Harnessing an mpg-C3N4 photocatalyst for the selective oxidative coupling of amines to yield azoaromatic compounds
Dinesh S.
Chaudhari
 a,
Rahul P.
Gaikwad
a,
Indrajeet R.
Warkad
a,
Rostislav
Langer
b,
Michal
Otyepka
a,
Rahul P.
Gaikwad
a,
Indrajeet R.
Warkad
a,
Rostislav
Langer
b,
Michal
Otyepka
 bc and
Manoj B.
Gawande
bc and
Manoj B.
Gawande
 *ad
*ad
aDepartment of Industrial and Engineering Chemistry, Institute of Chemical Technology, Mumbai, Marathwada Campus, Jalna 431213, Maharashtra, India. E-mail: mb.gawande@marj.ictmumbai.edu.in; mbgawande@gmail.com
bIT4Innovations, VSB – Technical University of Ostrava, 17. Listopadu 2172/15, 708 00 Ostrava-Poruba, Czech Republic
cRegional Centre of Advanced Technologies and Materials, The Czech Advanced Technology and Research Institute (CATRIN), Palacký University Olomouc, Šlechtitelů 27, 779 00 Olomouc, Czech Republic
dNanotechnology Centre, Centre for Energy and Environmental Technologies, VŠB – Technical University of Ostrava, 17. Listopadu 2172/15, 708 00 Ostrava-Poruba, Czech Republic
First published on 14th January 2026
Abstract
Developing sustainable and efficient methods for the oxidative coupling of arylamines to generate aromatic azo compounds under mild conditions remains a significant challenge in the realm of sustainable chemistry. In this study, a metal-free mesoporous graphitic carbon nitride (mpg-C3N4) photocatalyst was synthesized via a straightforward one-step thermal polymerization reaction of urea, resulting in a high surface area of 109.40 m2 g−1 and enhanced charge separation. The mpg-C3N4 photocatalyst was thoroughly characterized through XRD, XPS, HR-TEM, and BET analyses, confirming its mesoporous structure, crystallinity, and thermal stability. Under light irradiation in ambient air, mpg-C3N4 efficiently activated atmospheric O2 to generate reactive superoxide radicals, enabling selective azo formation with up to 99% conversion and selectivity across a range of aromatic amines. DFT calculations and adsorption studies revealed the role of π–π stacking and favorable adsorption Gibbs energies, underpinning effective substrate activation and surface interaction. Mechanistic investigations, aided by EPR and DFT studies, elucidated that photoexcited electrons reduced O2 to superoxide radicals, which mediated the oxidative coupling process. Reusability tests confirmed the catalyst's stability across five cycles, exhibiting no significant loss of activity or structural integrity. This sustainable, metal-free photocatalytic strategy, harnessing renewable light energy, not only demonstrates the scalable synthesis of azo compounds but also paves the way for new approaches to the green synthesis of functional azoaromatics.
1 Introduction
The increasing global population and rapid industrialization have caused substantial environmental problems, such as the exhaustion of resources, increased pollution, and global warming.1 As the demand for energy and chemical products continues to rise, traditional synthetic methods that often rely on non-renewable resources and generate hazardous wastes are exacerbating these issues.2,3 Consequently, there is an urgent need for sustainable and efficient chemical processes that minimize environmental impacts while meeting the growing demands of society. In a time marked by increasing environmental challenges and the continuous quest for sustainable alternatives, photocatalysis has emerged as a transformative approach to address these issues by utilizing solar energy to drive chemical reactions under mild conditions. This technology decreases the reliance on fossil fuels while promoting the adoption of renewable resources, aligning with the principles of green chemistry.4–9Photocatalysis, known for being both eco-friendly and energy-efficient, has emerged as a beacon of hope. This process is facilitated by suitable semiconductor photocatalysts that harness and utilize solar energy.10 Numerous semiconductor photocatalysts, including graphitic carbon nitride (g-C3N4), zinc oxide, titanium dioxide, tungsten trioxide, tin oxide, cadmium sulfide, zinc sulfide, copper oxide, and lead sulfide, are used as supports and catalysts for several important advanced chemical transformations and sustainable applications.11–15 Among these, g-C3N4 has garnered significant interest in the field of catalysis as it is a metal-free semiconductor with an ideal band gap of 2.7 eV, which allows for the efficient absorption of visible light.16–18 Additionally, the synthesis of g-C3N4 from readily available starting materials like urea, melamine, thiourea, and cyanamide makes it not only cost-effective but also easy to produce.3 As illustrated in Fig. 1a, it also allows for the straightforward tuning of its properties through synthetic modifications. Additionally, it is low in toxicity, has a suitable band gap, demonstrates exceptional chemical stability even when exposed to reactive species, and has abundant surface properties that endow it with catalytic performance similar to that of homogeneous photocatalysts.19–21 Since its first application as a metal-free photocatalyst for hydrogen generation by Wang et al.22 in 2009, it has been extensively explored for various photocatalytic processes including CO2 reduction, H2O2 production, organic transformations, and biomass valorization.19,23–25 However, despite the unique electronic and mechanical properties of g-C3N4, its non-porous nature and relatively low surface area (<10 m2 g−1) limit its effectiveness in applications that require significant surface area and pore volume.26 To overcome these limitations, the introduction of porosity into the carbon nitride matrix while preserving its inherent carbon and nitrogen chemistry offers a promising strategy for enhancing its performance in catalysis and adsorption.26
Aromatic azo compounds are important chemicals with diverse applications across pharmaceuticals, dyes, optical data storage, molecular switches, and numerous other industries.27–31 In the textile industry, these compounds are used as dyes for fabric coloration.32 Furthermore, the light-driven trans–cis isomerization of the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond in azo compounds leads to significant alterations in their physical properties. This transformation renders them particularly valuable for chemists in the development of protein probes, chemosensors, smart surface materials, and more.33–35 Traditional methods for synthesizing azoaromatics often require harsh reaction conditions and toxic reagents and generate substantial wastes, posing environmental and economic challenges (Fig. 1b).36 As the awareness of green chemistry increases, alternative sustainable synthetic methods are being investigated. These methods include the reductive coupling of nitrobenzenes, oxidative coupling of anilines, and oxidative dehydrogenation of hydrazobenzenes, all utilizing suitable catalytic systems to eliminate the use of hazardous diazonium intermediates.37–39 Of all these methods, the oxidative coupling of readily accessible anilines using atmospheric air is particularly appealing. To date, all known catalytic systems (homogeneous/heterogeneous) for the direct oxidation of anilines rely on metals and perform efficiently under thermal and photocatalytic conditions (Fig. 1c).28,40–48 Although efficient homogeneous and catalyst-free photocatalytic routes to azoaromatics have been reported, recyclable metal-free heterogeneous systems are still scarce.49,50 Notably, the photochemical conversion of arylamines to azoaromatics has recently demonstrated moderate to good yields when utilizing an Ir-based photocatalyst.27 While various catalysts, including noble metals, have been effectively investigated for the oxidative coupling of arylamines, significant challenges remain, like the need for high catalyst loading, stoichiometric reagents, strong oxidants, moderate yields, and a limited range of applicable substrates.27,28,40–42,44,51,52 Therefore, the development of metal-free, sustainable, efficient, and selective catalysts for the transformation of amines to azoaromatics is of paramount importance.
N double bond in azo compounds leads to significant alterations in their physical properties. This transformation renders them particularly valuable for chemists in the development of protein probes, chemosensors, smart surface materials, and more.33–35 Traditional methods for synthesizing azoaromatics often require harsh reaction conditions and toxic reagents and generate substantial wastes, posing environmental and economic challenges (Fig. 1b).36 As the awareness of green chemistry increases, alternative sustainable synthetic methods are being investigated. These methods include the reductive coupling of nitrobenzenes, oxidative coupling of anilines, and oxidative dehydrogenation of hydrazobenzenes, all utilizing suitable catalytic systems to eliminate the use of hazardous diazonium intermediates.37–39 Of all these methods, the oxidative coupling of readily accessible anilines using atmospheric air is particularly appealing. To date, all known catalytic systems (homogeneous/heterogeneous) for the direct oxidation of anilines rely on metals and perform efficiently under thermal and photocatalytic conditions (Fig. 1c).28,40–48 Although efficient homogeneous and catalyst-free photocatalytic routes to azoaromatics have been reported, recyclable metal-free heterogeneous systems are still scarce.49,50 Notably, the photochemical conversion of arylamines to azoaromatics has recently demonstrated moderate to good yields when utilizing an Ir-based photocatalyst.27 While various catalysts, including noble metals, have been effectively investigated for the oxidative coupling of arylamines, significant challenges remain, like the need for high catalyst loading, stoichiometric reagents, strong oxidants, moderate yields, and a limited range of applicable substrates.27,28,40–42,44,51,52 Therefore, the development of metal-free, sustainable, efficient, and selective catalysts for the transformation of amines to azoaromatics is of paramount importance.
Inspired by previous research, we developed a metal-free mesoporous carbon nitride (mpg-C3N4) photocatalyst for the oxidative coupling of arylamines to generate aromatic azo compounds under mild conditions (Fig. 1d). The mpg-C3N4 photocatalyst was produced via a straightforward one-step thermal polymerization reaction of urea, resulting in a higher surface area of 109.40 m2 g−1 and improved charge carrier transfer and separation compared to bulk g-C3N4. We explored the versatility of the mpg-C3N4 photocatalyst with various amine substrates, achieving conversion and selectivity up to 99% for the desired azoaromatics. This state-of-the-art mpg-C3N4 was compared with existing thermal catalysts and available photocatalysts for the oxidative coupling of amines. Mechanistic investigations supported by EPR and DFT studies demonstrated that the mpg-C3N4 photocatalyst can efficiently activate atmospheric O2 molecules to produce superoxide radical anions (O2˙−), which are highly reactive oxygen species. Importantly, this catalyst can be easily separated from the reaction medium and reused by simple centrifugation, highlighting its potential as an effective heterogeneous photocatalyst. We anticipate that this research expands the range of applications, especially in the sustainable production of value-added chemicals and fuels, thus contributing significantly to advancements in the field.
2 Results and discussion
2.1 Synthesis and characterization of catalysts
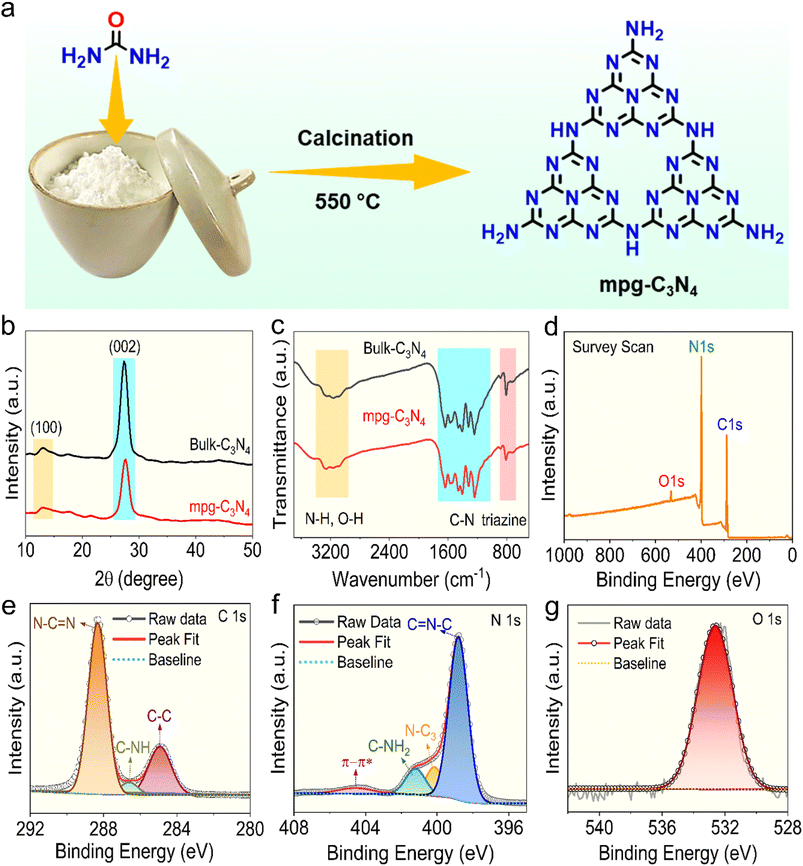
Mesoporous carbon nitride (mpg-C3N4) was synthesized using a one-step thermal polymerization method13 (Fig. 2a); the detailed procedure is outlined in the experimental section of the SI. For the synthesis of mpg-C3N4, urea was placed in a porcelain crucible and calcined in a muffle furnace at 550 °C. Bulk carbon nitride (bulk-C3N4) derived from melamine was also produced using the same approach to compare photocatalytic performance.Initially, powder X-ray diffraction (XRD) was used to examine the crystalline structure of mpg-C3N4 and bulk-C3N4 (Fig. 2b). The diffraction patterns of mpg-C3N4 and bulk-C3N4 displayed weak and strong peaks at 13.2° and 27.4°, respectively. The peak at 13.2° corresponds to the (100) plane and arises from the in-plane arrangement of tri-s-triazine motifs. Notably, the prominent peak at 27.4°, stemming from the (002) plane, is related to the stacking interactions between aromatic layers.53 Structural features and chemical bonding types in the samples were further characterized using Fourier transform infrared (FT-IR) spectroscopy, with similar spectral profiles observed for mpg-C3N4 and bulk-C3N4, indicating comparable structural components (Fig. 2c). Both spectra show broad absorption peaks around 3000 cm−1 to 3400 cm−1 ascribed to the N–H and O–H stretching vibrations.54 Additionally, a peak between 1150 cm−1 and 1750 cm−1 corresponds to the vibrational modes of the C–N heterocyclic groups, while sharp peaks at 810 and 890 cm−1 are attributed to the breathing mode of the tri-s-triazine units and N–H deformation mode.55
The XPS survey scan of mpg-C3N4 shows three peaks corresponding to C 1s, N 1s, and O 1s (Fig. 2d). The C 1s spectrum can be resolved into three distinct peaks with binding energies of 284.9, 286.6, and 288.3 eV (Fig. 2e). The peak at 284.9 eV is attributed to adventitious carbon (C–C), while the peaks at 286.6 eV and 288.3 eV correspond to C–NHx (sp2 hybridized carbon) and N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N within the tri-s-triazine structure of mpg-C3N4, respectively.55,56 The N 1s spectrum was resolved into four peaks, with a dominant peak at 398.8 eV, indicating nitrogen atoms in an sp2-hybridized state bonded to carbon. Additional peaks at 400.1 eV and 401.2 eV were ascribed to the tertiary N–C3 and –NHx functional groups associated with the tri-s-triazine backbone, respectively, while a minor peak at 404.5 eV was linked to π–π* excitation (Fig. 2f).57 The O 1s spectrum reveals a peak at 532.2 eV, corresponding to the surface-bound moisture (Fig. 2g).58,59
N within the tri-s-triazine structure of mpg-C3N4, respectively.55,56 The N 1s spectrum was resolved into four peaks, with a dominant peak at 398.8 eV, indicating nitrogen atoms in an sp2-hybridized state bonded to carbon. Additional peaks at 400.1 eV and 401.2 eV were ascribed to the tertiary N–C3 and –NHx functional groups associated with the tri-s-triazine backbone, respectively, while a minor peak at 404.5 eV was linked to π–π* excitation (Fig. 2f).57 The O 1s spectrum reveals a peak at 532.2 eV, corresponding to the surface-bound moisture (Fig. 2g).58,59
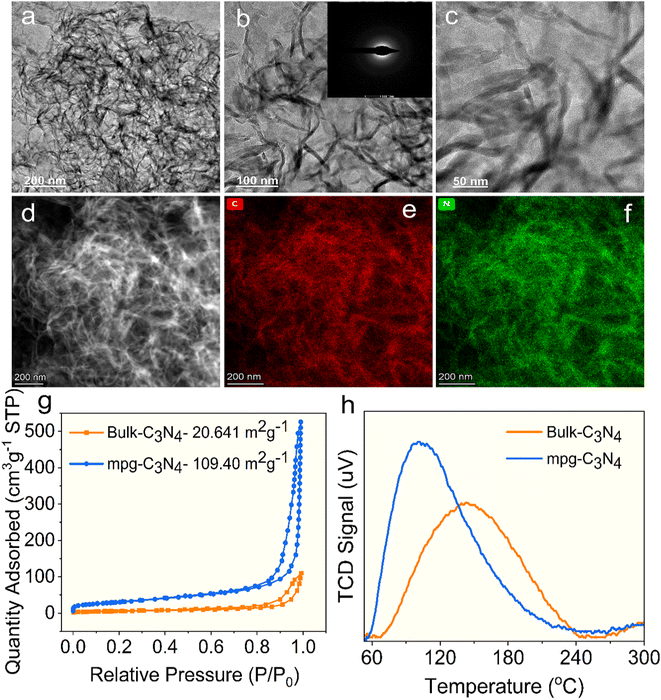
To examine the surface structure of prepared mpg-C3N4, scanning electron microscopic (SEM) and high-resolution transmission electron microscopic (HR-TEM) analyses were performed. The SEM images displayed a loosely interconnected structure with visible gaps and void regions, indicating a porous and non-compact morphology (Fig. S1). The HR-TEM images of the mpg-C3N4 photocatalyst are illustrated in Fig. 3. The images reveal the nanowire-like morphology of mpg-C3N4 (Fig. 3a–c), indicating an irregular shape. This morphology can be correlated with the decomposition behaviour of the precursor, as the nature of the starting material strongly influences structural evolution during polymerization.60,61 During the thermal breakdown of urea, gaseous species such as NH3 and CO2 are released, and these evolving gases act as in situ soft templates that promote the modification of the textural properties of the material.62,63 This process not only influences the morphology but also leads to the formation of pores within the carbon nitride framework during polymerization. Earlier studies have shown that the evolution of gases within g-C3N4 domains can lead to the formation of internal pores, which can directly result in the development of mesoporosity and an increased specific surface area.59,61 A similar phenomenon was observed in the mpg-C3N4 sample, as indicated by its significantly enhanced surface area (Fig. 3g and Table S1). This unique nanowire morphology of mpg-C3N4 is expected to enhance its photocatalytic activity.13,64,65 The amorphous nature of the mpg-C3N4 sample was further validated by the selected area electron diffraction (SAED) pattern shown in the inset of Fig. 3b, which displays diffraction rings for the (100) and (002) planes, aligning well with the XRD findings.66 Furthermore, EDX analysis indicated the presence of carbon and nitrogen in the mpg-C3N4 material, and elemental mapping corroborated these results (Fig. 3d–f).
The N2 adsorption–desorption isotherms along with the BJH pore-size distribution for both bulk and mpg-C3N4 photocatalysts are illustrated in Fig. 3g and S2. The specific surface area (SSA), pore size, and pore volume of the carbon nitride materials are detailed in Table S1. The isotherms displayed a type-IV hysteresis loop, consistent with mesoporosity and confirming mesopores in both bulk-C3N4 and mpg-C3N4 photocatalysts.67 The mpg-C3N4 sample exhibited an SSA of 109.40 m2 g−1, significantly higher than that of bulk-C3N4, which measured 20.64 m2 g−1 (Fig. 3g). Previous studies have shown that samples produced from urea as a precursor tend to have a greater SSA and pore volume than those derived from melamine.68–70 The BJH pore-size distribution of mpg-C3N4 indicates an average pore size of 28.17 nm. Additionally, mpg-C3N4 exhibits a larger pore volume of 0.8025 cm3 g−1, in contrast to bulk-C3N4, which has a pore volume of 0.1554 cm3 g−1 (Table S1). These results confirm that the synthesized mpg-C3N4 sample is indeed mesoporous in nature. The higher SSA and mesoporous structure of mpg-C3N4 may enhance the electron mobility, improve light reflection and scatter within the material, and provide more reactive sites for effective interaction with reactants.71,72 The number of basic sites in mpg-C3N4 and bulk-C3N4 was evaluated using the CO2 temperature-programmed desorption (TPD) (Fig. 3h). It is well established that surface basicity is essential for the chemical adsorption of amines and the formation of surface complexes.73 Both mpg-C3N4 and bulk-C3N4 displayed broad peaks in the range of 60 to 250 °C, which are associated with the physisorption and chemisorption of CO2.74,75 The basic site density was determined to be 0.091 mmol g−1 for mpg-C3N4 and 0.078 mmol g−1 for bulk-C3N4. The CO2-TPD results confirmed that mpg-C3N4 exhibited greater basicity, which was correlated with its higher reactivity in the photocatalytic oxidation of amines.
The optical and electronic characteristics of bulk-C3N4 and mpg-C3N4 were examined to assess their photocatalytic performance. The obtained UV-vis diffuse reflectance spectra (DRS) are shown in Fig. 4a. The absorption band of mpg-C3N4 shows a blue shift compared to that of bulk-C3N4.59 Using the Tauc plots, the bandgap energies were estimated to be approximately 2.70 eV for bulk-C3N4 and 2.94 eV for mpg-C3N4 (Fig. 4b). Furthermore, the charge separation and recombination dynamics of both materials were analyzed. The photoluminescence (PL) spectra of mpg-C3N4, excited at 385 nm, revealed that mpg-C3N4 displayed a lower PL intensity than that of bulk-C3N4 (Fig. 4c). The observed blue shift in the PL peak indicated a reduced recombination rate of photogenerated electron–hole pairs, which enhanced the photocatalytic performance of mpg-C3N4.59,76,77
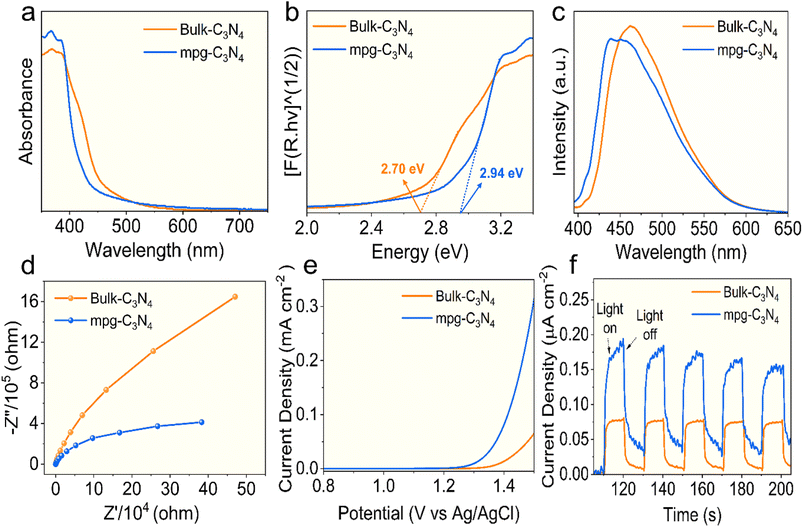 | ||
| Fig. 4 (a) UV DRS spectrum, (b) bandgap, (c) PL spectra, (d) Nyquist plot, (e) LSV curves, and (f) transient photocurrent curves of mpg-C3N4 and bulk-C3N4. | ||
To further investigate the transfer of photogenerated charge carriers, photo-electrochemical (PEC) studies were conducted. Electrochemical impedance spectroscopy (EIS) was performed to assess the electronic conductivity of the samples. The Nyquist plots showed that mpg-C3N4 exhibited a lower electronic resistance than that of bulk-C3N4, indicating reduced electron-transfer resistance (Fig. 4d). Additionally, linear sweep voltammetry (LSV) curves and transient photocurrent responses were recorded for both samples. Fig. 4e shows the current densities of bulk-C3N4 and mpg-C3N4 to be 0.063 mA cm−2 and 0.309 mA cm−2, respectively, under dark conditions at 0 V vs. Ag/AgCl, indicating greater charge separation and transfer.78,79Fig. 4f compares the transient photocurrent responses of bulk-C3N4 and mpg-C3N4. Under light, photocurrents for both samples rise to their peak values and remain relatively stable. Upon switching off the light, the photocurrents return to baseline. Notably, mpg-C3N4 exhibits a higher photocurrent than that of bulk-C3N4, indicating its superior capability for separating and transferring photogenerated electron–hole pairs.56 Overall, these findings clearly demonstrate that mpg-C3N4 exhibits enhanced light absorption, charge transfer, and charge separation, implying enhanced photocatalytic efficiency.
2.2 Photocatalytic activity
Considering the remarkable physicochemical properties and the successful characterization of mpg-C3N4, it was further explored for the oxidative coupling of aryl amines to generate azoaromatics under light irradiation. A series of reactions was carried out to optimize this photocatalytic transformation, with the various conditions summarized in Table 1. The standard conditions were p-anisidine (0.1 mmol) as the substrate, 30 mg of the mpg-C3N4 photocatalyst, K3PO4 (1 equivalent) as a base, and 3 mL of DMSO as the solvent. In air, the reactions were performed under illumination at 395 nm (36 W lamp), at 30–35 °C for 24 h (detailed procedure is given in the SI). Under these standard conditions, the oxidative coupling of p-anisidine (1a) affords the corresponding azoaromatics (2a) with outstanding conversion (99%) and selectivity (98%) (Table 1, entry 1). The mpg-C3N4 sample shows a higher activity than that of bulk-C3N4 derived from melamine and under the same reaction conditions (Table 1, entry 2). The improved performance of mpg-C3N4 is due to its larger surface area (see Fig. 3g) and enhanced charge transfer dynamics (see Fig. 4d–f) compared to bulk-C3N4. Table 1 presents a series of control experiments, in which essential components are excluded from the reaction mixture, emphasizing the crucial roles of light and the catalyst (entries 3 and 4) in facilitating photoirradiation-driven oxidative coupling of amines. The effect of thermal heating was evaluated by carrying out the reaction at 55 °C without light (Table 1, entry 5), which resulted in no conversion of 1a. Collectively, this finding underscores the significance of photocatalytic conditions. We also investigated the effect of light wavelength utilizing a 427 nm lamp (Table 1, entry 6), which afforded 47% conversion of 1a. The influence of various solvents, including ACN, H2O, and DMF as alternatives to DMSO was examined in the model reaction (Table 1, entries 7–9). ACN and H2O exhibited relatively low conversions of 1a (37% and 4%, respectively), while DMF achieved a higher conversion (78%), albeit with reduced selectivity toward 2a (34%). In contrast, using DMSO afforded 98% conversion and 99% selectivity, likely due to the improved solubility of the reactants under light irradiation. The reaction requires a base (Table 1, entry 10) since proton removal is a key step in the photocatalytic oxidative coupling of amines.49 Both inorganic base (KOAc) and organic base (NEt3) were evaluated, resulting in 81% and 13% conversions of 1a, respectively (Table 1, entries 11 and 12). Next, we investigated the effect of varying the K3PO4 concentration. Reducing the base to 0.5 equivalents decreased the conversion to 37%, indicating that an adequate concentration of base is essential for achieving optimal conversion (Table 1, entry 13). Moreover, we assessed the effect of catalyst loading and observed a significant reduction in the conversion of 1a to 79%, when the amount of mpg-C3N4 was decreased to 20 mg, highlighting the importance of maintaining an appropriate catalyst quantity (Table 1, entry 14).| Entry | Deviation from standard conditionsa | Conversion (1a)b (%) | Selectivity (2a)b (%) |
|---|---|---|---|
| a Standard conditions: p-anisidine (0.1 mmol), mpg-C3N4 (30 mg), K3PO4 (1 eq.), DMSO (3 mL), air atmosphere, 395 nm lamp (36 W), 30–35 °C, 24 h. b Conversion and selectivity were determined by GC-MS analysis using dodecane as an internal standard. c Reaction performed at 55 °C without light irradiation. d N.D. = not detected. | |||
| 1 | None | 99 | 98 |
| 2 | Bulk-gC3N4 | 73 | 96 |
| 3 | No light | N.D.d | N.D. |
| 4 | No catalyst | N.D. | N.D. |
| 5 | 55 °Cc | N.D. | N.D. |
| 6 | 427 nm | 47 | 98 |
| 7 | ACN instead of DMSO | 37 | 99 |
| 8 | H2O instead of DMSO | 4 | 99 |
| 9 | DMF instead of DMSO | 78 | 34 |
| 10 | No base | 4 | 99 |
| 11 | KOAc instead of K3PO4 | 81 | 96 |
| 12 | NEt3 instead of K3PO4 | 13 | 99 |
| 13 | 0.5 eq. of K3PO4 | 37 | 99 |
| 14 | 20 mg of mpg-C3N4 | 78 | 99 |
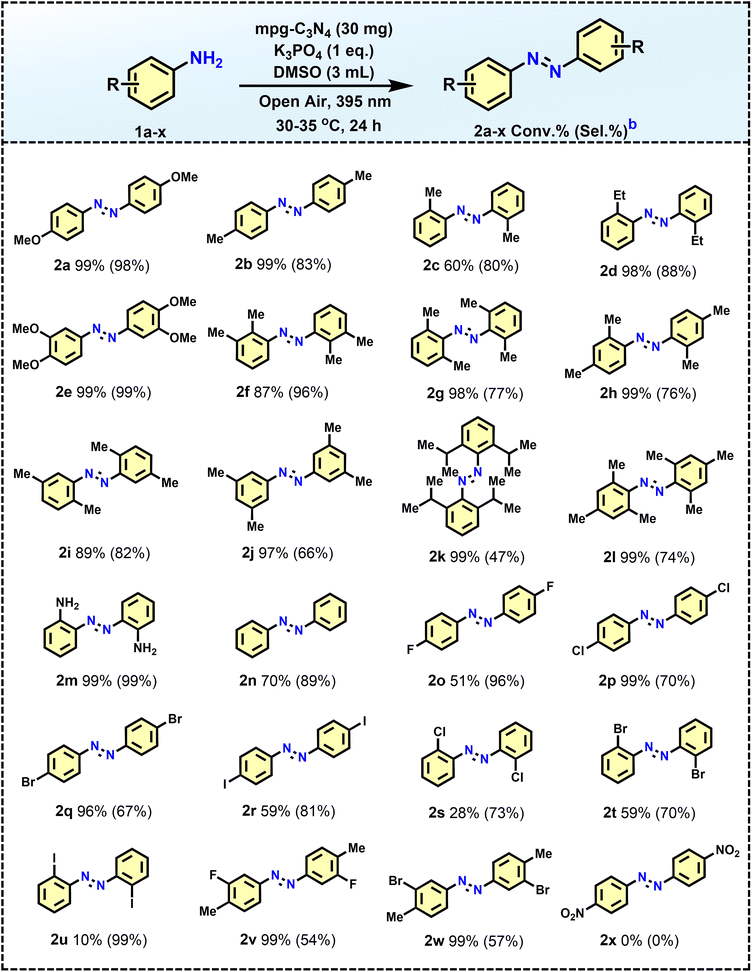
With the optimal conditions established, mpg-C3N4 photocatalysts were used to synthesize aromatic azo compounds with a broad substrate scope from readily accessible primary amines, including those bearing electron-rich and electron-deficient groups (Table 2). Generally, amines with electron-rich characteristics (Table 2, entries 2a–2n) exhibited significantly better conversion than those that were electron-deficient (Table 2, entries 2o–2x). The influence of steric factors was also noted, as para-substituted anilines yielded higher conversion than their ortho-substituted counterparts (Table 2, entries 2b and 2c). Additionally, disubstituted aryl amines were effectively transformed into the corresponding azoaromatics, achieving good conversion rates (Table 2, entries 2e–2k), although a slight decrease in product selectivity was observed for entries 2g and 2k due to the increased steric hindrance at the ortho position from the methyl (–CH3) and isopropyl (–C(CH3)2) groups, respectively. Notably, the sterically hindered and electron-rich trimethyl aniline produced the azo derivative with a conversion and selectivity of 99% and 74%, respectively (Table 2, entry 2l).
To further explore the behavior of aniline substrates during oxidative coupling in the presence of an amine group, a new substrate with an –NH2 group at the ortho position of anilines was subjected to photocatalytic testing. The findings revealed that only one amine group was involved in forming the oxidative coupled product, achieving both conversion and selectivity of 99% (Table 2, entry 2m). This selective coupling is attributed to steric hindrance from the additional –NH2 groups, which restricts the reaction of the second amine after the first has coupled. An azoaromatic product with 70% conversion was also obtained from unsubstituted anilines (Table 2, entry 2n). Importantly, halo-substituted anilines displayed good to moderate conversion, leading to the formation of halo-substituted azoaromatics (Table 2, entries 2o–2u), which could be further utilized in transformations. The effect of steric hindrance in the case of halo-substituted anilines was also studied, as the conversion of para-substituted halides (Table 2, entries 2o–2r) was significantly higher than that of the ortho-substituted halides (Table 2, entries 2s–2u). Excellent conversions with good selectivity were also achieved with arylamines containing both electron-rich and electron-deficient groups (Table 2, entries 2v–2w). In contrast, p-nitroaniline fails to produce any products, probably due to the significant electron deficiency of its amine group (Table 2, entry 2x).
These findings indicate that mpg-C3N4 photocatalysts possess a significant tolerance to a variety of functional groups. Substrates with electron-rich groups (such as para-, meta-, and ortho-substituted variants) as well as those with electron-deficient groups were effectively converted, achieving moderate to high conversion and selectivity. We also conducted a scale-up study by increasing the concentration of substrate 1a by 10-fold compared to the standard reaction conditions used during optimization (as shown in Table 1). The resulting azo product (2a) was obtained with a selectivity of 96% and a conversion rate of 52% after 24 hours. Extending the reaction time to 48 hours increased the conversion to 82% while maintaining consistent selectivity, underscoring the practical applicability of our approach (refer to Fig. S3).
A review of previously documented catalysts for the formation of azo bonds reveals that several metal-based catalysts provide efficient conversion, but require a high amount of catalyst, the use of stoichiometric reagents, and strong oxidizing agents (Table S2). Comparing these prior studies with our work allows us to evaluate the advancements in catalytic efficiency, reaction conditions, selectivity, and environmental sustainability. In a recent study, molybdenum-based catalysts were used for the thermal oxidative coupling of amines to produce azoaromatics. This process involved the use of H2O2 as the oxidant and Na2S2O3 as an additive.80 Qu and co-workers innovatively employed single-crystal MnOOH nanotubes to synthesize azo compounds from amines, demonstrating exceptional performance under thermal conditions, albeit requiring high-pressure oxygen (5 bar).81 Another research endeavour explored the utilization of Zr(OH)4 to oxidize anilines to azobenzenes, employing acetic acid as the solvent and TBHP as the oxidant.82 Numerous other studies have reported the synthesis of azoaromatics using strong oxidants like TBHP and H2O2, under both thermal and photocatalytic setups, often employing metal-based catalysts.44,46,47 Moreover, Vannucci and co-workers reported homogeneous Ir-based photocatalysts, showcasing their potential for azoaromatics synthesis from amines under aerial conditions; however, the notable drawbacks of high iridium loading (5 mol%) and limited substrate scope highlight certain constraints.27
In our investigation, we assessed the sustainability of our approach by examining factors such as metal cost, temperature, oxidant use, yield, substrate scope, and catalyst reusability, and compared these with the existing literature (Fig. 7g). Many reported catalysts often required high temperature, strong oxidants, and expensive noble or other transition metal-based catalysts, which may pose issues related to sustainability (Fig. 7g and Table S2). In contrast, mpg-C3N4 demonstrates strong photocatalytic activity in a fully metal-free environment, maintaining its performance even after five cycles. Our method emphasizes greener, low-cost, and more sustainable practices, delivering high efficiency and selectivity across various substrates, showcasing the advantages of photocatalysis over traditional metal-dependent systems.
2.3 Mechanistic studies
In order to investigate the reaction mechanism, a series of comparative experiments was conducted. Under ideal reaction conditions, air was chosen as the oxidant for the photo-induced oxidative coupling of amines to produce azoaromatics, highlighting the environmentally friendly nature of this reaction system. This suggests that the pathway involving oxygen is predominant, indicating that a reactive oxidative species (ROS) is generated from O2. To identify the specific type of ROS produced in this photocatalytic system, various radical scavengers were introduced to inhibit the production of singlet oxygen (1O2), hydroxyl radicals (˙OH), and superoxide anion radicals (O2˙−), with the results shown in Fig. 5a. The introduction of p-benzoquinone (p-BQ), a known scavenger for O2˙−, resulted in no conversion, underscoring the essential role of photogenerated O2˙− as a ROS in the reaction. Conversely, the addition of isopropanol (IPA), a scavenger for ˙OH, and sodium azide (NaN3), a scavenger for 1O2, did not significantly affect the reaction conversion, suggesting that ˙OH and 1O2 are not involved.83,84 Furthermore, conducting the reaction with 2,2,6,6-tetramethyl-1-piperidinyloxyl (TEMPO) led to a substantial decrease in conversion, providing strong evidence that the reaction follows an amine radical pathway.84 To support this hypothesis, a radical scavenging study was conducted using 2,6-di-tert-butyl-4-methylphenol (BHT) to capture the amine radical.56 After 24 hours of irradiation, the reaction mixture was analyzed by HR-MS and GC-MS, revealing a distinct HR-MS peak for the p-anisidine–BHT adduct with a molecular weight of 342.2416 g mol−1 (Fig. S5). No product was formed in the presence of BHT, which confirmed the involvement of amine radicals in the reaction. Additionally, the use of potassium persulfate (K2S2O8) as an electron scavenger56 or triethylamine (TEA) as a hole scavenger85 nearly halted the reaction, reinforcing the critical role of the photogenerated electron–hole pair in the photo redox process (Fig. 5a). Moreover, the generation of H2O2 as a byproduct from the superoxide radical was confirmed by adding a starch KI solution to the reaction mixture after photoreaction, which resulted in a dark blue-black colour (Fig. S6).86,87 In addition, quantitative analysis using KMnO4 redox titration88 confirmed that approximately 20 µmol of H2O2 was generated as a byproduct under the standard reaction conditions.The formation of O2˙− was convincingly validated using EPR spectroscopy, with 5,5-dimethyl-1-pyrroline N-oxide (DMPO) as a spin-trap under standard reaction conditions. Under conditions devoid of light, no free radical response was detected via EPR, underscoring the necessity of light for this reaction. However, upon introducing DMPO to the reaction mixture and irradiating it with 395 nm light for 5 min, a distinct pattern of four well-defined peaks corresponding to DMPO–O2˙− was observed (see Fig. 5b). The same pattern for the EPR spectra of mpg-C3N4 was observed under both dark and light irradiation conditions (Fig. S7). This result aligns well with previous studies, confirming that superoxide radical anions are the primary reactive species involved in the photocatalytic oxidative coupling of amines using mpg-C3N4.21,89 Additionally, to determine the possible involvement of oxygen-containing intermediates, such as nitroso or nitro compounds in the reaction pathway, control experiments were conducted under standard conditions, and the results of these experiments suggest that oxygenated intermediates are unlikely to be involved in the reaction mechanism (Fig. S4).
The photocatalytic performance extends beyond the sensitive optical response and the generation of stable electron–hole pairs. Alongside the photogenerated charge carriers described earlier, the nature of the surface-active sites is crucial in driving the oxidative coupling of amines. Notably, FT-IR spectroscopy, XPS, BET, and CO2-TPD analyses reveal that mpg-C3N4 features abundant nitrogen-rich functional motifs including –NHx groups, bridging C–N–C linkages, and electron-rich sp2 nitrogen species within the heptazine/triazine framework. These sites are known to facilitate substrate adsorption, activate amines, and stabilize nitrogen-centered radical intermediates.90–92 Furthermore, the mesoporous architecture and high surface area of mpg-C3N4 promote the accessibility of these active centers, leading to strengthened interactions among the catalyst surface, amines, and reactive oxygen species.93 These structural features collectively play a significant role in enhancing the photocatalytic efficiency.
Crucially, it is also essential to consider how reactive molecules, such as the substrate and O2, interact with the true active sites of the mpg-C3N4 photocatalyst. To this end, DFT calculations were performed to investigate the function of mpg-C3N4 in the photocatalytic aerobic oxidative coupling of aryl amines to afford azoaromatics, with particular focus on the activation of oxygen species. Guided by XPS characterization, a tri-s-triazine structural model of mpg-C3N4 was utilized for the simulations. The adsorption studies of p-anisidine on the catalyst revealed an adsorption Gibbs energy of −13.6 kcal mol−1. Notably, p-anisidine displayed π–π stacking interactions atop the tri-s-triazine unit (Fig. 5c), demonstrating a preference for this orientation over adsorption within the mesoporous cavities of mpg-C3N4. In the latter case, a more pronounced distortion of the mpg-C3N4 model was observed, reducing ΔGads to −4.1 kcal mol−1 (Fig. 5d). The dependence of adsorption strength on the adsorption site aligns with previous reports regarding the adsorption of benzyl alcohol on polymeric carbon nitride.94 Consequently, the structural integrity of mpg-C3N4 facilitates the efficient interaction of aromatic amines with the catalyst surface, thereby enhancing its role as a support in oxidative coupling reactions.
Furthermore, aligning with the radical trap experiments, the adsorption of superoxide radical (Fig. 5e) was examined to understand the activation of reactive oxygen species. The superoxide radical demonstrated an adsorption Gibbs energy of −10.9 kcal mol−1, and the optimized model of O2@mpg-C3N4 revealed an elongated O–O bond length of 1.3 Å in contrast to the 1.2 Å bond length of molecular O2, which was also observed for the Mn1/tri-CN photocatalyst.91 A Wiberg bond index (WBI) analysis indicated a bond order of 1.3, confirming the formation of the superoxide radical and implicating its key role in the oxidative coupling of aryl amines to generate azoaromatics. These findings underscore the critical role of mpg-C3N4 not only as a structural support but also as an active platform that facilitates and enhances the adsorption and activation of reactants in oxidative coupling reactions.
Taking into account the above-mentioned experimental and theoretical results, along with the previously reported reaction mechanism, we investigated the reaction pathway for the light-driven oxidative coupling of amines.27,95 Upon light irradiation, mpg-C3N4 generates electron–hole pairs; the photogenerated electrons reduce O2 to superoxide radicals (O2˙−), while the holes oxidize the amines to their corresponding radical cations.56 As illustrated in Fig. 6, the transformation of p-anisidine into azo compounds proceeds through a sequence of radical formation, proton transfer, homocoupling, and stepwise oxidation processes. After the adsorption of a reactant onto mpg-C3N4 with a Gibbs energy of −14 kcal mol−1, the reaction begins with the hole transfer and formation of superoxide radicals (Fig. 6, step a), a step that is thermodynamically feasible under direct light at a temperature of 313 K, with a reaction Gibbs energy of 22 kcal mol−1. This is followed by a key activation step where a superoxide radical (O2˙−) oxidizes the aniline to a nitrogen-centered radical, showing a reaction Gibbs energy of 9 kcal mol−1 (Fig. 6, step b). The pivotal event in the sequence is the homocoupling of two nitrogen-centered radicals (Fig. 6, step c), which forges the N–N bond, with a reaction Gibbs energy of −17 kcal mol−1. Subsequent oxidative steps mediated by ROS, such as superoxide and hydroperoxyl radicals (HOO˙), convert the hydrazine intermediate to azoaromatics, with each step proceeding through energetically favorable or accessible pathways (Fig. 6, steps d–f). These findings indicate that the catalytic role of mpg-C3N4 is related to the generation of superoxide radicals as well as the special organization of reactants on the catalyst surface (Fig. 6).
2.4 Stability studies
Sustainability and reusability are critical factors in assessing high-performance catalysts, particularly photocatalysts, where the effectiveness is closely related to their ability to be reused. To evaluate the reusability and stability of mpg-C3N4 photocatalysts, the catalyst was extracted from the reaction medium using centrifugation and thoroughly washed several times with DMSO, H2O, and EtOH. Recycling tests were conducted over five consecutive cycles under standard reaction conditions. As shown in Fig. 7a, mpg-C3N4 retained its catalytic efficiency with no significant decline after five cycles. Furthermore, the structural and morphological integrity of the recycled catalyst was examined through a range of characterization techniques such as XRD, FT-IR spectroscopy, and HR-TEM analysis. The XRD and FT-IR spectra of the recycled mpg-C3N4 sample after five cycles closely resembled those of the freshly synthesized mpg-C3N4 sample, demonstrating the remarkable stability of this heterogeneous photocatalyst (Fig. 7b and c). A slight enhancement in the FT-IR absorption intensity of the triazine (800 cm−1) and N–H/O–H (∼3000 cm−1) bands was observed for the reused catalyst after the 5th cycle, which could be due to minor structural modifications or the surface-adsorbed species that accumulate during repeated use (Fig. 7c). Furthermore, HR-TEM analysis confirmed that the nanowire morphology of mpg-C3N4 was preserved even after five cycles (Fig. 7d–f). In summary, the robust and reusable characteristics of mpg-C3N4 photocatalysts ensure reliable performance under standard reaction conditions, underscoring their potential for sustainable catalytic applications.3 Conclusions
In summary, we have successfully developed a metal-free mpg-C3N4 photocatalyst using a straightforward one-step thermal polymerization method and utilized it for the photocatalytic oxidative coupling of arylamines to generate aromatic azo compounds under mild conditions. This metal-free mpg-C3N4 photocatalyst exhibited exceptional activity across a diverse range of substrates, including both electron-rich and electron-deficient amines, achieving conversions and selectivities up to 99%. The ability of the mpg-C3N4 photocatalyst to harness light, combined with its high surface area, mesoporous structure, and efficient charge transfer and separation, significantly enhances its photocatalytic performance. The mechanism underlying the photocatalytic oxidative coupling of amines has been explored through a series of control experiments, EPR spectroscopy, and DFT studies. These mechanistic investigations revealed that the mpg-C3N4 photocatalyst can activate atmospheric O2 molecules to produce superoxide radical anions (O2˙−), which are highly reactive species. Importantly, this photocatalyst retained its catalytic efficiency over five cycles without any significant loss. The mild and environmentally friendly reaction conditions utilizing light as the irradiation source, an aerial atmosphere, and a metal-free photocatalyst render the mpg-C3N4-catalyzed process both green and sustainable. As the demand for eco-friendly approaches in chemical synthesis surges, our findings not only lay the foundation for ground-breaking advancement in photocatalyst design and application but also contribute significantly to the global quest for sustainable methods that reduce environmental impacts and enhance resource efficiency. In the future, this mpg-C3N4 catalyst may also be explored for other photocatalytic transformations, including oxidative or reductive functionalization, C–N/C–C bond-forming reactions, and the upgrading of biomass-derived substrates.Author contributions
Dinesh S. Chaudhari: writing – review & editing, writing – original draft, methodology, investigation, formal analysis, conceptualization. Rahul P. Gaikwad: writing – review & editing, formal analysis. Indrajeet R. Warkad: writing – review & editing, formal analysis. Rostislav Langer and Michal Otyepka: theoretical calculations, writing – DFT section. Manoj B. Gawande: writing – review & editing, visualization, validation, supervision, project administration, methodology, investigation, funding acquisition, formal analysis, data curation, conceptualization.Conflicts of interest
The authors confirm that there are no financial conflicts of interest or personal relationships that could have impacted the work described in this paper.Data availability
The data supporting this article are provided within the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta08970k.Acknowledgements
D. S. C. and I. R. W. express their gratitude to the Institute of Chemical Technology (ICT), Mumbai-Marathwada Campus, Jalna, for providing the facilities for doctoral studies. R. P. G. acknowledges the Council of Scientific and Industrial Research (CSIR), New Delhi, for granting the research fellowship for his doctoral research. D. S. C. and M. B. G. acknowledge the financial support from the DST-SERB Core Research Grant (CRG/2021/001738). This work was supported by the Ministry of Education, Youth and Sports of the Czech Republic through the e-INFRA CZ (ID: 90254). M. O. acknowledges the support of MEYS through the ERDF/ESF project TECHSCALE (No. CZ.02.01.01/00/22_008/0004587). M. O. also acknowledges the financial support from the European Union through the REFRESH project – Research Excellence For REgion Sustainability and High-tech Industries (CZ.10.03.01/00/22_003/0000048), funded via the Operational Programme Just Transition.References
- J. Huo and C. Peng, Resour. Policy, 2023, 86, 104049 CrossRef.
- J. Wang and W. Azam, Geosci. Front., 2024, 15, 101757 Search PubMed.
- W.-J. Ong, L.-L. Tan, Y. H. Ng, S.-T. Yong and S.-P. Chai, Chem. Rev., 2016, 116, 7159–7329 Search PubMed.
- B. Ran, L. Ran, Z. Wang, J. Liao, D. Li, K. Chen, W. Cai, J. Hou and X. Peng, Chem. Rev., 2023, 123, 12371–12430 CrossRef CAS PubMed.
- L. Buglioni, F. Raymenants, A. Slattery, S. D. A. Zondag and T. Noël, Chem. Rev., 2022, 122, 2752–2906 CrossRef CAS PubMed.
- A. Galushchinskiy, R. González-Gómez, K. McCarthy, P. Farràs and A. Savateev, Energy Fuels, 2022, 36, 4625–4639 Search PubMed.
- P. Sharma, S. Kumar, O. Tomanec, M. Petr, J. Z. Chen, J. T. Miller, R. S. Varma, M. B. Gawande and R. Zbořil, Small, 2021, 17, 2006478 Search PubMed.
- R. P. Gaikwad, I. R. Warkad, D. S. Chaudhari, S. Jiang, J. T. Miller, H. N. Pham, A. Datye and M. B. Gawande, J. Colloid Interface Sci., 2024, 676, 485–495 CrossRef CAS PubMed.
- P. Li, J. A. Terrett and J. R. Zbieg, ACS Med. Chem. Lett., 2020, 11, 2120–2130 CrossRef CAS PubMed.
- T. T. Salunkhe, T. R. Gurugubelli, B. Viswanadham, B. Babu, T. V. M. Sreekanth and K. Yoo, J. Chem., 2025, 2025, 8849437 Search PubMed.
- B. Singh, V. Sharma, R. P. Gaikwad, P. Fornasiero, R. Zbořil and M. B. Gawande, Small, 2021, 17, 2006473 CrossRef CAS PubMed.
- R. Paul, I. R. Warkad, S. Arulkumar, S. Parthiban, H. R. Darji, M. Naushad, R. G. Kadam and M. B. Gawande, Mol. Catal., 2022, 530, 112566 CAS.
- R. P. Gaikwad, D. R. Naikwadi, A. V. Biradar and M. B. Gawande, ACS Appl. Nano Mater., 2023, 6, 1859–1869 Search PubMed.
- X. Li, Q. Cheng, Y. Zhang, Y. Liu, Y. Pan, D. Zhao, S. Xiong, W. Liu, X. Jiang, J. Yan, X. Duan, Y. Tian and X. Li, Angew. Chem., Int. Ed., 2025, 64, e202424435 Search PubMed.
- C. Karthikeyan, P. Arunachalam, K. Ramachandran, A. M. Al-Mayouf and S. Karuppuchamy, J. Alloys Compd., 2020, 828, 154281 CrossRef CAS.
- J. Wang and S. Wang, Coord. Chem. Rev., 2022, 453, 214338 CrossRef CAS.
- G. F. S. R. Rocha, M. A. R. da Silva, A. Rogolino, G. A. A. Diab, L. F. G. Noleto, M. Antonietti and I. F. Teixeira, Chem. Soc. Rev., 2023, 52, 4878–4932 Search PubMed.
- S. Das, K. Murugesan, G. J. V. Rodríguez, J. Kaur, J. P. Barham, A. Savateev, M. Antonietti and B. König, ACS Catal., 2021, 11, 1593–1603 Search PubMed.
- A. Savateev, I. Ghosh, B. König and M. Antonietti, Angew. Chem., Int. Ed., 2018, 57, 15936–15947 Search PubMed.
- I. Ghosh, J. Khamrai, A. Savateev, N. Shlapakov, M. Antonietti and B. König, Science, 2019, 365, 360–366 Search PubMed.
- J. Bai, S. Yan, Z. Zhang, Z. Guo and C.-Y. Zhou, Org. Lett., 2021, 23, 4843–4848 CrossRef CAS PubMed.
- X. Wang, K. Maeda, A. Thomas, K. Takanabe, G. Xin, J. M. Carlsson, K. Domen and M. Antonietti, Nat. Mater., 2009, 8, 76–80 Search PubMed.
- A. Kumar, P. Raizada, V. K. Thakur, V. Saini, A. A. P. Khan, N. Singh and P. Singh, Chem. Eng. Sci., 2021, 230, 116219 Search PubMed.
- L. Ma, Y. Gao, B. Wei, L. Huang, N. Zhang, Q. Weng, L. Zhang, S. F. Liu and R. Jiang, ACS Catal., 2024, 14, 2775–2786 CrossRef CAS.
- Y. Zhao, Z. Wang, X. Wang, P. Sun, F. Meng, G. Yang, X. Zhou, J. Wang, X. Zhang and G. Lyu, Adv. Funct. Mater., 2025, 35, 2422264 CrossRef CAS.
- K. S. Lakhi, D.-H. Park, K. Al-Bahily, W. Cha, B. Viswanathan, J.-H. Choy and A. Vinu, Chem. Soc. Rev., 2017, 46, 72–101 Search PubMed.
- J. D. Sitter and A. K. Vannucci, J. Am. Chem. Soc., 2021, 143, 2938–2943 CrossRef CAS PubMed.
- A. Grirrane, A. Corma and H. García, Science, 2008, 322, 1661–1664 Search PubMed.
- S. Crespi, N. A. Simeth and B. König, Nat. Rev. Chem., 2019, 3, 133–146 Search PubMed.
- L. De Boni, E. Piovesan, L. Misoguti, S. C. Zílio and C. R. Mendonca, J. Phys. Chem. A, 2007, 111, 6222–6224 Search PubMed.
- A. Bafana, S. S. Devi and T. Chakrabarti, Environ. Rev., 2011, 19, 350–371 CrossRef CAS.
- N. N. Riaz, S. Iqbal, S. Amin, K. Ahmad, M. Atif, S. Muhammad, M. M. Ahmed and M. Ashfaq, J. Mol. Struct., 2025, 1321, 139705 Search PubMed.
- C. Fedele, P. A. Netti and S. Cavalli, Biomater. Sci., 2018, 6, 990–995 Search PubMed.
- B. Zhang, Y. Feng and W. Feng, Nano-Micro Lett., 2022, 14, 138 Search PubMed.
- F. Cuétara-Guadarrama, M. Vonlanthen, K. Sorroza-Martínez, I. González-Méndez and E. Rivera, Dyes Pigm., 2021, 194, 109551 Search PubMed.
- E. Merino, Chem. Soc. Rev., 2011, 40, 3835–3853 Search PubMed.
- L. Wang, A. Ishida, Y. Hashidoko and M. Hashimoto, Angew. Chem., Int. Ed., 2017, 56, 870–873 CrossRef CAS PubMed.
- M. K. Sahoo, K. Saravanakumar, G. Jaiswal and E. Balaraman, ACS Catal., 2018, 8, 7727–7733 Search PubMed.
- H. Zhu, X. Ke, X. Yang, S. Sarina and H. Liu, Angew. Chem., Int. Ed., 2010, 49, 9657–9661 Search PubMed.
- C. Zhang and N. Jiao, Angew. Chem., Int. Ed., 2010, 49, 6174–6177 CrossRef CAS PubMed.
- S. Cai, H. Rong, X. Yu, X. Liu, D. Wang, W. He and Y. Li, ACS Catal., 2013, 3, 478–486 Search PubMed.
- A. Saha, S. Payra, B. Selvaratnam, S. Bhattacharya, S. Pal, R. T. Koodali and S. Banerjee, ACS Sustainable Chem. Eng., 2018, 6, 11345–11352 Search PubMed.
- M. Kumar, K. Soni, G. D. Yadav, S. Singh and S. Deka, Appl. Catal., A, 2016, 525, 50–58 Search PubMed.
- H. Song, J. Wei, Z. Wang, Y. Liu, S. Zhao, X. Cai, Y. Xiao, L. Yang, P. Bai, L. Fang, F. Yang, S. Zheng, W. Zhang, J. Pan and C. Xu, ACS Catal., 2024, 14, 12372–12384 CrossRef CAS.
- X. Deng, X. Liu, S. Xia, H. Zhao, Y. Liu, Q. Ding and H. Zhang, Colloids Surf., A, 2023, 677, 132352 CrossRef CAS.
- S. Agarwal, B. Dowara, S. Kumar, V. Kumar and K. Deori, ACS Omega, 2022, 7, 48615–48622 CrossRef CAS PubMed.
- Q. Niu, Q. Huang, T.-Y. Yu, J. Liu, J.-W. Shi, L.-Z. Dong, S.-L. Li and Y.-Q. Lan, J. Am. Chem. Soc., 2022, 144, 18586–18594 CrossRef CAS PubMed.
- A. R. Patel, G. Patel, G. Maity, S. P. Patel, S. Bhattacharya, A. Putta and S. Banerjee, ACS Omega, 2020, 5, 30416–30424 CrossRef CAS PubMed.
- S. Singh, V. Agarwal, T. K. Sarma and T. K. Mukherjee, Green Chem., 2023, 25, 9109–9114 RSC.
- S. Singh and T. K. Mukherjee, Chem. Sci., 2024, 15, 13949–13957 RSC.
- W. Chen, H. Li, Y. Jin, C. Wu, Z. Yuan, P. Ma, J. Wang and J. Niu, Chem. Commun., 2022, 58, 9902–9905 RSC.
- X. Han, T. Zhang, X. Wang, Z. Zhang, Y. Li, Y. Qin, B. Wang, A. Han and J. Liu, Nat. Commun., 2022, 13, 2900 CrossRef CAS.
- C. Wang, B. Hu, X. Guo and L. Lei, Green Chem., 2024, 26, 6470–6479 RSC.
- M. Fronczak, E. Tálas, Z. Pászti, G. P. Szijjártó, J. Mihály, A. Tompos, P. Baranowski, S. K. Tiwari and M. Bystrzejewski, Diamond Relat. Mater., 2022, 125, 109006 CrossRef CAS.
- L. Florentino-Madiedo, E. Díaz-Faes and C. Barriocanal, Carbon, 2022, 187, 462–476 CrossRef CAS.
- A. Shi, K. Sun, Y. Wu, P. Xiang, I. B. Krylov, A. O. Terent'ev, X. Chen and B. Yu, J. Catal., 2022, 415, 28–36 Search PubMed.
- J. Lu, Z. Chen, Y. Shen, H. Yuan, X. Sun, J. Hou, F. Guo, C. Li and W. Shi, J. Colloid Interface Sci., 2024, 670, 428–438 CrossRef CAS PubMed.
- J. Xu, M. Fujitsuka, S. Kim, Z. Wang and T. Majima, Appl. Catal., B, 2019, 241, 141–148 CrossRef CAS.
- Y. Gao, J. Duan, X. Zhai, F. Guan, X. Wang, J. Zhang and B. Hou, J. Nanopart. Res., 2021, 23, 44 Search PubMed.
- P. Xia, G. Li, X. Li, S. Yuan, K. Wang, D. Huang, Y. Ji, Y. Dong, X. Wu, L. Zhu, W. He and L. Qiao, Crystals, 2022, 12, 1719 CrossRef CAS.
- D. Feng, Y. Cheng, J. He, L. Zheng, D. Shao, W. Wang, W. Wang, F. Lu, H. Dong, H. Liu, R. Zheng and H. Liu, Carbon, 2017, 125, 454–463 CrossRef CAS.
- J. Liu, T. Zhang, Z. Wang, G. Dawson and W. Chen, J. Mater. Chem., 2011, 21, 14398–14401 RSC.
- H. Chand, A. Kumar, S. Goswami and V. Krishnan, Fuel, 2024, 357, 129757 CrossRef CAS.
- T. Wasiak and D. Janas, J. Alloys Compd., 2022, 892, 162158 CrossRef CAS.
- M. Li, L. Zhang, X. Fan, M. Wu, M. Wang, R. Cheng, L. Zhang, H. Yao and J. Shi, Appl. Catal., B, 2017, 201, 629–635 CrossRef CAS.
- L.-y. Li, H. Gu, V. Šrot, P. van Aken and J. Bill, J. Mater. Sci. Technol., 2019, 35, 2851–2858 CrossRef CAS.
- S. Martha, A. Nashim and K. M. Parida, J. Mater. Chem. A, 2013, 1, 7816–7824 RSC.
- J. Oh, J. M. Lee, Y. Yoo, J. Kim, S.-J. Hwang and S. Park, Appl. Catal., B, 2017, 218, 349–358 CrossRef CAS.
- J.-P. Zou, L.-C. Wang, J. Luo, Y.-C. Nie, Q.-J. Xing, X.-B. Luo, H.-M. Du, S.-L. Luo and S. L. Suib, Appl. Catal., B, 2016, 193, 103–109 CrossRef CAS.
- C. Cheng, J. Shi, L. Wen, C.-L. Dong, Y.-C. Huang, Y. Zhang, S. Zong, Z. Diao, S. Shen and L. Guo, Carbon, 2021, 181, 193–203 CrossRef CAS.
- S. Yang, Y. Gong, J. Zhang, L. Zhan, L. Ma, Z. Fang, R. Vajtai, X. Wang and P. M. Ajayan, Adv. Mater., 2013, 25, 2452–2456 CrossRef CAS PubMed.
- J. Liu, J. Yan, H. Ji, Y. Xu, L. Huang, Y. Li, Y. Song, Q. Zhang, H. Xu and H. Li, Mater. Sci. Semicond. Process., 2016, 46, 59–68 CrossRef CAS.
- J. Chen, C. Shen, B. Guo, Y. Yu and L. Wu, Catal. Today, 2019, 335, 312–318 CrossRef CAS.
- M. Devi, M. H. Barbhuiya, B. Das, B. Bhuyan and S. S. Dhar, Sustain. Energy Fuels, 2020, 4, 3537–3545 RSC.
- M. Shen, L. Zhang, M. Wang, J. Tian, X. Jin, L. Guo, L. Wang and J. Shi, J. Mater. Chem. A, 2019, 7, 1556–1563 RSC.
- Y. Zhang, J. Liu, G. Wu and W. Chen, Nanoscale, 2012, 4, 5300–5303 RSC.
- M. A. Quintana, R. R. Solís, G. Blázquez, M. Calero and M. J. Muñoz-Batista, Appl. Surf. Sci., 2024, 656, 159717 CrossRef CAS.
- Y. Yang, G. Zeng, D. Huang, C. Zhang, D. He, C. Zhou, W. Wang, W. Xiong, B. Song, H. Yi, S. Ye and X. Ren, Small, 2020, 16, 2001634 CrossRef CAS.
- R. P. Gaikwad, I. R. Warkad, D. S. Chaudhari, H. N. Pham, A. K. Datye and M. B. Gawande, J. Colloid Interface Sci., 2025, 679, 54–63 CrossRef CAS PubMed.
- S. Han, Y. Cheng, S. Liu, C. Tao, A. Wang, W. Wei, H. Yu and Y. Wei, Angew. Chem., Int. Ed., 2021, 60, 6382–6385 CrossRef CAS PubMed.
- Y. Zou, M. Zhang, F. Cao, J. Li, S. Zhang and Y. Qu, J. Mater. Chem. A, 2021, 9, 19692–19697 RSC.
- J. Qin, Y. Long, F. Sun, P.-P. Zhou, W. D. Wang, N. Luo and J. Ma, Angew. Chem., Int. Ed., 2022, 61, e202112907 CrossRef CAS PubMed.
- W. Zou, X.-H. Liu, C. Xue, X.-T. Zhou, H.-Y. Yu, P. Fan and H.-B. Ji, Appl. Catal., B, 2021, 285, 119863 CrossRef CAS.
- G. Sportelli, G. Grando, M. Bevilacqua, G. Filippini, M. Melchionna and P. Fornasiero, Chem.–Eur. J., 2023, 29, e202301718 CrossRef CAS PubMed.
- A. Augustin, P. Ganguly, S. Shenoy, C. Chuaicham, S. C. Pillai, K. Sasaki, A. F. Lee and K. Sekar, Adv. Sustain. Syst., 2024, 8, 2400321 CrossRef CAS.
- L. Huang, J. Zhao, S. Guo, C. Zhang and J. Ma, J. Org. Chem., 2013, 78, 5627–5637 CrossRef CAS PubMed.
- M. K. Sahoo, G. Jaiswal, J. Rana and E. Balaraman, Chem.–Eur. J., 2017, 23, 14167–14172 CrossRef CAS PubMed.
- Y. Shiraishi, S. Kanazawa, Y. Sugano, D. Tsukamoto, H. Sakamoto, S. Ichikawa and T. Hirai, ACS Catal., 2014, 4, 774–780 CrossRef CAS.
- Y. Zhao and M. Antonietti, Angew. Chem., Int. Ed., 2017, 56, 9336–9340 CrossRef CAS.
- G. Grando, G. Sportelli, G. Filippini, M. Melchionna and P. Fornasiero, Nano Trends, 2023, 4, 100028 CrossRef.
- Z. Zhang, W. Liu, Y. Zhang, J. Bai and J. Liu, ACS Catal., 2021, 11, 313–322 CrossRef CAS.
- M. Yang, R. Lian, X. Zhang, C. Wang, J. Cheng and X. Wang, Nat. Commun., 2022, 13, 4900 CrossRef CAS.
- C. Xing, G. Yu, T. Chen, S. Liu, Q. Sun, Q. Liu, Y. Hu, H. Liu and X. Li, Appl. Catal., B, 2021, 298, 120534 CrossRef CAS.
- J. Ma, F. Zhang, Y. Tan, S. Wang, H. Chen, L. Zheng, H. Liu and R. Li, ACS Appl. Mater. Interfaces, 2022, 14, 18383–18392 CrossRef CAS PubMed.
- L. Kaur, M. Kumar and V. Bhalla, Green Chem., 2023, 25, 5240–5246 RSC.
| This journal is © The Royal Society of Chemistry 2026 |