DOI:
10.1039/D5SM01094B
(Paper)
Soft Matter, 2026,
22, 2270-2278
Osmotic forces modify lipid membrane fluctuations
Received
30th October 2025
, Accepted 30th January 2026
First published on 31st January 2026
Abstract
In hydrodynamic descriptions of lipid bilayers, the membrane is often approximated as being impermeable to the surrounding, solute-containing fluid. However, biological and in vitro lipid membranes are influenced by their permeability and the resultant osmotic forces—whose effects remain poorly understood. Here, we study the dynamics of a fluctuating, planar lipid membrane that is ideally selective: fluid can pass through it, while solutes cannot. We find that the canonical membrane relaxation mode, in which internal membrane forces are balanced by fluid drag, no longer exists over all wavenumbers. Rather, this mode only exists when it is slower than solute diffusion—corresponding to a finite range of wavenumbers. The well-known equipartition result, quantifying the size of membrane undulations due to thermal perturbations, is consequently limited in its validity to the aforementioned range. Moreover, this range shrinks as the membrane surface tension is increased, and above a critical tension, the membrane mode vanishes. Our findings are relevant when interpreting experimental measurements of membrane fluctuations, especially in vesicles at moderate to high tensions.
Introduction
Biological membranes are unique materials that surround the cell and compartmentalize its internal organelles. While such membranes are slowly permeable to water,1 large neutral molecules (such as sugars) cannot easily pass through them.2 The semipermeability of lipid membranes is relevant to both biological and in vitro scenarios—as in both cases, membranes are essentially always surrounded by solutes. While the general equations governing the coupled membrane, fluid, and solute dynamics were recently obtained,3 there is not yet a systematic characterization of membrane behavior. As a result, the effects of permeability and osmosis on lipid membrane dynamics remain poorly understood.
The present study investigates how planar lipid membranes act on, and are influenced by, their surrounding environment. Flat membranes are found in various biological settings, including the endoplasmic reticulum,4 Golgi complex,5 and neuromuscular junction.6 In addition, theories of impermeable bilayer fluctuations about a flat surface are often used to describe the thermal undulations of giant unilamellar vesicles (GUVs). We begin by investigating an impermeable membrane, where the dynamics of the solute are not considered. By starting with a well-understood system (see, e.g., ref. 7–19), we are better equipped to study our primary system of interest: the semipermeable membrane. The temporal evolution of the normal modes is determined for the linearized dynamics, and the relative time scale of membrane and solute relaxation is found to govern the nature of the coupled dynamics. When solutes relax more quickly, the dynamics are indistinguishable from the impermeable scenario. It is then straightforward to present an effective Langevin equation including thermal perturbations, and obtain the magnitude of the resultant height fluctuations. In contrast, when the membrane relaxes more quickly than the surrounding solutes, the resultant dynamics are qualitatively different from their impermeable counterpart. As a result, the character of thermal undulations—long assumed to be that of an impermeable membrane when analyzing fluctuating GUVs—is no longer known. Experimental consequences of our findings are highlighted, and avenues for future work are discussed.
The impermeable membrane
Consider a nearly planar lipid membrane surrounded by water. For the present analysis, we neglect solutes and make the standard approximation that the membrane is impermeable to fluid. Water is characterized by its three-dimensional mass density ρf, shear viscosity μf, and kinematic viscosity νf := μf/ρf. Relevant membrane parameters are the patch length ℓc, two-dimensional (2D) areal density ρm, surface tension λc, and bending modulus kb.20 It is important to note that surface tension is not a membrane property, but rather takes the requisite value to enforce areal incompressibility.21,22 As a result, the membrane surface tension can span a wide range of values and is often tuned in experiments.23,24 Characteristic values of all parameters are provided in Table 1.
Table 1 Membrane, fluid, and solute parameters
| Parameter |
Sym. |
Value |
Ref. |
| Patch size |
ℓ
c
|
102–103 nm |
4 and 6
|
| Density |
ρ
m
|
10−8 pg nm−2 |
28
|
| Bending modulus |
k
b
|
102 pN nm |
29
|
| Surface tension |
λ
c
|
10−4–10−1 pN nm−1 |
29,30
|
| Impermeability |
κ
|
103–105 pN μs nm−3 |
1,31
|
| Density |
ρ
f
|
10−9 pg nm−3 |
— |
| Shear viscosity |
μ
f
|
10−3 pN μs nm−2 |
— |
| Kinematic viscosity |
ν
f
|
106 nm2 μs−1 |
— |
| Concentration |
c
0
|
10−1 nm−3 |
32
|
| Diffusivity |
D
|
103 nm2 µs−1 |
33
|
Our base state is an unperturbed membrane sheet lying in the x–y plane, surrounded on both sides by a semi-infinite stationary fluid. We seek to understand how the coupled membrane and fluid system responds to small perturbations. To this end, we determine the linearized dynamics of the coupled membrane and fluid system about the base configuration. In what follows, an abbreviated analysis of the governing equations is presented; see Section S2 of the SI25 for further details.
The governing equations
The position x of the perturbed membrane is given by x(x,y,t) = xex + yey + h(x,y,t)ez, where x,y ∈ [0,ℓc] and |h| ≪ ℓc by construction. For planar geometries with a stationary base state, the in-plane and out-of-plane membrane equations are decoupled.26,27 To solve for the time evolution of the membrane height in the absence of thermal fluctuations, we require only the z-component of the linear momentum balance of the membrane—often called the shape equation and given by27| |  | (1) |
In eqn (1), α,β ∈ 1,2 denote directions in the x–y plane, with repeated indices summed over such that (for example) h,αα = h,xx + h,yy. Here, (·),t := ∂(·)/∂t is the partial time derivative and ⟦p⟧ := p+ − p− is the difference in the fluid pressure above (+) and below (−) the membrane. The pressure and velocity fields, p± and vj± (where Roman indices span {1,2,3} and repeated indices are summed over), satisfy the incompressible Navier–Stokes equations linearized about the base state: v±j,j = 0 and ρfv±j,t = μfv±j,kk − p±,j. With the no-slip boundary condition h,t = v±z at the membrane surface, water cannot flow through the membrane and we have a well-posed set of equations for the coupled system (ref. 25, Section 2).
At this point, we follow standard approaches to solve for the dynamics of the system. The membrane height is decomposed into planar Fourier modes as
| | 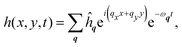 | (2) |
where the wavevector
q = (
qx,
qy) has corresponding wavenumber
q := (
qx2 +
qy2)
1/2 ∼ 10
−4–10
−1 nm
−1,
ωq is the associated relaxation frequency, and normal modes are assumed to be independent. Fluid unknowns are similarly decomposed into planar Fourier modes, with the functional dependence on
z to be solved for. The fluid pressure, for example, is expressed as
| | 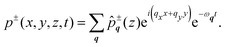 | (3) |
Our main task is to solve for all frequencies
ωq satisfying the governing equations. In doing so, we also solve for the pressure and velocity fields in the fluid.
The frequency solutions
As we limit ourselves to the linearized dynamics, all unknowns of a particular wavevector q are proportional to the membrane height ĥq. We assume ĥq ≠ 0 and combine all equations governing the membrane and fluid into a single equation for ωq, namely, the dispersion relation (see Section S2.3 of the SI25). Given the physically motivated requirement that all fluid perturbations decay far from the membrane surface, there are two frequencies at each wavenumber, which are plotted in Fig. 1. The aforementioned equation for ωq is not straightforward to interpret—though we find that the dynamics are well-approximated by| | | ρeffω2q− 4μfqωq + E = 0. | (4) |
 |
| | Fig. 1 Plot of the two impermeable frequency solutions as a function of wavenumber. Blue dashed and red dotted lines are the exact inertial and membrane branches, respectively, obtained numerically. The approximate solutions in eqn (5) are shown as thick, transparent bands. Relevant parameters are ρf = 10−9 pg nm−3, νf = 9 × 10−5 nm2 μs−1, ρm = 10−8 pg nm−2, λc = 2 × 10−3 pN nm−1, and kb = 102 pN nm. | |
Eqn (4), with the temporal ansatz h ∼ e−ωt in eqn (2), shows that the dynamics of the membrane are analogous to that of the spring–mass–damper from introductory mechanics. Here, ρeff := ρm + 4ρf/q ∼ 10−7–10−5 pg nm−2 approximates the combined inertia of the membrane and fluid, 4μfq ∼ 10−7–10−4 pN μs nm−3 is the well-known hydrodynamic drag from the surrounding fluid, and E := λcq2 + ½kbq4 ∼ 10−12–10−2 pN nm−3 captures the energetics of small membrane fluctuations: ½ℓc2E|ĥq2| is the energy of mode q to quadratic order.34 As the system is overdamped, 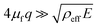 and the two solutions to eqn (4) are well-approximated by
and the two solutions to eqn (4) are well-approximated by
| | 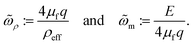 | (5) |
In
eqn (5), the ‘tilde’ accent indicates that
eqn (4) is only an approximation of the true dynamics; the subscripts
ρ and m designate the inertial and membrane branches. As shown in
Fig. 1, the frequencies in
eqn (5) are a good approximation to the true solution.
The membrane response.
Following eqn (4) and (5), let us denote the two exact frequency branches as ωρ and ωm. The membrane height, in real space, is then given by [cf.eqn (2)]| | 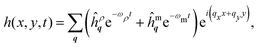 | (6) |
where ĥρq and ĥmq are determined upon specification of the initial conditions: h and ∂h/∂t at time t = 0. Since ωρ ≫ ωm over all wavenumbers, the inertial branch quickly decays and the dynamics of the coupled system are captured by the membrane branch. Prior studies are accordingly justified in neglecting inertial effects throughout their investigations of planar lipid membrane behavior.7–19,35–39
The fluid response.
To understand the dynamics of the fluid as the membrane relaxes, consider two disturbances to a specific mode q. In the first case, ĥmq = 0 and only the inertial mode is excited, while in the second case, ĥρq = 0 and only the membrane mode is excited [see eqn (6)]. In both cases, the pressure field (3) is given by ![[p with combining circumflex]](https://www.rsc.org/images/entities/i_char_0070_0302.gif) ±q = ∓½(ρmωq2 + E)ĥqe−q|z|. The magnitude of the pressure at z = 0 is determined using eqn (1), and the decay length 1/q is a consequence of the pressure field being harmonic: p±,jj = 0. In contrast, fluid velocities decay over the two length scales 1/q and 1/kν, where kν := (q2 − ω/νf)1/2 is a modified wavenumber that accounts for the inertia of the fluid (see Section S94 of ref. 40 and Section S2.1 of the SI25). As a consequence of the time scale separation ωρ ≫ ωm, 1/
±q = ∓½(ρmωq2 + E)ĥqe−q|z|. The magnitude of the pressure at z = 0 is determined using eqn (1), and the decay length 1/q is a consequence of the pressure field being harmonic: p±,jj = 0. In contrast, fluid velocities decay over the two length scales 1/q and 1/kν, where kν := (q2 − ω/νf)1/2 is a modified wavenumber that accounts for the inertia of the fluid (see Section S94 of ref. 40 and Section S2.1 of the SI25). As a consequence of the time scale separation ωρ ≫ ωm, 1/![[q with combining macron]](https://www.rsc.org/images/entities/i_char_0071_0304.gif) ≈1/kν on the membrane branch and 1/
≈1/kν on the membrane branch and 1/![[q with combining macron]](https://www.rsc.org/images/entities/i_char_0071_0304.gif) ≪ 1/kν on the inertial branch.
≪ 1/kν on the inertial branch.
The semipermeable membrane
With an understanding of the impermeable membrane, we are primed to investigate the primary system of interest: a semipermeable membrane, in which fluid flows through the membrane, while solutes dissolved in the fluid do not. Solutes are characterized by a base concentration c0 ∼ 0.1 nm−3 (ref. 32) and a diffusion constant D ∼ 103 nm2 μs−1.33 In addition, the membrane impermeability κ relates the flux of water through the bilayer to its driving force;3 here, κ ∼ 103–105 pN μs nm−3.1,31 Dynamical effects from water permeation are of order μfq/κ: a dimensionless parameter that we call the permeability number 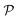 , ranging from 10−7 to 10−12 over wavenumbers q ∈ (10−4, 10−1) nm−1. We will show
, ranging from 10−7 to 10−12 over wavenumbers q ∈ (10−4, 10−1) nm−1. We will show  in the limit of an impermeable membrane; lipid bilayers are thus nearly impermeable.
in the limit of an impermeable membrane; lipid bilayers are thus nearly impermeable.
The governing equations
The membrane shape equation (1) remains unchanged in the presence of solutes.3 The perturbed concentrations c± above and below the membrane, with |c±| ≪ c0, evolve according to the diffusion equation c±,t = Dc±,jj. Solute impermeability is enforced by requiring the total solute flux through the membrane—comprising diffusive and convective contributions—to be zero:| | | −Dc±,z + c0 (v±z − h,t) = 0. | (7) |
In eqn (7), velocities and concentration gradients are evaluated at the membrane surface. The equation governing water permeation through an arbitrarily curved and deforming lipid membrane was recently obtained by A. M. Alkadri and K. K. Mandadapu,3 who extended several seminal findings.41–43 These authors used techniques from irreversible thermodynamics and differential geometry to show that fluid flow through the membrane is driven by differences in hydrodynamic tractions and osmotic pressures across the membrane surface. For nearly planar geometries and an ideally selective membrane, the findings from ref. 3 simplify to| | | κ(v±z − h,t) = kBϑ⟦c⟧ − ⟦p⟧, | (8) |
where kB is Boltzmann's constant, ϑ is the absolute temperature, and ⟦c⟧ := c+ − c− is the jump in solute concentration at the membrane surface. In the language of irreversible thermodynamics,44,45eqn (8) presents a linear relationship between the thermodynamic driving force (kBϑ⟦c⟧ − ⟦p⟧) and the corresponding thermodynamic flux (v±z − h,t), with the latter being a measure of fluid flow through the membrane. The phenomenological parameter κ, understood as a transport coefficient,3 captures the resistance to flow: for a given driving force, a larger value of κ corresponds to a smaller water flux through the membrane. Eqn (8) is equivalently expressed as v±z − h,t = (kBϑ⟦c⟧ − ⟦p⟧)/κ; in the limit of κ → ∞ or  we find v±z → h,t and recover the impermeable scenario.
we find v±z → h,t and recover the impermeable scenario.
The diffusive time scale.
The presence of solutes in the surrounding fluid introduces a diffusive frequency| | ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D := q2D. D := q2D. | (9) |
Fig. 2a shows ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D plotted on top of the curves in Fig. 1; note that the diffusive and impermeable membrane frequencies can be comparable. Let us denote q±0 as the two wavenumbers at which the diffusive and membrane time scales are equal. From eqn (5) and (9), we find
D plotted on top of the curves in Fig. 1; note that the diffusive and impermeable membrane frequencies can be comparable. Let us denote q±0 as the two wavenumbers at which the diffusive and membrane time scales are equal. From eqn (5) and (9), we find| |  | (10) |
with ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m <
m < ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D when q ∈ (q−0,q+0), and
D when q ∈ (q−0,q+0), and ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m >
m > ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D otherwise. Since the base surface tension λc ∼ 10−4–10−1 pN nm−1 is not a material parameter, but rather varies—and can be tuned—across systems, q±0 is plotted at different tensions λc in Fig. 2b to reveal a dome-like envelope. There exists a critical base tension
D otherwise. Since the base surface tension λc ∼ 10−4–10−1 pN nm−1 is not a material parameter, but rather varies—and can be tuned—across systems, q±0 is plotted at different tensions λc in Fig. 2b to reveal a dome-like envelope. There exists a critical base tension| |  | (11) |
at the top of the dome, above which no real q0 exists. We also note that the bilayer tears at the lysis tension λℓ ∼ 1–10 pN nm−1.1 Thus, when the base membrane tension λc lies between  and λℓ, we have
and λℓ, we have ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m >
m > ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D over all wavenumbers. As we will see, the relative values of
D over all wavenumbers. As we will see, the relative values of ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m and
m and ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D—and thus whether a mode is “inside” (i.e. below) or “outside” (i.e. above) the dome in Fig. 2b—govern different regimes of membrane behavior.
D—and thus whether a mode is “inside” (i.e. below) or “outside” (i.e. above) the dome in Fig. 2b—govern different regimes of membrane behavior.
 |
| | Fig. 2 Relevance of the diffusive time scale to the dynamics of the semipermeable system. (a) An overlay of ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D (solid green line) on top of the impermeable frequencies in Fig. 1. There are two wavenumbers for which the impermeable membrane frequency ωimpm equals D (solid green line) on top of the impermeable frequencies in Fig. 1. There are two wavenumbers for which the impermeable membrane frequency ωimpm equals ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D; approximating the former as D; approximating the former as ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m yields the two crossover wavenumbers q−0 and q+0(10)—shown as vertical, dashed lines. (b) Plot of q±0 over a range of the base membrane tension λc, which varies across systems. The surface tension from (a) of λc = 2 × 10−3 pN nm−1 is shown as the horizontal dashed line. There exists a critical tension m yields the two crossover wavenumbers q−0 and q+0(10)—shown as vertical, dashed lines. (b) Plot of q±0 over a range of the base membrane tension λc, which varies across systems. The surface tension from (a) of λc = 2 × 10−3 pN nm−1 is shown as the horizontal dashed line. There exists a critical tension 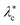 (11) above which (11) above which ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m > m > ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D for all wavenumbers. (c) Plot of the real parts of the semipermeable frequencies (dotted or dashed solid lines), overlaid on top of the impermeable frequencies (thick, transparent bands) for comparison. The inertial branch is essentially unchanged. The membrane branch only exists when ωm < D for all wavenumbers. (c) Plot of the real parts of the semipermeable frequencies (dotted or dashed solid lines), overlaid on top of the impermeable frequencies (thick, transparent bands) for comparison. The inertial branch is essentially unchanged. The membrane branch only exists when ωm < ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D and is thus confined to the range q ∈ (q−0,q+0). For base membrane tensions D and is thus confined to the range q ∈ (q−0,q+0). For base membrane tensions  , the membrane branch vanishes altogether. , the membrane branch vanishes altogether. | |
The frequency solutions.
Membrane and fluid unknowns are expressed as in the impermeable analysis, with the concentration expanded in planar normal modes as [cf.eqn (3)]| |  | (12) |
In our linearized description, all perturbed bulk quantities are again proportional to the membrane height. Assuming a nontrivial solution (ĥq ≠ 0), it is straightforward to arrive at the dispersion relation, as shown in Section S3.3 of the SI.25 While ωq cannot be solved for analytically, we take advantage of the smallness of  : since the membrane is nearly impermeable, semipermeable frequencies are close to their impermeable counterparts. In what follows, we investigate how the dynamics of the impermeable branches are altered by the presence of solutes in the surrounding medium.
: since the membrane is nearly impermeable, semipermeable frequencies are close to their impermeable counterparts. In what follows, we investigate how the dynamics of the impermeable branches are altered by the presence of solutes in the surrounding medium.
The inertial branch.
We begin by assuming that concentration modes evolve with the impermeable inertial frequency ωimpρ, such that for a single mode, c±,t = −ωimpρc± (see eqn (12)). As a consequence, the diffusion equation c±,t = Dc±,jj for the mode q simplifies to −ωimpρĉ±q = D(−q2ĉ±q+ d2ĉ±q/dz2)—which after some rearrangement yields| |  | (13) |
is a positive, real, dimensionless parameter defined for notational convenience. Eqn (13) admits the two purely oscillatory solutions ĉ+q ∼ e±iq![[small eta, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e110.gif) z. As these standing waves do not decay as z → ∞, we seek the lowest-order concentration correction due to the semipermeability of the membrane.†
z. As these standing waves do not decay as z → ∞, we seek the lowest-order concentration correction due to the semipermeability of the membrane.†
We first recognize that water permeation alters the hydrodynamic drag on the membrane by the surrounding fluid. Eqn (7) and (8) together yield ⟦p⟧ = kBϑ⟦c⟧ − (κD/c0)c,±z, which—along with eqn (1)—shows that standing waves in the concentration add a hydrodynamic drag force 90° out of phase with the membrane velocity. We show in the SI (ref. 25, Section 3.2) that an approximate dispersion relation for the semipermeable membrane is given by [cf. eqn (4)]
| |  | (14) |
In
eqn (14),

is a dimensionless parameter that arises when
eqn (7) and (8) are combined. The solution of
eqn (14) closest to
ωimpρ, to lowest order in the product

of small parameters, is given by
| |  | (15) |
Membrane permeability and osmotic forces accordingly introduce exceedingly slow temporal oscillations to the inertial dynamics of the coupled system.
The modification to the inertial frequency in eqn (15) also alters the spatial concentration of the solutes. Substituting eqn (15) into the diffusion equation and solving for the concentration field yield
| |  | (16) |
Importantly,
ĉ+q → 0 as
z → ∞ and concentration perturbations due to the fluctuating membrane decay far from the membrane surface. We have thus found a physically meaningful solution for both the frequency
(15) and concentration
(16) over all wavenumbers. The validity of our approximate analysis is confirmed in
Fig. 2c: the impermeable branch is visually indistinguishable from the real portion of its semipermeable counterpart.
The membrane branch when diffusion is slow.
As in the analysis of the inertial branch, we start by assuming that the solute concentration evolves at the frequency of the impermeable system—in this case, the membrane frequency ωimpm. For wavenumbers q ∉ (q−0,q+0), diffusion is slow relative to the membrane and ωimpm > ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D. The solute diffusion equation once again simplifies to eqn (13), where now
D. The solute diffusion equation once again simplifies to eqn (13), where now ![[small eta, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e110.gif) involves ωimpm instead of ωimpρ. With this change, and following a similar analysis to that described in the inertial scenario, we once again arrive at eqn (14). The membrane frequency is then found to be given by
involves ωimpm instead of ωimpρ. With this change, and following a similar analysis to that described in the inertial scenario, we once again arrive at eqn (14). The membrane frequency is then found to be given by| |  | (17) |
Importantly, the sign of the imaginary part of ωm is opposite to that of ωρ [cf.eqn (15)], and the out-of-phase drag force alters the inertial and membrane dynamics in different ways. Substituting eqn (17) into the diffusion equation gives the concentration field [cf.eqn (16)],| |  | (18) |
The concentration field in eqn (18) diverges as z → ∞ and is not physically meaningful. As a consequence, the membrane branch vanishes when q ∉ (q−0,q+0). A representative result at low tension is shown in Fig. 2(c), and at tensions larger than  (11), the membrane branch vanishes entirely.
(11), the membrane branch vanishes entirely.
The membrane branch when diffusion is fast.
When ωimpm < ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D and q∈ (q−0,q+0), the analysis of the coupled system is straightforward in the limit of small
D and q∈ (q−0,q+0), the analysis of the coupled system is straightforward in the limit of small  . The diffusion equation, assuming ω = ωimpm at lowest order, simplifies to d2ĉ±q/dz2 = (q2 − ωimpm/D)ĉ±q. We thus find the exponentially decaying concentration solutions
. The diffusion equation, assuming ω = ωimpm at lowest order, simplifies to d2ĉ±q/dz2 = (q2 − ωimpm/D)ĉ±q. We thus find the exponentially decaying concentration solutions| |  | (19) |
With no oscillations in the lowest-order concentration field, modifications to the hydrodynamic drag felt by the membrane are in phase with the membrane velocity. Moreover, as such corrections are of order 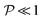 , we expect the membrane branch to be indistinguishable from its impermeable counterpart—as confirmed in Fig. 2(c).
, we expect the membrane branch to be indistinguishable from its impermeable counterpart—as confirmed in Fig. 2(c).
Theoretical implications.
As mentioned previously, solutes are present in the fluid surrounding nearly all artificial and biological membranes. Our findings demonstrate that such systems can only be approximated as impermeable for wavenumbers “inside the dome,” where q ∈ (q−0,q+0) and there is a slow frequency ω ≈ E/(4μfq). In contrast, only a quickly decaying inertial mode exists for wavenumbers q∉ (q−0,q+0) “outside the dome.” For this case, (i) the membrane response is independent of kb and λc at linear order and (ii) thermal perturbations (discussed subsequently) drive membrane undulations to continuously grow until saturated by nonlinear forces—both of which are surprising results.
The predictions described above motivate us to question whether our normal mode ansatz is indeed appropriate outside the dome. Here, we examine this issue from a statistical mechanical perspective. By starting with a microscopic Hamiltonian of the lipid, water, and solute molecules, one can integrate over fast degrees of freedom to obtain a free energy functional that depends only on the concentration field and membrane height.46 When solutes are fast relative to the membrane, concentration degrees of freedom can also be integrated over to yield a free energy which depends only on the membrane height. We thus recover a description similar to that of the impermeable scenario. If instead solutes are slow relative to the membrane, the concentration field is not equilibrated for a given, instantaneous bilayer shape. Upon integrating over the slow solute degrees of freedom, the resultant membrane equation inerits this slowness via a memory kernel—for which the time evolution of the membrane depends on its history, and there is no associated free energy functional or equipartition result.47 While such a result is incompatible with our normal mode ansatz, it justifies the membrane behaving qualitatively differently inside and outside the dome. Further analysis, likely involving nonlinear simulations of the membrane and surrounding fluid,48–54 is required to understand the dynamics of the coupled system. In any case, however, we emphasize that the standard impermeable result is not applicable for wavenumbers outside the dome.
Experimentally characterizing GUVs
In experimental investigations of lipid bilayers, one often seeks to characterize membrane material properties prior to further manipulation. When GUVs are involved, their fluctuations from thermal disturbances can be imaged and—with a theory of membrane undulations—processed to determine kb and λc.29,31,55–61 While most experimental investigations apply the well-understood impermeable theory over all wavenumbers, the present study reveals that only modes inside the dome should be treated as impermeable. Moreover, as the behavior of undulations outside the dome is not currently well-understood, experimental measurements involving these modes should be excluded when determining membrane properties. In what follows, we discuss how to incorporate thermal perturbations into our theory and then analyze GUV fluctuation data from experiments.
The impermeable Langevin equation.
When the lipid bilayer is treated as impermeable, its linear response to perturbations is well-known. Since inertial modes decay much more quickly than their membrane counterparts (see eqn (6) with ωρ ≫ ωm), inertia can be neglected entirely; the membrane frequency ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m presented in eqn (5) is then exact. Decomposing the membrane height as
m presented in eqn (5) is then exact. Decomposing the membrane height as  leads to the Langevin equation [cf. eqn (2) and (4)]
leads to the Langevin equation [cf. eqn (2) and (4)]| |  | (20) |
In eqn (20), ![[small xi, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e119.gif) q(t) is a complex Gaussian thermal noise satisfying the fluctuation–dissipation theorem, with two-time correlations proportional to δ(t − t′)kBϑ/(4µfqℓc2).18Eqn (20) can be solved exactly to yield62
q(t) is a complex Gaussian thermal noise satisfying the fluctuation–dissipation theorem, with two-time correlations proportional to δ(t − t′)kBϑ/(4µfqℓc2).18Eqn (20) can be solved exactly to yield62| |  | (21) |
which is the well-known result of the equipartition theorem as applied to planar membranes.34
The Langevin equation inside the dome.
For modes with q ∈ (q−0,q+0), there is no discernible difference between the impermeable and semipermeable dynamics. Thus, eqn (20) once again applies, and membrane fluctuations satisfy the equipartition result (21).
The Langevin equation outside the dome.
As discussed previously, we are currently unsure if our normal mode ansatz is appropriate outside the dome. If the ansatz is indeed valid, then we effectively see only an inertial mode. The corresponding Langevin equation describing membrane fluctuations is given by| |  | (22) |
where ω(r)ρ is the real part of the frequency and ![[small xi, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e119.gif) q is the same thermal noise as in eqn (20). There is no elastic restoring force in eqn (22), which is analogous to the equation of motion for a colloidal particle in a fluid. Just as a colloid diffuses freely at long times,62 the variance of membrane height undulations grows linearly in time according to eqn (22). Eventually, the membrane height will grow to a point where our assumption of linearity is no longer valid. At this point, membrane bending and surface tension forces will presumably prevent height fluctuations from continuing to grow—yet a description of such behavior is beyond the scope of the present work. Thus, irrespective of whether our ansatz is valid, membrane fluctuations are not expected to satisfy the equipartition result outside the dome where q ∉ (q−0,q+0).
q is the same thermal noise as in eqn (20). There is no elastic restoring force in eqn (22), which is analogous to the equation of motion for a colloidal particle in a fluid. Just as a colloid diffuses freely at long times,62 the variance of membrane height undulations grows linearly in time according to eqn (22). Eventually, the membrane height will grow to a point where our assumption of linearity is no longer valid. At this point, membrane bending and surface tension forces will presumably prevent height fluctuations from continuing to grow—yet a description of such behavior is beyond the scope of the present work. Thus, irrespective of whether our ansatz is valid, membrane fluctuations are not expected to satisfy the equipartition result outside the dome where q ∉ (q−0,q+0).
The standard contour analysis of a GUV.
Fig. 3(a) shows the canonical analysis of a GUV undergoing thermal fluctuations, the details of which can be found in ref. 29. In short, the equatorial cross-section of a GUV is imaged at frequent time intervals. For each snapshot, the radial deviation from a circle of radius rv is measured and decomposed into normal modes. As GUVs are large and weakly curved, it is often convenient to apply the planar membrane theory29—which is simpler than its spherical counterpart.31,58–60 The position x is defined as the distance along the circle, with h(x,t) being the radial perturbation, ℓc = 2πrv, and qx = 2πm/ℓc = m/rv for positive integers m. The one-dimensional (1D) fluctuation spectrum 〈|![[h with combining tilde]](https://www.rsc.org/images/entities/i_char_0068_0303.gif) qx|2〉 is calculated from experimental measurements, as shown in Fig. 3(a). To compare the 1D data with the theoretical 2D equipartition result of eqn (21), the latter is averaged over all qy as29
qx|2〉 is calculated from experimental measurements, as shown in Fig. 3(a). To compare the 1D data with the theoretical 2D equipartition result of eqn (21), the latter is averaged over all qy as29| |  | (23) |
The bending modulus kb and surface tension λc are chosen so as to minimize the difference between the measured fluctuation spectrum and prediction of eqn (23)—which, in Fig. 3(a), are shown as black circles and an orange line, respectively. Note that short-wavelength fluctuations cannot be visualized in experiments due to a lower bound ℓopt ≈ 250 nm on the optical resolution, which sets an upper limit qopt := 2π/ℓopt ≈ 0.025 nm−1 beyond which undulations cannot be observed.31 In practice, short-wavelength modes are excluded when fitting kb and λc. Techniques also exist to correct for the finite camera integration time29 and vertical focal projections,58 but are not discussed here.
 |
| | Fig. 3 Analysis of GUV contour fluctuations, modified due to membrane semipermeability. (a) Canonical protocol to calculate the bending modulus and surface tension of a fluctuating GUV. In a spherical vesicle of radius rv (left), the equatorial cross-section is imaged (center) at successive time intervals. Radial deviations from a circle are recorded as h(x,t), where x is the arclength along the unperturbed circle. The shape disturbances h(x,t) are decomposed into one-dimensional Fourier modes, whose thermally averaged amplitudes (right; adapted from ref. 55) are fit to eqn (23) by tuning the values of kb and λc (right; solid orange line).29 Theory and experiment deviate at large qx due to finite optical resolution in the latter. (b) Limitations of the equipartition result (21) due to solutes in the surrounding fluid. (left) Modes inside the dome (red lines) satisfy eqn (21); those outside the dome (green dots) do not. Membrane undulations cannot be visualized above qopt ≈ 0.025 nm−1 (gray shading) due to limitations in the optical resolution.31 (right) For a GUV with surface tension 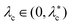 , modes are characterized in the qx–qy plane. The equipartition result (21) is limited to modes with amplitude q > q−0, and large-q modes cannot be visualized. The domains of integration in eqn (23), corresponding to two different values of qx, are shown as dashed vertical lines. The result of eqn (23) is invalid when qx < q−0, as in such cases, the integral contains modes outside the dome. (c) Plot of experimental membrane fluctuations, over a range of reported surface tensions λc, from prior studies: the studies by Faizi et al.59 (3.1 × 10−6 pN nm−1), Rautu et al.58 (1.4 × 10−5 pN nm−1), Pécréaux et al.29 (1.7 × 10−4 pN nm−1), Park et al.57 (7 × 10−4–3 × 10−3 pN nm−1), and Takatori and Sahu55 (4 × 10−3 pN nm−1). The y-axis is chosen to be independent of the vesicle radius and depends only on λc and kb [see eqn (23)]. At large qx, the data saturate due to optical resolution limitations. All data points below the dashed black line are outside the dome and should be excluded when determining membrane parameters. Fluctuation data from the high-tension vesicle in ref. 56 are not publicly available, and so cannot be plotted—though the first 88 modes should be excluded based on the reported value of λc = 0.025 pN nm−1 (ref. 25, Section 4). , modes are characterized in the qx–qy plane. The equipartition result (21) is limited to modes with amplitude q > q−0, and large-q modes cannot be visualized. The domains of integration in eqn (23), corresponding to two different values of qx, are shown as dashed vertical lines. The result of eqn (23) is invalid when qx < q−0, as in such cases, the integral contains modes outside the dome. (c) Plot of experimental membrane fluctuations, over a range of reported surface tensions λc, from prior studies: the studies by Faizi et al.59 (3.1 × 10−6 pN nm−1), Rautu et al.58 (1.4 × 10−5 pN nm−1), Pécréaux et al.29 (1.7 × 10−4 pN nm−1), Park et al.57 (7 × 10−4–3 × 10−3 pN nm−1), and Takatori and Sahu55 (4 × 10−3 pN nm−1). The y-axis is chosen to be independent of the vesicle radius and depends only on λc and kb [see eqn (23)]. At large qx, the data saturate due to optical resolution limitations. All data points below the dashed black line are outside the dome and should be excluded when determining membrane parameters. Fluctuation data from the high-tension vesicle in ref. 56 are not publicly available, and so cannot be plotted—though the first 88 modes should be excluded based on the reported value of λc = 0.025 pN nm−1 (ref. 25, Section 4). | |
The modified contour analysis of a GUV.
When solutes are present in the fluid surrounding the membrane, the 2D equipartition result (21) is only valid for modes q with magnitude |q| ∈ (q−0,q+0). We seek to determine how the experimental analysis of contour fluctuations is altered by such a restriction. To this end, we first note that qopt < q+0, as shown in Fig. 3(b). Short-wavelength undulations outside the dome thus cannot be observed in experiments and do not contribute to the determination of kb and λc. Long-wavelength fluctuations outside the dome, on the other hand, are captured experimentally. Fig. 3(b) characterizes modes in the qx–qy plane as being either outside the dome (|q| < q−0), inside the dome and visually accessible (q−0 < |q| < qopt), or visually inaccessible (|q| > qopt). Here, domains of the contour integral in eqn (23) for two values of qx are shown as dashed vertical lines. If qx < q−0, this domain contains modes for which the equipartition result (21) does not apply. The result of eqn (23), which relies on eqn (21), is accordingly valid only when qx > q−0—with the understanding that as qx increases, fewer modes are optically accessible.
To determine kb and λc from GUV contour fluctuations, it is useful to plot fluctuation amplitudes as shown in Fig. 3(c)—which contains experimental data from several prior investigations.29,55,57–59 Here, both the x-axis and y-axis are chosen to be independent of rv [cf.eqn (23)]. Next, experimental data inside and outside the dome need to be identified. To this end, we combine eqn (10) and (23) to obtain the fluctuation amplitudes of 1D modes qx = q−0 on the dome, for tensions  :
:
| |  | (24) |
Importantly,
kb is not known a priori, and so
eqn (24) can be approximated as
ℓc〈|
![[h with combining tilde]](https://www.rsc.org/images/entities/i_char_0068_0303.gif) qx
qx|
2〉 ≈
kBϑ/(8
μfqx2D)—valid when
kbqx ≪ 8
μfD—in a first analysis of the data.
Eqn (24) is plotted as the dashed black line in
Fig. 3(c); all of the data points below it are outside the dome and should be excluded when determining
kb and
λc.
Our aggregation and analysis of the experimental data in Fig. 3(c) are detailed in Section S4 of the SI.25 Most often, vesicle tensions λc ∼ 10−7–10−4 pN nm−1 are low29,58–60 and all data lie inside the dome. As it turns out, a vesicle at low tension is experimentally favorable because its undulations are large and hence easy to image. In many cases, the vesicle's osmotic environment is manipulated to elicit a low tension. Notable exceptions are the so-called active vesicles containing either self-propelled Janus particles or motile bacteria.55–57 For these systems, moderate vesicle tensions are required for active particles to not overcome the elastic membrane restoring force and form a tube. Given the fluctuation data for active vesicles in Fig. 1 of ref. 55 and Fig. 6(c) of ref. 57, between one and eight long-wavelength modes lie outside the dome (ref. 25, Section 4)—though the overall findings of these studies are unaffected. In contrast, for the high-tension (λc = 0.025 pN nm−1) active vesicle in Fig. 1(g) of ref. 56, the first 88 long-wavelength modes lie outside the dome. It is likely that upon excluding these modes, the calculated values of kb and λc would be affected. Unfortunately, the experimental fluctuation data for this vesicle are not provided, and we cannot investigate further. Nevertheless, we find that osmotic forces can indeed alter membrane fluctuations in biologically relevant scenarios.
Concluding remarks
In this work, we investigated the linearized dynamics of a planar lipid membrane, surrounded by a Newtonian fluid in which solutes are dissolved. Though the lipid bilayer is only weakly permeable to water, the presence of solutes can significantly alter its dynamics. More specifically, when ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D<
D<![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m and diffusion is slow relative to the impermeable membrane, undulations of the semipermeable system no longer decay at a frequency near
m and diffusion is slow relative to the impermeable membrane, undulations of the semipermeable system no longer decay at a frequency near ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m. In such cases, either (i) the ansatz that all quantities can be expressed in terms of planar normal modes as in eqn (2), (3), and (12) is no longer valid or (ii) the bilayer only decays at the fast inertial frequency ωρ ≫ ωm. When thermal perturbations are incorporated into the theory, the well-known equipartition result (21) is found to only be experimentally relevant when q∈ (q−0,qopt)—a range that narrows as λc is increased. Our results are relevant when membrane tensions are large, as is the case with active vesicles55–57 and a variety of biological scenarios.30,63–74
m. In such cases, either (i) the ansatz that all quantities can be expressed in terms of planar normal modes as in eqn (2), (3), and (12) is no longer valid or (ii) the bilayer only decays at the fast inertial frequency ωρ ≫ ωm. When thermal perturbations are incorporated into the theory, the well-known equipartition result (21) is found to only be experimentally relevant when q∈ (q−0,qopt)—a range that narrows as λc is increased. Our results are relevant when membrane tensions are large, as is the case with active vesicles55–57 and a variety of biological scenarios.30,63–74
The vanishing of the membrane mode at low q and large λc opens several lines of inquiry. We are particularly interested to see if membranes in spherical or cylindrical geometries respond in the same fashion; the former is needed for a careful analysis of GUV undulations,58–60 while the latter could be relevant to neuronal systems.30,63–66 In all scenarios where the membrane branch vanishes, the behavior of the coupled membrane–fluid–solute system can be interrogated with nonlinear simulations.48–54 Additional levels of complexity can be introduced by considering membranes that are not ideally selective,3 have a finite thickness,75–78 or are surrounded by charged solutes—with the latter requiring extensive theoretical developments.76–80
Conflicts of interest
There are no conflicts to declare.
Data availability
The code used to generate all data in the present study is publicly available at https://github.com/sahu-lab/osmosis-flat.
Supplementary information (SI): theoretical techniques used and methods of numerical solution.25 See DOI: https://doi.org/10.1039/d5sm01094b.
Acknowledgements
We express our most sincere gratitude to Prof. Kranthi Mandadapu for his perspective on lipid membrane behavior. We are also thankful to have discussed ref. 3 with Dr. Ahmad Alkadri and Kranthi, as well as refs. 57 and 61 with Dr. Kisung Lee, Dr. Myoenggon Park, and Prof. Steve Granick. It is a pleasure to thank Dr. Joël Tchoufag for many stimulating discussions, Profs. Brian Camley and Tine Curk for their thoughts on membrane fluctuations, and Prof. Sho Takatori for introducing us to the experimental analysis of GUV fluctuations. This work was partially supported by the Welch Foundation via grant no. F-2208-20240404.
References
- K. Olbrich, W. Rawicz, D. Needham and E. Evans, Biophys. J., 2000, 79, 321–327 CrossRef CAS PubMed.
- R. E. Wood, F. P. Wirth Jr and H. E. Morgan, Biochim. Biophys. Acta, 1968, 163, 171–178 Search PubMed.
- A. M. Alkadri and K. K. Mandadapu, Phys. Rev. E, 2025, 112, 034413 Search PubMed.
- M. Terasaki,
et al.
, Cell, 2013, 154, 285–296 CrossRef CAS PubMed.
- K. Tanaka and H. Fukudome, J. Elec. Microsc. Tech., 1991, 17, 15–23 CrossRef CAS PubMed.
- S. Watanabe,
et al.
, eLife, 2013, 2, e00723 CrossRef PubMed.
- J. Prost and R. Bruinsma, Europhys. Lett., 1996, 33, 321 CrossRef CAS.
- R. Granek, J. Phys. II France, 1997, 7, 1761–1788 CrossRef CAS.
- F. L. H. Brown, Biophys. J., 2003, 84, 842–853 CrossRef CAS PubMed.
- H.-Y. Chen, Phys. Rev. Lett., 2004, 92, 168101 CrossRef PubMed.
- N. S. Gov, Phys. Rev. Lett., 2004, 93, 268104 CrossRef CAS PubMed.
- L. C.-L. Lin and F. L. H. Brown, Phys. Rev. Lett., 2004, 93, 256001 CrossRef PubMed.
- D. Lacoste and A. W. C. Lau, Europhys. Lett., 2005, 70, 418 CrossRef CAS.
- M. A. Lomholt, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2006, 73, 061914 CrossRef PubMed.
- E. Ben-Isaac,
et al.
, Phys. Rev. Lett., 2011, 106, 238103 CrossRef PubMed.
- B. Loubet, U. Seifert and M. A. Lomholt, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2012, 85, 031913 CrossRef PubMed.
- R. Alert, J. Casademunt, J. Brugués and P. Sens, Biophys. J., 2015, 108, 1878–1886 CrossRef CAS PubMed.
- K. Sapp and L. Maibaum, Phys. Rev. E, 2016, 94, 052414–052423 CrossRef PubMed.
-
H. Turlier and T. Betz, Fluctuations in active membranes, in Physics of Biological Membranes, ed. P. Bassereau and P. Sens, Springer, Cham, 2018, pp. 581–619 Search PubMed.
- Some studies define kb/2 as the bending modulus.
- D. J. Steigmann, Arch. Ration. Mech. Anal., 1999, 150, 127–152 CrossRef.
-
A. Sahu, Irreversible Thermodynamics and Hydrodynamics of Biological Membranes, PhD thesis, University of California at Berkeley, 2022.
- R. S. Gracià, N. Bezlyepkina, R. L. Knorr, R. Lipowsky and R. Dimova, ASoft Matter, 2010, 6, 1472–1482 RSC.
- Y. A. Ayala, R. Omidvar, W. Römer and A. Rohrbach, Nat. Commun., 2023, 14, 65 CrossRef CAS PubMed.
- See the SI, where the governing equations are presented and dispersion relations are obtained. Methods of numerical solution are described, and all associated codes are present in the repository https://github.com/sahu-lab/osmosis-flat.
- A. Sahu, R. A. Sauer and K. K. Mandadapu, Phys. Rev. E, 2017, 96, 042409 CrossRef PubMed.
- A. Sahu, A. Glisman, J. Tchoufag and K. K. Mandadapu, Phys. Rev. E, 2020, 101, 052401 Search PubMed.
- P. Parkkila, M. Elderdfi, A. Bunker and T. Viitala, Langmuir, 2018, 34, 8081–8091 CrossRef CAS PubMed.
- J. Pécréaux, H.-G. Döbereiner, J. Prost, J.-F. Joanny and P. Bassereau, Eur. Phys. J. E, 2004, 13, 277–290 CrossRef PubMed.
- J. Dai, M. P. Sheetz, X. Wan and C. E. Morris, J. Neurosci., 1998, 18, 6681–6692 CrossRef CAS PubMed.
-
J. H. Ipsen, A. G. Hansen and T. Bhatia, Vesicle fluctuation analysis, in The Giant Vesicle Book, ed. R. Dimova and C. Marques, CRC Press, Boca Raton, 1st edn, 2020, pp. 333–345 Search PubMed.
- R. Dimova,
et al.
, J. Phys.: Condens. Matter, 2006, 18, S1151–S1176 CrossRef CAS PubMed.
- L. J. Gosting and M. S. Morris, J. Am. Chem. Soc., 1949, 71, 1998–2006 CrossRef CAS.
- F. Brochard and J. F. Lennon, J. Phys., 1975, 36, 1035–1047 CrossRef.
- B. A. Camley and F. L. H. Brown, Phys. Rev. Lett., 2010, 105, 148102 CrossRef PubMed.
- N. Oppenheimer and H. Diamant, Biophys. J., 2009, 96, 3041–3049 CrossRef CAS PubMed.
- H. A. Stone and H. Masoud, J. Fluid Mech., 2015, 781, 494–505 CrossRef CAS.
- B. A. Camley, M. G. Lerner, R. W. Pastor and F. L. H. Brown, J. Chem. Phys, 2015, 143, 243113 CrossRef PubMed.
- V. C. Suja, N. Oppenheimer and H. A. Stone, Phys. Rev. Fluids, 2025, 10, L041101 Search PubMed.
-
S. Chandrasekhar, Hydrodynamic and Hydromagnetic Stability, Dover, New York, 1981 Search PubMed.
- E. H. Starling, J. Physiol., 1896, 19, 312–326 Search PubMed.
- A. J. Staverman, Recl. Trav. Chim. Pays-Bas, 1951, 70, 344–352 CrossRef CAS.
- O. O. Kedem and A. Katchalsky, Biochim. Biophys. Acta, 1958, 27, 229–246 CrossRef CAS PubMed.
-
I. Prigogine, Introduction to Thermodynamics of Irreversible Processes, Interscience Publishers, 1961 Search PubMed.
-
S. R. de Groot and P. Mazur. Non-Equilibrium Thermodynamics, Dover, 1984 Search PubMed.
-
D. T. Limmer, Statistical Mechanics and Stochastic Thermodynamics: A Textbook on Modern Approaches in and out of Equilibrium, Oxford University Press, 2024 Search PubMed.
-
R. W. Zwanzig, Nonequilibrium Statistical Mechanics, Oxford University Press, New York, 2001 Search PubMed.
- V. Narsimhan, A. P. Spann and E. S. G. Shaqfeh, J. Fluid Mech., 2015, 777, 1–26 Search PubMed.
- A. Torres-Sánchez, D. Millán and M. Arroyo, J. Fluid Mech., 2019, 872, 218–271 CrossRef.
- D. A. Rower, M. Padidar and P. J. Atzberger, J. Comp. Phys., 2022, 455, 110994 CrossRef.
- P. D. Tran, T. A. Blanpied and P. J. Atzberger, Phys. Rev. E, 2022, 106, 044402 CrossRef CAS PubMed.
- A. Torres-Sánchez, M. K. Winter and G. Salbreux, PLOS Comput. Biol., 2022, 18, e1010762 Search PubMed.
- A. Sahu, J. Fluid Mech., 2025, 1020, A46 CrossRef.
-
D. Wörthmüller, G. Ferraro, P. Sens and M. Castellana, arXiv, 2025, preprint, arXiv:2506.17827 DOI:10.48550/arXiv.2506.17827.
- S. C. Takatori and A. Sahu, Phys. Rev. Lett., 2020, 124, 158102 Search PubMed.
- H. R. Vutukuri,
et al.
, Nature, 2020, 586, 52–56 CrossRef CAS PubMed.
- M. Park, K. Lee and S. Granick, Soft Matter, 2022, 18, 6419–6425 Search PubMed.
- S. A. Rautu,
et al.
, Soft Matter, 2017, 13, 3480–3483 Search PubMed.
- H. A. Faizi, C. J. Reeves, V. N. Georgiev, P. M. Vlahovska and R. Dimova, Soft Matter, 2020, 16, 8996–9001 RSC.
- H. A. Faizi, R. Granek and P. M. Vlahovska, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2413557121 CrossRef CAS PubMed.
- K. Lee, G. Chommanov, H.-S. Jang and S. Granick, J. Phys. Chem. A, 2020, 124, 4512–4516 CrossRef CAS PubMed.
- S. Chandrasekhar, Rev. Mod. Phys., 1943, 15, 1–89 CrossRef.
- J. Dai and M. P. Sheetz, Biophys. J., 1995, 68, 988–996 CrossRef CAS PubMed.
- A. Datar,
et al.
, Biophys. J., 2019, 117, 880–891 CrossRef CAS PubMed.
- P. A. Pullarkat, P. Dommersnes, P. Fernández, J.-F. Joanny and A. Ott, Phys. Rev. Lett., 2006, 96, 048104 CrossRef PubMed.
- J. Tchoufag, A. Sahu and K. K. Mandadapu, Phys. Rev. Lett., 2022, 128, 068101 CrossRef CAS PubMed.
- R. E. Goldstein, P. Nelson, T. Powers and U. Seifert, J. Phys. II, 1996, 6, 767–796 CrossRef CAS.
- T. R. Powers and R. E. Goldstein, Phys. Rev. Lett., 1997, 78, 2555–2558 CrossRef CAS.
- S. Boulant, C. Kural, J.-C. Zeeh, F. Ubelmann and T. Kirchhausen, Nat. Cell. Biol., 2011, 13, 1124–1131 CrossRef CAS PubMed.
- S. C. Al-Izzi, G. Rowlands, P. Sens and M. S. Turner, Phys. Rev. Lett., 2018, 120, 138102 CrossRef CAS PubMed.
- Z. Shi, Z. T. Graber, T. Baumgart, H. A. Stone and A. E. Cohen, Cell, 2018, 175, 1769–1779 CrossRef CAS PubMed.
-
S. C. Al-Izzi, M. S. Turner and P. Sens, arXiv, 2024, preprint, arXiv:2409.14835 DOI:10.48550/arXiv.2409.14835.
- M. Janssen, S. Liese, S. C. Al-Izzi and A. Carlson, Phys. Rev. E, 2024, 109, 044403 CrossRef CAS PubMed.
- A. Venkatesh, A. Bhargava and V. Narsimhan, J. Fluid Mech., 2025, 1003, A18 CrossRef.
- Z. G. Lipel, Y. A. D. Omar and D. Fraggedakis, Phys. Rev. Fluids, 2025, 10, 103101 CrossRef.
- Y. A. D. Omar, Z. G. Lipel and K. K. Mandadapu, Phys. Rev. E, 2024, 109, 054401 Search PubMed.
- Y. A. D. Omar, Z. G. Lipel and K. K. Mandadapu, Phys. Rev. E, 2025, 112, 024406 CrossRef CAS PubMed.
- Y. A. D. Omar, Z. G. Lipel and K. K. Mandadapu, Phys. Rev. E, 2025, 112, 024407 Search PubMed.
- K. D. Fong, H. K. Bergstrom, B. D. McCloskey and K. K. Mandadapu, AIChE J., 2020, 66, e17091 Search PubMed.
- Z. Yu, S. Zhao, M. J. Miksis and P. M. Vlahovska, Phys. Rev. E, 2025, 112, 054408 CrossRef CAS PubMed.
Footnote |
| † For conciseness, only the concentration field above the membrane is presented; see the SI (ref. 25, Sections 3.2 and 3.3) for all details. |
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article

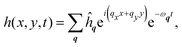
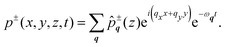

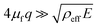 and the two solutions to eqn (4) are well-approximated by
and the two solutions to eqn (4) are well-approximated by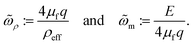
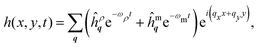
![[p with combining circumflex]](https://www.rsc.org/images/entities/i_char_0070_0302.gif) ±q = ∓½(ρmωq2 + E)ĥqe−q|z|. The magnitude of the pressure at z = 0 is determined using eqn (1), and the decay length 1/q is a consequence of the pressure field being harmonic: p±,jj = 0. In contrast, fluid velocities decay over the two length scales 1/q and 1/kν, where kν := (q2 − ω/νf)1/2 is a modified wavenumber that accounts for the inertia of the fluid (see Section S94 of ref. 40 and Section S2.1 of the SI25). As a consequence of the time scale separation ωρ ≫ ωm, 1/
±q = ∓½(ρmωq2 + E)ĥqe−q|z|. The magnitude of the pressure at z = 0 is determined using eqn (1), and the decay length 1/q is a consequence of the pressure field being harmonic: p±,jj = 0. In contrast, fluid velocities decay over the two length scales 1/q and 1/kν, where kν := (q2 − ω/νf)1/2 is a modified wavenumber that accounts for the inertia of the fluid (see Section S94 of ref. 40 and Section S2.1 of the SI25). As a consequence of the time scale separation ωρ ≫ ωm, 1/![[q with combining macron]](https://www.rsc.org/images/entities/i_char_0071_0304.gif) ≈1/kν on the membrane branch and 1/
≈1/kν on the membrane branch and 1/![[q with combining macron]](https://www.rsc.org/images/entities/i_char_0071_0304.gif) ≪ 1/kν on the inertial branch.
≪ 1/kν on the inertial branch.
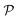 , ranging from 10−7 to 10−12 over wavenumbers q ∈ (10−4, 10−1) nm−1. We will show
, ranging from 10−7 to 10−12 over wavenumbers q ∈ (10−4, 10−1) nm−1. We will show  in the limit of an impermeable membrane; lipid bilayers are thus nearly impermeable.
in the limit of an impermeable membrane; lipid bilayers are thus nearly impermeable.
 we find v±z → h,t and recover the impermeable scenario.
we find v±z → h,t and recover the impermeable scenario.
![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D := q2D.
D := q2D.![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D plotted on top of the curves in Fig. 1; note that the diffusive and impermeable membrane frequencies can be comparable. Let us denote q±0 as the two wavenumbers at which the diffusive and membrane time scales are equal. From eqn (5) and (9), we find
D plotted on top of the curves in Fig. 1; note that the diffusive and impermeable membrane frequencies can be comparable. Let us denote q±0 as the two wavenumbers at which the diffusive and membrane time scales are equal. From eqn (5) and (9), we find
![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m <
m < ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D when q ∈ (q−0,q+0), and
D when q ∈ (q−0,q+0), and ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m >
m > ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D otherwise. Since the base surface tension λc ∼ 10−4–10−1 pN nm−1 is not a material parameter, but rather varies—and can be tuned—across systems, q±0 is plotted at different tensions λc in Fig. 2b to reveal a dome-like envelope. There exists a critical base tension
D otherwise. Since the base surface tension λc ∼ 10−4–10−1 pN nm−1 is not a material parameter, but rather varies—and can be tuned—across systems, q±0 is plotted at different tensions λc in Fig. 2b to reveal a dome-like envelope. There exists a critical base tension
 and λℓ, we have
and λℓ, we have ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m >
m > ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D over all wavenumbers. As we will see, the relative values of
D over all wavenumbers. As we will see, the relative values of ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m and
m and ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D—and thus whether a mode is “inside” (i.e. below) or “outside” (i.e. above) the dome in Fig. 2b—govern different regimes of membrane behavior.
D—and thus whether a mode is “inside” (i.e. below) or “outside” (i.e. above) the dome in Fig. 2b—govern different regimes of membrane behavior.

![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D (solid green line) on top of the impermeable frequencies in Fig. 1. There are two wavenumbers for which the impermeable membrane frequency ωimpm equals
D (solid green line) on top of the impermeable frequencies in Fig. 1. There are two wavenumbers for which the impermeable membrane frequency ωimpm equals ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D; approximating the former as
D; approximating the former as ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m yields the two crossover wavenumbers q−0 and q+0(10)—shown as vertical, dashed lines. (b) Plot of q±0 over a range of the base membrane tension λc, which varies across systems. The surface tension from (a) of λc = 2 × 10−3 pN nm−1 is shown as the horizontal dashed line. There exists a critical tension
m yields the two crossover wavenumbers q−0 and q+0(10)—shown as vertical, dashed lines. (b) Plot of q±0 over a range of the base membrane tension λc, which varies across systems. The surface tension from (a) of λc = 2 × 10−3 pN nm−1 is shown as the horizontal dashed line. There exists a critical tension 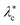 (11) above which
(11) above which ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m >
m > ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D for all wavenumbers. (c) Plot of the real parts of the semipermeable frequencies (dotted or dashed solid lines), overlaid on top of the impermeable frequencies (thick, transparent bands) for comparison. The inertial branch is essentially unchanged. The membrane branch only exists when ωm <
D for all wavenumbers. (c) Plot of the real parts of the semipermeable frequencies (dotted or dashed solid lines), overlaid on top of the impermeable frequencies (thick, transparent bands) for comparison. The inertial branch is essentially unchanged. The membrane branch only exists when ωm < ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D and is thus confined to the range q ∈ (q−0,q+0). For base membrane tensions
D and is thus confined to the range q ∈ (q−0,q+0). For base membrane tensions  , the membrane branch vanishes altogether.
, the membrane branch vanishes altogether.
 : since the membrane is nearly impermeable, semipermeable frequencies are close to their impermeable counterparts. In what follows, we investigate how the dynamics of the impermeable branches are altered by the presence of solutes in the surrounding medium.
: since the membrane is nearly impermeable, semipermeable frequencies are close to their impermeable counterparts. In what follows, we investigate how the dynamics of the impermeable branches are altered by the presence of solutes in the surrounding medium.

![[small eta, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e110.gif) z. As these standing waves do not decay as z → ∞, we seek the lowest-order concentration correction due to the semipermeability of the membrane.†
z. As these standing waves do not decay as z → ∞, we seek the lowest-order concentration correction due to the semipermeability of the membrane.†

 is a dimensionless parameter that arises when eqn (7) and (8) are combined. The solution of eqn (14) closest to ωimpρ, to lowest order in the product
is a dimensionless parameter that arises when eqn (7) and (8) are combined. The solution of eqn (14) closest to ωimpρ, to lowest order in the product  of small parameters, is given by
of small parameters, is given by

![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D. The solute diffusion equation once again simplifies to eqn (13), where now
D. The solute diffusion equation once again simplifies to eqn (13), where now ![[small eta, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e110.gif) involves ωimpm instead of ωimpρ. With this change, and following a similar analysis to that described in the inertial scenario, we once again arrive at eqn (14). The membrane frequency is then found to be given by
involves ωimpm instead of ωimpρ. With this change, and following a similar analysis to that described in the inertial scenario, we once again arrive at eqn (14). The membrane frequency is then found to be given by

 (11), the membrane branch vanishes entirely.
(11), the membrane branch vanishes entirely.
![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D and q∈ (q−0,q+0), the analysis of the coupled system is straightforward in the limit of small
D and q∈ (q−0,q+0), the analysis of the coupled system is straightforward in the limit of small  . The diffusion equation, assuming ω = ωimpm at lowest order, simplifies to d2ĉ±q/dz2 = (q2 − ωimpm/D)ĉ±q. We thus find the exponentially decaying concentration solutions
. The diffusion equation, assuming ω = ωimpm at lowest order, simplifies to d2ĉ±q/dz2 = (q2 − ωimpm/D)ĉ±q. We thus find the exponentially decaying concentration solutions
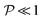 , we expect the membrane branch to be indistinguishable from its impermeable counterpart—as confirmed in Fig. 2(c).
, we expect the membrane branch to be indistinguishable from its impermeable counterpart—as confirmed in Fig. 2(c).
![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m presented in eqn (5) is then exact. Decomposing the membrane height as
m presented in eqn (5) is then exact. Decomposing the membrane height as  leads to the Langevin equation [cf. eqn (2) and (4)]
leads to the Langevin equation [cf. eqn (2) and (4)]
![[small xi, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e119.gif) q(t) is a complex Gaussian thermal noise satisfying the fluctuation–dissipation theorem, with two-time correlations proportional to δ(t − t′)kBϑ/(4µfqℓc2).18Eqn (20) can be solved exactly to yield62
q(t) is a complex Gaussian thermal noise satisfying the fluctuation–dissipation theorem, with two-time correlations proportional to δ(t − t′)kBϑ/(4µfqℓc2).18Eqn (20) can be solved exactly to yield62

![[small xi, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e119.gif) q is the same thermal noise as in eqn (20). There is no elastic restoring force in eqn (22), which is analogous to the equation of motion for a colloidal particle in a fluid. Just as a colloid diffuses freely at long times,62 the variance of membrane height undulations grows linearly in time according to eqn (22). Eventually, the membrane height will grow to a point where our assumption of linearity is no longer valid. At this point, membrane bending and surface tension forces will presumably prevent height fluctuations from continuing to grow—yet a description of such behavior is beyond the scope of the present work. Thus, irrespective of whether our ansatz is valid, membrane fluctuations are not expected to satisfy the equipartition result outside the dome where q ∉ (q−0,q+0).
q is the same thermal noise as in eqn (20). There is no elastic restoring force in eqn (22), which is analogous to the equation of motion for a colloidal particle in a fluid. Just as a colloid diffuses freely at long times,62 the variance of membrane height undulations grows linearly in time according to eqn (22). Eventually, the membrane height will grow to a point where our assumption of linearity is no longer valid. At this point, membrane bending and surface tension forces will presumably prevent height fluctuations from continuing to grow—yet a description of such behavior is beyond the scope of the present work. Thus, irrespective of whether our ansatz is valid, membrane fluctuations are not expected to satisfy the equipartition result outside the dome where q ∉ (q−0,q+0).
![[h with combining tilde]](https://www.rsc.org/images/entities/i_char_0068_0303.gif) qx|2〉 is calculated from experimental measurements, as shown in Fig. 3(a). To compare the 1D data with the theoretical 2D equipartition result of eqn (21), the latter is averaged over all qy as29
qx|2〉 is calculated from experimental measurements, as shown in Fig. 3(a). To compare the 1D data with the theoretical 2D equipartition result of eqn (21), the latter is averaged over all qy as29

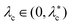 , modes are characterized in the qx–qy plane. The equipartition result (21) is limited to modes with amplitude q > q−0, and large-q modes cannot be visualized. The domains of integration in eqn (23), corresponding to two different values of qx, are shown as dashed vertical lines. The result of eqn (23) is invalid when qx < q−0, as in such cases, the integral contains modes outside the dome. (c) Plot of experimental membrane fluctuations, over a range of reported surface tensions λc, from prior studies: the studies by Faizi et al.59 (3.1 × 10−6 pN nm−1), Rautu et al.58 (1.4 × 10−5 pN nm−1), Pécréaux et al.29 (1.7 × 10−4 pN nm−1), Park et al.57 (7 × 10−4–3 × 10−3 pN nm−1), and Takatori and Sahu55 (4 × 10−3 pN nm−1). The y-axis is chosen to be independent of the vesicle radius and depends only on λc and kb [see eqn (23)]. At large qx, the data saturate due to optical resolution limitations. All data points below the dashed black line are outside the dome and should be excluded when determining membrane parameters. Fluctuation data from the high-tension vesicle in ref. 56 are not publicly available, and so cannot be plotted—though the first 88 modes should be excluded based on the reported value of λc = 0.025 pN nm−1 (ref. 25, Section 4).
, modes are characterized in the qx–qy plane. The equipartition result (21) is limited to modes with amplitude q > q−0, and large-q modes cannot be visualized. The domains of integration in eqn (23), corresponding to two different values of qx, are shown as dashed vertical lines. The result of eqn (23) is invalid when qx < q−0, as in such cases, the integral contains modes outside the dome. (c) Plot of experimental membrane fluctuations, over a range of reported surface tensions λc, from prior studies: the studies by Faizi et al.59 (3.1 × 10−6 pN nm−1), Rautu et al.58 (1.4 × 10−5 pN nm−1), Pécréaux et al.29 (1.7 × 10−4 pN nm−1), Park et al.57 (7 × 10−4–3 × 10−3 pN nm−1), and Takatori and Sahu55 (4 × 10−3 pN nm−1). The y-axis is chosen to be independent of the vesicle radius and depends only on λc and kb [see eqn (23)]. At large qx, the data saturate due to optical resolution limitations. All data points below the dashed black line are outside the dome and should be excluded when determining membrane parameters. Fluctuation data from the high-tension vesicle in ref. 56 are not publicly available, and so cannot be plotted—though the first 88 modes should be excluded based on the reported value of λc = 0.025 pN nm−1 (ref. 25, Section 4). :
:
![[h with combining tilde]](https://www.rsc.org/images/entities/i_char_0068_0303.gif) qx|2〉 ≈ kBϑ/(8μfqx2D)—valid when kbqx ≪ 8μfD—in a first analysis of the data. Eqn (24) is plotted as the dashed black line in Fig. 3(c); all of the data points below it are outside the dome and should be excluded when determining kb and λc.
qx|2〉 ≈ kBϑ/(8μfqx2D)—valid when kbqx ≪ 8μfD—in a first analysis of the data. Eqn (24) is plotted as the dashed black line in Fig. 3(c); all of the data points below it are outside the dome and should be excluded when determining kb and λc.
![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) D<
D<![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m and diffusion is slow relative to the impermeable membrane, undulations of the semipermeable system no longer decay at a frequency near
m and diffusion is slow relative to the impermeable membrane, undulations of the semipermeable system no longer decay at a frequency near ![[small omega, Greek, tilde]](https://www.rsc.org/images/entities/i_char_e10f.gif) m. In such cases, either (i) the ansatz that all quantities can be expressed in terms of planar normal modes as in eqn (2), (3), and (12) is no longer valid or (ii) the bilayer only decays at the fast inertial frequency ωρ ≫ ωm. When thermal perturbations are incorporated into the theory, the well-known equipartition result (21) is found to only be experimentally relevant when q∈ (q−0,qopt)—a range that narrows as λc is increased. Our results are relevant when membrane tensions are large, as is the case with active vesicles55–57 and a variety of biological scenarios.30,63–74
m. In such cases, either (i) the ansatz that all quantities can be expressed in terms of planar normal modes as in eqn (2), (3), and (12) is no longer valid or (ii) the bilayer only decays at the fast inertial frequency ωρ ≫ ωm. When thermal perturbations are incorporated into the theory, the well-known equipartition result (21) is found to only be experimentally relevant when q∈ (q−0,qopt)—a range that narrows as λc is increased. Our results are relevant when membrane tensions are large, as is the case with active vesicles55–57 and a variety of biological scenarios.30,63–74
