Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Hydrogen gas production and storage cycle with benzyl alcohol/benzaldehyde
Tomoyuki
Ito†
a,
Takumi
Ichimura†
a,
Hitoshi
Kasai
 a and
Kouki
Oka
a and
Kouki
Oka
 *abc
*abc
aInstitute of Multidisciplinary Research for Advanced Materials, Tohoku University, 2-1-1 Katahira, Aoba-ku, Sendai, Miyagi 980-8577, Japan. E-mail: oka@tohoku.ac.jp
bCarbon Recycling Energy Research Center Ibaraki University, 4-12-1 Nakanarusawa, Hitachi, Ibaraki 316-8511, Japan
cDeuterium Science Research Unit, Center for the Promotion of Interdisciplinary Education and Research Kyoto University, Yoshida, Sakyo-ku, Kyoto 606-8501, Japan
First published on 25th March 2026
Abstract
Hydrogen gas (H2) has attracted significant attention as a clean energy source. To realise a sustainable society, the development of methods for green H2 production from water and safe H2 storage is essential. Organic hydrides, which can store H2via covalent bonding under ambient conditions, are promising H2 storage materials. Primary alcohols, which can be produced from biomass and release H2 under mild conditions (80–220 °C) due to their small dehydrogenation enthalpies, have garnered attention as new candidates for application as organic hydrides. In this work, we demonstrated a reversible H2 storage cycle with primary alcohols/aldehydes. Specifically, we focused on benzyl alcohol and relatively stable benzaldehyde. Benzyl alcohol was completely dehydrogenated by warming (180 or 220 °C) in the presence of a metal complex catalyst to obtain benzaldehyde, which was then completely hydrogenated to benzyl alcohol using the same catalyst under H2 (1 atm), thereby accomplishing a reversible H2 storage cycle. Additionally, we used alcoholic fermentation with baker's yeast as a hydrogenation method without requiring precious metal catalysts or H2, and benzaldehyde was completely hydrogenated to store hydrogen directly from water and nicotinamide adenine dinucleotide (NADH). This work revealed the reversible H2 storage capability of primary alcohols/aldehydes as organic hydrides, and conceptually demonstrated a green H2 production and storage cycle by combining hydrogen storage using alcoholic fermentation and H2 release.
Introduction
Hydrogen gas (H2) is indispensable for industries (e.g., petroleum refining,1 steel manufacturing,2 and ammonia synthesis3). Additionally, H2 has attracted attention as a clean energy source alternative to fossil fuels4 due to its high gravimetric energy density (140.4 MJ kg−1),5 which is higher than that of coke (28.8 MJ kg−1) and gasoline (40.0 MJ kg−1), and its combustion (H2 + (1/2)O2 = H2O) is not accompanied by CO2 emissions.6 However, current H2 production methods mainly use fossil fuels, which in turn lead to CO2 emissions.7–9 Additionally, current H2 storage and transportation methods, such as compressed H2 (300–700 bar) and liquid hydrogen (−253 °C), require high pressure or cryogenic temperature and face safety risks (e.g., flammability and explosiveness).10,11 Therefore, for the practical use of hydrogen energy, the development of efficient methods for green H2 production from water and for safe H2 storage and transportation is required.12Porous materials (80 bar, −200 °C), hydrogen storage alloys (1–8 bar, −40–20 °C), and organic hydrides have been mainly reported as hydrogen storage materials.13,14 Among these, organic hydrides, which can store H2 reversibly via covalent bonding under ambient conditions, have attracted attention as safe H2 storage materials. Additionally, most organic hydrides exist as liquids under ambient conditions and are expected to enable hydrogen storage and transport by using the existing infrastructure for gasoline.15 From these advantages, organic hydrides are one of the promising hydrogen storage materials that enable relatively cost-effective and large-scale storage and transportation of hydrogen.16 However, most typical organic hydrides (e.g., toluene/methylcyclohexane) are produced from fossil resources,17 and they require high temperature conditions (e.g., >250 °C) for H2 release due to their large dehydrogenation enthalpies.18,19 These high temperature conditions consume 25–30% of the energy obtained from the released H2.20 Nitrogen-containing heterocyclic compounds have attracted attention due to their small dehydrogenation enthalpies, but their high melting points (e.g., N-ethylcarbazole: 69 °C)21 make them difficult to transport using existing gasoline infrastructure.22 Based on the above situation, the use of organic compounds, which have small dehydrogenation enthalpies and exist as liquids under ambient conditions, is desirable.20
As shown in Table 1, alcohols, which have small dehydrogenation enthalpies, can release H2 under milder conditions (80–220 °C)26–33 than those of typical organic hydrides and often exist as liquids under ambient conditions. Therefore, alcohols have recently attracted attention as new candidates for organic hydrides.34 Among them, primary alcohols can be easily produced from biomass35,36 and are expected to be used as sustainable and environmentally friendly organic hydrides.
| Entry | Organic hydride | Dehydrogenation enthalpy (kJ mol−1) | Hydrogen storage density (wt%) | Liquid range (°C) (H2 adduct/H2 acceptor) | Ref. |
|---|---|---|---|---|---|
| 1 |
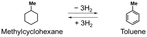
|
205 (68 per H2) | 6.2 | −127–101/−95–111 | 23 |
| 2 |
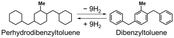
|
585 (65 per H2) | 6.2 | −58–371/−34–398 | 23 |
| 3 |

|
300 (50 per H2) | 5.2 | <20–280/68–270 | 23 |
| 4 (This work) |
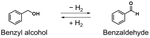
|
54 | 1.9 | −15–205/−56–179 | 24 |
| 5 |
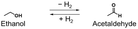
|
68 | 4.4 | −114–79/−123–20 | 25 |
| 6 |
|
— | 6.5 | −13–195/15–51 | — |
| 7 |
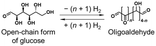
|
— | <5.6 | 146–NA/NA–NA | — |
In this work, we achieved reversible H2 storage using primary alcohols/aldehydes via hydrogenation and dehydrogenation reactions under mild conditions. Specifically, to conceptually demonstrate a reversible H2 storage cycle with primary alcohols/aldehydes, we focused on benzyl alcohol (H2 adduct), which can be produced from biomass,37 and benzaldehyde (H2 acceptor), which is relatively stable.38 Benzyl alcohol was completely dehydrogenated to benzaldehyde by simply warming it with an iridium catalyst.39 Subsequently, benzaldehyde was completely hydrogenated to benzyl alcohol with the same iridium catalyst under H2 (1 atm), thereby accomplishing a reversible H2 storage cycle with benzyl alcohol/benzaldehyde. Additionally, we used alcoholic fermentation with baker's yeast26 as a hydrogenation method without requiring precious metal catalysts or H2, and benzaldehyde was completely hydrogenated to store hydrogen directly from water and nicotinamide adenine dinucleotide (NADH) contained in baker's yeast. As shown in Fig. 1, this work conceptually demonstrated a green H2 production and storage cycle by combining hydrogen storage using alcoholic fermentation and H2 release.
Results and discussion
Reversible H2 storage cycle
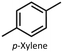
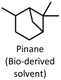
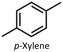
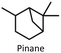
In this work, we used aqua(2,2′-bipyridine-6,6′-dionato)(penta-methylcyclopentadienyl)iridium(III) (Ir cat.) as the hydrogenation and dehydrogenation catalyst.39Ir cat. is known for dehydrogenating alcohols under mild conditions (<250 °C) without using oxidants in various solvents,26,39 and has a high durability and long lifetime with a catalytic turnover number exceeding 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. In addition, Ir cat. enables the reversible transformation between secondary alcohols and ketones (e.g., at least 8 cycles for 2-propanol/acetone39), and the technology for recovering iridium is mature.40 Therefore, Ir cat. also has reusability and recyclability. As shown in Fig. 1, benzyl alcohol was dehydrogenated with Ir cat. in p-xylene, which is often used in the dehydrogenation of alcohols by Ir cat.39 From the perspective of sustainability, the use of p-xylene is undesirable in the future, and therefore, p-xylene was used only for the analysis and demonstration of the dehydrogenation and hydrogenation of benzyl alcohol/benzaldehyde. In other parts, from the perspective of sustainability, pinane was used as an environmentally friendly solvent alternative to p-xylene.
000. In addition, Ir cat. enables the reversible transformation between secondary alcohols and ketones (e.g., at least 8 cycles for 2-propanol/acetone39), and the technology for recovering iridium is mature.40 Therefore, Ir cat. also has reusability and recyclability. As shown in Fig. 1, benzyl alcohol was dehydrogenated with Ir cat. in p-xylene, which is often used in the dehydrogenation of alcohols by Ir cat.39 From the perspective of sustainability, the use of p-xylene is undesirable in the future, and therefore, p-xylene was used only for the analysis and demonstration of the dehydrogenation and hydrogenation of benzyl alcohol/benzaldehyde. In other parts, from the perspective of sustainability, pinane was used as an environmentally friendly solvent alternative to p-xylene.
As shown in Table 2, Entry 1, and Fig. S1, benzyl alcohol was warmed in the presence of Ir cat. to be completely dehydrogenated to benzaldehyde. This full conversion was achieved by conducting the dehydrogenation reaction in an open system, where the evolved H2 that can serve as a reactant for the hydrogenation reaction is continuously released outside the system to proceed the dehydrogenation reaction only. Additionally, as shown in Fig. S2, benzyl alcohol was dehydrogenated in a sealed system, and the evolved gas was analysed by a gas chromatography (GC) analysis. Even in a sealed system, where the evolved H2 can remain, the endothermic nature of the dehydrogenation reaction can shift the equilibrium of the transformation between benzyl alcohol and benzaldehyde towards the production of benzaldehyde and H2 at high temperatures. As shown in Fig. S3 and S4, the dehydrogenation of benzyl alcohol produced benzaldehyde and H2 without any gaseous or liquid by-products. In addition, the amount of released H2 corresponding to the conversion of benzyl alcohol to benzaldehyde determined by 1H NMR spectroscopy was confirmed.
The dehydrogenation reaction of organic hydrides requires significant energy under high temperature conditions, making its kinetic analysis generally considered more important than that of the hydrogenation reaction.41,42 Therefore, as shown in Fig. 2, we conducted a kinetic analysis of the dehydrogenation reaction of benzyl alcohol with Ir cat. (details of the kinetic analysis and the reaction rate equations are shown in the SI). Based on the reaction rate constants summarised in Table S1 and the Arrhenius plot shown in Fig. 2b, the activation energy for the dehydrogenation of benzyl alcohol with Ir cat. was estimated to be +39.2 kJ mol−1. This activation energy is lower than the standard reaction enthalpy for dehydrogenation of benzyl alcohol to benzaldehyde and H2 (ΔH° = +53.9 kJ mol−1).24 This low activation energy is ascribed to the unique catalytic mechanism of Ir cat. (as shown in Fig. S5),43 and is expected to reduce the energy consumption for H2 release.
 | ||
| Fig. 2 (a) Dehydrogenation of benzyl alcohol with Ir cat. at 100–130 °C. (b) Arrhenius plots used to estimate the activation energy for the dehydrogenation of benzyl alcohol with Ir cat. | ||
As mentioned in the aforementioned part, p-xylene is produced from fossil resources,44 and therefore, the use of bio-derived solvents is desirable to develop sustainable H2 release methods. In this work, we replaced p-xylene with pinane, which is a green solvent derived from pine tree oil.45–47 As shown in Table S2, the dehydrogenation of benzyl alcohol in pinane was revealed to proceed more slowly than that in p-xylene (Fig. S6), and increasing the amount of Ir cat. achieved reaction rates and conversion comparable to those in p-xylene (Table 2, Entry 2 and Fig. S7). In our ongoing work, our objective is to make the H2 release method for primary alcohols more environmentally friendly by using non-precious, earth-abundant metal catalysts (e.g., manganese48).
Subsequently, as shown in Fig. 1, benzaldehyde was hydrogenated with Ir cat. in p-xylene or pinane under H2 (1 atm). As shown in Table 3 and Fig. S8 and S9, benzaldehyde was completely hydrogenated not only in p-xylene but also in pinane to produce benzyl alcohol and store H2. From the above results of hydrogenation and dehydrogenation, in this work, a reversible H2 storage cycle with benzyl alcohol/benzaldehyde is accomplished, and benzyl alcohol/benzaldehyde was conceptually demonstrated as an organic hydride (as shown in Fig. 1).
Green H2 production & storage cycle
As shown in Table 4, Entry 1, conventional H2 storage methods for organic hydrides require pure high-pressure H2, and the entire process of H2 production and storage (such as H2 production via water electrolysis, purification, and the hydrogenation of organic hydrides with high-pressure H2) consists of multiple steps and accompanied by energy consumption.49–51 Recently, as shown in Table 4, Entry 2, electrochemical hydrogenation has emerged as a simple method that can store hydrogen directly from water into organic hydrides without using H2.16 However, even in the electrochemical hydrogenation, precious metal catalysts are often required. Therefore, from the perspective of sustainability, the development of hydrogen storage methods without requiring precious metal catalysts is desirable.| Entry | Hydrogenation method | Processes | Catalyst | Hydrogen source | Temperature (°C) | Ref. |
|---|---|---|---|---|---|---|
| 1 | Conventional H2 storage methods | Multiple processes | Precious metal catalyst | H2 (>30 bar) | >120 | 17 |
| 2 | Electrochemical hydrogenation | Single process | Precious metal catalyst | H2O | 25 | 16 |
| 3 | Alcoholic fermentation | Single process | Baker's yeast | H2O and NADH | 25 | 26 |
In this context, as shown in Table 4, Entry 3, alcoholic fermentation with baker's yeast can hydrogenate aldehydes to primary alcohols using water and nicotinamide adenine dinucleotide (NADH), which is a coenzyme contained in baker's yeast, as hydrogen sources.26,52 During alcoholic fermentation, NADH is oxidised to NAD+, but NAD+ can be regenerated to NADH by adding glucose.53,54 Therefore, NADH can be used as a renewable hydrogen source. In addition, alcoholic fermentation with baker's yeast can hydrogenate aldehydes under ambient conditions, which are milder than those of the conventional hydrogenation methods (e.g., >120 °C). Furthermore, the hydrogen storage process based on alcoholic fermentation without using H2 does not require the conventional multiple H2 storage processes, which are energy-intensive. From the above, alcoholic fermentation with baker's yeast can be used as a novel environmentally friendly hydrogenation method for aldehydes, and the series of alcoholic fermentation and dehydrogenation processes is suggested to be more energy-efficient than that for conventional organic hydrides.
In this work, as shown in Fig. 1, we used alcoholic fermentation with baker's yeast as a hydrogenation method for benzaldehyde without requiring precious metal catalysts or H2. Coexistence of benzaldehyde with baker's yeast (1 g) in water allowed benzaldehyde to be completely hydrogenated and to store hydrogen directly from water and NADH after stirring for 6 h (Fig. S10 and Table 5, Entry 1). In addition, increasing the amount of baker's yeast to 3 g allowed the complete hydrogenation of benzaldehyde within 2 h (Fig. S11 and Table 5, Entry 2). These reaction times were comparable to or shorter than those required for the hydrogenation with Ir cat., indicating that alcoholic fermentation with baker's yeast has the potential to store hydrogen with a time scale and efficiency comparable to the hydrogenation with Ir cat. In addition, baker's yeast can be easily separated from the produced alcohols,26 enabling its combination with the subsequent dehydrogenation process without the deactivation of baker's yeast caused by the temperatures required for the dehydrogenation of alcohols. From the above results, we achieved direct hydrogen storage without requiring precious-metal catalysts or H2 by using alcoholic fermentation. As shown in Fig. 1, a green H2 production and storage cycle with benzyl alcohol/benzaldehyde was conceptually demonstrated by combining hydrogen storage using alcoholic fermentation and H2 release.
Conclusions
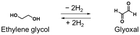
This work achieved the dehydrogenation of primary alcohols (e.g., benzyl alcohol) and hydrogenation of aldehydes (e.g., benzaldehyde) under H2 (1 atm) in pinane, a bio-derived solvent, and, for the first time, demonstrated that primary alcohols/aldehydes serve as organic hydrides. In addition, we used alcoholic fermentation with baker's yeast, in which water and NADH serve as the hydrogen sources, to achieve direct hydrogen storage without requiring precious metal catalysts or H2. Furthermore, a green H2 production and storage cycle with primary alcohols/aldehydes was conceptually demonstrated by combining alcoholic fermentation with the mild H2 release method. This cycle using primary alcohols/aldehydes can contribute to the development of an H2 release method under mild conditions and environmentally friendly methods for green H2 production and storage. In addition, as shown in Table 1, Entries 6 and 7, the use of ideal polyhydric primary alcohols with high hydrogen storage densities should lead to the development of promising organic hydrides with performance comparable to that of other representative candidates (e.g., methylcyclohexane/toluene), while having small dehydrogenation enthalpies. In our ongoing work, we comprehensively consider the characteristics (e.g., hydrogen storage density and volatility) for organic hydrides and aim to apply this green H2 production and storage cycle to more ideal primary alcohols/aldehydes (e.g., ethylene glycol/glyoxal) and to biomass (e.g., open-chain form of glucose).Author contributions
Tomoyuki Ito: methodology, formal analysis, data curation, investigation, validation, writing – original draft, writing – review & editing. Takumi Ichimura: methodology, formal analysis, data curation, investigation, writing – original draft, writing – review & editing, and funding acquisition. Hitoshi Kasai: writing – review &editing. Kouki Oka: conceptualization, methodology, formal analysis, data curation, investigation, project administration, supervision, writing – review & editing, funding acquisition, resources.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: materials and methods, 1H NMR spectra, gas chromatograms, mechanism for the dehydrogenation of alcohols with Ir cat., and tables. See DOI: https://doi.org/10.1039/d5se01721a.Acknowledgements
This work was partially supported by Grants-in-Aid for Scientific Research (JP23K17945, JP23H03827, JP24K01552, and JP25KJ0623) from MEXT, Japan. In addition, this work was partially supported by the Environment Research and Technology Development Fund (JPMEERF20241RA4) of the Environmental Restoration and Conservation Agency provided by the Ministry of the Environment of Japan. K. O. also acknowledges support from Shorai Foundation for Science and Technology, TEPCO Memorial Foundation, Amano Industry Technology Laboratory, the Yamada Science Foundation, Kenjiro Takayanagi Foundation, Kansai Research Foundation for Technology Promotion, Yashima Environment Technology Foundation, JACI Prize for Encouraging Young Researcher, Foundation for Interaction in Science and Technology, Iketani Science and Technology Foundation, and Ichimura Foundation for New Technology.References
- X. Shan, G. Shu, K. Li, X. Zhang, H. Wang, X. Cao, H. Jiang and H. Weng, Fuel, 2017, 194, 291–296 CrossRef CAS.
- M. Flores-Granobles and M. Saeys, Energy Environ. Sci., 2020, 13, 1923–1932 RSC.
- V. Madadi Avargani, S. Zendehboudi, N. M. Cata Saady and M. B. Dusseault, Energy Convers. Manage., 2022, 269, 115927 CrossRef CAS.
- T. Zhang, J. Uratani, Y. Huang, L. Xu, S. Griffiths and Y. Ding, Renewable Sustainable Energy Rev., 2023, 176, 113204 CrossRef CAS.
- X. Xu, Q. Zhou and D. Yu, Int. J. Hydrogen Energy, 2022, 47, 33677–33698 CrossRef CAS.
- M. G. Rasul, M. A. Hazrat, M. A. Sattar, M. I. Jahirul and M. J. Shearer, Energy Convers. Manage., 2022, 272, 116326 CrossRef CAS.
- L. Mosca, J. A. Medrano Jimenez, S. A. Wassie, F. Gallucci, E. Palo, M. Colozzi, S. Taraschi and G. Galdieri, Int. J. Hydrogen Energy, 2020, 45, 7266–7277 CrossRef CAS.
- J. Incer-Valverde, A. Korayem, G. Tsatsaronis and T. Morosuk, Energy Convers. Manage., 2023, 291, 117294 CrossRef CAS.
- K. Oka, B. Winther-Jensen and H. Nishide, Adv. Energy Mater., 2021, 11, 2003724 CrossRef CAS.
- H. Li, X. Cao, Y. Liu, Y. Shao, Z. Nan, L. Teng, W. Peng and J. Bian, Energy Rep., 2022, 8, 6258–6269 CrossRef.
- M. R. Usman, Renewable Sustainable Energy Rev., 2022, 167, 112743 CrossRef CAS.
- E. Rivard, M. Trudeau and K. Zaghib, Materials, 2019, 12, 1973 Search PubMed.
- G. Nazir, A. Rehman, S. Hussain, S. Aftab, K. Heo, M. Ikram, S. A. Patil and M. Aizaz Ud Din, Adv. Sustainable Syst., 2022, 6, 2200276 CrossRef CAS.
- D. Zhao, X. Wang, L. Yue, Y. He and B. Chen, Chem. Commun., 2022, 58, 11059–11078 RSC.
- T. Shimbayashi and K.-I. Fujita, Tetrahedron, 2020, 76, 130946 Search PubMed.
- T. Ichimura, H. Kasai and K. Oka, Energy, 2025, 3, 100028 Search PubMed.
- C. Chu, K. Wu, B. Luo, Q. Cao and H. Zhang, Carbon Resour. Convers., 2023, 6, 334–351 CAS.
- R. Tan, Q. Ji, Y. Ling and L. Li, Chem. Commun., 2024, 60, 8186–8203 RSC.
- E. Spatolisano, F. Restelli, A. Matichecchia, L. A. Pellegrini, A. R. de Angelis, S. Cattaneo and E. Roccaro, Int. J. Hydrogen Energy, 2024, 52, 703–717 CrossRef CAS.
- A. Lin and G. Bagnato, Int. J. Hydrogen Energy, 2024, 63, 315–329 CrossRef CAS.
- E. Díaz, P. Rapado-Gallego and S. Ordóñez, J. Energy Storage, 2023, 59, 106511 CrossRef.
- T. He, Q. Pei and P. Chen, J. Energy Chem., 2015, 24, 587–594 CrossRef.
- M.-J. Zhou, Y. Miao, Y. Gu and Y. Xie, Adv. Mater., 2024, 36, 2311355 CrossRef CAS PubMed.
- S. P. Verevkin and T. V. Vasiltsova, J. Chem. Eng. Data, 2004, 49, 1717–1723 CrossRef CAS.
- A. Kumar, Ind. Eng. Chem. Res., 2021, 60, 16561–16576 CrossRef CAS.
- T. Ichimura, H. Maeda, T. Shimbayashi, K. Okubo, M. Fukushima, N. Tohnai, K.-I. Fujita and K. Oka, Int. J. Hydrogen Energy, 2024, 72, 815–819 CrossRef CAS.
- R. Kato, K. Oka, K. Yoshimasa, M. Nakajima, H. Nishide and K. Oyaizu, Macromol. Rapid Commun., 2019, 40, 1900139 CrossRef PubMed.
- R. Kato, K. Yoshimasa, T. Egashira, T. Oya, K. Oyaizu and H. Nishide, Nat. Commun., 2016, 7, 13032 CrossRef CAS PubMed.
- K. Oka, M. Kataoka, Y. Kaiwa and K. Oyaizu, Bull. Chem. Soc. Jpn., 2021, 94, 2770–2773 CrossRef CAS.
- K. Oka, Y. Tobita, M. Kataoka, S. Murao, K. Kobayashi, S. Furukawa, H. Nishide and K. Oyaizu, Polym. J., 2021, 53, 799–804 Search PubMed.
- K. Oka, Y. Tobita, M. Kataoka, K. Kobayashi, Y. Kaiwa, H. Nishide and K. Oyaizu, Polym. Int., 2022, 71, 348–351 Search PubMed.
- T. Ichimura, H. Kasai and K. Oka, ChemPlusChem, 2025, 90, e202400639 Search PubMed.
- K. Oka, Y. Kaiwa, M. Kataoka, K.-I. Fujita and K. Oyaizu, Eur. J. Org Chem., 2020, 2020, 5876–5879 Search PubMed.
- R. H. Crabtree, Chem. Rev., 2017, 117, 9228–9246 CrossRef CAS PubMed.
- S. Saini, A. K. Chandel and K. K. Sharma, J. Cleaner Prod., 2020, 268, 122357 Search PubMed.
- A. Aui, Y. Wang and M. Mba-Wright, Renewable Sustainable Energy Rev., 2021, 145, 111098 CrossRef CAS.
- S. Pugh, R. McKenna, I. Halloum and D. R. Nielsen, Metab. Eng. Commun., 2015, 2, 39–45 CrossRef PubMed.
- S. H. Hilal, L. L. Bornander and L. A. Carreira, QSAR Comb. Sci., 2005, 24, 631–638 CrossRef CAS.
- R. Kawahara, K.-I. Fujita and R. Yamaguchi, Angew. Chem., Int. Ed., 2012, 51, 12790–12794 CrossRef CAS PubMed.
- C. Fan, K. Quan, Z. Han, F. Han, Z. Li, J. Liu and X. Liu, J. Sustain. Metall., 2023, 9, 909–926 Search PubMed.
- I. Rey, V. L. Barrio and I. Agirre, Int. J. Hydrogen Energy, 2025, 142, 616–626 CrossRef CAS.
- Z. Feng, X. Chen and X. Bai, Environ. Sci. Pollut. Res., 2020, 27, 36172–36185 CrossRef CAS PubMed.
- G. Zeng, S. Sakaki, K.-I. Fujita, H. Sano and R. Yamaguchi, ACS Catal., 2014, 4, 1010–1020 CrossRef CAS.
- N. Chakinala and A. G. Chakinala, Ind. Eng. Chem. Res., 2021, 60, 5331–5351 Search PubMed.
- M. Allenspach and C. Steuer, Phytochemistry, 2021, 190, 112857 CrossRef CAS PubMed.
- L. Xie, X. Wang, F. Yu, B. Yuan, C. Xie and S. Yu, RSC Adv., 2017, 7, 51452–51459 RSC.
- P. Wu, L. Zhang, S. Zhao, C. Wang, Y. Guo and W. Fang, J. Chem. Eng. Data, 2021, 66, 2706–2716 CrossRef CAS.
- Y. Wang, M. Wang, Y. Li and Q. Liu, Chem, 2021, 7, 1180–1223 CAS.
- P. M. Modisha, C. N. M. Ouma, R. Garidzirai, P. Wasserscheid and D. Bessarabov, Energy Fuels, 2019, 33, 2778–2796 Search PubMed.
- G. Bernardo, T. Araújo, T. da Silva Lopes, J. Sousa and A. Mendes, Int. J. Hydrogen Energy, 2020, 45, 7313–7338 CrossRef CAS.
- Z. Abdin, C. Tang, Y. Liu and K. Catchpole, iScience, 2021, 24, 102966 Search PubMed.
- R. Csuk and B. I. Glaenzer, Chem. Rev., 1991, 91, 49–97 CrossRef CAS.
- N. Xie, L. Zhang, W. Gao, C. Huang, P. E. Huber, X. Zhou, C. Li, G. Shen and B. Zou, Signal Transduct. Targeted Ther., 2020, 5, 227 CrossRef CAS PubMed.
- M. Benoit, A. Rodrigues, Q. Zhang, E. Fourré, K. De Oliveira Vigier, J.-M. Tatibouët and F. Jérôme, Angew. Chem., Int. Ed., 2011, 50, 8964–8967 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |