Realizing a thermoelectric conversion efficiency of 10% with long-term stability in a kesterite Cu2ZnSn(S1−xSex)4 single-leg device
Akira
Nagaoka
 *ab,
Shoma
Miura
c,
Keita
Nomoto
*ab,
Shoma
Miura
c,
Keita
Nomoto
 d,
Kangwei
Chen
d,
Naoki
Sato
d,
Kangwei
Chen
d,
Naoki
Sato
 e,
Kenji
Yoshino
e,
Kenji
Yoshino
 ab and
Kensuke
Nishioka
ab and
Kensuke
Nishioka
 ab
ab
aElectrical and Electronic System Program, University of Miyazaki, Miyazaki 889-2192, Japan. E-mail: nagaoka.akira.m0@miyazaki-u.ac.jp
bGX Research Center, University of Miyazaki, Miyazaki 889-2192, Japan
cGraduate School of Engineering, University of Miyazaki, Miyazaki 889-2192, Japan
dSchool of Aerospace, Mechanical and Mechatronic Engineering, Australian Centre for Microscopy and Microanalysis, The University of Sydney, Sydney, 2006, Australia
eResearch Center for Materials Nanoarchitectonics (MANA), National Institute for Materials Science (NIMS), Ibaraki 305-0044, Japan
First published on 15th January 2026
Abstract
The application of thermoelectric (TE) technology, contributing to carbon neutrality by the recovery of global waste heat, is hindered by low conversion efficiencies and high costs, demonstrating a strong demand for high-performance TE materials with environmentally friendly characteristics. Multinary Cu2ZnSnS4 (CZTS)-based materials have attracted considerable attention for TE power generation owing to their cost-effectiveness and earth-abundant features. However, their dimensionless figure of merit (ZT), which is the critical measure of TE performance, is relatively lower than that of other TE-material-based systems. Herein, we report the discovery of p-type Cu2ZnSn(S1−xSex)4 (CZTSSe) single crystals with a record-high ZT of 1.9 at 800 K and high TE conversion efficiency of ∼10% at a temperature difference of 473 K with long-term stability over 1000 h. The exceptional performance originates from a high power factor and intrinsically low thermal conductivity owing to the pseudo-cubic structure, alkali element doping, and short-range lattice distortion. This study demonstrates the potential of CZTS-based TE materials for intermediate-temperature TE harvesting.
Introduction
Carbon neutrality, that is, the realization of net-zero CO2 emissions, is attracting global attention owing to the depletion of non-renewable energy sources driven by increasing energy consumption and the resultant environmental deterioration. The generation of clean energy is critical to achieving carbon neutrality by the mid-21st century. Thermoelectric (TE) technology shows promise in enabling reversible conversion between thermal energy, such as industrial waste heat and automobile exhaust gas, and electricity. The total amount of global waste heat is roughly estimated at the order of ‘exajoules’,1,2 which is comparable to the energy output of several hundreds of nuclear power plants. In particular, high-temperature waste heat in the range of >500 °C is emitted from the iron and steel industry. This energy- and emission-intensive sector accounts for ∼8% of annual global anthropogenic CO2 emissions (>2800 Mt),3 to whose decarbonization the recovery of high-temperature waste heat contributes.TE applications with high conversion efficiency have been developed for the recovery of waste heat over various temperature ranges, which are based on Te-related compounds. High-performance and cost-effective TE materials are required for industrial applications toward carbon neutrality. However, state-of-the-art TE materials typically comprise expensive, scarce, or toxic elements, such as Pb, Bi, Sb, Ge, and Te.4 Recently, sulfide and selenide TE materials have been receiving much attention because the earth abundances of S and Se (420 ppm and 0.05 ppm, respectively) are higher than that of Te (0.001 ppm).5 A comparison of the costs of chalcogenides (Fig. 1A) indicates that the cost of sulfide materials is an order of magnitude lower than that of Te element-based materials; however, their performance is lower than that of Te element-based state-of-the-art chalcogenides.6 Te-free technologies must be developed for large-scale fabrication of TE devices.
The high TE conversion efficiency is quantified using the dimensionless figure of merit ZT = S2σT/κ, where S, σ, T, and κ are the Seebeck coefficient, electrical conductivity, temperature, and thermal conductivity, respectively. Over the past few decades, great efforts have been devoted to improving the ZT value for various TE materials such as PbTe, GeTe, Bi2Te3, filled skutterudites, and half-Heuslers.4 Current proven strategies are typified by the concepts of phonon-glass electron-crystals7 and phonon-liquid electron-crystals,8 electronic band engineering for improvement of the Seebeck coefficient,9–12 and nanostructure modification to reduce thermal conductivity or enhance electrical properties.13–18 The pseudo-cubic structure has great potential to maximize the ZT value, as it leads to low crystal-field splitting and, consequently, a high power factor (PF) owing to valley degeneracy at the valence band maximum.19 We demonstrated a high ZT value of 1.6 realized in a totally environmentally benign p-type kesterite Cu2ZnSnS4 (CZTS) single crystal based on a pseudo-cubic strategy.20,21
Doping techniques have contributed to the development of various materials including high performance TEs and photovoltaics (PVs). In particular, it has been recognized that the incorporation of alkali elements (alkali effect) is crucial to obtaining high efficiency for improving the electrical properties of PV devices based on chalcogenide materials such as CZTS and Cu(In,Ga)Se2.22,23 In the field of TE materials, the higher Na concentration leads to band convergence of valence bands in PbTe suppressing bipolar conduction and increasing the Seebeck coefficient.24 Furthermore, alkali element doping in CZTS leads to high TE performance owing to controlling point defects and enhancement in the electronic density of states (DOS), respectively.21,25 Although the preliminary results of the alkali effect indicate a positive influence on the properties of PV and TE materials, the details such as mechanism and local structure are still unknown.
Considering the remarkable potential of thermal energy utilization, research on practical aspects of TE devices/modules is attracting greater attention than traditional studies on achieving higher ZT. Developing a TE device is highly challenging and thus, from an industrial viewpoint, progress in this area remains slow because it is difficult to optimize device structure. A practical TE module consists of two or more p- and n-type TE legs that are electrically connected by a metal electrode in series. Currently, practical TE device application requires overcoming some issues such as TE material coupling, contact resistance between the TE material and electrodes, mechanical properties including thermal expansion, and long-term stability at operating temperatures. Considering current TE device progress, the development of the single-leg device is useful to evaluate the potential output power and conversion efficiency leading to optimal device structure for the module. However, there are few practical TE single-leg devices even though TE materials with a high ZT value are applied. The p-type GeTe alloy-based TE single-leg with a high ZT of 2.5 indicates a conversion efficiency of 14% at a temperature gradient of 440 K.26 The n-type Mg3Sb1.5Bi0.5-based TE single-leg with a high ZT of 1.5 indicates the conversion efficiency of 13% at a temperature gradient of 495 K.27 In addition, several TE single-legs demonstrate the conversion efficiency of 3–10% at a temperature gradient (ΔT) range of 164–600 K based on p-type CZTS with ZT of 1.6,28 p-type SnS with ZT of 1.6,5 p-type SnSe with ZT of 2.4,29 p-type AgSbTe2 with ZT of 1.77,30 p-type α-MgAgSb with ZT of 1.2,31 p-type half-Heuslers ZrCoBi with ZT of 1.42,32 p-type PbSe with ZT of 1.1,33 p-type PbTe with ZT of 2.2,34 p-type Bi0.4Sb1.6Te3 with ZT of 1.35,35 and n-type PbTe with ZT of 1.3.34 These conversion efficiencies lag behind their theoretical equivalents because of the relatively large interfacial resistance of the TE material–electrode interface.4,27 Thus, interface properties significantly affect the application of newly fabricated TE materials.
One further step that has to be taken to realize efficient utilization of waste heat sources in future carbon-neutral society is the fabrication of high efficiency TE devices with environmentally friendly characteristics, using various material strategies. In this study, a noteworthy TE performance comparable to that of state-of-the-art TE materials was achieved by revealing the alkali effect and anion substitution to simultaneously optimize the electrical and TE properties in CZTS. A record-high ZT of ∼1.9 at 800 K was achieved using p-type K-doped Cu2ZnSn(S1−xSex)4 (CZTSSe) single crystals. The CZTSSe/Mo(SSe)2-barrier-layer/Mo-contact single-leg TE device shown in Fig. 1B exhibited decent ohmic properties and chemical stability between the TE material and Cu electrode, enabling high energy-conversion efficiency and long-term stability. A maximum conversion efficiency (ηmax) of 10.4% and output power (Pmax) of 160 mW at a temperature difference of 473 K was achieved, demonstrating the remarkable potential of CZTSSe for low-cost TE power generation (Fig. 1C). The reduction in efficiency and output power within 10% was found after the thermal aging test of the CZTSSe single-leg over 1000 h, demonstrating excellent long-term stability (Fig. 1D).
Results and discussion
Fundamental properties of CZTSSe as a TE material
In the present study, 5% Cu-deficient and 0.1 mol% K-doped samples were fabricated with different anion compositions (x), including non-doped Cu1.9ZnSn(S1−xSex)4 (x = 0.2, 0.5, and 0.8 for samples 1, 2, and 3, respectively) and K-doped Cu1.9ZnSn(S1−xSex)4 (x = 0.5 and 1.0 for samples 4 and 5, respectively), using the Sn-solvent-based traveling heater method (THM).36 The single crystals were cut in the transverse (⊥) and longitudinal (∥) directions (perpendicular and parallel to the c-axis, respectively) for TE measurements. Their structural and compositional properties are shown in Fig. S1–S3 and Tables S1, S2. High-quality p-type CZTSSe single crystals with a homogeneous composition were obtained. The pseudo-cubic approach associated with the Cu/Zn disordered structure in CZTSSe is described in Fig. S4.The degrees of order in kesterite CZTSSe samples must be determined for a comprehensive study because the order–disorder transition likely affects TE properties, the details of which are discussed in the SI. Consequently, the order–disorder transition was found to not significantly affect the TE properties of CZTSSe, and all investigated samples had a disordered structure in this study.
TE properties
The temperature-dependent electrical and TE properties of the various compositional CZTSSe single crystals from 300 to 800 K were examined (Fig. 2). Here, σ systematically increased with increasing Se content over the entire temperature range (Fig. 2A). The larger size of the selenide anions than that of the sulfide anions led to a higher degree of covalency and, consequently, a lower band effective mass and higher carrier mobility, resulting in enhanced σ values with increasing Se content. Therefore, the Se alloying and K doping of sample 5 performed to enhance its electrical properties led to a metallic behavior. Results related to the Hall-effect measurements and electrical conduction parameters are listed in Table S3. The conductivity data of samples 1–4 were quantitatively described based on typical thermal activation: where σB, EA, and kbare are the pre-factor, band-conduction-associated activation energy, and Boltzmann constant, respectively.37 As an intrinsic defect common to Cu-based kesterites, the acceptor level introduced by VCu is always shallow and dominant under the Cu-deficient conditions employed in this study. The EA values of CZTSSe (23–75 meV; Table S3) are consistent with the theoretically predicted shallow VCu acceptor level present 20–30 meV above the valence band maximum.38 The Cu/Zn disorder readily leads to a high Cu concentration on the Zn antisite (CuZn) acceptor defect in CZTSSe. The acceptor defect level corresponding to CZTSe (110 meV) is relatively lower than that of CZTS (150 meV) because of the weaker Cu–Se p-d hybridization and its higher valence band,38 which corroborates the enhancement in hole concentration and electrical conduction with increasing Se content at a low thermal activation energy.
where σB, EA, and kbare are the pre-factor, band-conduction-associated activation energy, and Boltzmann constant, respectively.37 As an intrinsic defect common to Cu-based kesterites, the acceptor level introduced by VCu is always shallow and dominant under the Cu-deficient conditions employed in this study. The EA values of CZTSSe (23–75 meV; Table S3) are consistent with the theoretically predicted shallow VCu acceptor level present 20–30 meV above the valence band maximum.38 The Cu/Zn disorder readily leads to a high Cu concentration on the Zn antisite (CuZn) acceptor defect in CZTSSe. The acceptor defect level corresponding to CZTSe (110 meV) is relatively lower than that of CZTS (150 meV) because of the weaker Cu–Se p-d hybridization and its higher valence band,38 which corroborates the enhancement in hole concentration and electrical conduction with increasing Se content at a low thermal activation energy.
The S value monotonically increased with increasing temperature because more charge carriers were thermally activated to high-energy states originating from the pseudo-cubic structure (Fig. 2B); this tendency is also observed in TE kesterites.39–42 The anisotropy of S was caused by the anisotropy of the effective hole mass, indicating higher S values along the a-axis than those along the c-axis.21S decreased with increasing Se content and K doping because of enhancements in the hole concentration. The combination of high σ and S values led to a PF value of 2.2 × 10−3 W mK−2 at 800 K for sample 4 along the a-axis (Fig. 2C). A comparison of the PF values of various TE chalcogenides at 300–800 K (Fig. 2D) indicated that the aforementioned PF exceeded that of state-of-the-art chalcogenides, such as SnSe (ZT = 2.6 (ref. 8)), Cu2Se (ZT = 2.6 (ref. 7)), PbTe (ZT = 2.2 (ref. 15)), AgSbTe2 (ZT = 1.77 (ref. 30)), PbS (ZT = 1.7 (ref. 18)), Cu2−xS (ZT = 1.7 (ref. 16)), Cu12Sb4S13 (ZT = 1.15 (ref. 43)) and polycrystalline CZTS (ZT = 1.1 (ref. 42)).
The temperature-dependent thermal conductivity analysis of the CZTSSe single crystals from 300 to 800 K (Fig. 3A) showed a weak anisotropy of thermal conductivity, where κ⊥ was ∼3% greater than κ∥. Phonon-Boltzmann-transport-based theoretical calculations have indicated that the lattice thermal conductivity along the a-axis in kesterite is ∼5% higher than that along the c-axis.44 All investigated samples exhibited an overall T−1 dependence on thermal conductivity, indicating that the lattice thermal conductivity was primarily driven by phonon–phonon Umklapp scattering. Therefore, the CZTSSe system reported herein is suitable for studying the effects of anion substitution on the decrease in lattice thermal conductivity. Here, κ decreased with increasing Se content (Fig. 3A), with the lowest values among the examined specimens (2.56 and 0.93 W mK−1 at 300 and 800 K, respectively) exhibited by Cu2ZnSnS2Se2 (x = 0.5). The Callaway model was adopted to analyze the lattice thermal conductivity of CZTSSe to elucidate the phonon scattering mechanisms, including the mass and strain differences between S and Se.45,46 Assuming that the Umklapp and point defect scattering were the dominant scattering mechanisms, the ratio of the lattice thermal conductivity of alloyed samples (κL) to that of the pure sample (κL0) can be expressed using eqn (1) and (2).
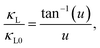 | (1) |
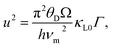 | (2) |
 | (3) |
Overall, the outstanding electrical properties and relatively low thermal conductivity of K-doped CZTSSe (sample 4) resulted in a TE material with a ZT of 1.9 at 800 K (Fig. 4A). This value is comparable to the highest ZT reported for other related quaternary polycrystalline materials, because the PF reported herein (2.22 × 10−3 W mK−2) is 2–18 times greater than those reported in the literature (Table S5). The combined uncertainty of all measurements involved in the calculation of ZT was estimated to be ∼10%. Additionally, the high ZT was proven to be reproducible by conducting repeated measurements for ten thermal cycles (Fig. 4B); the values for each cycle were within 10%. Compared with state-of-the-art chalcogenides,5,7,8,15,16,18,30,43 CZTSSe enables the investigated class of materials to be highly efficient for intermediate-temperature waste heat recovery. The approach based on the nature of single crystals in multinary compounds is comparable with existing strategies for discovering low-toxicity earth-abundant TE materials.
Potassium doping effect on TE properties
Revealing the location of the K atom in CZTSSe (sample 4) is crucial for discussing its doping effects on TE properties. Atomically resolved high-angle annular dark-field scanning TEM (HAADF-STEM) in Fig. S7 and integrated differential phase contrast (iDPC) images in Fig. 5A represent the positions of each element. The locations of Sn, Cu/Zn, and Se/S are identified byZ-contrast, aligning with the expected lattice sites of the pseudo-cubic structure. While revealing the positions of dopant atoms in TEM is challenging, some regions in the iDPC image show distortions around several Sn atoms (circled in yellow), which may suggest that subtle variations in the local electromagnetic environment are caused by impurity atoms, in this case, K.Complementing the TEM result, atom probe tomography (APT) provides further insights into the location of the dopant atoms. The APT data was calibrated using a {013} crystal lattice plane, with the lattice spacing in the spatial distribution map corresponding to the theoretical distance of 0.302 nm (Fig. 5B). Cluster analysis applied to the region of interest in the calibrated data revealed 165 K – Sn clusters, in contrast to 130 K – Cu and 59 K – Zn pairs. This suggests that K tends to cluster with Sn or locate at interstitial sites near the Sn atoms.
The effect of potassium doping on the electronic band structure and transport properties of CZTS was investigated using density functional theory (DFT) calculations. To reproduce the experimentally observed K atom positions near Sn atoms, a K atom was introduced into an interstitial site in a 2 × 2 × 1 supercell (Fig. S8). Fig. 5C and D shows the unfolded effective band structures of the supercells without and with interstitial K atom in CZTS, respectively. The presence of an interstitial K atom induces changes in both the band gap and the fine structure near the valence band maximum (VBM). In pristine CZTS, the VBM is split into two bands by crystal field effects, and the energy difference between the two bands (ΔCF) is 0.058 eV. Upon interstitial K-doping, the reduced symmetry lifts the degeneracy of the second band, resulting in further splitting; however, the energy difference from the topmost band is reduced to 0.045 eV. In addition, the band dispersion along the Γ− (0, 0, 0.5) direction becomes slightly flatter. Consistent with the changes in the band structure, the DOS near the VBM increases in the energy range of approximately 0.05–0.10 eV below the band edge, and the slope of the DOS also becomes steeper (Fig. 5E). These features qualitatively explain the experimental results that K-doping enhances the electrical conductivity while maintaining a high Seebeck coefficient, thereby achieving a remarkably high PF. Similar trends are observed in the band structure of CZTSSe with 50% Se substitution for S upon interstitial K doping (Fig. S8B).
Several studies have reported simultaneous enhancements in hole concentration and carrier mobility induced by alkali–metal incorporation in kesterite compounds; however, the microscopic mechanism responsible for these improvements remains insufficiently understood.21,25,52,53 A previous study on Na-doped CZTS proposed that Na substitution on the Cu site (NaCu) suppresses the dominant donor Zn on the antisite Cu (ZnCu) defect, thereby increasing the effective hole concentration and mobility through a reduction in the degree of carrier compensation. In this study, we found that K atoms tend to form clusters with Sn or occupy interstitial sites adjacent to Sn atoms, indicating that K preferentially resides in the local chemical environment around Sn. A recent study on K-doped CZTSe thin films similarly attributed the defect modification to doping-induced homogenization of local composition, which suppresses Sn-related deep defects such as Sn on antisite Sn (SnZn) and Sn vacancies (VSn).52 Suppression of these defects reduces carrier compensation and ionized impurity scattering, resulting in increased hole concentration and mobility. The presence of K–Sn clusters or K in interstitial sites adjacent to Sn may influence Sn diffusion by increasing local diffusion barriers and stabilizing Sn at its original lattice sites. Nevertheless, the possible role of K in modifying Sn diffusion kinetics remains speculative and represents an important direction for future research.
High TE conversion efficiency in the CZTSSe single-leg
TE devices operating at high temperatures must be thermodynamically stable, with the TE-material–contact interface being particularly important in this regard. Therefore, a Mo(SSe)2 layer was introduced as a diffusion barrier between CZTSSe and the electrode on the hot side of the TE device because of the presence of mobile atoms such as Cu and K in CZTSSe. Moreover, the Mo(SSe)2/Mo layer is traditionally used as a back-contact in CZTSSe-based PV devices because of its decent ohmic properties and bonding with CZTSSe.54 The TE single-leg device structure of the CZTSSe/Mo(SSe)2 barrier layer/Mo-contact was applied in this study. The hot-side interface was investigated to confirm the effectiveness of the Mo(SSe)2 layer as a diffusion barrier. A cross-sectional image of the interfaces in the aforementioned structure was obtained by scanning TEM (STEM) and energy-dispersive X-ray spectroscopy (EDX) (Fig. 6A). Chemical-diffusion-free uniform elemental distributions were observed by EDX-based compositional profiling across the CZTSSe/Mo(SSe)2/Mo interfaces (Fig. 6B). The K-atom concentration in CZTSSe/Mo(SSe)2/Mo was <1 at% over the entire measured region. The ∼5-µm-thick Mo(SSe)2 diffusion barrier had a smooth interface with ∼100-nm-sized roughness undulations. STEM images obtained before and after a 1100 h aging test conducted at 800 K (Fig. S9) revealed that the CZTSSe/Mo(SSe)2/Mo interfaces hindered chemical diffusion. Although the roughness undulations of the interface locally intensified after the aging test, no significant change of contact resistance was observed in the TE device properties.TE devices can be optimized by minimizing the electrical contact resistance to ensure a low internal resistance. The total electrical resistance, including that of the Mo(SSe)2 diffusion barrier, Mo contact, and Ag adhesive was estimated using a four-probe technique at 300 K to be as low as ∼0.55 mΩ (Fig. 6C), which corresponded to an interfacial contact resistivity of ∼88 µΩ cm2, which is higher than the benchmark established by the Bi2Te3–Ni interface (1–5 µΩ cm2).55
Output voltage (V), output power (P), and conversion efficiency (η) of a CZTSSe single-crystal-based single-leg TE device were monitored as a function of current (I) at different ΔT with a fixed cold-side temperature of 300 K (Fig. 7A–C). Current-dependent output heat flow (Qout) from the cold side (Fig. S10B) was used to calculate the conversion efficiency, and the details of the calculation are discussed in the SI. The device's theoretical properties were simulated through the COMSOL Multiphysics software with a heat-transfer module using the measured Seebeck coefficient, electrical conductivity, and thermal conductivity in Fig. S11.
The y-intercept and slope of linear V–I plots enable estimation of open-circuit voltage (VOC) and internal resistance (Rin), respectively. All device parameters that are related to the maximum conversion efficiency are listed in Table 1, wherein all the experimental parameters show good agreement with the simulated results. The increase in VOC and decrease in Rin with increasing temperature can be explained by the increase in S and σ of CZTSSe. The output power P increased with increasing current, reached a maximum when the external load resistance matched Rin, and subsequently decreased. The maximum output power (Pmax) at the hot-side temperature of 773 K (Pmax = 163 mW) corresponded to a high output-power-density of 6.52 kW m−2. An ηmax value of 10.4% was achieved by the CZTSSe single-leg device at a temperature gradient of 473 K. It is worth noting that this experimental value of ηmax is plotted together with that of other reported single-leg TE devices5,26,29–35,43,56,57 as a function of ΔT in Fig. 7D. Although additional device-related developments are required, such as achieving low contact resistivity, these TE systems have remarkable potential for use at intermediate temperatures. The experimentally obtained value of ηmax (10.4% at ΔT = 473 K) in this study is slightly lower than that of p-type GeTe (14% at ΔT = 438 K). However, our record value of 10.4% is higher than that of p-type half-Heusler ZrCoBi and other p-type chalcogenide-based single-leg devices such as PbTe, SnSe, AgSbTe2 and Cu12Sb4S13. In addition to its high conversion efficiency, the single-leg TE device operated at ΔT = 473 K showed decent thermal stability after various aging tests conducted at 800 K (Fig. S12). The maximum increase in Rin was ∼7% after the 1100 h aging test, and no distinct cracks were observed in the device by optical microscopy (Fig. S13). Such exceptional long-term service reliability at high temperature up to 800 K has rarely been reported in related-chalcogenide-based single-leg devices, marking a substantial advancement in TE device reliability.
| ΔT (K) | V OC (mV) | R in (mΩ) | P max (mW) | η max (%) | |
|---|---|---|---|---|---|
| Measured values | 73 | 16.6 | 40.1 | 1.7 | 0.4 |
| 173 | 42.3 | 38.9 | 11.6 | 1.4 | |
| 273 | 71.7 | 35.3 | 37.2 | 3.7 | |
| 373 | 93.2 | 32.1 | 66.7 | 5.2 | |
| 473 | 145.1 | 31.8 | 163.3 | 10.4 | |
| Simulated values | 73 | 20.2 | 31.7 | 3.2 | 0.8 |
| 173 | 48.4 | 28.7 | 20.3 | 2.6 | |
| 273 | 77.0 | 26.8 | 55.0 | 5.5 | |
| 373 | 108.9 | 25.3 | 116.3 | 9.0 | |
| 473 | 150.6 | 23.9 | 233.3 | 14.3 |
Conclusions
This study was aimed at achieving competitive ZT values for kesterite CZTSSe via a synergistic strategy involving a pseudo-cubic approach with a disordered structure and alloying. A record-high ZT of 1.9 was achieved at 800 K using a K-doped Cu1.9ZnSn(S1−xSex)4 single crystal (x = 0.5), whose PF (2.22 × 10−3 W mK−2) can compete with that of state-of-the-art TE materials. Importantly, a high conversion efficiency of 10.4% was achieved using a CZTSSe-based single-leg TE device with long-term stability. The following conclusions were drawn from this study: (i) interstitial K-doping (alkali effect) in CZTSSe enhances the electrical conductivity while maintaining a high Seebeck coefficient due to the steeper slope of DOS; (ii) the CZTSSe/Mo(SSe)2-barrier-layer/Mo-contact device structure led to decent ohmic properties and impeded chemical diffusion; (iii) thermal stability with small fluctuations of device parameters was ensured at aging times of up to 1100 h at 800 K. The results can pave the way for intermediate-temperature TE harvesting and accelerate the development of TE technology.Materials and methods
Single-crystal growth and fabrication of a single-leg TE device
CZTSSe single crystals were grown using the Sn-solvent-based traveling heater method (THM at an axial temperature gradient of 40 °C cm−1, a growth temperature of 850 °C, and a growth rate of 4 mm day−1). Details are provided in the SI. A diamond blade was used to dice the CZTSSe (x = 0.5) single crystals to a leg size of approximately 5 mm × 5 mm × 10 mm. The grown crystals and legs were mechanically polished with 0.01 µm Al2O3 powder and subsequently etched with HCl solution for 5 min to remove the saw and polishing damage. First, an ∼100-µm-thick Mo-contact layer was deposited on both the top and bottom sides of the leg by RF sputtering at 300 K in an Ar atmosphere. The Mo(SSe)2 barrier layer was formed by high-temperature annealing at 700 °C for 10 h under high vacuum (∼10−4 Pa), which enabled the Mo contact to react with the S and Se in CZTSSe. Subsequently, both sides and the Cu electrode were attached using Ag paste by heat treatment at 250 °C for 1 h in a N2 atmosphere.Characterization
The structural properties were analyzed by powder X-ray diffraction (XRD; Panalytical X'Pert PRO) and Raman spectroscopy (HORIBA T64000). The XRD measurements were performed at 40 kV and 40 mA using a Cu Kα radiation source. For the Raman analysis, a 532 nm laser excitation source was focused on the sample via an objective lens with a numerical aperture of 0.55, resulting in a laser power of 100 mW on the sample. The spectra were calibrated based on the 520 cm−1 Si peak. The orientation and crystallinity of the CZTSSe single crystals were investigated by X-ray rocking curve (XRC) analysis (Panalytical X'Pert PRO). The XRC measurements were performed at 45 kV and 40 mA with a step width of 0.002° using a Cu Kα radiation source. The crystallographic orientation of all samples can be observed using a scanning electron microscope (SEM) equipped with an electron backscatter diffraction (EBSD; Quanta 600) system shown in Fig. S3. The samples were imaged at 30 kV using an SEM probe and that used an off-axis backscatter detector.The detailed composition of the crystals was determined by inductively coupled plasma-atomic emission spectroscopy (ICP-AES; SII NanoTechnology SPS3520UV). Samples were dissolved in a mixture of HNO3 and HCl to facilitate this analysis.
Microstructure and composition of the CZTSSe single crystal and single-leg were determined by TEM and STEM (JEOL 2800) at 200 kV using two simultaneous solid-state EDX detectors. Spectral image acquisition was performed over a series of consecutive subsecond frames with drift correction between frames and a total acquisition time of less than 20 min. The EDX data were processed using Thermo Scientific Image Analysis software. The analytical certainty associated with EDX profiling was within 0.5 at%.
Atomic structure investigations were carried out by using TEM and APT. HAADF-STEM and iDPC images were acquired using a Themis-Z S/TEM (Thermo Fisher Scientific), operated at 300 kV, equipped with double-aberration correctors. The beam convergence angle in both HAADF and iDPC modes was 17.9 mrad, and the collection angles in HAADF and iDPC modes were 48–200 and 9–35 mrad, respectively. The iDPC images were acquired using a quadrant segmented dark field detector. APT was conducted using a CAMECA Local Electrode Atom Probe (LEAP) 4000X Si equipped with a picosecond-pulse ultraviolet laser. A laser pulse energy of 50 pJ and laser frequency of 200 kHz were used. APT was performed at ∼50 K and the data reconstruction and analysis were performed via CAMECA's AP Suite 6.3 software. The reconstruction was calibrated using the method described by Gault et al.58,59 Site-specific TEM and APT specimens were prepared by standard lift-out techniques using a Helios G4 plasma focused ion beam scanning electron microscope (Thermo Fisher Scientific).
The electrical conductivity σ and Seebeck coefficient S of samples with dimensions of approximately 2 mm × 2 mm × 10 mm were simultaneously measured in a helium atmosphere at 300–800 K using a commercial system (ZEM3; ADVANCE RIKO). The thermal conductivity κ was calculated using the equation κ = λCpD, where λ is the thermal diffusivity, Cp is the specific heat capacity, and D is the density. The thermal diffusivity of rectangular samples (8 mm × 8 mm × 1 mm) was measured using the laser flash method (Netzsch; LFA467); the samples were coated with a thin layer of graphite to minimize errors due to the emissivity of the material. The specific heat capacity of samples measuring 3 mm × 3 mm × 3 mm was determined by differential scanning calorimetry (DSC; Rigaku Thermo plus EVO2 DSCvesta). The mass of all samples was measured by using thermogravimetric and differential thermal analysis (TG-DTA; Rigaku Thermo plus EVO2 TG-DTA8122). Densities in the 4.4–4.5 g cm−3 range were measured using the Archimedes method at room temperature in ultrapure water.
The hole concentration p and mobility µ were determined based on the temperature dependence of the Hall effect (TOYO; ResiTest8400) in a 0.45 T magnetic field with the van der Pauw geometry in a He atmosphere at 300 K. For the Hall measurements, 200–300-nm-thick Au contacts with a diameter of 1 mm were evaporated onto the corners of each CZTSSe bulk single crystal with dimensions of 5 mm × 5 mm × 0.8 mm.
A four-probe technique was used to measure the electrical resistance of the single-leg CZTSSe device. A movable probe measured the voltage along the sample length when an alternating current was applied across the leg. The conversion efficiency of the CZTSSe-based single-leg TE device was evaluated by measuring electrical output power and output heat flow under vacuum (∼10−2 Pa) using an M612-B tester (MOTTAINAI ENERGY).
Computational details
The DFT calculations were performed using the Quantum ESPRESSO (QE) package60,61 with projector augmented wave pseudopotentials.62 The generalized gradient approximation functional with Perdew–Burke–Ernzerhof parametrization (GGA-PBE)63 was chosen for the exchange and correlation potentials. We applied the Hubbard U correction to the strongly correlated Cu 3d and Zn 3d orbitals. We chose Ueff (= U − V) values as 5.2 eV and 6.5 eV for Cu and Zn, respectively. Here, U, V, and Ueff are the on-site Coulomb interaction strength, the on-site exchange parameter, and the resulting effective repulsion applied to the localized d states, respectively. Spin–orbit coupling was not considered in the present study. The conventional unit cell with a tetragonal lattice containing 16 atoms was fully relaxed until the residual forces became less than 10−6 eV Å−1. A cutoff energy of 130 Ry for the plane wave basis and an 8 × 8 × 4 k-grid were used for structure relaxation. The 2 × 2 × 1 supercell was constructed based on the relaxed unit cell. Then, one K atom was introduced at an interstitial site identified using the pymatgen-analysis-defect module.64 Although there are two candidate interstitial sites suggested by Voronoi tessellation, the one that reproduces the experimental atomic position obtained from STEM observation was adopted. To model the alloying in the (S,Se)-site in CZTSSe, the special quasirandom structure (SQS) method implemented in the Alloy Theoretic Automated Toolkit (ATAT) was used.65 The experimentally determined lattice parameters were used and atomic positions in the supercell were fully relaxed until the residual forces became less than 10−3 eV Å−1. A cutoff energy of 130 Ry for the plane wave basis and a 4 × 4 × 4 k-grid were used for supercell relaxation. The effective band structures of the supercells in the primitive Brillouin zone were obtained by using the band unfolding method implemented in BandUP code.66Author contributions
All authors discussed the results and provided feedback on the manuscript. Conceptualization: A. Nagaoka; methodology: A. Nagaoka, S. Miura, K. Nomoto, and N. Sato; investigation: A. Nagaoka, S. Miura, K. Nomoto, K. Chen, and N. Sato; visualization: A. Nagaoka, S. Miura, K. Nomoto, K. Chen, and N. Sato; supervision: K. Yoshino and K. Nishioka; writing—original draft: A. Nagaoka; writing—review & editing: A. Nagaoka, K. Nomoto, N. Sato, and K. Nishioka.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Single crystal growth by Traveling Heater Method (THM). Characterizations for structural properties, pseudo-cubic structure, degree of order in CZTSSe, compositional properties, and thermoelectric conversion efficiency. Fig. S1 to S13; Tables S1 to S5; references. See DOI: https://doi.org/10.1039/d5ta08640j.Acknowledgements
This study was supported by JSPS KAKENHI Grant Number JP23K26603 (A. Nagaoka), JSPS KAKENHI Grant Number JP22K14505 (N. Sato), TEPCO Memorial Foundation No. 23-006 (A. Nagaoka), and the Australian Research Council Discovery Early Career Researcher Award (DE220100527) (K. Nomoto).References
- C. Forman, I. K. Muritala, R. Pardemann and B. Meyer, Renewable Sustainable Energy Rev., 2016, 57, 1568–1579 CrossRef.
- A. Firth, B. Zhang and A. Yang, Appl. Energy, 2019, 235, 1314–1334 CrossRef.
- Y. Sun, S. Tian, P. Ciais, Z. Zeng, J. Meng and Z. Zhang, Nat. Commun., 2022, 13, 297 CrossRef CAS.
- X. L. Shi, J. Zou and Z. G. Chen, Chem. Rev., 2020, 120, 7399–7515 CrossRef CAS.
- W. He, D. Wang, H. Wu, Y. Xiao, Y. Zhang, D. He, Y. Feng, Y. J. Hao, J. F. Dong, R. Chetty, L. Hao, D. Chen, J. Qin, Q. Yang, X. Li, J. M. Song, Y. Zhu, W. Xu, C. Niu, X. Li, G. Wang, C. Liu, M. Ohta, S. J. Pennycook, J. He, J. F. Li and L. D. Zhao, Science, 2019, 365, 1418–1424 CrossRef CAS.
- Mineral Commodity Summaries 2024, United States Geological Survey Search PubMed.
- A. A. Olvera, N. A. Moroz, P. Sahoo, P. Ren, T. P. Bailey, A. A. Page, C. Uher and P. F. P. Poudeu, Energy Environ. Sci., 2017, 10, 1668–1676 RSC.
- L. D. Zhao, S. H. Lo, Y. Zhang, H. Sun, G. Tan, C. Uher, C. Wolverton, V. P. Dravid and M. G. Kanatzidis, Nature, 2014, 508, 373–377 CrossRef CAS PubMed.
- J. Sui, J. Li, J. He, Y.-L. Pei, D. Berardan, H. Wu, N. Dragoe, W. Cai and L.-D. Zhao, Energy Environ. Sci., 2013, 6, 2916–2920 RSC.
- C. Fu, S. Bai, Y. Liu, Y. Tang, L. Chen, X. Zhao and T. Zhu, Nat. Commun., 2015, 6, 8144 CrossRef PubMed.
- Y. Luo, J. Yang, Q. Jiang, W. Li, D. Zhang, Z. Zhou, Y. Cheng, Y. Ren and X. He, Adv. Energy Mater., 2016, 6, 1600007 CrossRef.
- J. Zhang, L. Song, S. H. Pedersen, H. Yin, L. T. Hung and B. B. Iversen, Nat. Commun., 2017, 8, 13901 CrossRef CAS.
- K. F. Hsu, S. Loo, F. Guo, W. Chen, J. S. Dyck, C. Uher, T. Hogan, E. K. Polychroniadis and M. G. Kanatzidis, Science, 2004, 303, 818–821 CrossRef CAS PubMed.
- X. Shi, J. Yang, J. R. Salvador, M. Chi, J. Y. Cho, H. Wang, S. Bai, J. Yang, W. Zhang and L. Chen, J. Am. Chem. Soc., 2011, 133, 7837–7846 CrossRef CAS.
- K. Biswas, J. He, I. D. Blum, C. I. Wu, T. P. Hogan, D. N. Seidman, V. P. Dravid and M. G. Kanatzidis, Nature, 2012, 489, 414–418 Search PubMed.
- Y. He, T. Day, T. Zhang, H. Liu, X. Shi, L. Chen and G. J. Snyder, Adv. Mater., 2014, 26, 3974–3978 CrossRef CAS.
- S. I. Kim, K. H. Lee, H. A. Mun, H. S. Kim, S. W. Hwang, J. W. Roh, D. J. Yang, W. H. Shin, X. S. Li, Y. H. Lee, G. J. Snyder and S. W. Kim, Science, 2015, 348, 109–114 CrossRef CAS.
- M. Ibanez, Z. Luo, A. Genc, L. Piveteau, S. Ortega, D. Cadavid, O. Dobrozhan, Y. Liu, M. Nachtegaal, M. Zebarjadi, J. Arbiol, M. V. Kovalenko and A. Cabot, Nat. Commun., 2016, 7, 10766 CrossRef CAS.
- J. Zhang, R. Liu, N. Cheng, Y. Zhang, J. Yang, C. Uher, X. Shi, L. Chen and W. Zhang, Adv. Mater., 2014, 26, 3848–3853 CrossRef CAS.
- A. Nagaoka, T. Masuda, S. Yasui, T. Taniyama and Y. Nose, Appl. Phys. Express, 2018, 11, 051203 CrossRef.
- A. Nagaoka, K. Yoshino, T. Masuda, T. D. Sparks, M. A. Scarpulla and K. Nishioka, J. Mater. Chem. A, 2021, 9, 15595–15604 RSC.
- P. M. P. Salomé, H. Rodriguez-Alvarez and S. Sadewasser, Sol. Energy Mater. Sol. Cells, 2015, 143, 9–20 Search PubMed.
- D. Sharma, N. Nicoara, P. Jackson, W. Witte, D. Hariskos and S. Sadewasser, Nat. Energy, 2024, 9, 163–171 Search PubMed.
- P. Jood, J. P. Male, S. Anand, Y. Matsushita, Y. Takagiwa, M. G. Kanatzidis, G. J. Snyder and M. Ohta, J. Am. Chem. Soc., 2020, 142, 15464–15475 CrossRef CAS.
- A. Nagaoka, H. Miyake, T. Taniyama, K. Kakimoto, Y. Nose, M. A. Scarpulla and K. Yoshino, Appl. Phys. Lett., 2014, 104, 152101 CrossRef.
- Z. Bu, X. Zhang, B. Shan, J. Tang, H. Liu, Z. Chen, S. Lin, W. Li and Y. Pei, Sci. Adv., 2021, 7, eabf2738 CrossRef CAS PubMed.
- X. Wu, Y. Lin, Z. Han, H. Li, C. Liu, Y. Wang, P. Zhang, K. Zhu, F. Jiang, J. Huang, H. Fan, F. Cheng, B. Ge and W. Liu, Adv. Energy Mater., 2022, 12, 2203039 CrossRef CAS.
- A. Nagaoka, K. Nagatomo, K. Nakashima, Y. Hirai, Y. Ota, K. Yoshino and K. Nishioka, Mater. Trans., 2023, 64, 2535–2541 CrossRef CAS.
- X. L. Shi, W. D. Liu, M. Li, Q. Sun, S. D. Xu, D. Du, J. Zou and Z. G. Chen, Adv. Energy Mater., 2022, 12, 2200670 CrossRef CAS.
- B. C. Chen, K. K. Wang and H. J. Wu, Small, 2024, 2401723 CrossRef CAS.
- Z. Liu, J. Mao, J. Sui and Z. Ren, Energy Environ. Sci., 2018, 11, 23–44 RSC.
- H. Zhu, R. He, J. Mao, Q. Zhu, C. Li, J. Sun, W. Ren, Y. Wang, Z. Liu, Z. Tang, A. Sotnikov, Z. Wang, D. Broido, D. J. Singh, G. Chen, K. Nielsch and Z. Ren, Nat. Commun., 2018, 9, 2497 CrossRef.
- S. Liu, Y. Wen, S. Bai, H. Shi, Y. Qin, B. Qin, D. Liu, Q. Cao, X. Gao, L. Su, C. Chang, X. Zhang and L. D. Zhao, Adv. Mater., 2024, e2401828 CrossRef.
- P. Sauerschnig, P. Jood and M. Ohta, Adv. Mater. Technol., 2023, 8, 2201295 Search PubMed.
- X. Y. Tan, J. Dong, J. Liu, D. Zhang, S. F. D. Solco, K. Saglik, N. Jia, I. You, S. W. Chien, X. Wang, L. Hu, Y. Luo, Y. Zheng, D. X. Y. Soo, R. Ji, K. C. H. Goh, Y. Jiang, J. F. Li, A. Suwardi, Q. Zhu, J. Xu and Q. Yan, Adv. Sci., 2024, e2400870 CrossRef.
- A. Nagaoka, R. Katsube, S. Nakatsuka, K. Yoshino, T. Taniyama, H. Miyake, K. Kakimoto, M. A. Scarpulla and Y. Nose, J. Cryst. Growth, 2015, 423, 9–15 CrossRef CAS.
- A. Nagaoka, H. Miyake, T. Taniyama, K. Kakimoto and K. Yoshino, Appl. Phys. Lett., 2013, 103, 112107 CrossRef.
- S. Chen, A. Walsh, X. G. Gong and S. H. Wei, Adv. Mater., 2013, 25, 1522–1539 Search PubMed.
- M.-L. Liu, F.-Q. Huang, L.-D. Chen and I. W. Chen, Appl. Phys. Lett., 2009, 94, 202103 Search PubMed.
- Y. Dong, H. Wang and G. S. Nolas, Inorg. Chem., 2013, 52, 14364–14367 Search PubMed.
- Y. Dong, H. Wang and G. S. Nolas, Phys. Status Solidi, 2014, 8, 61–64 Search PubMed.
- Q. Jiang, H. Yan, Y. Lin, Y. Shen, J. Yang and M. J. Reece, J. Mater. Chem. A, 2020, 8, 10909–10916 Search PubMed.
- H. Hu, H. L. Zhuang, Y. Jiang, J. Shi, J. W. Li, B. Cai, Z. Han, J. Pei, B. Su, Z. H. Ge, B. P. Zhang and J. F. Li, Adv. Mater., 2021, 33, e2103633 Search PubMed.
- T. Gürel, C. Sevik and T. Çağın, Phys. Rev. B: Condens. Matter Mater. Phys., 2011, 84, 205201 Search PubMed.
- J. Callaway and H. C. von Baeyer, Phys. Rev., 1960, 120, 1149–1154 Search PubMed.
- B. Abeles, Phys. Rev., 1963, 131, 1906–1911 Search PubMed.
- H. Wang, A. D. LaLonde, Y. Pei and G. J. Snyder, Adv. Funct. Mater., 2013, 23, 1586–1596 CrossRef CAS.
- C. P. Heinrich, T. W. Day, W. G. Zeier, G. J. Snyder and W. Tremel, J. Am. Chem. Soc., 2014, 136, 442–448 Search PubMed.
- X. He, J. Pi, Y. Dai and X. Li, Acta Metall. Sin., 2013, 26, 285–292 Search PubMed.
- S.-H. Lo, J. He, K. Biswas, M. G. Kanatzidis and V. P. Dravid, Adv. Funct. Mater., 2012, 22, 5175–5184 CrossRef CAS.
- W. G. Zeier, A. LaLonde, Z. M. Gibbs, C. P. Heinrich, M. Panthofer, G. J. Snyder and W. Tremel, J. Am. Chem. Soc., 2012, 134, 7147–7154 CrossRef CAS PubMed.
- S. Tao, L. Dong, J. Han, Y. Wang, Q. Gong, J. Wei, M. Zhao and D. Zhuang, J. Mater. Chem. A, 2023, 11, 9085–9096 RSC.
- L. Wang, J. Ban, L. Han, Z. Zhou, W. Zhou, D. Kou, Y. Meng, Y. Qi, S. Yuan and S. Wu, J. Mater. Chem. A, 2024, 12, 25643–25677 RSC.
- W. Wang, M. T. Winkler, O. Gunawan, T. Gokmen, T. K. Todorov, Y. Zhu and D. B. Mitzi, Adv. Energy Mater., 2014, 4, 1301465 CrossRef.
- S. P. Feng, Y. H. Chang, J. Yang, B. Poudel, B. Yu, Z. Ren and G. Chen, Phys. Chem. Chem. Phys., 2013, 15, 6757–6762 RSC.
- R. Chetty, Y. Kikuchi, Y. Bouyrie, P. Jood, A. Yamamoto, K. Suekuni and M. Ohta, J. Mater. Chem. C, 2019, 7, 5184–5192 RSC.
- J. Jiang, H. Zhu, Y. Niu, Q. Zhu, S. Song, T. Zhou, C. Wang and Z. Ren, J. Mater. Chem. A, 2020, 8, 4790–4799 RSC.
- B. Gault, F. de Geuser, L. T. Stephenson, M. P. Moody, B. C. Muddle and S. P. Ringer, Microsc. Microanal., 2008, 14, 296–305 Search PubMed.
- B. Gault, M. P. Moody, F. de Geuser, G. Tsafnat, A. La Fontaine, L. T. Stephenson, D. Haley and S. P. Ringer, J. Appl. Phys., 2009, 105, 034913 CrossRef.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.:Condens. Matter, 2009, 21, 395502 CrossRef.
- P. Giannozzi, O. Andreussi, T. Brumme, O. Bunau, M. Buongiorno Nardelli, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, M. Cococcioni, N. Colonna, I. Carnimeo, A. Dal Corso, S. de Gironcoli, P. Delugas, R. A. DiStasio Jr, A. Ferretti, A. Floris, G. Fratesi, G. Fugallo, R. Gebauer, U. Gerstmann, F. Giustino, T. Gorni, J. Jia, M. Kawamura, H. Y. Ko, A. Kokalj, E. Kucukbenli, M. Lazzeri, M. Marsili, N. Marzari, F. Mauri, N. L. Nguyen, H. V. Nguyen, A. Otero-de-la-Roza, L. Paulatto, S. Ponce, D. Rocca, R. Sabatini, B. Santra, M. Schlipf, A. P. Seitsonen, A. Smogunov, I. Timrov, T. Thonhauser, P. Umari, N. Vast, X. Wu and S. Baroni, J. Phys.:Condens. Matter, 2017, 29, 465901 CrossRef CAS.
- P. E. Blochl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS.
- S. P. Ong, W. D. Richards, A. Jain, G. Hautier, M. Kocher, S. Cholia, D. Gunter, V. L. Chevrier, K. A. Persson and G. Ceder, Comput. Mater. Sci., 2013, 68, 314–319 CrossRef CAS.
- A. van de Walle, Calphad, 2009, 33, 266–278 CrossRef CAS.
- P. V. C. Medeiros, S. Stafström and J. Björk, Phys. Rev. B: Condens. Matter Mater. Phys., 2014, 89, 041407 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−
exp(−



