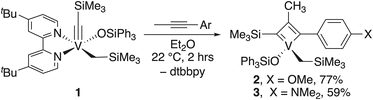Open Access Article
Open Access ArticleToward vanadium-mediated alkyne metathesis
Shirley
Hernandez
 a,
Vasilisa
Krivovicheva
a,
Vasilisa
Krivovicheva
 a,
Adenilson
Sousa-Silva
a,
Adenilson
Sousa-Silva
 b,
Xavier
Solans-Monfort
b,
Xavier
Solans-Monfort
 b and
Konstantin V.
Bukhryakov
b and
Konstantin V.
Bukhryakov
 *a
*a
aDepartment of Chemistry and Biochemistry, Florida International University, Miami, FL 33199, USA. E-mail: kbukhrya@fiu.edu
bDepartament de Química, Universitat Autònoma de Barcelona, 08193 Bellaterra, Spain
First published on 18th March 2026
Abstract
Alkyne metathesis, a widely used method for the synthesis of chemicals containing carbon–carbon triple bonds, relies exclusively on catalysts based on second and third-row transition metals. The development of a first-row metal-mediated alkyne metathesis would be a remarkable achievement from both fundamental and sustainability perspectives. In this study, we lay the groundwork for V-mediated alkyne metathesis. Thus, we demonstrated that V alkylidynes can react with various alkynes to produce cycloaddition products (metallacyclobutadienes), which are critical intermediates in the alkyne metathesis. Additionally, we conducted comprehensive computational studies to confirm the feasibility of the cycloreversion step needed to complete the alkyne metathesis transformation. Finally, we synthesized and characterized a series of new V alkylidynes, which can serve as essential starting materials for future development.
Introduction
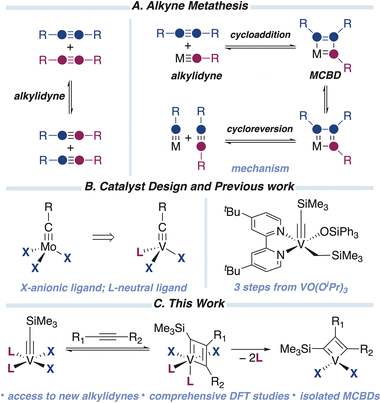
Alkyne metathesis, the redistribution of R groups between alkynes (Fig. 1A), is an important reaction utilized to produce valuable chemicals containing C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C triple bonds.1–7 The resulting alkynes are essential starting points for producing a large variety of organic compounds, including natural products and pharmaceuticals, taking advantage of the chemical versatility of a triple bond. Furthermore, alkyne metathesis has been extensively utilized in material science to produce polymers containing triple bonds.8–12 Two crucial steps of the classical Katz's mechanism13 of alkyne metathesis are shown in Fig. 1A and include a cycloaddition reaction between alkyne and metal alkylidyne (a complex containing M
C triple bonds.1–7 The resulting alkynes are essential starting points for producing a large variety of organic compounds, including natural products and pharmaceuticals, taking advantage of the chemical versatility of a triple bond. Furthermore, alkyne metathesis has been extensively utilized in material science to produce polymers containing triple bonds.8–12 Two crucial steps of the classical Katz's mechanism13 of alkyne metathesis are shown in Fig. 1A and include a cycloaddition reaction between alkyne and metal alkylidyne (a complex containing M![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C triple bond) to form metallacyclobutadiene (MCBD), followed by the cycloreversion step to form a new alkylidyne and alkyne product. Cycloaddition/cycloreversion steps resemble the mechanism of olefin metathesis. However, unlike olefin metathesis, the number of alkyne metathesis catalytic systems is somewhat limited. Indeed, the currently used systems rely exclusively on second and third-row transition metal alkylidynes such as Mo, W, and Re.7,14–17
C triple bond) to form metallacyclobutadiene (MCBD), followed by the cycloreversion step to form a new alkylidyne and alkyne product. Cycloaddition/cycloreversion steps resemble the mechanism of olefin metathesis. However, unlike olefin metathesis, the number of alkyne metathesis catalytic systems is somewhat limited. Indeed, the currently used systems rely exclusively on second and third-row transition metal alkylidynes such as Mo, W, and Re.7,14–17
From a fundamental standpoint, the question is whether the first-row mediated alkyne metathesis is achievable. From the practical standpoint, a shift to more sustainable and cost-effective first-row systems is desirable. In addition, the unique electronic structure of base metals can offer an unusual reactivity, for example, the V-mediated carbon isotope exchange of terminal alkenes, which enables the labeling of biologically active compounds.18,19
Among 3d transition metals, olefin metathesis catalysts based on V are the most developed,20–23 presumably due to the diagonal relationship between V and Mo.24 Since V is a group 5 metal, one anionic ligand should be replaced by a neutral ligand to transfer properties of Mo d0 alkylidenes to V counterparts, the principle utilized by our group previously.25–29 Therefore, we decided to utilize a similar approach to V alkylidynes (Fig. 1B). Furthermore, theoretical studies on the cycloaddition/cycloreversion steps involving group 5 metal alkylidynes suggest that those complexes “can be targeted for the development of alkyne metathesis catalysts”.30 However, those DFT studies suggested that the cycloreversion would be the rate-limiting step due to the increased stability of V MCBD.
First-row metal alkylidynes are rare.31 The number of high-oxidation state (d0) V alkylidynes is even more scarce. The majority reported V(+5) alkylidynes are based on β-diketiminate (NacNac) ligand prepared by Mindiola.32,33 Those complexes can react with terminal alkynes to form α,α-disubstituted MCBDs, followed by an intramolecular proton-shuttling event to generate a deprotiovanadacycle. This reactivity enables the formation of cyclic polyolefins via low-valent V species, facilitated by the non-innocent nature of the NacNac ligand.34 Reactions with nitriles and phosphaalkynes yield α-aza-MCBD and β-phospha-MCBD, respectively.35
Recently, we reported an easy and scalable method to prepare (d0) V alkylidynes in three steps from commercially available precursors with high overall yield (Fig. 1B).36 The resulting complexes resemble Mo alkylidynes and would be suitable candidates for probing alkyne metathesis.
Herein, we report the cycloaddition reaction involving V alkylidynes, a key step in alkyne metathesis; comprehensive DFT studies of V-based alkyne metathesis; and the synthesis of new V alkylidynes, which can serve as suitable starting points for developing V-mediated alkyne metathesis (Fig. 1C).
Results and discussion
Complex 1 readily reacts with various alkynes to form MCBDs (SI, Fig. S1) and free dtbbpy. However, most of the resulting MCBDs are not crystalline, which hinders their isolation and characterization. Fortunately, we were able to isolate complexes 2 and 3 (Fig. 2). In both cases, we observed the formation of only one isomer in which the aryl group is attached to the α-carbon atom of MCBD, as confirmed by the NOESY experiment for 2 (SI, Fig. S17) and by X-ray single crystal diffraction studies for 3.Notably, V-based MCBDs are extremely rare.34,37,38 To our knowledge, trisubstituted V MCBDs have not been isolated previously.
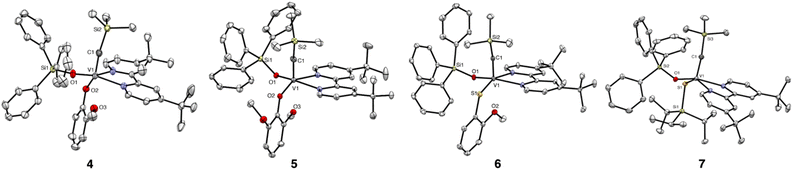
The X-ray structure of 3 revealed a four-coordinate complex with a distorted tetrahedral geometry at the V center (Fig. 3). The comparison of the structure details of 3 to the only known analogous metallacyclobuta-(1,3)-diene prepared by the Mindiola group,34 showed that the V–C1 and V–C3 bond distances (1.821(2)Å and 1.841(2)Å, respectively) are in the range of reported MCBD (1.788 and 1.891 Å) and V–C2 distance is slightly longer (2.058(2)Å vs. reported 2.004(8)Å).
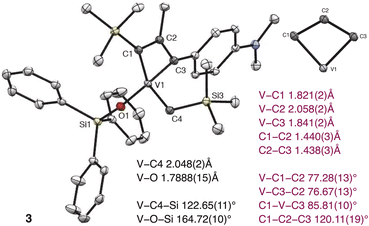 | ||
| Fig. 3 Perspective view of the crystal structure of complex 3 with thermal ellipsoids shown at 30% probability. Hydrogen atoms are omitted for clarity. | ||
A relatively short V–O bond length (1.7888(15)Å),36 and the large V–O–Si (164.72(10)°) suggest significant π-donation from the oxygen atom to V.7,39 The V–C4 bond distance (2.048(2)Å) is typical for a V–C single bond, and the V–C4–Si angle (122.65(11)°) is slightly larger compared to this in alkylidyne 1 (116.8(3)°).
We were somewhat surprised that the dtbbpy ligand dissociates during the cycloaddition step, which can be clearly seen by 1H NMR spectroscopy. We have not observed the cycloreversion step for complexes 2 and 3 even under heating, UV irradiation, and the addition of neutral ligands, such as pyridines, phosphines, and N-heterocyclic carbenes. The increased stability of four-coordinated V MCBDs is predicted theoretically and might be a challenging step in V-based alkyne metathesis.30 We concluded that the current ligand set is unsuitable for the efficient cycloreversion step, which would be required for alkyne metathesis, and we need theoretical support to guide our next steps.
To probe the feasibility of V-based alkyne metathesis, we performed DFT (B3LYP-D3)40–42 studies of cycloaddition and cycloreversion steps with alkylidyne 1 (Fig. 4). The dissociation of dtbbpy to form 3-coordinate V1-I is highly unfavorable. We could not de-coordinate one N atom of the bipyridine ligand from 1, all calculations go back to the coordination of the two N atoms. Consequently, we believe the reaction proceeds via an associative pathway, in which an octahedral MCBD V1-IIa is formed.
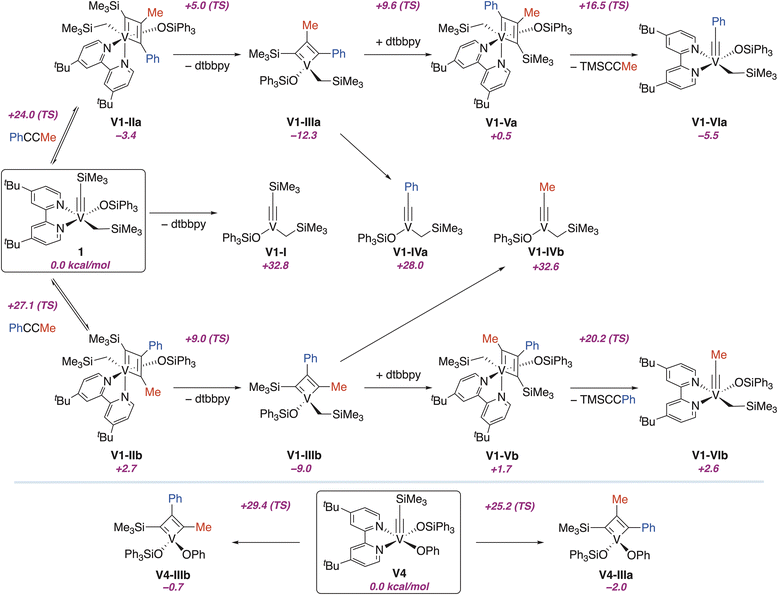 | ||
| Fig. 4 Calculated relative Gibbs free energies under benzene solvation (B3LYP-D3) in kcal mol−1 with respect to 1 (top) and V4 (bottom). | ||
Several isomers of V1-IIa can be envisaged depending on the ligand in trans to the incoming alkyne during the cycloaddition step. We considered several of these isomers and found that they are all close in energy, the largest difference being 8.7 kcal mol−1 (SI, Fig. S11). In addition, we were able to locate the associated transition states for the cycloaddition/cycloreversion step (1 → V1-IIa and 1 → V1-IIb) involving several of these isomers (SI, Fig. S12 and S13). The most favorable pathway involves coordination of the alkyne trans to the alkyl group, the strongest σ-donating ligand, which aligns well with the known data.43,44 The computed energy barrier is 24.0 kcal mol−1, which is significantly lower than that for the other pathways.
Therefore, cycloaddition via the associative pathway is feasible and can proceed at room temperature in accordance with the experimental data. This finding highlights the difference between classical alkyne metathesis systems and V reactivity. Thus, the Schrock catalysts are 4-coordinate alkylidynes that form 5-coordinate MCBDs.45 We believe the cycloaddition reaction via the associative pathway arises from the high electrophilicity of the V atom, as confirmed spectroscopically. Thus, the 13C NMR chemical shift of the alkylidyne carbon is a valuable tool to access the electrophilicity of the metal center.46 The alkylidyne carbon in complex 1 has a chemical shift of 421 ppm, significantly higher than the corresponding chemical shift in typical Mo-based catalysts (∼300 ppm).
The dissociation of the bipyridine ligand has a very low energy barrier and leads to thermodynamically favorable V1-IIIa, which is consistent with the experiments. The direct cycloreversion from V1-IIIa to form 3-coordinate V1-IVa is uphill by 40.3 kcal mol−1. Therefore, the binding of bipyridine or other neutral ligands is necessary for achieving an alkylidyne such as V1-VIa, which is lower in Gibbs energies than the initial reactants (ΔG = −5.5 kcal mol−1). Indeed, the binding of the dtbbpy to V1-IIIa is feasible and leads to V1-Va with a Gibbs reaction energy of 12.8 kcal mol−1. However, the Gibbs energy barrier for cycloreversion is 16.0 kcal mol−1 from V1-Va, leading to a Gibbs energy span of 28.8 kcal mol−1 with respect to V1-IIIa. Therefore, according to calculations, the tetracoordinated metallacyclobutadiene is too stable relative to both reactants and products; thus, cycloreversion would require an energy barrier of 28.8 kcal mol−1, which can only be overcome through substantial heating. Unfortunately, V MCBDs 2 and 3 decompose above 80 °C in the presence of dtbbpy.
The formation of isomer V1-IIIb is less favourable for kinetic and thermodynamic reasons, which agrees with the experiment. Similar to our studies of V alkylidenes,18 we believe that the V–C bond is highly polarized, with a significant negative charge at the α-carbon in MCBD, which is stabilized by the phenyl group in V1-IIIa (Table S6).
Next, we decided to explore the influence of the anionic ligand at V on the relative stability of alkylidyne and the resulting MCBD. We found that the substitution of the alkyl group in 1 with a phenoxide ligand (V4, Fig. 4, bottom) increases the energy of the corresponding MCBDs (V4-IIIa and V4-IIIb, Fig. 4) relative to the starting alkylidyne, which might make the cycloreversion step more feasible. Indeed, the cycloaddition transition states are 25.2 kcal mol−1 and 29.4 kcal mol−1, respectively. It suggests that the increase in MCBD energy is not associated with a too demanding cycloaddition process.
With the aim of getting insight into the change of the MCBD energy, we computed the V–L Wiberg bond orders (Table S7). Results show that bonds are strengthened at the MCBD when compared to the initial alkylidynes and the pseudo-octahedral intermediates V1-IIa and V4-IIa. The observed MCBDs V1-IIIa and V4-IIIa are four-coordinate complexes, where anionic ligands do not have ligands in the trans position. This is not the case for the initial alkylidynes and the cycloaddition intermediates. We hypothesize that introducing a strong σ-donating anionic ligand, such as an alkyl group, trans to a neutral ligand (bipy), destabilizes the alkylidyne due to the trans effect, but has a smaller effect on MCBD stability. As a result, the difference in the ground state between alkylidyne 1 and MCBD V1-IIIa becomes larger compared to the energy difference between V4 and V4-IIIa (−12.3 kcal mol−1vs. −2.0 kcal mol−1), since the phenoxide ligand is a weaker σ-donor than the alkyl group. Consequently, the anionic ligand substitution has a greater stabilizing effect on the alkylidynes than on the MCBDs.
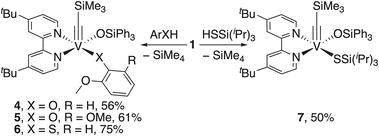
To substitute the alkyl group on complex 1, we tested its reactivity toward various phenols, thiophenols, alcohols, and thiols to form corresponding alkylidynes (SI, Fig. S2). We were able to isolate, characterize, and obtain X-ray structures of complexes 4–7 (Fig. 5).
The X-ray structures of 4–7 confirmed the presence of a V alkylidyne ligand. Thus, the V![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C bond distances in 4–7 are comparable to those in 1 (1.707(6)Å, Table 1). Complexes 4–6 have a distorted octahedral geometry, if considering an interaction between the methoxy group and V. Complex 7 has a distorted square pyramidal geometry (τ = 0.21) with alkylidyne in the apical position, which is similar to 1 (τ = 0.18). Remarkably, the V
C bond distances in 4–7 are comparable to those in 1 (1.707(6)Å, Table 1). Complexes 4–6 have a distorted octahedral geometry, if considering an interaction between the methoxy group and V. Complex 7 has a distorted square pyramidal geometry (τ = 0.21) with alkylidyne in the apical position, which is similar to 1 (τ = 0.18). Remarkably, the V![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Si angle is the most tilted in complex 4 (158.72(13)°), which is the highest deviation from the linear geometry among all reported V alkylidynes.32,36 This V
C–Si angle is the most tilted in complex 4 (158.72(13)°), which is the highest deviation from the linear geometry among all reported V alkylidynes.32,36 This V![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Si bending appears to be related to crystal packing, as a DFT optimization of the isolated complex yields a V
C–Si bending appears to be related to crystal packing, as a DFT optimization of the isolated complex yields a V![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Si angle close to 180°. At the same time, the V–O–Si angle in 4 (168.78(11)°) exceeds that of MCBD 3 (164.72(10)°, Fig. 3), suggesting significant π-donation from the oxygen atom to V.7,39 As expected, the V–S–R angles in 6 and 7 are lower compared to V–O2–Ar in 4 and 5, due to decreased π-donation from the sulfur atom compared to oxygen.47
C–Si angle close to 180°. At the same time, the V–O–Si angle in 4 (168.78(11)°) exceeds that of MCBD 3 (164.72(10)°, Fig. 3), suggesting significant π-donation from the oxygen atom to V.7,39 As expected, the V–S–R angles in 6 and 7 are lower compared to V–O2–Ar in 4 and 5, due to decreased π-donation from the sulfur atom compared to oxygen.47
| 1 (X = CH2, R = TMS) | 4 (X = O2, R = Ar) | 5 (X = O2, R = Ar) | 6 (X = S1, R = Ar) | 7 (X = S1, R = Si1) | |
|---|---|---|---|---|---|
V![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C [Å] C [Å] |
1.707(6) | 1.705(2) | 1.6995(16) | 1.694(3) | 1.6873(17) |
| V–O1 [Å] | 1.853(4) | 1.8757(14) | 1.8881(10) | 1.879(2) | 1.8707(11) |
| V–X [Å] | 2.089(6) | 1.9127(13) | 1.9253(10) | 2.3102(10) | 2.3331(4) |
| V–OMe [Å] | — | 2.409 | 2.436 | 2.506 | — |
V![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–Si [°] C–Si [°] |
167.6(4) | 158.72(13) | 164.55(11) | 164.7(2) | 164.21(12) |
| V–O1–Si [°] | 147.3(2) | 168.78(11) | 135.69(6) | 146.66(13) | 142.56(7) |
| V–X–R [°] | 116.8(3) | 122.56(13) | 123.39(9) | 106.24(11) | 111.24(2) |
We explored the reactivity of the complexes 4–7 with various alkynes (SI, Fig. S3). Interestingly, complex 5 does not react with tested alkynes even at 80 °C (5 decomposes above 80 °C). This observation supports the associative pathway. Thus, we hypothesize that the bulky 2,6-(MeO)2C6H3O ligand prevents coordination of the alkyne to the V center, which is crucial for initiating the cycloaddition step. Sulfur-containing alkylidynes 6 and 7 react with alkynes with decomposition. We were unable to confirm the formation of corresponding MCBDs by 1H NMR spectroscopy in both cases. Also, we did not observe any cycloreversion products by 1H NMR spectroscopy in reactions involving alkylidynes 5–7.
The reactions between 4 and PhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CMe and p-MeOC6H4C
CMe and p-MeOC6H4C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CMe at 60 °C showed formation of broad signals by 1H NMR spectrometry (Fig. S4 and S5), which might be explained by the formation of paramagnetic V complexes and/or polymers. Thus, in both cases, more than 1 equiv. of the alkyne was consumed, suggesting the formation of oligomers or polymers via MCBD ring-expansion, as previously described for Mo alkylidynes.6,48 We observed trace amounts of a cycloreversion product by 1H NMR spectroscopy (TMSC
CMe at 60 °C showed formation of broad signals by 1H NMR spectrometry (Fig. S4 and S5), which might be explained by the formation of paramagnetic V complexes and/or polymers. Thus, in both cases, more than 1 equiv. of the alkyne was consumed, suggesting the formation of oligomers or polymers via MCBD ring-expansion, as previously described for Mo alkylidynes.6,48 We observed trace amounts of a cycloreversion product by 1H NMR spectroscopy (TMSC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CMe, Fig. S4 and S5). As with reactions 6 and 7, we cannot confirm the formation of MCBDs in reaction with alkylidyne 4.
CMe, Fig. S4 and S5). As with reactions 6 and 7, we cannot confirm the formation of MCBDs in reaction with alkylidyne 4.
The destabilization of MCBDs is necessary for efficient alkyne metathesis but also introduces more complex reactivity that requires further investigation. While we do observe trace formation of cycloreversion products in the case of 4, we cannot confidently conclude that they arise from classical alkyne metathesis, since we did not observe the formation of MCBDs resulting from the cycloaddition step, and corresponding alkylidynes resulted from the cycloreversion step.
Conclusions
In conclusion, we showed that V alkyl alkylidynes can react with disubstituted alkynes to form corresponding 4-coordinate metallacyclobutadienes, which can be isolated and structurally characterized. Importantly, according to DFT studies, the cycloaddition step proceeds via a 6-coordinate intermediate, followed by the dissociation of a neutral ligand, in contrast to the classical Mo/W systems, where the 5-coordinate metallacycle is formed in one step directly from a 4-coordinate alkylidyne. The resulting V alkyl metallacycles are thermodynamically stable, as supported by experimental and computational studies, and do not readily undergo the cycloreversion reaction to complete the alkyne metathesis cycle. Based on DFT results, we found that introducing less σ-donating anionic ligands at the V alkylidyne can decrease the energy difference between alkylidyne and resulting metallacyclobutadiene, which can make the cycloreversion step more feasible. We successfully synthesized and characterized a series of V alkylidynes containing phenoxide, thiophenoxide, and thiolate ligands. However, some of them showed complex reactivity toward alkynes, likely due to the lower stability of the corresponding 4-coordinate MCBDs. One direction to address this issue is the introduction of polydentate ligands to facilitate the formation of 5-coordinate MCBDs that are isoelectronic to the classical Schrock systems. Nevertheless, we demonstrated the cycloaddition step involving V alkylidynes, a key step in alkyne metathesis, and provided theoretical insights into the mechanisms of cycloaddition and cycloreversion, which are essential milestones in the development of the first-row-mediated alkyne metathesis.Author contributions
S. H. and V. K. performed the synthetic experiments and analyzed the data. A. S. and X. S. performed DFT calculations. K. B. and X. S. wrote the manuscript. K. B. conceived and supervised the project.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2499223 (4) 2493563 (3) 2493564 (5) 2493565 (6) and 2493566 (7) contain the supplementary crystallographic data for this paper.49a–fAll experimental data associated with this work are available in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc01683a.
Acknowledgements
Research reported in this publication was supported by NSF under award number CHE-2442392.Notes and references
- A. Fürstner and P. W. Davies, Chem. Commun., 2005, 2307–2320, 10.1039/B419143A.
- R. R. Schrock and C. Czekelius, Adv. Synth. Catal., 2007, 349, 55–77 CrossRef CAS.
- W. Zhang and J. S. Moore, Adv. Synth. Catal., 2007, 349, 93–120 CrossRef CAS.
- A. Fürstner, Angew. Chem., Int. Ed., 2013, 52, 2794–2819 CrossRef PubMed.
- H. Ehrhorn and M. Tamm, Chem.–Eur. J., 2019, 25, 3190–3208 CrossRef CAS PubMed.
- D. Lee, I. Volchkov and S. Y. Yun, in Organic Reactions, 2020, pp. 613–931, DOI:10.1002/0471264180.or102.02.
- A. Fürstner, J. Am. Chem. Soc., 2021, 143, 15538–15555 CrossRef PubMed.
- F. R. Fischer and C. Nuckolls, Angew. Chem., Int. Ed., 2010, 49, 7257–7260 CrossRef CAS PubMed.
- S. von Kugelgen, R. Sifri, D. Bellone and F. R. Fischer, J. Am. Chem. Soc., 2017, 139, 7577–7585 CrossRef CAS PubMed.
- X. Jiang, J. D. Laffoon, D. Chen, S. Pérez-Estrada, A. S. Danis, J. Rodríguez-López, M. A. Garcia-Garibay, J. Zhu and J. S. Moore, J. Am. Chem. Soc., 2020, 142, 6493–6498 CrossRef CAS PubMed.
- S. Huang, Z. Lei, Y. Jin and W. Zhang, Chem. Sci., 2021, 12, 9591–9606 RSC.
- A. M. Beauchamp, J. Chakraborty, I. Ghiviriga, K. A. Abboud, D. W. Lester and A. S. Veige, J. Am. Chem. Soc., 2023, 145, 22796–22802 CrossRef PubMed.
- T. J. Katz and J. McGinnis, J. Am. Chem. Soc., 1975, 97, 1592–1594 CrossRef CAS.
- R. R. Schrock, Acc. Chem. Res., 1986, 19, 342–348 CrossRef CAS.
- R. R. Schrock, Chem. Rev., 2002, 102, 145–180 CrossRef CAS PubMed.
- R. R. Schrock, M. R. Buchmeiser, J. Groos, M. J. Benedikter, G. Parkin, K. Meyer and D. O’hare, in Comprehensive Organometallic Chemistry IV, Elsevier, Oxford, 2022, pp. 671–773, DOI:10.1016/B978-0-12-820206-7.00062-7.
- M. Cui, H. H. Y. Sung, I. D. Williams and G. Jia, J. Am. Chem. Soc., 2022, 144, 6349–6360 CrossRef CAS PubMed.
- D. S. Belov, C. M. Acosta, M. Garcia-Molina, K. Reu, X. Solans-Monfort and K. V. Bukhryakov, Organometallics, 2022, 41, 2897–2902 CrossRef CAS.
- V. Krivovicheva, M. S. R. Gangireddy, A. Liu, L. B. Maya, A. M. Mebel and K. V. Bukhryakov, J. Am. Chem. Soc., 2025, 147, 20212–20217 CrossRef CAS PubMed.
- D. S. Belov, G. Tejeda and K. V. Bukhryakov, ChemPlusChem, 2021, 86, 924–937 CrossRef CAS PubMed.
- W. S. Farrell, Z. Anorg. Allg. Chem., 2021, 647, 584–592 CrossRef CAS.
- K. Nomura, E. C. Constable, G. Parkin and L. Que Jr, in Comprehensive Coordination Chemistry III, Elsevier, Oxford, 2021, pp. 237–298 Search PubMed.
- K. Nomura, G. Parkin, K. Meyer and D. O’hare, in Comprehensive Organometallic Chemistry IV, Elsevier, Oxford, 2022, pp. 587–650, DOI:10.1016/B978-0-08-102688-5.00005-2.
- K. Mashima, Organometallics, 2021, 40, 3497–3505 CrossRef CAS.
- D. S. Belov, G. Tejeda, C. Tsay and K. V. Bukhryakov, Chem.–Eur. J., 2021, 27, 4578–4582 CrossRef CAS PubMed.
- D. S. Belov, D. A. Fenoll, I. Chakraborty, X. Solans-Monfort and K. V. Bukhryakov, Organometallics, 2021, 40, 2939–2944 CrossRef CAS.
- G. Tejeda, D. S. Belov, D. A. Fenoll, K. L. Rue, C. Tsay, X. Solans-Monfort and K. V. Bukhryakov, Organometallics, 2022, 41, 361–365 CrossRef CAS.
- W. S. Farrell, G. Tejeda, X. Solans-Monfort, É. Sá and K. V. Bukhryakov, J. Organomet. Chem., 2023, 996, 122753 CrossRef CAS.
- S. Aguero, V. Krivovicheva, M. S. R. Gangireddy and K. V. Bukhryakov, ACS Catal., 2025, 15, 18326–18332 CrossRef CAS.
- C. H. Suresh and G. Frenking, Organometallics, 2012, 31, 7171–7180 CrossRef CAS.
- M. Cui and G. Jia, J. Am. Chem. Soc., 2022, 144, 12546–12566 CrossRef CAS PubMed.
- F. Basuli, B. C. Bailey, D. Brown, J. Tomaszewski, J. C. Huffman, M.-H. Baik and D. J. Mindiola, J. Am. Chem. Soc., 2004, 126, 10506–10507 CrossRef CAS PubMed.
- D. Adhikari, F. Basuli, J. H. Orlando, X. Gao, J. C. Huffman, M. Pink and D. J. Mindiola, Organometallics, 2009, 28, 4115–4125 CrossRef CAS.
- M. G. Jafari, J. B. Russell, H. Lee, B. Pudasaini, D. Pal, Z. Miao, M. R. Gau, P. J. Carroll, B. S. Sumerlin, A. S. Veige, M.-H. Baik and D. J. Mindiola, J. Am. Chem. Soc., 2024, 146, 2997–3009 CrossRef CAS PubMed.
- M. G. Jafari, J. B. Russell, H. Myung, S. Kwon, P. J. Carroll, M. R. Gau, M.-H. Baik and D. J. Mindiola, Chem. Sci., 2024, 15, 19752–19763 RSC.
- S. Hernandez, D. S. Belov, V. Krivovicheva, S. Senthil and K. V. Bukhryakov, J. Am. Chem. Soc., 2024, 146, 18905–18909 CrossRef CAS PubMed.
- J. B. Russell, D. Konar, T. M. Keller, M. R. Gau, P. J. Carroll, J. Telser, D. W. Lester, A. S. Veige, B. S. Sumerlin and D. J. Mindiola, Angew. Chem., Int. Ed., 2024, 63, e202318956 CrossRef CAS PubMed.
- J. B. Russell, S. M. Smith, M. R. Gau and D. J. Mindiola, Polyhedron, 2024, 253, 116913 CrossRef CAS.
- A. Haack, J. Hillenbrand, M. van Gastel, A. Fürstner and F. Neese, ACS Catal., 2021, 11, 9086–9101 CrossRef CAS.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS.
- C. Lee, W. Yang and R. G. Parr, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785–789 CrossRef CAS PubMed.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed.
- C. Copéret, Z. J. Berkson, K. W. Chan, J. de Jesus Silva, C. P. Gordon, M. Pucino and P. A. Zhizhko, Chem. Sci., 2021, 12, 3092–3115 RSC.
- A. Poater, X. Solans-Monfort, E. Clot, C. Copéret and O. Eisenstein, J. Am. Chem. Soc., 2007, 129, 8207–8216 CrossRef CAS PubMed.
- Q.-H. Zhu, L. Zhang, G.-H. Zhang, G.-H. Tao, S. Qin, H. Chen, W.-L. Yuan, Y.-H. Wang, Y. Jin, L. Ma, L. He and W. Zhang, Mol. Catal., 2022, 531, 112696 CAS.
- D. P. Estes, C. P. Gordon, A. Fedorov, W.-C. Liao, H. Ehrhorn, C. Bittner, M. L. Zier, D. Bockfeld, K. W. Chan, O. Eisenstein, C. Raynaud, M. Tamm and C. Copéret, J. Am. Chem. Soc., 2017, 139, 17597–17607 CrossRef CAS PubMed.
- S. A. DiFranco, N. A. Maciulis, R. J. Staples, R. J. Batrice and A. L. Odom, Inorg. Chem., 2012, 51, 1187–1200 CrossRef CAS PubMed.
- L. G. McCullough and R. R. Schrock, J. Am. Chem. Soc., 1984, 106, 4067–4068 CrossRef CAS.
- (a) CCDC 2499223: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pwn3c; (b) CCDC 2493563: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pprjp; (c) CCDC 2493564: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pprkq; (d) CCDC 2493565: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pprlr; (e) CCDC 2493566: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pprms; (f) CCDC 2493567: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pprnt.
| This journal is © The Royal Society of Chemistry 2026 |