Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Salt complexation drives liquid crystalline self-assembly in crown ether–amino acid hybrids†
Aileen R.
Raab
*a,
Tanja Robin
Grießer
a,
Daniel
Rück
a,
Zhuoqing
Li
bc,
Anna
Zens
 a,
Johanna R.
Bruckner
a,
Johanna R.
Bruckner
 *f,
Patrick
Huber
*f,
Patrick
Huber
 bc,
Andreas
Schönhals
bc,
Andreas
Schönhals
 de,
Paulina
Szymoniak
*d and
Sabine
Laschat
de,
Paulina
Szymoniak
*d and
Sabine
Laschat
 *a
*a
aUniversität Stuttgart, Institut für Organische Chemie, Pfaffenwaldring 55, 70569 Stuttgart, Germany. E-mail: sabine.laschat@oc.uni-stuttgart.de; aileen-rebecca.raab@oc.uni-stuttgart.de
bHamburg University of Technology, Institute for Materials and X-ray Physics, Denickestr. 15, 21073 Hamburg, Germany
cCentre for X-ray and Nano Science CXNS, Deutsches Elektronen-Synchrotron DESY, Notkestr. 85, 22607 Hamburg, Germany
dBundesanstalt für Materialforschung und -prüfung, Unter den Eichen 87, 12205 Berlin, Germany. E-mail: paulina.szymoniak@bam.de
eTechnische Universität Berlin, Institut für Chemie, Straße des 17. Juni 135, 10623 Berlin, Germany
fUniversität Stuttgart, Institut für Physikalische Chemie, Pfaffenwaldring 55, 70569 Stuttgart, Germany. E-mail: johanna.bruckner@ipc.uni-stuttgart.de
First published on 6th February 2026
Abstract
Crown ether–amino acid hybrids represent a promising class of amphiphilic molecules combining ion recognition with self-assembly capabilities. Despite extensive studies on their binding properties, the influence of inorganic salt complexation on their liquid crystalline behaviour remains underexplored. Here we synthesized amphiphilic [18]-crown-6 derivatives of L-dihydroxyphenylalanine and tetrahydroisoquinoline analogues, systematically investigating the effects of alkyl chain length and salt type on mesophase formation. Complexation with various salts induced liquid crystalline phases, transitioning from smectic A to columnar hexagonal structures as anion size and alkyl chain length increased. Structural analyses and electron density mapping revealed assembly into charged superdiscs forming columnar stacks with tunable ion channels. Broadband dielectric spectroscopy highlighted differences in molecular mobility and conductivity linked to molecular design. These findings establish salt complexation as a key strategy to control self-assembly and ion transport in crown ether–amino acid hybrids, advancing their potential in responsive soft materials and ion-conductive applications.
1. Introduction
Hybrid materials combining crown ethers and amino acids are highly attractive from several perspectives. The ability of crown ethers to form supramolecular complexes with metal salts as well as amino acid and peptide derivatives is useful for chiral separation, molecular recognition and sensing applications. Most of the work has exploited the binding affinities of crown ethers for ammonium and guanidinium units of amino acids and peptides in order to develop chiral stationary phases for HPLC,1–14 supercritical fluid chromatography15,16 or capillary electrophoresis.17,18 Other reports dealt with chiral recognition in solution monitored by colorimetry,19 guided ion beam mass spectrometry,20,21 luminescence22,23 and NMR24,25 among other methods. Supramolecular interactions between amino acids and crown ethers were also utilized for chiral NMR shift agents.26,27 The underlying proton transfer has been studied both experimentally and theoretically,28–30 resulting in the development of mono- or (hetero)ditopic receptors,31,32 liquid membranes,23 optical,33 MRI34,35 and electrochemical sensors.36,37 Moreover, the selective ion recognition of crown ethers towards small spherical cations has inspired the development of Li+ ion receptors38 and ionophores for model membranes39–42 as well as ion conductors.43,44Despite this wealth of information on materials based on crown ether/amino acid interactions and reported liquid crystalline crown ethers38,43,45–62 and mesogenic amino acids,63–74 surprisingly little is known about liquid crystalline hybrids combining both crown ethers and amino acid entities in one molecular scaffold. Such a combination might lead to novel properties of the hybrid materials, such as ionic conductivity caused by the anions, while the chiral amphiphilic amino acid moiety should self-assemble into mesophases with long-range orientational order.
Depending on the presence or absence of charge, mesophase stabilization may arise from Coulomb interactions, van der Waals forces, π–π stacking of aromatic units, hydrogen bonding, and free-volume minimization, which together drive the nanosegregation of immiscible parts. In an earlier study on liquid crystals (LCs) derived from crown ether–amino acid hybrids, we demonstrated that the presence, number, and position of charges exerted a pronounced influence on the mesomorphic properties of these hybrid materials.64 In this previous work the crown ether was directly connected with the aryl unit of the amino acid 1 (Fig. 1). Complexation with NaI changed the columnar rectangular (Colr) mesophase of 1 to a smectic A (SmA) mesophase for 1·NaI.
 | ||
| Fig. 1 Our previous work on ionic liquid crystals derived from crown ether–amino acid hybrids (top),64 and the current work on DOPA(n) and THIQ(n) based crown ether hybrids (bottom). | ||
We recently published the dynamical properties and conductivity of L-dihydroxyphenylalanine (L-DOPA)-based guanidinium chlorides, which result from their self-assembly into superdiscs in Colh phases.75
Here we demonstrate for the first time, that the salt complexation of crown ether amino acid hybrids is essential for liquid crystalline self-assembly and also controls their conductive behaviour. For this purpose, we designed the amphiphilic hybrids c-DOPA(n) consisting of an N-terminally bound [18]-crown-6 unit and three flexible alkyl side chains with equal chain length (n = number of carbon atoms per chain) at the C-terminal ester and catechol ether groups of the amino acid DOPA (Fig. 1). To obtain structurally related hybrids with reduced conformational flexibility of the amino acid scaffold, the corresponding rigid tetrahydroisoquinoline crown ether hybrids c-THIQ(n) were synthesized. The synthesized hybrids and their salt complexes c-DOPA(n)·MX, c-THIQ(n)·MX were investigated by polarizing optical microscopy (POM), differential scanning calorimetry (DSC) and X-ray diffraction (XRD) to characterize the mesophase behaviour. The self-assembly of the molecules into mesophases is governed not only by intermolecular interactions but also by molecular mobility.67,75,76 Therefore, the molecular mobility for selected compounds was further investigated by broadband dielectric spectroscopy. Moreover, as the electrical conductivity of these ionic compounds is essential for applications, this property is also considered for the same selected compounds.
2. Results and discussion
2.1. Synthesis
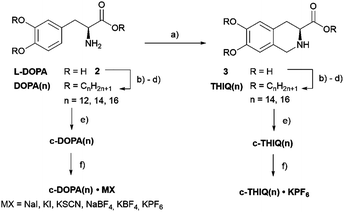
As outlined in Fig. 2 mesogens DOPA(n) and THIQ(n) were prepared from enantiopure L-DOPA 2. Thus, L-DOPA 2 was protected with Boc2O in the presence of NEt3, followed by a one-pot, base-catalysed etherification/esterification with the corresponding n-alkyl bromides in the presence of K2CO3 and NaI. The removal of the Boc group with TFA results in DOPA(n) (n = 12, 14, 16) in yields of 43–74% over 3 steps.65DOPA(n) were then reacted with [18]crown-6-benzoic acid [18]c-6-OH in the presence of ethylene diamine carbodiimide hydrochloride (EDC⋯HCl) and 4-(dimethylamino) pyridinium 4-toluene-sulfonate (DPTS) as well as hydroxybenzotriazole monohydrate (HOBt·H2O) to the amides c-DOPA(n) in yields of 18–84%.For the synthesis of c-THIQ(n), a Pictet–Spengler reaction with aqueous formaldehyde and 0.5 N H2SO4 was carried out in 77% yield. Compound 3 was then converted by a similar sequence as described for the DOPA(n) series to c-THIQ(n) in yields of 51–65% over 4 steps. Complexation with different salts (MX = KSCN, NaI, KI, NaBF4, KBF4, KPF6) provided the materials c-DOPA(n)·MX in yields of 83–98% and c-THIQ(n)·KPF6 in yields 83–99%.
2.2. Thermotropic properties
Investigation of the hybrids by POM suggested a strong dependence of the mesomorphic properties of the complexes on both salts and side chain lengths. While members of the salt-free series c-DOPA(n), c-THIQ(n) did not show liquid crystalline properties, for the complexes c-DOPA(n)·MX (MX = KSCN, NaI, KI, NaBF4 and KPF6) and c-THIQ(n)·MX textures were observed, which indicated the presence of a liquid crystalline phase.For the c-DOPA(12)·MX series with short alkyl chains (Fig. 3, top row, from left to right) c-DOPA(12)·KSCN with the smallest anion size showed predominantly homeotropic regions with fine fan-shaped textures and focal conic defect textures, which are typical for SmA phases. Complex c-DOPA(12)·NaI showed mostly homeotropic regions with fine Maltese crosses, while the KI-complex features fan-shaped textures. The textures of both iodide complexes are typical for SmA as well. The textures of c-DOPA(12)·NaBF4 resemble those observed for c-DOPA(12)·KI. Therefore, we assume a SmA phase for c-DOPA(12)·NaBF4. Complex c-DOPA(12)·KPF6 with the largest anion exhibited broad fan-shaped textures. Thus, the whole c-DOPA(12)·MX series exhibited SmA phases.
 | ||
| Fig. 3 Textures of the c-DOPA(n)·MX complexes during cooling observed by polarizing optical microscopy (crossed polarizer), unless otherwise stated on untreated slides with cooling rate: 1 K min−1. | ||
For the c-DOPA(14)·MX series with medium chain length similar textures were observed (Fig. 3, middle row, from left to right). c-DOPA(14)·KSCN and c-DOPA(14)·NaI featured predominantly homeotropic regions with fine uncharacteristic textures that resemble bâtonnets and small Maltese crosses, respectively. Both c-DOPA(14)·KI and c-DOPA(14)·NaBF4 featured fan-shaped textures. Therefore, these complexes also were assigned to form a SmA phase. However, the textures of the complex c-DOPA(14)·KPF6 were significantly different from that observed for the other complexes of the c-DOPA(14)·MX series. The spherulitic textures and dendritic growth indicate a columnar hexagonal (Colh) mesophase. This suggested a change of the type of the mesophase within the c-DOPA(14)·MX series from a SmA to a Colh mesophase with increasing anion size. The preference to form columnar hexagonal mesophases instead of layered phases for ionic liquid crystals with large anions like PF6 has been previously reported by Neidhardt.63
For the c-DOPA(16)·MX series with the longest alkyl chains (Fig. 3, bottom row) spherulitic textures were observed for the NaI, KI, KSCN and KPF6 complexes. Additionally dendritic growth was observed for the complex c-DOPA(16)·KPF6. c-DOPA(14)·NaBF4 complex showed fan-shaped focal conic textures with straight line defects, which are characteristic for columnar hexagonal phases. Therefore, the mesophase types of all complexes in this series were assigned to Colh phases.
Tetrahydroisoquinoline derivative c-THIQ(14)·KPF6 displayed homeotropic regions featuring small Maltese crosses, indicating a SmA phase (Fig. S99). For c-THIQ(16)·KPF6 spherulitic textures with dendritic growth were observed (Fig. S99b), which are typical of the Colh phase and similar to the textures for c-DOPA(14)·KPF6 and c-DOPA(16)·KPF6 (Fig. 3, middle right and bottom right), respectively.
DSC results summarized in Table 1 confirmed that both salt-free DOPA hybrids c-DOPA(n) and THIQ hybrids c-THIQ(n) were not liquid crystalline for the considered chain lengths with n = 12, 14 and 16 (entries 1, 8, 15, 21). The melting points of c-DOPA(n) occurred at higher temperatures compared to c-THIQ(n) (n = 14, 16) and did not dependent on the side chain length, whereas the melting temperatures for c-THIQ(n) increased with increasing n. The complexation of the crown ether unit with salt had a pronounced influence on the melting behaviour. Except for non-mesomorphic c-DOPA(n)·KBF4 (n = 12, 14) (entries 6, 13) all other salt complexes exhibited a liquid crystalline behaviour.
| # | n | MX | Temperature/°C (enthalpy in kJ mol−1)c | |
|---|---|---|---|---|
| a The results of the 1st cooling cycle are listed. b The results of the 6th cooling cycle are listed. c The following phases were observed Cr: crystal, I: isotropic phase, G: glass, SmA: smectic A, Colh: hexagonal columnar. | ||||
| c-DOPA(n)·MX | ||||
| (1) | 12 | — | Cr 100 (70.6) I | |
| (2) | 12 | KSCN | G 61 SmA 91 (1.6) I | |
| (3) | 12 | NaI | G 81 SmA 83 (1.0) I | |
| (4) | 12 | KI | G 63 SmA 77 (1.2) I | |
| (5) | 12 | NaBF4 | Cr −19 (2.6) G 53 SmA 115 (0.1) I | |
| (6) | 12 | KBF4 | Cr 97 (61.1) I | |
| (7) | 12 | KPF6 | G 69 SmA 101 (0.4) I | |
| (8) | 14 | — | Cr 101 (70.0) I | |
| (9) | 14 | KSCN | G 61 SmA 88 (1.2) I | |
| (10) | 14 | NaI | G 81 SmA 83 (1.0) I | |
| (11) | 14 | KI | G 57 SmA 98 (0.4) I | |
| (12) | 14 | NaBF4 | Cr 12 (2.2) G 58 SmA 112 (0.5) I | |
| (13) | 14 | KBF4 | Cr 99 (69.1) I | |
| (14) | 14 | KPF6 | G 57 Colh 134 (0.8) I | |
| (15) | 16 | — | Cr 100 (49.1) I | |
| (16) | 16 | KSCN | Cr 53 (17.3) Colh 106 (0.6) I | |
| (17) | 16 | NaI | Cr 22 (13.8) G 75 Colh 137 (0.2) I | |
| (18) | 16 | KI | Cr 30 (17.5) G 54 Colh 116 (0.4) I | |
| (19) | 16 | NaBF4 | Cr 34 (28.1) G 50 Colh 122 (0.5) I | |
| (20) | 16 | KPF6 | Cr 28 (18.0) G 69 Colh 133 (0.4) I | |
| c-THIQ(n)·MX | ||||
| (21) | 14 | — | Cr 33 (35.0) I | |
| (22) | 14 | KPF6 | G 65 SmA 112 (0.4) I | |
| (23) | 16 | — | Cr 49 (62.6) I | |
| (24) | 16 | KPF6 | Cr 31 (18.1) G 54 Colh 132 (0.4) I | |
For the c-DOPA(n)·MX series, enantiotropic mesomorphism was observed for n = 12 with NaI and KPF6 (entries 3, 7), for n = 14 with NaI, NaBF4 and KPF6 (entries 10, 12, 14), and for n = 16 with KSCN, NaI, KI, NaBF4 and KPF6 (entries 16–20). In contrast, monotropic mesomorphism was detected for n = 12 with KSCN, KI and NaBF4 (entries 2, 4, 5) and for n = 14 with KSCN, and KI (entries 9, 11). For c-THIQ(n)·MX, enantiotropic mesomorphism was observed for n = 14 and 16 with KPF6 (entries 22, 24). The cation seems to play some role in the mesomorphic properties. Despite the one hundred times higher association constant of the potassium cation with a [18]-crown-6 ether as compared to that of the sodium cation,77,78 the mesophases of c-DOPA(12)·KI and c-DOPA(14)·KI were only monotropic, while the corresponding sodium complexes c-DOPA(12)·NaI and c-DOPA(14)·NaI were enantiotropic. The absence of mesomorphism for c-DOPA(n)·KBF4 (n = 12, 14) might be due to the combination of the soft potassium cation with the hard tetrafluoroborate anion (Table S7, SI), which destabilized the mesophase79–82 (for all DSC curves see Fig. S96–S101, SI).
The complexation of c-DOPA(n) with metal salts resulted in significant decrease of the glass transition temperature, with the exception of the KBF4 derivatives (Fig. 4a and b). As c-DOPA(n) (n = 12, 14) and the corresponding complexes c-DOPA(n)·KBF4 showed almost identical phase transitions in the DSC curves, we checked complex formation via19F NMR. Initial attempts to record 19F NMR spectra of KBF4 or NaBF4 in CDCl3 were unsuccessful due to insolubility of the salts. In contrast, the respective complexes c-DOPA(n)·KBF4 and c-DOPA(n)·NaBF4 (n = 12, 14) were soluble and allowed 19F NMR measurements in CDCl3. In addition, 19F NMR spectra of pure KBF4 and NaBF4 in D2O were measured (Fig. S92 and S93). The resulting 19F signals differ clearly from those of c-DOPA(n)·KBF4 and c-DOPA(n)·NaBF4 (n = 12, 14), further supporting complex formation (Fig. S55, S70 as well as Fig. S52, S67). Regarding changes of 1H NMR shifts see Fig. S94, S95. For the two series c-DOPA(12)·MX and c-DOPA(14)·MX a combination of hard anions with hard cations (MX = NaBF4) or soft anions with soft cations (MX = KSCN, KI) reduced the glass transition temperature, stabilizing the mesophase, while a combination of soft anions with hard cations (MX = NaI) destabilized the mesophase, resulting in higher glass transition temperatures and mesophase ranges of only 2 K. This is likely due to enhanced nanosegregation of the mesogens with stronger interactions of hard-hard or soft-soft ion pairs (for the polarizability of the ions see Table S7). By further cooling of c-DOPA(12)·NaBF4 after vitrification of the sample a crystallization is also observed at −19 °C (Fig. S101a). The other complexes were only cooled to 0 °C, hence we assume they exhibit a similar crystallisation in a comparable temperature range. Additionally, for c-DOPA(14)·NaBF4 a crystallization also seems to occur below the glass transition temperature (see Fig. S103a).
 | ||
| Fig. 4 Mesophase ranges of crown ether complexes c-DOPA(n)·MX for (a) n = 12, (b) n = 14, (c) n = 16 as well as (d) c-THIQ(n)·MX in agreement with Table 1, monotropic phases are dashed, cooling rate: 5 K min−1. | ||
For the potassium complexes c-DOPA(16)·KX (KX = KSCN, KI, KPF6) the glass transition temperatures increased with increasing anion size, while decreasing with increasing anion size for the sodium complexes c-DOPA(16)·NaX (NaX = NaI, NaBF4) (Fig. 4c). Except for the KPF6-complexes the glass transition temperatures or melting point of the c-DOPA(16)·MX series were observed at lower temperatures than of the c-DOPA(14)·MX series due to the change of the mesophase type from SmA to Colh. Furthermore for c-DOPA(16)·MX (MX = KSCN, NaI, KI, NaBF4 and KPF6) a broadened crystallization transition was detected below the glass transition in the range of 22–34 °C (see Fig. 4c and Fig. S104). If the samples were completely vitrified a crystallisation at lower temperature should be suppressed, due to the restricted mobility of the frozen molecules. This indicates that during cooling first partial vitrification of the sample occurs due to freezing of the alkyl chains. Upon further decrease of the temperature the hydrophilic part of the hybrid materials finally crystallizes.
Complexation of c-DOPA(n) with metal salts resulted in decreased clearing points in the n = 12, 14 series compared to the melting point of the non-complexed compounds, except for the KPF6 and the NaBF4 derivatives (Fig. 4a and b). However, no general trend regarding anion size was visible. The lowest clearing transition was detected for c-DOPA(12)·NaI. In contrast, for the c-DOPA(16)·MX series the temperatures of the clearing transition increased significantly upon complexation. Clearing temperatures increased with increasing anion size resulting in broad mesophases with 49–102 K phase widths except for c-DOPA(16)·NaI (Fig. 4c).
Comparison of complexes c-DOPA(n)·KPF6 revealed decreasing glass and increased clearing temperatures with increasing chain length, resulting in broader temperature ranges of the mesophases (64–77 K) for n = 14, 16 in the Colh phase compared to 32 K for n = 12 in the SmA phase. In case of c-THIQ(n)·KPF6 glass transitions temperatures increased slightly with increasing n, while clearing transitions increased significantly, resulting in temperature ranges of the mesophases of 47 K for n = 14 in the SmA phase and 70 K for n = 16 in the Colh phase. It should be noted that the core rigidity of c-DOPA(n)versusc-THIQ(n) is not the only parameter that contributes to the thermal stabilization of LC phases and molecular assembled structures. In addition, the presence (in case of c-DOPA(n)) or absence (in case of c-THIQ(n)) of hydrogen bonds seems to play a major role as well. Thus, hydrogen bonds stabilize the crystalline phase of c-DOPA(n) (n = 14, 16), resulting in much higher melting temperatures as compared to c-THIQ(n). The hydrogen bonding interactions are weakened for the corresponding KPF6 complexes, likely due to the high fluidity of the molecules in the mesophase and the disorder in the glassy state. Fluidity and disorder become more relevant with increasing chain lengths. Thus, no significant stabilization of the mesophase of c-DOPA(16)·KPF6 compared to c-THIQ(16)·KPF6 was observed in contrast to the higher clearing temperature and broader mesophase range of c-DOPA(14)·KPF6 as compared to c-THIQ(14)·KPF6.
Additionally in the heating cycles of c-DOPA(14)·NaBF4, c-DOPA(16)·NaI, c-DOPA(16)·NaBF4 and c-THIQ(16)·KPF6 Cr → Cr transitions were observed (Fig. S103a, S104c and S105d). Especially the glass transitions are hardly visible. Typical examples are c-DOPA(12)·KPF6 (Fig. 5a), c-DOPA(14)·KPF6 (Fig. 5b) and c-DOPA(16)·KPF6 (Fig. 5c), where the glass transition in the cooling cycle is barely detectable.
 | ||
| Fig. 5 Representative DSC thermograms of c-DOPA(n)·KPF6 with (a) n = 12 (b) n = 14 (c) n = 16, heating and cooling rate: 5 K min−1. | ||
Additionally, measurements using fast scanning calorimetry (FSC) performed with a Flash DSC with high heating rates of up to 10.000 K s−1 on c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6 also showed a glass transition above the crystallisation during heating (Fig. S181 and S182). Due to the high heating rates the change of the heat flow at the glass transition is enhanced by FSC compared to conventional DSC measurements. Thus, it can be surmised that the amino acid-based hybrids c-DOPA(n)·MX and c-THIQ(n)·MX exhibit a mesophase (M)→ G transition during cooling, which is accompanied by a crystallisation (G → Cr). The only exception of this observation is c-DOPA(16)·KSCN, which crystallizes immediately from the mesophase during cooling and does not form a glass.
In order to assign phase geometries, XRD studies were carried out. Smectic phases, identified based on POM textures, showed a distinct small angle reflection assigned to the (001) reflex of a smectic structure (Fig. 6b). For the KI-complexes c-DOPA(12)·KI and c-DOPA(14)·KI a second distinct reflection was observed in the small angle section, which was assigned to the (002) reflex (Fig. 6a and Fig. S105, S106, S115–S117) that originates from the locally increased electron density of the comparably electron-rich KI.83 In the wide-angle region a broad halo was visible for all smectic complexes, which was caused by the average distance of the liquid-like alkyl side chains (Fig. S102–S122 and S158–S161).84 Using Bragg's law the periodicity distance d was determined in dependence of the temperature from the reflexes of the SAXS experiments by the following equation:
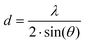 | (1) |
For c-DOPA(12)·KI the ratio of the 2θ-values of the (001) and (002) reflexes was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 confirming a SmA phase with a layer spacing of 36.9 Å. Considering the calculated molecular lengths of c-DOPA(12)Lcalc = 29.5 Å (obtained via Avogadro molecular modelling software),85 the ratio dobs/Lcalc = 1.25 suggests the presence of a SmAd phase, i.e. a bilayer organization of the crown ether amino acid hybrids with a strong degree of interdigitation of the alkyl chains as well as at least a partial overlap of the hydrophilic region of the mesogens, e.g. by stacking of the crown ether units.
2 confirming a SmA phase with a layer spacing of 36.9 Å. Considering the calculated molecular lengths of c-DOPA(12)Lcalc = 29.5 Å (obtained via Avogadro molecular modelling software),85 the ratio dobs/Lcalc = 1.25 suggests the presence of a SmAd phase, i.e. a bilayer organization of the crown ether amino acid hybrids with a strong degree of interdigitation of the alkyl chains as well as at least a partial overlap of the hydrophilic region of the mesogens, e.g. by stacking of the crown ether units.
Similar ratios for the 2θ-values were obtained for c-DOPA(14)·KI supporting the initial assignments of a SmA-type phase for c-DOPA(12)·MX (MX = KSCN, NaI, KI, NaBF4, KPF6), c-DOPA(14)·MX (MX = KSCN, NaI, KI, NaBF4) as well as c-THIQ(14)·KPF6, based on the POM studies (Fig. 3 and Fig. S99). The results are listed in Table 2, the diffractograms are depicted in the SI (Fig. S102–S122 and Fig. S158–S161). The smectic layer spacing dobs increases with increasing chain length. The values of dobs increase with increasing anion size for c-DOPA(12)·MX from dobs = 36.9 Å (KI) to 45.1 Å (KPF6) and for c-DOPA(14)·MX from dobs = 39.4 Å (KI) to 46.3 Å (KPF6). Meanwhile c-THIQ(14)·KPF6 exhibited dobs = 36.4 Å and a dobs/Lcalc ratio of 1.31 (Fig. S174). The reduction of the layer thickness by addition of the CH2-bridging unit is related to a change of the molecular shape, which will be discussed below assisted by Fig. 7–9.
| n | MX | T/°C | Phase type | hkl | d/Å | Lattice constant a/Å and Z-value |
|---|---|---|---|---|---|---|
| c-DOPA(n)·MX | ||||||
| 12 | KSCN | 85 | SmAd | 001 | 38.8 | — |
| halo | 4.5 (br) | |||||
| 12 | NaI | 77 | SmAd | 001 | 37.9 | — |
| halo | 4.1 (br) | |||||
| 12 | KI | 70 | SmAd | 001 | 36.9 | — |
| 002 | 18.5 (18.4) | |||||
| halo | 4.4 (br) | |||||
| 12 | NaBF4 | 85 | SmAd | 001 | 41.9 | — |
| halo | 4.5 (br) | |||||
| 12 | KPF6 | 83 | SmAd | 001 | 45.1 | — |
| halo | 4.1 (br) | |||||
| 14 | KSCN | 85 | SmAd | 001 | 39.4 | — |
| halo | 4.5 (br) | |||||
| 14 | NaI | 75 | SmAd | 001 | 39.4 | — |
| halo | 4.5 (br) | |||||
| 14 | KI | 80 | SmAd | 001 | 40.1 | — |
| 002 | 20.0 (20.1) | |||||
| halo | 4.2 (br) | |||||
| 14 | NaBF4 | 80 | SmAd | 001 | 46.3 | — |
| halo | 4.4 (br) | |||||
| 14 | KPF6 | 60 | Colh | 100 | 49.6 | a = 57.2 |
| 110 | 28.7 (28.6) | Z = 6 | ||||
| 200 | 24.8 (24.8) | |||||
| halo | 4.5 (br) | |||||
| 16 | KSCN | 85 | Colh | 100 | 48.7 | a = 56.2 |
| 110 | 28.3 (28.1) | Z = 6 | ||||
| 200 | 24.3 (24.4) | |||||
| halo | 4.6 (br) | |||||
| 16 | NaI | 93 | Colh | 100 | 53.7 | a = 61.7 |
| 110 | 30.8 (31.0) | Z = 6 | ||||
| 200 | 26.9 (26.8) | |||||
| halo | 4.4 (br) | |||||
| 16 | KI | 80 | Colh | 100 | 53 | a = 61.1 |
| 110 | 30.6 (30.6) | Z = 6 | ||||
| 200 | 26.7 (26.5) | |||||
| halo | 4.5 (br) | |||||
| 16 | NaBF4 | 80 | Colh | 100 | 47 | a = 54.2 |
| 110 | 27.1 (27.1) | Z = 5 | ||||
| 200 | 23.7 (23.5) | |||||
| halo | 4.6 (br) | |||||
| 16 | KPF6 | 74 | Colh | 100 | 56.8 | a = 65.6 |
| 110 | 33.1 (32.8) | Z = 7 | ||||
| 200 | 28.6 (28.4) | |||||
| halo | 4.4 (br) | |||||
| c-THIQ(n)·MX | ||||||
| 14 | KPF6 | 80 | SmAd | 001 | 46.3 | — |
| halo | 4.5 (br) | |||||
| 16 | KPF6 | 80 | Colh | 100 | 50.6 | a = 58.5 |
| 110 | 29.4 (29.2) | |||||
| 200 | 25.4 (25.3) | |||||
| halo | 4.5 (br) | |||||
For c-DOPA(14)·MX with MX = KSCN, NaI and KI a small increase of the layer thickness with increasing temperature was observed (Fig. S175). Such a positive thermal expansion is atypical for SmA phases86–91 and could be related to a compensating effect of the alkyl chains by strong interdigitation as well as a stronger interaction between neighbouring crown ether units at low temperatures.92,93 Only for larger anions X = BF4−, PF6− an increase of the layer thickness with decreasing temperature was observed, which possibly is due to a reduction of stacking interactions between neighbouring crown ether units caused by the larger anions (Fig. S175).
Comparison of the Avogadro-optimized molecular geometry of c-DOPA(14) with c-THIQ(14) revealed a smaller angle between the DOPA aryl unit and the benzocrown unit as compared to the angle between the THIQ aryl unit and benzocrown unit respectively (Fig. 7). This results in a larger steric demand of c-THIQ(n) compared to c-DOPA(n) in the hydrophilic region, which could accommodate for larger anions like PF6− in the smectic mesophase.
Since the KI-complexes exhibited two reflexes in the small angle region the relative electron density distribution ρ(z) in z-direction, i.e. parallel to the layer normal of the smectic layers, could be calculated with the approximation:
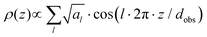 | (2) |
The accuracy of electron density profiles increases with the number of reflexes, hence for these complexes with just two reflexes they will not perfectly fit with regions of differing electron densities. As exemplified for c-DOPA(14)·KI the region of higher electron density corresponds to the crown ether units with the free anions connected by phenyl spacers to the aromatic core. The region with low electron density corresponds to the aliphatic chains of the mesogens, which are interdigitated with the aliphatic chains of the neighbouring smectic layer. The crown ether units are stacked on each other. Based on this proposed molecular arrangement the increase of the layer spacing with larger anions can be explained by a larger steric demand of the anions, which pushes the molecules outwards leading to an undulation of the stacked crown ether units. At the same time, the increase of the layer spacing allows the interdigitated alkyl chains of adjacent layers to decrease their lateral distance which is evident in the reduction of the intralamellar distance referred to as halo in Table 2. The crown ether unit of the c-THIQ(n) system forms a larger angle to the aromatic core, changing the effective shape of the molecule to a cylindrical form. Thus, the molten alkyl chains are less extended or stronger interdigitated to enable a space filling packing of the molecules in comparison to the c-DOPA molecules which possess a more truncated cone-like shape. This explains the small layer spacing for c-THIQ(14)·KPF6 among the KPF6-complexes.
Accordingly, a proposed molecular arrangement of c-DOPA(12)·KPF6 and c-THIQ(14)·KPF6 in the SmAd phase is depicted in Fig. 9.
All complexes, which were assigned Colh phases based on the textures determined by POM (Fig. 3 and Fig. S99), exhibited three distinct peaks in the small-angle regions (Fig. 10) and a broad halo in the wide-angle region. The 2θ-values of the peaks in the small angle region, which were assigned to the (100), (110) and (200) reflex, had a relation of 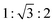 , confirming a hexagonal columnar (Colh) phase with p6mm space group.
, confirming a hexagonal columnar (Colh) phase with p6mm space group.
 | ||
| Fig. 10 SAXS diffractogram of c-DOPA(14)·KPF6 at 60 °C (inset: 2D diffraction pattern) during cooling from the isotropic phase. | ||
The lattice parameter a was calculated from the periodicity distance d100 of the (100) reflex with the equation:
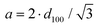 | (3) |
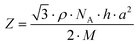 | (4) |
The lattice parameter a of c-DOPA(n)·KPF6 increased with increasing chain length from a = 57.2 Å (n = 14) to 65.6 Å (n = 16). Despite n = 16 for c-THIQ(16)·KPF6, the lattice parameter is only 58.5 Å due to the shorter molecular length caused by the bridging CH2 group. These correspond to an a/Lcalc ratio of 1.79 for c-DOPA(14)·KPF6 and 1.55 for c-THIQ(16)·KPF6, revealing a much stronger interdigitation of the alkyl chains of adjacent columns for the latter component. The a/Lcalc ratios for the c-DOPA(16)·MX complexes showed a strong dependence on the salt: for the potassium complexes (KSCN, KI, KPF6) the a/Lcalc ratio increased with increasing anion size and decreasing polarizability, ranging from 1.63 (KSCN) to 1.90 (KPF6) while for the sodium complexes the lattice parameter increased with increasing anion polarizability, with a/Lcalc ranging from 1.36 (NaBF4) to 1.79 (NaI) (Fig. S176).
All complexes exhibited a negative thermal expansion, with a noticeable decrease of the lattice parameter with increasing temperatures. For example, for c-DOPA(16)·KPF6a decreased from 65.6 Å at 70 °C to 59.5 Å at 130 °C (Fig. 11). A similar thermal expansion was previously observed for DOPA-based guanidinium chlorides and could be caused by increased intracolumnar distance between the supramolecular disks at higher temperatures.67
Not only the intercolumnar space but also the intracolumnar space was affected by the salt. A comparison of the intracolumnar distances, denoted as halo in Table 2, for the potassium complexes c-DOPA(16)·KX with X = SCN−, I− and PF6− showed a decrease of the average distance of the alkyl side chains with increasing anion size and decreasing polarizability. This indicates, similar to the results observed for the SmAd mesophases, that larger anions, due to their increased space requirement, push the molecules slightly out of the column centre which results in an increased lattice parameter. As a consequence, the alkyl chains need to spread and flatten to still guarantee a space filling packing, which in turn reduces the intracolumnar distance between the alkyl chains (Fig. S177).
The Z-values calculated according to eqn (4) range between 5 and 7. Since for most complexes a value of Z = 6 was obtained if the density was merely assumed to be 1.0 g cm−3, we propose for c-DOPA(14)·KPF6, c-DOPA(16)·MX and c-THIQ(16)·KPF6 that six molecules of the amino acid or tetrahydroisoquinoline crown ether complexes self-assemble to charged superdiscs with the crown ether cation complexes and anions located in the centre of the column surrounded by the aromatic core of the mesogens similar to the arrangement for DOPA-based ILCs with guanidinium headgroups known in literature (Fig. 13).65,67,101–111 Space filling between the columns is maintained by the alkyl side chains due to their tendency to maximize London dispersion interactions. Interdigitation and deviation from the all-trans configuration of the alkyl chains result in a cell constant larger than the molecular length but significantly smaller than the two-fold molecular length.
For the different complexes the two-dimensional electron density distribution ρ(xy), with the xy-plane being orthogonal to the director, was calculated according to
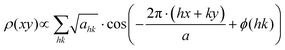 | (5) |
For the complexes c-DOPA(14)·KPF6, c-DOPA(16)·KSCN and DOPA(16)·KPF6 the 1D electron density profiles along the unit cell axis are depicted in Fig. 12. A representative model for the proposed molecular arrangement is depicted in Fig. 13 for c-THIQ(16)·KPF6. The region of higher electron density is located in the middle of the column, with an ion channel of the anions located in the centre, which are surrounded by the crown ether units and subsequently the aromatic part of the molecules. The aliphatic chains are located in the intercolumnar space and correspond to the region of lower electron density. Larger anions like PF6− require more space in the centre of the column, resulting in a flattening and broadening of the superdiscs. This effect is partially compensated for c-THIQ(16)·KPF6, which enables the accommodation of larger anions due to the difference of the shape in the hydrophilic region accommodating for larger anions.
 | ||
| Fig. 12 1D electron density distribution of c-DOPA(14)·KPF6 (orange), c-DOPA(16)·KSCN (black) and DOPA(16)·KPF6 (cyan) along the unit cell axis. | ||
The results of the electron density profiles also imply, that the six mesogens of a cell unit do assemble to a flat superdisc like it was proposed for the guanidinium chlorides by Neidhardt.65 Calculation of a 2D electron density profile reinforce this proposed molecular arrangement, as depicted for c-THIQ(16)·KPF6 in Fig. 14. Further 2D electron density maps are depicted in Fig. S179. To learn more about the location of the anion, we constructed an electron density difference map by subtracting the electron density distribution of c-DOPA(16)·NaBF4 from the one of c-DOPA(16)·NaI (Fig. S180 and corresponding discussion). This difference map further supports the proposed structure model of anion channels being located in the middle of the columns.
 | ||
| Fig. 14 2D electron density distribution in the x,y-plane with schematic depiction of the molecular arrangement for c-THIQ(16)·KPF6 with indicated self-assembly of the superdiscs into a Colh phase. | ||
2.3. Retardation measurements
Temperature-dependent optical retardation measurements112,113 were conducted for c-THIQ(16)·KPF6 (Fig. 15) and c-DOPA(16)·KPF6 (Fig. S178) to characterize the collective thermotropic alignment of discotic amino acid crown ether hybrids in cells of different surface hydrophobicity. The experimental setup employed a He–Ne laser (wavelength 632.8 nm) as a linearly polarized light source.The liquid crystalline powders were heated to approximately 20 °C above the liquid crystalline–isotropic phase transition temperature, then introduced into the bulk cells via capillary forces resulting in spontaneous imbibition. The bulk cells consisted of two glass substrates separated by a defined gap of 1.6 µm. The inner surfaces of the glass plates were chemically treated to produce either hydrophilic or hydrophobic surface conditions, thereby imposing distinct anchoring environments for the confined liquid crystals (for details, see Fig. S177). From the optical retardation curves the optical birefringence can be inferred (see SI, Chapter S9).
The retardation curves c-THIQ(16)·KPF6 allow for the extraction of meaningful information in both anchoring environments, as seen in Fig. 15. In both cases, the optical retardation signal is well-reproducible in the low-temperature regime below 320 K. A distinct transition from the liquid crystalline to the crystalline phase is observed near 315 K for both hydrophilic and hydrophobic surface treatments.
Within the liquid crystalline phase, c-THIQ(16)·KPF6 also displays signs of a multi-domain structure, as evidenced by moderate fluctuations in the optical retardation signal. Nonetheless, a weak and somewhat broad isotropic to liquid crystalline (I–LC) transition can still be discerned. This transition appears between 400 K and 410 K upon cooling, and between 360 K and 370 K during heating, albeit less sharply defined than the crystalline to liquid crystalline (Cr–LC) transition. The experimental setup does not allow the simultaneous confirmation of the multi-domain structure via POM.
The optical retardation response of c-DOPA(16)·KPF6 (Fig. S178) exhibits irregular fluctuations within a relatively narrow range and lacks reproducibility. This behaviour is likely attributable to weak surface anchoring, leading to the formation of a multi-domain structure within the bulk cell. The presence of randomly oriented domains results in minimal net optical anisotropy, which manifests as a fluctuating and poorly defined optical retardation signal which will not be discussed further.
It is well established that conventional LC materials typically exhibit thermal hysteresis in their phase transitions, where the liquid crystalline to isotropic transition (LC–I) occurs at a higher temperature upon heating and at a lower temperature during cooling. This behaviour arises from the first-order nature of the transition, which involves a discontinuous change in order parameters such as molecular orientation or positional order, and is associated with latent heat and a free energy barrier separating the two phases. Upon heating, the LC phase can persist until a superheating limit is reached, beyond which thermal energy is sufficient to overcome the energy barrier, resulting in a transition to the isotropic phase. During cooling, the isotropic phase may remain metastable below the equilibrium transition temperature until nucleation of the LC phase occurs – often delayed by surface anchoring conditions, defects, or impurities – leading to a lower apparent transition temperature.
However, in the present case, we observe an inverse hysteresis: the LC–Iso transition occurs at a lower temperature during heating and at a higher temperature during cooling. This atypical behaviour may be attributed to slow relaxation dynamics within the system. For instance, high viscosity, structural defects, or molecular crowding can hinder complete reorganization during the heating cycle, causing the LC structure to lose order prematurely and resulting in an early transition to the isotropic phase. Conversely, during cooling, nucleation of the LC phase may be facilitated by surface anchoring or residual molecular order, causing the transition to occur at a comparatively higher temperature. This reversal of hysteresis thus reflects the complex interplay between kinetic constraints and interfacial effects in the phase behaviour of the material.
The observed difference in collective orientation behaviour between the bridged and non-bridged molecules in the planar LC cell can be primarily attributed to differences in molecular rigidity and shape persistence. The bridged molecule, being structurally more rigid due to its covalent constraint, likely maintains a more anisotropic and well-defined molecular geometry. This rigidity favours stronger and more coherent intermolecular interactions, thereby enhancing long-range orientational order within the cell. In contrast, the non-bridged molecule possesses greater conformational flexibility, which can lead to a broader distribution of molecular orientations and a higher likelihood of forming disordered or multidomain structures. Additionally, rigid molecules typically have higher elastic constants (especially for splay and bend deformations), making it energetically more favourable for them to adopt uniform alignment under the influence of surface anchoring. In contrast, flexible molecules can more easily distort or accommodate random orientations, resulting in weaker macroscopic birefringence signals due to poor collective alignment.114,115
2.4. Dielectric studies
Dielectric investigations were carried out for two samples c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6. These samples were selected for the following reasons: first, both samples have a columnar mesophase which is important for applications like ion or water transport. Second, dielectric spectroscopy will allow to directly investigate the change in the molecular mobility due to the bridged structure in the core of c-THIQ(16)·KPF6 compared to that of c-DOPA(16)·KPF6. Additionally, the electrical conductivity was measured. The dielectric spectrum of c-THIQ(16)·KPF6 is shown in Fig. 16 as dielectric loss ε″ versus frequency and temperature in a 3D representation for the columnar mesophase and the low-temperature phase, which could be identified as a plastic crystalline phase. Plastic crystalline phases are solid-state phases that combine long-range positional order with orientational or conformational mobility of the constituent molecules resulting in detectable molecular mobility.116 Several dielectric relaxation processes are observed, indicated as peaks in ε″ besides a conductivity contribution. An analogous behaviour was observed for c-DOPA(16)·KPF6 (Fig. S179).The relaxation processes were labeled from 1 to 3 in order of increasing temperature. At higher temperatures the dielectric loss increases strongly with decreasing frequency and increasing temperature due to electrical conductivity related to the drift motion of charge carriers like ions and conductivity related processes like Maxwell/Wagner/Sillars and/or electrode polarization. By a derivative approach (conduction free loss)118 it could be shown that a further relaxation is hidden by the conductivity which is called relaxation process 4 or α-relaxation (see Chapter S10, SI).
The analysis of the dielectric spectra is based on the fitting of the model function of Havriliak-Negami (HN) to the data.117,119 For details (see Chapter S10, SI). From the fits the frequency of maximal dielectric loss for each process fp (relaxation rate) is derived.
Fig. 17 depicts the temperature dependence of the relaxation rate fpversus inverse temperature (Arrhenius diagram). The temperature dependence of the relaxation rates for process 1 follows the Arrhenius equation which is given by
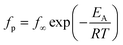 | (6) |
 | ||
| Fig. 17 Temperature dependence of the relaxation rates versus inverse temperature: (blue) – c-DOPA(16)·KPF6, (red) – c-THIQ(16)·KPF6, (green) – data for polyethylene taken from the literature.117 (squares) – Relaxation process 1, (circles) – relaxation process 2, (asterisk) – relaxation process 3 and (diamonds) – relaxation process 4 (α-relaxation). Solid lines are fits of the Arrhenius equation to the corresponding data. The dashed-dotted line indicate fits of the VFT equation to the corresponding data of the α-relaxation. The dashed line indicates the phase transition from the platic crystalline state to the columnar ordered liquid crystalline phase taken from the analysis of the DC conductivity (see below). | ||
Fig. 18 compares the dielectric spectra for the samples at 90 °C, which is in the temperature range of relaxation process 2. For c-DOPA(16)·KPF6 a weak peak is observed in the spectra indicating a relaxation process where for c-THIQ(16)·KPF6 only a shoulder on the low frequency side of the relaxation process 1 is visible. For both materials process 1 and 2 can be separated by analysing the dielectric spectra by fitting the HN-function to the data.119 This means that for c-THIQ(16)·KPF6 the intensity of relaxation process 2 is smaller than that of c-DOPA(16)·KPF6. The only difference between the samples is that c-THIQ(16)·KPF6 has no CH2 bridge in the aromatic core. The CH2 bridge provides an additional flexibility and mobility to the aromatic core of c-DOPA(16)·KPF6. Therefore, the relaxation process 2 is assigned to the small angle rotational or bend fluctuations in the aromatic core. For the sample c-THIQ(16)·KPF6 the flexibility is restricted leading to a lower intensity. Moreover, relaxation process 2 for c-THIQ(16)·KPF6 is observed at higher temperatures than that of c-DOPA(16)·KPF6 (see Fig. 18) supporting the assignment. For the activation energy of relaxation process 2 a value of ca. 48 kJ mol−1 was estimated for both materials pointing to a localized relaxation process in agreement with the proposed assignment.
 | ||
| Fig. 18 Dielectric loss versus frequency at T = 183.1 K (90 °C): (blue) – c-DOPA(16)·KPF6, (red) – c-THIQ(16)·KPF6. | ||
The temperature dependence of the relaxation rates of relaxation process 3 also follows the Arrhenius law with estimated apparent activation energies of 70.6 kJ mol−1 for c-DOPA(16)·KPF6 and 59.5 kJ mol−1 for c-THIQ(16)·KPF6, which are too high for a localized relaxation process. Therefore, it is assumed that relaxation process 3 is due to cooperative processes like glassy dynamics corresponding to a glass transition. As the activation energies of relaxation process 3 for c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6 are different it can be further concluded that relaxation process 3 is related to the core and is assigned to the glass transition of the cores. A similar assignment was made also for conventional and ionic liquid crystals.76,121–123 Due to the CH2 bridge this process becomes more localized for c-THIQ(16)·KPF6 in comparison to the c-DOPA(16)·KPF6 resulting in a lower apparent activation energy.
For c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6 a glass transition was detected with conventional DSC as well as with fast scanning calorimetry. As discussed above by a derivative approach it could be proven that a further relaxation (relaxation process 4 or α-relaxation) is hidden under the conductivity contribution for c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6. The analysis of the α-relaxation is given in the SI, Chapter S10 and Fig. S180.
The relaxation rates of relaxation process 4 are included in Fig. 17. The temperature dependence of relaxation process is curved when plotted versus invers temperature which can be described by the Vogel/Fulcher/Tammann (VFT-) equation which reads124–126
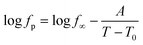 | (7) |
The glassy dynamics of c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6 is independent of the core. Therefore, it is concluded that the α-relaxation in these compounds is due to the glassy dynamics of alkyl chains in the intercolumnar space.
Due to the salt character of the hybrid materials it is interesting to consider the electrical conductivity. The complex dielectric function is related to the complex conductivity as detailed discussed in the SI, Chapter S10. Examples for the conductivity spectra and the estimation of the electrical DC conductivity σDC are also given in Fig. S181. As the samples are not oriented between the electrodes the conductivity should be considered as an averaged one.
In Fig. 19 the estimated values for the DC conductivity for c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6 are plotted versus inverse temperature in Arrhenius coordinates.
In the plastic crystalline phase, the DC conductivity is low, and its temperature dependence is characterized by an Arrhenius law with an activation energy of ca. 71 kJ mol−1. In the temperature range of the phase transition from the glass to the hexagonal ordered liquid crystalline phase σDC changes step-like to higher values. This step-like change in the temperature dependence of σDC indicates the phase transition. In the temperature range of the hexagonal ordered liquid crystalline phase the DC conductivity increase with increasing temperature also according to the Arrhenius law but with a higher activation energy of ca. 154 kJ mol−1. In the temperature range of the Colh phase to the isotropic phase a decreased activation energy is observed. This change is more pronounced for c-THIQ(16)·KPF6 in comparison to c-DOPA(16)·KPF6.
The DC conductivity values of c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6 are similar in the Colh phase. As the ions in both samples are the same it is concluded that the conductivity in the considered materials is due to drift motions of the mobile anion. Furthermore, the temperature dependence of the DC conductivity follows the Arrhenius law where the temperature dependence of the relaxation rates of the α-relaxation follows the VFT equation. Therefore, the conduction mechanism is not linked to the α-relaxation of the alkyl chains in the intercolumnar space but is due to the drift motion of anions in the channels of the columns formed by the superdiscs.
3. Conclusions
In order to understand the influence of metal salts MX on the liquid crystalline self-assembly as well as molecular mobility, electrical conductivity and long-range orientational order of selected samples within LC cells, two series of amino acid crown ether hybrid materials c-DOPA(n)·MX (n = 12, 14, 16; MX = KSCN, NaI, KI, NaBF4, KBF4, KPF6) and tetrahydroisoquinoline crown ether hybrid materials c-THIQ(n)·KPF6 (n = 14, 16) with three linear alkyl side chains were synthesized from L-DOPA. The THIQ series differs from the DOPA series by the presence of a bridging CH2-unit between the aromatic core and the N-terminus resulting in reduced rotational freedom of the amino acid core. Moreover, in the c-DOPA(n) series hydrogen bonds of the amide unit contribute to the liquid crystalline self-assembly. The investigation of the mesomorphic properties by DSC, POM and XRD revealed that complexation of the crown ether unit with a metal salt is essential for the formation of a mesophase where either SmAd or Colh phases were observed. Within the c-DOPA(n)·MX series (n = 12, 14) those complexes with KSCN, NaI, KI, NaBF4 in combination with shorter side chains (n = 12, 14) favoured the formation of SmAd phases, whereas c-DOPA(n)·MX with long side chains (n = 16) favoured the Colh phase. The results revealed that the combination of chain lengths and anion size is important for the mesophase formation and leads to a “switch” from SmAd phases for c-DOPA(14)·KPF6 and c-THIQ(12)·KPF6 to Colh states for c-DOPA(16)·KPF6 and c-THIQ(14)·KPF6, respectively.The choice of the complexation salt also plays a significant role in the stability of the mesophases. All SmAd phases were monotropic except for the NaI- and KPF6-complexes, which exhibited enantiotropic SmAd phases. Complexation with NaI in the c-DOPA(n)·MX series yielded only small mesophase widths of less than 5 K for the hybrid material with short and medium alky chain length (n = 12, 14) in the enantiotropic SmAd phase, while the KPF6 complex significantly stabilized the enantiotropic mesophase, exhibiting the broadest mesophase range with a temperature width of more than 75 K. XRD experiments revealed a bilayer structure of the SmAd phase with a strong interdigitation of the alkyl chains, deviating from the all-trans configuration as well as stacking of the complexed crown ether units in the centre of the bilayer structure. The proposed molecular arrangements within the Colh phase by superdisc formation of the wedge-shaped mesogens of the c-DOPA(n)·MX and c-THIQ(n)·MX series were supported by calculations of electron density maps.
The optical retardation studies show that molecular rigidity plays a decisive role in governing collective alignment in discotic liquid crystals. The non-bridged c-DOPA(16)·KPF6 displays weak anchoring and multidomain disorder, whereas the bridged c-THIQ(16)·KPF6 achieves more coherent orientational order and clearer phase transition signatures. The observed inverse thermal hysteresis further indicates slow relaxation dynamics and interfacial effects influencing phase stability. Overall, covalent rigidification enhances shape persistence and elastic resistance to deformation, providing an effective strategy for improving long-range alignment and optical anisotropy in discotic liquid crystalline materials.
To investigate the conductivity and molecular mobility two selected members c-DOPA(16)·KPF6 and c-THIQ(16)·KPF6 of the two series differing only in the presence of the bridging CH2-unit in their molecular structure were examined by broadband dielectric spectroscopy. These complexes exhibited both a broad enantiotropic Colh phase in similar temperature ranges. With increasing temperature three low temperature relaxation processes were observed for both complexes, all of which followed the Arrhenius law. The first relaxation process, due to localized fluctuations of the alkyl chains, is independent of the molecular structure of the aromatic core and similar relaxation energies were observed with 35.4 kJ mol−1 and 36.6 kJ mol−1, respectively, for both crown ether complexes. The second relaxation process with an estimated activation energy of 48 kJ mol−1, is assigned to a localized relaxation process of the aromatic core. The intensity of process 2 was significantly less for c-THIQ(16)·KPF6, due to the constraining effect of the bridging CH2-unit at the aromatic core. The third relaxation process with activation energies of 70.6 kJ mol−1 and 59.5 kJ mol−1, respectively, is related to a glass transition of the aromatic cores, which due to the bridging CH2-unit is more localized for c-THIQ(16)·KPF6, resulting in a lower activation energy. Finally, in the temperature region of the glass transition a fourth relaxation process was observed for both complexes, which was independent of the aromatic core and followed the VFT-temperature dependence. The process was assigned to glassy dynamics (α-relaxation) of the alkyl chains in the intercolumnar space.
For both complexes electrical conductivity was observed in the dielectric loss spectra. The temperature dependence estimated DC followed the Arrhenius law upon heating from the plastic crystalline phase a step wise increase of conductivity was observed. In the mesophases an activation energy of approximately 154 kJ mol−1 was estimated for both complexes. Further heating into the isotropic state resulted in a slight decrease of the activation energy, which was slightly more pronounced for c-THIQ(16)·KPF6. The conductivity was attributed to the drift motion of the PF6− anions within the channels formed by columnar structures of the mesophase, as for both complexes the activation energy was independent of the presence of the CH2-bridging unit.
Our results have shown that salt complexation controls both self-assembly and induces ion transport in amino acid crown ether hybrids, paving the way towards ion-conducting applications.
Author contributions
A. R. R. conceived the project and did a part of the synthesis, most of the characterization, analysis and data curation and wrote the first draft of the manuscript. T. R. G. did a part of the synthesis and characterization. P. S. and A. S. supervised the dielectric measurement. D. R. performed a part of the dielectric measurements and the fast-scanning calorimetry experiments and reviewed the manuscript. P. S. and A. S. contributed their expertise regarding analysis of the dielectric measurement. A. S. helped with the manuscript preparation and checked the data. Z. L. performed the optical retardation studies and helped with the manuscript preparation. A. Z. checked the data and helped with the manuscript preparation. P. H. contributed his expertise regarding XRD analysis. J. R. B. assisted with conceiving the packing models and calculating electron densities. S. L supervised the project and wrote the manuscript. All co-authors checked and proofread the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this publication are included in the supplementary information (SI) file accompanying this article. Supplementary information: 1H NMR and 13C NMR and 19F NMR spectra; details of X-Ray diffraction, DSC, FSC, POM, retardation and dielectric measurements. See DOI: https://doi.org/10.1039/d5tc04129e.Acknowledgements
Marcel Freund assisted in conceiving the synthesis. Soeren M. Bauch and Sebastian B. Wachsmann conceived a plug-in utilized in the software for the analysis. Generous financial support by the Deutsche Forschungsgemeinschaft (DFG grant # LA 907/21-1; HU 850/13-1; SCHO 470/26-1, INST 41/897-1 FUGG for 700 MHz NMR, INST 41/1136-1 FUGG for Orbitrap LC-MS), the Carl-Schneider Stiftung Aalen (shared instrumentation grant) and the Ministerium für Wissenschaft, Forschung und Kunst des Landes Baden-Württemberg are gratefully acknowledged.Notes and references
- T. Öztepe, N. B. Kale, T. Reçber, İ. Baysal, S. Yabanoğlu-Çiftçi, M. Gumustas, S. Kır, B. Chankvetadze and E. Nemutlu, J. Chromatogr. A, 2022, 1683, 463529 CrossRef PubMed.
- T. Németh, G. Dargó, J. L. Petró, Z. Petrik, S. Lévai, B. Krámos, Z. Béni, J. Nagy, G. T. Balogh, P. Huszthy and T. Tóth, Chirality, 2017, 29, 522–535 CrossRef PubMed.
- E. S. Cho, J. Y. Sung, J. S. Jin and M. H. Hyun, J. Sep. Sci., 2018, 41, 1192–1198 Search PubMed.
- T. Upmanis and H. Kažoka, J. Chromatogr. A, 2022, 1673, 463059 Search PubMed.
- R. Berkecz, G. Németi, A. Péter and I. Ilisz, Molecules, 2021, 26, 4648 CrossRef CAS PubMed.
- I. Kawamura, B. Mijiddorj, Y. Kayano, Y. Matsuo, Y. Ozawa, K. Ueda and H. Sato, Biochim. Biophys. Acta, Proteins Proteomics, 2020, 1868, 140429 Search PubMed.
- G. Carenzi, S. Sacchi, M. Abbondi and L. Pollegioni, Amino Acids, 2020, 52, 849–862 Search PubMed.
- K. Hirose, J. Yongzhu, T. Nakamura, R. Nishioka, T. Ueshige and Y. Tobe, J. Chromatogr. A, 2005, 1078, 35–41 CrossRef CAS PubMed.
- P. Gerbaux, J. De Winter, D. Cornil, K. Ravicini, G. Pesesse, J. Cornil and R. Flammang, Chem. – Eur. J., 2008, 14, 11039–11049 Search PubMed.
- Y. Konya, M. Taniguchi, M. Furuno, Y. Nakano, N. Tanaka and E. Fukusaki, J. Chromatogr. A, 2018, 1578, 35–44 Search PubMed.
- K. Yoshikawa, M. Furuno, N. Tanaka and E. Fukusaki, J. Biosci. Bioeng., 2020, 130, 437–442 CrossRef CAS PubMed.
- C. Yuan, W. Jia, Z. Yu, Y. Li, M. Zi, L. M. Yuan and Y. Cui, J. Am. Chem. Soc., 2022, 144, 891–900 Search PubMed.
- J. Y. Sung, S. M. Jin, S. Lee, S. Y. An and J. S. Jin, Talanta, 2021, 235, 122739 Search PubMed.
- Y. Nakano, M. Taniguchi and E. Fukusaki, J. Biosci. Bioeng., 2019, 127, 520–527 CrossRef CAS PubMed.
- L. Miller and L. Yue, Chirality, 2020, 32, 981–989 CrossRef CAS PubMed.
- Y. Konya, Y. Izumi, K. Hamase and T. Bamba, J. Chromatogr. A, 2022, 1677, 463305 CrossRef CAS PubMed.
- S. Lee, S. J. Kim, E. Bang and Y. C. Na, J. Chromatogr. A, 2019, 1586, 128–138 CrossRef CAS PubMed.
- F. E. O. Suliman, S. K. Al Burtomani, A. A. Elbashir and O. J. Schmitz, Electrophoresis, 2021, 42, 1800–1809 Search PubMed.
- K. Tsubaki, D. Tanima, M. Nuruzzaman, T. Kusumoto, K. Fuji and T. Kawabata, J. Org. Chem., 2005, 70, 4609–4616 Search PubMed.
- Y. Chen and M. T. Rodgers, J. Am. Chem. Soc., 2012, 134, 5863–5875 CrossRef CAS PubMed.
- Y. Chen and M. T. Rodgers, J. Am. Chem. Soc., 2012, 134, 2313–2324 CrossRef CAS PubMed.
- M. Kruppa, C. Mandl, S. Miltschitzky and B. König, J. Am. Chem. Soc., 2005, 127, 3362–3365 CrossRef CAS PubMed.
- Á. Golcs, B. Á. Ádám, V. Horváth, T. Tóth and P. Huszthy, Molecules, 2020, 25, 2571 CrossRef PubMed.
- O. H. Rubio, R. Taouil, F. M. Muñiz, L. M. Monleón, L. Simón, F. Sanz and J. R. Morán, Org. Biomol. Chem., 2017, 15, 477–485 Search PubMed.
- H. Ma, D. Luo, Q. Zhao, R. Liu, Z. Zhang, X. Hou, X. Sun and Y. Wang, J. Mol. Liq., 2022, 345, 117884 Search PubMed.
- Q. Y. Feng, S. Tong, S. Z. Vatsadze and M. X. Wang, Tetrahedron, 2023, 131, 133207 CrossRef CAS.
- Y. C. Rodriguez, T. M. Duarte, Z. Szakonyi, E. Forrõ, F. Fülöp and T. J. Wenzel, Chirality, 2015, 27, 708–715 Search PubMed.
- J. R. Avilés-Moreno, G. Berden, J. Oomens and B. Martínez-Haya, Phys. Chem. Chem. Phys., 2018, 20, 4067–4073 Search PubMed.
- J. R. Avilés-Moreno, G. Berden, J. Oomens and B. Martínez-Haya, Phys. Chem. Chem. Phys., 2017, 19, 31345–31351 Search PubMed.
- C. P. McNary, Y. W. Nei, P. Maitre, M. T. Rodgers and P. B. Armentrout, Phys. Chem. Chem. Phys., 2019, 21, 12625–12639 RSC.
- D. Jagleniec, N. Walczak, Ł. Dobrzycki and J. Romański, Int. J. Mol. Sci., 2021, 22, 10754 CrossRef CAS PubMed.
- P. Dominique, M. Schnurr and B. Lewandowski, Chem. Commun., 2021, 57, 3476–3479 RSC.
- A. Weißenstein, C. R. Saha-Möller and F. Würthner, Chem. – Eur. J., 2018, 24, 8009–8016 CrossRef PubMed.
- F. Oukhatar, S. Même, W. Même, F. Szeremeta, N. K. Logothetis, G. Angelovski and É. Tóth, ACS Chem. Neurosci., 2015, 6, 219–225 Search PubMed.
- F. Oukhatar, H. Meudal, C. Landon, N. K. Logothetis, C. Platas-Iglesias, G. Angelovski and É. Tóth, Chem. – Eur. J., 2015, 21, 11226–11237 CrossRef CAS PubMed.
- N. F. Atta, A. Galal and Y. M. Ahmed, J. Electrochem. Soc., 2019, 166, B623–B630 CrossRef.
- N. F. Atta, Y. M. Ahmed and A. Galal, Electroanalysis, 2019, 31, 1204–1214 Search PubMed.
- Y. Luo, N. Marets and T. Kato, Chem. Sci., 2018, 9, 608–616 Search PubMed.
- F. Otis, M. Auger and N. Voyer, Acc. Chem. Res., 2013, 46, 2934–2943 Search PubMed.
- J. Shen, R. Ye and H. Zeng, Angew. Chem., Int. Ed., 2021, 60, 12924–12930 Search PubMed.
- Y. Li, J. Dong, W. Gong, X. Tang, Y. Liu, Y. Cui and Y. Liu, J. Am. Chem. Soc., 2021, 143, 20939–20951 Search PubMed.
- P. A. Paquet-Côté, J. P. Paradis, M. Auger and N. Voyer, Biochim. Biophys. Acta, Biomembr., 2020, 1862, 183261 CrossRef PubMed.
- M. Lehmann, P. Weinberger, M. Finze, G. A. Giffin, M. Weller, S. Bachmann and A. C. Pöppler, J. Mater. Chem. C, 2024, 12, 7233–7239 RSC.
- V. Conejo-Rodríguez, C. Cuerva, R. Schmidt, M. Bardají and P. Espinet, J. Mater. Chem. C, 2019, 7, 663–672 Search PubMed.
- T. Wöhrle, I. Wurzbach, J. Kirres, A. Kostidou, N. Kapernaum, J. Litterscheidt, J. C. Haenle, P. Staffeld, A. Baro, F. Giesselmann and S. Laschat, Chem. Rev., 2016, 116, 1139–1241 Search PubMed.
- I. Carrasco, P. Ehni, M. Ebert, N. Dumait, G. Taupier, M. Amela-Cortes, C. Roiland, S. Cordier, J. A. Knöller, E. Jacques, S. Laschat and Y. Molard, ACS Appl. Mater. Interfaces, 2023, 15, 39752–39764 Search PubMed.
- P. Staffeld, M. Kaller, P. Ehni, M. Ebert, S. Laschat and F. Giesselmann, Crystals, 2019, 9, 74 CrossRef.
- K. Guy, P. Ehni, S. Paofai, R. Forschner, C. Roiland, M. Amela-Cortes, S. Cordier, S. Laschat and Y. Molard, Angew. Chem., Int. Ed., 2018, 57, 11692–11696 CrossRef CAS PubMed.
- J. Kirres, K. Schmitt, I. Wurzbach, F. Giesselmann, S. Ludwigs, M. Ringenberg, A. Ruff, A. Baro and S. Laschat, Org. Chem. Front., 2017, 4, 790–803 RSC.
- P. Ehni, K. Guy, M. Ebert, S. Beardsworth, K. Bader, R. Forschner, A. Bühlmeyer, N. Dumait, C. Roiland, Y. Molard and S. Laschat, Dalton Trans., 2018, 47, 14340–14351 RSC.
- J. Li, Z. He, H. Gopee and A. N. Cammidge, Org. Lett., 2010, 12, 472–475 Search PubMed.
- U. Beginn, G. Zipp and M. Möller, Adv. Mater., 2000, 12, 510–513 CrossRef CAS.
- U. Beginn, G. Zipp and M. Möller, Chem. – Eur. J., 2000, 6, 2016–2023 CrossRef CAS PubMed.
- U. Beginn, G. Zipp, A. Mourran, P. Walther and M. Möller, Adv. Mater., 2000, 12, 513–516 CrossRef CAS.
- G. Johansson, V. Percec, G. Ungar and D. Abramic, J. Chem. Soc., Perkin Trans. 1, 1994, 447 RSC.
- G. Ungar, S. V. Batty, V. Percec, J. Heck and G. Johansson, Adv. Mater. Opt. Electron., 1994, 4, 303–313 CrossRef CAS.
- V. Percec, G. Johansson, J. Heck, G. Ungarb and S. V. Battyb, J. Chem. Soc., Perkin Trans. 1, 1993, 1411 RSC.
- T. Basova, F. Latteyer, D. Atilla, A. G. Gürek, A. Hassan, V. Ahsen, H. Peisert and T. Chassè, Thin Solid Films, 2010, 518, 5745–5752 Search PubMed.
- C. W. Ong, C. Q. Yan, M.-C. Yeh and M.-C. Tzeng, Mol. Cryst. Liq. Cryst., 2015, 610, 249–254 CrossRef CAS.
- B. Zhang, J. Wang, Z.-Q. Yu, S. Yang, A.-C. Shi and E.-Q. Chen, J. Mater. Chem. C, 2014, 2, 5168 RSC.
- V. Percec, G. Zipp, G. Johansson, U. Beginn and M. Möller, Macromol. Chem. Phys., 1997, 198, 265–277 CrossRef CAS.
- U. Beginn, G. Zipp, M. Möller, G. Johansson and V. Percec, Macromol. Chem. Phys., 1997, 198, 2839–2852 CrossRef CAS.
- M. M. Neidhardt, M. Wolfrum, S. Beardsworth, T. Wöhrle, W. Frey, A. Baro, C. Stubenrauch, F. Giesselmann and S. Laschat, Chem. – Eur. J., 2016, 22, 16494–16504 CrossRef CAS PubMed.
- K. Bader, M. M. Neidhardt, T. Wöhrle, R. Forschner, A. Baro, F. Giesselmann and S. Laschat, Soft Matter, 2017, 13, 8379–8391 RSC.
- M. M. Neidhardt, K. Schmitt, A. Baro, C. Schneider, U. Bilitewski and S. Laschat, Phys. Chem. Chem. Phys., 2018, 20, 20371–20381 RSC.
- M. A. Grunwald, S. E. Hagenlocher, L. Turkanovic, S. M. Bauch, S. B. Wachsmann, L. A. Altevogt, M. Ebert, J. A. Knöller, A. R. Raab, F. Schulz, M. A. Kolmangadi, A. Zens, P. Huber, A. Schönhals, U. Bilitiewski and S. Laschat, Phys. Chem. Chem. Phys., 2023, 25, 17639–17656 RSC.
- Z. Li, A. Raab, M. A. Kolmangadi, M. Busch, M. Grunwald, F. Demel, F. Bertram, A. V. Kityk, A. Schönhals, S. Laschat and P. Huber, ACS Nano, 2024, 18, 14414–14426 CrossRef CAS PubMed.
- Z. Huang, J. Zhang, Y. Liu, A. Song and J. Hao, J. Mol. Liq., 2020, 301, 112399 CrossRef CAS.
- S. Fujiwara, H. Ohno, M. Yoshio, T. Kato and T. Ichikawa, Bull. Chem. Soc. Jpn., 2018, 91, 1–5 Search PubMed.
- S. Fujiwara, H. Ohno and T. Ichikawa, Mol. Syst. Des. Eng., 2018, 3, 668–676 RSC.
- K. Fujimura, T. Ichikawa, M. Yoshio, T. Kato and H. Ohno, Chem. – Asian J., 2016, 11, 520–526 CrossRef CAS PubMed.
- T. Ichikawa, K. Fujimura, M. Yoshio, T. Kato and H. Ohno, Chem. Commun., 2013, 49, 11746 RSC.
- S. Z. Mohammady, M. Pouzot and R. Mezzenga, Biophys. J., 2009, 96, 1537–1546 CrossRef CAS PubMed.
- R. Mezzenga, J. M. Seddon, C. J. Drummond, B. J. Boyd, G. E. Schröder-Turk and L. Sagalowicz, Adv. Mater., 2019, 31, 1900818 CrossRef PubMed.
- M. A. Kolmangadi, A. R. Raab, P. Szymoniak, Z. Li, P. Huber, S. Laschat and A. Schönhals, Phys. Chem. Chem. Phys., 2025, 27, 18162–18178 RSC.
- M. A. Kolmangadi, A. Yildirim, K. Sentker, M. Butschies, A. Bühlmeyer, P. Huber, S. Laschat and A. Schönhals, J. Mol. Liq., 2021, 330, 115666 CrossRef CAS.
- C. J. Pedersen, Angew. Chem., 1988, 100, 1053–1059 CrossRef CAS.
- L. X. Dang, J. Am. Chem. Soc., 1995, 117, 6954–6960 CrossRef CAS.
- G. J. Yuan, Y. T. Luan, J. Bin Miao, C. F. Wang, L. Chen, C. Chen, H. Chen, L. Li and X. M. Ren, Eur. J. Inorg. Chem., 2023, e202300523 CrossRef CAS.
- J. W. Steed, Coord. Chem. Rev., 2001, 215, 171–221 CrossRef CAS.
- R. G. Pearson, Inorg. Chim. Acta, 1995, 240, 93–98 CrossRef CAS.
- P. W. Ayers, M. Mohamed and F. Heidar-Zadeh, Conceptual Density Functional Theory: Towards a New Chemical Reactivity Theory, Wiley-VCH, Weinheim, 2022, vol. 2 Search PubMed.
- C. Haege, S. Jagiella and F. Giesselmann, Chem. Phys. Chem., 2019, 20, 2466–2472 CrossRef CAS PubMed.
- G. W. Stewart, Phys. Rev., 1928, 32, 558–563 CrossRef CAS.
- M. D. Hanwell, D. E. Curtis, D. C. Lonie, T. Vandermeersch, E. Zurek and G. R. Hutchison, J. Cheminform., 2012, 4, 17 CrossRef CAS PubMed.
- N. Kapernaum, C. Müller, S. Moors, M. C. Schlick, E. Wuckert, S. Laschat and F. Giesselmann, Chem. Phys. Chem., 2016, 17, 4116–4123 CrossRef CAS PubMed.
- A. De Vries, Mol. Cryst. Liq. Cryst., 1970, 11, 361–383 Search PubMed.
- A. De Vries, Mol. Cryst. Liq. Cryst., 1977, 41, 27–31 CrossRef CAS.
- K. Bader, C. Müller, Y. Molard, A. Baro, P. Ehni, J. Knelles and S. Laschat, RSC Adv., 2020, 10, 23999–24016 RSC.
- F. Schulz, B. Wank, P. Nacke, W. Frey and S. Laschat, Mater. Adv., 2023, 4, 1306–1313 RSC.
- F. Schulz, B. Lutz, D. Rück, D. Batman, W. Frey and S. Laschat, Soft Matter, 2023, 19, 2397–2406 RSC.
- D. R. Rueda, A. Nogales, J. J. Hernández, M. C. García-Gutiérrez, T. A. Ezquerra, S. V. Roth, M. G. Zolotukhin and R. Serna, Langmuir, 2007, 23, 12677–12681 CrossRef CAS PubMed.
- L. Yuan, J. Zhu, S. Wu and C. Chi, Chem. Commun., 2022, 58, 1302–1305 RSC.
- P. Davidson and L. Strzelecki, Liq. Cryst., 1988, 3, 1583–1595 CrossRef CAS.
- D. M. Smilgies, Rev. Sci. Instrum., 2002, 73, 1706–1710 CrossRef CAS.
- S. Aeffner, T. Reusch, B. Weinhausen and T. Salditt, Eur. Phys. J. E., 2009, 30, 205–214 CrossRef CAS PubMed.
- S. Laschat, A. Baro, N. Steinke, F. Giesselmann, C. Hägele, G. Scalia, R. Judele, E. Kapatsina, S. Sauer, A. Schreivogel and M. Tosoni, Angew. Chem., Int. Ed., 2007, 46, 4832–4887 CrossRef CAS PubMed.
- E. Grelet, S. Dardel, H. Bock, M. Goldmann, E. Lacaze and F. Nallet, Eur. Phys. J. E., 2010, 31, 343–349 CrossRef CAS PubMed.
- V. Percec, C. M. Mitchell, W. D. Cho, S. Uchida, M. Glodde, G. Ungar, X. Zeng, Y. Liu, V. S. K. Balagurusamy and P. A. Heiney, J. Am. Chem. Soc., 2004, 126, 6078–6094 CrossRef CAS PubMed.
- J. Xu, ACS Mater. Au, 2023, 3, 450–455 CrossRef CAS PubMed.
- K. V. Axenov and S. Laschat, Materials, 2010, 4, 206–259 CrossRef PubMed.
- L. Douce, J. M. Suisse, D. Guillon and A. Taubert, Liq. Cryst., 2011, 38, 1653–1661 CrossRef CAS.
- T. Kato and M. Yoshio, Liquid Crystalline Ionic Liquids, in Electrochemical Aspects of Ionic Liquids, ed. H. Ohno, John Wiley & Sons, Hoboken, 2011, pp. 307–320 Search PubMed.
- S. Chen and S. H. Eichhorn, Isr. J. Chem., 2012, 52, 830–843 CrossRef CAS.
- V. Causin and G. Saielli, Ionic Liquid Crystals, in Green Solvents II: Properties and Applications of Ionic Liquids, ed. A. Mohammad and D. Inamuddin, Springer, Dordrecht, 2012, pp. 79–118 Search PubMed.
- M. Mansueto and S. Laschat, Ionic Liquid Crystals, in Handbook of Liquid Crystals, ed. J. W. Goodby, C. Tschierske, P. Raynes, H. Gleeson, T. Kato and P. J. Collings, Wiley-VCH, Weinheim, 2014, vol. 6 Search PubMed.
- K. Goossens, K. Lava, C. W. Bielawski and K. Binnemans, Chem. Rev., 2016, 116, 4643–4807 CrossRef CAS PubMed.
- S. J. Devaki and R. Sasi, Ionic Liquids/Ionic Liquid Crystals for Safe and Sustainable Energy Storage Systems, in Progress and Developments in Ionic Liquids, ed. S. Handy, Intech, 2017 Search PubMed.
- V. Cîrcu, Ionic Liquid Crystals Based on Pyridinium Salts, in Progress and Developments in Ionic Liquids, ed. S. Handy, Intech, 2017 Search PubMed.
- T. Ichikawa, T. Kato and H. Ohno, Chem. Commun., 2019, 55, 8205–8214 Search PubMed.
- K. Salikolimi, A. A. Sudhakar and Y. Ishida, Langmuir, 2020, 36, 11702–11731 CrossRef CAS PubMed.
- K. Sentker, A. Yildirim, M. Lippmann, A. W. Zantop, F. Bertram, T. Hofmann, O. H. Seeck, A. V. Kityk, M. G. Mazza, A. Schönhals and P. Huber, Nanoscale, 2019, 11, 23304–23317 RSC.
- A. V. Kityk, M. Wolff, K. Knorr, D. Morineau, R. Lefort and P. Huber, Phys. Rev. Lett., 2008, 101, 187801 CrossRef PubMed.
- C. Tschierske, Angew. Chem., Int. Ed., 2013, 52, 8828–8878 CrossRef CAS PubMed.
- P. G. de Gennes and J. Prost, The physics of liquid crystals, Clarendon Press, 2001 Search PubMed.
- S. Das, A. Mondal and C. M. Reddy, Chem. Soc. Rev., 2020, 49, 8878–8896 Search PubMed.
- S. Havriliak and S. Negami, J. Polym. Sci. C, Polym. Symp., 1966, 14, 99–117 CrossRef.
- M. Wübbenhorst and J. van Turnhout, J. Non-Cryst. Solids, 2002, 305, 40–49 CrossRef.
- A. Schönhals and F. Kremer, Analysis of Dielectric Spectra, in Broadband Dielectric Spectroscopy, ed. F. Kremer and A. Schönhals, Springer, Berlin, Heidelberg, 2003, pp. 59–98 Search PubMed.
- O. Van Den Berg, W. G. F. Sengers, W. F. Jager, S. J. Picken and M. Wübbenhorst, Macromolecules, 2004, 37, 2460–2470 Search PubMed.
- A. Yildirim, C. Krause, P. Huber and A. Schönhals, J. Mol. Liq., 2022, 358, 119212 Search PubMed.
- A. Yildirim, M. A. Kolmangadi, A. Buhlmeyer, P. Huber, S. Laschat and A. Schönhals, J. Phys. Chem. B, 2020, 124, 8728–8739 CrossRef CAS PubMed.
- M. A. Kolmangadi, G. J. Smales, L. Zhuoqing, A. Yildirim, E. Wuckert, S. Eutionnat, F. Demel, P. Huber, S. Laschat and A. Schönhals, J. Phys. Chem. C, 2022, 126, 10995–11006 CrossRef CAS.
- H. Vogel, Phys. Z., 1921, 22, 645–646 CAS.
- G. S. Fulcher, J. Am. Ceram. Soc., 1925, 8, 339–355 CrossRef CAS.
- G. Tammann and W. Hesse, Z. Anorg. Allg. Chem., 1926, 156, 245–257 CrossRef CAS.
Footnote |
| † Dedicated to Professor Gerhard Erker on the occasion of his 80th birthday. |
| This journal is © The Royal Society of Chemistry 2026 |