DOI:10.1016/j.jwpe.2025.107333.
- A. Marotta, A. Borriello, M. R. Khan, S. Cavella, V. Ambrogi and E. Torrieri, Boosting Food Packaging Sustainability Through the Valorization of Agri-Food Waste and By-Products, Polymers, 2025, 17(6), 1–33, DOI:10.3390/polym17060735.
- Z. U. Arif, M. Y. Khalid, M. F. Sheikh, A. Zolfagharian and M. Bodaghi, Biopolymeric Sustainable Materials and Their Emerging Applications, J. Environ. Chem. Eng., 2022, 10(4), 1–24, DOI:10.1016/j.jece.2022.108159.
- L. G. Pinaeva and A. S. Noskov, Biodegradable Biopolymers: Real Impact to Environment Pollution, Sci. Total Environ., 2024, 947, 1–25, DOI:10.1016/j.scitotenv.2024.174445.
- C. Selvaraj, D. C. Dinesh, K. Rajaram, S. Sundaresan and S. K. Singh, Macromolecular Chemistry: An Introduction, in In Silico Approaches to Macromolecular Chemistry, ed. M. Elizabeth Thomas, J. Thomas, S. Thomas and H. Kornweitz, Elsevier, 2023, pp. 71–128, DOI:10.1016/B978-0-323-90995-2.00007-2.
- M.-H. Lee, K.-H. Teng, Y.-Y. Liang, C.-F. Ding and Y.-C. Chen, Flexible Biodegradable Wearables Based on Conductive Leaf Networks, Sustain. Mater. Technol., 2025, 43, 1–8, DOI:10.1016/j.susmat.2025.e01263.
- G. H. Matar and M. Andac, Recent Advances in Sustainable Biopolymer Films Incorporating Vanillin for Enhanced Food Preservation and Packaging, Polym. Bull., 2025, 1–27, DOI:10.1007/s00289-025-05661-2.
- T. Kibet, D. N. Githinji and P. Nziu, Natural Fibre–Reinforced Starch Biocomposites and Their Effects on the Material Mechanical Properties: A Review, Adv. Mater. Sci. Eng., 2025, 2025(1), 1–20, DOI:10.1155/amse/9905014.
- M. Piryaei, M. M. Abolghasemi and M. Sadeghi Raked, Eco-Friendly and Smart Biopolymer with Green Synthesis Nanoparticles, Anthocyanins, Sodium Alginate, Pectin, and Checking the Degree of Greenness of the Method, Results Surf. Interfaces, 2025, 18, 1–11, DOI:10.1016/j.rsurfi.2025.100433.
- M. Kogje, A. Satdive, S. Mestry and S. T. Mhaske, Biopolymers: A Comprehensive Review of Sustainability, Environmental Impact, and Lifecycle Analysis, Iran. Polym. J., 2025, 1–44, DOI:10.1007/s13726-024-01449-9.
- U. Tukenmez Emre, S. Sirin, S. Nigdelioglu Dolanbay and B. Aslim, Harnessing Polysaccharides for Sustainable Food Packaging, Polym. Bull., 2025, 1–47, DOI:10.1007/s00289-025-05659-w.
- A. B. Maslinda, M. S. A. Majid, M. J. M. Ridzuan, M. Afendi and A. G. Gibson, Effect of Water Absorption on the Mechanical Properties of Hybrid Interwoven Cellulosic-Cellulosic Fibre Reinforced Epoxy Composites, Compos. Struct., 2017, 167, 227–237 CrossRef.
- M. Cao, Y. Hu, W. Cheng, S. Huan, T. Bai, Z. Niu, Y. Zhao, G. Yue, Y. Zhao and G. Han, Lignin-Based Multi-Scale Cellular Aerogels Assembled from Co-Electrospun Nanofibers for Oil/Water Separation and Energy Storage, Chem. Eng. J., 2022, 436, 1–10, DOI:10.1016/j.cej.2022.135233.
- F. Genua, I. Lancellotti and C. Leonelli, Geopolymer-Based Stabilization of Heavy Metals, the Role of Chemical Agents in Encapsulation and Adsorption: Review, Polymers, 2025, 17(5), 1–29, DOI:10.3390/polym17050670.
- J. Parades-Aguilar, S. Agustin-Salazar, P. Cerruti, V. Ambrogi, K. Calderon, N. Gamez-Meza and L. A. Medina-Juarez, Agro-Industrial Wastes and Their Application Perspectives in Metal Decontamination Using Biocomposites and Bacterial Biomass: A Review, World J. Microbiol. Biotechnol., 2025, 41(1), 1–31, DOI:10.1007/s11274-024-04227-0.
- M. M. Aljohani, S. D. Al-Qahtani, M. Alshareef, M. G. El-Desouky, A. A. El-Bindary, N. M. El-Metwaly and M. A. El-Bindary, Highly Efficient Adsorption and Removal Bio-Staining Dye from Industrial Wastewater onto Mesoporous Ag-MOFs, Process Saf. Environ. Prot., 2023, 172, 395–407, DOI:10.1016/j.psep.2023.02.036.
- M. Sultana, M. H. Rownok, M. Sabrin, M. H. Rahaman and S. M. N. Alam, A Review on Experimental Chemically Modified Activated Carbon to Enhance Dye and Heavy Metals Adsorption, Clean Eng. Technol., 2022, 6, 1–14, DOI:10.1016/j.clet.2021.100382.
- R. Zhang, B. Liu, J. Ma and R. Zhu, Preparation and Characterization of Carboxymethyl Cellulose/Chitosan/Alginic Acid Hydrogels with Adjustable Pore Structure for Adsorption of Heavy Metal Ions, Eur. Polym. J., 2022, 179, 1–10, DOI:10.1016/j.eurpolymj.2022.111577.
- M. Chen, A. Long, W. Zhang, Z. Wang, X. Xiao, Y. Gao, L. Zhou, Y. Li, J. Wang, S. Sun, M. Tang, Y. Peng and H. Wang, Recent Advances in Alginate-Based Hydrogels for the Adsorption–Desorption of Heavy Metal Ions from Water: A Review, Sep. Purif. Technol., 2025, 353, 1–24, DOI:10.1016/j.seppur.2024.128265.
- E. Khademian, E. Salehi, H. Sanaeepur, F. Galiano and A. Figoli, A Systematic Review on Carbohydrate Biopolymers for Adsorptive Remediation of Copper Ions from Aqueous Environments-Part A: Classification and Modification Strategies, Sci. Total Environ., 2020, 738, 1–20, DOI:10.1016/j.scitotenv.2020.139829.
- M. Tagavifar, S. H. Jang, H. Sharma, D. Wang, L. Y. Chang, K. Mohanty and G. A. Pope, Effect of PH on Adsorption of Anionic Surfactants on Limestone: Experimental Study and Surface Complexation Modeling, Colloids Surf., A, 2018, 538, 549–558, DOI:10.1016/j.colsurfa.2017.11.050.
- T. Wang, W. Liu, L. Xiong, N. Xu and J. Ni, Influence of PH, Ionic Strength and Humic Acid on Competitive Adsorption of Pb(II), Cd(II) and Cr(III) onto Titanate Nanotubes, Chem. Eng. J., 2013, 215–216, 366–374, DOI:10.1016/j.cej.2012.11.029.
- P. Ananthi, K. Hemkumar and A. Pius, Antibacterial, Biodegradable Polymeric Films Loaded with Co-MOF/ZnS Nanoparticles for Food Packaging and Photo-Degradation Applications, ACS Food Sci. Technol., 2024, 4(6), 1462–1471, DOI:10.1021/acsfoodscitech.4c00087.
- A. J. Lorbeer, S. Charoensiddhi, J. Lahnstein, C. Lars, C. M. M. Franco, V. Bulone and W. Zhang, Sequential Extraction and Characterization of Fucoidans and Alginates from Ecklonia Radiata, Macrocystis Pyrifera, Durvillaea Potatorum, and Seirococcus Axillaris, J. Appl. Phycol., 2017, 29(3), 1515–1526, DOI:10.1007/s10811-016-0990-5.
- P. Kajla, V. Chaudhary, A. Dewan, S. P. Bangar, S. Ramniwas, S. Rustagi and R. Pandiselvam, Seaweed-Based Biopolymers for Food Packaging: A Sustainable Approach for a Cleaner Tomorrow, Int. J. Biol. Macromol., 2024, 274, 1–16, DOI:10.1016/j.ijbiomac.2024.133166.
- H. P. S. Abdul Khalil, C. K. Saurabh, Y. Y. Tye, T. K. Lai, A. M. Easa, E. Rosamah, M. R. N. Fazita, M. I. Syakir, A. S. Adnan, H. M. Fizree, N. A. S. Aprilia and A. Banerjee, Seaweed Based Sustainable Films and Composites for Food and Pharmaceutical Applications: A Review, Renewable Sustainable Energy Rev., 2017, 77, 353–362, DOI:10.1016/j.rser.2017.04.025.
- H. Zhang, J. Cheng and Q. Ao, Preparation of Alginate-Based Biomaterials and Their Applications in Biomedicine, Mar. Drugs, 2021, 19(5), 1–24, DOI:10.3390/md19050264.
- M. Jönsson, L. Allahgholi, R. R. R. Sardari, G. O. Hreggviðsson and E. Nordberg Karlsson, Extraction and Modification of Macroalgal Polysaccharides for Current and Next-Generation Applications, Molecules, 2020, 25(4), 1–29, DOI:10.3390/molecules25040930.
- T. M. Joseph, A. Sathian, K. S. Joshy, D. K. Mahapatra, J. T. Haponiuk and S. Thomas, Chemical Modifications of Alginate-Based Biopolymers, in Handbook of Natural Polymers, Elsevier, 2024, vol. 2, pp. 97–122, DOI:10.1016/B978-0-323-99856-7.00016-1.
- B. Kumar, N. Singh and P. Kumar, A Review on Sources, Modification Techniques, Properties and Potential Applications of Alginate-Based Modified Polymers, Eur. Polym. J., 2024, 213, 1–25, DOI:10.1016/j.eurpolymj.2024.113078.
- Y. Fei and Y. H. Hu, Design, Synthesis, and Performance of Adsorbents for Heavy Metal Removal from Wastewater: A Review, J. Mater. Chem. A, 2022, 10(3), 1047–1085, 10.1039/D1TA06612A.
- S. N. Pawar and K. J. Edgar, Alginate Derivatization: A Review of Chemistry, Properties and Applications, Biomaterials, 2012, 33(11), 3279–3305, DOI:10.1016/j.biomaterials.2012.01.007.
- S. Dash, P. Gutti, B. Behera and D. Mishra, Anionic Species from Multivalent Metal Salts Are Differentially Retained during Aqueous Ionic Gelation of Sodium Alginate and Could Fine-Tune the Hydrogel Properties, Int. J. Biol. Macromol., 2024, 265, 2478–2488, DOI:10.1016/j.ijbiomac.2024.130767.
- G. A. Martău, M. Mihai and D. C. Vodnar, The Use of Chitosan, Alginate, and Pectin in the Biomedical and Food Sector—Biocompatibility, Bioadhesiveness, and Biodegradability, Polymers, 2019, 11(11), 1837, DOI:10.3390/polym11111837.
- S. Dong, Y. Li, K. Zhu, C. Wang and S. Zhai, Advances in Structure Designing and Function Tailoring Strategy toward Alginate-Based Hydrogels for Efficient Water Remediation: A Review, Int. J. Biol. Macromol., 2025, 304, 1–29, DOI:10.1016/j.ijbiomac.2025.140801.
- A. Benettayeb, J. M. Masamvu, R. M. Chitepo, B. Haddou, M. Sillanpaa and S. Ghosh, Facile Fabrication of New Bioadsorbents from Moringa Oleifera and Alginate for Efficient Removal of Uranium(VI), J. Radioanal. Nucl. Chem., 2024, 333(5), 2369–2387, DOI:10.1007/s10967-024-09470-1.
- R. Sun, S. Gao, K. Zhang, W.-T. Cheng and G. Hu, Recent Advances in Alginate-Based Composite Gel Spheres for Removal of Heavy Metals, Int. J. Biol. Macromol., 2024, 268, 1–19, DOI:10.1016/j.ijbiomac.2024.131853.
- B. Wang, Y. Wan, Y. Zheng, X. Lee, T. Liu, Z. Yu, J. Huang, Y. S. Ok, J. Chen and B. Gao, Alginate-Based Composites for Environmental Applications: A Critical Review, Crit. Rev. Environ. Sci. Technol., 2019, 49(4), 318–356, DOI:10.1080/10643389.2018.1547621.
- T. Lu, T. Xiang, X.-L. Huang, C. Li, W.-F. Zhao, Q. Zhang and C.-S. Zhao, Post-Crosslinking towards Stimuli-Responsive Sodium Alginate Beads for the Removal of Dye and Heavy Metals, Carbohydr. Polym., 2015, 133, 587–595, DOI:10.1016/j.carbpol.2015.07.048.
- J. D. Guerrero, E. R. Arias and L. B. Gutierrez, Enhancing Copper and Lead Adsorption in Water by In-Situ Generation of Calcium Carbonate on Alginate/Chitosan Biocomposite Surfaces, Int. J. Biol. Macromol., 2024, 266, 1–13, DOI:10.1016/j.ijbiomac.2024.131110.
- G. R. Chavhan and L. N. Wankhade, Improvement of the Mechanical Properties of Hybrid Composites Prepared by Fibers, Fiber-Metals, and Nano-Filler Particles – A Review, Mater. Today Proc., 2020, 27, 72–82, DOI:10.1016/j.matpr.2019.08.240.
- Y. Si, J. Li, B. Cui, D. Tang, L. Yang, V. Murugadoss, S. Maganti, M. Huang and Z. Guo, Janus Phenol–Formaldehyde Resin and Periodic Mesoporous Organic Silica Nanoadsorbent for the Removal of Heavy Metal Ions and Organic Dyes from Polluted Water, Adv. Compos. Hybrid Mater., 2022, 5(2), 1180–1195, DOI:10.1007/s42114-022-00446-x.
- J. Nikić, M. Watson, A. Tubić, M. Šolić and J. Agbaba, Recent Trends in the Application of Magnetic Nanocomposites for Heavy Metals Removal from Water: A Review, Sep. Sci. Technol., 2024, 59(2), 293–331, DOI:10.1080/01496395.2024.2315626.
- R. Zhao, B. Wang, P. Wu, Q. Feng, M. Chen, X. Zhang and S. Wang, Calcium Alginate-NZVI-Biochar for Removal of Pb/Zn/Cd in Water: Insights into Governing Mechanisms and Performance, Sci. Total Environ., 2023, 894, 1–16, DOI:10.1016/j.scitotenv.2023.164810.
- M.-L. Liu, C.-X. Zhang, M.-J. Tang, S.-P. Sun, W. Xing and Y. M. Lee, Evolution of Functional Nanochannel Membranes, Prog. Mater. Sci., 2023, 139, 1–28, DOI:10.1016/j.pmatsci.2023.101162.
- Y. Shi, Q. Chang, T. Zhang, G. Song, Y. Sun and G. Ding, A Review on Selective Dye Adsorption by Different Mechanisms, J. Environ. Chem. Eng., 2022, 10(6), 1–33, DOI:10.1016/j.jece.2022.108639.
- X. Niu, R. Zhao, S. Yan, Z. Pang, H. Li, X. Yang and K. Wang, Chiral Materials: Progress, Applications, and Prospects, Small, 2023, 19(38), 1–50, DOI:10.1002/smll.202303059.
- T. Zhou, G. Che, L. Ding, D. Sun and Y. Li, Recent Progress of Selective Adsorbents: From Preparation to Complex Sample Pretreatment, TrAC, Trends Anal. Chem., 2019, 121, 1–19, DOI:10.1016/j.trac.2019.115678.
- K. Zhang, Y. Wang, L. Li and L. Jia, Fabrication of Alginate-Based Nanofibers Loaded with ZnO Nanoparticles for Adsorption of Tetracyclines from Environmental Waters, Mater. Today Commun., 2023, 34, 1–20, DOI:10.1016/j.mtcomm.2022.105214.
- M. Esmaeili Bidhendi, E. Parandi, M. Mahmoudi Meymand, H. Sereshti, H. Rashidi Nodeh, S.-W. Joo, Y. Vasseghian, N. Mahmoudi Khatir and S. Rezania, Removal of Lead Ions from Wastewater Using Magnesium Sulfide Nanoparticles Caged Alginate Microbeads, Environ. Res., 2023, 216, 1–11, DOI:10.1016/j.envres.2022.114416.
- W. Mao, L. Zhang, Y. Zhang, Y. Wang, N. Wen and Y. Guan, Adsorption and Photocatalysis Removal of Arsenite, Arsenate, and Hexavalent Chromium in Water by the Carbonized Composite of Manganese-Crosslinked Sodium Alginate, Chemosphere, 2022, 292, 1–12, DOI:10.1016/j.chemosphere.2021.133391.
- Q. Wang, L. Li, Y. Tian, L. Kong, G. Cai, H. Zhang, J. Zhang, W. Zuo and B. Wen, Shapeable Amino-Functionalized Sodium Alginate Aerogel for High-Performance Adsorption of Cr(VI) and Cd(II): Experimental and Theoretical Investigations, Chem. Eng. J., 2022, 446, 1–11, DOI:10.1016/j.cej.2022.137430.
- H. Ren, Z. Gao, D. Wu, J. Jiang, Y. Sun and C. Luo, Efficient Pb(II) Removal Using Sodium Alginate–Carboxymethyl Cellulose Gel Beads: Preparation, Characterization, and Adsorption Mechanism, Carbohydr. Polym., 2016, 137, 402–409, DOI:10.1016/j.carbpol.2015.11.002.
- M. Tally and Y. Atassi, Synthesis and Characterization of PH-Sensitive Superabsorbent Hydrogels Based on Sodium Alginate-g-Poly(Acrylic Acid-Co-Acrylamide) Obtained via an Anionic Surfactant Micelle Templating under Microwave Irradiation, Polym. Bull., 2016, 73(11), 3183–3208, DOI:10.1007/s00289-016-1649-8.
- M. M. Lakouraj, F. Mojerlou and E. N. Zare, Nanogel and Superparamagnetic Nanocomposite Based on Sodium Alginate for Sorption of Heavy Metal Ions, Carbohydr. Polym., 2014, 106, 34–41, DOI:10.1016/j.carbpol.2014.01.092.
- Ş. Parlayıcı and Y. Baran, Fruit Peel Incorporated Alginate Based Magnetic Hydrogel Bio-Composite Beads for Removal of Hexavalent Chromium, Int. J. Biol. Macromol., 2025, 284, 1–23, DOI:10.1016/j.ijbiomac.2024.137946.
- B. Hu, B. Zhang, W. Xie, X. Jiang, J. Liu and Q. Lu, Recent Progress in Quantum Chemistry Modeling on the Pyrolysis Mechanisms of Lignocellulosic Biomass, Energy Fuels, 2020, 34(9), 10384–10440, DOI:10.1021/acs.energyfuels.0c01948.
- M. Irimia-Vladu and N. S. Sariciftci, Natural Polymers for Emerging Technological Applications: Cellulose, Lignin, Shellac and Silk, Polym. Int., 2025, 74(2), 71–86, DOI:10.1002/pi.6697.
- A. Farrán, C. Cai, M. Sandoval, Y. Xu, J. Liu, M. J. Hernáiz and R. J. Linhardt, Green Solvents in Carbohydrate Chemistry: From Raw Materials to Fine Chemicals, Chem. Rev., 2015, 115(14), 6811–6853, DOI:10.1021/cr500719h.
- E. Ojogbo, E. O. Ogunsona and T. H. Mekonnen, Chemical and Physical Modifications of Starch for Renewable Polymeric Materials, Mater. Today Sustain., 2020, 7–8, 1–25, DOI:10.1016/j.mtsust.2019.100028.
- Q. Zhang, E. Zhu, T. Li, L. Zhang and Z. Wang, High-Value Utilization of Cellulose: Intriguing and Important Effects of Hydrogen Bonding Interactions – A Mini-Review, Biomacromolecules, 2024, 25(10), 6296–6318, DOI:10.1021/acs.biomac.4c00823.
- T. Aziz, W. Li, J. Zhu and B. Chen, Developing Multifunctional Cellulose Derivatives for Environmental and Biomedical Applications: Insights into Modification Processes and Advanced Material Properties, Int. J. Biol. Macromol., 2024, 278, 1–32, DOI:10.1016/j.ijbiomac.2024.134695.
- Y. Pei, L. Wang, K. Tang and D. L. Kaplan, Biopolymer Nanoscale Assemblies as Building Blocks for New Materials: A Review, Adv. Funct. Mater., 2021, 31(15), 1–30, DOI:10.1002/adfm.202008552.
- J. Zhao, Z. Wei, L. Sun, Y. Wang, X. Wu, T. Wang, Z. Wang and Y. Fu, A Novel Cellulose-Based Composite Hydrogel Microsphere Material: For Efficient Adsorption of Co(II) and Ni(II) Ions in Water, J. Inorg. Organomet. Polym. Mater., 2025, 35(2), 898–918, DOI:10.1007/s10904-024-03323-w.
- Y. Wang, Q. Chen, Y. Lei, M. G. A. Kaya, K. L. Goh and K. Tang, Identification, Deterioration, and Protection of Organic Cultural Heritages from a Modern Perspective, npj Heritage Sci., 2025, 13(1), 1–19, DOI:10.1038/s40494-025-01601-5.
- S. Kumari and G. S. Chauhan, New Cellulose–Lysine Schiff-Base-Based Sensor–Adsorbent for Mercury Ions, ACS Appl. Mater. Interfaces, 2014, 6(8), 5908–5917, DOI:10.1021/am500820n.
- S. Tiwari, P. Boylla, Y. Atescan-Yuksek, I. J. Badruddin, A. O. Orisawayi, P. D. Venkatraman, K. Salonitis and S. S. Rahatekar, Natural Dyes and Regenerated Cellulose Fibers Blending Using Ionic Liquid as a Common Platform for Sustainable Textiles/Fashion Applications, Carbohydr. Polym. Technol. Appl., 2025, 12, 1–16, DOI:10.1016/j.carpta.2025.101027.
- A. S. Abdul Rahman, A. N. S. Fizal, N. A. Khalil, A. N. Ahmad Yahaya, M. S. Hossain and M. Zulkifli, Fabrication and Characterization of Magnetic Cellulose–Chitosan–Alginate Composite Hydrogel Bead Bio-Sorbent, Polymers, 2023, 15(11), 1–14, DOI:10.3390/polym15112494.
- W. Al-Gethami, M. A. Qamar, M. Shariq, A.-N. M. A. Alaghaz, A. Farhan, A. A. Areshi and M. H. Alnasir, Emerging Environmentally Friendly Bio-Based Nanocomposites for the Efficient Removal of Dyes and Micropollutants from Wastewater by Adsorption: A Comprehensive Review, RSC Adv., 2024, 14(4), 2804–2834, 10.1039/D3RA06501D.
- W. Zhang, J. Ou, B. Wang, H. Wang, Q. He, J. Song, H. Zhang, M. Tang, L. Zhou, Y. Gao and S. Sun, Efficient Heavy Metal Removal from Water by Alginate-Based Porous Nanocomposite Hydrogels: The Enhanced Removal Mechanism and Influencing Factor Insight, J. Hazard. Mater., 2021, 418, 1–12, DOI:10.1016/j.jhazmat.2021.126358.
- H. Zhao, J. Sun, Y. Du, M. Zhang, Z. Yang, J. Su, X. Peng, X. Liu, G. Sun and Y. Cui, In-Situ Immobilization of CuMOF on Sodium Alginate/Chitosan/Cellulose Nanofibril Composite Hydrogel for Fast and Highly Efficient Removal of Pb2+ from Aqueous Solutions, J. Solid State Chem., 2023, 322, 1–13, DOI:10.1016/j.jssc.2023.123928.
- Y. Chen, X. Liu, R. Zhou, J. Qiao, J. Liu, R. Cai, J. Liu, J. Rong and Y. Chen, Porous Sodium Alginate/Cellulose Nanofiber Composite Hydrogel Microspheres for Heavy Metal Removal in Wastewater, Int. J. Biol. Macromol., 2024, 278, 1–11, DOI:10.1016/j.ijbiomac.2024.135000.
- X. He, H. Jia, N. Sun, M. Hou, Z. Tan and X. Lu, Fluorescent Hydrogels Based on Oxidized Carboxymethyl Cellulose with Excellent Adsorption and Sensing Abilities for Ag+, Int. J. Biol. Macromol., 2022, 213, 955–966, DOI:10.1016/j.ijbiomac.2022.06.029.
- Y. Ma, B. Nasri-Nasrabadi, X. You, X. Wang, T. J. Rainey and N. Byrne, Regenerated Cellulose Fibers Wetspun from Different Waste Cellulose Types, J. Nat. Fibers, 2021, 18(12), 2338–2350, DOI:10.1080/15440478.2020.1726244.
- J. Wei, Y. Long, B. Wang, H. Wu, H. Gao and Y. Nie, Structure and Properties Variations of Regenerated Cellulose Fibers Induced by Metal Ion Impurity, Int. J. Biol. Macromol., 2024, 255, 1–10, DOI:10.1016/j.ijbiomac.2023.128124.
- I. O. Saheed, S. O. Azeez and F. B. M. Suah, Imidazolium Based Ionic Liquids Modified Polysaccharides for Adsorption and Solid-Phase Extraction Applications: A Review, Carbohydr. Polym., 2022, 298, 1–17, DOI:10.1016/j.carbpol.2022.120138.
- X. Zhang, M. Liu, C. Zhang, Z. Yuan and H. Chi, Real-Time Uranyl Ion Adsorption Monitoring Based on Cellulose Hydrogels, ACS Appl. Polym. Mater., 2024, 6(21), 13193–13201, DOI:10.1021/acsapm.4c02411.
- Z. Zhang, Y. Lu, S. Gao and S. Wu, Sustainable and Efficient Wastewater Treatment Using Cellulose-Based Hydrogels: A Review of Heavy Metal, Dye, and Micropollutant Removal Applications, Separations, 2025, 12(3), 1–50, DOI:10.3390/separations12030072.
- H. G. Castellanos, Y. Aryanfar, S. Mohtaram, A. Keçebaş, G. Karaca-Dolgun, S. Ahmad, A. N. M. Asiri and S. Islam, The Efficacy of Nano-cellulose-based Composites in Heavy Metal Removal from Wastewater: A Comprehensive Review, J. Chem. Technol. Biotechnol., 2024, 1–12, DOI:10.1002/jctb.7775.
- Z. Zhang, Y. Lu, Y. Zhao, L. Cui, C. Xu and S. Wu, Current Developments in Chitosan-Based Hydrogels for Water and Wastewater Treatment: A Comprehensive Review, ChemistrySelect, 2025, 10(6), 1–39, DOI:10.1002/slct.202404061.
- A. Shahzad, M. W. Ullah, J. Ali, K. Aziz, M. A. Javed, Z. Shi, S. Manan, M. Ul-Islam, M. Nazar and G. Yang, The Versatility of Nanocellulose, Modification Strategies, and Its Current Progress in Wastewater Treatment and Environmental Remediation, Sci. Total Environ., 2023, 858, 1–22, DOI:10.1016/j.scitotenv.2022.159937.
- R. Zeng, J. Zheng, Y. Zuo, C. Xiao and Y. Zhu, Synergistic and Simultaneous Removal of Heavy Metal Ions over Waste Bamboo Shoot Particles Encapsulated Carboxymethyl Cellulose/Gelatin Composite Hydrogel, Int. J. Biol. Macromol., 2024, 283, 1–13, DOI:10.1016/j.ijbiomac.2024.137578.
- K. Kalaiselvi, S. Mohandoss, N. Ahmad, M. R. Khan and R. K. Manoharan, Adsorption of Pb2+ Ions from Aqueous Solution onto Porous Kappa-Carrageenan/Cellulose Hydrogels: Isotherm and Kinetics Study, Sustainability, 2023, 15(12), 1–15, DOI:10.3390/su15129534.
- P. Li, M. Zhou, H. Liu, H. Lei, B. Jian, R. Liu, X. Li, Y. Wang and B. Zhou, Preparation of Green Magnetic Hydrogel from Soybean Residue Cellulose for Effective and Rapid Removal of Copper Ions from Wastewater, J. Environ. Chem. Eng., 2022, 10(5), 1–10, DOI:10.1016/j.jece.2022.108213.
- J. Ding, Q. Li, X. Xu, X. Zhang, Y. Su, Q. Yue and B. Gao, A Wheat Straw Cellulose-Based Hydrogel for Cu (II) Removal and Preparation Copper Nanocomposite for Reductive Degradation of Chloramphenicol, Carbohydr. Polym., 2018, 190, 12–22, DOI:10.1016/j.carbpol.2018.02.032.
- S.-S. Li, Y.-L. Song, H.-R. Yang, Q.-D. An, Z.-Y. Xiao and S.-R. Zhai, Carboxymethyl Cellulose-Based Cryogels for Efficient Heavy Metal Capture: Aluminum-Mediated Assembly Process and Sorption Mechanism, Int. J. Biol. Macromol., 2020, 164, 3275–3286, DOI:10.1016/j.ijbiomac.2020.08.186.
- T. Hajeeth, P. N. Sudha, K. Vijayalakshmi and T. Gomathi, Sorption Studies on Cr (VI) Removal from Aqueous Solution Using Cellulose Grafted with Acrylonitrile Monomer, Int. J. Biol. Macromol., 2014, 66, 295–301, DOI:10.1016/j.ijbiomac.2014.02.027.
- J. Wang, L. Wei, Y. Ma, K. Li, M. Li, Y. Yu, L. Wang and H. Qiu, Collagen/Cellulose Hydrogel Beads Reconstituted from Ionic Liquid Solution for Cu(II) Adsorption, Carbohydr. Polym., 2013, 98(1), 736–743, DOI:10.1016/j.carbpol.2013.06.001.
- L. V. A. Gurgel, O. Karnitz Júnior, R. P. d. F. Gil and L. F. Gil, Adsorption of Cu(II), Cd(II), and Pb(II) from Aqueous Single, Bioresour. Technol., 2008, 99, 3077–3083 CrossRef CAS.
- S. Hokkanen, E. Repo, T. Suopajärvi, H. Liimatainen, J. Niinimaa and M. Sillanpää, Adsorption of Ni(II), Cu(II) and Cd(II) from Aqueous Solutions by Amino Modified Nanostructured Microfibrillated Cellulose, Cellulose, 2014, 21(3), 1471–1487, DOI:10.1007/s10570-014-0240-4.
- H. H. Sokker, Y. H. Gad and S. A. Ismail, Synthesis of Bifunctional Cellulosic Adsorbent by Radiation Induced Graft Polymerization of Glycidyl Methacrylate- Co -methacrylic Acids, J. Appl. Polym. Sci., 2012, 126(S1), 1–9, DOI:10.1002/app.34220.
- Y. Tang, Q. Ma, Y. Luo, L. Zhai, Y. Che and F. Meng, Improved Synthesis of a Branched Poly(Ethylene Imine)-modified Cellulose-based Adsorbent for Removal and Recovery of Cu(II) from Aqueous Solution, J. Appl. Polym. Sci., 2013, 129(4), 1799–1805, DOI:10.1002/app.38878.
- J. Chen, X. Wang, X. Huang, Z. Tong, J. Zhou, Y. Shen and C. Hao, Construction of Cellulose-Based Hydrogel Compounded with Modified Kaolin and Its Removal Performance for Heavy Metal Ions and Dyes in Water, Int. J. Biol. Macromol., 2025, 306, 1–12, DOI:10.1016/j.ijbiomac.2025.141398.
- L. Zhang, M. Qiao, H. Zheng, T. Vancov, V. Antoniadis, S. M. Shaheen, S. Joseph, C. Chen, S. Shan, H. Chen and H. Wang, Integrating Spectroscopic Analysis and Theoretical Calculations to Elucidate the Adsorption Efficiency and Mechanisms of Cd, Pb, and Cu Using Novel Carboxymethyl Cellulose/Pectin-Based Hydrogel Beads, Int. J. Biol. Macromol., 2025, 305, 1–12, DOI:10.1016/j.ijbiomac.2025.141028.
- J. P. F. Carvalho, N. S. Lameirinhas, M. C. Teixeira, J. L. Luís, H. Oliveira, J. M. Oliveira, A. J. D. Silvestre, C. Vilela and C. S. R. Freire, All-Cellulose Hydrogel-Based Bioinks for the Versatile 3D Bioprinting of Different Cell Lines, Biomacromolecules, 2025, 26(3), 1761–1770, DOI:10.1021/acs.biomac.4c01546.
- A. P. Bonto, J. P. Bantang, M. Sucaldito, M. O. S. Lobregas, F. M. dela Rosa, F. Wang and C. Delattre, Polysaccharide-Based Water Purifying Materials, in Biopolymers for Water Purification, Wiley, 2025, pp. 371–420, DOI:10.1002/9783527835904.ch12.
- J. Sangkaworn, W. Limprasart, M. V. Höfler, T. Gutmann, S. Pornsuwan, T. Bunchuay and J. Tantirungrotechai, Copper-Supported Thiol-Functionalized Cellulose as a Paper-Based Catalyst for Imine Synthesis, Sci. Rep., 2025, 15(1), 1–11, DOI:10.1038/s41598-025-95144-1.
- N. Mahfoudhi and S. Boufi, Nanocellulose as a Novel Nanostructured Adsorbent for Environmental Remediation: A Review, Cellulose, 2017, 24(3), 1171–1197, DOI:10.1007/s10570-017-1194-0.
- H. Jiang, S. Wu and J. Zhou, Preparation and Modification of Nanocellulose and Its Application to Heavy Metal Adsorption: A Review, Int. J. Biol. Macromol., 2023, 236, 1–19, DOI:10.1016/j.ijbiomac.2023.123916.
- A. El Mahdaoui, S. Radi, A. Elidrissi, M. A. F. Faustino, M. G. P. M. S. Neves and N. M. M. Moura, Progress in the Modification of Cellulose-Based Adsorbents for the Removal of Toxic Heavy Metal Ions, J. Environ. Chem. Eng., 2024, 12(5), 1–35, DOI:10.1016/j.jece.2024.113870.
- S. Hao, S. Shen, A. O. Orisawayi, S. Tiwari, I. J. Badruddin, K. Koziol and S. S. Rahatekar, Pioneering Microsphere-Dope Dyeing for Sustainable Cellulosic Fibre Colouring, Int. J. Biol. Macromol., 2025, 320, 1–14, DOI:10.1016/j.ijbiomac.2025.146007.
- Y. Liao, C. H. Loh, M. Tian, R. Wang and A. G. Fane, Progress in Electrospun Polymeric Nanofibrous Membranes for Water Treatment: Fabrication, Modification and Applications, Prog. Polym. Sci., 2018, 77, 69–94, DOI:10.1016/j.progpolymsci.2017.10.003.
- M. F. Amin, T. Ariwibowo, S. A. Putri and D. Kurnia, Moringa Oleifera: A Review of the Pharmacology, Chemical Constituents, and Application for Dental Health, Pharmaceuticals, 2024, 17(1), 100–122, DOI:10.3390/ph17010142.
- V. Babu, Y. B. C. Basha, S. Srinivasan, S. B. S. Sadik and A. K. Pandurangan, A Comprehensive Review on the Phytochemical and Pharmacological Benefits of Moringa Oleifera: An Update, Curr. Pharmacol. Rep., 2024, 11(1), 1–13, DOI:10.1007/s40495-024-00383-x.
- L. F. Cusioli, H. B. Quesada, A. L. de Brito Portela Castro, R. G. Gomes and R. Bergamasco, Development of a New Low-Cost Adsorbent Functionalized with Iron Nanoparticles for Removal of Metformin from Contaminated Water, Chemosphere, 2020, 247, 1–4, DOI:10.1016/j.chemosphere.2020.125852.
- M. Imran, K. Anwar, M. Akram, G. M. Shah, I. Ahmad, N. Samad Shah, Z. U. H. Khan, M. I. Rashid, M. N. Akhtar, S. Ahmad, M. Nawaz and R. J. Schotting, Biosorption of Pb(II) from Contaminated Water onto Moringa Oleifera Biomass: Kinetics and Equilibrium Studies, Int. J. Phytorem., 2019, 21(8), 777–789, DOI:10.1080/15226514.2019.1566880.
- S. Vigneshwaran, P. Karthikeyan, P. Sirajudheen and S. Meenakshi, Optimization of Sustainable Chitosan/Moringa. Oleifera as Coagulant Aid for the Treatment of Synthetic Turbid Water – A Systemic Study, Environ. Chem. Ecotoxicol., 2020, 2, 132–140, DOI:10.1016/j.enceco.2020.08.002.
- K. Ravikumar and A. K. Sheeja, Heavy Metal Removal from Water Using Moringa Oleifera Seed Coagulant and Double Filtration, 2013, pp. 9–12 Search PubMed.
- K. Ravikumar and J. Udayakumar, Preparation and Characterisation of Green Clay-Polymer Nanocomposite for Heavy Metals Removal, Chem. Ecol., 2020, 36(3), 270–291, DOI:10.1080/02757540.2020.1723559.
- V. U. Nwagbara, K. Chigayo, W. A. Iyama and H. M. Kwaambwa, Removal of Lead, Cadmium, and Copper from Water Using Moringa Oleifera Seed Biomass, J. Water Clim. Change, 2022, 13(7), 2747–2760, DOI:10.2166/wcc.2022.091.
- A. Benettayeb, E. Guibal, A. Morsli and R. Kessas, Chemical Modification of Alginate for Enhanced Sorption of Cd(II), Cu(II) and Pb(II), Chem. Eng. J., 2017, 316, 704–714, DOI:10.1016/j.cej.2017.01.131.
- A. J. Varkey, Purification of River Water Using Moringa Oleifera Seed and Copper for Point-of-Use Household Application, Sci. Afr., 2020, 8, 1–8, DOI:10.1016/j.sciaf.2020.e00364.
- R. Ngulube, N. Nombona and L. Pillay, Sustainable Pb(II) Remediation: Efficacy and Selectivity of Moringa Oleifera Composite Nanofibers, Discov. Appl. Sci., 2025, 7(10), 1–15, DOI:10.1007/s42452-025-06931-4.
- L. A. de Moraes Pinto, F. de Oliveira Tavares, R. Bergamasco, M. F. Vieira and A. M. S. Vieira, Biosorption of Manganese Using Moringa Oleifera Seed Pods: A Sustainable Approach to Water Treatment, Separations, 2025, 12(9), 1–22, DOI:10.3390/separations12090246.
- H. F. AbdEl-Halim and M. S. Afifi, Wastewater Treatment Using Moringa Oleifera (Lam.) and Eichhornia Crassipes (Mart.) as Neutral-Carbon Options within the Framework of COP 27 Recommendations, Environ. Sci. Pollut. Res., 2024, 32(2), 514–532, DOI:10.1007/s11356-024-35659-8.
- C. Teng, X. Jing, H. Zhang, W. Chen, C. Zhou, Z. Wang and Z. Xu, Spectroscopic Investigation of the Binding Behavior of Cd2+, Cu2+, Cr3+ and Fe3+ with Humic Acid of Varying Molecular Weights, J. Water Proc. Eng., 2025, 71, 1–10, DOI:10.1016/j.jwpe.2025.107434.
- W. Liu, T. Wang, A. G. L. Borthwick, Y. Wang, X. Yin, X. Li and J. Ni, Adsorption of Pb2+, Cd2+, Cu2+ and Cr3+ onto Titanate Nanotubes: Competition and Effect of Inorganic Ions, Sci. Total Environ., 2013, 456–457, 171–180, DOI:10.1016/j.scitotenv.2013.03.082.
- T. T. Li, Y. Zhong, M. Yan, W. Zhou, W. Xu, S. Y. Huang, F. Sun, C. W. Lou and J. H. Lin, Synergistic Effect and Characterization of Graphene/Carbon Nanotubes/Polyvinyl Alcohol/Sodium Alginate Nanofibrous Membranes Formed Using Continuous Needleless Dynamic Linear Electrospinning, Nanomaterials, 2019, 9(5), 1–13, DOI:10.3390/NANO9050714.
- H. R. Rafiei, M. Shirvani and O. A. Ogunseitan, Removal of Lead from Aqueous Solutions by a Poly(Acrylic Acid)/Bentonite Nanocomposite, Appl. Water Sci., 2016, 6(4), 331–338, DOI:10.1007/s13201-014-0228-0.
- D. H. K. Reddy, S.-M. Lee and K. Seshaiah, Removal of Cd(II) and Cu(II) from Aqueous Solution by Agro Biomass: Equilibrium, Kinetic and Thermodynamic Studies, Environ. Eng. Res., 2012, 17(3), 125–132, DOI:10.4491/eer.2012.17.3.125.
- A. Benettayeb, M. Usman, C. C. Tinashe, T. Adam and B. Haddou, A Critical Review with Emphasis on Recent Pieces of Evidence of Moringa Oleifera Biosorption in Water and Wastewater Treatment, Environ. Sci. Pollut. Res., 2022, 29(32), 48185–48209, DOI:10.1007/s11356-022-19938-w.
- C. K. O. Dzuvor, S. Pan, C. Amanze, P. Amuzu, C. Asakiya and F. Kubi, Bioactive Components from Moringa Oleifera Seeds: Production,Functionalities and Applications – a Critical Review, Crit. Rev. Biotechnol., 2022, 42(2), 271–293, DOI:10.1080/07388551.2021.1931804.
- F. O. Adetuyi, E. S. Akintimehin, K. O. Karigidi and A. O. Orisawayi, Safety Evaluation of Fermented and Nonfermented Moringa Oleifera Seeds in Healthy Albino Rats: Biochemical, Haematological, and Histological Studies, Int. J. Food Sci., 2025, 2025(1), 1–12, DOI:10.1155/ijfo/2694100.
- M. D. Ortolá, S. Pageo, F. J. García-Mares, M. Juan-Borrás and M. L. Castelló, Characterization of Partially Defatted Moringa Seed Flour Obtained at Different Temperatures, LWT, 2024, 198, 1–8, DOI:10.1016/j.lwt.2024.115901.
- M. M. Tosif, A. Najda, A. Bains, R. Kaushik, S. B. Dhull, P. Chawla and M. Walasek-Janusz, A Comprehensive Review on Plant-Derived Mucilage: Characterization, Functional Properties, Applications, and Its Utilization for Nanocarrier Fabrication, Polymers, 2021, 13(7), 1–24, DOI:10.3390/polym13071066.
- R. Srinivasan, Natural Polysaccharides as Treatment Agents for Wastewater, in Green Materials for Sustainable Water Remediation and Treatment, The Royal Society of Chemistry, 2013, pp. 51–81, 10.1039/9781849735001-00051.
- N. Kumari and S. Mishra, Synthesis, Characterization and Flocculation Efficiency of Grafted Moringa Gum Based Derivatives, Carbohydr. Polym., 2022, 281, 1–14, DOI:10.1016/j.carbpol.2021.119079.
- H. R. Badwaik, A. A. Hoque, L. Kumari, K. Sakure, M. Baghel and T. K. Giri, Moringa Gum and Its Modified Form as a Potential Green Polymer Used in Biomedical Field, Carbohydr. Polym., 2020, 249, 1–13, DOI:10.1016/j.carbpol.2020.116893.
- S. Mehta, P. Joshi, R. N. Goswami, O. P. Sharma and O. P. Khatri, Adsorptive Separation and Simultaneous Reduction of Highly Toxic Chromium Oxyanions by Agroforestry Biomass-Derived N-Rich Activated Carbon, Ind. Eng. Chem. Res., 2025, 64(3), 1555–1566, DOI:10.1021/acs.iecr.4c02792.
- J. L. Aleman-Ramirez, P. U. Okoye, S. Saldaña-Trinidad, S. Torres-Arellano and P. J. Sebastian, The Role of Moringa Oleifera in the Development of Alternative Biofuels, under the Concept of an Integral One-tree Biorefinery: A Minireview, Biofuels, Bioprod. Biorefin., 2025, 1–21, DOI:10.1002/bbb.2738.
- A. Fakhar, S. J. C. Galgo, R. C. Canatoy, M. Rafique, R. Sarfraz, A. A. Farooque and M. I. Khan, Advancing Modified Biochar for Sustainable Agriculture: A Comprehensive Review on Characterization, Analysis, and Soil Performance, Biochar, 2025, 7(1), 1–25, DOI:10.1007/s42773-024-00397-0.
- R. Zou, M. Qian, C. Wang, W. Mateo, Y. Wang, L. Dai, X. Lin, Y. Zhao, E. Huo, L. Wang, X. Zhang, X. Kong, R. Ruan and H. Lei, Biochar: From by-Products of Agro-Industrial Lignocellulosic Waste to Tailored Carbon-Based Catalysts for Biomass Thermochemical Conversions, Chem. Eng. J., 2022, 441, 2–17, DOI:10.1016/j.cej.2022.135972.
- K. A. Abdalkarim, D. I. Tofiq, R. F. Hamarawf, H. Q. Hassan, B. S. Ahmad, D. S. Muhammad and S. B. Aziz, MOF/Chitosan Composites: An Emerging Class of Multifunctional Materials for Diverse Applications, J. Inorg. Organomet. Polym. Mater., 2025, 1–53, DOI:10.1007/s10904-024-03572-9.
- K. Mishra and S. Sinha, Biodegradable Green Composite Film Developed from Moringa Oleifera (Sahajana) Seed Filler and PVA: Surface Functionalization, Characterization and Barrier Properties, J. Thermoplast. Compos. Mater., 2023, 36(1), 345–371, DOI:10.1177/08927057211007550.
- H. El-Gendi, J. S. Albrahim, H. Alenezi, E. M. El-Fakharany, Y. A. El-Maradny and A. K. Saleh, Bioactive Bacterial Cellulose/Chitosan/Sodium Alginate Composite Film Functionalized with Moringa Oleifera Seed Extract: Antimicrobial, Anticancer, and Molecular Docking Studies, Int. J. Biol. Macromol., 2025, 307, 1–19, DOI:10.1016/j.ijbiomac.2025.141958.
- V. O. Arief, K. Trilestari, J. Sunarso, N. Indraswati and S. Ismadji, Recent Progress on Biosorption of Heavy Metals from Liquids Using Low Cost Biosorbents: Characterization, Biosorption Parameters and Mechanism Studies, Clean, 2008, 36(12), 937–962, DOI:10.1002/clen.200800167.
- M. A. Butt, S. R. Ahmad, M. N. Chaudhary, M. Zaheer, R. Nazir, M. Zia-ur-rehman and N. Hussain, Environment Friendly Synthesis of Novel Schiff Base-Derived Nano Metal Complexes Using Green Solvents for Enhanced Biological Activity, Pol. J. Environ. Stud., 2025, 34(3), 2023–2035, DOI:10.15244/pjoes/188043.
- E. Torres, Biosorption: A Review of the Latest Advances, Processes, 2020, 8(12), 1–23, DOI:10.3390/pr8121584.
- W. Huang and Z. Liu, Biosorption of Cd(II)/Pb(II) from Aqueous Solution by Biosurfactant-Producing Bacteria: Isotherm Kinetic Characteristic and Mechanism Studies, Colloids Surf., B, 2013, 105, 113–119, DOI:10.1016/j.colsurfb.2012.12.040.
- F. Adekola, H. Adegoke, O. Arowosaiye and G. Olatunji, Kinetic and Thermodynamic Studies of Sorption of Lead and Cadmium from Aqueous Solution by Moringa Oleifera Pod Wastes, Int. J. Environ. Waste Manage., 2020, 25(1), 58–82, DOI:10.1504/IJEWM.2020.104347.
- A. K. Gautam, Markandeya, N. B. Singh, S. P. Shukla and D. Mohan, Lead Removal Efficiency of Various Natural Adsorbents (Moringa Oleifera, Prosopis Juliflora, Peanut Shell) from Textile Wastewater, SN Appl. Sci., 2020, 2(2), 1–11, DOI:10.1007/s42452-020-2065-0.
- D. Masekela, N. C. Hintsho-Mbita and N. Mabuba, Diethylamine Functionalised Moringa Oleifera Leaves for Theremoval of Chromium(VI) and Bacteria from Wastewater, Int. J. Environ. Anal. Chem., 2022, 102(13), 3002–3022, DOI:10.1080/03067319.2020.1762873.
- A.-E. Swelam, S. Saied and A. Hafez, Removal Comparative Study for Cd(II) Ions from Polluted Solutions by Adsorption and Coagulation Techniques Using Moringa Oleifera, Egypt. J. Chem., 2019, 62(8), 1499–1517, DOI:10.21608/ejchem.2019.6801.1568.
- S. Ranote, B. Ram, D. Kumar, G. S. Chauhan and V. Joshi, Functionalization of Moringa Oleifera Gum for Use as Hg2+ Ions Adsorbent, J. Environ. Chem. Eng., 2018, 6(2), 1805–1813, DOI:10.1016/j.jece.2018.02.032.
- Z. Shirani, C. Santhosh, J. Iqbal and A. Bhatnagar, Waste Moringa Oleifera Seed Pods as Green Sorbent for Efficient Removal of Toxic Aquatic Pollutants, J. Environ. Manage., 2018, 227, 95–106, DOI:10.1016/j.jenvman.2018.08.077.
- B. Garcia-Fayos, J. M. Arnal, J. Piris and M. Sancho, Valorization of Moringa Oleifera Seed Husk as Biosorbent: Isotherm and Kinetics Studies to Remove Cadmium and Copper from Aqueous Solutions, Desalination Water Treat., 2016, 57(48–49), 23382–23396, DOI:10.1080/19443994.2016.1180473.
- A. P. Meneghel, A. C. Gonçalves, C. R. T. Tarley, J. R. Stangarlin, F. Rubio and H. Nacke, Studies of Pb2+ Adsorption by Moringa Oleifera Lam. Seeds from an Aqueous Medium in a Batch System, Water Sci. Technol., 2014, 69(1), 163–169, DOI:10.2166/wst.2013.627.
- T. Sumathi and G. Alagumuthu, Adsorption Studies for Arsenic Removal Using Activated Moringa Oleifera, Int. J. Chem. Eng., 2014, 2014, 1–6, DOI:10.1155/2014/430417.
- A. P. Meneghel, A. C. Gonçalves, F. Rubio, D. C. Dragunski, C. A. Lindino and L. Strey, Biosorption of Cadmium from Water Using Moringa (Moringa Oleifera Lam.) Seeds, Water, Air, Soil Pollut., 2013, 224(3), 1–13, DOI:10.1007/s11270-012-1383-2.
- A. P. Meneghel, A. C. Gonçalves Jr, L. Strey, F. Rubio, D. Schwantes and J. Casarin, Biosorption and Removal of Chromium from Water by Using Moringa Seed Cake (Moringa Oleifera Lam.), Quim. Nova, 2013, 36(8), 1104–1110, DOI:10.1590/S0100-40422013000800005.
- D. H. K. Reddy, K. Seshaiah, A. V. R. Reddy and S. M. Lee, Optimization of Cd(II), Cu(II) and Ni(II) Biosorption by Chemically Modified Moringa Oleifera Leaves Powder, Carbohydr. Polym., 2012, 88(3), 1077–1086, DOI:10.1016/j.carbpol.2012.01.073.
- D. H. K. Reddy, D. K. V. Ramana, K. Seshaiah and A. V. R. Reddy, Biosorption of Ni(II) from Aqueous Phase by Moringa Oleifera Bark, a Low Cost Biosorbent, Desalination, 2011, 268(1–3), 150–157, DOI:10.1016/j.desal.2010.10.011.
- M. Helen Kalavathy and L. R. Miranda, Moringa Oleifera—A Solid Phase Extractant for the Removal of Copper, Nickel and Zinc from Aqueous Solutions, Chem. Eng. J., 2010, 158(2), 188–199, DOI:10.1016/j.cej.2009.12.039.
- D. H. K. Reddy, K. Seshaiah, A. V. R. Reddy, M. M. Rao and M. C. Wang, Biosorption of Pb2+ from Aqueous Solutions by Moringa Oleifera Bark: Equilibrium and Kinetic Studies, J. Hazard. Mater., 2010, 174(1–3), 831–838, DOI:10.1016/j.jhazmat.2009.09.128.
- K. Mohanrasu, A. C. Manivannan, H. J. R. Rengarajan, R. Kandaiah, A. Ravindran, L. Panneerselvan, T. Palanisami and C. I. Sathish, Eco-Friendly Biopolymers and Composites: A Sustainable Development of Adsorbents for the Removal of Pollutants from Wastewater, npj Mater. Sustain., 2025, 3(1), 1–21, DOI:10.1038/s44296-025-00057-9.
- X. Zhang, Y. Zhu, F. Zhang, Y. Mo, Y. Zhang, W. Fang and J. Jin, Hydrophilic/Hydrophobic Nanofibres Intercalated Multilayer Membrane with Hierarchical Structure for Efficient Oil/Water Separation, Sep. Purif. Technol., 2022, 288, 1–9, DOI:10.1016/j.seppur.2022.120672.
- S. Tang, J. Yang, L. Lin, K. Peng, Y. Chen, S. Jin and W. Yao, Construction of Physically Crosslinked Chitosan/Sodium Alginate/Calcium Ion Double-Network Hydrogel and Its Application to Heavy Metal Ions Removal, Chem. Eng. J., 2020, 393, 1–11, DOI:10.1016/j.cej.2020.124728.
- B. M. Dasari, K. K. Aradhi and D. Banothu, Evaluation of Heavy Metal Contamination and Their Distribution in Waters Around Oil and Natural Gas Drilling Sites, Water, Air, Soil Pollut., 2023, 234(7), 1–10, DOI:10.1007/s11270-023-06426-1.
- I. Hegazy, M. E. A. Ali, E. H. Zaghlool and R. Elsheikh, Heavy Metals Adsorption from Contaminated Water Using Moringa Seeds/Olive Pomace Byproducts, Appl. Water Sci., 2021, 11(6), 1–14, DOI:10.1007/s13201-021-01421-5.
- Y. Ma, B. Nasri-Nasrabadi, X. You, X. Wang, T. J. Rainey and N. Byrne, Regenerated Cellulose Fibers Wetspun from Different Waste Cellulose Types, J. Nat. Fibers, 2021, 18(12), 2338–2350, DOI:10.1080/15440478.2020.1726244.
- N. Mohammed, N. Grishkewich, R. M. Berry and K. C. Tam, Cellulose Nanocrystal–Alginate Hydrogel Beads as Novel Adsorbents for Organic Dyes in Aqueous Solutions, Cellulose, 2015, 22(6), 3725–3738, DOI:10.1007/s10570-015-0747-3.
- A. O. Orisawayi, P. Boylla, K. K. Koziol and S. S. Rahatekar, Sustainable Wet-Spun Cellulose- Moringa Oleifera Composite Fibres for Potential Water Purification, RSC Adv., 2025, 15(22), 17730–17745, 10.1039/D5RA02386F.
- T. Nypelö, S. Asaadi, G. Kneidinger, H. Sixta and J. Konnerth, Conversion of Wood-Biopolymers into Macrofibers with Tunable Surface Energy via Dry-Jet Wet-Spinning, Cellulose, 2018, 25(9), 5297–5307, DOI:10.1007/s10570-018-1902-4.
- A. Ullah and S. Ahmed, Green Biopolymers for Packaging Applications, CRC Press, Boca Raton, 2024, DOI:10.1201/9781003455356.
- R. Ribeiro-Santos, M. Andrade and A. Sanches-Silva, Application of Encapsulated Essential Oils as Antimicrobial Agents in Food Packaging, Curr. Opin. Food Sci., 2017, 14, 78–84, DOI:10.1016/j.cofs.2017.01.012.
- H. Niu, T. Lin and X. Wang, Needleless Electrospinning. I. A Comparison of Cylinder and Disk Nozzles, J. Appl. Polym. Sci., 2009, 114(6), 3524–3530, DOI:10.1002/app.30891.
- J. Zhang, H. Kitayama, Y. Gotoh, A. Potthast and T. Rosenau, Non-Woven Fabrics of Fine Regenerated Cellulose Fibers Prepared from Ionic-Liquid Solution via Wet Type Solution Blow Spinning, Carbohydr. Polym., 2019, 226, 1–8, DOI:10.1016/j.carbpol.2019.115258.
- Y. Liu, L. Chen, W. Li, J. Pu, Z. Wang, B. He, S. Yuan, J. Xin, L. Huang, Z. Luo, J. Xu, X. Zhou, H. Zhang, Q. Zhang and L. Wei, Scalable Production of Functional Fibers with Nanoscale Features for Smart Textiles, ACS Nano, 2024, 18(43), 29394–29420, DOI:10.1021/acsnano.4c10111.
- Z. Xu, D. Chen, X. Duan, Y. Chen, C. Li, S. Li, Y. Ma, B. Huang and X. Pan, Collaboratively Removal of Phosphate and Glyphosate from Wastewater by a Macroscopic Zr-SA/Ce-UIO-66 Adsorbent: Performance, Mechanisms and Applicability, J. Hazard. Mater., 2025, 484, 1–13, DOI:10.1016/j.jhazmat.2024.136786.
- R. Salas, R. Villa, F. Velasco, F. G. Cirujano, S. Nieto, N. Martin, E. Garcia-Verdugo, J. Dupont and P. Lozano, Ionic Liquids in Polymer Technology, Green Chem., 2025, 27(6), 1620–1651, 10.1039/D4GC05445H.
| This journal is © The Royal Society of Chemistry 2026 |





