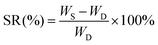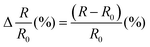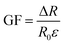A multifunctional anti-swelling hydrogel with high stretchability and adhesion for wearable flexible sensors
Qingyu
Han
ab,
Manzhou
Li
a,
Chi
Liu
ab,
Ying
Qu
ab,
Surui
Li
ab and
Shuang
Guan
 *ab
*ab
aCollege of Chemistry and Life Sciences, Changchun University of Technology, Changchun 130012, China. E-mail: guanshuang@ccut.edu.cn
bAdvanced Institute of Materials Science, Changchun University of Technology, Changchun 130012, China
First published on 24th October 2025
Abstract
Hydrogels have properties such as softness and stretchability, making them widely used in smart wearable electronics. However, they tend to swell underwater, limiting their application scenarios. This work presents an ACTA-SP hydrogel prepared using chitosan (CS) and a copolymer (AA-co-SBMA) consisting of acrylic acid and [2-(methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl) ammonium hydroxide, tannic acid (TA), Al3+ metal ions, and the conductive polymer poly(3,4-ethyl-enedioxythiophene)/poly(styrene sulfonate) (PEDOT:PSS). This multifunctional hydrogel exhibits outstanding anti-expansion properties in various solution environments. Meanwhile, it exhibits underwater adhesion to various substrates and excellent tensile strength (1072%). In the field of sensor applications, this hydrogel can achieve a wide detection range of 0–500% and a high strain coefficient of GF ≈ 6.66. It is notable that the ACTA-SP hydrogel can achieve precise, continuous and real-time dynamic monitoring of human movement postures and transmit command information via Morse code. This study provides an innovative approach to design multifunctional hydrogel-based underwater wearable sensors and expands the prospects for the application of hydrogels in flexible wearable devices.
1. Introduction
Various smart, electronic cutting-edge technologies have emerged with the continuous intellectualization and informatization of the times. In recent decades, intelligent wearable sensors have been widely used for behavior capture,1–3 health monitoring,4–6 human–computer interactions,7–9 electronic skin,10–12 and soft robotics.13–15 There is an urgent need for materials that possess flexibility, electrical conductivity, and excellent mechanical properties. Hydrogels, consisting of three-dimensional crosslinked polymer networks, are known for their excellent flexibility, biocompatibility, and tunable properties, endowing them with great potential for wearable sensor applications. However, the inherent hydrophilicity of hydrogels makes them prone to expansion in underwater or humid environments, damaging the internal structure and leading to a decline in mechanical properties. Furthermore, the hydration at the contact interface between the hydrogel and the substrate will significantly weaken its adhesion in this environment. It hinders the immediacy and accuracy of the sensing signal and limits the application scenarios of hydrogel-based flexible wearable sensors. Therefore, the development of multifunctional hydrogels with both anti-expansion performance and underwater adhesion performance is of great significance for achieving precise monitoring of human behavior signals in underwater or humid environments.There are generally three approaches to solving the swelling problem of hydrogels.16 The first is a solvent exchange strategy that utilizes the tunability and reversibility of noncovalent bonding to improve the swelling resistance of hydrogels. Pan et al. successfully developed an anti-swelling hydrogel by introducing hydrophobic substances and Zr4+ into the system through π–π stacking and cation–π interactions.17 The second strategy is to design the molecular structure to increase the crosslink density, thus limiting the swelling of the hydrogel. Sun et al. proposed a supramolecular strategy to develop hydrogels with excellent anti-swelling performance by introducing hydrogen bonds, coordination bonds, and cationic–π interactions between “rigid” conductive polymers and “flexible” hydrogel matrices.18 The third strategy is surface modification, whereby the surface is rendered hydrophobic using appropriate methods to inhibit water penetration. Yang et al. obtained an anti-swelling hydrogel by polymerizing hydroxyethyl methacrylate (HEMA) in ethylene glycol and then immersing it in water. The synergistic effect of hydrogen bonds and hydrophobic interactions endows the hydrogel with anti-swelling properties in water.19 Du et al. randomly polymerized the hydrophilic monomers N-(2-hydroxyethyl) acrylamide (HEAA) and AA with the hydrophobic monomer methyl methacrylate (MMA) in the presence of cationic surfactant cetyltrimethylammonium bromide (CTAB) micelles. Chitosan undergoes chain entanglement during soaking in sodium phytate (PANa), thereby forming a hydrophobic hydrogel with excellent anti-swelling performance.20 Although the hydrogels in these studies each have their own advantages, the hydrogels used as underwater sensors still need to exhibit excellent electrochemical performance, long-term stability, and, more importantly, adhesion to different substrates in terrestrial and underwater environments.
The excellent adhesion of hydrogels underwater is mainly due to the unique hydrophobic structure and surface microstructure on the gel surface penetrating the hydration layer. Ling et al. constructed DCP hydrogels with good adhesion to animal tissues based on reversible dynamic chemical bonds and physical interactions.21 However, these hydrogels suffer from relatively low adhesion strength, and their swelling behavior prevents them from maintaining stable and accurate signal output underwater. Additionally, a narrow sensing response range, low sensitivity, and poor accuracy further limit their practical applications in underwater environments.
This study proposes a one-pot method for preparing multiplex cross-linked hydrogels based on CS and P(AA-co-SBMA). The system's multiple intermolecular interactions such as hydrogen bonds, ionic bonds and electrostatic interactions form a dense cross-linked network. The introduction of PEDOT:PSS improves the hydrogel's mechanical properties and enhances its electrical conductivity. Amphoteric SBMA enables hydrogels to exhibit anti-swelling properties in different solution environments and adhesion to various materials in underwater environments. The introduction of TA containing catechol groups into the system improved the underwater adhesion performance of the hydrogel. The obtained ACTA-SP hydrogel also has a wide strain detection range (1–500%) and a high strain coefficient (GF ≈ 6.66). It has been confirmed that the strain sensor assembled using the ACTA-SP hydrogel can effectively detect the movement of human joints and muscles. In order to fully verify the practical potential and market applicability of the ACTA-SP hydrogel as an underwater wearable sensor, this study successfully applied it to the underwater Morse code information transmission scenario. The experimental results show that this sensor exhibits excellent signal transmission stability, sensitivity and durability in complex underwater environments. This experiment demonstrated not only its broad application prospects in professional fields such as deep-sea exploration and underwater search and rescue, but also its significant application potential in consumer markets such as underwater sports safety monitoring. This material provides a reliable technical means for solving underwater human–machine interactions, equipment status monitoring and emergency communication, and lays the key technical foundation and market competitiveness for developing high-performance and low-cost underwater intelligent equipment.
2. Experimental section
2.1. Materials
Acrylic acid (AA, 99%), chitosan (CS, 95%), aluminum chloride hexahydrate (AlCl3·6H2O, 97%), poly(3,4-ethyl-enedioxythiophene)/poly(styrene sulfonate) (PEDOT:PSS, 1.5% in water), [2-(methacryloyloxy)ethyl]dimethyl-(3-sulfopropyl)ammonium hydroxide (SBMA, 99%), glacial acetic acid (HAc), N,N'-methylene-bis-acrylamide (MBA), sodium chloride (NaCl), potassium chloride (KCl), magnesium sulfate (MgSO4), magnesium chloride (MgCl2) and calcium chloride (CaCl2) were acquired from Aladdin (Shanghai, China). Tannic acid (TA) and ammonium persulfate (APS, 98%) were purchased from Shanghai Macklin Biochemical Co., Ltd (Shanghai, China). Artificial seawater was prepared by dissolving specific concentrations of NaCl (26.7 g L−1), KCl (7 g L−1), MgSO4 (3.2 g L−1), MgCl2 (2.3 g L−1), and CaCl2 (1.15 g L−1) in water.2.2. Preparation of ACTA-SP hydrogels
CS was completely dissolved in a 5% acetic acid solution. Then, the monomer, TA, the amphoteric substance SBMA, Al3+ metal ions, and different masses of the high-molecular-weight conductive polymer PEDOT:PSS (3 wt%, 6 wt%, 9 wt%, and 12 wt%) were added into the solution and the solution was stirred until it was completely dissolved. Subsequently, the crosslinking agent N,N′-methylene-bis-acrylamide (MBA, 0.2 wt%) and the heat initiator ammonium persulfate (APS, 0.5 wt%) were added into the solution. After thorough stirring for one hour, ultrasonic treatment was carried out for 20 minutes to eliminate bubbles. The solution was poured into a mold and a heat-initiated polymerization reaction was conducted at 70 °C for six hours to form ACTA-SP hydrogels.2.3. FT-IR analysis
The characteristic functional groups of the pure AA hydrogel, AC hydrogel, ACT hydrogel, ACTA hydrogel, ACTA-S hydrogel, and ACTA-SP hydrogel were analyzed within the spectral range of 500–4000 cm−1 using a Fourier transform infrared spectrometer (FT-IR, Bruker ALPHA II, GER; resolution: 4 cm−1, number of scans: 32).2.4. Mechanical tests
The test was conducted using a universal tensile testing machine (AGS-X, Shimadzu, Japan) at a tensile speed of 80 mm min−1. The samples used for the test were cut into dumbbell shapes with a width of 4 mm and a thickness of 2 mm to standardize the sample size. After the initial loading, different tensile strains were applied, and cyclic tensile tests were conducted under the same holding conditions. The experimental conditions for single and cyclic tests were the same. The toughness of the hydrogels is calculated using the area under the integral stress–strain curve. The dissipated energy is calculated using the area of the closed graph enclosed by the loading stress–strain curve.A cylindrical hydrogel sample with a height of 8 mm and a diameter of 20 mm was subjected to a cyclic compression test at a speed of 2 mm s−1 using a texture analyzer. The error bar data in this article are based on three independent and repeated experiments.
2.5. Anti-swelling tests
The prepared hydrogel samples were molded into the same shape (a circular shape with a diameter of 12 mm and a thickness of 2 mm) and immersed in deionized water, artificial seawater and different solvents. They were taken out regularly, wiped dry with a dust-free cloth, and weighed with a balance, and the changes in weight over time were recorded. The swelling rate (SR) is calculated using the following formula:where WD represents the original hydrogel's weight and WS represents the weight of the hydrogel after swelling.
2.6. Adhesiveness tests
The adhesion properties between multi-component hydrogels and various substrates were tested using Lap-shear bonding tests (AGS-X, Shimadzu, Japan). The selected base materials are stainless steel, copper, zinc, silicone and glass. The contact area of the hydrogel samples with different substrate surfaces was 10 mm × 10 mm. Lap shear tensile tests were performed immediately after compression, and the bond strength was obtained by dividing the maximum load by the contact area.2.7. Electrical tests
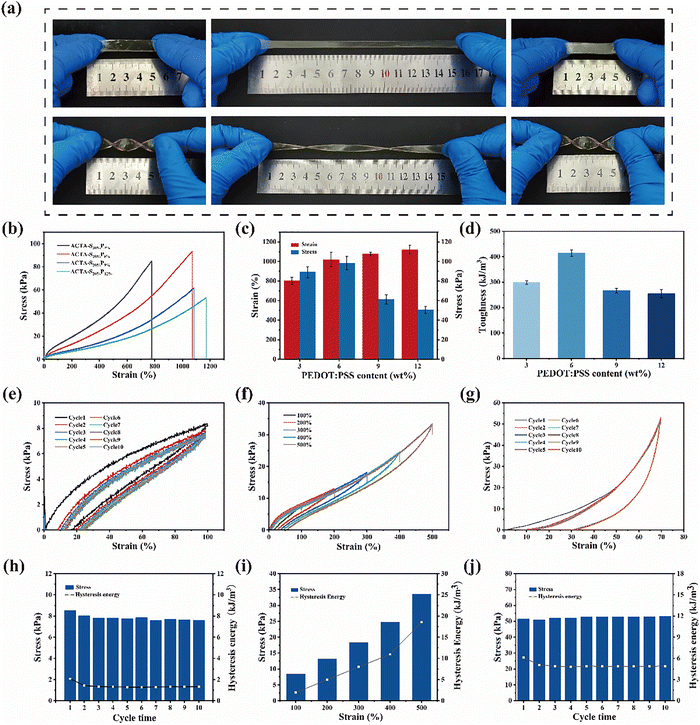
A digital multimeter (B35T + 1/2 digit, Owon, China) coupled with a universal testing machine (AGS-X, Shimadzu, Japan) was used to record the electrical signal response of the ACTA-SP hydrogel. The digital multimeter is responsible for recording the output resistance. The relative change in the resistance of the hydrogel during the testing process is calculated using the following formula:where R0 is the initial resistance of the hydrogel during testing and R is the real-time resistance during testing. The strain coefficient (GF) is calculated using the following formula:
where ε is the applied strain.
2.8. Strain-gel sensor testing and motion monitoring
The strain sensor was fabricated by attaching copper foil tapes to each end of a rectangular hydrogel sample measuring 45 × 10 × 2 mm3. An electrochemical workstation (PARSTAT3000, USA) was used to monitor real-time changes. The sensors were then fixed to various body parts, including fingers, wrists, elbows, and knees, to track the body's movements. Fingers and wrists were also placed underwater to monitor the sensor's movement tracking performance. All the experimental procedures involved in this study were carried out in accordance with the principles of the Helsinki Declaration. All participants had read and signed the written informed consent form before participating in the experiment.3. Results and discussion
3.1. Preparation of the ACTA-SP hydrogel and the gel forming mechanism
The multifunctional ACTA-SP hydrogel was prepared by a one-pot synthesis method: first, CS was completely dissolved, and monomers AA and TA, amphoteric substance SBMA, metal ions Al3+ and conductive polymer PEDOT:PSS were added and allowed to dissolve completely; then the crosslinking agent MBA and the heat initiator APS were added and stirred thoroughly for 1 hour. After ultrasonic treatment, the solution was poured into a mold and subjected to a thermally initiated polymerization reaction to form the ACTA-SP hydrogel. In the ACTA-SP hydrogel, the P(AA-co-SBMA) chain is composed of AA and SBMA, forming a hydrogel network with CS, TA and Al3+. As shown in Fig. 1a, after the introduction of PEDOT:PSS, the sulfonate ions carried by PSS in its components can form hydrogen bonds with the hydroxyl groups in P(AA-co-SBMA) and chitosan molecules. Meanwhile, strong dipole–dipole interactions occur between the zwitterionic groups in the system. These interactions collectively facilitate the effective stabilization of the three-dimensional network structure of the hydrogel by the zwitterionic SBMA. Additionally, the phenolic hydroxyl groups in TA form stronger hydrogen bonds with the carboxyl groups on P(AA-co-SBMA), while O− establishes electrostatic interactions with the quaternary ammonium cations in the network, further enhancing the stability of the hydrogel network structure.In addition, in order to further confirm the internal structure of the hydrogels, FTIR and SEM characterization tests were conducted. By FTIR spectroscopy, we analyzed the chemical structures of the hydrogels (Fig. 1b and c), and it can be seen that from AA to AC, the peak at 3343 cm−1 shifted to a smaller wavelength, confirming the existence of hydrogen bonding and electrostatic interactions between the AA chains and CS.22 The peak at 1702 cm−1 is attributed to the bending vibration of the carboxyl group C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, which is caused by AA–Al3+ chelation or electrostatic interactions between AA and CS.23,24 This peak for the other hydrogels undergoes a redshift and becomes sharper, indicating that a large number of hydrogen bonds are formed and strengthened in the hydrogel network. The peak at 2948 cm−1 indicates a stretching vibration of the hydroxyl group in AA, and the fact that this peak becomes sharper in ACT implies that a hydrogen bond has formed between the hydroxyl groups of AA and TA.25 The two peaks at 3357 cm−1 and 3253 cm−1 became sharper than those of the AC hydrogel and shifted to the left. This is due to the overlap of the phenolic hydroxyl group of TA with the hydroxyl and amine groups of CS, as well as the formation of hydrogen bonds and electrostatic interactions between TA and CS.26,27
O, which is caused by AA–Al3+ chelation or electrostatic interactions between AA and CS.23,24 This peak for the other hydrogels undergoes a redshift and becomes sharper, indicating that a large number of hydrogen bonds are formed and strengthened in the hydrogel network. The peak at 2948 cm−1 indicates a stretching vibration of the hydroxyl group in AA, and the fact that this peak becomes sharper in ACT implies that a hydrogen bond has formed between the hydroxyl groups of AA and TA.25 The two peaks at 3357 cm−1 and 3253 cm−1 became sharper than those of the AC hydrogel and shifted to the left. This is due to the overlap of the phenolic hydroxyl group of TA with the hydroxyl and amine groups of CS, as well as the formation of hydrogen bonds and electrostatic interactions between TA and CS.26,27
The characteristic peaks at 1122 cm−1 and 1043 cm−1 in ACTA-S are observed, which are attributed to the symmetric and asymmetric tensile vibrations of S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, which are due to the presence of sulfonic acid groups in the hydrogel,28 indicating the successful introduction of SBMA. After the addition of PEDOT:PSS, the peak at 985 cm−1 in ACTA-SP is attributed to the C–C stretching vibration on the thiophene ring of PEDOT, the C
O, which are due to the presence of sulfonic acid groups in the hydrogel,28 indicating the successful introduction of SBMA. After the addition of PEDOT:PSS, the peak at 985 cm−1 in ACTA-SP is attributed to the C–C stretching vibration on the thiophene ring of PEDOT, the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond absorption peak in PSS at 1637 cm−1, and the C–O stretching vibration on the thiophene ring at 1227 cm−1. The results showed that the ACTA-SP hydrogel was successfully synthesized. Fig. 1d shows the SEM image of the AA-CS-TA-Al3+ (ACTA) and AA-CS-TA-Al3+-SBMA-PEDOT:PSS (ACTA-SP) hydrogels. Hydrogels containing SBMA and PEDOT:PSS formed a denser structure. Thanks to this structure, their tensile properties and anti-swelling performance were synergistically enhanced.
C double bond absorption peak in PSS at 1637 cm−1, and the C–O stretching vibration on the thiophene ring at 1227 cm−1. The results showed that the ACTA-SP hydrogel was successfully synthesized. Fig. 1d shows the SEM image of the AA-CS-TA-Al3+ (ACTA) and AA-CS-TA-Al3+-SBMA-PEDOT:PSS (ACTA-SP) hydrogels. Hydrogels containing SBMA and PEDOT:PSS formed a denser structure. Thanks to this structure, their tensile properties and anti-swelling performance were synergistically enhanced.
3.2. Mechanical properties
Excellent mechanical properties are an important prerequisite for hydrogels as sensors. In this study, a complete, dense, and stable 3D hydrogel network was constructed based on the synergistic effect of multiple interactions, such as hydrogen bonding, ionic bonding, and electrostatic interactions, between different substances. In this paper, systematic mechanical property tests were conducted on the ACTA-SP hydrogel to demonstrate its excellent mechanical properties. As shown in Fig. 2a, the ACTA-SP hydrogel can elongate three times its original length in curled and straightened states without breaking, and it can recover its original state immediately after removal of the external force. These phenomena intuitively show that the ACTA-SP hydrogel has excellent toughness and self-recovery properties. There are strong dipole–dipole interactions between the amphoteric ionic groups. Therefore, the amphiphilic component SBMA in the system effectively stabilized the three-dimensional network structure of the hydrogel, thereby significantly enhancing its mechanical properties. The introduction of chitosan and the chelation effect of Al3+ further promoted the entanglement between polymer chains, enhancing the stability of the hydrogel network. Additionally, the hydroxyl groups in the TA molecule formed strong hydrogen bonds with the carboxyl groups on the P(AA-co-SBMA) chain, while the ionization of TA produced O−, which interacted electrostatically with the quaternary ammonium cations in SBMA, jointly contributing to the improvement of the water gel's toughness.We further investigated the effect of different PEDOT:PSS contents on the mechanical properties of the hydrogels. From the tensile stress–strain curves shown in Fig. 2b, it can be seen that the hydrogel with 6 wt% PEDOT:PSS content exhibits the best mechanical properties, with a fracture strength of 93 kPa (Fig. 2c), a fracture strain of up to 1072% (Fig. 2c), and a toughness of 413 kJ m−3 (Fig. 2d). Compared with hydrogels with other PEDOT:PSS contents, this hydrogel exhibits a significant improvement in fracture stress and toughness. As the polystyrene sulfonate ions in PEDOT:PSS can form hydrogen bonds with the hydroxyl groups on P(AA-co-SBMA) and chitosan, the physical entanglement effect in the polymer network is enhanced, thereby contributing to the formation of a denser hydrogel network structure. Therefore, as the amount of PEDOT:PSS increases, the tensile strength of the hydrogel also increases. However, excessive PEDOT:PSS will weaken the above two interactions, which may be due to its self-aggregation behavior, resulting in weakened hydrogen bond effects and damage to the integrity of the polymer network structure. Thus, when the content of PEDOT:PSS exceeds 6 wt%, the mechanical properties of the hydrogel decline, which is consistent with the results of the rheological test (Fig. S1). Therefore, in this system, when the content of PEDOT:PSS is 6 wt%, the ACTA-SP hydrogel exhibits optimal mechanical properties.
Cyclic loading–unloading tests were conducted to further prove the fatigue resistance and rebound ability of the ACTA-SP hydrogel. As shown in Fig. 2e, the tensile curve of the hydrogel loading and unloading 10 times under 100% strain is presented. The area within the loading–unloading curve represents the energy dissipation of the hydrogel during the stretching process. It can be seen that there is a large hysteresis line in the first stretching cycle, and the hysteresis lines become smaller and overlap in the following nine stretching cycles. It can be seen from Fig. 2h that the stress slightly decreases during ten cycles of stretching of the hydrogel. The dissipated energy is 2.07 kJ m−3 in the first cycle and promptly decreases to 1.42 kJ m−3 in the following cycles, but remains above 1.33 kJ m−3 in the tenth cycle. This further explains that most energy dissipation occurs in the first cycle. This reflects the excellent fatigue resistance of the ACTA-SP hydrogel. Afterwards, the hydrogel was subjected to loading–unloading tests at different strains (100–500%). As shown in Fig. 2f, with the increase of tensile strain, the area surrounded by hysteresis lines gradually increased, and the overlapping area between every two adjacent hysteresis lines also gradually increased. From Fig. 2i, it can be seen that the dissipated energy increases with increasing strain during stretching at different strains, implying that the hydrogel can realize energy dissipation through multiple interactions in the 3D network. The hydrogen bonds formed by PSS with TA, P(AA-co-SBMA), and chitosan ensure that the hydrogen bonds within the hydrogel cross-linking network can play the “sacrificial structure” role. This structure can achieve effective energy dissipation, causing the hydrogel to undergo rapid local rebound after being unloaded, indicating that it possesses excellent elasticity and fatigue resistance.
Furthermore, the compressive elastic recovery of ACTA-SP hydrogels was evaluated by 70% cyclic compressive loading–unloading experiments. As shown in Fig. 2g, the 10-cycle compression loading–unloading cycle curves almost completely overlapped. The stresses and dissipated energies in Fig. 2j show similar results to cyclic stretching. This indicates that the ACTA-SP hydrogel has excellent elasticity and fatigue resistance, enabling its continuous application in practice.
3.3. Anti-swelling properties
The swelling resistance of hydrogels is critical for application in underwater sensors. Due to the hydrophilic groups present in the structure of traditional hydrogels, they expand in water or other solvents, causing damage to the internal structure and resulting in breakage. Therefore, this study investigates the swelling behavior of the ACTA-SP hydrogel. Fig. 3a and b show the swelling behavior of the ACTA-SP hydrogel placed in water and seawater for 30 days. It can be observed that the hydrogel reaches the swelling equilibrium after 4 days of immersion and maintains a low swelling rate. The low swelling rate is mainly attributed to the electrostatic interactions and hydrogen bonds within the hydrogel network. Specifically, on one hand, the sulfonic acid anions in SBMA strongly attract the quaternary ammonium cations; on the other hand, the sulfonic acid anions can also form hydrogen bonds with the acrylic units, thereby enhancing the cross-linking between the molecular chains of P(AA-co-SBMA). These interactions collectively inhibit the penetration of water molecules effectively, enabling the hydrogel to possess excellent anti-swelling properties. In the seawater environment, the hydrogel exhibits a high swelling rate, which may be attributed to the shielding effect of low-molecular-weight electrolyte salts on the surface charge of the hydrogel, facilitating the hydration of zwitterions,29 thereby causing osmotic swelling. The above results indicate that this hydrogel has good stability under pure water and seawater conditions. Additionally, to systematically evaluate the anti-swelling performance of the hydrogel in different solvent environments, the ACTA-SP hydrogel was immersed in chloroform, hexane, ethyl acetate, and petroleum ether. As shown in Fig. 3c, the hydrogel exhibited excellent anti-swelling ability in these organic solvents. However, due to the influence of solvation, the hydrogel underwent a more significant swelling in ethanol.In addition, the swelling behavior of the ACTA-SP hydrogel in different pH environments was also investigated in this study (Fig. 3d). It can be seen that the swelling rate of the hydrogel under alkaline conditions is significantly higher than that under neutral and acidic conditions. This is mainly because the higher ionic concentration in the alkaline environment inhibits the ionization of the polyelectrolyte, triggering the anti-electrolyte effect, causing dissociation of the P(AA-co-SBMA) network. The internal interactions are disrupted, thereby promoting the swelling of the hydrogel.30 Mechanical properties after immersion are also crucial for hydrogel sensors that are used in underwater environments. As shown in Fig. 3e, after being immersed for 72 hours (3 days) under different pH conditions, the fracture stress of the hydrogel decreased only slightly. When the immersion time was extended to 7 days and 10 days (Fig. 3f), the fracture stress significantly decreased, but then stabilized. The difference in values between 7 days and 10 days was not significant. This indicates that the ACTA-SP hydrogel can maintain stable mechanical properties under different acidic and alkaline conditions. In conclusion, the ACTA-SP hydrogel shows stable anti-swelling properties in different water environments (seawater, different pH solutions, and different solvents).
3.4. Adhesive properties
For hydrogel sensors, good adhesion can avoid the gap problem at the interface between hydrogel sensors and skin or other substrates, improving the stability and consistency of the strain sensing process. As shown in Fig. 4a, the ACTA-SP hydrogel can adhere stably to surfaces of different materials and various shapes, including silica gel, metals, plastic, rubber, wood, and ceramics. As a result, we investigated the adhesion performance of the hydrogel toward the surfaces of different substrates through shear experiments (Fig. 4b). Fig. 4c shows the adhesion strength between the hydrogel and copper, zinc, silicone, and glass, with the adhesion strength to copper reaching a maximum of 65.35 kPa. The sulfonic acid groups and quaternary ammonium ions in the oppositely charged amphiphilic SBMA can achieve interfacial binding through interactions with polar or charged surfaces.31,32 Meanwhile, the catechol groups in the TA molecule can rapidly form non-covalent interactions such as hydrogen bonds and metal–ligand bonds on various substrates. These properties enable the ACTA-SP hydrogel to effectively adhere to different substrate surfaces.33,34Afterwards, the excellent adhesion strength between the hydrogel and different substrates was further confirmed by peel force tests. As shown in Fig. 4d, the 90° peel force tests between the hydrogel and pig skin, aluminum sheets, titanium sheets, silicone, plastic and glass were conducted. Among them, the peel force between the hydrogel and pigskin was the maximum, reaching 279 N m−1. The ACTA-SP hydrogel exhibits strong adhesion to pig skin, which is mainly due to the presence of charged groups in the pig skin collagen. These groups can form electrostatic interactions with the charged groups of CS and SBMA in the hydrogel, thereby creating strong interfacial bonding.35 At the same time, the TA in the hydrogel contains abundant catechol groups, which can undergo Michael addition reactions with the amino groups in the pig skin to form dynamic covalent bonds, further enhancing the adhesion effect.36,37 Additionally, the adhesion of this hydrogel to metal surfaces stems from the coordination interactions between P(AA-co-SBMA), CS, and TA and metal ions. In addition, the repeated adhesion ability of the hydrogel was also investigated. Fig. 4e shows the repeated peel force test of the hydrogel and the aluminum sheet. After ten repeated peeling cycles, the adhesion strength decreased, mainly due to the contamination or damage of the sample surface during the peel test, but its strength remained above 60 N m−1. The excellent adhesion performance of the ACTA-SP hydrogel in air provides favorable conditions for its application in sensors.
3.5. Underwater adhesiveness properties
In addition to its excellent adhesion in dry environments, the ACTA-SP hydrogel maintained a certain level of adhesion in underwater environments. As shown in Fig. 5a, the hydrogel can establish adhesion with pork rinds and shrimps underwater, and lift up the pork rinds and shrimp. The shells of crustaceans similar to shrimp are mainly composed of positively charged chitin, calcium minerals and proteins. These components can achieve underwater adhesion of the hydrogel through cooperative electrostatic interactions and the dynamic catechol reaction on the TA.38 Subsequently, we combined the ACTA-SP hydrogel with different substrates and tested their shear adhesion strength after being immersed in water for 30 minutes and 10 days (Fig. 5b). The results showed that the adhesion strength decreased by a relatively small amount after 30 minutes of immersion; even after 10 days of immersion, the strength remained above 30 kPa. Additionally, peel force tests (Fig. 5c) and repeated peel tests (Fig. 5d) were conducted on the samples after 30 minutes of immersion. It is noteworthy that after six repeated peel cycles on the porcine skin substrate, although the adhesion performance slightly declined, it remained above 120 N m−1. This indicates that the adhesion of the hydrogel underwater is reproducible. This is mainly because multiple electrostatic interactions within the system effectively suppress the hydration effect at the water–gel interface. The strong electrostatic coordination bonds formed between PAA, TA and Al3+ provide additional crosslinking sites, while also hindering the formation of hydrogen bonds between PAA and H2O or between TA and H2O, thereby significantly reducing the swelling rate.39 Furthermore, the dynamic crosslinking structure within the three-dimensional network of the hydrogel can effectively mitigate the damage to the interface caused by external forces through the energy dissipation mechanism, thereby maintaining its underwater adhesion performance to a certain extent.403.6. Electrical properties
The ACTA-SP hydrogel has excellent mechanical properties, anti-swelling properties, and underwater adhesion performance, highlighting its potential as a hydrogel sensor. Fig. 6a–c show that the water gel sensor can sensitively monitor the changes in relative resistance under different tensile strains and exhibits accurate responsiveness to a wide range of strains, including minor (1%, 3%, 5%, and 7%), medium (30%, 50%, 70%, and 90%), and large (100%, 200%, 300%, 400%, and 500%). According to different strains, (R − R0)/R0 has a clear distinction, and even a strain as small as 1% can have good sensitivity and accuracy. As the content of PEDOT:PSS increases, the electrical conductivity of the ACTA-SP hydrogel also increases. When the content reaches 6 wt%, the electrical conductivity is 0.278 S m−1 (Fig. S2). Furthermore, this hydrogel exhibits responsiveness and stability to different strain frequencies (Fig. S3). Fig. 6d shows the changes in relative resistance upon application of stepwise strains to the ACTA-SP hydrogel sensor. It is observed that the hydrogel sensor stably records the electrical signals of different strains from small to large, and there are significant differences. Furthermore, during the strain recovery process, there is no significant difference in the intensity of the relative resistance change corresponding to the deformation, indicating that the hydrogel has good self-recovery performance. Response time and recovery time refer to the time interval between the strain change and the corresponding resistance change, and they are also necessary for the sensor to respond to stimuli quickly and effectively. The rapid response time of the ACTA-SP hydrogel during the loading process was 100 ms, and the recovery time during the unloading process was 130 ms (Fig. 6e), which was approximately the same as the response time of human skin (about 100 ms). | ||
| Fig. 6 Relative resistance changes of the ACTA-SP hydrogel at different strains: (a) small strains (1–7%), (b) medium strains (30–90%), and (c) large strains (100–500%). (d) Changes in the relative resistance of ACTA-SP hydrogel sensors reversibly stretched at different angles (0, 25, 50, and 75%). (e) Response time of the ACTA-SP hydrogel at 25% strain. (f) The variation curve of relative resistance over 1000 consecutive loading–unloading cycles with a strain of 10%. (g) Relative resistance–strain curves of GF values (sensitivity) represented by ACTA-SP hydrogel sensors at different strains. (h) The relative resistance–strain curves of GF values (sensitivity) represented by the ACTA-SP hydrogel sensor at different strains after immersion in water for 72 hours. (i) Comparison of this work with the comprehensive performance of recently reported sensors.41–43 | ||
In addition, as shown in Fig. 6f, the change of (R − R0)/R0 was recorded during 1000 consecutive stretching cycles at 10% strain to investigate the long-term stability of the hydrogel. The ACTA-SP hydrogel still maintained stable and sensitive sensing capability after 1000 cycles. However, the sensing performance curve deviated throughout the cyclic stretching process. This phenomenon is mainly attributed to the temperature changes during the long-term testing process, which may accelerate water loss in the hydrogel and increase resistance. It is noteworthy that both the magnitude and shape of the strain response signal are consistent and stable for the process despite the drift of the curve. The strain factor (GF) is the relative rate of resistance change of a hydrogel at different tensile strains, indicating the sensor's sensitivity. As shown in Fig. 6g, the GF of the ACTA-SP hydrogel was 1.45 in the 1–9% strain range, increased to 3.10 in the 30–90% strain range, and was as high as 6.66 in the extensive strain range of 100–500%. With the increase of strain, the GF also increased, indicating that the ACTA-SP hydrogel has excellent strain sensitivity. The positively charged quaternary ammonium salt cationic groups and negatively charged sulfonic acid groups on the amphiphilic SBMA ions in the system will form ion migration channels under the action of an external electric field. The formation of this channel endows the ACTA-SP hydrogel with sensitive responsiveness. The GF of the ACTA-SP hydrogel after being placed underwater for 72 hours is shown in Fig. 6i. There is a slight decrease compared to that before immersion, indicating that the hydrogel is still able to have highly sensitive responsiveness even when it is exposed to water for an extended period. The change in LED brightness can be visualized by stretching and releasing the hydrogel in a connecting circuit in air and water (Fig. S4). The sensitive response, underwater stability, and reusability of the ACTA-SP hydrogel enable it to provide stable sensing signals as a sensor over a long period of repeated use. Compared with other reported hydrogel sensors, the ACTA-SP hydrogel sensor exhibits comprehensive advantages in multiple aspects such as stretchability, adhesion, sensitivity and anti-swelling properties (Fig. 6i). These characteristics make it a highly promising material for underwater flexible electronic devices, enabling stable operation in humid or underwater environments.
3.7. Strain sensing behavior
The ACTA-SP hydrogel is adhesive, can adhere directly and stably to the skin, and can capture and monitor human movement signals. Fig. 7 shows the relevant applications of hydrogels as sensors for detecting human movements. The ACTA-SP hydrogel sensor is attached to the finger. As the finger bends and straightens, the hydrogel deforms, causing fluctuations in the relative resistance, which are recorded. As shown in Fig. 7a, the (R − R0)/R0 value changed with finger movements and gradually recovered when the bending was gradually restored to the straightened state. In addition to the finger, the hydrogel has also adhered to other joints of the body to monitor human motion, and the changes in relative resistance signals generated by the wrist (Fig. 7b), elbow (Fig. 7c), and knee (Fig. 7d) during bending motion were recorded. It is also worth emphasizing that the signal peaks generated by repeated limb movements clearly distinguish height and width. And it can respond quickly to the real-time resistance changes of limb movements, indicating that the ACTA-SP hydrogel, as a sensor, has good reliability and repeatability.In addition to monitoring strenuous body movements in real time, the ACTA-SP hydrogel sensor can also capture subtle body changes. As shown in Fig. 7e, with the tightening and relaxation of the eyebrows, the (R − R0)/R0 value of the hydrogel shows regular changes; the (R − R0)/R0 value of the hydrogel sensor attached to the neck changed according to the swallowing action (Fig. 7f), and the current signal changes in both of these cases could be clearly distinguished. This indicates that the hydrogel has the ability and sensitivity to strain recognition for tiny movements.
Furthermore, as shown in Fig. 7g and h, the hydrogel is still able to follow the movement of the finger and wrist underwater with the change of resistance, and the change of the electrical signal can be recorded stably. However, there is a slight gap in the peak shape of the signal from the hydrogel sensor due to the difference in human limb movements, which may affect the hydrogel's monitoring accuracy.
3.8. Underwater communication sensor
With the continuous exploitation of marine resources and the increase in the number of diving enthusiasts, underwater communication devices that transmit real-time information have received widespread attention. Taking advantage of the strain sensitivity of the ACTA-SP hydrogel and its excellent underwater sensing performance, it was assembled as a hydrogel sensor. The hydrogel sensor was attached to the finger to monitor signals (Fig. 8a). According to the encoding principle of Morse code (Fig. 8b), minor angle bending (0°) and large angle bending (90°) on the finger were denoted as “·” and “-”. Underwater personnel can send distress or command messages by regularly bending their finger to form a specific Morse code. For example, three repetitions of bending the finger at 45° to form an “S” and three repetitions of bending the finger at 90° to form an “O” can be combined to produce the international distress signal “SOS” (Fig. 8c). The international distress signal “SOS” can be decoded by combining the messages. Similarly, other complex messages that can be encoded using Morse code with 26 letters can also be arranged and combined through different degrees of finger bending to transmit messages. Information such as “HELP” (Fig. 8b), “UP” and “DOWN” (Fig. 8e) can be transmitted through the ACTA-SP hydrogel sensor based on Morse code. Thus, it can be seen that the ACTA-SP hydrogel sensor has good application potential in various fields such as underwater communication, rescue and detection. The exploration of wearable sensors in the field of communication in underwater environments has great potential. | ||
| Fig. 8 (a) Illustration of an ACTA-SP hydrogel sensor used underwater. (b) Detailed signals in Morse code. (c) (d) and (e) The signals transmitted by fingers at different degrees of bending in water. | ||
4. Conclusion
In conclusion, we prepared multiple cross-linked hydrogels based on CS and P(AA-co-SBMA) through a one-pot method. The multiple intermolecular interactions such as hydrogen bonds, ionic bonds and electrostatic interactions existing in the system form a dense cross-linked network. The introduction of PEDOT:PSS endows the hydrogel with excellent mechanical properties and enhances its electrical conductivity. SBMA and Al3+ endow hydrogels with anti-swelling properties in different solution environments. In addition, the introduction of TA and amphoteric SBMA enables the hydrogel to have adhesion properties to various materials in underwater environments. The obtained hydrogel also has a wide detection range (0–500%) and a high strain coefficient (GF ≈ 6.66). It has been proved that the strain sensor assembled using the ACTA-SP hydrogel can effectively detect human movements (such as limb joint movements from fingers, elbows, wrists and knees, as well as facial muscle movements) and can provide a stable signal output for strain in underwater environments. In conclusion, this research has addressed the issues of underwater swelling, adhesion and sensing of hydrogels, providing new ideas for the application and development of flexible wearable device materials in underwater environments. It further promotes the development of hydrogel flexible sensors in multiple application fields, such as underwater operations, deep-sea rescue, and motion detection.Author contributions
Qingyu Han: investigation, methodology, formal analysis, and writing – original draft. Manzhou Li: review and data curation. Chi Liu: conceptualization and supervision. Ying Qu: methodology, review and editing. Surui Li: data curation and formal analysis. Shuang Guan: writing – review and editing, supervision, funding acquisition, and project administration.Conflicts of interest
There are no conflicts to declare.Data availability
The authors confirm that the data supporting the findings of this study are available within the article.The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5tc02686e.
Acknowledgements
The work was supported by the Science and Technology Development Plan of Jilin Province, sourced from the Department of Science and Technology (20200403025SF).References
- L. Zeng and G. Gao, ACS Appl. Mater. Interfaces, 2023, 15, 28993–29003 CrossRef CAS PubMed.
- J.-Y. Yu, S. E. Moon, J. H. Kim and S. M. Kang, Nano-Micro Lett., 2023, 15, 51 CrossRef CAS PubMed.
- S. Liu, J. Zhang, Y. Zhang and R. Zhu, Nat. Commun., 2020, 11, 5615 CrossRef CAS PubMed.
- X. Wang, H. Ji, L. Gao, R. Hao, Y. Shi, J. Yang, Y. Hao and J. Chen, Chem. Eng. J., 2024, 495, 153382 CrossRef CAS.
- Z. Wang, R. Xue, H. Zhang, Y. Zhang, X. Tang, H. Wang, A. Shao and Y. Ma, ACS Nano, 2024, 18, 7596–7609 CrossRef CAS.
- Y. Yu, Z. Zhou, H. Ruan and Y. Li, Chem. Eng. J., 2025, 505, 158877 CrossRef CAS.
- Z. Fu, D. Li, H. Liu, R. Liu, Q. Lyu, Y. Han, Y. Wang, K. Zhang, G. Chen and Y. Tian, Chem. Eng. J., 2024, 488, 150775 CrossRef CAS.
- W. Li, S. Wu, M. Kang, X. Zhang, X. Zhong, H. Qiao, J. Chen, P. Wang and L. Tao, J. Mater. Sci. Technol., 2024, 201, 130–138 CrossRef.
- W. Wang, H. Zhou, Z. Xu, Z. Li, L. Zhang and P. Wan, Adv. Mater., 2024, 36, 2401035 CrossRef CAS PubMed.
- C. Tian, S. A. Khan, Z. Zhang, X. Cui and H. Zhang, ACS Sens., 2024, 9, 840–848 CrossRef CAS PubMed.
- F.-L. Yi, F.-L. Guo, Y.-Q. Li, D.-Y. Wang, P. Huang and S.-Y. Fu, ACS Appl. Mater. Interfaces, 2021, 13, 32084–32093 CrossRef CAS.
- K. Tao, J. Yu, J. Zhang, A. Bao, H. Hu, T. Ye, Q. Ding, Y. Wang, H. Lin, J. Wu, H. Chang, H. Zhang and W. Yuan, ACS Nano, 2023, 17, 16160–16173 CrossRef CAS.
- C. S. Park, Y.-W. Kang, H. Na and J.-Y. Sun, Prog. Polym. Sci., 2024, 150, 101791 CrossRef CAS.
- Y. Ye, Z. Wan, P. D. S. H. Gunawardane, Q. Hua, S. Wang, J. Zhu, M. Chiao, S. Renneckar, O. J. Rojas and F. Jiang, Adv. Funct. Mater., 2024, 34, 2315184 CrossRef CAS.
- Y. Xin, X. Zhou, M. R. J. Tan, S. Chen, P. Huang, Y. Jiang, W. Wu, D. Gao, J. Lv, S. Magdassi and P. S. Lee, Adv. Mater., 2025, 37, 2415210 CrossRef CAS PubMed.
- S. Wang, J. Liu, L. Wang, H. Cai, Q. Wang, W. Wang, J. Shao and X. Dong, Adv. Mater. Technol., 2023, 8, 2201477 CrossRef CAS.
- S. Pan, C. Chang, G. Wu, Z. He, C. Zhang, S. Xiong, G. Tong and X. Zhu, ACS Appl. Mater. Interfaces, 2025, 17, 9760–9773 CrossRef CAS PubMed.
- Z. Sun, C. Dong, B. Chen, W. Li, H. Hu, J. Zhou, C. Li and Z. Huang, Small, 2023, 19, 2303612 CrossRef CAS.
- Y. Yang, C. Lv, C. Tan, J. Li and X. Wang, ACS Appl. Mater. Interfaces, 2023, 15, 46417–46427 CrossRef CAS.
- P. Du, J. Wang, Y.-I. Hsu and H. Uyama, Chem. Mater., 2024, 36, 1318–1332 CrossRef CAS.
- Q. Ling, W. Liu, J. Liu, L. Zhao, Z. Ren and H. Gu, ACS Appl. Mater. Interfaces, 2022, 14, 24741–24754 CrossRef CAS.
- M.-H. Lee, M.-K. Chun and H.-K. Choi, Arch. Pharmacal Res., 2008, 31, 932–937 CrossRef CAS PubMed.
- J. Yang, G. Mao, T. Yao, L. Shen and Y. Yu, ACS Appl. Mater. Interfaces, 2025, 64, e202420413 CAS.
- G. Shin, J. Y. Baek, J. H. Kim, J. H. Lee, H. J. Kim, B. J. So, Y. Choi, S. Yun, T. Kim, J. G. Jeon and T. J. Kang, Angew. Chem., Int. Ed., 2025, 35, 2412524 CAS.
- K. Driver, S. Baco and V. V. Khutoryanskiy, Eur. Polym. J., 2013, 49, 4249–4256 CrossRef CAS.
- V. Rubentheren, T. A. Ward, C. Y. Chee and P. Nair, Cellulose, 2015, 22, 2529–2541 CrossRef CAS.
- S. Rivero, M. A. García and A. Pinotti, Carbohydr. Polym., 2010, 82, 270–276 CrossRef CAS.
- M. Qi, D. Zhang, Y. Guo, H. Zhang, J. Shao, Y. Ma, C. Yang and R. Mao, J. Mater. Chem. A, 2024, 12, 16839–16853 RSC.
- L. Sun, B. Luo, Z. Sun, X. Ren and J. Xu, Mater. Today Commun., 2025, 46, 112605 CrossRef CAS.
- Y. Yu, H. Yuk, G. A. Parada, Y. Wu, X. Liu, C. S. Nabzdyk, K. Youcef-Toumi, J. Zang and X. Zhao, Adv. Funct. Mater., 2019, 31, 1807101 Search PubMed.
- L. Wang, G. Gao, Y. Zhou, T. Xu, J. Chen, R. Wang, R. Zhang and J. Fu, ACS Appl. Mater. Interfaces, 2018, 11, 3506–3515 CrossRef PubMed.
- X. Pei, H. Zhang, Y. Zhou, L. Zhou and J. Fu, Mater. Horiz., 2020, 7, 1872–1882 RSC.
- J. Shi, X. Hao, H. Yang, Z. He, J. Lu, Y. Li, L. Luan and Q. Zhang, Carbohydr. Polym., 2024, 342, 122397 CrossRef CAS.
- M. Liang, D. Wei, Z. Yao, P. Ren, J. Dai, L. Xu, T. Zhang and Q. Zhang, Biomater. Sci., 2022, 10, 1486–1497 RSC.
- B. Balakrishnan, D. Soman, U. Payanam, A. Laurent, D. Labarre and A. Jayakrishnan, Acta Biomater., 2017, 53, 343–354 CrossRef CAS.
- H. Qiao, P. Qi, X. Zhang, L. Wang, Y. Tan, Z. Luan, Y. Xia, Y. Li and K. Sui, ACS Appl. Mater. Interfaces, 2019, 11, 7755–7763 CrossRef CAS.
- L. Han, L. Yan, K. Wang, L. Fang, H. Zhang, Y. Tang, Y. Ding, L.-T. Weng, J. Xu, J. Weng, Y. Liu, F. Ren and X. Lu, NPG Asia Mater., 2017, 9, e372–e372 CrossRef CAS.
- X. Su, Y. Luo, Z. Tian, Z. Yuan, Y. Han, R. Dong, L. Xu, Y. Feng, X. Liu and J. Huang, Mater. Horiz., 2020, 7, 2651–2661 RSC.
- L. Wu, S. Wang, J. Mao, Z. Guo and Y. Hu, Eur. Polym. J., 2023, 197, 112373 CrossRef CAS.
- S. He, B. Guo, X. Sun, M. Shi, H. Zhang, F. Yao, H. Sun and J. Li, ACS Appl. Mater. Interfaces, 2022, 14, 45869–45879 CrossRef CAS.
- J. Ren, G. Chen, H. Yang, J. Zheng, S. Li, C. Zhu, H. Yang and J. Fu, ACS Appl. Mater. Interfaces, 2024, 36, 2412162 CAS.
- H. Yuan, T. Zhu, Y. Huang, Z. Wang, P. Han, L. Tan, J. Wu, X. Chen, P. Yao, C. Zhu and J. Xu, Adv. Funct. Mater., 2024, 34, 2409703 CrossRef CAS.
- Y. Liu, G. Tian, Y. Du, P. Shi, N. Li, Y. Li, Z. Qin, T. Jiao and X. He, Adv. Funct. Mater., 2024, 34, 2315813 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |