Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
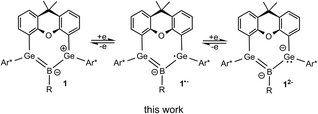
Three reversibly interconvertible redox states of boradigermaallyl: syntheses of radical allyl anion and allyl dianion
Stefan F.
Miehe
a,
Klaus
Eichele
a,
Hartmut
Schubert
a,
Holger F.
Bettinger
 b,
Christian P.
Sindlinger
b,
Christian P.
Sindlinger
 *c and
Lars
Wesemann
*c and
Lars
Wesemann
 *a
*a
aInstitut für Anorganische Chemie, Eberhard Karls Universität Tübingen, Auf der Morgenstelle 18, 72076 Tübingen, Germany. E-mail: lars.wesemann@uni-tuebingen.de
bInstitut für Organische Chemie, Eberhard Karls Universität Tübingen, Auf der Morgenstelle 18, 72076 Tübingen, Germany. E-mail: holger.bettinger@uni-tuebingen.de
cInstitut für Anorganische Chemie, Universität Stuttgart, Pfaffenwaldring 55, 70569 Stuttgart, Germany. E-mail: christian.sindlinger@iac.uni-stuttgart.de
First published on 2nd March 2026
Abstract
A methyl derivate of our previously published chloro-boradigermaallyl, which features a borylene unit stabilized by a chelating bis-germylene ligand, is synthesized by addition of MeBBr2 to the bis(germylene) A followed by KC8 reduction. Both derivatives, the BCl (1a) and BMe (1b) boradigermaallyls, feature an allyl-type delocalized Ge–B–Ge 2π-electron system. In this work, the reversible two step reduction of both compounds to the persistent radical anions and dianions is presented. EPR data and particularly hyperfine coupling constants to the 11B and 73Ge nuclei confirm allyl-type delocalization of the radicals. Computed spin densities illustrate the structural analogy to the organic allyl radical. Cyclic voltammetry measurements of the boradigermaallyl compounds exhibit one reduction wave within the accessible electrochemical window indicating a reduction potential of the dianionic species beyond −2.2 V [vs. (Ag/Ag+)].
Introduction
The allyl cation, radical and anion are small resonance stabilized organic compounds that represent examples of fundamental structural building blocks in organic matter.1–5 With three aligned p-orbitals occupied with two, three or four π-electrons, these molecules belong to the group of elementary conjugated systems. As intermediates in organic chemistry, the allyl cation and allyl radical play an important role in many synthetic transformations.6–9 The allyl anion on the other hand, which is used as allyl lithium or allyl magnesium halide for example, is a versatile starting material for organic and organometallic syntheses.10–14 In heavier group-14 element chemistry the search for heavy analogues of unsaturated organic molecules is a field of major interest (for example alkenes,15–21 allenes,22 alkynes,23–25 cyclopropenes,26 benzene,27,28 vinylidene29,30).26,31–33 A variety of group-15 element analogues of the allyl anion has been published.34–38 Heavy analogues of allyl cations have been presented for bismuth, silicon and germanium.39–42 Schleyer and coworkers studied the structures of the trisilaallyl anion [Si3H5]− using quantum chemical computations and discussed hyperconjugation as a substantial stabilization for the nonplanar structures.43 Sekiguchi and coworkers synthesized an allyl-type radical and anion of silicon reducing the cyclotetrasilenylium cation [(t-Bu2MeSiSi)3Sit-Bu2]+ with alkali metals Li, Na or potassium graphite (KC8), vide infra.44–47With the insertion of boron trichloride into a bis(germylene) A (Scheme 1), followed by a two-electron reduction of the addition product B, we have recently synthesized an allyl cation analogue 1a featuring delocalization of two electrons in the Ge–B–Ge π-system generated by linear combination of the three vacant p-orbitals.48
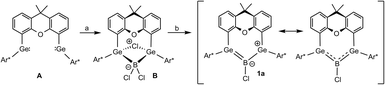 | ||
| Scheme 1 Synthesis of boradigermaallyl [(a) Me2S·BCl3, (b) {(MesNacnac)Mg}2] [Ar* = C6H3-2,6-(Trip)2, Trip = 2,4,6-C6H2-iPr3, MesNacnac = {[(Mes)NC(Me)]2CH}−, Mes = 2,4,6-C6H2Me3].48,49 | ||
With the aim of reductively breaking the remaining B–Cl bond in 1a, we discovered that 1a can be reduced (Scheme 2). This led us to investigate the redox properties of boradigermaallyl.
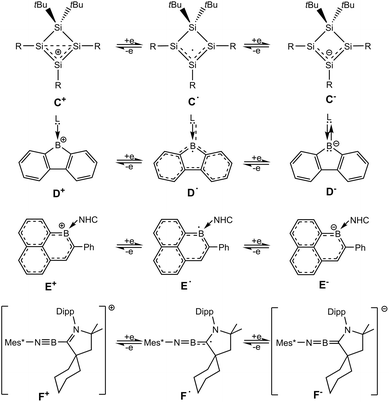
Molecular compounds that can be transferred reversibly into three isolable redox states are recently of interest in main group element chemistry.37,50–64 However, because of redox instabilities, compounds accessible in three redox states are rare. The cyclotetrasilenylium cation (C+, Scheme 3) was reduced by Sekiguchi and co-workers treating the cation with Li, Na or KC8 to give the red-purple coloured radical.46 The subsequent reduction of the cyclotetrasilenyl radical was carried out using lithium as reducing agent. The reversibility of the reduction has been demonstrated treating the anion with [Et3Si(C6H6)][B(C6F5)4] to give the radical as the product of a one-electron oxidation and finally the radical is oxidized after addition of [Ph3C][B(C6F5)4].45,46
Reduction of a CAAC- or NHC-adduct of borafluorene cations yields the radical species (D˙, Scheme 3).65,66 Monoanionic borafluorene adducts were obtained by reducing the 9-bromo-9-borafluorene adducts with strong reducing reagents (Li-naphthalenide, Na, KC8).66–70 The interconversion of a NHC-adduct of boraphenalene (E+, Scheme 3) into three different redox states has been presented with the reversible one- and two-electron reduction using KC8 and oxidation in reaction with AgSbF6.71 The CAAC-adduct of a phenyl substituted iminoboryl compound (F+, Scheme 3) shows a one-electron reduction in reaction with cobaltocene Cp*2Co to give the radical and the anion is formed after addition of Li/naphthalene.72 A T-shaped organoboron dication with a PNP-pincer ligand was reversibly reduced to the radical cation and neutral base-stabilized borylene.63 Furthermore, both oxidation products of a BN analogue of Thiele's hydrocarbon, a radical cation and after the second oxidation the dication, have been isolated and characterized.73 Most recently the reversible electron transfer in the formation of an anionic radical and dianionic 1,4-diborabutatriene has been reported.74
In this manuscript we present the synthesis of methyl-substituted boradigermaallyl, the reversible stepwise two-electron reduction of boradigermaallyl derivatives 1 and discuss the structural and spectroscopic findings of the reduction products (Scheme 2).
Results and discussion
In the first step of this project, we synthesized a methyl substituted derivative of boradigermaallyl because we wanted to compare the properties of the chloro- and methyl-substituted allyl system 1. The oxidative addition of MeBBr2·SMe2 to the bis(germylene) A is a straightforward reaction (Scheme 4) and the product 2 was isolated as yellow crystals in 88% yield. Reduction was carried out treating the bromide 2 with two equivalents of KC8 in Et2O at room temperature. The solution turned green and finally blue to yield blue crystals of the methyl boradigermaallyl 1b in a yield of 82% after crystallization at −40 °C.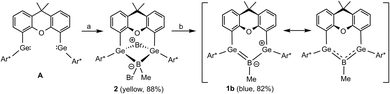 | ||
| Scheme 4 Synthesis of the methyl derivative 1b of boradigermaallyl ((a) n-pentane, 1 equiv. MeBBr2·SMe2, 2 h, rt; (b) Et2O, 2 equiv. KC8, rt, 30 min). | ||
The molecular structure of the MeBBr2 addition product 2, presented in Fig. 1, can be compared with the BCl3 addition product B (Scheme 1) and shows Ge–B distances in the same range [B: Ge–B 2.121(2), 2.117(2) Å]. Methyl-boradigermaallyl 1b, the molecular structure of which is shown in Fig. 1, exhibits short Ge–B distances of 1.948(5) and 1.962(4) Å which are almost as long as those in the chloro derivative 1a [Ge–B 1.960(2), 1.962(2) Å] and can be compared to Ge–B double bonds of germaborenes [1.886(2)–1.967(4) Å].20,75–77 The Ge–B–Ge angle of 121.4(2)° is slightly more acute than in 1a [Ge–B–Ge 126.7(1)°]. The Ge–B–C angles in 1b [118.4(1), 120.1(1)°] are slightly larger than the Ge–B–Cl angles in 1a [116.2(1), 117.1(1)°], which is in line with Bent's rule.78,79 The signal in the 11B NMR spectrum of 1b was observed at 41.9 ppm (1a 42.4 ppm). The solution UV-Vis spectrum of 1b features an absorption band at 636 nm. Based on TD-DFT calculations we qualitatively assign this absorption to the HOMO → LUMO transition (see SI).
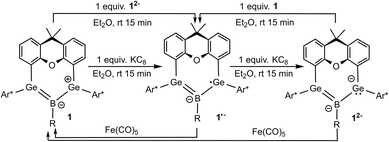
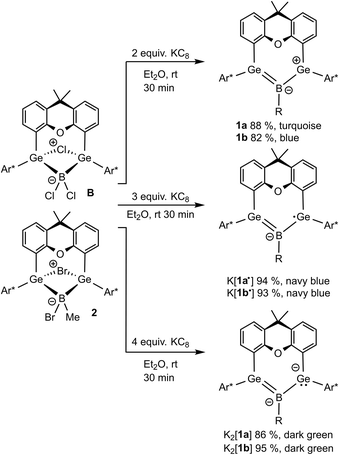
The reduction of boradigermaallyl derivatives 1a and 1b (Fig. 3 and Scheme 5) was initially carried out by reaction with one equivalent of KC8 in diethyl ether at room temperature to yield the persistent radical anions 1˙− as potassium salts. However, the reduction of the halides 2 and B has been established as a straightforward synthesis of radical anions 1a˙− and 1b˙− (Scheme 6). Since three equivalents of KC8 are used in this procedure, the weighing error is comparatively smaller and there is a smaller chance of over-reduction. During our investigations, no decomposition of 1˙− was observed at room temperature in solution over a period of several weeks.
 | ||
| Scheme 5 Reduction of 1a, 1b and radical anions 1a˙− and 1b˙−. oxidation of dianions 1a2−, 1b2− and radical anions 1a˙− and 1b˙− ((a) R = Cl, (b) R = Me). | ||
 | ||
| Scheme 6 Syntheses of boradigermaallyl (1a, 1b), radical anion salts (K[1a˙], K[1b˙]) and dianion salts (K2[1a], K2[1b]) ((a) R = Cl, (b) R = Me). | ||
The reversibility of the reduction was demonstrated by oxidation of 1˙− to the starting material 1 through the addition of iron pentacarbonyl (Scheme 5). Crystallization at −40 °C in diethyl ether gives navy blue crystals of K[1a˙] and K[1b˙]. The molecular structure of the potassium salt of the reduction product K[1a˙] is depicted in Fig. 2.
The radical anion 1a˙− exhibits almost no elongation of the Ge–B bonds lengths in comparison to the starting material 1a. The Ge–B–Ge angle of 131.8(1)° is significantly more obtuse than in 1a [Ge–B–Ge 126.7(1)°]. However, isolated crystals of K[1b˙] were of low quality, and only the positions of the heavy atoms, which have a structural motif analogous to K[1a˙], could be determined. The solution UV-Vis spectra of K[1a˙] and K[1b˙] feature a broadened absorption around 606 nm (K[1a˙]) and 650 nm (K[1b˙]). Based on TD-DFT calculations we qualitatively assign these absorptions to the SOMO → LUMO + n (n = 0,1,2) transitions (see SI).
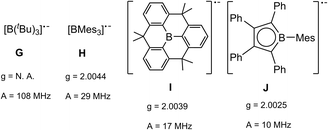
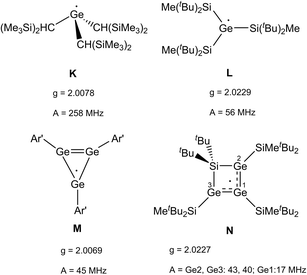
The radical character of K[1a˙] and K[1b˙] became evident by the fact that the compounds are NMR silent. The continuous-wave EPR spectra were measured in diethyl ether at room temperature and subsequently simulated (see SI for details). The spectra (Fig. 4, see SI for EPR of 1b˙−) of 1a˙− and 1b˙− yield isotropic g-factors of giso = 2.0116 (1a˙−) and giso = 2.0161 (1b˙−).80,81 Due to hyperfine coupling with the 11B (I = 3/2, 80.1% abundance) and 73Ge (I = 9/2, 7.8% abundance) isotopes 1˙− exhibit spectra with a central four-line resonance [Aiso(11B) = −14.1 MHz (1a˙−, 1b˙−)] featuring 73Ge hyperfine coupling [Aiso(73Ge) = −59.65 MHz (1a˙−) and −50.69 MHz (1b˙−)]. The EPR parameters of 1a˙− and 1b˙− were computed using different density functional methods, dispersion corrections and solvation models (see SI). The computed giso values of 1a˙− and 1b˙− are in excellent agreement with the experimental data across all methods [1a˙− (giso = 2.0131–2.0116), 1b˙− (giso = 2.0124–2.015)]. Regarding the hyperfine coupling constants, the ωB97X-V//CAM-B3LYP method provides values for 1a˙− which are very close to the experimental reference, however, the experimental values of 1b˙− could not be reproduced as accurately using the same method [results of computations: Aiso(73Ge) = −58.35 MHz (1a˙−) and −41.87 (1b˙−), Aiso(11B) = −14.12 MHz (1a˙−) and −20.64 (1b˙−)]. Spin density distribution of 1a˙− (Fig. 4) was computed using different functionals (see SI). A dominant localization of the spin density on the two germanium atoms with a small negative spin density at the boron centre and partial delocalization onto the xanthene backbone was found. Mulliken atomic spin densities of the boradigermaallyl radical anion 1a˙− [ωB97X-V/CPCM(Et2O), Ge1: 0.565, Ge2: 0.563, B: −0.224] indicate a structural resemblance to the allyl radical (C1H2 = C2H–C3H2˙; C1, C3: 0.575 (ref. 82)/0.583,83 C2: −0.179 (ref. 82)/−0.170 (ref. 83)) showing comparable spin densities and illustrate the textbook character of the boradigermaallyl system. The hyperfine coupling constant (hfcc) Aiso(11B) for the boron atom in 1a˙− and 1b˙− of −14.1 MHz is smaller in magnitude than those observed in G and H (Scheme 7). This indicates a less localized spin density on boron atoms in 1a˙− and 1b˙− and can be compared with delocalized spin densities found in I (A = 17 MHz) and J (A = 10 MHz) featuring comparable hfccs for the boron atom. In the case of the hyperfine coupling constants Aiso(73Ge) for the germanium atoms [Aiso(73Ge) = −59.65 MHz (1a˙−), −50.69 MHz (1b˙−)] the observed small hfccs also indicate a delocalized π-radical. The values are much smaller than the hfcc found in the pyramidal radical K featuring s-orbital character in the SOMO (Scheme 8).84–86 The hfcc of 1a˙− and 1b˙− can be compared with the values documented for radicals L, M and N (Scheme 8). Compound L is a planar π-radical with the 3pz orbital as the SOMO and the unpaired electron in radicals M and N reside in an orbital of π-symmetry.42,47,87,88
Reduction of the radical anions (Fig. 3) with a further equivalent of KC8 yields the dianions K2[1a] and K2[1b] (Scheme 5). As the best preparative procedure for the syntheses of the dianions the reduction of the BCl3 and MeBBr2 insertion products B and 2 with four equivalents of KC8 at room temperature in diethyl ether was developed (Scheme 6). Crystallization was carried out at −40 °C in n-pentane to give dark green crystals of K2[1a] and K2[1b]. The molecular structures of the salts are depicted in Fig. 5 and further coordination of the potassium cations in aromatic moieties of the organic ligands and coordination at dianionic BGe2-units is shown. Both dianions feature a slight elongation of one of the Ge–B bonds [1a2− Ge2–B 2.045(2), 1b2− Ge2–B 2.017(6) Å] in comparison to the boradigermaallyl structures [1a 1.960(2), 1.962(2), 1b 1.962(4), 1.948(5) Å] which could possibly be due to the population of the nonbonding combination of the three aligned p-orbitals upon reduction (Fig. 3, right). The Ge1–B–Ge2 angle in 1a2− of 129.4(1)° only slightly deviates from the corresponding radical [1a˙− Ge1–B–Ge2 131.8(1)°]. Interestingly, the boron atom in 1a2− shows a slight pyramidalization in the solid-state structure with a sum of bond angles around boron of 350.9°. Both salts show a signal in the 11B NMR spectrum at higher frequencies (K2[1a] 57.0, K2[1b] 55.5 ppm) in comparison to the boradigermaallyl derivatives (1a 42.4, 1b 41.9 ppm). On first sight, this observation may appear puzzling. However, quantum chemical calculations of magnetic shielding tensors of 1a and K2[1a] reproduce the experimental findings and indicate that this effect is caused by a significant deshielding contribution in σ11 along the direction of the B–Cl bond. This contribution arises from magnetic dipole allowed efficient mixing of the HOMO in 1a2− with low lying σ* orbitals (see SI for more details). The excited state spectra of K2[1a] and K2[1b] were investigated by TD-DFT calculations (see SI). HOMO–LUMO + n (n = 1,2,3,4) transitions are in the visible range and are there assigned to cause the dark green colour of the compounds. It is worth noting, that due to the high sensitivity of K2[1a] and K2[1b] it is experimentally challenging to obtain meaningful UV-Vis spectra even from highest purity solvents upon dilution.
Oxidation of the dianions K2[1a] and K2[1b] to yield the radical anions K[1a] and K[1b] (Scheme 5) was achieved by reaction with one equivalent of boradigermaallyl 1a and 1b, respectively. Oxidation by treating the dianions K2[1a] and K2[1b] with two equivalents of iron pentacarbonyl led in both cases to the formation of 1a and 1b (Scheme 5).
Electrochemical properties of the boradigermaallyl derivatives 1a and 1b were analyzed by cyclic voltammetry in THF using the silver/silver cation (Ag/Ag+) redox couple as reference and [Bu4N][Al(OC{CF3}3)4] salt as supporting electrolyte (Fig. 6, see SI for cyclic voltammogram of 1b).95 Both compounds reveal a reduction wave (1a −1.38 V, 1b −1.67 V) and an oxidation wave (1a 0.10 V, 1b 0.16 V).
The observed difference of reduction potentials is in accordance with the pronounced electron-withdrawing character of the chloro substituent. In the case of the reduction wave of 1a, which was obtained at different scan rates, a one electron transfer process can be estimated using the modified Randles-Ševčík equation for quasi-reversible processes (see SI).96 Redox potentials were computed at the r2SCAN-3c/CPCM(THF) level of theory analogously to a protocol published by Grimme and co-workers.97 Since the potassium counterion was not considered, the absolute values should be interpreted with caution. Nevertheless, the computed data clearly show a separation of the reduction potentials of redox couples 1|1˙− and 1˙−|12− by approximately 0.5 V (see SI). This leads us to the conclusion that the recorded reduction wave corresponds to the reaction 1 + e− ⇌ 1˙− and not to a single-step two electron reduction. A second reduction step is not observed within the accessible electrochemical window, as the potential required exceeds the stability limit of THF below −2.2 V. The electrochemical oxidation of the low valent boron compounds 1a and 1b was investigated by preparative oxidation reactions with Ag[Al(OC{CF3}3)4], NOPF6 and [Cp2Fe][PF6]. However, we were not able to isolate any product of the oxidation.98
Conclusion
In summary, methyl- and chloroborylene, stabilized by a chelating bis(diarylgermylene), form delocalized neutral allyl-cation analogues that exhibit stepwise two-electron reduction. After isolation of the persistent radical anioni and dianion as the products of reduction, their preparative oxidation confirms the interconvertible nature of the redox products. The radical boradigermaallyl species exhibit delocalization of the radical on the Ge–B–Ge moiety with spin densities comparable to the C3H5 allyl radical.Author contributions
Investigations, computational investigations, writing, review S. F. M.; special NMR experiments K. E.; X-ray measurements and structure determinations H. S.; supervision, funding acquisition, manuscript writing and review H. F. B.; electrochemistry, supervision, funding acquisition, manuscript writing and review C. P. S.; supervision, funding acquisition, manuscript writing and review L. W.Conflicts of interest
The authors declare no conflicts of interest.Data availability
Full experimental and computation details are provided as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc00727a.CCDC 2504370–2504374 contain the supplementary crystallographic data for this paper.99a–e
Acknowledgements
The authors acknowledge support by the state of Baden-Württemberg through bwHPC and the German Research Foundation (DFG) through grant no. INST 40/575-1 FUGG (Justus 2 cluster) as well as WE 1876/16-1 and BE 3183/13-1.Notes and references
- A. Gobbi and G. Frenking, J. Am. Chem. Soc., 1994, 116, 9275–9286 CrossRef CAS.
- G. E. Douberly, A. M. Ricks, P. v. R. Schleyer and M. A. Duncan, J. Chem. Phys., 2008, 128, 021102 CrossRef PubMed.
- V. Mišić, K. Piech and T. Bally, J. Am. Chem. Soc., 2013, 135, 8625–8631 CrossRef.
- L. Radom, P. C. Hariharan, J. A. Pople and P. V. R. Schleyer, J. Am. Chem. Soc., 1973, 95, 6531–6544 CrossRef CAS.
- M. Alagia, E. Bodo, P. Decleva, S. Falcinelli, A. Ponzi, R. Richter and S. Stranges, Phys. Chem. Chem. Phys., 2013, 15, 1310–1318 RSC.
- B. M. Trost and M. L. Crawley, Chem. Rev., 2003, 103, 2921–2944 CrossRef CAS PubMed.
- H.-M. Huang, P. Bellotti and F. Glorius, Chem. Soc. Rev., 2020, 49, 6186–6197 RSC.
- L. Debien, B. Quiclet-Sire and S. Z. Zard, Acc. Chem. Res., 2015, 48, 1237–1253 CrossRef CAS PubMed.
- G. E. Keck and J. B. Yates, J. Am. Chem. Soc., 1982, 104, 5829–5831 CrossRef CAS.
- H. Köster and E. Weiss, Chem. Ber., 1982, 115, 3422–3426 CrossRef.
- P. West, J. I. Purmort and S. V. McKinley, J. Am. Chem. Soc., 1968, 90, 797–798 CrossRef CAS.
- Y. Yamamoto and N. Asao, Chem. Rev., 1993, 93, 2207–2293 CrossRef CAS.
- C. Lichtenberg and J. Okuda, Angew. Chem., Int. Ed., 2013, 52, 5228–5246 CrossRef CAS PubMed.
- I. Georg, M. Bursch, B. Endeward, M. Bolte, H.-W. Lerner, S. Grimme and M. Wagner, Chem. Sci., 2021, 12, 12419–12428 RSC.
- R. West, M. J. Fink and J. Michl, Science, 1981, 214, 1343–1344 CrossRef CAS PubMed.
- V. Y. Lee, S. Aoki, M. Kawai, T. Meguro and A. Sekiguchi, J. Am. Chem. Soc., 2014, 136, 6243–6246 CrossRef CAS PubMed.
- N. Nakata and A. Sekiguchi, J. Am. Chem. Soc., 2006, 128, 422–423 CrossRef CAS PubMed.
- H. Meyer, G. Baum, W. Massa, S. Berger and A. Berndt, Angew. Chem., Int. Ed., 1987, 26, 546–548 CrossRef.
- M. Driess and H. Grützmacher, Angew. Chem., Int. Ed., 1996, 35, 828–856 CrossRef CAS.
- B. Rao and R. Kinjo, Angew. Chem., Int. Ed., 2020, 59, 3147–3150 CrossRef CAS PubMed.
- B. Pampuch, W. Saak and M. Weidenbruch, J. Organomet. Chem., 2006, 691, 3540–3544 CrossRef CAS.
- J. Escudié, H. Ranaivonjatovo and L. Rigon, Chem. Rev., 2000, 100, 3639–3696 CrossRef PubMed.
- L. Pu, A. D. Phillips, A. F. Richards, M. Stender, R. S. Simons, M. M. Olmstead and P. P. Power, J. Am. Chem. Soc., 2003, 125, 11626–11636 CrossRef CAS PubMed.
- M. Stender, A. D. Phillips, R. J. Wright and P. P. Power, Angew. Chem., Int. Ed., 2002, 41, 1785–1787 CrossRef CAS.
- T. Sasamori, T. Sugahara, T. Agou, K. Sugamata, J.-D. Guo, S. Nagase and N. Tokitoh, Chem. Sci., 2015, 6, 5526–5530 RSC.
- A. Sekiguchi and V. Y. Lee, Chem. Rev., 2003, 103, 1429–1448 CrossRef CAS PubMed.
- K. Abersfelder, A. J. P. White, H. S. Rzepa and D. Scheschkewitz, Science, 2010, 327, 564–566 CrossRef CAS.
- T. Sugahara, J.-D. Guo, D. Hashizume, T. Sasamori and N. Tokitoh, J. Am. Chem. Soc., 2019, 141, 2263–2267 CrossRef CAS PubMed.
- A. Rit, J. Campos, H. Niu and S. Aldridge, Nat. Chem., 2016, 8, 1022–1026 CrossRef CAS PubMed.
- D. Scheschkewitz, Angew. Chem., Int. Ed., 2004, 43, 2965–2967 CrossRef CAS PubMed.
- P. P. Power, Chem. Rev., 1999, 99, 3463–3504 CrossRef CAS PubMed.
- R. C. Fischer and P. P. Power, Chem. Rev., 2010, 110, 3877–3923 CrossRef CAS PubMed.
- P. P. Power, Acc. Chem. Res., 2011, 44, 627–637 CrossRef CAS PubMed.
- A. Hinz, A. Schulz and A. Villinger, Chem. Commun., 2015, 51, 11437–11440 RSC.
- E. Niecke, B. Kramer and M. Nieger, Organometallics, 1991, 10, 10–11 CrossRef CAS.
- H.-W. Lerner, I. Sänger, F. Schödel, A. Lorbach, M. Bolte and M. Wagner, Dalton Trans., 2008, 787–792 RSC.
- M. Stubenhofer, C. Kuntz, M. Bodensteiner, U. Zenneck, M. Sierka and M. Scheer, Chem.–Eur. J., 2010, 16, 7488–7495 CrossRef CAS PubMed.
- L. S. H. Dixon, L. K. Allen, R. J. Less and D. S. Wright, Chem. Commun., 2014, 50, 3007–3009 RSC.
- D. Spinnato, N. Nöthling, M. Leutzsch, M. van Gastel, L. Wagner, F. Neese and J. Cornella, Nat. Chem., 2025, 17, 265–270 CrossRef CAS PubMed.
- M. Tsukasa, S. Katsunori, F. Tomohide, L. Baolin, I. Mikinao, S. Yoshiaki, O. Takashi, L. Liangchun, K. Megumi, H. Makoto, T. Yoshiyuki, H. Daisuke, F. Takeo, F. Aiko, L. Yongming, T. Hayato and T. Kohei, Bull. Chem. Soc. Jpn., 2011, 84, 1178–1191 CrossRef.
- J. Keuter, A. Hepp, A. Massolle, J. Neugebauer, C. Mück-Lichtenfeld and F. Lips, Angew. Chem., Int. Ed., 2022, 61, e202114485 CrossRef CAS PubMed.
- M. M. Olmstead, L. Pu, R. S. Simons and P. P. Power, Chem. Commun., 1997, 1595–1596 RSC.
- A. A. Korkin and P. v. R. Schleyer, J. Am. Chem. Soc., 1992, 114, 8720–8722 CrossRef CAS.
- A. Sekiguchi, T. Matsuno and M. Ichinohe, J. Am. Chem. Soc., 2000, 122, 11250–11251 CrossRef CAS.
- A. Sekiguchi, T. Matsuno and M. Ichinohe, J. Am. Chem. Soc., 2001, 123, 12436–12437 CrossRef CAS PubMed.
- T. Matsuno, M. Ichinohe and A. Sekiguchi, Angew. Chem., Int. Ed., 2002, 41, 1575–1577 CrossRef CAS.
- V. Y. Lee, Y. Ito, O. A. Gapurenko, R. M. Minyaev, H. Gornitzka and A. Sekiguchi, J. Am. Chem. Soc., 2020, 142, 16455–16460 CrossRef CAS PubMed.
- R. H. Kern, M. Schneider, K. Eichele, H. Schubert, H. F. Bettinger and L. Wesemann, Angew. Chem., Int. Ed., 2023, 62, e202301593 CrossRef CAS PubMed.
- S. P. Green, C. Jones and A. Stasch, Science, 2007, 318, 1754–1757 CrossRef CAS PubMed.
- K. Deuchert and S. Hünig, Angew. Chem., Int. Ed., 1978, 17, 875–886 CrossRef.
- Y. Li, K. C. Mondal, P. P. Samuel, H. Zhu, C. M. Orben, S. Panneerselvam, B. Dittrich, B. Schwederski, W. Kaim, T. Mondal, D. Koley and H. W. Roesky, Angew. Chem., Int. Ed., 2014, 53, 4168–4172 CrossRef CAS PubMed.
- M. M. Hansmann, M. Melaimi, D. Munz and G. Bertrand, J. Am. Chem. Soc., 2018, 140, 2546–2554 CrossRef CAS PubMed.
- M. M. Hansmann, M. Melaimi and G. Bertrand, J. Am. Chem. Soc., 2018, 140, 2206–2213 CrossRef CAS PubMed.
- P. W. Antoni and M. M. Hansmann, J. Am. Chem. Soc., 2018, 140, 14823–14835 CrossRef CAS PubMed.
- P. W. Antoni, T. Bruckhoff and M. M. Hansmann, J. Am. Chem. Soc., 2019, 141, 9701–9711 CrossRef CAS PubMed.
- J. Messelberger, A. Grünwald, S. J. Goodner, F. Zeilinger, P. Pinter, M. E. Miehlich, F. W. Heinemann, M. M. Hansmann and D. Munz, Chem. Sci., 2020, 11, 4138–4149 RSC.
- L. A. Freeman, A. D. Obi, H. R. Machost, A. Molino, A. W. Nichols, D. A. Dickie, D. J. D. Wilson, C. W. Machan and R. J. Gilliard, Chem. Sci., 2021, 12, 3544–3550 RSC.
- D. Munz, J. Chu, M. Melaimi and G. Bertrand, Angew. Chem., Int. Ed., 2016, 55, 12886–12890 CrossRef CAS PubMed.
- A. Mahata, S. Chandra, A. Maiti, D. K. Rao, C. B. Yildiz, B. Sarkar and A. Jana, Org. Lett., 2020, 22, 8332–8336 CrossRef CAS PubMed.
- D. D. Hebert, A. Puri, D. Ye, A. McAninch, A. Chisholm, M. A. Siegler, M. Swart and I. Garcia-Bosch, Inorg. Chem., 2025, 64, 11204–11218 CrossRef CAS PubMed.
- A. D. Ready, Y. A. Nelson, D. F. T. Pomares and A. M. Spokoyny, Acc. Chem. Res., 2024, 57, 1310–1324 CrossRef CAS PubMed.
- J. Xu, S. Yao, V. Postils, E. Matito, C. Lorent and M. Driess, Chem. Sci., 2025, 16, 10826–10832 RSC.
- W. Lv, Y. Dai, R. Guo, Y. Su, D. A. Ruiz, L. L. Liu, C.-H. Tung and L. Kong, Angew. Chem., Int. Ed., 2023, 62, e202308467 CrossRef CAS PubMed.
- L. Zhu, C. Du, Y. Yang and C. Cui, J. Am. Chem. Soc., 2025, 147, 31042–31048 CrossRef CAS PubMed.
- W. Yang, K. E. Krantz, L. A. Freeman, D. A. Dickie, A. Molino, G. Frenking, S. Pan, D. J. D. Wilson and R. J. Gilliard Jr, Angew. Chem., Int. Ed., 2020, 59, 3850–3854 CrossRef CAS PubMed.
- K. K. Hollister, K. E. Wentz and R. J. Gilliard Jr, Acc. Chem. Res., 2024, 57, 1510–1522 CrossRef CAS PubMed.
- C. J. Berger, G. He, C. Merten, R. McDonald, M. J. Ferguson and E. Rivard, Inorg. Chem., 2014, 53, 1475–1486 CrossRef CAS PubMed.
- H. Budy, T. Kaese, M. Bolte, H.-W. Lerner and M. Wagner, Angew. Chem., Int. Ed., 2021, 60, 19397–19405 CrossRef CAS PubMed.
- J. Gilmer, H. Budy, T. Kaese, M. Bolte, H.-W. Lerner and M. Wagner, Angew. Chem., Int. Ed., 2020, 59, 5621–5625 Search PubMed.
- K. E. Wentz, A. Molino, S. L. Weisflog, A. Kaur, D. A. Dickie, D. J. D. Wilson and R. J. Gilliard Jr., Angew. Chem., Int. Ed., 2021, 60, 13065–13072 Search PubMed.
- C.-L. Deng, K. K. Hollister, A. Molino, B. Y. E. Tra, D. A. Dickie, D. J. D. Wilson and R. J. Gilliard Jr, J. Am. Chem. Soc., 2024, 146, 6145–6156 CrossRef CAS PubMed.
- X. Mao, S. Qiu, R. Guo, Y. Dai, J. Zhang, L. Kong and Z. Xie, J. Am. Chem. Soc., 2024, 146, 10917–10924 CrossRef CAS PubMed.
- Y. K. Loh, P. Vasko, C. McManus, A. Heilmann, W. K. Myers and S. Aldridge, Nat. Commun., 2021, 12, 7052 Search PubMed.
- L. Zhu, C. Du, Y. Yang and C. Cui, J. Am. Chem. Soc., 2025, 147, 31042–31048 CrossRef CAS PubMed.
- D. Raiser, H. Schubert, H. F. Bettinger and L. Wesemann, Chem.–Eur. J., 2021, 27, 1981–1983 CrossRef CAS.
- D. Raiser, C. P. Sindlinger, H. Schubert and L. Wesemann, Angew. Chem., Int. Ed., 2020, 59, 3151–3155 CrossRef CAS PubMed.
- C. Reik, L. W. Jenner, H. Schubert, K. Eichele and L. Wesemann, Chem. Sci., 2024, 15, 11358–11366 RSC.
- H. A. Bent, Chem. Rev., 1961, 61, 275–311 CrossRef CAS.
- H. A. Bent, J. Chem. Educ., 1960, 37, 616 CrossRef CAS.
- P. P. Power, Chem. Rev., 2003, 103, 789–810 Search PubMed.
- Y. Su and R. Kinjo, Coord. Chem. Rev., 2017, 352, 346–378 CrossRef CAS.
- F. Aquilante, K. P. Jensen and B. O. Roos, Chem. Phys. Lett., 2003, 380, 689–698 CrossRef CAS.
- D. Lazdins and M. Karplus, J. Chem. Phys., 1966, 44, 1600–1611 CrossRef CAS.
- J. D. Cotton, C. S. Cundy, D. H. Harris, A. Hudson, M. F. Lappert and P. W. Lednor, Chem. Commun., 1974, 651–652 RSC.
- A. Hudson, M. F. Lappert and P. W. Lednor, Dalton Trans., 1976, 2369–2375 Search PubMed.
- M. P. Egorov, O. M. Nefedov, T.-S. Lin and P. P. Gaspar, Organometallics, 1995, 14, 1539–1541 CrossRef CAS.
- V. Y. Lee, M. Nakamoto and A. Sekiguchi, Chem. Lett., 2007, 37, 128–133 Search PubMed.
- W. D. Woodul, E. Carter, R. Müller, A. F. Richards, A. Stasch, M. Kaupp, D. M. Murphy, M. Driess and C. Jones, J. Am. Chem. Soc., 2011, 133, 10074–10077 CrossRef CAS PubMed.
- A. Berndt, H. Klusik and K. Schlüter, J. Organomet. Chem., 1981, 222, c25–c27 Search PubMed.
- R. G. Griffin and H. V. Willigen, J. Chem. Phys., 1972, 57, 86–90 CrossRef CAS.
- C. Elschenbroich, P. Kühlkamp, A. Behrendt and K. Harms, Chem. Ber., 1996, 129, 859–869 CrossRef CAS.
- T. J. DuPont and J. L. Mills, J. Am. Chem. Soc., 1975, 97, 6375–6382 CrossRef CAS.
- T. Kushida and S. Yamaguchi, Organometallics, 2013, 32, 6654–6657 CrossRef CAS.
- H. Braunschweig, V. Dyakonov, J. O. C. Jimenez-Halla, K. Kraft, I. Krummenacher, K. Radacki, A. Sperlich and J. Wahler, Angew. Chem., Int. Ed., 2012, 51, 2977–2980 CrossRef CAS PubMed.
- I. Raabe, K. Wagner, K. Guttsche, M. Wang, M. Grätzel, G. Santiso-Quiñones and I. Krossing, Chem.–Eur. J., 2009, 15, 1966–1976 CrossRef CAS PubMed.
- M. G. Trachioti, A. C. Lazanas and M. I. Prodromidis, Microchim. Acta, 2023, 190, 251 CrossRef CAS PubMed.
- H. Neugebauer, F. Bohle, M. Bursch, A. Hansen and S. Grimme, J. Phys. Chem. A, 2020, 124, 7166–7176 Search PubMed.
- I. Krossing, Chem.–Eur. J., 2001, 7, 490–502 CrossRef CAS.
- (a) CCDC 2504370: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q204z.; (b) CCDC 2504371: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q2050; (c) CCDC 2504372: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q2061; (d) CCDC 2504373: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q2072; (e) CCDC 2504374: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q2083.
| This journal is © The Royal Society of Chemistry 2026 |