Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Quasi-one-dimensional magnetism of transition-metal oxide in Fe-based inorganic–organic hybrid nanosheets
Takayuki
Nakane
 *a,
Takashi
Naka
ba,
Kazuyoshi
Sato
c,
Noriki
Terada
a,
Pascal
Manuel
d,
Ahmed
Ibrahim
e,
Shiro
Kubuki
*a,
Takashi
Naka
ba,
Kazuyoshi
Sato
c,
Noriki
Terada
a,
Pascal
Manuel
d,
Ahmed
Ibrahim
e,
Shiro
Kubuki
 e,
Chiya
Numako
f,
Dimitry
Khalyavin
e,
Chiya
Numako
f,
Dimitry
Khalyavin
 d,
Anne
de Visser
g and
Hiroya
Abe
h
d,
Anne
de Visser
g and
Hiroya
Abe
h
aNational Institute for Materials Science, 1-2-1, Sengen, Tsukuba, Ibaraki 305-0047, Japan
bUniversity of the Ryukyus, 1 Senbaru, Nishihara-cho, Nakagami-gun, Okinawa 903-0213, Japan
cGunma University, 1-5-1 Tenjin-cho, Kiryu, Gunma 376-8515, Japan
dISIS Facility, STFC Rutherford Appleton Laboratory, Chilton, Didcot, Oxfordshire OX11 0QX, UK
eTokyo Metropolitan University, 1-1 Minami-Osawa, Hachi-Oji, Tokyo 192-0397, Japan
fGraduate School of Science, Chiba University, Chiba 263-8522, Japan
gUniversity of Amsterdam, Science Park 904, 1098 XH, The Netherlands
hOsaka University, 11-1 Mihogaoka, Ibaraki, Osaka 567-0047, Japan
First published on 10th February 2026
Abstract
Low dimensionality provides an exciting research field for investigating quantum effects in functional materials. This work reports a novel quasi one-dimensional (1D) spin system configured in an inorganic–organic hybrid nanosheet. This nanosheet was solvothermally synthesized from FeCl3, CH3COOK, and ethylene glycol (C2H6O2) as the solvent. The minimum chemical composition of this hybrid nanosheet is Fe7O4.77(C2H4O2)9, and the quantum magnetic properties arise from a quasi-1D spin system of Fe3+. Remarkably, this quasi-1D antiferromagnetism exhibits a critical temperature at 21 K, implying a relatively low magnetic fluctuation state compared with an ideal 1D spin system. The unique characteristics of this novel inorganic–organic hybrid nanosheet are therefore considered attractive for tailoring novel low-dimensional quantum devices.
1. Introduction
Low-dimensional materials provide an exciting research platform for investigating functional quantum phenomena,1–9 such as superconductivity,1,2 metal–insulator transitions,3 and multistage magnetic ordering.4 The unique characteristics and potential of one-dimensional (1D) materials were actively predicted in the early stages of modern materials science.10–14 However, the fabrication of 1D materials and the detection of their distinctive features were challenging, although recent advances in nanotechnology have drastically boosted this research field.Quantum properties generally arise in nanoscale regions. Consequently, the functional signal from an individual quantum region is typically weak. Therefore, the effective alignment of quantum regions to bundle these weak responses is of paramount importance for investigating the functionality of 1D materials. To address this issue, fabrication techniques of hybrid nanomaterials have recently come to the fore as effective approaches to tailor novel quantum materials.15–18 A hybrid nanomaterial is generally defined as a compound consisting of a nanostructured matrix within a framework of organic molecules.19,20 Thus, it is not a simple mixture of components; rather, the constituents form an original crystal structure through chemical bondings.
Here, we report the discovery of a novel hybrid material in a precursor nanosheet used for synthesizing shape-controlled Fe3O4 microcrystals.21 The uniqueness of this hybrid nanosheet resides in its quasi-1D properties, displaying a phase transition that originates from the distinctive structure of the iron oxide within the material. The crystal structure remains to be clarified. This work precisely characterizes this novel hybrid nanosheet involving a quasi-1D spin system of Fe3+ (HNS-1DFe).
2. Experiments
2.1. Synthesis and fundamental characterization
HNS-1DFe was solvothermally synthesized from 0.5 M FeCl3 (96.0%, Kanto Chemical: Japan) and 3 M CH3COOK (97.0%, Kishida Chemical: Japan) in dehydrated anhydrous ethylene glycol (EG: 99.8%, Sigma-Aldrich: USA) as the solvent.21 These solutions were mixed, sealed into a reaction tube with a high allowable inner pressure, and heated at 180 °C for 2 h in an oil bath. After synthesis, the product was extracted, washed three times with EG using centrifugation under 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 G for 10 min, and finally dried for 1 week. This fabrication procedure reproducibly yielded a dark yellow nanosheet powder; however, the colour was slightly different among samples synthesized on different days. Therefore, all evaluations in this study were conducted using a single sample batch to maintain consistency in the discussion.
000 G for 10 min, and finally dried for 1 week. This fabrication procedure reproducibly yielded a dark yellow nanosheet powder; however, the colour was slightly different among samples synthesized on different days. Therefore, all evaluations in this study were conducted using a single sample batch to maintain consistency in the discussion.
The chemical composition of the product HNS-1DFe was evaluated by inductively coupled plasma optical emission spectroscopy (ICP-OES: Ultima-2, HORIBA: Japan) and organic elemental analysis (OEA: TruSpec Micro CHN, LECo: USA). Tht thermal stability of HNS-1DFe was verified by thermogravimetric analysis (TG: TGD-7000RH/SP, ULVAC: Japan) under flowing N2.
2.2. Morphology and structural characterizations
The morphology of HNS-1DFe was observed by field-emission scanning electron microscopy (FE-SEM: SU-8000, Hitachi High-Tech: Japan), and the crystal structure was verified from field-emission transmission electron microscopy (FE-TEM: JEM-2000F, Japan Electron: Japan) images acquired with an accelerating voltage of 200 kV.Spectroscopic structural analysis was also carried out using Fourier transform infrared spectroscopy (FT-IR: FT/IR-680 Plus, JASCO: Japan) with the specimen prepared as KBr pellets, and Raman spectroscopy (NanoFinder-1000, Tokyo Instrument: Japan) with a 532 nm excitation laser, respectively. The powder X-ray diffraction (XRD) pattern was measured using a conventional diffractometer (MiniFlex-600, RIGAKU: Japan) with copper Kα radiation. Powder Neutron diffraction (PND) measurements were conducted on a long-wavelength diffractometer (WISH, ISIS Neutron and Muon Source: UK), which covers long d-spacing values up to 100 Å in magnetic and large unit-cell systems.22
The main valence state of iron ions and their cationic configurations in HNS-1DFe were also evaluated by Fe K-edge X-ray absorption fine structure (XAS) spectra measured at BL-9A, photon factory, KEK, Japan, using the transmission method. Data processing of the X-ray absorption near-edge structure (XANES) region and extended X-ray absorption fine structure (EXAFS) region in the XAS spectra was carried out using the data processing program, ATHENA.23 Measurements of 57Fe Mössbauer spectra were carried out under constant acceleration mode. A γ-ray source of 57Co(Rh) (1.85 GBq, MCo7.124/74/20, Rietverc, verified on 02.12. 2020) was attached to the transducer, and α-Fe (30 µm thickness, MRA 2.6/30.20) was used as a reference for zero-velocity and velocity-scale calibration, with the sample placed in front of the proportional counter (45431, Niki-Kogei). For the measurements, 40 mg of the well-pulverized sample was shaped into a 10 mm diameter pellet and fixed on cellophane tape. The γ-ray from the source through the sample was detected by the proportional counter under an applied voltage of 2 kV supplied by a high-voltage power supply (556, ORTEC: USA).
2.3. Measurement of physical properties
The physical properties of HNS-1DFe were characterized in terms of dc-magnetization and specific heat by using a Magnetic Properties Measurement System (MPMS-XL, Quantum Design: USA) and a Physical Properties Measurement System (PPMS Dynacool, Quantum Design: USA), respectively. The magnetic contribution of Fe3+ ions in the specific heat, Cmag, was estimated as Cmag/T = [Cmol – Clattice]/T for HNS-1DFe. Here, Clattice denotes the lattice contribution to the specific heat, and the estimation procedure is given in the SI (SI-4). Then, the magnetic entropy, Smag(T), was calculated as follows.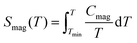 | (1) |
3. Results
3.1. Verification of the hybrid structure
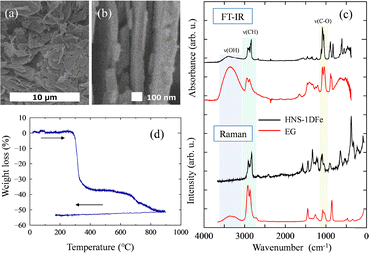
The solvothermal synthesis in this study yielded a dark yellow powder as the product (HNS-1DFe) with each particle in nanosheet form (see Fig. 1(a)). Details of this fundamental characterization are described in the SI (SI-1). The HNS-1DFe shows a flake-like morphology with a size of 1–5 µm. The thickness of HNS-1DFe can be estimated from Fig. 1(b) and is about 50–100 nm. Compositional analysis for HNS-1DFe revealed the minimum chemical composition as Fe7O4.77(C2H4O2)9. This result indicates the existence of an organic component, which is additionally verified by spectral analysis. Fig. 1(c) shows FT-IR and Raman spectra of HNS-1DFe and ethylene glycol (EG, C2H6O2) used as the solvent during synthesis. Most of the peaks above 800 cm−1 for HNS-1DFe can be assigned to EG, while several fine structures with peak splitting and small shifts in wavenumber are also observed. The difference between the peaks of HNS-1DFe and those of simple EG is attributed to the solidification of EG molecules.25 For example, absorption bands at around 3700, 2900 and 1060 cm−1 are assigned to strong stretching modes of OH, CH, and C–O, respectively, in the case of liquid-state EG. However, the strong absorbance of the OH stretching-vibration at ∼3700 cm−1 is diminished in HNS-1DFe. This indicates that HNS-1DFe includes EG-derived organic molecules lacking hydrogen atoms.Fig. 1(d) shows the weight loss of HNS-1DFe as a function of increasing and decreasing temperature. The weight loss starts at around 300 °C. This temperature is apparently higher than the boiling point of EG (≈ 198 °C), and it defines the phase formation point of Fe3O4 from HNS-1DFe.21 These observations lead to the conclusion that EG in HNS-1DFe does not exist as an adsorbed organic molecule but instead forms chemical bonds that allow its stabilization within the structure.
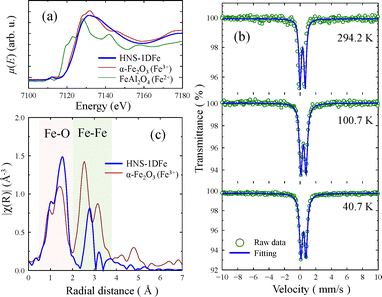
For characterizing the nanostructured matrix in HNS-1DFe, XAS and Mössbauer spectra were measured. The results are shown in Fig. 2. Fig. 2(a) compares the XANES spectrum of HNS-1DFe with those of α-Fe2O3 and FeAl2O4 measured as reference materials,26 indicating Fe3+ and Fe2+, respectively. Pre-edge peaks observed at around 7115 eV are due to 1s-to-3d forbidden transition of Fe K-edge absorption, and the position is almost the same for HNS-1DFe and α-Fe2O3. This peak position is known to depend on the valence state of iron ions.27,28 Hence, these data indicate that the valence state of Fe in HNS-1DFe is similar to that in α-Fe2O3 rather than FeAl2O4. This trend is also identified from the absorption edge of XANES spectra. The absorption edge of HNS-1DFe is almost identical to that of α-Fe2O3, which shows that the valence state of Fe ions are all 3+ with the spin state, S = 5/2, in HNS-1DFe. Mössbauer spectra shown in Fig. 2(b) further support this observation; Fe2+ was not identified. Analysis details are given in SI (SI-2).
Fig. 2(c) plots the radial distribution function against the radial distance with respect to the XAS spectra of Fe in HNS-1DFe and in that of α-Fe2O3. For α-Fe2O3, the origin of the first peaks (red area) is assigned to Fe–O distances (distances to the nearest-neighbour anions), and that of the second peaks (green area) is assigned to Fe–Fe distances (distances to the nearest-neighbour cations). This figure reveals that the radial distances of Fe–O bonds in α-Fe2O3 are almost the same as those between Fe and the nearest-neighbour-atom in HNS-1DFe, even though the densities differ. The average distance between Fe and its nearest-neighbour atoms is clearly shorter than the value in metallic iron (≈2.5 Å)29–31 and in iron-based complexes (≈2.0 Å).31–33 Therefore, the nanostructured matrix of HNS-1DFe is inferred to be an iron oxide.
3.2. Evaluation of low dimensionality of HNS-1DFe
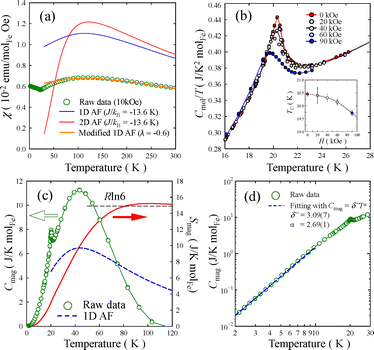
Temperature dependence of the magnetic susceptibility, χ(T), of HNS-1DFe is presented in Fig. 3(a) (see also SI-3 in SI). The field-cooled (FC) χ(T), measured in a field of 10 kOe, shows a round maximum at Tmax ≈ 120 K and a kink at around 21 K. The kink is indicative of a magnetic transition, while the broad maximum at higher temperature suggests magnetic order with low dimensionality of the spin system.34 Therefore, we attempted to fit the data with 1D and 2D models for antiferromagnetic (AF) spin systems by applying the Fisher classical model35 and the Lines model,36 respectively, instead of the conventional Curie–Weiss law, which is established for 3D materials. For the Fisher classical model, χ(T) of a 1D AF material, χ1D(T), was calculated from formulas (2) and (3).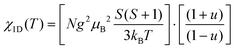 | (2) |
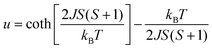 | (3) |
On the other hand, the Lines model calculates the χ(T) of 2D AF material, χ2D(T), by formulas (4) and (5).
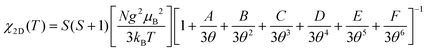 | (4) |
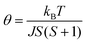 | (5) |
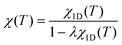 | (6) |
Fig. 3(a) shows the successful fit by the modified 1D model (orange line) obtained by applying formula (6) with λ ≈ −0.6 to χ1D(T) with J/kB = −13.6 K. These results indicate quasi-1D antiferromagnetism with the contribution of J′ in HNS-1DFe.
Low dimensionality of the spin system in HNS-1DFe was also verified by heat capacity measurements. First, Fig. 3(b) shows the specific heat divided by temperature, Cmol/T. A small peak appears at 20.4 K, corresponding to the critical transition temperature, TC, in Fig. 3(a). This peak temperature is slightly depressed by applying a magnetic field (see the inset in Fig. 3(b)). Next, the magnetic contribution of Fe3+ ions to the specific heat, Cmag, is plotted in Fig. 3(c). Remarkably, Cmag(T) exhibits a broad maximum at Tmax ≈ 44 K above the magnetic transition at TC. This indicates that the magnetic degrees of freedom of Fe3+ ions persist at high temperatures, T ≫ TC. Thus, the existence of strong AF spin correlations or low-dimensional spin fluctuations in the spin system is considered the origin of the broad maximum in Cmag(T). This point is verified by a theoretical calculation for the 1D AF spin system,38 and the computed line is drawn (bold dashed blue line) in Fig. 3(c). Here, theoretical calculations reproduce draw the Cmag(T) behaviour using formulas (7) and (8). Then, a2 = 11.097, a2 = 11.097, a3 = −0.8511, a4 = 0.1799, b1 = 0.0195, b2 = 0.5845, and γ = 1.8081 are used as the optimized parameters for S = 5/2.37
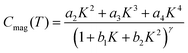 | (7) |
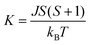 | (8) |
The model curve does not represent the experimental curve well, and this discrepancy is attributed to the existence of J′ between the 1D chains of the spin system in HNS-1DFe.
The magnetic entropy, Smag(T), is also plotted in Fig. 3(c). It is worth noting that Smag(T) reaches a saturation value of Smag = 15 J K−1 molFe−1 at temperatures as high as T = 100 K, which corresponds well to the theoretically expected value of Sspin = Rln(2S + 1) = 14.9 J K−1 molFe−1 for S = 5/2. This is consistent with the previous results.
On the other hand, the kink at 21 K is remarkable because such a transition phenomenon is not expected for a typical 1D AF spin system.38 This is further evidence of the intrinsic quasi-1D characteristics of the spin system in HNS-1DFe. The Cmag(T) at T ≪ TC is expected to follow a cubic power law, Cmag = δ″Tα (α ≈ 3).24 In our case, Cmag(T) follows the power law Cmag = δ″T2.69(1) with δ″ = 3.09(7) mJ K−2.69 below 10 K (Fig. 3(d)). The theoretical exponent is discussed as dmag/n,24 where dmag is the dimension of the spin system and n is an index indicating ferromagnetic (n = 2) or AF (n = 1) interactions between spins. That is, the exponent value of a ferromagnetic spin system should be 0.5–1.5 and that of a 3D AF system should be ≈3. An exponent value of 2.69(1) is much higher than 1.5 and clearly lower than 3. This result is in-line with a low-dimensional AF systems with the existence of J′ in the low-temperature phase of HNS-1DFe.
3.3. Structural characterization of HNS-1DFe
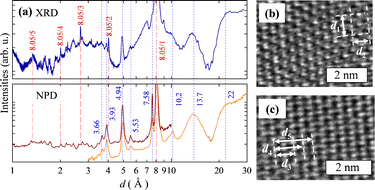
Experimental details and the raw data are presented in SI (SI-5). In Fig. 4(a), the XRD and PND patterns of HNS-1DFe are plotted against the lattice distance, d. This figure truncates the extremely intense peak observed at d ≈ 8.05 Å in order to focus on other small peaks. There are several sharp peaks in the XRD pattern, which can be assigned to fractional numbers of the main intense peak considered to originate from the iron oxide matrix (see the red line in Fig. 4(a)). This series of peaks can be indexed as 00l, with a lattice constant of ≈8.05 Å. On the other hand, both diffraction patterns display many broad peaks that can be attributed to the organic molecules in HNS-1DFe. However, assignment of these peaks is not as easy as that of the 00l peaks; hence, HNS-1DFe appears to belong to a crystal system with an axial angle ≠90°. Fig. 4(a) shows the position of these typical peaks to clarify the characteristic distances in HNS-1DFe.The nanosheet surface of HNS-1DFe was studied by TEM observation. The most frequently observed surface area is shown in Fig. 4(b), while Fig. 4(c) and the mesophases (speculated) between them were also observed in the surrounding regions. Gerber et al. reported a hybrid nanosheet of 1D iron oxide synthesized under similar conditions to those used for HNS-1DFe.39 Their hybrid nanosheet configures zebra array structure, in which organic molecules and 1D iron oxide align alternately along the nanosheet surface. However, our TEM observation couldn’t find this zebra array structure for HNS-1DFe. Instead, Fig. 4(b), which is a typical TEM image of HNS-1DFe, shows a hexagonal arrangement of atoms, where the observed atoms are considered to be Fe. The symbolic lengths described in Fig. 4(b) as d1 and d2 are estimated as 10 and 8 Å, respectively. (Note that it cannot be concluded that these lengths correspond to 8.05 Å and 10.2 Å, obtained from XRD and PND data. The atomic planes with d ≈ 8 Å have another atomic plane between them, which will scatter in anti-phase, making the intensity of the reflection at 8 Å zero.) In this case, the distance between two Fe atoms was about 5 Å, significantly longer than the typical distance (<4 Å) in iron oxides. Additionally, it is much longer than the calculated radial distance extracted from Fig. 2(c) (≈2.8 Å).
On the other hand, we were able to observe other views for HNS-1DFe. Fig. 4(c) shows that atoms form short-bond stacking while sliding their positions uniformly. In that case, distances of 8 Å, 14 Å, and 20 Å (≈8.05 Å, ≈13.7 Å, and <22 Å) corresponding to d3, d4, and d5 drawn in Fig. 4(c), respectively. This image is considered to relate to the periodicity of the 1D iron oxide chain.
4. Discussion
4.1. Low-dimensional structure of iron oxide
In the previous sections we have demonstrated that the spin system in HNS-1DFe orders with a quasi-1D nature. This is attributed to the magnetic moments of Fe3+ in the iron oxide matrix. Magnetism in transition-metal oxides is usually mediated by superexchange interaction; hence a 1D (or quasi-1D) network of the chemical bonds between Fe3+ and O2− should structurally exist in HNS-1DFe. However, this does not directly indicate the structural low dimensionality of iron oxide matrix in HNS-1DFe. This section discusses the structural low dimensionality of HNS-1DFe in more detail.This low dimensionality of iron oxide in HNS-1DFe is inferred from the Mössbauer data described in SI (SI-2). Here, the isomer shift, δ, of Fe3+ is relatively small (=0.316 mm s−1 at 294.2 K) compared to the typical value for Fe3+-based oxides. Based on the general understanding of the analysis of 57Fe Mössbauer spectra for 3D materials, this small δ suggests that Fe3+ in HNS-1DFe locally has a tetrahedral coordination rather than an octahedral one. However, this is not common for simple Fe3+ oxides. In contrast, the small δ value is reasonable if the iron oxide matrix has a low-dimensional configuration with a relatively small number of surrounding anions. This result indicates the structural low dimensionality of iron oxide in HNS-1DFe, since the origin of the magnetism is only Fe3+ in the case of this material. The Debye temperature, ΘD, calculated from the Mössbauer spectra also implies this trend. The calculated ΘD of HNS-1DFe is 245 K, consistent with the value ΘD = 237 K obtained from the fitting process of Cmol (see SI SI-2 and SI-4). These ΘD values are smaller than those of Fe2O3 (300–330 K)40 and suggest weaker bonds around Fe3+ in HNS-1DFe than those in Fe2O3 with a 3D structure.
On the other hand, Fig. 2(c) shows that the density of the distances to the nearest-neighbour cation (assigned as Fe–Fe distances) of Fe3+ in HNS-1DFe is smaller than that in α-Fe2O3. This indicates that the number of surrounding ions is relatively small in the case of Fe3+ in HNS-1DFe. This observation is in agreement with the structural low dimensionality of the iron oxide matrix in HNS-1DFe.
From these data, we consider that the magnetic low dimensionality of HNS-1DFe is strongly correlated with the structural uniqueness of the 1D iron oxide matrix.
4.2. Quasi-1D structure of iron oxide
The preceding section argues for the structural uniqueness of the 1D iron oxide matrix in HNS-1DFe. In general, a 1D network is assumed to be a continuous straight chain structure, and thus of the term “chain” is used to describe the 1D network for clarity. However, some experimental data of HNS-1DFe indicate the contribution of J′ to overall 1D magnetism. This requires further structural verification for the 1D network of Fe3+ and O2−. Therefore, this section examines the structure of the 1D network.The simplest structural model for the 1D iron oxide is a continuous straight chain (see the illustration in Fig. 5(a)). However, Fig. 2(c) indicates that the average distance between Fe3+ and O2− is about 1.55 Å. Then, the average distance between Fe3+ and Fe3+ is about 2.8 Å. Therefore, if one calculates a bond angle of Fe–O–Fe bonds, it is about 129°. This indicates that the 1D iron oxide in HNS-1DFe is not a straight chain like that shown in Fig. 5(a) but instead has at least a zigzag structure. Additionally, the chemical composition of HNS-1DFe would be expected to be Fe7O7(C2H4O2)9 in the case of a simple continuous chain. However, the oxygen content of the 1D iron oxide in HNS-1DFe is calculated as 4.77, which is considered smaller than expected. The oxygen content was not directly measured, and thus the accuracy is limited. There is the possibility that the residual oxygen content is 7 if we measured it exactly. However, further evidence for a residual oxygen content of 4.7 comes from considering the isolated 1D iron oxides in the inorganic–organic hybrid structure. For example, atomic ordering as shown in Fig. 5(b) may generate quasi-1D iron oxide with some magnetic frustration, and the chemical composition becomes Fe3O2. The chemical composition of Fe3O2 can correspond to Fe7O4.7 if the Fe content is calculated as 7, taking into account Fe7O4.77(C2H4O2)9 as the minimum chemical composition of HNS-1DFe. This possibility is therefore also reasonable for explaining the origin of the phase transition phenomenon at TC ≈ 21 K, since the quasi-1D iron oxide structure is expected to be more rigid than a simple chain. Fig. 5(b) is a 2D image drawn with 120° of Fe–O–Fe bonds, and HNS-1DFe involves organic molecules of EG in the hybrid structure; therefore, the real crystal structure is considered more complex. However, this speculation implies the possibility that the 1D iron oxides in HNS-1DFe form a quasi-1D structure rather than simple zigzag chains.
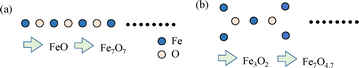 | ||
| Fig. 5 Simplified model of the 1D iron oxide structure in HNS-1DFe. (a) Continuous straight chain model. (b) Modified model with Fe3O2 composition. | ||
5. Summary
This investigation reports the discovery of HNS-1DFe as a novel hybrid nanosheet. Interestingly, the spin system of this material shows quasi-1D magnetism. This characteristic property was carefully verified through several experiments performed independently, and the obtained results were all consistent with the proposed structural model.Notably, the results indicate that the iron oxide matrix in HNS-1DFe has the potential to give rise to a phase-transition phenomenon, since its quasi-1D structure is expected to be more rigid than a simple 1D straight chain. Transition-metal oxides are widely known as effective platforms for investigating novel functional materials. Therefore, the rigid structure of iron oxide in HNS-1DFe is expected to pave the way for tailoring low-dimensional functional properties. HNS-1DFe is a hybrid nanomaterial, and the hybrid nanomaterial has an advantage for controlling the overall structure by arranging the organic molecules. The potential for tailoring novel functional devices using this nanosheet warrants a future detailed investigation of HNS-1DFe.
Author contributions
The manuscript was written through the contributions of all authors. T. Nakane, T. Naka and HA contributed equally to this work. Material preparations were performed by HA. Fundamental evaluations were exhibited by HA, T. Nakane and KS. Structural characterization and the analysis were exhibited by T. Nakane, NT, DDK and PM. Measurement of XAS spectra and the analysis was performed by CN and T. Naka. On the other hand, evaluation of the magnetism and the analysis was conducted by T. Naka and T. Nakane. Specific heat measurement and the analysis was exhibited and analysed by T. Naka and AdV. Mössbauer spectra were collected and analysed by AI and SK. Finally, all authors have approved the final version of the manuscript.Conflicts of interest
The authors declare that they have no competing interest.Data availability
All relevant data are within the manuscript and supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5tc03791c.Acknowledgements
This work was supported by a Grant-in-Aid for the Cooperative Research Project of Design & Engineering by Joint Inverse Innovation for Materials Architecture (DEJI2MA) of the Ministry of Education, Culture, Sports, Science and Technology (MEXT), and JSPS KAKENHI Grant Number (21H01637, 16K13999 and 24K08507). This work was conducted with experimental supports of the NIMS Electron Microscopy Unit and the NIMS Surface and Bulk Analysis Unit.References
- D. Jérome, A. Mazaud, M. Ribault and K. Bechgaard, Superconductivity in a synthetic organic conductor (TMTSF)2PF6, J. Phys., Lett., 1980, 41, L95–L98 CrossRef.
- M. Fujioka, M. Jeem, K. Sato, M. Tanaka, K. Morita, T. Shibuya, K. Takahashi, S. Iwasaki, A. Miura, M. Nagao, S. Demura, H. Sakata, M. Ono, H. Kaiju and J. Nishii, Intercalation on Transition Metal Trichalcogenides via a Quasi-Amorphous Phase with 1D Order, Adv. Funct. Mater., 2023, 33, 2208702 CrossRef CAS.
- F. Kagawa, T. Itou, K. Miyagawa and K. Kanoda, Transport criticality of the first-order Mott transition in the quasi-two-dimensional organic conductor k-(BEDT-TTF)2Cu[N(CN)2]Cl, Phys. Rev. B: Condens. Matter Mater. Phys., 2004, 69, 64511 CrossRef.
- Y. Shimizu, K. Miyagawa, K. Kanoda, M. Maesato and G. Saito, Spin Liquid State in an Organic Mott Insulator with a Triangular Lattice, Phys. Rev. Lett., 2003, 91, 107001 CrossRef CAS PubMed.
- A. Ishii and A. Miyasaka, A. Direct detection of circular polarized light in helical 1D perovskite-based photodiode, Sci. Adv., 2020, 6, eabd3274 CrossRef CAS PubMed.
- A. F. Santander-Syro, O. Copie, T. Kondo, F. Fortuna, S. Pailhès, R. Weht, X. G. Qiu, F. Bertran, A. Nicolaou, A. Taleb-Ibrahimi, P. Le Fèvre, G. Herranz, M. Bibes, N. Reyren, Y. Apertet, P. Lecoeur, A. Barthélémy and M. J. Rozenberg, Two-dimensional electron gas with universal subbands at the surface of SrTiO3, Nature, 2011, 469, 189–193 CrossRef CAS PubMed.
- S. Iijima, Helical microtubules of graphitic carbon, Nature, 1991, 354, 56–58 CrossRef CAS.
- H. Ishii, H. Kataura, H. Shiozawa, H. Yoshioka, H. Otsubo, Y. Takayama, T. Miyahara, S. Suzuki, Y. Achiba, M. Nakatake, T. Narimura, M. Higashiguchi, K. Shimada, H. Namatame and M. Taniguchi, Direct observation of Tomonaga–Luttinger-liquid state in carbon nanotubes at low temperatures, Nature, 2003, 426, 540–544 CrossRef CAS PubMed.
- A. K. Geim and K. S. Novoselov, The Rise of Graphene, Nat. Mater., 2007, 6, 183–191 CrossRef CAS PubMed.
- S. Tomonaga, Remarks on Bloch's Method of Sound Waves applied to Many-Fermion Problems, Prog. Theor. Phys., 1950, 5, 544–569 CrossRef.
- J. M. Luttinger, An Exactly Soluble Model of a Many-Fermion System, J. Math. Phys., 1963, 4, 1154–1162 Search PubMed.
- D. C. Mattis and E. H. Lieb, Exact Solution of a Many-Fermion System and Its Associated Boson Field, J. Math. Phys., 1965, 6, 304–312 CrossRef CAS.
- F. D. M. Haldane, Continuum dynamics of the 1-D Heisenberg antiferromagnet: Identification with the O(3) nonlinear Sigma model, Phys. Lett. A, 1983, 93, 464–468 Search PubMed.
- F. D. M. Haldane, Nonlinear Field Theory of Large-Spin Heisenberg Antiferromagnets: Semiclassically Quantized Solitons of the One-Dimensional Easy-Axis Néel State, Phys. Rev. Lett., 1983, 50, 1153–1156 Search PubMed.
- A. Kojima, K. Teshima, Y. Shirai and T. Miyasaka, Organometal Halide Perovskites as Visible-Light Sensitizers for Photovoltaic Cells, J. Am. Chem. Soc., 2009, 131, 6050–6051 CrossRef CAS PubMed.
- M. G. Yamada, H. Fujita and M. Oshikawa, Designing Kitaev Spin Liquids in Metal-Organic Frameworks, Phys. Rev. Lett., 2017, 119, 057202 Search PubMed.
- T. Takenaka, K. Ishihara, M. Roppongi, Y. Miao, Y. Mizukami, T. Makita, J. Tsurumi, S. Watanabe, J. Takeya, M. Yamashita, K. Torizuka, Y. Uwatoko, T. Sasaki, X. Huang, W. Xu, D. Zhu, N. Su, J. G. Cheng, T. Shibauchi and K. Hashimoto, Strongly correlated superconductivity in a copper-based metal-organic framework with a perfect kagome lattice, Sci. Adv., 2021, 7, eabf3996 Search PubMed.
- K. Wang, L. Jin, Y. Gao, A. Liang, B. P. Finkenauer, W. Zhao, Z. Wei, C. Zhu, T. F. Guo, L. Huang and L. Dou, Lead-Free Organic−Perovskite Hybrid Quantum Wells for Highly Stable Light-Emitting Diodes, ACS Nano, 2021, 15, 6316–6325 CrossRef CAS PubMed.
- M. Kondo, T. Yoshitomi, K. Seki, H. Matsuzaka and S. Kitagawa, Three-Dimensional Framework with Channeling Cavities for Small Molecules: {[M2(4, 4’-bpy)3(NO3)4]·xH2O}n (M = Co, Ni, Zn), Angew. Chem., Int. Ed. Engl., 1997, 36, 1725–1727 CrossRef CAS.
- H. Li, M. Eddaoudi, T. L. Groy and O. M. Yaghi, Establishing Microporosity in Open Metal−Organic Frameworks:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Gas Sorption Isotherms for Zn(BDC) (BDC = 1,4-Benzenedicarboxylate), J. Am. Chem. Soc., 1998, 120, 8571–8572 CrossRef CAS.
Gas Sorption Isotherms for Zn(BDC) (BDC = 1,4-Benzenedicarboxylate), J. Am. Chem. Soc., 1998, 120, 8571–8572 CrossRef CAS. - H. Abe, T. Naka, K. Sato, K. Suzuki and M. Nakano, Shape-Controlled Syntheses of Magnetite Microparticles and Their Magnetorheology, Int. J. Mol. Sci., 2019, 20, 3617–3627 CrossRef CAS PubMed.
- L. C. Chapon, P. Manuel, P. G. Radaelli, C. Benson, L. Perrott, S. Ansell, N. J. Rhodes, D. Raspino, D. Duxbury, E. Spill and J. Norris, Wish: The new powder and single crystal magnetic diffractometer on the second target station, Neutron News, 2011, 22, 22–25 Search PubMed.
- B. Ravel and M. Newville, ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT, J. Synchrotron. Rad., 2005, 12, 537–541 CrossRef CAS PubMed.
- L. J. de Jongh and A. R. Miedema, Experiments on simple magnetic model systems, Adv. Phys., 1974, 23, 1–260 CrossRef CAS.
- K. Krishman and R. S. Krishman, Raman and Infrared Spectra of Ethylene Glycol, Proc. Indian Acad. Sci. Sect. A, 1966, 64, 111–122 CrossRef.
- T. Nakane, S. Ishii, T. Naka and T. Uchikoshi, Influence of Fabrication Conditions on the Structural Characteristics and the Magnetic Properties of FeAl2O4, J. Am. Ceram. Soc., 2023, 106, 2317–2325 Search PubMed.
- M. Wilke, F. Farges, P.-E. Petit, G. E. Brown and F. Martin, Oxidation state and coordination of Fe in minerals: An Fe K-XANES spectroscopic study, Am. Mineral., 2001, 86, 714–730 CrossRef CAS.
- R. Murao, T. Harano, M. Kimura and I.-H. Jung, Thermodynamic Modeling of the SFCA Phase Ca2(Fe,Ca)6(Fe,Al,Si)6O20, ISIJ Int., 2018, 58, 259–266 CrossRef CAS.
- N. V. Ershov, Y. A. Babanov and V. R. Galakhov, EXAFS Study of Crystalline Cu, Ni, and Fe, Phys. Stat. Solidi B, 1983, 117, 749–753 CrossRef CAS.
- P. Srivastava, L. Lemke, H. Wende, R. Chauvistré, N. Haack, K. Baberschke, J. Hunter-Dunn, D. Arvanitis, N. Mårtensson, A. Ankudinov and J. J. Rehr, Magnetic extended x-ray absorption fine structure at the L3,2 edges of Fe and Co on Cu(001), J. Appl. Phys., 1998, 83, 7025–7027 CrossRef CAS.
- N. Parsai and A. Mishra, Study of XAFS of some Fe compounds and determination of first shell radial distance, J. Phys.: Conf. Ser., 1998, 836, 012045 Search PubMed.
- V. G. Vlasenko, A. T. Shuvaev, T. I. Nedoseikina, A. L. Nivorozhkin, A. I. Uraev, A. D. Garnovskii and O. Y. Korshunov, EXAFS studies of the novel iron(III) complexes with an N/S(Se) chromophore simulating ligand environment of the active site of nitrile hydratase, J. Synchrotron Rad., 1999, 6, 406–408 Search PubMed.
- Y. T. Chen, K. H. C. Chen and M. Fukuda, DFT Study for Evaluation of the Interraction of Coordinated Molecule to Fe(III) in Various Iron(III)Chloride Complexes, J. Comput. Chem. Jpn., 2017, 16, 1–16 Search PubMed.
- H. Lu, T. Yamamoto, W. Yoshimune, N. Hayashi, Y. Kobayashi, Y. Ajiro and H. A. Kageyama, Nearly Ideal One-Dimensional S = 5/2 Antiferromagnet FeF3(4,4-bpy) (4,4-bpy =4,4-bipyridyl) with Strong Intrachain Interactions, J. Am. Chem. Soc., 2015, 137, 9804–9807 Search PubMed.
- M. E. Fisher, Magnetism in One-Dimensional Systems - The Heisenberg Model for Infinite Spin, Am. J. Phys., 1964, 32, 343–346 CrossRef.
- M. E. Lines, The quadratic-layer antiferromagnet, J. Phys. Chem. Solids, 1970, 31, 101–116 CrossRef CAS.
- T. de Neef, A. J. M. Kuipers and K. Kopinga, Magnetic specific heat of isotropic linear chains for S ⩽ 5/2, J. Phys. A: Math., Nucl. Gen., 1974, 7, L171–L175 CrossRef CAS.
- E. Ijing, Beitrag zur Theorie des Ferromagnetismus, Z. Phys., 1925, 31, 253–258 CrossRef.
- O. Gerber, B. P. Pichon, D. Ihiawakrim, I. Florea, S. Moldovan, O. Ersen, D. Begin, J.-M. Grenèche, S. Lemonnier, E. Barraud and S. Begin-Colin, Synthesis engineering of iron oxide raspberry-shaped nanostructures, Nanoscale, 2017, 9, 305–313 RSC.
- A. Ibrahim, K. Tani, K. Hashi, B. Zhang, Z. Homonnay, E. Kuzmann, A. Bafti, L. Pavić, S. Krehula, M. Marciuš and S. Kubuki, Debye Temperature Evaluation for Secondary Battery Cathode of α-SnxFe1−xOOH Nanoparticles Derived from the 57Fe- and 119Sn-Mössbauer Spectra, Int. J. Mol. Sci., 2024, 25, 2488–2513 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |