 Open Access Article
Open Access ArticleStudy of reduced states of Ce1−yFeyO2 and Ce1−yUyO2 for the thermochemical water splitting to hydrogen
Hicham
Idriss

Department of Chemistry, University College London, London WC1E 6BT, UK
First published on 14th April 2026
Abstract
Solar thermochemical water splitting (TCWS) is a promising approach for sustainable hydrogen production using concentrated solar energy. Cerium oxide (CeO2) is one of the most studied redox materials for this process due to its fast oxidation kinetics and structural stability at high temperature. However, its practical implementation remains limited by the high reduction energy required to reach significant non-stoichiometry, which restricts hydrogen yields. In this review, key fundamental aspects of ceria reduction and defect formation are discussed, with a focus on strategies to enhance reducibility through partial substitution of Ce cations by transition metals and actinides. In particular, substitution with Fe or U promotes the formation of oxygen vacancies (for different reasons, as discussed) and stabilizes reduced states, thereby increasing the extent of reduction and improving hydrogen production. Experimental and theoretical studies on Ce1−yFeyO2−δ and Ce1−yUyO2±δ are reviewed, including insights from temperature-programmed reduction, core- and valence-level X-ray photoelectron spectroscopy, and DFT+U calculations. Test reactions show, in line with spectroscopic results, that small fractions of Fe3+ or U4+ gave highest Ce3+ and TCWS reaction yields, while increasing the dopant fraction decreases the reaction yield. The correlations between reduction behavior, oxygen vacancy formation, charge-transfer phenomena, and hydrogen yields are discussed, with emphasis on the need for fundamental understanding of reduction energetics as a key factor for advancing solar thermochemical hydrogen generation.
1. Introduction
The Thermo-Chemical Water Splitting (TCWS) reaction is a solar driven H2 production from water using a redox type process of suitable materials. This short review's focus is on reducible oxides as the material of choice. The reaction has received considerable attention these last few decades1–3 and the process is a two-step reaction. The first one is endothermic, in which an oxide material in binary or mixed phases is reduced at a high temperature in an inert environment releasing a fraction of its oxygen atoms as O2. Temperatures as high as 1500 °C are typically needed to ensure a reduction suitable for studies and with a potential for application (eqn (1)). The second step is exothermic, where the reduced oxide(s) is exposed to H2O vapor, typically conducted a few hundred degrees lower in temperature. In this second step the reduced oxide is oxidized back and H2 is released (eqn (2)). Therefore, H2 and O2 are produced separately. The following two equations present these two steps.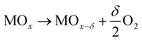 | (1) |
| MOx−δ + δH2O → MOx + δH2 | (2) |
This reaction performs similarly for CO2 to CO reduction. When the energy input is provided from the sun, it uses a large fraction of the solar spectrum making it among of the most efficient4,5 known systems for H2 generation from water with theoretical efficiencies >40%. It is still in the research phase largely because of the cost associated to the solar concentrators needed for the reduction.
The dynamics of the reduction and oxidation of metal oxides affect their properties and these in turn, dictates their performance in many applications, and that includes other processes beside the TCWS or CO2 reduction such as in redox-driven reactions in catalysis6 and gas sensors.7 Cerium oxide (CeO2/CeO2−x, x < 0.5) stands out as one of the most active and stable reducible metal oxides8 and is therefore prone to many fundamental studies largely because of its relatively simple structure (fluorite), simple surface termination dominated by the (111) face, and localized charges upon the creation of oxygen defects; when compared to many other solar materials.
It is worth making a distinction between the TCWS cycle and a hydrocarbon-based catalytic cycle as it is in practice at present. In catalysis, the reduction of an oxide, is conducted by using a chemical compound (providing the chemical energy), such as H2 (ref. 9) or CO.10 H2 and CO are largely generated by steam methane reforming and therefore contribute to CO2 emission, that is why solar thermal-driven alternatives are desired when looking beyond a hydrocarbon-based energy system. The main challenge for this alternative process is the large input of energy needed for the reduction step. To this end, considerable amount of work addressing the reduction of CeO2 in order to understand its steps at the fundamental and applied levels is pursued. Many computational studies have indicated that the energy needed to create an oxygen vacancy in the bulk of CeO2 is about 3 eV,11–13 while the energy required for atomic oxygen diffusion14 is less ≈1 eV. Despite the relatively fast reaction kinetics for the redox cycle, which is highly desired for the two steps described by eqn (1) and (2), the high energy cost makes the process unsustainable. At present, estimates of the levelized cost of H2 [LCOH: the cost of making H2 before making profit after a number of years, typically 20 years] produced by the TCWS method is close to 10 USD per kg, which is about five times higher than the present cost from fossil fuels.15 Because the TCWS reaction solely relies on the O-defect density, which is, per mass, low in CeO2 many approaches have been pursued to mix CeO2 with other metal cations to decrease the reduction energy and consequently increase the density of the reduced state.16
Among the few methods used the following two are of focus of this review.
(A) Charge transfer: a fraction of Ce4+ cations is substituted with metal cations that can donate electrons and themselves be oxidized. For example, the substitution of Ce4+ with U4+ was found to enhance the reduction of CeO2, particularly at low levels, ref. 12 and 13. Upon the removal of an oxygen atom (two electrons are left in the lattice), in a pure CeO2 two Ce4+ cations are reduced by these two electrons to Ce3+ cations. However, in the presence of U4+ three Ce3+ cations are formed (instead of two), and one U4+ cation is oxidized to a U5+ cation, as per the following equation.
| Ce4+(4f0) + U4+(5f2) → Ce3+(4f1) + U5+(5f1) | (3) |
For a charge-transfer mechanism to occur between two cations in a mixed oxide, several conditions should ideally be satisfied:
(i) The substituting cation should have an ionic radius close to that of the host cation,
(ii) It should possess the same (initial) oxidation state, and
(iii) The corresponding oxide should have a similar crystal structure to the host material, which increases the likelihood of forming homogeneous solid solutions.17
Yet, the optimal fractional substitution for the reduction of Ce cations is not straight forward to know because statistical entropy (distribution of U4+ cations around Ce4+ cations) affect the electron transfer process, and in addition phase segregation may still occur depending on the environment (oxidizing vs. reducing). The redox properties of such a system have been experimentally studied in the past.18–21 Experimental evidence of charge transfer was found, and the optimal charge transfer between Ce and U cations occurred at low % of U. In these studies, the mixed oxide Ce1−yUyO2±δ was reduced by H2 at 973 K to prevent phase segregation or by Ar+ sputtering for the core level spectroscopy measurements to monitor the charge transfer. For hydrogen production from water, experimental results showed, in line with spectroscopy, that low levels of U substitutions were better than higher levels, with an experimental optimal of about 10%. Moreover, spectroscopic studies of the Ce and U oxidation states of an epitaxy of the mixed oxide with (111) surface termination have largely confirmed the polycrystalline results.22
(B) Charge compensation (alio-valency): this is generally viewed as due to lattice distortion because of the difference in the cations' size and charge. The substitution of Ce4+ with these metal cations, of a lower oxidation state, creates vacancies. These vacancies are not associated to excess electrons and therefore there is no increase/decrease in electron charge. A large number of elements were studied for this, and most showed that indeed Ce4+ reduction to Ce3+ was enhanced. The addition of cobalt (Co) enhanced CeO2 reduction during the photoreduction23 of CO2 to CH4 and COS hydrolysis24 reactions. Praseodymium (Pr), where Pr3+ increases the creation of oxygen vacancies and Pr4+ increases the oxygen storage capacity.25 Manganese (Mn), enhanced oxygen mobility via vacancy formation.26 Computationally, a large body of work has also been conducted to study the creation of oxygen vacancies upon alio-valent cation substitution, Table 1. Among them: Mn (DFT+U and HSE06),27 Cu (DFT+U),28 Ni (DFT+U and HSE06)29 and (DFT+U),30 and Fe (DFT+U). These results indicated that the oxygen vacancy formation energy is lower when compared to ceria alone.31,32
Among the aliovalent systems, solid solutions of CeO2 containing Fe3+ have been studied in some details. In one of these works it was found that substituting a fraction of Ce4+ by Fe3+ cations in CeO2 led to a decrease of the activation energy for the selective catalytic reduction reaction of NO by half.39 This was attributed to the formation of a distorted Fe–O–Ce structure. DFT calculation and titration via pyridine-adsorption indicated that this Fe–O–Ce structure has increased the number of Lewis acid sites as well as the charge density around Ce cations when compared to those of pure CeO2. The authors also found that low Fe fraction (x < 0.3) kept the fluorite structure (solid solution) via a vacancy compensation mechanism, leading to an increase in oxygen vacancies which was linked to increase of the catalytic performance. Other work has shown the presence of a considerable fraction of Ce3+ cations by analyzing the XPS Ce 3d lines (at ca. 885 eV and 904 eV (V′ and U′ lines)) upon the introduction of Fe3+ cations into the lattice of Ce0.8Fe0.2O2−δ40,41 (δ: deviation from stoichiometry). Also, other have shown that Fe substitution of less than 30% increased the number of oxygen vacancies42 and improved CO conversion to CO2 the latter is attributed to the formation of more mobile oxygen atoms in the redox cycle.43 In another study, Fe 5 at% was tested at 1823 K and found to be more active (a higher production rate per unit weight) and faster (a higher rate of release of hydrogen) than CeO2 alone for the same reaction. Phase segregation, however, occurred due to the very high thermal reduction, 1823 K, temperature.44
The performance of pristine and cation substituted CeO2 in TCWS has been systematically studied, revealing that these metal cation can lower reduction temperatures, increase δ, and improve H2 yield per cycle. However, trade-offs with stability and phase segregation remain critical (Table 2).
| Material | Conditions (T, atm) | δ or O2 released | H2 yield per cycle | References |
|---|---|---|---|---|
| a Reduced by Ar ions sputtering. | ||||
| CeO2 (bulk) | 1773 K | δ < 0.02 | ∼10 mL H2 per g | 45–48 |
| Ce0.9Zr0.1O2 | 1673 K | δ ∼30% vs. CeO2 | ∼10 mL H2 per g | 49 |
| Ce0.9Fe0.1O2−δ | 1673–1823 K | Up to 50 µmol O2 per g | ∼20–25 mL H2 per g | 50 |
| Ce0.9U0.1O2±δ | 973–1200 Ka | Ce3+ fractiona >50% | ∼18–22 mL H2 per g | 20 |
| Ce0.8Pr0.2O2−δ | 1773 K | ∼15 mL O2 per g−1 | ∼4 mL H2 per g | 51 and 52 |
| Ce0.8Mn0.2O2−δ | 1773 K | Fast vacancy migration | ∼4 mL H2 per g | 53 and 54 |
The review focuses on two systems for which complementary information has been obtained using a combination of techniques, including spectroscopy, diffraction, microscopy, and thermochemical performance measurements. This two systems are composed of Fe and U cations used (separately) as substituents to enhance the reduction of CeO2. A particular attention is given on phase segregation studied by XRD, charge transfer studied by core- and valence-level spectroscopy, and reduction temperature by TPR, all on polycrystalline samples. Also, core level spectroscopy results of single crystals of the mixed Ce1−yUyO2 grown by epitaxy complemented by DFT+U computations are presented.
2. X-ray diffraction (Ce1−yFeyO2 and Ce1−yUyO2)
Fig. 1 presents XRD patterns of CeO2, and Ce0.95Fe0.05O2 that were heated at the indicated temperatures. | ||
| Fig. 1 (A) XRD patterns of CeO2 together with the extracted crystallite size and cubic parameter from the (111) lines; inset: a zoom on the (111) diffraction line to highlight the shift in angle and narrowing of the Full Width Half Maximum (FWHM) with increasing temperature in ambient environment. (B) Contains similar results for Ce0.95Fe0.05O2. From ref. 50, permission License number 6133081449846. | ||
The diffraction pattern of CeO2 shows a nano size dimension (L ≈ about 14 nm) for the “as prepared” oxide, with a lattice parameter (a) = 0.543 nm (Fig. 1A). Crystalline CeO2 gives rise to strong lines at 2θ = 28.5°, 33.0°, 47.5°, 56.4° for the (111), (200), (220) and (311) respectively, whose positions and Full Width Half Maximum (FWHM) being sensitive to its crystallite dimension and degree of crystallinity. No change in these parameters is seen up to ca. 900 °C (1173 K). At this temperature L considerably increased to 39 nm (due to sintering) together with an increase of a (0.545 nm) (due to bulk reduction). At 1100 °C, L doubled to 80.2 nm and a further increase of a (0.546 nm) is seen. Hematite Fe2O3 gives lines at 2θ = 33.3°, 35.7° and 62.3° for the (104), (110) and (214) lines, respectively. Because Fe3+ (0.6 Å) ions are much smaller than Ce4+ ions (ca. 1 Å in octahedral coordination) their incorporation into the lattice can be monitored mostly by a positive shift of 2θ which is associated with a broadening of the lines due to the formation of smaller crystallites.55 Therefore, both the shift and the absence of Fe2O3 lines are often taken as a strong indication of the presence of a solid solution. Depending on the preparation method this is found to be up to about 0.3–0.4 atomic ratio Fe3+/Ce4+ cations.56 The addition of Fe to CeO2 appears to be substitutional since no other diffraction lines attributed to Fe2O3 were seen up to 900 °C or so. Segregation occurred at 1100 °C where diffraction lines corresponding to Fe2O3 (110) and (003) are observed. Table 3 compares the parameters of three oxides at 500 °C and 1100 °C.
| Oxide | 2θ CeO2 (111) at 773 K | Crystallite size at 773 K (nm) | Cubic lattice parameter at 773 K (nm) | Crystallite size at 1373 K (nm) | Cubic lattice parameter at 1373 K (nm) |
|---|---|---|---|---|---|
| CeO2 | 28.46° | 14 | 0.543 | 80.2 | 0.545 |
| Ce0.95Fe0.05O2 | 28.68° | 9.5 | 0.540 | 73.6 | 0.542 |
| Ce0.75Fe0.25O2 | 28.84° | 6.5 | 0.540 | 61.3 | 0.544 |
Fig. 2 presents XRD of as prepared Ce0.5U0.5O2 and after it has been heated, in situ under N2 atmosphere, at the indicated temperatures. XRD patterns related to the fluorite structure are identified with prominent (111), (200), (220) and (311) peaks at 2θ = 28.54°, 33.11°, 47.58°, and 56.39°, respectively. The crystallite dimension calculated from the (111) diffraction line is found close to 12 nm for the fresh oxide. Upon heating to 300 °C (573 K) other diffraction patterns at 2θ = 21.38°, 26.16°, 33.95°, 43.58°, and 51.68° are seen. The patterns are attributed to other uranium oxide phases: orthorhombic α U3O8 or hexagonal α U2O5, or orthorhombic U2O5.57,58 Previous work has indicated that in the process of oxidation of UO2 to UO3, U3O8 is formed59 as the most stable phase; however, metastable U2O5 is also formed that eventually transform to α U3O8. The small crystallite structures and the broadening of the lines did not allow for further analysis. It is clear however, that some U cations in the solid solution segregate forming a more stable higher oxide that is attributed to either α U3O8 (or U2O5). Because the process occurs under N2 environment, it is expected that CeO2 left in the fluorite structure is reduced to CeO2−δ; this point will be reviewed in more details below.
 | ||
| Fig. 2 In situ XRD of Ce0.5U0.5O2 that was heated at the indicated temperatures. The numbers at the right hand side of the figure are those of crystallites size based on the (111) diffraction line of the fluorite structure at 2θ = 28.54° (d = 3.12 Å). The numbers (in red) on top of the dashed vertical lines in the figure are the diffraction pattern of α U3O8. From ref. 20, permission license number 6133100874303. | ||
3. Transmission electron microscopy (TEM); Ce1−yFeyO2 and Ce1−yUyO2
Fig. 3 presents the TEM, Selected Area Electron Diffraction pattern (SAED) and Energy Dispersive X-ray patterns (EDX) of Ce0.95Fe0.05O2−δ, and Ce0.75Fe0.25O2−δ that were calcined at 773 K. Like the data of XRD, both presented the fluorite structure of CeO2; no Fe2O3 phase is seen. The d spacing shows a slight decrease in the case of Ce0.75Fe0.25O2−δ when compared to Ce0.95Fe0.05O2−δ. Therefore, both microscopic and macroscopic diffraction methods indicate that the crystallite size of CeO2 decreases with Fe substitution. From TEM the average crystallite size of Ce0.95Fe0.05O2−δ is 8–10 nm while that of Ce0.75Fe0.25O2−δ is 5–7 nm. The decrease of the crystallite size might be due to a decrease of the surface energy (more stable) when compared to that of CeO2. EDX shows the presence of Fe in both mixed oxides with an intensity qualitatively tracking the expected concentration. | ||
| Fig. 3 Transmission Electron Microscopy (TEM), Selected Area Electron diffraction pattern (SAED) and Energy Dispersive X-ray patterns (EDX) of Ce0.95Fe0.05O2−δ, and Ce0.75Fe0.25O2−δ that were calcined at 773 K. The values in the tables in the middle are extracted from the SAED in the inset of the TEM images and ring numbers starts with the center of the concentric circles (the bright spot of each inset). From ref. 50, permission license number 6133081449846. | ||
A series of polycrystalline Ce1−xUxO2±δ was synthesized and studied for their thermochemical water splitting to hydrogen. XRD, as shown above for the as-prepared mixed oxides, present a fluorite structure, in line with numerous other studies in which Ce and U cations have high miscibility60–62 for the as prepared oxide. Fig. 4A (HR-TEM) indicates that particles are crystalline with a size of about 10 nm. Like in the case of Fe, data indicate a high miscibility of U cations in CeO2. For example, while Fig. 4B (STEM) shows no distinction between the atoms while the EELS spectra of the same area, Fig. 4C, shows the presence of both U and Ce; the Ce 3d → 4f and U 4d → 5f transitions. The lattice fringes obtained from the diffraction pattern (Fig. 4D) related to Fig. 4B are nearly identical to those of CeO2.
 | ||
| Fig. 4 (A) High-resolution transmission electron microscopy (HRTEM) of Ce0.75U0.25O2 polycrystalline mixed oxide. (B) Scanning transmission electron microscopy (STEM) of a selected area from (A). (C) Electron energy loss spectroscopy (EELS) spectrum of the area shown in (B). (D) Selected area electron diffraction (SAED) of (B). From ref. 22 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
4. Temperature programmed reduction (TPR) (Ce1−yFeyO2 and Ce1−yUyO2)
Fig. 5 shows TPR results for a series of Ce1−yUyO2 oxides. The addition of U cations to CeO2 resulted in lower reduction temperature and higher hydrogen consumption overall. Because the size of U4+ is very close to that Ce4+ the increase in reduction is not due to lattice compensation but most likely due to the presence of U cations in higher oxidation state than +4 in addition to possible charge transfer. Table 4 shows a summary of the H2 consumption of the series, together with BET surface areas and the calculated fraction of oxygen removed. The addition of U cations further increases the reduction of CeO2 (from 9% for CeO2 alone to 26% of oxygen atoms removed in the case of Ce0.25U0.75O2). There is also a decrease of the “overall” reduction temperatures when compared to CeO2 or UO2 alone. The case of the 0.5 fraction of each cation is intriguing as it presents one single large reduction peak; this however was not further investigated. | ||
| Fig. 5 TPR of a series of Ce1−yUyO2: CeO2, Ce0.75U0.25O2, Ce0.5U0.5O2, Ce0.25U0.75O2 and UO2; ramping rate (10 °C min−1). The weight used in each run is indicated on the right-hand side of the curves. Adapted from ref. 63, permission license number 6133100874303. | ||
| Oxide | Amount of H2 consumed mL goxide−1 | % of removed O atoms | BET surface area m2 goxide−1 |
|---|---|---|---|
| CeO2 | 24.3 | 9 | 71 |
| Ce0.75U0.25O2 | 21.8 | 10 | 33 |
| Ce0.5U0.5O2 | 32.8 | 16 | 34 |
| Ce0.25U0.75O2 | 38.9 | 21 | 30 |
| UO2+x | 27.9 | Excess | 17 |
Next, TPR of Ce1−yFeyO2 together with that of Fe2O3 is presented, Fig. 6. The four profiles are plotted as monitored without multiplication and with offset for clarity. The weight of the oxide used is given on the left side of each profile and the total amount of consumed hydrogen is given on the right side. The stoichiometry of the oxides, based on their formulae units, is given next to each line. The table in the inset presents the stoichiometry based on hydrogen consumption (loss of oxygen). Fe2O3 is expected to be completely reduced to metallic iron. The needed amount of hydrogen to reduce it (ca. 420 mL g−1) is about 80% of that observed (Fe2O3 + 3H2 → 2Fe + 3H2O); it may indicate experimental errors of 20% or so but can also, partly be due to excess oxygen (of surface hydroxyls from adsorbed water).
 | ||
| Fig. 6 TPR of CeO2, Ce0.95Fe0.05O2−δ, Ce0.75Fe0.25O2−δ, and Fe2O3. All oxides were pre-calcined at 500 °C. The computed values of 2 − δ are given beside each line in the formulae units. The inset rectangle has the computed formulae units based on the oxygen loss during TPR. The amount of hydrogen used to fully reduce Fe2O3 to Fe may give an indication on the errors, about 20%. The highlighted regions I and II are those of surface and bulk reduction of CeO2 while region III is that of bulk reduction of Fe2O3. The first peak in the TPR of Fe2O3 might be due to surface reduction. From ref. 50, permission license number 6133081449846. | ||
The results of the mixed oxides can be qualitatively explained based on those obtained from the pure ones. The two peaks of CeO2 labeled I and II may represent surface and bulk reductions. These are qualitatively similar to those reported in Fig. 5, although it is from a different sample batch. Based on XRD (and TEM) results the addition of Fe cations to CeO2 is substitutional and resulted in decreasing the crystallite size. The decrease in crystallite size would in turn results in increasing the surface to bulk ratio which explains the increase in peak I compared to peak II ratio in the CeFe oxides. Peak III is that of pure Fe2O3; because it is also present in the case of Ce0.75Fe0.25O2−δ some Fe cations may have segregated out of the fluorite structure. The increase in peak II of Ce1−yUyO2 oxides compared to CeO2 alone might be due to the ease of bulk reduction due to the presence of Fe cations inside the fluorite structure.
The increased reduction of CeO2 using Fe was studied by Temperature Programmed Reduction (TPR) in other works. Pure CeO2 was seen to be reduced in two temperature domains at about 773 K and 1073 K.63 The first has been attributed to surface reduction while the second to bulk reduction. The reduction is mild (leads to the removal of a small fraction of oxygen anions typically 10–20%) and the formation of Ce3+ cations within the investigated temperatures in TPR (typically up to 1273 K). Only few Thermo-Gravimetric Analysis (TGA) experiments of doped CeO2 with Fe cations are available. In a one particular study devoted to the effect of doping CeO2 with metal cations, it was found that the incorporation of Fe3+ increased the mass loss of CeO2 (due to reduction). In this case for Ce0.9Fe0.1O2−δ, 50.0 µmol of O2 per gmaterial were removed at each cycle during ten thermochemical cycles in which the thermal reduction step was performed at 1673 K. These were active sites since nearly stoichiometric CO production (by CO2 reduction) 96.3 µmol gmaterial−1·cycle at 1273 K was seen.64
5. Valence band (Ce1−yFeyO2)
Fig. 7A and B presents the valence band and shallow core levels (Ce 5p, O 2s, and Ce 5s) of Ce0.75Fe0.25O2−δ and Ce0.95Fe0.05O2−δ before and after argon ion sputtering for reduction. This reduction method has advantages and disadvantages when compared to reduction in the presence of H2 or thermally in inert environment. Thermal reduction of these oxides requires heating to at least 1773 K. The mixed oxide at this temperature largely segregates, and therefore, the analysis may not be conducted. Reduction using H2 in UHV is not possible because of its very low sticking coefficient on oxides. Argon ion sputtering to reduce oxides relies on the mass difference between M and O and the stability of the reduced phase. Ce3+, Fe2+, and Fe0 are stable in UHV conditions for a few hours because of the low H2O and O2 partial pressures. One of the disadvantages of Ar ion sputtering is the loss of surface structure (the formation of an amorphous layer due to the high energy used, 1 kV). | ||
| Fig. 7 (A) Valence band XPS (green shaded) and Ce 5p, O 2s (blue-shaded) and Ce 5s spectra of Ce0.75Fe0.25O2−δ before (0 minute) and after argon ion sputtering (x minutes). (B) Valence band XPS (green shaded) and Ce 5p, O 2s (blue-shaded) and Ce 5s spectra of Ce0.95Fe0.05O2−δ before (0 min) and after argon ion sputtering (x minutes). From ref. 16 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
The spectra of the as-prepared oxides (0 min) are dominated by the O 2p, O 2s, Ce 5p and Ce 5s lines, marginal changes are seen between both oxides. There are some, surface hydroxyls (–OH, 3σ) at a binding energy of ca. 10 eV. There is also a minor contribution from reduced Ce cations (Ce 4f, Ce3+) and reduced Fe cations (Fe 3d, oxidation state <+3), both have lower binding energies (at 0.5–2 eV with respect to Fe) than that of the O 2p binding energy band.
Upon argon ions sputtering, one notices the following:
(i) An increase of the signal below the O 2p line due to reductions (due to increased concentrations of Ce3+ and Fex+, x < +3).
(ii) An increase in surface hydroxyls.
(iii) A relative increase in Ce 5p signal with respect to the O 2s signal.
For (i) the increase is expected and is treated in more detail to extract quantitative information below. For (ii) the increase, that has been seen before, is due to the increase in the sticking coefficient of the traces of dissociatively adsorbed water over a reduced metal oxide when compared to its stoichiometric form. Ions bombardment causes a reduction due to oxygen removal (as atoms), and the remaining two electrons, per oxygen atom removed (VO), are transferred to Ce4+ (and/or Fe3+) to reduce them. The creation of VOs leads to the preferential dissociative adsorption of H2O. This results in the formation of two pairs of surface hydroxyls for each oxygen vacancy healed.
For (iii), this observation is treated qualitatively below.
Fig. 8A and B presents the valence band region, in which the signal below the XPS O 2p line was fitted by two peaks: at about 0.4 eV and at ca. 1.5 eV, binding energy. There are no noticeable changes in the large O 2p lines' shapes upon reduction. For both oxides, the signal attributed to the Ce 4f1 orbital is larger than that of the Fe 3d orbitals and is more pronounced for Ce0.25Fe0.05O2−δ when compared to Ce0.75Fe0.25O2−δ. This is consistent with core levels measurements and with the TCWS results (see below). For Ce0.95Fe0.05O2−δ, increasing the reduction time affects mostly the Ce cations, while for Ce0.75Fe0.25O2−δ, it favors Fe reduction. This might be due to the probability of hitting the atoms during bombardment. At a high Fe%, the probability of oxygen removal adjacent to Fe atoms is high, and therefore more Fe is reduced, while at a low Fe%, the oxide is more homogeneous and the chemical effect on the reduction is expected to be higher.
 | ||
| Fig. 8 (A) Valence band XPS of as-prepared Ce0.75Fe0.25O2−δ after 1, 2 and 5 minutes of Ar ion sputtering. (B) Valence band XPS of as-prepared Ce0.95Fe0.05O2−δ after 1, 2 and 5 minutes of Ar ion sputtering. (C and D) Computed peak areas of fitted Ce 4f (FWHM = 1.5 eV) and Fe 3d (FWHM = 1.5 eV) signals. From ref. 16 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
Fig. 9A and B presents the valence band and the Ce 5p and O 2s lines. The spectra are baseline-subtracted, then normalized to highlight the differences. Ions sputtering results in a preferential increase in the Ce 5p signal when compared to the O 2s lines. In addition, the O 2s line becomes narrower. There is no noticeable shift in the binding energy before or after ion bombardment. Similar experiments were conducted on CeO2, and no change was seen. Some qualitative information may be drawn upon comparisons with previous work conducted by others. In one study also using ions bombardment65 of CeO2, (Fig. 3a and b of ref. 65), the relative ratio has increased in favor of the Ce 5p orbital, like in this work. The exact position of the O 2s orbital with respect to the Ce 5p1/2 orbital is not clear. Here, it is put after the Ce 5p1/2 lines similar to other work, although others have put it in between the Ce 5p3/2 and Ce 5p1/2 energy positions based on the relativistic computation of CeO8 and Ce63O216 clusters.66 The O 2s and Ce 5p lines give information on charge transfer; because of their quasi-degenerate energy positions they are sensitive to the oxidation state of Ce cations. The spectra in Fig. 9 are similar to those reported for a thin film of CeO2 grown on Rh(111) excited with photon energy equal to 125 eV (Ce 4d–Ce 4f resonance).67 The spectra are also similar to those of irradiated (with Xe ions) CeO2 thin film and bulk [ref. 65]. The authors pointed out to the final state effect (3d94f1OVMO−1 (outer valence molecular orbital, OVMO) and 3d95p5np1 (inner valence molecular orbital, INVO)). The Ce 5p atomic orbitals participate in the formation of both OVMO and IVMO, where a large part of the latter is taken by the filled Ce 5p1/2, 5p3/2 and O 2s atomic shells.68
The increase in the Ce 5p/O 2s intensity ratio after sputtering reflects the creation of oxygen vacancies and the associated reduction of Ce4+ to Ce3+. Removal of oxygen decreases the O 2s contribution to the inner valence molecular orbitals while electrons left by the vacancy populate Ce 4f states. The resulting modification of the Ce–O valence manifold enhances the Ce-derived component of the IVMO. From ref. 16 (no permission is needed: Creative Commons Attribution 4.0 License).
6. X-ray photoelectron spectroscopy (XPS) (Ce1−yFeyO2)
Fig. 10 presents XPS Ce 3d lines of Ce cations in the fresh and reduced samples. The presence of both oxidation states of Ce cations is clear. The lines' positions and attributions are shown (3d5/2: u, u″ and u‴, and 3d3/2: v, v″ and v‴ for Ce4+ cations, and 3d5/2: uo and u′ and 3d3/2: vo and v′ for Ce3+ cations). In Fig. 10 and the inset table, a comparison between the fresh and the most reduced samples is made. The presence of Fe at a 5% concentration resulted in a more pronounced Ce3+ concentration when compared to the presence of Fe at a 25% concentration. This is in line with the valence band results presented in Fig. 7 and 8. CeO2 alone showed a very mild increase upon reduction; this is also in line with the virtually no change in the Ce 5p/O 2s lines. While Ar ions sputtering relies on momentum transfer of incoming ions (in this case, 1 keV of kinetic energy), which results in breaking the chemical bonds there should be no difference between CeO2 and Fe-substituted CeO2 since the incoming ions have much more energy than the chemical bond, assuming complete energy transfer. However, this is a cascade reaction where the energy transfer occurs consecutively, and therefore the last steps of an incoming ion (before it leaves the material or is implanted in it irreversibly) would be more efficient for weaker bonding. | ||
| Fig. 10 XPS Ce 3d spectra of CeO2 (left panel), Ce0.75Fe0.25O2−δ and Ce0.95Fe0.05O2−δ (right panel) after 5 minutes of Ar ion sputtering. Also shown are the computed contributions of the Ce3+ cations. From ref. 16 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
Based on the Ce 4f/O 2p, Ce 5p/O 2s and Ce 3d (for Ce4+ and Ce3+) XPS signals, Table 5 is made to provide an estimate of the reduction of Ce cations.
| Oxide | Ce3+/Ce4+ (Ce 3d) | [Ce 4f + Fe 3dx]/O 2p | Fe0/Fe3+ (Fe 2p) | Ce 5p/O 2s |
|---|---|---|---|---|
| CeO2 | 0.2 | ≈0 | ≈0 | 1.1 |
| Ce0.75Fe0.25O2−δ | 2.3 | 0.3 | 0.5 | 1.6 |
| Ce0.95Fe0.05O2−δ | 3.6 | 0.4 | 0.6 | 1.75 |
The XPS core levels of iron oxides are among the most studied of oxide materials.69 There are three common oxidation states: Fe3+, such as in Fe2O3, Fe2+, such as in FeO, and Fe3O4, in addition to Fe0. The spectra are complicated by the presence of satellites,70–72 iron hydroxide (FeOOH)73,74 and many multiplets.75 The binding energy of the XPS Fe 2p signal is about 707 eV for Fe0, 710 eV for Fe2+ and 711 eV for Fe3+. In Ce1−yFeyO2, a further complication arises from the presence of Ce Auger lines in the Fe 2p region. Although charge neutralization was used to acquire the spectra, the as-prepared oxide always had wider peaks when compared to that reduced upon ion sputtering. Therefore, peak areas are to be taken as an estimate and binding energies within a 0.5 eV accuracy. Both oxides show similar spectra and trends upon reduction (Fig. 11). While, the as-prepared oxides contained Fe3+ cations, they, however, also contained Fe2+ and some Fe0. The small % of Fe2+ might be formed during the preparation. The presence of metallic iron was not expected and might be due to interstitial atoms formed due to strong lattice distortion. It is also possible that this peak is due to another Auger line of Ce cations. The insets in both figures show the trend during the reduction. In both cases, the amount of Fe2+ increases, similar to a previous study,76 then decreases to zero, indicating that within the reduction time studied, all reducible Fe3+ cations were transformed to Fe0. In line with the valence band results, it appears that both Ce4+ and Fe3+ cations are more efficiently reduced when in small amounts (Fe0.05), although the difference is not as pronounced.
 | ||
| Fig. 11 (A) XPS Fe 2p spectra of as-prepared Ce0.75Fe0.25O2−δ after 5 minutes of Ar ions sputtering. (B) XPS Fe 2p spectra of as-prepared Ce0.95Fe0.05O2−δ after 5 minutes of Ar ions sputtering. Insets in (A and B): quantitative analysis of XPS Fe 2p spectra of Ce0.75Fe0.25O2−δ and Ce0.95Fe0.05O2−δ before and after sputtering at the indicated time. Data acquisitions were conducted using an Al Kα X-rays. From ref. 16 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
7. Epitaxy (Ce1−yUyO2)
Growing the mixed metal oxide as an epitaxial film in a controlled environment allows more accurate studies of the charge transfer process. It also offers an alternative prototype material for the reaction because both heat and mass transfer limitations would be largely removed. Many previous works have shown epitaxial films of CeO2 on metal single crystals such as Ru(0001)77–79 and Pt(111)80,81 surfaces. Here the characterization of a series of Ce1−yUyO2±δ (111) thin films of about 6 nm thick grown on Ru(0001)/Al2O3(0001) templates is presented and discussed.Fig. 12 shows the Low Energy Electron Diffraction (LEED) pattern of (111)-oriented epitaxial thin films for a series of Ce1−yUyO2±δ. Samples with low U content present a faint ring associated with a fraction of randomly oriented islands. Increasing the U fraction translates into a blurring of the spots due to a higher disorder and a larger density of structural defects. The absence of Ru(0001) spots indicates a complete coverage of the whole substrate.
 | ||
| Fig. 12 LEED patterns: (A) clean Ru(0001)/Al2O3(0001) substrate. The hexagonal pattern indicated by solid violet circles corresponds to the Ru spots, while the dashed green circles indicate the (2 × 1) reconstruction of adsorbed oxygen. (B) and (C), CeO2 and UO2 (111) reference thin films; solid circles (blue and red) indicate their spots in azimuthal registry with the Ru(0001). For the CeO2 pattern, the dashed blue arc indicates the faint ring associated with a small fraction of randomly oriented islands. (D)–(F) LEED patterns of Ce1−yUyO2±δ thin films with y = 0.05, 0.2, and 0.6, respectively. Solid orange circles indicate their spots in azimuthal registry with the Ru(0001). From ref. 22 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
In situ XPS measurements of Ce 3d and U 4f regions are presented in Fig. 13A and B for CeO2 and UO2 references, respectively. The Ce 3d lines are dominated by the Ce4+ cations (v = 882.6 eV, v‴ = 898.7 eV, and u‴ = 916.9 eV) for the UHV annealed film. The small contribution from Ce3+ cations (v′ ca. 886 eV) disappears upon heating in O2. For the UHV annealed UO2 film, XP U 4f spectra indicate the presence of U4+ cations (U 4f7/2 = 380.2 eV and its satellite at 6.9 eV above). Heating in the presence of O2 resulted in the oxidation of a fraction of U4+ cations to U5+ cations (U 4f7/2 = 381.1, U 4f5/2 = 391.8 and its satellite at ca. 8 eV above; the U 4f5/2 component of the U4+ cations obscures the U 4f7/2 satellite of the U5+ cations).82 The UHV annealed films are mostly composed of Ce4+ (for CeO2) and exclusively composed of U4+ (for UO2).
 | ||
| Fig. 13 (A) XPS Ce 3d of CeO2 thin film after annealing in UHV (bottom) and heating in 5 × 10−7 mbar O2 for 30 minutes (top). (B) XPS U 4f of UO2 thin film after annealing in UHV (bottom) and heating in 5 × 10−7 mbar O2 for 30 minutes (top). The insets show the LEED images of the UHV annealed films. From ref. 22 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
8. Charge transfer from U4+ to Ce4+ cations
8.1. Experimental
A noticeable difference is seen for the mixed oxides samples. Fig. 14A and B presents the same XPS regions as those shown in Fig. 13 for the Ce0.8U0.2O2±δ. XPS Ce 3d indicates (Fig. 14A) that the UHV annealed film contains a significant fraction of Ce3+ (which was not observed for CeO2±δ); the estimated ratio Ce3+/Ce4+ is equal to 2.7. XPS U 4f lines of the same film contain a large fraction of U5+ (the UHV annealed of UO2 did not contain U5+); the U5+/U4+ was found equal to 1.5. Similar trends were obtained for other investigated mixed oxides, yet quantitative differences were observed. In particular, the charge transfer occurs during the annealing process (as indicated by eqn (3)). Heating in O2 resulted in preferential oxidation back to Ce4+, yet under these conditions, part of the oxygen consumed for the oxidation was taken or transferred from U5+–O because the oxidation trend has reversed (Fig. 14B). This indicates that the dynamic of the electron transfer between both metal cations is sensitive to the partial pressure of O2 in addition to the Ce to U ratio. | ||
| Fig. 14 (A) XPS Ce 3d of Ce0.8U0.2O2±δ after annealing in UHV (bottom) and heating in 5 × 10−7 mbar O2 for 30 minutes (top). (B) XPS U 4f of Ce0.8U0.2O2±δ after annealing in UHV (bottom) and heating in 5 × 10−7 mbar O2 for 30 minutes (top). The inset shows the LEED images of the UHV annealed samples. The arrows in (B) point to the ΔE of the satellites (6.9 eV for U4+ and 8.2 eV for U5+). From ref. 22 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
8.2. Compositional effect (Ce1−yUyO2)
Increasing the percentage of U decreased the fraction of oxidized U cations Fig. 15A and B. Also, the ratio Ce3+/Ce4+ decreased compared to that of the lower U content. This trend indicates that the reduction of Ce4+ decreases when U cations content is significant and is not solely explained by the charge transfer described in eqn (3). However, Ce3+ cations were still present even when heated in O2, while U cations were oxidized to U5+ and a small fraction to U6+. As mentioned above, previous works on polycrystalline Ce0.5U0.5O2 showed segregation of the uranium oxide upon heating,20 with diffraction lines that are attributed to either/or U2O5 and U3O8. Taking these observations together and the fact that UO2 tends to become UO2+x, one may conclude that it may segregate to the top of the thin film, shielding CeO2 from being oxidized, and that is why Ce3+ cations are still present even when the film was annealed in the presence of O2. While Ce3+ cations are formed, when the percentage of U cations is high, the former will not be as active because they are in deeper layers, thus explaining the decrease of the activity of the mixed oxide for water splitting (as presented below) with increasing U content after the optimal concentration. | ||
| Fig. 15 (A) XPS Ce 3d of Ce0.4U0.6O2±δ after annealing in UHV (bottom) and heating in 5 × 10−7 mbar O2 for 30 minutes (top). (B) XPS U 4f of Ce0.4U0.6O2±δ after annealing in UHV (bottom) and heating in 5 × 10−7 mbar O2 for 30 minutes (top). The inset shows the LEED image of the UHV annealed sample. The arrows in (B) point to the ΔE of the satellites (6.9 eV for U4+ and 8.2 eV for U5+). The red line is at the position of a U6+ satellite; this, together with the large FWHM of the main lines (U 4f7/2,5/2), indicates that U cations have a wide range of oxidation states from +4 to +6. From ref. 22 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
8.3. Computation
Fig. 16(A and B) presents the results of the DFT+U computation of Ce31UO64 before and after creating an oxygen vacancy to make a Ce31UO63 super-cell. There are no Ce 4f electrons before the removal of the oxygen ion, while three electrons on three Ce cations (Ce 4f orbitals) are seen after the creation of the VO. Removing an oxygen ion from the lattice has resulted in the transfer of two electrons towards two Ce4+ cations, plus the additional reduction of a third Ce4+ to Ce3+ due to charge transfer from U4+ that becomes U5+. Fig. 16(C and D) presents the bond distances between Ce (U) and oxygen ions and the Bader charge analysis in the presence of VO. Contraction in the U–O bonds and expansions in the Ce–O bonds is observed. Quantitative description is given in Table 6, in which the focus is made on the bonds of seven oxygen anions with Ce and U cations (the oxygen atoms are labeled 1 to 7). Ce3+ cations directly adjacent to the vacancy experience an expansion in their bond distance to the nearest oxygen by 2.7%. Also, Ce4+ cations adjacent to the nearest oxygen atoms, labeled Ced−i, experience an even larger bond distance expansion of 6.7%. On the contrary, the U5+ cation shows a contraction of its bond distances to the seven oxygen ions. The contraction is, anisotropic and spans from 2.5 to 6.6%. | ||
| Fig. 16 (A) A section of the super cell (Ce1−xUxO2: (2 × 2 × 2) model (31Ce, 1U, and 64O atoms); U% = 3.1%) showing the U cation (purple), Ce cations (yellow) and oxygen anions (red). The arrow points to the oxygen ion to be removed. Note the 5f orbitals of the U4+ cation that contain two electrons and the absence of 4f electrons on the Ce4+ cations. (B) The same section as in (A) after removing the oxygen anion. Three Ce cations have accommodated one electron each in their 4f states, while the U cation has lost one electron. (C) Bond distances of the Ce and U cations adjacent to the oxygen vacancy (Ce–O and U–O distances of the stoichiometric cells are Ce–O = 2.372 Å and U–O = 2.342 Å). (D) Bader charges on the three Ce cations and U cation in the presence of VO. From ref. 22 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
| Atom | O1,2,3 | O4,5,6 | O7 |
|---|---|---|---|
| Cea,b,c | 2.436 (+2.7%) | ||
| U | 2.188 (−6.6%) | 2.283 (−2.5%) | 2.244 (−4.2%) |
| Ced,e,f,g,h,i | 2.531 (+6.7%) |
To study the configurational effect of U cations on the reduction of Ce4+ cations, computation of a system in which U cations having the same concentration but in two different positions were conducted. Fig. 17A and B presents results from a super cell (4 × 2 × 1) containing 3 U cations of a Ce29U3O64. In one case (Fig. 17A) they are surrounding a Ce4+ cation, representing a high local concentration, while in the second case (Fig. 17B) one of the three cations is moved away, three units to the left on the x-axis and one unit up on the y-axis (a unit is defined as the Ce4+ to Ce4+ distance, which is equal to 3.86 Å).
 | ||
| Fig. 17 (A and B) Effect of U arrangement around Ce ions on electron transfer. U concentration is 9.3% of the (4 × 2 × 1) bulk model (Ce29U3O64 (Ce0.907U0.093O2)). (i) and (iv) U, Ce, and O are in blue, yellow, and red; top view. (ii) and (v) Projected orbitals on U and Ce ions; top view. (iii) and (vi) Projected orbitals on U and Ce ions; side view. (C) U concentration is 6.2% of the (2 × 2 × 2) bulk model (Ce30U2O63 (Ce0.937U0.063O1.969)) in the presence of VO. (D) Bond distances and Bader charge on Ce in the presence of VO (from C). From ref. 22 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
No electron transfer occurred in the first configuration, while in the second, in which only two U cations were adjacent to Ce4+, transfer did occur to the Ce cation at equidistance from the two U cations. This occurred in the absence of VO. Fig. 17C and D presents a specific case of Fig. 17B. The removal of an oxygen ion in this case did not result in the formation of three Ce3+ but in two. In this case, the presence of the two U cations adjacent to each other prevented the charge transfer upon the removal of an oxygen ion.
Based on the results on polycrystalline and epitaxial Ce1−yUyO2 samples, it was observed (in both cases) that charge transfer from U to Ce cations occurs. Its extent, however, has a considerable entropic factor. While low concentrations of U cations are needed, this is not solely related to a dilution effect. Also, the analysis of model Ce1−yUyO2 (111) thin films indicates that the fraction of Ce4+ reduced to Ce3+ is larger than that of U4+ oxidation to U5+ (or U6+) by a factor far exceeding experimental uncertainties (more details can be found in ref. 22). At high U concentrations, U cations migrate to the shell of the crystallites. Besides the fact that UO2 is easily oxidized to higher oxidation states, O2 adsorption on UO2(111) is possible (with a computed adsorption energy of −0.24 eV),83 whereas there are no known experimental reports of O2 irreversible adsorption on CeO2(111). Together, these facts would result in a core–shell structure (UOz/Ce1−yUyO2), preventing Ce3+ cations from being oxidized in the core of the crystallite. Such element migration and phase separation are not uncommon in metallic nanoparticles; for example, an alloy of RhPd or RhPt particles sees migration of Rh to the surface in an oxidized environment84 because it is easier to oxidize Rh when compared to Pt or Pd. The reduction of Ce cation in the presence of U cations, while occurs through charge transfer, is therefore sensitive to the configuration of the metal cations, and, as pointed out computationally before (ref. 11), its energy is sensitive to statistical entropy contribution.
9. Hydrogen production from water
9.1. Polycrystalline Ce1−yFeyO2
Fig. 18 presents hydrogen production on CeO2, Ce0.95Fe0.05O2−δ and Ce0.75Fe0.25O2−δ from water, these were prior reduced with hydrogen 973 K (700 °C). The use of hydrogen allows reduction at temperature low enough were phase segregation is minimal. Moreover, while the TCWS technology relies on direct energy input from the sun using solar concentrators, with its considerable challenges,85 at the laboratory scale heat is provided by electrical furnaces instead in order to maintain a controlled and stable energy source. The objective here is to relate H2 production (performance) to the observed reduced states (spectroscopy) of the oxides. Because, as indicated by XRD, the two mixed oxides are not segregated when heated to this temperature, a link to spectroscopic measurements is possible. CeO2 alone shows negligible activity, while the Fe-substituted cerium oxides are active. The activity of the Ce0.95Fe0.05O2−δ is almost twice that of the Ce0.75Fe0.25O2−δ catalyst. This suggests that the activity is more linked to Ce3+ than to Fe0 (based on the XPS Ce 3d and XPS Ce 5p/O 2s spectra). | ||
| Fig. 18 Hydrogen production from water over CeO2, Ce0.75Fe0.25O2−δ and Ce0.95Fe0.05O2−δ, which were previously reduced with hydrogen at 973 K at one atmosphere (reaction temperature = 973 K). The total H2 production was found equal to 0.2, 7.4 and 11.4 µmol goxide−1 for CeO2, Ce0.75Fe0.25O2−δ and Ce0.95Fe0.05O2−δ, respectively. From ref. 16 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
9.2. Polycrystalline Ce1−yUyO2
The water splitting to hydrogen at 973 K for the series polycrystalline Ce1−xUxO2±δ (where 0.05 ≤ x ≤ 0.75, including pure CeO2 and UO2) was further studied to see the effect of substituting Ce4+ by U4+ cations on the reaction. Fig. 19A presents H2 production from water (vapor pressure 23 torr) as a function of time over a prior reduced Ce0.95U0.05O2. The calculated total H2 production per weight over these oxides is presented in Fig. 19B. The binary oxides (CeO2 and UO2) show a small activity at both ends of the plot. The presence of U in small amounts resulted in an enhancement of H2 production by ca. 30 times when compared to CeO2. Increasing the amounts of U, while still showed enhancement compared to CeO2, was less pronounced. Also presented are the percentage of Ce3+ states (as obtained from XPS Ce 3d lines20) that were produced upon Ar-ions sputtering of the same polycrystalline mixed oxide series. The trend is similar to H2 production: substituting Ce by U cations increased the reduction of Ce3+ considerably. Fig. 19C presents some of the data of the H2 production shown in Fig. 19B as a function of bulk VO formation energy (EVO), computed using DFT+U (Ueff = 4 eV for U and 5 eV for Ce) taken from ref. 13. EVO represents the energy required to remove an oxygen atom from the oxide lattice and is defined as the energy difference between a reduced slab and the sum of the energies of the corresponding stoichiometric slab and the removed oxygen atoms. If oxygen vacancies were isolated and non-interacting, one expects a linear relationship between EVO and the extent of reduction δ, taking in consideration that H2 production is proportional to δ/2 (as per eqn (2)). | ||
| Fig. 19 (A) H2 production from water at 973 K on pre-reduced Ce0.95U0.05O2. (B) H2 production on a series of Ce1−xUxO2±δ mixed oxide as a function of U content (right y-axis). The percentage of Ce3+ (XPS Ce 3d lines from ref. 20) cations obtained upon reduction with Ar-ions prior to XPS collection is shown in the left y-axis. (C) H2 production on a selected samples from the series presented in (B) as a function of VO formation energy computed using DFT+U, from ref. 12. From ref. 22 (no permission is needed: Creative Commons Attribution 4.0 License). | ||
The introduction of U considerably decreased the energy needed to remove bulk oxygen atoms. The effect is offset by increasing U content, because it is harder to reduce U4+ cations.
10. Thermal water splitting (Ce1−yFeyO2)
Fig. 20 presents the activity of reduced CeO2 and reduced Ce0.95Fe0.05O2−δ for thermal water splitting at 1200 °C while Fig. 21 presents the STEM, EDX and EELS results of the Ce0.95Fe0.05O2−δ oxide after the reaction. The reduction was conducted upon heating to 1550 °C for two hours in the absence of H2 (under N2). The incorporation of Fe (Fe/Ce = 0.05) into CeO2 has resulted in two main effects. It increased the kinetic of H2 production (Fig. 20C) and the yield; total amount has almost doubled (per unit weight of oxide) (Fig. 20B and C). This is in line with spectroscopic results where there is an increased reduction of CeO2 due to the presence of Fe cations (up to a threshold level). The FWHM of the production peak decreased from ca. 35 min to about 10 min which gives a simple observation on the kinetic effect. To further see the effect of Fe on the reduction kinetics, the decay part of the peaks was fitted with an exponential function. Attempts with a single exponential decay fit were not successful, a double exponential fit was found to be adequate with R2 of 0.999. Table 7 presents the different parameters of the decay for both oxides. The addition of Fe has increased the time constant for water splitting by a factor of 2 when compared to CeO2 alone. However, increasing the number of reduced sites, should not change the decay time but mostly affect the amplitude (pre-factors A1 and A2) if these sites were all of similar nature and do not interact with each other. The increase of the time constant by a factor of two indicates that the reduced sites are more reactive than those of reduced CeO2 alone. It is not simple to attribute a separate physical meaning to both decay constants. It is possible that the (t2) decay describes events requiring more (or additional) energy than the first one; such as defects diffusion from the bulk to the surface. Initially defects are statistically distributed in the bulk and on the surface, then with the reaction progress more available defects are healed (and the process would more dependent on diffusion of O2− (or VO) within the oxide), and this decreases the reaction rate. In other words, the first decay might be largely independent from defects distribution and migration while the second is affected. | ||
| Fig. 20 Thermo-chemical water splitting over CeO2 (A) and Ce0.95Fe0.05O2−δ (B) at 1200 °C (1473 K), the oxides were reduced under N2 at 1550 °C (1823 K) for two hours prior to use. The total amount of H2 from water per g of oxide is indicated. (C) Normalized fitting of the decay part of H2 production using a bi-exponential function for both oxides. From ref. 50, permission license number 6133081449846. | ||
 | ||
| Fig. 21 STEM, EDX, EELS of Ce0.95Fe0.05O2−δ after the reaction presented in Fig. 20. (A) and (B) are for one particle and (C) and (D) are for another particle. (D) An elemental EELS map of the particle in (C). The numbers 1 to 4 in (E) are labeled in (B). While segregation of iron to the edges of CeO2 is seen some iron is still present within the crystallite (also note the presence of FeOx on the large CeO2 crystallite in (C)). The crystallite size (TEM) is close to 1 µm (an increase of about 200 times when compared to that calcined at 500 °C). Yellow for cerium, blue of iron and red for oxygen atoms. From ref. 50, permission license number 6133081449846. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e−x/t1 + A2
e−x/t1 + A2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e−x/t2a
e−x/t2a
The activation energy for oxygen diffusion in CeO2 is typically a small fraction from that needed to reduce it (10–20% or so).86 In the process of oxidation of the reduced CeO2 with water vapor two chemical steps occur. (i) Water dissociative adsorption and (ii) oxygen anions diffusion from the surface to the bulk (or VO diffusion from the bulk to the surface). (i) The first one is different on a stoichiometric surface when compared to defected ones and therefore their kinetic effect on the reaction is expected to be different (water dissociative adsorption over reduced CeO2 has a stronger energy than that on stoichiometric surface87). (ii) The particles have considerably sintered (Fig. 21) due to heat, so intra particle pore diffusion of water molecules may be neglected. While the increase of the volume to surface ratio upon sintering makes the process mostly bulk driven (since the majority of the oxygen defects would be located in the bulk) the reaction would still be surface driven first if the adsorption of water is a limiting step. While a fraction of Fe oxides has segregated out there is still some Fe inside as seen by EDX and EELS (Fig. 21). It is therefore possible that, in some locations, the interface has the needed gradient concentration of Fe cations to affect the reaction rate when compared to pure CeO2.
The three following equations summarize the main reactions that occur during water oxidation. Large crystallites of CeO2 are (111) O-terminated in the fluorite structure and these terminated oxygen anions are bonded to three Ce cations in the second layer, yet for the sake of simplicity it is easier to write them as (Ce–O–Ce) bearing in mind that they are not in the same plane.
10.1. Reaction steps
Electron transfer reaction: water dissociative adsorption on surface oxygen defect followed by oxidation and hydrogen generation.| H2O + [Ce3+–VsO–Ce3+]surface → H2 + Ce4+–O2−–Ce4+ | (4) |
VsO is for a surface oxygen defect and Ce3+ is for a reduced cation in the second layer of the (111) terminated surface.
Acid–base interaction: water dissociative adsorption over stoichiometric sites (no charge transfer).
| H2O + [Ce4+–O2––Ce4+]surface → Ce4+(−OH)–O2−(H+)–Ce4+ | (5) |
Diffusion: surface oxygen diffusion into the bulk (or VO diffusion to the surface) driven by heat and water dissociative adsorption (eqn (4) and (5)).
| [Ce4+–O2−–Ce4+]surface + [Ce3+–VbO–Ce3+]bulk → [Ce3+–VsO–Ce3+]surface + [Ce4+–O2−–Ce4+]bulk | (6) |
VbO is bulk oxygen defect.
The two protons in eqn (5) (of the two surface hydroxyls) become one molecule of hydrogen upon the reaction with two electrons from eqn (6).
The substitution of a fraction of Ce cations by Fe cations on the surface and in the bulk affect eqn (4) and (6). Eqn (5) is an acid–base exchange reaction that is largely not affected by the change in the nature of a metal cation. Although it can be affected by the degree of coordination88 (such as in different surface structures) this may be neglected here because of the small energy difference. The concentration of VO has increased in the presence of Fe cations and this affects primarily eqn (4). However, the faster kinetics indicate that the dissociative adsorption of water on oxygen vacancy sites is also accelerated when Fe cations are present. Therefore, in the presence of Fe cations in the CeO2 lattice, eqn (7) bellow, is expected to be faster than eqn (4) if the dissociative adsorption energy is higher on Fe–VsO–Ce when compared to Ce–VsO–Ce centers.
| H2O + [Ce3+–VsO–Fe2+]surface → H2 + Ce4+–O2−–Fe3+ | (7) |
11. Conclusions
The chemical states of Ce3+ cations in Ce1−yFeyO2 are analyzed by their deep (XPS Ce 3d) and shallow (XPS Ce 5p) core levels in addition to their valence level (XPS Ce 4f) spectroscopy at different Fe3+ concentrations. Moreover, the states of Ux+ in Ce1−yUyO2 were further analyzed by their core levels and DFT+U methods. HR-STEM and EELS for the as prepared mixed oxides showed, in line with XRD results, that the cations (Fe3+ or U4+) are homogeneously distributed inside CeO2. While the addition of Fe3+ cations doubled the reaction yield and considerably increased the cycle kinetics, phase segregation occurred upon thermochemical water splitting reaction when the mixed oxide was reduced by heating in an inert environment at 1550 °C. A three-step mechanism for the water splitting reaction is presented in which the dissociative adsorption, the bulk diffusion of oxygen anions and the hydrogen re-combinative desorption are discussed. Results point to the role of Ce–O–Fe sites in improving the reaction kinetics, yet the considerable sintering indicates that maintaining oxide homogeneity is needed for long-term stability. In the case of Ce1−yUyO2, the TCWSR to H2 is sensitive to the content of U, where low percentages of U cations considerably enhanced the production. This enhancement is attributed to the increase in Ce4+ cations reduction to Ce3+ cations. The trend of this Ce4+/Ce3+ conversion studied by in situ XPS of single crystalline mixed oxides showed evidence of charge transfer (Ce4+ + U4+ → Ce3+ + U5+) at low U content. Evidence of charge transfer was further obtained computationally using the DFT+U method, giving more support to the experimental Ce4+/Ce3+ and U4+/U5+/U6+ results. This charge-transfer effect is sensitive to the local distribution of U4+ cations in the ceria lattice. One of the key conclusions is the need to lower the reduction temperature of the mixed oxide phase considerably to prevent phase separation. There is also a need to further study the entropical effect of the mixed oxides on the reduction of the host material, as it appears to be an important factor for the charge transfer reaction. Several strategies are discussed to mitigate phase segregation in ceria-based redox materials, including maintaining low dopant concentrations to preserve solid solutions, selecting dopants with compatible ionic radii and crystal structures, and employing synthesis methods that promote homogeneous cation distribution.Conflicts of interest
The author declares no conflicts of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.References
- C. L. Muhich, B. D. Ehrhart, I. Al-Shankiti, B. J. Ward, C. B. Musgrave and A. W. Weimer, WIREs Energy Environ., 2016, 5, 261–287, DOI:10.1002/wene.174.
- D. Schweke, Y. Mordehovitz, M. Halabi, S. Lee and S. Hayun, Adv. Mater., 2018, 30, 1706300, DOI:10.1002/adma.201706300.
- I. Al-Shankiti, Y. Man Choi, F. M. Al-Otaibi and H. Idriss, Catalyst for thermochemical water splitting, US Pat., US9421537, 2016US Pat., US9675961, 2017. Search PubMed.
- S. Chuayboon and S. Abanades, Int. J. Hydrogen Energy, 2020, 45, 25783–25810, DOI:10.1016/j.ijhydene.2020.04.098.
- Y. Mao, Y. Gao, W. Dong, H. Wu, Z. Song, Z. Xiao, J. Sun and W. Wang, Appl. Energy, 2020, 267, 114860, DOI:10.1016/j.apenergy.2020.114860.
- F. M. Pinto, V. Y. Suzuki, R. C. Silva and F. A. La Porta, Front. Mater., 2019, 6, 260, DOI:10.3389/fmats.2019.00260.
- T. Lin, X. Lv, Z. Hu, A. Xu and C. Feng, Sensors, 2019, 19, 233, DOI:10.3390/s19020233.
- T. Montini, M. Melchionna, M. Monai and P. Fornasiero, Chem. Rev., 2016, 116, 5987–6041, DOI:10.1021/acs.chemrev.5b00603.
- U. Johnson Etim, Y. Song and Z. Zhong, Front. Energy Res., 2020, 8, 545431, DOI:10.3389/fenrg.2020.545431.
- K. Taniya, Y. Horie, R. Fujita, Y. Ichihashi and S. Nishiyama, Appl. Catal., B, 2023, 330, 122568, DOI:10.1016/j.apcatb.2023.122568.
- D. A. Andersson, S. I. Simak, N. V. Skorodumova and I. A. Abrikosov, Börje Johansson, Appl. Phys. Lett., 2007, 90, 031909, DOI:10.1063/1.2431775.
- B. E. Hanken, C. R. Stanek, N. Grønbech-Jensen and M. Asta, Phys. Rev. B, 2011, 84, 085131, DOI:10.1103/PhysRevB.84.085131.
- J. Scaranto and H. Idriss, Top. Catal., 2015, 58, 143–148, DOI:10.1007/s11244-014-0353-x.
- M. Kato, M. Watanabe, S. Hirooka and R. Vauchy, Front. Nucl. Eng., 2023, 1, 1081473, DOI:10.3389/fnuen.2022.1081473.
- M. Moser, M. Pecchi and T. Fend, Energies, 2019, 12, 352, DOI:10.3390/en12030352.
- H. Idriss, Inorganics, 2024, 12, 42, DOI:10.3390/inorganics12020042.
- Y. Hinatsu and T. Fujino, J. Solid State Chem., 1988, 76, 124–130, DOI:10.1016/0022-4596(88)90199-5.
- L. Shelly, D. Schweke, S. Zalkind, N. Shamir, S. Barzilai, T. Gouder and S. Hayun, Chem. Mater., 2018, 30, 8650–8660, DOI:10.1021/acs.chemmater.8b03894.
- D. Prieur, J.-F. Vigier, K. Popa, O. Walter, O. Dieste, Z. Varga, A. Beck, T. Vitova, A. C. Scheinost and P. M. Martin, Inorg. Chem., 2021, 60, 14550–14556, DOI:10.1021/acs.inorgchem.1c01071.
- Y. Al-Salik, I. Al-Shankiti and H. Idriss, J. Electron Spectrosc. Relat. Phenom., 2014, 194, 66–73, DOI:10.1016/j.elspec.2013.11.013.
- H. Idriss, I. Al-Shankiti, Y.-M. Choi and F. M. Al-Otaibi, Catalyst for thermochemical water splitting, US Pat., US9675961, 2017 Search PubMed.
- C. Morales, R. Tschammer, T. Gouder, Y. M. Choi, D. Anjum, A. Baunthiyal, J.-O. Krisponeit, J. Falta, J. I. Flege and H. Idriss, J. Phys.: Energy, 2025, 7, 025012, DOI:10.1088/2515-7655/adbad9.
- L. Cheng, X. Yue, J. Fan and Q. Xiang, Adv. Mater., 2022, 34, e2200929, DOI:10.1002/adma.202200929.
- S. Zhao, D. Kang, Y. Liu, Y. Wen, X. Xie, H. Yi and X. Tang, ACS Catal., 2020, 10, 11739–11750, DOI:10.1021/acscatal.0c02832.
- K. Ahn, D.-S. Yoo, D. H. Prasad, H.-W. Lee, Y.-C. Chung and J.-H. Lee, Chem. Mater., 2012, 24, 4261–4267, DOI:10.1021/cm3022424.
- M. Zhang, D. Jiang and H. Jiang, Mater. Res. Bull., 2012, 47, 4006–4012, DOI:10.1016/j.materresbull.2012.07.012.
- M. D. Krcha and M. J. Janik, Langmuir, 2013, 29, 10120–10131, DOI:10.1021/la401747n.
- G. Righi, S. Benedetti and R. Magri, J. Phys.: Condens. Matter, 2022, 34, 204010, DOI:10.1088/1361-648X/ac58d9.
- M. Nolan, J. Mater. Chem., 2011, 21, 9160–9168, 10.1039/C1JM11238D.
- X. Wang, M. Shen, J. Wang and S. Fabris, J. Phys. Chem. C, 2010, 114, 10221–10228, DOI:10.1021/jp101100f.
- S. Grieshammer, B. O. H. Grope, J. Koettgen and M. Martin, Phys. Chem. Chem. Phys., 2014, 16, 9974–9986, 10.1039/C3CP54811B.
- X.-S. Liu, Xu-D. Wang, M. Yao, W. Cui and H. Yan, Catal. Commun., 2015, 63, 35–40, DOI:10.1016/j.catcom.2014.09.032.
- H.-T. Chen and J.-G. Chang, J. Phys. Chem. C, 2011, 115(30), 14745–14753, DOI:10.1021/jp201231d.
- Y. Yang, M. Zhang, H. Shi, J. Qu, Y. Xiong, W. Tan, X. Ge and Q. Jin, J. Environ. Sci., 2025, 158, 39–49, DOI:10.1016/j.jes.2025.02.001.
- S. Grieshammer, S. Eisele and J. Koettgen, J. Phys. Chem. C, 2018, 122, 18809–18817, DOI:10.1021/acs.jpcc.8b04361.
- Z. Hu and H. Metiu, J. Phys. Chem. C, 2011, 115, 17898–17909, DOI:10.1021/jp203774u.
- M. Alaydrus, M. Sakaue and H. Kasai, Phys. Chem. Chem. Phys., 2016, 18, 12938–12946, 10.1039/C6CP00637J.
- S. P. Waldow and R. A. De Souza, J. Phys.: Energy, 2020, 2, 024001, DOI:10.1088/2515-7655/ab5cfd.
- H. Wang, Z. Qu, H. Xie, N. Maeda, L. Miao and Z. Wang, J. Catal., 2016, 338, 56–67, DOI:10.1016/j.jcat.2016.02.009.
- M. Lykaki, S. Stefa, S. A. C. Carabineiro, P. K. Pandis, V. N. Stathopoulos and M. Konsolakis, Catalysts, 2019, 9, 371, DOI:10.3390/catal9040371.
- S. Halima Beguma, C.-T. Hung, Y.-T. Chen, S.-J. Huang, P.-H. Wu, X. Han and S.-B. Liu, J. Mol. Catal. A: Chem., 2016, 423, 423–432, DOI:10.1016/j.molcata.2016.07.036.
- H. Wang, Z. Qu, H. Xie, N. Maeda, M. Lei and W. Zhong, J. Catal., 2016, 338, 56–67, DOI:10.1016/j.jcat.2016.02.009.
- M. Lykaki, S. Stefa, S. A. C. Carabineiro, P. K. Pandis, V. N. Stathopoulos and M. Konsolakis, Catalysts, 2019, 9, 371, DOI:10.3390/catal9040371.
- M. Orfila, D. Sanz, M. Linares, R. Molina, R. Sanz, J. Marugán and J. Ángel Botas, Int. J. Hydrogen Energy, 2021, 46, 17458–17471, DOI:10.1016/j.ijhydene.2020.04.222.
- A. Onigbajumo, P. Swarnkar, G. Will, T. Sundararajan, A. Taghipour, S. Couperthwaite, T. Steinberg and T. Rainey, J. Cleaner Prod., 2022, 371, 133303, DOI:10.1016/j.jclepro.2022.133303.
- W. C. Chueh, C. Falter, M. Abbott, D. Scipio, P. Furler, S. M. Haile and A. Steinfeld, Science, 2010, 330, 1797–1801, DOI:10.1126/science.1197834.
- S. Ackermann, J. Scheffe and A. Steinfeld, J. Phys. Chem. C, 2014, 118, 5216–5225, DOI:10.1021/jp411717m.
- R. Grau-Crespo, N. H. de Leeuw, S. Hamad and U. V. Waghmare, Proc. R. Soc. A, 2011, 467, 1925–1938, DOI:10.1098/rspa.2010.0582.
- A. Le Gal, S. Abanades and G. Flamant, Energy Fuels, 2011, 25, 4836–4845 CrossRef CAS.
- S. Al-Taweel, M. A. Nadeem and H. Idriss, Energy Technol., 2022, 10, 2100491, DOI:10.1002/ente.202100491.
- Ki Hwan Ahn, D. S. Yoo, D. H. Prasad, H.-W. Lee, Y.-C. Chung and J.-H. Lee, Chem. Mater., 2012, 24, 4261–4267, DOI:10.1021/cm3023399.
- D. Arifin, A. Ambrosini, S. A. Wilson, B. Mandal, C. L. Muhich and A. W. Weimer, Int. J. Hydrogen Energy, 2020, 45, 160–174, DOI:10.1016/j.ijhydene.2019.10.177.
- H. Kaneko, T. Miura, H. Ishihara, S. Taku, T. Yokoyama, H. Nakajima and Y. Tamaura, Energy, 2007, 32, 656–663, DOI:10.1016/j.energy.2006.06.018.
- M. Zhang, D. Jiang and H. Jiang, Mater. Res. Bull., 2012, 47, 4006–4012, DOI:10.1016/j.materresbull.2012.07.012.
- T. Dhanni, S. Jayalekshmi, M. C. S. Kumar, T. P. Rao and A. C. Bose, J. Phys. Chem. Solids, 2010, 71, 1020–1025, DOI:10.1016/j.jpcs.2010.04.011.
- G. D. Takalkar, R. R. Bhosale, A. Kumar, F. AlMomani, M. Khraisheh, R. A. Shakoor and R. B. Gupta, Sol. Energy, 2018, 172, 204–211, DOI:10.1016/j.solener.2018.03.022.
- M. Strehle, X-Ray Photoelectron Spectroscopy (XPS) Study of Single Crystal UO2 and U3O8 on R-Plane and Y Stabilized Zirconium (YSZ) Substrates, MSc thesis, University of Illinois at Urbana-Champaign, 2011 Search PubMed.
- H. R. Hoekstra, S. Siegel and F. X. Gallagher, J. Inorg. Nucl. Chem., 1970, 32, 3237–3248, DOI:10.1016/0022-1902(70)80206-8.
- G. C. Allen and N. R. Holmes, J. Nucl. Mater., 1995, 223, 231–237, DOI:10.1016/0022-3115(95)00025-9.
- A. Magnéli, L. Kihlborg, H. Holtermann, J. S. Sörensen and N. A. Sörensen, Acta Chem. Scand., 1951, 5, 578–580, DOI:10.3891/acta.chem.scand.05-0578.
- H. Tagawa, T. Fujino, K. Watanabe, Y. Nakagawa and K. Saita, Bull. Chem. Soc. Jpn., 1981, 54, 138–142, DOI:10.1246/bcsj.54.138.
- K. Suresh Kumar, T. Mathews, H. P. Nawada and N. P. Bhat, J. Nucl. Mater., 2004, 324, 177–182, DOI:10.1016/j.jnucmat.2003.09.014.
- I. Al-Shankiti, F. Al-Otaibi, Y. Al-Salik and H. Idriss, Top. Catal., 2013, 56, 1129–1138, DOI:10.1007/s11244-013-0079-1.
- S. Halima Beguma, C.-T. Hunga, Y.-T. Chena, H. Shing-Jong, P.-H. Wu, X. Hand and L. Shang-Bin, J. Mol. Catal. A: Chem., 2016, 423, 423–432, DOI:10.1016/j.molcata.2016.07.036.
- K. I. Maslakov, Y. A. Teterin, A. J. Popel, A. Yu. Teterin, K. E. Ivanov, S. N. Kalmykov, V. G. Petrov, P. K. Petrov and I. Farnan, Appl. Surf. Sci., 2018, 448, 154–162 CrossRef CAS.
- K. I. Maslakov, Y. A. Teterin, M. V. Ryzhkov, A. J. Popel, A. Yu. Teterin, K. E. Ivanov, S. N. Kalmykov, V. G. Petrov, P. K. Petrov and I. Farnan, Phys. Chem. Chem. Phys., 2018, 20, 16167–16175, 10.1039/C8CP01442F.
- S. Eck, C. Castellarin-Cudia, S. Surnev, M. G. Ramsey and F. P. Netzer, Surf. Sci., 2002, 520, 173–185, DOI:10.1016/S0039-6028(02)02272-0.
- Y. A. Teterin, S. V. Stefanovskij, S. V. Yudintsev, G. N. Bek-Uzarov, A. Yu. Teterin, K. I. Maslakov and I. O. Utkin, Nucl. Technol. Radiat. Prot., 2004, 19, 31–38, DOI:10.2298/NTRP0401031T.
- G. S. Parkinson, Surf. Sci. Rep., 2016, 71, 272–365, DOI:10.1016/j.surfrep.2016.02.001.
- A. N. Mansour and R. A. Brizzolara, Surf. Sci. Spectra, 1996, 4, 345–350, DOI:10.1116/1.1247831.
- M. Preisinger, M. Krispin, T. Rudolf, S. Horn and D. R. Strongin, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 71, 165409, DOI:10.1103/PhysRevB.71.165409.
- T. Radu, C. Iacovita, D. Benea and R. Turcu, Appl. Surf. Sci., 2017, 405, 337–343, DOI:10.1016/j.apsusc.2017.02.002.
- A. P. Grosvenor, B. A. Kobe and N. S. McIntyre, Surf. Sci., 2004, 572, 217–227, DOI:10.1016/j.susc.2004.08.035.
- W. Temesghen and P. Sherwood, Anal. Bioanal. Chem., 2002, 373, 601–608, DOI:10.1007/s00216-002-1362-3.
- S. B. Paul, C. J. Nelin, C. R. Brundle, B. Vincent Crist, N. Lahiri and K. M. Ross, J. Chem. Phys., 2021, 154, 094709, DOI:10.1063/5.0039765.
- M. Fondell, M. Gorgoi, M. Boman and A. Lindblad, J. Electron Spectrosc. Relat. Phenom., 2018, 224, 23–26, DOI:10.1016/j.elspec.2017.09.008.
- T. Hasegawa, S. M. F. Shahed, Y. Sainoo, A. Beniya, N. Isomura, Y. Watanabe and T. Komeda, J. Chem. Phys., 2014, 140, 044711, DOI:10.1063/1.4849595.
- M. Sauerbrey, J. Höcker, M. Wellbrock, M. Schowalter, J.-O. Krisponeit, K. Müller-Caspary, A. Rosenauer, G. Wei, L. C. Ciacchi, J. Falta and J. I. Flege, Cryst. Growth Des., 2016, 16, 4216–4224, DOI:10.1021/acs.cgd.6b00192.
- J. I. Flege, J. Höcker, B. Kaemena, T. O. Menteş, A. Sala, A. Locatelli, S. Gangopadhyay, J. T. Sadowski, S. D. Senanayake and J. Falta, Nanoscale, 2016, 8, 10849–10856, 10.1039/C6NR02393B.
- D. C. Grinter and G. Thornton, J. Phys.: Condens. Matter, 2022, 34, 253001, DOI:10.1088/1361-648X/ac5d89.
- Y. Zhang, W. Feng, F. Yang and X. Bao, Chin. J. Catal., 2019, 40, 204–213, DOI:10.1016/S1872-2067(18)63171-7.
- G. El Jamal, T. Gouder, R. Eloirdi, H. Idriss and M. Jonsson, J. Phys. Chem. C, 2023, 127, 14222–14231, DOI:10.1021/acs.jpcc.3c02795.
- I. Arts, R. Saniz, G. Baldinozzi, G. Leinders, M. Verwerft and D. Lamoen, J. Nucl. Mater., 2024, 599, 155249, DOI:10.1016/j.jnucmat.2024.155249.
- F. Tao, M. E. Grass, Y. Zhang, D. R. Butcher, J. R. Renzas, Z. Liu, J. Y. Chung, B. S. Mun, M. Salmeron and G. A. Somorjai, Science, 2009, 322, 932–934, DOI:10.1126/science.1164170.
- A. Lidor, Y. Aschwanden, J. Häseli, P. Reckinger, P. Haueter and A. Steinfeld, Appl. Energy, 2023, 329(1–11), 120211, DOI:10.1016/j.apenergy.2022.120211.
- D. Arifin, A. Ambrosini, S. A. Wilson, B. Mandal, C. L. Muhich and A. W. Weimer, Int. J. Hydrogen Energy, 2020, 45, 160–174, DOI:10.1016/j.ijhydene.2019.10.177.
- D. R. Mullins, P. M. Albrecht, T.-L. Chen, F. C. Calaza, M. D. Biegalski, H. M. Christen and S. H. Overbury, J. Phys. Chem. C, 2012, 116, 19419–19428, DOI:10.1021/jp306444h.
- S. Fuente, M. M. Branda and F. Illas, Theor. Chem. Acc., 2012, 131, 1190, DOI:10.1007/s00214-012-1190-2.
| This journal is © The Royal Society of Chemistry 2026 |

