Synergistic optimization of thermoelectric performance in polycrystalline and crystalline SnS via Na doping and Se alloying
Shan
Liu
a,
Shulin
Bai
a,
Yi
Wen
a,
Bingchao
Qin
 a,
Yixuan
Hu
a,
Tian
Gao
a,
Yichen
Li
a,
Yixuan
Hu
a,
Tian
Gao
a,
Yichen
Li
 a,
Lei
Wang
a,
Cheng
Chang
a,
Lei
Wang
a,
Cheng
Chang
 *ab and
Li-Dong
Zhao
*ab and
Li-Dong
Zhao
 *ac
*ac
aSchool of Materials Science and Engineering, Beihang University, Beijing, 100191, China. E-mail: changchengcc@buaa.edu.cn; zhaolidong@buaa.edu.cn
bState Key Laboratory of Tropic Ocean Engineering Materials and Materials Evaluation, Beihang University, Beijing 100191, PR China
cTianmushan Laboratory, Beihang University, Hangzhou 311115, China
First published on 30th December 2025
Abstract
The development of high-performance and eco-friendly thermoelectric materials is crucial for sustainable energy conversion. Tin sulfide (SnS) has emerged as a promising candidate owing to its low intrinsic thermal conductivity, abundance, and non-toxicity. However, its thermoelectric performance is severely limited by poor electrical conductivity, originating from a wide bandgap and low carrier mobility. Herein, we present a notable improvement in the thermoelectric performance of SnS via a synergistic strategy of Na doping and Se alloying. Na doping optimizes the electrical transport behaviors effectively through boosting the carrier concentration to ∼1019 cm−3. Furthermore, Se alloying is crucial for modifying the electronic band structure by narrowing the bandgap and inducing valence band convergence, which enhances the Seebeck coefficient and weighted mobility. Concurrently, mass and strain field fluctuations between S and Se atoms lead to an ultralow lattice thermal conductivity of ∼0.6 W m−1 K−1 at 873 K. Consequently, a peak ZT of ∼1.0 is achieved at 873 K in the polycrystalline Sn0.98Na0.02S0.55Se0.45 sample, representing a fourfold improvement over pristine polycrystalline SnS. Moreover, the corresponding crystal sample exhibits a record-high ZT of ∼1.6 at 623 K, attributed to a remarkable PF of ∼25 µW cm−1 K−2. This study demonstrates that the synergistic regulation of phonon and charge transport through dual-element doping/alloying and crystal growth is a highly effective strategy for developing thermoelectric materials.
Introduction
Thermoelectric materials realize the direct conversion of thermal energy to electrical energy and vice versa, based on the combined action of the Seebeck effect and the Peltier effect, finding potential uses in waste heat recovery and electronic cooling.1–6 Owing to their distinctive benefits like lightweight design and precise controllability, these materials have garnered considerable attention from the global research community.7,8 However, the energy conversion efficiency of these materials is primarily quantified by the dimensionless figure of merit, ZT = (S2σT)/κtot, where S, σ, T and κtot represent the Seebeck coefficient, electrical conductivity, absolute temperature, and total thermal conductivity, respectively.9,10 Therefore, achieving high energy conversion efficiency requires a material to retain a large S, high σ, and low κtot across a broad temperature range, thereby demanding the decoupling of the intricate interplay between charge and phonon transport.11,12 Essentially, these physical parameters are intrinsically linked to the carrier concentration. Consequently, a synergistic optimization strategy proves more effective than individually optimizing a single parameter in enhancing thermoelectric properties. For instance, strategies such as electronic band engineering (involving band convergence and resonant states) can optimize electronic transport,13,14 while multiscale microstructural features (such as point defect scattering and nano-precipitates) can suppress phonon transport.15,16 Furthermore, texture processing and grain boundary engineering can regulate grain orientation, thereby controlling the anisotropy of transport properties.17,18 Together, these approaches facilitate the realization of an ideal “two-dimensional (2D) phonon and three-dimensional (3D) charge” model, ultimately leading to a remarkable improvement in the power factor and a marked reduction in thermal conductivity.19–22These optimization strategies have achieved notable progress in traditional commercial Bi2Te3, which currently attains a room-temperature ZT value of ∼1.1.23,24 However, its narrow bandgap substantially limits high-temperature thermoelectric performance, thereby restricting its application in power generation. Moreover, the high cost and scarcity of tellurium have increasingly constrained its commercial applications in recent years. Therefore, it is imperative to seek alternative materials that can replace Bi2Te3 while offering wider bandgaps and lower costs, such as PbSe,25–29 PbS,8,30 Mg3Sb2,31–34 CoSb3,35–37 and SnSe.38–40 Among these, SnSe has attracted extensive attention due to its unique two-dimensional layered crystal structure, ultralow intrinsic thermal conductivity, and strong anharmonic lattice vibrations, emerging as one of the most promising candidates.39 Specifically, in n-type SnSe, the decoupling of charge and phonon transport has been successfully achieved through its 2D/3D transport and the control of crystal symmetry, yielding an average ZT (ZTave) of ∼1.7 at 300–773 K.19,20 Similarly, the combination of multiband transport and lattice plainification strategies led to a remarkable enhancement in carrier mobility in p-type SnSe, achieving a ZTave of ∼2.2 at 300–773 K.7,41
Based on the achievements of layered crystal SnSe in thermoelectrics, another tin chalcogenide, SnS, has emerged as a promising eco-friendly thermoelectric candidate due to its analogous crystal structure, higher elemental abundance, lighter weight, and nontoxicity.17,42–44 Despite its ultralow thermal conductivity, which originates from an orthorhombic layered structure (Pnma) and strong lattice anharmonicity induced by Sn2+ lone-pair electrons, the electrical transport performance of SnS remains suboptimal.45 This limitation stems primarily from a wide bandgap (∼46kBT), which restricts carrier concentration,22,46 together with interlayer energy barriers and lone-pair electron scattering that suppress carrier mobility.44
Our previous work has established that the synergistic coupling strategy of silver (Ag) doping and selenium (Se) alloying significantly enhances hole carrier concentration,46 narrows the bandgap, and promotes carrier transport. We extend this strategy by substituting Ag with Na to further optimize carrier concentration.47 On one hand, the shallow acceptor nature of Na+ effectively reduces the energy barrier for hole excitation, leading to higher doping efficiency.42,45 On the other hand, the mass fluctuation resulting from the difference in ionic radii between Na+ and Sn2+ intensifies point defect scattering, thereby decreasing the lattice thermal conductivity.48
In this study, we aim to address the critical challenge of low electrical properties in p-type SnS by proposing a synergistic strategy of Na doping and Se alloying. We hypothesize that aliovalent Na doping can effectively enhance the hole carrier concentration, while isovalent Se alloying is anticipated to modulate the electronic band structure. The intricate interplay between these two approaches is systematically investigated to decouple the phonon and charge transport. Furthermore, to fundamentally understand the role of Se alloying, we employ density functional theory (DFT) calculations to examine the evolution of the band structure. Ultimately, we extend our optimization to crystal growth, seeking to elucidate the thermoelectric potential of SnS by minimizing the detrimental effects of grain boundaries. This study provides a comprehensive understanding of the synergistic effects of dual-element regulation in optimizing the thermoelectric performance of SnS.
Results and discussion
To further enhance the carrier concentration in this study, Na was employed as a dopant to replace Ag. Given the metallic nature of sodium, a high-temperature solid-state reaction was adopted to ensure efficient and homogeneous incorporation of Na into the SnS lattice through a molten-state process. Phase and structural characterization results of the Sn1−xNaxS (x = 0, 0.01, 0.02, 0.03, and 0.04) samples are summarized in Fig. S1. The X-ray diffraction (XRD) patterns show that all diffraction peaks align well with the standard reference (PDF no. 73-1859), confirming the orthorhombic SnS structure (space group Pnma) in all samples (Fig. S1a). Notably, even at high Na doping levels, no secondary phases are detected, indicating high phase purity. Rietveld refinement of the lattice parameters reveals nearly constant unit cell dimensions across different doping concentrations (Fig. S1b), structurally corroborating the successful substitution of Na+ for Sn2+ without significant lattice distortion. Combined with the high relative density observed in microstructural analysis (Fig. 3), these results affirm the reliability of the present synthesis route in producing high-purity Na-doped polycrystalline SnS materials.As shown in Fig. 1a, the σ values of Na-doped Sn1−xNaxS (x = 0, 0.01, 0.02, 0.03, and 0.04) samples exhibit a pronounced non-monotonic trend within 300–873 K. This behavior originates from the competing effects of carrier excitation and scattering mechanisms that evolve with temperature, a phenomenon which thus cannot be explained by a single conduction mechanism and is consistent with reports on other layered thermoelectric systems such as polycrystalline SnSe. Specifically, in the low-temperature range (300–423 K), electrical conductivity increases with temperature. Although carrier transport is limited by grain boundary scattering, the ionization of Na acceptors significantly enhances the carrier concentration, emerging as the dominant factor governing the rise in σ. In the mid-temperature range (423–673 K), conductivity decreases with increasing temperature, exhibiting metal-like transport characteristics. This trend is attributed to the saturation of donor ionization, with the transition from grain boundary scattering to acoustic phonon scattering as the primary mechanism limiting carrier mobility (µ). In the high-temperature region (673–873 K), σ increases again, driven by the onset of intrinsic excitation, which leads to an increase in carrier concentration. Additionally, a structural transition from the Pnma to Cmcm phase occurs in SnS. According to effective medium theory, the coexistence of these two phases may provide additional conduction pathways, which synergizes with intrinsic excitation and leading to a peak conductivity of ∼45 S cm−1 at 873 K in Sn0.98Na0.02S. Although Na doping markedly enhances electrical conductivity, Sn0.98Na0.02S retains competitive S values of ∼200 µV K−1 at 300 K and about 350 µV K−1 at 873 K, as shown in Fig. 1b. Ultimately, the synergistic optimization of S and σ yields a substantially enhanced power factor (PF) of ∼6 µW cm−1 K−2 at 873 K for Sn0.98Na0.02S (Fig. 1c). To gain a better understanding of the underlying cause of the PF enhancement, Hall measurements were performed (Table S1). At 300 K, the carrier concentration increases systematically with Na doping content, increasing from ∼3.2 × 1017 cm−3 in pristine SnS to ∼2.3 × 1019 cm−3 in Sn0.96Na0.04S. In contrast, the carrier mobility decreases with higher doping levels, primarily due to enhanced ionized impurity scattering introduced by Na dopants (Fig. 1d). Furthermore, we systematically analyzed the relationship between the S and n, and constructed a Pisarenko plot (Fig. S2). A comparison of the experimental data with theoretical curves calculated using a single parabolic band (SPB) model reveals minimal change in the effective mass (m*). In summary, Na doping successfully achieves significant optimization of the electrical transport properties in polycrystalline SnS.
Owing to its unique crystal structure and strong lattice anharmonicity, SnS exhibits intrinsically low thermal conductivity. The total thermal conductivity (κtot) consists of electronic (κele) and lattice (κlat) components (κtot = κele + κlat). Fig. 1e displays the temperature-dependent (κtot) and (κlat) for the Sn1−xNaxS samples, both showing a general declining tendency with increasing temperature. At 873 K, Sn0.98Na0.02S achieves a κtot of ∼0.65 W m−1 K−1 and a κlat of ∼0.59 W m−1 K−1. As a result, leveraging the nearly threefold enhancement in PF combined with low κtot the figure of merit ZT increases markedly from 0.3 for undoped SnS to ∼0.75 for Sn0.98Na0.02S at 873 K (Fig. 1f), demonstrating substantial improvement of the thermoelectric properties of p-type polycrystalline SnS.
Building upon the preliminary optimization of thermoelectric performance in Na-doped SnS, we recognize that this single strategy is inherently constrained by the wide bandgap of pristine SnS, which limits doping efficiency. To achieve further breakthroughs, we draw on the successful experience from our previous work by implementing a synergistic approach combining aliovalent Na doping with isovalent Se alloying. The incorporation of Se at S sites is expected to narrow the bandgap, thereby enhancing carrier concentration and mobility, and better leveraging the multiband electronic structure of SnS to further promote electrical transport.
To investigate the effect of Se alloying on the crystal structure of Sn0.98Na0.02S, XRD analysis was performed on the Sn0.98Na0.02S1−ySey (y = 0, 0.1, 0.2, 0.3, 0.4, and 0.45) samples. As shown in Fig. 2a, all diffraction peaks match well with the standard reference (PDF no. 73-1859), and no secondary phases are detected, indicating that Se alloying does not alter the Pnma structure. Furthermore, the lattice parameters a, b, and c obtained from Rietveld refinement exhibit a gradual expansion with increasing Se content, suggesting a slight lattice expansion in SnS (Fig. 2b). This result provides a quantitative explanation for the observed shift of diffraction peaks toward lower angles in Fig. 2a, confirming that Se has been successfully incorporated into the Sn0.98Na0.02S matrix and the formation of a homogeneous single-phase solid solution.
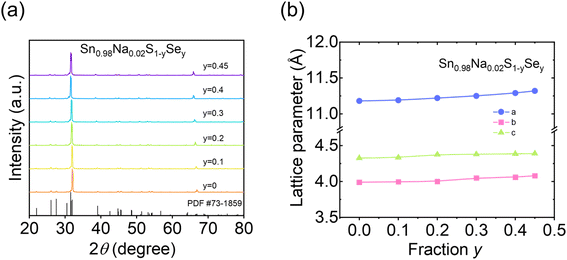 | ||
| Fig. 2 (a) XRD powder patterns and (b) calculated lattice parameters for Sn0.98Na0.02S1−ySey (y = 0, 0.1, 0.2, 0.3, 0.4, and 0.45). | ||
To further verify the microstructural homogeneity of the Sn0.98Na0.02S1−ySey solid solutions and directly observe their grain morphology, scanning electron microscopy (SEM) and energy dispersive spectroscopy (EDS) were employed. Fig. 3a and b present the SEM images of Sn0.98Na0.02S and Sn0.98Na0.02S0.55Se0.45, respectively. The Se-alloyed sample exhibits a dense microstructure with uniform grain morphology and only limited porosity, indicating the successful fabrication of highly compact bulk materials. To investigate the elemental distribution at the microscale, EDS elemental mapping was performed in the same region, as shown in Fig. 3c–f. The corresponding distribution maps of Sn, S, Se, and Na reveal a spatially uniform presence of all elements within the scanned area. In summary, both SEM and EDS analyses are fully consistent with the XRD results, thereby establishing a solid structural foundation for the evaluation of the thermoelectric properties in these compositions.
 | ||
| Fig. 3 Secondary electron imaging from SEM for (a) Sn0.98Na0.02S and (b) Sn0.98Na0.02S0.55Se0.45. (c–f) Elemental energy dispersive spectroscopy (EDS) mapping for Sn0.98Na0.02S0.55Se0.45. | ||
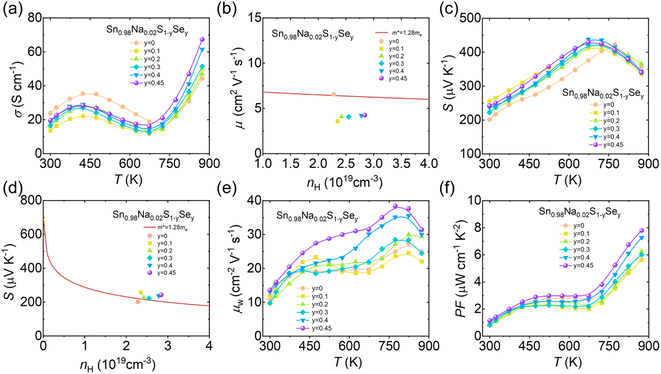
To explore the effect of Se alloying on the electrical transport of polycrystalline SnS, we measured the temperature-dependent σ of Sn0.98Na0.02S1−ySey (y = 0, 0.1, 0.2, 0.3, 0.4, 0.45) samples (Fig. 4a). The results reveal that σ increases with higher Se content over the full temperature range, which is due to a synergistic doping effect between Se and Na. The introduction of Se effectively reduces the ionization energy of Na acceptors, thereby enhancing doping efficiency and further increasing the carrier concentration. Hall measurements confirm that the n at 300 K increases continuously with increasing y, while the mobility µ decreases due to enhanced alloy scattering (Fig. 4b). Notably, despite the elevated carrier concentration, the S of the Se-alloyed samples is enhanced overall. For instance, Sn0.98Na0.02S0.55Se0.45 exhibits S values of ∼242 µV K−1 at 300 K and ∼430 µV K−1 at 673 K, representing an improvement of ∼50 µV K−1 compared to the Sn0.98Na0.02S sample (Fig. 4c). This enhancement stems from the band convergence induced by Se alloying. In the multiband structured SnS, the cooperative effect of Na and Se leads to band convergence, resulting in an increased effective mass. The associated gain in the Seebeck coefficient outweighs the suppression caused by the higher carrier concentration. Furthermore, the Pisarenko plot based on the 300 K data shows that the Seebeck coefficients of Se-alloyed samples deviate from the conventional single parabolic band model, indicating a moderate increase in m* (Fig. 4d). To comprehensively assess the coupled influence of m* and µ on charge transport, we calculated the weighted mobility (µw), as presented in Fig. 4e. The results demonstrate that µw gradually increases with Se content, indicating that the benefit of enhanced m* sufficiently compensates for the loss in µ, thereby achieving simultaneous optimization of the weighted mobility and Seebeck coefficient. Ultimately, the synergistic improvement in σ and S leads to a high PF of ∼8 µW cm−1 K−2 at 873 K for Sn0.98Na0.02S0.55Se0.45 (Fig. 4f), highlighting the remarkable effectiveness of Se alloying in optimizing the electrical transport properties of p-type SnS.
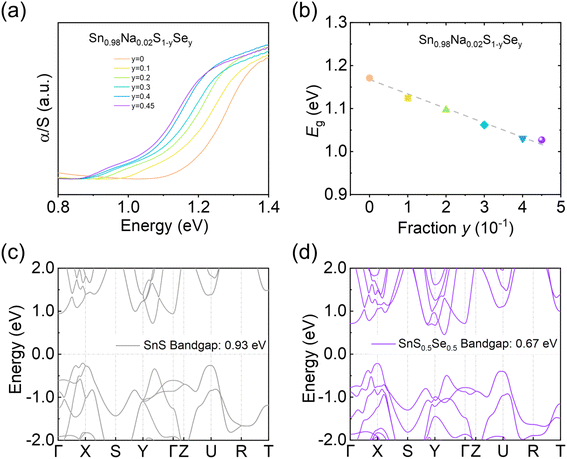
To elucidate the underlying mechanism of Se alloying at the electronic structure level, we systematically investigated the bandgap evolution of Sn0.98Na0.02S1−ySey samples using UV-vis absorption spectra (Fig. 5a). The results indicate that the bandgap of p-type SnS decreases linearly from ∼1.17 eV to ∼1.03 eV with increasing Se content (Fig. 5b). This bandgap narrowing modifies the electronic band structure and effectively mobilizes additional hole carriers into electrical transport processes. Furthermore, to clarify the origin of the improved electrical transport properties, DFT calculations were performed to analyze the electronic band structures of SnS and SnS0.5Se0.5 (Fig. 5c and d); when Se atoms occupy 50% of the S sites, the theoretically calculated bandgap decreases from ∼0.93 eV to ∼0.67 eV, consistent with the optical measurements. The band structure calculations further reveal a band convergence phenomenon in SnS0.5Se0.5, providing a coherent explanation for the previously observed increase in the Seebeck coefficient with increasing n in the measurements of electrical transport.
To elucidate the influence of Se alloying on the thermal transport behaviors of polycrystalline SnS, we systematically characterized the temperature-dependent κlat and κtot of Sn0.98Na0.02S1−ySey (y = 0, 0.1, 0.2, 0.3, 0.4, 0.45) samples (Fig. 6a and c). As revealed in Fig. 6a, κlat exhibits a continuous reduction with increasing Se content across the entire temperature range. Remarkably, Sn0.98Na0.02S0.55Se0.45 achieves an ultralow κlat of ∼0.9 W m−1 K−1 at 300 K, representing a 50% decrease relative to the unalloyed Sn0.98Na0.02S sample. This significant suppression in κlat is attributed to the synergistic enhancement of mass and strain field fluctuations induced by the substantial differences in mass and atomic radius between S and Se atoms, which effectively shorten the phonon mean free path. Further quantitative analysis via the Callaway model at 300 K shows close agreement between the calculated and experimental κlat values (Fig. 6b), strongly confirming point defect scattering as the dominant phonon scattering mechanism at 300 K. Although bandgap narrowing due to Se alloying activates bipolar diffusion at elevated temperatures (773–873 K), leading to a slight increase in κtot, the Sn0.98Na0.02S0.55Se0.45 sample still retains an exceptionally low κlat of ∼0.6 W m−1 K−1 at 873 K, benefiting from its intrinsically low lattice thermal conductivity (Fig. 6c). Eventually, through the cooperative optimization of Na doping and Se alloying, a ZT value of ∼1.0 is achieved at 873 K for Sn0.98Na0.02S0.55Se0.45 (Fig. 6d), representing a fourfold enhancement over pristine p-type SnS and underscoring a notable breakthrough in thermoelectric performance.
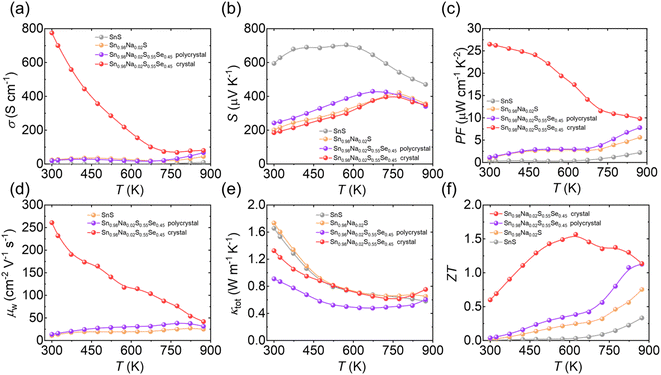
Finally, the optimal polycrystalline composition Sn0.98Na0.02S0.55Se0.45 was grown into a crystal via the temperature gradient method, enabling a systematic comparison of thermoelectric transport properties between crystalline and polycrystalline forms. As illustrated in Fig. 7, the Sn0.98Na0.02S0.55Se0.45 crystal exhibits significantly enhanced electrical transport performance, achieving an electrical conductivity as high as 800 S cm−1 at 300 K, showing a decline with increasing temperature, demonstrating typical semiconducting behavior (Fig. 7a). Although the S of the crystal is lower than that of its polycrystalline counterpart (Fig. 7b), the exceptionally high electrical conductivity still yields a remarkable PF of ∼26 µW cm−1 K−2 at 300 K (Fig. 7c). The weighted mobility follows a trend similar to that of the power factor (Fig. 7d), further confirming that the reduction in grain boundary scattering in the crystal effectively improves µ, thereby enhancing electrical performance. In terms of thermal transport, due to the lack of strong phonon scattering by grain boundaries, the thermal conductivity of the crystal is ∼0.2–0.4 W m−1 K−1 higher compared with the polycrystalline sample across the entire temperature range (Fig. 7e). Nevertheless, owing to its outstanding power factor, the crystal achieves ZT values of ∼0.5 at 300 K and a peak ZT of ∼1.6 at 623 K (Fig. 7f), significantly surpassing the previously reported performance limits for p-type SnS-based materials. The theoretical calculation of conversion efficiency for this crystal is estimated to be ∼19% at 873 K, which is superior to the reported SnS-based systems (Fig. S3). This work illustrates that the synergistic combination of band engineering and crystal growth strategies effectively decouples charge and phonon transport, highlighting the considerable potential and application prospects of SnS-based crystalline materials for achieving high thermoelectric performance.
Conclusion
We have demonstrated an effective strategy for synergistically optimizing the thermoelectric performance of p-type SnS through a combination of Na doping and Se alloying. Initial doping with Na alone was shown to significantly enhance the electrical conductivity by increasing the n. This approach yielded a respectable ZT of ∼0.75 at 873 K for the Sn0.98Na0.02S polycrystalline sample. Building on this, the incorporation of Se was proven to be a pivotal step for achieving a breakthrough performance. Se alloying effectively narrows the bandgap of SnS, further boosting the carrier concentration and inducing band convergence that remarkably enhances the S and µw. Concurrently, the mass and strain field fluctuations between S and Se atoms intensified phonon scattering, leading to an ultralow κlat. The synergistic effect in the Sn0.98Na0.02S0.55Se0.45 composition resulted in a dramatically improved ZT of ∼1.0 at 873 K. Furthermore, the growth of a crystal based on this optimal composition unveiled the full potential of the electronic transport properties in SnS. The elimination of grain boundary scattering in the crystal led to a high PF of ∼25 µW cm−1 K−2 and a record-high ZT of ∼1.6 at 623 K for SnS-based systems. The synergistic combination of band engineering by Na doping and Se alloying and microstructure control by crystal growth provides a powerful pathway to decouple the interdependent charge and phonon transport. This study not only establishes SnS-based materials as a compelling and eco-friendly alternative to traditional thermoelectrics but also offers a generalizable strategy for developing high-performance energy conversion materials.Author contributions
The manuscript was written through the contributions of all authors. All authors have approved the results and conclusions of this work. S. L. performed the sample synthesis, property measurements, data curation, and original draft writing. S. B. performed theoretical calculations. Y. W. performed microstructure characterization. Y. H. and T. G. performed data analysis and validation. Y. L. and L. W. performed data analysis. L.-D. Z., C. C. and S. L. conceived the idea, designed the experiments, and conducted revision of the draft.Conflicts of interest
The authors declare no conflict of interest.Data availability
The authors declare that the data supporting the findings of this study are available upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ta09386d.
Acknowledgements
This work was supported by the National Key Research and Development Program of China (2024YFA1210400), the National Science Fund for Distinguished Young Scholars (52525101), the National Natural Science Foundation of China (52450001, 22409014, and 12374023), the International Cooperation and Exchange of the National Natural Science Foundation of China (52411540237), and the Tencent Xplorer Prize. Y. W. is thankful for the support from the China Postdoctoral Science Foundation (2024M754059).References
- Z. Zhang, Z. Si, Y. Wei, Y. Wen, J. Kang, P. Chen, Y. Li, Y. Hu, J. Peng, Y. Jin, S. Liu, H. Shi, X. Gao, D. Gao, H. Xie and L.-D. Zhao, Adv. Energy Mater., 2025, 15, 2501184 CrossRef CAS.
- Y. Li, J. Su, Y. Wen, D. Gao, P. Chen, Z. Zhao, T. Gao, S. Wang, S. Liu and Y. Hu, Small, 2025, 2506188 CrossRef CAS.
- S. Bai, H. Shi, Y. Wen, Y. Hu, J. Zheng, Y. Qin, L. Su, S. Liu, D. Liu, T. Gao, T. Hong, X. Gao, F. Zhu, B. Qin and L.-D. Zhao, Phys. Rev. X, 2025, 15, 031033 CAS.
- S. Bai, X. Zhang and L.-D. Zhao, Acc. Chem. Res., 2023, 56, 3065–3075 CrossRef CAS PubMed.
- W. He, D. Wang, J.-F. Dong, Y. Qiu, L. Fu, Y. Feng, Y. Hao, G. Wang, J. Wang and C. Liu, J. Mater. Chem. A, 2018, 6, 10048–10056 RSC.
- B. Yuan, Y. Wen, L. Wang, Z. Li, H.-C. Wang, C. Chang and L.-D. Zhao, Acta Metall. Sin., 2025, 38, 803–810 CrossRef CAS.
- D. Liu, D. Wang, T. Hong, Z. Wang, Y. Wang, Y. Qin, L. Su, T. Yang, X. Gao and Z. Ge, Science, 2023, 380, 841–846 CrossRef CAS.
- L. Wang, Y. Wen, S. Bai, C. Chang, Y. Li, S. Liu, D. Liu, S. Wang, Z. Zhao and S. Zhan, Nat. Commun., 2024, 15, 3782 Search PubMed.
- H. Shi, L. Su, S. Bai, B. Qin, Y. Wang, S. Liu, C. Chang and L.-D. Zhao, Energy Environ. Sci., 2023, 16, 3128–3136 RSC.
- Q. Yan and M. G. Kanatzidis, Nat. Mater., 2022, 21, 503–513 Search PubMed.
- B. Qin and L.-D. Zhao, Mater. Lab, 2022, 1, 220004 Search PubMed.
- H. Pang, Y. Qiu, D. Wang, Y. Qin, R. Huang, Z. Yang, X. Zhang and L.-D. Zhao, J. Am. Chem. Soc., 2021, 143, 8538–8542 CrossRef CAS PubMed.
- S. Liu, S. Bai, Y. Wen, J. Lou, Y. Jiang, Y. Zhu, D. Liu, Y. Li, H. Shi and S. Liu, Science, 2025, 387, 202–208 CrossRef CAS PubMed.
- B. Qin, D. Wang, X. Liu, Y. Qin, J.-F. Dong, J. Luo, J.-W. Li, W. Liu, G. Tan and X. Tang, Science, 2021, 373, 556–561 CrossRef CAS PubMed.
- B. Jiang, Y. Yu, J. Cui, X. Liu, L. Xie, J. Liao, Q. Zhang, Y. Huang, S. Ning and B. Jia, Science, 2021, 371, 830–834 CrossRef CAS PubMed.
- Y. Jiang, J. Dong, H.-L. Zhuang, J. Yu, B. Su, H. Li, J. Pei, F.-H. Sun, M. Zhou and H. Hu, Nat. Commun., 2022, 13, 6087 CrossRef CAS PubMed.
- Y. Li, T. Hong, X. Li, H. An, Z. Yin, M. Wu, H. Liang, X. Wang, L. Su, X. Qian, C. Zhou, Y. Xiao, W. He and L.-D. Zhao, J. Mater. Chem. A, 2025, 13, 28063–28069 RSC.
- W. He, D. Wang, H. Wu, Y. Xiao, Y. Zhang, D. He, Y. Feng, Y.-J. Hao, J.-F. Dong and R. Chetty, Science, 2019, 365, 1418–1424 CrossRef CAS.
- L. Su, D. Wang, S. Wang, B. Qin, Y. Wang, Y. Qin, Y. Jin, C. Chang and L.-D. Zhao, Science, 2022, 375, 1385–1389 CrossRef CAS PubMed.
- C. Chang, M. Wu, D. He, Y. Pei, C.-F. Wu, X. Wu, H. Yu, F. Zhu, K. Wang and Y. Chen, Science, 2018, 360, 778–783 Search PubMed.
- C. Chang, G. Tan, J. He, M. G. Kanatzidis and L. D. Zhao, Chem. Mater., 2018, 30, 7355–7367 CrossRef CAS.
- Y. Hu, S. Bai, D. Gao, L. Su, H. Shi, T. Gao, S. Zhan, S. Liu, S. Liu, Y. Li, P. Chen, R. Liu, B. Qin and L.-D. Zhao, J. Am. Chem. Soc., 2025, 147, 44572–44580 CrossRef CAS PubMed.
- J. Peng, D. Liu, S. Bai, Y. Wen, H. Liang, L. Su, X. Qian, D. Wang, X. Gao, Z. Ding, Q. Cao, Y. Pei, B. Qin and L.-D. Zhao, Adv. Energy Mater., 2025, 15, 2404653 CrossRef CAS.
- Y. Li, S. Bai, Y. Wen, Z. Zhao, L. Wang, S. Liu, J. Zheng, S. Wang, S. Liu, D. Gao, D. Liu, Y. Zhu, Q. Cao, X. Gao, H. Xie and L.-D. Zhao, Sci. Bull., 2024, 69, 1728–1737 CrossRef CAS.
- J. Cai, J. Yang, G. Liu, L. Xu, X. Wang, H. Hu, X. Tan and J. Jiang, Adv. Energy Mater., 2022, 12, 2103287 CrossRef CAS.
- D. Gao, Y. Wen, S. Bai, S. Wang, S. Liu, Y. Li, L. Wang, W. Zang, X. Su, X. Gao, H. Xie and L.-D. Zhao, Small, 2024, 20, 2407556 CrossRef CAS.
- M. E. Manley, O. Hellman, N. Shulumba, A. F. May, P. J. Stonaha, J. W. Lynn, V. O. Garlea, A. Alatas, R. P. Hermann, J. D. Budai, H. Wang, B. C. Sales and A. J. Minnich, Nat. Commun., 2019, 10, 1928 Search PubMed.
- J. Sun, Y. Zhang, Y. Fan, X. Tang and G. Tan, Chem. Eng. J., 2022, 431, 133699 CrossRef CAS.
- Q. Deng, F. Zhang, P. Nan, Z. Zhang, L. Gan, Z. Chen, B. Ge, H. Dong, H.-k. Mao and R. Ang, Adv. Funct. Mater., 2024, 34, 2310073 CrossRef CAS.
- Y. Li, Z. Wu, J. Lin, Y. Wang, J. Mao, H. Xie and Z. Li, Mater. Sci. Semicond. Process., 2021, 121, 105393 CrossRef CAS.
- J. Mao, H. Zhu, Z. Ding, Z. Liu, G. A. Gamage, G. Chen and Z. Ren, Science, 2019, 365, 495–498 Search PubMed.
- X. Tan, G.-Q. Liu, H. Hu, H. Shao, J. Xu and J. Jiang, J. Mater. Chem. A, 2019, 7, 8922–8928 RSC.
- X. Shi, X. Wang, W. Li and Y. Pei, Small Methods, 2018, 2, 1800022 Search PubMed.
- Z. Liang, C. Xu, S. Song, X. Shi, W. Ren and Z. Ren, Adv. Funct. Mater., 2023, 33, 2210016 CrossRef CAS.
- B. R. Ortiz, C. M. Crawford, R. W. McKinney, P. A. Parilla and E. S. Toberer, J. Mater. Chem. A, 2016, 4, 8444–8450 Search PubMed.
- G. Li, U. Aydemir, M. Wood, W. A. Goddard III, P. Zhai, Q. Zhang and G. J. Snyder, Chem. Mater., 2017, 29, 3999–4007 Search PubMed.
- Z.-H. Zheng, J.-Y. Niu, D.-W. Ao, B. Jabar, X.-L. Shi, X.-R. Li, F. Li, G.-X. Liang, Y.-X. Chen, Z.-G. Chen and P. Fan, J. Mater. Sci. Technol., 2021, 92, 178–185 Search PubMed.
- A. Banik and K. Biswas, Joule, 2019, 3, 636–638 CrossRef CAS.
- L.-D. Zhao, C. Chang, G. Tan and M. G. Kanatzidis, Energy Environ. Sci., 2016, 9, 3044–3060 RSC.
- M. Jin, Z. Chen, X. Tan, H. Shao, G. Liu, H. Hu, J. Xu, B. Yu, H. Shen, J. Xu, H. Jiang, Y. Pei and J. Jiang, ACS Energy Lett., 2018, 3, 689–694 CrossRef CAS.
- D. Liu, S. Bai, Y. Tian, J. Peng, S. Liu, H. Shi, H. Liang, Y. Qin, L. Su, X. Qian, B. Qin and L.-D. Zhao, Adv. Mater., 2025, 37, 2506999 Search PubMed.
- B. Zhou, S. Li, W. Li, J. Li, X. Zhang, S. Lin, Z. Chen and Y. Pei, ACS Appl. Mater. Interfaces, 2017, 9, 34033–34041 Search PubMed.
- Y. Hu, S. Bai, B. Qin and L. D. Zhao, Electromag. Sci., 2025, 3, 0100611–0100613 Search PubMed.
- W. He, R. Ang and L. D. Zhao, Sci. China Mater., 2022, 65, 1143–1155 CrossRef CAS.
- H. Q. Yang, X. Y. Wang, H. Wu, B. Zhang, D. D. Xie, Y. J. Chen, X. Lu, X. D. Han, L. Miao and X. Y. Zhou, J. Mater. Chem. C, 2019, 7, 3351–3359 RSC.
- S. Liu, D. Liu, Y. Qiu, B. Qin and L.-D. Zhao, Chin. Sci. Bull., 2025, 70, 685–696 Search PubMed.
- X. Su, X. Tang and Z. Li, ACS Appl. Mater. Interfaces, 2022, 14, 6916–6925 Search PubMed.
- H. Wu, K. Peng, B. Zhang, X. Gong and X. Zhou, Mater. Today Phys., 2020, 14, 100221 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |