 Open Access Article
Open Access ArticleApplicability of a bipolar electrode arrangement involving ion-selective electrodes in vertical farming, with the self-controlled supply of macro- and microcomponents
Ewa
Jaworska
 ,
Agata
Michalska
,
Agata
Michalska
 and
Krzysztof
Maksymiuk
and
Krzysztof
Maksymiuk
 *
*
University of Warsaw, Faculty of Chemistry, Pasteura 1, 02-093 Warsaw, Poland. E-mail: kmaks@chem.uw.edu.pl
First published on 29th January 2026
Abstract
All-solid-state potentiometric ion-selective electrodes (ISEs) with plastic membranes are attractive analytical tools due to the simplicity of the apparatus and their ease of use. The implementation of ISEs into a bipolar electrode system working in self-powered mode would open new and attractive possibilities, far beyond typical analytical purposes. In the present work, the applicability of such systems for vertical farming, e.g., in hydroponics, is evaluated as a proof of concept. This approach can be used for long-term monitoring of the concentration of electrolytes (macrocomponents, e.g., potassium or chloride ions) in solutions. Another innovative application proposed herein utilizes the properties of the bipolar electrode system to allow for self-adjustment of the concentrations of components via spontaneous charge transfer processes occurring at both poles of the bipolar electrode. We demonstrate that a self-powered bipolar electrode system composed of an active metal (zinc) and an ISE can be applied for the controlled release of minute amounts of microelements, in this case, zinc ions. The ion flux is selectively controlled by a chemical stimulus, namely, the composition and concentration of a neutral electrolyte (macrocomponents), as well as the properties of the bipolar electrode system.
Introduction
Vertical farming is a method of growing plants in vertically and horizontally ordered layers, usually under controlled and optimized conditions.1,2 For this purpose, soilless farming techniques, e.g., hydroponics, are used. The main advantage of vertical farming is its considerably higher crop yield compared to that expected using traditional farming methods, which results from the small area required and the use of beneficial cultivation methods.2 Moreover, this arrangement is separated from the environment and outdoor conditions, i.e., by growing plants in buildings. Therefore, it is not affected by disadvantageous weather conditions, water shortages, etc., nor is it harmful to local animals and plants.3However, due to the soilless conditions, this farming method requires a continuous supply of the nutrients and minerals necessary for growth, which include both macro- and microelements. Therefore, there is increasing demand for the precise monitoring and adjustment of nutrient concentrations, preferably via maintenance-free methods in a self-regulating mode.
We propose an approach to solve this problem through the application of simple devices: bipolar electrode systems working in self-powered mode. A typical bipolar arrangement involves two electrodes connected by a conductor and immersed in the sample solution. Due to the appropriate redox potential difference at both ends (electrodes), a spontaneous redox process (oxidation and reduction at opposite electrodes) can be induced, resulting in current flow between the electrodes.4–6 This concept has found various, mainly analytical applications, e.g., in enzymatic biosensors, enzyme screening7–13 and other types of sensors.14–16 In a recent paper, we proposed the use of such systems in the chemically stimulated oxidation of silver ions for possible medical applications.17
The implementation of ion-selective electrodes into a bipolar electrode arrangement is expected to provide additional improvement. Ion-selective electrodes (ISEs) belong to the most popular group of simple electrochemical sensors. The potential of the ISE (or more precisely, the voltage of a cell containing an ISE and a reference electrode characterized by a constant potential) is selectively and linearly dependent on the logarithm of the concentration of analyte ion (activity) in the sample solution, as described by the Nernst/Nikolsky equation. The significant advantages of ISEs are their high selectivity and signal stability, as well as their cheap and simple instrumentation.18 ISEs and modified ISEs can be applied for environmental analysis, e.g., as sensors working in optical response mode for freshwater analysis.19
It has been found that the application of ISEs in bipolar electrode systems significantly enhances the analytical possibilities of this group of sensors.20–23 A bipolar electrode arrangement has also been used to lower the detection limit for an ISE, in which the ion-selective electrode was bypassed by an active metal (zinc) wire.24
Taking into account the advantages of these simple systems, in this work, we propose different, novel arrangements of bipolar electrodes involving ISEs, extending their applicability far beyond analytical issues. We propose, as a proof of concept, a simple approach that is potentially useful in vertical farming to monitor and control the amounts of chemicals used as well as to supply either macroelements (in the model, potassium or chloride ions) or microelements (e.g., zinc ions). We also propose methods allowing the self-regulation of the concentrations of these components, as well as ways of coupling their concentrations, such that a chemical stimulus (such as an increase in the potassium ion concentration) would affect the supply rate of microelements (e.g., zinc ions).
The first example presented in this work demonstrates the monitoring and control of the electrolyte concentration in solutions. In coulometric mode, this approach can be used for long-term monitoring of electrolyte concentration changes without the need for continuous measurement, e.g., recording changes in the open circuit potential. The second example presents a procedure for the self-adjustment of electrolyte concentrations via the spontaneous charge transfer processes occurring at both poles of the bipolar electrode. In the third example, one of the ISEs is replaced by an active metal for the controlled release of metal ions (in this case, zinc cations), with the rate being selectively controlled by a chemical stimulus (potassium ions) via the use of a potassium-selective ISE.
Experimental
Apparatus and chemicals
In the open circuit potentiometric experiments, a multichannel data acquisition setup and software (Lawson Labs. Inc., 3217 Phoenixville Pike, Malvern, PA 19355, USA) were used to record stable (within ± 0.2 mV) potential readings. The 700 Dosino and 711 Liquino pump systems (Metrohm, Herisau, Switzerland) were used to obtain sequential dilutions of the calibration solutions. For other electrochemical measurements, a galvanostat–potentiostat (CH-Instruments model 760A, Austin, TX, USA) and a conventional three-electrode cell were used.Tetrahydrofuran (THF), poly(vinyl chloride) (PVC), bis(2-ethylhexyl)sebacate (DOS), sodium tetrakis[3,5-bis(trifluoromethyl)phenyl]borate (NaTFPB), the ionophore valinomycin, the monomer pyrrole and inorganic salts were obtained from Merck (Germany).
1,6-Hexanediol diacrylate (HDDA), 2,2-dimethoxy-2-phenylacetophenone (DMPP), n-butyl acrylate (nBA), hexyl acrylate, poly(vinyl alcohol) (PVA), tris(hydroxymethyl)aminomethane (Tris), pyrene (puriss p.a. for fluorescence), 1-(2-pyridylazo)-2-naphthol (PAN), and multiwalled carbon nanotubes (MWCNTs) were purchased from Sigma Aldrich (Germany).
Pyrrole was purified before use by passing it through an alumina gel mini-column. Freshly deionized and distilled water (resistance 18.2 MΩ cm, MilliQ plus, Millipore, Austria) was used throughout this work.
Potassium-selective membranes
The potassium-selective membranes for the ion-selective electrodes (ISE-K) contained 1.3% (m/m) NaTFPB, 2.8% valinomycin, 33.1% poly(vinyl chloride) and 62.8% of the plasticizer DOS. A total of 100 mg of the membrane components was dissolved in 1 mL of THF and applied as described below.Chloride-selective membranes
The chloride-selective membranes for ion-selective electrodes (ISE-Cl) contained 5.1% (m/m) NaTFPB, 32.7% poly(vinyl chloride) and 62.2% of the plasticizer DOS. A total of 100 mg of the membrane components was dissolved in 1 mL of THF and applied as described below.Electrodes
Glassy carbon disc electrodes with an area of 0.07 cm2 were used in the experiments with polypyrrole/membrane electrodes. These electrodes were polished with 0.3 μm Al2O3. A mirror-smooth polish was avoided to prevent peeling of the relatively thick polymer film. A double-junction silver/silver chloride reference electrode with 1 M lithium acetate in the outer sleeve was used.Unless otherwise stated, glassy carbon electrodes were covered with polypyrrole films under a constant potential of 0.9 V from a solution containing 0.1 M KNO3 and 0.5 M pyrrole. The polymerization charge was 200 mC, except where specified. The obtained nitrate-ion-doped polymer exhibits anion-exchanging properties.
Electrodes with solid contact obtained as described above were positioned with the polymer film facing up. Typically, the electrode surface was coated by pipetting 30 μL of a THF solution containing the membrane components, yielding a membrane thickness of ca. 200 μm. Electrodes with membranes were conditioned overnight in a 1 mM KCl solution.
Ag/AgCl electrodes were prepared by immersing a silver wire (diameter 1.0 mm, purity 99.9%, Sigma Aldrich) in a 1 M FeCl3 solution for 10 min and then rinsing it with distilled water.
Zinc wire (diameter 1.0 mm) from Sigma Aldrich with a purity of 99.999% was used.
Coulometric measurements
To measure the spontaneous charge flow in a bipolar electrode system composed of two ISEs connected by a conductor, the setup shown in Fig. 1 (top panel) was used. The ISEs were immersed in separate KCl solutions of the same concentration. Additionally, two silver/silver chloride electrodes were immersed in these solutions and connected with a potentiostat. The charge flowing in the system (between the Ag/AgCl electrodes and the same current between the two ISEs) was measured in two-electrode chronocoulometric mode, applying a voltage of 0 V.Fluorimetric measurements
The poly(n-butyl acrylate) microspheres used in the fluorimetric experiments were prepared using the Hall method,25 according to a procedure described elsewhere.26 The absorption of dyes (either pyrene or PAN) was performed by adding to 1 mL of a microsphere suspension 200 μL of a THF solution containing 0.12 mg of PAN and 0.1 mg of pyrene (resulting in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio). The mixture was placed in an ultrasound bath for 20 min to allow absorption to occur. The microspheres were then centrifuged, and the obtained microspheres were suspended in 1 mL of 1% (w/v) PVA solution. The microspheres were then kept in a refrigerator until use. The concentrations of PAN and pyrene in the microspheres were estimated by taking into account the determined incorporation effectiveness, the amount of microspheres used, and the microsphere volume (assuming that the volume of spheres is equal to the volume of monomers used, taking into account the effectiveness of the polymerization procedure), and calculated to be 4.8 × 10−3 and 5 × 10−3 M, respectively.
1 molar ratio). The mixture was placed in an ultrasound bath for 20 min to allow absorption to occur. The microspheres were then centrifuged, and the obtained microspheres were suspended in 1 mL of 1% (w/v) PVA solution. The microspheres were then kept in a refrigerator until use. The concentrations of PAN and pyrene in the microspheres were estimated by taking into account the determined incorporation effectiveness, the amount of microspheres used, and the microsphere volume (assuming that the volume of spheres is equal to the volume of monomers used, taking into account the effectiveness of the polymerization procedure), and calculated to be 4.8 × 10−3 and 5 × 10−3 M, respectively.
A 1 mL sample of zinc ions was transferred into 2 mL of 0.1 M Tris-buffer solution (pH 7.4), to which 20 μL of a poly(n-butyl acrylate) microsphere dispersion containing pyrene and PAN was then added. This solution was stirred briefly, and its fluorescence emission intensity was measured after 5 min.
Results and discussion
Applicability of bipolar electrodes for monitoring long-term concentration changes
In this part, we propose the application of self-powered bipolar electrodes for monitoring unwanted changes in the concentration of electrolytes on a longer time scale, e.g., due to natural water evaporation. Maintaining the constant composition and concentration of solutions is an important requirement in many application areas, especially vertical farming.Bipolar ion-selective electrode systems (BISE) were obtained by simply short-circuiting two typical ion-selective electrodes with a polypyrrole solid contact. In this case, a potassium-ion-selective electrode (K-ISE) was connected to a chloride-ion-selective electrode (Cl-ISE), and both electrodes were immersed in the same electrolyte solution.
The linear potentiometric characteristics (dependence of potential on the logarithm of activity) of the K-ISE and Cl-ISE electrodes were characterized separately over the KCl concentration range of 0.1 M to 10−5 M, and exhibited nearly Nernstian slopes (positive and negative for K-ISE and Cl-ISE, respectively, Fig. 2). At KCl concentrations higher than 10−2 M, the potential of the K-ISE is higher than that of the Cl-ISE. When the electrodes are connected, after immersion in solution, current flow should occur due to the potential difference between the two ISEs. In this case, electron flow from the Cl-ISE to K-ISE (at KCl concentrations higher than 10−2 M) is expected, because the potential of K-ISE is higher than that of Cl-ISE. However, at KCl concentrations equal to or lower than 10−2 M, the potential of the K-ISE is lower than that of the Cl-ISE, and thus, electron flow in the opposite direction is expected. These electron flow processes are coupled with the oxidation and reduction of the corresponding solid contacts (polypyrrole) of the ISEs, as shown in the bottom panel of Fig. 1 for the example of reduction at the K-ISE.
 | ||
| Fig. 2 (A) Scheme of the self-adjustment of the KCl concentration in the presence of a BISE composed of a K-ISE and Cl-ISE. (B) Individual potentiometric calibration plots for the Cl-ISE and K-ISE. | ||
The quantification of the charge flow between the connected ISEs was carried out in the system shown in Fig. 1, top panel. Fig. 3A shows the dependence of the recorded charge on time over 3600 s. Following more significant changes at the beginning of the experiment, almost linear dependences were obtained, with the slope dependent on the KCl concentration. The linear dependence of the charge on time indicates a constant value of current inside the system. For the 0.1 M KCl solution, the slope is positive. For lower concentrations, the slopes are negative and become increasingly negative with decreasing concentration of KCl. The sign of the linear dependences of charge vs. time strictly corresponds to the expected direction of current flow due to the potential difference between the ISEs, as discussed above. Additionally, Fig. 3B shows the almost linear dependence of the charge recorded for 3600 s on the logarithm of the concentration of KCl (slope = 5 × 10−5 C dec−1, R2 = 0.91).
The observed linear dependences of charge on time and the significant influence of the KCl concentration on the observed slope provide an opportunity for applications related to monitoring possible changes in KCl concentration on a longer time scale. It should be underlined that this approach in direct mode has not been considered before. Although similar applications of ISEs have been introduced by Bakker and coworkers,27 the important benefit of our system is its simplicity and related reliability. We propose here a setup in which a connected K-ISE and Cl-ISE are immersed in the solution under study, with a capacitor implemented between the electrodes (Fig. 1C). In this configuration, the capacitor would accumulate charge due to the internal current flowing between the K-ISE and Cl-ISE, with a constant rate for a constant electrolyte concentration. The recorded charge for a constant concentration of electrolyte, called reference one, can be stored and reported at the end of the measurement. However, if any change in the electrolyte concentration occurs during the measurement, it will affect the flowing charge, as confirmed by Fig. 3. Therefore, at the end of the measurement, any deviation of the recorded charge (read from the capacitor) from the reference value will be an indication of change in the electrolyte concentration during the course of the charge collection.
Taking advantage of the linear dependence of the charge on time as well as the linear dependence of charge on the logarithm of concentration, a simple linear plot can be constructed showing the dependence of the percentage deviation of the recorded charge from the reference value on the change of pC × time. This approach will allow the quantification of concentration changes. From the linear dependence of charge on the logarithm of concentration, the charge value corresponding to a concentration change equal to one logarithmic unit (pC = 1) for a time of 1 h can be determined. This value can then be divided by the charge recorded after 1 h for a selected electrolyte concentration (reference value) and expressed in %. For periods shorter than 1 h, an appropriate fraction of the calculated value will be used (e.g., a fraction of 1/2 for a time of 30 minutes). Advantageously, a positive or negative deviation of the charge indicates a concentration increase or decrease, respectively. As shown in Fig. 4, for 0.1 M KCl, the percent change in charge for a concentration deviation reaching one logarithmic unit and a time duration of 10 minutes (pC × time = 10 minutes) will be 30%, which is the same as that for a concentration change of 0.5 logarithmic unit and 20 minutes (in this case also pC × time = 10 minutes). For a KCl concentration equal to 1 mM, deviations in concentration would result in smaller percentage deviations from the reference charge. Generally, as shown in Fig. 4, the proposed method can report possible electrolyte concentration changes occurring on the time scale of minutes.
 | ||
| Fig. 4 Dependence of the percent deviation of the charge from the reference value on pC × time for KCl concentrations of 0.1 M (black) and 10−3 M (green). | ||
A limitation of this method may be the possible influence of other components in the case of more-complicated samples or insufficiently high capacitance of the applied capacitor. Moreover, in this case, it is not possible to separate the effect of time and concentration change, as the deviation in the charge value reflects the cumulative influence of pC × time.
However, the method is a very simple apparatus-free option that does not require continuous voltage measurement or continuous data collection. Only the inclusion of a capacitor in the circuit is needed, and it would be sufficient to read the charge (or capacitor voltage) just after the measurement.
Applicability of BISEs for the self-correction of undesirable concentration changes
To maintain a constant electrolyte concentration in solutions, both long-term monitoring and an automatic self-adjustment of this concentration would be very advantageous. Therefore, we propose a simple self-operating system that is able to eliminate undesirable small changes in concentration. A bipolar electrode system containing ISEs could also be useful for this application.As above, the proposed bipolar system consists of two poles exhibiting opposite ion-exchange properties, with a BISE containing a K-ISE and Cl-ISE with a polypyrrole solid contact as the model example.
As shown in the potentiometric calibration plots for K-ISE and Cl-ISE with polypyrrole solid contacts in Fig. 2, an intersection point in the linear dependences for K-ISE and Cl-ISE is observed for concentrations between 0.01 M and 0.1 M KCl. For KCl concentrations higher or lower than that corresponding to the intersection point, the internal currents between Cl-ISE and K-ISE flow in opposite directions.
Above the intersection point, the reduction process at the K-ISE is accompanied by the incorporation of potassium ions from the solution, while the oxidation reaction at the Cl-ISE is coupled with the incorporation of a stoichiometric (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) number of chloride ions. Therefore, in summary, KCl is removed from the solution and (in the form of ions) incorporated into the membranes and then the solid contact materials. Conversely, for concentrations lower than that corresponding to the intersection point, the processes will be accompanied by the release of potassium cations from the K-ISE and chloride anions from the Cl-ISE, leading to the release of KCl into the solution. Therefore, if a constant concentration of KCl equal to that at the intersection point is maintained, any deviation from this concentration, e.g., concentration increase due to water evaporation, would trigger a process mitigating this effect (the removal of KCl at too-high KCl concentration or the release of KCl at too-low concentration). Therefore, a kind of feedback effect would be observed. The concentration corresponding to the intersection point, i.e., the constant concentration to be maintained, can be adjusted, e.g., by using different solid contact materials for the ISEs. For instance, replacing the polypyrrole solid contact of the K-ISE with carbon nanotubes results in a shift of the calibration plot for K-ISE to higher potentials and thus a shift of the intersection point to KCl concentration to ∼10−4 M.
1) number of chloride ions. Therefore, in summary, KCl is removed from the solution and (in the form of ions) incorporated into the membranes and then the solid contact materials. Conversely, for concentrations lower than that corresponding to the intersection point, the processes will be accompanied by the release of potassium cations from the K-ISE and chloride anions from the Cl-ISE, leading to the release of KCl into the solution. Therefore, if a constant concentration of KCl equal to that at the intersection point is maintained, any deviation from this concentration, e.g., concentration increase due to water evaporation, would trigger a process mitigating this effect (the removal of KCl at too-high KCl concentration or the release of KCl at too-low concentration). Therefore, a kind of feedback effect would be observed. The concentration corresponding to the intersection point, i.e., the constant concentration to be maintained, can be adjusted, e.g., by using different solid contact materials for the ISEs. For instance, replacing the polypyrrole solid contact of the K-ISE with carbon nanotubes results in a shift of the calibration plot for K-ISE to higher potentials and thus a shift of the intersection point to KCl concentration to ∼10−4 M.
Based on the obtained coulometric results and the linear dependence of charge on time (Fig. 3), the constant current flowing between the K-ISE and Cl-ISE can be estimated. The current magnitude in a 10−4 M KCl solution, which is related to the release of K+ and Cl− ions, is close to 10−7 A. For a sample volume of 1 mL, this current would correspond to an ion release rate of around 5 μM h−1. If the decrease in the actual concentration due to external conditions does not exceed this limit, the changes can be mitigated using the above-described method. Additionally, the current flowing between the electrodes could be tailored; for example, using a thinner membrane would be expected to yield higher currents due to the lower resistance.
The number of ions accumulated or released is limited by the amount of solid contact material. However, for typical layers of conducting polymers, e.g., polypyrrole with a film thickness of a few micrometers, the exchange of charge in the range of 10–20 mC is possible. Again, for a sample volume equal to 1 mL, this amount of charge would correspond to a change of around 0.1–0.2 mM in the ion concentration in the solution.
Due to the internal charge flow, concentration polarization effects are possible. Taking the potential difference of the K-ISE and Cl-ISE and the estimated membrane resistance into account, the initial current expected in the BISE can be calculated from Ohm's law to be around 1 × 10−7 A, which is consistent with the experimental coulometric results (Fig. 3). The polarization effect can be estimated using eqn (1):28
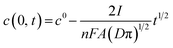 | (1) |
On the other hand, for the above-mentioned charge related to the oxidation/reduction of the solid contact and current value on the order of 10−7 A, the working time of the device can be estimated to be over 20 hours, i.e., one day.
Zinc ion release selectively controlled by a chemical stimulus
A similar arrangement to that described in the above section can be proposed for the precise (in terms of amount and selectivity) release of metal ions, in this example, zinc ions. For this purpose, the above-described BISE arrangement was modified by replacing the Cl-ISE with a zinc wire to obtain a K-ISE–Zn bipolar electrode system. Zinc is an active metal that easily undergoes oxidation, as well as an important microelement in hydroponic farming. Therefore, the proposed bipolar electrode setup is potentially applicable for the continuous, controlled release of zinc ions.For the K-ISE–Zn system, the charge flow was recorded using a method analogous to that used for the BISE with a Cl-ISE and K-ISE (Fig. 1D). Fig. 5A shows the significant and linear dependence of charge on time tested over a time range of 3600 s. The slope of charge vs. time is highly dependent on the KCl concentration; however, the charge flow direction is always the same, related to the oxidation of zinc and reduction of the solid contact of the K-ISE. The dependence of charge on the logarithm of the concentration of KCl (Fig. 5B) is linear over a range of 0.1 M to 10−6 M KCl (slope = 5 × 10−4 C dec−1, R2 = 0.99). Thus, the slope (sensitivity) for the same time range is over 10 times higher than in the case of the BISE.
The maximum charge flowing in the system with zinc is slightly above 2 mC, which corresponds to the oxidation of zinc and reduction of the polypyrrole solid contact. The polymerization charge of the applied polypyrrole solid contact is 200 mC, which corresponds to a maximum oxidation charge of around 20 mC. This indicates that the reported charge of 2 mC is only 10% of the maximum oxidation charge; in other cases (at lower concentrations or in the BISE), this amount will be even lower. Therefore, for this low consumption of oxidized polypyrrole, the linearity of the dependence of charge on time (or current constancy) is not biased.
Additional fluorimetric experiments were carried out to confirm the release of zinc ions into the solution. Fig. 6 presents the fluorescence spectra obtained for the system with zinc and the K-ISE after 24 h of contact with KCl solutions of two concentrations, 0.1 M and 0.01 M (the polymerization charge of the polypyrrole solid contact was 800 mC, to avoid significant reduction of polypyrrole). These spectra reveal two maxima at wavelengths of 370 nm and 393 nm; for concentration determination, the maximum at 393 nm was used. For the electrode immersed in 0.1 M KCl solution, the estimated zinc ion concentration (based on the linear calibration plot of fluorescence intensity on the logarithm of the concentration of zinc ions within the range of 10−7 M to 10−4 M, inset to Fig. 6) was found to be around 3.7 × 10−5 M. The concentration estimated from coulometric results, assuming a linear dependence of charge on time and also determined over a multi-hour time range, was close to 3.4 × 10−5 M. Thus, this value is consistent with that obtained from fluorimetric measurements within the range of experimental error. For 0.01 M KCl, the zinc ion concentration estimated from the fluorimetric measurement was lower (2.2 × 10−5 M), but close to that obtained from coulometric measurement (2.4 × 10−5 M).
In the case of the K-ISE immersed in a 0.1 M NaCl solution, the recorded fluorescence intensity was significantly smaller than that in a 0.1 M KCl solution; this difference was significant at the confidence level of 0.95. The response in 0.1 M NaCl solution is similar to that recorded in water, confirming the selective response of the ion-selective electrode and ultimately the bipolar electrode system with zinc and the K-ISE to potassium ions. In this way, the proposed system enables the release of zinc ions at a constant rate that can be selectively controlled by a chemical stimulus, in this case, the composition and concentration of the applied electrolyte. The recorded charge is quite low as a result of the high resistance of the ion-selective membrane and the relatively small potential difference between the ISE and zinc. However, as shown by Kisiel et al.,24 the magnitude of the flowing charge can be to some extent modified by the properties of the bipolar electrode, e.g., by the resistance incorporated between the K-ISE and zinc, as well as by changing the thickness of the ion-selective membrane.
Due to the observed linear dependence of the charge on time, the above-described system is an apparatus-free source of a constant nA-range current under long-term conditions. This simple source of a constant current could find analytical applications, e.g., for low-current chronopotentiometry. Another advantage is the constant in time release rate of chemicals, e.g., zinc ions. In the present case of the K-ISE–Zn bipolar electrode, as shown in Fig. 5, the flowing charge is on the order of a single mC, corresponding to an increase in the concentration of released zinc ions at a rate of around 10–20 μM h−1 (for a sample volume of 1 mL).
It should be noted that the recorded rate of zinc ion release is consistent with the zinc ion concentration in some liquid fertilizers for green plants and hydroponics (around 30 μM).30 Therefore, the proposed bipolar electrode setup could be useful for the continuous, controlled dosage of the microelement zinc for vertical farming purposes, stimulated by the concentration of microelements, e.g. sodium or potassium ions.
Conclusions
The use of ion-selective electrodes in a self-powered bipolar electrode system offers significant advantages for vertical farming, e.g., hydroponics. They offer the potential for (i) simple long-term monitoring of electrolyte concentration changes and/or (ii) a self-operating mode for adjusting undesirable ion concentration changes by triggering oxidation/reduction processes coupled with ion incorporation or release at the poles, as shown for the example of KCl.Additionally, they can be used for the controlled release of minute amounts of chemicals, with the release rate selectively tailored by the composition and concentration of a neutral electrolyte. The studied example confirmed the applicability of the system to release zinc ions at a rate consistent with vertical farming requirements. Moreover, in the studied system composed of zinc and a potassium-selective ISE, a higher release rate of zinc ions can be obtained by the application of a higher KCl concentration in solution, with discrimination in the presence of NaCl. This effect provides the opportunity to actuate the release rate using other components of the solution.
Author contributions
Ewa Jaworska: investigation, supervision, preparation of figures. Agata Michalska: methodology, supervision, review and editing. Krzysztof Maksymiuk: conceptualization, writing – original draft, writing – review and editing, project administration, funding acquisition.Conflicts of interest
The authors declare that they have no conflict of interest.Data availability
Data for this article are available on request from authors: E-mail: kmaks@chem.uw.edu.pl.Acknowledgements
Financial support from the National Science Centre (NCN, Poland), project 2018/31/B/ST5/02687, in the years 2019–2025, is gratefully acknowledged.References
- J. Birkby, Vertical Farming, ATTRA Sustainable Agriculture Program, Retrieved 15 October 2025.
- K. Benke and B. Tomkins, Sustain. Sci. Pract. Policy, 2017, 13(1), 13–26, DOI:10.1080/15487733.2017.1394054.
- L. M. Navarro and H. M. Pereira, Ecosystems, 2012, 15, 900–912, DOI:10.1007/s10021-012-9558-7.
- R. L. Arechederra and S. D. Minteer, Anal. Bioanal. Chem., 2011, 400, 1605–1611, DOI:10.1007/s00216-011-4782-0.
- M. Zhou, Electroanalysis, 2015, 27, 1786–1810, DOI:10.1002/elan.201500173.
- F. Wu, P. Yu and L. Mao, Chem. Soc. Rev., 2017, 46, 2692–2704, 10.1039/C7CS00148G.
- C. Fischer, A. Fraiwan and S. Choi, Biosens. Bioelectron., 2016, 79, 193–197, DOI:10.1016/j.bios.2015.12.020.
- M. Grattieri and S. D. Minteer, ACS Sens., 2018, 3, 44–53, DOI:10.1021/acssensors.7b00818.
- E. Katz, A. F. Buckmann and I. Willner, J. Am. Chem. Soc., 2001, 123, 10752–10753, DOI:10.1021/ja0167102.
- G. Slaughter and T. Kulkarni, Biosens. Bioelectron., 2016, 78, 45–50, DOI:10.1016/j.bios.2015.11.022.
- D. P. Hickey, R. C. Reid, R. D. Milton and S. D. Minteer, Biosens. Bioelectron., 2016, 77, 26–31, DOI:10.1016/j.bios.2015.09.013.
- A. N. Sekretaryova, V. Beni, M. Eriksson, A. A. Karyakin, A. P. F. Turner and M. Y. Vagin, Anal. Chem., 2014, 86, 9540–9547, DOI:10.1021/ac501699p.
- L. Deng, C. Chen, M. Zhou, S. Guo, E. Wang and S. Dong, Anal. Chem., 2010, 82, 4283–4287, DOI:10.1021/ac100274s.
- X. Zhang, L. Zhang, Q. Zhai, W. Gu, J. Li and E. Wang, Anal. Chem., 2016, 88, 2543–2547, DOI:10.1021/acs.analchem.6b00054.
- X. Zhang, Y. Jing, Q. Zhai, Y. Yu, H. Xing, J. Li and E. Wang, Anal. Chem., 2018, 90, 11780–11784, DOI:10.1021/acs.analchem.8b02838.
- H. Lee, J. Kim, M. Hwang and J. Kim, ACS Sens., 2023, 8, 4374–4383, DOI:10.1021/acssensors.3c01807.
- A. Kisiel, A. Ruszczyńska, A. Michalska and K. Maksymiuk, J. Solid State Electrochem., 2025, 29, 4891–4901, DOI:10.1007/s10008-025-06354-6.
- K. N. Mikhelson, Ion-Selective Electrodes, Springer, 2013 Search PubMed.
- J. Zhai, L. Yang, X. Du and X. Xie, Anal. Chem., 2018, 90, 12791–12795, DOI:10.1021/acs.analchem.8b03213.
- S. Jansod and E. Bakker, Anal. Chem., 2021, 93, 4263–4269, DOI:10.1021/acs.analchem.0c05117.
- Y. Wu and E. Bakker, ACS Sens., 2022, 7, 3201–3207, DOI:10.1021/acssensors.2c01826.
- Y. Wu, A. Qileng and E. Bakker, Sens. Actuators, B, 2023, 396, 134561, DOI:10.1016/j.snb.2023.134561.
- E. Jaworska, A. Michalska and K. Maksymiuk, Electrochim. Acta, 2018, 284, 321–327, DOI:10.1016/j.electacta.2018.07.130.
- A. Kisiel, A. Michalska and K. Maksymiuk, Analyst, 2022, 147, 2764–2772, 10.1039/d2an00458e.
- M. J. Ruedas-Rama and E. A. H. Hall, Analyst, 2006, 131, 1282–1291, 10.1039/b608901a.
- E. Woznica, K. Maksymiuk and A. Michalska, Anal. Chem., 2014, 86, 411–418, DOI:10.1021/ac4033142.
- Y. Wu, Y. Zhang, A. Qileng and E. Bakker, Anal. Chem., 2024, 96, 18401–18407, DOI:10.1021/acs.analchem.4c03081.
- Z. Galus, Fundamental of Electrochemical Analysis, Ellis Horwood, PWN, 1994 Search PubMed.
- R. A. Robinson and R. H. Stokes, Electrolyte Solutions, Butterworth, London, 1959 Search PubMed.
- https://zielony-parapet.pl/nawozy/3684-nawoz-plynny-do-roslin-zielonych-i-hydroponiki-2281967578254.html?srsltid=AfmBOoqQHjYWsftl19bqCt2Y52wWxKGYnJqx1g3RloX_3_JTNbaXojt7, state: 2 July 2025.
| This journal is © The Royal Society of Chemistry 2026 |




