Polymer dielectrics enabled by molecular engineering design and charge trap regulation for high-temperature energy storage
Guangyu
Duan
 *a,
Fengying
Hu
a,
Yiran
He
a,
Zuming
Hu
b,
Ming
Tian
ac and
Junwei
Zha
d
*a,
Fengying
Hu
a,
Yiran
He
a,
Zuming
Hu
b,
Ming
Tian
ac and
Junwei
Zha
d
aCollege of Materials Engineering, Henan University of Engineering, Zhengzhou 450007, China. E-mail: duanguangyu@haue.edu.cn
bState Key Laboratory for Modification of Chemical Fibers and Polymer Materials, Donghua University, Shanghai 201620, China
cCollege of Materials Science and Engineering, Beijing University of Chemical Technology, Beijing, 101500, China
dCollege of Chemistry and Biological Engineering, Beijing University of Science and Technology, Beijing, 100083, China
First published on 11th November 2025
Abstract
Aromatic polyimides (PIs) are widely regarded as promising high-temperature polymer dielectric films because of their excellent thermal stability. However, the extensive intra- and inter-chain migration of delocalized π-electrons through aromatic rings, coupled with a sharp increase in the density of injected and thermally activated charge carriers, significantly weakens the high-temperature energy storage performance of conventional PI. In this work, a novel strategy combining molecular engineering design with modulation of charge trap levels was employed to fabricate a series of fluorinated polyimide/magnesium oxide (FPI/MgO) dielectric films incorporating –CF3 groups and wide-bandgap MgO nanoparticles. Compared with conventional PI, the introduction of –CF3 and MgO effectively suppresses π-electron migration and thermally excited charge carriers and hinders the propagation and development of breakdown pathways along the electric field direction. These synergistic effects effectively inhibit bulk-limited hopping conduction, leading to significantly improved breakdown strength (Eb) and enhanced high-temperature capacitive performance of the FPI/MgO dielectric film. At 150 °C, the FPI dielectric film with 0.10 wt% MgO exhibits a high Eb of 508 MV m−1, representing an enhancement of 46.4% over that of conventional PI (347 MV m−1). Benefiting from the superior Eb and suppressed leakage current density, the FPI dielectric film with 0.10 wt% MgO achieves a maximal discharge energy density (Ud) of 4.97 J cm−3 at 150 °C, which is 4.3 times that of conventional PI (1.15 J cm−3) and exceeds that of most previously reported high-temperature polymer dielectric films. This work presents a new paradigm for developing PI-based dielectrics with superior high-temperature energy storage performance.
1. Introduction
Due to the high energy density and ultrafast charge–discharge characteristics, polymer film capacitors constitute critical elements for high-frequency energy storage and conversion, offering substantial potential for integration into advanced electronics, medical devices and high-pulse systems.1–3 As modern electronic systems progress toward miniaturization and higher functional integration, film capacitors are increasingly required to operate under extreme conditions-including elevated temperatures and high electric fields-such as those encountered in underground oil and gas exploration, wind energy conversion, and electromagnetic launchers.4,5 In these demanding operational scenarios, the high-temperature reliability of polymer dielectrics becomes paramount to ensuring capacitor performance and service longevity. Nevertheless, conventional polymer dielectrics often suffer from significantly increased conduction losses, diminished insulating strength, and impaired capacitive stability at elevated temperatures, effects primarily driven by enhanced bulk-limited and electrode-limited conduction mechanisms.6 For instance, biaxially oriented polypropylene (BOPP), a widely deployed dielectric in power inverters, exhibits pronounced capacitive deterioration when operating above 85 °C.7,8 Accordingly, developing novel high-temperature polymer dielectric films that demonstrate robust thermal stability and reliable energy storage performance across wide temperature ranges is essential for advancing next-generation polymer film capacitor technologies.Aromatic polyimides (PIs) are considered exceptional candidates for high-temperature polymer dielectrics due to their outstanding thermal stability, mechanical strength, and chemical resistance-properties stemming from the π–π conjugated aromatic backbone.9,10 However, at elevated temperatures and high electric fields, the π–π conjugated structure facilitates considerable π-electron delocalization and significantly promotes the formation of intra- and inter-molecular charge transfer complexes (CTCs) (Fig. 1a), resulting in a sharp rise in leakage current density and substantial Joule heating.11,12 The accumulation of Joule heat and elevated current density collectively deteriorate the dielectric breakdown strength and energy storage performance, ultimately impairing the operational reliability of PI dielectric films under extreme conditions. The incorporation of wide-bandgap (Eg) inorganic nanofillers, such as Al2O3,13,14 SiO2,15 and TiO2 (ref. 16), has widely been employed to introduce deep charge traps within PI nanocomposites, effectively restraining charge carrier transport and mobility at elevated temperatures. Nevertheless, this approach often encounters challenges related to poor organic–inorganic interfacial compatibility, which causes localized electric field distortion and filler agglomeration at higher loadings that severely compromises flexibility, uniformity, and processability. These limitations hinder the scalable production of high-performance composite dielectric films.17–20 In contrast, molecular engineering offers a distinctive strategy to concurrently enhance the service performance and processability of polymer dielectrics. The π–π stacking and inter-chain charge transfer can be effectively suppressed by incorporating non-coplanar alicyclic structures21,22 or crosslinking sites23,24 into the PI backbone and noticeably reducing the conduction losses at elevated temperatures and high electric fields.
 | ||
| Fig. 1 Possible migration and motion of charge carriers in the (a) PI and (b) FPI/MgO films. (c) Schematic illustration of the fabrication process for FPI and FPI/MgO films. | ||
In this work, guided by molecular engineering principles and the strategy of introducing deep charge trap levels, a novel high-temperature PI dielectric film with superior electrical insulation and enhanced energy storage performance at elevated temperatures was designed. Specifically, an aromatic fluorinated polyimide (FPI) featuring four trifluoromethyl (–CF3) groups per repeating unit was synthesized via a straightforward solution polymerization route. The combination of experimental and density functional theory (DFT) analyses reveals that the steric hindrance of –CF3 groups enlarges the interchain distance, leading to the disruption of π–π conjugation and consequent suppression of delocalized electron migration along the FPI backbone. Moreover, to further impede the transport and mobility of injected and thermally activated charge carriers under extreme thermal conditions, trace amounts of wide-bandgap magnesium oxide (MgO) nanoparticles were incorporated into the FPI matrix via in situ polymerization. The results demonstrate that the well-dispersed MgO nanoparticles not only mitigate agglomeration and local electric field distortion, but also significantly reduce high-temperature conduction loss and leakage current density by deepening trap energy levels and increasing trap density (Fig. 1a and b). As a result of suppressed electron delocalization and reduced leakage current density, the FPI/MgO dielectric films exhibit markedly improved high-temperature energy storage performance. At 150 °C, the FPI dielectric film with only 0.10 wt% MgO achieves an outstanding Ud of 4.97 J cm−3, which is 4.3 times that of conventional PI (1.15 J cm−3). This work presents a novel strategy for designing high-performance PI dielectric films capable of operating under extreme conditions, paving the way for their application in advanced electronic devices.
2. Experimental section
2.1 Materials
Pyromellitic dianhydride (PMDA, 99.0%) and 4,4′-diaminodiphenyl ether (ODA, 98.0%) were provided by Shanghai Macklin Biochemical Co., Ltd. 2,2′-Bis(trifluoromethyl)benzidine (TFMB, 98.0%) and 4,4′-(hexafluoroisopropylidene) diphthalic anhydride (6FDA, 99.0%) and magnesium oxide nanoparticles (MgO, ∼50 nm in diameter) were purchased from Shanghai Aladdin Industrial Corporation. N-Methylpyrrolidone (NMP, 99.0%) was sourced from Shanghai Shaen Chemical Co., Ltd. All chemical reagents were directly used as received without further purification.2.2 Preparation of the FPI/MgO dielectric film
The FPI/MgO dielectric films were fabricated via in situ polymerization followed by high-temperature thermal imidization procedures. Specifically, MgO nanoparticles were first dispersed in N-methylpyrrolidone (NMP) via ultrasonic treatment at 250 W and 53 kHz for 2 h in a three-neck flask maintained at 25 °C to obtain a homogeneous suspension. Under a nitrogen atmosphere, 0.02 mol of TFMB was completely dissolved into the resulting mixture. Subsequently, 0.0202 mol of 6FDA was gradually added to the flask to adjust the solid content to 18 wt%, and the reaction solution was stirred continuously for 12 h under nitrogen protection to yield a transparent and viscous poly(amic acid) (FPAA) precursor solution containing –CF3 groups. By adjusting the MgO content relative to the total mass of the two monomers to 0.00 wt%, 0.05 wt%, 0.10 wt%, and 0.25 wt%, a series of precursor solutions-denoted as FPAA, FPAA/MgO-0.05, FPAA/MgO-0.10, and FPAA/MgO-0.25-were successfully prepared.To obtain FPI/MgO dielectric films, the method of solution coating was further utilized. In brief, the precursor solution was coated onto a clean glass substrate using a 75 µm coating blade and then subjected to an oven at 80 °C for 12 h. Then, the glass substrate was then transferred to a high-temperature oven and subjected to thermal imidization with a heating rate of 2 °C min−1, with successive temperature plateaus of 2 h at 100 °C, 2 h at 200 °C, 1 h at 250 °C and 1 h at 300 °C, respectively. After cooling to room temperature, the glass was placed in deionized water until the film detached, and the obtained dielectric film was dried at 80 °C for 12 h. For the convenience of subsequent description, the different dielectric films were named FPI, FPI/MgO-0.05, FPI/MgO-0.10 and FPI/MgO-0.25, with the thickness in the range of 8–12 µm. The synthetic route of FPI and fabrication process of FPI/MgO dielectric films are illustrated in Fig. 1c.
2.3 Preparation of the conventional PI film
Briefly, ODA was initially added into a three-necked flask with NMP under a nitrogen atmosphere. After the diamine was completely dissolved, PMDA was slowly added into above solution and the mixed solution was stirred for 12 h under the nitrogen protection at room temperature to obtain a yellow and viscous PAA solution; the synthetic route of PAA is displayed in Fig. S1. Considering the kinetic differences observed during the polymerization of different monomers, the solid content of the conventional PAA system was set at 12 wt% to prevent premature gelation of the solution that would otherwise result from an excessively high concentration. Consistent with the preparation process of FPI/MgO dielectric films, the PAA solution initially was coated on a glass substrate using a 50 µm coating blade, followed by a thermal imidization process. The PI based on PMDA/ODA was chosen as the conventional benchmark because it integrates exceptional thermal stability and a scalable processing window and is widely regarded as the most industrially viable high-temperature polymer dielectric, offering a robust reference for evaluating the dielectric performance of 6FDA/TFMB fluorinated polyimide at coupled high-field and high-temperature extremes.252.4 Characterization and simulation
Details of sample characterization are given in Section S1, SI. Molecular dynamics (MD) simulation, density functional theory (DFT) calculations and finite element simulation are provided in Section S2, SI.3. Results and discussion
To elucidate the influence of molecular configuration on the condensed-state structure, molecular dynamics simulations were utilized to analyze the free volume fractions of PI and FPI models, as illustrated in Fig. S2, 2a and b. The PI model exhibits a free volume fraction of only 26.5%, which can be attributed to the strong π–π stacking between aromatic rings that enhances intermolecular interactions and increases chain-packing density. In contrast, the incorporation of bulky –CF3 groups into the FPI backbone significantly attenuates interchain interactions and expands interchain spacing by disrupting conjugation among aromatic rings, resulting in a higher free volume fraction of 33.7%. The X-ray diffraction (XRD) patterns of PI and FPI films are displayed in Fig. 2c. The diffraction peak of FPI shifts to a lower 2θ angle compared to that of PI. According to Bragg's law (2d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin
sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ = nλ), where d is the interplanar spacing, θ is the diffraction angle, n is the diffraction order, and λ is the X-ray wavelength, a decrease in 2θ corresponds to an increase in d-spacing. Consequently, the calculated d-spacing of FPI increases to 6.15 Å from 4.19 Å for conventional PI, consistent with the simulation predictions.26,27
θ = nλ), where d is the interplanar spacing, θ is the diffraction angle, n is the diffraction order, and λ is the X-ray wavelength, a decrease in 2θ corresponds to an increase in d-spacing. Consequently, the calculated d-spacing of FPI increases to 6.15 Å from 4.19 Å for conventional PI, consistent with the simulation predictions.26,27
Owing to its potent electron-withdrawing capability, the –CF3 group restricts π-electron delocalization via the inductive effect, thereby truncating electron transport pathways along the conjugated backbone.28,29 This leads to a significant reduction in the electrical conductivity of FPI, which contributes to maintaining high capacitive performance and Eb at elevated temperatures. To further explain the suppressive effect of –CF3 groups on π-electron migration, DFT calculations were employed to analyze the electrostatic potential (ESP) distribution and spatial area of PI and FPI, as shown in Fig. 2d and e. In the ESP maps, blue and red regions correspond to electron-rich and electron-deficient areas, respectively. Notably, FPI exhibits a pronounced positive electrostatic potential around the –CF3 groups, confirming the strong electron-withdrawing character. Furthermore, the integrated ESP area of FPI is larger than that of PI, suggesting an enhanced ability to mitigate the influence of external electric fields.12
It has been reported that disrupting π–π conjugation in aromatic systems can effectively widen the Eg, thus remarkably suppressing nonlinear conduction mechanisms in polymer dielectrics at high temperatures.27 Accordingly, the highest occupied molecular orbital (HOMO), lowest unoccupied molecular orbital (LUMO), and Eg for PI and FPI were computed, as displayed in Fig. 2f and g. The results indicate that FPI possesses a lower HOMO and higher LUMO, yielding a larger HOMO–LUMO gap of 4.01 eV compared to 2.79 eV for conventional PI. This computational trend is qualitatively consistent with experimental optical band gap measurements (Fig. S3). Additionally, due to the expanded interchain spacing and constrained π-electron mobility, both inter- and intramolecular charge transfer complex (CTC) effects are significantly attenuated. This outcome leads to high optical transparency in the precursor FPAA solution and FPI film, as depicted in Fig. 2h and i. Collectively, these computational and experimental findings indicate that the FPI film exhibits superior high-temperature energy storage performance and breakdown strength.
The above findings demonstrate that the molecular engineering strategy contributes to potently suppressing the delocalization of π-electrons and inhibiting the formation of intra- and inter-molecular CTCs. However, FPI still exhibits a substantial increase in leakage current density at elevated temperatures, originating from charge carrier de-trapping and injection from metal electrodes. Therefore, wide-bandgap MgO nanoparticles were incorporated into the FPI matrix to introduce deep charge traps, which considerably impede charge de-trapping and suppress the injection and transport of thermally excited electrons. The XRD pattern of MgO nanoparticles in Fig. 3a displays characteristic diffraction peaks at 2θ = 36.8°, 42.7°, 62.2°, 74.5°, and 78.5°, respectively, corresponding to the (100), (200), (220), (222), and (311) planes of the cubic crystal structure (PDF# 45-0946).30 The scanning electron microscopy (SEM) image in Fig. 3b reveals that the MgO nanoparticles possess an average size of approximately 50 nm, with energy-dispersive spectroscopy (EDS) mapping confirming homogeneous distribution of Mg and O elements.
X-Ray photoelectron spectroscopy (XPS) analysis was conducted to examine the surface elemental composition of the various polymer films (Fig. 3c). A distinct F 1s peak appears at approximately 690.0 eV in the FPI film, which is absent in the conventional PI film. Notably, no characteristic MgO signals are observed in the XPS spectra, likely due to the small particle size and low concentration of the incorporated MgO. Furthermore, the FPI/MgO films exhibit smooth surfaces and retain excellent mechanical flexibility (Fig. S4 and 3d), indicating favorable structural integrity and processability. Cross-sectional SEM images of PI, FPI, and FPI/MgO films (Fig. S5 and 3e) reveal dense, defect-free morphologies with homogeneous dispersion of MgO nanoparticles within the FPI matrix. It should be noted that the high specific surface area of low-dimensional MgO nanoparticles can lead to slight agglomeration at higher loading concentrations, as observed in the FPI/MgO-0.25 film.
Fig. 3f presents the Fourier transform infrared spectroscopy (FTIR) spectra of PI, FPI and FPI/MgO films. Characteristic absorption peaks are observed at 1780 cm−1, 1720 cm−1 and 721 cm−1, corresponding to the symmetric stretching, asymmetric stretching, and bending vibrations of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups in the imide ring, respectively.31,32 The peak at 1365 cm−1 is attributed to the C–N stretching vibration of the imide moiety. In comparison with PI, all FPI films exhibit a new absorption band in the range of 950–1000 cm−1, assigned to the –CF3 groups. Furthermore, in the FTIR spectra of FPI/MgO films, the features between 600 and 800 cm−1 may be associated with Mg–O–Mg and Mg–O stretching vibrations.33
O groups in the imide ring, respectively.31,32 The peak at 1365 cm−1 is attributed to the C–N stretching vibration of the imide moiety. In comparison with PI, all FPI films exhibit a new absorption band in the range of 950–1000 cm−1, assigned to the –CF3 groups. Furthermore, in the FTIR spectra of FPI/MgO films, the features between 600 and 800 cm−1 may be associated with Mg–O–Mg and Mg–O stretching vibrations.33
It is noteworthy that although the bulky –CF3 groups weaken π–π conjugation, they also reduce the inter-molecular interactions between polymer chains, hence influencing the glass transition temperature (Tg) of FPI. As shown in Fig. 3g, FPI shows a lower Tg than PI, which can be ascribed to the increased free volume facilitating chain segment mobility. Interestingly, it is seen that the Tg of FPI/MgO films increases with increasing content of MgO nanoparticles. For instance, the Tg increases from 307.4 °C for FPI to 316.3 °C for FPI/MgO-0.25. This enhancement may be attributed to the following reason. The surface atoms of MgO are coordinatively unsaturated and can form weak interactions, such as van der Waals forces and dipole–dipole interactions, with the polar groups (e.g., C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) in the polymer chains.34,35 This spatial confinement reduces the free volume available for segmental motion, thereby markedly restraining the mobility of polymer chains at elevated temperatures. Despite the reduction in Tg, all FPI/MgO films retain a high Tg exceeding 300 °C, satisfying the thermal stability requirements for next-generation high-temperature energy storage devices. Moreover, the thermogravimetric (TGA) and dynamic mechanical (DMA) analysis curves presented in Fig. 3h and S6 demonstrate that the PI, FPI, and FPI/MgO films decompose only at above 530 °C and maintain exceptional mechanical integrity, indicating the excellent thermal stability at elevated temperatures. These thermal properties affirm that the developed FPI and FPI/MgO films are suitable for use in high-temperature energy storage and conversion applications.
O) in the polymer chains.34,35 This spatial confinement reduces the free volume available for segmental motion, thereby markedly restraining the mobility of polymer chains at elevated temperatures. Despite the reduction in Tg, all FPI/MgO films retain a high Tg exceeding 300 °C, satisfying the thermal stability requirements for next-generation high-temperature energy storage devices. Moreover, the thermogravimetric (TGA) and dynamic mechanical (DMA) analysis curves presented in Fig. 3h and S6 demonstrate that the PI, FPI, and FPI/MgO films decompose only at above 530 °C and maintain exceptional mechanical integrity, indicating the excellent thermal stability at elevated temperatures. These thermal properties affirm that the developed FPI and FPI/MgO films are suitable for use in high-temperature energy storage and conversion applications.
Fig. 4a presents the frequency-dependent dielectric properties of PI, FPI and FPI/MgO films measured at room temperature. The dielectric constant gradually decreases with increasing frequency, which can be attributed to the inability of dipole orientation polarization to follow the rapid alternation of the electric field at higher frequencies.36,37 At a fixed frequency, the dielectric constant of conventional PI is higher than that of FPI. This behavior can be explained as follows: (i) the introduction of the strongly electron-withdrawing –CF3 group reduces the electronic polarizability of FPI by modifying electron density distribution under an external electric field.29 (ii) The steric effect of the –CF3 group increases interchain spacing, thereby decreasing dipole density per unit volume. However, for the FPI/MgO films, the dielectric constant increases continuously with higher MgO filler content. This can be ascribed to the high dielectric constant of MgO (∼10) and the emergence of interfacial polarization at organic–inorganic interfaces.38,39 Furthermore, all samples exhibit low dielectric losses (<0.015) over the full range of frequencies, which is beneficial for maintaining high charge–discharge efficiency (η). In addition, the FPI and FPI/MgO films show lower dielectric loss compared to conventional PI, particularly at high frequencies (>104 Hz). This reduction can be attributed to: (i) the electron-withdrawing –CF3 groups suppressing delocalized π-electron migration along the conjugated aromatic backbones, thereby shortening conduction pathways and reducing bulk conductivity. (ii) The introduction of wide-bandgap MgO nanoparticles creates deep charge traps, inhibiting the de-trapping and migration of injected and thermally activated charges. Consequently, the constrained electron delocalization and diminished conduction loss are identified as key factors to reduce the dielectric loss at high frequencies.
The high-temperature dielectric stability is critical for practical applications. In this paper, the dielectric properties of PI, FPI and FPI/MgO films were evaluated over a frequency range of 102–106 Hz at 200 °C and across a temperature range of 25–200 °C at 1 kHz, as displayed in Fig. 4b and c. Notably, all samples manifest a stable dielectric constant and dielectric loss across broad frequency and temperature ranges, indicating their suitability for high-temperature capacitive energy storage.
Breakdown strength (Eb), a critical parameter for evaluating the dielectric withstand voltage performance of insulating films, was characterized using the two-parameter Weibull distribution for all samples. As shown in Fig. S7, 4d and e, the FPI/MgO films display significantly enhanced Eb compared to FPI and PI films. Among them, FPI/MgO-0.10 demonstrates the highest Eb across all tested temperatures. Specifically, at room temperature, the Eb of FPI/MgO-0.10 reaches 545 MV m−1, which is 32.0% higher than that of conventional PI (413 MV m−1). Even at 150 °C, FPI/MgO-0.10 maintains an outstanding Eb of 508 MV m−1, surpassing that of conventional PI (347 MV m−1) by 46.4%. The remarkable enhancement in breakdown strength is intimately associated with the following mechanisms. On the one hand, the –CF3 functional groups suppress both intra- and inter-molecular charge transfer, thereby diminishing the overall conductivity of the composite film. On the other hand, as a wide bandgap inorganic filler, MgO introduces deep traps at the interface between the inorganic filler and the polymer matrix, playing the role of scattering and trapping of charge carriers.16 However, it is noteworthy that the Eb of FPI/MgO-0.25 decreases to 466 MV m−1 at room temperature and 410 MV m−1 at 150 °C. This decline may be ascribed to the local electric field distortion between the organic–inorganic interface under high loading of MgO nanoparticles, which induces premature electrical breakdown.39 Despite the reduction in Eb at higher MgO content, FPI and FPI/MgO films still outperform the conventional PI film. This underscores the role of suppressed delocalized electron migration along conjugated aromatic rings and the introduction of deep charge traps in improving the electrical insulation performance of FPI/MgO films. Moreover, the higher shape parameter β of FPI/MgO films compared to that of FPI and PI films over the entire temperature range attests to the greater credibility of Eb.
To gain deeper insight into the enhanced Eb, the electrical conduction behavior of samples was investigated, as displayed in Fig. 4f. The leakage current densities of PI, FPI, and FPI/MgO-0.10 were measured at 150 °C. Clearly, FPI/MgO-0.10 exhibits the lowest leakage current density across the entire range of applied electric fields compared to FPI and PI. For instance, under an applied field of 200 MV m−1, the leakage current density for FPI/MgO-0.10 is 5.60 × 10−8 A cm−2, which is lower than the values of 1.09 × 10−7 A cm−2 for FPI and 2.50 × 10−7 A cm−2 for PI. These results indicate that the incorporation of –CF3 groups and MgO nanoparticles are conducive to significantly reducing the leakage current density and conduction loss at elevated temperatures. Previous research studies suggest that the introduction of deep charge traps can effectively impede charge carrier hopping and transport.23,26 So, to clarify the intrinsic conduction mechanism of samples, the current density-electric field curves were fitted using the hopping conduction model as follows:
In addition to the thermal runaway originating from a rapid increase in leakage current density, the electro-mechanical breakdown mechanism driven by coulombic forces is also recognized as a critical factor governing the breakdown strength (Eb) of polymer dielectric materials. According to Stark & Garton's theory, 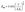 , where Eem, Y, εr and ε0 represent electro-mechanical/strength, the elastic modulus, the relative dielectric constant and the vacuum dielectric constant, respectively.40,41 Clearly, the enhanced Y enables the polymer dielectric to more effectively counteract the Coulomb forces resulting from charge accumulation at the metal–dielectric interface. Atomic force microscopy (AFM) is a powerful tool for characterizing the local surface properties of polymer films – including topography, surface potential, and mechanical properties – with high spatial resolution.42–44 To further elucidate the mechanism behind the variation in Eb from an electro-mechanical perspective, AFM was employed to probe the nanomechanical properties of PI, FPI, and FPI/MgO-0.10 films. As shown in Fig. 5a, all films exhibit a maximum height roughness of less than 2 nm within the scan area, indicating a sufficiently smooth interface for uniform distribution of space charge at the electrode–dielectric interface. Fig. 5b and c present the surface Y mappings and corresponding distribution histograms of the various films. Notably, the average surface Y of the FPI film slightly decreases to 2.18 GPa compared to 2.24 GPa for the conventional PI film, which can be ascribed to the incorporation of bulky –CF3 groups along the polymer backbone. These –CF3 groups increase the free volume and reduce intermolecular interactions. In contrast, the FPI/MgO-0.10 film shows a pronounced shift toward higher modulus values, with an average surface Y of 2.70 GPa. This improvement is attributed to (i) the inherent high stiffness of the MgO nanoparticles, which reinforces the film's mechanical structure.30 (ii) The enhanced intermolecular interactions facilitated by the interface between the FPI matrix and low-dimensional MgO nanoparticles. The increased surface Y of the FPI/MgO-0.10 film effectively suppresses electro-mechanical breakdown under high electric fields.
, where Eem, Y, εr and ε0 represent electro-mechanical/strength, the elastic modulus, the relative dielectric constant and the vacuum dielectric constant, respectively.40,41 Clearly, the enhanced Y enables the polymer dielectric to more effectively counteract the Coulomb forces resulting from charge accumulation at the metal–dielectric interface. Atomic force microscopy (AFM) is a powerful tool for characterizing the local surface properties of polymer films – including topography, surface potential, and mechanical properties – with high spatial resolution.42–44 To further elucidate the mechanism behind the variation in Eb from an electro-mechanical perspective, AFM was employed to probe the nanomechanical properties of PI, FPI, and FPI/MgO-0.10 films. As shown in Fig. 5a, all films exhibit a maximum height roughness of less than 2 nm within the scan area, indicating a sufficiently smooth interface for uniform distribution of space charge at the electrode–dielectric interface. Fig. 5b and c present the surface Y mappings and corresponding distribution histograms of the various films. Notably, the average surface Y of the FPI film slightly decreases to 2.18 GPa compared to 2.24 GPa for the conventional PI film, which can be ascribed to the incorporation of bulky –CF3 groups along the polymer backbone. These –CF3 groups increase the free volume and reduce intermolecular interactions. In contrast, the FPI/MgO-0.10 film shows a pronounced shift toward higher modulus values, with an average surface Y of 2.70 GPa. This improvement is attributed to (i) the inherent high stiffness of the MgO nanoparticles, which reinforces the film's mechanical structure.30 (ii) The enhanced intermolecular interactions facilitated by the interface between the FPI matrix and low-dimensional MgO nanoparticles. The increased surface Y of the FPI/MgO-0.10 film effectively suppresses electro-mechanical breakdown under high electric fields.
 | ||
| Fig. 5 (a) AFM surface morphology, (b) elastic modulus distribution maps and (c) corresponding modulus distribution histograms for PI, FPI and FPI/MgO-0.10 films. | ||
To further illuminate the breakdown mechanism of the FPI/MgO film, finite element simulations were performed to track the temporal evolution of breakdown pathways. As illustrated in Fig. 6a, the breakdown path in the conventional PI film develops almost perpendicular to the electric field direction and rapidly traverses the entire film, resulting in premature dielectric failure. In contrast, it is seen from Fig. 6b and c that the propagation of breakdown paths is markedly suppressed in FPI and FPI/MgO films. In particular, at identical simulation times, the growth of electrical trees is markedly slower, and the time required to form a complete breakdown path is substantially extended. The incorporation of MgO nanoparticles as a reinforcing phase further inhibits the progression of electrical trees. As a result, the trees develop pronounced branching when interacting with MgO nanoparticles, highlighting the effectiveness of wide-bandgap fillers in obstructing breakdown propagation. These simulation results are consistent with the experimental trends observed in Eb and leakage current density, corroborating the effectiveness of MgO nanoparticles in enhancing dielectric reliability.
 | ||
| Fig. 6 Simulated breakdown paths development as a function of time in (a) PI, (b) FPI and (c) FPI/MgO-0.10 films. | ||
The electric displacement–electric field (D–E) hysteresis loops of conventional PI, FPI and FPI/MgO films were measured to assess the energy storage performances at room temperature and 150 °C (Fig. S11–S13). As illustrated in Fig. 7a, FPI and FPI/MgO films display slimmer D–E hysteresis loops and lower remanent polarization under the same electric field compared to conventional PI, indicating a reduction in energy loss during charge–discharge cycles. Owing to this suppressed conduction loss and enhanced Eb, the FPI/MgO film exhibits a significant improvement in energy storage performance over FPI and conventional PI films. In particular, it is seen from Fig. 7b that the FPI/MgO-0.10 film shows a maximal Ud of 7.15 J cm−3 with η of 74.4% at room temperature, 250.9% higher than that of the conventional PI film (2.85 J cm−3).
As previously discussed, the –CF3 groups serve as localized charge traps, hindering the migration of delocalized electrons along aromatic chains. Meanwhile, the wide-bandgap MgO nanoparticles introduce deeper trap energy levels, thereby reducing the number of thermally activated charge carriers. These synergistic effects are conducive to reducing the leakage current density in FPI/MgO films at elevated temperatures. It is observed from Fig. 7c–e that the FPI/MgO-0.10 film maintains a lower remanent polarization and a slimmer D–E hysteresis loop compared to the other films at 150 °C. Accordingly, the FPI/MgO-0.10 film exhibits a maximal Ud of 4.97 J cm−3 with an η of 68.3% at 150 °C, representing a 332.2% improvement over that of conventional PI (1.15 J cm−3). Evidently, the prepared FPI/MgO films display noticeably improved energy storage performance, which stems from the tailored molecular structure of FPI and the incorporation of wide-bandgap MgO nanoparticles. Furthermore, the maximal Ud reported in this work compares competitively with those of other high-temperature polymer dielectric films. As shown in Fig. 7f, it is clearly seen that the maximal Ud of 4.97 J cm−3 achieved for the FPI/MgO-0.10 film at 150 °C significantly surpasses those of previously reported PI-based dielectric films, highlighting the promising application potential of FPI/MgO films as high-temperature polymer dielectrics.23,31,32,36–38,42,45–49Fig. 7g and h display the long-cycle capacitive performance of FPI/MgO-0.10 at 150 °C and 200 MV m−1. It can be seen that even after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles, it still maintains a relatively narrow D–E loop and stable Ud and η, indicating its long-term reliable stability at elevated temperatures.
000 charge–discharge cycles, it still maintains a relatively narrow D–E loop and stable Ud and η, indicating its long-term reliable stability at elevated temperatures.
4. Conclusion
In summary, this work successfully fabricates a series of novel FPI/MgO dielectric films incorporating trace amounts of MgO nanoparticles via a strategy based on molecular engineering and energy level modulation. The theoretical and experimental results demonstrate that the –CF3 groups in the FPI serve as localized charge traps, effectively inhibiting the transport of delocalized π-electrons along conjugated aromatic backbones. Furthermore, the introduction of MgO nanoparticles creates deeper charge-trap energy levels, which significantly restricts the migration and de-trapping of charge carriers. These synergistic effects effectively suppress the leakage current density and conduction loss, while simultaneously enhancing the breakdown strength (Eb) and capacitive energy storage performance over a wide temperature range. As a result, the FPI/MgO film with 0.10 wt% MgO nanoparticles achieves a high Eb of 508 MV m−1 and an outstanding discharge energy density (Ud) of 4.97 J cm−3 at 150 °C, far exceeding those of the conventional PI-based dielectric film. This research provides valuable insights into the molecular-level design of high-temperature PI dielectric films with superior energy storage performances.Author contributions
Guangyu Duan: funding acquisition, conceptualization, data curation, formal analysis, investigation, methodology, visualization, writing – original draft, writing – review and editing. Fengying Hu: validation, methodology. Yiran He: software, formal analysis, investigation. Zuming Hu: funding acquisition, formal analysis, writing-review and editing, project administration. Ming Tian: methodology, writing – review and editing. Junwei Zha: writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
All data are available within the manuscript and supplementary information (SI).Supplementary information: characterization section, simulation section, synthetic route of PI, molecular structures, optical band gaps, SEM images, cross-sectional images, DMA curves, calculation for the breakdown strength, O 1s orbital spectrum of MgO nanoparticles, UPS spectra, energy band diagrams, schematic D–E hysteresis loop of a dielectric material, and D–E hysteresis loops of PI, FPI and PFI/MgO at room temperature and 150 °C. See DOI: https://doi.org/10.1039/d5ta07395b.
Acknowledgements
The authors wish to express their gratitude to Yize Duan and for the financial support of the National Key R&D Program of China (2021YFB3700101), Henan Provincial Science and Technology Research Project (252102231078), and Key Research Project of Higher Education Institutions in Henan Province (25A430042). The authors acknowledge the assistance on structure characterization from the Analysis and Testing Center of Henan University of Engineering.References
- Z. Xie, L. Fan, H. Li, Z. Ran, S. Lai, X. Liu, A. Deatherage, Y. Wang, Q. Li, Y. Yin and Y. Liu, Prog. Polym. Sci., 2025, 164, 101957 CrossRef CAS.
- Q.-K. Feng, S.-L. Zhong, J.-Y. Pei, Y. Zhao, D.-L. Zhang, D.-F. Liu, Y.-X. Zhang and Z.-M. Dang, Chem. Rev., 2022, 122, 3820–3878 CrossRef CAS.
- M. Singh, S. K. Tiwary and A. Karim, Nat. Energy, 2024, 9, 113–114 CrossRef CAS.
- H. Li, Y. Zhou, Y. Liu, L. Li, Y. Liu and Q. Wang, Chem. Soc. Rev., 2021, 50, 6369–6400 RSC.
- Q. Xie, W. Liao, W. Gong, C. Huang, S. Huang and Q. Zhang, Nano Lett., 2025, 25, 7793–7800 CrossRef CAS PubMed.
- M. Yang, Y. Zhao, Z. Wang, H. Yan, Z. Liu, Q. Li and Z.-M. Dang, Energy Environ. Sci., 2024, 17, 1592–1602 RSC.
- Y. Guo, W. Zhao, D. Li, J. Liu, J. Qian, L. Pang, T. Zhou, W. Liu, Z. Liu, H. Huang, J. Zhai and D. Zhou, Adv. Mater., 2025, 37, 2415652 CrossRef CAS PubMed.
- Y. Zhou, Y. Chen, Y. Cui, Y. Li, Z. Li, C. Zhou, L. Cheng and W. Liu, Energy Storage Mater., 2024, 72, 103715 CrossRef.
- C. Yan, J. Zhou, A. Xu, H. Long, H. Luo and S. Chen, Small, 2025, 21, 2501050 CrossRef CAS PubMed.
- G. Duan, F. Hu, R. Zhang and Z. Hu, High Volt., 2023, 8, 630–639 CrossRef.
- W. Sima, Y. Mai, P. Sun, M. Yang, T. Yuan, B. Chen and Y. Yang, Energy Storage Mater., 2025, 74, 103974 CrossRef.
- C. Zhang, J. Zhang, T. Zhang, Q. Zhang, X. Tong, T. Zhang, Y. Shang, Z. Meng and Q. Chi, Energy Storage Mater., 2024, 72, 103757 CrossRef.
- D. Ai, Y. Han, Z. Xie, X. Pang, Y. Chang, H. Li, C. Wu, Y. Cheng and G. Wu, Nano Res., 2024, 17, 7746–7755 CrossRef CAS.
- D. Ai, H. Li, Y. Zhou, L. Ren, Z. Han, B. Yao, W. Zhou, L. Zhao, J. Xu and Q. Wang, Adv. Energy Mater., 2020, 10, 1903881 CrossRef CAS.
- P. Yin, Z. Shi, L. Sun, P. Xie, D. Dastan, K. Sun and R. Fan, Polym. Compos., 2021, 42, 3000–3010 CrossRef CAS.
- L. Ren, J. Qiao, C. Wang, W. Zheng, H. Li, X. Zhao, L. Yang and R. Liao, Mater. Today Energy, 2022, 30, 101161 CrossRef CAS.
- F. Zhang, X. Li, Z. Lan, N. Zhang, J. Yang and Y. Wang, Carbohydr. Polym., 2025, 366, 123934 CrossRef CAS PubMed.
- G. Duan, Y. Cao, J. Quan, Z. Hu, Y. Wang, J. Yu and J. Zhu, J. Mater. Sci., 2020, 55, 8170–8184 CrossRef CAS.
- H. Zhu, J. Tang, H. Chen, C. Chen and T. Yang, Compos. Commun., 2025, 57, 102475 CrossRef.
- Q. Yu, H. Wang and Y. Ma, Composites, Part B, 2025, 304, 112648 CrossRef CAS.
- J. Dong, L. Li, Y. Niu, Z. Pan, Y. Pan, L. Sun, L. Tan, Y. Liu, X. Xu, X. Guo, Q. Wang and H. Wang, Adv. Energy Mater., 2024, 14, 2303732 CrossRef CAS.
- J. Zhou, A. Xu, H. Tan, H. Zhong, L. Deng, H. Luo and S. Chen, J. Energy Storage, 2025, 105, 114789 CrossRef.
- A. Xu, J. Zhou, C. Yan, Z. Liu, H. Luo and S. Chen, J. Energy Storage, 2024, 86, 111209 CrossRef.
- Y. Wan, P. Hu, B. Sun, S. Shen, H. Luo, S. Zhang, D. Zhang, C.-W. Nan and Y. Shen, Adv. Funct. Mater., 2025, 35, 2506635 CrossRef CAS.
- G. He, H. Luo, Y. Liu, Y. Wan, B. Peng, D. Hu, F. Wang, X. Li, J. Peng, H. Wang and D. Zhang, Energy Environ. Sci., 2025, 18, 2405–2414 RSC.
- X. Yang, H. Luo, S. Chen, J. Peng, G. He, R. Guo and D. Zhang, Composites, Part A, 2025, 196, 109019 CrossRef CAS.
- X. Dong, Y. Wang, Y. Cao, N. Li, J. Fu, Y. Wang, J. Yu and Z. Hu, Chem. Eng. J., 2024, 500, 157312 CrossRef CAS.
- X. Yang, Y. Feng, P. Liu, L. Jiang, S. Zhang, Y. Wu and S. Li, Energy Storage Mater., 2025, 77, 104206 CrossRef.
- H. Ye, L. Chen, H. Jiang and L. Xu, ACS Appl. Polym. Mater., 2024, 6, 2524–2533 CrossRef CAS.
- N. Zhang, C. Zhang, H. Guo, J. Bai and H. Zhao, Chem. Eng. J., 2025, 508, 161058 CrossRef CAS.
- D. Ai, C. Wu, Y. Han, Y. Chang, Z. Xie, H. Yu, Y. Ma, Y. Cheng and G. Wu, J. Mater. Sci. Technol., 2025, 210, 170–178 CrossRef CAS.
- D. Wu, X. Zhao, X. Li, J. Dong and Q. Zhang, Compos. Commun., 2023, 42, 101653 CrossRef.
- S. Abbas, B. Uzair, S. Sajjad, S. A. K. Leghari, S. Noor, M. B. K. Niazi, I. Farooq and H. Iqbal, Arabian J. Sci. Eng., 2022, 47, 5895–5909 CrossRef CAS.
- Z. Gao, S. Cai, D. Min, Y. Hao, M. Ji, Q. Wu, S. Li and W. Liu, Surf. Interfaces, 2024, 44, 103807 CrossRef CAS.
- Y. Lin, L. Liu, G. Xu, D. Zhang, A. Guan and G. Wu, J. Phys. Chem. C, 2015, 119, 12956–12966 CrossRef CAS.
- K. Gao, F. Liu, F. Lv, N. Li, M. Liu, Z. Ye, M. Yu, R. Yin, C. Zhang, Y. Huang and W. Zhao, ACS Appl. Mater. Interfaces, 2025, 17, 2093–2104 CrossRef CAS.
- P. Yuan, R. Xue, Y. Wang, Y. Su, B. Zhao, C. Wu, W. An, W. Zhao, R. Ma and D. Hu, J. Colloid Interface Sci., 2024, 662, 1052–1062 CrossRef CAS.
- B. Sun, Z. Guo, J. Wang, Z. Zhao, T. Yu, Z. Shen, Y. Shen and P. Hu, Small Methods, 2025, 9, 2401059 CrossRef CAS PubMed.
- Z. Lin, W. Wang, Y. Su, N. Yang, Z. Zhang, Z. Liao, P. Dong, R. Zhang, D. Hu and H. Fan, ACS Appl. Mater. Interfaces, 2025, 17, 32838–32847 CrossRef CAS PubMed.
- J. Dong, L. Li, P. Qiu, Y. Pan, Y. Niu, L. Sun, Z. Pan, Y. Liu, L. Tan, X. Xu, C. Xu, G. Luo, Q. Wang and H. Wang, Adv. Mater., 2023, 35, 2211487 CrossRef CAS.
- Z. Chang, Z. Shi, Y. Liu, L. Lei, L. Sun, Q. Tang, R. Fan, H. Cui and H. Wang, Small, 2024, 20, 2407299 CrossRef CAS.
- G. Duan, F. Hu, Y. Wang, W. Shao, R. Xu, D. Lu and Z. Hu, ACS Appl. Nano Mater., 2025, 8, 543–551 CrossRef CAS.
- M. Shi, X. Li, Y. Jiang, S. Li, B. Li, X. Zhang, S. Zhang and C. W. Nan, Adv. Electron. Mater., 2023, 9, 2300187 CrossRef CAS.
- Z. Wang, Y. Zhao, M. Yang, H. Yan, C. Xu, B. Tian, C. Zhang, Q. Xie and Z. M. Dang, Adv. Energy Mater., 2025, 15, 2405411 CrossRef CAS.
- B. Y. Zhou, Z. T. Ma, S. L. Zhong, Z. J. Huang, Q. Guo, D. C. Feng, Z. K. Yuan, S. L. Lu, Y. Z. Yang, C. Wang, D. S. Yu and X. D. Chen, Chin. J. Polym. Sci., 2024, 42, 1038–1048 CrossRef CAS.
- Y. Chen, T. Yu, X. Chang, Z. Guo, Z. Zhao, Z. Huang and P. Hu, Chem. Eng. J., 2024, 497, 154779 CrossRef CAS.
- H. Qin, Z. Li, S. Qin, Y. Jiang, M. Liu, L. Zhou, S. Yu, S. Wang and C. Xiong, J. Power Sources, 2025, 627, 235849 CrossRef CAS.
- S. Peng, R. Wang, Z. Liang and X. Du, J. Energy Storage, 2024, 99, 113381 CrossRef.
- S. Yu, Y. Liu, C. Ding, X. Liu, Y. Liu, D. Wu, H. Luo and S. Chen, J. Energy Storage, 2023, 62, 106868 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |





