Open Access Article
Open Access ArticleElectrochemical control enables high-concentration, one-pot macrolactonization
Siying
Mao†
,
Dengchao
Wei†
,
Kun
Xu
 * and
Chengchu
Zeng
* and
Chengchu
Zeng

College of Chemistry and Life Science, Beijing University of Technology, Beijing 100124, China. E-mail: kunxu@bjut.edu.cn
First published on 17th February 2026
Abstract
State-of-the-art macrolactone synthesis predominantly relies on the end-to-end cyclization of seco-acids, a strategy that requires high-dilution conditions (<5 mM) to suppress oligomerization. To address this limitation, we report an electrochemical macrolactonization protocol that operates at a significantly higher concentration (50 mM). Leveraging an iodide/PPh3 dual-mediation system, this method provides access to 5- to 21-membered lactones and accommodates O-, S-, and N-heteroatom functionalization. Its efficacy originates from the gradual, charge-consumption-dependent formation of an acyloxyphosphonium intermediate, which maintains a relatively low local concentration despite high substrate loading.
Introduction
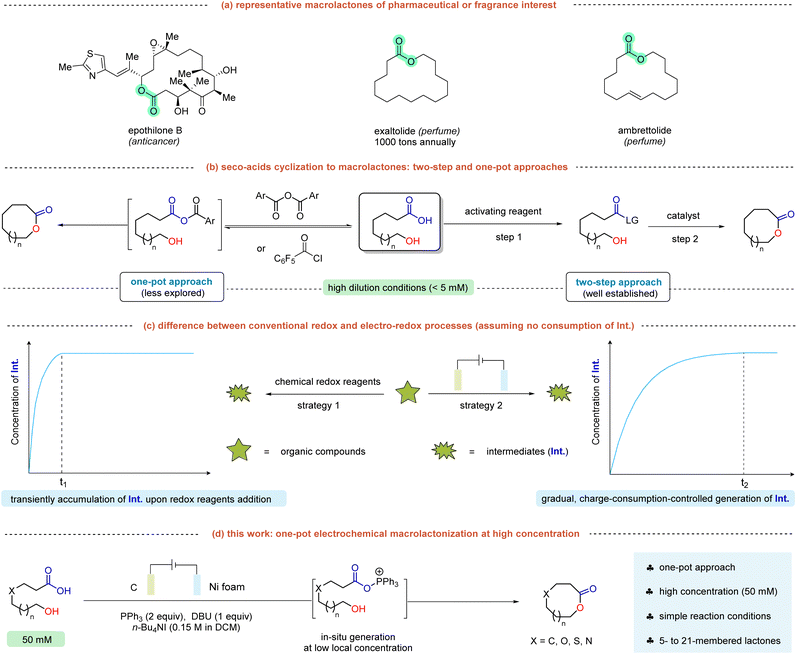
Macrolactones are important structural motifs prevalent in diverse natural products and pharmaceuticals, and they also serve as key ingredients in the flavor and fragrance industries (Scheme 1a).1 For instance, Exaltolide®, a widely used musk, is produced on an industrial scale exceeding 1000 tons annually for perfumery applications. Given their pronounced significance, the development of practical macrolactonization methods has attracted considerable research interest.2 Among various methods, intramolecular cyclization of readily accessible seco-acids remains the predominant route to macrolactones (Scheme 1b). However, due to the entropic cost,3 this end-to-end macrocyclization strategy faces the well-known challenge of competing intermolecular esterification. Currently, a widely recognized method in this field is the Yamaguchi cyclization,4 which proceeds in two steps by first activating the carboxylic acid as a mixed anhydride, followed by base-promoted lactonization (Scheme 1b, right). Inspired by this concept, some other two-step macrolactonization strategies were subsequently developed,5–9 including the Corey–Nicolaou,5 Mukaiyama,6 Trost–Kita,7 and Zhao methods.8 Beyond these two-step protocols, the group of Shiina introduced a single-pot macrolactonization strategy using substituted benzoic anhydrides as activating reagents (Scheme 1b, left).10 However, it necessitates a slow addition of the reagents over 15 hours to maintain the low concentrations of the active intermediate. Addressing this limitation, the pioneering work by the groups of Collins11 and Lebœuf12 leverages Hf(IV) catalysis or non-covalent interactions to enable highly efficient one-pot macrolactonization without the need for slow addition. Despite significant progress, these methods are hindered by the requirement for high-dilution conditions (typically <5 mM) to suppress oligomerization, and often necessitate elevated temperatures to drive the cyclization. These limitations diminish the practicality and scalability of these methods. In fact, oligomerization at high substrate concentrations arises from the rapid formation of activated acyl surrogates upon addition of activating reagents in a single portion. The transient accumulation of these active intermediates at high concentrations promotes undesired intermolecular esterifications. Therefore, a strategy that allows controlled and progressive generation of the activated species could potentially enable macrolactonization at higher substrate loadings.Organic electrochemistry, which utilizes electrons as clean redox reagents, has enriched the toolkit for modern synthesis.13 A key advantage of this approach is the precise control over electron-transfer kinetics by adjusting the current. In this context, electrochemical activation allows the gradual formation of intermediates at low concentrations through controlled charge consumption (Scheme 1c, right).14 This feature is a notable contrast to conventional redox reactions, where concentrated reactive intermediates are transiently produced upon redox reagent addition (Scheme 1c, left). Building on our research interest in synthetic electrochemistry,15 we sought to develop an electrochemical method to enable one-pot, high-concentration macrolactonization of seco-acids. Recently, significant progress has been made in PPh3 mediated electrochemical deoxygenative functionalizations,16 with alkoxy- or acyloxyphosphonium intermediates serving as key species. Inspired by these elegant precedents, we report an electrochemical, high concentration (50 mM), one-pot macrolactonization protocol enabled by iodide/PPh3 (Scheme 1d). Featuring broad substrate scope, this method facilitates the synthesis of 5- to 21-membered lactones and accommodates O-, S-, and N-heteroatom functionalization. Crucially, this method relies on the gradual, charge-consumption-controlled generation of the alkoxyphosphonium intermediate at a low local concentration, thereby preventing undesired oligomerization despite the high bulk substrate loading. Given that the by-product Ph3PO can be reduced to PPh3 by various methods,17 this electrochemical approach represents an attractive alternative for macrolactone synthesis.
Results and discussion
Initially, we chose the lactonization of seco-acid 1a as a model reaction to optimize the reaction conditions (Table 1). To lower the operating potential, iodide was introduced as a redox mediator to activate PPh3.18 When the electrolysis was performed at 50 mM concentration with graphite and Ni foam as the electrodes, 12-membered lactone 2a was obtained in 50% yield (entry 1). At room temperature, the yield dropped to 34%. This indicates that elevated temperature may accelerate both the generation of the alkoxyphosphonium intermediate and the cyclization step. Replacing the graphite anode with Pt decreased the yield to 17% (entry 2). Similarly, replacing the Ni foam cathode with either Pt or graphite gave lower yields of 23% and 16%, respectively (entry 3). Halide electrolyte was critical for this reaction: n-Bu4NBr reduced the yield to 19% (entry 4), whereas only trace product was obtained with n-Bu4NBF4 or n-Bu4NClO4 (entry 5). DBU was found to be the superior base, delivering a higher yield than pyridine, Et3N and KH2PO4 (entry 6). Further optimization of the solvent identified DCM as the optimal choice; acetone, DMF, or MeCN failed to give the desired product (entry 7). Performing the reaction in air afforded a much lower yield of 2a (11%, entry 8). Control experiments confirmed the necessity of both PPh3 and electrolysis, as no desired product was detected in the absence of either component (entries 9 and 10).| Entry | Deviation from standard conditions | Yield (%) |
|---|---|---|
| a Reaction conditions: graphite plate anode (immersed area: 1 × 1 cm2), nickel foam cathode (1 × 1 cm2), 1a (0.2 mmol), PPh3 (0.4 mmol), DBU (0.2 mmol), n-Bu4NI in DCM (0.15 M, 4 mL), undivided cell, 10 mA, Ar, 40 °C, 3 h, isolated yield. b r.t. | ||
| 1 | None | 50(34)b |
| 2 | Pt anode | 17 |
| 3 | Pt or C cathode | 23, 16 |
| 4 | n-Bu4NBr instead of n-Bu4NI | 19 |
| 5 | n-Bu4NBF4 or n-Bu4NClO4 instead of n-Bu4NI | Trace |
| 6 | Pyridine, Et3N or KH2PO4 instead of DBU | 25, 22, 20 |
| 7 | Acetone, DMF and MeCN instead of DCM | 0 |
| 8 | In air | 11 |
| 9 | No PPh3 | 0 |
| 10 | No electricity | 0 |
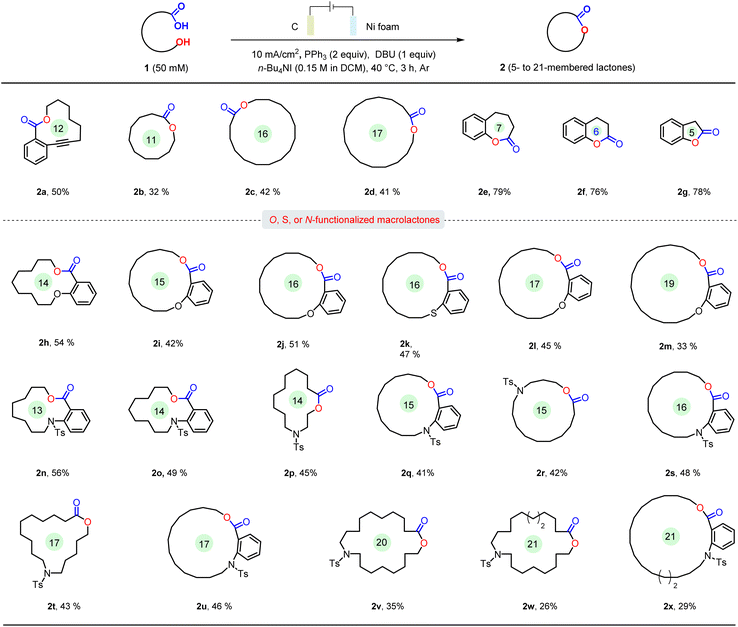
With the optimized reaction conditions established, the generality of this protocol was evaluated. As shown in Scheme 2, this electrochemical method exhibited good compatibility with the synthesis of macro-sized lactones, affording 12- to 17-membered ring products (2a, 2c, 2d) in up to 50% yield. The internal alkyne moiety present in 2a may serve as a versatile handle for late-stage derivatization. In addition to macrolactones, the medium-sized lactone (2b) was also obtained in 32% yield.11,12 While the construction of macrolactones (≥12-membered rings) poses a significant challenge, the formation of normal-sized lactones is typically more facile. Under the optimized conditions, 5- to 8-membered ring products (2e–g) were obtained in up to 79% yield. Extending its utility, this electrochemical approach proved effective for synthesizing O- or S-functionalized macrolactones. A series of such compounds (2h–m) were obtained in synthetically useful yields (up to 54%) across ring sizes of 14 to 19 members. Furthermore, the method was successfully applied to nitrogen-containing hydroxycarboxylic acids, providing access to N-functionalized macrolactones (2n–x) with ring sizes ranging from 14 to 21 members. However, when the ring size was increased to 20 atoms or more, the reaction yield decreased significantly (2v–x).
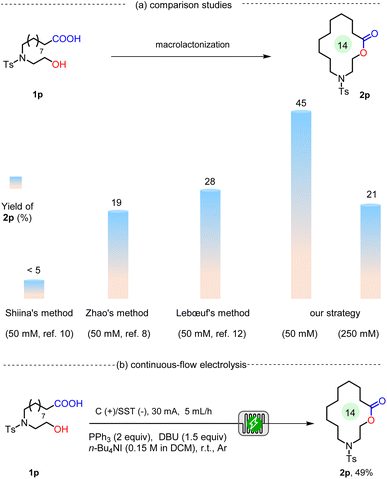
Key features of this electrochemical macrolactonization include a simple one-pot synthesis in an undivided cell under constant-current conditions, without the need for slow addition or high dilution to prevent intermolecular esterification. To evaluate the practical utility of our protocol, a comparison with three standard macrolactonization methods was conducted, employing the synthesis of 2p as a model reaction (Scheme 3a). Under Shiina's conditions, slow addition of 1p to an anhydride solution at 50 mM afforded 2p in <5% yield.10 Zhao's method relies on an ynamide mediator via a two-step acid-catalyzed pathway.8 When 1p was reacted at 50 mM concentration, 2p was obtained in only 19% yield. Lebœuf's approach utilizes a mixed-anhydride strategy for one-pot macrolactonization and demonstrates broad compatibility with various seco-acids at 1.5 mM.12 When the concentration was increased to 50 mM, 2p was obtained in 28% yield. In contrast, our method provided 2p in 45% yield at 50 mM, and 21% yield was achieved even at a concentration of 250 mM (see the SI for additional details).
Batch electrolysis suffers from yield variability due to many influencing factors, making it difficult for other synthetic chemists to reproduce expected results.19 To improve reaction reproducibility, we conducted continuous-flow electrolysis experiments (Scheme 3b).20 Taking the synthesis of N-functionalized 14-membered lactone 2p as the model reaction, the reaction afforded a maximum yield of 49% when a graphite plate and stainless steel (SST) were employed as electrodes with an interelectrode distance of 0.5 mm. More importantly, by assembling four continuous-flow electrolytic cells in parallel, the reaction produced 2p with an average yield of 40% (1.17 g). In contrast, directly scaling up the reaction under batch conditions afforded 2p in only 11% yield (see the SI for additional details).
To investigate the reaction mechanism, a series of mechanistic experiments were conducted (Fig. 1). Initially, cyclic voltammetry (CV) was performed to identify the electrochemically active species. As shown in Fig. 1a, left, an irreversible oxidation peak at 1.39 V (vs. Ag/AgNO3) was observed for PPh3 (curve a). TBAI displayed two pairs of quasi-reversible redox peaks (curve b), with anodic peak potentials at 0.39 V and 0.71 V (vs. Ag/AgNO3), corresponding to the I3−/3I− and 3I2/2I3− redox couples, respectively. However, substrate 1e showed no noticeable oxidation current in the potential range of 0–2.0 V. Upon addition of one or two equivalents of PPh3 to TBAI, a new anodic peak emerged at around 0.48 V, accompanied by a decrease in the current of the second oxidation peak (3I2/2I3−) (curves c and d). These results indicate that TBAI is more readily oxidized than PPh3 and 1e, and the new oxidation peak is attributed to the reaction between PPh3 and I3−, leading to the formation of the iodotriphenylphosphonium cation (I-PPh3+). To further confirm the formation of I-PPh3+, we added substrate 1e to a mixture of TBAI and PPh3 (Fig. 1, right). When 1e, TBAI, and PPh3 were mixed in a 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 ratio, only a slight increase in oxidation current was observed (curve e). However, upon addition of a base to the above mixture, a significant enhancement in oxidation current was detected (curve f). This is mainly because the presence of the base facilitates nucleophilic attack on I-PPh3+ by 1e, thereby releasing I−.
2 ratio, only a slight increase in oxidation current was observed (curve e). However, upon addition of a base to the above mixture, a significant enhancement in oxidation current was detected (curve f). This is mainly because the presence of the base facilitates nucleophilic attack on I-PPh3+ by 1e, thereby releasing I−.
 | ||
| Fig. 1 Mechanistic studies. (a) CV analysis to identify the electrochemically active species. (b) Confirming carboxyl activation other than hydroxyl activation. (c) Mechanistic proposal. | ||
Given that I-PPh3+ can activate both the hydroxyl and carboxyl groups to form alkoxy- or acyloxy-phosphonium intermediates, it is critical to establish which pathway predominates. To this end, high-concentration hexafluoroisopropanol (HFIP) was employed as a weak nucleophile to intercept the intramolecular cyclization (Fig. 1b). Upon addition of HFIP (1 mL), the intermolecular esterification byproduct 3 was isolated in 37% yield, whereas the intramolecular lactonization product 2p was not observed. This result indicates that the reaction of the carboxyl group with I-PPh3+ to generate the acyloxytriphenylphosphonium ion is the predominant pathway.
Based on the results described above and relevant literature,16 a plausible mechanism for the electrochemical macrolactonization is proposed. As illustrated in Fig. 1c, anodic oxidation of iodide in the presence of PPh3 produces the iodotriphenylphosphonium intermediate I and liberates two iodide anions. The latter species undergoes further oxidation at the anode to yield I3−. Meanwhile, I undergoes nucleophilic attack by the carboxylic acid group in the presence of DBU, yielding the acyloxyphosphonium ion III while regenerating iodide. Once intermediate III is formed, it rapidly undergoes intramolecular esterification to give macrolactone product IV, accompanied by the elimination of triphenylphosphine oxide. Concurrently, protons are reduced at the cathode to release hydrogen gas. The generation of I3− is controlled by charge consumption (E-step), so that its subsequent reaction with PPh3 (C-step) and nucleophilic attack by II (C-step) to afford III proceed gradually over the course of the reaction. Accordingly, the reactive alkoxyphosphonium intermediate (III) is generated in a stepwise fashion via an ECC mechanism. This controlled, incremental release maintains a persistently low local intermediate concentration, thereby reducing oligomerization even under high substrate loading conditions.
Conclusions
In conclusion, we have developed a one-pot electrochemical macrolactonization strategy via the synergistic mediation of iodide and PPh3. This operationally simple approach enables access to a diverse range of lactones with ring sizes from 5 to 21 members. Notably, the protocol is also applicable to a variety of macrolactones functionalized with O, S, or N heteroatoms. In contrast to previous methods, this system operates efficiently at elevated substrate concentrations (50 mM) and eliminates the need for slow-addition or azeotropic techniques. Besides batch electrolysis, this method can also be extended to continuous-flow electrolysis to improve reproducibility. The success of this strategy arises from a key design principle: the reactive alkoxyphosphonium intermediate is released gradually under charge-consumption control, thereby maintaining a low local concentration and effectively reducing oligomerization even under high substrate loading.Experimental
Electrolysis was performed in an undivided cell equipped with graphite plate (1.0 × 1.0 cm2) and nickel foam (1.0 × 1.0 cm2) electrodes, using a DC constant current supply. To the cell were added seco-acid (1, 0.2 mmol), PPh3 (0.4 mmol, 2 equiv.), n-Bu4NI (0.6 mmol, 3 equiv.), DBU (0.2 mmol, 1 equiv.), and dry DCM (4 mL). The reaction was conducted in Ar at 40 °C under constant current conditions (10 mA, 3 h). Workup began with the addition of saturated NaCl (15 mL), followed by extraction with DCM (3 × 10 mL). The organic phase was dried with MgSO4, filtered, and concentrated. Purification by silica gel chromatography (ethyl acetate/petroleum ether = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 to 1
20 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4) yielded macrolactone 2.
4) yielded macrolactone 2.
Author contributions
S. M. and D. W.: investigation, data curation; K. X. and C. Z.: conceptualization, writing & editing. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
All data supporting the findings of this study are available within the article and its supplementary information (SI) file. Supplementary information: general information, experimental procedures, characterization data, and spectroscopic data. See DOI: https://doi.org/10.1039/d6sc00702c.Acknowledgements
We are grateful to the National Natural Science Foundation of China (22271009, 22571011 and 22171015). We also extend our thanks to the Large-scale Instruments and Equipment Sharing Platform of Beijing University of Technology for help with NMR experiments.References
- (a) E. M. Driggers, S. P. Hale, J. Lee and N. K. Terrett, The exploration of macrocycles for drug discovery–an underexploited structural class, Nat. Rev. Drug Discovery, 2008, 7, 608–624 CrossRef CAS PubMed; (b) M. D. Delost, D. T. Smith, B. J. Anderson and J. T. Njardarson, From oxiranes to oligomers: architectures of U.S. FDA approved pharmaceuticals containing oxygen heterocycles, J. Med. Chem., 2018, 61, 10996–11020 CrossRef CAS PubMed; (c) G. Ohloff, W. Pickenhagen, P. Kraft and F. Grau, Scent and Chemistry: The Molecular World of Odors, Wiley-VCH, Weinheim, 2nd edn, 2022 Search PubMed.
- For related reviews, see: (a) M. V. Hoof, G. Force and D. Lebœuf, Modern macrolactonization techniques, Synthesis, 2024, 56, 714–732 CrossRef; (b) K. T. Mortensen, T. J. Osberger, T. A. King, H. F. Sore and D. R. Spring, Strategies for the diversity-oriented synthesis of macrocycles, Chem. Rev., 2019, 119, 10288–10317 CrossRef CAS PubMed.
- (a) C. J. White and A. K. Yudin, Contemporary strategies for peptide macrocyclization, Nat. Chem., 2011, 3, 509–524 CrossRef CAS PubMed; (b) F. Gou, D. Shi, B. H. Kou, Z. Li, X. S. Yan, X. Wu and Y. B. Jiang, One-pot cyclization to large peptidomimetic macrocycles by in situ-generated β-turn-enforced folding, J. Am. Chem. Soc., 2023, 145, 9530–9539 CrossRef CAS PubMed.
- J. Inanaga, K. Hirata, H. Saeki, T. Katsuki and M. Yamaguchi, A rapid esterification by means of mixed anhydride and its application to large-ring lactonization, Bull. Chem. Soc. Jpn., 1979, 52, 1989–1993 CrossRef CAS.
- E. J. Corey and K. C. Nicolaou, Efficient and mild lactonization method for the synthesis of macrolides, J. Am. Chem. Soc., 1974, 96, 5614–5616 CrossRef CAS.
- T. Mukaiyama, M. Usui and K. Saigo, The facile synthesis of lactones, Chem. Lett., 1976, 5, 49–50 CrossRef.
- (a) B. M. Trost and J. D. Chisholm, An acid-catalyzed macrolactonization protocol, Org. Lett., 2002, 4, 3743–3745 CrossRef CAS PubMed; (b) Y. Ohba, M. Takatsuji, K. Nakahara, H. Fujioka and Y. Kita, A highly efficient macrolactonization method via ethoxyvinyl ester, Chem.–Eur. J., 2009, 15, 3526–3537 CrossRef CAS PubMed.
- M. Yang, X. W. Wang and J. F. Zhao, Ynamide-mediated macrolactonization, ACS Catal., 2020, 10, 5230–5235 CrossRef CAS.
- (a) E. P. Boden and G. E. Keck, Proton-transfer steps in steglich esterification: a very practical new method for macrolactonization, J. Org. Chem., 1985, 50, 2394–2395 CrossRef CAS; (b) T. Kurihara, Y. Nakajima and O. Mitsunobu, Synthesis of lactones and cycloalkanes. cyclization of ω-hydroxy acids and ethyl α-cyano-ω-hydroxycarboxylates, Tetrahedron Lett., 1976, 17, 2455–2458 CrossRef; (c) F. Kopp, C. F. Stratton, L. B. Akella and D. S. Tan, A diversity-oriented synthesis approach to macrocycles via oxidative ring expansion, Nat. Chem. Biol., 2012, 8, 358–365 CrossRef CAS PubMed; (d) J. L. Zhao, J. R. Shi and Y. Li, Benzyne-mediated esterification reaction, Org. Lett., 2021, 23, 7274–7278 CrossRef CAS PubMed; (e) I. Zalessky, J. M. Wootton, J. K. F. Tam, D. E. Spurling, W. C. Glover-Humphreys, J. R. Donald, W. E. Orukotan, L. C. Duff, B. J. Knapper, A. C. Whitwood, T. F. N. Tanner, A. H. Miah, J. M. Lynam and W. P. Unsworth, A modular strategy for the synthesis of macrocycles and medium-sized rings via cyclization/ring expansion cascade reactions, J. Am. Chem. Soc., 2024, 146, 5702–5711 CrossRef CAS PubMed and references cited therein..
- I. Shiina, M. Kubota and R. Ibuka, A novel and efficient macrolactonization of ω-hydroxycarboxylic acids using 2-methyl-6-nitrobenzoic anhydride (MNBA), Tetrahedron Lett., 2002, 43, 7535–7539 CrossRef CAS.
- M. de Léséleuc and S. K. Collins, Direct macrolactonization of seco acids via hafnium(IV) catalysis, ACS Catal., 2015, 5, 1462–1467 CrossRef.
- G. Force, A. Perfetto, R. J. Mayer, I. Ciofini and D. Lebœuf, Macrolactonization reactions driven by a pentafluorobenzoyl group, Angew. Chem., Int. Ed., 2021, 60, 19843–19851 CrossRef CAS PubMed.
- (a) M. Yan, Y. Kawamata and P. S. Baran, Synthetic organic electrochemical methods since 2000: on the verge of a renaissance, Chem. Rev., 2017, 117, 13230–13319 CrossRef CAS PubMed; (b) A. Wiebe, T. Gieshoff, S. Möhle, E. Rodrigo, M. Zirbes and S. R. Waldvogel, Electrifying organic synthesis, Angew. Chem., Int. Ed., 2018, 57, 5594–5619 CrossRef CAS PubMed; (c) Y. L. Wang, S. Dana, H. Long, Y. Xu, Y. J. Li, N. Kaplaneris and L. Ackermann, Electrochemical late-stage functionalization, Chem. Rev., 2023, 123, 11269–11335 CrossRef CAS PubMed; (d) S. Blank, Z. Nguyen, D. G. Boucher and S. D. Minteer, Electrochemical cascade reactions for electro-organic synthesis, Curr. Opin. Electrochem., 2022, 35, 101049 CrossRef CAS; (e) J. Rein, S. B. Zacate, K. N. Mao and S. Lin, A tutorial on asymmetric electrocatalysis, Chem. Soc. Rev., 2023, 52, 8106–8125 RSC; (f) P. Villo, A. Shatskiy, M. D. Kärkäs and H. Lundberg, Electrosynthetic C–O bond activation in alcohols and alcohol derivatives, Angew. Chem., Int. Ed., 2023, 62, e202211952 CrossRef CAS PubMed; (g) G. Hilt, Recent advances in paired electrolysis and their application in organic electrosynthesis, Curr. Opin. Electrochem., 2024, 43, 101425 CrossRef CAS; (h) Z. Tan, H. Zhang, K. Xu and C. Zeng, Electrochemical radical-polar crossover: a radical approach to polar chemistry, Sci. China: Chem., 2024, 67, 450–470 CrossRef CAS; (i) B. C. Hawkins, J. M. Chalker, M. L. Coote and A. C. Bissember, Electrochemically generated carbocations in organic synthesis, Angew. Chem., Int. Ed., 2024, 63, e202407207 CrossRef CAS PubMed; (j) Z. L. Yang, W. Y. Shi, H. Alhumade, H. Yi and A. W. Lei, Electrochemical oxidative C(sp3)–H cross-coupling with hydrogen evolution, Nat. Synth., 2023, 2, 217–230 CrossRef CAS; (k) C. Ma, P. Fang, D. Liu, K. J. Jiao, P. S. Gao, H. Qiu and T. S. Mei, Transition metal-catalyzed organic reactions in undivided electrochemical cells, Chem. Sci., 2021, 12, 12866–12873 RSC.
- A. G. O'Brien, A. Maruyama, Y. Inokuma, M. Fujita, P. S. Baran and D. G. Blackmond, Radical C-H functionalization of heteroarenes under electrochemical control, Angew. Chem., Int. Ed., 2014, 53, 11868–11871 CrossRef PubMed.
- (a) L. Y. Lan, K. Xu and C. C. Zeng, The merger of electro-reduction and hydrogen bonding activation for a radical smiles rearrangement, Chem. Sci., 2024, 15, 13459–13465 RSC; (b) L. Y. Lan, Y. Gao, C. B. Liu, B. Zhang, K. Xu and C. C. Zeng, In situ sulfur modification enables electrochemical selective deuterated thiol–yne reactions, Angew. Chem., Int. Ed., 2025, 64, e202511623 CrossRef CAS PubMed.
- (a) H. Ohmori, S. Nakai, M. Sekiguchi and M. Masui, Anodic oxidation of organophosphorus compounds. III. anodic alkoxylation and thioalkoxylation of triphenylphosphine, Chem. Pharm. Bull., 1980, 28, 910–915 CrossRef CAS; (b) Z. J. Li, W. X. Sun, X. X. Wang, L. Y. Li, Y. Zhang and C. Li, Electrochemically enabled, nickel-catalyzed dehydroxylative cross-coupling of alcohols with aryl halides, J. Am. Chem. Soc., 2021, 143, 3536–3543 CrossRef CAS PubMed; (c) S. Nagahara, Y. Okada, Y. Kitano and K. Chiba, Biphasic electrochemical peptide synthesis, Chem. Sci., 2021, 12, 12911–12917 RSC; (d) K. H. Sun, Z. M. Xu, V. Ramadoss, L. F. Tian and Y. H. Wang, Electrochemical deoxygenative reduction of ketones, Chem. Commun., 2022, 58, 11155–11158 RSC; (e) Q. P. Guo, Y. Y. Jiang, R. J. Zhu, W. H. Yang and P. F. Hu, Electrochemical azo-free mitsunobu-type reaction, Angew. Chem., Int. Ed., 2024, 63, e202402878 CrossRef CAS PubMed; (f) E. A. Hale and Q. L. Zhu, Electrochemical synthesis of aziridines, pyrrolidines and oxazolines enabled by azo-free alcohol amination, Chem. Sci., 2025, 16, 15216–15222 RSC; (g) X. Y. Ying, Y. Zhang and C. Li, Cobalt-catalyzed photoelectrochemical dehydration of primary alcohols, J. Am. Chem. Soc., 2025, 147, 45315–45323 CrossRef CAS PubMed.
- (a) D. Hérault, D. H. Nguyen, D. Nuel and G. Buono, Reduction of secondary and tertiary phosphine oxides to phosphines, Chem. Soc. Rev., 2015, 44, 2508–2528 RSC; (b) J. S. Elias, C. Costentin and D. G. Nocera, Direct electrochemical P(V) to P(III) reduction of phosphine oxide facilitated by triaryl borates, J. Am. Chem. Soc., 2018, 140, 13711–13718 CrossRef CAS PubMed; (c) S. Manabe, C. M. Wong and C. S. Sevov, Direct and scalable electroreduction of triphenylphosphine oxide to triphenylphosphine, J. Am. Chem. Soc., 2020, 142, 3024–3031 CrossRef CAS PubMed.
- (a) F. Lian, K. Xu and C. C. Zeng, Indirect electrosynthesis with halogen ions as mediators, Chem. Rec., 2021, 21, 2290–2305 CrossRef CAS PubMed; (b) R. Francke, Electrogenerated hypervalent iodine compounds as mediators in organic synthesis, Curr. Opin. Electrochem., 2019, 15, 83–88 CrossRef CAS; (c) X. W. Hu, Z. M. Cao, M. Chen, C. D. Huang, S. F. Ni, S. K. Zhang and Z. X. Ruan, Electrochemical late-stage stitching of tryptophan peptides via N–S bond formation, Angew. Chem., Int. Ed., 2025, 64, e202517101 CrossRef CAS PubMed.
- C. Kingston, M. D. Palkowitz, Y. Takahira, J. C. Vantourout, B. K. Peters, Y. Kawamata and P. S. Baran, A survival guide for the “electro-curious”, Acc. Chem. Res., 2020, 53, 72–83 CrossRef CAS PubMed.
- (a) A. A. Folgueiras-Amador and T. Wirth, Perspectives in flow electrochemistry, J. Flow Chem., 2017, 7, 94–95 CrossRef CAS; (b) T. Noël, Y. Cao and G. Laudadio, The fundamentals behind the use of flow reactors in electrochemistry, Acc. Chem. Res., 2019, 52, 2858–2869 CrossRef PubMed; (c) N. Tanbouza, T. Ollevier and K. Lam, Bridging lab and industry with flow electrochemistry, iScience, 2020, 23, 101720 CrossRef CAS PubMed; (d) X. Sun, F. Meng, K. Xu and C. Zeng, Continuous-flow electrochemistry enables 1,2-arylheteroarylation of alkenes, Org. Lett., 2025, 27, 12784–12789 CrossRef CAS PubMed; (e) T. S. Chen, P. Xiong and H.-C. Xu, Continuous flow electrochemistry unlocks broadly applicable arene C-H amination, Angew. Chem., Int. Ed., 2025, 64, e202513864 CrossRef CAS PubMed.
Footnote |
| † Siying Mao and Dengchao Wei contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |