Interfacial charge transfer engineering in V3O5/ZnO p–n heterostructures for enhanced SERS performance
Shan
Liang
 ,
Xiaolan
Wu
,
Chao
Yang
,
Yuhan
Liu
,
Jifei
Wang
,
Meiying
Leng
,
Yaxin
Zhai
,
Xiaolan
Wu
,
Chao
Yang
,
Yuhan
Liu
,
Jifei
Wang
,
Meiying
Leng
,
Yaxin
Zhai
 * and
Xingang
Zhang
*
* and
Xingang
Zhang
*
Key Laboratory of Low-Dimensional Quantum Structures and Quantum Control of Ministry of Education, Key Laboratory for Matter Micro-structure and Function of Hunan Province, Department of Physics and Synergetic Innovation Center for Quantum Effects and Applications, School of Physics and Electronics, Hunan Normal University, Changsha 410081, China. E-mail: zxg@hunnu.edu.cn; yzhai@hunnu.edu.cn
First published on 9th December 2025
Abstract
Surface-enhanced Raman scattering (SERS) substrates based on transition metal oxides have garnered significant attention due to their structural diversity, tunable properties, and unique optical characteristics. However, their practical applications have been severely hindered by relatively weak Raman enhancement effects and poorly understood charge transfer mechanisms. To address these limitations, this study developed a novel charge-transfer-enhanced SERS substrate by constructing a V3O5/ZnO p–n semiconductor heterojunction. The optimized heterostructure demonstrates a detection limit of 10−8 M and an enhancement factor of 5.9 × 104 for methylene blue (MB), while maintaining excellent stability under extreme conditions (30–70 °C and pH 3–10). Using picosecond-resolution transient absorption spectroscopy, the charge transfer dynamics at the heterojunction interface were precisely monitored, providing systematic insights into the microscopic mechanism underlying the Raman enhancement. This study provides a strategy for designing high-performance SERS substrates based on non-precious metals and insights into the charge transfer mechanism revealed by transient absorption spectroscopy.
1. Introduction
Surface-enhanced Raman scattering (SERS), a rapid, non-destructive analytical technique, enables precise molecular identification through vibrational fingerprint detection. Current theoretical frameworks primarily attribute SERS signal amplification to electro-magnetic (EM) and chemical (CM) enhancement mechanisms.1,2 EM originates from surface plasmon resonance (SPR) induced by laser excitation on nanostructured noble metals, generating localized electromagnetic field amplification that elevates molecular Raman signals by 104–1011-fold, constituting the predominant mechanism in SERS.3,4 However, SERS substrates based on noble metals not only face disadvantages such as high manufacturing costs, poor biocompatibility, strong spectral background, and low repeatability, but also lack selectivity for molecular detection, which seriously limits their practical applications.5 CM arises from molecule–substrate charge transfer interactions that elevate molecular polarization, thereby augmenting the Raman scattering cross section through altered electronic states—a mechanism distinct from electromagnetic effects.6,7 Semiconductor materials have become the main research focus for providing CM due to their diverse types and highly adjustable band structures.8–11 CM based SERS substrates have shown significant advantages in biocompatibility and selective detection, but the low Raman enhancement ability has always been a key limiting factor for their applications.In order to prepare high-performance CM based SERS substrates, researchers have extensively studied materials such as semiconductor oxides,12 transition metal sulfides,13,14 two-dimensional materials,15,16 MXenes,17,18etc., exploring their potential as high-performance SERS substrates. Research has found that defects,19,20 element ratios,21,22 element doping,23,24 and crystal phase25,26 in materials can greatly affect their Raman enhancement ability. Rational modulation of material parameters enables significant optimization of substrate–molecule charge-transfer processes, thereby amplifying molecular Raman responses. This principle is exemplified in surface defect engineering strategies, as demonstrated by Cong et al.‘s work, where controlled oxygen vacancies in ultrathin WO3 nanosheets elevated charge-carrier density, triggering interfacial electronic coupling effects that achieved ∼100-fold Raman signal amplification.27 Xu et al. constructed SnSe2−x nanoflake arrays; by adjusting the ratio of Sn and Se, SnSe1.75 markedly improves R6G's Raman spectral response, primarily due to selenium vacancy formation.28 Jiang et al. optimized SnO2's band structure by varying Ce dopant concentration, improving charge transfer between the substrate and target molecule, which led to enhanced Raman signals of the molecule.29 Meng et al. used Xe lamp irradiation to transform crystalline MoO3 into an amorphous state and found that amorphous MoO3 has a higher Raman enhancement ability. This enhancement is attributed to its higher electron delocalization and electronic state density, which facilitate electron transfer between the substrate and target molecules.30
Beyond modifying individual materials, constructing heterojunctions effectively enhances the performance of non-noble-metal SERS substrates. Zhou et al. demonstrated that sponge-like Cu-doped SnO2–NiO p–n heterostructures show superior Raman enhancement, resulting from the combined effects of p–n junction-enhanced charge separation efficiency and Cu doping-induced charge transfer resonance.31 Xie et al. developed TiO2@MoOx nanorod heterojunctions for SERS, achieving a 10−8 M detection limit for R6G. Their mechanistic study revealed that the staggered band alignment in TiO2@MoOx enables photo-generated electron–hole separation in the depletion layer under laser excitation. Electrons migrate to MoOx surfaces and subsequently transfer to adsorbed molecules, amplifying SERS signals.32 This demonstrates that optimizing interfacial charge transfer is critical for enhancing molecular Raman detection.
Herein, we assemble Zn-based metal organic framework (MOF) nanoparticles and V-based MOF nanowires into hybrid MOF-on-MOF heterostructures, which are further annealed to transform into V3O5/ZnO p–n semiconductor heterostructures as an effective charge transfer SERS substrate. Experimentally, methylene blue (MB) served as a Raman probe to assess the SERS activity of materials. Comparative tests under 532 nm laser excitation revealed that the V3O5/ZnO heterojunction outperformed standalone V3O5 nanowires and ZnO nanoparticles, achieving an MB enhancement factor of 5.9 × 104 and a detection limit of 10−8 M. Transient absorption (TA) spectroscopy confirmed that the heterojunction's superior SERS performance stems from intensified charge-transfer interactions with MB, which amplify molecular polarization and boost Raman signals.
2. Results and discussion
2.1 Preparation of V3O5/ZnO heterojunctions
The preparation process of V3O5/ZnO heterojunctions is shown in Fig. 1. Firstly, V-MOF nanowires with uniform structure were synthesized by the hydrothermal method, and then the Zn-MOF was grown on the surface of V-MOF nanowires to construct tomatoes-on-sticks shaped hybrid MOF-on-MOF heterostructures. Finally, after annealing treatment, the hybrid MOF-on-MOF heterostructures were converted into V3O5/ZnO heterojunctions.As shown in Fig. 2a, V-MOF nanowires exhibit uniform surfaces with an average diameter of ∼210 nm via scanning electron microscopy (SEM) analysis. Transmission electron microscopy (TEM) imaging (Fig. 2b and c) corroborates their structural uniformity, aligning with SEM observations. X-ray diffraction (XRD) patterns (Fig. S1) reveal high crystallinity through sharp peaks matching prior reports.33 These V-MOF nanowires were subsequently employed as host frameworks to construct MOF-on-MOF heterostructures. By directly immersing V-MOF nanowires into a methanol solution of 2-methyllimidazole and Zn2+, a V-MOF/Zn-MOF heterostructure was synthesized. As shown in Fig. 2d, each V-MOF nanowire is strung with a number of regular dodecahedron Zn-MOF particles with a size of 400–500 nm, forming a shape similar to the tomatoes-on-sticks string. With the increase of V-MOF nanowire feeding (Fig. S2), the number of Zn-MOF particles on its surface can also be well controlled. The Zn-MOF nanoparticles were synthesized as shown in Fig. S3, presenting a regular dodecahedron. The V-MOF/Zn-MOF heterostructure morphology was confirmed by TEM (Fig. 2e) and EDS mapping (Fig. 2f). XRD analysis (Fig. S4) verified its crystallinity, with all diffraction peaks corresponding to pristine V-MOF and Zn-MOF phases.33,34 The compositions of V-MOF, Zn-MOF, and V-MOF/Zn-MOF were further validated by Raman spectroscopy (Fig. S5), with results consistent with previously reported literature.35–37
 | ||
| Fig. 2 (a) SEM and (b and c) TEM images of V-MOF; (d) SEM, (e) TEM images, and (f) EDS mapping of the V-MOF/Zn-MOF heterostructure. | ||
Through annealing treatment (300 to 500 °C), the V-MOF/Zn-MOF heterostructure is converted into a V3O5/ZnO nanowire heterostructure. After annealing at 600 °C, the V3O5/ZnO nanowires transform into agglomerated nanoparticles (Fig. S6). Moreover, V-MOF and Zn-MOF are transformed into V3O5 (Fig. S7) and ZnO (Fig. S8), respectively, after annealing at 500 °C, while still maintaining their nanowire and nanoparticle structures. The thermal stability of the as-obtained V-MOF/Zn-MOF (annealed at 500 °C) was investigated by thermogravimetric analysis (TGA) (Fig. S9). The results revealed two distinct degradation stages: the first stage (0–100 min) corresponds to the volatilization of solvent molecules (H2O), while the second stage (100–300 min) is attributed to the decomposition of organic linkers and the MOF structure.38 The TEM image indicates that V3O5/ZnO nanowires exhibit a rough surface (Fig. 3a). In the HRTEM image (Fig. 3b), the (002) and (110) crystal planes of ZnO and the (310) crystal plane of V3O5 can be seen, both of which have good crystallinity. The clear boundary indicates the synthesis of the V3O5/ZnO heterojunction. The elemental mapping images of V3O5/ZnO nanowires confirm its hybrid structure (Fig. 3c). The concentrations of Zn, V, and O in V3O5/ZnO are estimated to be 38.7 wt%, 35.9 wt%, and 19.4 wt%, respectively, via a quantitative calculation of the EDX spectrum (Fig. S10). XRD spectra of V3O5/ZnO nanowires were also collected, as shown in Fig. 3d. Specifically, most of the sharp diffraction peaks are attributed to V3O5 (PDF#71-0039), indicating that V3O5 has good crystallinity. Meanwhile, we also observed diffraction peaks of ZnO, which further confirm the synthesis of the V3O5/ZnO heterojunction, consistent with the HRTEM image. XRD analysis of the V-MOF product after high-temperature annealing reveals a composition of V3O5 and V2O3, while the Zn-MOF-derived product is a composite of ZnO and MOF, indicating that 500 °C annealing is insufficient for complete pyrolysis of pure Zn-MOFs (Fig. S11). X-ray photoelectron spectroscopy (XPS) analysis of the V3O5/ZnO heterojunction (Fig. S12) confirmed the presence of Zn, V, O, and C. The Zn 2p XPS spectrum of the sample exhibits a typical spin–orbit splitting with a 2p3/2 peak (blue) at 1020.9 eV and a 2p1/2 peak (green) at 1044.2 eV (Fig. 3e). The corresponding splitting energy of ∼23 eV is consistent with the inherent electronic characteristics of zinc. Here, the 2p3/2 binding energy aligns closely with that of Zn2+ (e.g., ZnO, ∼1021.3 eV) and is significantly lower than that of metallic Zn0 (1021.7 eV), confirming that Zn is in the +2 oxidation state. Additionally, the weak and broad loss feature observed (rather than a sharp plasmon peak) is consistent with the electronic structure of ZnO semiconductor materials, thus providing auxiliary evidence for the existence of Zn2+.39 XPS characterization of the V 2p spectrum (Fig. 3f) reveals that vanadium atoms in the material exhibit mixed valence states of V4+ and V3+. V4+ is identified as the dominant species, with its 2p3/2 peak centered at 516.4 eV and its 2p1/2 peak at 523.9 eV, corresponding to a spin–orbit splitting energy of approximately 7.5 eV. These spectral features are consistent with the typical characteristics of tetravalent vanadium, as exemplified by VO2. V3+ exists as a minor component, showing 2p3/2 and 2p1/2 peaks at 515.6 eV and 522.6 eV, respectively, with a splitting energy of ∼7.0 eV, matching well with the standard binding energies of trivalent vanadium compounds such as V2O3. The estimated area ratio of V4+ to V3+ peaks is 2.07![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, suggesting that the material may consist of a mixed vanadium oxide.40 Notably, compared with the binding energy values of the corresponding oxidation states in the literature,39,41 the binding energy peak of the Zn oxidation state in the heterojunction shows a negative shift of approximately −0.4 eV, while the binding energy peak of the V oxidation state exhibits a slight positive shift (+0.1 eV). These results indicate that the electron density around Zn increases, whereas the electron density around V decreases. This difference in charge distribution promotes the formation of a built-in electric field at the V3O5–ZnO interface, which is directed from V3O5 to ZnO. The O 1s peak exhibits a multi-component structure (Fig. S13). It primarily consists of two well-assigned chemical states: the peak at 529.4 eV is attributed to the Zn–O bond (lattice oxygen in zinc oxide), while the peak at 530.7 eV corresponds to the V–O bond (lattice oxygen in vanadium oxide). Additionally, a weak, unlabeled signal is observed around 531.9 eV, which may originate from minor oxygen species, such as surface-adsorbed hydroxyl groups (–OH) or oxygen defects.39 These findings collectively demonstrate the successful fabrication of V3O5/ZnO nanowire heterostructures.
1, suggesting that the material may consist of a mixed vanadium oxide.40 Notably, compared with the binding energy values of the corresponding oxidation states in the literature,39,41 the binding energy peak of the Zn oxidation state in the heterojunction shows a negative shift of approximately −0.4 eV, while the binding energy peak of the V oxidation state exhibits a slight positive shift (+0.1 eV). These results indicate that the electron density around Zn increases, whereas the electron density around V decreases. This difference in charge distribution promotes the formation of a built-in electric field at the V3O5–ZnO interface, which is directed from V3O5 to ZnO. The O 1s peak exhibits a multi-component structure (Fig. S13). It primarily consists of two well-assigned chemical states: the peak at 529.4 eV is attributed to the Zn–O bond (lattice oxygen in zinc oxide), while the peak at 530.7 eV corresponds to the V–O bond (lattice oxygen in vanadium oxide). Additionally, a weak, unlabeled signal is observed around 531.9 eV, which may originate from minor oxygen species, such as surface-adsorbed hydroxyl groups (–OH) or oxygen defects.39 These findings collectively demonstrate the successful fabrication of V3O5/ZnO nanowire heterostructures.
2.2 SERS performance of V3O5/ZnO heterojunctions
To assess SERS performance, MB was utilized as the probe molecule. In a typical experiment, V3O5/ZnO, V3O5, and ZnO were immersed in a 10−5 M MB aqueous solution for 2 hours, and the corresponding Raman spectra were collected under 532 nm laser excitation. Fig. 4a presents the Raman spectra of MB molecules collected from each sample, and these peak positions are consistent with literature reports.16,22 Meanwhile, the V3O5/ZnO heterojunction has the best Raman enhancement performance, indicating that the heterojunction interaction between V3O5 and ZnO has a potential role in enhancing the Raman spectra of MB molecules. Moreover, we also compared the effect of annealing temperature on the Raman enhancement ability of the V3O5/ZnO heterojunction. As shown in Fig. S14, as the temperature increases from 300 °C to 500 °C, the Raman enhancement ability of the V3O5/ZnO heterojunction gradually improves, which can be attributed to the better crystallinity of the V3O5/ZnO heterojunction at higher temperatures. Thus, we chose samples annealed at 500 °C for subsequent experiments. Subsequently, MB aqueous solutions of different concentrations (10−5–10−9 M) were used to evaluate the detection ability of the V3O5/ZnO heterojunction. From Fig. 4b, it can be seen that even at an MB concentration as low as 10−8 M, the Raman signal can still be resolved. Even on fish surfaces, MB with 10−7 M can also be detected (Fig. S15). Based on the above Raman measurement data, we calculated the enhancement factor (EF) of the V3O5/ZnO heterojunction to be 5.9 × 104 when the concentration of MB is 10−8 M (see the Experimental section). Moreover, to investigate the SERS enhancement capability for other molecules, we detected R6G, malachite green, and methyl orange (common organic dye pollutants) using V3O5/ZnO. The results showed clear Raman peaks even at concentrations as low as 10−6 M (Fig. S16), indicating that the V3O5/ZnO heterojunction's SERS effect is not limited to MB molecules. The above results indicate that the V3O5/ZnO heterojunction has superior Raman enhancement ability for MB molecules.Raman spectra collected from ∼20 random points on the V3O5/ZnO heterojunction were analysed by calculating the standard deviation of the 1623 cm−1 peak intensity, which showed minimal variation (RSD = 4.4%, Fig. 4c), demonstrating uniform SERS activity and homogeneous MB molecule adsorption across the heterojunction. Chemical stability is also an important indicator of SERS substrates, and SERS substrates with good chemical stability can function normally under extreme conditions. Therefore, we evaluated the detection ability of the V3O5/ZnO heterojunction for MB molecules at different pH values (from 3 to 10) and temperatures (30–70 °C). The Raman spectra of MB molecules showed almost no change (Fig. 4d and e). The above results indicate that the V3O5/ZnO heterojunction SERS substrate has potential applications under extreme conditions.
2.3 Enhancement mechanism
To reveal the Raman enhancement mechanism of the V3O5/ZnO heterojunction, electrochemical impedance spectroscopy (EIS) was employed to investigate the resistive behaviour of samples. The Nyquist plots of all samples exhibited semicircles in the high-frequency region and inclined straight lines in the low-frequency region (Fig. S17). The semicircle diameter reflects the charge transfer resistance at the electrolyte/electrode interface, with larger diameters indicating higher resistance. The linear portion corresponds to Warburg diffusion resistance, representing ion diffusion rates. Comparative analysis of the EIS spectra reveals that V3O5/ZnO demonstrates the highest conductivity. The superior electrical conductivity of V3O5/ZnO facilitates efficient electron transfer between the sample and MB molecules. Then, we conducted N2 adsorption–desorption isotherm measurements and analyzed the pore-size distribution of the samples (Fig. S18). The test results demonstrate that the Brunauer–Emmett–Teller (BET) surface areas of V3O5, ZnO, and V3O5/ZnO are 99.01 m2 g−1, 1144.08 m2 g−1, and 120.15 m2 g−1, respectively, with the average pore size primarily distributed in the 2–10 nm range. The ZnO nanoparticles inherit the porous structure of Zn-MOF particles, exhibiting a significantly larger specific surface area. Compared to the individual V3O5, upon combining ZnO with V3O5, the specific surface area is markedly enhanced (from 99.01 m2 g−1 to 120.15 m2 g−1), thereby providing more adsorption sites for target molecules. Additionally, the larger pore size ensures efficient accessibility of target molecules to the active sites within the material. Moreover, ultraviolet-visible (UV-vis) absorption spectra were recorded to investigate the interactions between MB and the V3O5/ZnO heterojunction (Fig. 4f). As we can see, the MB molecule exhibits a distinct absorption peak at 655 nm, accompanied by an absorption shoulder at 615 nm, which is consistent with the literature report.27 Due to the synergistic effects, the V3O5/ZnO heterostructure presents significant broadband absorption in the ultraviolet to near-infrared spectral region. It is noteworthy that upon adsorption of MB onto the heterojunction, the corresponding characteristic absorption peak undergoes significant broadening. This observation suggests that effective electron transfer occurs between the molecule and the semiconductor. Moreover, we compared the adsorption of MB molecules by V3O5, ZnO, and V3O5/ZnO composites using UV-vis absorption spectroscopy. Equal masses of each material were separately added to equal volumes of 10−5 M MB solutions, and the mixtures were stirred at room temperature for 1 h. After centrifugation, the supernatant was collected and analyzed by UV-vis absorption spectroscopy (Fig. S19). The results revealed that V3O5 exhibited the weakest MB adsorption effect, while V3O5/ZnO exhibited comparable adsorption efficiency to ZnO, despite ZnO's significantly higher specific surface area compared to V3O5/ZnO (1144.08 m2 g−1vs. 99.01 m2 g−1). This indicates that V3O5/ZnO has superior adsorption properties for MB molecules. To further understand the possible electron transition mechanism in the heterostructure, the corresponding Tauc plots of bare ZnO and V3O5 nanostructures prepared under the same conditions are displayed in Fig. 4g and h. Taking into account the Tauc eqn (1),42| (αhν)1/γ = A(hν − Eg) | (1) |
| EF = −Vfb(vs. SHE)·e − 4.4 eV | (2) |
The corresponding EF values for V3O5 and ZnO are determined to be −5.24 eV and −4.86 eV, respectively. Considering that the Fermi level of an n-type semiconductor (p-type semiconductor) is slightly lower (higher) than its conduction band bottom (valence band top) by approximately 0.3 eV, combined with the energy band gap obtained above, we can obtain the energy level arrangement of the heterostructure. A typical staggered-gap heterojunction of V3O5/ZnO is depicted in the inset of Fig. 4i, which facilitates the rapid separation of interface photogenerated carriers. Moreover, the bare V3O5 and ZnO present positive and negative slopes in the Mott–Schottky plot, respectively, indicating that V3O5 is a p-type semiconductor, while ZnO is an n-type semiconductor. Therefore, when p-type V3O5 encounters n-type ZnO, the majority carriers (electrons) in ZnO will migrate to V3O5 to obtain a Fermi level equilibrium. As a result, an internal electric field from ZnO toward V3O5 will be established, which will further accelerate the rapid extraction of photoelectrons at the interface, providing an advantageous platform for molecular Raman detection.
Furthermore, to uncover the charge transfer pathways in more depth, we investigated the charge transfer processes in heterostructures by transient absorption (TA) spectroscopy. Fig. 5a compares the TA spectra at 1 ps for thin films of V3O5, MB, and the V3O5/MB heterostructure excited at 3.1 eV. The V3O5 spectrum shows three features: a negative ground-state photo-bleaching (PB) at around 1.7 eV, and two positive peaks at 2.1 eV and higher energies, indicating photo-induced absorption (PIA) from the first excited state to higher states, see Fig. 5b. The MB thin film exhibits a strong negative PB at 2.1 eV and a weak PIA peak at higher energy. In the V3O5/MB heterostructure, the PB and PIA from MB are significantly enhanced, while those from V3O5 are suppressed, indicating efficient charge transfer from V3O5 to MB within our time resolution (∼100 fs). Fig. 5c shows the time evolution of the TA signal in the V3O5/MB heterostructure at 1 ps, 2 ps, and 10 ps. Fig. 5d compares the TA spectra at 1 ps for V3O5, MB, ZnO, and the V3O5/ZnO/MB heterostructure. Due to its UV-range bandgap, the ZnO thin film shows only a broad PIA peak centered at 2.3 eV. In the V3O5/ZnO/MB heterostructure, the PIA peaks of ZnO are significantly enhanced, while the PB and PIA signals from both V3O5 and MB are weakened, indicating charge transfer from V3O5 and MB to ZnO (Fig. 5e). Unlike the rapid charge transfer in the V3O5/MB heterostructure, the PIA signal in the V3O5/ZnO/MB heterostructure increases over the first 10 ps, indicating relatively slower charge transfer, as shown in Fig. 5f. To confirm these findings, we measured the TA spectra for V3O5, MB, ZnO, and their mixed materials in solutions, as shown in Fig. S20. Due to weak molecular interactions, the spectra of the mixed solutions are simply a combination of the components, with no charge-transfer processes observed. The TA spectra indicate significant charge transfer between the V3O5/ZnO heterostructure and the MB molecule, effectively amplifying the molecular polarizability.
3. Conclusions
In summary, V3O5/ZnO heterostructures derived from hybrid MOF-on-MOF heterostructures were prepared. As a Raman enhanced sensor, a detection limit of 1 × 10−8 M and an EF of 5.9 × 104 for MB molecules were achieved on the V3O5/ZnO heterostructure. Meanwhile, the sensor can work normally under strong acidic and alkaline conditions and at high temperatures, showing a wide range of potential applications. The superior SERS properties of the V3O5/ZnO heterostructure are attributed to the chemical enhancement. The charge transfer mechanism between the V3O5/ZnO heterostructure and MB molecules was investigated in detail by TA spectroscopy, and the charge transfer process was observed on the picosecond scale. This study provides strategies for the design of high-performance chemically enhanced Raman substrates, as well as insights into the charge transfer mechanism revealed by TA spectroscopy.4. Experimental section
4.1 Preparation of the V-MOF
The MOF synthesis commenced with preparing an aqueous precursor solution (4 mM) containing stoichiometric VCl3 (Sinopharm Chemical Reagent Co., Ltd) and terephthalic acid under vigorous stirring (30 min, ambient conditions). Subsequent addition of CTAB (Sinopharm Chemical Reagent Co., Ltd) as a structure-directing agent preceded hydrothermal treatment (200 °C, 48 h) in a sealed autoclave. Post-synthesis processing involved (i) cooling to ambient temperature, (ii) triple-phase washing (deionized water/ethanol cycles), and (iii) vacuum drying at 60 °C for 12 h.4.2 Preparation of the Zn-MOF
The Zn-MOF was synthesized via ambient-temperature coprecipitation. Stoichiometric Zn·(NO3)2·6H2O (0.024 M) (Sinopharm Chemical Reagent Co., Ltd) and 2-methylimidazole (2.4 M) (Sinopharm Chemical Reagent Co., Ltd) aqueous solutions were rapidly mixed under vigorous stirring (3 h, 25 °C), yielding a milky suspension. This suspension was centrifuged at 5000 rpm for 5 minutes to separate the precipitate and supernatant. The supernatant was removed and the precipitate was collected. The precipitate was washed three times with methanol solution to remove impurities. Finally, the cleaned precipitate was dried at 60 °C for 12 hours to obtain white Zn MOF powder.4.3 Preparation of the V-MOF/Zn-MOF heterostructure
A certain amount of dried V-MOF was weighed and dispersed in 4.5 mL of methanol. Meanwhile, 138.5 mg of Zn·(NO3)2·6H2O and 154 mg of 2-methylimidazole were dissolved in 4 mL of ethanol solution, respectively. V-MOF solution was added to 2-methylimidazole solution; then the mixed solution was placed on a magnetic stirrer and stirred at 35 rpm for 5 minutes at room temperature to ensure uniform mixing. Afterwards, the Zn·(NO3)2·6H2O solution was quickly added to the mixed solution and stirring was continued at the same speed for 3 hours until the solution turns light green. The solution was centrifuged at 5000 rpm for 5 minutes and then redispersed in 8 mL of ethanol solution; this process was repeated three times, and the precipitate was collected. Finally, the obtained precipitate was dried in a drying oven at 60 °C for 12 hours. Finally, a light green V-MOF/Zn-MOF powder was obtained.4.4 Preparation of the V3O5/ZnO heterostructure
The V-MOF/Zn-MOF heterostructure was placed into a tube furnace, and argon was allowed to flow at room temperature for half an hour; then the high-temperature box furnace was heated up to the required temperature (300–600 °C) at a rate of 5 °C min−1, and maintained at that temperature for 3 hours under an Ar atmosphere. After cooling naturally to room temperature, the brown V3O5/ZnO powder was collected. The preparation methods for V3O5 and ZnO powders is consistent with the procedure described above.4.5 Sample characterization
The morphology and microstructure of samples were characterized using a field-emission transmission scanning electron microscope (SEM450) coupled with an energy-dispersive spectrometer (EDS) and a transmission electron microscope (TEM, Tecnai G2 F20 S-TWIN). The crystal structure and surface chemistry of the samples were characterized by X-ray diffraction (XRD, AXS, D8 Discover) and X-ray photoelectron spectroscopy (XPS, ESCALAB250Xi). Time-resolved spectroscopy was performed on an ultrafast transient absorption spectroscopy system (Femto-TA100). SERS measurements were performed on a Horiba LabRAM HR Evolution, with a 532 nm laser focused through a 100× objective onto the sample. The integration time for SERS measurements was 10 s.4.6 SERS performance evaluation methods
The SERS performance of the substrate was comprehensively evaluated through four key metrics: (1) detection sensitivity (quantified by the limit of detection), (2) enhancement factor (calculated via the ratio of SERS to normal Raman signals), (3) spatial uniformity (assessed via the standard deviation of Raman spectral intensity), and (4) stability under extreme conditions (including exposure to strong acids/bases and elevated temperatures).4.7 Calculation of Raman enhancement factor (EF)
The Raman enhancement factor (EF) of the V3O5/ZnO heterostructure was calculated through the following equation:| (ISERS/NSERS)/(Ibulk/Nbulk) | (3) |
In the SERS experiments, 20 µL of MB solution was drop-cast on a 1 cm2 SERS material followed by gentle drying. NSERS is given by NSERS = cVNAARaman/Asub, where c is the MB concentration, V is the MB droplet volume, NA is the Avogadro constant, ARaman is the laser spot area, and Asub is the substrate area. For Nbulk, 0.1 M MB solution was drop-cast onto bare glass. After gentle drying, bulk MB crystals were formed. The density of MB was used to calculate the number of molecules. Nbulk is given by NSERS = ρhNAARaman/M, where ρ is the density of MB (1.757 g cm−3); the laser penetration depth h can be calculated using the equation h = 2λ/NA2, where NA is the numerical aperture (0.5). M is the molar mass of MB (319.86 g mol−1). Taking all the above mentioned factors into account, the EF in the MB/V3O5/ZnO measurements can be derived as:
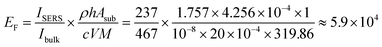 | (4) |
4.8 Transient absorption (TA) measurements
Transient absorption (TA) measurements were conducted using a pump-probe spectrometer setup. Initially, a Ti: sapphire amplifier generated a fundamental laser pulse with a wavelength of 800 nm, operating at a repetition rate of 1 kHz. This fundamental pulse was then split into two branches by a beam splitter. One branch was directed toward a β-barium borate (BBO) crystal for second harmonic generation of the 800 nm pulse to generate an excitation pump at 400 nm. The pump pulse, modulated at a frequency of 500 Hz, underwent attenuation via neutral-density filter wheels. Simultaneously, the other branch of the fundamental pulse was focused into a sapphire crystal to produce a white-light continuum spanning from 350 nm to 1600 nm, utilized as the probe. Time delays between the pump and probe pulses were achieved using a motorized translation stage with a retro-reflecting mirror. TA measurements in the thin films were performed under a microscope. The pump and probe beams were combined using a 425 nm long-pass dichroic mirror, which transmits probe beams with wavelengths longer than 450 nm while reflecting pump beams with wavelengths of 380–410 nm. In this study, the probe pulse spans from 450 nm to 800 nm. Then the combined beams were directed towards the microscope objective (Olympus, 50×magnification, numerical aperture 0.75, and working distance 3 mm). The pump and probe beams were spatially overlapped on the sample surface, both incident on the sample normally. The focused spot size of the pump beam on the sample surface was approximately 1 µm. The transmitted probe beam of the sample was focused onto another 50× objective and collected through a fiber optic spectrometer.Author contributions
Zhang X., Liang S., and Zhai Y. conceived the concept, designed the research, and wrote the manuscript. Wu X. and Yang C. performed the experiments. Liu Y., Wang J. and Zhai Y. conducted transient absorption spectroscopy measurements and analysis. All authors contributed to the general discussion.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the electronic supplementary information (SI). No additional datasets or code were generated or analysed in this study. Supplementary information: SEM images and XRD patterns; Raman spectra and TG/DTG curves of V-MOF/Zn-MOF; EDX spectra and XPS spectrum of V3O5/ZnO heterostructure; SERS spectra of molecules detected on V3O5/ZnO heterostructure; EIS curves, N2 adsorption–desorption isotherms and the pore-size distribution of V3O5, ZnO, and V3O5/ZnO; UV-Vis absorption spectra and time-resolved spectroscopy. See DOI: https://doi.org/10.1039/d5ta04365d.Acknowledgements
This research was supported by the Science & Technology Department (Grant No. 2024JJ5250, 2023ZJ1010 and 2024JJ2041), the Education Department of Hunan Province (Grant No. 23A0047), and the National Natural Science Foundation of China (NSFC) (Grant No. 12204167, 62522404, and 12421005).References
- T. Itoh, M. Procházka, Z. Dong, W. Ji, Y. Yamamoto, Y. Zhang and Y. Ozaki, Chem. Rev., 2023, 12, 1552–1634 CrossRef PubMed.
- H. Ma, S. Pan, W. Wang, X. Yue, X. Xi, S. Yan, D. Wu, X. Wang, G. Liu and B. Ren, ACS Nano, 2024, 18, 14000–14019 CrossRef CAS PubMed.
- E. Ru and B. Auguié, ACS Nano, 2024, 18, 9773–9783 CrossRef PubMed.
- S. Ding, J. Yi, J. Li, B. Ren, D. Wu, R. Panneerselvam and Z. Tian, Nat. Rev. Mater., 2016, 1, 1–16 Search PubMed.
- X. Liu, Z. Ye, Q. Xiang, X. Liu, Z. Ye, Q. Xiang, Z. Xu, W. Yue, C. Li, Y. Xu, L. Wang, X. Cao and J. Zhang, Chem, 2023, 9, 1464–1476 CAS.
- I. Chaudhry, G. Hu, H. Ye and L. Jensen, ACS Nano, 2024, 18, 20835–20850 CrossRef CAS PubMed.
- S. Cong, X. Liu, Y. Jiang, W. Zhang and Z. Zhao, Innovation, 2020, 1, 100051 CAS.
- Q. Wang, K. Chang, Q. Yang and W. Wu, Trends Food Sci. Technol., 2024, 104460 CrossRef CAS.
- S. Li, H. Rao, C. Tang, T. Bao, P. She and J. Qin, Coord. Chem. Rev., 2024, 510, 215803 CrossRef CAS.
- D. Cialla-May, A. Bonifacio, T. Bocklitz, A. Markin, N. Markina, S. Fornasaro, A. Dwivedi, T. Dib, E. Farnesi, C. Liu, A. Ghosh and J. Popp, Chem. Soc. Rev., 2024, 53, 8957–8979 RSC.
- X. Tang, Q. Hao, X. Hou, L. Lan, M. Li, L. Yao, X. Zhao, Z. Ni, X. Fan and T. Qiu, Adv. Mater., 2024, 2312348 CrossRef CAS PubMed.
- S. Cong, Z. Wang, W. Gong, Z. Chen, W. Lu, J. Lombardi and Z. Zhao, Nat. Commun., 2019, 10, 678 CrossRef CAS PubMed.
- Z. Li, L. Zhai, Q. Zhang, W. Zhai, P. Li, B. Chen, C. Chen, Y. Yao, Y. Ge, H. Yang, P. Qiao, J. Kang, Z. Shi, A. Zhang, H. Wang, J. Liang, J. Liu, Z. Guan, L. Liao, V. Neacsu, C. Ma, Y. Chen, Y. Zhu, C. Lee, L. Ma, Y. Du, L. Gu, J. Li, Z. Tian, F. Ding and H. Zhang, Nat. Mater., 2024, 23, 1–8 CrossRef PubMed.
- Y. Peng, C. Lin, Y. Li, Y. Gao, J. Wang, J. He, Z. Huang, J. Liu, X. Luo and Y. Yang, Matter, 2022, 5, 694–709 CrossRef CAS PubMed.
- J. Jin, Z. Guo, D. Fan and B. Zhao, Mater. Horiz., 2023, 10, 1087–1104 RSC.
- Q. Lv, J. Tan, Z. Wang, P. Gu, H. Liu, L. Yu, Y. Wei, L. Gan, B. Liu, J. Li, F. Kang, H. Cheng, Q. Xiong and R. Lv, Nat. Commun., 2023, 14, 2717 CrossRef CAS PubMed.
- Z. He, T. Rong, Y. Li, J. Ma, Q. Li, F. Wu, Y. Wang and F. Wang, ACS Nano, 2022, 16, 4072–4083 CrossRef CAS PubMed.
- G. Yang, F. Liu, J. Zhao, L. Fu, Y. Gu, L. Qu, C. Zhu, J. Zhu and Y. Lin, Chem. Rev., 2023, 479, 215002 CAS.
- P. Zuo, L. Jiang, X. Li, P. Ran, B. Li, A. Song, M. Tian, T. Ma, B. Guo, L. Qu and Y. Lu, Nanoscale, 2019, 11, 485–494 RSC.
- G. Song, S. Cong and Z. Zhao, Chem. Sci., 2022, 13, 1210–1224 RSC.
- Y. Liu, Z. Gao, M. Chen, Y. Tan and F. Chen, Adv. Funct. Mater., 2018, 28, 1805710 CrossRef.
- X. Tang, X. Fan, J. Zhou, S. Wang, M. Li, X. Hou, K. Jiang, Z. Ni, B. Zhao, Q. Hao and T. Qiu, Nano Lett., 2023, 23, 7037–7045 CrossRef CAS PubMed.
- Z. Zheng, S. Cong, W. Gong, J. Xuan, G. Li, W. Lu, F. Geng and Z. Zhao, Nat. Commun., 2017, 8, 1993 CrossRef PubMed.
- Q. Lv, J. Tan, Z. Wang, L. Yu, B. Liu, J. Lin, J. Li, Z. Huang, F. Kang and R. Lv, Adv. Funct. Mater., 2022, 32, 2200273 CrossRef CAS.
- Y. Zhang, Z. Shi, H. Cui, Q. Xia, F. Liu, Z. Wang, J. Wang, H. Fan, C. Shu, B. Chen, H. Li, Z. Lai, Z. Luo, W. Zheng, L. Wang and Z. Huang, Nano Lett., 2024, 24, 14293–14301 CrossRef CAS PubMed.
- L. Tao, Z. Li, K. Chen, Y. Zhou, H. Li, X. Wang, R. Zhan, X. Hou, Y. Zhao, J. Xu, T. Qiu, X. Wan and J. Xu, Cell Rep. Phys. Sci., 2021, 2, 100526 CrossRef CAS.
- G. Song, W. Gong, S. Cong and Z. Zhao, Angew. Chem., 2021, 60, 5505–5511 CrossRef CAS PubMed.
- G. Xu, R. Dong, D. Gu, H. Tian, L. Xiong, Z. Wang, W. Wang, Y. Shao, W. Li, G. Li, X. Zheng, Y. Yu, Y. Feng, Y. Dong, G. Zhong, B. Zhang, W. Li, L. Wei, C. Yang and M. Chen, J. Phys. Chem. Lett., 2022, 13, 1453–1463 CrossRef CAS PubMed.
- L. Jiang, J. Zhang, H. Li, C. He, R. Yuan, Y. Zhao, J. Huang and X. Yang, Chem. Eng. J., 2023, 461, 142102 CrossRef CAS.
- X. Meng, J. Yu, W. Shi, L. Qiu, K. Qiu, A. Li, L. Zhen, Y. Wang, J. Wu, J. Lin, X. Wang and L. Guo, Angew. Chem., 2024, 136, e202407597 CrossRef.
- Y. Zhou, Q. Gu, T. Qiu, X. He, J. Chen, R. Qi, R. Huang, T. Zheng and Y. Tian, Angew. Chem., 2021, 133, 26464–26471 CrossRef.
- S. Xie, K. Lai, C. Gu, T. Jiang, L. Zhou, X. Zheng, X. Shen, J. Han and J. Zhou, Mater. Today Nano, 2022, 18, 100179 CrossRef CAS.
- H. Luo, B. Wang, F. Wang, J. Yang, F. Wu, Y. Ning, Y. Zhou, D. Wang, H. Liu and S. Dou, ACS Nano, 2020, 14, 7328–7337 CrossRef CAS PubMed.
- R. Li, S. Chen, X. Zhang, F. Zeng, X. Song, J. Yin and C. Jiang, Sci. China Mater., 2024, 67, 2985–2994 CrossRef CAS.
- G. Kumari, K. Jayaramulu, T. Maji and C. Narayana, J. Phys. Chem., 2013, 117, 11006–11012 CrossRef CAS PubMed.
- Y. Ma, D. Xiong, Y. Meng, Y. Lu, P. Duan, B. Chen, Y. Yang, X. Chen, L. Mao, X. Wu and L. Yang, Sci. China Mater., 2025, 1–11 Search PubMed.
- L. Colombo, V. VolovSek, K. Furik and J. Durig, J. Raman Spectrosc., 1990, 21, 169–175 CrossRef CAS.
- X. Li, F. Zhang, M. Zhang, X. Zhou and H. Zhang, ACS Omega, 2021, 6, 35548–35558 CrossRef CAS PubMed.
- R. AlGaashani, S. Radiman, A. Daud, N. Tabet and Y. Al-Douri, Ceram. Int., 2013, 39, 2283–2292 CrossRef CAS.
- G. Silversmit, D. Depla, H. Poelman, G. Marin and R. Gryse, J. Electron Spectrosc. Relat. Phenom., 2004, 135, 167–175 CrossRef CAS.
- S. K. Das, S. K. Nandi, C. V. Marquez, A. Rua, M. Uenuma, E. Puyoo, S. K. Nath, D. Albertini, N. Baboux, T. Lu, Y. Liu, T. Haeger, R. Heiderhoff, T. Riedl, T. Ratcliffe and R. G. Elliman, Adv. Mater., 2023, 35, 2208477 CrossRef CAS PubMed.
- M. Tarannum and S. Egusa, J. Phys. Chem. C, 2024, 128, 4125–4223 CrossRef.
- L. Zhang, J. Ran, S. Z. Qiao and M. Jaroniec, Chem. Soc. Rev., 2019, 48, 5184–5206 RSC.
| This journal is © The Royal Society of Chemistry 2026 |






