Open Access Article
Open Access ArticleWearable enzyme-free glucose sensor using a flexible sericin-based conductive bio-composite
Xiaorui
Huang†
a,
Yi
Li†
*a,
Boxiang
Yang†
a,
Yuxuan
Wu
a,
Zhuocheng
Jiang
a,
Jiuxi
Sui
a,
Siyi
Xing
a,
Xue
Zhang
a,
Hualin
Lan
c,
Hanyan
Zou
*b,
Yuchan
Zhang
*a and
Guangchao
Zang
 *a
*a
aBiomedical Innovation and Entrepreneurship Practice Base, Lab Teaching & Management Center, Chongqing Medical University, Chongqing, China. E-mail: liyicqmu@cqmu.edu.cn; zhangyc@cqmu.edu.cn; zangguangchao@cqmu.edu.cn
bInstitute for Food and Drug Control, Chongqing, China. E-mail: zouhanyan@cqifdc.org.cn
cChongqing Quality Testing & Inspection Center for Medical Devices, Chongqing, China
First published on 16th January 2026
Abstract
With over 500 million people affected by diabetes worldwide, there is an urgent need for painless, continuous glucose monitoring. However, current wearable sensors are limited by enzyme instability, mechanical rigidity, and poor correlation with blood glucose levels. Here, we present an eco-friendly flexible biosensor fabricated from silk sericin, an abundant cocoon by-product, crosslinked with dialdehyde HPMC and functionalized with AuNPs–AMWCNTs. This dual-crosslinking strategy integrates sericin's biocompatibility with the high conductivity and catalytic activity of the nanocomposite, yielding a robust sensing layer that withstands 200 folding cycles without performance loss. The sensor exhibits a low detection limit of 4 μM and a sensitivity of 13.43 μA mM−1 cm−2 in the physiologically relevant 25–400 μM range. This work offers a sustainable and reliable platform for real-time, continuous, and noninvasive glucose monitoring, with significant potential for next-generation wearable health devices.
1. Introduction
With the increasing interest in body health monitoring, new skin biosensors have attracted more attention and improved rapidly in recent years.1 Among various biosensors and sensing platforms, electrochemical sensors that detect physiological components in sweat and convert them into electronic signals for monitoring life indicators have recently gained much research interest.2 Many disorders, such as diabetes, can be monitored by analyzing glucose levels in sweat. Sweat glands are distributed across the body; thus, sweat can be collected continuously and noninvasively, which is more convenient than collecting other body fluids. Traditional blood glucose testing by finger-prick is invasive and causes discomfort, highlighting the need for less invasive and more user-friendly approaches such as sweat-based flexible skin sensors.Flexible skin sensors are typically fabricated from biocompatible materials, which helps reduce skin irritation.3 In addition, a series of eco-friendly biosensors can be created from natural, biodegradable, and non-toxic materials.4 Sericin, a natural protein derived from silkworm cocoons, exhibits good biocompatibility, low toxicity, and environmental friendliness.5 It contains multiple modifiable chemical groups,6 such as carboxyl, amino, and hydroxyl, giving it great potential for electrochemical detection.7 Hydroxypropyl methyl cellulose (HPMC) is a water-soluble, biocompatible thickening agent with excellent film-forming ability.8 It can be selectively oxidized by periodate to produce dialdehyde HPMC (DHMC).9 Cross-linking sericin with DHMC can theoretically improve its mechanical properties and broaden its application potential.10 Therefore, sericin and DHMC were chosen as the raw materials in this study to prepare a flexible bio-composite membrane.
Improving the electrical conductivity of biosensors is also essential. Amino-functionalized multi-walled carbon nanotubes (AMWCNTs) are widely used due to their strong catalytic activity.11 When modified with metal nanoparticles, AMWCNTs provide abundant catalytic sites for glucose oxidation and show minimal toxicity, making them highly suitable for enzyme-free glucose detection.12 Modifying AMWCNTs with AuNPs further enhances conductivity, dispersibility, and thermal stability.13
In this work, we developed a flexible bio-composite film by combining silk sericin (SS), DHMC, and AuNPs–AMWCNTs to achieve excellent biocompatibility and electrocatalytic performance for enzyme-free glucose detection. The Schiff-base crosslinking reaction ensured strong structural integrity, while the AuNPs–AMWCNT network enhanced electron transfer and catalytic efficiency. The film was further integrated onto a screen-printed electrode to construct a wearable platform for real-time sweat glucose monitoring. This strategy offers a sustainable and reliable approach for continuous, noninvasive glucose sensing, with promising potential for personalized health management.
2. Materials and methods
2.1 Materials and instruments
Silkworm cocoons were obtained from a local sericulture farm in Chongqing, China. Hydroxypropyl methylcellulose (HPMC) was purchased from Tianjin Guangfu Fine Chemical Research Institute. Sodium periodate (NaIO4) and chloroauric acid (HAuCl4·3H2O) were purchased from Shanghai Adamas-beta Co., Ltd. Aminated multi-walled carbon nanotubes (AMWCNTs, purity >95%) were acquired from Nanjing XFNANO Materials Technology Co., Ltd. All other chemicals were of analytical grade and used without further purification.Cell fluorescence imaging was performed using an Eclipse Ti-U inverted fluorescence microscope (Nikon, Japan), while material surface morphologies and elemental compositions were characterized using an SU8010 field-emission scanning electron microscope (Hitachi, Japan) coupled with an X-MaxN energy-dispersive X-ray spectrometer (Oxford Instruments). X-ray diffraction (XRD) patterns were obtained using a D8 ADVANCE diffractometer (Bruker, Germany). Fourier-transform infrared (FTIR) spectra were collected on an iS50 spectrometer (Nicolet, US) over a wavelength range of 400 to 4000 cm−1. Electrochemical tests employed a three-electrode system consisting of the conductive composite film as the working electrode (CHI660E, Shanghai, China), an Ag/AgCl (saturated KCl) reference electrode, and a platinum wire counter electrode. All instruments were calibrated according to the manufacturers' standard protocols prior to use.
2.2 Preparation of the sensor
2.3 Biocompatibility evaluation
2.4 Electrochemical characterization of conductive composite films
The area (1 cm × 1 cm) of composite films with distinct films was detected by CV from −0.55 V to 0.65 V at a scan rate of 100 mV s−1 in a 0.1 mM pH = 7.0 PBS solution and electrochemical impedance spectroscopy (EIS) in a 0.05 M KCl solution containing 0.5 mM [Fe(CN)6]3−/4−. In a stationary solution, linearity tests for DPV and CV were performed. The DPV method was used to examine several interfering chemicals. The anti-jamming ability was assessed by comparing the current value before and after the addition of various interferents.3. Results and discussion
3.1 Physical and chemical characterization of SS–DHMC–AuNPs–AMWCNTs
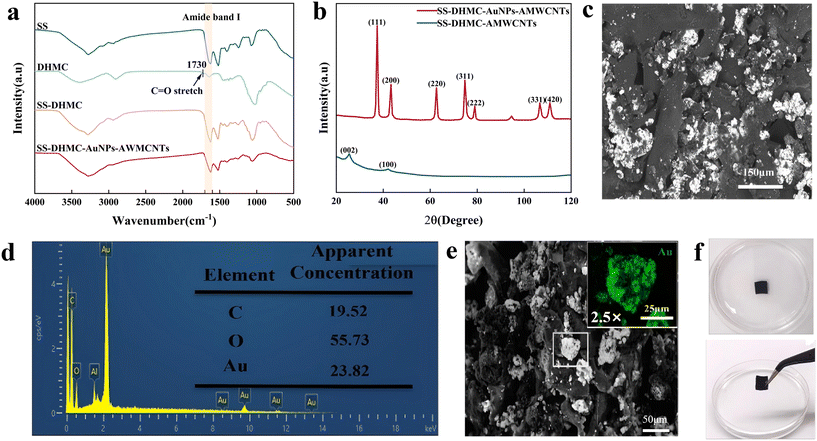
Comprehensive analyses that employ multiple characterization techniques can reveal key information regarding chemical composition, microstructure, crystal structure, and elemental distribution.15 In this study, systematic characterization of the SS–DHMC–AuNPs–AMWCNT composite film was used to verify the preparation of the material and to provide a theoretical foundation for understanding its performance advantages.First, the crosslinking state of the composite film was analyzed by FT-IR spectroscopy (Fig. 2a). Oxidation of HPMC with NaIO4 led to the appearance of an absorption peak at 1730 cm−1 that is not present in the reported spectrum of HPMC.16 This peak was attributed to aldehyde carbonyl vibration, and its presence is consistent with the formation of DHMC. However, the absorption spectrum of SS–DHMC showed no peak at 1730 cm−1, indicating that the aldehyde groups had undergone chemical reactions in the presence of SS.17 The characteristic vibration peak of the Schiff base C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond (around 1660 cm−1) was not observed in SS–DHMC, likely due to masking by the amide I band (1600–1690 cm−1) of SS.18 After crosslinking, the FT-IR spectrum of SS–DHMC–AuNPs–AMWCNTs was consistent with a slight transformation in the secondary structure of SS, from random coil to α-helix.19
N bond (around 1660 cm−1) was not observed in SS–DHMC, likely due to masking by the amide I band (1600–1690 cm−1) of SS.18 After crosslinking, the FT-IR spectrum of SS–DHMC–AuNPs–AMWCNTs was consistent with a slight transformation in the secondary structure of SS, from random coil to α-helix.19
XRD analysis (Fig. 2b) was used to investigate the crystal structure of the material. The peaks in the spectrum of SS–DHMC–AMWCNTs at 25.85° and 42.6° were attributed to the (002) and (100) crystal planes of AMWCNTs,20 while the newly emerged diffraction peaks of SS–DHMC–AuNPs–AMWCNTs at 38.15°, 44.35°, 64.6°, 77.6°, 81.9°, 110.85°, and 115.3° were assigned to the (111), (200), (220), (311), (222), (331), and (422) crystal planes of AuNPs, respectively.21 The appearance of these characteristic peaks confirmed the successful loading of AuNPs–AMWCNTs, and this indicated the presence of numerous high-index crystal planes that could increase the surface energy relative to free AuNPs,22 thereby enhancing the material's reactivity and amenability to use in electrochemical detection.
We next performed SEM analysis of the to investigate the microstructure of the composite film, as microstructure is key to the performance of any material. As shown in Fig. 2c, the SS–DHMC–AuNPs–AMWCNT film exhibited a uniform porous structure, suggesting that the material has a relatively high specific surface area and capacity for efficient mass transfer.23 The microporosity might also increase the number of electrochemically active sites,24 greatly facilitating subsequent electrochemical detection.
An elemental quantitative analysis (Fig. 2d) confirmed that the proportions of C, O, and Au were consistent with expectations, accounting for 19.52%, 55.73%, and 23.82%, respectively, of the total mass of the material, providing quantitative evidence for the composition of the composite. EDS mapping (Fig. 2e) showed that Au was uniformly dispersed in the material, indicating no significant aggregation of AuNPs, with most nanoparticles having a diameter of 50 nm, further increasing the surface area of the porous structure.25
The stability of the material is a key factor determining its practical applicability as a flexible sensor substrate.26 We found that the SS–DHMC–AuNPs–AMWCNT film maintained its structural integrity after immersion in water, with no noticeable damage or detachment of the AuNPs (Fig. 2f). This stability likely stems from the covalent crosslinked network formed between SS and DHMC, which provides a robust framework, and the incorporation of AMWCNTs, which enhances the mechanical strength of the material.14,27 Moreover, it's swelling rate in water remained around 2.15 after 96 hours and tended to stabilize, which has favourable and stable water absorption (Fig. S1).
Compared to traditional polymer-based composites,28 the composite material prepared in this work exhibits significantly better stability while maintaining a well-defined structure. We attribute this improvement primarily to the multi-level crosslinking strategy employed in the material design, as Schiff base reactions are known to form a stable covalent network at the molecular level.29 In addition, the interactions between AuNPs–AMWCNTs and the matrix at the nanoscale further enhance structural stability.30 Not only do the uniformly dispersed AuNPs provide abundant active sites, but they may also synergistically improve the conductivity of the material conductivity through interactions with AMWCNTs.31
In summary, systematic characterization confirmed the successful preparation of the SS–DHMC–AuNPs–AMWCNT composite film, and it also provided important insights into its structure–performance relationship. The excellent stability demonstrated by this material lays a solid foundation for its practical application in sensor technologies, particularly for sensing systems that require long-term stability. Further research is needed to determine and optimize the long-term stability of the material under various environmental conditions and to establish quantitative relationships between its sensing performance and microstructure.
3.2 Biocompatibility of the composite materials
Biocompatibility is a critical parameter when evaluating the potential clinical applicability of biomedical materials.32,33 In this study, we systematically investigated the biocompatibility of the SS–DHMC–AuNPs–AMWCNT composite through both in vitro cell experiments and in vivo animal experiments (Fig. 3). In vitro, both SS–DHMC–AMWCNTs and SS–DHMC–AuNPs–AMWCNTs exhibited excellent cytocompatibility in comparison with the control treatment. Specifically, fluorescence microscopy analyses (Fig. 3a) demonstrated that HeLa cells adhered with higher cell densities onto both composite surfaces as compared with a control surface, and the adhered cells exhibited an apparently normal morphology. When L929 cells were incubated in the presence of SS–DHMC–AMWCNTs and SS–DHMC–AuNPs–AMWCNTs, the proliferation rates relative to control were found by the CCK-8 assay to be 106.5% and 109.4%, respectively (Fig. 3b). Similar results were observed for other research, in which the microporous structure was demonstrated to be directly responsible for increased adhesion of the cell.34 Taken together, these results indicated an apparent lack of cytotoxicity and suggested that the porous structure of the material might promote cell adhesion and proliferation.To evaluate the potential safety of epidermal biosensors created from the material, three composite films (SS–DHMC, SS–DHMC–AMWCNTs and SS–DHMC–AuNPs–AMWCNTs) were implanted subcutaneously in the dorsal skin of rats. All samples were processed under endotoxin-minimizing conditions and sterilized prior to biological testing. At one week after implantation, no significant swelling or inflammatory exudation was observed in any treatment group (Fig. 3d and e). However, while rats implanted with SS–DHMC and SS–DHMC–AMWCNTs showed no obvious inflammatory responses (Fig. 3f, g and i), rats implanted with SS–DHMC–AuNPs–AMWCNTs exhibited mild localized inflammatory cell infiltration after one week (Fig. 3h). Histological analyses showed that the inflammatory cells primarily accumulated at the interface between the material and the tissue interface, as indicated by blue-stained regions in H&E staining, potentially due to mechanical stimulation from the rigid structure of the AMWCNTs and to the generation of trace reactive oxygen species that activated immune cells.35 Importantly, this inflammatory response remained mild and within a physiological range that is normally observed upon biomaterial implantation.
The intent of the sensor is to be used for short-term (24 h) monitoring on the epidermis, which possesses an intact stratum corneum barrier and fewer immune cells compared to dermis.36,37 Accordingly, our results suggest that the material demonstrates superior biocompatibility in epidermal applications. Human skin patch tests (Fig. S2) confirmed that the SS–DHMC–AuNPs–AMWCNT film caused no skin irritation or allergic reactions when applied for 48 h. Thus, the material performed comparably to commercial sterile dressings, thereby validating its safety for epidermal biosensing and supporting its clinical potential. The current in vivo assessment focused on short-term responses; longer-term degradation/eluate testing and surface-engineered variants will be evaluated in future work to further de-risk inflammatory responses for prolonged wear.
Kus-Liśkiewicz38 proposed a series of in vitro and in vivo assays to be employed in the evaluation of the cytotoxicity and biocompatibility of gold nanoparticles (AuNPs); the SS–DHMC–AuNPs–AMWCNT composite demonstrates excellent biocompatibility in epidermal applications, meeting the biological requirements for biocompatibility. Compared with previously reported carbon-based nanocomposites,39 this material shows significant advantages in biocompatibility. The biologically friendly interface constructed by the natural polymer matrix (SS–DHMC) provides an optimal microenvironment for cell growth,40 while the uniform dispersion of nanomaterials achieved through process optimization avoids reactions caused by locally high concentrations of potential irritants.41,42 The synergistic effects of these advantages endow the composite with reliable safety and stability, establishing a solid foundation for its application in medical monitoring tools. In future work, we plan to optimize the surface modifications of the AMWCNTs, such as introducing hydrophilic groups or biocompatible coatings, to more completely reduce potential inflammatory risks and expand its application potential in long-term implantable devices.
The mild, localized infiltration observed in the subcutaneous model should be interpreted in light of the sensor's short-term epidermal use, where the intact stratum corneum limits cellular exposure. To further reduce risk, future versions will apply anti-fouling surface modifications to the nanofillers (PEGylation or zwitterionic coatings; polydopamine-anchored hyaluronic acid/gelatin/sericin shells; thiolated-PEG end-capping on AuNPs) and mild oxidative/acid functionalization of CNTs to improve hydrophilicity. Antioxidant functional layers (e.g., polydopamine/tannic acid) will be explored to scavenge ROS near the interface. Regarding degradation, the SS–DHMC matrix is expected to yield peptides and cellulose-derived fragments, whereas metallic/carbonaceous fillers are largely insoluble; therefore, our follow-up will emphasize eluate testing (mass-loss/FTIR, ICP-MS for Au, CNT shedding by Raman), together with eluate cytotoxicity and macrophage cytokine assays to quantify inflammatory potential.
3.3 Electrochemical properties of SS–DHMC–AuNPs–AMWCNTs
Differential pulse voltammetry (DPV; Fig. 4b) revealed a distinct oxidation peak near 0.07 V for SS–DHMC–AuNPs–AMWCNTs, with a significantly higher current response compared to SS–DHMC and SS–DHMC–AMWCNTs, confirming the critical role of AuNPs in improving sensor sensitivity. Chronoamperometry (CA; Fig. 4c) further demonstrated that the SS–DHMC–AuNPs–AMWCNT composite film exhibited a substantially enhanced current response in glucose detection. This pronounced catalytic enhancement effect underscores the pivotal role of AuNPs in electrochemical glucose sensing, providing a strong foundation for developing other high-performance biosensors.
An electrochemical impedance spectroscopy (EIS) analysis based on the Randles equivalent circuit model47–49 further elucidated the electron transfer properties at the material interface (Fig. 4d). The charge transfer resistance (RCT) of SS–DHMC–AuNPs–AMWCNTs (537.8 Ω) was 24.2% lower than that of SS–DHMC–AMWCNTs (709.5 Ω). This kinetic result confirms that the high conductivity of AuNPs provides additional electron transfer pathways, while the synergistic interface formed with AMWCNTs reduces the interfacial energy barrier and promotes electron transfer.50,51 An additional factor supporting the sensitivity of the detector is that the porous structure of the nanocomposite (Fig. 2c) contributes to an increased effective electrode area.
To elucidate the electrocatalytic mechanism of AuNPs in the oxidation of glucose, we systematically investigated the electrochemical behavior of SS–DHMC–AuNPs–AMWCNT-modified electrodes at varying scan rates (50–500 mV s−1, Fig. 4g). The oxidation peak potential shifted positively while the reduction peak potential shifted negatively with increasing scan rates, characteristic of a quasi-reversible redox system.53 Notably, the oxidation peak current showed strong linearity with the scan rate (Fig. S4), confirming a surface-adsorption-controlled process rather than a diffusion-controlled process.54–56 This kinetic feature highlights the efficient catalytic activity of the surface sites of the AuNPs and the facilitating exposure of active sites by the porosity of the composite.57 The kinetic characteristics also suggest that the sensor response speed is unaffected by limitations of glucose diffusion and that sensitivity could be further enhanced through surface area optimization. The unique advantage of this study lies in achieving uniform dispersion of AuNPs and AMWCNTs within the flexible SS–DHMC network, maintaining high catalytic activity while preventing nanoparticle aggregation.58,59
The DPV response was measured over a pH range from 6.2 to 7.4 (Fig. 4i), and the shift in the oxidation peak potentials was found to correlate inversely with pH (R2 = 0.987, Fig. 4j). The slope of the correlation line (58.4 mV pH−1) was consistent with the theoretically ideal slope of a pH probe (59 mV pH−1),63,64 confirming that proton-coupled electron transfer occurred during glucose oxidation.21
Previous studies have reported that AuNPs can directly catalyze glucose oxidation to produce gluconic acid and hydrogen peroxide.65 This catalytic process requires the participation of hydroxide ions (OH−), in a similar manner to the catalytic activity of glucose oxidase (GOx). The pH-dependence observed for our system can be mechanistically explained as follows: under acidic conditions (pH < 7.0), insufficient hydroxyl groups are adsorbed on the surface of the gold, leading to limited catalytic activity. In contrast, under weakly basic conditions, the increased presence of hydroxide can lead to the formation of oxidized layers on the gold surfaces (e.g., Au(OH)3), which passivate the active sites and inhibit further glucose oxidation,66 resulting in compromised oxidative capability. The optimal performance observed at neutral pH represents a balanced state where adequate OH− is available to participate in the reaction while excessive formation of gold oxides is prevented, thereby ensuring both catalytic activity and material stability. This observation is consistent with previous reports demonstrating that gold electrodes exhibit maximum current response at neutral pH.67
In summary, the SS–DHMC–AuNPs–AMWCNT composite material achieves outstanding glucose sensing performance through rational structural design. The high sensitivity originates from the synergistic effect between the catalytic activity of AuNPs and the conductive network formed by AMWCNTs, while the porous structure ensures rapid mass transport and sufficient exposure of active sites. The confirmation of the importance of these parameters provides valuable insights into developing next-generation flexible biosensors.
3.4 Sensing performance of an SS–DHMC–AuNPs–AMWCNT electrode
To further evaluate the glucose-specific recognition capability of our sensor, we determined its electrochemical responses to 2 mM concentrations of the compounds themselves, in the absence of glucose. As shown in Fig. 5d, when testing these interferents separately, the obtained peak currents were significantly lower than those obtained when measuring a solution containing 2 mM glucose. These findings confirm the sensor's specificity toward glucose detection, and they provide valuable mechanistic insights into the molecular recognition process to inform the design of other, highly selective non-enzymatic glucose sensors.
As actual sweat contains much lower concentrations of the tested compounds than those analyzed here, and considering the limit of detection of 4 μM glucose, we concluded that the sensor exhibits sufficient performance for real-world applications. The high selectivity of the detector is particularly important for the prevention of false positive results emanating from exercise-induced fluctuations in metabolites (e.g., lactate) during dynamic monitoring.74
Long-term stability was assessed by (i) storage at 25 °C, ∼70% RH for 1–4 weeks, (ii) continuous PBS (pH 7.0, 37 °C) immersion for 1–6 days, (iii) temperature cycling between 20–40 °C. The SS–DHMC–AuNPs–AMWCNT composite film maintained stable glucose oxidation peak currents after continuous immersion in PBS for 6 d (Fig. 5f and g). Subsequent monthly monitoring tests (Fig. 5h) demonstrated current response fluctuations within 7%, fully meeting the long-term stability requirements for wearable devices. These comprehensive stability validation experiments, conducted across multiple testing dimensions, conclusively demonstrate that the SS–DHMC–AuNPs–AMWCNT sensor possesses the reliability and stability necessary for practical implementation.
Furthermore, evaluation of the mechanical durability performance (Fig. 5j–l) showed that after 0/50/100/150/200 cycles of bending the sensor 180° (Fig. S7), the LED circuit brightness exhibited no significant change, indicating that the integrity of the conductive network was maintained. The lack of change of the DPV response current confirmed that the electrochemical active sites remained undamaged after repeated folding. These comprehensive tests verified the sensor's excellent environmental adaptability and mechanical stability under simulated usage conditions, meeting the stringent requirements for wearable health monitoring applications.
In summary, the SS–DHMC–AuNPs–AMWCNT sensor achieves highly sensitive, selective, and stable glucose detection due to the design of the material and the optimization of its performance. The exceptional mechanical properties and environmental adaptability of this system make it an ideal candidate for wearable health monitoring. This work provides important ideas for non-enzymatic glucose sensor development while offering novel design concepts for flexible electronic devices.
3.5 In vivo deployment of wireless sweat glucose monitoring
When applied in a simulated environment, the SS–DHMC–AuNPs–AMWCNT sensor demonstrated exceptional reliability. Three healthy adult volunteers (age 20–35 years; 2 males, 1 females) were recruited under informed consent. All experiments were performed in accordance with the guidelines and approved by the Ethics Committee of Chongqing Medical University (Reference number: 2023011). Informed consents were obtained from human participants of this study. Application to artificial sweat yielded glucose recovery rates of 87.7 to 102.9% with RSD values of 1.3 to 2.6% (Table S2). Clinical validation using 10-fold diluted postprandial blood samples from the three volunteers showed recovery rates of 92.6 to 104.4%, with RSD values of 1.9 to 8.9% (Table S3), matching commercial glucometer readings. The slightly higher RSD values in blood testing reflect biological sample complexity, but the variability remained within acceptable limits. These results confirm the sensor's accuracy in physiological glucose detection and, combined with established sweat-blood glucose correlations,78 demonstrate its potential for noninvasive glycemic monitoring.
Notably, no skin irritation or discomfort was reported by any of the volunteers, confirming the technology's safety and wearability. Agreement with the commercial glucometer was evaluated using Bland–Altman analysis (bias and 95% limits of agreement). A strong correlation (R2 = 0.981) was observed between DPV current signals and glucometer readings (Fig. S6). Pre-/post-prandial monitoring (Fig. 6c) and OGTT (Fig. 6d) revealed a high correlation between sweat and blood glucose concentrations. This strong agreement can be attributed to the dynamic equilibrium between sweat glucose and blood glucose levels82 and the close association between dermal interstitial fluid glucose and blood glucose.83
4. Conclusion
In this study, we successfully transformed waste SS into high-performance flexible sensing material, leading to the development of a glucose sensor based on SS–DHMC–AuNPs–AMWCNTs with a demonstrated dual value in terms of both environmental and economic benefits. Through crosslinking reactions between SS and DHMC combined with synergistic modification strategies employing AuNPs and AMWCNTs, we significantly enhanced the material's mechanical properties and electrochemical activity while pioneering new applications for natural polymers in wearable devices. The fabricated flexible sensor exhibits exceptional performance in noninvasive blood glucose monitoring, with strong correlation to commercial glucometers (R2 = 0.981) confirming its clinical translation potential. This work not only provides a successful paradigm for high-value utilization of biological waste but also establishes a critical foundation for developing next-generation non-invasive health monitoring devices, showing broad application prospects in personalized medicine and chronic disease management. This work establishes a sustainable, flexible, enzyme-free sensing platform and demonstrates initial on-body feasibility; clinical validation in diabetic cohorts remains a necessary next step.Author contributions
Xiaorui Huang: data curation, formal analysis, and writing – original draft. Yi Li: conceptualization, data curation, investigation, writing – review & editing. Boxiang Yang: data curation, visualization. Yuxuan Wu: data curation, and visualization. Zhuocheng Jiang: data curation and visualization. Jiuxi Sui: data curation and visualization. Siyi Xing: data curation and visualization. Xue Zhang: data curation and visualization. Hualin Lan: writing – review & editing. Hanyan Zou: conceptualization, data curation, investigation, writing – review & editing. Yuchan Zhang: conceptualization, data curation, funding acquisition, investigation, writing – review & editing. Guangchao Zang: conceptualization, data curation, funding acquisition, investigation, writing – review & editing.Conflicts of interest
The authors declare no conflict of interest.Data availability
Data will be made available upon request.Supplementary information (SI): Fig. S1. The swelling rate of SS–DHMC–AuNPs–AMWCNTs film (n = 3). Fig. S2. Biocompatibility evaluation of SS–DHMC–AuNPs–AMWCNTs patch on human skin. Fig. S3. Relationship between redox peak currents and glucose concentration for SS–DHMC–AuNPs–AMWCNTs electrode. Fig. S4. Relationship between redox peak currents and scan rate for SS–DHMC–AuNPs–AMWCNTs electrode. Fig. S5. Cyclic voltammetry curves of SS–DHMC–AuNPs–AMWCNTs with varying contents of conductive fillers in PBS (pH 7.0). Fig. S6. Correlation analysis between non-invasive sweat glucose test results and commercial blood glucose meter measurements. Fig. S7. Flexible sensor folds 180 degrees. Fig. S8. (a) and (b) are the pre-fluid of SS–DHMC and SS–DHMC–AuNPs–AMWCNTs. (c) and (d) are the congealed SS–DHMC and SS–DHMC–AuNPs–AMWCNTs liquid after 24 hours. Table S1. Comparative analysis of detection performance between SS–DHMC–AuNPs–AMWCNTs and other non-enzymatic glucose sensors. Table S2. The detection of glucose in human sweat samples. Table S3. The detection of glucose in human blood samples. See DOI: https://doi.org/10.1039/d5sd00188a.
Acknowledgements
This work was supported by grants from the Natural Science Foundation of Chongqing (2024NSCQ-MSX0915, CSTB2023NSCQ-MSX0232); the project supported by the National Training Program of Innovation and Entrepreneurship for Undergraduates (202510631031); the JinFeng Laboratory Foundation, Chongqing, China (jfkyjf202203001); the Project of Tutorial System of Medical Undergraduate in Lab Teaching & Management Center in Chongqing Medical University (LTMCMTS202434, LTMCMTS202435, LTMCMTS202457).References
- J. Y. Xiao, et al., Microfluidic Chip-Based Wearable Colorimetric Sensor for Simple and Facile Detection of Sweat Glucose, Anal. Chem., 2019, 91(23), 14803–14807 Search PubMed.
- Y. J. Lin, et al., Porous Enzymatic Membrane for Nanotextured Glucose Sweat Sensors with High Stability toward Reliable Noninvasive Health Monitoring, Adv. Funct. Mater., 2019, 29(33), 1–8 Search PubMed.
- H. B. Lee, et al., A wearable lab-on-a-patch platform with stretchable nanostructured biosensor for non-invasive immunodetection of biomarker in sweat, Biosens. Bioelectron., 2020, 156, 112133 Search PubMed.
- H. Guan, et al., Processing Natural Wood into a High-Performance Flexible Pressure Sensor, ACS Appl. Mater. Interfaces, 2020, 12(41), 46357–46365 Search PubMed.
- F. Wang, et al., Transgenic PDGF-BB/sericin hydrogel supports for cell proliferation and osteogenic differentiation, Biomater. Sci., 2020, 8(2), 657–672 Search PubMed.
- M. C. Arango, et al., Silk sericin as a biomaterial for tissue engineering: a review, Int. J. Polym. Mater. Polym. Biomater., 2021, 70(16), 1115–1129 Search PubMed.
- T. T. Cao and Y. Q. Zhang, Processing and characterization of silk sericin from Bombyx mori and its application in biomaterials and biomedicines, Mater. Sci. Eng., C, 2016, 61, 940–952 Search PubMed.
- V. Bampidis, et al., Safety and efficacy of hydroxypropyl methyl cellulose for all animal species, EFSA J., 2020, 18(7), 6214 Search PubMed.
- S. Koprivica, et al., Regeneration of Aqueous Periodate Solutions by Ozone Treatment: A Sustainable Approach for Dialdehyde Cellulose Production, ChemSusChem, 2016, 9(8), 825–833 CrossRef CAS PubMed.
- P. Wang, et al., Cross-linking of dialdehyde carboxymethyl cellulose with silk sericin to reinforce sericin film for potential biomedical application, Carbohydr. Polym., 2019, 212, 403–411 CrossRef CAS PubMed.
- R. S. Singh and K. Chauhan, Immobilization of Inulinase on Aminated Multiwalled Carbon Nanotubes by Glutaraldehyde Cross-Linking for the Production of Fructose, Catal. Lett., 2019, 149(10), 2718–2727 CrossRef CAS.
- S. Alim, et al., Recent uses of carbon nanotubes & gold nanoparticles in electrochemistry with application in biosensing: A review, Biosens. Bioelectron., 2018, 121, 125–136 Search PubMed.
- M. S. Tamore, et al., Effect of functionalized multi-walled carbon nanotubes on physicomechanical properties of silicone rubber nanocomposites, J. Compos. Mater., 2019, 53(22), 3157–3168 Search PubMed.
- Y. Chen, et al., A wearable non-enzymatic sensor for continuous monitoring of glucose in human sweat, Talanta, 2024, 278, 126499 CrossRef CAS PubMed.
- J. E. Baio, D. J. Graham and D. G. Castner, Surface analysis tools for characterizing biological materials, Chem. Soc. Rev., 2020, 49(11), 3278–3296 RSC.
- U. Kim, et al., Cellulose-chitosan beads crosslinked by dialdehyde cellulose, Nanomaterials, 2017, 24(10), 5517–5528 Search PubMed.
- P. Wang, et al., Cross-linking of dialdehyde carboxymethyl cellulose with silk sericin to reinforce sericin film for potential biomedical application, Carbohydr. Polym., 2019, 212, 403–411 Search PubMed.
- H. Tan, et al., Collagen cryogel cross-linked by naturally derived dialdehyde carboxymethyl cellulose, Carbohydr. Polym., 2015, 129, 17–24 CrossRef CAS PubMed.
- X. Li, et al., CNT/Sericin Conductive Nerve Guidance Conduit Promotes Functional Recovery of Transected Peripheral Nerve Injury in a Rat Model, ACS Appl. Mater. Interfaces, 2020, 12(33), 36860–36872 CrossRef CAS PubMed.
- F. A. Awadallah and S. Al-Muhtaseb, Carbon Nanoparticles-Decorated Carbon Nanotubes, Sci. Rep., 2020, 10(1), 4878 Search PubMed.
- Q. Peng, et al., Glucose determination behaviour of gold microspheres-electrodeposited carbon cloth flexible electrodes in neutral media, Anal. Chim. Acta, 2021, 1159, 338442 Search PubMed.
- R. Li, et al., Electrochemical biosensor based on the integration of maple leaf-like gold nanocrystal and truncated aptamer for detection of alpha-amanitin with high sensitivity, selectivity and rapidity, Food Chem., 2024, 453, 139639 CrossRef CAS PubMed.
- H. Yu, et al., Research Progress on Porous Carbon-Based Non-Precious Metal Electrocatalysts, Materials, 2023, 16(8), 3283 CrossRef CAS PubMed.
- D. Zhang, et al., Effect of porous structure on the electrochemical performance of FeS2 for lithium ion batteries, J. Nanosci. Nanotechnol., 2014, 14(8), 6095–6102 CrossRef CAS PubMed.
- X. Yang, et al., Monodisperse Carbon Nanospheres with Hierarchical Porous Structure as Electrode Material for Supercapacitor, Nanoscale Res. Lett., 2017, 12(1), 550 CrossRef PubMed.
- X. He, Fundamental Perspectives on the Electrochemical Water Applications of Metal-Organic Frameworks, Nano-Micro Lett., 2023, 15(1), 148 Search PubMed.
- K. Jayaramulu, et al., Graphene-Based Metal-Organic Framework Hybrids for Applications in Catalysis, Environmental, and Energy Technologies, Chem. Rev., 2022, 122(24), 17241–17338 CrossRef CAS PubMed.
- K. Singh, K. K. Maurya and M. Malviya, Recent progress on nanomaterial-based electrochemical sensors for glucose detection in human body fluids, Microchim. Acta, 2025, 192(2), 110 Search PubMed.
- Y. P. Mo, et al., The intramolecular H-bonding effect on the growth and stability of Schiff-base surface covalent organic frameworks, Phys. Chem. Chem. Phys., 2016, 19(1), 539–543 Search PubMed.
- I. S. Kucherenko, et al., Advances in nanomaterial application in enzyme-based electrochemical biosensors: a review, Nanoscale Adv., 2019, 1(12), 4560–4577 Search PubMed.
- Y. Zhang, et al., Enzyme-catalyzed electrochemical aptasensor for ultrasensitive detection of soluble PD-L1 in breast cancer based on decorated covalent organic frameworks and carbon nanotubes, Anal. Chim. Acta, 2023, 1282, 341927 CrossRef CAS PubMed.
- L. Tang and W. Hu, Molecular determinants of biocompatibility, Expert Rev. Med. Devices, 2005, 2(4), 493–500 Search PubMed.
- V. Rosa, et al., Guidance on the assessment of biocompatibility of biomaterials: Fundamentals and testing considerations, Dent. Mater., 2024, 40(11), 1773–1785 CrossRef CAS PubMed.
- L. Zheng, et al., Engineering of Aerogel-Based Biomaterials for Biomedical Applications, Int. J. Nanomed., 2020, 15, 2363–2378 CrossRef CAS PubMed.
- J. Zhang, et al., Tumoral NOX4 recruits M2 tumor-associated macrophages via ROS/PI3K signaling-dependent various cytokine production to promote NSCLC growth, Redox Biol., 2019, 22, 101116 Search PubMed.
- S. de Szalay and P. W. Wertz, Protective Barriers Provided by the Epidermis, Int. J. Mol. Sci., 2023, 24(4), 3145 CrossRef CAS PubMed.
- P. R. Murphy, D. Narayanan and S. Kumari, Methods to Identify Immune Cells in Tissues With a Focus on Skin as a Model, Curr. Protoc., 2022, 2(7), e485 Search PubMed.
- M. Kus-Liśkiewicz, P. Fickers and I. Ben Tahar, Biocompatibility and Cytotoxicity of Gold Nanoparticles: Recent Advances in Methodologies and Regulations, Int. J. Mol. Sci., 2021, 22(20), 10952 Search PubMed.
- B. R. C. Menezes, et al., Recent advances in the use of carbon nanotubes as smart biomaterials, J. Mater. Chem. B, 2019, 7(9), 1343–1360 RSC.
- X. Yang, et al., AR regulates porcine immature Sertoli cell growth via binding to RNF4 and miR-124a, Reprod. Domest. Anim., 2021, 56(3), 416–426 Search PubMed.
- D. R. Seshadri, et al., Flexible and Scalable Dry Conductive Elastomeric Nanocomposites for Surface Stimulation Applications, IEEE Trans. Biomed. Eng., 2023, 70(12), 3461–3468 Search PubMed.
- V. Castranova, P. A. Schulte and R. D. Zumwalde, Occupational nanosafety considerations for carbon nanotubes and carbon nanofibers, Acc. Chem. Res., 2013, 46(3), 642–649 Search PubMed.
- H. Z. Zhao, et al., Mechanisms for the direct electron transfer of cytochrome c induced by multi-walled carbon nanotubes, Sensors, 2012, 12(8), 10450–10462 CrossRef CAS PubMed.
- Y. Yang, et al., Flexible enzymatic biofuel cell based on 1, 4-naphthoquinone/MWCNT-Modified bio-anode and polyvinyl alcohol hydrogel electrolyte, Biosens. Bioelectron., 2022, 198, 113833 Search PubMed.
- X. Z. Zhang, D. Q. Wu and C. C. Chu, Synthesis, characterization and controlled drug release of thermosensitive IPN-PNIPAAm hydrogels, Biomaterials, 2004, 25(17), 3793–3805 Search PubMed.
- X. Chen, et al., Terminal deoxynucleotidyl transferase induced activators to unlock the trans-cleavage of CRISPR/Cpf 1 (TdT-IU-CRISPR/Cpf 1): An ultrasensitive biosensor for Dam MTase activity detection, Biosens. Bioelectron., 2020, 163, 112271 Search PubMed.
- H. Wang, et al., Electrochemical impedance spectroscopy applied to microbial fuel cells: A review, Front. Microbiol., 2022, 13, 973501 CrossRef PubMed.
- E. B. Bahadir and M. K. Sezginturk, A review on impedimetric biosensors, Artif. Cells, Nanomed., Biotechnol., 2016, 44(1), 248–262 Search PubMed.
- I. Turcan, et al., Dielectrophoretic and Electrical Impedance Differentiation of Cancerous Cells Based on Biophysical Phenotype, Biosensors, 2021, 11(10), 401 Search PubMed.
- J. Im, et al., Functionalized Gold Nanoparticles with a Cohesion Enhancer for Robust Flexible Electrodes, ACS Appl. Nano Mater., 2022, 5(5), 6708–6716 CrossRef CAS PubMed.
- M. Mehrban, et al., Fabrication of impedimetric sensor based on metallic nanoparticle for the determination of mesna anticancer drug, Sci. Rep., 2023, 13(1), 11381 CrossRef CAS PubMed.
- P. Pongsanon, et al., Effect of Gold Nanoparticle Size on Regulated Catalytic Activity of Temperature-Responsive Polymer-Gold Nanoparticle Hybrid Microgels, Gels, 2024, 10(6), 357 CrossRef CAS PubMed.
- S. Zhao, et al., Glucose oxidase/colloidal gold nanoparticles immobilized in Nafion film on glassy carbon electrode: Direct electron transfer and electrocatalysis, Bioelectrochemistry, 2006, 69(2), 158–163 Search PubMed.
- Y. Liu, et al., MOF-polymer composites with well-distributed gold nanoparticles for visual monitoring of homocysteine, Analyst, 2024, 149(5), 1658–1664 Search PubMed.
- W. Wang, et al., Effects of Gold Nanospheres and Nanocubes on Amyloid-beta Peptide Fibrillation, Langmuir, 2019, 35(6), 2334–2342 CrossRef CAS PubMed.
- H. Xiang, et al., Au Nanozyme Driven Cascading Catalysis in Tollens' Reaction: An Insight of Glucose Oxidase-Like Mechanism, Chemistry, 2023, 29(34), e202300454 Search PubMed.
- H. Zhangsun, et al., NiCu nanoalloy embedded in N-doped porous carbon composite as superior electrochemical sensor for neonicotinoid determination, Food Chem., 2022, 384, 132607 Search PubMed.
- B. Liu, et al., Highly-dispersed NiFe alloys in-situ anchored on outer surface of Co, N co-doped carbon nanotubes with enhanced stability for oxygen electrocatalysis, J. Colloid Interface Sci., 2023, 635, 208–220 Search PubMed.
- X. Cai, et al., Molecular Mechanisms, Characterization Methods, and Utilities of Nanoparticle Biotransformation in Nanosafety Assessments, Small, 2020, 16(36), e1907663 Search PubMed.
- Y. Chen, et al., Advanced Metal-Organic Frameworks-Based Catalysts in Electrochemical Sensors, Front. Chem., 2022, 10, 881172 Search PubMed.
- I. Sarhid, et al., Hexacyano Ferrate (III) Reduction by Electron Transfer Induced by Plasmonic Catalysis on Gold Nanoparticles, Materials, 2019, 12(18), 3012 CrossRef CAS PubMed.
- M. H. Salehi, et al., Electrically conductive biocompatible composite aerogel based on nanofibrillated template of bacterial cellulose/polyaniline/nano-clay, Int. J. Biol. Macromol., 2021, 173, 467–480 Search PubMed.
- W. Sun, et al., Direct electrocatalytic oxidation of adenine and guanine on carbon ionic liquid electrode and the simultaneous determination, Biosens. Bioelectron., 2008, 24(4), 994–999 Search PubMed.
- C. S. Lee, S. H. Yu and T. H. Kim, One-Step Electrochemical Fabrication of Reduced Graphene Oxide/Gold Nanoparticles Nanocomposite-Modified Electrode for Simultaneous Detection of Dopamine, Ascorbic Acid, and Uric Acid, Nanomaterials, 2017, 8(1), 17 CrossRef PubMed.
- N. J. Lang, B. Liu and J. Liu, Characterization of glucose oxidation by gold nanoparticles using nanoceria, J. Colloid Interface Sci., 2014, 428, 78–83 CrossRef CAS PubMed.
- M. Pasta, F. Mantia and C. Yi, Mechanism of glucose electrochemical oxidation on gold surface, Electrochim. Acta, 2010, 55(20), 5561–5568 CrossRef CAS.
- M. H. Hassan, et al., Recent Advances in Enzymatic and Non-Enzymatic Electrochemical Glucose Sensing, Sensors, 2021, 21(14), 4672 CrossRef CAS PubMed.
- D. R. Seshadri, et al., Wearable sensors for monitoring the physiological and biochemical profile of the athlete, npj Digit. Med., 2019, 2, 72 Search PubMed.
- T. Kangkamano, et al., Chitosan Cryogel with Embedded Gold Nanoparticles Decorated Multiwalled Carbon Nanotubes Modified Electrode for Highly Sensitive Flow Based Non-Enzymatic Glucose Sensor, Sens. Actuators, B, 2017, 246(10), 854–863 CrossRef CAS.
- S. Hu, et al., Enzyme-Free Tandem Reaction Strategy for Surface-Enhanced Raman Scattering Detection of Glucose by Using the Composite of Au Nanoparticles and Porphyrin-Based Metal-Organic Framework, ACS Appl. Mater. Interfaces, 2020, 12(49), 55324–55330 CrossRef CAS PubMed.
- J. das Neves, R. Sverdlov and A. Sosnik, Molecular and cellular cues governing nanomaterial-mucosae interactions: from nanomedicine to nanotoxicology, Chem. Soc. Rev., 2020, 49(14), 5058–5100 RSC.
- C. Kim, T. Kim and J. Cho, Selective Charge Carrier Transport and Bipolar Conduction in an Inorganic/Organic Bulk-Phase Composite: Optimization for Low-Temperature Thermoelectric Performance, ACS Appl. Mater. Interfaces, 2024, 16(4), 5036–5049 Search PubMed.
- S. Y. Oh, et al., Skin-Attachable, Stretchable Electrochemical Sweat Sensor for Glucose and pH Detection, ACS Appl. Mater. Interfaces, 2018, 10(16), 13729–13740 CrossRef CAS PubMed.
- A. Philp, A. L. Macdonald and P. W. Watt, Lactate--a signal coordinating cell and systemic function, J. Exp. Biol., 2005, 208(Pt 24), 4561–4575 Search PubMed.
- A. Sonawane, P. Manickam and S. Bhansali, Stability of Enzymatic Biosensors for Wearable Applications, IEEE Rev. Biomed. Eng., 2017, 10, 174–186 Search PubMed.
- J. Liu, et al., Graphene-Based Wearable Temperature Sensors: A Review, Nanomaterials, 2023, 13(16), 110971 CrossRef PubMed.
- A. M. V. Mohan, V. Rajendran, R. K. Mishra and M. Jayaraman, Recent advances and perspectives in sweat based wearable electrochemical sensors, TrAC, Trends Anal. Chem., 2020, 131, 116024 Search PubMed.
- H. Zafar, et al., Comprehensive Review on Wearable Sweat-Glucose Sensors for Continuous Glucose Monitoring, Sensors, 2022, 22(2), 638 CrossRef CAS PubMed.
- J. Moyer, et al., Correlation between sweat glucose and blood glucose in subjects with diabetes, Diabetes Technol. Ther., 2012, 14(5), 398–402 CrossRef CAS PubMed.
- Z. Zhang, et al., Sweat as a source of non-invasive biomarkers for clinical diagnosis: An overview, Talanta, 2024, 273, 125865 CrossRef CAS PubMed.
- Q. Chen, et al., Silk-Based Electrochemical Sensor for the Detection of Glucose in Sweat, Biomacromolecules, 2022, 23(9), 3928–3935 CrossRef CAS PubMed.
- E. V. Karpova, E. E. Karyakina and A. A. Karyakin, Wearable non-invasive monitors of diabetes and hypoxia through continuous analysis of sweat, Talanta, 2020, 215, 120922 CrossRef CAS PubMed.
- T. Saha, et al., A Passive Perspiration Inspired Wearable Platform for Continuous Glucose Monitoring, Adv. Sci., 2024, 11(41), e2405518 CrossRef PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |