Novel soluble sulfonyl-containing conjugated polymers as highly efficient photocatalysts for CO2 reduction reaction
Cai
Cheng-Wei†
a,
Kuang-Hao
Cheng†
ab,
Palraj
Ranganathan†
a,
Kuei-Jhong
Lin
e,
Ching-I.
Huang
c,
Kun-Han
Lin
 e,
Jyh-Chien
Chen
e,
Jyh-Chien
Chen
 b and
Leeyih
Wang
b and
Leeyih
Wang
 *acd
*acd
aCenter for Condensed Matter Sciences, National Taiwan University, No. 1, Sec. 4, Roosevelt Rd, Taipei 10617, Taiwan. E-mail: leewang@ntu.edu.tw
bDepartment of Materials Science and Engineering, National Taiwan University of Science and Technology, 43, Sec. 4, Keelung Road, Taipei 10607, Taiwan
cInstitute of Polymer Science and Engineering, National Taiwan University, Taipei 10617, Taiwan
dCenter of Atomic Initiative for New Materials, National Taiwan University, Taipei 10617, Taiwan
eDepartment of Chemical Engineering, National Tsing Hua University, 101, Section 2, Kuang-Fu Road, Hsinchu 30044, Taiwan
First published on 19th November 2025
Abstract
Solution-processable polymeric photocatalysts offer a promising advantage for photocatalytic CO2 reduction reaction (CO2 RR), including tunable optoelectronic properties, structural versatility, and scalable fabrication. In this work, we developed two sulfonyl-functionalized acceptor monomers, 2,5-dibromo-3-hexylthiophene 1,1-dioxide and 3,7-dibromo-2,8-bis(octyloxy)dibenzo[b,d]thiophene 5,5-dioxide, both incorporating long alkyl side chains to improve solubility and processability, and copolymerized them with 2,5-bis(trimethylstannyl)thiophene to build donor–acceptor (D–A) type conjugated polymers, P3HTO-T and PDBTOOC8-T. For comparison, a sulfonyl-free P3HT-T analogue was also synthesized. These resulting polymers were used as photocatalysts for CO2 RR under solar light, without needing a co-catalyst or sacrificial agents, demonstrating exclusive CO production with no detectable H2 evolution, thereby highlighting their high selectivity for CO2 RR. Incorporating the sulfonyl group in the P3HTO-T and PDBTOOC8-T polymer structures markedly enhanced the key charge dynamics, as evidenced by prolonged exciton lifetime from time-resolved photoluminescence, amplified current response in transient photocurrent measurements, and markedly reduced charge-transfer resistance observed by electrochemical impedance spectroscopy. Interestingly, PDBTOOC8-T achieved the highest CO yield of 256.3 µmol g−1 h−1, while P3HTO-T delivered 134.2 µmol g−1 h−1, both vastly outperforming the sulfonyl-free control polymer P3HT-T (27.0 µmol g−1 h−1). Both sulfonyl-functionalized polymers maintained steady catalytic performance over 24 h during photocatalytic CO2 RR. This work highlights a critical role of sulfone functionalization in enhancing the performance of polymer photocatalysts, offering a new pathway for designing efficient organocatalysts for CO2 RR.
Introduction
Global warming, exacerbated by rising fossil-derived fuel consumption and greenhouse gas emissions, poses one of Earth's greatest critical ecological threats.1 In light of this, the quest for innovative strategies to convert CO2 into valuable chemicals and fuels has gained urgency as a vital measure to combat emissions. Photocatalytic processes powered by solar energy stand out as a promising and sustainable approach toward achieving this goal.2,3 Until now, extensive research has focused on inorganic materials as photocatalysts for CO2 reduction reactions (CO2 RR).4–6 Nevertheless, only a few metal-free organic photocatalysts have been reported for CO2 RR.7 Recently, linear and cross-linked polymers based on donor–acceptor (D–A) configurations, featuring an alternating arrangement of electron-rich and electron-deficient units, have garnered considerable attention in the current research.8–11 The inherent D–A structure facilitates effective charge separation and directs electron transport, promoting CO2 RR efficiency, making them potential next-generation multifunctional photocatalysts.12–14 A key challenge is that cross-linked porous photocatalysts are often synthesized as insoluble and unprocessable powders, restraining their practicality.15 For polymeric photocatalysts to be practically applied, they must be processable, enabling continuous and scalable photoreactions. Linear conjugated polymers (LCPs) are primarily solution-processable, showing promise for CO2 photoreduction applications, though research in this area remains in its infancy.16,17 Recently, our research group demonstrated the effectiveness of solution-processable naphthalene diimide-based D–A type LCPs and small molecules in photocatalytic CO2 RR applications.18,19 Similarly, McCulloch and co-workers developed solution-processable LCPs with rigid iptycene groups for gas phase CO2 photoreduction, achieving a maximum CO rate of 175 µmol h−1 m−2 with copper iodide as a co-catalyst, demonstrating sustained activity for over 100 hours.20 Nonetheless, the overall efficiencies of these materials must be enhanced for scalability, particularly in the absence of cocatalysts or sacrificial agents. Thus, the design and synthesis of novel D–A motif organic photocatalysts are of paramount importance.In the realm of D–A polymer photocatalysts, the role of acceptor units is as vital as that of donor units for achieving high performance.21–23 Within such D–A polymer systems, the contrasting electron affinities between donor and acceptor segments lead to a significantly stronger dipole moment along the D–A polymer backbone. During photocatalytic operation, this dipole could induce charge transfer from the electron-rich donor units to the electron-deficient acceptor units. The photoelectrons might preferentially accumulate on the acceptor moieties in the excited state, which would potentially enhance the separation of charge carriers.24 Therefore, the photoelectrons concentrated in the acceptor unit might contribute to promoting the CO2 RR. Following this strategy, only a handful of acceptors (e.g., benzothiadiazole, benzotriazole, and occasionally their derivatives) have been reported and focused on for CO2 RR.11,25–28 However, progress in developing novel acceptor units has lagged behind advancements in donor materials. Recently, dibenzothiophene-S,S-dioxide (DBTO) has emerged as a promising electron-accepting unit in polymer photocatalysts due to its strong electron-withdrawing characteristics, which facilitate efficient photoinduced charge separation.29–31 For instance, Cooper et al.30 pioneered its use by synthesizing DBTO-benzene copolymer (P7), which achieved a hydrogen (H2) evolution rate of 3.68 mmol h−1 g−1 under visible light. Since then, several research teams have synthesized DBTO-based polymer photocatalysts that outshone their DBTO-lacking counterparts.32 Building on this, Wang et al.33 explored the role of sulfone content in conjugated polymers, demonstrating that higher sulfone densities pointedly augment electron-capturing ability. This was supported by amplified SUMcharges-P values and greater electron density localized on the oxygen atoms of sulfone groups, leading to markedly improved photocatalytic activity for H2 evolution.
Beyond H2 production, DBTO-based polymers have shown strong potential in photocatalytic CO2 RR. Cooper's group later reported that both P7 and its homopolymer analogue P10 unveiled high CO evolution rates of 959.1 and 839.7 µmol g−1 h−1, respectively, under visible-light irradiation in the presence of CoCl2, 2,2′-bipyridine, and triethanolamine in a MeCN/H2O (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) medium.34 Despite these successes, this DBTO-based polymers often suffer from poor solubility, limited scalability, reliance on co-catalysts and sacrificial agents, and lacks a D–A structure, underscoring the need for solution-processable sulfone-containing systems that function efficiently under these additive-free conditions.
1) medium.34 Despite these successes, this DBTO-based polymers often suffer from poor solubility, limited scalability, reliance on co-catalysts and sacrificial agents, and lacks a D–A structure, underscoring the need for solution-processable sulfone-containing systems that function efficiently under these additive-free conditions.
To address these limitations, we focused on the strategic design of sulfone-based acceptor monomers that combine strong electron-withdrawing capacity with enhanced solubility and processability, which may facilitate the development of D–A type LCPs for efficient photocatalytic CO2 reduction. We hypothesized that introducing sulfonyl-based acceptor units featuring long alkyl side chains into the D–A conjugated polymer backbone can enhance photocatalytic efficiency for CO2 reduction while improving solubility and solution processability. Furthermore, we anticipate that functionalizing the DBTO core with electron-donating substituents, such as octyloxy groups, can increase the electron density localized on the sulfone oxygen atoms, which is favorable for improving the photocatalytic efficiency. Guided by this rationale, we designed and synthesized two novel sulfonyl-based monomers: 2,5-dibromo-3-hexylthiophene 1,1-dioxide (3HTO-2Br) and 3,7-dibromo-2,8-bis(octyloxy)dibenzo[b,d]thiophene 5,5-dioxide (DBTOOC8-2Br) as an acceptor unit. These monomers were synthesized via a sulfide oxidation approach from thiophene and DBT precursors. The introduction of sulfonyl groups increases the electron-deficient character of the core structure, while the addition of solubilizing hexyl and octyloxy side chains ensures compatibility with solution processing. This dual-functional design makes these acceptors particularly attractive for constructing high-performance conjugated polymer photocatalysts. We employed thiophene as the electron donor, chosen for its planar six-π-electron configuration that facilitates electron transfer to the conjugated backbone and enhances charge mobility.35,36 The thiophene donor unit was then coupled with the sulfonyl-based acceptor units (3HTO-2Br and DBTOOC8-2Br) to yield two new conjugated polymers: P3HTO-T and PDBTOOC8-T. A sulfonyl-free analogue, P3HT-T, was also synthesized for comparison purposes. Our results demonstrate that incorporating sulfonyl units into the P3HTO-T and PDBTOOC8-T leads to efficient photocatalysts for CO2 reduction without needing co-catalysts or sacrificial agents. Remarkably, both P3HTO-T and PDBTOOC8-T achieved significantly higher CO evolution rates (134.2 and 256.3 µmol g−1 h−1, respectively), compared to only 27.0 µmol g−1 h−1 for P3HT-T. This work represents the first demonstration of solution-processable sulfonyl-based acceptors integration onto LCPs to achieve high-performance photocatalytic CO2 RR.
Experimental
Materials
Starting materials, such as 2,5-dibromothiophene, 2,5-dibromo-3-hexylthiophene (3HT-2Br), and 2,8-dibenzo[b,d]thiophene (DBT), were sourced from commercial suppliers and used directly without purification. Additionally, all reagents and catalysts utilized in the reactions were acquired from Acros, Sigma-Aldrich, and other suppliers and were employed as received. The synthetic route to the monomers (2,5-dibromo-3-hexylthiophene 1,1-dioxide (2; 3HTO-2Br), 2,8-dibromodibenzo[b,d]thiophene (4), 2,8-dimethoxydibenzo[b,d]thiophene (5), dibenzo[b,d]thiophene-2,8-diol (6), 2,8-bis(octyloxy)dibenzo[b,d]thiophene (7), 2,8-bis(octyloxy)dibenzo[b,d]thiophene 5,5-dioxide (8), 3,7-dibromo-2,8-bis(octyloxy)dibenzo[b,d]thiophene 5,5-dioxide (9; DBTOOC8-2Br), and 2,5-bis(trimethylstannyl)thiophene (11; T-2tin)) is outlined in the SI, detailing the procedures and reagents used in their preparation.Synthesis of polymer
1H NMR (600 MHz, CDCl3), δ (ppm) = 7.15–7.02 (br, 3H), 2.81 (br, 2H), 1.72 (br, 2H), 1.45 (br, 2H), 1.37–1.28 (br, 4H), 0.94 (br, 3H).
1H NMR (600 MHz, CDCl3), δ (ppm) = 7.66–7.62 (br, 1H), 6.83–6.63 (br, 2H), 2.73–2.68 (br, 2H), 2.39–2.28 (br, 2H), 2.20–2.03 (br, 2H), 1.35–1.28 (br, 4H), 0.92 (br, 3H).
1H NMR (600 MHz, CDCl3), δ (ppm) = 6.977–6.878 (br, 4H), 5.959–5.797 (br, 2H), 3.687–3.597 (br, 4H), 1.788–1.716 (br, 4H), 1.438–1.282 (br, 20H), 0.972 (br, 6H).
Photocatalytic CO2 RR
In a standard photocatalytic experiment, 0.2 mg of polymer catalyst was dissolved in 1 mL of chloroform (CHCl3) and coated onto glass or molecular sieve substrates, followed by evaporation of the solvent via air drying. The coated substrate was placed in a custom photoreactor containing 0.5 mL of pure water, suspended at the bottom. The glass reactor (47 mL internal volume) with a defined illuminated area of 2 cm × 2 cm (4 cm2) was vacuumed three times and then backfilled with CO2 to a pressure of 800 torr at room temperature. Illumination was provided for 3 hours using an AM 1.5G solar simulator (intensity = 100 mW cm−2). After irradiation, 0.5 mL of the gaseous mixture was sampled for analysis with a Shimadzu gas chromatograph equipped with helium ionization (HID) and flame ionization (FID) detectors. The liquid reaction solution was subjected to liquid chromatography analysis. For isotope-labelled experiments, C18O2 and H218O were used in place of C16O2 and H216O, with gas products analysed by gas chromatography-mass spectrometry (GC-MS).Results and discussion
Molecular synthesis and characterization
The synthetic route for two novel sulfonyl-based acceptor monomers, namely 2,5-dibromo-3-hexylthiophene 1,1-dioxide (2; 3HTO-2Br) and 3,7-dibromo-2,8-bis(octyloxy)dibenzo[b,d]thiophene 5,5-dioxide (9; DBTOOC8-2Br), along with the donor monomer, 2,5-bis(trimethylstannyl)thiophene (11; T-2tin), is delineated in Scheme 1. As shown in Scheme 1a, 3HTO-2Br (2) was synthesized by the oxidation of 2,5-dibromo-3-hexylthiophene (1; 3HT-2Br) using meta-chloroperoxybenzoic acid (mCPBA) as the oxidizing agent. Meanwhile, Scheme 1b details the multi-step synthesis of DBTOOC8-2Br (9), starting from dibenzo[b,d]thiophene (3; DBT). Initial bromination of DBT (3) with Br2 in chloroform at room temperature yielded 2,8-dibromodibenzo[b,d]thiophene (4). Subsequent copper-catalyzed methoxylation of 4 afforded 2,8-dimethoxydibenzo[b,d]thiophene (5) in good yield (86.3%). Demethylation of 5 using boron tribromide in chloroform yielded dibenzo[b,d]thiophene-2,8-diol (6), which was further alkylated with 1-bromooctane in the presence of K2CO3 and 4-methyl-2-pentanone to afford 2,8-bis(octyloxy)dibenzo[b,d]thiophene (7). Oxidation of 7 using 30% aqueous hydrogen peroxide in acetic acid resulted in the formation of sulfone derivative, 2,8-bis(octyloxy)dibenzo[b,d]thiophene 5,5-dioxide (8). Finally, bromination of 8 with Br2 yielded the target monomer DBTOOC8-2Br (9) as a white powder in 71.9% yield. Scheme 1c outlines the synthesis of the donor monomer T-2tin (11) via lithiation of 2,5-dibromothiophene (10), followed by quenching with trimethyltin chloride. Detailed synthetic procedures for all these monomers are provided in Section S1 (SI), and their structures were confirmed by 1H NMR (see Fig. S1–S8, SI).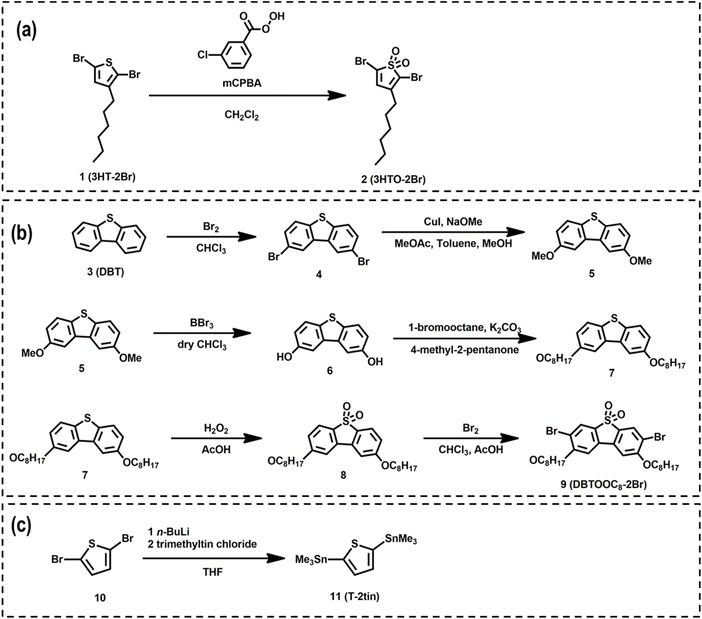 | ||
| Scheme 1 Synthetic steps and structures of the (a) 3HTO-2Br, (b) DBTOOC8-2Br, and (c) T-2tin monomers. | ||
As depicted in Scheme 2, target polymers P3HTO-T and PDBTOOC8-T were synthesized via coupling T-2tin (11) with sulfonyl-based monomers 3HTO-2Br (2) and DBTOOC8-2Br (9) through the Stille route, using Pd(PPh3)4 as the catalyst. For comparison, P3HT-T, a control polymer lacking the sulfonyl functionality, was also synthesized under identical conditions by coupling T-2tin (11) with 3HT-2Br (1), to systematically evaluate the impact of the sulfonyl group on photocatalytic CO2 RR. Purification of the resulting polymers was conducted by sequential Soxhlet extraction with methanol, acetone, n-hexane, and dichloromethane to eliminate oligomers and residual metals. The chemical structures of all polymers were affirmed by 1H NMR spectroscopy (Fig. S9–S11, SI), verifying successful synthesis. All polymer demonstrates excellent solubility in organic solvents such as chlorobenzene, chloroform, and tetrahydrofuran. Gel-permeation chromatography analysis in tetrahydrofuran eluent revealed that P3HT-T, P3HTO-T, and PDBTOOC8-T possessed number-average molecular weights (Mn) of 6.1, 5.4, and 5.4 kDa, weight-average molecular weights (Mw) of 10.7, 9.2, and 5.6 kDa, and polydispersity indices (Đ) of 1.75, 1.70, and 1.04, respectively (Table S1, SI).
Energy level calculation
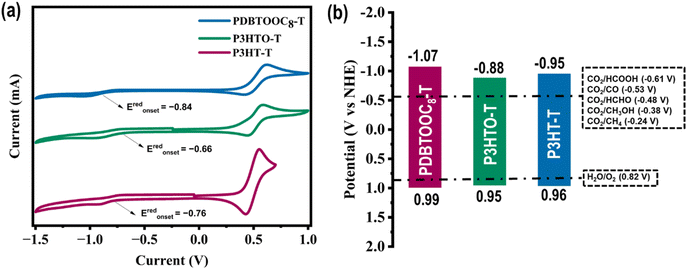
The molecular energy levels of P3HT-T, P3HTO-T, and PDBTOOC8-T were determined using both cyclic voltammetry (CV) and UV-visible absorption spectroscopy (UV-vis). Electrochemical measurements were performed using a standard three-electrode setup, with ferrocene/ferrocenium (Fc/Fc+) as the internal standard and 0.1 M tetrabutylammonium hexafluorophosphate (TBAPF6) in ultra-dried acetonitrile as the supporting electrolyte. The results obtained from the CV analysis (Fig. 1a) are summarized in Table S2 (SI). The onset reduction potential (Eredonset) found from the CV curves, shown in Fig. 1a, were −0.76, −0.66, and −0.84 V (vs. Ag/AgCl) for P3HT-T, P3HTO-T, and PDBTOOC8-T, respectively. Utilizing these values, the LUMO energy levels (vs. vacuum) were calculated using the equation: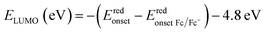 , where
, where 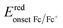 is the onset reduction potential of Fc/Fc+ (vs. Ag/AgCl), summarized in Table S2 (SI). Applying this equation yielded LUMO energy levels of −3.55 eV (P3HT-T), −3.62 eV (P3HTO-T), and −3.43 eV (PDBTOOC8-T) (vs. vacuum). To facilitate comparison with redox potential commonly used in CO2 photocatalysis, the LUMO values were also converted to the normal hydrogen electrode (NHE) scale using ELUMO (V vs. NHE) = −ELUMO (eV vs. vacuum) − 4.5, resulting LUMO values of −0.95 V (P3HT-T), −0.88 V (P3HTO-T), and −1.07 V (PDBTOOC8-T) vs. NHE.37
is the onset reduction potential of Fc/Fc+ (vs. Ag/AgCl), summarized in Table S2 (SI). Applying this equation yielded LUMO energy levels of −3.55 eV (P3HT-T), −3.62 eV (P3HTO-T), and −3.43 eV (PDBTOOC8-T) (vs. vacuum). To facilitate comparison with redox potential commonly used in CO2 photocatalysis, the LUMO values were also converted to the normal hydrogen electrode (NHE) scale using ELUMO (V vs. NHE) = −ELUMO (eV vs. vacuum) − 4.5, resulting LUMO values of −0.95 V (P3HT-T), −0.88 V (P3HTO-T), and −1.07 V (PDBTOOC8-T) vs. NHE.37
The optical bandgap (Eg) of the polymers was derived from the onset absorption wavelengths (λonset) of their thin-film UV-vis spectra, as presented in Fig. S12 (SI), using the formula Eg = 1240/λonset. The calculated Eg values were 1.91 eV (P3HT-T), 1.83 eV (P3HTO-T), and 2.06 eV (PDBTOOC8-T). The corresponding λonset values were 649 nm (P3HT-T), 677 nm (P3HTO-T), and 601 nm (PDBTOOC8-T), while their maximum absorption peaks (λmax) appeared at 536, 483, and 442 nm, respectively. Finally, the highest occupied molecular orbital energy levels (HOMO) vs. NHE were estimated by subtracting the Eg from the LUMO levels, based on EHOMO = Eg − ELUMO, giving HOMO values of 0.96 V (P3HT-T), 0.95 (P3HTO-T), and 0.99 (PDBTOOC8-T) vs. NHE, respectively.
The differences in energy levels among the polymers arise from their structural modifications. P3HT-T, composed entirely of thiophene units, represents a donor-only type structure, exhibiting a relatively higher LUMO level (−0.95 V vs. NHE) due to the absence of any pronounced electron-withdrawing groups. Rather, the introduction of a sulfone moiety in P3HTO-T introduces strong electron-withdrawing character, effectively deepens the LUMO to −0.88 V vs. NHE, and narrows the bandgap to 1.83 eV. In PDBTOOC8-T, the introduction of the DBTOOC8-2Br unit, comprising a thiophene fused with a benzene ring, has the largest twisted angle, which shortens the conjugation length, leading to a wider bandgap (2.06 eV) due to the reduced charge transfer ability. Simultaneously, the integration of octyloxy side chains, acting as electron-donating groups, elevates the PDBTOOC8-T polymer LUMO energy, leading to a wider Eg. The combined effect of shorter conjugation length and electron-donating octyloxyl side-chain substituents accounts for the wider Eg observed in PDBTOOC8-T relative to P3HT-T and P3HTO-T. Fig. 1b illustrate the energy band diagram of the polymers, demonstrating that these polymers are theoretically well-suited for photocatalytic CO2 reduction and H2O oxidation.
Photocatalytic performance for CO2 reduction
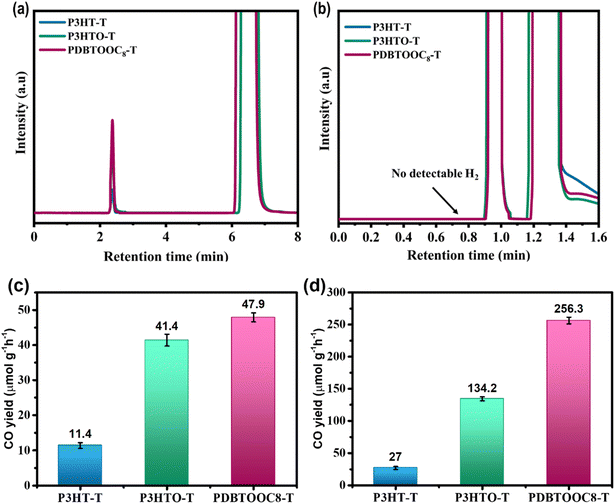
Encouraged by the favourable band alignments, we conducted CO2 RR experiments using P3HT-T, P3HTO-T, and PDBTOOC8-T as photocatalysts. The polymers were initially applied as films, prepared by drop-casting from CHCl3 onto glass substrates and placed within a sealed chamber containing 0.5 mL of H2O at the base. The chamber was subsequently charged with CO2 to a pressure of 880 torr and irradiated under simulated solar light (AM 1.5G, 100 mW cm−2) for 3 h. No co-catalysts, photosensitizers, or sacrificial agents were utilized in this reaction. Post-irradiation, the gaseous and aqueous portions were analysed by gas chromatography (GC) equipped with flame ionization (GC-FID) and helium ionization (GC-HID) detectors. No products were detected in the aqueous phase. To accurately identify the retention times of gaseous products after photocatalysis, a certified standard gas mixture containing H2, CO, and CH4 was first analysed using GC-FID and GC-HID detectors. The resulting chromatograms, shown in Fig. S13a and b (SI), were used as references for assigning product peaks. As shown in Fig. 2a, the GC profiles confirmed that CO was the sole detectable product from photocatalytic CO2 reduction, exhibiting a sharp, high-resolution peak that precisely matched the retention time (2.3 min) of the standard CO signal. Importantly, no signal was detected at the retention time corresponding to H2 (0.75 min), as illustrated in Fig. 2b, affirming that H2 evolution was effectively suppressed under the employed photocatalytic conditions. The N2/O2 peaks represent instrumental background signals from the GC system. To accurately quantify the amount of CO produced during photocatalysis, calibration curves were established by plotting the GC peak areas against known CO concentrations from three certified standard CO gas cylinders (Fig. S14, SI). The CO yield for each polymer was then determined by integrating the GC signal area of CO and referencing the corresponding calibration curve. The calculated CO yields, summarized in Fig. 2c, enable direct comparison of the photocatalytic activity among the different polymer systems. We exclusively observed CO as the reduction product, with no detectable formation of H2, CH4, or any liquid-phase species.Fig. 2c shows the average CO yield of the P3HT-T, P3HTO-T, and PDBTOOC8-T polymers, each coated on a glass substrate. The photocatalytic activity of P3HT-T was lowest among the tested polymers, with a CO yield of 11.4 ± 0.8 µmol g−1 h−1. The illuminated area for each individual reaction is detailed in Tables S3 and S4 (SI). Intriguingly, the synthesized P3HTO-T, which features a sulfonyl group, achieved a CO yield of 41.4 ± 1.7 µmol g−1 h−1, marking a 3.6-fold increase compared to the CO yield of the structurally similar P3HT-T without the sulfonyl group. Such a significant increase could reveal that sulfonyl groups are beneficial for improving the photocatalytic CO2 reduction activity of P3HTO-T. Remarkably, PDBTOOC8-T achieved the highest efficiency among the tested polymers, with a CO yield of 47.9 ± 1.3 µmol g−1 h−1, which is approximately 4.2 times higher than that of P3HT-T. Its superior performance is attributed to the sulfonyl acceptor unit combined with an octyloxy side chain, which enhances electron density and transport.
The superior solubility of these polymers in organic solvents allows for their application onto porous substrates, increasing the interface with CO2. Accordingly, we coated these polymers onto a porous molecular sieve (MS) substrate, which was subsequently utilized for CO2 RR. After 3 hours of irradiation, no products were noticed from the pure MS substrates, verifying that the CO2 reduction activity was solely due to the coated polymers (Fig. S15, SI). As shown in Fig. 2d, the photocatalytic behavior of the P3HT-T, P3HTO-T, and PDBTOOC8-T polymers on the MS substrate followed similar trends to those on the glass substrate. Under the simulated solar light, the CO production yield pointedly increased to 27.0 ± 2.3, 134.2 ± 3.1, and 256.3 ± 5.2 µmol g−1 h−1P3HT-T, P3HTO-T, and PDBTOOC8-T, respectively, after 3 h in pure H2O (0.5 mL). Enhanced CO production witnessed with all three polymers on the MS substrate can be attributed to the larger interfacial area provided by the porous material, compared to the glass substrate. Notably, PDBTOOC8-T achieves a higher CO production than hitherto reported LCPs (Table S5, SI), positioning it as a strong contender. These findings mark a significant advancement in photocatalytic CO2 RR, demonstrating that polymers incorporating sulfonyl-functionalized acceptor units combined with octyloxy side chains can serve as highly effective catalysts in this field.
Recent studies have suggested that residual Pd in polymer matrices might enhance CO2 reduction as a co-catalyst.38,39 To evaluate this, we analyzed the residual Pd and Sn content in three polymers using inductively coupled plasma mass spectrometry (ICP-MS). The residual Pd levels were found to be 5.9 ppm in P3HTO-T, 2.4 ppm in PDBTOOC8-T, and 6.1 ppm in P3HT-T, while the residual Sn levels were 2.5 ppm, 1.6 ppm, and 26.6 ppm for the respective polymers. It can be clear that the PDBTOOC8-T proved the superior photocatalytic performance compared with both P3HT-T and P3HTO-T, despite having the lowest residual Pd and Sn content. Disparities in photocatalytic activity can be ascribed not only to structural variations in the polymer backbone and the nature of the acceptor units, but also to the synergistic influence of residual Pd species.40,41 Our results suggest that Pd acts as an auxiliary co-catalyst that facilitates charge transfer and surface reaction kinetics; however, a higher Pd content does not necessarily lead to superior activity. The most efficient photocatalyst in our series contains only a moderate Pd level, indicating that intrinsic factors—such as the sulfone-mediated enhancement of CO2 adsorption and the superior optoelectronic characteristics of PDBTOOC8-T—play a more decisive role. These mechanistic insights are further supported by our simulation results and are discussed in detail in the Theoretical investigation section.
Control and isotopic experiments were conducted using the top-performing photocatalysts, P3HTO-T and PDBTOOC8-T, to pinpoint the source of CO. No CO was generated in the absence of light or photocatalyst, demonstrating that light alone does not drive CO formation (Fig. S15, SI). These results confirm that the observed CO2 reduction with H2O under light is indeed a photocatalytic process involving the polymers. Further validation was achieved through isotope-labelling experiments, where C18O2 and H218O were utilized instead of C16O2 and H216O to trace the carbon source. Analysis by GC-MS revealed a prominent peak at m/z = 30, corresponding to C18O, confirming that the CO originates from the CO2 reactant under AM 1.5 irradiation, rather than from degradation of the photocatalyst (Fig. 3a and b). We also did the reaction with 13CO2 instead of natural-abundance CO2 and observed the formation of 13CO (m/z = 29) by GC-MS, confirming that CO originates from CO2 reduction rather than polymer decomposition (Fig. S16, SI).
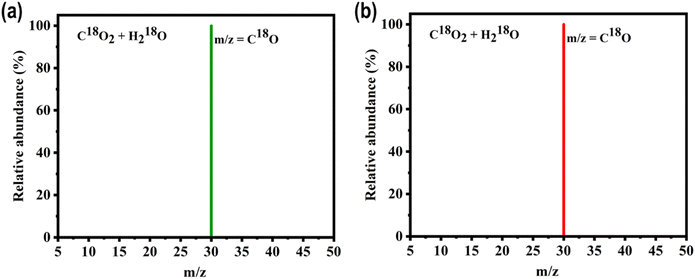 | ||
| Fig. 3 Mass spectra of products generated from CO2 RR utilizing (a) P3HTO-T and (b) PDBTOOC8-T as catalysts, with C18O2 and H218O as reactants. | ||
We further evaluated the apparent quantum yield (AQY) of the top-performing polymer PDBTOOC8-T under various light wavelengths to assess its potential for solar-to-chemical energy conversion. Fig. S17 (SI) shows the AQY values as a function of wavelength, with PDBTOOC8-T achieving a peak AQY of 0.012% at 430 nm. A notable decline in AQY with increasing wavelengths reflects diminished light absorption, a trend corroborated by the corresponding UV-vis absorption spectra, reaffirming that the CO2 RR process is driven by light absorption.
Photocatalytic mechanism analysis
To comprehend the divergences in photocatalytic CO2 reduction activity among the P3HT-T, P3HTO-T, and PDBTOOC8-T polymers, we cautiously evaluated their charge dissociation and migration attributes employing time-resolved photoluminescence (TRPL) (Fig. 4a), transient photocurrent analysis (Fig. 4b), and electrochemical impedance spectroscopy (EIS) (Fig. 4c). The TRPL lifetime for each polymer was measured at an excitation wavelength of 460 nm (Fig. 4a), with the average values calculated from the fitted PL decay curves listed in Table 1. Herein, the short lifetime (τ1) is associated with nonradiative relaxation processes (Sn → S1), whereas the long lifetime (τ2) corresponds to the recombination of separated charge carriers (Sn → S0).42P3HT-T and P3HTO-T exhibit τ1 values of 0.42 ns and 0.49 ns, and τ2 values of 1.37 ns and 1.89 ns, respectively. The increase in both τ1 and τ2 for P3HTO-T suggests that the sulfonyl group reduces nonradiative decay and prolongs exciton lifetimes, resulting in improved charge separation and slower recombination of photogenerated charge carriers. PDBTOOC8-T further prolongs exciton lifetimes, with τ1 and τ2 values of 2.77 ns and 5.66 ns, respectively, demonstrating that it affords greater opportunities for free charge carriers (electrons and holes) to partake in surface photo redox reactions before recombination, which is advantageous for enhancing CO2 RR.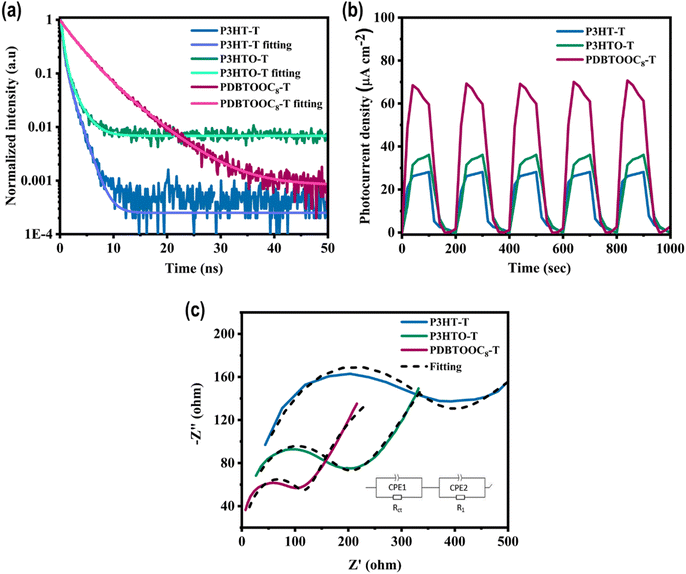 | ||
| Fig. 4 (a) TR-PL intensity decay spectra, (b) TPC response, and (c) EIS Nyquist plots of P3HT-T, P3HTO-T, and PDBTOOC8-T polymers. | ||
| Polymers | A 1 (%) | τ 1 (ns) | A 2 (%) | τ 2 (ns) |
|---|---|---|---|---|
| P3HT-T | 54.7 | 0.42 | 45.3 | 1.37 |
| P3HTO-T | 58.2 | 0.49 | 41.8 | 1.83 |
| PDBTOOC8-T | 54.5 | 2.77 | 45.5 | 5.66 |
The photocurrent response of the three polymers was monitored under cyclic on/off visible light irradiation (Fig. 4b). Among the samples, P3HT-T exhibited the lowest photocurrent density. After introducing the sulfone units, a higher photocurrent density was observed for P3HTO-T and PDBTOOC8-T compared to P3HT-T. Remarkably, PDBTOOC8-T displayed the highest photocurrent density among the three polymers. The significantly higher photocurrent in PDBTOOC8-T confirms that photoinduced charge carriers are more effectively generated and transferred within this polymer, which directly correlates with its improved CO2 RR performance. Additionally, EIS was carried out on three polymers to get deeper insights into the charge transfer resistances and transport dynamics under solid–gas phase photocatalytic CO2 RR. The resulting Nyquist plots (Fig. 4c) were fitted using an equivalent circuit model shown in the inset of Fig. 4c, which consists of charge transfer resistance (Rct), ionic diffusion resistance (R1), and two constant phase elements (CPE1 and CPE2).43,44 In this circuit, Rct explicitly represents the resistance to charge transfer between the photocatalyst surface and adsorbed reactants (e.g., CO2 and H2O). Generally, a smaller diameter of the Nyquist semicircle directly corresponds to a lower Rct, signifying more efficient interfacial charge transfer dynamics. As depicted in Table S6 (SI), the Rct trends follow: PDBTOOC8-T < P3HTO-T < P3HT-T. Notably, PDBTOOC8-T and P3HTO-T exhibit significantly lower Rct values of 106.5 ohms and 156.9 ohms, respectively, compared to P3HT-T (323.0 ohms). The significantly reduced Rct of PDBTOOC8-T indicates more efficiency for the transport of charge carriers, contributing to its superior photocatalytic CO2 reduction performance.
Stability tests
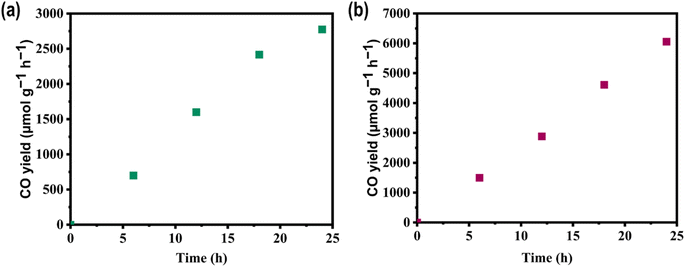
For practical photocatalytic CO2 reduction, a catalyst with firm and durable performance is needed. Building on the remarkable photocatalytic activity observed, we investigated the stability of the top-performing catalysts, P3HTO-T and PDBTOOC8-T. Fig. 5a and b illustrate a steady increase in CO generation over time, with both catalysts maintaining consistent CO output during a 24-hour visible light test. Such results demonstrate the exceptional catalytic stability of P3HTO-T and PDBTOOC8-T, confirming their structural robustness and emphasizing their suitability for sustained CO2 RR applications.Theoretical investigation
To elucidate how molecular design modulates electronic structure and thereby photocatalytic activity, we conducted computations using density functional theory (DFT), a machine-learning-assisted conformer search, and DLPNO-CCSD(T). The frontier orbitals and energies of the constituent molecular building blocks are summarized in Fig. S18 and S19 (SI). Comparing the LUMO levels shows that incorporating sulfone groups into the backbone lowers the LUMO, a trend consistent with our monomer and dimer calculations (Fig. S20, SI). However, the experimentally measured LUMO of P3HT-T is not the highest, seemingly at odds with computation. This discrepancy is plausibly due to differences in degree of polymerization across the three polymers, which may alter conjugation length—another key determinant of HOMO/LUMO positions.We next assessed CO2 physical binding affinity across different functional groups using monomer models of the three polymers (3HT, 3HTO, and DBTOOC). Preliminary conformers search was performed with the AIMNet2-wB97MD3 potential (see SI), and the analysis is summarized in Fig. S21–S23 (SI). The sulfone motif binds CO2 slightly more strongly than other sites such as thiophene S or methoxy O, likely because the more negatively charged sulfone O atoms electrostatically attract the partially positive carbon of CO2 (Fig. S24). Across all sites, DFT-computed adsorption energies are around −0.1 to −0.2 eV, corresponding to weak physical adsorption of CO2.
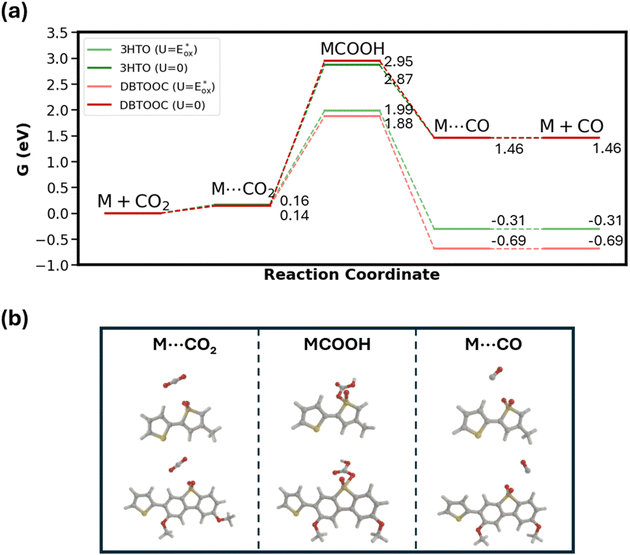
Finally, to identify active sites for CO2 photoreduction, we computed Gibbs free-energy diagrams at U = 0 and 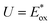 (photocatalytic conditions; see SI), shown in Fig. 6. Among the three polymers, P3HT-T cannot form a stable MCOOH intermediate and is therefore omitted from the diagram. A stable MCOOH forms only in the presence of sulfone groups. The highest barrier occurs at the MCOOH formation, ∼3 eV at U = 0 and ∼2 eV at
(photocatalytic conditions; see SI), shown in Fig. 6. Among the three polymers, P3HT-T cannot form a stable MCOOH intermediate and is therefore omitted from the diagram. A stable MCOOH forms only in the presence of sulfone groups. The highest barrier occurs at the MCOOH formation, ∼3 eV at U = 0 and ∼2 eV at 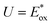 . This is substantially larger than on Pd, where the rate-limiting step is CO desorption (barrier ∼1 eV at U = 0 and pH = 7).45 These results indicate that, although sulfones can stabilize MCOOH, Pd more plausibly serves as the true active site. Although sulfones are not catalytic sites, they have been reported to facilitate electron transfer from the polymer to Pd, thereby enhancing photocatalytic H2 evolution.46 Taken together, we attribute the higher CO2-reduction activity of sulfone-functionalized polymers primarily to improved electron transfer from sulfone to Pd, rather than to sulfone groups acting as intrinsic catalytic sites. The superior performance of PDBTOOC8-T over P3HTO-T—despite both containing sulfones—may stem from the longer excited-state lifetime of PDBTOOC8-T.
. This is substantially larger than on Pd, where the rate-limiting step is CO desorption (barrier ∼1 eV at U = 0 and pH = 7).45 These results indicate that, although sulfones can stabilize MCOOH, Pd more plausibly serves as the true active site. Although sulfones are not catalytic sites, they have been reported to facilitate electron transfer from the polymer to Pd, thereby enhancing photocatalytic H2 evolution.46 Taken together, we attribute the higher CO2-reduction activity of sulfone-functionalized polymers primarily to improved electron transfer from sulfone to Pd, rather than to sulfone groups acting as intrinsic catalytic sites. The superior performance of PDBTOOC8-T over P3HTO-T—despite both containing sulfones—may stem from the longer excited-state lifetime of PDBTOOC8-T.
Conclusions
This study successfully demonstrates sulfone functionalization as a powerful strategy to enhance the photocatalytic performance of D–A type LCP materials for CO2 RR. We successfully synthesized two sulfonyl-functionalized acceptor monomers, 3HTO-2Br (2) and DBTOOC8-2Br (9), and copolymerized them with T-2tin (11) to construct two D–A type conjugated polymers: P3HTO-T and PDBTOOC8-T. The inclusion of electron-donating octyloxy side chains in PDBTOOC8-T not only enhanced solubility but also supported electron donation to the sulfone acceptor unit, benefiting photocatalytic performance. Photophysical and electrochemical analyses, including time-resolved photoluminescence (TR-PL), transient photocurrent (TPC), and electrochemical impedance spectroscopy (EIS), revealed that sulfonyl groups significantly prolong exciton lifetime, promote charge transport, and mitigate interfacial resistance. These polymers were also solution processed on glass and porous molecular sieves, making them suitable for practical applications. Both P3HTO-T and PDBTOOC8-T efficiently catalyzed CO2 reduction, producing CO as the sole product without intentional sacrificial agents or co-catalysts. As thin films on glass, PDBTOOC8-T exhibited the best performance (47.9 µmol g−1 h−1), while P3HTO-T gave 41.4 µmol g−1 h−1, both exceeding the sulfone-free control P3HT-T (11.4 µmol g−1 h−1). When deposited onto porous molecular sieves to increase the catalyst/CO2 interface, their performance was further enhanced, with PDBTOOC8-T reaching the highest CO yield at 256.3 µmol g−1 h−1, followed by P3HTO-T at 134.2 µmol g−1 h−1, whereas P3HT-T remained low at 27.0 µmol g−1 h−1. Our computations further indicate that sulfone units promote electron transfer to Pd, enabling a Pd-assisted pathway that lowers the barrier for *COOH formation; thus, residual metal can influence the observed activity. Nevertheless, activity does not scale monotonically with Pd content, and PDBTOOC8-T remains most favorable owing to its sulfone-enabled CO2 proximity and superior electronic structure. Remarkably, both P3HTO-T and PDBTOOC8-T showed stable catalytic performance for up to 24 hours. This study underscores the effectiveness of sulfonyl functionalization combined with tailored side chains for creating efficient, solution-processable photocatalysts, offering new pathways for developing advanced materials for CO2 reduction.Author contributions
Kuang-Hao Cheng: data curation, formal analysis, writing – original draft. Cai Cheng-Wei: data curation, formal analysis, writing – original draft. Palraj Ranganathan: data curation, formal analysis, writing – original draft. Ching-I Huang: investigation. Jyh-Chien Chen: formal analysis, investigation. Leeyih Wang: conceptualization, formal analysis, supervision, writing – review & editing, funding acquisition. Kuei-Jhong Lin: data curation, DFT simulation, Kun-Han Lin: data curation, DFT simulation.Conflicts of interest
There are no conflicts to declare.Data availability
Should any raw data files be needed in another format they are available from the corresponding author upon reasonable request. Source data are provided with this paper.The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information (SI). Supplementary information: experimental details, NMR data, long-term stability, performance comparison. See DOI: https://doi.org/10.1039/d5ta07296d.
Acknowledgements
This research received financial support from the National Science and Technology Council of Taiwan (NSTC 111-2113-M-002-009, 111-2113-M-002-022, and 113-2221-E-011-009), Dragon Gate Program (112-2926-I-002-514-G) and the Center of Atomic Initiative for New Materials, National Taiwan University through the Featured Areas Research Center Program under the Higher Education Sprout Project by the Ministry of Education, Taiwan (113 L9008 and 111 668 L9008) is acknowledged. The authors gratefully acknowledge the use of facilities and instrumentation at the Instrumentation Center of National Taiwan Normal University (NTNU) (Bruker AVIII HD-600/NMR000400) and the MOST Instrumental Center at National Yang Ming Chiao Tung University (NYCU) (Trace 1300GC+ ISQ MS+ auto-sampler TriPlus RSH and NIST2011 + Wiley 10th edition), Hsinchu, Taiwan. K.-H. L. acknowledges the financial support from the National Science and Technology Council, Taiwan (NSTC 114-2628-E-007-002) and the computational and storage resources from National Center for High-performance Computing (NCHC) of National Institutes of Applied Research (NIAR) of Taiwan.References
- P. Gabrielli, L. Rosa, M. Gazzani, R. Meys, A. Bardow, M. Mazzotti and G. Sansavini, One Earth, 2023, 6, 682–704 CrossRef
.
- S. Fang, M. Rahaman, J. Bharti, E. Reisner, M. Robert, G. A. Ozin and Y. H. Hu, Nat. Rev. Methods Primers, 2023, 3, 61 CrossRef CAS
.
- J. Lv, J. Xie, A. G. A. Mohamed, X. Zhang, Y. Feng, L. Jiao, E. Zhou, D. Yuan and Y. Wang, Nat. Rev. Chem., 2023, 7, 91–105 CrossRef PubMed
.
- H.-J. Son, C. Pac and S. O. Kang, Acc. Chem. Res., 2021, 54, 4530–4544 CrossRef CAS PubMed
.
- S. Trivedi, D. Prochowicz, A. Kalam, M. M. Tavakoli and P. Yadav, Renewable Sustainable Energy Rev., 2021, 145, 111047 CrossRef CAS
.
- H. L. Nguyen, Adv. Energy Mater., 2020, 10, 2002091 CrossRef CAS
.
- H. Shen, T. Peppel, J. Strunk and Z. Sun, Sol. RRL, 2020, 4, 1900546 CrossRef CAS
.
- Y. Jiao, L. Đorđević, H. Mao, R. M. Young, T. Jaynes, H. Chen, Y. Qiu, K. Cai, L. Zhang, X.-Y. Chen, Y. Feng, M. R. Wasielewski, S. I. Stupp and J. F. Stoddart, J. Am. Chem. Soc., 2021, 143, 8000–8010 CrossRef CAS PubMed
.
- T. Banerjee, F. Podjaski, J. Kröger, B. P. Biswal and B. V. Lotsch, Nat. Rev. Mater., 2021, 6, 168–190 CrossRef CAS
.
- P. Roy, A. Jha, V. B. Yasarapudi, T. Ram, B. Puttaraju, S. Patil and J. Dasgupta, Nat. Commun., 2017, 8, 1716 CrossRef PubMed
.
- C. Yang, B. Cheng, J. Xu, J. Yu and S. Cao, EnergyChem, 2024, 6, 100116 CrossRef CAS
.
- S. Xiang, C. Han, C. Shu, C. Zhang and J.-X. Jiang, Sci. China Mater., 2022, 65, 422–430 CrossRef CAS
.
- Y. Qian, Y. Han, X. Zhang, G. Yang, G. Zhang and H.-L. Jiang, Nat. Commun., 2023, 14, 3083 CrossRef CAS PubMed
.
- D. Ye, L. Liu, Y. Zhang, J. Qiu, Z. Tan, Y. Xing and S. Liu, Molecules, 2023, 28, 2203 CrossRef CAS PubMed
.
- R. Y. Liu, S. Guo, S.-X. L. Luo and T. M. Swager, Nat. Commun., 2022, 13, 2775 CrossRef CAS PubMed
.
- B. Xu and J. Hou, Adv. Energy Mater., 2018, 8, 1800022 CrossRef
.
- P. Zhao, L. Wang, Y. Wu, T. Yang, Y. Ding, H. G. Yang and A. Hu, Macromolecules, 2019, 52, 4376–4384 CrossRef CAS
.
- S.-H. Wang, F. Khurshid, P.-Z. Chen, Y.-R. Lai, C.-W. Cai, P.-W. Chung, M. Hayashi, R.-J. Jeng, S.-P. Rwei and L. Wang, Chem. Mater., 2022, 34, 4955–4963 CrossRef CAS
.
- S.-H. Wang, P.-Z. Chen, Y.-Y. Chen, F. Khurshid, C.-W. Cai, Y.-Y. Lai, P.-W. Chung, R.-J. Jeng, S.-P. Rwei and L. Wang, ACS Appl. Mater. Interfaces, 2022, 14, 43109–43115 CrossRef CAS PubMed
.
- F. Moruzzi, W. Zhang, B. Purushothaman, S. Gonzalez-Carrero, C. M. Aitchison, B. Willner, F. Ceugniet, Y. Lin, J. Kosco, H. Chen, J. Tian, M. Alsufyani, J. S. Gibson, E. Rattner, Y. Baghdadi, S. Eslava, M. Neophytou, J. R. Durrant, L. Steier and I. McCulloch, Nat. Commun., 2023, 14, 3443 CrossRef CAS
.
- L. Wang and W. Zhu, Adv. Sci., 2024, 11, 2307227 CrossRef CAS
.
- C. Han, S. Xiang, S. Jin, L.-W. Luo, C. Zhang, C. Yan and J.-X. Jiang, J. Mater. Chem. A, 2022, 10, 5255–5261 RSC
.
- K. Li, Y. Su, Z. Sun, Z. Li, Q. Sun, Y. Tian, B. Liu, G. Yue, J. Xue, Y. Tian and X. Xiong, Sep. Purif. Technol., 2025, 356, 129942 CrossRef CAS
.
- S. Fukuzumi, K. Ohkubo and T. Suenobu, Acc. Chem. Res., 2014, 47, 1455–1464 CrossRef CAS PubMed
.
- Q. Li, J. Li, W.-R. Wang, L.-N. Liu, Z.-W. Xu, G. Xie, J. Li, J. Yao and W.-S. Li, Chin. J. Chem., 2022, 40, 2457–2467 CrossRef CAS
.
- C. Yang, W. Huang, L. C. da Silva, K. A. I. Zhang and X. Wang, Chem.–Eur. J., 2018, 24, 17454–17458 CrossRef CAS PubMed
.
- B. Chen, F. Tian, M. Wang and C. Peng, Appl. Catal., A, 2022, 638, 118618 CrossRef CAS
.
- T. Wang, M. Li, Y. Chen, X. Che, F. Bi, Y. Yang, R. Yang and C. Li, ACS Catal., 2023, 13, 15439–15447 CrossRef CAS
.
- X. Zhao, B. Chen, W. Dong, S. Wang, Y. Xiang and H. Chen, J. Mater. Chem. A, 2021, 9, 10208–10216 RSC
.
- R. S. Sprick, B. Bonillo, R. Clowes, P. Guiglion, N. J. Brownbill, B. J. Slater, F. Blanc, M. A. Zwijnenburg, D. J. Adams and A. I. Cooper, Angew. Chem., Int. Ed., 2016, 55, 1792–1796 CrossRef CAS
.
- W.-C. Lin, J. Jayakumar, C.-L. Chang, L.-Y. Ting, T.-F. Huang, M. H. Elsayed, A. M. Elewa, Y.-T. Lin, J.-J. Liu, Y.-T. Tseng and H.-H. Chou, J. Mater. Chem. A, 2022, 10, 6641–6648 RSC
.
- W. Zhang, X. Luo, H. Tang, Z. Tang, F. Huang, Q. Wan, G. Yu and H. Yang, J. Mater. Chem. A, 2025, 13, 4746–4793 RSC
.
- Z.-A. Lan, W. Ren, X. Chen, Y. Zhang and X. Wang, Appl. Catal., B, 2019, 245, 596–603 CrossRef CAS
.
- Z. Fu, A. Vogel, M. A. Zwijnenburg, A. I. Cooper and R. S. Sprick, J. Mater. Chem. A, 2021, 9, 4291–4296 RSC
.
- C. Yang, S. Zhang and J. Hou, Aggregate, 2022, 3, e111 CrossRef CAS
.
- J. Wu, G. Li, J. Fang, X. Guo, L. Zhu, B. Guo, Y. Wang, G. Zhang, L. Arunagiri, F. Liu, H. Yan, M. Zhang and Y. Li, Nat. Commun., 2020, 11, 4612 CrossRef CAS PubMed
.
- C. M. Cardona, W. Li, A. E. Kaifer, D. Stockdale and G. C. Bazan, Adv. Mater., 2011, 23, 2367–2371 CrossRef CAS PubMed
.
- M. Sachs, H. Cha, J. Kosco, C. M. Aitchison, L. Francàs, S. Corby, C.-L. Chiang, A. A. Wilson, R. Godin, A. Fahey-Williams, A. I. Cooper, R. S. Sprick, I. McCulloch and J. R. Durrant, J. Am. Chem. Soc., 2021, 143, 524 CrossRef CAS PubMed
.
- J. Kosco and I. McCulloch, ACS Energy Lett., 2018, 3, 2846–2850 CrossRef CAS
.
- B. Cai, M. Axelsson, S. Zhan, M. V. Pavliuk, S. Wang, J. Li and H. Tian, Angew. Chem., Int. Ed., 2023, 62, e202312276 CrossRef CAS PubMed
.
- W. Wu, C. Feng, M. Chen, Q. Tan, Y. Deng, C. Zeng, L. Zhong and C. Dai, Green Chem., 2023, 25, 9335–9342 RSC
.
- C. Cheng, J. Yu, D. Xu, L. Wang, G. Liang, L. Zhang and M. Jaroniec, Nat. Commun., 2024, 15, 1313 CrossRef CAS PubMed
.
- C.-H. Chen, F. Brosa Planella, K. O'Regan, D. Gastol, W. D. Widanage and E. Kendrick, J. Electrochem. Soc., 2020, 167, 080534 CrossRef CAS
.
- X. Wang, Q. Xiang, B. Liu, L. Wang, T. Luo, D. Chen and G. Shen, Sci. Rep., 2013, 3, 2007 CrossRef PubMed
.
- S. Zhu, X. Qin, Q. Wang, T. Li, R. Tao, M. Gu and M. Shao, J. Mater. Chem. A, 2019, 7, 16954–16961 RSC
.
- S. A. J. Hillman, R. S. Sprick, D. Pearce, D. J. Woods, W.-Y. Sit, X. Shi, A. I. Cooper, J. R. Durrant and J. Nelson, J. Am. Chem. Soc., 2022, 144, 19382–19395 CrossRef CAS PubMed
.
Footnote |
| † Cai Cheng-Wei, Kuang-Hao Cheng and Palraj Ranganathan contributed equally to this paper. |
| This journal is © The Royal Society of Chemistry 2026 |