Orchestrating the d-band and p-band centroids with local Lewis acid sites accelerates O2 evolution performance in ultralow Ru-doped NiFe LDH†
Suvankar
Deka
ab,
Manju Kumari
Jaiswal
ab,
Tanmoy
Kalita
c,
Kangkon
Saikia
d,
Parasmani
Rajput
ef,
Som Datta
Kaushik
g,
Dhruba Jyoti
Kalita
 c and
Biswajit
Choudhury
c and
Biswajit
Choudhury
 *ab
*ab
aMaterials and Energy Laboratory, Physical Sciences Division, Institute of Advanced Study in Science and Technology (IASST), Paschim Boragaon, Vigyan Path, Assam 781035, India. E-mail: biswajitchoudhury@iasst.gov.in; biswa.tezu@gmail.com
bAcademy of Scientific and Innovative Research (AcSIR), Ghaziabad 201002, India
cDepartment of Chemistry, Gauhati University, Guwahati, Assam 781014, India
dZymolent Biosciences Pvt. Ltd., Mirza, Assam 781125, India
eBeamline Development and Application Section, Bhabha Atomic Research Centre, Trombay, Mumbai 400085, India
fHomi Bhabha National Institute, Anushakti Nagar, Mumbai 400 094, India
gUGC-DAE Consortium for Scientific Research Mumbai Centre, R5 Shed, BARC, Mumbai 400085, India
First published on 10th November 2025
Abstract
Accelerating the oxygen evolution reaction (OER) entails the dynamic stabilisation of its reaction intermediates to minimise energy barriers and enhance reaction kinetics. Nonetheless, optimisation of adsorption and desorption equilibria remains a formidable bottleneck. Our work highlights an ultralow Ru-doped NiFe LDH (0.43%, NRu3) framework coupled with tuned d-band and p-band centroid positions that deploys close to a quasi-industrial-scale current density of 746 mA cm−2 in 1 M KOH (25 °C). EXAFS reveals a perturbed Ru–O environment, suggesting the presence of Lewis acid sites. These sites act as electron acceptors and metastabilise Ru, facilitating a better charge redistribution in NRu3. DFT calculations assert that the site of Ru doping, along with oxygen vacancies, plays a pivotal role in charge redistribution and optimisation of the centroid positions. Notably, NRu3 manifested extraordinary OER performance under both alkaline (η = 173 mV and Tafel slope 33.4 mV dec−1) and simulated-seawater conditions (η = 196 mV and Tafel slope 51.2 mV dec−1). Moreover, NRu3 displays an outstanding AEMWE performance, delivering a 66.7 fold enhancement in activity-to-price efficiency over the standard RuO2 catalyst. Furthermore, a neural network (NN) driven multi-layer perceptron model was designed to predict the electrocatalytic activity across varying temperatures. Collectively, our work highlights the exceptional potential of NRu3 towards scalable and sustainable water-splitting technologies.
1. Introduction
“Heal the world, make it a better place for you and for me and the entire human race”, the song by celebrated musical figure Michael Jackson resonates as an epitome tagline for today's campaign towards a sustainable future. Growing environmental concerns and escalating energy crises accentuate the urgent need for developing renewable energy solutions. In this context, water splitting emerges as the most promising route for sustainable production of high-purity oxygen and hydrogen, without leaving any carbon footprint.1,2 Notwithstanding this, water electrolysis (WE) is acutely curtailed due to the inherent sluggish kinetics of the OER, thereby diminishing the overall water-splitting efficiency.3,4 Despite remarkable advances in non-precious metal catalysts, noble metal-based systems (IrO2, RuO2, and IrRuOx) continue to stand as distinguished benchmarks in the field of OER catalysis.5 Situated near the apex of ΔG*O − ΔG*OH, these materials display perfect equilibrium between oxophilicity and catalytic reactivity.6 Nonetheless, its commercial utilization is acutely disrupted because of its expensiveness and low availability.7 Therefore, developing electrocatalysts that prioritise cost-effectiveness and sustainability stands as a vital step toward scalable water-splitting technologies.Over the past few years, NiFe-based systems have garnered profound attention as OER electrocatalysts owing to their earth abundance, tuneable internal architecture, high surface area, and synergistic interaction between Ni and Fe atoms.8–10 Despite this, the OER performance is restricted by the limited number of active sites, poor conductivity and durability.11 Furthermore, the energy barriers associated with the adsorption/desorption of OER intermediates in NiFe LDH drastically impair its catalytic activity.12,13 To overcome this shortcoming, a myriad of strategies were employed. Among them, electronic modulation via heteroatom doping appears to be a rational route in optimising the intrinsic activity of electrocatalysts.14 Reports suggest that introducing an electron-deficient element into NiFe LDH can create auxiliary electron-accepting (e-A) sites and assist in better redistribution of π-symmetry electrons.11,14,15 In addition, such elements (V, Ce, and Ru) can effectively stabilise the Fe sites susceptible to dissolution under prolonged OER conditions, thereby sustaining the structural stability and catalytic activity of NiFe LDH.16,17 For instance, Chen et al. demonstrated that decorating Ru nanoclusters on defective NiFe LDH induces an upshift of the Ni 3d-band centre, which precisely fine-tunes the adsorption of oxygen intermediates, enhancing the OER performance.18 Furthermore, Wang and his coworkers described that Ag and Ru could synergistically enhance the reaction kinetics and corrosion resistance of NiFe LDH in seawater electrolysis.4 In light of this, Ru can be recognised as the most viable choice of dopant as (a) it is the least expensive among all the noble metals, (b) it exhibits high intrinsic catalytic activity and (c) it displays favorable adsorption energetics for oxygen-containing intermediate adsorption.18,19 Capitalising on this, tuning the d-band (εd) and p-band (εp) centroids can decisively optimise the adsorption–desorption energetics of oxygenated intermediates towards the Sabatier optimum enhancing the water oxidation process.20,21 Nonetheless, confusion surrounds! While some reports say that a downward shift of εd is advantageous, others argue that an upward shift seems more beneficial.12,22,23 Therefore, it is imperative to understand “how much shift of εd concerning Ef benefits the OER process?” In addition, limited efforts have been devoted to designing OER catalysts with an exceptionally minimal quantity of Ru. Lastly, only countable reports bestow mathematical insight into charge transportation pathways in NiFe LDH.
Guided by the aforementioned thoughts, we rationally design an OER catalyst by incorporating an ultralow concentration of Ru into NiFe LDH (NRu3). First-principles calculations assert that the doping concentration and site selectivity hold paramount importance in modulating the p-band and d-band centroid positions along with the M–O covalency. This is attributed to Lewis acid sites that act as electron-accepting (e-A) centres and aid in metastabilising Ru, which sets the stage for efficient redistribution of charges in the NiFe LDH lattice. Furthermore, computations based on the mathematical modelling of NRu3 established that Ru, in the presence of Lewis sites, assists in minimising the charge conduction pathway, thereby boosting the charge transport efficiency in NiFe LDH. Spectacularly, ultralow Ru-doped NiFe LDH exhibited an outstanding water oxidation performance under both alkaline and simulated seawater conditions. Especially in 1 M KOH, the catalyst entails only an overpotential of 289 mV to deploy a close to quasi-industrial-scale current density of 746 mA cm−2. Beyond this, the integration of NRu3‖Ni foam into an anion-exchange membrane water electrolyser (AEMWE) attains an exceptional activity-to-price ratio of 6.98 A$−1, approximately 66.7 fold higher than that of standard RuO2‖Ni foam. In a nutshell, our study establishes an unprecedented balance between cost-effectiveness and catalytic performance, showcasing the potential of ultralow Ru-doped NiFe LDH towards economically sustainable and scalable OER systems. Furthermore, the article offers an in-depth fundamental exposition of how precise tuning of d-band and p-band centroids can accelerate the OER dynamics in Ru-doped NiFe LDH systems.
2. Materials and methods
2.1. Chemicals
Nickel(II) nitrate hexahydrate (Ni(NO3)2·6H2O), iron(III) nitrate nonahydrate (Fe(NO3)2·9H2O), urea (NH2CONH2), ammonium fluoride (NH4F), potassium hydroxide (KOH), ruthenium(IV) oxide (RuO2) and ethanol were purchased from Merck. Nafion perfluorinated resin solution (5 wt%), ruthenium(III) chloride hydrate (RuCl3·xH2O) and platinum on carbon (10 wt% loading matrix activated carbon support) were purchased from Sigma-Aldrich. Nickel foam (1.6 mm thickness, purity > 99.99%) was purchased from THE ELECTRODE STORE. Deionised (DI) Milli-Q water (15 MΩ cm−1 at 27 °C) was used to prepare all the aqueous solutions. All the reagents were of analytical grade and used without any further purification.2.2. Synthesis of NiFe LDH and Ru-doped NiFe LDH
NiFe LDH (NF) and Ru-doped NiFe LDH were synthesised by employing a facile one-pot hydrothermal approach. To explore the influence of Ru doping, three distinct concentrations of RuCl3·xH2O – 27 µmol, 18 µmol, and 9 µmol were introduced into NiFe LDH. From here onwards, NiFeRu (27 µmol), NiFeRu (18 µmol) and NiFeRu (9 µmol) are labelled as NRu1, NRu2 and NRu3, respectively. Sections S1 and S2 incorporate a detailed description of the synthesis protocol and material characterisation.2.3. Neutron diffraction experiments and EXAFS measurements
Neutron diffraction studies were conducted to determine the phase composition and crystal structure of our prepared samples. The experiments were performed at the UGC-DAE Consortium for Scientific Research Mumbai Centre at the National Facility for Neutron Beam Research (NFNBR), Dhruva reactor, Mumbai (India). Section S3a outlines the experimental setup with information on data processing. Meanwhile, XANES and EXAFS measurements were deployed to extract information on the local atomic structure and bonding environment. A comprehensive overview of the experimental procedure and analysis steps is presented in Section S3b.2.4. Computational details
The Quantum ESPRESSO (QE) software package was utilised to conduct the theoretical calculations on NiFe LDH and Ru-doped NiFe LDH systems.24 An in-depth description is outlined in Section S4.2.5. Electrochemical measurements
The electrocatalytic oxygen evolution reaction (OER) was investigated in a Gamry EC workstation (Interface 1010E Potentiostat/Galvanostat/ZRA, 27102) employing a standard three-electrode configuration. Detailed information is provided in Section S5 in the SI. Furthermore, the overall water splitting was assessed in a membrane-electrode assembly (MEA) based anion exchange membrane water electrolyser (model: RSI-ELY-AEM-300, Serial No. 20250401, 2025, RESEARCH SUPPORTERS INDIA PVT. LTD.). Additionally, we have detailed the cost assessment of sample production, O2 production and faradaic efficiency, as well as AEMWE in Sections S6 to S8, respectively.2.6. Machine learning model construction
A Multi-Layer Perceptron (MLP) model, featuring a customised architecture and optimised hyperparameters, was meticulously designed to capture the intricate relationships inherent in the LSV data. The model aims to predict the LSV profiles across various temperatures relevant to our investigation. Section S9 in the SI provides an in-depth description.3. Results and discussion
3.1. Structural, morphological, and surface chemical composition features
Fig. 1a presents the X-ray diffraction (XRD) pattern of our synthesised samples, with its characteristic diffraction peaks revealing the formation of hydrotalcite-like LDH (PDF no. 01-082-8040).25,26 Based on the (003) reflection, the interlayer distance of NF was determined to be 7.59 Å and the lattice constant, ‘c’, to be 22.7 Å. As ‘c’ is precisely three times that of interlayer spacing, this suggests that the 3R stacking arrangement aligns with the structure of CO32− intercalated LDH.27,28 Stepping into the Ru-doped samples, a subtle shift in the (003) plane has been observed for NRu1 with interplanar spacing, ‘d003’ = 7.57 Å. Nevertheless, no observable shift in the peak positions was observed upon successive reduction of the Ru concentration in NiFe LDH (i.e. for NRu2 and NRu3 samples). This suggests that NRu2 and NRu3 experience negligible expansion or contraction, indicating no alteration in the crystal system, thereby preserving the LDH structure. Nonetheless, a distinct shift of the (003) plane to a lower 2θ value was observed in NiFe LDH with high Ru loading (NRu1). This outlines that Ru doping might have led to local distortion in the crystal lattice with alteration in periodicity and interplanar spacing. Furthermore, neutron diffraction (ND) experiments were performed to complement the information deduced from X-ray diffraction analysis, featuring additional clarity on the crystal structure and phase purity of our synthesized samples. Fig. 1b illustrates the ND pattern with well-defined peaks for NF, NRu1 and NRu3. The collected ND data were subjected to refinement by employing Le Bail refinement, which confirms that the samples crystallized into a hexagonal phase with the R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group. The obtained cell parameters exhibit a minimal change after introducing Ru into NiFe LDH (Table S1). Convincingly, ND findings align with the X-ray diffraction analysis, confirming the phase purity and structural integrity of the synthesized material. Meanwhile, Inductively Coupled Plasma Mass Spectrometry (ICP-MS) was performed for a detailed quantification of metallic components involving NF, NRu1, NRu2, and NRu3 (Table S2). Notably, NRu3 exhibits an ultralow concentration of Ru (0.43 wt%), remarkably lower than the reported values for Ru-loaded NiFe LDH. Subsequent to this, SEM images were captured to visualize the surface morphology of the prepared samples. An overall morphological perspective of NF is presented in Fig. S1a. The image features regular and uniform “origami marigold-like” morphology with an average diameter of 10.30 µm derived from the distribution plot in Fig. S1b. Similarly, the highest Ru-doped sample, NRu1, too exhibited “origami marigold-like” morphology akin to that of NF with a diameter of 12.86 µm (shown in Fig. 1c). FE-SEM elemental mapping manifests the coexistence of Ni, Fe, and Ru and their uniform distribution throughout the material, which is presented in Fig. S1c–e. The high-resolution TEM image (Fig. 1d) displays a smooth layered nanosheet morphology in NRu3, with an average lateral size of 176 nm. Furthermore, the AFM image of NRu3, as shown in Fig. 1e, confirms the formation of nanosheets. A few distinct flakes marked with ‘a’ and ‘b’ were selected to measure the thickness of the nanosheets. Their corresponding height profiles, as depicted in Fig. S1f, reveal thicknesses of 51 nm and 18 nm, respectively. Subsequent to this, Raman scattering was performed to investigate the vibrational properties and structural details, providing crucial insights into lattice dynamics and the bonding environment. Fig. 1f displays the Raman profile of the prepared samples within a range of 200 cm−1 to 1200 cm−1. For NF, the characteristic peaks observed at 456.6 cm−1 and 534.6 cm−1 can be ascribed to Ni–O and Fe–O vibrations. Typically, the vibrations are assigned to E2g and A1g phononic modes. A small peak evolving at 1053.1 cm−1 is observed, which can be attributed to the intercalation of CO32− anionic species within the layers. Strikingly for NRu1 (the sample with the highest Ru concentration), a significant redshift in both Ni–O (450.7 cm−1) and Fe–O (530.9 cm−1) peaks was observed compared to pristine NF. A similar scenario can be well realized with NRu2, where both the E2g and A1g phononic modes were found redshifted. Nevertheless, for NRu3, a minimal red shift has been observed. We infer that Ru doping induces a redistribution of electron density, weakening the bonding between the metal centres (Ni and Fe) and O within the lattice. Consequently, this weakening of the M–O bonds can be attributed to shifting the vibrational phononic modes to lower frequencies. X-ray photoelectron spectroscopy (XPS) was performed to decipher the chemical composition and valence states of the electrocatalyst. Tables S3 and S4 incorporate the fitted binding energy positions of Ni 2p, Fe 2p and O 2p and percentage compositions of chemical species in NF, NRu1, NRu2, and NRu3, respectively. Fig. S2a presents the deconvolution of the Ni 2p envelope for NF. The peaks at 855.16 eV and 872.80 eV convey the presence of Ni2+ states, whereas the peaks at 856.31 eV and 874.36 can be recognised as belonging Ni3+ valence states. Akin to NF, Ru-doped samples highlight similar information on the presence of valence species (Fig. S2b–d). Notably, NRu3 showcases ample Ni3+ states among all the samples. Reports convey that when the eg occupancy of the transition metal cation is close to 1, OER activity profoundly accelerates.29,30 As Ni3+ inherits a unique configuration of t62g and e1g, an appreciable oxidation performance can be anticipated for NRu3.30 Fig. S2e–h depicts the deconvolution of the core level Fe 2p spectra for NF, NRu1, NRu2 and NRu3. For NF, the peaks typically identified at 711.77 eV and 725.38 eV can be indexed to Fe 2p3/2 and Fe 2p1/2 of Fe3+. Fig. S2i–l presents the deconvoluted O 1s XPS core level spectra for all the samples, revealing three distinct components. The fitted peaks O(I) correspond to the M–O bond, whereas O(II) and O(III) can be ascribed to M–OH and surface-adsorbed water species, respectively.
m space group. The obtained cell parameters exhibit a minimal change after introducing Ru into NiFe LDH (Table S1). Convincingly, ND findings align with the X-ray diffraction analysis, confirming the phase purity and structural integrity of the synthesized material. Meanwhile, Inductively Coupled Plasma Mass Spectrometry (ICP-MS) was performed for a detailed quantification of metallic components involving NF, NRu1, NRu2, and NRu3 (Table S2). Notably, NRu3 exhibits an ultralow concentration of Ru (0.43 wt%), remarkably lower than the reported values for Ru-loaded NiFe LDH. Subsequent to this, SEM images were captured to visualize the surface morphology of the prepared samples. An overall morphological perspective of NF is presented in Fig. S1a. The image features regular and uniform “origami marigold-like” morphology with an average diameter of 10.30 µm derived from the distribution plot in Fig. S1b. Similarly, the highest Ru-doped sample, NRu1, too exhibited “origami marigold-like” morphology akin to that of NF with a diameter of 12.86 µm (shown in Fig. 1c). FE-SEM elemental mapping manifests the coexistence of Ni, Fe, and Ru and their uniform distribution throughout the material, which is presented in Fig. S1c–e. The high-resolution TEM image (Fig. 1d) displays a smooth layered nanosheet morphology in NRu3, with an average lateral size of 176 nm. Furthermore, the AFM image of NRu3, as shown in Fig. 1e, confirms the formation of nanosheets. A few distinct flakes marked with ‘a’ and ‘b’ were selected to measure the thickness of the nanosheets. Their corresponding height profiles, as depicted in Fig. S1f, reveal thicknesses of 51 nm and 18 nm, respectively. Subsequent to this, Raman scattering was performed to investigate the vibrational properties and structural details, providing crucial insights into lattice dynamics and the bonding environment. Fig. 1f displays the Raman profile of the prepared samples within a range of 200 cm−1 to 1200 cm−1. For NF, the characteristic peaks observed at 456.6 cm−1 and 534.6 cm−1 can be ascribed to Ni–O and Fe–O vibrations. Typically, the vibrations are assigned to E2g and A1g phononic modes. A small peak evolving at 1053.1 cm−1 is observed, which can be attributed to the intercalation of CO32− anionic species within the layers. Strikingly for NRu1 (the sample with the highest Ru concentration), a significant redshift in both Ni–O (450.7 cm−1) and Fe–O (530.9 cm−1) peaks was observed compared to pristine NF. A similar scenario can be well realized with NRu2, where both the E2g and A1g phononic modes were found redshifted. Nevertheless, for NRu3, a minimal red shift has been observed. We infer that Ru doping induces a redistribution of electron density, weakening the bonding between the metal centres (Ni and Fe) and O within the lattice. Consequently, this weakening of the M–O bonds can be attributed to shifting the vibrational phononic modes to lower frequencies. X-ray photoelectron spectroscopy (XPS) was performed to decipher the chemical composition and valence states of the electrocatalyst. Tables S3 and S4 incorporate the fitted binding energy positions of Ni 2p, Fe 2p and O 2p and percentage compositions of chemical species in NF, NRu1, NRu2, and NRu3, respectively. Fig. S2a presents the deconvolution of the Ni 2p envelope for NF. The peaks at 855.16 eV and 872.80 eV convey the presence of Ni2+ states, whereas the peaks at 856.31 eV and 874.36 can be recognised as belonging Ni3+ valence states. Akin to NF, Ru-doped samples highlight similar information on the presence of valence species (Fig. S2b–d). Notably, NRu3 showcases ample Ni3+ states among all the samples. Reports convey that when the eg occupancy of the transition metal cation is close to 1, OER activity profoundly accelerates.29,30 As Ni3+ inherits a unique configuration of t62g and e1g, an appreciable oxidation performance can be anticipated for NRu3.30 Fig. S2e–h depicts the deconvolution of the core level Fe 2p spectra for NF, NRu1, NRu2 and NRu3. For NF, the peaks typically identified at 711.77 eV and 725.38 eV can be indexed to Fe 2p3/2 and Fe 2p1/2 of Fe3+. Fig. S2i–l presents the deconvoluted O 1s XPS core level spectra for all the samples, revealing three distinct components. The fitted peaks O(I) correspond to the M–O bond, whereas O(II) and O(III) can be ascribed to M–OH and surface-adsorbed water species, respectively.
3.2. Local coordination dynamics
X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) spectra were utilised to ascertain the electronic interactions and atomic arrangement of the synthesised samples. Typically, elements in higher oxidation states display an elevated absorption threshold position, signifying a direct correlation between the oxidation state and edge energy. In XANES spectra, the K-edge features of Ni, Fe and Ru can be attributed to electron transitions from 1s to 4p orbitals.11Fig. 2a shows that the absorption threshold position of NF closely aligns with that of NiO, evincing Ni to be in the 2+ oxidation state. NRu1, NRu2 and NRu3 also appreciably overlap with NF, referring to an oxidation state similar to that of NF. The Fe K-edges of all the samples with the reference compounds Fe foil and Fe2O3 are presented in Fig. 2b. The edge positions of all samples asymptotically align with that of Fe2O3, conveying the valence of Fe to be Fe(III). Notably, XANES spectra for the Ru K-edge (as shown in Fig. 2c) depict that the absorption threshold for NRu3 is close to that of RuCl2, indicating the valence of Ru in the +2 state. For NRu1, the absorption threshold exhibits a minimal shift towards higher energy. Apparently, the white line intensity in NRu3 is more pronounced than that of NRu1, which may describe an increased density of unoccupied states.
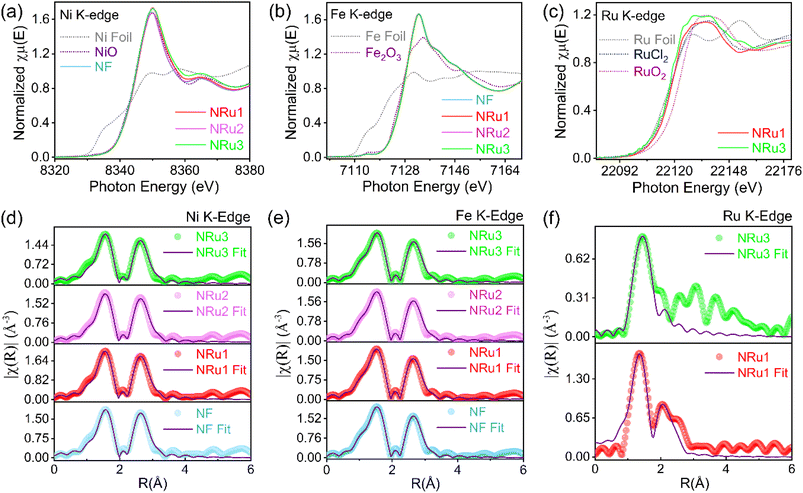 | ||
| Fig. 2 XANES and Fourier transform (FT)-EXAFS spectra of (a and d) Ni K-edge, (b and e) Fe K-edge and (c and f) Ru K-edge. | ||
Fig. 2d–f displays the Fourier transform (FT) EXAFS oscillations of Ni, Fe and Ru K-edges for NF, NRu1, NRu2 and NRu3 samples. In the case of Ni and Fe K-edges, the k-range spans from 2.5–10.0 Å−1 whereas for the Ru K-edge, it is in the range of 2.5–9.0 Å−1. The fitting of the EXAFS data was meticulously accomplished to derive the coordination number (CN), interatomic distance and Debye–Waller (DW) factor corresponding to M–O and M–M neighbours from Ni, Fe and Ru sites (Tables S5–S7). Two distinct characteristic peaks were observed for both the Ni/Fe edges for all the samples. The first peak primarily corresponds to the nearest-neighbour interaction between metal and oxygen (M–O), while the second peak arises from the nearest-neighbour correlation between metal atoms (M–M). EXAFS analysis of the Ni K-edge and Fe K-edge reveals almost identical CN values for Ni(Fe)–O and Ni(Fe)–Ni(Fe) bonds corresponding to the Ni(Fe) site. The value of CN hovers around 5.5 to 5.8, suggesting a slight deviation from the ideal sixfold coordination. Looking towards the bond length, it is evident that the Ni–O bond contracts to 2.038 Å (NRu1) from 2.169 Å, in contrast to the pristine system. Nonetheless, Ni–O relaxes back to 2.049 Å as Ru content decreases. Fascinatingly, the Debye–Waller factor for Ni–O coordination decreases with the increasing Ru concentration, whereas for the Fe–O environment it remains almost identical (Table S5). This conveys that Ru promotes a structural stabilizing effect on Ni–O coordination. Nonetheless, NRu3 (0.0043 Å2) displays a marginal increase in static/dynamic disorder in the Ni–O shell compared to NRu1 and NRu2. In a similar fashion, the Fe K-edge describes that the CN for Fe–O and Fe–Fe remains essentially the same for all samples. This suggests that each Fe centre maintains a consistent average number of next-nearest-neighbour Fe atoms thereby preserving the overall layered structure. Nevertheless, shorter Fe–O bonds and slightly higher DW factors were perceptible in NRu1 and NRu2 samples, implying perturbation in the immediate Fe–O environment. Fig. 2f illustrates the EXAFS oscillations for the Ru K-edge for NRu1 (highest concentration of Ru) and NRu3 (lowest concentration of Ru) samples. Strikingly, NRu1 depicts an elevated CN of Ru–O (3.8) compared to NRu3. A closer inspection of the EXAFS fitting (Fig. 2f) manifests a clear second shell of the Ru–Ru path for NRu1. The findings illustrate that Ru atoms may tend to form domains or clusters in the NiFe LDH matrix, with relatively well-defined Ru–Ru neighbours of ∼2.621 Å. Subsequently, in the context of NRu3, the absence of a Ru–Ru shell signifies an exceptionally low Ru concentration to yield a distinct Ru–Ru EXAFS signal with the NiFe LDH lattice. The CN of Ru–O in NRu3 drastically shrinks from 3.8 (NRu1) to 2.2, accompanied by elongation in the Ru–O bond length (1.887 Å), as the Ru concentration decreases. The DW factor also tends to increase in NRu3 compared to NRu1. Ergo, the findings reflect a more perturbed local environment around Ru sites. Eventually, we can say that as we gradually decrease the Ru concentration from high to low, it changes the Ru's local structure from clustering (Ru–Ru) to more isolated Ru sites (due to the absence of the Ru–Ru shell) with an increase in disorder around Ru at low concentrations with lower atomic coordination.
3.3. Computational study
DFT calculations were carried out to explore the electronic structure of pristine NiFe LDH and Ru-doped NiFe LDH, emphasizing how Ru doping influences the density of states (DOS), charge distribution, and Bader charges. Essentially, we have considered five different models for the theoretical calculations. Fig. 3a–f illustrate the models and their associated DOS plot for pristine NiFe LDH (model I), Ru doped at the Ni site (model II), and Ru doped at the Fe site (model III), respectively. Parenthetically, the models for Ru-doped at Ni and Fe sites incorporating oxygen vacancies (Ov) are presented in Fig. S3a and b, respectively. Fig. S3c and d present the corresponding DOS plot of the theoretical models. As anticipated, doping of Ru into NiFe LDH (irrespective of the Ni/Fe site) leads to the creation of additional states near the Fermi level (Fig. 3e and f). Nevertheless, a “dip or valley” seems to develop between the newly created and pre-existing states, indicating an alteration in the local electronic environment. Fascinatingly, the moment Ov were introduced (models IV and V), the “dip” like feature disappeared and a continuous DOS plot was obtained. | ||
| Fig. 3 First-principles study. (a–f) Computational models with their respective density of states (DOS) plot. Assignments: NiFe LDH (model I), Ru at the Ni site (model II), and Ru at the Fe site (model III). Visualization through VESTA.31. (g and h) d-Band centre of Ni 3d and Fe 3d and (i) p-band centre of O 2p for all the computational models. | ||
Fig. 3g–i showcases the positions of the d-band centre (εd) and p-band centre (εp) for Ni 3d, Fe 3d and O 2p orbitals, respectively. Observations reveal that for both the scenarios—Ru doped at the Ni site and Ru doped at the Fe site, the εd for Ni and Fe shifts away from the Fermi level. Nevertheless, a closer inspection of the models featuring oxygen vacancies (i.e. for models IV and V) reveals that εd of Ni and Fe move closer towards the Fermi level. Similarly, replacing a Ni/Fe site (models II and III) with Ru displays a minimal downward shift of the O p-band centre. When Ru is doped at the Ni/Fe site and oxygen vacancies are introduced, the εp even shifts more towards higher energies (−4.771 eV for model IV and −4.434 eV for model V). This conveys that systems IV and V experience a rearrangement and redistribution of electron density. As a consequence of the charge compensation mechanism, the neighbouring atoms may form strong bonds with Ru, thereby pushing the O 2p band centre to higher energy.
Table S8 showcases the band-centre difference (Δε) for Ni–O and Fe–O in the pristine NiFe LDH system along with other Ru-doped models, showcasing how strongly Ni and Fe bind in the lattice. Considering model I as the reference, an increase in Fe–O covalency was observed upon Ru doping in both the models – model II (ΔεFe,O = 0.126 eV) and model III (ΔεFe,O = 0.106 eV). Nevertheless, regardless of the doping site, the ΔεNi,O and ΔεRu,O acutely increase, suggesting weaker Ni–O and Ru–O orbital mixing. This implies that, although Ru may redistribute the charge in the NiFe LDH system, it cannot bring forth an optimum condition for balancing the adsorption/desorption of OER intermediates. Nonetheless, a quick glance at model IV reveals that Ru doping with oxygen vacancies can be a great boon. Concerning model V, although Ov aids in increasing the Ru–O covalency to a greater extent among all the models, it does so at the expense of Ni–O and Fe–O hybridisation. Evidently, model IV may be reckoned as a paragon that can deliver the most enhanced OER performance among all the listed models. But what prompted us to think so? Let us understand. Substitution of Ru at the Ni site with Ov reshapes the electronic structure of NiFe LDH into a more catalytically and energetically favourable environment. If you closely observe the d-band and p-band centroids, it's quite clear that the centroids edge closer towards the EF. This upward shift of εd relative to EF will lead to the elevation of the antibonding orbitals. Ergo, only a few electrons can occupy these antibonding states, which ultimately strengthens the metal–adsorbate interaction. Furthermore, the presence of Ov brings the system to a balanced Ni–O, Fe–O, and Ru–O covalency, which can foster a better delocalisation of electrons within the NiFe LDH system. Unequivocally, this outlines that model IV can fine-tune the adsorption strength of OER intermediates, which is vital in facilitating an optimised water oxidation process.32
Bader charge analysis of the computationally designed models is presented in Table S9. For simplicity, each Ni and Fe atom in the pristine NiFe LDH system is assigned a specific number (as shown in Fig. S4), facilitating easy identification of charge variations at Ni and Fe sites across different models. Considering model I as the reference, we find that substituting a Ni atom with a Ru atom at the Ni4 site (model II) reduces the charge on hexacoordinated Ni atoms (Ni8, Ni10, Ni16, Ni20, Ni23, and Ni25) surrounding the Ru site. Parenthetically, the charge on Fe atoms at positions Fe1, Fe4, Fe5, and Fe8 exhibits a marginal increment, while the Fe atoms (Fe2 and Fe7), located diagonally to the Ru atom, show a minimal decrease in charge. Consequently, this suggests that Ru steers redistribution of electron density in NiFe LDH. Moving to model III, where Ru replaces Fe at the Fe7 site, the charge on Fe1, Fe3, and Fe4 is enhanced, whereas the charge trends in Ni atoms follow a similar trend to those in model II. Intriguingly, once oxygen vacancies were introduced near the Ru site (i.e. for models IV and V), a notable depletion in Ni charge was witnessed accompanied by an increase in Fe charge. Convincingly, the metrics of the Bader charge for model IV spotlight an “elevated and better” charge distribution in the Ni–O–Ru–O–Fe motif among all the models. Unequivocally, we can conclude that Ru effectively facilitates tuning the electronic structure of NiFe LDH. In particular, the specific site of doping – whether at the Ni site or Fe site in the presence of oxygen vacancies plays a pivotal role in guiding the extent of charge distribution in the –M–O–M– network.
3.4. Electrocatalytic performance
The electrocatalytic OER performance of the prepared samples was evaluated by conducting linear sweep voltammetry (LSV) in N2 saturated 1 M KOH electrolyte. Fig. 4a displays the iR-drop corrected polarisation curves of NF and Ru-doped NF samples. Fascinatingly, we observed that lowering the concentration of Ru in NiFe LDH enhanced its water oxidation activity. Among the synthesised catalysts, the lowest Ru-loaded sample, NRu3, exhibited an outstanding current density, jgeo, of 746 mA cm−2 (close to quasi-industrial-scale current density) at an overpotential of merely 289 mV. In contrast, NRu2 and NRu3 deliver comparatively lower current densities of 508 mA cm−2 and 249 mA cm−2, respectively. A closer evaluation reveals that NRu3 entails an overpotential of 173 mV to deliver a benchmark current density of 10 mA cm−2 followed by NRu2 (178 mV) and NRu1 (192 mV), each surpassing pristine NF which requires an overpotential of 202 mV. Table 1 summarises the extracted values of kinetic descriptors (Tafel slope and charge transfer coefficient, α) and activity descriptor (Tafel constant) from the LSV profile. Fig. 4b presents the Tafel slope of our synthesised samples. For pristine NF, the Tafel slope was found to be 69.0 mV dec−1; however, upon Ru doping, the Tafel slope decreased following the trend NRu1 > NRu2 > NRu3.| Sample | Overpotential, ηiR (mV) (at various current densities) | Tafel constant (mV) | Tafel slope at 10 mA cm−2 (mV dec−1) | Charge transfer coefficient, αa | ||
|---|---|---|---|---|---|---|
| η 10 | η 100 | η 500 | ||||
| NF | 202 | 290 | 393 | 162 | 69.0 | 0.21 |
| NRu1 | 192 | 248 | 335 | 163 | 47.6 | 0.31 |
| NRu2 | 178 | 221 | 290 | 153 | 40.6 | 0.36 |
| NRu3 | 173 | 213 | 266 | 148 | 37.9 | 0.39 |
Essentially for a broader performance benchmark, the OER activity of NRu3 was even compared with that of commercially available standards RuO2 and Pt/C (as shown in Fig. S5). Remarkably, NRu3 outperforms both the catalysts with considerably low overpotential and faster reaction kinetics, highlighting the critical role of Ru content in accelerating the OER dynamics.
Essentially, voltammetric techniques include contributions from interfacial capacitance hindering the accurate assessment of catalytic activity. This is because the acquired current and overpotential from voltammograms were not measured under steady conditions, thereby exhibiting variations with time. That being so, we utilized chronoamperometry (CA) as a steady-state polarization technique to provide sufficient time for the catalytic interface to reach equilibrium, facilitating a precise evaluation of the catalyst's intrinsic activity. Table S10 displays the steady-state current obtained for NF, NRu1, NRu2 and NRu3 at various overpotentials. By employing these obtained values, the logarithmic current densities were plotted against iR corrected overpotential to obtain the Tafel slope. Fig. 4c shows the corresponding Tafel plot for NF, NRu1, NRu2 and NRu3. Compared to the Tafel slopes extracted from LSV, CA exhibits a lower Tafel slope for NF (62.0 mV dec−1) and NRu3 (33.4 mV dec−1).
We further evaluated the (i) mass activity and (ii) TOFs (turnover frequencies) of our catalysts at different overpotentials ranging from 100 mV to 250 mV. Fig. 4d displays the mass activity of the prepared samples. At an overpotential of 250 mV, pristine NF showcases a mass activity of 5.0 A g−1. In contrast, a 2.4-fold, 5.2-fold, and 7.8-fold increase has been observed for NRu1, NRu2, and NRu3 samples, respectively. Notably, NRu3 surpasses the other samples with an impressive mass activity of 39.3 A g−1. Furthermore, to unveil turnover frequency (TOF) values, the surface concentration of active sites related to the redox species must be calculated. Therefore, the corresponding reduction peaks are illustrated in Fig. S6. A detailed procedure for the TOF calculation can be found in Section S5. Fig. 4e depicts the TOF merits for pristine NF, NRu1, NRu2 and NRu3. At an overpotential of 250 mV, NRu3 exhibited a TOF value of 0.315 s−1, approximately 2.26-fold greater than that of pristine NF which stands at a value of 0.139 s−1. Convincingly, the results indicate that NRu3 has a higher atom utilization efficiency and faster kinetics per active site.33
Furthermore, we delved into electrochemical impedance spectroscopy (EIS) to unveil insights into the kinetics of the OER. Essentially, the idea is to comprehensively understand the electron transfer process and its associated conductivity. For this purpose, EIS measurements were carried out at various potentials starting from 1.404 V to 1.484 V vs. RHE as displayed in Fig. S7. Subsequently, Fig. 4f presents a comparison of the Nyquist plots for NF, NRu1, NRu2, and NRu3, recorded at a potential of 1.464 V vs. RHE. NRu3 displays the most diminutive semicircle, indicating minimal resistance experienced throughout the OER process. An equivalent circuit model, namely the 2R-CPE model (Fig. S8), has been used to fit the plots. The charge transfer resistance (Rct) was found to be in the order of NF > NRu1 > NRu2 > NRu3. The impressive charge transfer and low resistance of NRu3 align closely with the results obtained from the Tafel plot. Though equivalent circuit models (ECMs) provide a simplified representation of the impedance data, yet they depend on a priori assumptions which restrict the analysis to a specific set of polarisation processes. Consequently, the models might not accurately represent and interpret the system under study. In this context, the distribution of relaxation times (DRT) was employed to unveil a more precise picture of the different polarisation processes by transforming the impedance data into a time-based distribution without predefining assumptions. Fig. 4g shows the DRT plot corresponding to the impedance curves recorded at a potential of 1.464 V vs. RHE. Primarily, the DRT plot features two time constants for all samples. Here, τ1 refers to the charge transfer (CT) kinetics associated with the OER process, whereas τ2 reflects fast interfacial relaxation processes. As the relaxation intensity associated with τ1 is higher than that of τ2, it suggests that CT is the rate-determining step that dominates the overall impedance process. It is evident from Fig. 4g that, for NRu2 and NRu3, the spectral impedance δ, associated with τ1, demonstrates an accelerated declination from 0.45 ohm s−1 to 0.20 ohm s−1 compared to NF and NRu. A similar declination trend in the δ value (NRu1 > NRu2 > NRu3) for the τ2 component was observed in Ru-doped samples compared to that of pristine NF. Convincingly, the findings suggest that lowering the Ru concentration in NiFe LDH aided in lowering the polarising resistance, facilitating facile charge transfer and easier adsorption and desorption of oxygenated species.
Another key parameter in evaluating an electrocatalyst's performance is the availability of electrochemically active surface areas (ECSAs). Therefore, ECSAs of the prepared catalyst were estimated through the double-layer capacitance (Cdl) measured from EIS (details provided in Section S5). EIS was employed to serve the purpose because it stands unaffected by the potential drop caused by series resistance, a limitation often observed in DC techniques such as cyclic voltammetry. Let's find out what our Cdl value showcases. Calculation unveils that it follows the trend – NRu3 (9.10 mF) > NRu2 (8.21 mF) > NRu1 (6.20 mF) > NF (3.96 mF). Now advancing towards ECSA, Fig. 4h displays the ECSAs obtained for the prepared catalysts. The low percentage loading of Ru into NiFe LDH, i.e. NRu3, was found to show the highest ECSA of 227.54 cmECSA−2 followed by NRu2 (205.2 cmECSA−2) and NRu1 (154.9 cmECSA−2).
Afterwards, we tried to obtain the electrochemical density of states (eDOS) at potentials of 1.424 V and 1.464 V vs. RHE. Section S5 provides the relevant details for eDOS calculation. Strikingly, it was found that eDOS for NRu3 soars to a value of 6.10 × 1029 eV m−3, approximately 5.8 fold higher than that of pristine NF (1.05 × 1029 eV m−3). Such augmentation in eDOS indicates higher availability of electronic states near the Fermi level, accelerating the oxidation kinetics. Our results are further supported by the DFT calculations, which reveal that Ru doping in NiFe LDH introduces additional electronic states near the Fermi level. This modification enhances the electronic structure, potentially improving the catalytic dynamics. Following this, we step towards another indispensable attribute of an efficient catalyst – its long-term stability (LTS). In fact, it stands out as one of the defining attributes in evaluating its real-world applicability. That being so, we embarked on an ambitious LTS assessment spanning over 144.8 h. For this purpose, chronoamperometric measurements were performed at an overpotential of 284 mV (1.514 V vs. RHE) for three consecutive cycles of 50 h each. Spectacularly, NRu3 demonstrated excellent stability across all cycles, as depicted in Fig. 4i. In the initial cycle (0–50 h), the catalyst preserved 88.3% of its activity, establishing a stable baseline performance. In the subsequent cycle (2nd cycle, 50–94.8 h), the activity remained essentially unchanged at 88.8% for 44.8 h. Intriguingly, in the 3rd cycle (94.8–144.8 h), NRu3 featured only a marginal 2.6% decrease in current density, showcasing an exceptional stability of 97.4%. Nevertheless, considering the complete time frame i.e. from 0–144.8 h, the catalyst retained 83.5% of its initial activity. Convincingly, the outcomes highlight the robustness and resilience of our catalyst under prolonged operation of 144.8 h, thereby reinforcing its prospects in practical deployment.
In succession, with an outlook to understand the interfacial characteristics, the EIS was recorded at three different overpotentials, as shown in Fig. 5d. “Nevertheless, we land in some serious twist”. The modified Randel's circuit legitimately fits all the EIS curves, albeit meticulous examination reveals a sudden shift in the rate-limiting step of charge transfer kinetics, transitioning from the low-frequency R3-CPE2 loop to the high-frequency R2-CPE1 loop (Table S11). That being so, we immediately pulled the notion of DRT into this problem. Indeed, our conjecture appeared to be correct. DRT indicates that “the rate-limiting step has changed”. When we leap at 1.464 V (the catalytically active region), we notice a fronting asymmetry and peak broadening in the characteristic peak (Fig. S9). Apparently, this conveys overlapping contributions from charge transfer and interfacial relaxation processes. This suggests an alteration in the reaction pathways induced by some additional secondary processes. Hold on! While the “twist” has been recognised, we are still left with an incomplete answer to a pair of questions – (a) which factors contributed to the rate-limiting step and overall impedance of the system? and (b) What made the LSV curves shift towards higher potentials under SSW conditions? Of course, we came up with a convincing and compelling answer. The crux lies in – (a) competitive adsorption between Cl− and OH− ions and (b) surface reconstruction with active site poisoning.34,35 Cl− ions disrupt the surface coverage of the OER intermediates by binding to the active sites. This initiates alteration in reaction pathways (parasitic reactions) and acutely shifts and slows down the rate-determining steps which is evident and well supported from our DRT plot (Fig. S9). Furthermore, the chlorine evolution reaction (CER) overlaps with the OER at higher potentials. This may facilitate the formation of Ni/Fe oxychloride or other chlorinated phases, triggering changes in local pH, surface deactivation and shifting of Ni/Fe redox transitions. This eventually shifts the polarisation curves to higher overpotential (as perceptible from Fig. 5a), exacerbating the OER performance.
Encompassing all, our findings legitimately highlight that optimising the Ru concentration into NiFe LDH profoundly enhances water oxidation activity, demonstrating excellent catalytic performance in both alkaline and simulated seawater environments. Convincingly, compared to various Ru-incorporated NiFe LDH studies (Table S12), our article showcases unparalleled OER performance with an exceptionally low concentration of Ru.
3.5. Deciphering the OER mechanism
Unequivocally, our experimental and theoretical findings unveil the indispensable role of Ru in effectively elevating the water oxidation performance in NiFe LDH. Nonetheless, projecting a comprehensive understanding of the mechanistic insights is profoundly essential. That being so, permit us to navigate along two exclusive “cornerstones” to decipher the OER mechanism.| G = (V, E, φ); |
| E = {(vi, vj)|vi, vj ∈ V and vi ≠ vj} |
is a function that assigns weight wi to each edge eij.
This graph theory system is conceptualised in such a way that all nodes and edges mimic the atoms and their associated bonds of NF and NRu3, respectively. The weight (wi) of all edges is defined from the interatomic distance (R, Å) characterized by EXAFS. The key notion relies on the quest for recognizing the least resistive charge conduction pathway in the weighted graph. That being so, we employed Dijkstra's algorithm to (a) map the shortest pathway and (b) map the length of the shortest charge conduction pathway. Section S10 incorporates the Python code for the corresponding computation. Furthermore, an infographic on the node positions of various atoms for NF and NRu3 is presented in Fig. S12. The shortest pathway has been mapped for four specific positions (1 → 7), (1 → 13), (1 → 19) and (1 → 22) for both the systems NF and NRu3. Among them, the shortest pathway mapped from (1 → 22) in both the systems is illustrated in Fig. 6b. In this context, we have a key statement to emphasize. Our algorithm identifies two equally probable paths of identical length. Nonetheless, it chooses and showcases only one of the possible paths because the algorithm operates solely on path cost minimization, without an inherent tie-breaking mechanism. Ergo, within the domain of our study, this equivalency is not a limitation but a rational outcome coherent with the fundamental principles of network theory. Meanwhile, the shortest path length for NF and NRu3 has been computed to be 29.211 Å and 27.723 Å, respectively. Decisively, the data interpret the essence of Ru in optimising the transportation pathway in NRu3. Parenthetically, permit us to describe what happens in the presence of the Lewis sites. As these sites can facilitate the adsorption of OER intermediates, we impartially assign the 8th node as the source node (in NRu3). The shortest path length from (8 → 19) and (8 → 22) was calculated to be 14.001 Å and 17.892 Å, respectively. As the 8th node is not a catalytically active centre in NF, therefore, considering the 7th node as the source node, the path length from (7 → 19) and (7 → 22) increases to a value of 16.692 Å and 20.865 Å. Unequivocally, Ru in tandem with Lewis sites can act as a cornerstone, carving an “optimised path” to augment the charge transport efficiency in NiFe LDH.
4. Conclusion
Origami marigold-like ultralow Ru-doped NiFe LDH unveils extraordinary water oxidation activity under both alkaline and simulated seawater conditions. DFT findings convey that the site of Ru doping is vital in modulating the M–O covalency and d–p centroid positions. An optimised electronic environment was achieved when the Ru atom was doped at a Ni site, accompanied by Ov. Furthermore, EXAFS, in concert with first-principles calculations, suggests that the Lewis acid sites in the local environment of Ru metastabilise Ru and aid in better redistribution of charge in the NiFe LDH lattice. Our argument on “better redistribution of charge” is further validated via “graph theory”. Calculation reveals that Ru in conjunction with Lewis acid sites aids in elevating the charge transport efficiency in NiFe LDH. Remarkably, in an alkaline environment, NRu3 exhibited an incredibly low overpotential of 173 mV, featuring a Tafel slope of 37.9 mV dec−1. Nevertheless, under seawater conditions, the overpotential and Tafel slopes increase to 196 mV and 51.2 mV dec−1, respectively. Following this, we also designed a multi-layer perceptron architecture to predict the electrochemical behaviour of Ru-doped NiFe LDH across different temperatures. The predicted results were in close agreement with the experimental results, conveying its efficacy and reliability. Furthermore, within the AEMWE configuration, NRu3‖Ni foam delivers unprecedented activity-to-price performance, outperforming the benchmark RuO2‖Ni foam. To summarise, our research highlights the exceptional catalytic efficacy, economic promise, and scalable aspect of ultralow Ru-doped NiFe LDH, paving the way for its integration into next-generation water splitting technologies. In addition, the investigation provides a mechanistic framework elucidating how the interplay of centroids, eg occupancy, and Lewis acid sites serves as a yardstick in shaping the OER dynamics.Author contributions
Suvankar Deka: conceptualization, methodology, software, validation, formal analysis, investigation, data curation, writing original draft, writing – review and editing, and visualization; Manju Kumari Jaiswal: methodology, software, validation, formal analysis, writing – review and editing; Tanmoy Kalita and Dhruba Jyoti Kalita led the investigation and analysis associated with the DFT calculations. Parasmani Rajput and Som Datta Kaushik contributed to the investigation and analysis of EXAFS and neutron scattering experiments, respectively. Kangkon Saikia contributed to the design and implementation of the machine learning models and the computational tasks associated with the mathematical modelling. Biswajit Choudhury: supervision, validation, resources, writing – review and editing, project administration, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data that supports the findings of this study are available from the corresponding author upon reasonable request.Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta05192d.
Acknowledgements
Suvankar Deka and Biswajit Choudhury would like to thank IASST for the in-house project funding (2023–2025) and SAIC, IASST, for the instrumentation facility. The authors would like to acknowledge the Raja Ramanna Centre for Advanced Technology (RRCAT), a unit under the Department of Atomic Energy, Government of India, for supporting EXAFS characterisation. We also appreciate the Central Research Facility, IIT Delhi, for equipping us with the XPS facility. Tanmoy Kalita and Dhruba Jyoti Kalita are indebted to the Department of Science and Technology (SB/FT/CS-077/2013, CRG/2022/001313), India, for the financial support. They would also like to thank the University Grants Commission for the UGC-BSR Research start-up grant (No. F.30.-122/2015(BSR)) and Gauhati University for providing essential research facilities and supplementary funding.References
- J. Song, C. Wei, Z. F. Huang, C. Liu, L. Zeng, X. Wang and Z. J. Xu, Chem. Soc. Rev., 2020, 49, 2196–2214 RSC.
- Y. Yang, H. Gao, C. Zhu, W. Sun, J. Zeng, X. Wang and X. Tian, Adv. Funct. Mater., 2025, e14599 CrossRef.
- C. Feng, M. B. Faheem, J. Fu, Y. Xiao, C. Li and Y. Li, ACS Catal., 2020, 10, 4019–4047 CrossRef CAS.
- H. Chen, R. T. Gao, H. Chen, Y. Yang, L. Wu and L. Wang, Adv. Funct. Mater., 2024, 34, 2315674 CrossRef CAS.
- J. Rossmeisl, Z. W. Qu, H. Zhu, G. J. Kroes and J. K. Nørskov, J. Electroanal. Chem., 2007, 607, 83–89 CrossRef CAS.
- I. C. Man, H. Y. Su, F. Calle-Vallejo, H. A. Hansen, J. I. Martínez, N. G. Inoglu, J. Kitchin, T. F. Jaramillo, J. K. Nørskov and J. Rossmeisl, ChemCatChem, 2011, 3, 1159–1165 CrossRef CAS.
- D. Zhao, R. Zhang, M. Dai, H. Liu, W. Jian, F. Q. Bai and X. Wu, Small, 2022, 18, 2107268 CrossRef CAS.
- L. Yu, J. F. Yang, B. Y. Guan, Y. Lu and X. W. D. Lou, Angew. Chem., Int. Ed., 2018, 57, 172–176 CrossRef CAS PubMed.
- J. Zhang, L. Yu, Y. Chen, F. Lu, S. Gao, X. Wen, D. Lou, J. T. Zhang, L. Yu, X. F. Lu, X. W. Lou, Y. Chen and S. Y. Gao, Adv. Mater., 2020, 32, 1906432 CrossRef CAS PubMed.
- P. Li, M. Wang, X. Duan, L. Zheng, X. Cheng, Y. Zhang, Y. Kuang, Y. Li, Q. Ma, Z. Feng, W. Liu and X. Sun, Nat. Commun., 2019, 10(1), 1–11 CrossRef.
- Y. Lin, H. Wang, C.-K. Peng, L. Bu, C.-L. Chiang, K. Tian, Y. Zhao, J. Zhao, Y.-G. Lin, J.-M. Lee, L. Gao, Y. Lin, L. Bu, K. Tian, Y. Zhao, J. Zhao, L. Gao, H. Wang, J. Lee, C. Peng and C. Chiang, Small, 2020, 16, 2002426 CrossRef CAS.
- X. Wang, W. Pi, S. Hu, H. Bao, N. Yao and W. Luo, Nano-Micro Lett., 2025, 17, 1–12 CrossRef CAS PubMed.
- Y. Yang, Q. N. Yang, Y. B. Yang, P. F. Guo, W. X. Feng, Y. Jia, K. Wang, W. T. Wang, Z. H. He and Z. T. Liu, ACS Catal., 2023, 13, 2771–2779 CrossRef CAS.
- M. Liu, K. A. Min, B. Han and L. Y. S. Lee, Adv. Energy Mater., 2021, 11, 2101281 CrossRef CAS.
- J. Jiang, F. Sun, S. Zhou, W. Hu, H. Zhang, J. Dong, Z. Jiang, J. Zhao, J. Li, W. Yan and M. Wang, Nat. Commun., 2018, 9(1), 1–12 CrossRef PubMed.
- D. Y. Chung, P. P. Lopes, P. F. B. D. Martins, H. He, T. Kawaguchi, P. Zapol, H. You, D. Tripkovic, D. Strmcnik, Y. Zhu, S. Seifert, S. Lee, V. R. Stamenkovic and N. M. Markovic, Nat. Energy, 2020, 5, 222–230 CrossRef.
- Y. Yang, W.-J. Wang, Y.-B. Yang, P.-F. Guo, B. Zhu, K. Wang, W.-T. Wang, Z.-H. He and Z.-T. Liu, J. Electrochem. Soc., 2022, 169, 024503 CrossRef CAS.
- Y. Chen, Y. Liu, W. Zhai, H. Liu, T. Sakthivel, S. Guo and Z. Dai, Adv. Energy Mater., 2024, 14, 2400059 CrossRef CAS.
- Z. W. She, J. Kibsgaard, C. F. Dickens, I. Chorkendorff, J. K. Nørskov and T. F. Jaramillo, Science, 2017, 355, eaad4998 CrossRef.
- X. Wang, M. Liu, N. Li, Z. Li, C. Zhang and S. Liu, Adv. Energy Mater., 2025, 15, 2402923 CrossRef CAS.
- H. Ooka, J. Huang and K. S. Exner, Front. Energy Res., 2021, 9, 654460 CrossRef.
- Y. Du, F. Xie, M. Lu, R. Lv, W. Liu, Y. Yan, S. Yan and Z. Zou, Nat. Commun., 2024, 15, 1–11 CAS.
- Y. Zhu, Y. Chen, Y. Feng, X. Meng, J. Xia and G. Zhang, Adv. Mater., 2024, 36, 2401694 CrossRef CAS PubMed.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. De Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.: Condens. Matter, 2009, 21, 395502 CrossRef.
- K. M. Nair, S. Ajith, F. Paul, S. Pallilavalappil, N. Thomas, S. J. Hinder, L. Manjakkal and S. C. Pillai, Small, 2025, 21, 2409959 CrossRef CAS PubMed.
- E. Duquesne, S. Betelu, A. Seron, D. Bricault, M. Goldmann, I. Ignatiadis, D. Limagne, A. Vlad, A. Resta, H. Perrot, O. Sel and C. Debiemme-Chouvy, J. Mater. Chem. A, 2022, 10, 24783–24792 RSC.
- D. Sokol, A. N. Salak, M. G. S. Ferreira, A. Beganskiene and A. Kareiva, J. Sol-Gel Sci. Technol., 2018, 85, 221–230 CrossRef CAS.
- C. Busetto, G. Del Piero, G. Manara, F. Trifirò and A. Vaccari, J. Catal., 1984, 85, 260–266 CrossRef.
- J. Suntivich, K. J. May, H. A. Gasteiger, J. B. Goodenough and Y. Shao-Horn, Science, 2011, 334, 1383–1385 CrossRef CAS PubMed.
- Z. Xu, Y. Ying, G. Zhang, K. Li, Y. Liu, N. Fu, X. Guo, F. Yu and H. Huang, J. Mater. Chem. A, 2020, 8, 26130–26138 RSC.
- K. Momma and F. Izumi, J. Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS.
- S. Sun, X. Zhou, B. Cong, W. Hong and G. Chen, ACS Catal., 2020, 10, 9086–9097 CrossRef CAS.
- Y. Luo, Y. Wu, D. Wu, C. Huang, D. Xiao, H. Chen, S. Zheng and P. K. Chu, ACS Appl. Mater. Interfaces, 2020, 12, 42850–42858 CrossRef CAS PubMed.
- M. Krivec, R. Dillert, D. W. Bahnemann, A. Mehle, J. Štrancar and G. Dražić, Phys. Chem. Chem. Phys., 2014, 16, 14867–14873 RSC.
- G. Bahuguna and F. Patolsky, ACS Mater. Lett., 2024, 6, 3202–3217 CrossRef CAS.
Footnote |
| † In cherished memory of Zubeen Garg – an Aurelian voice that inspired generations and will continue transcending eternity. |
| This journal is © The Royal Society of Chemistry 2026 |