 Open Access Article
Open Access ArticlePhotoluminescence of ligand-protected gold nanoclusters: progress in experimental and theoretical studies
Kang
Li
a,
Pu
Wang
 *a and
Yong
Pei
*a and
Yong
Pei
 *ab
*ab
aDepartment of Chemistry, Key Laboratory of Environmentally Friendly Chemistry and Applications of Ministry of Education, Xiangtan University, Hunan Province 411105, China. E-mail: ypei2@xtu.edu.cn; 90wangpu@xtu.edu.cn
bCollege of Chemistry & Chemical Engineering, Hunan Normal University, Changsha, Hunan 410081, China
First published on 10th March 2026
Abstract
Ligand-protected gold nanoclusters, with their small sizes and atomically precise structures, exhibit unique photophysical properties that position them as promising candidates for applications in bioimaging, sensing, and optoelectronics. However, except for a few ligand-protected gold nanoclusters, their photoluminescence quantum yield (PLQY) is still relatively low, and the underlying emission mechanisms are not yet fully elucidated. This review highlights recent experimental and theoretical advances aimed at enhancing and elucidating the photoluminescence (PL) mechanisms of ligand-protected gold nanoclusters. Experimentally, strategies such as heterometallic doping, ligand engineering, and structural rigidification have been employed to promote radiative transitions and suppress nonradiative decay, resulting in substantial improvements in PLQY. Theoretically, methods including density functional theory (DFT), time-dependent density functional theory (TDDFT), and nonadiabatic molecular dynamics (NA-MD) have provided important insights into the PL origin, emission pathways, and excited-state dynamics of ligand-protected gold nanoclusters. These advances will deepen our understanding of structure–property relationships in ligand-protected gold nanoclusters and pave the way for the rational design of highly emissive gold nanoclusters.
1. Introduction
Owing to their ultrasmall size (1–3 nm) and atomically precise structures, ligand-protected gold nanoclusters exhibit unique physical and chemical properties that distinguish them from traditional gold nanoparticles (>3 nm, characterized primarily by surface plasmon resonance). In particular, they display rich photoluminescence (PL) behaviors across the visible and near-infrared (NIR) regions, which has established them as a research focus in the field of advanced functional nanomaterials. This atomic-level precision is specifically reflected in the well-defined “metal core—ligand shell” configuration of ligand-protected gold nanoclusters, which not only provides a structural basis for regulating PL performance but also renders them an ideal model for elucidating the correlation between structure and PL properties. Notably, the PL properties of ligand-protected gold nanoclusters are highly aligned with the demands of biological applications. Their excellent biocompatibility, relatively low toxicity, and persistent NIR emission capable of penetrating biological tissues render them ideal fluorescent probes for bioimaging, disease diagnostics, and therapeutic applications.1–7However, research on the PL of ligand-protected gold nanoclusters still faces two major challenges. First, from an experimental perspective, although systems with high photoluminescence quantum yield (PLQY) have been synthesized in recent years (e.g., Ir@Au12 (PLQY = 87%) and Au16Cu6 (PLQY ≈ 100%)),8–13 the PLQY of most gold nanoclusters is still relatively low, particularly in the NIR-II region, which fails to meet the signal intensity requirements for practical applications. Second, from a mechanistic perspective, the complex electronic dynamics induced by core–ligand interactions in these gold nanoclusters make it challenging to experimentally determine the origin of PL (core-dominated vs. surface-ligand-involved). Furthermore, the strong intrinsic spin–orbit coupling (SOC) of Au atoms efficiently promotes intersystem crossing (ISC) and reverse intersystem crossing (RISC) between singlet and triplet states,14–16 leading to fluorescence, phosphorescence, thermally activated delayed fluorescence (TADF), and the coexistence of multiple emission phenomena.17–23 The overlapping emission peaks arising from the small singlet–triplet state energy gap (ΔEST) further hinder the differentiation of emission types (phosphorescence or TADF).
Recently, experimental studies have made substantial progress in exploring the PL mechanism of ligand-protected gold nanoclusters via various characterization techniques, such as steady-state and time-resolved spectroscopy.18,24,25 These efforts have provided clues to the origin of PL but cannot fully reveal the microscopic mechanisms underlying excited-state dynamics and nonradiative relaxation. First-principles theoretical calculations have become a key tool to address these bottlenecks. Specifically, they can directly overcome the limitations of experimental characterization and provide atomic-level insights for the direct exploration of electronic structures, excited-state relaxation dynamics, and energy/charge transfer processes. With the ongoing advancement of computational methods and the increasing computational efficiency and computing resources, theoretical calculations have become an indispensable partner in experiments, offering theoretical references for the modulation of PL properties in these gold nanoclusters.
In this review, we summarize the latest experimental advances in enhancing the PL intensity of ligand-protected gold nanoclusters, as well as theoretical studies focused on exploring their excited-state properties. First, we summarize the experimental strategies employed to boost PL efficiency in recent years, including enhancing radiative decay (e.g., heterometal doping) and suppressing nonradiative pathways (e.g., ligand rigidification). Subsequently, we introduce the key computational methods utilized to study the excited-state properties of these gold nanoclusters, including time-dependent density functional theory (TDDFT) and nonadiabatic molecular dynamics (NA-MD), and analyze the applicability and accuracy boundaries of each method. Finally, we discuss theoretical investigations into the excited-state properties of these gold nanoclusters, including the origins of photoluminescence, SOC-driven emission pathways, and excited-state nonradiative relaxation dynamics, aiming to provide theoretical references for the precise design of ligand-protected gold nanoclusters featuring high PL performance.
2. Experimental advances in photoluminescence enhancement
Research on the photoluminescence of ligand-protected gold nanoclusters faces two primary challenges, including low PLQY and unclear photoluminescence mechanisms, which severely constrain the rational design of high-performance gold nanoclusters. Over the past decade, substantial experimental efforts have been devoted to addressing these issues, resulting in a range of effective strategies for PLQY enhancement, including size control,26–28 heterometallic doping,8,29–31 tailored ligand engineering,9,32–34 and aggregation-induced emission (AIE).35–37 The availability of ligand-protected gold nanoclusters with atomically precise structures and systematic spectroscopic data has also provided an essential experimental foundation for theoretical studies. Such well-defined cluster systems serve as benchmark models for validating computational approaches. For example, in TDDFT calculations, the assessment and selection of exchange–correlation functionals still rely largely on comparisons with experimental optical spectra (see Section 3).Nevertheless, most existing reviews emphasize synthetic strategies and potential applications of gold nanoclusters with high PLQY, while discussions on how different experimental strategies enhance PLQY via photophysical modulation are fragmented across the literature. A unified photophysical framework linking experimental strategies to the key factors that determine PLQY is still lacking. In this context, the present section provides a systematic overview of recent experimental advances in improving the PLQY of ligand-protected gold nanoclusters from a photophysical perspective. By focusing on the key physical parameters governing PLQY, this section highlights major experimental breakthroughs, outlines unresolved challenges, and underscores the indispensable role of computational methods in elucidating PLQY enhancement mechanisms and guiding rational experimental design.
2.1 Photophysical basis for PLQY enhancement
According to the PLQY expression, Φ = kr/(kr + knr), the decay of the excited states in ligand-protected gold clusters proceeds primarily through two competing pathways, i.e., radiative and nonradiative transitions (as illustrated in Fig. 1). The relative magnitudes of the corresponding rate constants ultimately determine the PLQY of the system. Upon photon absorption, an electron is promoted from an occupied orbital to an unoccupied orbital with higher energy, driving the system from the ground state to an excited state. In accordance with Kasha's rule, excitation to higher-lying excited states is typically followed by rapid nonradiative relaxation processes, including internal conversion (IC), ISC, and vibrational relaxation, leading to population of the lowest excited state (S1 or T1), from which radiative decay to the ground state mainly occurs. In the radiative process, an excited-state molecule releases energy and returns to the ground state through photon emission. The efficiency of this process is quantified using Einstein's spontaneous emission rate formula: | (1) |
 | ||
| Fig. 1 Schematic illustration of PL processes in ligand-protected gold nanoclusters (left) and the key factors influencing PLQY of ligand-protected gold nanoclusters (right). | ||
Within the framework of Einstein's theory of spontaneous emission, both a larger emission energy Eem and a larger transition dipole moment µ favor an increased radiative decay rate. Meanwhile, excited states can also return to the ground state through nonradiative pathways. The corresponding nonradiative rate constant knr is commonly governed by the energy gap law and decreases exponentially with increasing excited–ground state energy gap, which is often correlated with a higher emission energy Eem. Consequently, increasing Eem not only enhances the radiative transition rate but also suppresses nonradiative decay. Because the equilibrium geometries of the ground and excited states differ, photoexcitation is followed by structural relaxation on the excited-state potential energy surface. This relaxation involves an energetic cost associated with geometric reorganization, which is commonly described by the reorganization energy λ. This relaxation leads to an emission energy Eem that is lower than the vertical excitation energy Eex defined at the ground-state equilibrium geometry. Therefore, for a given electronic level structure, suppressing excited-state structural relaxation can reduce energy loss and increase Eem. Beyond indirect modulation via emission energy, direct control of nonradiative decay channels is also critical. In particular, weakening nonadiabatic coupling between electronic states and reducing electron–vibrational coupling strength can effectively suppress nonradiative processes, thereby lowering knr.
In summary, enhancing the PLQY of ligand-protected gold nanoclusters can be achieved by engineering their electronic structures to increase the emission energy and transition dipole moment, while simultaneously suppressing excited-state structural relaxation and nonradiative decay through ligand engineering or structural rigidification. Such strategies enable larger radiative rates and smaller nonradiative rates, ultimately leading to improved photoluminescence efficiency. On this basis, the following sections will systematically review recent experimental efforts aimed at enhancing the PLQY of ligand-protected gold nanoclusters, with particular emphasis on strategies that promote radiative transitions and suppress nonradiative processes.
2.2 Promoting the efficiency of the radiative process
In the radiative process, an excited-state molecule releases energy and returns to the ground state through photon emission. According to Einstein's theory of spontaneous emission, the radiative rate is mainly governed by two key factors: the emission energy Eem and the transition dipole moment µ. Accordingly, strategies for enhancing the radiative rate can be broadly classified into two aspects: (i) increasing the emission energy of the lowest excited state through electronic structure modulation, and (ii) enhancing the transition dipole moment to accelerate radiative decay.Previous studies have demonstrated that the HOMO–LUMO gap Eg in ligand-protected gold nanoclusters can be effectively tuned through various experimental strategies, including cluster size control,39–41 ligand modification,33,42–44 and metal doping.8,45,46 Among these strategies, metal doping is an effective strategy to enhance the luminescence efficiency by enlarging the Eg, and it has been widely applied in experiments. Takano and co-workers showed that replacing the central atom of the Au13 core with elements lighter than Au (in the order of Au > Pt > Ir in atomic weight) can widen the Eg (Fig. 2A and B).47 Using this principle, they successfully synthesized a series of heterometal-doped clusters, including [RuAu12(dppm)6]2+, [RhAu12(dppm)6]3+, and [IrAu12(dppm)6]3+ (dppm = 1,2-bis(diphenylphosphino)methane). Notably, [RuAu12(dppm)6]2+ exhibits intense room-temperature phosphorescence in degassed CH2Cl2 solution, with a PLQY of 37%.8 Using a similar doping strategy, a series of clusters with the general formula [MAu12(dppe)5Cl2] (M = Au, Pd, Pt, Rh, Ir; dppe = Ph2PCH2CH2PPh2) were also successfully prepared.30 Experimental results showed that heterometal doping substantially enlarges the Eg of the clusters (Fig. 2B), thereby accelerating radiative transitions while suppressing nonradiative decay pathways. As a result, the PLQY of these systems is significantly enhanced (Fig. 2C). Importantly, such doped cluster systems with well-defined atomic structures and systematic spectroscopic data not only validate the physical mechanism of enhancing radiative efficiency through HOMO–LUMO gap engineering, but also provide ideal experimental models and reliable benchmarks for subsequent excited-state electronic-structure calculations using TDDFT and methodological assessment.
 | ||
| Fig. 2 (A) Two-step jellium potentials for doping lower (left) and higher (right) valent atoms into Au13. Reproduced with permission.47 Copyright 2021, American Chemical Society. (B) Plot of the HOMO–LUMO gaps of M@Au12(dppe)5Cl2 (M = Ir, Pt, Au, Rh, Pd). (C) Plot of the PLQYs of M@Au12(dppe)5Cl2 (MAu12) and M@Au12(dppm)6 (MAu12-m) versus the energy of their PL peaks. Reproduced with permission.30 Copyright 2022, Wiley-VCH. | ||
 | ||
| Fig. 3 (A) Isosurface and irreducible representation of the frontier orbitals of Au38S2(S-Adm)20 and Au30(S-Adm)18. Reproduced with permission.49 Copyright 2024, American Chemical Society. (B) Optical absorption spectra of Au42, Au60, Au78, Au96, and Au114; (C) the HOMO and LUMO orbitals and electric transition dipole moment for transitions from the ground state to the excited states of Au42(SCH3)32, Au60(SH)44, Au78(SH)56, and Au96(SH)68. Reproduced with permission.40 Copyright 2024, National Academy of Sciences. (D) Transversal and longitudinal dipole moment transitions of Au76(SR)44. Reproduced with permission.53 Copyright 2016, American Chemical Society. | ||
The HOMO–LUMO gap Eg can be indirectly obtained from experimental techniques such as optical absorption spectroscopy and can be effectively tuned through modulation of cluster size, choice of heterometal dopants, or ligand electronic effects. In contrast, the intensity of an apparent absorption peak in the experimental spectrum may arise from the superposition of multiple excited-state transitions. As a result, it is experimentally challenging to directly correlate the intensity of the longest-wavelength absorption peak with the transition dipole moment of the lowest excited state. Fundamentally, the transition dipole moment is determined by the spatial distribution and symmetry characteristics of the orbitals involved in the electronic transition, which generally require excited-state electronic structure calculations for detailed analysis. This issue becomes particularly important in the context of applications such as bioimaging, where NIR emission is desired and blue-shifting the emission (i.e., increasing the emission energy) is no longer an option. Under such conditions, enhancing the transition dipole moment represents a more viable approach for improving radiative efficiency. Therefore, establishing more general and design-oriented relationships between the cluster structure and transition dipole moment through theoretical calculations is essential for guiding the rational design of highly emissive gold clusters.
2.3 Suppressing nonradiative processes
In addition to radiative transitions, excited-state ligand-protected gold nanoclusters can also undergo nonradiative deactivation pathways, which diminish the PLQY. Consequently, beyond enhancing radiative efficiency, suppressing nonradiative decay channels is another critical strategy for improving the PL performance of gold clusters. In fact, most reported ligand-protected gold nanoclusters still exhibit relatively low PLQY, indicating that nonradiative channels generally dominate the excited-state decay processes.Structurally, ligand-protected gold nanoclusters adopt a core–shell structure consisting of a metallic core surrounded by an organic ligand shell.54 In such systems, various vibrational modes introduced by both the metal core and the ligands can couple with electronic excited states, providing efficient pathways for nonradiative energy dissipation. Recent studies have emphasized strategies like ligand shell rigidification and suppression of surface vibrations to mitigate nonradiative relaxation pathways and consequently enhance PLQY.
In recent years, a series of NHC-protected gold clusters with exceptionally high PLQY have been reported. Luo et al. synthesized chiral NHC-protected Au13 clusters with PLQY as high as 61% (ref. 62) (Fig. 4A). By combining ligand-exchange experiments with theoretical simulations, they revealed that noncovalent inter-ligand interactions, including C–H⋯N, C–H⋯π, and π⋯π interactions, play a crucial role in enhancing the overall rigidity of the ligand shell and suppressing excited-state structural relaxation. Subsequently, Kulkarni et al. reported bidentate NHC-protected Au13 clusters with PLQY reaching 62% (ref. 10) (Fig. 4B), attributing the enhanced performance to increased ligand rigidity and strong interligand interactions. For diphosphine-protected gold nanoclusters, Mutoh and co-workers combined Ir doping with tailor-made diphosphine ligands to synthesize [IrAu12(bbpe)6]3+ (bbpe = bis[benzo[b]phosphindole]ethane), which exhibits an exceptionally high PLQY of 87%.9 Similar ligand-engineering strategies have also been successfully applied to thiolate-protected gold nanoclusters. Enhancing the π⋯π interactions by reducing the methyl group at the end of the thiol ligand, Wang et al. improved the PLQY of the Au52(SR)32 cluster from 3.8% to 18.3% (ref. 34) (Fig. 4C). And this approach was subsequently applied to the Au36(SR)24 cluster, achieving a similar PLQY enhancement63 (Fig. 4D).
 | ||
| Fig. 4 (A) The chiral NHC-stabilized Au13 cluster with high PLQY (61%). Reproduced with permission.62 Copyright 2023, Wiley-VCH. (B) The bis-NHC-protected Au13 clusters with π-extended aromatic systems and high PLQY. Reproduced with permission.10 Copyright 2025, American Chemical Society. (C) The PLQY of Au52(SR)32 with four different aromatic thiolate ligands. Reproduced with permission.34 Copyright 2023, American Chemical Society. (D) Emission spectra of Au36(SR)24 with four different aromatic thiolate ligands. Reproduced with permission.63 Copyright 2024, American Chemical Society. | ||
Collectively, these studies indicate that enhancing interligand interactions to rigidify the ligand shell is an effective strategy for suppressing excited-state structural relaxation and thereby reducing nonradiative decay. However, most ligand-modification approaches reported to date have focused on hydrophobic ligands. Meanwhile, hydrophilic ligands are essential for ligand-protected gold nanoclusters aimed at biological applications,4,18,64,65 and the surrounding aqueous environment plays a decisive role in governing their PL properties. From a theoretical perspective, a faithful description of these aqueous environment effects requires going beyond implicit-solvent treatments of bulk polarity and instead adopting explicit, dynamic multiscale simulations (e.g., QM/MM), coupled with nonadiabatic excited-state dynamics to capture solvent-coupled structural relaxation as well as charge- and energy-transfer processes.66–72 Notably, such multiscale and nonadiabatic approaches have been less frequently applied to ligand-protected gold nanoclusters and merit further development.
 | ||
| Fig. 5 (A) Atomic structure of Au38S2(S-Adm)20 determined from single-crystal diffraction. Reproduced with permission.48 Copyright 2021, American Chemical Society. (B) PLQY of Au42 dissolved in deaerated 2-MeTHF containing DMBA (N,N-dimethylbenzamide) with different concentrations. (C) PL spectra of Au42 dissolved in C2Cl4 and embedded in a polystyrene (PS) film. Reproduced with permission.12 Copyright 2024, American Chemical Society. (D) Schematic diagram of intramolecular cross-linking of ligands by the formation of bis-Schiff linkages on the Au22(SG)18 surface. (E) PL spectra of Au22(SG)18 and PDA-Au22(SG)18 NCs. Reproduced with permission.77 Copyright 2022, Springer Nature. (F) Comparison of the emission spectra of Au16Cu6 and Au22 in CH2Cl2. Reproduced with permission.11 Copyright 2024, American Association for the Advancement of Science. | ||
Au–S staple motion can also be constrained through external molecular environments. By introducing amide molecules to further restrict staple vibrations in Au42(PET)32 clusters, the room-temperature PLQY was increased to 50% and even reached 75% when embedded in a polymer matrix containing amide molecules12 (Fig. 5B and C). In addition to environmental confinement, surface modification engineering can achieve similar effects. Deng et al. employed bis-Schiff base cross-links to restrict the movement of Au–S staples in individual Au22(SG)18 clusters (SG = glutathione), achieving a PLQY of 48% (ref. 77) (Fig. 5D and E). Similar to thiolate-protected systems, alkynyl-protected gold nanoclusters also feature Au–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CR staple motifs. Recently, metal doping within staple motifs provides another effective means of suppressing nonradiative decay in alkynyl-protected gold nanoclusters. Shi et al. doped six Cu atoms into the surface staple units of Au22(tBuPhC
CR staple motifs. Recently, metal doping within staple motifs provides another effective means of suppressing nonradiative decay in alkynyl-protected gold nanoclusters. Shi et al. doped six Cu atoms into the surface staple units of Au22(tBuPhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)18, yielding a more compact Au16Cu6(tBuPhC
C)18, yielding a more compact Au16Cu6(tBuPhC![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C)18 cluster with a near-unity PLQY approaching 100% (ref. 11) (Fig. 5F). From a theoretical perspective, suppressing staple vibrations can be viewed as mitigating excited-state structural relaxation and vibronic coupling at the surface, thereby reducing nonradiative decay.
C)18 cluster with a near-unity PLQY approaching 100% (ref. 11) (Fig. 5F). From a theoretical perspective, suppressing staple vibrations can be viewed as mitigating excited-state structural relaxation and vibronic coupling at the surface, thereby reducing nonradiative decay.
In summary, experimental studies on the photoluminescence of ligand-protected gold nanoclusters in recent years have established a clear and reproducible set of structure–property relationships. Although the specific experimental strategies differ in their practical implementations, their underlying mechanisms can be consistently rationalized in terms of effective modulation of key parameters governing excited-state decay processes. On the one hand, radiative decay rates are enhanced by increasing the emission energy and the transition dipole moment. On the other hand, nonradiative decay is efficiently suppressed by strengthening the rigidity of ligands and surface motifs and by reducing the coupling between vibrational modes and electronic excited states.
It should be emphasized that these experimental observations cannot be fully understood solely on the basis of static structural parameters or ground-state electronic structures. Instead, they critically depend on excited-state electronic structure characteristics, including energy levels, coupling parameters, and the dynamical evolution of excited-state decay processes. Therefore, to achieve a systematic mechanistic understanding and predictive capability for the PLQY of ligand-protected gold nanoclusters, it is essential to introduce theoretical and computational frameworks centered on excited states. Beyond identifying the key factors governing PLQY, theoretical methods are also indispensable for determining emission types, elucidating emission origins, and describing the dynamics of nonradiative relaxation processes. This includes not only calculations of excited-state energies and transition properties based on TDDFT, but also quantitative descriptions of SOC effects, ISC, and vibrational coupling accompanied by structural relaxation. On this basis, the following section will focus on the crucial role of excited-state theoretical approaches in addressing these issues.
3. Theoretical tools for understanding photoluminescence
Although extensive experimental efforts have been devoted to enhancing the PLQY of ligand-protected gold nanoclusters and to elucidating their excited-state properties and emission pathways,18,19,24,31,41 there is still a lack of a comprehensive understanding of their PL mechanisms, which hinders the rational design of clusters with specific emission wavelengths and lifetimes. Specifically, experimental characterization faces three limitations. Firstly, the origin of photoluminescence remains ambiguous due to the inherent core–shell structure of ligand-protected gold nanoclusters. Experimental researchers have proposed several competing explanations, including core-dominated emission, ligand participation, ligand-to-metal charge transfer (LMCT), and metal-to-ligand charge transfer (MLCT). These hypotheses remain under debate because the electronic distribution and transfer processes in the excited states of the clusters cannot be directly observed. Secondly, the strong SOC in gold atoms often results in the coexistence of multiple emission types, including fluorescence, phosphorescence, and TADF. However, the experimentally observed fluorescence and phosphorescence emission bands usually overlap owing to their broad bandwidths and small energy separations, making it challenging to unambiguously distinguish and assign the emission types. Finally, although ultrafast spectroscopy provides powerful means to probe the timescales of excited-state relaxation in these clusters, capturing the atomic-scale details of processes such as structural relaxation (e.g., core distortion and ligand vibration) and charge or energy transfer remains highly challenging.Theoretical calculations can complement these experimental limitations and serve as indispensable tools for elucidating PL mechanisms. In light of the challenges mentioned above, this section systematically reviews two major computational approaches employed to investigate the PL mechanisms of ligand-protected gold nanoclusters and clarifies their relevance to the unresolved experimental issues (as illustrated in Fig. 6). The first category involves electronic structure methods, including density functional theory (DFT), TDDFT, and multiconfigurational methods. By selecting an appropriate theoretical methods, theoretical calculations can reproduce experimental absorption and emission spectra, thereby providing a reliable foundation for subsequent rate-constant evaluations and excited-state property analyses. In particular, analytical tools such as natural transition orbital (NTO) and electron–hole analysis78 enable direct visualization of charge distribution and transfer characteristics in excited states, helping to elucidate the origin of photoluminescence. The second category focuses on the simulation of nonradiative processes, encompassing static rate-constant calculations based on Fermi's golden rule and NA-MD methods that explicitly account for electron–nuclear coupling. By computing and comparing rate constants associated with different decay pathways, one can distinguish emission types, estimate PLQY, and unravel the underlying mechanisms. Moreover, NA-MD simulations provide time-resolved insights into electronic relaxation dynamics, revealing transition times and pathways among excited states. Together, these computational strategies establish a robust methodological framework for in-depth mechanistic analysis of photoluminescence and for the rational design of ligand-protected gold nanoclusters with enhanced PL performance.
 | ||
| Fig. 6 Computational approaches for investigating PL mechanisms in ligand-protected gold nanoclusters. | ||
3.1 Electronic structure methods
The selection of electronic structure methods is pivotal for accurately modeling the electronic configuration and deriving dependable photophysical characteristics, serving as a fundamental prerequisite for valid theoretical interpretations. DFT and TDDFT are widely used to analyze the electronic structure and excited-state properties of ligand-protected gold nanoclusters, owing to their favorable balance between computational efficiency and accuracy.43,55,79,80 However, the reliability of theoretical predictions, such as UV-vis absorption spectra, emission energies, and excited-state lifetimes, strongly depends on the choice of the exchange–correlation functional.80–83Generalized gradient approximation (GGA) functionals, such as BP86 (ref. 84) and PBE,85 are frequently selected in computational studies of ligand-protected gold nanoclusters due to their computational efficiency and acceptable accuracy. However, they tend to underestimate excitation energies, thus limiting their applicability in excited-state properties. Recently, in the computational investigation of X@Cu14 (X = Au, Cl) clusters, Luo et al. assessed the performance of three GGA/meta-GGA functionals (BP86, PBE, and TPSS86,87) and nine hybrid functionals (PBE0,88 B3LYP,89,90 revPBE38,91 TPSSh,92 TPSS0,93 M06, M06-2X,94 CAM-B3LYP,95 and ωB97X-D4 (ref. 96)) for the calculation of fluorescence and phosphorescence emission energies.97 For Au@Cu14, hybrid functionals with high Hartree–Fock exchange (e.g., revPBE38) most accurately replicate the experimental T1 → S0 phosphorescence emission energy (Fig. 7), while for Cl@Cu14, GGA/meta-GGA functionals (BP86, PBE, and TPSS) align more closely with the experiment. Notably, the calculated ΔE(S1–T1) increased with higher HF exchange fractions, resulting in a growing deviation between the calculated fluorescence and phosphorescence emission properties and experimental data. Additionally, the oscillator strengths of fluorescence radiation show an increasing trend with rising HF exchange percentage in the functional. For phosphorescence radiation, except for M06-2X, other functionals showed minimal influence on the oscillator strengths. Qian and co-workers evaluated the performance of various functionals for ligand-protected noble-metal clusters, finding that the proportion of HF exchange predominantly influences simulated spectral features, regardless of the hybrid functional type.98 Benchmarking against experimental spectra indicates that functionals incorporating approximately 10–20% HF exchange yield the most accurate spectral predictions. Furthermore, She et al. examined the fluorescence mechanisms and doping effects in heteroatom-doped M@Au12(dppe)6x−2 clusters, with M representing a 4d or 5d transition metal, and x denoting the nominal charge. They benchmarked several exchange–correlation functionals by comparing the calculated optical absorption and fluorescence spectra of [Au13(dppe)5Cl2]3+ with experimental data.79 They found that the PBE functional markedly underestimates emission energies, while HSE06 (ref. 99) offers the best consistency with experimental observations. Taken together, these studies indicate that no universal exchange–correlation functional has yet been established for ligand-protected gold nanoclusters. The optimal choice remains system-dependent and typically requires benchmarking against experimental spectra or higher-level calculations.
 | ||
| Fig. 7 Calculated T1 → S0 emission energies of Au@Cu14 using functionals with different fractions of Hartree–Fock exchange. | ||
In theoretical calculations, electronic excitation properties (such as local and charge-transfer excitations) can be characterized using NTO or hole–electron analysis. Specifically, such analyses are performed to calculate the excited-state properties of clusters via DFT and TDDFT methods combined with optimal exchange–correlation functionals, to further obtain the orbital compositions and transition contributions of the excited-state electronic structure, and ultimately to quantitatively determine the respective contributions of the metal core and ligand shell in the excited-state transitions, thereby elucidating the origin of photoluminescence. For instance, Weerawardene et al. utilized TDDFT to study Au25(SR)18− clusters and revealed that the emission primarily originates from core-based transitions (i.e., excitations out of superatom P orbitals into the lowest two superatom D orbitals).100
While DFT and TDDFT provide an advantageous compromise between computational efficiency and accuracy, their computational cost escalates rapidly as the size of gold nanoclusters (e.g., beyond Au50) increases, limiting the feasibility of simulating larger systems. Concurrently, the electronic and steric influences of protecting ligands markedly affect the photophysical characteristics of gold nanoclusters.9,34,79 This presents a methodological challenge: simplifying ligand structures to reduce computational demand introduces systematic discrepancies, whereas employing realistic ligand models substantially elevates computational resource requirements. In their study of the dual-emission mechanism of the Au14Cd(S-Adm)12 cluster,101 Havenridge and co-workers observed that the absorption spectra are highly sensitive to ligands. Simplifying ligands, such as substituting Adm groups with hydrogen or methyl groups, may lead to deviations from the authentic photophysical responses of the actual system. They thus applied a combined approach of TDDFT and tight-binding approximation (TDDFT + TB)102 to simulate absorption spectra of Au14Cd(S-Adm)12 protected by full ligands. This approach yielded computational efficiency comparable to that of TDDFT while significantly reducing computational effort. Subsequently, they developed an innovative approximation method, termed TDDFT-aas (time-dependent density functional theory approximate auxiliary s-function), which does not depend on tight-binding parameters.103 Within higher computational efficiency, the calculated absorption spectra of silver and gold nanoparticles using the TDDFT-aas method are in good agreement with the results of TDDFT and TDDFT + TB. Furthermore, the implementation of analytical excited-state gradients within the TDDFT-aas framework facilitates efficient calculation of emission energies for larger nanoclusters.
It is worth noting that TDDFT may struggle to accurately describe potential energy surfaces near conical intersections or state crossings, where multi-configurational methods (e.g., CASSCF) become essential. In 2016, Wu et al. investigated the photoluminescence mechanisms of ultrasmall ligand-protected gold nanoclusters using the CASPT2//CASSCF approach, which enabled highly accurate predictions of absorption and emission wavelengths in excellent agreement with experimental data.104 For example, when applied to the cluster [Au7(dppp)4]3+ (dppp = 1,3-bis(diphenylphosphino)propane), the calculated phosphorescence emission energy was 644 nm, matching the experimental value (642 nm) with a deviation of only 2 nm. However, the high computational cost and the need for careful active-space selection limit the applicability of multi-configurational methods to larger or more complex systems.105
3.2 Simulation of nonradiative processes
The PLQY of ligand-protected gold nanoclusters is determined by the competition between radiative and nonradiative decay pathways, serving as a critical parameter for evaluating their photoluminescence efficiency. However, experimental approaches often face challenges in directly resolving the microscopic mechanisms of nonradiative relaxation processes (such as excited-state structural relaxation and electron-nuclear coupling interactions) and in quantitatively assessing their influence on PLQY. Consequently, theoretical calculations become essential for providing deeper mechanistic insight. To theoretically determine the PLQY value, the primary goal is to calculate the radiative rate constant kr and the nonradiative rate constant knr,106–108 which in turn allows for a clear determination of the reasons behind low PLQY. Notably, early theoretical studies of ligand-protected gold nanoclusters focused primarily on their absorption and emission spectra, oscillator strengths, emission energies, and radiative rate constants.The radiative rate constant kr is predominantly determined using Einstein's spontaneous emission rate expression (eqn (1)), which requires prior calculation of the emission energy and transition dipole moment through methods such as TDDFT. In contrast, modeling nonradiative processes remains a significant challenge in theoretical chemistry and is critical for accurately predicting the PLQY of ligand-protected gold nanoclusters. According to the energy gap law,109 the IC rate constant kIC increases exponentially as the emission energy Eem decreases, which can be expressed as kIC = Ae−αEem. However, since vibronic coupling phenomena between electronic states are not explicitly incorporated, this empirical expression provides only a rough estimate and has limited applicability. Additionally, the strong relativistic effect in gold atoms induces significant SOC, which facilitates ISC from singlet to triplet states. Consequently, ligand-protected gold nanoclusters can exhibit phosphorescence, TADF, or dual emission17–23,50 involving triplet states and the associated ISC and RISC processes. Therefore, a thorough analysis of both ISC and RISC pathways is essential for understanding the luminescence properties and mechanisms of these nanoclusters. Typically, ΔEST and spin–orbit coupling matrix elements (SOCMEs) are utilized to estimate the competition between ISC and RISC via Fermi's golden rule, thereby helping to identify the emission type of ligand-protected gold nanoclusters. Furthermore, direct evaluation of ISC rate constant kISC and RISC rate constant kRISC, and their comparison with kr and IC rate constant kIC, enables comprehensive mapping of excited-state decay pathways and facilitates quantitative prediction of excited-state lifetimes and PLQY.106
However, nonradiative processes, such as IC, ISC, and RISC, involve intricate couplings among electronic and nuclear degrees of freedom,110,111 making accurate theoretical treatment particularly challenging. According to Fermi's golden rule, the nonradiative transition rate constant knr governing transitions between two electronic states can be expressed as follows:
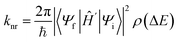 | (2) |
Recent advances by Shuai,107,112 Marian,113,114 Santoro,115 Valiev116 and others have established general rate-constant formulations that incorporate vibrational overlap and the Duschinsky rotation effect, which have been implemented in computational packages for evaluating nonradiative rate constants. For instance, Xie et al.51 applied Lin's formalism117,118 to compute the ISC and RISC rate constants (6.87 × 107 s−1 and 2.87 × 102 s−1, respectively). By combining these with the fluorescence and phosphorescence radiative rate constants (1.53 × 108 s−1 and 8.52 × 103 s−1, respectively), they provided a theoretical explanation of the dual-emission behavior observed in the Au42(PET)32 cluster. Recently, Li et al. computed nonradiative rate constants for Au38S2(SR)20 and Au30(SR)18 with the FCclasses3 program.49 Based on the computed rate constants and the law of mass action, exciton population evolution diagrams were subsequently constructed to analyze the excited-state decay pathways and emission characteristics of these clusters. In addition to rate-constant calculations, analyses of the Huang–Rhys factors and reorganization energies, which were decomposed into contributions from individual vibrational normal modes, provided further insight into how cluster structure influences nonradiative decay processes. However, the excited-state lifetimes and PLQYs predicted by the current methods tend to be significantly lower than experimental values, primarily due to the neglect of anharmonic vibrational effects.108,116,119,120 Moreover, these Fermi's golden rule-based approaches require the computation of excited-state vibrational frequencies, which becomes computationally prohibitive for larger clusters. As a practical alternative to Fermi's golden rule-based approaches, Marcus theory has been employed to estimate the ISC and RISC rate constants:121–124
 | (3) |
Beyond the static excited-state decay models based on Fermi's golden rule discussed above, nonadiabatic molecular dynamics (NA-MD) has emerged as a powerful tool for elucidating the ultrafast excited-state relaxation pathways of ligand-protected gold nanoclusters.125,126 NA-MD simulations can provide detailed information on the electronic relaxation dynamics, including state-to-state transition times and mechanisms. For instance, NA-MD simulations of the Au25(SR)18− cluster revealed that the experimentally observed picosecond-scale processes likely originate from core-to-core transitions rather than the previously proposed core-to-half-ring transitions.102 However, most current NA-MD studies of ligand-protected gold nanoclusters neglect SOC effects, thereby omitting ISC between singlet and triplet states. For ligand-protected gold nanoclusters, reproducing ISC processes that accompany dynamical structural transformations within NA-MD remains highly challenging. This difficulty primarily arises from the need to compute triplet-state energy levels and SOCMEs along nuclear trajectories. Additionally, earlier nonadiabatic simulations of nanoclusters primarily relied on the independent-particle approximation, in which excited states are represented as single-particle excitations constructed from Kohn–Sham orbitals. Within this framework, the state energies and nonadiabatic couplings (NACs) are evaluated using single Slater determinants, thereby neglecting critical carrier–carrier interactions. Recently, Xie and co-workers127 compared simulation results obtained from both single-particle and many-body (MB) treatments in their NA-MD study of Au25(SR)18−. Their findings demonstrated that the inclusion of MB effects significantly enhanced the NAC between excited states, resulting in ultrafast IC from higher excited states on a sub-picosecond timescale, which is consistent with previous experimental observations. Future developments of NA-MD simulation methods in ligand-protected gold nanoclusters need to incorporate relativistic effects, ISC processes, and carrier–carrier interactions to more accurately reproduce the excited-state dynamics of ligand-protected gold nanoclusters.71,72,127–129
4. Mechanistic insights into photoluminescence and excited‐state dynamics
Section 3 has already systematically introduced the applicability logic of core methods such as DFT, TDDFT, and NA-MD in the study of ligand-protected gold nanoclusters. Therefore, this section will adopt a “Computational Method → Mechanism Elucidation” framework to systematically summarize the current theoretical research status concerning the photoluminescence origin, excited-state relaxation dynamics, and emission mechanisms, using case studies from typical cluster systems. This summary will explicitly define the core role of different computational methods in elucidating these mechanisms, thereby providing a valuable reference for subsequent theoretical studies and rational structure design.4.1 Origin of photoluminescence
The core–shell structure of ligand-protected gold nanoclusters, consisting of a metal core and a ligand shell (as illustrated in Fig. 8), gives rise to diverse PL origins, including core-dominated emission, ligand-involved emission, and charge-transfer processes such as LMCT and MLCT. In some systems, structural relaxation and nonradiative processes (IC or ISC) can reshape the character of the excited state, enabling conversion between initially populated charge-transfer states and a core-dominated excitation state before emission. Furthermore, the intricate interactions between the metal core and ligand shell lead to complex excited-state properties and electronic dynamics, which make experimental identification of the PL origin highly challenging. TDDFT, combined with NTO analysis or electron–hole analysis, has thus emerged as a powerful tool for clarifying excited-state electronic behavior. This method serves as an important tool for elucidating the dominant PL origin and understanding the structural factors that modulate emission by enabling direct comparison among different cluster systems. At present, research on the PL origin in ligand-protected gold nanoclusters can be broadly classified into two major categories.(1) Core-dominated excitation: for the majority of ligand-protected gold nanoclusters, PL originates primarily from core-localized electronic excitation. In this case, the emission is governed by electronic transitions within the metal core, whereas the ligands mainly modulate the orbital energy levels through electron-donating or -withdrawing effects and influence core structural relaxation via steric hindrance. This conclusion has been consistently validated across multiple systems using TDDFT calculations. For example, early transient absorption spectroscopy (TAS) studies on the Au25(SR)18− cluster suggested that its PL might arise from ligand–metal, charge-transfer, or semi-ring states.130 To resolve this controversy, Weerawardene et al. employed TDDFT to calculate the excited-state electronic structures of Au25(SR)18− clusters clearly modified with different R groups (R = H, CH3, C2H5, C3H7).100 Their results clearly demonstrated that the emission originates predominantly from core-based transitions rather than charge-transfer or semi-ring states (Fig. 9A), thereby providing a theoretical resolution to the long-standing debate over this system's emission origin. Subsequent TDDFT calculations on systems such as Au38(SR)24,131 Au@Cu14,97 Au22(C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) CR)18,132 Au42(PET)32 (Fig. 9B),52 and Au38S2(S-Adm)20 (ref. 49) further confirmed the dominance of core-localized excitation. Furthermore, Weerawardene et al.100 illustrated the ligand-dependent modulation of emission energy for Au25(SH)18−, Au25(SCH3)18−, and Au25(SCH2CH2CH3)18−, and the fluorescence emission energies were calculated to be 0.829 eV, 0.655 eV, and 0.512 eV, respectively.
CR)18,132 Au42(PET)32 (Fig. 9B),52 and Au38S2(S-Adm)20 (ref. 49) further confirmed the dominance of core-localized excitation. Furthermore, Weerawardene et al.100 illustrated the ligand-dependent modulation of emission energy for Au25(SH)18−, Au25(SCH3)18−, and Au25(SCH2CH2CH3)18−, and the fluorescence emission energies were calculated to be 0.829 eV, 0.655 eV, and 0.512 eV, respectively.
 | ||
| Fig. 9 (A) Kohn–Sham orbitals and orbital energies for the S1 state of Au25(SH)18−. Reproduced with permission.94 Copyright 2016, American Chemical Society. (B) Distributions of hole and electron pairs during the electronic transitions of Au42 and Au60 (blue regions represent holes and green regions represent electrons). Reproduced with permission.53 Copyright 2025, American Chemical Society. (C) Distribution of hole and electron pairs during the T1 → S0 transition at the optimized T1 geometry of Au@Cu14 and Cl@Cu14. Reproduced with permission.91 Copyright 2025, American Chemical Society. | ||
(2) Charge transfer: in contrast, for certain Au(I) clusters and specific ligand-protected systems, the ligands participate directly in electronic transitions, giving rise to LMCT. For instance, studies on the PL properties of Au(I) and Cu(I) clusters protected by NHC ligands,133 which exhibit highly efficient TADF, revealed through NTO analysis that the S1 state in all clusters displayed LMCT characteristics. Similarly, in the Cl-doped Cl@Cu14 cluster, the T1 state exhibits charge-transfer features (Fig. 9C),97 whereas the PL origin in the Au@Cu14 system remains core-localized excitation. Furthermore, He et al. demonstrated that Au doping in AuAg12 and Au3Ag10 induces significant charge-transfer characteristics.134
4.2 SOC and emission pathways
Ligand-protected gold nanoclusters, which comprise several tens of gold atoms or more, exhibit pronounced SOC owing to the heavy-atom effect of gold. Serving as a pivotal mediato, SOC accelerates ISC between singlet and triplet states and, by regulating the RISC rate, directly dictates the emission pathway, namely fluorescence, phosphorescence, or TADF (Fig. 10 left). Early studies often overlooked SOC and the associated ISC/RISC processes, which obscured the interpretation of complex photoluminescence behaviors. Here, we summarize advances in elucidating emission types of ligand-protected gold nanoclusters based on quantitative rate-constant calculations and analyses, with particular emphasis on how SOC is incorporated into the models. By computing ISC and RISC rate constants, we clarify the determinative roles of SOC and the ΔEST in selecting the final emission channel, thereby establishing a “Structural Factors → SOC Effects → Emission Type” regulatory framework (Fig. 10 right). | ||
| Fig. 10 Jablonski diagram of fluorescence, TADF, and phosphorescence emission processes (left), and factors influencing ISC and RISC processes (right). | ||
(1) Differences of emission in isomers: in 2020, the groups of Jin and Wu independently reported two isomeric Au28(SR)20 clusters (i.e., Au28i(SR)20 and Au28ii(SR)20).74,75 Although both possess an identical Au14 core, differences in their ligand-staple motifs led to markedly different PLQYs. An earlier DFT/TDDFT study by Li et al.135 proposed an anti-Kasha S2 fluorescence mechanism but did not incorporate SOC effects or triplet states. More recently, Mazumder et al. combined temperature and oxygen quenching experiments with calculations of ISC and RISC rate constants,20 revealing the emission type and PL mechanism of two Au28(SR)20 isomers (Fig. 11A). Au28i(SR)20 exhibited a small ΔEST (0.126 eV), resulting in comparable T1 → S1 RISC and S1 → T1 ISC rate constants (2.90 × 1010 s−1 and 3.53 × 1010 s−1, respectively), thus showing both phosphorescence and TADF. In contrast, Au28ii(SR)20 has a larger ΔEST (0.240 eV), which suppresses RISC (9.87 × 103 s−1) and leads exclusively to phosphorescence via the ISC pathway. Furthermore, rate constant calculations also indicated that the higher-lying T2 state provides an indirect and efficient nonradiative channel from the excited singlet states to the triplet states, reinforcing the conclusion that SOC directly regulates the emission type.
 | ||
| Fig. 11 Simulated mechanism of photoluminescence for (A) Au28i(SR)20 and Au28ii(SR)20. Reproduced with permission.20 Copyright 2024, American Chemical Society. (B) Au24, Au42, and Au60 clusters. Reproduced with permission.52 Copyright 2025, American Chemical Society. (C) Au5Ag11 and Pt1Ag16. Reproduced with permission.136 Copyright 2022, Wiley-VCH. (D) Au@Cu14 and Cl@Cu14. Reproduced with permission.97 Copyright 2025, American Chemical Society. | ||
(2) Three-state modeling of dual emission mechanism: to rationalize the dual emission of Au42(PET)32, Xie et al. established a three-state kinetic model (S1, T1, and S0) and simulated the ISC and RISC processes.51 Because S1 and T1 share similar localized electronic character, the SOCME between S1 and T1 is relatively weak (46.49 cm−1), consistent with the El-Sayed rule. In addition, the S1 state has a large oscillator strength, yielding a high fluorescence radiative rate constant (1.53 × 108 s−1) that is comparable to the relatively modest S1 → T1 ISC rate constant (6.87 × 107 s−1). Consequently, a fraction of excitons return directly to S0via fluorescence, while the remainder populate T1 through S1 → T1 ISC. From T1, the subsequent phosphorescence rate (8.52 × 103 s−1) substantially exceeds the RISC rate (2.87 × 102 s−1), producing phosphorescence. Overall, the competition among these sequential pathways accounts for the coexistence of fluorescence and phosphorescence in Au42(PET)32.
(1) Heavy-atom effect of core atom doping: a comparison of calculations between Au@Cu14 and Cl@Cu14 clearly illustrates the mechanism of heavy-atom regulation effect (Fig. 11D).97 Owing to the incorporated Au, Au@Cu14 shows a substantially larger SOCME value (2.17 cm−1) compared to that of Cl@Cu14 (0.77 cm−1). This stronger SOC, together with a smaller ΔEST(S1–T2) of 0.03 eV, results in a faster S1 → T2 ISC rate (1.58 × 1010 s−1). As a result, Au@Cu14 exhibits strong phosphorescence (PLQY = 71.3%) via the S1 → T2 → T1 → S0 pathway. In contrast, the weaker SOC in Cl@Cu14 leads to competition between ISC and RISC among S1, T1, and T2, producing the coexistence of TADF and phosphorescence. This comparison clarifies the enhancing role of heavy-atom doping in strengthening SOC.
(2) Alloying effects: to investigate how doping concentration influences emission, He et al. performed theoretical calculations on Ag13, Au13, and the model alloy clusters AuAg12 and Au3Ag10, all of which share the identical icosahedral core structure and superatom electronic configuration (1S21P6).134 For Au13, which exhibits strong NIR emission, the large T2–T1 energy gap (0.55 eV) allows T2 phosphorescence to compete effectively with the T2 → T1 IC process, indicating that its emission primarily originates from the T2 → S0 transition. In contrast to the localized S1 and T1 states found in Au13 and Ag13, the T1 state in AuAg12 and Au3Ag10 exhibits pronounced charge-transfer character induced by Au doping (Fig. 12). This modification enhances the SOCMEs (23.97 cm−1 for AuAg12 and 45.55 cm−1 for Au3Ag10) and reduces the ΔEST relative to Ag13 (about 0.01 eV for AuAg12 and 0.03 eV for Au3Ag10). These effects accelerate ISC and increase the overall PLQY.
 | ||
| Fig. 12 Energy diagrams and images of the hole and electron pairs of (A) Ag13, (B) Au1Ag12, (C) Au3Ag10 and (D) Au13. The Sr index is defined as the full space integration of a function (Sr(r)) describing the overlap between electron and hole distributions, and the D index is the distance between a hole and an the electron center of mass. Reproduced with permission.134 Copyright 2024, Oxford University Press on behalf of the Chinese Academy of Sciences. | ||
Investigation into the analogous dopant effects of various metals on photoluminescence has also been conducted for Au5Ag11 (fluorescence) and Pt1Ag16 (phosphorescence).136 The computational analysis revealed the photoluminescence mechanisms of Au5Ag11 and PtAg16 (Fig. 11C). For Au5Ag11, only the T1 state lies below the S1 state, with a relatively larger ΔEST between S1 and T1 (0.034 eV) than that of Pt1Ag16 (the ΔEST between S1 and T2 is about 0.002 eV), resulting in a small ISC rate constant that favors radiative emission as fluorescence. In contrast, in Pt1Ag16, the S1 and T2 states are nearly degenerate (ΔE ≈ 0.002 eV), which promotes an efficient ISC from S1 to T2. This facilitates an efficient S1 → T2 → T1 → S0 decay pathway, ultimately leading to phosphorescence.136
(3) Size effects: in a theoretical study of one-dimensional ultrathin rod-like ligand-protected gold nanoclusters (Au24, Au42, and Au60), Luo and co-workers combined rate-constant calculations with multistate model analysis (Fig. 11B).52 They showed that the size indirectly controls the emission type by modulating the SOC effect. The smaller Au24 cluster exhibits dual fluorescence, attributable to substantial excited-state structural distortion that creates two minima (S1-MIN1 and S1-MIN2) on the S1 potential energy surface. These minima yield large ΔEST values (0.5 eV and 0.81 eV, respectively), which render ISC negligible. By contrast, for the longer rod-like clusters Au42 and Au60, the large transition dipole moments lead to fluorescence radiative rates (1.13 × 108 s−1 and 1.71 × 108 s−1, respectively) comparable to ISC rates (1.23 × 108 s−1 and 8.68 × 108 s−1, respectively). Together with larger ΔEST values (0.39 eV and 0.57 eV) that suppress RISC, the result is dual emission comprising fluorescence and phosphorescence.
(4) Ligand substituent effects: rate-constant calculations were performed on chiral Au(I)Cu(I) clusters protected by NHC ligands (R/S-py-X and R/S-ql-X, X = Cl, Br, I) to investigate the effect of the halide substituent (Fig. 13).133 The results show that as the halogen atomic mass increases, the ΔEST(S1–T1) narrows systematically (Fig. 13A): the R-ql-Cl cluster (0.294 eV), R-ql-Br cluster (0.193 eV), and R-ql-I cluster (0.116 eV). This narrowing of the ΔEST(S1–T1) corresponds to an increase in the RISC rate constant: R-ql-I (7.05 × 1010 s−1) > R-ql-Br (4.22 × 109 s−1) > R-ql-Cl (5.49 ×108 s−1). Consequently, the R-ql-I cluster exhibits the most efficient TADF owing to its minimal ΔEST(S1–T1).
 | ||
| Fig. 13 (A) Energy diagram of R-ql-Cl, R-ql-Br, and R-ql-I indicating TADF and phosphorescence emission processes. (B) The hole and electron pairs for S1/S0 and T1/S0 transitions were obtained by NTO analysis at the optimized S1 and T1 geometries of R-ql-Cl, R-ql-Br, and R-ql-I (isovalue of 0.02). Au, yellow; Cu, brown; N, dark blue; C, gray; Cl, blue; Br, green; I, light purple; H, white. Reproduced with permission.133 Copyright 2023, Springer Nature. | ||
In summary, recent studies establish that SOC, ISC, and RISC are pivotal in determining the photoluminescence of metal nanoclusters. The interaction between singlet and triplet states, which is quantified by SOCMEs and the ΔEST, governs the competition among fluorescence, phosphorescence, and TADF, thereby shaping the quantum yield and lifetime. Structural factors, including cluster architecture, heteroatom doping, ligand identity and rigidity, and heavy-atom contributions, tune SOCMEs and ΔEST and also modulate nonradiative channels, producing outcomes ranging from single-channel emission to dual emission. These insights underscore the need for multistate, rate-constant-based models that explicitly incorporate triplet states to explain and predict the diverse luminescent behaviors of metal nanoclusters.
4.3 Excited-state relaxation and nonradiative dynamics
Excited-state relaxation dynamics directly determine the cluster's excited-state lifetime and PLQY. These dynamics comprise IC and ISC among excited states, structural relaxation, charge transfer, and electron–hole recombination. Because such processes are not directly observable at the atomic scale, combining NA-MD with DFT/TDDFT enables atomistic elucidation of relaxation pathways, timescales, and nonradiative channels. Current research has successfully revealed how the ligand, metal core structure, and doping effects influence these excited-state relaxation dynamics.By modifying the energy gap and orbital interactions, ligands regulate relaxation rates. TDDFT and NA-MD on Au25(SR)18− showed that internal relaxation among high-lying singlets (S6 → S1) occurs on a sub-picosecond timescale, whereas the final S1 → S0 decay is about two orders of magnitude slower.126 Comparing Au25(SR)18− with different R groups (R = CH3, C2H5, C3H7, MPA, PET) (MPA = mercaptopropionic acid) revealed S1–S6 relaxation times of 2–18 ps, with MPA producing distinctive dynamics due to a narrower LUMO+1–LUMO+2 gap.137 For the phosphine-ligated Au13(dppe)5Cl23+, which shares the Au13 core, NA-MD indicated a slower excited-state decay (0.5–20.3 ps) than in thiolate-protected Au25(SR)18−, consistent with its larger HOMO–LUMO gap.138 TDDFT and NA-MD on Au38(SR)24 further identified low-frequency vibrations, such as the 24.2 cm−1 bending mode, as key channels for electron–phonon coupling and nonradiative decay,139 a behavior also observed in Au18(SH)14.140 Notably, NA-MD simulations on Au25(SR)18− by Xie et al. revealed that low-frequency modes, particularly the breathing and stretching modes of Au(core)–Au(core) and Au(core)–Au(ring), are strongly coupled to the electronic transitions.127 More recent NA-MD work shows that ligands can also modulate carrier lifetimes by altering SOC.129 For example, thiolate-protected Au25(SR)18− relaxed in 99 fs, compared with 132 fs for the diphosphine-protected Au133+ cluster.138
Studies of Au30(SR)18, Au36(SR)24, Au38S2(SR)20, and Au44(SR)28 demonstrate that core stacking adjusts excited-state relaxation.141 Au30(SR)18 with a HCP core exhibits stronger electron–phonon coupling than Au38S2(SR)20 with a body-centered cubic (BCC) core, resulting in a shorter lifetime. Additionally, nonadiabatic relaxation also depends on dopants.142 Yu et al. combined DFT with time-domain NA-MD to examine single-atom doping in MAu24(SR)18 (M = Pd, Pt, Cd, Hg), showing that dopants co-regulate relaxation through changes in the gap, phonon spectrum, and electron–phonon coupling strength.143 In diphosphine-ligated M@Au12 clusters, Ir doping enhances core–shell coupling and suppresses recombination, thereby extending the lifetime and confirming the dynamical impact of doping.144
Overall, real-time NA-MD based on DFT and TDDFT has become indispensable for resolving the excited-state dynamics of ligand-protected gold nanoclusters. These simulations show that larger electronic gaps lengthen lifetimes, stronger electron–phonon coupling accelerates nonradiative decay, and low-frequency vibrations critically mediate energy dissipation. Moreover, subtle structural variations, including isomerism145 and single-atom doping, can markedly reshape relaxation behavior and photophysical properties.
5. Conclusions and outlook
In this review, we summarize recent experimental advances in enhancing the PLQY of ligand-protected gold nanoclusters, with emphasis on strategies that promote radiative decay and suppress nonradiative pathways. Experimentally, increasing the radiative rate constant via HOMO–LUMO gap engineering, achieved through heterometal doping to widen the HOMO–LUMO gap and through enhancement of transition dipole moments, has proven highly effective. Suppressing nonradiative relaxation by rigidifying and constraining ligand motion in core–shell structures has likewise yielded substantial gains in quantum efficiency. Nevertheless, a deeper mechanistic understanding of how transition dipole moments are enhanced is still needed to further improve radiative efficiency. In addition, most ligand-protected gold nanoclusters are soluble only in organic solvents, limiting biomedical applications; consequently, simple and general strategies to convert organo-soluble clusters into water-soluble forms are highly desirable.64Theoretically, DFT/TDDFT methods and NA-MD have clarified photoluminescence origins, complex excited-state dynamics, and emission pathways—fluorescence, phosphorescence, TADF, and dual emission—governed by ΔEST, SOCMEs, and structural relaxation. Even so, improving the computational efficiency of excited-state optimizations and vibrational analyses remains essential, particularly for large clusters.146,147 Computing radiative and nonradiative rate constants has deepened the understanding of competing decay channels, yet accurately predicting PLQYs is still challenging. Looking ahead, multiscale modeling that incorporates environmental effects such as solvent and matrix interactions will provide more realistic descriptions.66–70 Moreover, machine-learning approaches trained on high-level quantum-chemical data promise to accelerate the discovery and screening of ligands, dopants, and structural motifs with desirable photophysical properties.148–154
Author contributions
All authors contributed to writing and editing this article.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The authors acknowledge financial support from NSFC (22373082), the science and technology innovation program of Hunan Province (2023RC1055), the project of innovation team of the ministry of education (IRT_17R90), and the Scientific Research Fund of Hunan Provincial Education Department (22B0154).References
- K. Zheng and J. Xie, Acc. Mater. Res., 2021, 2, 1104–1116 CrossRef CAS.
- Z. Guo, G. Yu, Z. Zhang, Y. Han, G. Guan, W. Yang and M. Y. Han, Adv. Mater., 2023, e2206700 CrossRef PubMed.
- Y. Huang, K. Chen, L. Liu, H. Ma, X. Zhang, K. Tan, Y. Li, Y. Liu, C. Liu, H. Wang and X.-D. Zhang, Small, 2023, 19, 2300145 CrossRef CAS PubMed.
- A. Baghdasaryan and H. Dai, Chem. Rev., 2025, 125, 5195–5227 CrossRef CAS PubMed.
- J. Yang, F. Yang, C. Zhang, X. He and R. Jin, ACS Mater. Lett., 2022, 4, 1279–1296 CrossRef CAS.
- Z. Yang, X. Yang, Y. Guo and H. Kawasaki, ACS Appl. Bio Mater., 2023, 6, 4504–4517 CrossRef CAS PubMed.
- N. Sharma, W. Mohammad, X. Le Guével and A. Shanavas, Chem. Biomed. Imaging, 2024, 2, 462–480 CrossRef CAS PubMed.
- S. Takano, H. Hirai, T. Nakashima, T. Iwasa, T. Taketsugu and T. Tsukuda, J. Am. Chem. Soc., 2021, 143, 10560–10564 CrossRef CAS PubMed.
- K. Mutoh, T. Yahagi, S. Takano, S. Kawakita, T. Iwasa, T. Taketsugu, T. Tsukuda and T. Nakashima, Chem. Sci., 2025, 16, 8240–8246 RSC.
- V. K. Kulkarni, E. L. Albright, E. Zeinizade, E. Steele, J. Chen, L. Ding, S. Malola, S. Takano, K. Harrington, N. Kwon, T. I. Levchenko, M. Nambo, T. Tsukuda, H. Häkkinen, K. Stamplecoskie, G. Zheng and C. M. Crudden, J. Am. Chem. Soc., 2025, 147, 4017–4025 CrossRef CAS PubMed.
- W.-Q. Shi, L. Zeng, R.-L. He, X.-S. Han, Z.-J. Guan, M. Zhou and Q.-M. Wang, Science, 2024, 383, 326–330 CrossRef CAS PubMed.
- L. Luo, Z. Liu, A. Mazumder and R. Jin, J. Am. Chem. Soc., 2024, 146, 27993–27997 CAS.
- X. Wang, Y. Zhong, T. Li, K. Wang, W. Dong, M. Lu, Y. Zhang, Z. Wu, A. Tang and X. Bai, Nat. Commun., 2025, 16, 587 CrossRef CAS PubMed.
- J. S. Yang, Y. J. Zhao, X. M. Li, X. Y. Dong, Y. B. Si, L. Y. Xiao, J. H. Hu, Z. Yu and S. Q. Zang, Angew Chem. Int. Ed. Engl., 2024, 63, e202318030 CrossRef CAS PubMed.
- K. Yoshida, D. Arima and M. Mitsui, J. Phys. Chem. Lett., 2023, 14, 10967–10973 CrossRef CAS PubMed.
- W. Zhang, L. Luo, Z. Liu, F. Zhao, J. Kong, R. Jin, Y. Luo and M. Zhou, Sci. Adv., 2025, 11, eadx2781 CrossRef CAS PubMed.
- Z. Liu, L. Luo, J. Kong, E. Kahng, M. Zhou and R. Jin, Nanoscale, 2024, 16, 7419–7426 RSC.
- Z. Liu, L. Luo and R. Jin, Adv. Mater., 2024, 36, e2309073 CrossRef PubMed.
- L. Luo, Z. Liu, X. Du and R. Jin, Commun. Chem., 2023, 6, 22 CrossRef CAS PubMed.
- A. Mazumder, K. Li, Z. Liu, Y. Wang, Y. Pei, L. A. Peteanu and R. Jin, ACS Nano, 2024, 18, 21534–21543 CrossRef CAS PubMed.
- C. Zhu, Z.-L. Chen, H. Li, L. Lu, X. Kang, J. Xuan and M. Zhu, J. Am. Chem. Soc., 2024, 146, 23212–23220 CrossRef CAS PubMed.
- W.-D. Si, C. Zhang, M. Zhou, W.-D. Tian, Z. Wang, Q. Hu, K.-P. Song, L. Feng, X.-Q. Huang, Z.-Y. Gao, C.-H. Tung and D. Sun, Sci. Adv., 2023, 9, eadg3587 CrossRef CAS PubMed.
- W. D. Si, C. Zhang, M. Zhou, Z. Wang, L. Feng, C. H. Tung and D. Sun, Sci. Adv., 2024, 10, eadm6928 CrossRef CAS PubMed.
- H. Lin, X. Song, O. J. H. Chai, Q. Yao, H. Yang and J. Xie, Adv. Mater., 2024, e2401002 CrossRef PubMed.
- Q. Li, C. J. t. Zeman, B. Kalkan, K. Kirschbaum, C. G. Gianopoulos, A. Parakh, D. Doan, A. C. Lee, J. Kulikowski, G. C. Schatz, G. Shen, M. Kunz and X. W. Gu, Nano Lett., 2023, 23, 132–139 CrossRef CAS PubMed.
- L. Zhang, D. Fan, Y. Shi, S. He, M. Cui, H. Yu and M. Zhu, RSC Adv., 2023, 13, 16034–16038 RSC.
- Y. Zhou, L. Liao, S. Zhuang, Y. Zhao, Z. Gan, W. Gu, J. Li, H. Deng, N. Xia and Z. Wu, Angew. Chem., Int. Ed., 2021, 60, 8668–8672 CrossRef CAS PubMed.
- Y. Wang, C. G. Gianopoulos, Z. Liu, K. Kirschbaum, D. Alfonso, D. R. Kauffman and R. Jin, JACS Au, 2024, 4, 1928–1934 CrossRef CAS PubMed.
- M. Suyama, S. Takano and T. Tsukuda, J. Phys. Chem. C, 2020, 124, 23923–23929 CrossRef CAS.
- H. Hirai, S. Takano, T. Nakashima, T. Iwasa, T. Taketsugu and T. Tsukuda, Angew Chem. Int. Ed. Engl., 2022, 61, e202207290 CrossRef CAS PubMed.
- Z. Liu, M. Zhou, L. Luo, Y. Wang, E. Kahng and R. Jin, J. Am. Chem. Soc., 2023, 145, 19969–19981 CrossRef CAS PubMed.
- B. Zhang, J. Chen, Y. Cao, O. J. H. Chai and J. Xie, Small, 2021, 17, e2004381 CrossRef PubMed.
- A. Kim, C. Zeng, M. Zhou and R. Jin, Part. Part. Syst. Charact., 2017, 34, 1600388 CrossRef.
- Y. Wang, Z. Liu, A. Mazumder, C. G. Gianopoulos, K. Kirschbaum, L. A. Peteanu and R. Jin, J. Am. Chem. Soc., 2023, 145, 26328–26338 CrossRef CAS PubMed.
- S. Kim, H. Kim, C. Lee, I. Park, Y. Kim, D. Moon, J. H. Shim, S. Ryu and S. S. Park, ACS Nano, 2024, 18, 29036–29044 CrossRef CAS PubMed.
- H. Chang, N. S. Karan, K. Shin, M. S. Bootharaju, S. Nah, S. I. Chae, W. Baek, S. Lee, J. Kim, Y. J. Son, T. Kang, G. Ko, S.-H. Kwon and T. Hyeon, J. Am. Chem. Soc., 2020, 143, 326–334 CrossRef PubMed.
- Z. Luo, X. Yuan, Y. Yu, Q. Zhang, D. T. Leong, J. Y. Lee and J. Xie, J. Am. Chem. Soc., 2012, 134, 16662–16670 CrossRef CAS PubMed.
- D. A. Buschmann, H. Hirai and T. Tsukuda, Inorg. Chem. Front., 2024, 11, 6694–6710 RSC.
- X. Du, T. Higaki, H. Ma, X. Zhang, G. Wang, H. Wang and R. Jin, Nanoscale, 2025, 17, 14408–14414 RSC.
- L. Luo, Z. Liu, J. Kong, C. G. Gianopoulos, I. Coburn, K. Kirschbaum, M. Zhou and R. Jin, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2318537121 CrossRef CAS PubMed.
- M. Zhou, T. Higaki, Y. Li, C. Zeng, Q. Li, M. Y. Sfeir and R. Jin, J. Am. Chem. Soc., 2019, 141, 19754–19764 CrossRef CAS PubMed.
- J. Wei, J. F. Halet, S. Kahlal, J. Y. Saillard and A. Munoz-Castro, Inorg. Chem., 2020, 59, 15240–15249 CrossRef CAS PubMed.
- A. Fihey, F. Maurel and A. Perrier, J. Phys. Chem. C, 2014, 118, 4444–4453 CrossRef CAS.
- T. Song, J. Kong, S. Tang, X. Cai, X. Liu, M. Zhou, W. W. Xu, W. Ding and Y. Zhu, Nano Res., 2023, 16, 19754–19764 Search PubMed.
- H. Hirai, S. Takano, T. Nakamura and T. Tsukuda, Inorg. Chem., 2020, 59, 17889–17895 CrossRef CAS PubMed.
- S. Wang, X. Meng, A. Das, T. Li, Y. Song, T. Cao, X. Zhu, M. Zhu and R. Jin, Angew. Chem., Int. Ed., 2014, 53, 2376–2380 CrossRef CAS PubMed.
- S. Takano and T. Tsukuda, J. Am. Chem. Soc., 2021, 143, 1683–1698 CrossRef CAS PubMed.
- Q. Li, C. J. Zeman, G. C. Schatz and X. W. Gu, ACS Nano, 2021, 15, 16095–16105 CrossRef CAS PubMed.
- K. Li, P. Wang and Y. Pei, J. Phys. Chem. Lett., 2024, 15, 9216–9225 CrossRef CAS PubMed.
- L. Luo, Z. Liu, X. Du and R. Jin, J. Am. Chem. Soc., 2022, 144, 19243–19247 CrossRef CAS PubMed.
- X. Y. Xie, K. Q. Cheng, W. K. Chen, W. Li, Q. Li, J. Han, W. H. Fang and G. Cui, J. Phys. Chem. Lett., 2023, 14, 10025–10031 CrossRef CAS PubMed.
- Y. Luo, K. Li, P. Wang and Y. Pei, JACS Au, 2025, 5, 4593–4603 CrossRef CAS PubMed.
- Z. Ma, P. Wang, G. Zhou, J. Tang, H. Li and Y. Pei, J. Phys. Chem. C, 2016, 120, 13739–13748 CrossRef CAS.
- B. Li, L. Kang, Y. Lun, J. Yu, S. Song and Y. Wang, Carbon Energy, 2024, 6, e547 CrossRef CAS.
- Y. Pei, P. Wang, Z. Ma and L. Xiong, Acc. Chem. Res., 2019, 52, 23–33 CrossRef CAS PubMed.
- S. Hossain, D. Hirayama, A. Ikeda, M. Ishimi, S. Funaki, A. Samanta, T. Kawawaki and Y. Negishi, Aggregate, 2022, 4, e255 CrossRef.
- Y. Negishi, S. Hashimoto, A. Ebina, K. Hamada, S. Hossain and T. Kawawaki, Nanoscale, 2020, 12, 8017–8039 RSC.
- R. H. Adnan, J. M. L. Madridejos, A. S. Alotabi, G. F. Metha and G. G. Andersson, Adv. Sci., 2022, 9, e2105692 CrossRef PubMed.
- Z. Lei, X.-K. Wan, S.-F. Yuan, Z.-J. Guan and Q.-M. Wang, Acc. Chem. Res., 2018, 51, 2465–2474 CrossRef CAS PubMed.
- E. L. Albright, T. I. Levchenko, V. K. Kulkarni, A. I. Sullivan, J. F. DeJesus, S. Malola, S. Takano, M. Nambo, K. Stamplecoskie, H. Hakkinen, T. Tsukuda and C. M. Crudden, J. Am. Chem. Soc., 2024, 146, 5759–5780 CrossRef CAS PubMed.
- M. R. Narouz, K. M. Osten, P. J. Unsworth, R. W. Y. Man, K. Salorinne, S. Takano, R. Tomihara, S. Kaappa, S. Malola, C. T. Dinh, J. D. Padmos, K. Ayoo, P. J. Garrett, M. Nambo, J. H. Horton, E. H. Sargent, H. Hakkinen, T. Tsukuda and C. M. Crudden, Nat. Chem., 2019, 11, 419–425 CrossRef CAS PubMed.
- P. Luo, X. J. Zhai, S. Bai, Y. B. Si, X. Y. Dong, Y. F. Han and S. Q. Zang, Angew. Chem., Int. Ed., 2023, 62, e202219017 CrossRef CAS PubMed.
- N. N. Eyyakkandy, A. Afreen, G. Vilangappurath, S. Gratious, K. V. Adarsh and S. Mandal, J. Phys. Chem. C, 2024, 128, 18828–18835 CrossRef CAS.
- A. I. Sullivan, E. A. Steele, S. Takano, E. Zeinizade, J. Chen, S. Malola, K. Siddhant, H. Häkkinen, K. G. Stamplecoskie, T. Tsukuda, G. Zheng and C. M. Crudden, J. Am. Chem. Soc., 2025, 147, 4230–4238 CrossRef CAS PubMed.
- Z. Liu, Y. Wang, W. Ji, X. Ma, C. G. Gianopoulos, S. Calderon, T. Ma, L. Luo, A. Mazumder, K. Kirschbaum, E. C. Dickey, L. A. Peteanu, D. Alfonso and R. Jin, ACS Nano, 2025, 19, 9121–9131 CrossRef CAS PubMed.
- T. Eskelinen and A. J. Karttunen, Inorg. Chem., 2025, 64, 9150–9162 CrossRef CAS PubMed.
- Y.-J. Gao, W.-K. Chen, Z.-R. Wang, W.-H. Fang and G. Cui, Phys. Chem. Chem. Phys., 2018, 20, 24955–24967 RSC.
- Q. Wang, Y.-J. Gao, T.-T. Zhang, J. Han and G. Cui, RSC Adv., 2019, 9, 20786–20795 RSC.
- J. Liu and X. He, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2023, 13, e1650 CAS.
- H. Li, T. Wang, J. Han, Y. Xu, X. Kang, X. Li and M. Zhu, Nat. Commun., 2024, 15, 5351 CrossRef CAS PubMed.
- G. Cui and W. Thiel, J. Chem. Phys., 2014, 141, 124101 CrossRef PubMed.
- Y.-G. Fang, L.-Y. Peng, X.-Y. Liu, W.-H. Fang and G. Cui, Comput. Theor. Chem., 2019, 1155, 90–100 CrossRef CAS.
- K. Pyo, V. D. Thanthirige, K. Kwak, P. Pandurangan, G. Ramakrishna and D. Lee, J. Am. Chem. Soc., 2015, 137, 8244–8250 CrossRef CAS PubMed.
- Y. Chen, M. Zhou, Q. Li, H. Gronlund and R. Jin, Chem. Sci., 2020, 11, 8176–8183 RSC.
- N. Xia, J. Yuan, L. Liao, W. Zhang, J. Li, H. Deng, J. Yang and Z. Wu, J. Am. Chem. Soc., 2020, 142, 12140–12145 CrossRef CAS PubMed.
- J. Dong, Z. Gan, W. Gu, Q. You, Y. Zhao, J. Zha, J. Li, H. Deng, N. Yan and Z. Wu, Angew Chem. Int. Ed. Engl., 2021, 60, 17932–17936 CrossRef CAS PubMed.
- H. Deng, K. Huang, L. Xiu, W. Sun, Q. Yao, X. Fang, X. Huang, H. A. A. Noreldeen, H. Peng, J. Xie and W. Chen, Nat. Commun., 2022, 13, 3381 CrossRef CAS PubMed.
- Z. Liu, T. Lu and Q. Chen, Carbon, 2020, 165, 461–467 CrossRef CAS.
- J. She, W. Pei, S. Zhou and J. Zhao, J. Phys. Chem. Lett., 2022, 13, 5873–5880 CrossRef CAS PubMed.
- P. N. Day, R. Pachter, K. A. Nguyen and R. Jin, J. Phys. Chem. A, 2019, 123, 6472–6481 CrossRef CAS PubMed.
- F. Muniz-Miranda, M. C. Menziani and A. Pedone, J. Phys. Chem. A, 2015, 119, 5088–5098 CrossRef CAS PubMed.
- H. Baek, J. Moon and J. Kim, J. Phys. Chem. A, 2017, 121, 2410–2419 CrossRef CAS PubMed.
- F. Muniz-Miranda, M. C. Menziani and A. Pedone, J. Phys. Chem. C, 2014, 118, 7532–7544 CrossRef CAS.
- J. P. Perdew, Phys. Rev. B:Condens. Matter Mater. Phys., 1986, 33, 8822–8824 CrossRef PubMed.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS PubMed.
- J. Tao, J. P. Perdew, V. N. Staroverov and G. E. Scuseria, Phys. Rev. Lett., 2003, 91, 146401 CrossRef PubMed.
- J. P. Perdew, J. Tao, V. N. Staroverov and G. E. Scuseria, J. Chem. Phys., 2004, 120, 6898–6911 CrossRef CAS PubMed.
- C. Adamo and V. Barone, J. Chem. Phys., 1999, 110, 6158–6170 CrossRef CAS.
- A. D. Becke, J. Chem. Phys., 1992, 96, 2155–2160 CrossRef CAS.
- P. J. Stephens, F. J. Devlin, C. F. Chabalowski and M. J. Frisch, J. Phys. Chem., 1994, 98, 11623–11627 CrossRef CAS.
- Y. Zhang and W. Yang, Phys. Rev. Lett., 1998, 80, 890 CrossRef CAS.
- V. N. Staroverov, G. E. Scuseria, J. Tao and J. P. Perdew, J. Chem. Phys., 2003, 119, 12129–12137 CrossRef CAS.
- S. Grimme, J. Phys. Chem. A, 2005, 109, 3067–3077 CrossRef CAS PubMed.
- Y. Zhao and D. G. Truhlar, Theor. Chem. Acc., 2008, 120, 215–241 Search PubMed.
- T. Yanai, D. P. Tew and N. C. Handy, Chem. Phys. Lett., 2004, 393, 51–57 CrossRef CAS.
- D. Jacquemin, E. A. Perpète, I. Ciofini and C. Adamo, Theor. Chem. Acc., 2011, 128, 127–136 Search PubMed.
- Y. Luo, P. Wang and Y. Pei, J. Phys. Chem. Lett., 2025, 16, 3705–3714 CrossRef CAS PubMed.
- L. Qian, Y. Gao, C. Xu and L. Cheng, J. Phys. Chem. Lett., 2025, 16, 8683–8689 CrossRef CAS PubMed.
- A. V. Krukau, O. A. Vydrov, A. F. Izmaylov and G. E. Scuseria, J. Chem. Phys., 2006, 125, 224106 CrossRef PubMed.
- K. L. D. M. Weerawardene and C. M. Aikens, J. Am. Chem. Soc., 2016, 138, 11202–11210 CrossRef CAS PubMed.
- S. Havenridge and C. M. Aikens, J. Chem. Phys., 2021, 155, 074302 CrossRef CAS PubMed.
- R. Rüger, E. van Lenthe, T. Heine and L. Visscher, J. Chem. Phys., 2016, 144, 184103 CrossRef PubMed.
- Y. Wang, S. Havenridge and C. M. Aikens, J. Chem. Phys., 2024, 161, 024101 CrossRef CAS PubMed.
- L. Wu, W. Fang and X. Chen, Phys. Chem. Chem. Phys., 2016, 18, 17320–17325 RSC.
- V. Veryazov, P. Å. Malmqvist and B. O. Roos, Int. J. Quantum Chem., 2011, 111, 3329–3338 CrossRef CAS.
- K. Veys, M. H. E. Bousquet, D. Jacquemin and D. Escudero, J. Chem. Theory Comput., 2023, 19, 9344–9357 CrossRef CAS PubMed.
- Z. Shuai, Chin. J. Chem., 2020, 38, 1223–1232 CrossRef CAS.
- A. Humeniuk, M. Buzancic, J. Hoche, J. Cerezo, R. Mitric, F. Santoro and V. Bonacic-Koutecky, J. Chem. Phys., 2020, 152, 054107 CrossRef CAS PubMed.
- R. Englman and J. Jortner, Mol. Phys., 1970, 18, 145–164 CrossRef CAS.
- M. Hagai, N. Inai, T. Yasuda, K. J. Fujimoto and T. Yanai, Sci. Adv., 2024, 10, eadk3219 CrossRef CAS PubMed.
- T. J. Penfold, E. Gindensperger, C. Daniel and C. M. Marian, Chem. Rev., 2018, 118, 6975–7025 CrossRef CAS PubMed.
- Q. Peng, Y. Niu, Q. Shi, X. Gao and Z. Shuai, J. Chem. Theory Comput., 2013, 9, 1132–1143 CrossRef CAS PubMed.
- C. M. Marian, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2011, 2, 187–203 Search PubMed.
- M. Etinski, J. Tatchen and C. M. Marian, J. Chem. Phys., 2011, 134, 154105 CrossRef PubMed.
- J. Cerezo and F. Santoro, J. Comput. Chem., 2023, 44, 626–643 CrossRef CAS PubMed.
- R. R. Valiev, B. S. Merzlikin, R. T. Nasibullin, A. Kurtzevitch, V. N. Cherepanov, R. R. Ramazanov, D. Sundholm and T. Kurten, Phys. Chem. Chem. Phys., 2023, 25, 6406–6415 RSC.
- S. H. Lin, W. Z. Xiao and W. Dietz, Phys. Rev. E, 1993, 47, 3698–3706 CrossRef CAS PubMed.
- S. H. Lin, J. Chem. Phys., 1966, 44, 3759–3767 CrossRef.
- C. R. Mulvihill, Y. Georgievskii and S. J. Klippenstein, J. Chem. Phys., 2023, 159, 174104 CrossRef CAS PubMed.
- Y. Wang, J. Ren and Z. Shuai, J. Chem. Phys., 2021, 154, 214109 CrossRef CAS PubMed.
- M. B. Ousmanou, J. Z. Yohana, F. Tchangnwa Nya, A. Malloum and J. Conradie, Mater. Sci. Semicond. Process., 2025, 185, 108894 CrossRef CAS.
- R. A. Marcus, Rev. Mod. Phys., 1993, 65, 599–610 Search PubMed.
- D. Beljonne, Z. Shuai, G. Pourtois and J. L. Bredas, J. Phys. Chem. A, 2001, 105, 3899–3907 CrossRef CAS.
- A. Köhler and H. Bässler, Mater. Sci. Eng., R, 2009, 66, 71–109 CrossRef.
- X. Chen, O. V. Prezhdo, Z. Ma, T. Hou, Z. Guo and Y. Li, Phys. Status Solidi B, 2016, 253, 458–462 CrossRef CAS.
- R. D. Senanayake, A. V. Akimov and C. M. Aikens, J. Phys. Chem. C, 2017, 121, 10653–10662 CrossRef CAS.
- J. Xie, W. Li and Y. Pei, J. Phys. Chem. Lett., 2025, 16, 11259–11266 CrossRef CAS PubMed.
- X. Y. Liu, W. K. Chen, W. H. Fang and G. Cui, J. Chem. Theory Comput., 2023, 19, 8491–8522 CrossRef CAS PubMed.
- Y. Zhou, J. Zhou, X. Yu, W. Pei, S. Zhou and J. Zhao, J. Phys. Chem. Lett., 2025, 16, 8613–8620 CrossRef CAS PubMed.
- S. A. Miller, J. M. Womick, J. F. Parker, R. W. Murray and A. M. Moran, J. Phys. Chem. C, 2009, 113, 9440–9444 CrossRef CAS.
- K. L. D. M. Weerawardene, E. B. Guidez and C. M. Aikens, J. Phys. Chem. C, 2017, 121, 15416–15423 CrossRef CAS.
- S. Havenridge and C. M. Aikens, J. Phys. Chem. A, 2023, 127, 9932–9943 CrossRef CAS PubMed.
- X.-H. Ma, J. Li, P. Luo, J.-H. Hu, Z. Han, X.-Y. Dong, G. Xie and S.-Q. Zang, Nat. Commun., 2023, 14, 4121 CrossRef CAS PubMed.
- W.-M. He, J.-H. Hu, Y.-J. Cui, J. Li, Y.-B. Si, S.-B. Wang, Y.-J. Zhao, Z. Zhou, L.-F. Ma and S.-Q. Zang, Natl. Sci. Rev., 2024, 11, nwae174 CrossRef CAS PubMed.
- J. Li, P. Wang and Y. Pei, J. Phys. Chem. Lett., 2022, 13, 3718–3725 CrossRef CAS PubMed.
- C. Zhu, J. Xin, J. Li, H. Li, X. Kang, Y. Pei and M. Zhu, Angew. Chem., 2022, 134, e202205947 CrossRef.
- R. D. Senanayake and C. M. Aikens, Phys. Chem. Chem. Phys., 2020, 22, 5272–5285 Search PubMed.
- K. L. D. M. Weerawardene, P. Pandeya, M. Zhou, Y. Chen, R. Jin and C. M. Aikens, J. Am. Chem. Soc., 2019, 141, 18715–18726 Search PubMed.
- R. D. Senanayake, E. B. Guidez, A. J. Neukirch, O. V. Prezhdo and C. M. Aikens, J. Phys. Chem. C, 2018, 122, 16380–16388 CrossRef CAS.
- R. D. Senanayake and C. M. Aikens, J. Chem. Phys., 2019, 151, 094702 Search PubMed.
- X. Yu, W. Pei, W. W. Xu, Y. Zhao, Y. Su and J. Zhao, Inorg. Chem., 2023, 62, 20450–20457 CrossRef CAS PubMed.
- P. Pandeya, R. D. Senanayake and C. M. Aikens, J. Chem. Phys., 2021, 154, 184303 CrossRef CAS PubMed.
- X. Yu, Y. Sun, W.-w. Xu, J. Fan, J. Gao, X. Jiang, Y. Su and J. Zhao, Nanoscale Horiz., 2022, 7, 1192–1200 RSC.
- W. Pei, L. Hou, J. Yang, S. Zhou and J. Zhao, Nanoscale, 2024, 16, 14081–14088 RSC.
- Y. Sun, X. Yu, P. Liu, W. Han, W.-w. Xu, Y. Su and J. Zhao, Nanotechnology, 2022, 34, 105701 CrossRef PubMed.
- W. K. Chen, W. H. Fang and G. Cui, J. Chem. Phys., 2023, 158, 044110 Search PubMed.
- J. Du, K. Liao, J. Ma, W. Li and S. Li, J. Chem. Theory Comput., 2022, 18, 7630–7638 Search PubMed.
- C. A. McCandler, A. Pihlajamäki, S. Malola, H. Häkkinen and K. A. Persson, ACS Nano, 2024, 18, 19014–19023 CrossRef CAS PubMed.
- A. Karmakar, Dhananjay and T. Karmakar, Nano Lett., 2025, 25, 10187–10192 CrossRef CAS PubMed.
- T. Chen, J. Li, P. Cai, Q. Yao, Z. Ren, Y. Zhu, S. Khan, J. Xie and X. Wang, Nano Res., 2022, 16, 4188–4196 CrossRef.
- L. Fang, J. Laakso, P. Rinke and X. Chen, J. Chem. Phys., 2024, 160, 094106 CrossRef CAS PubMed.
- A. Pihlajamäki, J. Hämäläinen, J. Linja, P. Nieminen, S. Malola, T. Kärkkäinen and H. Häkkinen, J. Phys. Chem. A, 2020, 124, 4827–4836 CrossRef PubMed.
- V. Tiwari and T. Karmakar, Nano Lett., 2025, 25, 5940–5946 CrossRef CAS PubMed.
- Y. Wang, D. S. N. D. Samarasinghe, H. Deng, B. Liu and C. M. Aikens, J. Chem. Inf. Model., 2025, 65, 3892–3902 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |




