Transient degradable electronics enabled by systems of conducting polymers and natural biopolymers
Xin
Sun
 ab,
David
Barker
ab,
David
Barker
 ab and
Jadranka
Travas-Sejdic
ab and
Jadranka
Travas-Sejdic
 *ab
*ab
aCentre for Innovative Materials for Health, School of Chemical Sciences, The University of Auckland, Auckland 1010, New Zealand. E-mail: j.travas-sejdic@auckland.ac.nz
bMacDiarmid Institute for Advanced Materials and Nanotechnology, Wellington 6140, New Zealand
First published on 5th December 2025
Abstract
Transient electronics hold great promise for reducing electronic waste and enabling advanced biomedical and environmental technologies. Achieving this requires conductive materials that combine reliable electrical performance with controlled degradability. Conducting polymer–biopolymer (CP–biopolymer) systems uniquely address this need by pairing the electroactivity of CPs with the inherent biodegradability, biocompatibility and functional versatility of natural polymers. Polysaccharides, polypeptides and polyphenols provide abundant functional groups for modification, crosslinking and grafting with CPs, while some, such as chitosan, also impart antibacterial activity. Transient CP–biopolymer systems have been reported in four main formats: blends, multilayer structures, hydrogels, and graft copolymers, each offering strategies to balance conductivity, processability and degradation. This review highlights recent progress in material design, outlines degradation pathways ranging from matrix to network disintegration, and surveys applications in neural interfaces, tissue engineering and skin-mounted sensors. Despite substantial progress, advancing transient CP–biopolymer systems toward next-generation implantable electronics will require deeper insight into their degradation mechanisms and associated immune responses, along with improved electrical stability, enhanced mechanical performance and scalable fabrication strategies. Critically, this progress must be accompanied by the development of novel chemistries that render conducting polymers intrinsically degradable.
1. Introduction
Transient electronics are an emerging class of materials engineered to disintegrate after a defined operational lifetime.13–15 Their controlled degradation is envisioned to enable devices that deliver reliable performance for a finite period and then disappear without leaving harmful residues. Applications range from implantable systems that eliminate the need for secondary surgeries16,17 to environmental sensors that function effectively before degrading in situ.25The transient electronics field was pioneered by Rogers and colleagues, who showed that monocrystalline silicon can undergo hydrolysis under aqueous conditions, though at a slow rate.30 This concept was later extended to dissolvable metals such as Mg and Mo, employed as conductive elements within degradable matrices.31,32 More recently, conducting polymers (CPs) have emerged as alternative conductive components.33–35 CPs combine metal-like conductivity with flexibility, processability and properties that can be tuned through molecular design.16,36,37 CP-based transient electronics have been developed by incorporating cleavable linkers or degradable segments into the polymer backbone38–42 and through integration with degradable polymers.36,43 Among these, CP integrated with naturally sourced biopolymers are particularly attractive, as biopolymers provide inherent biodegradability and biocompatibility, unlike many synthetic degradable polymers that may raise risks in vivo.44–47 Therefore, CP–biopolymer systems possess a unique advantage when employed in healthcare and biomedical fields.
Representative biopolymers include polysaccharides (e.g., cellulose, alginate, chitosan), polypeptides (e.g., gelatin, silk fibroin, elastin) and polyphenols (e.g., tannic acid derivatives).48 Their abundant functional groups, such as hydroxyl, carboxyl, amino and phenolic groups, allow for chemical modification, crosslinking and covalent grafting with CPs.36 In addition, some biopolymers provide inherent antibacterial activity; for example, chitosan disrupts microbial membranes through its cationic groups, offering added benefit for implantable and skin-contact devices.49 By integrating CPs with biopolymers, transient electronic systems can be engineered to degrade on demand while maintaining the electrical function of CPs. Such CP–biopolymer systems provide effective interfaces between electronics and living tissue, supporting long-term physiological monitoring and enabling the development of next-generation healthcare devices.36,37,50 It is worth noting that the degradability of CP–biopolymer electronics investigated to date originates solely from the biopolymer components, rather than the conducting polymers. The degradation performance of different CP–biopolymer systems can vary significantly, primarily due to the distinct interaction mechanisms between the conducting polymer and the biopolymer.
On this basis, the present review focuses on transient degradable electronics enabled by conducting polymers and natural biopolymers, addressing a gap left by earlier reviews that have largely emphasised synthetic degradable polymers.36,43 In contrast, natural CP–biopolymer-based transient systems remain comparatively underexplored. Here, we first outline material design strategies for transient polymer electronics, focusing on blends, multilayer structures, hydrogels and graft copolymers of biopolymers with CPs (Section 2). We then examine the degradation mechanisms of transient CP–biopolymer electronic systems (Section 3), followed by a summary of their applications in biomedical devices, wearable and environmental sensing and energy storage systems (Section 4). Finally, we provide perspectives on challenges and future opportunities in transient CP–biopolymer electronics.
2. Synthesis and fabrication strategies of CP–biopolymer systems
The main preparation strategies for transient polymer electronics involving CPs and biopolymers include blends, multilayer structures, hydrogels and graft copolymers of CPs and biopolymers, as illustrated in Fig. 1.2.1. Blends and multilayer structures of biopolymer and CPs
Blends and multilayer structures of conducting polymers (CPs) and biopolymers can be fabricated via direct physical mixing, surface deposition or in situ polymerisation of CPs in the presence of biopolymers (Fig. 1(A)). While biopolymers contribute desirable biodegradability, their inherently insulating nature often leads to reduced electrical conductivity in the resulting materials. Consequently, balancing degradability with adequate electrical performance remains a key challenge.Although blends and multilayer structures of biopolymer and CPs have contributed to the early development of CP–biopolymer transient systems, their degradation performance has not been thoroughly investigated. In these systems, overall degradability arises from the biopolymer component, which undergoes hydrolytic, enzymatic or oxidative cleavage. In contrast, conducting polymer components are intrinsically non-degradable, with their degradation limited to macroscale, such as physical disintegration or detachment from the biopolymer matrix.35,43
This section reviews recent advances in CP–biopolymer blends and multilayer structures, with a focus on their fabrication strategies, electrical properties and reported degradation behaviour.
Among these, cellulose has been widely employed in CP-based blend systems due to its abundance, renewability and mechanical robustness.48 In one example, as demonstrated in Fig. 2(A), seaweed-derived cellulose (s-cellulose), PEDOT:PSS and glycerol were combined to fabricate a thin film.9 This film reached a conductivity of 5.3 S cm−1, approximately one order of magnitude higher than pristine PEDOT:PSS (0.7 S cm−1), which was attributed to the glycerol-assisted chain rearrangement of PEDOT. This thin film also demonstrated biodegradability, with 50% mass loss observed after one week in soil and clear erosion at the film edges by the third week. It further showed effective Joule heating, reaching 51 °C at 1.5 V, indicating suitability for transient thermal applications. A similar cellulose/PEDOT:PSS blend incorporating glycerol and sodium alginate was developed by Engquist et al. as printable ink for flexible and patterned electronic papers via direct writing techniques (Fig. 2(B)).20
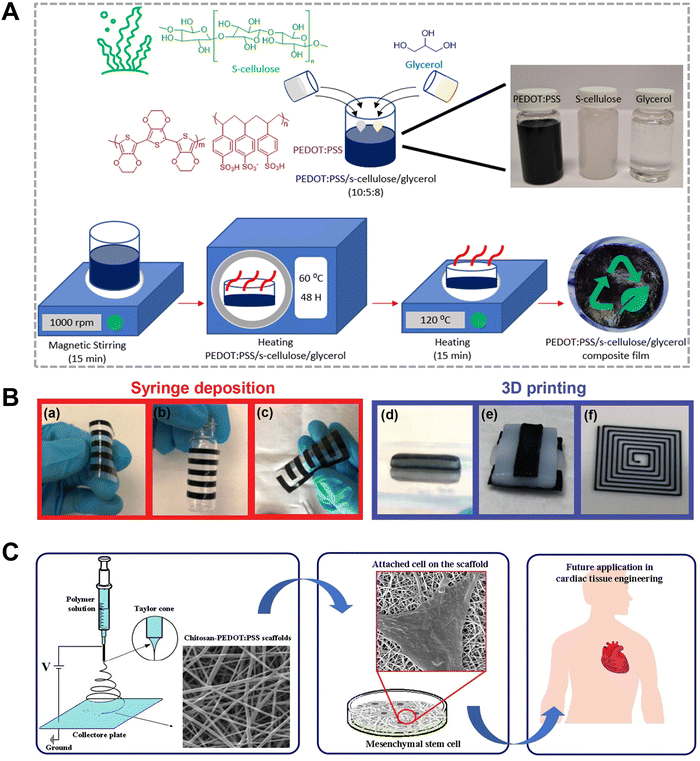 | ||
| Fig. 2 Direct blending of CPs with biopolymers. (A) Schematic representation of the fabrication of pedot:pss/s-cellulose/glycerol films, highlighting the chemical structures of PEDOT:PSS, s-cellulose and glycerol. Reproduced from the ref. 9 Licensed under CC BY-NC 3.0. (B) Direct ink writing of nanocellulose and PEDOT:PSS. (a)–(c) by syringe deposition: printed patterns. (d)–(f) 3D printing of structures. Reproduced from the ref. 20 Licensed under CC BY-NC 4.0. (C) Illustration of conductive nanofibrous chitosan/PEDOT:PSS as tissue engineering scaffolds. Reproduced from ref. 27 Copyright (2019), with permission from Elsevier. | ||
In addition, chitosan, a deacetylated derivative of chitin that is the second most abundant natural polymer, has also been extensively explored. Chitosan functions as a cationic polyelectrolyte under acidic conditions (pH 2–6).55 It has been extensively explored for applications in tissue engineering and as an antibacterial material. In one study, Narula et al. fabricated PEDOT:PSS/chitosan thin films by simply blending two components, with poly(vinyl alcohol) (PVA) added to enhance the mechanical properties.51 After drying at 50 °C for two days, the films exhibited a conductivity of 1.87 × 10−4 ± 0.04 S cm−1 (PEDOT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PSS
PSS![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) chitosan volume ratio = 1
chitosan volume ratio = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), substantially lower than that of pristine PEDOT:PSS (5.02 ± 0.02 S cm−1). This reduction was attributed to disrupted chain packing in the PEDOT:PSS matrix caused by the introduction of large amount of chitosan. The PEDOT:PSS/chitosan thin films demonstrated bactericidal activity against Gram positive bacteria, owing to the positively charged amino groups from chitosan, with a minimum inhibition growth concentration of 0.044 mg L−1. In another study, Tayebi et al. electrospun nanofiber scaffolds composed of chitosan/PVA/PEDOT:PSS blends, achieved a conductivity of 7.63 × 10−3 S m−1 (Fig. 2(C)).27 These scaffolds demonstrated enhanced mesenchymal stem cell adhesion and proliferation compared to the chitosan/PVA control group, indicating strong potential for tissue engineering.
3), substantially lower than that of pristine PEDOT:PSS (5.02 ± 0.02 S cm−1). This reduction was attributed to disrupted chain packing in the PEDOT:PSS matrix caused by the introduction of large amount of chitosan. The PEDOT:PSS/chitosan thin films demonstrated bactericidal activity against Gram positive bacteria, owing to the positively charged amino groups from chitosan, with a minimum inhibition growth concentration of 0.044 mg L−1. In another study, Tayebi et al. electrospun nanofiber scaffolds composed of chitosan/PVA/PEDOT:PSS blends, achieved a conductivity of 7.63 × 10−3 S m−1 (Fig. 2(C)).27 These scaffolds demonstrated enhanced mesenchymal stem cell adhesion and proliferation compared to the chitosan/PVA control group, indicating strong potential for tissue engineering.
While the aforementioned PEDOT:PSS systems are typically processed in aqueous environments, a further possibility is to formulate blends in organic solvents, particularly for non-water-dispersible CPs, such as polyaniline (PANI). One early report involved the preparation of PANI/gelatin blend nanofibers using 1,1,1,3,3,3-hexafluoro-2-propanol as the solvent.53 These fibers exhibited a conductivity of 0.021 S cm−1 and supported the adhesion, migration and proliferation of H9c2 rat cardiac myoblasts, confirming their biocompatibility and potential for cardiac tissue engineering. Similarly, PANI/chitosan52 and PANI/silk54 nanofibers were electrospun using trifluoroacetic acid/dichloromethane and formic acid, respectively. In particular, PANI/chitosan nanofibers have been investigated for wound healing applications, benefiting from the synergistic integration of hydrophilic chitosan and electroactive PANI.52
Spin-coating is a widely adopted and straightforward method to apply CPs onto biopolymer thin films. For instance, as shown in Fig. 3(A), Kim's research group developed a stretchable cellulose/PEDOT:PSS film by spin-coating PEDOT:PSS onto a cellulose substrate.6 To enhance its optical transmittance, they introduced 11-aminoundecanoic acid as a surface modifier. The use of conductivity enhancers and surfactants successfully reduced the film's sheet resistance to as low as 156 Ω sq−1. The resulting film exhibited durable stretchability, showing only a 21% increase in resistance after 100 stretch-release cycles. Notably, it dissolved completely in water within 15 minutes, confirming its degradability. These films were applied as stretchable electrodes in wearable healthcare sensors capable of detecting multiple physiological signals, such as from body movements, drinking, breathing and speaking, as well as temperature and humidity, with high sensitivity and low power consumption. In another example, PEDOT:PSS was spin-coated onto a polylactic acid (PLA) substrate derived from corn starch, followed by an additional PLA top layer via the same technique.56 The tri-layer structure was patterned by laser cutting, yielding breathable, stretchable electrodes with a sheet resistance of 150 Ω sq−1 (thickness: 80 nm). These were used to fabricate electrophysiological sensors for eye-movement monitoring, enabling human–machine interaction. While the above examples used drop-cast PLA or cellulose films as substrates, electrospun nanofiber scaffolds have also been explored. Du et al. coated chitosan nanofibers with PEDOT:PSS via spin-coating, resulting in a scaffold that promoted the growth of brain neuroglioma cells, with increased axon formation observed.17
 | ||
| Fig. 3 Coating CPs on a biopolymer matrix. (A) Spin-coating method. (a) Illustration of the cellulose/PEDOT:PSS hybrid film structure; (b) chemical structures of hydroxyethyl cellulose, glycerol and glucose; (c) schematic of the fabrication process for the cellulose/PEDOT:PSS hybrid film. Reprinted with permission from ref. 6 Copyright 2023 American Chemical Society. (B) Drop-casting method. PEDOT:PSS/PVA/Pullulan-coated Xuan paper applied as a sustainable electronic material for wearable dry ECG electrodes and flexible strain sensors. Reproduced from ref. 18 Copyright (2025), with permission from Elsevier. (C) Dip-coating method. Schematic representation of the one-step fabrication process for conductive RSF/PEDOT:PSS films. Reprinted with permission from ref. 19 Copyright 2022 American Chemical Society. | ||
Besides PEDOT:PSS, other polythiophene derivatives have been similarly applied. Bonard and colleagues spin-coated P3HT onto chitosan films and doped it with HAuCl4, leveraging the strong Au–S interaction to enhance hole injection.57 The conductivity of the doped P3HT-chitosan films ranged from 821.3 to 2017.4 S m−1. Similar strategies have been employed to fabricate biodegradable organic field-effect transistors (OFETs) by spin-coating polythiophene-based semiconductors onto biopolymer substrates such as chitosan58 and gelatin.59
Drop-casting and dip-coating offer equally simple alternatives for coating CPs onto biopolymer matrices. For example, a hybrid ink composed of PEDOT:PSS, PVA and pullulan was drop-cast onto Xuan paper (a cellulose-based substrate), yielding a flexible film with a low sheet resistance of 1.51 ± 0.013 Ω sq−1 and excellent mechanical stability under bending (Fig. 3(B)).60 These paper-based electrodes were used as strain sensors for detecting finger motion and as electrocardiography electrodes for monitoring cardiorespiratory signals.
As for dip-coating, regenerated silk fibroin (RSF) films were immersed in a PEDOT:PSS solution in H2O/ethanol cosolvent, Fig. 3(C).19 Post-treatment with the same cosolvent facilitated phase separation between PEDOT and PSS due to conformational transitions in RSF, which optimised the properties of these films. The resulting RSF/PEDOT:PSS films displayed high conductivity (1.003 S cm−1) and visible-light transmittance (>80%). After 4 weeks of enzymatic degradation in protease XIV solution (a commonly used enzyme to mimic proteolytic environments), over 20% weight loss was observed, indicating the partial disintegration of PEDOT:PSS along with the biodegradation of RSF substrate. Similarly, bacterial cellulose/PEDOT:PSS films were fabricated by the dip-coating method, with the assistance of nano-gold, which effectively supported the adhesion, growth and proliferation of MC3T3 cells.61
An inverse approach has also been explored, in which a biopolymer is coated onto a CP substrate. Miao et al. first spray-coated a multilayer film of single-wall carbon nanotube (SWCNT)/pristine graphene (PG)/PEDOT:PSS and then drop-cast an edible starch-chitosan solution over it.62 The integration of SWCNT and PG crosslinked with PEDOT chains results in low resistance and good flexibility. The resulting transparent and flexible electrode exhibited excellent optoelectronic performance, with a sheet resistance of 46 Ω sq−1 and 83.5% transmittance at 550 nm. It could be enzymatically degraded in just 8 minutes in a lysozyme solution at room temperature, leaving no toxic residues.
One of the earliest examples of in situ PPy polymerisation was reported by Wallace's group in 2009, where hyaluronic acid (HA) and chondroitin sulfate A (CS) were used as both matrix and dopant.63 The resulting conductive films enhanced myoblast adhesion and promoted their differentiation into multinucleated myofibres. This pioneering approach demonstrated the feasibility of incorporating extracellular matrix components directly into conductive polymers, yet the performance strongly depended on the choice of dopant and degradability were not addressed. Soon after, in 2010, Basavaraja et al. also reported a similar system PPy/alginate, in which alginate served both as the matrix and the dopant.64 The conductivity was observed to increase with higher alginate content in the blend, confirming the dopant function of alginate for PPy in the blend film. More recently, a PPy/alginate film was synthesised using silver nitrate as the oxidant and sodium dodecyl sulfate as the dopant. The resulting PPy/Ag/alginate film demonstrated a conductivity of 0.02 S cm−1, along with excellent anti-ultraviolet and antistatic properties.65 In addition to alginate, chitosan has also been explored as the matrix for in situ polymerisation of PPy. Typically, pre-formed chitosan thin films are immersed in pyrrole/oxidant solutions (e.g., FeCl3)8,66,67 or the polymerisation is directly carried out in chitosan solutions.68 Applications of these PPy/chitosan blends span electrochemical temperature sensors8,66 and supercapacitor electrodes.67 For instance, in Fig. 4(A), one study reported PPy/chitosan films with a conductivity of 4.55 S cm−1, which were utilised to investigate the influence of temperature on the reversible redox behaviour via cyclic voltammetry.8 Despite these advances, the degradation behaviour of CP–biopolymer blends remains largely unexplored.
 | ||
| Fig. 4 In situ formation of CPs in the presence of a biopolymer matrix. A. (a) Schematic illustration of the fabrication process for the PPy/CS hybrid film; (b) photograph of the pristine CS film; (c) photograph of the resulting PPy/CS hybrid film; (d) demonstration of the PPy/CS hybrid film's flexibility. Reproduced from the ref. 8 Licensed under CC BY-NC 3.0. (B) Schematic representation of the preparation process for BC and BC/PPy-P strips. Reproduced from ref. 10 Copyright (2025), with permission from Elsevier. (C) Schematic diagram illustrating the synthesis and fabrication of conductive CNF-PANI/NR hybrid elastomers with a hierarchical 3D network structure. Reproduced from ref. 22 Copyright (2019), with permission from Elsevier. | ||
Cellulose has also been explored as a matrix for in situ PPy polymerisation. In a recent study by Yuan et al. in 2025 (Fig. 4(B)), PPy nanoparticles were polymerised on bacterial cellulose nanofiber surfaces using p-toluenesulfonic acid (p-TSA) as the dopant, resulting in conductive PPy/bacterial cellulose microfibers.10 These microfibers exhibited a high conductivity of 7.19 S cm−1, excellent tensile strength of 210 MPa, and a Young's modulus of 2 GPa. When applied in resistive sensors for human motion detection, they showed fast response times (∼15 s) and remained stable over 1000 loading cycles. With regards to degradability, PPy/chitosan blends were shown to fully disintegrate within 96 hours in acidic cellulose solutions, with PPy particles precipitating and recoverable at the bottom of the solution.
Similarly, PANI was in situ polymerised within chitosan and cellulose matrices. For example, Mohammadi et al. prepared PANI/chitosan blends using potassium dichromate as the oxidant.69 The resulting spherical PANI particles (45–100 nm in diameter) coated the chitosan matrix, and the film's conductivity could be tuned by controlling PANI content and synthesis time. These films demonstrated antimicrobial and antifungal activities with inhibition zones of 187.27 mm2 (Escherichia coli) and 61.47 mm2 (Aspergillus niger), respectively. In another study on PANI/chitosan thin films, the incorporation of PANI into the chitosan matrix was found to decrease water solubility and vapor permeability while enhancing antioxidant activity, indicating their potential application in active food packaging.70
Further combinations include chitosan-modified graphene/PANI blends, where chitosan acts as a dispersing and stabilising agent for graphene oxide during PANI polymerisation.71 The resulting films, used as supercapacitor electrodes, showed improved capacitance and reduced graphene aggregation.
Cellulose nanofiber (CNF)/PANI blends have also been developed via in situ polymerisation of aniline on CNF surfaces, as shown in Fig. 4(C).22 These CNF/PANI structures were then blended with natural rubber (NR) latex through a latex co-coagulation process to form CNF/PANI/NR hybrid elastomers. These elastomers exhibited high conductivity (8.95 × 10−1 S m−1) along with excellent mechanical flexibility and stretchability, enabling their use in wearable sensors for finger movement detection.
2.2. Hydrogels of biopolymer and CPs
Hydrogels composed of CPs and biopolymers have attracted considerable attention, particularly in applications such as bioelectronics and electronic skin (e-skin), owing to their outstanding flexibility, biocompatibility and capacity for functional integration.44 A key advantage of CP–biopolymer hydrogels in transient electronics lies in their tuneable mechanical properties, which can mimic those of human tissues and thereby enable their utilisation as effective interfaces between human and machine. Typically, their formation relies on two major cross-linking strategies: physical and chemical (Fig. 1(C)).72–74 This section summarises recent progress in CP–biopolymer-based hydrogels, with an emphasis on their fabrication method, electrical conductivity and degradability. | ||
| Fig. 5 Hydrogels of biopolymers and CPs via physical cross-linking (hydrogen bonding and ionic cross-linking). (A) Hydrogen bonding cross-linked hydrogel. (a) Schematic illustration of the manipulation process for cell-laden nanopellets; (b) photograph of the obtained hydrogels. Reproduced with permission.5 Copyright 2011 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim. (B) Schematic representation of the preparation and synthesis process of hydrogen bonding cross-linked CNF-PPy/PB hydrogels. Reprinted with permission from ref. 24 Copyright 2018 American Chemical Society. (C) Sulfonated alginate/PEDOT polymers act as dispersible fillers in ionic cross-linked hydrogel prepolymers, enabling the fabrication of injectable and 3D-printable hydrogel bioelectronics for wearable physiological monitoring and wound closure applications. Reproduced from ref. 26 licensed under CC BY 4.0. | ||
One example of a hydrogel formed via hydrogen bonding was reported by Teshima et al.,5 who developed PEDOT:PSS/silk fibroin hydrogel nanopellets by immersing the mixed solution of those two components into methanol, which induced gelation through β-sheet formation in silk fibroin (Fig. 5(A)). During the gelation process of silk fibroin, hydrogen bonding was generated between the beta sheet of silk fibroin. The resulting hydrogen-bonded hydrogel nanopellets exhibited a conductivity of 50 µS sq−1 and showed excellent biocompatibility, supporting adhesion and manipulation of Chinese hamster ovary and hippocampal neural cells, which was attributed to their electroactivity.
CP–biopolymer hydrogels formed through hydrogen bonding between the two components have also been reported.24,76,77 For instance, Na–alginate and PEDOT:PSS were combined via hydrogen bonding to produce a hydrogel electrode material.76 This hydrogel exhibited an ionic conductivity of 7.6 × 10−2 S cm−1 and a high specific capacitance of 312 F g−1 at 80 mV s−1, making it suitable for supercapacitor applications. Another example involves a CNF/PPy hydrogel, where poly(vinyl alcohol) (PVA) was added to reinforce hydrogen bonding among CNF and PPy (Fig. 5(B)).24 The resulting hydrogel achieved a conductivity of 3.65 ± 0.08 S m−1 and a maximum specific capacitance of 236.9 F g−1, along with excellent mechanical strength and biocompatibility.
Ionically cross-linked hydrogels based on biopolymers and conducting polymers are typically formed through electrostatic interactions facilitated by ions. Biopolymers such as alginate and silk fibroin serve as effective matrices in these systems due to their ability to interact readily with divalent or trivalent cations, such as Ca2+ and Fe3+. As mentioned before, commercially available and water-soluble PEDOT:PSS is commonly integrated into these hydrogels to impart electronic conductivity. For example, in one study, alginate was mixed with PEDOT:PSS to form a mixed solution, which was then cross-linked by CaCl2 to produce an electroactive hydrogel.78,79 These hydrogels have been demonstrated as pressure sensor arrays78 and electroresponsive drug delivery systems.79 In a 2025 report by Montazerian and coworkers, a novel self-doped hydrogel was fabricated by in situ polymerisation of EDOT in sulfonated alginate using APS as the oxidant (Fig. 5(C)).26 The sulfonated alginate served as both the dopant and the ionic cross-linking agent. Exposure to FeCl3 triggered ionic gelation, yielding a PEDOT/sulfonated alginate hydrogel with a conductivity of around 7.5 × 10−2 S m−1. This hydrogel functioned effectively as a skin-interfaced electrode for physiological signal detection and was further integrated with gelatin-based bioadhesive to create injectable, pH-responsive smart sealants for wound monitoring. Notably, in vivo studies demonstrated reduced immune response to the sulfonated alginate/PEDOT system compared to conventional PEDOT:PSS, highlighting its enhanced biocompatibility.
Similarly, silk/PPy hydrogels were fabricated by in situ polymerisation of pyrrole within CaCl2-cross-linked silk fibroin hydrogels.80 These hydrogels demonstrated a high conductivity (26 S m−1) and were successfully applied as flexible, wearable strain sensors capable of detecting both large and subtle body motions with excellent sensitivity, reproducibility and mechanical stability.
Glutaraldehyde and genipin are widely utilised to form covalent bonds with free amine groups in biopolymer backbones. For example, Lee et al. (Fig. 6(A)) developed a PEDOT:PSS/gelatin hydrogel by introducing genipin, which reacts with the primary amine groups in gelatin.11 The resulting hydrogel patch demonstrated a surface resistance as low as 3.5 kΩ cm−2 and excellent adhesive properties to human skin (adhesive strength of 0.85 N), while minimising skin irritation compared to conventional adhesive electrodes. Similarly, Li's group reported a PPy/chitosan hydrogel fabricated utilising in situ polymerisation of pyrrole in a chitosan solution, followed by glutaraldehyde-mediated cross-linking.81 This injectable hydrogel improved electrical impulse propagation within the infarcted myocardium, which in turn contributed to the preservation of global cardiac function, demonstrating its applicability in cardiac tissue repair. Additionally, a multi-component hydrogel consisting of chitosan, gelatin, agar and PEDOT:PSS was developed utilising glutaraldehyde as the cross-linker.82 Agar served as a porogen, and was removed after freeze–thaw cycling, yielding a porous chitosan/gelatin/PEDOT:PSS network. The hydrogel achieved a conductivity of 3.35 × 10−4 S cm−1 and remained stable in PBS.
 | ||
| Fig. 6 Hydrogels of biopolymers and CPs via chemical cross-linking. A. Gelatin/PEDOT:PSS hydrogel electrode fabricated using genipin as the cross-linker. Reproduced from the ref. 11 Licensed under CC BY-NC 4.0. B. Methacryloyl (GelMA)/PEDOT:PSS hydrogel: (a) GelMA synthesis; (b) PEDOT:PSS preparation; (c) photopolymerisation of the precursor utilising Irgacure 2959 as the photoinitiator; and (d) resulting hydrogels with 0%, 0.1%, and 0.3% PEDOT:PSS. Reprinted with permission from ref. 28 Copyright 2018 American Chemical Society. C. Transparent and stretchable silk-based hydrogel electrodes: (a) PEGDE crosslinking with silk fibroin (SF) to form PEGylated SF (PSF); (b) thin silk film electrode formed by casting PSF solution onto PEDOT:PSS, followed by evaporation; (c) cross-sectional view of electrode with SF chains (yellow) penetrating the PEDOT:PSS interface; (d) solvent evaporation exposes silk micelle chains, increasing interaction with PSS. Reproduced with permission.29 Copyright 2021 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim. | ||
Carbodiimide chemistry, particularly using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) in combination with N-hydroxysuccinimide (NHS), is widely employed to facilitate amide bond formation between carboxyl and amine groups. Xu et al. developed a carboxymethyl chitosan (CMCS)/PEDOT:PSS hydrogel for neural tissue engineering by cross-linking CMCS with EDC/NHS, followed by in situ EDOT polymerisation.83 Hydrogen bonding between CMCS and PEDOT further stabilised the network. The hydrogel exhibited a conductivity of 4.68 ± 0.28 × 10−3 S cm−1, showed no cytotoxicity and supported PC12 cell adhesion and proliferation. In the degradation experiments, the hydrogels exhibited reduced weight loss with increasing PEDOT content. As noted in the original study,83 this inverse relationship was attributed to the decreased hydrophilicity imparted by higher PEDOT levels, which limited enzyme penetration and consequently slowed degradation. Such tuneable degradation behaviour suggests that the degradation rate of CMCS/PEDOT:PSS hydrogels can be tailored to match the timescale required for bridging nerve conduit gaps in tissue engineering applications.
A similar strategy was applied to synthesise poly-γ-glutamic acid/PEDOT hydrogels as flexible electrodes for supercapacitors, using EDC as the cross-linker and cysteamine to introduce amine functionality.21 The resulting hydrogel delivered a specific capacitance of 45.4 ± 0.7 mF cm−2, demonstrating long-term electrochemical stability and flexibility, making it suitable for supercapacitor applications. Nevertheless, while their electrochemical performance was well established, insight into their degradation behaviour is still lacking, and such information is essential for evaluating their potential as transient electrodes.
Biopolymers functionalized with methacrylate or acrylate groups (e.g., GelMA, polyethylene glycol diacrylate (PEGDA)) can form covalent networks upon UV irradiation in the presence of photoinitiators. Spencer et al. (Fig. 6(B)) fabricated GelMA/PEDOT:PSS hydrogels by dispersing PEDOT:PSS into GelMA prepolymer solution, followed by UV-induced polymerisation utilising Irgacure 2959.28 The hydrogels showed low impedance (as low as 261 kΩ at 1 Hz), promoted the spreading of C2C12 myoblasts and enhanced ex vivo electrostimulation of abdominal tissues, highlighting their potential as electroactive scaffolds for soft tissue interfaces. The degradation rate of GelMA/PEDOT:PSS hydrogels showed accelerated initial weight loss with higher PEDOT:PSS content, which was attributed to the presence of a higher fraction of un-crosslinked GelMA introduced by PEDOT:PSS incorporation. Wu et al. further developed GelMA/PANI conductive hydrogels via interfacial in situ polymerisation of aniline within pre-crosslinked GelMA networks, also using Irgacure 2959.84 The resulting hydrogels exhibited significantly enhanced conductivity and supported the viability and adhesion of murine mesenchymal progenitor cells. These hydrogels could also be patterned into complex architectures via digital light processing, enabling their application in 3D bioelectronics and scaffold engineering.
To further enhance mechanical properties, Testore et al. introduced PEGDA into a gelatin/PEDOT:PSS hydrogel system, using riboflavin (RF) as a biocompatible type II photoinitiator.85 In this system, gelatin served as both a co-initiator and bioactive matrix, while PEGDA contributed mechanical reinforcement. The optimized hydrogels exhibited a modulus of ∼20 kPa and supported cardiac fibroblast adhesion, indicating good mechanical and cellular compatibility with cardiac tissue. Importantly, PEGDA–gelatin hydrogels exhibited composition-dependent degradation, with higher PEGDA:gelatin ratios slowing weight loss due to increased crosslinking density. After two weeks, the hydrogels exhibited weight loss ranging from ∼62% (at the lowest PEGDA:gelatin ratio) to ∼43% (at the highest ratio). The observed tuneable degradation was primarily driven by dissolution of soluble components. Such controlled degradation is particularly important for proper integration of the materials, as biopolymer-based hydrogels typically degrade too rapidly owing to their high solubility, which is often considered a limitation.
Above are described three common types of chemical cross-linkers employed in the fabrication of CP–biopolymer hydrogels: amine-reactive, carbodiimide-based and photo-initiating systems. In addition to these, other cross-linking strategies have been explored to expand the functional versatility of such materials. For example, as shown in Fig. 6(C), poly(ethylene glycol) diglycidyl ether (PEGDE) was utilised to cross-link silk fibroin through nucleophilic addition reactions with primary amine and hydroxyl groups present in the protein backbone.29 The PEGDE-crosslinked silk fibroin solution was then deposited onto PEDOT:PSS films, forming a robust interpenetrating network. This PEDOT:PSS/silk hydrogel exhibited a low sheet resistance (∼160 ± 56 Ω sq−1) and remarkable stretchability (∼260%). Degradation studies revealed that the hydrogel lost more than 80% of its initial weight after 16 days in PBS with protease, while pure silk dissolved immediately under the same conditions. These results highlighted the controlled degradation behaviour of the hydrogel. Furthermore, the PEDOT:PSS/silk system was successfully demonstrated as a soft neural electrode, capable of monitoring neurovascular activity in rats.
2.3. Graft copolymers of biopolymer and CPs
Grafting has emerged as a versatile approach for functionalising conducting polymers with desired properties.43,73 For instance, our group has grafted stretchable polymers from conducting polymer backbones to produce stretchable electronic materials.86–88 More recently, grafting strategies have been increasingly applied to the design of transient (degradable) electronic systems.36,43 In this context, graft copolymers of biopolymers and conducting polymers (CPs) are synthesised by covalently attaching CP chains or oligomers from or onto a biopolymer backbone. Grafting generally refers to the covalent modification of a polymeric backbone with pendant chains. One key advantage of this strategy is that the grafted conducting polymer segments, guided by covalent bonding to a biopolymer backbone, tend to form interconnected crystalline domains, which can significantly enhance charge transport throughout the material12,23,89 In addition, the grafting structure often imparts distinct degradation behaviour, which is discussed further in Section 3.There are two primary approaches utilised in synthesising graft copolymers: the “grafting from” and “grafting to” methods (Fig. 1(D)). In the grafting from strategy, polymerisation of the conducting polymer occurs from monomeric or initiator sites pre-attached to the biopolymer. In contrast, the grafting to approach involves the covalent attachment of pre-formed conducting polymers or oligomers to reactive groups on the biopolymer backbone.
One of the earliest examples of this strategy was reported by Ünal et al., who synthesised a PPy-graft-chitosan copolymer via chemical oxidative polymerisation of pyrrole in the presence of chitosan.90 In this system, FeCl3 served both as an oxidant and as an initiator, attacking the saccharide units of the chitosan backbone to generate macroradicals, which subsequently coupled with pyrrole radicals during the radical polymerisation of pyrrole monomers. The resulting graft copolymer exhibited superior antibacterial activity compared with either PPy or chitosan alone, which was attributed to the dense distribution of positive charge along the chitosan chains. Similar strategies have since been applied to synthesise a variety of graft copolymers, including PANI-graft-gelatin,91 PANI-graft-chitosan,90,92 PPy-graft-chitosan,92 polythiophene-graft-chitosan,92 PPy-graft-Xanthan gum,93 PPy-graft-chitin,94 and poly(3-methyl thiophene) (P3MT)-graft-cellulose.95 These materials have been explored for applications in tissue engineering,91 antibacterial packaging,90,92 chemical sensing93 and flexible electronics.95 Although the mechanistic pathways proposed in these studies provide valuable insights, further work is still needed to fully confirm the underlying grafting mechanisms. Likewise, the degradation behaviour of many of these graft copolymers remains underexplored, leaving opportunities for future studies to clarify their stability and biodegradation profiles.
An alternative and more chemically controlled approach involve the pre-functionalization of the biopolymer with reactive groups that participate in the CP polymerisation. For example, in a study by Wang et al. (Fig. 7(A)), PPy-graft-gelatin was synthesized by means of in situ polymerisation of pyrrole in the presence of gelatin methacrylate (GelMA).4 Here, the pendant vinyl double bonds introduced by methacrylic anhydride acted as reactive sites that were attacked by pyrrole radical cations during oxidative polymerisation, thereby enabling covalent grafting of polypyrrole onto the GelMA backbone and yielding a PPy-graft-gelatin copolymer. NMR spectroscopy confirmed the disappearance of double bonds, confirming the reaction between PPy and MA. In addition to initiating pyrrole polymerisation, FeCl3 also coordinated with GelMA functional groups, reinforcing the hydrogel network structure. The resulting injectable hydrogel exhibited good electrical conductivity (0.016 S cm−1), along with self-healing ability and excellent biocompatibility. However, this study provided limited insight into how such graft copolymers degrade under physiological conditions, highlighting an important direction for future research.
 | ||
| Fig. 7 Graft copolymers of biopolymer and CPs via the “grafting from” strategy. A. Schematic illustration of the synthesis routes for GelMA and PPy-GelMA. Reprinted with permission from ref. 4. Copyright 2020 American Chemical Society. B. Grafting of EDOT-NH2 monomers onto heparin, followed by polymerisation of PEDOT side chains. Reproduced from the ref. 2. Licensed under CC BY-NC 4.0. C. Fabrication process of the glucose biosensor, highlighting enzyme immobilisation on electropolymerized Ch-PTh-PPy. Reproduced from ref. 7 Copyright (2020), with permission from Elsevier. | ||
Another strategy involves using CP monomers as reactive sites that enable covalent grafting during subsequent polymerisation. For example, p-phenylenediamine (aniline monomer) was first grafted onto oxidised cellulose nanocrystals functionalized with carboxylic acid groups.96 These pendant aniline moieties then served as initiation sites for side-chain growth, where oxidative radical polymerisation extended polyaniline (PANI) chains directly from the cellulose backbone. This approach yielded cellulose-PANI graft copolymers with well-defined covalent linkages and enhanced electrical properties. Similarly, a more recent example demonstrated the synthesis of a self-doped hydrogel composed of sulfated heparin and PEDOT side chains (Fig. 7(B)).2 In this work, EDOT-NH2 was covalently grafted onto the heparin backbone via EDC/NHS coupling. Polymerisation was initiated from these grafted sites using APS to produce the graft copolymer, heparin-ketone-PEDOT. Sulfate groups on the heparin backbone acted as internal dopants. Additionally, aminoacetone was grafted alongside EDOT-NH2 to introduce ketone groups that could react with a multi-arm amino-oxy PEG via oxime cross-linking, yielding an injectable hydrogel. The resulting material exhibited a modulus in the 104 Pa range, an adhesive strength of 31.6 kPa and a conductivity of 1.65 S m−1. This hydrogel was used as a bioelectronic patch capable of electrically pacing human induced pluripotent stem cell (iPSC)-derived cardiomyocytes, with excellent in vivo biocompatibility. When injected into myocardial tissue, the hydrogel formed a discrete black globule and degraded gradually over 4 weeks.
Moreover, a sequential grafting strategy has also been employed, wherein one conducting monomer is first grafted onto the biopolymer backbone, and this is subsequently used to initiate the polymerisation of a second type of conducting monomer from the grafted sites. In a study by Senel and coworkers (Fig. 7(C)), thiophene monomers were initially grafted onto chitosan, forming thiophene-functionalized chitosan.7 These grafted thiophene groups provided conjugated sites on the chitosan backbone that could participate in further electrochemical reactions. During subsequent electropolymerisation in a pyrrole monomer solution, the thiophene moieties served as active centres where pyrrole radical cations could couple and grow into polypyrrole chains. During the polymerisation process, glucose oxidase was simultaneously immobilised within the matrix. The resulting hydrogel-based electrode demonstrated enhanced glucose sensing performance, with high sensitivity and stability when compared to other reported biosensor platforms.
The earliest reported examples of this approach involved carboxyl-capped aniline pentamers (AP) grafted onto chitosan backbones via EDC (or DCC)/NHS coupling chemistry in a mixed aqueous/organic cosolvent system (acetic acid aqueous/DMSO/DMF).97,98 Due to the presence of two carboxyl groups on AP, partial cross-linking occurred during grafting. A similar system, AP-graft-gelatin, was reported by Liu et al. in 2012 (Fig. 8(A)), using the same coupling chemistry in a slightly modified cosolvent system.1 Following grafting, the copolymer was further cross-linked using EDC in 90% ethanol to form a hydrogel. The grafting density of AP could be tuned to yield different levels of AP incorporation. The resulting AP-graft-gelatin hydrogels demonstrated excellent in vitro biocompatibility, as evidenced by favourable adhesion and low cytotoxicity in MTT assays with rat SC96 cells. Compared to pristine AP, the graft copolymers and their degradation products showed reduced cytotoxicity, attributed to the biocompatible gelatin. Among tested variants, the copolymer with 1.9 wt% AP exhibited optimal properties for tissue engineering applications.
 | ||
| Fig. 8 Graft copolymers of biopolymer and CPs via “grafting to” strategy. (A) Schematic representation of the synthesis route for the aniline pentamer-grafted-gelatin copolymer (AP-g-GA). Reproduced with permission from ref. 1 Copyright 2011 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim. (B) Synthetic pathway of the Dex-AT-FA polymer (a) and preparation of the conductive, self-healing Dex-AT/CECS hydrogel (b). Reproduced from ref. 3 Copyright (2019), with permission from Elsevier. (C) Gelatin-graft-poly(3-hexylthiophene) copolymers developed for transient electronic applications. Reprinted with permission from ref. 12 Copyright 2024 American Chemical Society. (D) The fabrication of EPL-g-O3HT copolymers via amide bond formation between O3HT and the ε-poly-L-lysine backbone. Reproduced from the ref. 23 Licensed under CC BY-NC 4.0. | ||
Building upon these early works, Guo and coworkers developed a conductive and degradable hydrogel based on aniline tetramer (AT)-graft-dextran (Fig. 8(B)).3 First, AT was modified with hexamethylene diisocyanate (HDI) to yield HDI-AT, which was then grafted onto the hydroxyl groups of dextran. The resulting AT-graft-dextran was further modified by reaction with 4-formylbenzoic acid (FA) to introduce aldehyde groups. Finally, this intermediate was reacted with N-carboxyethyl chitosan, forming a dynamic Schiff base network between FA and the amine groups on chitosan, yielding a self-healing hydrogel. The hydrogel exhibited a conductivity of 2.7–3.4 × 10−5 S cm−1, comparable to that of human skeleton muscle. It also demonstrated injectability, in vivo degradability, and the ability to encapsulate cells during injection. The released cells retained proliferation capability, highlighting its potential as a scaffold or delivery carrier for skeleton muscle repair.
Inspired by these advances, our group further explored graft copolymers of biopolymers and CPs using the “grafting to” strategy. We reported the synthesis of P3HT-graft-gelatin (Fig. 8(C)), in which P3HT-COOH was covalently grafted onto the gelatin backbone.12 A mixed solvent system (THF/DMF/DMSO/H2O) was utilised to overcome solubility difference between P3HT and gelatin. The P3HT content in P3HT-graft-gelatin was up to 9.05 wt%. The thin-film morphology was optimized by using a THF/H2O cosolvent, which enhanced π–π stacking of P3HT and resulted in improved electroactivity. The final graft copolymer achieved a conductivity of 1.65 ± 0.02 × 10−7 S cm−1. Enzymatic degradation was achieved due to the amide bonds within gelatin and over five days yielded amphiphilic micellar nanoparticles. In vitro testing confirmed the biocompatibility of the material and its degradation products. The copolymer was successfully employed in pressure sensors, achieving a sensitivity of 58 Pa−1. This type of graft conducting copolymers represents a new approach to designing enzymatically degradable and biocompatible conducting polymer materials. In these systems, the short biopolymer chains help to shield the degradation products, thereby reducing potential toxicity. As a result, such materials are attractive for applications in implantable, wearable and environmentally degradable electronics.
While grafting long CP chains such as P3HT imparts electroactivity, the achievable grafting density and overall conductivity of the copolymer are limited, as the bulky polymer chains crowd each other on the backbone and hinder efficient chain packing. To address this issue, an optimized approach was developed whereby grafting of shorter conducting oligomers, specifically oligo 3-hexylthiophene (O3HT, ∼40 mers), onto ε-poly-L-lysine (EPL) (Fig. 8(D)) was employed.23 O3HT-graft-EPL copolymers were synthesized utilising O3HT functionalized with NHS esters and their reaction with the primary amines of EPL (in a THF/DMF/DMSO/H2O solvent system). Copolymers with 43, 65 and 90 wt% O3HT were successfully prepared. The resulting materials achieved conductivities of up to 1.13–1.98 ± 0.05 S m−1, markedly higher than those of P3HT-graft-gelatin. The O3HT-graft-EPL-1 copolymer (43 wt% O3HT) underwent complete enzymatic degradation within 12 days. In addition, EPL-graft-O3HT-1 exhibited strong broad-spectrum antibacterial activity against both Gram-negative and Gram-positive bacteria, which was attributed to its high ε-poly-L-lysine content. These graft copolymers also demonstrated high functional versatility in transient bioelectronics, serving as the channel material in organic electrochemical transistors (OECTs) and as electrodes in electromyography (EMG) sensors for monitoring muscle signals.
Together, these examples highlight the feasibility of grafting CPs or oligomers onto biopolymer backbones, facilitated by the use of cosolvent systems, enabling controlled tuning of grafting density, conductivity and degradation.
3. Degradation of biopolymer/CP transient electronic materials
Degradation represents a fundamental design parameter in transient electronic systems, which are engineered to undergo controlled disintegration following a predefined operational lifespan.37,99 The degradation process may proceed through a variety of mechanisms, including hydrolytic cleavage, dissolution, enzymatic catalysis, photodegradation, thermal decomposition, oxidative reactions and mechanical stress.100,101 Among these processes, enzymatic degradation, commonly referred to as biodegradation, is particularly relevant under environmentally and biologically realistic conditions.102,103 Biodegradation encompasses a multi-step biological process in which microorganisms, mainly bacteria and fungi, initiate surface deterioration, enzymatically fragment polymer chains into smaller molecules, and subsequently assimilate these fragments through metabolic pathways, ultimately converting them into CO2, water and biomass.102,103For biopolymer–CP transient electronic materials, degradation predominantly relies on the breakdown of the biopolymer backbone.15,36,43,104 Natural biopolymers, such as gelatin, chitosan, cellulose, alginate and silk fibroin, are inherently biodegradable and exhibit distinct enzymatic degradation pathways.105,106Table 1 summarises enzymes involved in the degradation of biopolymers. These polymer-specific pathways determine the rate at which the biopolymer backbone breaks down, thereby modulating film morphology and the subsequent physical disintegration of the conducting network. In contrast, CPs are generally chemically robust and do not undergo significant molecular degradation during enzymatic processes, largely due to their rigid conjugated backbones and the absence of easily hydrolysable bonds, such as esters, amides or glycosidic linkages.35,43
| Type of polymer | Enzyme type | Degradation type | Degradation mechanism |
|---|---|---|---|
| Protein | Protease | Hydrolysis | Cleavage of peptide bonds in proteins |
| Lipids | Lipases | Hydrolysis | Hydrolyses of ester bonds in lipids |
| Starch | Amylases | Hydrolysis | Breaking of α-1,4-glycosidic bonds in starch |
| Cellulose | Cellulases | Hydrolysis | Breaking of β-1,4-glycosidic bonds in cellulose |
| Chitin | Chitinases | Hydrolysis | Breaking of β-1,4-glycosidic bonds in chitin |
| Lignin | Laccases/peroxidases | Oxidation | Oxidation of phenolic groups in lignin/peroxide-driven oxidative cleavage |
As a result, the degradation behaviours of CP–biopolymer transient electronic materials are largely governed by the interaction between the two components. As shown in Fig. 9, the behaviour of these systems can be broadly categorised into two distinct modes: (I) matrix degradation, in which only the biopolymer component is enzymatically degraded while the CP remains largely unaffected, and (II) network degradation, where macromolecular structure disintegrates into byproducts, while covalent or strong physicochemical interactions between the biopolymer and CP persist.
 | ||
| Fig. 9 Degradation pathways of CP–biopolymer systems: matrix degradation leaves conducting polymers intact, while network degradation causes co-fragmentation of both components. | ||
3.1. Matrix degradation
Matrix degradation usually happens in physically blended systems, such as CP–biopolymer blends, multilayer structures and composite hydrogels. In these materials, the two components are merely mixed, blended or layered, and are held together only by weak physical interactions. Consequently, degradation of these systems is primarily governed by the degradation of the biopolymer matrix. Although some physical fragmentation of CP may occur during matrix degradation (e.g., cracking or delamination), the CP chains largely remain chemically intact. For instance, PEDOT: PSS/starch-chitosan-based multilayer films have been shown to degrade in lysozyme solution within 8 minutes,62 while PPy/chitosan blends disintegrate in acidic aqueous solutions within 96 hours, with PPy particles precipitating at the bottom.10 These examples demonstrate dissolution-driven degradation, one of the simplest mechanisms of material breakdown. This method is fundamentally distinct from enzyme-mediated degradation pathways, particularly in applications where microbial activity plays a significant role. The latter may offer more precise control over degradation performance and rate.Matrix degradation, where only the degradable biopolymer matrix is degraded, is generally sufficient for non-biological applications that aim to disintegrate the electronic material and reduce electronic waste. However, the limited natural degradation pathways available for CPs present a significant drawback for in vivo applications, where residual CPs may persist. This highlights the need for holistic degradation strategies for transient CP–biopolymer systems.
3.2. Network degradation
Network degradation occurs in systems where CPs are covalently bonded or physically coordinated to biopolymers, such as in graft copolymers or hydrogel networks. In these systems, the physical or covalent interactions between the two components remain stable under degradation conditions. These architectures enable coordinated disintegration of both structural and functional components, offering a more strategic and controlled biodegradation pathway. During the degradation of interconnected macromolecular network, only the biopolymer segments containing enzyme-cleavable sites are selectively broken down. This selective cleavage leads to the fragmentation of the CP–biopolymer macromolecule, resembling matrix degradation in some respects.However, the key distinction between matrix and network degradation lies in the fate of CP component. In network-integrated systems, the strong covalent or physicochemical interactions between biopolymers and CP ensure that the CP is carried away alongside biopolymer degradation. Consequently, the resulting degradation products are copolymeric fragments composed of short biopolymer chains and CP segments. Because CPs are typically considered resistant to metabolic degradation in vivo, their combination with biocompatible biopolymers opens up opportunities for more biocompatible clearance from the body, rather than long-term accumulation. For instance, heparin-graft-PEDOT-based hydrogels demonstrated excellent in vivo degradability, leaving no detectable residues within 4 weeks following injection into myocardial tissue in mice.2 Our group further investigated structure of degradation products for graft copolymers P3HT-graft-gelatin. In this system, the micelle-like nanoparticles were observed in degradation products, with short gelatin chains forming the hydrophilic shell and P3HT forming the hydrophobic core.12 P3HT-graft-gelatin also demonstrated great biocompatibility before and after degradation. This core–shell coverage strategy highlights the advanced nature of the network degradation pathway for CP–biopolymer-based transient electronic materials.
4. Applications
Transient electronic systems composed of biopolymers and CPs have shown broad potential in applications that require temporary functionality, high biocompatibility and environmental sustainability. Their transient nature, defined by controlled disintegration over time, makes them valuable in biomedical devices, wearable and environmental sensing and energy storage systems.In the biomedical field, transient CP–biopolymer electronics have been widely explored for devices designed for short-term operation followed by safe degradation.50 In tissue engineering, conductive scaffolds composed of biopolymers and CPs can provide both mechanical support and electrical stimulation to promote cell proliferation and tissue regeneration.50 Importantly, the degradation profile must be tailored to the requirements of specific applications. For example, in neural tissue engineering, particularly for nerve conduits designed to bridge long gaps, sustaining structural integrity for at least eight weeks is required to preserve conduit morphology and provide reliable guidance for axonal regeneration.83 Xu et al. reported that CMCS/PEDOT:PSS hydrogels function effectively as nerve regeneration scaffolds, supporting neurite outgrowth in PC12 cells.83 The incorporation of the PEDOT network was shown to hinder enzymatic penetration into the hydrogel matrix, thereby slowing degradation and adjusting the degradation rate with the timescale of nerve cell growth. Moreover, the conductivity of CP–biopolymer scaffolds must be carefully regulated to match the electrical characteristics of the target tissue, thereby enabling appropriate stimulation for regeneration. For instance, an aniline tetramer-graft-dextran hydrogel, with conductivity comparable to that of human skeletal muscle, has been employed as a cell delivery carrier for skeletal muscle repair.1 Furthermore, evaluation of the in vivo degradation performance is a critical step toward the clinical translation of transient electronics. A recent example is a heparin-ketone-PEDOT hydrogel (Fig. 10(A)), which was demonstrated to facilitate the electrical pacing of iPSC-derived cardiomyocytes.2 At 7 days post-myocardial injection, histological analysis showed no evidence of tissue necrosis, swelling or heightened inflammation in the surrounding areas. The hydrogel began to degrade after two weeks in rat models, with only a few localised remnants detected at four weeks, and no hydrogel was detected in most samples by eight weeks post-injection. This example successfully confirmed the in vivo degradability of CP–biopolymer systems as transient electronics.
 | ||
| Fig. 10 Representative applications of CP–biopolymer transient electronics. (A) Heparin-ketone-PEDOT hydrogels for implantable bioelectronics, showing intramyocardial injection and subsequent in vivo degradation. Reproduced from the ref. 2. Licensed under CC BY-NC 4.0. (B) Cellulose/PEDOT:PSS films as multifunctional wearable sensors, including mechanical flexibility, breathing monitoring, and temperature sensing. Reprinted with permission from ref. 6. Copyright 2023 American Chemical Society. (C) Poly-γ-glutamic acid/PEDOT hydrogels applied as transient supercapacitors for powering small electronic device. Reprinted with permission from ref. 21. Copyright 2018 American Chemical Society. | ||
Wearable transient electronics composed of CP–biopolymer systems benefit from the breathability and degradability of natural polymers, allowing the creation of practical wearable sensors for monitoring physiological signals. After fulfilling their function, these devices disintegrate and minimise skin irritation from prolonged contact. In such applications, both electrical and mechanical stability are critical to ensure reliable signal monitoring. For instance, cellulose/PEDOT: PSS films have served as stretchable electrodes in wearable healthcare sensors to detect various body motions, temperature, humidity, breathing and speech (Fig. 10(B)).6 The inherent flexibility was provided by the cellulose thin-film substrate, while the incorporation of D-glucose and glycerol further enhanced stretchability by forming dynamic supramolecular chains with cellulose. The surface organic modifier 11-aminoundecanoic acid (11-AA) was further introduced between the PEDOT:PSS and cellulose substrate, which increased the average transmittance of obtained films and enabled visual observation on human skin during real-time signal collection. The conductivity of the spin-coated PEDOT:PSS layers was improved through the addition of high-boiling-point solvents such as DMSO, which facilitated phase separation and enhanced charge transfer between PEDOT grains. In another example reported by our group, thin films of the biodegradable graft copolymer EPL-g-O3HT were demonstrated as transient electrodes for electromyography (EMG) sensors, enabling temporary muscle signal monitoring.23 The considerable conductivity of the EPL-g-O3HT thin films was attributed to the interconnected O3HT crystallites formed through the grafted copolymer structure, while the electrodes also maintained stable resistance after repeated bending cycles. CP–biopolymer transient electronics are also promising for environmental monitoring and smart agriculture. Biodegradable sensors can be employed to detect soil moisture, pH or heavy-metal pollutants and naturally disintegrate afterwards, thereby avoiding long-term environmental impact.107,108 In these settings, transient polymer electronics can breakdown after defined lifetime, eliminating the need for retrieval and preventing soil contamination. This controlled degradation also allows the device lifetime to align with crop cycles while reducing the accumulation of electronic waste.
Powering such systems requires degradable energy storage components. CP–biopolymer-based degradable supercapacitors have been reported using PPy-chitosan blends,67 PEDOT: PSS/sodium hydrogels,76 and poly-γ-glutamic acid/PEDOT hydrogels.21 For example, poly-γ-glutamic acid/PEDOT hydrogels exhibit a specific capacitance of 45.4 ± 0.7 mF cm−2 and stable electrochemical performance suitable for transient supercapacitor applications (Fig. 10(C)).21
Apart from the above aspects, transient CP–biopolymer electronics have also been applied to logic and signal-processing components, such as organic field-effect transistors (OFETs)58,59 and organic electrochemical transistors (OECTs).23 These devices serve as temporary switching and amplification elements in short-lived sensing circuits, enabling integrated biosignal processing and low-power operation within transient electronic platforms. It is worth noting that in non-biomedical applications, a key advantage of transient electronics lies in their degradability, which helps mitigate the accumulation of electronic waste. This sustainability benefit makes them particularly attractive for large-scale or short-lived deployments where device retrieval is impractical or non-efficient.
5. Conclusion and future prospects
The combination of conducting polymers with natural biopolymers has made CP–biopolymer systems an attractive option for transient degradable electronics. Conducting polymers provide adjustable electroactivity and conductivity, while biopolymers contribute biodegradability, biocompatibility, and chemical versatility. Together, they allow a balance between electrical performance and controlled degradation. With recent progress in processing, these systems have been developed as blends, multilayer structures, hydrogels and graft copolymers, each offering a way to couple conductivity with degradability. Beyond serving as degradable conductors, these systems enable devices that actively interface with biological tissues while eliminating the need for retrieval or reducing electronic waste. Demonstrated applications include neural interfaces, tissue scaffolds, implantable devices, wearable sensors and energy storage systems, highlighting their unique potential to integrate sustainable electronics with living systems.Despite these advances, several challenges remain before CP–biopolymer systems can be translated into practical, large-scale applications. The first challenge is the design and control of degradation pathways and degradation products. A clearer understanding of degradation kinetics is needed. More importantly, the immune response and biosafety of both the implantable materials and their degradation by-products, particularly their clearance pathways in the body, must be thoroughly assessed and understood. The second challenge is electrical stability. The conductivity of these materials remains lower than that of conventional electronic materials and is easily affected under physiological conditions owing to the swelling of hydrophilic biopolymers. Third, mechanical stability needs to be improved to withstand stresses, such as repeated bending in wearable devices while maintaining stable conductivity. Finally, a critical challenge is the limited scalability of current processing methods. Cost-effective, environmentally friendly, and scalable processing methods, such as roll-to-roll processing or high-throughput printing, will be key to moving these systems from lab-scale prototypes to commercially viable products.
Based on the above challenges, future perspectives for transient CP/biopolymer systems lie in several promising directions.
5.1. Biodegradable circuits
Research should move beyond transient conductors or single-function devices, such as supercapacitors, toward fully biodegradable circuits that combine conductors, semiconductors, substrates and encapsulants into complete systems.5.2. Bioresorbable electronics
Bioresorbable electronics represent a subclass of transient electronics that are not only biodegradable but also fully resorbable within the body. Such devices are completely metabolized and cleared through natural physiological processes, thereby eliminating the need for surgical removal and reducing long-term risks such as inflammation. CP-based bioresorbable electronics have been reported, primarily through the incorporation of cleavable linkers or degradable segments into the polymer backbone.16,38,42 By emphasizing bioresorbable characteristics and utilising the well-explored and new degradation pathways to CP-based bioresorbable electronics, transient CP–biopolymer systems, especially those with structure-controlled degradation mechanisms, hold strong potential for implantable applications, including temporary sensing and therapeutic devices. Important to these advances will be the development of novel chemistries that render conducting polymers intrinsically degradable, enabling fully biodegradable and bioresorbable CP–biopolymer electronic materials.5.3. Added benefits from biopolymer to CP–biopolymer systems
There is strong potential to utilise multifunctional biopolymers that not only provide structural support and biodegradability but also incorporate features such as antibacterial or adhesive functionalities. Multifunctionality and full biodegradability would enable the next generation of transient electronics, particularly in biomedical contexts.5.4. Broad applications
Expanding applications more into areas such as smart agriculture, environmental monitoring, and transient energy storage will further broaden the societal impact of transient polymer electronics.In conclusion, CP–biopolymer systems mark an important step toward next-generation transient electronics that are not only functional and degradable but also sustainable. Progress in molecular design, control of degradation and scalable processing will be key to moving these materials from the lab to practical applications. Ultimately, such systems hold strong promise for building a greener electronics landscape and contributing to a more sustainable world.
Conflicts of interest
The authors declare no conflict of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The authors kindly acknowledge the Marsden Fund (project number 20-UOA-314) and the financial support for this project provided by the Royal Society Te Apārangi, New Zealand. This research was supported by an AINSE Ltd. Postgraduate Research Award (PGRA).References
- Y. Liu, J. Hu, X. Zhuang, P. Zhang, Y. Wei, X. Wang and X. Chen, Macromol. Biosci., 2012, 12, 241–250 CrossRef CAS PubMed.
- D. Hachim, O. Hernandez-Cruz, J. E. J. Foote, R. Wang, M. W. Delahaye, D. J. Stuckey, Z. Feng, J. P. Wojciechowski, L. C. B. Salter, J. Lin, S. E. Harding and M. M. Stevens, Adv. Healthcare Mater., 2025, 14, e2403995 CrossRef PubMed.
- B. Guo, J. Qu, X. Zhao and M. Zhang, Acta Biomater., 2019, 84, 180–193 CrossRef CAS PubMed.
- S. Wang, J. Lei, X. Yi, L. Yuan, L. Ge, D. Li and C. Mu, ACS Appl. Polym. Mater., 2020, 2, 3016–3023 CrossRef CAS.
- T. Teshima, H. Nakashima, N. Kasai, S. Sasaki, A. Tanaka, S. Tsukada and K. Sumitomo, Adv. Funct. Mater., 2016, 26, 8185–8193 CrossRef CAS.
- A. F. Wibowo, J. W. Han, J. H. Kim, A. Prameswati, S. A. N. Entifar, J. Park, J. Lee, S. Kim, D. C. Lim and Y. Eom, ACS Appl. Mater. Interfaces, 2023, 15, 18134–18143 CrossRef CAS PubMed.
- M. Senel, M. Dervisevic, L. Esser, E. Dervisevic, J. Dyson, C. D. Easton, V. J. Cadarso and N. H. Voelcker, Int. J. Biol. Macromol., 2020, 143, 582–593 CrossRef CAS.
- A. Shabeeba, M. P. Sidheekha, L. Rajan and Y. A. Ismail, RSC Adv., 2022, 12, 31911–31922 RSC.
- M. Y. Teo, K. Lim, K. C. Aw, S. Kee and J. Stringer, RSC Adv., 2023, 13, 26267–26274 RSC.
- Z. Yuan, H. Yin, M. Zheng, X. Chen, W. Peng, H. Zhou, J. Xing, L. Wang and S. Hu, Carbohydr. Polym., 2025, 349, 122963 CrossRef CAS PubMed.
- Y. Lee, S. G. Yim, G. W. Lee, S. Kim, H. S. Kim, D. Y. Hwang, B. S. An, J. H. Lee, S. Seo and S. Y. Yang, Sensors, 2020, 20, 5737 CrossRef CAS PubMed.
- X. Sun, E. W. C. Chan, Q. Chen, N. Kirby, J. Yang, J. P. Mata, R. L. Kingston, D. Barker, L. Domigan and J. Travas-Sejdic, ACS Appl. Mater. Interfaces, 2024, 16, 23872–23884 CAS.
- W. B. Han, J. H. Lee, J. W. Shin and S. W. Hwang, Adv. Mater., 2020, 32, 2002211 CrossRef CAS PubMed.
- R. Jamshidi, M. Taghavimehr, Y. Chen, N. Hashemi and R. Montazami, Adv. Sustainable Syst., 2022, 6, 2100057 CrossRef.
- R. Li, L. Wang, D. Kong and L. Yin, Bioact. Mater., 2018, 3, 322–333 Search PubMed.
- Y. Zhang, G. Lee, S. Li, Z. Hu, K. Zhao and J. A. Rogers, Chem. Rev., 2023, 123, 11722–11773 CrossRef CAS PubMed.
- Q. Lv, Q. Li, P. Cao, C. Wei, Y. Li, Z. Wang and L. Wang, Adv. Mater., 2025, 37, e2411946 CrossRef PubMed.
- K.-H. Teng and M.-H. Lee, J. Environ. Chem. Eng., 2025, 13, 115716 CrossRef CAS.
- A. Zhuang, X. Huang, S. Fan, X. Yao, B. Zhu and Y. Zhang, ACS Appl. Mater. Interfaces, 2022, 14, 123–137 CrossRef CAS PubMed.
- M. Lay, M. G. Say and I. Engquist, Adv. Mater. Technol., 2023, 8, 2300652 CrossRef CAS.
- M. C. G. Saborío, S. Lanzalaco, G. Fabregat, J. Puiggalí, F. Estrany and C. Alemán, J. Phys. Chem. C, 2018, 122, 1078–1090 CrossRef.
- J. Han, K. Lu, Y. Yue, C. Mei, C. Huang, Q. Wu and X. Xu, Ind. Crops Prod., 2019, 128, 94–107 CrossRef CAS.
- X. Sun, E. W. C. Chan, F. R. Shiraz, B. Zhu, J. Yang, K. Matura, V. Sarojini, S. Tekoglu, D. Barker and J. Travas-Sejdic, Adv. Sci., 2025, e01726 CrossRef CAS PubMed.
- Q. Ding, X. Xu, Y. Yue, C. Mei, C. Huang, S. Jiang, Q. Wu and J. Han, ACS Appl. Mater. Interfaces, 2018, 10, 27987–28002 CrossRef CAS.
- C. Diacci, T. Abedi, J. W. Lee, E. O. Gabrielsson, M. Berggren, D. T. Simon, T. Niittylä and E. Stavrinidou, iScience, 2021, 24, 101966 CrossRef CAS PubMed.
- H. Montazerian, E. Davoodi, C. Wang, F. Lorestani, J. Li, R. Haghniaz, R. R. Sampath, N. Mohaghegh, S. Khosravi, F. Zehtabi, Y. Zhao, N. Hosseinzadeh, T. Liu, T. K. Hsiai, A. H. Najafabadi, R. Langer, D. G. Anderson, P. S. Weiss, A. Khademhosseini and W. Gao, Nat. Commun., 2025, 16, 3755 CrossRef CAS PubMed.
- A. Abedi, M. Hasanzadeh and L. Tayebi, Mater. Chem. Phys., 2019, 237, 121882 CrossRef CAS.
- A. R. Spencer, A. Primbetova, A. N. Koppes, R. A. Koppes, H. Fenniri and N. Annabi, ACS Biomater. Sci. Eng., 2018, 4, 1558–1567 CAS.
- Y. Cui, F. Zhang, G. Chen, L. Yao, N. Zhang, Z. Liu, Q. Li, F. Zhang, Z. Cui, K. Zhang, P. Li, Y. Cheng, S. Zhang and X. Chen, Adv. Mater., 2021, 33, e2100221 CrossRef PubMed.
- S.-W. Hwang, H. Tao, D.-H. Kim, H. Cheng, J.-K. Song, E. Rill, M. A. Brenckle, B. Panilaitis, S. M. Won and Y.-S. Kim, Science, 2012, 337, 1640–1644 CrossRef CAS PubMed.
- Y. Choi, J. Koo and J. A. Rogers, MRS Bull., 2020, 45, 103–112 CrossRef.
- W. B. Han, G.-J. Ko, J.-W. Shin and S.-W. Hwang, MRS Bull., 2020, 45, 113–120 CrossRef.
- G. J. Ko, V. R. Naganaboina, E. S. Goda, A. Dutta, H. Cheng and S. W. Hwang, Adv. NanoBiomed Res., 2024, 2400126 Search PubMed.
- B. Guo, L. Glavas and A.-C. Albertsson, Prog. Polym. Sci., 2013, 38, 1263–1286 CrossRef CAS.
- V. R. Feig, H. Tran and Z. Bao, ACS Cent. Sci., 2018, 4, 337–348 CrossRef CAS PubMed.
- Kenry and B. Liu, Biomacromolecules, 2018, 19, 1783–1803 CrossRef CAS PubMed.
- G. J. Ko, V. R. Naganaboina, E. S. Goda, A. Dutta, H. Cheng and S. W. Hwang, Adv. NanoBiomed Res., 2025, 5, 2400126 CrossRef CAS.
- A. Uva, Y. Kim, S. Michailovich, N. S. Y. Hsu, D. C. Bain, S. H. Huang, A. J. Musser and H. Tran, Polym. Chem., 2025, 16, 2817–2828 RSC.
- H. Park, Y. Kim, D. Kim, S. Lee, F. S. Kim and B. J. Kim, Adv. Funct. Mater., 2022, 32, 2106977 CrossRef CAS.
- T. Lei, M. Guan, J. Liu, H.-C. Lin, R. Pfattner, L. Shaw, A. F. McGuire, T.-C. Huang, L. Shao and K.-T. Cheng, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, 5107–5112 CrossRef CAS PubMed.
- A. Uva, S. Michailovich, N. S. Y. Hsu and H. Tran, J. Am. Chem. Soc., 2024, 146, 12271–12287 CrossRef CAS PubMed.
- N. Nozaki, A. Uva, T. Iwahashi, H. Matsumoto, H. Tran and M. Ashizawa, RSC Appl. Polym., 2025, 3, 257–267 RSC.
- E. W. C. Chan, X. Sun and J. Travas-Sejdic, Macromol., 2023, 56, 3755–3773 CrossRef CAS.
- R. Yao, X. Liu, H. Yu, Z. Hou, S. Chang and L. Yang, Int. J. Biol. Macromol., 2024, 278, 134694 CrossRef CAS PubMed.
- C. Cui, Q. Fu, L. Meng, S. Hao, R. Dai and J. Yang, ACS Appl. Bio Mater., 2020, 4, 85–121 CrossRef PubMed.
- C. Wang, T. Yokota and T. Someya, Chem. Rev., 2021, 121, 2109–2146 CrossRef CAS PubMed.
- Z. Wang, Z. Ma, J. Sun, Y. Yan, M. Bu, Y. Huo, Y.-F. Li and N. Hu, Polymers, 2021, 13, 813 CrossRef CAS PubMed.
- C. Wang, T. Yokota and T. Someya, Chem. Rev., 2021, 121, 2109–2146 CrossRef CAS PubMed.
- A. Verlee, S. Mincke and C. V. Stevens, Carbohydr. Polym., 2017, 164, 268–283 CrossRef CAS PubMed.
- B. Guo and P. X. Ma, Biomacromolecules, 2018, 19, 1764–1782 CrossRef CAS PubMed.
- S. Khan and A. K. Narula, Eur. Polym. J., 2016, 81, 161–172 CrossRef CAS.
- P. Moutsatsou, K. Coopman and S. Georgiadou, Polymers, 2017, 9, 687 CrossRef PubMed.
- M. Li, Y. Guo, Y. Wei, A. G. MacDiarmid and P. I. Lelkes, Biomaterials, 2006, 27, 2705–2715 CrossRef CAS PubMed.
- C. Y. Chen, S. Y. Huang, H. Y. Wan, Y. T. Chen, S. K. Yu, H. C. Wu and T. I. Yang, Polymers, 2020, 12 CAS.
- D. Gao, J. Lv and P. S. Lee, Adv. Mater., 2022, 34, e2105020 CrossRef PubMed.
- Y. Won, J. J. Lee, J. Shin, M. Lee, S. Kim and S. Gandla, ACS Sens., 2021, 6, 967–975 CrossRef CAS PubMed.
- S. Bonardd, N. Morales, L. Gence, C. Saldías, F. A. Angel, G. Kortaberria and A. Leiva, ACS Appl. Mater. Interfaces, 2020, 12, 13275–13286 CrossRef CAS PubMed.
- B. W. Du, S. Y. Hu, R. Singh, T. T. Tsai, C. C. Lin and F. H. Ko, Materials, 2017, 10, 1026 CrossRef PubMed.
- M. R. Kiran, Y. Yadav and S. P. Singh, Synth. Met., 2023, 297, 117398 CrossRef CAS.
- K.-H. Teng and M.-H. Lee, J. Environ. Chem. Eng., 2025, 13, 115716 CrossRef CAS.
- S. Khan, M. Ul-Islam, M. W. Ullah, M. Israr, J. H. Jang and J. K. Park, Int. J. Biol. Macromol., 2018, 107, 865–873 CrossRef CAS PubMed.
- J. Miao, H. Liu, Y. Li and X. Zhang, ACS Appl. Mater. Interfaces, 2018, 10, 23037–23047 CrossRef CAS.
- K. J. Gilmore, M. Kita, Y. Han, A. Gelmi, M. J. Higgins, S. E. Moulton, G. M. Clark, R. Kapsa and G. G. Wallace, Biomaterials, 2009, 30, 5292–5304 CrossRef CAS PubMed.
- C. Basavaraja, E. A. Jo, B. S. Kim, D. G. Kim and D. S. Huh, Macromol. Res., 2010, 18, 1037–1044 CrossRef CAS.
- F. Ji, X. Guo, A. Liu, P. Xu, Y. Tan, R. Wang and L. Hao, Carbohydr. Polym., 2021, 270, 118362 CrossRef CAS PubMed.
- A. Shabeeba and Y. A. Ismail, Mater. Res. Bull., 2022, 152, 111817 CrossRef CAS.
- A. Shabeeba, L. Rajan, M. P. Sidheekha, M. S. Thayyil and Y. A. Ismail, J. Energy Storage, 2022, 55, 105724 CrossRef.
- S. Yalçınkaya and Ş. Duran, Polym. Bull., 2022, 79, 10037–10052 CrossRef.
- B. Mohammadi, S. Pirsa and M. Alizadeh, Polym. Polym. Compos., 2019, 27, 507–517 CAS.
- S. Pirsa and B. Mohammadi, Main Group Chem., 2021, 20, 133–147 CrossRef CAS.
- K. Z. Htut, M. Kim, E. Lee, G. Lee, S. H. Baeck and S. E. Shim, Synth. Met., 2017, 227, 61–70 CrossRef CAS.
- D. Liu, C. Huyan, Z. Wang, Z. Guo, X. Zhang, H. Torun, D. Mulvihill, B. B. Xu and F. Chen, Mater. Horiz., 2023, 10, 2800–2823 RSC.
- M. Wang, P. Baek, A. Akbarinejad, D. Barker and J. Travas-Sejdic, J. Mater. Chem. C, 2019, 7, 5534–5552 RSC.
- H. Tan and K. G. Marra, Materials, 2010, 3, 1746–1767 CrossRef CAS.
- Y. Liu, X.-C. Sun, C. Lv and H. Xia, Smart Mater. Struct., 2021, 30, 125014 CrossRef CAS.
- N. M. Badawi, K. M. Batoo, R. Subramaniam, R. Kasi, S. Hussain, A. Imran and M. Muthuramamoorthy, Micromachines, 2023, 14, 1461 CrossRef PubMed.
- A. M. Youssef, M. S. Hasanin, M. E. A. El-Aziz and G. M. Turky, Int. J. Biol. Macromol., 2021, 167, 1435–1444 Search PubMed.
- I. Babeli, G. Ruano, J. Casanovas, M.-P. Ginebra, J. García-Torres and C. Alemán, J. Mater. Chem. C, 2020, 8, 8654–8667 Search PubMed.
- A. Puiggali-Jou, E. Cazorla, G. Ruano, I. Babeli, M. P. Ginebra, J. Garcia-Torres and C. Aleman, ACS Biomater. Sci. Eng., 2020, 6, 6228–6240 CrossRef CAS PubMed.
- Y. Han, L. Sun, C. Wen, Z. Wang, J. Dai and L. Shi, Biomed. Mater., 2022, 17, 024107 Search PubMed.
- A. Mihic, Z. Cui, J. Wu, G. Vlacic, Y. Miyagi, S.-H. Li, S. Lu, H.-W. Sung, R. D. Weisel and R.-K. Li, Circulation, 2015, 132, 772–784 CrossRef CAS PubMed.
- D. A. Ahmad Ruzaidi, M. M. Mahat, Z. Mohamed Sofian, N. A. Nor Hashim, H. Osman, M. A. Nawawi, R. Ramli, K. A. Jantan, M. F. Aizamddin, H. H. Azman, Y. H. Robin Chang and H. H. Hamzah, Polymers, 2021, 13, 2901 CrossRef CAS.
- C. Xu, S. Guan, S. Wang, W. Gong, T. Liu, X. Ma and C. Sun, Mater. Sci. Eng., C, 2018, 84, 32–43 CrossRef CAS PubMed.
- Y. Wu, Y. X. Chen, J. Yan, D. Quinn, P. Dong, S. W. Sawyer and P. Soman, Acta Biomater., 2016, 33, 122–130 CrossRef CAS PubMed.
- D. Testore, A. Zoso, G. Kortaberria, M. Sangermano and V. Chiono, Front. Bioeng. Biotechnol., 2022, 10, 897575 CrossRef PubMed.
- M. Wang, P. Baek, L. Voorhaar, E. W. C. Chan, A. Nelson, D. Barker and J. Travas-Sejdic, Eur. Polym. J., 2018, 109, 237–247 CrossRef CAS.
- M. Wang, S. Kee, D. Barker and J. Travas-Sejdic, Eur. Polym. J., 2020, 125, 109508 CrossRef CAS.
- M. Wang, S. Kee, P. Baek, M. S. Ting, Z. Zujovic, D. Barker and J. Travas-Sejdic, Polym. Chem., 2019, 10, 6278–6289 RSC.
- R. Noriega, J. Rivnay, K. Vandewal, F. P. V. Koch, N. Stingelin, P. Smith, M. F. Toney and A. Salleo, Nat. Mater., 2013, 12, 1038–1044 CrossRef CAS PubMed.
- M. Cabuk, Y. Alan, M. Yavuz and H. I. Unal, Appl. Surf. Sci., 2014, 318, 168–175 CrossRef CAS.
- L. Li, J. Ge, B. Guo and P. X. Ma, Polym. Chem., 2014, 5, 2880–2890 RSC.
- M. Cabuk, M. Yavuz, H. Ibrahim Unal and Y. Alan, Polym. Compos., 2015, 36, 497–509 CrossRef CAS.
- S. Pandey and J. Ramontja, Int. J. Biol. Macromol., 2016, 89, 89–98 CrossRef CAS.
- A. T. Ramaprasad, D. Latha and V. Rao, J. Phys. Chem. Solids, 2017, 104, 169–174 CrossRef CAS.
- O. A. T. Dias, S. Konar, A. L. Leao and M. Sain, Carbohydr. Polym., 2019, 220, 79–85 CrossRef CAS PubMed.
- Z. Xu, J. Zhou, D. Li, G. Zhu and N. Lin, ACS Sustainable Chem. Eng., 2023, 11, 10895–10905 CrossRef CAS.
- J. Hu, L. Huang, X. Zhuang, X. Chen, Y. Wei and X. Jing, J. Polym. Sci., Part A: Polym. Chem., 2008, 46, 1124–1135 CrossRef CAS.
- J. Hu, L. Huang, X. Zhuang, P. Zhang, L. Lang, X. Chen, Y. Wei and X. Jing, Biomacromolecules, 2008, 9, 2637–2644 CrossRef CAS PubMed.
- J. Min, Y. Jung, J. Ahn, J. G. Lee, J. Lee and S. H. Ko, Adv. Mater., 2023, 35, e2211273 CrossRef PubMed.
- J. Vohlídal, Chem. Teach. Int., 2021, 3, 213–220 CrossRef.
- W. L. Hawkins, Polymer degradation and stabilization, Springer, 1984, pp. 3–34 Search PubMed.
- N. Lucas, C. Bienaime, C. Belloy, M. Queneudec, F. Silvestre and J.-E. Nava-Saucedo, Chemosphere, 2008, 73, 429–442 CrossRef CAS PubMed.
- R. R. A. Silva, C. S. Marques, T. R. Arruda, S. C. Teixeira and T. V. de Oliveira, Macromol, 2023, 3, 371–399 CAS.
- S. W. Hwang, J. K. Song, X. Huang, H. Cheng, S. K. Kang, B. H. Kim, J. H. Kim, S. Yu, Y. Huang and J. A. Rogers, Adv. Mater., 2014, 26, 3905–3911 CrossRef CAS PubMed.
- W. Li, Q. Liu, Y. Zhang, C. A. Li, Z. He, W. C. Choy, P. J. Low, P. Sonar and A. K. K. Kyaw, Adv. Mater., 2020, 32, 2001591 CrossRef CAS PubMed.
- N. Nair, V. Sekhar, K. Nampoothiri and A. Pandey, Current developments in biotechnology and bioengineering, Elsevier, 2017, pp. 739–755 Search PubMed.
- N. S. M. Ramdzan, Y. W. Fen, N. A. A. Anas, N. A. S. Omar and S. Saleviter, Molecules, 2020, 25, 2548 CrossRef CAS PubMed.
- L. Madej-Kiełbik, K. Gzyra-Jagieła, J. Jóźwik-Pruska, R. Dziuba and A. Bednarowicz, Materials, 2022, 15, 7493 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |




