 Open Access Article
Open Access ArticleTransition metal dichalcogenides as platforms for biosensing, phototherapy, and drug delivery
Yilin
Tang†
a,
Ziheng
Huang†
a,
Keyue
Tian
a and
Feng
Luo
 *ab
*ab
aState Key Laboratory of Oral Diseases & National Center for Stomatology & National Clinical Research Center for Oral Diseases, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan, China. E-mail: luofeng0122@scu.edu.cn; Fax: +86 28 85503474; Tel: +86 28 85503671/13488904276
bDepartment of General Dentistry, West China School of Stomatology, Sichuan University, Chengdu 610041, China
First published on 17th March 2026
Abstract
Two-dimensional transition metal dichalcogenides (TMDCs) are advancing from model nanomaterials to clinically relevant biointerfaces. Their atomic thickness, tunable band structures, strong light–matter coupling, and chemically addressable surfaces enable this progress. This review connects structure–property–function relationships to synthesis routes, including mechanical exfoliation, chemical vapor deposition, and solution-phase exfoliation. These methods control layer number, phase, defect chemistry, and lateral size. We outline how covalent or non-covalent functionalization, heterostructuring, and doping expand colloidal stability, targeting specificity, and robustness in physiological media. TMDCs leverage their high surface area and semiconducting properties to enable ultrasensitive biosensing across field-effect, electrochemical, and fluorescence/colorimetric approaches. They also enable stimuli-responsive drug delivery with spatiotemporal control and multimodal bioimaging. Modalities span photoluminescence, photoacoustic, two-photon, and MRI-hybrid platforms. Broadband near-infrared absorption supports efficient photothermal and photodynamic cancer therapies, enabling synergistic theranostics. Mechanically compliant, conductive composites position TMDCs for tissue engineering and regenerative scaffolds. We critically assess biocompatibility and long-term fate, focusing on phase or defect chemistry, protein coronas, reactive oxygen species, and degradation pathways. Translational bottlenecks include batch variability, scalable manufacturing with phase control, deep-tissue activation, standardized characterization, and regulatory readiness. We map these to a practical roadmap integrating reproducible synthesis, harmonized materials descriptors, in vivo pharmacokinetics or toxicology standards, and interoperable device integration. Together, TMDCs are emerging as a coherent materials platform for precision diagnostics, targeted therapy, and regeneration, with milestones toward clinical adoption.
1. Introduction
Two-dimensional (2D) materials, particularly transition metal dichalcogenides (TMDCs), have emerged as key players in biomedicine due to their unique properties arising from their atomic thickness and composition. TMDCs, consisting of a transition metal (e.g., molybdenum [Mo] or tungsten [W]) and chalcogenides (e.g., sulfur [S], selenium [Se], or tellurium [Te]), offer a diverse range of electrical, optical, mechanical, and chemical characteristics that are ideal for various biomedical applications.1 These materials exhibit properties ranging from semiconducting to metallic, depending on their composition and structural phase.2 This versatility makes TMDCs particularly suitable for applications in sensing, imaging, and therapy. Their high surface-to-volume ratio promotes strong interactions with biomolecules, enhancing their sensitivity for detection and ability to load large amounts of drugs. Additionally, TMDCs possess unique optical properties, including strong light absorption and photoluminescence across the visible to near-infrared (NIR) range, making them ideal for bioimaging and phototherapies.2,3 The inherent mechanical strength and flexibility of TMDCs enable their integration into wearable and implantable devices without compromising functionality or comfort. Their chemical modifiability enables the conjugation of biomolecules, targeting agents, and polymers, thereby enhancing their biocompatibility and targeting specificity.4TMDCs, with a transition metal (M) sandwiched between two chalcogen atoms (X), typically sulfur, selenium, or tellurium (MX2), exhibit a range of electronic properties, from metallic to semiconducting.5 Synthesis methods for TMDCs, such as mechanical exfoliation, chemical vapor deposition (CVD), and atomic layer deposition (ALD), offer control over layer thickness and material purity, enabling the fine-tuning of their properties. While graphene has garnered significant attention for its properties, TMDCs differ in that they exhibit a wide range of band gaps, making them particularly attractive for optoelectronic applications and biosensing. The semiconducting nature of TMDCs, such as MoS2 and WS2, makes them particularly suitable for photothermal therapy (PTT) and drug delivery systems, where their light-absorption properties can be used to control drug release and target and destroy cancer cells. Their unique optical properties also enable fluorescence-based biosensing and imaging for diagnostics and in vivo monitoring.
TMDCs are explored for biosensing, drug delivery, bioimaging, photothermal and photodynamic therapy (PTT/PDT), and tissue engineering. TMDC-based sensors utilize electrical and optical properties for sensitive biomolecule detection. As carriers for therapeutics, they enable controlled release and targeted delivery, improving treatment efficacy and minimizing side effects. Their optical characteristics offer high-resolution imaging of biological structures. Strong NIR light absorption enables localized cancer therapy with minimal damage to healthy tissue. TMDCs are also incorporated into scaffolds to support cell growth and tissue regeneration, leveraging their mechanical and surface features.
The mechanisms behind these applications are rooted in TMDCs' atomic structure and material properties. In biosensing, the adsorption of biomolecules alters electrical properties, enabling the detection of specific analytes. For drug delivery, TMDCs' surfaces can be engineered to respond to environmental stimuli, releasing loaded drugs at target sites. Their optical properties are exploited in bioimaging and phototherapies, where interactions with light enable both visualization and therapeutic functionalities. Despite the promising potential of TMDCs in biomedicine, challenges remain, including ensuring long-term biocompatibility, mitigating potential toxicity, achieving efficient and targeted in vivo delivery, and scaling up production. Future research will focus on overcoming these challenges by developing novel synthesis and functionalization strategies, conducting comprehensive biocompatibility and toxicity studies, and exploring the integration of TMDCs with existing medical technologies to enable seamless clinical translation. The application of TMDCs in biomedicine is a rapidly growing field with the potential to revolutionize diagnostics, therapeutics, and regenerative medicine. As we continue to explore and harness the unique properties of TMDCs, their integration into biomedical applications promises innovative solutions to healthcare challenges, paving the way for advancements in medical technology and patient care.
While several excellent reviews have summarized the fundamental properties and general applications of TMDCs in biomedicine, this review distinguishes itself by providing a critical and forward-looking analysis focused on the translational pathway from laboratory innovation to clinical implementation. Unlike previous publications, we place a particular emphasis on the interplay between material synthesis, functionalization strategies, and their direct consequences for in vivo performance and biocompatibility.
2. Synthesis of TMDCs
2.1 Mechanical exfoliation
Mechanical exfoliation is a straightforward and effective method for synthesizing 2D TMDCs, such as MoS2, WS2, and WSe2. This technique, which played a crucial role in the initial isolation of graphene, involves physically peeling layers from a bulk TMDC crystal to produce thin layers or even single layers of the material. Despite its simplicity, mechanical exfoliation has been instrumental in the research and development of TMDCs, enabling the production of high-quality, atomically thin layers ideal for fundamental studies and small-scale device fabrication.3The process begins with a bulk TMDC crystal, characterized by van der Waals forces between layers that enable separation. Adhesive tape is applied and peeled off to cleave layers from the material. Repeating the peeling process with fresh tape or folding and unfolding the tape thins the layers further. The thinned layers on the tape are then transferred to a suitable substrate, such as a silicon wafer coated with silicon dioxide, by pressing the tape onto the substrate and peeling it off, leaving the TMDC layers on the surface.6,7 The transferred layers are characterized using optical microscopy, atomic force microscopy (AFM), or Raman spectroscopy.
This process is straightforward and requires minimal equipment, making it accessible for basic research laboratories. Mechanical exfoliation yields TMDC layers with minimal defects, preserving their intrinsic electronic and optical properties. However, the technique is not suitable for large-scale production due to its labor-intensive nature and the difficulty in consistently producing layers of uniform thickness.8 Typically, the process yields a small number of 2D layers, with the thickness and size of exfoliated flakes varying significantly.9 Despite these limitations, mechanical exfoliation remains a valuable technique for producing high-quality TMDC samples for research, especially for investigating their unique physical, chemical, and electronic properties. Additionally, prototype devices such as transistors, sensors, and photodetectors can be developed to explore the potential applications of TMDCs in electronics and optoelectronics.
Mechanical exfoliation does not work for industrial-scale production, but findings from this method drive progress in alternative, scalable synthesis methods.
2.2 CVD
CVD enables the controlled growth of TMDC layers—materials composed of transition metals and chalcogens—on various substrates. This method offers scalability and uniformity, which are essential for practical applications in electronics, optoelectronics, and biomedicine (Fig. 1). The process begins by placing a substrate, often a silicon wafer coated with silicon dioxide or another suitable material, inside a CVD chamber. Transition metal precursors, such as molybdenum oxide or tungsten oxide, and sulfur or selenium, which are sources of the chalcogen element, are introduced in solid or gas form. The chamber is then heated to high temperatures, usually between 600 °C and 900 °C, under a controlled atmosphere. Carrier gases such as argon or nitrogen flow through the chamber and transport the vaporized precursors to the substrate. Upon reaching the substrate, the metal and chalcogen vapors react to form TMDC layers that adhere to the surface.10 Process parameters, including temperature, pressure, gas flow rates, and precursor ratios, are carefully controlled to achieve the desired thickness and morphology.11,12 After growth, the system is allowed to cool before the substrate is removed with the synthesized TMDC layers. | ||
| Fig. 1 CVD of TMDCs. A shows the growth mechanism of AA stacking MoS2 bilayer grain and representative low-magnification optical images of MoS2 domains grown for 25 min, demonstrating the morphology during the CVD method for preparing TMDC.10 Copyright© American Chemical Society. B illustrates the general structure of TMDC materials and their common phases.13 Copyright© MDPI. C presents the microscopic images of hBN and WSe2 sheets that have undergone TFD coating treatment during the CVD process, along with their surrounding environment.14 Copyright© Royal Society of Chemistry. D iIllustrates a preparation method for achieving the desired thickness and morphology, as well as the electronic band structure analyzed by ARPES.11 Copyright© Royal Society of Chemistry. E shows a key technology that facilitates the exploration of carrier dynamics in 2D materials.11 Copyright© Royal Society of Chemistry. F shows a detailed process of growing WS2 flakes on a SiO2 surface.12 Copyright© Springer Nature. These figures are intended to provide a more vivid description of CVD. | ||
Building on these process advantages, CVD is particularly useful for producing large-area TMDC (transition metal dichalcogenide) films. This makes it suitable for industrial applications. The technique provides uniform layer thickness and high crystalline quality. These features are essential for device fabrication. Additionally, CVD offers scalability, precise thickness control, good coverage, and strong electronic properties. This enables the synthesis of heterostructures and complex architectures. However, the CVD process requires precise control over many parameters. This makes it more complex than mechanical exfoliation.13 High-quality CVD setups can be expensive and require specialized vacuum systems and precise temperature control. Despite these challenges, CVD-synthesized TMDCs have applications in electronics, optoelectronics, sensing, and biomedicine.
Applications include the fabrication of transistors, photodetectors, and other electronic devices that benefit from TMDCs' semiconducting properties.14 The development of light-emitting diodes (LEDs), lasers, and photovoltaic cells utilizes the direct bandgap and strong light-matter interaction in TMDCs. Furthermore, CVD-synthesized TMDCs are crucial for creating sensitive and selective sensors for chemical, biological, and environmental monitoring. These materials are also used in bioimaging, drug delivery systems, and biosensors, where their biocompatibility and functionalization are critical.
Appel and colleagues investigated the biocompatibility of 2D TMDC materials, MoS2 and WS2, prepared by mechanical exfoliation and CVD. Their findings revealed no toxic effects on mammalian and bacterial cells, and no significant alterations in ROS levels, suggesting the potential of these materials for medical device manufacturing. Similarly, Chen's team demonstrated the biocompatibility of MoS2 synthesized via CVD for biosensor applications, confirming its suitability as a semiconductor through in vitro and in vivo testing. Their research emphasizes the potential of integrating TMDC nanostructures into biodegradable and water-soluble electronic platforms for biomedical implants. As the understanding and control of CVD processes improve, the range of achievable materials and their applications in electronics, optoelectronics, sensing, and biomedicine is expected to expand significantly.15
2.3 Solution-based chemical exfoliation
Solution-based chemical exfoliation involves dispersing bulk TMDCs in suitable solvents, often assisted by sonication (to agitate particles), chemical reactions, or intercalation (inserting molecules between layers), to obtain thin or even single layers of TMDC in solution. Solution-based chemical exfoliation enables the production of TMDCs with a wide range of thicknesses and lateral sizes, making them suitable for various applications in electronics, optoelectronics, and biomedicine.16The process begins with selecting high-quality bulk TMDC crystals and appropriate solvents. The choice of solvent is crucial, typically selected to match the solvent's surface energy with that of the TMDC material, thereby promoting exfoliation.17 The bulk TMDCs are then dispersed in the selected solvent, followed by sonication (exposure to high-frequency sound waves) or mechanical agitation (vigorous mixing). This step helps intercalate (insert) solvent molecules between the TMDC layers, weakening the van der Waals forces (weak attractions between molecules) that hold the layers together and allowing the exfoliation (separation) of individual layers into solution.18 Chemical intercalation agents, such as alkali metals or organic molecules, can be introduced prior to sonication to further weaken interlayer forces and enhance exfoliation efficiency. After exfoliation, the solution is centrifuged (spun at high speed) or filtered to remove unexfoliated bulk material, yielding a dispersion of TMDC layers with the desired thicknesses and sizes.19 Surface functionalization (adding specific chemical groups) or chemical modification can be performed to enhance the dispersibility, stability, and functionality of exfoliated TMDCs.20
Solution-based chemical exfoliation enables large-scale production of TMDC dispersions. This method is suitable for applications that require substantial material quantities.21 By adjusting exfoliation conditions, TMDCs with controlled thicknesses and lateral dimensions can be synthesized. Exfoliated TMDCs in solution can also be easily functionalized with organic molecules, polymers, or biomolecules. This improves their compatibility and allows for use in specific applications. However, achieving uniform layer thickness and size distribution remains a challenge and may impact TMDC properties and performance. Chemical intercalation agents or solvent residues may also remain, so thorough cleaning is needed to ensure purity.
Thin, flexible TMDC layers produced via solution-based exfoliation are ideal for flexible transistors, sensors, and photodetectors. TMDC dispersions can also be added to polymers or other matrices to improve the mechanical, electrical, or thermal properties of composites.22 Functionalized TMDCs are explored for drug delivery, bioimaging, and biosensing due to their biocompatibility and surface functionality. Solution-based chemical exfoliation offers a scalable, versatile route to TMDC materials for many applications. While challenges in quality and purity persist, research and development focus on refining this technique to unlock the full potential of TMDCs in electronics, optoelectronics, and biomedicine (Fig. 2).
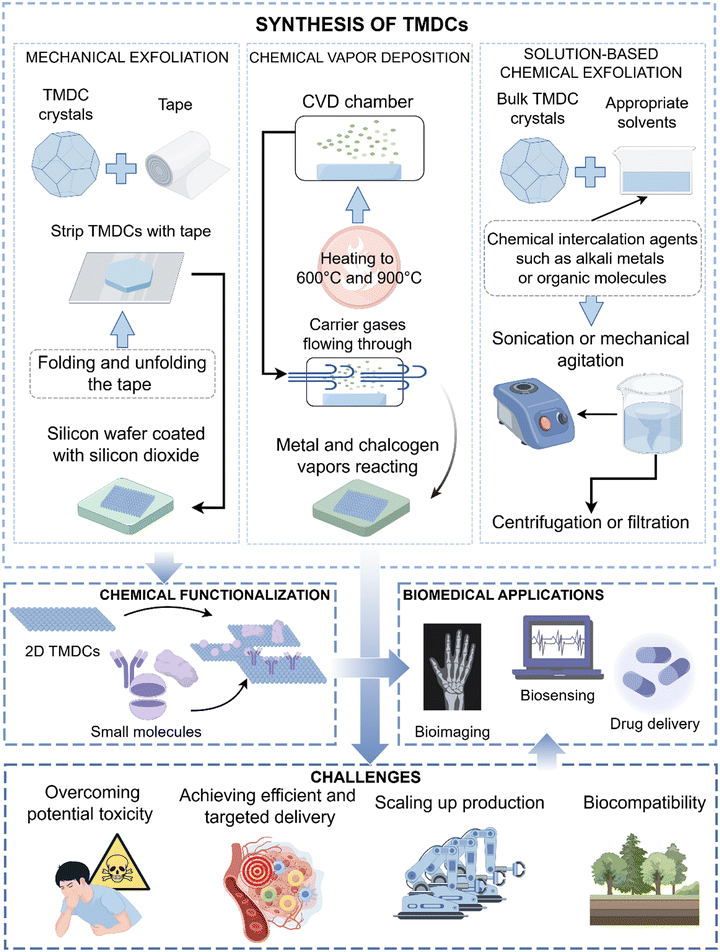 | ||
| Fig. 2 From synthesis to application. Three synthetic methods of TMDCs, chemical functionalization, biological applications and future challenges of TMDCs in the field of biomedicine | ||
3. Characteristics of TMDCs relevant to biomedical application
TMDCs, with their diverse and tunable properties, have garnered significant attention for their potential in biomedical applications. These 2D materials, composed of a transition metal (M) such as Mo or W sandwiched between two chalcogen atoms (X) such as S, Se, or Te, exhibit a variety of electronic, optical, and chemical properties relevant to biomedical applications. The characteristics of TMDCs that are particularly relevant to biomedical applications include:3.1 Biocompatibility
Biocompatibility ensures that TMDCs do not cause adverse reactions when introduced into a biological system. This property makes them safe for medical applications such as drug delivery, biosensors, and bioimaging agents. Goldman et al. examined the biocompatibility of WS2 Inorganic fullerene-like nanoparticles (IFWS2) and multiwall inorganic nanotubes (INT-WS2) in salivary gland cells. Their toxicity assessments in RSC and A5 cells at concentrations of 0.22–100 μg mL−1 showed no significant effects on growth or survival. These microstructures are localized in the cytoplasm and are enclosed by membranes, indicating good biocompatibility and potential medical applications.23 Appel et al. tested the cytotoxicity and genotoxicity of WS2 nanoparticles and nanosheets on S. typhimurium TA100 and HEK293f human kidney cells. Their tests showed that WS2 did not induce high ROS levels linked to cell death. S. typhimurium also showed no significant mutations after exposure to 100 μg mL−1 of WS2 for 24 hours.24Several factors influence the biocompatibility of TMDCs. The type of transition metal and chalcogenide, along with impurities or synthesis residues, can have a significant impact. Materials made with high-purity precursors and thoroughly cleaned after synthesis tend to exhibit better biocompatibility. The physical characteristics of TMDCs, such as lateral size, thickness, and morphology, also affect their interactions with biological systems. Smaller, well-defined nanoparticles can interact differently with cells compared to larger or irregularly shaped materials. Functionalizing TMDCs with biocompatible ligands or polymers can enhance their biocompatibility.25 Surface modifications help prevent direct interactions with cells and tissues, reduce toxicity, and improve solubility and stability in biological fluids. Biocompatibility also depends on concentration and exposure time; lower concentrations and shorter exposure times usually cause fewer adverse effects. Biocompatible TMDCs can carry therapeutic agents, enabling targeted delivery and controlled release to minimize side effects and enhance treatment efficacy. TMDCs with good biocompatibility also act as contrast agents in fluorescence, photoacoustic, and MRI imaging. This provides insight into biological processes without adverse effects. Biosensors made with biocompatible TMDCs can sensitively and selectively detect biomolecules, aiding in disease diagnosis and monitoring.26 Ongoing efforts to enhance biocompatibility through material design and functionalization are crucial for TMDC applications in healthcare. Functionalization with molecules such as polyethylene glycol (PEG), peptides, or antibodies can control TMDC interactions with biological systems for specific uses.27 For example, doxorubicin can adsorb onto MoS2 nanosheets, thereby improving their targeting and killing of cancer cells.28 Large molecules such as DNA and proteins can also bind TMDC surfaces via non-covalent interactions, enabling DNA detection.29 Functionalization improves solubility and stability, reduces non-specific binding, and enables targeting of specific cells or tissues for drug delivery or imaging.
3.2 High surface area
TMDCs' high surface area facilitates substantial loading of therapeutic agents, including drugs, genes, and proteins, enabling more efficient delivery per unit mass, potentially increasing treatment efficacy.30 The surface can also be chemically modified to control the release kinetics of loaded agents, enabling sustained release over time or responsive release triggered by specific stimuli (e.g., pH changes, enzymes, or light), which is particularly beneficial for targeted therapy.31 The extensive surface area also enables effective interaction with biomolecules, enhancing biosensing performance. By functionalizing the surface with specific receptors or antibodies, TMDCs can selectively bind to target biomolecules, such as DNA, RNA, proteins, or small molecules, leading to detectable changes in their physical or chemical properties.32 This high degree of interaction enhances the sensitivity of TMDC-based biosensors, allowing for the detection of low concentrations of biomarkers, which is crucial for early diagnosis and disease monitoring.In bioimaging, the large surface allows efficient loading with imaging agents or functionalization with targeting ligands. This improves contrast and specificity of imaging techniques. For fluorescence imaging, TMDCs can be combined with fluorescent dyes or quantum dots to amplify the signal. In photoacoustic imaging, strong optical absorption enables TMDCs to convert light into acoustic signals, yielding high-resolution images of biological tissues.33 The high surface area also promotes interaction with cells and tissues for therapies like PTT or PDT. TMDCs absorb light and create heat or generate ROS, damaging cancer cells while sparing healthy tissue.30
3.3 Optical properties
TMDC materials exhibit unique light absorption, photoluminescence (PL, the emission of light by a substance after absorbing photons), and nonlinear optical responses across the visible to near-infrared (NIR) spectrum, making them valuable for applications such as bioimaging, phototherapy, and biosensing.34 The quantum confinement and direct bandgap in monolayer TMDCs enhance their optical properties, distinguishing them from other nanomaterials and expanding their utility in biomedicine.35TMDCs such as MoS2 and WS2 exhibit strong PL when exfoliated to monolayer thicknesses, owing to their direct band gaps. This property is exploited in fluorescence bioimaging, where TMDCs serve as fluorescent probes or contrast agents.34 They can be functionalized with targeting molecules to bind specifically to biomarkers or cellular structures, providing high-contrast images of biological processes.36 TMDCs' photostability and tunable emission offer advantages over traditional organic dyes and quantum dots, reducing photobleaching and enabling customizable imaging windows. Their broad NIR absorption makes them excellent candidates for PTT, as they efficiently convert absorbed light into heat, inducing localized hyperthermia to selectively destroy cancer cells with minimal damage to healthy tissue. NIR absorption aligns with the biological tissue transparency window, allowing deeper tissue penetration and more effective tumor targeting.5 TMDCs, such as MoS2, have demonstrated high photothermal conversion efficiency, making them promising for minimally invasive cancer treatment.37 Additionally, some TMDCs can generate ROS upon light excitation, useful for PDT. Under specific light irradiation, TMDCs transfer energy to oxygen molecules, producing ROS that induce cell death through oxidative stress.38 TMDCs also exhibit significant nonlinear optical responses, including two-photon absorption, useful in multiphoton fluorescence microscopy. This technique enables deep-tissue imaging with reduced photodamage and photobleaching, providing high-resolution 3D images of biological structures.39
3.4 Electronic and sensing properties
TMDCs can be utilized to fabricate field-effect transistors (FETs) that serve as highly sensitive biosensors.40 The semiconducting nature of TMDCs, combined with their high surface-to-volume ratio, enables effective transduction of biological interactions at the surface into measurable electrical signals. When molecules bind to the surface of a TMDC-based FET, they alter the carrier concentration or surface potential, thereby modulating the device's conductivity. This principle is exploited to detect a wide range of biological analytes, including DNA, RNA, proteins, glucose, and ions, with high specificity and sensitivity. In addition, TMDC-based FETs typically exhibit high on/off current ratios and sensitivity, which are crucial for detecting low concentrations of biomolecules. The thinness of TMDC layers minimizes the distance between the sensing surface and the channel where charge carriers flow, enhancing the sensor's responsiveness to surface-bound molecules.41 This feature enables the early detection of disease biomarkers, potentially enabling prompt intervention and treatment. Beyond FET-based sensing, TMDCs also show promise in electrochemical sensing applications. Their unique electronic properties facilitate electron transfer in electrochemical reactions, making them suitable for the development of electrochemical biosensors.42 These sensors can detect analytes via redox reactions at the TMDC surface, offering another versatile platform for biosensing applications, ranging from monitoring metabolic markers to detecting pollutants and toxins.43 Moreover, the inherent flexibility of TMDCs, along with their electronic properties, makes them ideal candidates for integration into flexible and wearable biosensors. These sensors can conform to the skin or be embedded in textiles, enabling continuous, non-invasive monitoring of physiological parameters such as heart rate, sweat composition, and blood glucose levels.44 The combination of TMDCs' electronic properties with flexible substrates opens new avenues for personalized healthcare and mobile monitoring devices.453.5 Chemical versatility and functionalization
TMDCs can be functionalized via both covalent and non-covalent interactions, enabling attachment of a wide range of functional groups, biomolecules, polymers, and nanoparticles.46 This versatility facilitates the development of TMDC-based materials with enhanced solubility, stability, biocompatibility, and targeted interaction with biological entities. For covalent functionalization, this approach involves forming strong chemical bonds between the TMDC surface and functional molecules. It can be used to attach specific ligands, peptides, or polymers to enhance the dispersion of TMDCs in biological fluids, or to target specific cells or tissues for drug delivery and imaging applications. In contrast, non-covalent interactions, such as π–π stacking, electrostatic interactions, and van der Waals forces, enable reversible attachment of molecules to TMDCs without altering their intrinsic structure. This method is often used to adsorb drugs, DNA, proteins, or fluorescent tags for biosensing and bioimaging, preserving the electronic and optical properties of TMDCs.Besides, the edges of TMDC layers expose unsaturated metal and chalcogen sites that can be chemically functionalized to modify their electronic properties, catalytic activity, or biological interactions.47 Edge functionalization can enhance the reactivity of TMDCs, making them suitable for applications such as enzymatic biosensors and electrocatalysts in bioelectronic devices. Moreover, doping TMDCs with foreign atoms is another strategy to tailor their electronic, optical, and catalytic properties. Substitutional doping (replacing metal or chalcogen atoms) or intercalation doping (inserting atoms or molecules between the layers) can adjust the band structure, charge-carrier concentration, and reactivity of TMDCs, thereby optimizing them for specific sensing or therapeutic applications. Finally, the assembly of heterostructures by stacking different 2D materials, including TMDCs, graphene, and hexagonal boron nitride (h-BN), enables the creation of materials with novel properties not present in their individual components.
3.6 Mechanical properties
Known for its outstanding in-plane stiffness (∼0.2 TPa), single-layer TMDC is ideal for strain engineering due to its low bending modulus (∼9.61 eV) and strong strain endurance (∼10%). These properties arise from strong covalent bonds within the layers. Despite their thinness, TMDCs withstand significant stress without breaking—crucial for durable biomedical devices. Their elasticity allows stretching or compression without permanent deformation, supporting integration into flexible medical implants that adapt to dynamic bodily environments. This flexibility benefits wearable and implantable biosensors,48 enabling devices to conform to the body while maintaining function and comfort. Consistent contact with biological tissues enables accurate monitoring of physiological parameters, such as heart rate, blood pressure, and glucose levels, as well as targeted delivery of therapeutics.49 The mechanical properties of TMDCs underlie their biocompatibility and bioactivity, which are essential for biomedical materials. TMDCs endure physiological conditions without degrading, supporting long-term device stability. Their surfaces can be engineered to promote cellular adhesion and growth, making them suitable scaffolds for tissue engineering. By mimicking the mechanics of natural tissues, TMDCs support the regeneration of damaged skin, bone, or cardiac tissue. TMDCs also combine with polymers, proteins, or hydrogels to form composites with tailored properties,50 merging TMDCs' strength and flexibility with component-specific enhancements such as biocompatibility or environmental responsiveness.51 This enables the use of advanced materials for drug delivery, wound dressings, and regenerative medicine. However, challenges in scalability, integration, and long-term physiological compatibility remain. Future research will focus on optimizing TMDC synthesis, functionalization, composite formation, and in vivo performance.523.7 Environmental stability
Environmental stability allows TMDCs to retain their structure, composition, and function when exposed to diverse environments, including air, moisture, biological fluids, and environments within organisms. Several factors contribute to this stability. Strong covalent bonds within TMDC layers resist environmental degradation. The specific transition metal and chalcogen atoms affect resistance to oxidation and other reactions. Surface modifications and protective coatings, such as polymers, oxides, or organic molecules, strengthen environmental stability by serving as barriers against degradative agents. Functionalizing with biocompatible ligands further improves stability in biological settings. Combining TMDCs with other stable materials, such as graphene or h-BN, yields hybrids that enhance protection against mechanical, chemical, and biological degradation by leveraging the strengths of each material.53For TMDC-based medical devices such as implants and biosensors, environmental stability is key for reliable performance throughout their intended use. Devices that degrade too early may provide inaccurate readings or endanger patients. In drug delivery, TMDC stability ensures controlled therapeutic release without leakage or carrier breakdown. This is crucial for maintaining efficacy and reducing side effects. For bioimaging, stable TMDCs give consistent signals for accurate, long-term biological tracking. Although TMDCs are inherently stable, challenges remain in optimizing them for specific biomedical uses. Concerns such as long-term interactions with tissues, immune responses, and the effects of prolonged TMDC exposure require further study.54 Future directions include new coatings and functionalizations to improve biocompatibility and biofouling resistance, designing TMDC-based hybrids for greater stability, and conducting in vivo studies on stability and biodegradation in biological environments.
Despite their promise, TMDCs in biomedicine face challenges, such as ensuring long-term biocompatibility and stability, achieving targeted delivery, and scaling up production. Future efforts aim to overcome these barriers, explore new functionalizations, develop hybrid materials with other biocompatible substances, and advance device fabrication. TMDCs' unique, tunable properties make them strong candidates for advancing applications like bioimaging, drug delivery, biosensing, and tissue engineering. Continued research is expected to expand their role in new medical technologies, ultimately improving diagnostics, treatments, and patient outcomes (Table 1).
| Characteristic | Description | Biomedical applications | Ref. | ||
|---|---|---|---|---|---|
| Biocompatibility | Minimal cytotoxicity | TMDCs like MoS2 and WS2 have shown low cytotoxicity in various cell lines, making them suitable for in vivo applications | Drug delivery | TMDCs can be used as carriers for targeted drug delivery systems | 111 |
| Surface modification | Biocompatibility can be enhanced by functionalizing TMDCs with biocompatible molecules (e.g., PEGylation) | Tissue engineering | Scaffold materials for cell growth | 111 | |
| Bio-interface interaction | TMDCs interact favorably with biological membranes, proteins, and cells, which is crucial for biomedical applications | Protease detection | Monitoring protease activity by detecting the fluorescence intensity | 112 | |
| High surface area | 2D structure | The 2D layered structure of TMDCs provides a high surface area-to-volume ratio, enhancing interaction with biological molecules | Drug delivery | High loading capacity for therapeutic agents | 111 |
| Enhanced loading capacity | High surface area allows for increased loading of therapeutic agents, drugs, or biomolecules | Biosensors | Enhanced sensitivity due to increased surface interactions. | 113 | |
| Optical properties | Photoluminescence | TMDCs exhibit strong photoluminescence, which is tunable by altering the number of layers, allowing for applications in bioimaging | Bioimaging | Used in fluorescence and photoluminescence imaging | 114 |
| NIR absorption | TMDCs like MoS2 absorb in the NIR range, useful for PTT | PTT | NIR absorption for cancer treatment | 115 | |
| Fluorescence | Strong fluorescence signals enable TMDCs to be used in imaging applications | Biosensors | Functionalized with targeting molecules to provide high-contrast images | 116 | |
| Electronic and sensing properties | High carrier mobility | TMDCs exhibit excellent electronic properties, making them suitable for biosensing applications | Biosensors | Detect biomolecules and physiological parameters | 113 |
| Sensitive detection | High sensitivity to changes in the environment allows TMDCs to detect biomolecules, gases, or ions effectively | Electrochemical sensors | Monitor glucose, pH, and other analytes | 117 | |
| Conductivity | Tunable electronic properties for specific sensing applications by doping or modifying TMDCs | Transistor biosensors | Detect biological analytes including DNA, RNA, proteins, glucose, and ions | 118 | |
| Chemical versatility and functionalization | Surface chemistry | TMDCs can be chemically modified to introduce functional groups that enhance biocompatibility or target specificity | Targeted drug delivery | Functionalization for specific targeting | 111 |
| Functionalization | Enables covalent or non-covalent attachment of drugs, targeting ligands, or fluorescent tags | Hybrid systems | Integration with other materials for multifunctionality | 119 | |
| Versatile chemistry | Ability to form various derivatives and composites with polymers, nanoparticles, or biomolecules | PTT | Producing localized heat from the defect-rich structure to enhance the Fenton process | 120 | |
| Mechanical properties | Flexibility and strength | TMDCs possess high mechanical strength while being flexible, allowing them to conform to biological tissues | Flexible electronics | Wearable and implantable devices | 121 |
| Layered structure | Enables mechanical stability under physiological conditions | Tissue engineering | Scaffolds that mimic the mechanical properties of tissues | 111 | |
| Elasticity | Suitable for flexible and stretchable biomedical devices | Composites | Combine with other material to create composites with tailored properties | 122 | |
| Environmental stability | Chemical stability | TMDCs are resistant to oxidation and degradation under physiological conditions, ensuring long-term stability in biomedical applications | Implantable devices | Long-term stability for implants and sensors | 68 |
| Thermal stability | Maintains structural integrity under a range of temperatures relevant to biological processes | Therapeutic systems | Stability in diverse physiological conditions | 123 | |
| Biological stability | Offers stability in various physiological environments | Drug delivery | Stability ensures the controlled release without unintended leakage or degradation | 111 | |
4. Biomedical applications of TMDCs
TMDCs are a class of 2D materials that have attracted significant interest for their unique properties and potential medical applications. TMDCs, such as molybdenum disulfide (MoS2), tungsten diselenide (WSe2), and their compounds, exhibit diverse electrical, optical, and chemical characteristics, making them suitable for various biomedical applications. Here are some of the notable applications of TMDCs in medicine (Fig. 3).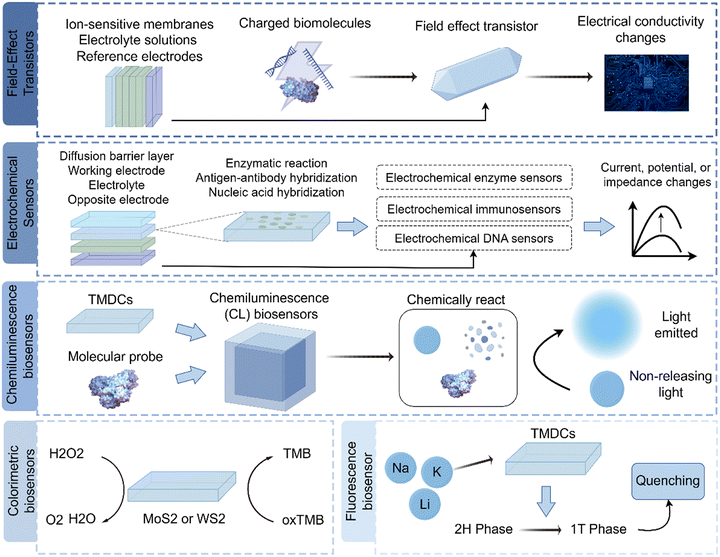 | ||
| Fig. 3 Biomedical applications of TMDCs. TMDCs operate as biosensors through several mechanisms, involving FETs, electrochemical sensors, chemiluminescence biosensor, and fluorescence biosensors. | ||
4.1 Biosensing
TMDCs are promising biosensing materials in biomedicine. They have high surface area, strong electrical and optical features, and chemical versatility.55 Common examples include MoS2, WS2, MoSe2, and WSe2. TMDCs can detect many biological substances with high sensitivity and specificity. They function as biosensors mainly via FETs, electrochemical methods, chemiluminescence, and fluorescence, each using a specific TMDC property.56Electrochemical enzyme sensors use immobilized enzymes as recognition units to detect the corresponding biomolecules. Under enzyme catalysis, biological molecules undergo chemical reactions to produce various ions at different concentrations. The production of this soluble ion alters electrical signals, which can be converted into measurable signals by a conversion unit. Thus, the concentration of the target biological substance is determined indirectly.
An electrochemical immunosensor combines immunology and electrochemical detection; it is a labeled immunoassay. When the antigen/antibody response reaches equilibrium, the electrical signal changes. Electrical signals are obtained through electrodes. Therefore, the measured signal strength is linearly related to the concentration of the target molecule within a certain range.
The principle of electrochemical DNA sensors is nucleic acid hybridization. The ssDNA probe is fixed to the electrode, and at the right temperature, pH, and ionic strength, the probe on the electrode surface can selectively hybridize with the target sequence to form dsDNA. Different interaction forces between ssDNA and dsDNA at the electrode surface can alter the electrode surface structure. The electrode is then used as a signal sensor to detect the hybridization reaction at its surface. Therefore, target sequences or specific genes can be detected by measuring these measurable electrical signals.
To quantitatively compare the performance of different TMDCs sensing platforms, the article presents representative data on their detection limits, linear ranges, response times, etc. (Table 2). TMDCs have extremely high sensitivity and can generally achieve detection limits as low as fM (10−15 M) or even lower, enabling them to detect trace biomarkers and being crucial for early disease diagnosis. Particularly, the FET-type sensors have very fast electrical responses, facilitating real-time monitoring of dynamic biological processes. The table also shows that TMDCs can be used for the detection of various analytes such as proteins, viruses, ions, and small molecules, highlighting the wide applicability of their platform technology.60–62
| Sensing mechanism | TMDC material | Target analyte | Limit of detection (LOD) | Linear range | Response time | Sample matrix | Ref. |
|---|---|---|---|---|---|---|---|
| Field-effect transistor (FET) | MoS2 | Tumor necrosis factor-alpha (TNF-α) | 60 fM | — | — | Buffer | 60 |
| MoS2 | Mercury ion (Hg2+) | 1000 fM | Semi-logarithmic relationship | Reversible and reusable | Aqueous solution | 61 | |
| WSe2 | SARS-CoV-2 spike protein | ∼0.066 fM | — | Real-time response | Phosphate buffer saline (PBS) | 124 | |
| Electrochemical sensor | WS2 | Carcinoembryonic antigen (CEA) | ∼2.78 fM | — | — | Clinical sample | 62 |
| Fluorescence/colorimetric sensor | MoS2 | microRNA | ∼3.4 fM | — | — | — | 62 |
| MoS2/WS2 | Glucose | — | — | — | — | 62 |
TMDCs also have their own advantages and disadvantages compared to other mainstream nanomaterials. Compared to graphene with zero band gap, TMDCs represented by MoS2, which possess natural semiconductor properties, enable FET sensors to have higher switching ratios and lower power consumption. They optimize the signal-to-noise ratio while achieving the detection limit of fM level. In electrochemical and colorimetric sensing, their adjustable energy band structure and abundant edge active sites provide catalytic activity superior to traditional carbon materials. However, compared to MXenes with metallic-level conductivity, the carrier mobility of TMDCs limits their application in ultrafast response sensing, and their long-term biological safety data are still scarce compared to graphene. More importantly, for practical application, TMDCs face challenges in large-scale production and uniformity, and their process maturity is far inferior to that of graphene. Therefore, the core competitiveness of TMDCs is not all-encompassing, but lies in their adjustable direct band gap, enzyme-like catalytic activity, and unique interfaces with biological molecules.
The characteristics of TMDC determine its application. The high surface area and the ability to functionalize TMDCs with specific receptor molecules allow for the development of highly sensitive and selective biosensors. TMDCs can be engineered to detect a wide range of biological targets, from small molecules and ions to large biomolecules like proteins and nucleic acids.63 The electrical and optical properties of TMDCs enable the detection of extremely low analyte concentrations, which is crucial for early disease diagnosis and monitoring.64 The mechanical flexibility of TMDCs enables their incorporation into wearable and implantable sensors, facilitating continuous monitoring of health parameters.65,66 TMDC-based biosensors have been applied in various areas, including disease diagnosis, environmental monitoring, and food safety.67,68 Detecting biomarkers specific to certain diseases, such as cancer, diabetes, and cardiovascular conditions, enables early diagnosis and personalized treatment plans.69 Sensing pollutants, toxins, and pathogens in the environment, ensuring public health and safety. Detecting contaminants, allergens, and pathogens in food products to prevent foodborne illnesses and ensure food quality.
The following situations can lead to the failure of TMDCs. Oxidation can lead to degradation of the material's edges and surface structure, altering its electrical properties; Aggregation can cause nanosheets to cluster due to high surface energy, reducing the effective surface area and dispersion, and affecting the uniformity of sensing; Instability of phase state can directly change the electrical behavior of the material, causing sensor signals to drift or fail; In addition, the electrode interface is prone to be affected by water molecule insertion or metal diffusion in complex environments, resulting in increased contact resistance and signal attenuation, while the material's own defects (such as vacancies, doping) can trap charge carriers, reducing fluorescence efficiency and carrier mobility, and further affecting the sensitivity and stability of the sensor. These factors collectively limit the reliability and service life of TMDC biosensors.
The characteristics of TMDC determine its application. While TMDCs offer significant potential for biosensing, challenges related to stability in biological environments, scalability of sensor fabrication, and integration with existing medical infrastructure need to be addressed. Future research directions include improving stability and biocompatibility, enhancing sensitivity and selectivity, and scalable fabrication. Developing surface modification strategies to enhance the stability and biocompatibility of TMDCs in complex biological fluids. Exploring novel functionalization techniques to increase the sensitivity and selectivity of TMDC-based biosensors for a broader range of analytes.96,108 Developing cost-effective and scalable methods for fabricating TMDC-based biosensors to facilitate widespread adoption.
4.2 Drug delivery
TMDCs are making significant inroads into drug delivery by leveraging their unique physical, chemical, and biological properties. These 2D materials, including MoS2, WS2, and their analogs, offer promising platforms for the controlled release and targeted delivery of therapeutic agents. Their high surface area, ease of functionalization, and favorable interactions with biological molecules enable the development of advanced drug delivery systems to enhance the efficacy and safety of treatments (Fig. 4).56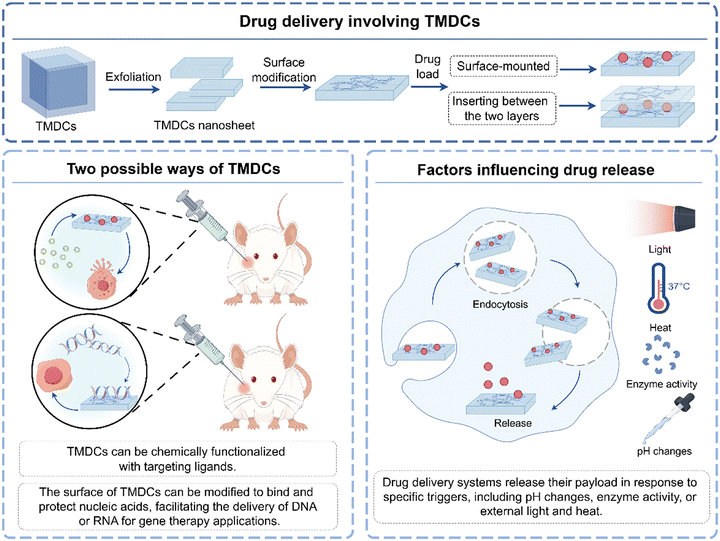 | ||
| Fig. 4 TMDCs mediate drug delivery. When a drug is loaded onto a TMDC, the TMDC interacts with the cell, and the drug is released into the cell by being affected by different factors. | ||
The large surface area and porous structure of TMDCs allow for high loading efficiencies of various drugs, including chemotherapeutic agents, proteins, and nucleic acids.25 Compared with graphene and its derivatives, drugs can not only be adsorbed on the surface but also between the layers of TMDCs, thereby achieving a much larger drug loading capacity. Besides, TMDCs can be chemically functionalized with targeting ligands, such as antibodies, peptides, or small molecules, that recognize specific receptors on the surface of diseased cells, such as cancer cells.70 This specificity facilitates the direct delivery of drugs to the target site, minimizing side effects on healthy tissues and enhancing therapeutic outcomes.71 For instance, studies have shown that by loading enhanced siRNA/doxorubicin drugs onto the two-dimensional planar structure of MoS2-peptide NSs, the mortality rate of C6 glioma cells upon near-infrared light irradiation reached approximately 90%.72 However, there may be differences between in vitro cell experiments and the actual in vivo therapeutic effects. The in vitro experiments are conducted in an environment that is highly complex and different from the in vivo conditions: Firstly, once the nanomaterials enter the bloodstream, they will quickly adsorb plasma proteins and form a “protein cap”, which may completely mask the carefully modified targeting ligands on their surface, causing them to be recognized and cleared by the immune system, thereby significantly reducing the targeting ability and tumor enrichment efficiency. Secondly, the two-dimensional in vitro cell culture models cannot simulate the high interstitial pressure and hypoxic microenvironment of in vivo tumor tissues. These biological physical barriers can severely impede the deep penetration of nanomedicines. Moreover, factors such as the intervention of the immune system (such as the clearance of MPS) and systemic metabolism, which are unique to the in vivo environment, cannot be reflected in simple in vitro models. Therefore, more advanced 3D tumor sphere models, organoids, and in vivo imaging techniques are urgently needed to more accurately predict their in vivo behavior. Moreover, the unique properties of TMDCs enable the design of stimuli-responsive drug delivery systems that release their payloads in response to specific triggers, such as pH changes, enzyme activity, or external light and heat.73 This controlled-release mechanism ensures that drugs are released at the right time and place, thereby maximizing their therapeutic effect. Li's team developed a novel WS2/Au lipid nanocomposite that combines the therapeutic effects of chemotherapy and photohyperthermia, achieving targeted drug delivery and dual-response release via nanotechnology, thereby improving treatment specificity and efficacy while maintaining low systemic toxicity.74 Sobhani et al. further attached fifth-generation dendritic macromolecules to the surface of WS2@NVCL-co-AAm nanocarriers and loaded the antidiabetic drug pioglitazone onto these nanocarriers. They then evaluated the drug's loading and release patterns in a simulated human blood environment (pH 7.4). The study found that the nanocarriers could release up to 98% PG within 6 hours in a simulated human blood environment at 50 °C, and about 18% PG within 6 hours at 37 °C, indicating that the nanocarriers are temperature-sensitive and can control drug release in response to changes in body temperature.75 Meanwhile, functionalized TMDCs have shown good biocompatibility and, in some cases, biodegradability. These properties are crucial for safe in vivo applications, as they minimize toxicity and ensure that TMDCs are metabolized or excreted after fulfilling their drug-delivery function.25
However, a comprehensive assessment of the biological safety of these substances urgently requires a clear understanding of their biological distribution, clearance, and degradation pathways. Studies have shown that after intravenous injection, TMDCs nanosheets that have been surface-modified (such as PEGylation) are mainly captured by the mononuclear phagocytic system (MPS), resulting in significant accumulation in the liver and spleen.76 This is the basis for their passive targeting of tumors, but it also brings the potential risk of long-term retention. Their clearance pathways are highly dependent on size and chemical stability: sufficiently small nanoparticles or degradation products may be excreted through glomerular filtration in urine; while larger aggregates mainly pass through the hepatic-biliary system and are eliminated through feces, and this process may last for several weeks. In terms of degradation, TMDCs may gradually degrade in the physiological environment through oxidation and enzymatic hydrolysis pathways. For example, MoS2 can be oxidized to soluble molybdate and sulfate in the presence of peroxidase (such as myeloperoxidase, MPO). However, the degradation rates and products of different TMDCs (such as WS2, WSe2) vary significantly, and their long-term in vivo fate and safety of metabolic products still require systematic assessment.72 Meanwhile, the long-term in vivo toxicity of all TMDCs and the biological effects of their degradation products (such as metal ions) remain largely unclear. This is the main weakness compared to the mature platforms such as liposomes or albumin nanoparticles that have already entered clinical trials.
TMDCs' ability to carry both therapeutic drugs and diagnostic agents also supports theragnostic applications, combining therapy and imaging for personalized cancer treatment. In addition, the surface of TMDCs can be modified to bind and protect nucleic acids, facilitating the delivery of DNA or RNA for gene therapy applications.77 This approach holds promise for treating genetic disorders by directly correcting defective genes within cells. Moreover, TMDCs such as MoS2 exhibit strong NIR absorption, making them suitable for PTT.78 They can be used to deliver heat-sensitive drugs, releasing them upon NIR irradiation for localized therapy, or as agents for combined drug delivery and photothermal cancer treatment.79 Based on the current publicly available research data, the R&D efforts of TMDC in the field of drug delivery are mainly concentrated on the proof-of-concept stage in the laboratory.
Despite their great potential, the transition of TMDCs-based drug delivery systems to clinical applications still faces several key bottlenecks. Firstly, there is insufficient understanding of their long-term biological safety and in vivo fate. The materials and their degradation products (such as metal ions) may remain in the body for months or even years, potentially triggering chronic inflammation or fibrosis and other systemic diseases.76 Secondly, to move from the laboratory to clinical application, the problem of large-scale production must be solved. The existing synthesis methods are difficult to produce high-quality materials with consistent size, layer number, crystal phase and defect density, and the batch-to-batch variations will directly affect the reproducibility of drug loading and release behavior. Moreover, at the application level, how to enable the carriers to effectively penetrate complex biological barriers such as the blood–brain barrier remains a major challenge. The delivery efficiency of existing functionalization strategies still needs to be improved. Finally, the absence of a regulatory scientific framework constitutes a fundamental constraint.72 Currently, no specific characterization methods, quality standards and review guidelines for such two-dimensional material drugs have been established globally, which brings significant uncertainty to their clinical transformation path.
4.3 Bioimaging
The application of TMDCs in bioimaging offers new avenues for disease diagnosis, tracking therapeutic interventions, and understanding biological processes at the molecular and cellular levels (Table 3).| Contents | Principle | Target object | Specific application | Advantages | Ref. |
|---|---|---|---|---|---|
| Fluorescence imaging | Utilizes TMDCs' photoluminescence for cellular and tissue imaging | Cells and tissues | Intracellular dynamic imaging to diagnose tumor pathologic status and monitor drug distribution | Photostability, tunable emission, and functionalizability | 117, 125 |
| Photoacoustic imaging | Exploits TMDCs' strong NIR absorption to generate acoustic waves | Deep tissue visualization | High-resolution imaging of vascular structures and tumors | Enhanced photoacoustic signals, potential for functional and molecular imaging | 126, 127 |
| Two-photon imaging | Leverages TMDCs' two-photon absorption for deep tissue imaging | Cellular components and processes | Observe the activity of neurons | Reduced photodamage and photobleaching, high spatial and temporal resolution | 128 |
| Magnetic resonance imaging (MRI) | Uses magnetic nanoparticles-functionalized TMDCs as contrast agents | Tissues for diagnostic imaging | Improved contrast and specificity in MRI images | Combines TMDCs' properties with magnetic nanoparticles' contrast | 129, 130 |
The application of TMDC in biological imaging is currently at the laboratory proof-of-concept stage. Before entering clinical research, the issue of material failure needs to be resolved. Oxidation can lead to degradation of the material's edges and surface structure, causing fluorescence quenching or photothermal signal attenuation;85 agglomeration can cause nanosheets to aggregate in the physiological environment due to high surface energy, reducing dispersibility and targeting efficiency, and possibly triggering embolism risks; unstable phase can irreversibly change the optical and electrical properties of the material, resulting in drift of imaging signals.86 These factors jointly restrict the reliability and clinical application prospects of TMDC-based biological imaging probes.
These include ensuring biocompatibility, achieving targeted and efficient accumulation in specific tissues, and preventing rapid clearance from the body. Future research will focus on enhancing biocompatibility, targeted delivery, and multimodal imaging. For instance, strategies are needed to improve the solubility, stability, and biocompatibility of TMDCs for in vivo imaging applications. Researchers are also engineering TMDCs with targeting molecules that recognize specific biomarkers. This can improve their accumulation in diseased tissues and reduce off-target effects. Combining different imaging modalities in TMDC-based systems provides complementary information and can improve diagnostic accuracy and treatment monitoring. TMDCs provide a versatile and powerful platform for bioimaging applications, leveraging their unique optical and electronic properties.
4.4 Photothermal and photodynamic therapy
TMDCs show considerable promise in PTT and PDT. They emerge as effective agents in minimally invasive treatment for various diseases, notably cancer. These 2D materials, such as MoS2, WS2, and their analogs, exploit their unique optical and electronic properties to absorb light. This absorption lets them generate localized heat for PTT or produce ROS for PDT. TMDCs in these therapies provide a targeted approach, minimizing damage to healthy tissues while attacking diseased cells (Table 4).| Aspect | Photothermal therapy (PTT) | Photodynamic therapy (PDT) | Ref. |
|---|---|---|---|
| Mechanism | • Converts NIR light into heat to destroy cancer cells | • Uses light-activated photosensitizers to generate ROS, inducing cell death | 131 |
| Light spectrum | • NIR light | • Specific wavelengths of light to activate photosensitizers | 132 |
| Targeting | • Passive targeting, active | • Same as PTT | 133 |
| • Targeting, ligand targeting | |||
| Minimal side effects | • Precision targeting | • Precise light activation | 133 |
| Therapeutic outcome | • Direct destruction | • Indirect destruction | 134 |
| Combination therapy | • Radiotherapy, shemotherapy, nanomedicine delivery, PDT, gene therapy, immunotherapy | • Radiotherapy, chemotherapy, nanomedicine delivery, PTT, gene therapy, immunotherapy | 135, 136 |
Compared with TMDCs, other materials have their own advantages and disadvantages. Graphene has high drug loading capacity and photothermal stability, but its zero band gap limits its own PDT activity and there is a problem of long-term bio-stagnation;90 MXenes have high photothermal conversion efficiency, but there is a lack of long-term biological safety data, and the surface chemical properties may limit its functionalization strategy;91,92 black phosphorus has excellent biodegradability and photothermal/photodynamic synergy performance, but it has poor environmental stability and is prone to rapid degradation, affecting the persistence of therapeutic efficacy.93 Overall, TMDCs demonstrate unique clinical translation potential in balancing degradation and therapeutic controllability.
TMDC can be designed with specific targeting molecules to direct the treatment towards specific diseased cells, such as immune cells and tumor cells. This approach can reduce collateral damage to healthy tissues and holds great promise for the treatment of cancer and immune-related diseases.89,94,95 Both PTT and PDT are minimally invasive, using light to activate their therapeutic effects, which can be applied externally or delivered via minimally invasive endoscopic techniques.96 TMDCs offer the flexibility to be used in combination therapies, integrating PTT and PDT with drug delivery for enhanced therapeutic outcomes. This multifunctional approach can overcome treatment resistance and achieve synergistic effects in combating diseases.97,98 The inherent or engineered optical properties of TMDCs can also enable real-time imaging of the therapeutic process, enabling monitoring of treatment efficacy and adjustments to therapy protocols.99 Based on current research, the application of TMDC in photothermal therapy and photodynamic therapy is currently mainly in the preclinical (in vivo) experimental verification stage (Fig. 5). While TMDCs present a novel and effective approach to PTT and PDT, challenges remain in their clinical translation, including biocompatibility and toxicity, light penetration, and targeting efficiency. Ensuring the long-term biocompatibility and minimizing potential toxicity of TMDCs, especially with repeated or prolonged exposure, is crucial for safe clinical applications. Enhancing the penetration depth of light for activating TMDCs in deep tissues remains a challenge, requiring innovative approaches such as upconversion nanoparticles or fiber-optic delivery systems. Improving the targeting and accumulation of TMDCs in diseased sites to maximize therapeutic effects while minimizing side effects is an ongoing area of research.
 | ||
| Fig. 5 Application of TMDCs in PTT and PDT therapy. A. The influence of PDT on the immune response.97 Copyright© MDPI. B. During the 9 day treatment with the MoS2–AuNRs-apt solution, the use of different therapies to track the wound closure due to MRPA infection demonstrated that MoS2–AuNRs-apt solution had a positive effect on wound healing.98 Copyright© John Wiley and Sons. C. Illustrates immunogenic cell death (ICD) in MC38 cells after various drug treatments in a self-assembled chimeric cross-linked nanovesicles (CCNVs) experiment by Chen et al.94 Copyright© American Chemical Society. D. Schematic diagram illustrating the mechanism of tumor cell apoptosis induced by PDT and the release of 1MT.95 Copyright© American Chemical Society. E. The release of sorafenib from ROS-activated nanoparticles under laser irradiation to enhance PDT's anti-tumor effects.89 Copyright© Elsevier. F. Displaying element mapping of MOS2–CuO heterogeneous nanocomposites.99 Copyright© Elsevier. The figures illustrate the excellent efficacy of TMDCs in treating specific diseased cells. | ||
5. Tissue engineering and regeneration
The 2D structure, combined with inherent mechanical strength, flexibility, and surface functionality, makes TMDCs promising candidates for developing scaffolds and matrices that support the growth, differentiation, and regeneration of various tissues.100 TMDCs, through their interaction with biological systems, offer potential solutions for repairing and regenerating tissues, ranging from skin and bone to cardiac and neural tissues.101TMDCs can be processed into porous scaffolds that mimic the extracellular matrix (ECM), providing a supportive framework for cell attachment, proliferation, and differentiation. The high surface area of TMDCs facilitates the adsorption of proteins and growth factors, thereby enhancing cell adhesion and signaling.102,103 Moreover, TMDCs can be combined with polymers, hydrogels, and other biocompatible materials to tailor the mechanical and degradation properties of the scaffolds, ensuring they meet the specific requirements of the targeted tissue. The surface of TMDCs can be chemically modified to introduce functional groups that improve interactions with cells and biological molecules. This functionalization can promote specific cellular responses, including enhanced stem cell differentiation and tissue integration.104 Additionally, incorporating TMDCs into scaffolds can enhance the material's conductivity, particularly beneficial for engineering electrically active tissues, such as nerve and cardiac tissue, by facilitating the transmission of electrical signals that guide tissue formation and function. TMDCs incorporated into tissue engineering scaffolds can also serve as platforms for localized drug delivery. Drugs, growth factors, or other bioactive molecules can be loaded onto TMDCs and released in a controlled manner, directly influencing the regenerating tissue. This approach can be used to enhance tissue regeneration, modulate immune responses, or prevent infection at the site of implantation.105
Besides, TMDCs can be applied in tissue engineering and regeneration.51 In bone tissue engineering, TMDCs have shown promise in supporting stem cell growth and osteogenic differentiation, making them suitable for bone tissue engineering applications. Their incorporation into biocompatible scaffolds can enhance bone regeneration in critical-sized defects or fractures. For skin regeneration, scaffolds incorporating TMDCs can promote wound healing by supporting keratinocyte proliferation and angiogenesis while providing antibacterial properties to prevent infection. Regarding cardiac and neural tissue engineering: TMDCs' electrical conductivity makes them attractive for engineering cardiac and neural tissues. They can be used to develop conductive scaffolds that restore electrical signal propagation in damaged heart tissue or support the regeneration of nerve pathways.102,104
Based on the current publicly available research data, the application of TMDC in the fields of tissue engineering and regenerative medicine is currently in a transitional period from the concept verification stage in the laboratory to the preclinical (in vivo) experimental verification stage. The main focus is on verifying the basic biological compatibility of the materials at the cellular level in vitro and assessing their potential to promote tissue regeneration and the actual effects and initial safety of repairing tissue defects such as bone, cartilage, and nerve in live animal models.
Despite their potential, TMDCs in tissue engineering and regeneration face specific challenges: scalability, long-term biocompatibility, and integration with host tissues. Addressing these requires future research to optimize scaffold design and fabrication to achieve precise control of architecture, porosity, and mechanical properties that mimic native tissue environments.106 Research should also prioritize evaluating degradation products for safety, minimizing adverse immune responses or toxicity. Finally, overcoming regulatory, manufacturing, and clinical translation hurdles is essential to advancing TMDC-based solutions from the lab to the clinic.
6. Characteristics and applications of different TMDCs
MoS2 demonstrates excellent biocompatibility and degradability in biomedical applications. Research indicates that its cytotoxicity is significantly lower than that of carbon nanomaterials like graphene oxide.107 After functionalization with molecules such as polyethylene glycol (PEG), MoS2 exhibits enhanced stability in physiological environments and can be metabolized in vivo via the liver and kidneys, eventually being excreted as ions in urine and feces, indicating good biological clearance. These properties make it particularly suitable for high-sensitivity biosensing (e.g., FET, electrochemical sensors with detection limits reaching nM–pM levels) and drug delivery systems. In therapy, MoS2 possesses a moderate photothermal conversion efficiency (approximately 20–40%) suitable for photothermal therapy (PTT), and its high specific surface area supports efficient drug loading.108The most prominent biological characteristics of WS2 are its superior photothermal performance and long-term retention tendency. It exhibits strong light absorption in the near-infrared region and a high photothermal conversion efficiency (PCE), often exceeding that of MoS2, with some systems reaching 40–60%. However, animal studies have shown that PEG-functionalized WS2 nanomaterials primarily accumulate in the reticuloendothelial system, such as the liver and spleen, and remain difficult to be completely metabolized even 30 days post-injection, suggesting a potential risk of long-term retention. Regarding biosafety, some studies suggest that the short-term cytotoxicity of WS2 to human bronchial epithelial cells (BEAS-2B) is relatively controllable, but long-term exposure may inhibit cell proliferation.109 Therefore, WS2 holds greater advantage in applications requiring highly efficient photothermal effects, such as photothermal therapy (PTT)/photodynamic therapy (PDT) and photoacoustic imaging.
The biological properties of WSe2 are closely related to its narrow bandgap semiconductor characteristics, giving it unique advantages in optical theranostics. Its monolayer direct bandgap (∼1.63 eV) is conducive to deep-tissue fluorescence imaging in the near-infrared window. Recent studies involving single-atom doping engineering (e.g., with Cu, Ru) have enabled WSe2 clusters to gain enzyme-mimicking activities (e.g., catalase-like or peroxidase-like), allowing them to regulate the intracellular redox balance and showing great potential for catalytic therapy.110 However, it is noteworthy that among the four materials, WSe2 exhibits relatively higher cytotoxicity in vitro, showing dose-dependent inhibition of viability in human lung cancer cells (A549), which requires attention in applications.107 Thus, WSe2 is more suited for development in cutting-edge fields such as imaging-guided catalytic therapy/photodynamic therapy (Table 5).
| TMDC Materia | Biological characteristics | Preferred synthesis method(s) | Common functionalization strategies | Primary biomedical applications | Representative performance metrics (with sources) | Ref. |
|---|---|---|---|---|---|---|
| MoS2 | Good biocompatibility; high specific surface area | CVD; solution-based chemical exfoliation | Covalent grafting (e.g., PEG, targeting peptides); polymer coating; heterostructure formation with metals/oxides | • Biosensing (FET, electrochemical) | • Limit of detection (LOD): ∼nM–pM for biomarker sensing | 137, 138 |
| • Drug delivery | • Photothermal conversion efficiency (PCE): 20–40% | |||||
| • Photothermal therapy (PTT) | • Drug loading capacity: Up to 150–200% (weight-based) | |||||
| WS2 | Good biocompatibility and low toxicity; strong near-infrared absorption; excellent photothermal conversion efficiency (PCE) | CVD; mechanical Exfoliation | Surface plasmon resonance tuning; NIR-II dye conjugation; phospholipid encapsulation for enhanced biocompatibility | • Photoacoustic imaging | • PCE: typically higher than MoS2, reported in the range of 40–60% | 137 |
| • Synergistic PTT/photodynamic therapy (PDT) | • Photoacoustic signal intensity: 2–3× higher than gold nanorods | |||||
| • Tissue regeneration scaffolds | • Bandgap: tunable, with monolayer being direct; relevant for optical applications | |||||
| WSe2 | The single-layer direct bandgap is beneficial for optical applications in deep layers; the hole conductive property | CVD | Bandgap engineering; lanthanide doping; nanocomposites with upconversion nanoparticles | • NIR fluorescence imaging | • Fluorescence quantum yield: higher than MoS2/WS2, beneficial for deep-tissue imaging | 137, 139 |
| • Photodynamic therapy (PDT) | • X-ray attenuation coefficient: superior to iodine-based contrasts | |||||
| • X-ray-enhanced radiotherapy | • Direct bandgap pressure coefficient (αA): 3.4 ± 0.1 meV kbar−1 |
7. Challenges and future directions
Despite significant advancements, the application of TMDCs in biomedicine faces several challenges that must be addressed to fully harness their potential. Understanding these challenges and exploring future directions is crucial for integrating TMDCs into clinical practice. One of the primary concerns with TMDCs is their biocompatibility and clinical potential toxicity, especially with long-term exposure. The degradation products of TMDCs and their interactions with biological systems need thorough investigation to ensure safety in medical applications. Producing TMDCs with consistent quality on a large scale remains challenging. Achieving uniformity in thickness, composition, and functionalization across batches is crucial for their reliable use in biomedical applications. For drug delivery and bioimaging applications, targeting TMDCs to specific cells or tissues with high efficiency and minimal off-target effects is a significant challenge. Enhancing targeting capabilities through surface modification and functionalization needs further development. Maintaining the stability of TMDCs in the complex and dynamic biological environment is challenging. Strategies to prevent rapid degradation or clearance from the body, while retaining their functional properties, are essential. Integrating TMDC-based technologies into existing medical frameworks and ensuring compatibility with current diagnostic and therapeutic protocols requires comprehensive evaluation and optimization.Therefore, further research is recommended to develop new methods for surface functionalizing TMDCs to improve their solubility, biocompatibility, targeting accuracy, and therapeutic efficacy. Exploring the development of hybrid materials that combine TMDCs with other biocompatible materials (e.g., polymers, liposomes, or other 2D materials) to enhance their properties and overcome current limitations. Conducting extensive in vivo studies and clinical trials to assess the long-term safety, efficacy, and therapeutic outcomes of TMDC-based biomedical applications, facilitating their translation from the lab to the clinic. Advancing the understanding of the interaction between TMDCs and biological systems at the molecular and cellular levels to design more effective and safer biomedical applications. Establishing clear regulatory pathways for the approval of TMDC-based medical products, ensuring that they meet safety and efficacy standards for clinical use. Promoting collaboration among material scientists, biologists, physicians, and engineers to address the multifaceted challenges of applying TMDCs in biomedicine and to accelerate the translation of research findings into practical applications.
8. Conclusion
TMDCs are a new class of 2D materials with exceptional properties that can transform biomedical applications. Their atomic thickness, high surface-to-volume ratio, tunable electronic and optical characteristics, and strong mechanical strength make them ideal for biosensing, drug delivery, bioimaging, and photothermal or photodynamic therapies. TMDCs offer strong biocompatibility and versatile chemistry, enabling functionalization to enhance interactions with biological systems and deliver precise, targeted therapies and diagnostics. They also fit well in flexible, wearable devices, supporting personalized healthcare. However, challenges remain in improving their biocompatibility, stability, scalability, and targeting for clinical use. Researchers must focus on advanced synthesis methods, surface modifications, and thorough in vivo studies to ensure safe and effective use in medicine. Interdisciplinary research combining materials science, biology, engineering, and medicine will drive the development of next-generation diagnostic and therapeutic tools. As research advances, TMDC-based systems will continue to evolve and could revolutionize healthcare through new solutions for disease detection, treatment, and tissue regeneration. The unique properties of TMDCs, along with progress in their functionalization and clinical applications, position them as key materials in modern medical technologies, potentially redefining biomedicine in the coming years.Author contributions
Y. T.: conceptualization, writing – review & editing, visualization, organizing the tables. Z. H.: conceptualization, writing – original draft & organizing the tables, visualization. K. T.: conceptualization, visualization. F. L.: conceptualization, funding, supervision, writing – review & editing. All authors have read and approved the article.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Abbreviations
| 2D | Two-dimensional |
| TMDCs | Transition metal dichalcogenides |
| Mo | Molybdenum |
| W | Tungsten |
| S | Sulfur |
| Se | Selenium |
| Te | Tellurium |
| NIR | Near-infrared |
| CVD | Chemical vapor deposition |
| PTT | Photothermal therapy |
| PDT | Photodynamic therapy |
| ROS | Reactive oxygen species |
| AFM | Atomic force microscopy |
| ALD | Atomic layer deposition |
| LEDs | Light-emitting diodes |
| PL | Photoluminescence |
| FETs | Field-effect transistors |
| IF-WS2 | Inorganic fullerene-like tungsten disulfide |
| INT-WS2 | Inorganic tungsten disulfide nanotubes |
| PEG | Polyethylene glycol |
| MRI | Magnetic resonance imaging |
| h-BN | Hexagonal boron nitride |
| ECM | Extracellular matrix |
| MOSFETs | Metal-oxide-semiconductor field-effect transistors |
| TMB | Tetramethylbenzidine |
| ELISA | Enzyme-linked immunosorbent assay |
| GOx | Glucose oxidase |
| PAI | Photoacoustic imaging |
| Cu2O | Cuprous oxide |
| MoS2 | Molybdenum disulfide |
| CL | Chemiluminescence |
Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by the Applied Basic Research Project of Sichuan Province (Grant number 2022NSFSC1345).References
- S. Nisar, G. Dastgeer, Z. M. Shazad, M. W. Zulfiqar, A. Rasheed, M. Z. Iqbal, K. Hussain, I. Rabani, D. Kim, A. Irfan and A. R. Chaudhry, 2D Materials in Advanced Electronic Biosensors for Point-of-Care Devices, Adv. Sci., 2024, 11, 2401386 CrossRef.
- S. Anju and P. V. Mohanan, Biomedical applications of transition metal dichalcogenides (TMDCs), Synth. Met., 2021, 271, 116610 CrossRef CAS.
- S. Tajik, Z. Dourandish, F. Garkani Nejad, H. Beitollahi, P. M. Jahani and A. Di Bartolomeo, Transition metal dichalcogenides: Synthesis and use in the development of electrochemical sensors and biosensors, Biosens. Bioelectron., 2022, 216, 114674 CrossRef CAS PubMed.
- B. L. Li, R. Li, H. L. Zou, K. Ariga, N. B. Li and D. T. Leong, Engineered functionalized 2D nanoarchitectures for stimuli-responsive drug delivery, Mater. Horiz., 2020, 7, 455–469 RSC.
- S. Meng, Y. Zhang, H. Wang, L. Wang, T. Kong, H. Zhang and S. Meng, Recent advances on TMDCs for medical diagnosis, Biomaterials, 2021, 269, 120471 CrossRef CAS PubMed.
- A. Gkountaras, Y. Kim, J. Coraux, V. Bouchiat, S. Lisi, M. W. Barsoum and T. Ouisse, Mechanical Exfoliation of Select MAX Phases and Mo4 Ce4 Al7 C3 Single Crystals to Produce MAXenes, Small, 2020, 16, e1905784 CrossRef PubMed.
- K. DiCamillo, S. Krylyuk, W. Shi, A. Davydov and M. Paranjape, Automated Mechanical Exfoliation of MoS2 and MoTe2 Layers for 2D Materials Applications, IEEE Trans. Nanotechnol., 2019, 18, 144–148 Search PubMed.
- Y. Sozen, J. J. Riquelme, Y. Xie, C. Munuera and A. Castellanos-Gomez, High-Throughput Mechanical Exfoliation for Low-Cost Production of van der Waals Nanosheets, Small Methods, 2023, 7, e2300326 Search PubMed.
- L. Ali, F. Subhan, M. Ayaz, S. S. ul Hassan, C. C. Byeon, J. S. Kim and S. Bungau, Exfoliation of MoS2 Quantum Dots: Recent Progress and Challenges, Nanomaterials, 2022, 12, 3465 Search PubMed.
- X. Luo, Z. Peng, Z. Wang and M. Dong, Layer-by-Layer Growth of AA-Stacking MoS2 for Tunable Broadband Phototransistors, ACS Appl. Mater. Interfaces, 2021, 13, 59154–59163 Search PubMed.
- F. Liu, Time- and angle-resolved photoemission spectroscopy (TR-ARPES) of TMDC monolayers and bilayers, Chem. Sci., 2023, 14, 736–750 Search PubMed.
- J. T. Mlack, P. Masih Das, G. Danda, Y.-C. Chou, C. H. Naylor, Z. Lin, N. P. López, T. Zhang, M. Terrones, A. T. C. Johnson and M. Drndić, Transfer of monolayer TMD WS2 and Raman study of substrate effects, Sci. Rep., 2017, 7, 43037 Search PubMed.
- M. F. Hossen, S. Shendokar and S. Aravamudhan, Defects and Defect Engineering of Two-Dimensional Transition Metal Dichalcogenide (2D TMDC) Materials, Nanomaterials, 2024, 14, 410 Search PubMed.
- T. Völzer, A. Schubert, E. von der Oelsnitz, J. Schröer, I. Barke, R. Schwartz, K. Watanabe, T. Taniguchi, S. Speller, T. Korn and S. Lochbrunner, Strong quenching of dye fluorescence in monomeric perylene orange/TMDC hybrid structures, Nanoscale Adv., 2023, 5, 3348–3356 Search PubMed.
- Transition metal dichalcogenide metaphotonic and self-coupled polaritonic platform grown by chemical vapor deposition | Nature Communications, https://www.nature.com/articles/s41467-022-33088-0, (accessed 7 February 2026).
- F. Chen, Y. Luo, X. Liu, Y. Zheng, Y. Han, D. Yang and S. Wu, 2D Molybdenum Sulfide-Based Materials for Photo-Excited Antibacterial Application, Adv. Healthcare Mater., 2022, 11, e2200360 Search PubMed.
- G. Jiang, T. Lin, Y. Qin, X. Zhang, L. Hou, Y. Sun, J. Huang, S. Liu and S. Zhao, Accelerating the peroxidase-like activity of MoSe2 nanosheets at physiological pH by dextran modification, Chem. Commun., 2020, 56, 10847–10850 Search PubMed.
- L. I. Silva, D. A. Mirabella, J. Pablo Tomba and C. C. Riccardi, Optimizing graphene production in ultrasonic devices, Ultrasonics, 2020, 100, 105989 Search PubMed.
- R. T. Busch, L. Sun, D. Austin, J. Jiang, P. Miesle, M. A. Susner, B. S. Conner, A. Jawaid, S. T. Becks, K. Mahalingam, M. A. Velez, R. Torsi, J. A. Robinson, R. Rao, N. R. Glavin, R. A. Vaia, R. Pachter, W. Joshua Kennedy, J. P. Vernon and P. R. Stevenson, Exfoliation procedure-dependent optical properties of solution deposited MoS2 films, npj 2D Mater. Appl., 2023, 7, 12 Search PubMed.
- A. T. Hoang, K. Qu, X. Chen and J.-H. Ahn, Large-area synthesis of transition metal dichalcogenides via CVD and solution-based approaches and their device applications, Nanoscale, 2021, 13, 615–633 RSC.
- High-Throughput Mechanical Exfoliation for Low-Cost Production of van der Waals Nanosheets - Sozen - 2023 - Small Methods - Wiley Online Library, https://onlinelibrary.wiley.com/doi/10.1002/smtd.202300326?af=R, (accessed 7 February 2026).
- D. Bhattacharya, S. Mukherjee, R. K. Mitra and S. K. Ray, Superior piezoelectric performance of chemically synthesized transition metal dichalcogenide heterostructures for self-powered flexible piezoelectric nanogenerator, Nanotechnology, 2023, 34(43) DOI:10.1088/1361-6528/ace97d.
- E. B. Goldman, A. Zak, R. Tenne, E. Kartvelishvily, S. Levin-Zaidman, Y. Neumann, R. Stiubea-Cohen, A. Palmon, A.-H. Hovav and D. J. Aframian, Biocompatibility of tungsten disulfide inorganic nanotubes and fullerene-like nanoparticles with salivary gland cells, Tissue Eng., Part A, 2015, 21, 1013–1023 Search PubMed.
- J. H. Appel, D. O. Li, J. D. Podlevsky, A. Debnath, A. A. Green, Q. H. Wang and J. Chae, Low Cytotoxicity and Genotoxicity of Two-Dimensional MoS2 and WS2, ACS Biomater. Sci. Eng., 2016, 2, 361–367 CrossRef CAS.
- M. Bahri, D. Yu, C. Y. Zhang, Z. Chen, C. Yang, L. Douadji and P. Qin, Unleashing the potential of tungsten disulfide: Current trends in biosensing and nanomedicine applications, Heliyon, 2024, 10, e24427 CrossRef CAS.
- A. S. Sethulekshmi, A. Saritha, K. Joseph, A. S. Aprem and S. B. Sisupal, MoS2 based nanomaterials: Advanced antibacterial agents for future, J. Controlled Release, 2022, 348, 158–185 CrossRef CAS.
- S. Roy, K. A. Deo, K. A. Singh, H. P. Lee, A. Jaiswal and A. K. Gaharwar, Nano-bio interactions of 2D molybdenum disulfide, Adv. Drug Delivery Rev., 2022, 187, 114361 CrossRef CAS PubMed.
- T. Liu, C. Wang, X. Gu, H. Gong, L. Cheng, X. Shi, L. Feng, B. Sun and Z. Liu, Drug delivery with PEGylated MoS2 nano-sheets for combined photothermal and chemotherapy of cancer, Adv. Mater., 2014, 26, 3433–3440 CrossRef CAS.
- F. Mirzapour, M. Shamsipur, K. Ahanjan and A. Pashabadi, Amplification-free sub-femtomolar monitoring of SARS-CoV-2-RNA in serum samples using cDNA-modified MoS2 field-effect transistor, Microchem. J., 2024, 207, 111955 Search PubMed.
- Y. Yang, J. Wu, D. H. Bremner, S. Niu, Y. Li, X. Zhang, X. Xie and L.-M. Zhu, A multifunctional nanoplatform based on MoS2-nanosheets for targeted drug delivery and chemo-photothermal therapy, Colloids Surf., B, 2020, 185, 110585 CrossRef CAS PubMed.
- C. Mo, Z. Wang, J. Yang, Y. Ouyang, Q. Mo, S. Li, P. He, L. Chen and X. Li, Rational assembly of RGD/MoS2/Doxorubicin nanodrug for targeted drug delivery, GSH-stimulus release and chemo-photothermal synergistic antitumor activity, J. Photochem. Photobiol., B, 2022, 233, 112487 Search PubMed.
- P. Štefík, A. Annušová, B. Lakatoš, K. Elefantová, L. Čepcová, M. Hofbauerová, A. Kálosi, M. Jergel, E. Majková and P. Šiffalovič, Targeting acute myeloid leukemia cells by CD33 receptor-specific MoS2-based nanoconjugates, Biomed. Mater., 2021, 16(5) DOI:10.1088/1748-605X/ac15b1.
- L. Liu, H. Jiang, J. Dong, W. Zhang, G. Dang, M. Yang, Y. Li, H. Chen, H. Ji and L. Dong, PEGylated MoS2 quantum dots for traceable and pH-responsive chemotherapeutic drug delivery, Colloids Surf., B, 2020, 185, 110590 Search PubMed.
- Graphene and graphene-like two-denominational materials based fluorescence resonance energy transfer (FRET) assays for biological applications - PubMed, https://pubmed.ncbi.nlm.nih.gov/27342369/, (accessed 7 February 2026).
- Y. Liu, Y. Zhang, W. Zhang, X. Wang, Y. Sun, Y. Huang, P. Ma, J. Ding and D. Song, Ratiometric fluorescent sensor based on MoS2 QDs and AuNCs for determination and bioimaging of alkaline phosphatase, Spectrochim. Acta, Part A, 2021, 262, 120087 Search PubMed.
- H. Dong, S. Tang, Y. Hao, H. Yu, W. Dai, G. Zhao, Y. Cao, H. Lu, X. Zhang and H. Ju, Fluorescent MoS2 Quantum Dots: Ultrasonic Preparation, Up-Conversion and Down-Conversion Bioimaging, and Photodynamic Therapy, ACS Appl. Mater. Interfaces, 2016, 8, 3107–3114 Search PubMed.
- D. Sahoo, S. P. Behera, J. Shakya and B. Kaviraj, Cost-effective synthesis of 2D molybdenum disulfide (MoS2) nanocrystals: An exploration of the influence on cellular uptake, cytotoxicity, and bio-imaging, PLoS One, 2022, 17, e0260955 Search PubMed.
- B. Luo, X. Li, P. Liu, M. Cui, G. Zhou, J. Long and X. Wang, Self-assembled NIR-responsive MoS2@quaternized chitosan/nanocellulose composite paper for recyclable antibacteria, J. Hazard. Mater., 2022, 434, 128896 Search PubMed.
- J. Wang, L. Sui, J. Huang, L. Miao, Y. Nie, K. Wang, Z. Yang, Q. Huang, X. Gong, Y. Nan and K. Ai, MoS2-based nanocomposites for cancer diagnosis and therapy, Bioact. Mater., 2021, 6, 4209–4242 Search PubMed.
- X. Li, J. Yang, H. Sun, L. Huang, H. Li and J. Shi, Controlled Synthesis and Accurate Doping of Wafer-Scale 2D Semiconducting Transition Metal Dichalcogenides, Adv. Mater., 2025, 37, e2305115 CrossRef.
- M. S. Zafar, G. Dastgeer, A. Kalam, A. G. Al-Sehemi, M. Imran, Y. H. Kim and H. Chae, Precise and Prompt Analyte Detection via Ordered Orientation of Receptor in WSe2-Based Field Effect Transistor, Nanomaterials, 2022, 12, 1305 Search PubMed.
- J. Park, J. W. Leem, M. Park, J. O. Kim, Z. Ku, W. Chegal, S.-W. Kang and Y. L. Kim, Heteronanostructured Field-Effect Transistors for Enhancing Entropy and Parameter Space in Electrical Unclonable Primitives, ACS Nano, 2024, 18, 1041–1053 Search PubMed.
- X. Chen, C. Liu and S. Mao, Environmental Analysis with 2D Transition-Metal Dichalcogenide-Based Field-Effect Transistors, Nanomicro Lett, 2020, 12, 95 CAS.
- D. Bhattacharya, S. Mukherjee, R. K. Mitra and S. K. Ray, TMDC ternary alloy-based triboelectric nanogenerators with giant photo-induced enhancement, Nanoscale, 2023, 15, 17398–17408 RSC.
- J. Fan and M. Sun, Transition Metal Dichalcogenides (TMDCs) Heterostructures: Synthesis, Excitons and Photoelectric Properties, Chem. Rec., 2022, 22, e202100313 Search PubMed.
- E. Dornsiepen, F. Pieck, R. Tonner and S. Dehnen, [{(PhSn)3SnS6}{(MCp)3S4}] (M = W, Mo): Minimal Molecular Models of the Covalent Attachment of Metal Chalcogenide Clusters on Doped Transition Metal Dichalcogenide Layers, J. Am. Chem. Soc., 2019, 141, 16494–16500 CrossRef CAS PubMed.
- S. H. Choi, H.-J. Kim, B. Song, Y. I. Kim, G. Han, H. T. T. Nguyen, H. Ko, S. Boandoh, J. H. Choi, C. S. Oh, H. J. Cho, J. W. Jin, Y. S. Won, B. H. Lee, S. J. Yun, B. G. Shin, H. Y. Jeong, Y.-M. Kim, Y.-K. Han, Y. H. Lee, S. M. Kim and K. K. Kim, Epitaxial Single-Crystal Growth of Transition Metal Dichalcogenide Monolayers via the Atomic Sawtooth Au Surface, Adv. Mater., 2021, 33, e2006601 CrossRef.
- M. Shao, H. Sheng, L. Lin, H. Ma, Q. Wang, J. Yuan, X. Zhang, G. Chen, W. Li, Q. Su, E. Xie, J. Wang, Z. Zhang and W. Lan, High-Performance Biodegradable Energy Storage Devices Enabled by Heterostructured MoO3 -MoS2 Composites, Small, 2023, 19, e2205529 Search PubMed.
- J. Dong, H. Yang, J. Zhao, L. Wen, C. He, Z. Hu, J. Li, D. Huo and C. Hou, Sandwich-type microRNA biosensor based on graphene oxide incorporated 3D-flower-like MoS2 and AuNPs coupling with HRP enzyme signal amplification, Microchim. Acta, 2022, 189, 49 Search PubMed.
- Y. Huang, X. Zhai, T. Ma, M. Zhang, H. Yang, S. Zhang, J. Wang, W. Liu, X. Jin, W. W. Lu, X. Zhao, W. Hou, T. Sun, J. Shen, H. Pan, Y. Du and C.-H. Yan, A Unified Therapeutic-Prophylactic Tissue-Engineering Scaffold Demonstrated to Prevent Tumor Recurrence and Overcoming Infection toward Bone Remodeling, Adv. Mater., 2023, 35, e2300313 CrossRef.
- K. A. Deo, M. K. Jaiswal, S. Abasi, G. Lokhande, S. Bhunia, T.-U. Nguyen, M. Namkoong, K. Darvesh, A. Guiseppi-Elie, L. Tian and A. K. Gaharwar, Nanoengineered Ink for Designing 3D Printable Flexible Bioelectronics, ACS Nano, 2022, 16, 8798–8811 CrossRef CAS PubMed.
- H. Chen, X. Zhang, L. Shang and Z. Su, Programmable Anisotropic Hydrogels with Localized Photothermal/Magnetic Responsive Properties, Adv. Sci., 2022, 9, e2202173 CrossRef PubMed.
- Z. Zhang, X. Yang, K. Liu and R. Wang, Epitaxy of 2D Materials toward Single Crystals, Adv. Sci., 2022, 9, e2105201 CrossRef.
- G. Ma, W. Shen, D. S. Sanchez, Y. Yu, H. Wang, L. Sun, X. Wang and C. Hu, Excitons Enabled Topological Phase Singularity in a Single Atomic Layer, ACS Nano, 2023, 17, 17751–17760 CrossRef CAS PubMed.
- H. Sun, D. Li, X. Yue, R. Hong, W. Yang, C. Liu, H. Xu, J. Lu, L. Dong, G. Wang and D. Li, A Review of Transition Metal Dichalcogenides-Based Biosensors, Front. Bioeng. Biotechnol., 2022, 10 DOI:10.3389/fbioe.2022.941135.
- J. Zhang, J. Chen, J. Zuo, J. Lan, Z. Jiang, C. Xiao, X. Wang and Y. Zuo, A novel electrochemical sensor with NiSx@MoS2 composite for efficient NO2− sensing, Food Chem., 2025, 462, 140947 CrossRef CAS PubMed.
- P. Fathi-Hafshejani, N. Azam, L. Wang, M. A. Kuroda, M. C. Hamilton, S. Hasim and M. Mahjouri-Samani, Two-Dimensional-Material-Based Field-Effect Transistor Biosensor for Detecting COVID-19 Virus (SARS-CoV-2), ACS Nano, 2021, 15, 11461–11469 CrossRef CAS PubMed.
- M. Raghunathan, A. Kapoor, P. Kumar, A. Laxshmivarahan, S. C. Tripathi, I. Ahmad and D. B. Pal, Nanostructured transition metal dichalcogenides-based colorimetric sensors: Synthesis, characterization, and emerging applications, Luminescence, 2024, 39, e4833 CrossRef CAS.
- T. Lin, L. Zhong, L. Guo, F. Fu and G. Chen, Seeing diabetes: visual detection of glucose based on the intrinsic peroxidase-like activity of MoS2 nanosheets, Nanoscale, 2014, 6, 11856–11862 RSC.
- M. Sedki, Y. Chen and A. Mulchandani, Non-Carbon 2D Materials-Based Field-Effect Transistor Biosensors: Recent Advances, Challenges, and Future Perspectives, Sensors, 2020, 20, 4811 CrossRef CAS PubMed.
- F. J. Urbanos, S. Gullace and P. Samorì, Field-effect-transistor-based ion sensors: ultrasensitive mercury(II) detection via healing MoS2 defects, Nanoscale, 2021, 13, 19682–19689 RSC.
- N. Chauhan, K. Saxena, M. Tikadar and U. Jain, Recent advances in the design of biosensors based on novel nanomaterials: An insight, Nanotechnol. Precis. Eng., 2021, 4(4) DOI:10.1063/10.0006524.
- L. Huang, A. Krasnok, A. Alú, Y. Yu, D. Neshev and A. E. Miroshnichenko, Enhanced light-matter interaction in two-dimensional transition metal dichalcogenides, Rep. Prog. Phys., 2022, 85(4) DOI:10.1088/1361-6633/ac45f9.
- Z. Qin, J. Zhang and S. Li, Molybdenum Disulfide as Tunable Electrochemical and Optical Biosensing Platforms for Cancer Biomarker Detection: A Review, Biosensors, 2023, 13(9) DOI:10.3390/bios13090848.
- X. Qiao, Y. Cai, Z. Kong, Z. Xu and X. Luo, A Wearable Electrochemical Sensor Based on Anti-Fouling and Self-Healing Polypeptide Complex Hydrogels for Sweat Monitoring, ACS Sens., 2023, 8, 2834–2842 CrossRef CAS PubMed.
- W. Sakong, H. Z. Gul, B. Ahn, S. Oh, G. Kim, E. Sim, J. Bahng, H. Yi, M. Kim, M. Yun and S. C. Lim, Optical Duality of Molybdenum Disulfide: Metal and Semiconductor, Nano Lett., 2022, 22, 5207–5213 CrossRef CAS PubMed.
- A. Axin Liang, B. Huipeng Hou, C. Shanshan Tang, D. Liquan Sun and E. Aiqin Luo, An advanced molecularly imprinted electrochemical sensor for the highly sensitive and selective detection and determination of Human IgG, Bioelectrochemistry, 2021, 137, 107671 CrossRef CAS.
- N. Mphuthi, L. Sikhwivhilu and S. S. Ray, Functionalization of 2D MoS2 Nanosheets with Various Metal and Metal Oxide Nanostructures: Their Properties and Application in Electrochemical Sensors, Biosensors, 2022, 12, 386 CrossRef CAS PubMed.
- P.-S. Ganesh, S. E. Elugoke, S.-H. Lee, H.-U. Ko, S.-Y. Kim and E. E. Ebenso, A bifunctional MoS2/SGCN nanocatalyst for the electrochemical detection and degradation of hazardous 4-nitrophenol, Ecotoxicol. Environ. Saf., 2024, 282, 116701 CrossRef CAS PubMed.
- S. Xu, Y. Zhong, C. Nie, Y. Pan, M. Adeli and R. Haag, Co-Delivery of Doxorubicin and Chloroquine by Polyglycerol Functionalized MoS2 Nanosheets for Efficient Multidrug-Resistant Cancer Therapy, Macromol. Biosci., 2021, 21, e2100233 CrossRef PubMed.
- S. Chibh, N. Aggarwal, N. Gupta, S. Ali, J. Mishra, S. Tiwari, M. E. Ali, D. P. Mishra and J. J. Panda, Photoresponsive and Shape-Switchable MoS2-Peptide-Hybrid Nanosystems for Enacting Photochemo and siRNA-Mediated Gene Therapy in Glioma, ACS Appl. Mater. Interfaces, 2025, 17, 29318–29340 CrossRef.
- S. K. Nandy, S. Das, S. Pandey, P. Kalita, M. K. Gupta, A. Kabra, P. Wadhwa and D. Kumar, The futuristic applications of transition metal dichalcogenides for cancer therapy, Luminescence, 2024, 39, e4771 CrossRef CAS PubMed.
- Z. Omrani, M. Pourmadadi, F. Yazdian and H. Rashedi, Preparation and characterization of pH-sensitive chitosan/starch/MoS2 nanocomposite for control release of curcumin macromolecules drug deliveryapplication in the breast cancer treatment, Int. J. Biol. Macromol., 2023, 250, 125897 CrossRef CAS PubMed.
- J. Li, X. Qi, P. Ye, M. Yang and M. Xie, Construction of WS2/Au-lipid drug delivery system for multiple combined therapy of tumor, J. Drug Delivery Sci. Technol., 2022, 76, 103747 CrossRef CAS.
- M. Sobhani, A. Zieglari, E. Moniri, H. Ahmad Panahi and M. Daghighi Asli, Graft hyper-branched dendrimer onto WS2 nanosheets modified Poly (N-Vinylcaprolactam) as a thermosensitive nanocarrier for Pioglitazone delivery using near-infrared radiation, Int. J. Pharm., 2021, 607, 120985 CrossRef CAS PubMed.
- L. Wang, D. Xu, L. Jiang, J. Gao, Z. Tang, Y. Xu, X. Chen and H. Zhang, Transition Metal Dichalcogenides for Sensing and Oncotherapy: Status, Challenges, and Perspective, Adv. Funct. Mater., 2021, 31, 2004408 CrossRef CAS.
- M. R. Kesama and S. Kim, DNA-Nanocrystal Assemblies for Environmentally Responsive and Highly Efficient Energy Harvesting and Storage, Adv. Sci., 2023, 10, 2206848 CrossRef CAS.
- Z. Chen, X. Wei, Y. Zheng, Z. Zhang, W. Gu, W. Liao, H. Zhang, X. Wang, J. Liu, H. Li and W. Xu, Targeted co-delivery of curcumin and erlotinib by MoS2 nanosheets for the combination of synergetic chemotherapy and photothermal therapy of lung cancer, J. Nanobiotechnol., 2023, 21, 333 CrossRef CAS PubMed.
- J. Liu, S. Smith and C. Wang, Photothermal Attenuation of Cancer Cell Stemness, Chemoresistance, and Migration Using CD44-Targeted MoS2 Nanosheets, Nano Lett., 2023, 23, 1989–1999 CrossRef CAS.
- H. S. Choi and H. K. Kim, Multispectral image-guided surgery in patients, Nat. Biomed. Eng., 2020, 4, 245–246 CrossRef.
- J. Chen, C. Liu, D. Hu, F. Wang, H. Wu, X. Gong, X. Liu, L. Song, Z. Sheng and H. Zheng, Single-Layer MoS2 Nanosheets with Amplified Photoacoustic Effect for Highly Sensitive Photoacoustic Imaging of Orthotopic Brain Tumors, Adv. Funct. Mater., 2016, 26, 8715–8725 CrossRef CAS.
- R.-Q. Yang, P.-Y. Wang, K.-L. Lou, Y.-Y. Dang, H.-N. Tian, Y. Li, Y.-Y. Gao, W.-H. Huang, Y.-Q. Zhang, X.-L. Liu and G.-J. Zhang, Biodegradable Nanoprobe for NIR-II Fluorescence Image-Guided Surgery and Enhanced Breast Cancer Radiotherapy Efficacy, Adv. Sci., 2022, 9, e2104728 CrossRef.
- W. Gu, Y. Yan, X. Cao, C. Zhang, C. Ding and Y. Xian, A facile and one-step ethanol-thermal synthesis of MoS2 quantum dots for two-photon fluorescence imaging, J. Mater. Chem. B, 2016, 4(1) 10.1039/c5tb01839k.
- J. Zheng, H. Nie, D. Zhang, D. Cao, Z. Xing, B. Li and L. Jia, Molybdenum disulfide-based hyaluronic acid-guided multifunctional theranostic nanoplatform for magnetic resonance imaging and synergetic chemo-photothermal therapy, J. Colloid Interface Sci., 2019, 548, 131–144 CrossRef.
- P.-J. Wyndaele, J.-F. de Marneffe, R. Slaets, B. Groven, A. Franquet, P. Brüner, T. Grehl and S. D. Gendt, 2D TMDC aging: a case study of monolayer WS2and mitigation strategies, Nanotechnology, 2024, 35(47) DOI:10.1088/1361-6528/ad72fb.
- B. Liu, W. Jiang, Y. Ye, L. Liu, X. Wei, Q. Zhang and B. Xing, 2D MoS2 Nanosheets Induce Ferroptosis by Promoting NCOA4-Dependent Ferritinophagy and Inhibiting Ferroportin, Small, 2023, 19, e2208063 CrossRef PubMed.
- D. Zhang, P. Cui, Z. Dai, B. Yang, X. Yao, Q. Liu, Z. Hu and X. Zheng, Tumor microenvironment responsive FePt/MoS2 nanocomposites with chemotherapy and photothermal therapy for enhancing cancer immunotherapy, Nanoscale, 2019, 11, 19912–19922 RSC.
- K. Li, K. Xu, S. Liu, Y. He, M. Tan, Y. Mao, Y. Yang, J. Wu, Q. Feng, Z. Luo and K. Cai, All-in-One Engineering Multifunctional Nanoplatforms for Sensitizing Tumor Low-Temperature Photothermal Therapy In Vivo, ACS Nano, 2023, 17, 20218–20236 CrossRef CAS PubMed.
- X. Sun, Z. Cao, K. Mao, C. Wu, H. Chen, J. Wang, X. Wang, X. Cong, Y. Li, X. Meng, X. Yang, Y.-G. Yang and T. Sun, Photodynamic therapy produces enhanced efficacy of antitumor immunotherapy by simultaneously inducing intratumoral release of sorafenib, Biomaterials, 2020, 240, 119845 CrossRef CAS.
- M. F. Hossen, S. Shendokar and S. Aravamudhan, Defects and Defect Engineering of Two-Dimensional Transition Metal Dichalcogenide (2D TMDC) Materials, Nanomaterials, 2024, 14, 410 CrossRef CAS PubMed.
- A. Koyappayil, S. G. Chavan, Y.-G. Roh and M.-H. Lee, Advances of MXenesPerspectives on Biomedical Research, Biosensors, 2022, 12, 454 CrossRef CAS PubMed.
- F. Seidi, A. Arabi Shamsabadi, M. Dadashi Firouzjaei, M. Elliott, M. R. Saeb, Y. Huang, C. Li, H. Xiao and B. Anasori, MXenes Antibacterial Properties and Applications: A Review and Perspective, Small, 2023, 19, e2206716 CrossRef PubMed.
- M. Zorrón, A. L. Cabrera, R. Sharma, J. Radhakrishnan, S. Abbaszadeh, M.-A. Shahbazi, O. A. Tafreshi, S. Karamikamkar and H. Maleki, Emerging 2D Nanomaterials-Integrated Hydrogels: Advancements in Designing Theragenerative Materials for Bone Regeneration and Disease Therapy, Adv. Sci., 2024, 11, e2403204 CrossRef.
- W. Yang, G. Zhu, S. Wang, G. Yu, Z. Yang, L. Lin, Z. Zhou, Y. Liu, Y. Dai, F. Zhang, Z. Shen, Y. Liu, Z. He, J. Lau, G. Niu, D. O. Kiesewetter, S. Hu and X. Chen, In Situ Dendritic Cell Vaccine for Effective Cancer Immunotherapy, ACS Nano, 2019, 13, 3083–3094 CrossRef CAS PubMed.
- W. Song, J. Kuang, C.-X. Li, M. Zhang, D. Zheng, X. Zeng, C. Liu and X.-Z. Zhang, Enhanced Immunotherapy Based on Photodynamic Therapy for Both Primary and Lung Metastasis Tumor Eradication, ACS Nano, 2018, 12, 1978–1989 CrossRef CAS PubMed.
- Y. Li, G. Dang, M. Rizwan Younis, Y. Cao, K. Wang, X. Sun, W. Zhang, X. Zou, H. Shen, R. An, L. Dong and J. Dong, Peptide functionalized actively targeted MoS2 nanospheres for fluorescence imaging-guided controllable pH-responsive drug delivery and collaborative chemo/photodynamic therapy, J. Colloid Interface Sci., 2023, 639, 302–313 CrossRef CAS.
- W. Domka, D. Bartusik-Aebisher, M. Przygoda, K. Dynarowicz, J. Tomik and D. Aebisher, PDT-Induced Activation Enhanced by Hormone Response to Treatment, Int. J. Mol. Sci., 2023, 24, 13917 CrossRef CAS PubMed.
- J. Lv, B. Li, T. Luo, C. Nie, W. Pan, X. Ge, J. Zheng, Y. Rui and L. Zheng, Selective Photothermal Therapy Based on Lipopolysaccharide Aptamer Functionalized MoS2 Nanosheet-Coated Gold Nanorods for Multidrug-Resistant Pseudomonas aeruginosa Infection, Adv. Healthcare Mater., 2023, 12, e2202794 CrossRef PubMed.
- F. Jiang, B. Ding, S. Liang, Y. Zhao, Z. Cheng, B. Xing, P. Ma and J. Lin, Intelligent MoS2-CuO heterostructures with multiplexed imaging and remarkably enhanced antitumor efficacy via synergetic photothermal therapy/ chemodynamic therapy/ immunotherapy, Biomaterials, 2021, 268, 120545 CrossRef CAS.
- V. de la Asunción-Nadal, C. Franco, A. Veciana, S. Ning, A. Terzopoulou, S. Sevim, X.-Z. Chen, D. Gong, J. Cai, P. D. Wendel-Garcia, B. Jurado-Sánchez, A. Escarpa, J. Puigmartí-Luis and S. Pané, MoSBOTs: Magnetically Driven Biotemplated MoS2 -Based Microrobots for Biomedical Applications, Small, 2022, 18, e2203821 CrossRef.
- Y. Deng, X. Ouyang, J. Sun, X. Shi, Y. Li, Y. K. Chan, W. Yang and S. Peng, Rapid sterilisation and diabetic cutaneous regeneration using cascade bio-heterojunctions through glucose oxidase-primed therapy, Bioact. Mater., 2023, 25, 748–765 CAS.
- H. Nazari, A. Heirani-Tabasi, E. Esmaeili, A.-M. Kajbafzadeh, Z. Hassannejad, S. Boroomand, M. H. Shahsavari Alavijeh, M. A. Mishan, S. H. Ahmadi Tafti, M. E. Warkiani and N. Dadgar, Decellularized human amniotic membrane reinforced by MoS2-Polycaprolactone nanofibers, a novel conductive scaffold for cardiac tissue engineering, J. Biomater. Appl., 2022, 36, 1527–1539 CrossRef CAS PubMed.
- S. S. Murugan, P. A. Dalavi, S. Surya, S. Anil, S. Gupta, R. Shetty and J. Venkatesan, Fabrication and characterizations of simvastatin-containing mesoporous bioactive glass and molybdenum disulfide scaffold for bone tissue engineering, APL Bioeng., 2023, 7, 046115 CrossRef CAS.
- K. Liang, Y. Xue, B. Zhao, M. Wen, Z. Xu, G. Sukhorukov, L. Zhang and L. Shang, Chirality-Dependent Angiogenic Activity of MoS2 Quantum Dots toward Regulatable Tissue Regeneration, Small, 2023, 19, e2304857 CrossRef.
- Y. Tang, Z. Qin, Y. Zhong, X. Yan, L. Kong, X. Yang, S. Yin, M. Li, Z. Liu and H. Sun, Bioinspired MoS2 Nanosheet-Modified Carbon Fibers for Synergetic Bacterial Elimination and Wound Disinfection, Adv. Healthcare Mater., 2023, 12, e2202270 CrossRef.
- A. Kumar, A. Sood and S. S. Han, Molybdenum disulfide (MoS2)-based nanostructures for tissue engineering applications: prospects and challenges, J. Mater. Chem. B, 2022, 10, 2761–2780 RSC.
- W. Z. Teo, E. L. K. Chng, Z. Sofer and M. Pumera, Cytotoxicity of exfoliated transition-metal dichalcogenides (MoS2 , WS2 , and WSe2 ) is lower than that of graphene and its analogues, Chemistry, 2014, 20, 9627–9632 CrossRef CAS.
- S. Gaba, M. Sahu, N. Chauhan and U. Jain, Unlocking the potential of low-dimensional MoS2 as a smart nanoplatform for environmental technologies, therapeutic strategies, and biomedical sensing, Talanta Open, 2025, 12, 100498 CrossRef.
- L. Zapór, L. Chojnacka-Puchta, D. Sawicka, K. Miranowicz-Dzierżawska and J. Skowroń, Cytotoxic and pro-inflammatory effects of molybdenum and tungsten disulphide on human bronchial cells, Nanotechnol. Rev., 2022, 11, 1263–1272 CrossRef.
- R. Zhao, L. Li, Y. Yan, X. Xue, Y. Huo, T. Liu, H. Wang, C. Liu, S. Zhang, S. Sun and X.-D. Zhang, Single-Atom Modulated Biocatalytic Selectivity on WSe2 Clusters, ACS Appl. Mater. Interfaces, 2025, 17, 38367–38378 CrossRef.
- M. Pourmadadi, A. Tajiki, S. M. Hosseini, A. Samadi, M. Abdouss, S. Daneshnia and F. Yazdian, A comprehensive review of synthesis, structure, properties, and functionalization of MoS2emphasis on drug delivery, photothermal therapy, and tissue engineering applications, J. Drug Delivery Sci. Technol., 2022, 76, 103767 CrossRef.
- H. Huang, W. Feng and Y. Chen, Two-dimensional biomaterials: material science, biological effect and biomedical engineering applications, Chem. Soc. Rev., 2021, 50, 11381–11485 RSC.
- S. Niknam, A. Dehdast, O. Pourdakan, M. Shabani and M. Koohi, Tungsten Disulfide Nanomaterials (WS2 NM) Application in Biosensors and Nanomedicine, A review, Nanomed. Res. J., 2022, 214–226 CAS.
- X. Hu, Y.-S. Zhang, Y.-C. Liu, N. Wang, X.-T. Zeng and L.-L. Zhang, Emerging photodynamic/sonodynamic therapies for urological cancers: progress and challenges, J. Nanobiotechnol., 2022, 20, 437 CrossRef.
- J. Wang, M. Xu, K. Wang and Z. Chen, Stable mesoporous silica nanoparticles incorporated with MoS2 and AIE for targeted fluorescence imaging and photothermal therapy of cancer cells, Colloids Surf., B, 2019, 174, 324–332 CrossRef CAS.
- B. Ryu, A Study of Biomedical Sensors Based on Layered Semiconductors: From Characteristics to Nanofabrication Approaches, 2020 Search PubMed.
- S. Meng, Y. Zhang, H. Wang, L. Wang, T. Kong, H. Zhang and S. Meng, Recent advances on TMDCs for medical diagnosis, Biomaterials, 2021, 269, 120471 CrossRef CAS PubMed.
- M. Sedki, Y. Chen and A. Mulchandani, Non-Carbon 2D Materials-Based Field-Effect Transistor Biosensors: Recent Advances, Challenges, and Future Perspectives, Sensors, 2020, 20, 4811 CrossRef CAS PubMed.
- P. I. P. Soares, J. Romão, R. Matos, J. C. Silva and J. P. Borges, Design and engineering of magneto-responsive devices for cancer theranostics: Nano to macro perspective, Prog. Mater. Sci., 2021, 116, 100742 CrossRef CAS.
- G. Guan, X. Wang, B. Li, W. Zhang, Z. Cui, X. Lu, R. Zou and J. Hu, “Transformed” Fe3S4 tetragonal nanosheets: a high-efficiency and body-clearable agent for magnetic resonance imaging guided photothermal and chemodynamic synergistic therapy, Nanoscale, 2018, 10, 17902–17911 RSC.
- D. Won, J. Bang, S. H. Choi, K. R. Pyun, S. Jeong, Y. Lee and S. H. Ko, Transparent Electronics for Wearable Electronics Application, Chem. Rev., 2023, 123, 9982–10078 CrossRef CAS.
- L. S. Pires, F. D. Magalhães and A. M. Pinto, New Polymeric Composites Based on Two-Dimensional Nanomaterials for Biomedical Applications, Polymer, 2022, 14, 1464 CAS.
- D. Monga, S. Sharma, N. P. Shetti, S. Basu, K. R. Reddy and T. M. Aminabhavi, Advances in transition metal dichalcogenide-based two-dimensional nanomaterials, Mater. Today Chem., 2021, 19, 100399 CrossRef CAS.
- Two-Dimensional-Material-Based Field-Effect Transistor Biosensor for Detecting COVID-19 Virus (SARS-CoV-2) | ACS Nano, https://pubs.acs.org/doi/10.1021/acsnano.1c01188, (accessed 7 February 2026).
- P. Yu, Y. Li, H. Sun, H. Zhang, H. Kang, P. Wang, Q. Xin, C. Ding, J. Xie and J. Li, Mimicking Antioxidases and Hyaluronan Synthase: A Zwitterionic Nanozyme for Photothermal Therapy of Osteoarthritis, Adv. Mater., 2023, 35, e2303299 CrossRef PubMed.
- J. Chen, C. Liu, D. Hu, F. Wang, H. Wu, X. Gong, X. Liu, L. Song, Z. Sheng and H. Zheng, Single-Layer MoS2 Nanosheets with Amplified Photoacoustic Effect for Highly Sensitive Photoacoustic Imaging of Orthotopic Brain Tumors, Adv. Funct. Mater., 2016, 26(47), 8715–8725 CrossRef CAS.
- W. Hu, T. Xiao, D. Li, Y. Fan, L. Xing, X. Wang, Y. Li, X. Shi and M. Shen, Intelligent Molybdenum Disulfide Complexes as a Platform for Cooperative Imaging-Guided Tri-Mode Chemo-Photothermo-Immunotherapy, Adv. Sci., 2021, 8, e2100165 CrossRef PubMed.
- W. Gu, Y. Yan, X. Cao, C. Zhang, C. Ding and Y. Xian, A facile and one-step ethanol-thermal synthesis of MoS2 quantum dots for two-photon fluorescence imaging, J. Mater. Chem. B, 2016, 4, 27–31 RSC.
- J. Liu, J. Zheng, H. Nie, D. Zhang, D. Cao, Z. Xing, B. Li and L. Jia, Molybdenum disulfide-based hyaluronic acid-guided multifunctional theranostic nanoplatform for magnetic resonance imaging and synergetic chemo-photothermal therapy, J. Colloid Interface Sci., 2019, 548, 131–144 CrossRef CAS.
- W. Xie, Q. Gao, D. Wang, Z. Guo, F. Gao, X. Wang, Q. Cai, S. Feng, H. Fan, X. Sun and L. Zhao, Doxorubicin-loaded Fe3O4@MoS2-PEG-2DG nanocubes as a theranostic platform for magnetic resonance imaging-guided chemo-photothermal therapy of breast cancer, Nano Res., 2018, 11, 2470–2487 CrossRef CAS.
- M. Overchuk, R. A. Weersink, B. C. Wilson and G. Zheng, Photodynamic and Photothermal Therapies: Synergy Opportunities for Nanomedicine, ACS Nano, 2023, 17, 7979–8003 CrossRef CAS.
- S. Liu, X. Pan and H. Liu, Two-Dimensional Nanomaterials for Photothermal Therapy, Angew. Chem., Int. Ed., 2020, 59, 5890–5900 CrossRef CAS PubMed.
- Y. Zhang, Y. Feng, Y. Huang, Y. Wang, L. Qiu, Y. Liu, S. Peng, R. Li, N. Kuang, Q. Shi, Y. Shi, Y. Chen, R. Joshi, Z. Wang, K. Yuan and W. Min, Tumor-Targeted Gene Silencing IDO Synergizes PTT-Induced Apoptosis and Enhances Anti-tumor Immunity, Front. Immunol., 2020, 11, 968 CrossRef CAS PubMed.
- L. He, D. Di, X. Chu, X. Liu, Z. Wang, J. Lu, S. Wang and Q. Zhao, Photothermal antibacterial materials to promote wound healing, J. Controlled Release, 2023, 363, 180–200 CrossRef CAS PubMed.
- X. Huang, Y. Lu, M. Guo, S. Du and N. Han, Recent strategies for nano-based PTT combined with immunotherapy: from a biomaterial point of view, Theranostics, 2021, 11, 7546–7569 CrossRef CAS.
- S. K. Mishra, A. C. Dhadve, A. Mal, B. P. K. Reddy, A. Hole, M. K. Chilakapati, P. Ray, R. Srivastava and A. De, Photothermal therapy (PTT) is an effective treatment measure against solid tumors which fails to respond conventional chemo/radiation therapies in clinic, Biomater. Adv., 2022, 143, 213153 CrossRef CAS.
- A. Zavabeti, A. Jannat, L. Zhong, A. A. Haidry, Z. Yao and J. Z. Ou, Two-Dimensional Materials in Large-Areas: Synthesis, Properties and Applications, Nanomicro Lett., 2020, 12, 66 CAS.
- H. Peng, R. Wang, L. Mei, Q. Zhang, T. Ying, Z. Qian, A. B. Farimani, D. Voiry and Z. Zeng, Transition metal dichalcogenide-based functional membrane: Synthesis, modification, and water purification applications, Matter, 2023, 6, 59–96 CrossRef CAS.
- Pressure coefficients for direct optical transitions in MoS2, MoSe2, WS2, and WSe2 crystals and semiconductor to metal transitions | Scientific Reports, https://www.nature.com/articles/srep26663, (accessed 7 February 2026).
Footnote |
| † These authors contributed equally: Yilin Tang and Ziheng Huang. |
| This journal is © The Royal Society of Chemistry 2026 |
