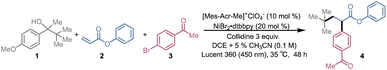Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Photocatalytic multicomponent alkene dicarbofunctionalization via PCET/nickel dual catalysis
Yeersen
Patehebieke
,
Victoria
Jansson
,
Jakob
Öberg
and
Carl-Johan
Wallentin
 *
*
Department of Chemistry and Molecular Biology, University of Gothenburg, Gothenburg SE 41390, Sweden. E-mail: carl.wallentin@chem.gu.se
First published on 19th February 2026
Abstract
Herein we report a synergistic photoredox PCET/nickel dual catalytic strategy that enables three-component alkene dicarbofunctionalization (DCF) using unprotected alcohols as alkyl radical precursors. This transformation merges concerted proton-coupled electron transfer (PCET) activation of alcohols with nickel-catalyzed cross-coupling, achieving sequential Giese addition and C(sp3)–C(sp3), C(sp3)–C(sp2) bond formation under visible light and redox-neutral conditions. The method proceeds directly from secondary and tertiary alcohols, exhibits broad functional-group tolerance. Notably, employing a chiral Ni catalyst enables the first enantioselective variant of this type of PCET mediated transformation. This work establishes a general platform for deconstructive alkene difunctionalization from simple alcohol feedstocks, expanding the synthetic reach of PCET in dual catalytic radical chemistry.
Introduction
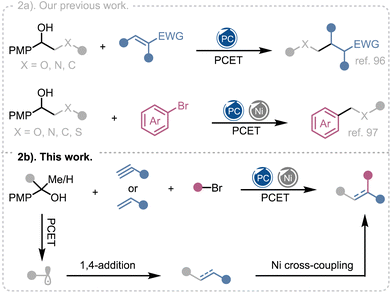
Multicomponent reactions (MCRs) provide a powerful platform for the rapid assembly of molecular complexity from simple precursors in a single operation.1,2 Their inherent convergence, efficiency, and atom economy have made them indispensable tools for the synthesis of pharmaceuticals, agrochemicals, and functional materials.3 By forming multiple bonds in a single step, MCRs minimize purification steps, reduce waste, and enable modular access to structurally diverse scaffolds. These features align MCRs with the principles of sustainable chemistry4 and make them highly attractive for late-stage diversification in medicinal chemistry.5 The challenge, however, lies in controlling selectivity when multiple reactive intermediates are generated simultaneously. Recent advances in catalysis, particularly visible-light photoredox catalysis, have opened new avenues to address this challenge by enabling the controlled generation of reactive radical intermediates under exceptionally mild and tunable conditions.6–8Photoredox catalysis relies on single-electron transfer (SET) processes from photoexcited catalysts to transform simple precursors into radicals with excellent control over reactivity and selectivity.9 As a result, photoredox catalysis has greatly expanded the scope of multicomponent transformations, enabling the direct incorporation of diverse alkyl fragments into complex molecular frameworks. Precursors such as carboxylic acids,10–15 alkyltrifluoroborates,16–28 oxalates,29–32N-(acyloxy)phthalimides (NHPI esters),33–44 alkyl halides,45–53 silicates,54–58 Katritzky pyridinium salts,59–61 aldehydes,62–65 alcohols,66–70 and even unactivated C(sp3)–H bonds66,68,71–75 have been converted into carbon-centered radicals and engaged in sequential bond-forming events with high functional-group tolerance (Fig. 1a).
 | ||
| Fig. 1 (a) Representative alkyl radical precursors in photocatalytic multicomponent reactions. (b) Photocatalytic Ni-catalyzed multicomponent reactions using alcohols as alkyl radical precursors. | ||
These developments have transformed MCRs into versatile tools for step-economic construction of C–C and C–heteroatom bonds and have set the stage for increasingly sophisticated radical cascades. Among the various strategies for radical generation, proton-coupled electron transfer (PCET) has emerged as a particularly powerful approach for activating strong X–H bonds under mild, redox-neutral conditions.76,77 By orchestrating electron transfer and proton transfer in a single concerted event, PCET overcomes the high bond dissociation energies of substrates such as alcohols (BDE ≈ 105 kcal mol−1), enabling direct access to alkoxy or carbon-centered radicals without prefunctionalization.76,78 Pioneering studies by Knowles,79–83 and others84–91 established the foundation for PCET activation of alkanols, enabling ring-opening reactions and Giese-type additions that rapidly build molecular complexity. These breakthroughs demonstrated that alcohols can serve as practical radical precursors, circumventing the need for laborious derivatization steps. More recently, PCET also has been integrated into dual catalytic systems, where the radicals generated from alcohols are intercepted by nickel, unlocking cross-coupling pathways that were previously inaccessible.92–95
Recently, our group developed a PCET based metal-free photocatalytic strategy for the β-scission of secondary and tertiary alcohols, enabling their direct conversion into alkyl radicals under visible light irradiation.96 This protocol allowed the efficient alkylation of diverse electron-deficient alkenes and demonstrated late-stage functionalization of pharmaceutically relevant scaffolds under mild, redox-neutral conditions (Fig. 2a). In subsequent work, we merged concerted PCET-mediated radical generation with nickel cross-coupling to achieved a practical deconstructive arylation of aliphatic alcohols (Fig. 2a).97 Together, these studies established that free alcohols can serve as versatile radical precursors for both Giese-type additions and Ni-catalyzed cross-couplings, thereby laying the foundation for combining these two manifolds in a single multicomponent transformation.
A few photocatalytic three-component reactions that combine radical 1,4-addition with nickel-catalyzed cross-coupling have been reported as powerful methods for vicinal difunctionalization of alkenes under mild conditions.98,99 In these processes carbon-centered radicals are derived from pre-activated species like NHPI esters,37–39,42,43 oxalates,29–31 or trifluoroborates16,18–20,24–28 which require multistep synthesis and limit late-stage applicability. Direct use of free alcohol in such three-component transformations remains rare (Fig. 1b).
In 2021, Molander and Gutierrez presented photoredox/nickel dual catalytic utilization of alcohols (among other feedstocks like ethers and amides) by performing photocatalyzed hydrogen atom transfer (photo-HAT) to activate aliphatic C–H bonds giving alkyl radicals. The subsequent steps involve Giese addition of the alkyl radicals and nickel-catalyzed coupling.66 Later in 2024, Nevado realized a similar alkene dicarbofunctionalization via an enantioselective photo-HAT/DCF process.68 In the same year, the MacMillan group utilized benzoxazolium activated primary alcohols as alkyl radical precursors in photoredox/nickel dual catalytic DCF of alkenes. The reaction proceeds through a triple radical sorting mechanism that merges photoredox and nickel-catalyzed bimolecular homolytic substitution (SH2) catalysis to achieve alkene dialkylation.69
There has not been any report that utilize PCET in photoredox/nickel dual catalytic DCF of alkenes. Combining PCET-mediated radical generation from unactivated alcohols would allow for the direct use of secondary and tertiary alcohols in DCF type transformations. Consequently, a PCET based approach would deliver a more general and modular three-component strategy towards molecular construction.
Herein, we report a synergistic photoredox PCET/nickel dual catalytic system that accomplishes the three-component dicarbofunctionalization of electron-deficient alkenes using unprotected alcohol as the radical source (Fig. 2b). The method features broad substrate scope, including challenging secondary and tertiary alcohols, and demonstrates excellent functional-group tolerance. We also demonstrated the first enantioselective variant of a PCET initiated DCF process.
Results and discussion
To evaluate whether our previously developed PCET-mediated alkyl radical generation from alcohols is compatible with three-component alkene dicarbofunctionalization, we selected tertiary alcohol 1, phenyl acrylate 2, and 4-bromoacetophenone 3 as model substrates. The reaction conditions were adapted from our previously reported nickel-catalyzed deconstructive cross-coupling methods.97 Using the Fukuzumi catalyst [Mes–Acr–Me]+ClO4− as the photocatalyst, NiBr2·dtbbpy (4,4′-di-tert-butyl-2,2′-dipyridyl) as cross-coupling catalyst, 2,4,6-collidine as the base, in a solvent mixture of 1,2-dichloroethane (DCE) + 5% MeCN at 35 °C under 450 nm blue light irradiation for 48 h. To our delight, the desired dicarbofunctionalized product 4 was obtained in a very good yield of 82% (Table 1, entry 1).| Entry | Deviation from standard conditions | Yieldb |
|---|---|---|
| a Reaction conditions: alcohol (0.3 mmol), alkene (0.2 mmol), aryl bromide (0.1 mmol), 2,4,6-collidine (0.3 mmol), [Mes–Acr–Me]+ClO4− (001 mmol), NiBr2·dtbbpy (0.02 mmol), DCE + 5% MeCN (3 mL), time (48 h), Lucent 360 photo reactor (450 nm, 50% light intensity), temperature (35 °C). b Yields were determined by 1H NMR analysis of the crude reaction mixtures using ethylene carbonate as an internal standard. | ||
| 1 | No deviation | 82% |
| 2 | DCE | 73% |
| 3 | MeCN | n.d. |
| 4 | DCM | 57% |
| 5 | DMF | Trace |
| 6 | [Mes–Acr–Me]+BF4− | 81% |
| 7 | 6 h | 73% |
| 8 | 12 h | 81% |
| 9 | 12 h, 100% intensity | 74% |
| 10 | 6 h, 75% intensity | 74% |
| 11 | 12 h, 75% intensity | 84% |
| 12 | 12 h, no photocatalyst | n.d. |
| 13 | 12 h, no nickel catalyst | n.d. |
| 14 | 12 h, no base | n.d. |
| 15 | 12 h, no light | n.d. |
Encouraged by this result, we performed additional screening to refine the conditions. Using DCE alone reduced the yield to 73% (entry 2), while MeCN as the sole solvent failed to produce the desired product (entry 3). Interestingly, emplying MeCN as a co-solvent improved the yield, possibly due to an optimal balance of polarity and Brønsted basicity that promotes both precursor complex formation and disaggregation of the successor complex, thereby mitigating non-productive back electron transfer.100 Other solvents gave significantly lower or trace yields (entries 4–5). Altering the reaction stoichiometry negatively impacted the outcome (see SI for details). Changing the photocatalyst counterion from ClO4− to BF4− had little effect (entry 6), and screening other acridinium photocatalysts resulted in decreased yields (see SI for details).
Shortening the reaction time to 6 h slightly reduced the yield to 73% (entry 7), with 1H NMR indicating residual starting alcohol. At 12 h, the alcohol was nearly consumed, and the yield improved to 81% (entry 8). Increasing light intensity to 100% accelerated alcohol consumption within 12 h but lowered the product yield to 74% (entry 9), likely due to increased side-product formation and photocatalyst degradation. Reducing light intensity to 75% and maintaining a 12 h reaction time afforded the highest yield of 84% (entry 11). Finally, control experiments confirmed the essential role of all components—photocatalyst, nickel catalyst, base, and light—as omission of any resulted in no product formation (entries 12–15).
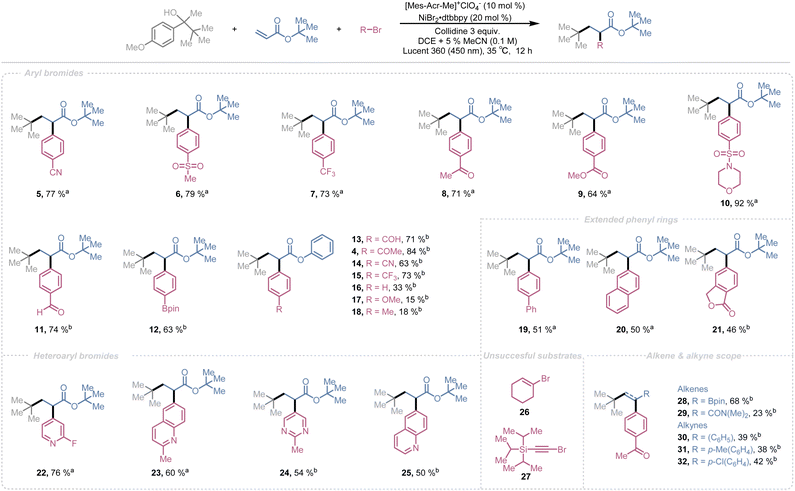
With the optimal reaction conditions established, we next explored the substrate scope of this transformation. We began by examining the aryl bromide scope. A wide range of aryl bromides bearing diverse functional groups were well tolerated under optimized conditions (Scheme 1). Substrates containing nitrile 5, sulfone 6 & 10, trifluoromethyl 7, ketone 8, ester 9, aldehyde 11, and boronic ester groups 12 afforded the desired products in good to excellent yields (92–63%). Notably, aryl bromides bearing aldehyde 11 or Bpin 12 groups posed purification challenges, as the target products co-eluted with the ketone byproduct and trace amounts of unreacted alcohol.
To address this, we replaced tert-butyl acrylate with phenyl acrylate, anticipating improved separation. Phenyl acrylate delivered good yields (84–63%) with electron-withdrawing substituents on the aryl halide 4, 13–15, however, electron-neutral 16 and electron-donating substituents 17 & 18 significantly diminished the yields. This limitation is consistent with known trends in photoredox/nickel cross-coupling reactions, where efficient oxidative addition to low-valent nickel species requires a polarized C–X bond and an electrophilic aryl carbon. Electron-donating groups reduce electrophilicity, slowing oxidative addition and increasing decomposition pathways.101 Unfortunately, switching to phenyl acrylate did not resolve the purification issue, so we reverted to tert-butyl acrylate as the alkene partner.
Extended aromatic systems such as biphenyl 19, naphthalene 20, and phthalide derivatives 21 provided decent yields (51–46%). Heteroaryl bromides, including pyridine 22, quinoline 23 & 25, and pyrimidine derivatives 24, also furnished good to moderate yields (76–50%). Attempts to employ alkyl bromides 26 or alkynyl bromides 27 as coupling partners were unsuccessful, with reactions failing under the optimized conditions.
After achieving satisfactory results with aryl halides, we investigated the scope of alkenes and alkynes. Electron-deficient olefins such as vinylboronic acid pinacol ester 28 and N,N-dimethylacrylamide (DMAA) 29 exhibited reactivity comparable to acrylates, delivering good to synthetically useful yields. Ethynylbenzene derivatives bearing electron-neutral 30, electron-donating 31, and electron-withdrawing substituents 32 also afforded reasonable yields.
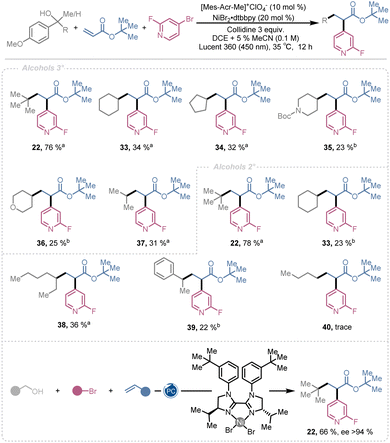
Finally, we examined the alcohol scope for alkyl radical generation (Scheme 2). Tertiary alcohols bearing alkyl, cycloalkyl, and heterocyclic groups, including tert-butyl 22, cyclohexyl 33, cyclopentyl 34, piperidine 35, tetrahydropyran 36, and isopropyl 37, provided dicarbofunctionalized products in yields ranging from 76% to 23%. Secondary alcohols with tertiary 22, secondary 33 & 38, benzylic 39, and primary 40 substituents exhibited similar trends, affording products in 78% to trace yield. The observed reactivity correlates with radical stability, following the order 3° > 2° > benzylic > 1°. More stable and sterically hindered radicals favor Giese-type addition to the olefin, after which the resulting radical engages the nickel catalytic cycle to furnish the difunctionalized product.
In contrast, less stable and less hindered radicals are more prone to nickel-mediated cross-coupling, competing with Giese addition and thereby reducing overall yields. These trends are clearly reflected in quantitative 1H NMR analysis of the crude reaction mixtures of 4, 33, and 40 (see SI). For the tertiary radical involved in the synthesis of 4, no cross-coupling product is detected; instead, the Giese-type addition product is formed in a 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 ratio relative to the targeted difunctionalized product 4. In contrast, secondary radicals predominantly yield the cross-coupled product, while the Giese-type and desired difunctionalized products are formed in comparable amounts (1.15
1.0 ratio relative to the targeted difunctionalized product 4. In contrast, secondary radicals predominantly yield the cross-coupled product, while the Giese-type and desired difunctionalized products are formed in comparable amounts (1.15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.00 for 33). The least stable and least sterically demanding primary radical likewise furnishes the cross-coupled product as the major component, with only trace amounts of the difunctionalized product observed, as exemplified by 40.
1.00 for 33). The least stable and least sterically demanding primary radical likewise furnishes the cross-coupled product as the major component, with only trace amounts of the difunctionalized product observed, as exemplified by 40.
Encouraged by the work of Chu and Gutierrez18 and other groups24,47,50,68,102 we wanted to explore the potential to install the aromatic moiety in an enantioenriched fashion. Consequently, we engaged the synthesis of 22 utilizing a chiral Ni catalyst employing the standard conditions. To our delight 22 was generated in 66% yield with an ee of >94% (Scheme 2). This illustrates that this dicarbofunctuionlization strategy indeed is compatible with enantioselective multicomponent based diversification of electron deficient alkenes. Studies focused on further optimization of the chiral ligand and reaction parameters, as well as expansion of the enantioselective scope are ongoing in our lab.
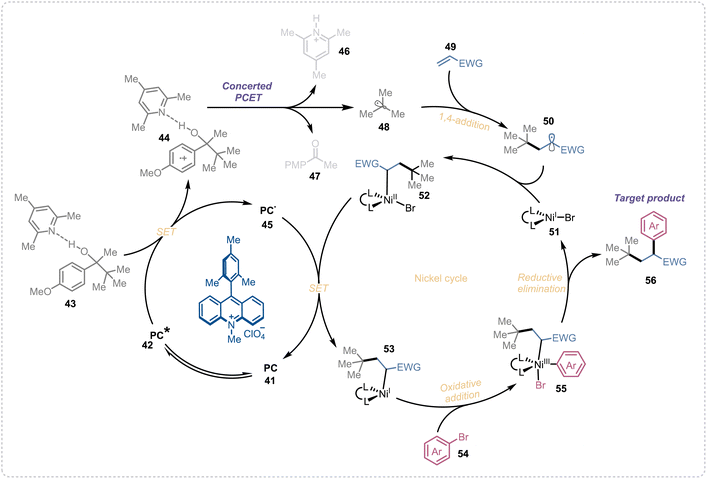
In our previous investigation of the reaction mechanism, we have concluded the concerted PCET is the main mechanism of alkyl radical generation from these aliphatic alcohols. So based on our previous mechanistic studies96,97 and the reported literature on photoredox/nickel dual catalytic cross-coupling reactions,103–105 we propose the following plausible mechanism for the alkene dicarbofunctionalization reaction (Fig. 3). Upon visible-light irradiation, the photocatalyst (41) in its excited state (42) oxidizes the base complex substrate (43) to its corresponding oxidized form (44). This oxidized species then undergoes a concerted proton-coupled electron transfer (PCET), generating the key alkyl radical (48) along with a protonated base (46) and a ketone byproduct (47). The alkyl radical subsequently adds to an electron-deficient olefin (49), forming a new radical intermediate (50). This intermediate then combines with a Ni(I)–Br complex (51) to yield a Ni(II)–alkyl–bromo species (52). Next, a single-electron transfer (SET) from the reduced photocatalyst (45) to the nickel complex (52) regenerates the ground-state photocatalyst and produces a Ni(I)–alkyl species (53). Oxidative addition of the aryl halide (54) to this Ni(I) complex forms a Ni(III)–alkyl–aryl–bromo intermediate (55), which undergoes reductive elimination to deliver the desired difunctionalized product (56) and a Ni(I)–bromo species (51), thereby completing the nickel catalytic cycle.
Conclusions
In conclusion, we have developed an efficient PCET mediated photoredox/nickel dual catalytic strategy that achieves three-component alkene dicarbofunctionalization directly from unprotected alcohols. This redox-neutral protocol merges concerted PCET-mediated alkyl radical generation with nickel-catalyzed cross-coupling, enabling the direct use of secondary and tertiary alcohols as alkyl radical precursors under visible light. The method proceeds under mild conditions, displays broad functional-group tolerance, and provides access to diverse vicinal aryl–alkyl disubstituted products. The observed reactivity trend across different alcohol classes correlates with radical stability and underlines the mechanistic synergy between photoredox and nickel catalysis. Product distribution analysis further reveals a radical stability dependent competition between conjugate addition and direct nickel capture, rationalizing diminished efficiency for less stabilized radicals. Moreover, the successful realization of an enantioselective example highlights the potential of this approach for asymmetric multicomponent synthesis. Ongoing efforts are directed toward developing broadly applicable enantioselective variants of this transformation through catalyst and ligand design. This work establishes a general and practical platform for deconstructive difunctionalization of alkenes from readily available alcohols and paves the way toward sustainable, atom-economical radical transformations driven by PCET.Author contributions
Y. P.: conceptualization (lead); methodology; investigation; validation; formal analysis; data curation; writing – original draft. V. J.: investigation; validation; formal analysis; data curation. J. Ö.: investigation; validation; formal analysis; data curation. C.-J. W.: conceptualization (lead); supervision; project administration; funding acquisition; writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): all experimental procedures, characterization data, and copies of NMR spectra for new compounds. See DOI: https://doi.org/10.1039/d6sc00382f.Acknowledgements
Financial support from the Knut and Alice Wallenberg Foundation (Grant 2019.0071) and The Swedish Research Council are gratefully acknowledged.Notes and references
- J. Zhu, Q. Wang and M. Wang, Multicomponent reactions in organic synthesis, John Wiley & Sons, 2015 Search PubMed.
- R. P. Herrera and E. Marqués-López, Multicomponent reactions: concepts and applications for design and synthesis, John Wiley & Sons, 2015 Search PubMed.
- A. Dömling, W. Wang and K. Wang, Chemistry and Biology Of Multicomponent Reactions, Chem. Rev., 2012, 112, 3083–3135 CrossRef PubMed.
- B. Martinengo, E. Diamanti, E. Uliassi and M. L. Bolognesi, Harnessing the 12 Green Chemistry Principles for Sustainable Antiparasitic Drugs: Toward the One Health Approach, ACS Infect. Dis., 2024, 10, 1856–1870 CrossRef CAS PubMed.
- E. Ruijter and R. V. A. Orru, Multicomponent reactions – opportunities for the pharmaceutical industry, Drug Discov. Today Technol., 2013, 10, e15–e20 Search PubMed.
- L. Marzo, S. K. Pagire, O. Reiser and B. König, Visible-Light Photocatalysis: Does It Make a Difference in Organic Synthesis?, Angew. Chem., Int. Ed., 2018, 57, 10034–10072 Search PubMed.
- J. Twilton, C. Le, P. Zhang, M. H. Shaw, R. W. Evans and D. W. C. MacMillan, The merger of transition metal and photocatalysis, Nat. Rev. Chem., 2017, 1, 0052 Search PubMed.
- C. K. Prier, D. A. Rankic and D. W. C. MacMillan, Visible Light Photoredox Catalysis with Transition Metal Complexes: Applications in Organic Synthesis, Chem. Rev., 2013, 113, 5322–5363 Search PubMed.
- J. M. R. Narayanam and C. R. J. Stephenson, Visible light photoredox catalysis: applications in organic synthesis, Chem. Soc. Rev., 2011, 40, 102–113 RSC.
- C. Zhu, H. Yue, B. Maity, I. Atodiresei, L. Cavallo and M. Rueping, A multicomponent synthesis of stereodefined olefins via nickel catalysis and single electron/triplet energy transfer, Nat. Catal., 2019, 2, 678–687 CrossRef CAS.
- R. S. Mega, V. K. Duong, A. Noble and V. K. Aggarwal, Decarboxylative Conjunctive Cross-coupling of Vinyl Boronic Esters using Metallaphotoredox Catalysis, Angew. Chem., Int. Ed., 2020, 59, 4375–4379 CrossRef CAS PubMed.
- H. Jiang, X. Yu, C. G. Daniliuc and A. Studer, Three-Component Aminoarylation of Electron-Rich Alkenes by Merging Photoredox with Nickel Catalysis, Angew. Chem., Int. Ed., 2021, 60, 14399–14404 CrossRef CAS PubMed.
- V. T. Nguyen, G. C. Haug, V. D. Nguyen, N. T. H. Vuong, G. B. Karki, H. D. Arman and O. V. Larionov, Functional group divergence and the structural basis of acridine photocatalysis revealed by direct decarboxysulfonylation, Chem. Sci., 2022, 13, 4170–4179 RSC.
- N. J. Venditto and J. A. Boerth, Photoredox-Catalyzed Multicomponent Synthesis of Functionalized γ-Amino Butyric Acids via Reductive Radical Polar Crossover, Org. Lett., 2023, 25, 3429–3434 CrossRef CAS PubMed.
- F. Pasca, Y. Gelato, M. Andresini, D. Serbetci, P. Natho, G. Romanazzi, L. Degennaro, M. Colella and R. Luisi, Continuous Flow Decarboxylative Monofluoroalkylation Enabled by Photoredox Catalysis, JACS Au, 2025, 5, 684–692 Search PubMed.
- M. W. Campbell, J. S. Compton, C. B. Kelly and G. A. Molander, Three-Component Olefin Dicarbofunctionalization Enabled by Nickel/Photoredox Dual Catalysis, J. Am. Chem. Soc., 2019, 141, 20069–20078 Search PubMed.
- J. Yi, S. O. Badir, R. Alam and G. A. Molander, Photoredox-Catalyzed Multicomponent Petasis Reaction with Alkyltrifluoroborates, Org. Lett., 2019, 21, 4853–4858 CrossRef CAS PubMed.
- L. Guo, M. Yuan, Y. Zhang, F. Wang, S. Zhu, O. Gutierrez and L. Chu, General Method for Enantioselective Three-Component Carboarylation of Alkenes Enabled by Visible-Light Dual Photoredox/Nickel Catalysis, J. Am. Chem. Soc., 2020, 142, 20390–20399 CrossRef CAS PubMed.
- W. C. Huang, S. Keess and G. A. Molander, Dicarbofunctionalization of 1.1.1 Propellane Enabled by Nickel/Photoredox Dual Catalysis: One-Step Multicomponent Strategy for the Synthesis of BCP-Aryl Derivatives, J. Am. Chem. Soc., 2022, 144, 12961–12969 CrossRef CAS PubMed.
- W. C. Huang, S. Keess and G. A. Molander, One step synthesis of unsymmetrical 1,3-disubstituted BCP ketones via nickel/photoredox-catalyzed 1.1.1 propellane multicomponent dicarbofunctionalization, Chem. Sci., 2022, 13, 11936–11942 Search PubMed.
- S. Li, P. W. Davies and W. Shu, Modular synthesis of α-arylated carboxylic acids, esters and amides via photocatalyzed triple C–F bond cleavage of methyltrifluorides, Chem. Sci., 2022, 13, 6636–6641 RSC.
- C. Zhang, Z. M. Tang, Y. J. Qiu, J. Tang, S. Q. Ye, Z. M. Li and J. Wu, Access to axially chiral styrenes via a photoinduced asymmetric radical reaction involving a sulfur dioxide insertion, Chem Catal., 2022, 2, 164–177 CAS.
- Q. Li, H. H. Sun, F. Y. Yan, Y. Y. Zhao, Y. C. Zhang, C. Zhou, M. Y. Han, H. J. Li and X. W. Sui, Modular construction of N-arylated amino acid esters enabled by a photoredox-catalyzed multicomponent reaction, Green Chem., 2023, 25, 6226–6230 RSC.
- X. Li, M. Yuan, F. Chen, Z. Huang, F.-L. Qing, O. Gutierrez and L. Chu, Three-component enantioselective alkenylation of organophosphonates via nickel metallaphotoredox catalysis, Chem, 2023, 9, 154–169 Search PubMed.
- Z.-K. Wang, Y.-P. Wang, Z.-W. Rao, C.-Y. Liu, X.-H. Pan and L. Guo, General Method for Selective Three-Component Carboacylation of Alkenes via Visible-Light Dual Photoredox/Nickel Catalysis, Org. Lett., 2023, 25, 1673–1677 Search PubMed.
- Q. Zeng, F. Y. Gao, J. Benet-Buchholz and A. W. Kleij, Stereoselective Three-Component Allylic Alkylation Enabled by Dual Photoredox/Ni Catalysis, ACS Catal., 2023, 13, 7514–7522 CrossRef CAS.
- X. He, E. X. Shi and J. H. Xiao, Enantioselective Synthesis of Benzylic Boronic Esters via Photoredox/Nickel-Catalyzed Multicomponent Cross-Couplings, Org. Lett., 2024, 26, 9728–9734 Search PubMed.
- L. J. Niu, S. Z. Jin, M. Q. Zhu, L. F. Wang and D. Y. Wang, Three-Component 1,2-Carboalkynylation of Alkenes Via Cooperative Nickel/Photoredox Catalysis, Org. Lett., 2025, 27, 3183–3187 Search PubMed.
- L. Guo, F. Song, S. Zhu, H. Li and L. Chu, syn-Selective alkylarylation of terminal alkynes via the combination of photoredox and nickel catalysis, Nat. Commun., 2018, 9, 4543 CrossRef PubMed.
- L. Guo, H.-Y. Tu, S. Zhu and L. Chu, Selective, Intermolecular Alkylarylation of Alkenes via Photoredox/Nickel Dual Catalysis, Org. Lett., 2019, 21, 4771–4776 CrossRef CAS PubMed.
- H. Li, L. Guo, X. Feng, L. Huo, S. Zhu and L. Chu, Sequential C–O decarboxylative vinylation/C–H arylation of cyclic oxalates via a nickel-catalyzed multicomponent radical cascade, Chem. Sci., 2020, 11, 4904–4910 RSC.
- W.-H. Sun, J.-Y. Zou, X.-J. Xu, J.-L. Wang, M.-L. Liu and X.-Y. Liu, Photo-Catalyzed Redox-Neutral 1,2-Dialkylation of Alkenes, Adv. Synth. Catal., 2022, 364, 2260–2265 CrossRef CAS.
- A. Tlahuext-Aca, R. A. Garza-Sanchez and F. Glorius, Multicomponent Oxyalkylation of Styrenes Enabled by Hydrogen-Bond-Assisted Photoinduced Electron Transfer, Angew. Chem., Int. Ed., 2017, 56, 3708–3711 Search PubMed.
- Y. Shen, M.-L. Shen and P.-S. Wang, Light-Mediated Chiral Phosphate Catalysis for Asymmetric Dicarbofunctionalization of Enamides, ACS Catal., 2020, 10, 8247–8253 CrossRef CAS.
- M. J. Cabrera-Afonso, A. Sookezian, S. O. Badir, M. El Khatib and G. A. Molander, Photoinduced 1,2-dicarbofunctionalization of alkenes with organotrifluoroborate nucleophiles via radical/polar crossover, Chem. Sci., 2021, 12, 9189–9195 RSC.
- W. Dong, E. Yen-Pon, L. Li, A. Bhattacharjee, A. Jolit and G. A. Molander, Exploiting the sp2 character of bicyclo[1.1.1]pentyl radicals in the transition-metal-free multi-component difunctionalization of [1.1.1]propellane, Nat. Chem., 2022, 14, 1068–1077 CrossRef CAS PubMed.
- S. Z. Duan, Y. J. Zi, L. L. Wang, J. L. Cong, W. Chen, M. Y. Li, H. B. Zhang, X. D. Yang and P. J. Walsh, α-Branched amines through radical coupling with 2-azaallyl anions, redox active esters and alkenes, Chem. Sci., 2022, 13, 3740–3747 RSC.
- Y. Li, J. Yang, X. Geng, P. Tao, Y. Shen, Z. Su and K. Zheng, Modular Construction of Unnatural α-Tertiary Amino Acid Derivatives by Multicomponent Radical Cross-Couplings, Angew. Chem., Int. Ed., 2022, 61, e202210755 CrossRef CAS PubMed.
- X. Xi, Y. Chen and W. Yuan, Nickel-Catalyzed Three-Component Alkylacylation of Alkenes Enabled by a Photoactive Electron Donor–Acceptor Complex, Org. Lett., 2022, 24, 3938–3943 CrossRef CAS PubMed.
- W. Z. Dong, S. Keess and G. A. Molander, Nickel-mediated alkyl-, acyl-, and sulfonylcyanation of 1.1.1 propellane, Chem Catal., 2023, 3, 100608 CAS.
- J. Xu, P. F. Zhang and W. M. Li, Synthesis of BCP nitriles enabled by a metallaphotoredox-based multi-component reaction, Chem Catal., 2023, 3, 100618 CAS.
- J. Z. Wang, E. Mao, J. A. Nguyen, W. L. Lyon and D. W. C. MacMillan, Triple Radical Sorting: Aryl-Alkylation of Alkenes, J. Am. Chem. Soc., 2024, 146, 15693–15700 CrossRef CAS PubMed.
- T. Zhou, Z.-W. Zhang, J. Nie, F. Y. Kwong, J.-A. Ma and C. W. Cheung, Metallaphotocatalytic triple couplings for modular synthesis of elaborate N-trifluoroalkyl anilines, Nat. Commun., 2024, 15, 9926 CrossRef CAS PubMed.
- P. Cao, W. Xiong and T. Yang, Modular Access to Chiral Benzylamines via Ni/Photoredox-Catalyzed Multicomponent Cross-Electrophile Coupling, Org. Lett., 2025, 27, 1475–1480 CrossRef CAS PubMed.
- H.-M. Huang, P. Bellotti, P. M. Pflüger, J. L. Schwarz, B. Heidrich and F. Glorius, Three-Component, Interrupted Radical Heck/Allylic Substitution Cascade Involving Unactivated Alkyl Bromides, J. Am. Chem. Soc., 2020, 142, 10173–10183 CrossRef CAS PubMed.
- S.-Z. Sun, Y. Duan, R. S. Mega, R. J. Somerville and R. Martin, Site-Selective 1,2-Dicarbofunctionalization of Vinyl Boronates through Dual Catalysis, Angew. Chem., Int. Ed., 2020, 59, 4370–4374 CrossRef CAS PubMed.
- P. C. Qian, H. X. Guan, Y. E. Wang, Q. Q. Lu, F. Zhang, D. Xiong, P. J. Walsh and J. Y. Mao, Catalytic enantioselective reductive domino alkyl arylation of acrylates via nickel/photoredox catalysis, Nat. Commun., 2021, 12, 6613 CrossRef CAS PubMed.
- Y.-F. Han, Y. Huang, H. Liu, Z.-H. Gao, C.-L. Zhang and S. Ye, Photoredox cooperative N-heterocyclic carbene/palladium-catalysed alkylacylation of alkenes, Nat. Commun., 2022, 13, 5754 CrossRef CAS PubMed.
- C. Zhu, H. Yue and M. Rueping, Nickel catalyzed multicomponent stereodivergent synthesis of olefins enabled by electrochemistry, photocatalysis and photo-electrochemistry, Nat. Commun., 2022, 13, 3240 CrossRef CAS PubMed.
- M. S. Liu and W. Shu, Rapid Synthesis of fl-Chiral Sulfones by Ni-Organophotocatalyzed Enantioselective Sulfonylalkenylation of Alkenes, JACS Au, 2023, 3, 1321–1327 CrossRef CAS PubMed.
- M. A. Smith, R. J. D. Kang, R. Kumar, B. Roy and M. J. Gaunt, Modular synthesis of α-branched secondary alkylamines via visible-light-mediated carbonyl alkylative amination, Chem. Sci., 2024, 15, 14888–14898 RSC.
- J. Zhu, Y. Hong, Y. Wang, Y. Guo, Y. Zhang, Z. Ni, W. Li and J. Xu, Synthesis of 1-(Halo)alkyl-3-heteroaryl Bicyclo[1.1.1]pentanes Enabled by a Photocatalytic Minisci-type Multicomponent Reaction, ACS Catal., 2024, 14, 6247–6258 CrossRef CAS.
- A. Gimnkhan, R. Kancherla, K. Muralirajan and M. Rueping, Selective Alkylarylation Difunctionalization of 1,3-Butadienes via Nickel/Photoredox Dual Catalysis, Org. Lett., 2025, 27, 5509–5514 CrossRef CAS PubMed.
- A. García-Domínguez, R. Mondal and C. Nevado, Dual Photoredox/Nickel-Catalyzed Three-Component Carbofunctionalization of Alkenes, Angew. Chem., Int. Ed., 2019, 58, 12286–12290 Search PubMed.
- S. Zheng, Z. Chen, Y. Hu, X. Xi, Z. Liao, W. Li and W. Yuan, Selective 1,2-Aryl-Aminoalkylation of Alkenes Enabled by Metallaphotoredox Catalysis, Angew. Chem., Int. Ed., 2020, 59, 17910–17916 Search PubMed.
- X. Yu, C. G. Daniliuc, F. A. Alasmary and A. Studer, Direct Access to α-Aminosilanes Enabled by Visible-Light-Mediated Multicomponent Radical Cross-Coupling, Angew. Chem., Int. Ed., 2021, 60, 23335–23341 Search PubMed.
- F. Ye, S. L. Zheng, Y. X. Luo, X. T. Qi and W. M. Yuan, Ligand-Controlled Regioreversed 1,2-Aryl-Aminoalkylation of Alkenes Enabled by Photoredox/Nickel Catalysis, ACS Catal., 2024, 14, 8505–8517 Search PubMed.
- W. H. Xiong, H. Y. Xiao, G. Y. Sun, P. T. Cao, Y. Chen, W. R. Yang, M. J. Koh and T. Yang, Enantioselective multicomponent 1,2-arylaminoalkylation of acrylates by photoredox/nickel dual catalysis, Cell Rep. Phys. Sci., 2025, 6(2), 102435 Search PubMed.
- F. J. R. Klauck, H. Yoon, M. J. James, M. Lautens and F. Glorius, Visible-Light-Mediated Deaminative Three-Component Dicarbofunctionalization of Styrenes with Benzylic Radicals, ACS Catal., 2019, 9, 236–241 Search PubMed.
- S. Nandi, P. Das, S. Das, S. Mondal and R. Jana, Visible-light-mediated β-acylative divergent alkene difunctionalization with Katritzky salt/CO2, Green Chem., 2023, 25, 3633–3643 Search PubMed.
- S. Das, S. A. Rahaman, K. Pradhan and R. Jana, Organophotoredox-Catalyzed Synthesis of Unnatural α/β Amino Acids and Peptides via Deaminative Three-Component Coupling, Org. Lett., 2024, 26, 6955–6960 CrossRef CAS PubMed.
- A. Trowbridge, D. Reich and M. J. Gaunt, Multicomponent synthesis of tertiary alkylamines by photocatalytic olefin-hydroaminoalkylation, Nature, 2018, 561, 522–527 CrossRef CAS.
- H.-M. Huang, P. Bellotti, S. Kim, X. Zhang and F. Glorius, Catalytic multicomponent reaction involving a ketyl-type radical, Nat. Synth., 2022, 1, 464–474 Search PubMed.
- D. Wang and L. Ackermann, Three-component carboacylation of alkenes via cooperative nickelaphotoredox catalysis, Chem. Sci., 2022, 13, 7256–7263 Search PubMed.
- T. Yang, W. H. Xiong, G. Y. Sun, W. R. Yang, M. D. Lu and M. J. Koh, Multicomponent Construction of Tertiary Alkylamines by Photoredox/Nickel-Catalyzed Aminoalkylation of Organohalides, J. Am. Chem. Soc., 2024, 146, 29177–29188 CrossRef CAS PubMed.
- M. W. Campbell, M. B. Yuan, V. C. Polites, O. Gutierrez and G. A. Molander, Photochemical C-H Activation Enables Nickel-Catalyzed Olefin Dicarbofunctionalization, J. Am. Chem. Soc., 2021, 143, 3901–3910 Search PubMed.
- L.-M. Feng, S. Liu, Y.-H. Tu, P.-X. Rui and X.-G. Hu, Radical Deoxygenative Three-Component Reaction of Alcohols, Aryl Alkenes, and Cyanopyridines, Org. Lett., 2024, 26, 6225–6229 CrossRef CAS PubMed.
- X. Hu, I. Cheng-Sánchez, W. Q. Kong, G. A. Molander and C. Nevado, Nickel-catalysed enantioselective alkene dicarbofunctionalization enabled by photochemical aliphatic C-H bond activation, Nat. Catal., 2024, 7, 655–665 CrossRef CAS PubMed.
- J. Z. Wang, W. L. Lyon and D. W. C. MacMillan, Alkene dialkylation by triple radical sorting, Nature, 2024, 628, 104–109 Search PubMed.
- B.-Q. He, L. Zhao, J. Zhang, W.-H. Bao, M. Yang and X. Wu, Alcohol Activation by Benzodithiolylium for Deoxygenative Alkylation Driven by Photocatalytic Energy Transfer, Angew. Chem., Int. Ed., 2025, 64, e202423795 Search PubMed.
- X.-H. Ouyang, Y. Li, R.-J. Song, M. Hu, S. Luo and J.-H. Li, Intermolecular dialkylation of alkenes with two distinct C(sp3)-H bonds enabled by synergistic photoredox catalysis and iron catalysis, Sci. Adv., 2019, 5, eaav9839 CrossRef CAS PubMed.
- S. Xu, H. Chen, Z. Zhou and W. Kong, Three-Component Alkene Difunctionalization by Direct and Selective Activation of Aliphatic C-H Bonds, Angew. Chem., Int. Ed., 2021, 60, 7405–7411 Search PubMed.
- Y. Hong, M.-Y. Dong, D.-S. Li and H.-P. Deng, Photoinduced Three-Component Carboarylation of Unactivated Alkenes with Protic C(sp3)–H Feedstocks, Org. Lett., 2022, 24, 7677–7684 CrossRef CAS PubMed.
- M. Kim, E. You, J. Kim and S. Hong, Site-Selective Pyridylic C–H Functionalization by Photocatalytic Radical Cascades, Angew. Chem., Int. Ed., 2022, 61, e202204217 Search PubMed.
- W. Liu, C. Liu, M. Wang and W. Kong, Modular Synthesis of Multifunctionalized CF3-Allenes through Selective Activation of Saturated Hydrocarbons, ACS Catal., 2022, 12, 10207–10221 Search PubMed.
- E. C. Gentry and R. R. Knowles, Synthetic Applications of Proton-Coupled Electron Transfer, Acc. Chem. Res., 2016, 49, 1546–1556 Search PubMed.
- P. R. D. Murray, J. H. Cox, N. D. Chiappini, C. B. Roos, E. A. McLoughlin, B. G. Hejna, S. T. Nguyen, H. H. Ripberger, J. M. Ganley, E. Tsui, N. Y. Shin, B. Koronkiewicz, G. Qiu and R. R. Knowles, Photochemical and Electrochemical Applications of Proton-Coupled Electron Transfer in Organic Synthesis, Chem. Rev., 2022, 122, 2017–2291 CrossRef CAS PubMed.
- R. Tyburski, T. Liu, S. D. Glover and L. Hammarström, Proton-Coupled Electron Transfer Guidelines, Fair and Square, J. Am. Chem. Soc., 2021, 143, 560–576 CrossRef CAS PubMed.
- H. G. Yayla, H. Wang, K. T. Tarantino, H. S. Orbe and R. R. Knowles, Catalytic Ring-Opening of Cyclic Alcohols Enabled by PCET Activation of Strong O–H Bonds, J. Am. Chem. Soc., 2016, 138, 10794–10797 Search PubMed.
- K. Zhao, G. Seidler and R. R. Knowles, 1,3-Alkyl Transposition in Allylic Alcohols Enabled by Proton-Coupled Electron Transfer, Angew. Chem., Int. Ed., 2021, 60, 20190–20195 CrossRef CAS PubMed.
- K. Zhao, K. Yamashita, J. E. Carpenter, T. C. Sherwood, W. R. Ewing, P. T. W. Cheng and R. R. Knowles, Catalytic Ring Expansions of Cyclic Alcohols Enabled by Proton-Coupled Electron Transfer, J. Am. Chem. Soc., 2019, 141, 8752–8757 CrossRef CAS PubMed.
- E. Ota, H. Wang, N. L. Frye and R. R. Knowles, A Redox Strategy for Light-Driven, Out-of-Equilibrium Isomerizations and Application to Catalytic C–C Bond Cleavage Reactions, J. Am. Chem. Soc., 2019, 141, 1457–1462 Search PubMed.
- D. Q. Thach and R. R. Knowles, Ring Expansions of 1,2-Amino Alcohols to Amine Heterocycles Enabled by Proton-Coupled Electron Transfer, J. Am. Chem. Soc., 2025, 147, 21818–21823 CrossRef CAS PubMed.
- T. Ji, X.-Y. Chen, L. Huang and M. Rueping, Remote Trifluoromethylthiolation Enabled by Organophotocatalytic C–C Bond Cleavage, Org. Lett., 2020, 22, 2579–2583 Search PubMed.
- T. Kikuchi, K. Yamada, T. Yasui and Y. Yamamoto, Synthesis of Benzo-Fused Cyclic Ketones via Metal-Free Ring Expansion of Cyclopropanols Enabled by Proton-Coupled Electron Transfer, Org. Lett., 2021, 23, 4710–4714 Search PubMed.
- X. Wang, Y. Li and X. Wu, Photoredox/Cobalt Dual Catalysis Enabled Regiospecific Synthesis of Distally Unsaturated Ketones with Hydrogen Evolution, ACS Catal., 2022, 12, 3710–3718 CrossRef CAS.
- K. Liao, F. Wu, J. Chen and Y. Huang, Catalytic cleavage and functionalization of bulky and inert Csp3–Csp3 bonds via a relayed proton-coupled electron transfer strategy, Cell Rep. Phys. Sci., 2022, 3, 100763 Search PubMed.
- K. Liao, C. Y. Chan, S. Liu, X. Zhang, J. Chen and Y. Huang, Photoredox Cleavage of a Csp3–Csp3 Bond in Aromatic Hydrocarbons, J. Am. Chem. Soc., 2023, 145, 12284–12292 Search PubMed.
- S. Horiuchi, M. Oishi, A. Mizutani, H. Takamura, I. Kadota and K. Tanaka, Generation of alkyl radicals via C(sp3)–C(sp3) bond cleavage of xanthene-based precursors for photocatalytic Giese-type reaction, Chem. Commun., 2025, 61, 16376–16379 RSC.
- X. Hu, L. Ribadeau-Dumas and C. Nevado, Redox-neutral photocatalytic cleavage and gem-difluoroalkenylation of lignin linkages, Sci. Adv., 2025, 11, eady2227 CrossRef CAS PubMed.
- T. Matsuo, M. Sano, Y. Sumida and H. Ohmiya, Organic photoredox-catalyzed unimolecular PCET of benzylic alcohols, Chem. Sci., 2025, 16, 3150–3156 RSC.
- L. Huang, T. Ji and M. Rueping, Remote Nickel-Catalyzed Cross-Coupling Arylation via Proton-Coupled Electron Transfer-Enabled C–C Bond Cleavage, J. Am. Chem. Soc., 2020, 142, 3532–3539 Search PubMed.
- L. Huang, T. Ji, C. Zhu, H. Yue, N. Zhumabay and M. Rueping, Bioinspired desaturation of alcohols enabled by photoredox proton-coupled electron transfer and cobalt dual catalysis, Nat. Commun., 2022, 13, 809 Search PubMed.
- N. Varabyeva, M. Barysevich, Y. Aniskevich and A. Hurski, Ti(OiPr)4-Enabled Dual Photoredox and Nickel-Catalyzed Arylation and Alkenylation of Cyclopropanols, Org. Lett., 2021, 23, 5452–5456 CrossRef CAS PubMed.
- J. Wang and X. Li, Asymmetric β-arylation of cyclopropanols enabled by photoredox and nickel dual catalysis, Chem. Sci., 2022, 13, 3020–3026 RSC.
- Y. Patehebieke, R. Charaf, H. P. Bryce-Rogers, K. Ye, M. Ahlquist, L. Hammarström and C.-J. Wallentin, β-Scission of Secondary Alcohols via Photosensitization: Synthetic Utilization and Mechanistic Insights, ACS Catal., 2024, 14, 585–593 CrossRef CAS.
- Y. Patehebieke, R. Charaf, K. B. Pal, B. M. Baamonde, A. Brnovic, L. Hammarström and C.-J. Wallentin, PCET-mediated deconstructive cross-coupling of aliphatic alcohols, Chem. Sci., 2025, 16, 7720–7729 Search PubMed.
- S. Q. Zhu, X. Zhao, H. Li and L. L. Chu, Catalytic three-component dicarbofunctionalization reactions involving radical capture by nickel, Chem. Soc. Rev., 2021, 50, 10836–10856 RSC.
- P. Gao, Y. J. Niu, F. Yang, L. N. Guo and X. H. Duan, Three-component 1,2-dicarbofunctionalization of alkenes involving alkyl radicals, Chem. Commun., 2022, 58, 730–746 RSC.
- J. W. Tucker and C. R. J. Stephenson, Shining Light on Photoredox Catalysis: Theory and Synthetic Applications, J. Org. Chem., 2012, 77, 1617–1622 CrossRef CAS PubMed.
- S. Ni, R. Halder, D. Ahmadli, E. J. Reijerse, J. Cornella and T. Ritter, C–heteroatom coupling with electron-rich aryls enabled by nickel catalysis and light, Nat. Catal., 2024, 7, 733–741 Search PubMed.
- X. Du, I. Cheng-Sánchez and C. Nevado, Dual Nickel/Photoredox-Catalyzed Asymmetric Carbosulfonylation of Alkenes, J. Am. Chem. Soc., 2023, 145, 12532–12540 CrossRef CAS PubMed.
- L. Anghileri, H. Baunis, A. R. Bena, C. Giannoudis, J. H. Burke, S. Reischauer, C. Merschjann, R. F. Wallick, T. Al Said, C. E. Adams, G. Simionato, S. Kovalenko, L. Dell'Amico, R. M. van der Veen and B. Pieber, Evidence for a Unifying NiI/NiIII Mechanism in Light-Mediated Cross-Coupling Catalysis, J. Am. Chem. Soc., 2025, 147, 13169–13179 CrossRef CAS PubMed.
- N. A. Till, L. Tian, Z. Dong, G. D. Scholes and D. W. C. MacMillan, Mechanistic Analysis of Metallaphotoredox C–N Coupling: Photocatalysis Initiates and Perpetuates Ni(I)/Ni(III) Coupling Activity, J. Am. Chem. Soc., 2020, 142, 15830–15841 Search PubMed.
- N. A. Till, S. Oh, D. W. C. MacMillan and M. J. Bird, The Application of Pulse Radiolysis to the Study of Ni(I) Intermediates in Ni-Catalyzed Cross-Coupling Reactions, J. Am. Chem. Soc., 2021, 143, 9332–9337 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |