 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synergistic plasmonic–semiconductor heterointerfaces enabling efficient CO2 hydrogenation to methanol under visible-light irradiation
Abdul
Malek
ac,
Anh-Tuan
Hoang
d,
Md. Tarekul
Islam
e,
Mohammad A.
Hasnat
f,
Tarikul
Islam
 *g and
Aminul
Islam
*g and
Aminul
Islam
 *abc
*abc
aDepartment of Petroleum and Mining Engineering, Jashore University of Science and Technology, Jashore 7408, Bangladesh. E-mail: aminul_pme@just.edu.bd
bCollege of Engineering, Korea University, 145 Anam-ro, Seongbuk-gu, Seoul 02841, Republic of Korea
cHydrogen Energy and CO2 Conversion Lab, Department of Petroleum and Mining Engineering, Jashore University of Science and Technology, Jashore 7408, Bangladesh
dEnergy-Fuel Technology and Applied Material Research Group, Dong Nai Technology University, Dong Nai, Vietnam
eDepartment of Leather Engineering, Faculty of Mechanical Engineering, Khulna University of Engineering and Technology, Khulna 9203, Bangladesh
fDepartment of Chemistry, School of Physical Sciences, Shahjalal University of Science and Technology, Sylhet 3114, Bangladesh
gDepartment of Textiles, Merchandising, and Interiors, University of Georgia, Athens, Georgia 30602, USA. E-mail: tarikul@uga.edu
First published on 8th January 2026
Abstract
The photocatalytic conversion of carbon-dioxide (CO2) to methanol (CH3OH) under mild conditions has been regarded as a promising, cost-effective, and environmentally sustainable approach for carbon utilization and renewable fuel generation. However, the process has been hindered by limited charge separation efficiency and insufficient CO2 activation. In this study, a heterostructured Ag–Si/MgO/ZnO photocatalyst was rationally designed and synthesized via a solid-phase reaction method. A CH3OH production rate of 357.53 µmol gcat−1 h−1 was achieved over the optimized 10% Ag–Si/MgO/ZnO composite catalyst at 250 °C, representing a substantial enhancement compared to the Si/ZnO and Si/MgO/ZnO photocatalysts. The CH3OH production performance was found to be higher in the photocatalyst/gas-phase system than that reported in comparable studies. The theoretical activation energy for Ag–Si/MgO/ZnO was found to be 158.14 kJ mol−1, which is lower than that of Si/MgO/ZnO (167.79 kJ mol−1) and Si/ZnO (177.97 kJ mol−1), indicating enhanced CO2 activation and higher CO2 conversion. More importantly, after more than 72 h of irradiation, the system still exhibited a high CH3OH production rate, demonstrating its potential for practical application.
1. Introduction
The average concentration of CO2 has jumped to 427.48 ppm in the atmosphere currently,1 although the concentration level of CO2 in the atmosphere shouldn't be higher than 350 ppm. The sharp increase in concentration of CO2 in the atmosphere is the main driver of the greenhouse effect which causes global warming and climate change, gravely affecting the ability of human beings to live normally. Therefore, limiting the release of CO2 is a significant task for humankind in an attempt to mitigate global warming. The production of liquid fuels and value-added chemicals via CO2 hydrogenation has recently gained significant attention due to its promising economic potential and environmental advantages.2–4 Various value-added chemicals including CH3OH, CH4, CH3CH2OH, olefins, DME etc. are obtained from hydrogenation of CO2.5,6 Among different products, CH3OH has extensive applications as a vital organic solvent. It is an essential organic feedstock for organic synthesis, including the production of HCHO, C3H6O3, C2H4, CH3COOH, and other substances. It is also used as a hydrogen carrier, an energy source for fuel cells, and a feedstock for gasoline production.7,8 Consequently, the process of hydrogenation of CO2 into CH3OH utilizing green H2 has garnered a lot of interest recently.9,10CO2 hydrogenation to CH3OH had been extensively studied. For instance, new active sites were introduced through nitrogen doping by Yang et al.,11 which resulted in significant enhancement of reaction activity and catalytic stability of In2O3. Using an In2O3 nano-catalyst, a CH3OH selectivity of 62.3% was achieved at 300 °C and 5 MPa. The ZrO2/Cu photocatalyst was reported to have exhibited strong activity for CH3OH formation. The hydrogenation of surface oxygenate intermediates and the activation of CO2 were found to have been significantly enhanced over these configurations. In Cu-based catalysts, the promotion of CH3OH synthesis was observed when Zn species were incorporated. The presence of Zn species was found to have increased the dispersion of Cu particles and prevented their agglomeration. As a result, a high copper surface area was maintained on the catalyst, which was considered to have improved its performance in CH3OH synthesis. Moreover, the incorporation of ZnO was observed to have strengthened the resistance of Cu particles to poisoning, improved CO2 adsorption on the catalyst surface, and provided a higher density of catalytically active sites for the reactants.
Single-component photocatalysts had typically been limited by poor visible-light utilization, fast electron–hole recombination, and unfavorable adsorption energetics for key CO2-reduction intermediates, whereas complementary metals had been shown to adjust charge-transfer pathways and intermediate binding in a controllable manner.12 In bimetallic systems, plasmonic nanoparticles had been expected to generate localized surface plasmon resonance under visible light. As a result, hot-carrier generation and near-field enhancement had been exploited to strengthen light excitation and accelerate electron injection into nearby semiconductor or oxide domains, as widely reported for plasmon-assisted CO2 photoreduction.13 Meanwhile, semiconductor domains had been introduced to tune the interfacial electronic structure and adsorption behavior. This approach had allowed early oxygenated intermediates to be stabilized on oxide sites, while hydrogenation-favored steps had been promoted on plasmonic metal sites, in agreement with the principle that bimetallic interfaces enabled spatial separation of adsorption/activation and hydrogenation functions.14 This mechanism had been expected to enhance both activity and selectivity by increasing the supply of reactive electrons from plasmonic components and by optimizing intermediate binding while suppressing unproductive pathways through interfacial site differentiation.15
Elemental Si, a visible-light-responsive semiconductor with a narrow indirect bandgap of ∼1.1 eV, enables broad solar absorption and efficient generation of charge carriers under light irradiation, making it highly suitable for solar-driven catalytic applications.16 When coupled with plasmonic components, Si primarily functions as an electron reservoir and transport medium, facilitating the directional migration of photogenerated or plasmon-induced hot electrons toward catalytically active surface sites. This process promotes interfacial charge separation and effectively suppresses electron–hole recombination through plasmonic–semiconductor coupling and Schottky junction formation.17 Similar Si-based photocatalytic architectures have been reported to enhance visible-light utilization, charge separation efficiency, and CO2 reduction performance through plasmonic–semiconductor coupling effects.18,19 Plasmonic–semiconductor photocatalysts can utilize up to ∼45% of the solar spectrum, significantly improving photon-to-chemical conversion efficiency and reducing external heating energy input.20 By coupling localized surface plasmon resonance (LSPR)–induced hot-electron generation with semiconductor charge transport, the system drives CO2 reduction under mild conditions, thus lowering operational energy requirements per unit methanol produced. From a techno-economic perspective, visible-light-assisted pathways may become cost-competitive as renewable H2 prices continue to decline and scalable synthesis routes for plasmonic materials (e.g., Ag and Cu) are further optimized.21 Recent analyses suggest that photocatalytic methanol production can be economically favorable when solar-to-fuel efficiencies exceed 5–8% and catalyst stability surpasses several thousand hours.22 Although further development is needed, the combination of low-temperature operation and reduced energy intensity, and the prospect of using abundant, sunlight-driven excitation underscore the long-term economic viability of plasmonic–semiconductor photocatalysts for sustainable CO2 valorization.
Visible-light-driven CH3OH synthesis over plasmonic Cu/ZnO catalysts, achieving a CH3OH production rate of 127.8 µmol g−1 h−1, was reported by Wang and co-workers.23 It was found that, despite these advancements, copper-based catalysts were still prone to favor the reverse water–gas shift (RWGS) reaction. This undesired pathway was identified as a major limitation to the selective hydrogenation of CO2 into CH3OH and other value-added products when thermal heterogeneous catalysts were employed. To mitigate sintering and enhance catalyst stability, various promoters such as Al2O3, MgO, SiO2, CeO2, and ZrO2 were commonly incorporated.24 Among these, MgO was recognized as a promising promoter owing to its unique physicochemical properties. MgO was reported to inhibit the RWGS reaction and function as an adsorbent carrier with desirable features such as large surface area, high density of adsorption sites, tunable surface properties, and high chemical reactivity. Mg-based catalysts were reported to exhibit excellent performance for CO2 hydrogenation to hydrocarbons. The incorporation of MgO as a catalyst component led to enhanced CO2 adsorption due to its basic nature and improved metal dispersion by increasing surface area as a result of its low density. Few studies had been conducted on MgO-based catalysts for CO2 hydrogenation to CH3OH, and MgO was generally combined with other catalytic components to improve overall performance. For example, the influence of Cu/MgO/Al2O3 catalysts was investigated by Dasireddy et al.,25 and the Cu/MgO/Al2O3 sample was reported to display the highest activity, which was attributed to the increased number of active sites for CO2 and H2 adsorption provided by MgO. Using a Cu–ZnO/MgO catalyst, Guo et al. achieved a CO2 conversion of 7.6% and a CH3OH selectivity of 91% at 200 °C and 5 MPa.26 These findings demonstrated that high CH3OH selectivity could be achieved through conventional heterogeneous catalysis. Nevertheless, because of the inherent thermodynamic stability of CO2, its hydrogenation to CH3OH was typically carried out under relatively harsh conditions, requiring elevated temperatures (>200 °C) and high pressures (>4 MPa).
Doping was adopted as a standard modification strategy and was increasingly focused upon by researchers. Among the various modification approaches applied to ZnO or MgO carriers, the incorporation of metals was predominantly employed. Noble metals were considered effective cocatalysts for charge carrier separation, as they were believed to act as electron sinks and solid-state mediators. Catalysts based on noble metals such as Pd, Pt, and Au were reported to exhibit excellent hydrogen spillover capacity, high stability, and strong anti-sintering ability in the CO2-to-CH3OH reaction.27 For instance, oxygen vacancies were introduced on the surface of Pd/In2O3 catalysts by Men et al.,28 resulting in a CH3OH yield of 8.9% with a CO2 conversion of 24.5%. Similarly, a 2 wt% Pd/CeO2 catalyst was reported by Pothu et al. to have achieved a CO2 conversion of 49.6% and a CH3OH selectivity of 69.5%.29 In another study, CO2 hydrogenation with H2 over a Pt/film/In2O3 catalyst was reported by Men and co-workers,28 achieving a CO2 conversion of 37.0% and a CH3OH selectivity of 62.6%. Sun et al. confirmed,30 both theoretically and experimentally, the feasibility of selective CO2 hydrogenation to CH3OH over Ag/In2O3. Under reaction conditions of 300 °C and 5 MPa, a CH3OH selectivity of 58.2% was maintained with a CO2 conversion of 13.6% and a space–time yield (STY) of 0.453 gmethanol g−1 h−1. The appropriate selection of a metal for doping into the metal oxide catalyst was reported to enhance interfacial charge transfer, thereby significantly improving CO2 adsorption. Although plasmonic–semiconductor hybrid systems have been investigated under photothermal conditions, where thermal effects predominantly govern the catalytic activity,31–34 studies focusing on photocatalytic CO2 conversion driven by plasmon-induced charge separation particularly in silicon-based, cost-effective semiconductor platforms remain limited.
In this study, a plasmonic Ag–Si/MgO/ZnO composite photocatalyst was synthesized via a solid-phase reaction method, and its photocatalytic performance was evaluated for CO2 conversion under visible-light irradiation. Bimetallic Ag and Si species were introduced as cocatalysts supported on an MgO promoter to enhance charge separation efficiency and overall catalytic activity of the Ag–Si/MgO/ZnO system. The photocatalytic performance was systematically investigated in terms of product yield and apparent quantum yield (AQY). Furthermore, surface activation, structural evolution, and catalyst stability under extended illumination were analyzed using a suite of advanced characterization techniques. The findings reveal that the engineered heterostructure exhibits excellent stability, high photocatalytic efficiency, and strong scalability potential, offering valuable insights into the rational design of next-generation plasmonic–semiconductor systems for solar-driven CO2 reduction.
2. Materials and methods
2.1 Materials
Mg powder (99%), Zn powder (99%), silver nitrate (AgNO3), tri-sodium citrate dehydrate (C6H5Na3O7·2H2O), and sodium borohydride (NaBH4) were purchased from Sigma-Aldrich (USA). Ultra-pure water was supplied through distillation of water. CO2 (99.99%) and N2 (99.99%) were collected from Spectra International Limited, Bangladesh. Water is electrolyzed to deliver renewable H2 (99.99%). The electrolyte, Na2SO4 (99.99%) utilized to electrolyze water, was purchased from Sigma-Aldrich (USA). All chemicals used in this work were employed without any additional treatment.2.2 Synthesis of SiO2
Through the calcination procedure, waste rice husk was converted into a precursor silica (SiO2) nanopowder. The wasted rice husk was first acid purified by immersing it in a 1000 mL aqueous solution of 20% HCl in a beaker and heating it to 100 °C for six hours. After being cleaned three times with pure water, the deep yellow rice husk was taken out from the beaker. The cleaned rice husk was then dried in an oven for 12 hours at 90 °C. To calcine the sample, the dried rice husk was placed in a tube furnace with the aid of a ceramic crucible and heated to 800 °C for two hours at a ramping rate of 10 °C min−1. The bright white SiO2 powder was collected when it had reached room temperature.2.3 Synthesis of Si/MgO/ZnO
To develop a semiconductor-based photocatalyst, the solid combustion approach was utilized in a specifically developed sealed stainless steel reactor. Employing a single-step magneto-thermic reduction technique, a Si/MgO/ZnO composite was developed in an enclosed reactor. Initially, 1 g of the obtained SiO2 nanopowder was finely ground with 0.65 g of Zn powder and 0.65 g of Mg powder by with the proportions of SiO2, Zn, and Mg being 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.65
0.65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.65. Then, the sealed stainless steel reactor was filled with the ground mixture of SiO2, Zn, and Mg powders. To execute the magneto-thermic conversion, the enclosed stainless steel reactor was placed within a muffle furnace and heated to 800 °C for 6 h, increasing the temperature by 5 °C per minute. The reactor was removed from the furnace once it reached room temperature. The built Si/MgO/ZnO material was removed from the reactor and finely powdered.
0.65. Then, the sealed stainless steel reactor was filled with the ground mixture of SiO2, Zn, and Mg powders. To execute the magneto-thermic conversion, the enclosed stainless steel reactor was placed within a muffle furnace and heated to 800 °C for 6 h, increasing the temperature by 5 °C per minute. The reactor was removed from the furnace once it reached room temperature. The built Si/MgO/ZnO material was removed from the reactor and finely powdered.
2.4 Synthesis of Ag nanoparticles (NPs)
Ag nanoparticles (Ag NPs) were synthesized via a chemical reduction method employing tri-sodium citrate (C6H5Na3O7·2H2O) and sodium borohydride (NaBH4) as reducing agents. Silver nitrate (AgNO3) and Na3C6H5O7 were used as precursors, and all solutions were freshly prepared using distilled water. In a typical procedure, 100 mL of 1 M Na3C6H5O7 solution was magnetically stirred, after which 100 mL of 0.25 mM AgNO3 solution was added dropwise under continuous stirring until a clear and homogeneous solution was obtained. Subsequently, 250 mL of 148 mM NaBH4 solution was introduced dropwise at a controlled rate of approximately one drop per second. The reaction mixture was stirred vigorously for 6 h, during which a gradual color transition from light yellow to dark yellow and finally to gray was observed, indicating nanoparticle formation and subsequent colloid destabilization. After completion of the reaction, stirring was stopped, and the solution was left undisturbed overnight to allow complete precipitation of the nanoparticles. The resulting Ag nanoparticles had been collected as a solid residue, repeatedly washed with distilled water to remove impurities, and subsequently subjected to high-temperature treatment (850 °C for 6 h) during photocatalyst preparation. Therefore, any residual species would have been expected to be removed through the post calcination process.2.5 Synthesis of Ag–Si/MgO/ZnO
With the aid of a hydraulic pressure machine, the fine Ag–Si/MgO/ZnO mixture powder was further sealed in a cylindrical stainless steel reactor under elevated pressures. Attempting to build a solid Ag–Si/MgO/ZnO structure, the reactor was then placed inside a muffle furnace and heated to 850 °C for 6 h, increasing the temperature by 5 °C per minute. When the reactor reached room temperature, it was removed from the furnace. Employing a hydraulic pressure machine, the Ag–Si/MgO/ZnO specimen with a solid structure was removed from the reactor. After the surface of the formed solid Ag–Si/MgO/ZnO specimen was cleaned with emery paper, it was heated in a muffle furnace for two hours at a temperature of 5 °C per minute. The solid-structured Ag–Si/MgO/ZnO composite was subsequently removed from the furnace and split it into several pieces to be used as a photocatalyst in a continuous flow reactor once it had reached room temperature. To assess the photocatalytic efficiency, additional Si/MgO/ZnO and Si/ZnO catalysts were developed without Ag or Ag/MgO, respectively. The overall synthesis process of the Ag–Si/MgO/ZnO photocatalyst is presented in Fig. S1.2.6 Characterization
The microstructure and morphology of the prepared materials were characterized by scanning electron microscopy (SEM, Sigma-300, Germany), while elemental composition and mapping were determined by energy-dispersive spectroscopy (EDS). Transmission electron microscopy (TEM) and high-resolution TEM (HRTEM) images with elemental mapping were obtained using FEI Talos F200X and Tecnai F20 instruments. The crystalline phases were analyzed by X-ray diffraction (XRD, Bruker AXS, Germany) using Cu Kα radiation (λ = 1.5418 Å). Optical absorbance and diffuse reflectance spectra were recorded using a Cary 5000 UV-vis spectrophotometer (Varian, USA) and a Thermo Scientific Evolution 220 UV-vis DRS instrument, respectively. Surface composition and chemical states were examined by X-ray photoelectron spectroscopy (XPS, ESCALAB 250Xi, Thermo Fisher, UK), and photoluminescence (PL) spectra of the solid powders were measured with a Hitachi F-7000 fluorescence spectrophotometer (Japan).2.7 Photo-electrochemical tests
A CorrTest electrochemical workstation (Wuhan Corrtest Instruments Comp. Ltd, China) was utilized to measure the transient photocurrent response of all samples. The electrochemical impedance spectroscopy and Mott–Schottky plots of catalyst specimens were performed on an Autolab workstation (Netherlands). A platinum wire served as the counter electrode. An Ag/AgCl electrode was used as the reference electrode. A conventional quartz cell with three electrodes was employed in both the electrochemical systems. A solution of 0.5 M Na2SO4 was used as the electrolyte. The series of photocatalyst films were coated on fluorine-tin-oxide (FTO) glasses of 1 cm2 area and used as a working electrode. The working electrodes were prepared using the doctor blade method. In short, before preparing the solution, the polyvinylidene difluoride (PVDF) binder was dried over an entire night. N-Methyl-2-pyrrolidone (NMP) solvent was utilized to combine 90% active material (photocatalyst) and 10% binder agent (PVDF) to prepare a paste. Employing a doctor blade approach, the as-prepared paste was placed on the functionalized surface of the FTO glass. The decorated FTO was placed in an oven for 24 hours at 60 °C for drying properly. A tungsten lamp (100 W) was utilized to illuminate the prepared samples. N2 gas was used to purge the electrolyte for 30 minutes prior to the tests. In a 0.5 M Na2SO4 solution, electrochemical impedance spectroscopy (EIS) was conducted at a frequency ranging from 0.1 Hz to 1000 Hz. In each instance, a sinusoidal potential amplitude of 5 mV was applied. Mott–Schottky plots were obtained utilizing the same setup (applied voltage: −0.5 V, frequency: 1000 Hz, AC voltage: 5 mV amplitude). Over the course of on–off cycle, the transient photocurrent responses were measured at 0 V potential bias versus Ag/AgCl.2.8 Photocatalytic performance test
The schematic diagram of the experimental set up for photocatalytic CO2 hydrogenation to CH3OH under visible light is presented in Fig. S2. Utilizing the prepared catalyst, a cylindrical fixed-bed continuous-flow quartz-glass reactor was employed to measure the hydrogenation of CO2 to CH3OH. The reactor is made up of a 130 mm length and 8 mm diameter column. 1 g of photocatalyst (40–50 mesh) was installed in the middle part of the column almost 10 mm in length. By filling both sides of the photocatalyst bed with quartz wool, the catalyst bed remained fixed. The reactor was installed in a cylindrical glass furnace 4 cm away from the light source and linked to the gas inlet and outlet. An electric heater is fitted into the furnace to control the temperature of the reactor. A thermocouple is fitted in the furnace to measure the inside temperature of the furnace. A mass flow controller was employed to control the flow rate of the reactant gas mixture. For maintaining a specified pressure, a back-pressure controller was integrated into the gas line which was connected to the outlet. The reaction was carried out at atmospheric pressure under specific reaction conditions, with an N2 flow, at temperatures up to 300 °C. After 3 h, H2 (generated by electrolysis) and CO2 (99.999%) were provided throughout the reaction. In all experiments using the reaction mixture, we used the ratio H2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CO2 = 3
CO2 = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 that reflects a stoichiometric equation for the CH3OH synthesis reaction (CO2 + 3H2 → CH3OH + H2O) and has been commonly used in related prior studies. The kinetic study on the reaction was carried out between 200 °C and 300 °C. The liquid phase products (CH3OH) resulting from photocatalytic CO2 reduction were collected in a cold trap and subsequently analyzed on a gas chromatograph (GC-2014, SHIMADZU) equipped with a flame ionization detector (FID). The production rate of CH3OH in the hydrogenation of CO2 was determined by using the following eqn (1):35
1 that reflects a stoichiometric equation for the CH3OH synthesis reaction (CO2 + 3H2 → CH3OH + H2O) and has been commonly used in related prior studies. The kinetic study on the reaction was carried out between 200 °C and 300 °C. The liquid phase products (CH3OH) resulting from photocatalytic CO2 reduction were collected in a cold trap and subsequently analyzed on a gas chromatograph (GC-2014, SHIMADZU) equipped with a flame ionization detector (FID). The production rate of CH3OH in the hydrogenation of CO2 was determined by using the following eqn (1):35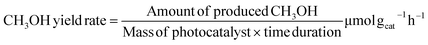 | (1) |
The apparent quantum yield of the as-prepared Ag–Si/MgO/ZnO photocatalyst for CH3OH production was evaluated by using the following eqn (2).35
Apparent quantum yield for CH3OH,
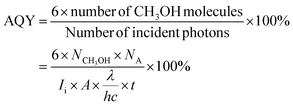 | (2) |
2.9 Computational methods
The distribution of the electro-magnetic (EM) field over Si/ZnO, Si/MgO, Si/ZnO/MgO, and Ag–Si/ZnO/MgO is conducted using the finite-difference time-domain (FDTD) method. 630 nm is used as the simulated excitation wavelength of all nano-structures. Ag–Si/ZnO/MgO is modeled as follows: isolated Si, Si/MgO and Ag–Si/MgO NPs assemblies are placed on the surface of ZnO nanosheets, respectively. A plane wave propagating in the z-direction with an electric field parallel to the x-direction was selected to estimate the electric field distribution. The dielectric functions of Ag, Si, MgO, and ZnO were taken from the literature. Density functional theory (DFT) calculations were carried out using the DMol3 module within the Materials Studio software package. The interaction between ionic cores and valence electrons was modeled using the projector augmented-wave (PAW) formalism. The plane-wave basis set was constructed with a kinetic energy cutoff of 500 eV to ensure adequate convergence. Exchange–correlation effects were accounted for within the framework of the generalized gradient approximation (GGA), employing the Perdew–Burke–Ernzerhof (PBE) functional. Brillouin zone integrations were performed using Monkhorst–Pack k-point meshes. For the slab models considered, a final k-point mesh of X × Y × 1 was adopted, where X and Y were determined from the convergence tests. A single k-point was used along the surface normal due to the large vacuum spacing. Self-consistent field (SCF) cycles were converged to a tolerance of 1 × 10−5 eV per atom, with convergence thresholds of 0.1 eV Å−1 for maximum forces and 5 × 10−3 Å for atomic displacements.The Gibbs free energy for all reactions was obtained by using the following formula eqn (3):36
| ΔG = ΔE + ΔZPE − TΔS | (3) |
| Reactions | Equations for change in ZPE | Value of ΔZPE (eV) | Equations for change in TS | Value of TΔS (eV) |
|---|---|---|---|---|
| CO2 + H = COOH | ΔZPE = ZPECOOH − (ZPECO2 + ZPEH) | 0.005238155 | TΔS = TSCOOH − (TSCO2 + TSH) | −1.01506546 |
| COOH + H = CO + H2O | ΔZPE = ZPECO + ZPEH2O − (ZPECOOH + ZPEH) | 0.010259345 | TΔS = TSCO + TSH2O − (TSCOOH + TSH) | 0.83977502 |
| CO + H = HCO | ΔZPE = ZPEHCO − (ZPECO + ZPEH) | −0.120043635 | TΔS = TSHCO − (TSCO + TSH) | −0.27203544 |
| HCO + H = H2CO | ΔZPE = ZPEH2CO − (ZPECO + ZPEH) | −0.114836475 | TΔS = TSH2CO − (TSCO + TSH) | −0.37171585 |
| H2CO + H = H3CO | ΔZPE = ZPEH3CO − (ZPEH2CO + ZPEH) | −0.132813575 | TΔS = TSH3CO − (TSH2CO + TSH) | −0.30651881 |
| H3CO + H = CH3OH | ΔZPE = ZPECH3OH − (ZPEH3CO + ZPEH) | −0.084275405 | TΔS = TSCH3OH − (TSH3CO + TSH) | −0.33720159 |
The adsorption energy (Eads) was calculated using the following expression:37
| Eads = Eadsorbate+surface − (Eadsorbate + Esurface) | (4) |
3. Results and discussion
The surface activation process for the Ag–Si/ZnO/MgO composite photocatalyst is shown in Fig. 1A. Scanning electron microscopy (SEM) analysis was employed to investigate the morphology of the fabricated Ag–Si/MgO/ZnO catalyst. The Ag–Si/MgO/ZnO catalyst exhibited densely packed, well-defined cuboidal/prismatic particles with faceted surfaces, indicative of high crystallinity and uniform growth. These particles exhibit a relatively narrow size distribution and form an aggregated network with close particle-to-particle contact, as depicted in Fig. 1B and C. The morphology of Ag–Si/MgO/ZnO catalysts was also studied using transmission electron microscopy (TEM). The TEM photograph shown in Fig. 1D and E displays particles that are well-defined and faceted. Their morphology is polyhedral and appears to be cuboidal or slightly truncated prismatic. The clear edges and uniform contrast show that these are single crystalline domains, not amorphous aggregates. The particle size seen in the TEM image is very similar to the size value proposed by SEM since the size is very monodispersed. Fig. 1G–L showing the EDX elemental mapping images of the synthesized Ag–Si/MgO/ZnO catalyst showed the distributions of elements Ag, Si, Zn, Mg, and O, thereby confirming successful preparation of the nanocomposite. The elemental mapping of the Ag–Si/MgO/ZnO nanostructured catalyst confirmed the uniform presence of Ag and Si on the nanocrystalline MgO/ZnO support, showing that these elements were evenly spread throughout the catalyst. The powder XRD analysis showed that all synthesized catalysts are highly crystalline. The XRD pattern of Ag–Si/MgO/ZnO shows peaks at 28.48°, 42.88°, 56.22°, 69.70°, and 77.38° depicting the indexed peaks (111), (220), (110), (400), and (331) of Si (JCPDS 01-089-2955). The presence of ZnO, MgO and Ag was confirmed by matching the characteristic peaks with their respective JCPDS files (03-65-2880, 00-045-0946, and 01-089-2955).MgO promotion had been specifically important because MgO had provided basic sites that had strengthened CO2 adsorption. A higher number of effective adsorption events was maintained compared with supports that were less basic or less able to stabilize highly dispersed metal particles.38 The plasmonic function of Ag had increased the flux of energetic electrons under visible light, while MgO had increased reactant uptake and stabilized interfacial active sites, and this dual promotion had been consistent with the experimentally observed highest methanol rate (357.53 µmol gcat−1 h−1 at 250 °C and 3 bar) and reduced apparent activation energy (158.14 kJ mol−1) for Ag–Si/MgO/ZnO versus the Ag-free counterparts. It was inferred from these results that strong ZnO–MgO interactions occurred which generated a high dispersion of Ag and Si species on the surface of the catalyst and small active site formation.
All samples were studied under the same conditions using the same excitation wavelength of 450 nm in the solid phase to study the charge-carrier dynamics and charge-separation efficiency using photoluminescence (PL) experiments (Fig. 2A). Lower PL intensity means charge separation is more efficient. There was a significantly lower number of recombination events in Ag–Si/MgO/ZnO indicating that the efficiency of charge-separation was excellent for this material. The introduction of Ag into Si/MgO/ZnO likely created additional vacancies that acted as traps for photo-generated charge carriers, thereby significantly suppressing their recombination. This property of keeping electrons in the conduction band for a longer period enhances their photocatalytic activity. The PL quenching effects seen in Ag–Si/MgO/ZnO showed that the charge transfer and separation in the system was enhanced. The outcomes of our PL tests have been confirmed to be consistent with the photocatalytic CO2 reduction data (Fig. 4B, C and E). This means that effective suppression of photo-induced e−/h+ recombination results in enhanced observed activity. Ag–Si/MgO/ZnO showed the best performance for CH3OH production among all tested samples.
Enhancing the photocatalytic conversion of CO2via the Ag–Si/MgO/ZnO heterostructure was due to efficient separation of photo-generated charge carriers. The presence of Ag in Ag–Si/MgO/ZnO made the charge more mobile and limited e−/h+ recombination. As a result, the transient photocurrent response (Fig. 2B) of Ag–Si/MgO/ZnO was about 7.5 times stronger than that of Si/ZnO and 1.8 times stronger than that of Si/MgO/ZnO. The response of the photocurrent remains stable upon repeated on off cycles which indicates good photoelectrochemical stability that is in line with PL results. Fig. 2C presents EIS Nyquist plots (frequency range: 0.1 Hz to 1 kHz, applied bias: −0.5 V). A minor arc radius in the Nyquist plot suggested less resistance to electron migration, signifying efficient charge-carrier separation. The Ag–Si/MgO/ZnO composite had a smaller arc radius than Si/ZnO and Si/MgO/ZnO with less charge recombination and improved electron–hole migration efficiency. These results were in agreement with the PL spectra. It can be concluded that Ag may play a crucial role in enhancing charge-separation efficiency by suppressing charge-recombination rates and improving the light-absorption behavior. Other researchers also observed that Ag loading effectively suppressed photo-induced electron–hole recombination and increased visible-light absorption, ultimately enhancing photocatalytic activity.39
UV-vis diffuse reflectance spectrophotometry was used to study optical absorption characteristics of the prepared photocatalysts. The UV-vis diffuse reflectance spectra of Si/MgO/ZnO and Ag–Si/MgO/ZnO are presented in Fig. 2D. The absorption band edge of Si/MgO/ZnO is observed at about 600 nm. After adding Ag nanoparticles, the absorption peak in the visible range was increased as well as red-shifted to about 630 nm. Thus, the edge of absorption was changed. The main reason for this change was due to the SPR behavior of the Ag species. Thus, inclusion of Ag into Ag–Si/MgO/ZnO was found to have greatly enhanced the light-absorption range into the visible region. Similar observations were made by Chen et al. who realized that Ag doping is responsible for visible light absorption due to plasmon resonance.39 A shift in UV-vis light absorption toward the visible region after Ag doping was also reported by Chen et al.40 However, it was noted that excessive Ag loading resulted in a decline in light absorption and photocatalytic CO2 reduction activity, likely due to a shielding effect, where surplus Ag partially blocked the light absorption of ZnO and MgO. The bandgap (Eg) values of the samples were estimated using Tauc's relation, as shown in eqn (5).41
| αhν = A(hν − Eg)n | (5) |
| Efb (vs. NHE) = Efb (vs. Ag/AgCl) + 0.197 | (6) |
The values of Efb (vs. NHE) for Si/ZnO, Si/MgO/ZnO and Ag–Si/MgO/ZnO samples were determined to be −1.013 V, −0.823 V and −0.783 V, respectively. It was known that the conduction band (CB) position of an n-type semiconductor lies approximately 0.1 eV below Efb.43 Hence, the CB potentials for Si/ZnO, Si/MgO/ZnO and Ag–Si/MgO/ZnO were calculated to be −1.113 V, −0.923 V and −0.883 V, respectively. The VB positions were then determined to be 1.157 V for Si/ZnO (−1.113 + 2.27), 1.097 V for Si/MgO/ZnO (−0.923 + 2.02) and 1.057 V for Ag–Si/MgO/ZnO (−0.883 + 1.94). Ag nanoparticles were found to act as electron-trapping centers, leading to enhanced photocatalytic performance.39 Based on these calculations, experimental data, and CO2 photoreduction results, a possible band diagram of the as-synthesized photocatalysts was proposed, and the mechanism of CO2 photoreduction to CH3OH over the Ag–Si/MgO/ZnO heterostructure was illustrated (Fig. 2G). The conduction band-edge potential of Ag–Si/MgO/ZnO was shown to be more negative than the reduction potential of CO2 to CH3OH. Furthermore, the bandgap of Ag–Si/MgO/ZnO was found to be smaller than that of Si/MgO/ZnO. Under visible light illumination, photo-generated electrons were efficiently transferred from the CB of Ag–Si/MgO/ZnO to the Ag NPs. The electrons accumulated on Ag NPs and then reduced CO2 to CH3OH. The difference in CO2 photoreduction efficiency between Ag-undoped and Ag-doped Si/MgO/ZnO heterostructures confirmed that the electron transfer rate from Ag NPs to the adsorbed carbonate species was enhanced by Ag integration, which improved carrier separation and overall CO2 photoreduction efficiency. Additionally, a synergistic effect between ZnO and MgO was postulated to suppress photo-induced charge recombination by transferring photo-generated species from ZnO to MgO within the heterostructure. Thus, the as-prepared Ag–Si/MgO/ZnO heterostructure was found to play a significant role in promoting CH3OH formation by increasing its production rate. To further elucidate the hydrogenation pathway for CO2 reduction to CH3OH over the Ag–Si dual-metal active sites of the Ag–Si/MgO/ZnO catalyst, DFT calculations were carried out using the Material Studio software. The surface electronic state, chemical composition, and bonding environment of the synthesized catalysts were analyzed by X-ray photoelectron spectroscopy (XPS). The XPS spectra of the Ag–Si/MgO/ZnO catalyst are presented in Fig. 3A–E. The full survey spectrum confirmed the presence of Ag, Si, Mg, Zn, and O elements within the sample, which was consistent with the EDS results, thereby indicating the successful synthesis of the catalyst. The characteristic peaks corresponding to Ag 3d, Si 2p, Mg 2p, Zn 2p, and O 1s were observed at their respective binding energies.
As shown in Fig. 3A, the Ag 3d spectrum exhibited two strong peaks at 367.7 and 374.4 eV, attributed to Ag 3d5/2 and Ag 3d3/2, respectively, confirming the presence of metallic Ag clusters. Two weak peaks corresponding to Ag+ were also observed, in agreement with the results of Tada et al.,45 who reported that XPS spectra displayed peaks for metallic Ag at 368.0 eV (Ag 3d5/2) and 374.0 eV (Ag 3d3/2), as well as weak peaks for Ag+ at 369.7 eV (Ag 3d5/2) and 375.7 eV (Ag 3d3/2).46,47 In the Si spectrum (Fig. 3B), peaks at 99.38 and 100.3 eV were assigned to Si 2p3/2 and Si 2p1/2, respectively, confirming the coexistence of Ag and Si in the composite. Cerofolini et al. reported that the 2p3/2 and 2p1/2 peaks in the range of 99–100.5 eV were characteristic of elemental silicon.48 The peak at 100.3 eV was assigned to Si–H bonds, while a feature at 532.1 eV indicated the presence of Si–OH species. The presence of Si–H dangling bonds has been associated with enhanced charge separation efficiency on Si-supported photocatalysts.49 These functional bonds acted as electron sinks, promoting charge transfer during photocatalysis. In the Mg spectrum (Fig. 3C), two peaks at 49.7 and 50.1 eV were attributed to Mg 2p3/2 and Mg 2p1/2, respectively. Fig. 3D shows the Zn 2p spectrum with peaks at 1021.24 eV and 1022.2 eV, corresponding to Zn 2p3/2 and Zn 2p1/2, confirming the coexistence of Zn2+ and Mg2+ species in the catalyst. A higher-binding-energy (∼1023.5 eV) in the Zn 2p3/2 region could be attributed to surface-defective Zn2+ species, originating from oxygen-deficient coordination and/or hydroxylated Zn sites, which result in reduced electronic screening compared to bulk ZnO.50 The O 1s spectrum (Fig. 3E) exhibited peaks at 529.7, 530.9, and 532.1 eV, assigned to lattice oxygen, oxygen vacancies, and surface hydroxyl groups, respectively. The 529.7 eV peak was consistent with MgO formed through solid-phase oxidation of Mg. The role of MgO as a support was attributed to its tunable acid–base properties, which regulate electron transfer between the support and active phase, as suggested by Julkapli et al.51 Additionally, Meshkani et al. reported that Lewis basicity of MgO enhanced chemisorption of surface functional groups and promoted electron transport.52 No detectable boron-related chemical states had been observed in the XPS spectra, indicating that residual boron-containing species were below the detection limit and negligible in the final material.
The total density of states (TDOS) for bimetallic Ag–Si/MgO/ZnO, single-metallic Si/MgO/ZnO, and Si/ZnO catalysts is presented in Fig. S3. The TDOS of bimetallic Ag–Si/MgO/ZnO exhibited a narrower bandgap compared to Si/MgO/ZnO and Si/ZnO, justifying its faster electron excitation and transfer to intermediate species. The bimetallic co-catalyst thus accelerated electron transport, enabling the six-electron reduction of CO2 to CH3OH. Projected partial density of states (PDOS) analysis (Fig. 3F) showed that the s-band center of Ag–Si/MgO/ZnO was located near the Fermi level (EF), favoring activation of O-species adsorption. The weak s–p orbital overlap indicated a long-range interlayer interaction, allowing CO2 molecules to pass through easily without significantly affecting the catalyst interface.
To understand the enhancement in photocatalytic performance after loading Ag and Si nanoparticles, finite-difference time-domain (FDTD) calculations were conducted in the visible range at different wavelengths (400–800 nm) to simulate the local electric field distribution between the Ag and Si nanoparticle assemblies on MgO/ZnO with an interparticle distance of 2 nm (Fig. 4A and S4(A–F)). The electric field intensity distribution was visualized on a logarithmic scale. Fig. S4(G) shows the electric field distribution, |E|2, around the surface of Ag–Si/MgO/ZnO as a function of wavelength using FDTD simulations. Upon light irradiation, the electric field distribution ∣E∣2 around the surface of Ag–Si/MgO/ZnO was enhanced, and a maximum value of ∣Emax∣2 of ≈800 was obtained at a wavelength of 457.143 nm. This improved local electric field, originating from the LSPR effect, was believed to have accelerated the transfer of hot electrons in the Ag–Si nanoparticles and promoted the separation of electron–hole pairs in MgO/ZnO. Furthermore, the visible-light absorbance was increased by Ag loading, thereby improving the photocatalytic activity, consistent with the photoluminescence results. The photocatalytic performance of the prepared samples was evaluated in a continuous-flow reactor system using CO2 as the reactant at temperatures up to 300 °C and atmospheric pressure. The H2/CO2 ratio was maintained at 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The results (Fig. 4B–F) showed that CH3OH was the predominant product. At 160 °C, CH3OH formation was detected under light irradiation, whereas no CH3OH was observed under dark conditions. These results suggested that below 160 °C, the Ag–Si/MgO/ZnO nanocrystal superstructures could only catalyze the RWGS reaction. CH3OH formation was most likely kinetically inhibited at low temperatures and pressures. The highest CH3OH production rate was obtained at 250 °C, reaching 357.53 µmol gcat−1 h−1 under light illumination (Fig. 4B and C). This value at 3 bar was higher than that produced over Si/MgO/ZnO and Si/ZnO composites by factors of 1.12 and 4.7, respectively. The as-prepared Ag–Si/MgO/ZnO photocatalyst exhibited a high apparent quantum yield (AQY) of 21.27% at an excitation wavelength of 420 nm for CH3OH production, demonstrating its excellent visible-light-driven photocatalytic efficiency. When the temperature was increased to 300 °C, the CH3OH production rate decreased to 121.84 µmol gcat−1 h−1. This decline was attributed to the exothermic nature of CH3OH synthesis (eqn (1)), which shifted the equilibrium toward the reactants at higher temperatures according to Le Chatelier's principle. The reaction thermodynamics at 300 °C favored the RWGS pathway, confirming that 250 °C was the optimal temperature for CH3OH formation.
1. The results (Fig. 4B–F) showed that CH3OH was the predominant product. At 160 °C, CH3OH formation was detected under light irradiation, whereas no CH3OH was observed under dark conditions. These results suggested that below 160 °C, the Ag–Si/MgO/ZnO nanocrystal superstructures could only catalyze the RWGS reaction. CH3OH formation was most likely kinetically inhibited at low temperatures and pressures. The highest CH3OH production rate was obtained at 250 °C, reaching 357.53 µmol gcat−1 h−1 under light illumination (Fig. 4B and C). This value at 3 bar was higher than that produced over Si/MgO/ZnO and Si/ZnO composites by factors of 1.12 and 4.7, respectively. The as-prepared Ag–Si/MgO/ZnO photocatalyst exhibited a high apparent quantum yield (AQY) of 21.27% at an excitation wavelength of 420 nm for CH3OH production, demonstrating its excellent visible-light-driven photocatalytic efficiency. When the temperature was increased to 300 °C, the CH3OH production rate decreased to 121.84 µmol gcat−1 h−1. This decline was attributed to the exothermic nature of CH3OH synthesis (eqn (1)), which shifted the equilibrium toward the reactants at higher temperatures according to Le Chatelier's principle. The reaction thermodynamics at 300 °C favored the RWGS pathway, confirming that 250 °C was the optimal temperature for CH3OH formation.
A preliminary evaluation of the temperature-dependent solar CH3OH production over Ag–Si/MgO/ZnO is shown in Fig. 4B. The largest amount of CH3OH was condensed between −8 °C and −16 °C (Fig. 4D). The stability of the Ag–Si/MgO/ZnO photocatalyst was tested during continuous operation for 72 h at 250 °C and 3 bar under light irradiation (Fig. 4E). Only a slight decrease in the CH3OH production rate was observed, indicating excellent long-term stability. CH3OH production rates of various photocatalysts and comparable systems are listed in Table 2.53–59 A significant enhancement of CH3OH generation from 159.84 to 357.53 µmol gcat−1 h−1 was achieved when the GHSV was increased from 120 to 480 mL gcat−1 h−1 at 250 °C and 3 bar (Fig. 4F). This improvement was attributed to the synergistic effect of Ag–Si bimetallic loading, consistent with previous findings by Chen et al.,40 who reported that Ag doping effectively suppressed electron–hole recombination and Si increased the catalyst surface area, jointly promoting visible-light photocatalysis. Plasmonic enhancement had been imparted by Ag because the optical response had been extended into the visible region and red-shifted toward ∼630 nm after Ag incorporation, and stronger charge separation/transport had been evidenced by pronounced PL quenching, higher transient photocurrent, and a smaller EIS semicircle, indicating that a larger fraction of photoexcited electrons had been delivered to surface reaction sites than in non-plasmonic semiconductor-only systems.60 This behavior had contrasted with common strategies such as band-gap narrowing by bulk doping or simple heterojunction formation, where photon harvesting and interfacial electron flux had often remained limited by weak visible absorption or fast recombination; in the present architecture, plasmonic near-field/hot-carrier effects had been expected to increase the local excitation rate and accelerate interfacial electron injection under visible irradiation, thereby increasing the effective electron supply for CO2/CO activation and hydrogenation.61
| Catalysts | T | P |
V
CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) VH2 VH2 |
Methanol yield | Reference |
|---|---|---|---|---|---|
| Au/In2O3 | — | — | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
320 µmol gcat−1 h−1 | 53 |
| CaCu3Ti4O12 | 250 °C | 8 bar | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
308.5 µmol gcat−1 h−1 | 54 |
| Ru/In2O3 | — | atmP | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
280.4 µmol gcat−1 h−1 | 55 |
| Cu/ZnO | 220 °C | atmP | 2.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
127.8 µmol g−1 h−1 | 56 |
| In2O3−x(OH)y-NR-14h | 250 °C | atmP | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
97.30 µmol gcat−1 h−1 | 57 |
| CoO/Co/TiO2 | 120 °C | atmP | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
39.6 µmol gcat−1 h−1 | 58 |
| HzIn2O3−x(OH)y | 300 °C | atmP | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
31.2 µmol gcat−1 h−1 | 59 |
| HzIn2O3−x(OH)y | 250 °C | atmP | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
14.92 µmol gcat−1 h−1 | 59 |
| Ag–Si/MgO/ZnO | 250 °C | 3 bar | 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
357.53 µmol gcat−1 h−1 | This work |
A schematic summary had been proposed in which CO2 had been adsorbed at basic MgO sites, COOH*/CO* formation had been favored at Ag–Si/MgO/ZnO and terminal hydrogenation to CH3OH completed predominantly at Ag sites supplied by efficiently separated photoelectrons under visible irradiation. The most favorable pathway for CO2 hydrogenation to CH3OH over this interface system was determined. Formate (HCOO*) and carboxyl (*COOH) species were identified as the key intermediates in the HCOO* and RWGS pathways. The Ag–Si/MgO/ZnO(100) system demonstrated the most energetically favorable *COOH pathway, with the lowest barrier (−4.93 eV) and a strongly exothermic reaction energy for CH3OH formation (−8.55961 eV). In contrast, the HCOO* pathway exhibited a high barrier (15.1 eV) and endothermic CO2 → HCOO* conversion. Therefore, the *COOH route was identified as the preferred pathway over Ag–Si/MgO/ZnO(100) (Fig. 5A). Starting from *COOH, the Gibbs free-energy profiles for the 6H+/6e− reduction of CO2 to CH3OH over Ag–Si/MgO/ZnO(100) and Si/MgO/ZnO(100) were analyzed (Fig. 5B). CO2 hydrogenation to *COOH was exothermic (−4.93 eV) over Ag–Si/MgO/ZnO(100) but endothermic (+1.95 eV) over Si/MgO/ZnO(100), demonstrating the greater favorability of COOH formation in the presence of Ag. Incorporation of MgO enhanced *COOH adsorption, enabling its conversion to *CO, which was readily hydrogenated to methoxy (CH3O*), as also reported by Li et al.62 Compared with Si/MgO/ZnO, Ag–Si/MgO/ZnO had little effect on the free energies of *COOH → *CO, *CO → CHO, or *CHO → *CH2O. However, the last three proton-transfer steps were identified as the decisive steps controlling performance. It was inferred that the first three intermediates were adsorbed on Si sites, while the final three were stabilized on Ag sites.
Partial density of states (PDOS) analysis at Ag sites demonstrated that Ag facilitated more rapid electron transfer from *CH2O, accelerating its hydrogenation to *CH3O. The free energy for the step CH3O + H+ + e− → CH3OH was reduced to 5.65 eV (Fig. 5B). Overall, CH3OH formation over Ag–Si/MgO/ZnO proceeded predominantly via the pathway:
| *COOH + H* → *CO + H* → *HCO + H* → *H2CO + H* → *CH3O + H* → CH3OH* |
Fig. 5D shows the structural CO2 hydrogenation pathways for methanol production within the Ag–Si/MgO/ZnO interface system. Ag and Si had synergistically generated a dual-site catalytic landscape on Ag–Si/MgO/ZnO(100), where the initial CO2 activation had been promoted through a COOH-mediated route that had been calculated as exothermic on Ag–Si/MgO/ZnO(100) (−4.93 eV) but endothermic on Si/MgO/ZnO(100) (+1.95 eV). This indicates that the bimetallic interface reduced the energy barrier for forming early oxygenated intermediates compared with the Ag-free surface. The decisive hydrogenation sequence had been described as spatially partitioned, such that early intermediates had been preferentially stabilized at Si-associated interfacial/oxide sites while the late hydrogenation steps (CHO* → CH3OH) had been stabilized at Ag-rich sites acting as electron-accepting and hydrogenation centers, which had been consistent with the reported DOS features near the Fermi level.64 This mechanistic assignment had been directly linked to experimental observables because suppressed recombination and improved charge transport had been evidenced by PL quenching, increased transient photocurrent, and a smaller EIS semicircle for Ag–Si/MgO/ZnO, and the same sample had delivered the highest methanol rate (357.53 µmol gcat−1 h−1 at 250 °C and 3 bar) with the lowest apparent activation energy (158.14 kJ mol−1), which had been aligned with the computed facilitation of key elementary steps.65
4. Conclusion
The Ag and Si–bimetal-doped MgO-promoted ZnO photocatalysts were synthesized via a solid-phase reaction approach. Using the 10 wt% Ag–Si/MgO/ZnO catalyst, the highest CH3OH production rate of 357.53 µmol gcat−1 h−1 was achieved for CO2 hydrogenation at 250 °C under light illumination. A strong interaction between ZnO and MgO, resulting in a synergistic effect, could be responsible for the excellent selectivity of the 10 wt% Ag–Si/MgO/ZnO catalyst in the photocatalytic hydrogenation of CO2 to CH3OH. No significant deactivation was observed after 72 h of reaction. CO2 adsorption on the Ag–Si/MgO/ZnO(100) surface (−1.04 eV) was 0.35 eV stronger than on Si/MgO/ZnO(100), which was inferred to have enhanced CO2 activation and promoted higher CO2 conversion. The stronger adsorption observed at the Ag–Si/MgO/ZnO(100) interface compared with the Si/MgO/ZnO(100) interface was inferred to enhance CO2 adsorption, thereby promoting higher CO2 conversion over the Ag–Si/MgO/ZnO(100) catalyst. This study thus provides new insights into the rational design of photocatalysts with well-defined heterojunctions for efficient photocatalytic CO2 reduction.Author contributions
AM: writing – original draft, methodology, formal analysis, data curation; A-TH: review & editing, writing; MTI: writing – review & editing, validation; MAH: writing – review & editing; TI: writing – review & editing, writing – original draft, visualization; AI: writing – review & editing, writing – original draft, supervision, funding acquisition, conceptualization.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.Supplementary information (SI): schematic synthesis procedure of the Ag–Si/MgO/ZnO nanocomposite (S1); experimental setup for photocatalytic CO2 hydrogenation to CH3OH under visible light (S2); DFT calculations of CO2 reduction and the total density of states for Ag–Si/MgO/ZnO, Si/MgO/ZnO, and Si/ZnO (S3); finite-difference time-domain (FDTD) simulation of electric field distribution (S4); apparent activation energy (Ea) evaluation (S5); and necessary equations. See DOI: https://doi.org/10.1039/d5se01485a.
Acknowledgements
This work was supported by the Bangladesh Energy and Power Research Council (BEPRC) under Research Grant No. March 27, 0000.000.39.001.17.651. The research was conducted at the Hydrogen Energy and CO2 Conversion Laboratory, Department of Petroleum and Mining Engineering, Jashore University of Science and Technology, Jashore, 7408, Bangladesh.References
- P. Ndem, O. Biose, E. Effiong, S. Udegbe, J. E. Ukpebor, I. H. Ifijen and E. E. Ukpebor, Environ. Monit. Assess., 2025, 197(8), 925 CrossRef CAS PubMed.
- C. Zhang, J. Fang, Z. Han, P. Chen, X. Xu, K. Wang, Y. Shi and J. Liao, iScience, 2025, 28(8), 113127 CrossRef CAS PubMed.
- F. Yan, H. Chen, T. Chi, J. Lu, X. Shen, F. Xie, P. Wang and Z. Zhang, Chem. Eng. J., 2025, 520, 166450 CrossRef CAS.
- J. He, G. Tian, D. Liao, Z. Li, Y. Cui, F. Wei, C. Zeng and C. Zhang, J. Energy Chem., 2025, 778–803 Search PubMed.
- J. Zhao, Z. Wen, R. Zhu, Z. Li, R. Ding, Y. Zhu, T. Gu, R. Yang and Z. Zhu, Chem. Eng. J. Adv., 2020, 3, 100029 CrossRef CAS.
- A. Baig, V. Goswami and Sonal, Asian J. Water, Environ. Pollut., 2024, 21(6), 49–57 CrossRef.
- F. A. Rafi, M. M. Rana, B. Agili and A. S. Al Shahrany, Eur. J. Energy Res., 2025, 5(2), 1–15 CrossRef.
- M. M. Rana, S. M. Alam, F. A. Rafi, S. B. Deb, B. Agili, M. He and M. H. Ali, IEEE Access, 2025, 35124–35156 Search PubMed.
- M. M. Rana, F. A. Rafi, B. Agili and A. S. Al Shahrany, Eur. J. Electr. Eng. Comput. Sci., 2024, 8(2), 1–8 CrossRef.
- S. Yasar Aurko, R. Salehdin, F. Allahma Rafi and K. Habibul Kabir, The Fourth Industrial Revolution and Beyond: Select Proceedings of IC4IR+, Springer Nature Singapore, Singapore, 2023, pp. 277–290 Search PubMed.
- Y. Yang, C. Shen, K. Sun, D. Mei and C. J. Liu, ACS Catal., 2023, 13(9), 6154–6168 CrossRef CAS.
- Q. Wu, S. Liang, T. Zhang, B. Louis and Q. Wang, Fuel, 2022, 313, 122963 CrossRef CAS.
- C. Clavero, Nat. Photonics, 2014, 8(2), 95–103 Search PubMed.
- M. M. J. Li and S. C. E. Tsang, Catal. Sci. Technol., 2018, 8(14), 3450–3464 RSC.
- M. L. Brongersma, N. J. Halas and P. Nordlander, Nat. Nanotechnol., 2015, 10(1), 25–34 CrossRef CAS PubMed.
- S. M. Sze and K. K. Ng, Physics of semiconductor devices: Sze/physics, Wiley-Blackwell, Chichester, England, 3rd edn, 2006 Search PubMed.
- H. A. Atwater and A. Polman, Nat. Mater., 2010, 9(3), 205–213 CrossRef CAS PubMed.
- W. Sheng, J. Yang, X. Li, G. Liu, Z. Lin, J. Long, S. Xiao, L. Tan and Y. Chen, Energy Environ. Sci., 2021, 14(6), 3532–3541 Search PubMed.
- T. Kong, A. Liao, Y. Xu, X. Qiao, H. Zhang, L. Zhang and C. Zhang, RSC Adv., 2024, 14(24), 17041–17050 CAS.
- J. Zhao, B. Liu, L. Meng, S. He, R. Yuan, Y. Hou, Z. Ding, H. Lin, Z. Zhang, X. Wang and J. Long, Appl. Catal., B, 2019, 256, 117823 CAS.
- A. O. Yusuf, R. Y. Tekle, H. K. Amusa and G. Palmisano, Case Stud. Chem. Environ. Eng., 2025, 11, 101172 CAS.
- M. T. Khalil, X. Wu, S. Liu, Y. Liu, S. Ashraf, R. Shen, H. Zhang, Z. Peng, J. Jiang and B. Li, Green Chem., 2025, 27(30), 9016–9054 Search PubMed.
- Z. J. Wang, H. Song, H. Pang, Y. Ning, T. D. Dao, Z. Wang, H. Chen, Y. Weng, Q. Fu, T. Nagao and Y. Fang, Appl. Catal., B, 2019, 250, 10–16 CAS.
- J. L. G. Fierro, M. A. Peña and M. C. Alvarez-Galvan, Supported Metal Catal., 2005, pp. 229–255 Search PubMed.
- V. D. Dasireddy and B. Likozar, Renewable Energy, 2019, 140, 452–460 CAS.
- Q. Chen, S. Meng, R. Liu, X. Zhai, X. Wang, L. Wang, H. Guo and Y. Yi, Appl. Catal., B, 2024, 342, 123422 CAS.
- Q. He, Z. Li, D. Li, F. Ning, Q. Wang, W. Liu, W. Zhang, Y. Cui, J. Zhang and C. Liu, Mol. Catal., 2024, 558, 114008 CAS.
- Y. L. Men, Y. Liu, Q. Wang, Z. H. Luo, S. Shao, Y. B. Li and Y. X. Pan, Chem. Eng. Sci., 2019, 200, 167–175 CAS.
- R. Pothu, H. Mitta, P. Banerjee, R. Boddula, R. K. Srivastava, P. K. Kalambate, R. Naik, A. B. Radwan and N. Al-Qahtani, Mater. Sci. Energy Technol., 2023, 6, 484–492 CAS.
- K. Sun, N. Rui, C. Shen and C. J. Liu, J. Phys. Chem. C, 2021, 125(20), 10926–10936 CAS.
- H. Liu, M. Zhao, J. Guo, S. Sun, D. Li, D. He and Y. Lei, ACS Sustainable Chem. Eng., 2025, 13(15), 5787–5795 CAS.
- X. Zhang, H. Liu, Y. Wang, S. Yang, Q. Chen, Z. Zhao, Y. Yang, Q. Kuang and Z. Xie, Chem. Eng. J., 2022, 443, 136482 CAS.
- Y. Wang, Y. Liu, L. Tan, X. Lin, Y. Fang, X. F. Lu, Y. Hou, G. Zhang and S. Wang, J. Mater. Chem. A, 2023, 11(48), 26804–26811 CAS.
- Y. Wang, B. Su, S. Wang, L. Tan, G. Zhang, Y. Fang and X. Wang, Angew. Chem., Int. Ed., 2025, e202508611 CAS.
- A. Islam, S. H. Teo, M. R. Awual and Y. H. Taufiq-Yap, J. Cleaner Prod., 2020, 244, 118805 CAS.
- C. Hu, Z. Jiang, Q. Wu, S. Cao, Q. Li, C. Chen, L. Yuan, W. Yang, J. Yang and J. Peng, Nat. Commun., 2023, 14(1), 4767 CAS.
- H. Yu, F. Chen, X. Li, H. Huang, Q. Zhang, S. Su, K. Wang, E. Mao, B. Mei, G. Mul and T. Ma, Nat. Commun., 2021, 12(1), 4594 CAS.
- H. J. Freund and G. Pacchioni, Chem. Soc. Rev., 2008, 37(10), 2224–2242 CAS.
- S. Chen, B. Lv, D. Wu and Y. Xu, J. Nanosci. Nanotechnol., 2013, 13(2), 1569–1573 CAS.
- Q. Chen, W. Shi, Y. Xu, D. Wu and Y. Sun, Mater. Chem. Phys., 2011, 125(3), 825–832 CAS.
- I. H. Alsohaimi, A. M. Nassar, T. A. S. Elnasr and B. Amar Cheba, J. Cleaner Prod., 2020, 248, 119274 CAS.
- N. N. Vu, S. Kaliaguine and T. O. Do, Adv. Funct. Mater., 2019, 29(31), 1901825 Search PubMed.
- N. Ojha, A. Bajpai and S. Kumar, J. Colloid Interface Sci., 2021, 585, 764–777 Search PubMed.
- N. Ojha, A. Bajpai and S. Kumar, Catal. Sci. Technol., 2019, 9(17), 4598–4613 Search PubMed.
- S. Tada, F. Watanabe, K. Kiyota, N. Shimoda, R. Hayashi, M. Takahashi, A. Nariyuki, A. Igarashi and S. Satokawa, J. Catal., 2017, 351, 107–118 Search PubMed.
- R. Grabowski, J. Słoczynski, M. Sliwa, D. Mucha, R. P. Socha, M. Lachowska and J. Skrzypek, ACS Catal., 2011, 1(4), 266–278 CAS.
- I. C. Freitas, J. M. R. Gallo, J. M. C. Bueno and C. M. Marques, Top. Catal., 2016, 59(2), 357–365 CAS.
- G. F. Cerofolini, C. Galati and L. Renna, Surf. Interface Anal., 2003, 35(12), 968–973 CAS.
- S. H. Teo, A. Islam, Y. H. Taufiq-Yap and M. R. Awual, J. Cleaner Prod., 2021, 313, 127909 CAS.
- P. Devaraji, M. Mapa, H. M. Abdul Hakkeem, V. Sudhakar, K. Krishnamoorthy and C. S. Gopinath, ACS Omega, 2017, 2(10), 6768–6781 CAS.
- N. M. Julkapli and S. Bagheri, Rev. Inorg. Chem., 2016, 36(1), 1–41 CAS.
- F. Meshkani and M. Rezaei, Catal. Commun., 2011, 12(11), 1046–1050 CrossRef CAS.
- L. Wang, D. Yao, C. Zhang, Y. Chen, L. Amirav and Z. Zhong, Chem Catal., 2024, 4(9), 101095 CAS.
- Z. Jiang, Z. Yuan, P. N. Duchesne, W. Sun, X. Lyu, W. Miao, C. J. Viasus Pérez, Y. Xu, D. Yang, B. Huang, Y. Dai, Z. Wang, H. He and G. A. Ozin, Chem Catal., 2023, 3(2), 100507 Search PubMed.
- B. Deng, H. Song, Q. Wang, J. Hong, S. Song, Y. Zhang, K. Peng, H. Zhang, T. Kako and J. Ye, Appl. Catal., B, 2023, 327, 122471 CAS.
- Z. J. Wang, H. Song, H. Pang, Y. Ning, T. D. Dao, Z. Wang, H. Chen, Y. Weng, Q. Fu, T. Nagao and Y. Fang, Appl. Catal., B, 2019, 250, 10–16 CrossRef CAS.
- L. Wang, M. Ghoussoub, H. Wang, Y. Shao, W. Sun, A. A. Tountas, T. E. Wood, H. Li, J. Y. Y. Loh, Y. Dong and M. Xia, Joule, 2018, 2(7), 1369–1381 Search PubMed.
- Z. H. He, C. S. Jiang, K. Wang, Z. Y. Wang, N. Li, W. T. Wang and Z. T. Liu, Catal. Today, 2020, 356, 579–588 CrossRef CAS.
- Z. Zhang, C. Mao, D. M. Meira, P. N. Duchesne, A. A. Tountas, Z. Li, C. Qiu, S. Tang, R. Song, X. Ding and J. Sun, Nat. Commun., 2022, 13(1), 1512 CrossRef CAS PubMed.
- S. Linic, P. Christopher and D. B. Ingram, Nat. Mater., 2011, 10(12), 911–921 CrossRef CAS PubMed.
- M. L. Brongersma, N. J. Halas and P. Nordlander, Nat. Nanotechnol., 2015, 10(1), 25–34 CAS.
- K. Li and J. G. Chen, ACS Catal., 2019, 9(9), 7840–7861 CrossRef CAS.
- X. Shan, G. Zhang, Y. Zhang, S. Zhang, F. Guo and Q. Xu, Catalysts, 2024, 14(11), 756 CAS.
- J. Greeley, T. F. Jaramillo, J. Bonde, I. B. Chorkendorff and J. K. Nørskov, Nat. Mater., 2006, 5(11), 909–913 Search PubMed.
- Z. Zeng, K. C. Chang, J. Kubal, N. M. Markovic and J. Greeley, Nat. Energy, 2017, 2(6), 17070 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





