Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Single-position ligand modifications tune CB2R activity by targeting the toggle switch
Rudolf L. Z.
Ganzoni†
 a,
Miroslav
Kosar†
a,
Miroslav
Kosar†
 a,
Yongqi
Han
a,
Yongqi
Han
 b,
Rosa Maria
Vitale
b,
Rosa Maria
Vitale
 c,
Pietro
Amodeo
c,
Pietro
Amodeo
 c,
Xiaoting
Li
c,
Xiaoting
Li
 d,
Zhonghua
Zha
d,
Zhonghua
Zha
 d,
Kacper J.
Patej
d,
Kacper J.
Patej
 a,
Bilal
Kicin
a,
Bilal
Kicin
 a,
Taddäus E. N.
Strunden
a,
Taddäus E. N.
Strunden
 a,
Lisa
Reichert
a,
Lisa
Reichert
 a,
Uxía
Gómez-Bouzó
a,
Uxía
Gómez-Bouzó
 a,
Themiya P.
Perera
a,
Themiya P.
Perera
 b,
Kenneth
Atz
b,
Kenneth
Atz
 e,
Wolfgang
Guba
e,
Wolfgang
Guba
 e,
Christian
Bartelmus
e,
Christian
Bartelmus
 e,
Raphael
Bigler
e,
Raphael
Bigler
 f,
Paolo
Tosatti
f,
Stephan
Bachmann
f,
Tian
Hua
f,
Paolo
Tosatti
f,
Stephan
Bachmann
f,
Tian
Hua
 d,
David A.
Sykes
d,
David A.
Sykes
 *b,
Dmitry B.
Veprintsev
*b,
Dmitry B.
Veprintsev
 *b,
Uwe
Grether
*b,
Uwe
Grether
 *e and
Erick M.
Carreira
*e and
Erick M.
Carreira
 *a
*a
aLaboratorium für Organische Chemie, Eidgenössische Technische Hochschule Zürich, Vladimir-Prelog-Weg 3, 8093 Zürich, Switzerland. E-mail: erickm.carreira@org.chem.ethz.ch
bFaculty of Medicine & Health Sciences, University of Nottingham, Centre of Membrane Proteins and Receptors (COMPARE), Nottingham NG7 2UH, UK. E-mail: david.sykes@nottingham.ac.uk; dmitry.veprintsev@nottingham.ac.uk
cInstitute of Biomolecular Chemistry, National Research Council, Via Campi Flegrei 34, 80078 Pozzuoli, Italy
diHuman Institute, ShanghaiTech University, Shanghai 201210, China
eRoche Pharma Research & Early Development, Roche Innovation Center Basel, F. Hoffmann-La Roche Ltd, 4070 Basel, Switzerland. E-mail: uwe.grether@roche.com
fDepartment of Process Chemistry and Catalysis, F. Hoffmann-La Roche Ltd, Basel 4070, Switzerland
First published on 18th March 2026
Abstract
Cannabinoid receptor type 2 (CB2R) is a prominent class A G protein-coupled receptor (GPCR) and is a therapeutic target of interest for inflammatory diseases, pain management, and neurodegenerative disorders. We report the development of ligands based on HU-308 that share a single central scaffold but bear diverse sidechains, enabling controlled modulation of GPCR activation. Structural modifications at a single position of the parent ligand allow modulation of the single-residue toggle switch of CB2R, Trp2586.48, and thereby control over receptor activity. A continuum of functional outcomes is achieved through interaction of the ligands with the CB2R toggle switch, leading to full agonism, partial agonism, neutral antagonism, or partial inverse agonism. Several low-efficacy ligands display protean behavior across assays, underscoring context-dependent modulation of CB2R and its importance in profiling such ligands. A notable compound within this series is CF3-substituted (S)-1, which displays distinct CB2R affinity, potency, and a biased CB2R signaling profile. We provide a rationale based on molecular dynamics simulations for the unique pharmacological profile observed and suggest that stabilization of an active receptor conformation occurs by close-contact interaction of (S)-1 with the CB2R toggle switch. Our findings demonstrate that strategic structural modifications of class A GPCR ligands may, by targeting a receptor's toggle switch, shift ligands to different positions along the efficacy spectrum, independent of their parent scaffold's original functional profile.
Introduction
G protein-coupled receptors (GPCRs) represent the largest family of membrane proteins in the human genome and are targets for approximately one-third of all approved drugs by the Food and Drug Administration (FDA),1,2 with class A GPCR subpopulations constituting approximately 86% of GPCR drug targets.3 Class A GPCRs have attracted substantial interest and investment,3–5 with the market for small-molecule modulators projected to grow significantly in the coming years.6 However, the challenge associated with discovering new ligands for a target is significant,7 and developing suitable leads into a drug is underscored by its prohibitive cost.8–10 The process of lead identification for GPCR targets is particularly difficult because of a variety of different binding and activation modes and the associated, diverse functional outcomes at the receptor.7,11The cannabinoid receptors are prominent examples of class A GPCRs which hold promise for treatment of various ailments12 but have fallen short of that promise so far.13 In recent years, cannabinoid receptor 2 (CB2R) especially has garnered significant interest as a therapeutic target. As CB2R is mainly expressed in immune cells in the periphery,14,15 selective pharmacological intervention at CB2R offers potential for the treatment of neuroinflammation,16 autoimmune disorders,17,18 chronic pain,19,20 as well as cancer21,22 while circumventing adverse effects associated with cannabinoid receptor 1 (CB1R) modulation.23 The promising therapeutic potential of selective CB2R modulation, however, has not yet successfully translated to the clinic,24,25 with recent discontinuation of development programs aimed at treatment of cancer26 and irritable bowel syndrome.27 Most recently, development of ajulemic acid (Lenabasum), a CB1R/CB2R dual agonist for the treatment of cystic fibrosis and dermatomyositis, was discontinued as of 2023.28
The apparent difficulty of advancing CB2R selective compounds in the clinic underpins the need for a better understanding of structural determinants of receptor activation. Indeed, there is a paucity of fine-tunable ligands to activate or deactivate CB2R over the entirety of its efficacy spectrum: agonists ↔ partial agonists ↔ neutral antagonists ↔ inverse agonists.
Despite the abundance of known CB2R agonists, CB2R-selective partial agonists are scarce.30–32 This scarcity reflects the challenges in designing ligands that finely tune receptor activity without inducing maximal activation or complete inhibition. Receptor density and coupling efficiency are two key factors known to influence the apparent degree of agonism.33,34 Consequently, the pharmacological behavior of partial agonists is highly context dependent: the same ligand may function as an agonist, neutral antagonist, or inverse agonist, depending on basal activity and cellular environment in native as well as overexpressing systems. In this respect, context dependence of ligand efficacy has been referred to as protean agonism. For example, as described in pioneering studies, in systems with high constitutive CB2R activity, low-efficacy partial agonists (AM-1241 and L768242) may function as neutral antagonists or inverse agonists, whereas in systems with reduced basal activity, the same ligands can activate the receptor.35
In the clinic, it has been suggested that partial agonists may offer better control of context-dependent modulation, thereby achieving desired therapeutic effects.4,36,37 For example, Δ9-tetrahydrocannabinol (THC), a CB1R/CB2R partial agonist, elicits milder psychotropic adverse effects compared to synthetic compounds known to be full agonists of CB1R (e.g. CP-55940, HU-210).38,39 In the context of CB2R, the partial CB2R-selective agonist LEI-102 was shown to effectively protect against cisplatin induced kidney injury in mice.40 This finding is particularly relevant given that CB2R activation by full agonists has negative implications for immunosuppression, leading to infection and sepsis.41,42 Analogously, limited downmodulation by partial inverse agonists may offer opportunities in ameliorating conditions which result from increased basal tone, without completely abolishing signaling and thereby disrupting the receptor's role in maintaining homeostasis.43,44
Previous work from our group focused on fluorescent imaging of CB2R.45 We detailed a C(2′) phenyl-substituted HU-308 (ref. 46) derivative that displayed inverse agonist activity. Notably, the ligand retains its inverse agonist functional profile, affinity, and selectivity independent of its conjugation to a range of fluorescent reporters (e.g. DY-480XL, fluorescein, Alexa488). We wondered whether variation of substituents at C(2′) (Fig. 1) would offer a venue for a novel approach to ligand design that leverages reutilization of known scaffolds for the purpose of covering a broader and fine-tuned range of accessible functionality.
 | ||
| Fig. 1 Ligand design based on the HU-308 scaffold and modification of the C(2′) position. Fine-tuned actuation of the CB2R Trp2586.48 single-residue toggle switch leads to a continuum of functional outcomes at the receptor.29 | ||
Herein, we provide a basis for strategic structural alterations at an established class A GPCR ligand to venture beyond switch-like, binary functional responses. Inspired by recent reports showcasing intermediate receptor states of class A GPCRs that populate an active-to-inactive continuum,47 we sought to elicit gradual functional outcomes at CB2R. It has recently been appreciated that the activity of many class A GPCRs hinges on conformational flexibility of residues within the receptor that function as molecular toggle switches.2,48 Targeting the precise modulation of a receptor's toggle switch therefore presents an opportunity to access its efficacy spectrum and elicit fine-tuned functional responses. In this respect, we employ a single, exemplary molecular scaffold to elicit functional outcomes spanning the efficacy spectrum of the receptor by virtue of targeted modulation of Tryptophan 2586.48 (Trp2586.48), the single-residue toggle switch of CB2R.40,49,50
Based on the canonical CB2R ligand HU-308 (ref. 46) and intuitive design principles guided by docking studies, we synthesized and studied a collection of CB2R ligands. The ligands we describe display high CB2R affinity and excellent selectivity for CB2R over CB1R. Profiling in a panel of in vitro (G protein recruitment) as well as in cellulo (cyclic adenosine monophosphate (cAMP) production, β-arrestin recruitment, G protein recruitment) functional assays revealed a spectrum of accessible ligand efficacy compared to its parent scaffold spanning agonist, partial agonist, neutral antagonist, and inverse agonist functionality. The assays were benchmarked with canonical balanced agonist CP-55,940 (ref. 51–53) and CB2R-selective inverse agonist SR-144,528.51 An exemplary compound within the series synthesized, C(2′) CF3-derivative (S)-1, a high-affinity partial agonist, was investigated by molecular dynamics (MD) simulations. This revealed a close contact interaction between (S)-1 and Trp2586.48 in the CB2R/ligand complex.
Results and discussion
In silico ligand design
Our studies were based on the hypothesis that varying substituent size at the C(2′) position of the HU-308-derived ligands would affect the functional outcome at the receptor. At the onset of our investigation, we conducted docking studies to rapidly evaluate the influence of various substitutions at the homobenzylic C(2′) position of the HU-308 sidechain. This approach was aimed at qualitatively assessing the potential to modulate the Trp2586.48 toggle switch by employing a simple-to-use model based on substituent size and to provide an initial gauge of the hypothesis' viability (Fig. 2, see SI). We focused on HU-308 derivatives where substitution at the C(2′) position would incrementally alter the steric bulk in the binding pocket and thereby affect the fit of these molecules in either the active or inactive state of CB2R. We sought to explore the extent to which larger or modified aryl groups could be accommodated in the cleft hosting the CB2R toggle switch and to assess their potential to elicit a more efficacious inverse agonist response. Accordingly, we expected smaller substituents to exhibit partial agonist or neutral antagonist activity, as they could be accommodated by both the active and inactive receptor states.The docking studies indicated that even moderately more spatially demanding groups than phenyl at C(2′), such as benzyl, would exceed the limits of the binding pocket (in both the active and the inactive receptor state) and likely diminish affinity for the receptor. Consequently, phenyl and para/meta-fluorine-substituted phenyl groups were selected as possessing maximal substituent volume likely to maintain a favorable fit, as judged by computational modeling (Fig. 2A and B). Our qualitative docking studies suggested that aryl modifications, as well as C(2′) n-propyl substitution (Fig. 2C) could be accommodated only within the inactive state of CB2R, whereas smaller aliphatic side chains were predicted to fit both the active and inactive receptor conformation (Fig. 2D–F). Based on this initial viability check, we proceeded to compile a focused panel of potential ligands.
To the best of our knowledge, the only other reported modification at the analogous C(2′) position involves the study of Δ8-THC-derivatives with CH3/H substitution at C(2′) and H/H substitution at C(1′), which is associated with CB2R selectivity over CB1R.54 Beyond that, there is a lack of studies elucidating how small aliphatic groups at this position influence receptor affinity or what drives the compounds' functional profiles. This further motivated our investigation into rational structural modification beyond methyl at the C(2′) position of HU-308, shifting ligand functionality away from full agonism. This emerged as a working model to tune ligand functionality across a broad efficacy spectrum.
Synthesis
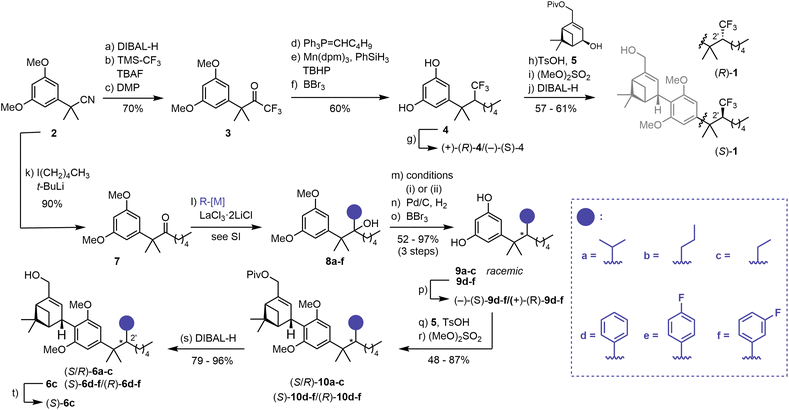
A modular synthetic approach was employed to access a panel of C(2′) substituted HU-308 derivatives. In the synthesis of fluorescent inverse agonist probes based on HU-308 amide derivatives, a dialkyl ketone readily underwent addition by phenyl lithium to give a tertiary alcohol that was subjected to dehydration and reduction.45 Implementation of this strategy to access CF3-substituted derivatives (R)-1 and (S)-1 failed and required a new approach. The synthesis of (R)-1/(S)-1 commenced with reduction of nitrile 2 to the corresponding aldehyde, which was then treated with TMS-CF3 (ref. 55) and tetrabutylammonium fluoride (TBAF) (Scheme 1).56 Subsequent oxidation with Dess-Martin Periodinane (DMP) afforded CF3-ketone 3 (70%, 3 steps). Introduction of the n-pentyl fragment proved problematic, as ketone reduction by the corresponding organometal reagent57,58 occurred preferentially over 1,2-addition. However, treatment of 3 with Ph3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CHC4H9 effected efficient olefination. Attempted olefin reduction under classical hydrogenation conditions (Pd/C, H2) proved challenging, as no conversion was observed at 1 atm, and reaction at elevated pressure (7 atm) was accompanied by week-long reaction times and overreduction of the resorcinol to the corresponding cyclohexane. Consequently, we employed a modified hydrogen atom transfer (HAT) procedure59 to effect smooth reduction of the unusual CF3-olefin. Subsequent demethylation afforded resorcinol 4 in 60% yield (3 steps). Separation by semi-preparative supercritical fluid chromatography (SFC) furnished the two enantiomers (+)-(R)-4 and (−)-(S)-4 in excellent enantiomeric excess (ee) (>99% and 98%, respectively). The absolute configuration of the C(2′) stereocenter was unambiguously established by X-ray crystallographic studies of a derivative (see SI and Schemes S1 and S2). The parallel synthesis of (R)-1/(S)-1 was completed by Friedel–Crafts allylation of (R)-4 and (S)-4 with allylic alcohol 5 in the presence of TsOH·H2O. This was followed by methylation and pivalate removal, affording the CF3-bearing HU-308-derivatives in 57 to 61% yield over three steps.
CHC4H9 effected efficient olefination. Attempted olefin reduction under classical hydrogenation conditions (Pd/C, H2) proved challenging, as no conversion was observed at 1 atm, and reaction at elevated pressure (7 atm) was accompanied by week-long reaction times and overreduction of the resorcinol to the corresponding cyclohexane. Consequently, we employed a modified hydrogen atom transfer (HAT) procedure59 to effect smooth reduction of the unusual CF3-olefin. Subsequent demethylation afforded resorcinol 4 in 60% yield (3 steps). Separation by semi-preparative supercritical fluid chromatography (SFC) furnished the two enantiomers (+)-(R)-4 and (−)-(S)-4 in excellent enantiomeric excess (ee) (>99% and 98%, respectively). The absolute configuration of the C(2′) stereocenter was unambiguously established by X-ray crystallographic studies of a derivative (see SI and Schemes S1 and S2). The parallel synthesis of (R)-1/(S)-1 was completed by Friedel–Crafts allylation of (R)-4 and (S)-4 with allylic alcohol 5 in the presence of TsOH·H2O. This was followed by methylation and pivalate removal, affording the CF3-bearing HU-308-derivatives in 57 to 61% yield over three steps.
The synthesis of a collection of HU-308 derived ligands, (S/R)-6a–c, (S)-6c, (S)-6d–f, and (R)-6d–f was also initiated from common nitrile precursor 2 (Scheme 1). Treatment of 2 with n-pentyl lithium, followed by imine hydrolysis, afforded dimethyl benzyl ketone 7. A variety of substituents were introduced by organometal 1,2-addition mediated by LaCl3·2LiCl,60 essential for the high efficiency of this step, providing tertiary alcohols 8a–f. Subsequent elimination of the alcohols generated the respective olefins. Cleavage of the methyl ethers and hydrogenation mediated by Pd/C afforded resorcinols 9a–f in good yields (52 to 97%, 3 steps). We carried out extensive efforts to develop an enantioselective hydrogenation protocol. However, the highest ee achieved was 80% (see SI, Table S1). Separation of 9a–f by semi-preparative SFC proved effective for isolation of enantiomers (−)- and (+)-9d–f (98–>99% ee). The assignment of the absolute configuration of (−)-(S)-9d and (+)-(R)-9d was carried out by independent synthesis from a previously characterized compound45 (see SI). For (−)-(S)-9e/(+)-(R)-9e and (−)-(S)-9f/(+)-(R)-9f the configuration is assigned by analogy based on SFC retention times and optical rotation (see SI).
Attempts at separation of the enantiomers for 9a–c were not met with success. More than 20 chiral SFC columns were screened, and separation was attempted at multiple synthetic stages. However, efforts to resolve the enantiomers were mostly unsuccessful. To nevertheless assess the influence of these intermediate-size C(2′) sidechains, 9a–c were then employed as racemates, affording inseparable C(2′) diastereomer mixtures in the subsequent steps. With resorcinols (−)-(S)-9d–f, (+)-(R)-9d–f and 9a–c in hand, we set our sights on the completion of the synthesis in analogy to (R)-1/(S)-1: Friedel–Crafts allylation with 5 proceeded smoothly and furnished, after methylation, pivalates (S)-10d–f, (R)-10d–f, and (S/R)-10a–c in good yields (48 to 87%, 2 steps). In a last step, reductive cleavage of the pivalate afforded HU-308 analogs (S)-6d–f, (R)-6d–f, and (S/R)-6a–c (79 to 96%). At this stage, one diastereomer of 6c could be separated by semi-preparative SFC, affording (+)-6c (99% ee), whose configuration was tentatively assigned as (S) by analogy to (R)-1/(S)-1 (see SI).
Affinity profiling: competition binding assay
To validate that the HU-308 derivatives synthesized interact with CB2R and retain the high selectivity of the parent compound, we determined their binding affinities at CB2R and CB1R. To that end, we employed our validated time-resolved Förster resonance energy transfer (TR-FRET) binding assay.45,61–64 HEK293 membrane preparations of SNAP-Lumi4-Tb labelled hCB2R and hCB1R were incubated with a validated fluorescent probe (see SI) and the novel ligands (Table 1).64 In accord with our previous findings with phenyl-modified HU-308 derivatives, compounds (S)-6d and (R)-6d showed high affinity for CB2R (pKi = 7.15 and 8.88, respectively), as well as excellent selectivity for CB2R over CB1R in the case of (R)-6d (243-fold). Similarly, CF3-bearing derivatives (R)-1 and (S)-1 displayed selectivity for CB2R (18- and 3615-fold, respectively) with (S)-1 standing out with its high affinity (pKi = 9.57). In contrast, (R)-1 showed attenuated CB2R affinity (pKi = 6.56). The trend of high binding affinities continued for derivatives bearing aliphatic sidechains, as apparent for (S/R)-6a (pKi = 9.87) as well as (S)-6c and (S/R)-6c (pKi = 8.96 and 9.76, respectively). (S)-6c Displayed six-fold reduced affinity compared to (S/R)-6c. (S/R)-6b Displayed good binding and selectivity at CB2R (pKi = 8.62, Ki ratio = 1996).| Ligand | C(2′)-R | pKi [nM] | ||
|---|---|---|---|---|
| CB2R | CB1R | K i ratio (CB1R/CB2R) | ||
| a Competition binding data (pKi) were determined in a TR-FRET assay at 37 °C with membrane preparations from either hCB2R-HEK293 or hCB1R-HEK293 cells. Data shown as a mean, N = 3–4 (±SEM). b C(2′) Diastereomeric mixture. c C(2′) Configuration assigned by analogy. d Previously reported in the same assay.64 | ||||
| (R)-1 | CF3 | 6.56 (±0.11) | 5.37 (±0.20) | 18 |
| (S)-1 | CF3 | 9.57 (±0.42) | 5.84 (±0.30) | 3615 |
| (S/R)-6ab | i-Pr | 9.87 (±0.21) | 5.50 (±0.12) | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 912 912 |
| (S/R)-6bb | n-Pr | 8.62 (±0.18) | 5.50 (±0.10) | 1996 |
| (S)-6cc | Et | 8.96 (±0.16) | 5.93 (±0.20) | 1189 |
| (S/R)-6cb | Et | 9.76(±0.16) | 5.75 (±0.37) | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 098 098 |
| (S)-6d | Ph | 7.15 (±0.07) | 6.30 (±0.26) | 10 |
| (R)-6d | Ph | 8.88 (±0.03) | 6.66 (±0.26) | 243 |
| (S)-6ec | p-F-Ph | 6.13 (±0.07) | 6.36 (±0.27) | 1 |
| (R)-6ec | p-F-Ph | 7.41 (±0.33) | 6.27 (±0.26) | 11 |
| (S)-6fc | m-F-Ph | 7.01(±0.18) | 5.85 (±0.20) | 15 |
| (R)-6fc | m-F-Ph | 9.12 (±0.20) | 6.26 (±0.05) | 600 |
| HU-308d | — | 7.57d | 5.41d | 145d |
| CP-55,940d | — | 7.51d | 8.32d | 6d |
| SR-144,528d | — | 8.55d | 6.43d | 131d |
Docking studies had predicted that fluorinated phenyl side chains would be accommodated within the CB2R binding pocket, and indeed we observed binding of these compounds at CB2R, albeit with a slight drop in affinity. Fluoroarene derivatives (S)-6e/(R)-6e (pKi = 6.13 and 7.41, respectively) and (S)-6f/(R)-6f (pKi = 7.01 and 9.12, respectively) exhibited reduced binding affinities compared to their non-fluorinated counterparts (S)-6d and (R)-6d. (R)-6f retained good selectivity (600-fold) and comparable affinity (pKi = 9.12) to (R)-6d for CB2R. These findings suggest that the para fluorine substituents on the C(2′) phenyl sidechains introduce steric bulk that is not optimally accommodated by the pocket that harbors the single-residue toggle switch Trp2586.48. Such substitution may lead to unfavorable interactions within the binding pocket of the receptor. A decrease in selectivity of these fluorinated HU-308 congeners for CB2R over CB1R may stem from their decreased affinity for CB2R, especially in the case of para-fluorination.
With binding profiles established, we next proceeded to systematically characterize the pharmacological responses elicited across established CB2R signaling pathways. Since the parent compound HU-308 behaves as a potent full agonist at CB2R,46,51 we were particularly interested to examine whether we could observe fine-tuned functional outcomes differing from full agonism.
Functional profiling: cAMP production, G protein recruitment, and β-arrestin recruitment
A canonical signaling pathway of CB2R involves its interaction with Gαi/o proteins, which transiently inhibit adenylyl cyclase, leading to a reduction in cellular cAMP levels and the suppression of protein kinase A activity.65 Accordingly, we focused our attention on elucidating the functional profiles of the HU-308 derivatives in a homogeneous time-resolved fluorescence (HTRF) cAMP assay and a bioluminescence resonance energy transfer (BRET) Gi-CASE assay (Table 2).66| Ligand | C(2′)-R | HTRF cAMP assay | Gi-CASE assay | ||
|---|---|---|---|---|---|
| pEC50 | E max/% | pEC50 | E max/% | ||
| a Potency (pEC50) and efficacy (Emax) data were obtained in a cAMP HTRF assay using hCB2R–CHO cells. b Potency (pEC50) and Emax data were obtained in a Gi-CASE BRET-based assay using membrane preparations from hCB2R-HEK293 T-REx cells. Data were normalized to agonist CP-55,940 response (100%) and basal level (0%). c Entries reflect pIC50. d Data were additionally normalized to inverse agonist SR-144,528 (ref. 51) (100%) and basal level (0%) and are shown in brackets. Data shown as a mean (±SEM), N = 3–4. e C(2′) Diastereomeric mixture. f C(2′) Configuration assigned by analogy. | |||||
| (R)-1 | CF3 | 6.59 (±0.12) | 90 (±6) | 6.48 (±0.13) | 62 (±10) |
| (S)-1 | CF3 | 7.97 (±0.09) | 95 (±4) | 9.42 (±0.19) | 78 (±1) |
| (S/R)-6ae | i-Pr | — | — | 8.31 (±0.20) | 40 (±4) |
| (S/R)-6be | n-Pr | 7.01 (±0.23)c | −33 (±5) [−45 (±7)]d | 7.15 (±0.03) | 24 (±6) |
| (S)-6cf | Et | 7.36 (±0.15) | 69 (±6) | 8.2 (±0.19) | 80 (±17) |
| (S/R)-6ce | Et | 6.87 (±0.11) | 81 (±5) | 8.72 (±0.25) | 67 (±5) |
| (S)-6d | Ph | 6.32 (±0.25)c | −25 (±4) [−34 (±5)]d | 8.29 (±0.22)c | −28 (±8) [−41 (±12)]d |
| (R)-6d | Ph | 6.61 (±0.17) c | −46 (±5) [−63 (±7)]d | 8.47 (±0.12)c | −43 (±9) [−64 (±13)]d |
| (S)-6ef | p-F-Ph | 5.66 (±0.31)c | −18 (±5) [−25 (±7)]d | 6.82 (±0.29)c | −29 (±7) [−42 (±10)]d |
| (R)-6ef | p-F-Ph | — | — | 7.22 (±0.10) | 25 (±3) |
| (S)-6ff | m-F-Ph | 5.97 (±0.41)c | −30 (±9) [−41 (±12)]d | 7.37 (±0.17)c | −24 (±8) [−36 (±12)]d |
| (R)-6ff | m-F-Ph | 6.76 (±0.13)c | −43 (±4) [−59 (±5)]d | 8.62 (±0.29)c | −40 (±10) [−59 (±15)]d |
| HU-308 | — | 7.76 (±0.11) | 89 (±5) | 7.40 (±0.03) | 91 (±4) |
| SR-144,528d | — | 5.56 (±0.13)c | −73 (±8) [−100 (±11)]d | 7.97 (±0.09)c | −68 (±4) [100 (±6)]d |
| CP-55,940 | — | 9.11 (±0.07) | 100 (±4) | 8.52 (±0.03) | 100 (±2) |
In the cAMP assay, the derivatives synthesized spanned a range of functionality from agonists (Table 2, e.g. (S)-1) to inverse agonist (Table 2, e.g. (R)-6d), with efficacies (Emax) ranging from 95 to −46%. Interestingly, both (R)-1 and (S)-1 displayed agonistic behavior, triggering decrease in cAMP levels, albeit with considerably differing potencies (pEC50 = 6.59 and 7.97, respectively).
Ethyl-substituted HU-308 derivatives (S)-6c and (S/R)-6c displayed partial agonist properties (Emax = 69% and 81%, respectively) in the assay together with high potencies (pEC50 = 7.36 and 6.87, respectively). HU-308 derivative (S/R)-6a with a branched i-Pr group displayed neutral antagonist behavior while elongated n-Pr-substituted derivative (S/R)-6b behaved as an inverse agonist (Emax = −33%). Substituents at the C(2′) position apparently mediate a fine-tuned grading of functionality from agonism to inverse agonism. These observations for the first time demonstrate the effect of relatively minor increases in sidechain size at C(2′) on the ligands' functional profiles.
The Gi-CASE data follow the trends observed with functional data obtained from the HTRF cAMP assay (Table 2, solid blue line Fig. 3). Agonists (R)-1 and (S)-1 displayed comparable pEC50 values to the cAMP assay (6.48 and 9.42, respectively) with attenuated efficacy (Emax = 62 and 78%, respectively) compared to the HU-308 parent ligand (Emax = 91%). Interestingly, (S)-1 displayed 100-fold increased potency relative to HU-308 (pEC50 = 7.40) in the assay. (S/R)-6a (Emax = 40%), (S)-6c (Emax = 80%), and (S/R)-6c (Emax = 67%) retained their partial agonist character in the Gi-CASE assay compared to the cAMP assay. However, in contrast to the cAMP functional readout as inverse agonist, (S/R)-6b displayed weak partial agonist properties (pEC50 = 7.15, Emax = 24%) in the Gi-CASE assay. We hypothesize that the ligands that show low efficacy may display protean behavior in terms of functionality across assays. Differences in the basal activity across assays or tissues may move partial agonists into the neutral antagonist/inverse agonist realm (and vice versa) – a known phenomenon for class A GPCRs.33,68,69 Indeed, other protean ligands at CB2R have been reported in the literature.35,70–72 Additionally, specific conditions of the assays may exhibit a bias in terms of accessible receptor conformations because of the truncated or modified cannabinoid receptors employed.49
Ligands bearing aromatic sidechains (S)-6d (Emax = −28%), (R)-6d (Emax = −43%), (S)-6e (Emax = −29%), (S)-6f (Emax = −24%), and (R)-6f (Emax = −40%) corroborated the functional profiles established by the HTRF cAMP assay. Only the results for para-F substituted (R)-6e deviated from the trend (pEC50 = 7.22, Emax = 25%), suggesting that (R)-6e may act as protean agonists in a context dependent fashion. The compound's reduced affinity could be a driving factor behind its observed functionality.
Notably, compounds (S/R)-6a and (S/R)-6b did not induce measurable recruitment of effector proteins in the β-arrestin as well as in the mini-Gi assay (Table 3). This lack of recruitment is consistent with their low efficacy and assay-dependent behavior observed in cAMP and Gi-CASE assays. (S)-6c and (S/R)-6c, more efficacious agonists in the previous assays, showed moderate potency and efficacy in the β-arrestin assay (pEC50 = 7.48, Emax = 41% and pEC50 = 7.91, Emax = 43%, respectively) as well as in the mini-Gi assay (pEC50 = 6.97, Emax = 51% and pEC50 = 7.40, Emax = 50%, respectively). Interestingly, while (S)-1 displayed robust Gi and β-arrestin recruitment (pEC50 = 7.44, Emax = 86% and pEC50 = 7.12, Emax = 72%, respectively), (R)-1 failed to elicit a response in both assays (mini-Gi and β-arrestin) and did not measurably recruit either effector protein. An observable trend for this set of essays is that only very efficacious agonists elicited a response, suggesting that β-arrestin and mini-Gi assays are well-suited for comparative assessment for the profiling of efficacious ligands because of their unamplified and associative nature. Conversely, more sensitive assays such as Gi-CASE (lower receptor reserve)66 and HTRF cAMP (downstream amplification) may be better suited for the identification of protean ligands.
| Ligand | C(2′)-R | Mini-Gi assay | β-arrestin assay | ||
|---|---|---|---|---|---|
| pEC50 | E max/% | pEC50 | E max/% | ||
| a Potency (pEC50) and Emax data were obtained in a mini-Gi BRET-based assay using hCB2R-HEK293 T-REx cells. b Potency (pEC50) and Emax data were obtained in a β-arrestin BRET-based assay using hCB2R-HEK293 T-REx cells. c Functional data were normalized to agonist CP-55,940 response (100%) and basal level (0%). Data shown as a mean (±SEM), N = 6–8. d C(2′) Diastereomeric mixture. e C(2′) Configuration assigned by analogy. | |||||
| (R)-1 | CF3 | — | — | — | — |
| (S)-1 | CF3 | 7.12 (±0.17) | 72 (±5) | 7.44 (±0.10) | 86 (±4) |
| (S/R)-6ad | i-Pr | — | — | — | — |
| (S/R)-6bd | n-Pr | — | — | — | — |
| (S)-6ce | Et | 6.97 (±0.07) | 51 (±5) | 7.48 (±0.20) | 41 (±5) |
| (S/R)-6cd | Et | 7.40 (±0.17) | 50 (±2) | 7.91 (±0.08) | 43 (±3) |
| HU-308 | — | 6.93 (±0.11) | 79 (±9) | 7.07 (±0.10) | 94 (±13) |
| CP-55,940 | — | 8.37 (±0.06) | 100 (±1) | 8.14 (±0.16) | 100 (±2) |
Data from the compounds that were able to elicit a response in the mini-Gi and β-arrestin assays was furthermore used to assess whether the ligands showed biased signaling behavior according to the method developed by Bouvier and co-workers.80 Indeed, (S)-1, (S)-6c, (S/R)-6c displayed bias towards β-arrestin signaling, mirroring the behavior of HU-308 (see SI Table S1 and Fig. S3). This suggests that C(2′) substitution has a limited impact on biased signaling and that the scaffold's preference for β-arrestin recruitment is governed by other structural determinants which are beyond the scope of this work.
Of the compounds with C(2′) aliphatic substitution, (S)-1 is an especially interesting case, as the introduction of a CF3 residue leads to a significant increase in potency as well as efficacy compared to the parent compound HU-308 in the Gi-CASE assay. In contrast, diastereomer (R)-1 displays a stark drop in affinity and potency at CB2R and did not recruit effector proteins in the mini-Gi and β-arrestin assays.
Of note, although Gi-CASE, HTRF cAMP, and mini-Gi assays all probe G protein-coupled signaling, they can yield markedly different outcomes, particularly for ligands that do not act as full agonists. Evaluating ligands across multiple functional assays is important, particularly for G protein-coupled pathways of signaling, since results can vary depending on where in the signaling cascade the measurement is taken.67 Context-dependency influenced by basal signaling tone as well as assay conditions and sensitivity can lead to functional profiles that may not be recapitulated across other assays targeting the same pathway. For instance, (R)-1 did not significantly recruit mini-Gi but did act as a partial agonist in the Gi-CASE (Emax = 62%) and HTRF cAMP (Emax = 90%) assays. Our results are consistent with previous observations that show how narrow functional profiling may lead to premature identification of seemingly clear-cut functional profiles, highlighting the importance of early, thorough profiling across multiple and comparable assay platforms.51
Molecular dynamics investigation of (R)-1 and (S)-1
Given the distinct pharmacological profile of compounds (R)-1 and (S)-1, as discussed above, molecular dynamics (MD) simulations were employed. We selected the X-ray structure of CB2R in its inactive conformation (PDB: 5ZTY) as the starting point for the MD simulations in order to investigate ligand-induced transition toward the active state.81,82 Starting from the inactive state rather than the active state is an approach to explore potential differences in receptor activation triggered by the two compounds (R)-1 and (S)-1. Within the orthosteric binding site, both epimers are predicted to exhibit the previously reported, canonical HU-308 L-shaped conformation40,49,50 which is stable over the simulated time of 1 µs (Fig. 5 and see SI). The hydroxyl group on the pinene core of both compounds (Fig. 5A for (R)-1, Fig. 5B for (S)-1) is predicted to form a hydrogen-bond with His952.65.The structure of the CB2R/(R)-1 simulated complex reflects an inactive state of the receptor. Conversely, in the simulation, the CF3 group of (S)-1 engages the indole of Trp2586.48 in a close contact interaction that promotes a reorientation of the Trp2586.48 sidechain. This interaction is reflected by a shorter distance between the fluorine atoms of (S)-1 and the NE1 atom of Trp2586.48 in the CB2R/(S)-1 complex (≤3.5 Å in 50.07% of the MD frames) compared to the CB2R/(R)-1 complex over the simulation period (see SI Fig. S5). Furthermore, only in the CB2R/(S)-1 simulated complex is the ionic lock (not shown in Fig. 5) between Arg1313.50 and Asp2406.30 disrupted, indicating a conformational shift towards the activated form of CB2R (see SI Fig. S6).40 Finally, the difference in free energy of binding (ΔΔG) between the two epimers, obtained by performing molecular mechanics/Poisson–Boltzmann (Generalized Born) surface area (MM/PB(GB)SA), shows that (S)-1 in complex is more stable by 3.55 kcal mol−1 (MM/GBSA) and 3.53 kcal mol−1 (MM/PBSA) than (R)-1 (see SI), providing insight for the marked difference in affinity between the two compounds. Overall, the MD simulations suggest that (S)-1 may demonstrate both higher receptor affinity and an agonist-like profile, providing a compelling rationale for its experimentally observed greater propensity for inducing CB2R activation and a greater affinity for the receptor compared to (R)-1.
Conclusions
We have demonstrated that peripheral structural changes to an established agonist scaffold enable access to the entire efficacy spectrum at CB2R. In line with our initial hypothesis that varying substituent size at the C(2′) position of HU-308-derived ligands would modulate the CB2R toggle switch and thereby affect functional outcome, targeting the modulation of Trp2586.48 enables graded functional outcomes. Our design considerations for reutilizing the canonical CB2R ligand HU-308 were guided by a qualitative docking model that allowed for assessment of interaction with the toggle switch. Synthesis of a panel of designed ligands was achieved by a unified approach that required a modified HAT-reduction for access to CF3-bearing derivatives. The ligands' subsequent pharmacological profiling revealed a wide range of accessible functionalities, from full agonists to partial agonists and neutral antagonists to inverse agonists. Notably, (S)-1 showed high potency and β-arrestin bias, whereas its (R)-epimer exhibited reduced potency and failed to recruit effector proteins in both mini-Gi and β-arrestin assays. This exemplifies the marked impact that minor modifications, such as the introduction of a CF3 group, can have on receptor binding affinity and functional outcomes. These findings were further substantiated by molecular dynamics simulations, which provided mechanistic insights into the proposed effect of a close-contact interaction with the toggle switch residue and its influence on the increased affinity and activity of (S)-1 compared to (R)-1 and HU-308.Our study, exemplified by precision pharmacology at CB2R, demonstrates how mechanistic understanding of class A GPCR activation facilitates rational design of fine-tunable ligands based on a single chemical scaffold and its binding modes in the orthosteric pocket. Derivatives with smaller aliphatic sidechains generally promote partial agonist activity that can switch into antagonist territory in a context-dependent manner. In contrast, sterically more demanding aromatic sidechains tend to induce inverse agonism or neutral antagonism, with those derivatives revealing a tight size tolerance for the cleft harboring the CB2R single-residue toggle switch.
More broadly, this work underscores the potential of scaffold reutilization on the basis of rational ligand design to achieve functional outcomes at designated positions on the efficacy spectrum. The approach presented herein is possible because of diversity-oriented synthesis strategies that enable modification at C(2′) of the HU-308 scaffold. Traditional focused library synthesis leads to variety of structures aimed typically at optimizing for one outcome. The present approach leverages targeted structural diversification to access diversity across a range of functionality.
Reutilizing known GPCR ligand scaffolds in this manner may be useful for early-stage discovery by leveraging pre-existing pharmacological and synthetic knowledge. Designing class A GPCR ligands that target toggle switches based on a single scaffold may provide a practical strategy to rapidly produce families of ligands with minimal modifications that display distinct functional profiles.
Author contributions
E. M. C., D. B. V., D. A. S., U. G. conceived and oversaw the project. M. K. and R. L. Z. G. conceived the project, conceived chemical experiments and synthetic routes to tool compounds and derivatives, synthesized compounds, and analyzed data of chemical synthesis as well as biological data. U. G. further provided project administration and logistics. Y. H. and T. P. P. performed selected biological assays (Gi-CASE). Y. H. performed data analysis and comparative data analysis on selected biological assays (Gi-CASE, HTRF cAMP). R. M. V. and P. A. performed the molecular dynamics (MD) simulations. T. H. conceived oversaw the performed HTRF cAMP assays. X. L. and Z. Z. performed HTRF cAMP assays and provided data analysis thereof. D. A. S. conceived and performed biological assays and provided data analysis thereof as well as resulting calculations (Tb-FRET, Gi-CASE, m-Gi, β-arrestin). W. G. conceived and oversaw the docking studies. K. A. and W. G. performed the docking studies. R. B., P. T., S. B., and C. B. conducted experiments and analytics pertaining to the attempted asymmetric hydrogenation. K. J. P., B. K., T. E. N. S., L. R., and U. G.-B. performed the synthesis of selected compounds. R. L. Z. G., M. K., and E. M. C. wrote the manuscript. All authors participated in discussion and preparation of the manuscript.Conflicts of interest
The authors declare the following competing financial interest: M. K., B. K., W. G., U. G., and E. M. C. have filed a patent on CB2R selective modulators and fluorescent probes.Data availability
Experimental data associated with this work are available in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc00062b.CCDC 2450722 contains the supplementary crystallographic data for this paper.83
Acknowledgements
We thank Eric Bald for his help with chiral separations and Isabelle Kaufmann for her assistance with logistics and administration. We are grateful to Dr Kurt Püntener and Manuela Müller for their expertise, insight, and experimental help with high-pressure hydrogenations. We thank René Arnold, Rainer Frankenstein, and Stephan Burkhardt for their help with NMR measurements, Jan Kovacovic for his expertise with high-pressure hydrogenation, and the MoBiAS team for MS analysis. We are grateful to Dr Nils Trapp and Michael Solar for X-ray crystallographic analysis. M. K. gratefully acknowledges a fellowship by the Scholarship Fund of the Swiss Chemical Industry (SSCI). T. H. was supported by the National Natural Science Foundation of China grant 32471267. MD studies were funded by project code PIR01_00011 “IBISCo”, PON 2014–2020.Notes and references
- A. S. Hauser, S. Chavali, I. Masuho, L. J. Jahn, K. A. Martemyanov, D. E. Gloriam and M. M. Babu, Pharmacogenomics of GPCR Drug Targets, Cell, 2018, 172, 41–54 CrossRef CAS PubMed.
- M. Zhang, T. Chen, X. Lu, X. Lan, Z. Chen and S. Lu, G protein-coupled receptors (GPCRs): advances in structures, mechanisms, and drug discovery, Signal Transduction Targeted Ther., 2024, 9, 88 CrossRef CAS PubMed.
- J. S. Lorente, A. V. Sokolov, G. Ferguson, H. B. Schioth, A. S. Hauser and D. E. Gloriam, GPCR drug discovery: new agents, targets and indications, Nat. Rev. Drug Discovery, 2025, 24, 458–479 CrossRef CAS PubMed.
- D. Yang, Q. Zhou, V. Labroska, S. Qin, S. Darbalaei, Y. Wu, E. Yuliantie, L. Xie, H. Tao, J. Cheng, Q. Liu, S. Zhao, W. Shui, Y. Jiang and M. W. Wang, G protein-coupled receptors: structure- and function-based drug discovery, Signal Transduction Targeted Ther., 2021, 6, 7 CrossRef CAS PubMed.
- A. S. Hauser, M. M. Attwood, M. Rask-Andersen, H. B. Schioth and D. E. Gloriam, Trends in GPCR drug discovery: new agents, targets and indications, Nat. Rev. Drug Discovery, 2017, 16, 829–842 CrossRef CAS PubMed.
- The Insight Partners 2023, Small Molecule Drug Discovery Market Key Players and Opportunities by 2031, https://www.theinsightpartners.com/reports/small-molecule-drug-discovery-market, accessed 18 Dec 2025 Search PubMed.
- P. Morales, M. M. Scharf, C. P. Johnson, A. Di Pizio and D. Hilger, Editorial: New approaches for the discovery of GPCR ligands, Front. Endocrinol., 2023, 14, 1272700 CrossRef PubMed.
- O. J. Wouters, M. McKee and J. Luyten, Estimated Research and Development Investment Needed to Bring a New Medicine to Market, 2009-2018, JAMA, 2020, 323, 844–853 CrossRef PubMed.
- A. Sertkaya, T. Beleche, A. Jessup and B. D. Sommers, Costs of Drug Development and Research and Development Intensity in the US, 2000-2018, JAMA Netw. Open, 2024, 7, e2415445 CrossRef PubMed.
- S. M. Paul, D. S. Mytelka, C. T. Dunwiddie, C. C. Persinger, B. H. Munos, S. R. Lindborg and A. L. Schacht, How to improve R&D productivity: the pharmaceutical industry's grand challenge, Nat. Rev. Drug Discovery, 2010, 9, 203–214 CrossRef CAS PubMed.
- P. Addis, U. Bali, F. Baron, A. Campbell, S. Harborne, L. Jagger, G. Milne, M. Pearce, E. M. Rosethorne, R. Satchell, D. Swift, B. Young and J. F. Unitt, Key aspects of modern GPCR drug discovery, SLAS Discovery, 2024, 29, 1–22 CrossRef CAS PubMed.
- M. Maccarrone, V. Di Marzo, J. Gertsch, U. Grether, A. C. Howlett, T. Hua, A. Makriyannis, D. Piomelli, N. Ueda and M. van der Stelt, Goods and Bads of the Endocannabinoid System as a Therapeutic Target: Lessons Learned after 30 Years, Pharmacol. Rev., 2023, 75, 885–958 CrossRef CAS PubMed.
- F. Shahbazi, V. Grandi, A. Banerjee and J. F. Trant, Cannabinoids and Cannabinoid Receptors: The Story so Far, iScience, 2020, 23, 101301 CrossRef CAS PubMed.
- S. Galiegue, S. Mary, J. Marchand, D. Dussossoy, D. Carriere, P. Carayon, M. Bouaboula, D. Shire, G. Le Fur and P. Casellas, Expression of central and peripheral cannabinoid receptors in human immune tissues and leukocyte subpopulations, Eur. J. Biochem., 1995, 232, 54–61 CrossRef CAS PubMed.
- P. Pacher and R. Mechoulam, Is lipid signaling through cannabinoid 2 receptors part of a protective system?, Prog. Lipid Res., 2011, 50, 193–211 CrossRef CAS PubMed.
- K. Bala, P. Porel and K. R. Aran, Emerging roles of cannabinoid receptor CB2 receptor in the central nervous system: therapeutic target for CNS disorders, Psychopharmacology, 2024, 241, 1939–1954 CrossRef CAS PubMed.
- E. D. Goncalves and R. C. Dutra, Cannabinoid receptors as therapeutic targets for autoimmune diseases: where do we stand?, Drug Discovery Today, 2019, 24, 1845–1853 CrossRef CAS PubMed.
- H. Khan, F. K. Ghori, U. Ghani, A. Javed and S. Zahid, Cannabinoid and endocannabinoid system: a promising therapeutic intervention for multiple sclerosis, Mol. Biol. Rep., 2022, 49, 5117–5131 CrossRef CAS PubMed.
- N. J. van den Hoogen, E. K. Harding, C. E. D. Davidson and T. Trang, Cannabinoids in Chronic Pain: Therapeutic Potential Through Microglia Modulation, Front. Neural Circuits, 2021, 15, 816747 CrossRef CAS PubMed.
- Y. Shang and Y. Tang, The central cannabinoid receptor type-2 (CB2) and chronic pain, Int. J. Neurosci., 2017, 127, 812–823 CrossRef CAS PubMed.
- C. Laezza, C. Pagano, G. Navarra, O. Pastorino, M. C. Proto, D. Fiore, C. Piscopo, P. Gazzerro and M. Bifulco, The Endocannabinoid System: A Target for Cancer Treatment, Int. J. Mol. Sci., 2020, 21, 747–767 CrossRef CAS PubMed.
- M. B. Faiz, F. Naeem, M. Irfan, M. A. Aslam, L. M. Estevinho, D. A. Atessahin, A. M. Alshahrani, D. Calina, K. Khan and J. Sharifi-Rad, Exploring the therapeutic potential of cannabinoids in cancer by modulating signaling pathways and addressing clinical challenges, Discover Oncol., 2024, 15, 490 CrossRef PubMed.
- F. A. Moreira, M. Grieb and B. Lutz, Central side-effects of therapies based on CB1 cannabinoid receptor agonists and antagonists: focus on anxiety and depression, Best Pract. Res., Clin. Endocrinol. Metab., 2009, 23, 133–144 CrossRef CAS PubMed.
- J. L. Wilkerson, J. A. Bilbrey, J. S. Felix, A. Makriyannis and L. R. McMahon, Untapped endocannabinoid pharmacological targets: Pipe dream or pipeline?, Pharmacol., Biochem. Behav., 2021, 206, 173192 CrossRef CAS PubMed.
- M. Kosar, L. Mach, E. M. Carreira, M. Nazare, P. Pacher and U. Grether, Patent review of cannabinoid receptor type 2 (CB(2)R) modulators (2016-present), Expert Opin. Ther. Pat., 2024, 34, 665–700 CrossRef CAS PubMed.
- TT-816 As Monotherapy or in Combination with a PD-1 Inhibitor in Patients with Advanced Cancers (SEABEAM) (MK3475-E88), https://clinicaltrials.gov/study/NCT05525455, accessed 4. Jan 2025.
- Olorinab in Irritable Bowel Syndrome With Predominant Constipation (IBS-C) and Irritable Bowel Syndrome With Predominant Diarrhea (IBS-D) (CAPTIVATE), https://clinicaltrials.gov/study/NCT04043455, accessed 4. Jan 2025.
- Corbus Pharmaceuticals – Lenabasum, https://adisinsight.springer.com/drugs/800007180, accessed 4. Jan 2026.
- R. Ganzoni, Figure created in BioRender, 2025, Figure_1_part1: https://BioRender.com/m8n0bwm; Figure_1_part2: https://BioRender.com/gak69nf; TOC: https://BioRender.com/vbn1xif.
- P. Mukhopadhyay, M. Baggelaar, K. Erdelyi, Z. Cao, R. Cinar, F. Fezza, B. Ignatowska-Janlowska, J. Wilkerson, N. van Gils, T. Hansen, M. Ruben, M. Soethoudt, L. Heitman, G. Kunos, M. Maccarrone, A. Lichtman, P. Pacher and M. Van der Stelt, The novel, orally available and peripherally restricted selective cannabinoid CB2 receptor agonist LEI-101 prevents cisplatin-induced nephrotoxicity, Br. J. Pharmacol., 2016, 173, 446–458 CrossRef CAS PubMed.
- A. I. Uba, H. Aluwala, H. Liu and C. Wu, Elucidation of partial activation of cannabinoid receptor type 2 and identification of potential partial agonists: Molecular dynamics simulation and structure-based virtual screening, Comput. Biol. Chem., 2022, 99, 107723 CrossRef CAS PubMed.
- K. J. Valenzano, L. Tafesse, G. Lee, J. E. Harrison, J. M. Boulet, S. L. Gottshall, L. Mark, M. S. Pearson, W. Miller, S. Shan, L. Rabadi, Y. Rotshteyn, S. M. Chaffer, P. I. Turchin, D. A. Elsemore, M. Toth, L. Koetzner and G. T. Whiteside, Pharmacological and pharmacokinetic characterization of the cannabinoid receptor 2 agonist, GW405833, utilizing rodent models of acute and chronic pain, anxiety, ataxia and catalepsy, Neuropharmacology, 2005, 48, 658–672 CrossRef CAS PubMed.
- H. Kurose and S. G. Kim, Pharmacology of Antagonism of GPCR, Biol. Pharm. Bull., 2022, 45, 669–674 CrossRef CAS PubMed.
- T. Kenakin, Efficacy at G-protein-coupled receptors, Nat. Rev. Drug Discovery, 2002, 1, 103–110 CrossRef CAS PubMed.
- I. Mancini, R. Brusa, G. Quadrato, C. Foglia, P. Scandroglio, L. S. Silverman, D. Tulshian, A. Reggiani and M. Beltramo, Constitutive activity of cannabinoid-2 (CB2) receptors plays an essential role in the protean agonism of (+)AM1241 and L768242, Br. J. Pharmacol., 2009, 158, 382–391 CrossRef CAS PubMed.
- M. Wu, E. Carballo-Jane, H. Zhou, P. Zafian, G. Dai, M. Liu, J. Lao, T. Kelly, D. Shao, J. Gorski, D. Pissarnitski, A. Kekec, Y. Chen, S. F. Previs, G. Scapin, Y. Gomez-Llorente, S. A. Hollingsworth, L. Yan, D. Feng, P. Huo, G. Walford, M. D. Erion, D. E. Kelley, S. Lin and J. Mu, Functionally selective signaling and broad metabolic benefits by novel insulin receptor partial agonists, Nat. Commun., 2022, 13, 942 CrossRef CAS PubMed.
- R. I. Ohlsen and L. S. Pilowsky, The place of partial agonism in psychiatry: recent developments, J. Psychopharmacol., 2005, 19, 408–413 CrossRef CAS PubMed.
- E. L. Theunissen, K. P. C. Kuypers, N. L. Mason and J. G. Ramaekers, A Comparison of Acute Neurocognitive and Psychotomimetic Effects of a Synthetic Cannabinoid and Natural Cannabis at Psychotropic Dose Equivalence, Front. Psychiatry., 2022, 13, 891811 CrossRef PubMed.
- T. S. Thorsen, Y. Kulkarni, D. A. Sykes, A. Boggild, T. Drace, P. Hompluem, C. Iliopoulos-Tsoutsouvas, S. P. Nikas, H. Daver, A. Makriyannis, P. Nissen, M. Gajhede, D. B. Veprintsev, T. Boesen, J. S. Kastrup and D. E. Gloriam, Structural basis of THC analog activity at the Cannabinoid 1 receptor, Nat. Commun., 2025, 16, 486 CrossRef CAS PubMed.
- X. Li, H. Chang, J. Bouma, L. V. de Paus, P. Mukhopadhyay, J. Paloczi, M. Mustafa, C. van der Horst, S. S. Kumar, L. Wu, Y. Yu, R. van den Berg, A. P. A. Janssen, A. Lichtman, Z. J. Liu, P. Pacher, M. van der Stelt, L. H. Heitman and T. Hua, Structural basis of selective cannabinoid CB(2) receptor activation, Nat. Commun., 2023, 14, 1447 CrossRef CAS PubMed.
- B. Csoka, Z. H. Nemeth, P. Mukhopadhyay, Z. Spolarics, M. Rajesh, S. Federici, E. A. Deitch, S. Batkai, P. Pacher and G. Hasko, CB2 cannabinoid receptors contribute to bacterial invasion and mortality in polymicrobial sepsis, PLoS One, 2009, 4, e6409 CrossRef PubMed.
- J. Chen, F. Wang, S. Zhang, Q. Lin, H. Xu, T. Zhu, L. Peng, F. Cen, F. Li, Z. Wang, C. G. Feng, Z. Yin, Y. Liu and G. Zhang, Activation of CD4(+) T Cell-Derived Cannabinoid Receptor 2 Signaling Exacerbates Sepsis via Inhibiting IL-10, J. Immunol., 2022, 208, 2515–2522 CrossRef CAS PubMed.
- L. M. Wingler and R. J. Lefkowitz, Conformational Basis of G Protein-Coupled Receptor Signaling Versatility, Trends Cell Biol., 2020, 30, 736–747 CrossRef CAS PubMed.
- R. A. de Ligt, A. P. Kourounakis and I. J. AP, Inverse agonism at G protein-coupled receptors: (patho)physiological relevance and implications for drug discovery, Br. J. Pharmacol., 2000, 130, 1–12 CrossRef CAS PubMed.
- M. Kosar, R. C. Sarott, D. A. Sykes, A. E. G. Viray, R. M. Vitale, N. Tomasevic, X. Li, R. L. Z. Ganzoni, B. Kicin, L. Reichert, K. J. Patej, U. Gomez-Bouzo, W. Guba, P. J. McCormick, T. Hua, C. W. Gruber, D. B. Veprintsev, J. A. Frank, U. Grether and E. M. Carreira, Flipping the GPCR Switch: Structure-Based Development of Selective Cannabinoid Receptor 2 Inverse Agonists, ACS Cent. Sci., 2024, 10, 956–968 CrossRef CAS PubMed.
- L. Hanus, A. Breuer, S. Tchilibon, S. Shiloah, D. Goldenberg, M. Horowitz, R. G. Pertwee, R. A. Ross, R. Mechoulam and E. Fride, HU-308: a specific agonist for CB(2), a peripheral cannabinoid receptor, Proc. Natl. Acad. Sci. U. S. A., 1999, 96, 14228–14233 CrossRef CAS PubMed.
- X. Wang, C. Neale, S. K. Kim, W. A. Goddard and L. Ye, Intermediate-state-trapped mutants pinpoint G protein-coupled receptor conformational allostery, Nat. Commun., 2023, 14, 1325 CrossRef CAS PubMed.
- S. Filipek, Molecular switches in GPCRs, Curr. Opin. Struct. Biol., 2019, 55, 114–120 CrossRef CAS PubMed.
- X. Li, T. Hua, K. Vemuri, J. H. Ho, Y. Wu, L. Wu, P. Popov, O. Benchama, N. Zvonok, K. Locke, L. Qu, G. W. Han, M. R. Iyer, R. Cinar, N. J. Coffey, J. Wang, M. Wu, V. Katritch, S. Zhao, G. Kunos, L. M. Bohn, A. Makriyannis, R. C. Stevens and Z. J. Liu, Crystal Structure of the Human Cannabinoid Receptor CB2, Cell, 2019, 176, 459–467 CrossRef CAS PubMed.
- T. Hua, X. Li, L. Wu, C. Iliopoulos-Tsoutsouvas, Y. Wang, M. Wu, L. Shen, C. A. Brust, S. P. Nikas, F. Song, X. Song, S. Yuan, Q. Sun, Y. Wu, S. Jiang, T. W. Grim, O. Benchama, E. L. Stahl, N. Zvonok, S. Zhao, L. M. Bohn, A. Makriyannis and Z. J. Liu, Activation and Signaling Mechanism Revealed by Cannabinoid Receptor-Gi Complex Structures, Cell, 2020, 180, 655–665 CrossRef CAS PubMed.
- M. Soethoudt, U. Grether, J. Fingerle, T. W. Grim, F. Fezza, L. de Petrocellis, C. Ullmer, B. Rothenhausler, C. Perret, N. van Gils, D. Finlay, C. MacDonald, A. Chicca, M. D. Gens, J. Stuart, H. de Vries, N. Mastrangelo, L. Xia, G. Alachouzos, M. P. Baggelaar, A. Martella, E. D. Mock, H. Deng, L. H. Heitman, M. Connor, V. Di Marzo, J. Gertsch, A. H. Lichtman, M. Maccarrone, P. Pacher, M. Glass and M. van der Stelt, Cannabinoid CB(2) receptor ligand profiling reveals biased signalling and off-target activity, Nat. Commun., 2017, 8, 13958 CrossRef CAS PubMed.
- W. A. Devane, F. A. Dysarz 3rd, M. R. Johnson, L. S. Melvin and A. C. Howlett, Determination and characterization of a cannabinoid receptor in rat brain, Mol. Pharmacol., 1988, 34, 605–613 CrossRef CAS PubMed.
- M. Herkenham, A. B. Lynn, M. D. Little, M. R. Johnson, L. S. Melvin, B. R. de Costa and K. C. Rice, Cannabinoid receptor localization in brain, Proc. Natl. Acad. Sci. U. S. A., 1990, 87, 1932–1936 CrossRef CAS PubMed.
- J. W. Huffman, S. M. Bushell, S. N. Joshi, J. L. Wiley and B. R. Martin, Enantioselective synthesis of 1-methoxy- and 1-deoxy-2'-methyl-delta8-tetrahydrocannabinols: new selective ligands for the CB2 receptor, Bioorg. Med. Chem., 2006, 14, 247–262 CrossRef CAS PubMed.
- I. Ruppert, K. Schlich and W. Volbach, Die ersten CF3-substituierten organyl(chlor)silane, Tetrahedron Lett., 1984, 25, 2195–2198 CrossRef CAS.
- G. K. S. Prakash, R. Krishnamurti and G. A. Olah, Synthetic methods and reactions. 141. Fluoride-induced trifluoromethylation of carbonyl compounds with trifluoromethyltrimethylsilane (TMS-CF3). A trifluoromethide equivalent, J. Am. Chem. Soc., 1989, 111, 393–395 CrossRef CAS.
- V. Grignard, Mixed organomagnesium combinations and their application in acid, alcohol, and hydrocarbon synthesis, Ann. Chim., 1901, 24, 433–490 CAS.
- M. S. Kharasch and S. Weinhouse, Grignard Reagents – their Reducing Action and Rates of Addition, J. Org. Chem., 1936, 01, 209–230 CrossRef CAS.
- K. Iwasaki, K. K. Wan, A. Oppedisano, S. W. Crossley and R. A. Shenvi, Simple, chemoselective hydrogenation with thermodynamic stereocontrol, J. Am. Chem. Soc., 2014, 136, 1300–1303 CrossRef CAS PubMed.
- A. Krasovskiy, F. Kopp and P. Knochel, Soluble lanthanide salts (LnCl3.2LiCl) for the improved addition of organomagnesium reagents to carbonyl compounds, Angew Chem. Int. Ed. Engl., 2006, 45, 497–500 CrossRef CAS PubMed.
- M. Kosar, D. A. Sykes, A. E. G. Viray, R. M. Vitale, R. C. Sarott, R. L. Ganzoni, D. Onion, J. M. Tobias, P. Leippe, C. Ullmer, E. A. Zirwes, W. Guba, U. Grether, J. A. Frank, D. B. Veprintsev and E. M. Carreira, Platform Reagents Enable Synthesis of Ligand-Directed Covalent Probes: Study of Cannabinoid Receptor 2 in Live Cells, J. Am. Chem. Soc., 2023, 145, 15094–15108 CrossRef CAS PubMed.
- R. C. Sarott, A. E. G. Viray, P. Pfaff, A. Sadybekov, G. Rajic, V. Katritch, E. M. Carreira and J. A. Frank, Optical Control of Cannabinoid Receptor 2-Mediated Ca(2+) Release Enabled by Synthesis of Photoswitchable Probes, J. Am. Chem. Soc., 2021, 143, 736–743 CrossRef CAS PubMed.
- R. C. Sarott, M. V. Westphal, P. Pfaff, C. Korn, D. A. Sykes, T. Gazzi, B. Brennecke, K. Atz, M. Weise, Y. Mostinski, P. Hompluem, E. Koers, T. Miljus, N. J. Roth, H. Asmelash, M. C. Vong, J. Piovesan, W. Guba, A. C. Rufer, E. A. Kusznir, S. Huber, C. Raposo, E. A. Zirwes, A. Osterwald, A. Pavlovic, S. Moes, J. Beck, I. Benito-Cuesta, T. Grande, N. E. S. Ruiz de Marti, A. Yeliseev, F. Drawnel, G. Widmer, D. Holzer, T. van der Wel, H. Mandhair, C. Y. Yuan, W. R. Drobyski, Y. Saroz, N. Grimsey, M. Honer, J. Fingerle, K. Gawrisch, J. Romero, C. J. Hillard, Z. V. Varga, M. van der Stelt, P. Pacher, J. Gertsch, P. J. McCormick, C. Ullmer, S. Oddi, M. Maccarrone, D. B. Veprintsev, M. Nazare, U. Grether and E. M. Carreira, Development of High-Specificity Fluorescent Probes to Enable Cannabinoid Type 2 Receptor Studies in Living Cells, J. Am. Chem. Soc., 2020, 142, 16953–16964 CrossRef CAS PubMed.
- L. Borrega-Roman, B. L. Hoare, M. Kosar, R. C. Sarott, K. J. Patej, J. Bouma, M. Scott-Dennis, E. J. Koers, T. Gazzi, L. Mach, S. Barrondo, J. Salles, W. Guba, E. Kusznir, M. Nazare, A. C. Rufer, U. Grether, L. H. Heitman, E. M. Carreira, D. A. Sykes and D. B. Veprintsev, A universal cannabinoid CB1 and CB2 receptor TR-FRET kinetic ligand-binding assay, Front. Pharmacol, 2025, 16, 1469986 CrossRef CAS PubMed.
- G. Wolfgang, M. Nazare and U. Grether, in New Tools to Interrogate Endocannabinoid Signalling: From Natural Compounds to Synthetic Drugs, ed. M. Maccarrone, The Royal Society of Chemistry, 2021, pp. 89–167, 10.1039/9781839160752.
- M. Scott-Dennis, F. A. Rafani, Y. Yi, T. Perera, C. R. Harwood, W. Guba, A. C. Rufer, U. Grether, D. B. Veprintsev and D. A. Sykes, Development of a membrane-based Gi-CASE biosensor assay for profiling compounds at cannabinoid receptors, Front. Pharmacol, 2023, 14, 1158091 CrossRef CAS PubMed.
- W. Stallaert, A. Christopoulos and M. Bouvier, Ligand functional selectivity and quantitative pharmacology at G protein-coupled receptors, Expert Opin. Drug Discovery, 2011, 6, 811–825 CrossRef CAS PubMed.
- F. Gbahou, A. Rouleau, S. Morisset, R. Parmentier, S. Crochet, J. S. Lin, X. Ligneau, J. Tardivel-Lacombe, H. Stark, W. Schunack, C. R. Ganellin, J. C. Schwartz and J. M. Arrang, Protean agonism at histamine H3 receptors in vitro and in vivo, Proc. Natl. Acad. Sci. U. S. A., 2003, 100, 11086–11091 CrossRef CAS PubMed.
- J. McDonald, T. A. Barnes, H. Okawa, J. Williams, G. Calo, D. J. Rowbotham and D. G. Lambert, Partial agonist behaviour depends upon the level of nociceptin/orphanin FQ receptor expression: studies using the ecdysone-inducible mammalian expression system, Br. J. Pharmacol., 2003, 140, 61–70 CrossRef CAS PubMed.
- B. B. Yao, S. Mukherjee, Y. Fan, T. R. Garrison, A. V. Daza, G. K. Grayson, B. A. Hooker, M. J. Dart, J. P. Sullivan and M. D. Meyer, In vitro pharmacological characterization of AM1241: a protean agonist at the cannabinoid CB2 receptor?, Br. J. Pharmacol., 2006, 149, 145–154 CrossRef CAS PubMed.
- B. Bosier and E. Hermans, Versatility of GPCR recognition by drugs: from biological implications to therapeutic relevance, Trends Pharmacol. Sci., 2007, 28, 438–446 CrossRef CAS PubMed.
- B. Bosier, G. G. Muccioli, E. Hermans and D. M. Lambert, Functionally selective cannabinoid receptor signalling: therapeutic implications and opportunities, Biochem. Pharmacol., 2010, 80, 1–12 CrossRef CAS PubMed.
- Y. K. Peterson and L. M. Luttrell, The Diverse Roles of Arrestin Scaffolds in G Protein-Coupled Receptor Signaling, Pharmacol. Rev., 2017, 69, 256–297 CrossRef CAS PubMed.
- M. Patel, C. Matti, N. L. Grimsey, D. F. Legler, J. A. Javitch, D. B. Finlay and M. Glass, Delineating the interactions between the cannabinoid CB(2) receptor and its regulatory effectors; beta-arrestins and GPCR kinases, Br. J. Pharmacol., 2022, 179, 2223–2239 CrossRef CAS PubMed.
- P. Morales, M. M. Scharf, M. Bermudez, A. Egyed, R. Franco, O. K. Hansen, N. Jagerovic, J. Jakubik, G. M. Keseru, D. J. Kiss, P. Kozielewicz, O. Larsen, M. Majellaro, A. Mallo-Abreu, G. Navarro, R. Prieto-Diaz, M. M. Rosenkilde, E. Sotelo, H. Stark, T. Werner and L. M. Wingler, Progress on the development of Class A GPCR-biased ligands, Br. J. Pharmacol., 2024, 182, 3249–3300 CrossRef PubMed.
- M. S. Ibsen, M. Connor and M. Glass, Cannabinoid CB(1) and CB(2) Receptor Signaling and Bias, Cannabis Cannabinoid Res., 2017, 2, 48–60 CrossRef CAS PubMed.
- E. Wouters, J. Walraed, S. D. Banister and C. P. Stove, Insights into biased signaling at cannabinoid receptors: synthetic cannabinoid receptor agonists, Biochem. Pharmacol., 2019, 169, 113623 CrossRef CAS PubMed.
- M. Patel, N. L. Grimsey, S. D. Banister, D. B. Finlay and M. Glass, Evaluating signaling bias for synthetic cannabinoid receptor agonists at the cannabinoid CB(2) receptor, Pharmacol. Res. Perspect., 2023, 11, e01157 CrossRef CAS PubMed.
- T. A. Tummino, C. Iliopoulos-Tsoutsouvas, J. M. Braz, E. S. O'Brien, R. M. Stein, V. Craik, N. K. Tran, S. Ganapathy, F. Liu, Y. Shiimura, F. Tong, T. C. Ho, D. S. Radchenko, Y. S. Moroz, S. R. Rosado, K. Bhardwaj, J. Benitez, Y. Liu, H. Kandasamy, C. Normand, M. Semache, L. Sabbagh, I. Glenn, J. J. Irwin, K. K. Kumar, A. Makriyannis, A. I. Basbaum and B. K. Shoichet, Virtual library docking for cannabinoid-1 receptor agonists with reduced side effects, Nat. Commun., 2025, 16, 2237 CrossRef CAS PubMed.
- E. T. van der Westhuizen, B. Breton, A. Christopoulos and M. Bouvier, Quantification of ligand bias for clinically relevant beta2-adrenergic receptor ligands: implications for drug taxonomy, Mol. Pharmacol., 2014, 85, 492–509 CrossRef PubMed.
- W. I. Weis and B. K. Kobilka, The Molecular Basis of G Protein-Coupled Receptor Activation, Annu. Rev. Biochem., 2018, 87, 897–919 CrossRef CAS PubMed.
- O. Diaz, J. A. R. Dalton and J. Giraldo, Revealing the Mechanism of Agonist-Mediated Cannabinoid Receptor 1 (CB1) Activation and Phospholipid-Mediated Allosteric Modulation, J. Med. Chem., 2019, 62, 5638–5654 CrossRef CAS.
- CCDC 2450722: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n85kp.
Footnote |
| † Rudolf L. Z. Ganzoni and Miroslav Kosar contributed equally to this work and therefore share co-first authorship. |
| This journal is © The Royal Society of Chemistry 2026 |