Novel redox-active vanadium(V)-dithiolene complexes for efficient supercapacitive energy storage: role of selenium functionalization on pseudocapacitive properties
Sangita
Mondal
 a,
T. Kedara
Shivasharma
a,
T. Kedara
Shivasharma
 b,
Sujit
Das
a,
Mayur
Thosare
b,
Christel Livia
Mascarenhas
a,
Babasaheb R.
Sankapal
b,
Sujit
Das
a,
Mayur
Thosare
b,
Christel Livia
Mascarenhas
a,
Babasaheb R.
Sankapal
 *b and
Kartik
Chandra Mondal
*b and
Kartik
Chandra Mondal
 *a
*a
aDepartment of Chemistry, Indian Institute of Technology Madras, Chennai, Tamil Nadu 600036, India. E-mail: csdkartik@iitm.ac.in
bNano Materials and Device Laboratory, Department of Physics, Visvesvaraya National Institute of Technology, South Ambazari Road, Nagpur-440010, M.S., India
First published on 14th January 2026
Abstract
Supercapacitors have been established as efficient storage devices to address the intermittent output issue of renewable energy systems, bridging the gap between batteries and conventional capacitors by offering high power density, rapid charge–discharge rates, and long cycle life; however, achieving superior electrochemical performance remains a key challenge. In this regard, the present work emphasizes the synthesis and characterization of two photo-redox-active anionic V(V)-dithiolene complexes [(THF)4Li][V(V)(SS–NHC = E)3] (E = S (1), Se (2)), by reacting dithiolene radical anions [(THF)2Li(SS-NHC = S˙−)] and [(THF)2Li(SS-NHC = Se˙−)] with V(III)Cl3 in THF, respectively. These complexes were thoroughly characterized using UV-vis-NIR, IR, Raman and EPR spectroscopy, confirming their structural and electronic properties, which are well correlated with their electrochemical energy storage capabilities using cyclic voltammetry (CV), galvanostatic charge–discharge (GCD), electrochemical impedance spectroscopy (EIS) and stability tests. Electrochemical studies reveal that both complexes can exhibit remarkable pseudocapacitive behavior in transition-metal-dithiolene-based materials with excellent charge storage capacity and cycling stability. The unique electronic structures of these vanadium-dithiolene complexes contribute to their efficient redox activity, making them a promising class of supercapacitor material. Moreover, variations in electrolyte composition significantly influenced their performance, highlighting the crucial role of ion–electrode interactions. Impedance analysis further confirmed their low charge transfer resistance, suggesting efficient ion diffusion and active redox processes. The materials exhibited good cycling stability, highlighting the robust redox-active sites and their suitability for long-term energy storage applications. Complex 1 exhibited a slightly better specific capacitance of 82.82 F g−1 (areal 82.82 mF cm−2) than complex 2 (76.56 F g−1, areal 76.56 mF cm−2) at 5 mV s−1, while complex 2 exhibited excellent capacitive retention of 104.39% after 2000 cycles. Complex 1 showed some degradation over prolonged cycling. This study also highlights the potential of redox non-innocent dithiolene ligands in the design of advanced and next-generation energy storage materials for the first time.
1. Introduction
Electrochemical energy storage devices, particularly supercapacitors, have attracted considerable attention due to their high power density, rapid charge–discharge capabilities, and long cycle life, making them ideal for applications in portable electronics, electric vehicles, and grid stabilization.1 Among the various materials investigated for supercapacitor electrodes, transition metal complexes2,3 have emerged as promising candidates due to their tunable redox properties and versatile coordination environments. Vanadium, in particular, is a noteworthy element in this regard because of its ability to adopt multiple oxidation states, which enables rich redox chemistry and enhances charge storage mechanisms.4–6 Among the various ligand systems explored for redox-active metal complexes in electrochemical energy storage, dithiolene ligands have gained significant attention due to their unique electronic properties and non-innocent behavior.7 These ligand systems have been extensively explored in coordination chemistry for their strong electron-donating ability and unique “non-innocent” redox behavior, allowing them to influence the electronic properties of the coordinated metal center. This has been investigated both experimentally and theoretically,8 revealing that dithiolenes can adopt multiple electronic states, from ene-1,2-dithiolate to dithioketone, influencing molecular geometry, spectroscopic properties, and reactivity. These properties make dithiolenes highly versatile, leading to diverse structural and electronic variations in metal complexes, including those of vanadium.9–13 Electrochemically active vanadium(IV/V)-dithiolene complexes are structurally robust14 with diverse electronic stability, making them suitable for various applications. Spectroscopic and electrical properties of oxovanadium(IV)-dithiolene complexes have also been studied.15 Vanadium tris(dithiolene) complexes, such as [V(S2C2H2)3], show enhanced stability and unique electronic features, often displaying a nearly perfect D3h geometry.16 Additionally, vanadium-(mnt2−)-dithiolene complexes have been shown to serve as single-species electrolytes in symmetric nonaqueous redox flow batteries (RFBs) due to their multiple redox processes, enhancing their potential in molecular conductors and energy storage.17Beyond their diverse redox and electronic properties, vanadium complexes have also been explored for their potential in energy storage applications, particularly supercapacitors.18,19 Supercapacitors, also known as electrochemical capacitors, have a longer lifetime and higher power densities than traditional batteries.20 The pseudocapacitive behavior of electrode materials, which relies on fast, reversible faradaic reactions occurring at or near the electrode surface, is essential for achieving high-performance supercapacitors. Recent studies have shown that vanadium-based materials, including oxides, their composites with carbon nanomaterials, V-based MOFs, sulfides or polyoxometalates, can deliver outstanding pseudocapacitive performances due to their variable oxidation states and efficient redox kinetics.21a–c These materials are typically developed as nanocomposite systems, often embedded in conductive carbon matrices to facilitate electrical transport.21d–f Within these systems, the chelation of redox non-innocent dithiolene ligands to vanadium ions further influences electron transfer and ion diffusion, thereby promoting the diffusion-limited charge storage mechanism characteristic of pseudocapacitors. Despite the promising attributes of vanadium-dithiolene complexes, the literature on hexacoordinated vanadium(V) dithiolene systems, in the context of energy storage featuring NHC carbene-derived non-innocent radical ligands, remains relatively scarce.15–17
Addressing this gap, the present work reports the synthesis and isolation of two novel hexa-coordinate V(V)-dithiolene complexes, reacting the dithiolene radical anions [(THF)2Li(SS-NHC = S)]22 and [(THF)2Li(SS-NHC = Se)]23 with anhydrous V(III)Cl3 in THF to isolate six-coordinated V-dithiolene complexes [(THF)4Li][V(V)(SS–NHC = E)3] (E = S (1), Se (2)). The crystalline anionic dithiolene radical ligand, [(THF)2Li(SS-NHC = S)] was isolated initially by Robinson et al. in 2017, and its unique redox non-innocent aspects were explored through cyclic voltammetry studies.24 An octahedral aluminum-tri-radical complex with the same ligand was recently synthesized and characterized by different techniques.25 The C2–C3 bond length of 1.418 Å in the Al-tri-radical compound suggests electron delocalization within the C2S2 unit, retaining its π-delocalized ligand-centered radical character.25 Both the complexes (1 and 2) were structurally characterized by X-ray single-crystal diffraction, EPR, IR, and UV-vis-NIR spectroscopy, and further studied using cyclic voltammetry (CV) measurements. Comprehensive electrochemical evaluation for the two complexes was carried out using cyclic voltammetry (CV), galvanostatic charge–discharge (GCD), and electrochemical impedance spectroscopy (EIS), where investigation of the pseudocapacitive behavior of these materials was performed. Our findings demonstrate that the complexes exhibit diffusion-limited, reversible faradaic processes and possess excellent cycling stability—attributes crucial for their application as pseudocapacitor electrodes. Both vanadium(V)-dithiolene complexes have been extensively studied. Between them, the Se-analogue [Li(THF)4][V(SS-NHC = Se)3] (2) exhibits better pseudocapacitive behavior than complex 1, considering its stability, as evident from its electrolyte-dependent capacitance, response at different scan rates, and galvanostatic charge–discharge trends, which are described here in depth.
A comparative data table (Table S1), summarizing reported vanadium-based supercapacitor materials alongside our complexes, has been added to the SI. Many literature reports achieve high capacitance through bulk redox processes and conductive substrates like Ni foam or graphite, which improve performance but increase cost and limit scalability. In contrast, our work introduces V(+5)-dithiolene complexes as discrete molecular redox-active units showing pseudocapacitive behavior with moderate capacitance and good cycling stability, deposited directly onto cost-effective stainless steel. We acknowledge that the performance could be further enhanced by forming composites with EDLC materials, which we see as a promising direction for future research.
2. Experimental section
2.1 Materials and methods
All experiments were performed using either standard Schlenk-line techniques under an argon atmosphere or in an argon-filled MBraun Eco Plus glove box, where O2 and H2O levels were maintained below 0.1 ppm at all times. Solvents obtained from an MBraun Solvent Purification System (SPS) were further dried by refluxing with Na/K alloy for two days, followed by vacuum distillation over 4 Å molecular sieves. N-Heterocyclic carbene was synthesized according to the procedures reported in the literature. [(THF)2Li(SS-NHC = S)] was prepared following the reported synthetic procedure.22 Melting points were measured in sealed glass tubes on a Rashmi Melting Point Apparatus. Absorption spectra were obtained on a Shimadzu UV-3100 UV-Vis-NIR Recording Spectrophotometer in the 200–2000 nm range. Solid-state FTIR measurements were made on a JASCO FT/IR-4100 spectrometer using KBr pellets. Cyclic voltammetry studies were performed on a CH instrument CHI608B (serial no. I0620, 220 V AC 0.4 A).2.2 Single-crystal X-ray diffraction
X-ray single-crystal diffraction data were collected on a Bruker VENTURE D8 APEX4 HEED X-ray diffractometer equipped with Helios optics mono-chromated Mo-Kα (λ = 0.71073 Å) radiation at 100 K. All data were integrated using SAINT PLUS, and absorption correction was done using the multi-scan absorption correction method (SADABS). Crystallographic data are summarized in Table 1. CCDC 2424329 and 2427545 contain the SI crystallographic data for this paper.| Complex | 1 (E = S) | 2 (E = Se) |
|---|---|---|
| Empirical formula | C97H134LiN6O4S9V | C97H134LiN6O4S6Se3V |
| Formula weight | 1794.51 | 1935.21 |
| Crystal size [mm] | 0.28 × 0.28 × 0.11 | 0.3 × 0.23 × 0.22 |
| Wavelength [Å] | 0.71073 | 0.71073 |
| Crystal system | Monoclinic | Monoclinic |
| Space group | P21/n | P21/n |
| a [Å] | 15.4498(12) | 15.4863(5) |
| b [Å] | 26.372(2) | 26.4050(9) |
| c [Å] | 24.676(2) | 24.7686(9) |
| α [°] | 90 | 90 |
| β [°] | 91.465(2) | 91.593(1) |
| γ [°] | 90 | 90 |
| V [Å3] | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 050.6(14) 050.6(14) |
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 124.4(6) 124.4(6) |
| Z | 4 | 4 |
| Temperature [K] | 100 | 100 |
| r [Mg m−3] | 2–1.186 | 1.27 |
| m [mm−1] | 0.34 | 1.35 |
| F (000) | 3840 | 4056 |
| θ-area [°] | 2.2–22.7 | 2.3–28.7 |
| Reflections collected | 486![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 516 516 |
137![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 675 675 |
| Independent reflections | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 368 368 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 892 892 |
| Reflections with I > 2σ(I) | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 163 163 |
15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 115 115 |
| R int | 0.105 | 0.050 |
| Number of restraints | 0 | 12 |
| Parameters | 1087 | 1097 |
| R 1 [I > 2 s(I)] | 0.051 | 0.081 |
| wR 2 [I > 2 s(I)] | 0.125 | 0.251 |
| R 1 [all data] | 0.076 | 0.091 |
| wR 2 [all data] | 0.142 | 0.264 |
| GooF | 1.03 | 1.08 |
| Largest diff. peak/hole/e Å−3 | 1.06/−0.56 | 2.17/−2.75 |
| CCDC number | 2424329 | 2427545 |
2.3 Electrode preparation for cyclic voltammetry
Electrolytic solutions were prepared in an argon-filled glove box. Cyclic voltammetry (CV) studies of complexes 1 and 2 were carried out with 0.1 M [n-Bu4N][PF6] as supporting electrolyte in alloy-distilled tetrahydrofuran (THF) solvent at scan rates of 50, 100 and 150 mV s−1. Blank cyclic voltammograms were also recorded at these scan rates. The electrochemical setup consisted of a glassy carbon (GC) working electrode (WE), an Ag-reference electrode (RE) and a Pt counter electrode (CE). All data were referenced to the ferrocene/ferrocenium couple.2.4 Supercapacitor electrode formation
A slurry of active material was prepared by mixing it with PVDF, and carbon black in N-methylpyrrolidone (NMP) at a weight ratio of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and it was coated on a mirror-polished stainless steel (SS) substrate. The electrochemical performances of the as-prepared electrode were investigated with the help of a Gamry instrument 1010E in three-electrode configuration, with Ag/AgCl as the reference electrode, SS coated with the active material as the working electrode and platinum wire as the counter electrode.
1, and it was coated on a mirror-polished stainless steel (SS) substrate. The electrochemical performances of the as-prepared electrode were investigated with the help of a Gamry instrument 1010E in three-electrode configuration, with Ag/AgCl as the reference electrode, SS coated with the active material as the working electrode and platinum wire as the counter electrode.
3. Results and discussion
3.1 Synthesis, structure and morphological studies
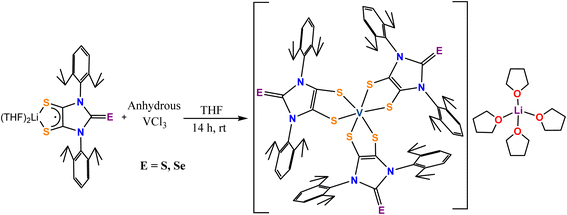
Both complex 1 [Li(THF)4][V(SS-NHC = S)3] and complex 2 [Li(THF)4][V(SS-NHC = Se)3] were synthesized by reacting the respective ligands [(THF)2Li(SS-NHC = S)] and [(THF)2Li(SS-NHC = Se)] with anhydrous vanadium(III)trichloride in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ligand-to-metal ratio in dry THF solvent. The synthesized complexes crystallized in the monoclinic P21/n space group. Single-crystal X-ray diffraction analysis reveals that complexes 1 and 2 contain a V(V) center coordinated by three S2-donor dithiolene dianion ligands [Fig. 1(a) and (b)]. There is an Li(THF)4 unit as a counter cation to balance the charge. The central vanadium ion in both complexes adopts a six-coordinated prismatic coordination geometry (VS6).15,16 The V–S bond distances in both complexes are comparable, ranging from 2.3634(9) to 2.4383(9) Å in complex 1 and from 2.3694(14) to 2.4369(14) Å in complex 2, indicating similar metal–ligand interactions. These bond lengths are longer than those reported for V(dddt)32− (2.340(4) Å) or V(S2C2Ph2)3 (2.337(6) Å), but are comparable to those of other V(dithiolene)32− complexes, such as V(dmit)32− (2.378(4) Å), V(dmt)32− (2.375(3) Å), and V(mnt)32− (2.36(1) Å), indicating a similar metal–ligand bonding environment influenced by the radical character of the ligands.11,25 The C
1 ligand-to-metal ratio in dry THF solvent. The synthesized complexes crystallized in the monoclinic P21/n space group. Single-crystal X-ray diffraction analysis reveals that complexes 1 and 2 contain a V(V) center coordinated by three S2-donor dithiolene dianion ligands [Fig. 1(a) and (b)]. There is an Li(THF)4 unit as a counter cation to balance the charge. The central vanadium ion in both complexes adopts a six-coordinated prismatic coordination geometry (VS6).15,16 The V–S bond distances in both complexes are comparable, ranging from 2.3634(9) to 2.4383(9) Å in complex 1 and from 2.3694(14) to 2.4369(14) Å in complex 2, indicating similar metal–ligand interactions. These bond lengths are longer than those reported for V(dddt)32− (2.340(4) Å) or V(S2C2Ph2)3 (2.337(6) Å), but are comparable to those of other V(dithiolene)32− complexes, such as V(dmit)32− (2.378(4) Å), V(dmt)32− (2.375(3) Å), and V(mnt)32− (2.36(1) Å), indicating a similar metal–ligand bonding environment influenced by the radical character of the ligands.11,25 The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S bond lengths in complex 1 (1.661(3)–1.665(3) Å) and C
S bond lengths in complex 1 (1.661(3)–1.665(3) Å) and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se bond lengths in complex 2 (1.769(5)–1.799(5) Å) align with the standard values for these bonds, indicating their expected structural characteristics.
Se bond lengths in complex 2 (1.769(5)–1.799(5) Å) align with the standard values for these bonds, indicating their expected structural characteristics.
Surface morphological studies were carried out through field emission scanning electron microscopy (FE-SEM). Fig. 2(a) and (b) depict the micrographs of complexes 1 and 2, respectively. The FE-SEM image of complex 1 displays a heterogeneous surface morphology composed of irregularly shaped fine particles scattered across the surface, along with larger crystalline aggregates. These fine particles might provide active sites for electrochemical activity. In contrast, complex 2 exhibits a significantly different microstructure, characterized by densely packed, plate-like crystalline domains with well-defined edges and layered arrangements. The distinct morphological difference between the two complexes suggests variations in packing and structural order, which may contribute to the enhanced electrochemical performance observed for complex 2.
 | ||
| Fig. 2 Field emission scanning electron microscopy (FE-SEM) images of (a) complex 1 and (b) complex 2, showing differences in surface morphology. | ||
Fig. S25(a) and (b) illustrate the EDAX spectra recorded for complex 1 and complex 2, respectively. The spectrum of complex 1 confirms the presence of C, N, O, S, and V, with corresponding atomic percentages of 71.6, 1.2, 3.6, 18.9 and 2.4%. In comparison, the spectrum for complex 2 reveals the presence of C, N, O, S, Se and V, with atomic percentages of 63.7, 0.1, 4.5, 13.8, 11.4, and 5.8%, respectively.
Thermogravimetric analysis (TGA) under an N2 atmosphere indicates that both complexes (1 and 2) (Fig. S1) exhibit thermal stability up to approximately 250 °C, beyond which they undergo significant ligand decomposition in the temperature range 250–300 °C (loss of N-Dip groups). Initially, a gradual loss of solvent molecules (THF) is observed, attributed to the detachment of the THF molecules weakly coordinated to the lithium ion of the Li(THF)4 unit. A similar thermal decomposition pattern is observed for complex 2, although it exhibits a slightly higher initial loss of solvent than complex 1. At elevated temperatures (∼900 °C), a stable residual mass of LiV(S2C2H4)3 is obtained (SI). Both complexes are completely soluble in tetrahydrofuran (THF) solvent, nearly so in toluene/benzene, and partially soluble in n-hexane and pentane. Magnetic susceptibility measurement showed that they are diamagnetic in nature.
3.2 Cyclic voltammetry
Both complexes 1 and 2 exhibit redox activity, as revealed by their cyclic voltammetry results. Complex 1 shows two quasi-reversible reduction processes (SI) arising from the chelated dithiolene ligand. This indicates electrochemical changes L2− → L˙− (E1/2 = −1.62 V, ΔEp = 0.205 V) followed by L˙− → L0 (E1/2 = −0.83 V, ΔEp = 0.179 V), where (L2−) indicates the dianionic dithiolene ligand, followed by the formation of radical anion (L•−) and then a neutral dithiolene ligand (L0). Additionally, a reversible one-electron reduction (Fig. 3) at E1/2 = −0.375 V [(ΔEp = 0.087 V) vs. Fc+/Fc] is attributed to the reduction of the vanadium center from V5+ to V4+ (1 to 1˙−). In the case of complex 2, where the ligand is modified with Se functionalization as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se, a significant shift in redox potential is observed, with reversible one-electron oxidation (Fig. 4) at E1/2 = 0.095 V (ΔEp = 0.14 V) vs. Fc+/Fc (2 to 2˙−). The higher electron density on the Se atom of the Se-functionalized dithiolene ligand stabilizes V5+, making its reduction easier and shifting the redox potential towards the more positive side. This shift contributes further to its better pseudocapacitive nature and potential as an efficient electrochemical energy storage material. Complex 2 also exhibits analogous redox behavior to complex 1, but at relatively positive potentials with E1/2 = −0.789 V and −0.367 V, indicating that this ligand also follows a similar radical pathway (SI). To further understand the electronic influence of S and Se functionalization, DFT calculations were performed to analyze the spin density distribution, particularly on the metal center and ligand framework.
Se, a significant shift in redox potential is observed, with reversible one-electron oxidation (Fig. 4) at E1/2 = 0.095 V (ΔEp = 0.14 V) vs. Fc+/Fc (2 to 2˙−). The higher electron density on the Se atom of the Se-functionalized dithiolene ligand stabilizes V5+, making its reduction easier and shifting the redox potential towards the more positive side. This shift contributes further to its better pseudocapacitive nature and potential as an efficient electrochemical energy storage material. Complex 2 also exhibits analogous redox behavior to complex 1, but at relatively positive potentials with E1/2 = −0.789 V and −0.367 V, indicating that this ligand also follows a similar radical pathway (SI). To further understand the electronic influence of S and Se functionalization, DFT calculations were performed to analyze the spin density distribution, particularly on the metal center and ligand framework.
 | ||
| Fig. 3 Cyclic voltammogram of complex 1 in THF solution of 0.1 M [n-Bu4N]PF6 with RE: Ag, WE: GC, and CE: Pt. | ||
 | ||
| Fig. 4 Cyclic voltammogram of complex 2 in THF solution of 0.1 M [n-Bu4N]PF6 with RE: Ag, WE: GC, and CE: Pt. | ||
3.3 Electron paramagnetic resonance (EPR) spectroscopy
The redox-active diamagnetic complexes 1 and 2 were reduced with one equivalent of KC8 in THF to produce their doublet radical anions (1˙−, 2˙−). The X-band EPR spectrum of radical anion 1˙− displays a well-resolved eight-line hyperfine pattern, characteristic of V4+ (d1, S = 1/2, I = 7/2 for the 51V nucleus) species. Interestingly, an additional set of eight smaller lines is observed at the center of the main signal (SI, Fig. S10). This suggests the possible presence of a second magnetically distinct V4+ site, likely arising from an alternative conformer or coordination geometry. Similar spectral behavior has been reported in VO2+-doped lattices, where substitution at different lattice sites or orientations results in multiple EPR-active environments with distinct spectral signatures.26a,b The observed pattern supports the presence of at least two V4+ environments in the complex, potentially due to conformational flexibility or subtle differences in local ligand fields. In contrast to the delocalized mixed-valent system seen in [V2S4(nBu2dtc)4](BF4), which gives a 15-line (2nI + 1) pattern due to coupling with two equivalent 51V nuclei,14 the present spectrum supports localized electronic environments at vanadium centers. The EPR of complex 1˙− was also measured at 77 K in THF (SI, Fig. S11). The experimental EPR spectrum (blue line; Fig. 5) of 1˙− was fitted with the 51V (I = 7/2) nucleus, producing an isotropic hyperfine coupling constant of A(51V) = 305.0132 MHz. This value lies in the range of previously reported V4+ systems.26b,27 The slight difference can be attributed to variations in the ligand environment and electronic distribution around the vanadium center, which influence the spin density at the 51V-nucleus. A similar spectrum was obtained for radical anion 2˙− (see SI).The absence of the isopropyl CH septet in the 1H NMR spectrum of complex 1 can be attributed to conformational heterogeneity and paramagnetic broadening, consistent with EPR data indicating two distinct V environments. In contrast, complex 2, being structurally more rigid and symmetric with a single V center, allows clean NMR observation of the Dip (diisopropylphenyl) septet. These NMR spectra are consistent with similar ones reported in the literature.22
3.4 Geometry optimization and energy profile determination
The geometry optimizations of the complexes [Li(THF)4][V(SS-NHC = S)3] (1) and [Li(THF)4][V(SS-NHC = Se)3] (2) (SI) and their radical anions [(1′)˙− and (2′)˙−] were carried out at the BP86-D3(BJ)/Def2-TZVPP level of theory by replacing Dip groups with Me in singlet (1′–2′) and doublet electronic (1′˙−–2′˙−) states, both in the gaseous phase (1a′ and 2a′) and a solvent medium (THF) (1b′ and 2b′), with the help of DFT. NBO analysis of 1′ and 2′ in the doublet dianionic state [(1′)˙− and (2′)˙−] shows that the SOMO is primarily centered on the V ion with 94.5–94.7% spin density localized on the V-metal, confirmed by Mulliken spin density analysis plots (Fig. 6a and c) consistent with the SOMO distribution in the model complexes. The molecular orbital diagrams of vanadium tris–dithiolene complexes (1′ and 2′) are shown for both anionic singlet and dianionic doublet states (SI). In the singlet monoanion (1′/2′), all electrons are paired, while an additional electron is present in the doublet dianion state, resulting in a SOMO and its corresponding LUMO (V4+). Notably, the LUMO of the singlet monoanion corresponds to the SOMO of the doublet dianion, indicating that the extra electron occupies this d-orbital upon reduction.3.5 Spectroscopic analysis and stability
Various spectroscopic analyses provided valuable insights into the electronic properties and atomic behavior of complexes 1 and 2. Infrared, UV-vis-NIR, and EPR spectroscopy enabled a detailed understanding of the complexes, which was further correlated with their structure and electrochemical properties.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibration at 1425 cm−1, attributed to the imidazole ring of the ligand. A distinct band at 1628 cm−1 is assigned to C
C stretching vibration at 1425 cm−1, attributed to the imidazole ring of the ligand. A distinct band at 1628 cm−1 is assigned to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C or C
C or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching, indicative of an aromatic or conjugated system. In the fingerprint region, the absorption at 1180 cm−1 corresponds to C–N stretching, suggesting the presence of an amine functional group. The peak at 1040 cm−1 is attributed to C–S stretching, consistent with previous reports on dithiolene ligands.28 Raman spectroscopic analysis (Table S4, SI) further supports the coordination environment in the complexes (Fig. S25–S26). Strong bands observed at approximately 342, 349, 410, and 415 cm−1 are attributed to V–S stretching and deformation modes, which are consistent with reported values for vanadium–sulfur bonds.29a Additional weaker bands in the 596–770 cm−1 range correspond to C–S stretching vibrations,29b while a distinct band at 967 cm−1 is assigned to the C
N stretching, indicative of an aromatic or conjugated system. In the fingerprint region, the absorption at 1180 cm−1 corresponds to C–N stretching, suggesting the presence of an amine functional group. The peak at 1040 cm−1 is attributed to C–S stretching, consistent with previous reports on dithiolene ligands.28 Raman spectroscopic analysis (Table S4, SI) further supports the coordination environment in the complexes (Fig. S25–S26). Strong bands observed at approximately 342, 349, 410, and 415 cm−1 are attributed to V–S stretching and deformation modes, which are consistent with reported values for vanadium–sulfur bonds.29a Additional weaker bands in the 596–770 cm−1 range correspond to C–S stretching vibrations,29b while a distinct band at 967 cm−1 is assigned to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se stretching mode,29b in agreement with values reported in the literature. These vibrational features collectively confirm the coordination of vanadium with sulfur and support the incorporation of chalcogen-functionalized ligands in the complexes.
Se stretching mode,29b in agreement with values reported in the literature. These vibrational features collectively confirm the coordination of vanadium with sulfur and support the incorporation of chalcogen-functionalized ligands in the complexes.
TD-DFT computation (BP86/def2tzvpp, SMD, THF) of the model complexes 1b′ and 2b′ (b denotes in solvent) reveals absorption bands in an almost similar region to that of the experimental bands. However, a substantial difference between the experimental and theoretical λmax values is observed for both complexes in the NIR region. Such slight differences in the absorption bands are attributed to the structural differences between the modeled structures (1b′ and 2b′) and the experimental structures (1, 2).
In complex 1, the UV-vis-NIR spectrum exhibits a broad absorption band at 937 nm, attributed to a ligand-to-metal charge transfer (LMCT) transition. This assignment is corroborated by time-dependent density functional theory (TD-DFT) calculations of model complex 1b′, which predict a corresponding transition at 914 nm. This transition originates from the HOMO and HOMO’ of the ligand to the LUMO of the metal (Fig. 8), with the presence of two transitions contributing to the broadness of this band. Additionally, a second broad and less intense band appears at 648 nm, which is assigned to a formally forbidden d–d transition within the metal center, supported by TD-DFT computation showing a transition from HOMO − 1 to LUMO + 1. A significant absorption band at 416 nm is also observed, arising from an LMCT30 transition from the HOMO − 7 and HOMO − 7′ of the ligand to the LUMO of the metal. Similarly, complex 2 exhibits an absorption band at 964 nm, assigned to an LMCT transition from the HOMO and HOMO’ of the ligand to the LUMO of the metal, as supported by TD-DFT calculations. Another LMCT transition is observed at 660 nm, corresponding to a transition from HOMO’ to LUMO. Additionally, a band at 481 nm is attributed to a transition from HOMO’ to LUMO + 2. In both complexes, although the vanadium center is in the +5 oxidation state with no unpaired electrons, the ligand features electron density localized on the sulfur atoms, which are in close proximity to the metal center. This electron density likely facilitates partial electron donation to the metal, leading to the observed d–d transitions in both cases.
Upon reduction with KC8, for complex 1, where vanadium is in the +5 oxidation state, the UV-vis-NIR band intensity at 937 decreases, while the bands at 514 and 649 nm increase in intensity and shift to 531 and 623 nm, respectively, with an isosbestic point at 778 nm [SI, Fig. S13(a)]. These spectral changes indicate the reduction of the V(V) center to V(IV). Similarly, in complex 2, the UV-vis-NIR spectrum initially displays bands at 964, 660, and 537 nm. Upon reduction with KC8, new bands appear at 951 and 593 nm, with an isosbestic point at 825 nm [SI, Fig. S13(b)]. The gradual decrease in intensity and shift of the 964 nm band to 951 nm suggest the formation of a V(IV) center. Meanwhile, the bands at 660 and 537 nm increase in intensity, merging into a broader band at 593 nm, further confirming the reduction process. Additionally, the appearance of a new band at 406 nm suggests changes in ligand field effects upon reduction.31
3.6 Electrochemical analysis
 | ||
| Fig. 9 CV plots of (a) complex 1, (c) complex 2 and specific capacitance of (b) complex 1, (d) complex 2 with varying scan rates of 5–100 mV s−1. | ||
Complex 2 exhibited reversible oxidation and reduction peaks in CV plots, while complex 1 exhibited quasi-rectangular CV curves, owing to its surface capacitive and intercalation type of charge storage, indicating pseudocapacitive behavior for both electrodes.33 Complexes 1 and 2 achieved maximum specific capacitance 82.82 F g−1 (areal 82.82 mF cm−2) and 76.56 F g−1 (areal 76.56 mF cm−2) at a scan rate of 5 mV s−1, respectively [Fig. 9(b) and (d)] which are lower than those achieved by traditional materials, such as V2O5 (112 F g−1) or Ni-doped V2O5 (152 F g−1)34 in 1 M Na2SO4 or porous V2O5 (735 F g−1)19 in 2 M LiClO4. In comparison, a V2O5-MWCNT composite35 exhibited 629 F g−1 at 2 A g−1, whereas another vanadium-based complex, (NH4)2[H6(VIVO)3(O){O3P–C(OH)(CH2-4-C5NH4)-PO3}3]·26H2O35 demonstrated 140.8 F g−1 at 1 A g−1, and layered vanadium oxide, (NH4)2V10O25·8H2O (in 1 M (NH4)2SO4 with 5 mM NH4I added)36 achieved 187 F g−1 at 10 A g−1. Additionally, 3d–4d mixed-metal polyoxometalates with MWCNT composites37 showed 640 F g−1 at 5 mV s−1. However, it should be noted that these materials are pure inorganic oxides or composites in nature. While the dithiolene complexes (1 and 2) showed lower capacitance, their novelty and stability make them promising candidates for further performance tuning, especially through composite formation. The novelty of these dithiolene-based systems stands out, particularly given the absence of prior reports on such complexes in this field. At lower scan rates, the electrolyte ions (1 and 2) possess sufficient time to interact with the electrode material, resulting in an enhancement in specific capacitance.38 Electrochemical charge storage in electrode materials is governed primarily by two fundamental mechanisms: non-faradaic (capacitive) and faradaic (redox) processes. These mechanisms correspond to surface-controlled and diffusion-governed charge storage behaviors, respectively. The kinetic nature of these processes can be assessed using the power–law relationship:
| i = avb |
 | ||
| Fig. 10 Power–law plots of (a) complex 1, (c) complex 2 and corresponding b-values of (b) complex 1, (d) complex 2, obtained from the slopes of the power–law plots. | ||
For deeper insight into the storage mechanism, Trasatti analysis was used to qualitatively separate the total charge storage into surface capacitive (Qs) and diffusion-controlled (Qdα v−0.5) contributions. The total charge stored follows the following equations:
| Qt = Qs + Qd |
| Qt = Qs + kv−0.5 |
A plot of the total charge Qt against the inverse square root of the scan rate was used to extract Qs from the y-intercept for complexes 1 and 2 [(Fig. 11(a) and (c)]. The percentage contributions from each mechanism are illustrated in Fig. 11(b) and (d). While the surface capacitive contribution remains constant across scan rates, the diffusion-controlled mechanism becomes increasingly dominant at lower scan rates. At 100 mV s−1, complex 1 shows a diffusion-controlled contribution of approximately 26.73% to the total charge, which rises to 57.95% at 5 mV s−1 [Fig. 11(b)]. In contrast, complex 2 exhibits a predominantly diffusion-controlled mechanism with 96.99% at 100 mV s−1, which increases to 99.29% at 5 mV s−1 [Fig. 11(d)].
Fig. 12(a and c) present the galvanostatic charge–discharge (GCD) curves of 1 and 2 within the identical potential window to that used in cyclic voltammetry. Complex 2 exhibits redox nature in the GCD plots, with a sudden voltage drop due to the resistive nature, followed by a non-linear curve. Complex 1 exhibits a small voltage drop followed by a near–linear curve.33 Complex 2 achieved a maximum specific capacitance of 21.90 F g−1 (areal 21.90 mF cm−2) at 0.6 mA cm−2, retaining 8.78 F g−1 (areal 8.78 mF cm−2) even at a high current of 1.4 mA cm−2 [Fig. 12(d)]. While its S-analogue, complex 1, exhibited the highest specific capacitance of 38.02 Fg−1 (areal 38.02 mF cm−2) at 0.2 mA cm−2, reaching 21.35 F g−1 (areal 21.35 mF cm−2) at a high current of 1.0 mA cm−2 [Fig. 12(b)].
 | ||
| Fig. 12 Galvanostatic charge–discharge plots of (a) complex 1 and (c) complex 2 with different current densities. Specific capacitance of (b) complex 1 and (d) complex 2vs. current density. | ||
Fig. 13 illustrates the Ragone plot comparing the energy and power performance of electrodes based on complex 1 and complex 2. Specifically, complex 1 demonstrates a maximum energy density of 0.69 Wh kg−1 at a corresponding power density of 900 W kg−1, and achieves the highest power density of 4500 W kg−1 while maintaining an energy density of 0.59 Wh kg−1. Complex 2 shows a peak energy density of 2.19 Wh kg−1 at a power density of 255 W kg−1, and reaches its highest power density of 595 W kg−1 when the energy density is 0.88 Wh kg−1.
Electrochemical impedance spectroscopy (EIS) measurements of 2 and 1 electrodes were carried out in the frequency range of 10 kHz to 0.1 Hz. Fig. 14(a) and (b) represent the Nyquist plot (real vs. imaginary impedance) of 1 and 2 electrodes fitted with an equivalent circuit. The solution resistance (RS)—arising from interactions at the electrode–electrolyte interface, ionic resistance, and intrinsic substrate resistance—was determined to be 6.97 and 2.00 Ω cm2 for the 1 and 2 electrodes, respectively. The charge transfer resistance (RCT), indicative of reversible faradaic redox activity, was found to be 2.91 and 0.52 Ω cm2 for 1 and 2, respectively. The low values of RS and RCT suggest effective electrode–electrolyte interactions.39 The mass transport limitation at the electrode–electrolyte interface was observed in the low-frequency linear region.40 The constant phase element (CPE) impedance was defined by the equation ZCPE = T(jω)−n, where T and n are frequency-independent empirical constants, and ω represents the angular frequency.
The fitted circuit [Fig. 14(a) and (b)] revealed n-values of 0.8 and 0.5 for 1 and 2, respectively, further confirming their pseudocapacitive nature.41 The equivalent circuit also included double-layer capacitance (CDL). Both electrodes also include leakage resistance (RL), which resists the self-discharge of the electrode material. The Warburg element (W) in 1 indicates the diffusion of electrolyte ions into the electrode material. The Bode plot [SI, Fig. S24(a) and (b)] illustrates the phase angle vs. frequency, which shows maximum phase angles of −65.49° for 1 and 2, respectively, indicating pseudocapacitive charge storage. The characteristic frequency at −45° was used to distinguish between resistive and capacitive behavior. The time constants (τ0 = 1/f0) for 1 and 2 were calculated as 0.79 s and 50 ms, respectively.41
Lastly, stability tests were performed on both electrodes over 2000 CV cycles at 100 mV s−1 to assess their long-term applicability. The capacitive retention of complexes 1 and 2 after 2000 cycles was 63.13 and 104.39%, respectively [Fig. 14(c) and (d)]. The electrode made of 2 showed excellent capacitive retention even after 2000 CV cycles, surpassing that of many reported systems, including a study on 3d–4d mixed-metal polyoxometalates of vanadium, chromium, and manganese combined with MWCNTs achieving 94% retention after 2000 cycles,37 while the capacitive retention of the electrode made from 1 was reduced significantly, which might be due to material degradation. Recently, several research groups have been exploring inorganic–organic vanadium-based complexes for supercapacitor applications. Some notable examples include a vanadium-based complex35 showing 79.23% retention after 4500 cycles, and a layered vanadium oxide system36 achieving 74.86% retention after 5000 cycles. Porous V2O5 intermixed flakes19 were synthesized by B. Pandit et al. and applied in supercapacitor applications, showing 71% retention over 1000 cycles, while a V2O5-MWCNT composite5 achieved 93% retention after 4000 cycles. Compared to these, the vanadium-dithiolene complex 2 in the present work stands out, with higher (104.39%) retention after 2000 cycles, highlighting its exceptional stability and potential as a novel supercapacitor material. This self-improving behavior suggests the potential for electrochemical activation or structural reorganization, contributing to enhanced long-term performance. Additionally, impedance studies confirm low charge transfer resistance, supporting efficient ion diffusion and redox activity—an analysis often overlooked in comparable literature. While the current capacitance values are modest, the stability and novelty of the complexes provide a strong foundation for further optimization. Future efforts could focus on incorporating conductive carbon materials, such as MWCNTs or graphene, to improve conductivity and specific capacitance. Furthermore, synthesizing mixed-metal dithiolene variants, inspired by the success of multi-metal polyoxometalates, could leverage multiple oxidation states to enhance redox activity and overall performance. This work, therefore, not only introduces a new class of materials for supercapacitor applications but also paves the way for innovative hybrid systems with improved electrochemical properties.
To summarize and directly compare the electrochemical performance of the two complexes, the key parameters obtained from cyclic voltammetry (CV), galvanostatic charge–discharge (GCD) measurements, and stability studies are presented in Table 2.
| Parameters | Complex 1 | Complex 2 |
|---|---|---|
| Electrolyte | 1 M Na2SO4 | 1 M LiClO4 |
| Specific capacitance (CV) F g−1 | 82.82 @ 5 mV s−1 | 76.56 @ 5 mV s−1 |
| Specific capacitance (GCD) F g−1 | 38.02 @ 0.2 mA cm−2 | 21.90 @ 0.6 mA cm−2 |
| Potential window (V) | 0.8 | 0.85 |
| Stability @ 2000 cycles | 63.13% | 104.39% |
4. Conclusion
In summary, two photo-redox-active anionic vanadium(V)-dithiolene complexes [(THF)4Li][V(V)(SS–NHC = E)3] [E = S (1) and Se (2)] were synthesized, characterized by different techniques, and their electrochemical energy storage potential was further investigated, demonstrating impressive pseudocapacitive behavior. These mono-anionic complexes were shown to be reduced to their dianionic doublets [1/2 to (1/2)˙−], which were characterized by EPR measurement in solution. The electrochemical behavior of complex 2 changes significantly due to the presence of three comparatively more polar C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Se groups. Electrochemical studies, including cyclic voltammetry, galvanostatic charge–discharge, and electrochemical impedance spectroscopy, confirmed their high charge storage capacity, excellent redox activity, and good cycling stability. The capacitance was found to depend strongly on scan rate and current density, indicative of a diffusion-limited charge storage mechanism characteristic of pseudocapacitors. The working redox potential for displaying supercapacitive properties was tuned by functionalization of an N-hetero cyclic carbene (NHC)-based dithiolene ligand by Se at the C2 position of NHC, leading to the better performance of complex 2 (E = Se) than 1 (E = S). Additionally, electrolyte composition significantly influenced electrochemical performance, emphasizing the crucial role of ion–electrode interactions. Among the tested electrolytes, 1 M Na2SO4 and LiClO4 provided the best electrochemical performance for complexes 1 and 2, respectively, with specific capacitances of 82.82 and 76.56 F g−1 at a scan rate of 5 mV s−1. Notably, complex 2 exhibited excellent cycling stability with a capacitive retention of 104.39% after 2000 cycles, while complex 1 showed some degradation over prolonged cycling. Impedance studies further confirmed low charge transfer resistance, supporting efficient ion diffusion and redox activity. A plethora of metal complexes of different dithiolene ligands has been known from the mid-1960s until now.14–16,42,43 However, this unprecedented report has shown vanadium-dithiolene-based molecular materials with prominent pseudo-supercapacitive properties storing energy for the rapid recharge of electronic devices. This finding not only establishes photo-redox-active vanadium-dithiolene complexes as promising candidates for next-generation supercapacitors but also underscores the potential of redox non-innocent dithiolene ligands. Our studies showed that structural modifications can further enhance their electrochemical performance and stability, which will be crucial in the design and synthesis of advanced functional materials for energy storage in the future.
Se groups. Electrochemical studies, including cyclic voltammetry, galvanostatic charge–discharge, and electrochemical impedance spectroscopy, confirmed their high charge storage capacity, excellent redox activity, and good cycling stability. The capacitance was found to depend strongly on scan rate and current density, indicative of a diffusion-limited charge storage mechanism characteristic of pseudocapacitors. The working redox potential for displaying supercapacitive properties was tuned by functionalization of an N-hetero cyclic carbene (NHC)-based dithiolene ligand by Se at the C2 position of NHC, leading to the better performance of complex 2 (E = Se) than 1 (E = S). Additionally, electrolyte composition significantly influenced electrochemical performance, emphasizing the crucial role of ion–electrode interactions. Among the tested electrolytes, 1 M Na2SO4 and LiClO4 provided the best electrochemical performance for complexes 1 and 2, respectively, with specific capacitances of 82.82 and 76.56 F g−1 at a scan rate of 5 mV s−1. Notably, complex 2 exhibited excellent cycling stability with a capacitive retention of 104.39% after 2000 cycles, while complex 1 showed some degradation over prolonged cycling. Impedance studies further confirmed low charge transfer resistance, supporting efficient ion diffusion and redox activity. A plethora of metal complexes of different dithiolene ligands has been known from the mid-1960s until now.14–16,42,43 However, this unprecedented report has shown vanadium-dithiolene-based molecular materials with prominent pseudo-supercapacitive properties storing energy for the rapid recharge of electronic devices. This finding not only establishes photo-redox-active vanadium-dithiolene complexes as promising candidates for next-generation supercapacitors but also underscores the potential of redox non-innocent dithiolene ligands. Our studies showed that structural modifications can further enhance their electrochemical performance and stability, which will be crucial in the design and synthesis of advanced functional materials for energy storage in the future.
Author contributions
Sangita Mondal: synthesis, primary characterization and analysis, writing – original draft. T Kedara Shivasharma: methodology, investigation, analysis, writing. Sujit Das: methodology, investigation. Mayur Thosare: analysis, writing. Babasaheb R. Sankapal: conceptualization, writing – review & editing, supervision, and funding acquisition. Kartik Chandra Mondal: conceptualization, writing – review & editing, supervision, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2424329 and 2427545 contain the supplementary crystallographic data for this paper.44a,bThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed synthetic procedures, spectroscopic and analytical characterization data (TGA, IR, UV-vis, EPR, and NMR); theoretical calculation details; and additional electrochemical measurements, including cyclic voltammetry and capacitance analysis. See DOI: https://doi.org/10.1039/d5ta05686a.
Acknowledgements
S.M. and C.L.M. thank DST-INSPIRE for providing (DST/INSPIRE/03/2021/000816 and SP23241803CYDSTX008694) fellowship. S. D. thanks CSIR for fellowship. K. C. M. thanks SERB, New Delhi (ECR/2016/000890). T.K.S is thankful to DST (DST/INSPIRE/03/2022/000141) for providing INSPIRE fellowship. B.R.S gratefully acknowledge DST-FIST (SR/FST/PSI/2017/5 (C)) projects. K. C. M. IIT Madras for research facilities, HPCE Aqua Cluster of IIT Madras for theoretical calculation facilities, and IISER Tirupati for single-crystal XRD facilities.References
- G. Wang, L. Zhang and J. Zhang, Chem. Soc. Rev., 2012, 41, 797–828 Search PubMed
.
- M. Cui and X. Meng, Nanoscale Adv., 2020, 2, 5516–5528 Search PubMed
.
- R. Liu, A. Zhou, X. Zhang, J. Mu, H. Che, Y. Wang, T.-T. Wang, Z. Zhang and Z. Kou, Chem. Eng. J., 2021, 412, 128611 Search PubMed
.
-
K. S. Ngai, Encyclopedia of Energy Storage, 2022, vol. 4, pp. 341–350 Search PubMed
.
- B. Pandit, D. P. Dubal, P. Gómez-Romero, B. B. Kale and B. R. Sankapal, Sci. Rep., 2017, 7, 1–12 Search PubMed
.
- B. Pandit, S. S. Karade and B. R. Sankapal, ACS Appl. Mater. Interfaces, 2017, 9, 44880–44891 CrossRef CAS PubMed
.
- P. J. Cappillino, H. D. Pratt III, N. S. Hudak, N. C. Tomson, T. M. Anderson and M. R. Anstey, Adv. Energy Mater., 2014, 4, 1300566 Search PubMed
.
- G. Periyasamy, N. A. Burton, I. H. Hillier, M. A. Vincent, H. Disley, J. McMaster and C. D. Garner, Faraday Discuss., 2007, 135, 469–488 RSC
.
- A. Davison, N. Edelstein, R. H. Holm and A. H. Maki, J. Am. Chem. Soc., 1964, 86, 2799–2805 Search PubMed
.
- J. H. Waters, R. Williams, H. B. Gray, G. N. Schrauzer and H. W. Finck, J. Am. Chem. Soc., 1964, 86, 4198–4199 Search PubMed
.
- J. H. Welch, R. D. Bereman and P. Singh, Inorg. Chem., 1988, 27, 2862–2868 Search PubMed
.
- R. Eisenberg and H. B. Gray, Inorg. Chem., 1967, 6, 1844–1849 CrossRef CAS
.
- S. Sproules, T. Weyhermüller, S. DeBeer and K. Wieghardt, Inorg. Chem., 2010, 49, 5241–5261 Search PubMed
.
- I. S. Fomenko, A. L. Gushchin, V. A. Nadolinny, N. N. Efimov, Y. A. Laricheva and M. N. Sokolov, Eur. J. Inorg. Chem., 2018, 2018, 2965–2971 Search PubMed
.
- G. Matsubayashi, K. Akiba and T. Tanaka, Inorg. Chem., 1988, 27, 4744–4749 Search PubMed
.
- M. Kondo, S. Minakoshi, K. Iwata, T. Shimizu, H. Matsuzaka, N. Kamigata and S. Kitagawa, Chem. Lett., 1996, 25, 489–490 Search PubMed
.
- R. W. Hogue, C. G. Armstrong and K. E. Toghill, ChemSusChem, 2019, 12, 4506–4515 Search PubMed
.
- B. Pandit, L. K. Bommineedi and B. R. Sankapal, J. Energy Chem., 2018, 31, 79–88 CrossRef
.
- B. Pandit, D. P. Dubal and B. R. Sankapal, Electrochim. Acta, 2017, 242, 382–389 CrossRef CAS
.
- M. Czagany, S. Hompoth, A. K. Keshri, N. Pandit, I. Galambos, Z. Gacsi and P. Baumli, Materials, 2024, 17, 702 Search PubMed
.
-
(a) Y. Yan, B. Li, W. Guo, H. Pang and H. Xue, J. Power Sources, 2016, 329, 148–169 CrossRef CAS
; (b) A. G. Temam, A. Alshoaibi, S. A. Getaneh, C. Awara, A. C. Nwanya, P. M. Ejikeme and F. I. Ezema, Curr. Opin. Electrochem., 2023, 38, 101239 CrossRef CAS
; (c) M. S. Shruti, S. Khilari, E. J. J. Samuel, H. Han and A. K. Nayak, J. Energy Storage, 2023, 63, 107006 CrossRef
; (d) H. Qin, S. Liang, L. Chen, Y. Li, Z. Luo and S. Chen, Sustain. Energy Fuels, 2020, 4, 4902–4933 Search PubMed
; (e) J. Zhu, X. Chen, A. Q. Thang, F. Li, D. Chen, H. Geng, X. Rui and Q. Yan, SmartMat, 2022, 3, 384–416 Search PubMed
; (f) H. MP and A. C. Bose, Energy Fuels, 2023, 37, 10799–10826 CrossRef CAS
.
- Y. Wang, H. P. Hickox, Y. Xie, P. Wei, S. A. Blair, M. K. Johnson, H. F. Schaefer III and G. H. Robinson, J. Am. Chem. Soc., 2017, 139, 6859–6862 Search PubMed
.
- See SI for the procedure.
- Y. Wang, Y. Xie, P. Wei, H. F. Schaefer III and G. H. Robinson, Dalton Trans., 2019, 48, 3543–3546 Search PubMed
.
- P. M. Tran, Y. Wang, B. Dzikovski, M. E. Lahm, Y. Xie, P. Wei, V. V. Klepov, H. F. Schaefer III and G. H. Robinson, J. Am. Chem. Soc., 2024, 146, 16340–16347 CrossRef CAS PubMed
.
-
(a) E. Bozkurt, B. Karabulut, İ. Kartal and Y. S. Bozkurt, Chem. Phys. Lett., 2009, 477, 65–69 Search PubMed
; (b) P. Singh, S. Schlittenhardt, D. Thakre, S. K. Kushvaha, S. Kumar, H. S. Karnamkkott, M. Ruben, M. Ibrahim, A. Banerjee and K. C. Mondal, Cryst. Growth Des., 2022, 22, 5666–5679 Search PubMed
.
- L. Tesi, E. Lucaccini, I. Cimatti, M. Perfetti, M. Mannini, M. Atzori, E. Morra, M. Chiesa, A. Caneschi, L. Sorace and R. Sessoli, Chem. Sci., 2015, 7, 2074–2083 Search PubMed
.
- R. A. Nyquist and W. J. Potts, Spectrochim. Acta, 1959, 15, 514 Search PubMed
.
-
(a) R. J. H. Clark and P. C. Turtle, J. Chem. Soc., Dalton Trans., 1978, 1714–1721 RSC
; (b) G. Socrates, Infrared and Raman Characteristic Group Frequencies: Tables and Charts, John Wiley & Sons, 2004 Search PubMed
.
- R. Borah, S. Lahkar, N. Deori and S. Brahma, RSC Adv., 2022, 12, 13740–13748 RSC
.
- S. Menati, H. A. Rudbari, M. Khorshidifard and F. Jalilian, J. Mol. Struct., 2015, 1103, 94–102 Search PubMed
.
- T. K. Shivasharma, N. Upadhyay, T. B. Deshmukh and B. R. Sankapal, ACS Omega, 2023, 8, 37685–37719 CrossRef CAS PubMed
.
- Y. Jiang and J. Liu, E. Environ. Mag., 2019, 2, 30–37 Search PubMed
.
- V. U. Shankar, D. Govindarajan, P. Christuraj, M. J. Salethraj, F. J. Johanson and M. D. Raja, Mater. Today Proc., 2022, 50, 2675–2678 Search PubMed
.
- D. Thakre, A. Anandan Vannathan, A. Banerjee and S. S. Mal, Ionics, 2022, 28, 4001–4010 Search PubMed
.
- X. Wang, H. Mei, P. Chang, M. Zhang, T. Hu, L. Cheng and L. Zhang, Appl. Surf. Sci., 2022, 605, 154688 CrossRef CAS
.
- K. S. Morla, D. Thakre, T. B. Deshmukh, S. Malo, S. Ahamed, R. K. Aparna, S. Sahoo, D. Sarma, K. C. Mondal, B. R. Sankapal and A. Banerjee, J. Mate. Chem. A, 2024, 12, 22013–22029 Search PubMed
.
- T. K. Shivasharma, A. C. Mendhe, R. Sahu and B. R. Sankapal, J. Colloid Interface Sci., 2024, 676, 739–754 CrossRef PubMed
.
- A. Ch. Lazanas and M. I. Prodromidis, ACS Meas. Sci. Au, 2023, 3, 162–193 Search PubMed
.
- K. Sheng, Y. Sun, C. Li, W. Yuan and G. Shi, Sci. Rep., 2012, 2, 247 Search PubMed
.
- T. K. Shivasharma, L. K. Bommineedi and B. R. Sankapal, Inorg. Chem. Commun., 2021, 135, 109083 Search PubMed
.
-
Dithiolene Chemistry: Synthesis, Properties, and Applications, ed. E. I. Stiefel, Progress in Inorganic Chemistry, John Wiley & Sons, Inc., 2003, vol. 52, DOI:10.1002/0471471933
.
-
(a) A. L. Balch, I. G. Dance and R. H. Holm, J. Am. Chem. Soc., 1968, 90, 1139 CrossRef CAS
; (b) I. G. Dance and T. R. Miller, Inorg. Chem., 1974, 13, 525 Search PubMed
; (c) G. N. Schrauzer, V. P. Mayweg, H. W. Finck and W. Heinrich, J. Am. Chem. Soc., 1966, 88, 4604 Search PubMed
; (d) D. Sellmann, M. Geek, F. Knoch, G. Ritter and J. Dengler, J. Am. Chem. Soc., 1991, 113, 3819 Search PubMed
; (e) K. Ray, A. Begum, T. Weyhermüller, S. Piligkos, J. van Slageren, F. Neese and K. Wieghardt, J. Am. Chem. Soc., 2005, 127, 4403–4415 CrossRef CAS
; (f) K. Ray, E. Bill, T. Weyhermüller and K. Wieghardt, J. Am. Chem. Soc., 2005, 127, 5641–5654 Search PubMed
; (g) K. Ray, T. Weyhermüller, F. Neese and K. Wieghardt, Inorg. Chem., 2005, 44, 5345–5360 CrossRef CAS PubMed
; (h) A. K. Patra, E. Bill, E. Bothe, K. Chlopek, F. Neese, T. Weyhermüller, K. Stobie, M. D. Ward, J. A. McCleverty and K. Wieghardt, Inorg. Chem., 2006, 45, 7877–7890 CrossRef CAS PubMed
; (i) A. K. Patra, E. Bill, T. Weyhermüller, K. Stobie, Z. Bell, M. D. Ward, J. A. McCleverty and K. Wieghardt, Inorg. Chem., 2006, 45, 6541–6548 CrossRef CAS PubMed
; (j) R. Yu, K. Arumugam, A. Manepalli, Y. Tran, R. Schmehl, H. Jacobsen and J. P. Donahue, Inorg. Chem., 2007, 46, 5131–5133 CrossRef CAS PubMed
; (k) S. R. Boone, G. H. Purser, H. R. Chang, M. D. Lowery, D. N. Hendrickson and C. G. Pierpont, J. Am. Chem. Soc., 1989, 111, 2292–2299 CrossRef CAS
.
-
(a)
CCDC 2424329: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2mcq5x
; (b) CCDC 2427545: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2mh1x3
.
| This journal is © The Royal Society of Chemistry 2026 |