Open Access Article
Open Access ArticleDynamic covalent organic frameworks
Ling
Zhang
 and
Florian
Auras
and
Florian
Auras
 *
*
Faculty of Chemistry and Food Chemistry, TUD Dresden University of Technology, 01217 Dresden, Germany. E-mail: florian.auras@tu-dresden.de
First published on 9th March 2026
Abstract
Covalent organic frameworks (COFs) have emerged as one of the most promising classes of crystalline and porous materials as they offer precise control over structural, catalytic, and optical properties via the design of monomers and their connectivity. While most COFs are rigid materials with fixed geometry, porosity, and properties, recently developed dynamic COFs can change these properties in response to external stimuli such as solvents, vapours, temperature, or light. This research field is rapidly developing and new generations of both 2D and 3D dynamic COFs now offer large and fully reversible changes of unit cell volumes and porosities as well as full control over the expansion/contraction along pre-defined crystal directions. They hold great promise for various applications ranging from economically important uses in gas separation and monomer purification to future technologies including adaptive sensing and spin-optoelectronics. While the design, synthesis and applications of rigid COFs have been the subject of several recent reviews, a comprehensive summary of the current state of the art, challenges and opportunities of dynamic COFs has remained elusive. In this review, we provide an overview of the dynamic COFs reported to date and analyse their structures and underlying design principles. We discuss the different origins of structural flexibility, and summarise first applications and future challenges and opportunities of these intriguing materials.
1. Introduction
Covalent organic frameworks (COFs) are an emerging class of crystalline porous polymers that are constructed by interconnecting rigid or semi-rigid organic building blocks.1–5 Since their initial reports by Yaghi and co-workers,6–8 COFs have sparked an enormous surge in scientific interest due to their modular design, structural predictability, tuneable porosity and wide range of bespoke chemical and electronic functionalities.9–19COFs can be covalently linked in all three dimensions (3D COFs),20–25 or they can be composed of covalently linked layers (2D COFs)26–32 or ribbons (1D COFs)33–36 that are held together by non-covalent interactions (Fig. 1). The framework topology, geometry and porosity are primarily determined by the selection of the building blocks37–41 and can be tuned further via the synthesis conditions employed during the COF polymerisation.42–46
 | ||
| Fig. 1 Representative examples of (a) 2D and (b) 3D COFs.20,58 | ||
The exceptional crystallinity of COFs compared to conventional polymers is primarily due to the monomer design47–51 and the linkage chemistry.52–56 COFs are typically constructed from two building blocks equipped with complementary functional groups for polymerisation, and extensive research has gone into optimising monomer geometries.57–60 The covalent linkages between building blocks are chosen such that they are slightly reversible, allowing bonds to be formed, broken and re-formed during the synthesis.61–63 This provides an effective error correction mechanism, enabling the materials to approach their thermodynamically stable, long-range ordered state.
A wide range of functional monomers has been developed, giving rise to bespoke frameworks with well-defined structural, catalytic, optical and electronic properties. The resulting functional COFs are highly promising for future applications in gas storage and separation, catalysis, energy storage, optoelectronics and sensing.64–78
While most COFs are rigid frameworks with permanent porosity, an increasing number of COFs have now been found to switch their geometry or porosity reversibly when exposed to gases or solvent vapours.79–81 This intriguing behaviour is known from several classes of metal–organic frameworks (MOFs) and the respective materials are referred to as soft porous crystals or dynamic frameworks.82,83 Here, well-engineered flexibility along selected crystal directions enables these materials to switch cooperatively between two or more crystalline phases with the same topology but different unit cell parameters, symmetries and porosities.84,85 Framework dynamics is predominantly triggered by the introduction or extraction of guest molecules, but can also be induced by applied electric fields, temperature or light irradiation.86–92 Potential applications of dynamic frameworks have focused predominantly on selective adsorption and separation,93,94 but these materials could also provide new opportunities for manipulating optoelectronic and spin-electronic properties,95,96 highlighting their potential impact across multiple research fields and disciplines.
In this review article, we aim to provide a comprehensive overview of dynamic COFs, with an emphasis on the COF designs and underlying principles to generate structural flexibility. We will begin with a summary of known dynamic COFs with various 3D and 2D topologies, followed by an in-depth analysis of the factors that govern framework flexibility and dynamics. We will also explore how framework dynamics influences emerging applications, and outline current challenges and opportunities in designing next-generation COFs with adaptive, multifunctional capabilities.
2. Covalent organic frameworks
COFs combine the traits of organic polymers and framework materials. Chemically, COFs are polymers formed through covalent linkages. Structurally, COFs share characteristics with MOFs and other frameworks, i.e., their geometry being primarily determined by the shape of the building blocks. This unique combination opens the door to a plethora of tailor-made materials with customizable structural, chemical, and electronic properties.The general design of COFs as well as their synthesis and applications have been reviewed in a number of recent articles.97–122 We will thus provide only a brief overview before focusing on dynamic COFs.
2.1. Monomer and framework design
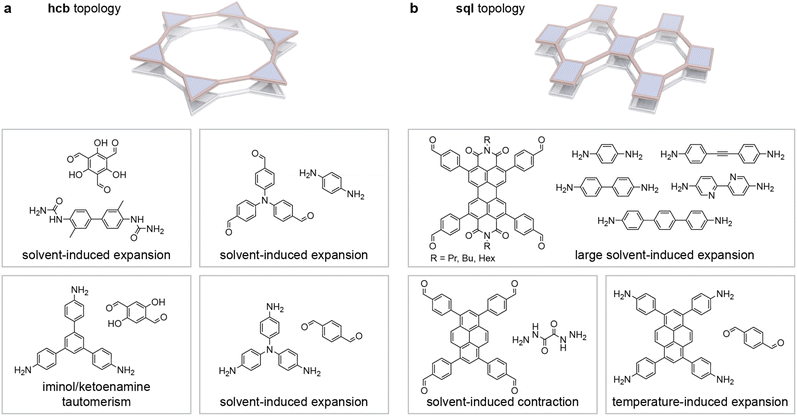
Analogous to DNA and proteins, COFs can be described by a primary, secondary, and tertiary structure. COF monomers are generally selected to be sufficiently rigid with a predictable molecular geometry. Additionally, for generating interconnected networks, at least one of the monomers must be able to form more than two bonds with neighbouring building blocks. These multitopic building blocks are often referred to as “nodes”. 3- and 4-connected nodes are most common,123–132 but also 6-, 8-, and 16-connected variants have been employed successfully.133–137 The other monomer(s) can be ditopic (often referred to as “linkers”) or multitopic.Polymerisation of these building blocks generates the primary structure of the framework. If the monomer geometries and synthesis conditions are chosen appropriately, this process simultaneously creates a periodic secondary structure with a predictable topology. For example, the combination of (pseudo)tetragonal tetratopic and angled ditopic monomers generates 1D ribbons,36 while the combination of the same nodes with linear counterparts generates extended 2D sheets with sql topology (Fig. 1a).27,58 Non-planar multitopic monomers can give rise to 3D secondary structures such as the diamond-like (dia) and quartz (qtz) frameworks generated by (pseudo)tetrahedral units (Fig. 1b)20,44 and pts networks composed of tetrahedral and square planar nodes.24,138
These covalently linked objects form periodic tertiary structures via non-covalent interactions. 2D COF layers stack via electrostatic and dispersive forces. This is often referred to as π-stacking although the attraction between aromatic molecules can be due to a combination of several different mechanisms.139,140 Likewise, the ribbons of 1D COFs are typically π-stacked. Additionally, they are geometrically interlocked or postsynthetically crosslinked along the third dimension.33
3D COFs can in some cases be realised as non-interpenetrated frameworks without a tertiary structure.141,142 However, most 3D COF lattices are highly porous with void fractions often ranging between 50 and 90%. Thus, the formation of interpenetrated lattices is favoured, leading to more densely packed frameworks with altered symmetries and reduced porosities. The number of interpenetrated lattices can range between 2-fold interpenetration for most pts networks138 up to 15-fold for dia COFs comprising large building blocks.143
2.2. Linkage chemistry
The realisation of crystalline covalently interconnected networks imposes substantial challenges to the polymerisation processes that need to be overcome by careful selection and optimisation of the linkage chemistry. Monomers need to attach in the correct orientation and must form all bonds with the correct partners in order to avoid lattice strain and defects in the growing COF crystals.144 High crystallinity thus requires an effective error correction mechanism, i.e., sufficient reversibility to allow bonds to be formed, broken and re-formed during the synthesis. This dynamic covalent chemistry enables the frameworks to approach their thermodynamically stable, long-range ordered geometry rather than becoming trapped in an amorphous state. Suitable building block designs or pre-organisation of the monomers using surfactants or micelles can reduce the number of error correction cycles necessary to obtain high crystallinity.57,145,146 but there is a general trend that more reversible linkages afford more crystalline COFs.52While the first COFs were constructed from boroxines or boronate esters,6,8 imines have evolved into the most widely used COF linkages.20,26 Generated via acid-catalysed condensation of primary amines and aldehydes, they represent an excellent trade-off between crystallinity and stability and are compatible with a large variety of functional monomers. Single-crystalline imine-linked COFs have recently become accessible through modulator strategies, in which monofunctional reagents such as aniline are used to suppress rapid nucleation and polymerisation.22,55
Linkages that provide enhanced stability at the expense of reduced crystallinity include phenazines147–149 and imides.150–152 Enhanced chemical stability can also be obtained via multistep reactions, where the initial reversible condensation provides crystalline pre-organisation of the framework, while subsequent irreversible reactions generate more stable bonds. Typical examples include imidazoles,153 thiazoles154,155 and β-ketoenamines.156,157
Vinylene linkages, formed via base-catalysed Knoevenagel condensation or acid- or base-catalysed aldol condensation,54,158–160 can endow the frameworks with improved chemical stability and enhanced electronic conjugation. For improved crystallinity, linker exchange strategies have opened up viable routes for converting imine-linked COF single crystals into vinylene-linked single-crystalline COFs.56,161
2.3. Dynamic COFs
Dynamic behaviour describes the ability of some frameworks to undergo fully reversible crystal-to-crystal transformations under preservation of the framework topology, i.e., no chemical bonds are formed or broken.Since their structure is predominantly defined by the geometry and connectivity of the building blocks, COFs are almost always porous with void fractions often above 50% and much lower densities than molecular crystals or linear polymers.50,142 These voids do not only create accessible pores, but can also alter the mechanical properties of the frameworks as they allow subunits to rotate, bend and slide without substantially compressing/straining chemical bonds or π–π contacts. Depending on the COF geometry and topology, this can result in structural flexibility at low activation energies.
Dynamic transformations typically involve two or more crystalline phases of different porosities that are commonly referred to as contracted-pore (cp), open-pore (op), and (if applicable) intermediate-pore (ip) phases. Phase transitions can be step-wise or continuous, and are categorised as ‘gate opening’ if an initially contracted framework expands upon guest uptake, or ‘breathing’ if the framework contracts upon initial guest uptake and re-opens when guest loading is increased.83,162
Constructing dynamic COFs requires careful engineering of the framework flexibility along selected crystal directions, as well as optimising the delicate balance between flexibility and stability. Ideally, a well-designed dynamic COF should respond to an external trigger with a pre-defined cooperative geometry change and return to its initial state when the stimulus is removed. At the same time, sufficient robustness is required to ensure that the COF can be toggled between its phases without degradation or loss of long-range order.
Framework dynamics in COFs has so far mostly been induced by the uptake or release of guest molecules. Other triggers that have been employed include temperature changes, external pressure, and the light-induced photoisomerisation of photoswitch moieties.
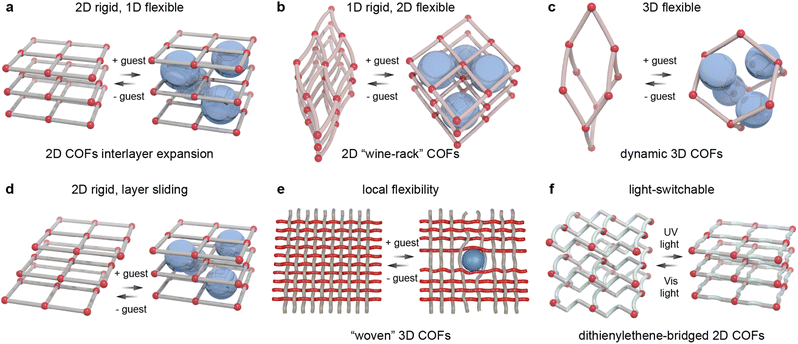
Framework dynamics in COFs can take different forms, depending on the combination of rigid and flexible elements in the COF design. In the simplest case, frameworks are rigid in two dimensions and can expand or contract along the third (Fig. 2a). “Wine rack” type COFs are flexible along two dimensions, but rigid along the third (Fig. 2b). This concept can be realised for instance with sql lattice 2D COFs or their 3D COF counterparts with scu topology.71,81,163
Conversely, most dynamic 3D COFs show framework flexibility along all three dimensions (Fig. 2c). This is for example the case with dia frameworks where expansion along two dimensions upon guest uptake causes contraction along the third, and vice versa.164
In 2D COFs, guest uptake can also result in altered stacking of the COF layers (Fig. 2d).165 COFs can also exhibit local flexibility upon guest uptake without macroscopic changes of the unit cell parameters or porosity (thus strictly speaking not counted as dynamic frameworks) (Fig. 2e). This type of flexibility has been observed for “woven” COFs.166 In addition to guest-induced framework dynamics, some photoswitch-modified COFs can display small unit cell changes upon photoisomerisation (Fig. 2f). For instance, dithienylethene-bridged 2D COFs contract along the stacking direction upon photoisomerisation from the open to the more planar closed configuration.167
3. Dynamic 3D COFs
3D COFs can be very porous materials depending on their framework topology, size and geometry of building blocks, and interpenetration. This provides a wide design space for dynamic frameworks with tuneable host–guest interactions. An overview of the most important 3D COF topologies and the nodes used for constructing dynamic 3D COFs is shown in Fig. 3.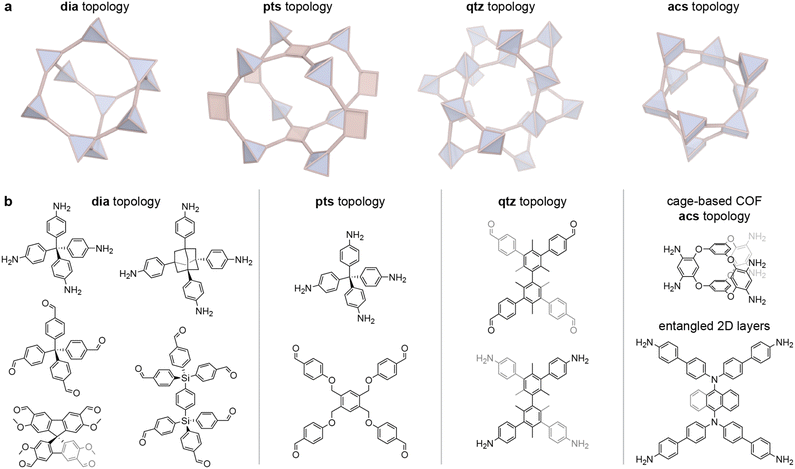 | ||
| Fig. 3 (a) Illustrations of the most important topologies of 3D COFs. (b) Chemical structures of the node-forming tetratopic and 6-connected monomers used for constructing dynamic 3D COFs. | ||
3.1. COFs with dia topology
COFs with dia topology, constructed from tetrahedral and linear or two complementary tetrahedral building blocks such as functionalised tetraphenylmethanes (TPMs), are among the most common 3D COFs. dia COFs can be described as a quasi-infinite lattice of edge-sharing adamantane-like cages (Fig. 3a). Due to the very high void fraction of a single lattice, most dia COFs crystallise with interpenetrated lattices.168The first imine-linked 3D COF (COF-300) was realised via co-condensation of tetrakis(4-aminophenyl)methane (TAM) with terephthalaldehyde.20 This COF was microporous due to a 5-fold interpenetrated structure, but unlike previously reported COFs, the Ar sorption isotherm displayed a pronounced hysteresis and a step around p/p0 = 0.1. This feature was explained by the dynamic response of the framework, specifically a gate-opening process where higher pressure is required to activate the pores. Similar gate-opening was observed for the larger homologue, the biphenyl-linked COF-320.21 Additionally, the structure of this COF was found to be temperature-dependent. At room temperature, the COF had pores with a square cross-section, while at 89 K the framework was significantly contracted with rectangular pores.
Dynamic COFs were first described in more detail by Wang and colleagues, who studied LZU-301, a COF-320 analogue with a 3,3′-bipyridine linker.79 Exposure of this material to solvents such as tetrahydrofuran (THF) led to a shift and splitting of the main reflections in the powder X-ray diffraction (PXRD) patterns, indicating a symmetry-breaking lattice expansion with up to 35% increase in unit cell volume. The observed phase transformations were fully reversible upon drying. The framework dynamics was further investigated using variable temperature 129Xe NMR. At room temperature, the COF adopted its contracted phase with low Xe loading due to the small pore size. Upon cooling to 193 K, an increasing amount of Xe was adsorbed by the COF, resulting in framework expansion. This was accompanied by a downfield shift of the 129Xe NMR signals caused by stronger Xe–Xe interactions of the densely packed Xe atoms.
To date, COF-300 remains one of the most widely studied dynamic 3D COFs. Crystals suitable for single crystal X-ray diffraction (SC-XRD) have been achieved via modulators that control the nucleation and growth.22,55 Depending on the synthesis conditions, this COF can crystallise in two isomers with 5- and 7-fold interpenetration, respectively.168 In the 5-fold interpenetrated variant, the conformation of the TAM nodes is close to ideal tetrahedral geometry. This isomer has a pore diameter of about 8 Å, while the 7-fold interpenetrated COF-300 is elongated along the c-axis and features very narrow pores with only 4 Å diameter.
Upon guest uptake, the 7-fold interpenetrated COF-300 expands and the initially distorted adamantane-like cages assume a less elongated geometry (Fig. 4, right).164 Notably, this guest-loaded COF is very similar to the 5-fold interpenetrated COF-300 isomer discussed above. This study also revealed that the geometry of the solvated framework is almost identical for a broad range of solvents, including THF, toluene, xylenes and acetonitrile.164 Moreover, in situ PXRD conducted during CO2 adsorption and desorption confirmed that the COF undergoes well-defined transitions between crystalline phases, as opposed to swelling that would manifest as gradual shifts of the PXRD reflections.
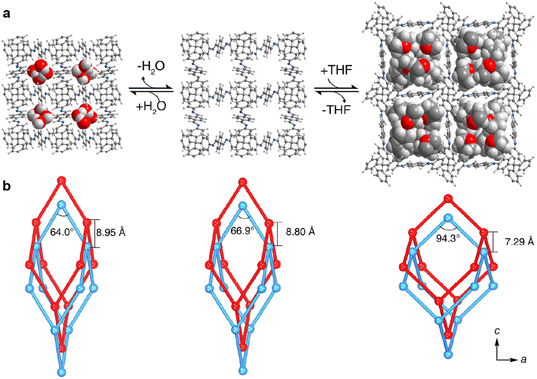 | ||
| Fig. 4 Guest-triggered structural deformations of COF-300. (a) The COF expands upon THF uptake, but contracts when water is adsorbed in the pores due to strong host–guest interactions. (b) Guest-induced distortion of the adamantane-like cages. Reproduced with permission from ref. 164. Copyright 2019 American Chemical Society. | ||
While the uptake of most guest molecules causes dynamic COFs to expand, strongly interacting guests can also trigger a framework contraction. SC-XRD and cryo-electron diffraction tomography studies on 7-fold interpenetrated COF-300 revealed that water molecules can form hydrogen bonds with the imine linkages, causing a substantial contraction of the hydrated framework (Fig. 4, left).22,164,169
Another key aspect for understanding dynamic COFs is the arrangement of the guest molecules within the pores. Zhang and co-workers employed COF-300 single crystals grown via a diffusion gradient transimination protocol to study the adsorption of various guests via SC-XRD and in situ PXRD during solvent vapour adsorption (Fig. 5).86 The adsorption of BuOH caused phase transitions from the guest free tetragonal cp phase to an orthorhombic ip phase at low BuOH loading and a tetragonal op phase at higher BuOH loading. SC-XRD revealed that the phases correspond to the adsorption of two and four BuOH molecules, respectively, per pore and COF repeat unit. A comparison of a variety of linear, branched, and aromatic solvents revealed that the guest molecules assume specific periodic arrangements in the COF pores depending on their shape, size, and interactions with the COF host.
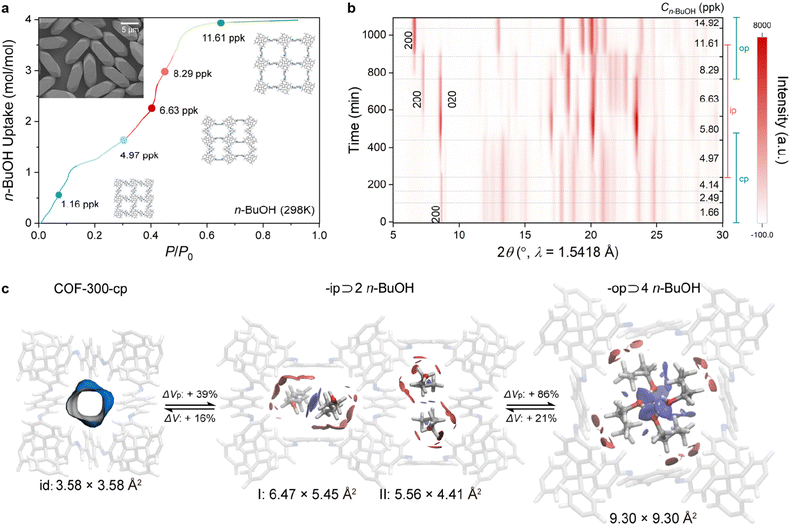 | ||
| Fig. 5 Guest alignment in the pores of COF-300. (a) n-BuOH adsorption isotherm. The colours indicate the cp, ip and op phases, respectively. (b) Corresponding in situ PXRD patterns recorded during BuOH adsorption. (c) Single crystal structures of the three COF phases, highlighting the alignment of the n-BuOH guest molecules. Reproduced from ref. 86. CC-BY-NC-ND 4.0. | ||
Host–guest interactions were further illustrated by a recent study of extended aromatic guests, including anthracene, pyrene and perylene in the pores of COF-300.87 It was found that the COF adapts its symmetry and pore size to match the steric requirements and preferred packing of the guests. These dynamic transformations can result in expansion or contraction of whole COF crystals. Optical dark-field microscopy revealed that exposure of COF-300 single crystals to chloroform triggered observable shape changes within 60–80 s.170
While COF-300 is synthesised from an amine-functionalised tetrahedral node, the structurally related COF-303 is obtained by reacting tetrakis(4-formylphenyl)methane with 1,4-phenylenediamine.55 This COF was found to display similar dynamic behaviour when exposed to solvents, transforming from cp and ip phases with rectangular pore cross-sections to an op phase with square prismatic pores.
Replacing terephthalaldehyde in COF-300 with biphenyl-4,4′-dicarbaldehyde generates the larger COF-320.21 However, this COF can exist in different isomeric forms depending on the synthesis conditions. Solvothermal synthesis at 120 °C resulted in the more rigid COF-320 with permanent porosity, whereas low-temperature synthesis at 65 °C led to the strongly contracted COF-320-A.171 While both frameworks share the same chemical composition, they differ in their crystal structure. Notably, COF-320-A was found to be dynamic when exposed to gases like C2H4, C2H2, or CO2. Structural analysis using continuous rotation electron diffraction (cRED) revealed that the imine bonds in COF-320-A adopt a cis configuration, enabling the rotation and bending of the biphenyl bridge unit at low activation energies. In contrast, the trans configuration of the imines in standard COF-320 restricts this rotation, rendering it more rigid.
Preparation of this COF via a ventilation-vial synthetic protocol produced COF-320-V, which had a similarly contracted geometry in its guest-free cp phase.172 Upon exposure to 1,4-dioxane, this COF expanded substantially with a volume change of 78%. Butane adsorption at 298 K revealed the existence of two ip phases with two and four butane molecules per pore and unit cell, respectively. In the fully expanded op phase, five butane molecules were adsorbed, resulting in a volume expansion of 65% relative to the cp phase.
TAM-based COFs provide ample opportunities for tailoring chemical, structural, and electronic properties via functionalisation of the linear linkers.173 Especially the hydroxy-functionalised aldehyde monomers allow for combining electronic and structural changes. COF-301, synthesised by co-condensation of TAM with 2,5-dihydroxyterephthalaldehyde, was found to change its colour from yellow to light brown when the framework contracts upon desolvation.95,146 When the COF was exposed to water, its colour changed further to dark brown. This effect was explained by an enolimine–ketoenamine tautomerism, where the solvated COF supports the enolimine form, but contraction stabilises the cis-ketoenamine. Similar behaviour was also observed for COF-310, which is constructed from a 2,6-dihydroxynaphthalene-1,5-dicarbaldehyde linker.77
Combining TAM with an anthracene-based linker yielded the dynamic COF-330 with the photoactive acene moieties in an acceptor–donor–acceptor configuration.78 The guest-free COF was highly contracted, causing effective fluorescence quenching due to close contacts between the chromophores. Uptake of acetone vapour led to a step-wise expansion of the COF with three observed phases, corresponding to 2, 16, and 20 solvent molecules per unit cell, respectively. These phase changes were accompanied by fluorescence turn-on due to increased anthracene distances and distinct emission spectra for each phase.
Frameworks with dia topology can also be realised with other tetrahedral building blocks. Fang et al. employed a methoxy-functionalised spiro-bifluorene tetraaldehyde monomer in combination with benzidine-derived linear linkers to construct a series of non-interpenetrated 3D COFs.174 The framework dynamics was controlled via steric hindrance and the rigidity of the benzidine linkers. Unmodified benzidine proved too flexible and the guest-free COF assumed a contracted microporous structure. The 2,2′,6,6′-tetramethylbiphenyl variant, on the other hand, was substantially more rigid and permanently mesoporous. Notably, the 2,2′-dimethylbiphenyl-bridged COF was semi-flexible and showed gate-opening dynamics.
Sun et al. constructed dia frameworks by incorporating the 6-connected 1,4-phenylenebis(tris(4-formylphenyl)silane) in place of two tetrahedral nodes.175 The resulting COFs lost their long-range order upon drying, but their crystallinity was recovered by exposure to THF.
3.2. 3D COFs with pts topology
Frameworks with pts topology are generated by combining tetrahedral with square or rectangular building blocks.138 Depending on the size and steric demands of the monomers, the COFs can be non-interpenetrated or interpenetrated.24,176 Due to the combination of two tetratopic nodes of different geometry, pts COFs tend to be more rigid than dia frameworks and realising dynamic materials requires careful building block design.Wang et al. used the co-condensation of rectangular 1,2,4,5-tetrakis[(4-formylphenoxy)methyl]benzene (TFMB) and tetrahedral TAM to construct the dynamic FCOF-5 with a six-fold interpenetrated pts topology (Fig. 6).80,94 The TFMB moiety is inherently flexible due to its four –CH2–O– single bonds. The guest-free FCOF-5 was highly contracted, and single-crystal cRED data showed that this was due to buckling of the TFMB moiety. Upon solvation with THF, the TFMB expanded to a more planar geometry, allowing the COF to assume an op configuration. Analysis of the gas-triggered dynamics of this COF via in situ PXRD/gas adsorption revealed a single-step gate-opening step upon adsorption of CO2, C2H6, and other C2 and C3 hydrocarbons. Moreover, FCOF-5 was found to possess significantly higher affinity for C3H4 over C3H6, enabling the selective removal of C3H4 from binary gas mixtures.
 | ||
| Fig. 6 Dynamics of FCOF-5 based on a flexible building block. (a) Phase transformation upon exposure to THF vapour. (b) Single-crystal structure of the guest-free COF showing the distorted geometry of the flexible moiety. (c) Structure of the expanded THF-filled COF. Reproduced with permission from ref. 80. Copyright 2021 American Chemical Society. | ||
3.3. 3D COFs with other topologies
While in most cases combining two tetrahedral building blocks yields COFs with dia topology, the hexagonal qtz topology can be accessible depending on synthesis conditions and building block flexibility.44–46 Jiang et al. combined the pseudo-tetrahedral bimesityl tetra-aniline and bimesityl tetra-benzaldehyde to generate USTB-5 with a three-fold interpenetrated qtz topology.72 This COF showed gate-opening type dynamics with contraction upon solvent removal and reversible expansion upon adsorption of solvents or N2. Notably, the expansion was one-dimensional along the helical axis of the COF, accompanied by a contraction along the other two dimensions. While most dia COFs lose their dynamic behaviour upon linkage conversion (see Section 5.2), the imine linkages of USTB-5 could be reduced or oxidised and the framework dynamics was retained. This series of COFs also showed strong temperature-dependent expansion by up to 8% upon heating from 100 to 300 K.Dynamic COFs can also be constructed from higher-connectivity nodes. This was achieved by combining an amine-functionalised molecular cage with 2,5-dihydroxyterephthalaldehyde to produce 3D-CageCOF-1 with an interpenetrated acs topology.25 In the dry state, this COF was contracted due to a rotation of the cage moieties, in conjunction with flexing of the linear bridges. Exposure to N,N-dimethylformamide (DMF) led to straightening of the bridges and reversible expansion of the framework.
In a different approach, a dynamic 3D COF was constructed from entangled 2D layers.177 Here, the design strategy was to induce sufficient steric hindrance to prevent π-stacking of the 2D layers, while allowing two sets of layers to form an entangled structure. This COF showed dynamic framework expansion when exposed to THF vapour due to torsion between the entangled layers as well as multiple conformational changes within the 2D layers.
4. Dynamic 2D COFs
2D COFs are layered structures with covalent linkages within the 2D sheets and non-covalent interlayer interactions (Fig. 7). Framework dynamics can affect both the in-plane and out-of-plane geometries.4.1. 2D COFs with hcb topology
Honeycomb-like (hcb) COFs are typically generated by combination of trigonal and linear or two complementary trigonal building blocks. They are mostly low-density materials with close to eclipsed (AA) stacking (alternating AB and ABC layer sequences are less common) and very high porosities.40,178 While most hcb COFs are rigid, there are a few examples of dynamic COFs.Semi-flexible urea-linked COFs displayed very high crystallinity when immersed in MeCN.179 Unlike the smaller-pore COF-117, the biphenyl-bridged COF-118 retained its crystallinity upon desolvation. Its unit cell, however, contracted from 36.1 Å to 35.3 Å along a and b, and expanded from 3.4 Å to 3.8 Å along the stacking direction (c axis). These changes were fully reversible upon exposure to MeCN or MeOH vapour.
Similar contraction upon solvent removal was also observed for triphenylamine-based COFs.180 A comparison of two structural isomers with different orientation of the imine linkages revealed more pronounced dynamics for the tris(4-formylphenyl)amine-based IISERP-COF2, which was attributed to its slightly different geometry and less polarised imines.
Like the hydroxy-functionalised 3D COFs discussed in Section 3.1, the 2,5-dihydroxyterephthalaldehyde-based TAPB-PDA-OH COF showed reversible iminol-to-ketoenamine tautomerism when exposed to water.181 In this case, however, the re-arrangement of the in-plane linkages led to an expansion of the framework from 28 Å to 32 Å, which, as the COF dried, relaxed to its original position.
4.2. 2D COFs with sql topology
Square lattice (sql) COFs are obtained by combining square or rectangular nodes with linear linkers, or by polymerising two geometrically compatible square or rectangular nodes. Layer stacking in sql COFs follows typically a slightly inclined AA sequence, leading to straight and accessible pores with a rhombic cross-section.58,63While most pyrene-based COFs are rigid frameworks with very high porosity, linker exchange of an imine-linked tetrakis(4-formylphenyl)pyrene/p-phenylenediamine COF using oxalyldihydrazide afforded the semi-flexible SF-COF.182 Unlike the parent framework, this COF contracted by about 2 Å along a and b when immersed in THF.
Dynamic 2D COFs that can be reversibly switched between a non-porous cp phase and porous ip and op phases were realised via a “wine rack” COF design (Fig. 8).81 These COFs consisted of rigid columns of π-stacked PDIs that were interconnected by flexible imine-linked bridges, allowing the frameworks to expand and contract like foldable wine racks. For example, upon exposure to toluene vapour, the buPDI-1P COF achieved up to 40% expansion relative to its cp phase. These substantial geometry changes were found to affect also the electronic coupling between the PDI columns, leading to a switch from h-aggregation in the op phase to ‘null-aggregates’ with monomer-like behaviour in the cp phase COF.
 | ||
| Fig. 8 (a) 2D “wine rack” type dynamic COFs display step-wise pore opening upon guest uptake.163 (b and c) Toluene vapour adsorption and corresponding in situ PXRD patterns of the dynamic buPDI-1P COF.81 Toluene uptake triggers two phase transformations that appear as steps in the sorption isotherms, accompanied by a shift of the 110 reflection from 6.1° (cp phase) to 5.5° (ip phase) to 4.6° (op phase). Copyright 2024 the authors of ref. 81, under exclusive license to Springer Nature. | ||
Isostructural extension of these frameworks using longer biphenyl and terphenyl linkers produced a series of strongly dynamic COFs with very large solvent-induced framework expansion.163 Upon loading with cyclohexane, the PDI-3P COF exhibited an elongation of its a axis from 20.4 Å to 35.7 Å, which translated into a record-high volume change of 85% relative to the cp phase.
While the above COFs show in-plane expansion or contraction upon guest uptake, their layer-to-layer distances remain relatively unaffected. In contrast, polyhedral crystal films of tetraphenylpyrene-based COFs exhibited pronounced temperature-induced and fully reversible expansion of the interlayer distances from 3.76 Å at 50 °C to 3.99 Å at 350 °C.183 Notably, this temperature-induced dynamics was uniaxial, leaving the in-plane geometry unchanged during the temperature cycles.
4.3. Interlayer sliding
A unique type of framework dynamics found in 2D COFs is the solvent-induced layer sliding. Here, the in-plane geometry and layer-to-layer distances remain largely unchanged, but the stacking pattern changes, typically between eclipsed and inclined (slip-stacked) or serrated configurations.Studies of triphenylbenzene-based COFs with various linear linkers revealed that the frameworks can lose their long-range order upon solvent treatment, but this can be largely restored by annealing in supercritical CO2.184
Layer sliding transformations between two crystalline COF phases were first observed for the tetrathiafulvalene-based TTF-DMTA COF.165 In its guest-free state, this material assumed a slip-stacked arrangement with narrow pores. Solvent uptake led to a re-arrangement of the layers to approximate an eclipsed configuration with increased pore diameter.
On the other hand, solvent-induced interlayer shifting has also been reported for examples where the guest-free COF assumes an AA stacking sequence.185,186 Here, solvent or gas uptake causes in these cases a reversible phase transformation to a staggered AB configuration.
4.4. Photoswitchable COFs
Photoswitches, i.e., moieties that can be toggled between two isomers with different geometries by light irradiation could be a facile solution for achieving remote control over COF geometries. While most photoswitch-modified COFs reported to date are rigid frameworks in which the photoswitches can be used to control their porosity,187,188 small changes of the layer spacing have been achieved in some frameworks.Dithienylethenes are capable of reversible photocyclization when exposed to UV light. This functionality was successfully incorporated in a dithienylethene-bridged COF.167 Irradiation with 365 nm light led to cyclisation of the photoswitch from its open to the closed form, concomitant with an increase in electrical conductivity. Since the closed form is more planar, this led to a contraction of the framework along the stacking direction from 6.6 Å to 6.3 Å. This process could be reversed by illumination with 550 nm visible light.
In a different study, an azobenzene-modified COF was found to expand slightly when the photoswitches isomerised to their more bulky Z configuration.189
5. Origins of the framework dynamics
Most COFs are rigid frameworks with permanent porosity, which can adsorb and release guests without changing their crystal structures. The ability of dynamic COFs to adapt their geometries to guest molecules reversibly while fully retaining their crystallinity requires well-engineered framework flexibility. Dynamic COFs must be sufficiently flexible to respond to external triggers with well-defined structural changes, but at the same time robust enough to avoid collapse of the pores and amorphization upon desolvation. This carefully balanced flexibility is usually achieved with a combination of factors, which we will discuss in the following.5.1. Distortion of tetraphenylmethane (TPM) nodes
TPM-based building blocks are the most prominent monomers for constructing dynamic dia net COFs such as COF-300. These frameworks are composed of interconnected adamantane-like cages with 10 TPM moieties located at the corners (Fig. 9a).A key feature of the TPM moiety is its flexibility about the central sp3 carbon atom. The four σ bonds allow not only for a semi-confined rotation of the phenyl rings, but also for considerable distortion of the local geometry.
Studies of COF-300 synthesised under different conditions revealed an interpenetration isomerism with 5-fold and 7-fold interpenetrated lattices.168 Interestingly, the number of interpenetrated lattices had a stark effect on the geometry of the TPM nodes and the adamantane cages. In dia-c5 COF-300, the angles of the adamantane cages were 103.4° and 112.6° for the smaller and larger angle, respectively, which is close to ideal tetrahedral geometry of 109.5°. The adamantane cages of dia-c7 COF-300, on the other hand, are highly distorted to 66.9° and 134.1°, respectively, which has been attributed to decreasing nonbond energy and van der Waals forces.
Single crystal structures of various solvated and hydrated COF-300 samples showed very similar geometries, where the solvent-loaded COFs approximate ideal tetrahedral geometry, whereas the water-containing COFs feature highly distorted adamantane cages (Fig. 4).22,164,169 These observations are not limited to COF-300. The adamantane cages in isostructural frameworks such as the biphenyl-bridged COF-320 or functionalized COFs such as COF-301 exhibit very similar angles.
The angles measured between the nodes of the adamantane cages, however, are influenced by at least two factors, that is the geometry of the TPM moieties as well as the orientation and angles of the interconnecting imine bonds (see discussion below). We analysed the reported crystal structures of 12 TPM-based dynamic COFs and determined the node angles θ and θ′, measured between the outermost carbon atoms of the approximately face-to-face oriented phenyls (Fig. 9b and c).21,22,77–79,86,87,95,164,168,169,172 In most highly symmetric COFs θ = θ′, but there are some exceptions.95,172 In molecular crystals of TPM or TAM, the deviation from ideal tetrahedral geometry is small with θ ≈ 105°.190,191 The TPM moieties in fully solvated or gas-loaded COFs adopt similar geometries with θ ranging from 95° to 106°, depending on the specific COF and guest. In hydrated COFs, on the other hand, the TPM nodes adopt a heavily distorted geometry with θ around 85° for all reported structures. TPMs in the guest-free, activated COFs can adopt two different geometries with θ either being around 85° like in the hydrated COFs, or θ ranging from 95° to 105°, like in the solvated COFs. Interestingly, which of these two TPM configurations is present depends not only on the chemical structure of the respective COFs, but can be synthesis-dependent. In particular for COF-300, both geometries have been found in guest-free single crystals (Fig. 9c, green squares). Synthesis via a ventilation-vial protocol at 65 °C furnished COFs with strongly distorted TPM nodes, while the modulator-aided crystallisation at room temperature resulted in more tetrahedral TPM geometries with θ = 101°.22,164
5.2. Imine bond rotation
In imine-linked dynamic COFs, the conformational changes of the imine bonds provide a key contribution to the framework flexibility and dynamics. Rotation of the –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N– moiety leads to a pedal-like motion that can narrow or widen the COF pores depending on the imine orientation.79 Systematic studies on COF-300 single crystals revealed that the terephthalic diimine bridge units can toggle between anti-periplanar, syn-periplanar, and anticlinal configurations, leading to crystallographic symmetry-breaking along with framework expansion or contraction.86 Throughout the conformational transition, the dominant interaction within the framework changed from node–edge to both node–node and edge–node interactions, demonstrating the flexibility of the moieties within the dynamic framework. In the larger homologue, COF-320, framework contraction is facilitated by a bridge geometry where both imines share almost the same orientation with a small torsion angle of 38° (Fig. 10), leading to buckling of the biphenyl unit.172 The very large solvent-induced expansion of this COF is achieved by rotation of the imines to a torsion angle of 113°, causing the biphenyl to straighten and increasing the node-to-node distances from 21.8 to 23.2 Å.
N– moiety leads to a pedal-like motion that can narrow or widen the COF pores depending on the imine orientation.79 Systematic studies on COF-300 single crystals revealed that the terephthalic diimine bridge units can toggle between anti-periplanar, syn-periplanar, and anticlinal configurations, leading to crystallographic symmetry-breaking along with framework expansion or contraction.86 Throughout the conformational transition, the dominant interaction within the framework changed from node–edge to both node–node and edge–node interactions, demonstrating the flexibility of the moieties within the dynamic framework. In the larger homologue, COF-320, framework contraction is facilitated by a bridge geometry where both imines share almost the same orientation with a small torsion angle of 38° (Fig. 10), leading to buckling of the biphenyl unit.172 The very large solvent-induced expansion of this COF is achieved by rotation of the imines to a torsion angle of 113°, causing the biphenyl to straighten and increasing the node-to-node distances from 21.8 to 23.2 Å.
 | ||
| Fig. 10 (a) Water- and solvent-induced geometry changes of COF-320-V. (b) Guest-induced conformational changes of the imine bonds and biphenyl linker. Reproduced from ref. 172. CC BY-NC-ND 4.0. | ||
Imine rotation requires sufficient space between interpenetrated lattices or COF layers. The interpenetrated lattices of dia net 3D COFs are typically displaced by about 8 Å along the crystallographic c axis, allowing for relatively unimpeded imine rotation. Most 2D COFs, however, are closely π-stacked with layer-to-layer distances around 3.5 Å and only small (if any) lateral offsets below 1.5 Å.40,58 Hence, 2D COFs are usually rigid with permanent porosity.
An exception to this is the recently developed family of PDI-based dynamic 2D COFs, where a substantial lateral offset provides the –Ph–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–(Ph)n–N
N–(Ph)n–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–Ph– (n = 1–3) bridge units with sufficient flexibility to enable the imine rotation (Fig. 11).81,163 Consequently, these COFs are highly dynamic with two-step pore opening and solvent-induced expansions of up to 85% relative to the cp phase.
C–Ph– (n = 1–3) bridge units with sufficient flexibility to enable the imine rotation (Fig. 11).81,163 Consequently, these COFs are highly dynamic with two-step pore opening and solvent-induced expansions of up to 85% relative to the cp phase.
 | ||
| Fig. 11 Lateral offset between COF layers generates dynamic 2D COFs. PDI moieties are strongly slip-stacked, breaking the π–π interactions between adjacent linear bridge units (highlighted in orange). Copyright 2024 the authors of ref. 81, under exclusive license to Springer Nature. | ||
The importance of revolving imine bonds for the framework dynamics is further highlighted by studies where imine bonds were reduced to amines and formamides. It would be expected that the greater flexibility of the secondary amine linkages would translate into highly flexible COFs. However, integrating amine linkages into otherwise rigid COFs did not produce dynamic frameworks.192,193 Imine reduction starting from the dynamic COF-300 furnished the similarly dynamic COF-300-AR.194 But the further conversion into formamides caused a rigidification of the COF despite their otherwise almost identical structure.
In two further studies, reduction or oxidation of the imine linkages both resulted in a rigidification of the respective COFs, which was ascribed to the hindered rotation of the amines or amides.72,170
5.3. Flexible building blocks
Additional flexibility can be introduced via suitably designed flexible building blocks. While pts COFs are typically rigid with permanent porosity, the introduction of a flexible 1,2,4,5-tetra(phenoxymethyl)benzene (TFMB) building block comprising four σ-bonded –CH2–O– moieties yielded the dynamic FCOF-5 (Fig. 6).80,94 Here, the framework dynamics is due to buckling/stretching of the TFMB unit.In a similar approach, Cooper et al. used an organic cage as a triangular prism node to construct a cage-based 3D COF (Fig. 12).25 This COF crystallised with a 2-fold interpenetrated acs topology. Owing to its slightly flexible ether-linked cage node and conformational changes of the imine linkages, it exhibited reversible dynamic behaviour, switching between a contracted small-pore and a large-pore structure upon exposure to solvents such as DMF.
 | ||
| Fig. 12 (a) Chemical structures of the cage monomer with flexible ether moieties (red) and the linear 2,5-dihydroxyterhephthalaldehyde linker. (b) The cage-based 3D-CageCOF-1 can be toggled between its small-pore (sp) and large-pore (lp) structural isomers by loading and removal of DMF, respectively. Adapted from ref. 25. CC-BY. | ||
5.4. Framework topology and symmetry
Besides the precise control of the local or short-range flexibility via the strategies discussed above, the second key component for designing dynamic COFs is the framework topology and symmetry.Chemical bonds possess very high resistance towards compression or elongation, whereas the energy barriers for bending or twisting deformations are much lower. Thus, dynamic COFs require topologies that allow for volume changes solely via bending of flexible moieties without substantial compression or elongation.
In 2D COFs, these criteria are met by the hcb and sql topologies (Fig. 13a), but not by Kagome (kgm) nets,28,57 whose triangular components translate any shear stress into compressive forces. Accordingly, several examples of dynamic 2D COFs with sql and hcb structure have been reported, but no dynamic kgm COFs.
The same considerations also apply to 3D COFs. This field is dominated by dia frameworks, where the main mode of framework dynamics is the distortion of the adamantane cages (Fig. 4). In contrast, the more interconnected pts networks require highly flexible building blocks to exhibit framework dynamics, and the dynamic volume changes are significantly smaller (Fig. 13b, purple). Truss-like topologies such as bor or scu nets have so far only produced rigid frameworks.8,135
The influence of the topology is further illustrated by comparing COFs with isostructural lattices but different interpenetration patterns. Deng et al. constructed COF-308, a COF-300 analogue from TAM and 2,3-dimethoxyterephthalaldehyde.195 The interpenetrated dia lattices of COF-300 share the same orientation and are stacked along the c axis, allowing for synchronous distortion of all adamantane cages. Conversely, COF-308 crystallises with an uncommon [4 + 2] interpenetration, where two lattices were rotated by 180°. This was found to hinder the simultaneous adamantane cage distortion, rendering COF-308 rigid and permanently porous.
Framework dynamics requires the concerted motion of all flexible moieties within a crystal domain, and effective synchronisation could be supported (or hindered) by the framework geometry. In order to derive design rules for highly dynamic COFs, we compared the most dynamic COFs reported to date with different topologies and symmetries.25,72,78–81,86,95,163,164,172,179,181,182 We found that both the majority of examples and the strongest unit-cell changes were associated with two topologies – the 3D dia and the 2D sql lattices (Fig. 13b). It is noteworthy that the node monomers employed in constructing these COFs exhibit only two-fold rotational symmetry (Fig. 3b and 7), and all nodes share the same orientation in the respective frameworks. This reduces the crystal symmetry, resulting in a preferred direction for guest-triggered distortions of flexible moieties and thereby the desired synchronisation. Indeed, the highly dynamic PDI-based sql COFs expand along the a axis, whereas the b axis remains relatively unaffected upon guest uptake. Likewise, the adamantane cages of dia COFs deviate from tetrahedral symmetry (Fig. 4 and 9), causing a preferred contraction along c, accompanied by expansion along the other two dimensions.
In contrast, hcb lattices have three-fold or six-fold rotational symmetry, resulting in multiple possible directions for guest-induced expansion/contraction, which could hinder the concerted geometry changes. This could be one of the reasons for the small guest-induced volume changes of these frameworks despite their very large void fractions. The importance of concerted framework flexibility is further corroborated by the observation that hcb COFs based on flexible building blocks can be rigid and highly porous.196
5.5. Local flexibility of woven and entangled COFs
COFs containing metal complex nodes can form interwoven or entangled networks of low-dimensional threads.23,197 These frameworks can be subsequently demetalated and remetalated to change their mechanical properties and porosities.In the demetallated state, these materials are held together solely by the mechanical interlaced threads and non-covalent interactions. This enables substantial local flexibility and thereby the “adaptive inclusion” of larger guest molecules.166
In another work, Yaghi et al. constructed 3D COFs from interlocking 1D square ribbon.198 In the demetalated form, the individual 1D ribbons are held together only by mechanical interlocking of rings, which allows their collective movement to produce a narrow-pore form. Adsorption of THF vapour resulted in a gradual re-opening of the COF pores and the COF adopted a geometry that was close to the structure of the metalated COF.
6. Applications of dynamic COFs
Dynamic COFs are an intriguing materials platform that can be tailored towards applications in several different fields. Emerging applications as adsorbents or membrane materials utilize their adaptive pore size and shape, whereas future applications as sensor materials make use of geometry-dependent electronic coupling within the frameworks and host–guest interactions.6.1. Adsorption and separation
Dynamic COFs can be high-capacity sponges for the adsorption of pollutants. A COF constructed from an extended adamantane-based tetrahedral node was employed as a high-capacity adsorbent for iodine vapour.199 At 75 °C, 1 g of this COF was able to adsorb 4.7 g I2. The high capacity was ascribed to the formation of a charge transfer complex between the iodine and the pore walls as well as the structural flexibility, allowing the COF to adapt its pore geometry to the iodine guest while fully retaining its connectivity. Extension of this concept to acetylene- and diacetylene-bridged 3D COFs yielded materials with adsorption capacities of up to 0.59 g g−1.200The selective adsorption capacity of dynamic COFs has also been applied for the separation of mixtures of solvents or gases. COF-300 variants with multiple functionalised linkers have been employed in the separation of benzene and cyclohexane vapours.173 In breakthrough experiments benzene was retained by the COFs with selectivity coefficients of up to 8.3 due to its stronger adsorption. COF-300 showed also preferential adsorption of C2H2 over C2H4, suggesting a potential for application in gas separation.194
The technologically relevant separation of trace amounts of C3H4 from C3H6 was successfully demonstrated with the pts net FCOF-5.94 This process is a key step in the production of polymer-grade C3H6 and adsorption-based separation could be an energy-efficient alternative to current technology. Here, the different gate-opening behaviours in response to the two gases despite their similar size and kinetic diameters led to a high adsorption selectivity of 11.6 for C3H4 and thus efficient retention in breakthrough experiments.
6.2. Photocatalysis
The conformational flexibility of dynamic COFs can also be employed to fine-tune their photocatalytic activity. This concept has been illustrated for the photocatalytic oxidation of iodine.201 Single crystals of COF-300 were treated with various solvents to obtain a series of COF geometries with differently twisted diimine linkers, allowing for optimisation of the singlet-triplet energy gap to boost the intersystem crossing. The resulting better charge carrier separation led to improved iodine oxidation performance.6.3. Optoelectronic properties and sensing
Taking advantages of the structural dynamics of 3D COFs, Zhang et al. reported the synthesis of an anthracene-modified dynamic 3D COF with interpenetrated dia topology (Fig. 14).78 In the guest-free state, this COF was strongly contracted, leading to efficient fluorescence quenching of the anthracene moieties. Upon exposure to acetone and dioxane vapours or butane gas, the material showed a step-wise expansion and adaptive guest inclusion, accompanied by fluorescence turn-on. Owing to host–guest interactions, the emission spectra were sensitive to the specific guests and their concentration. | ||
| Fig. 14 (a) Acetone adsorption triggers a fluorescence turn-on in the anthracene-containing COF-330. (b) Evolution of the fluorescence spectra. (c) Correlation between emission intensity and acetone uptake. (d) Synchrotron PXRD data and extracted lattice parameters showing four different COF phases. (e) Evolution of the crystal structure during adaptive inclusion of various numbers of acetone molecules per unit cell. Reproduced from ref. 78. CC-BY-NC-ND 4.0. | ||
Interactions between semiconductor moieties in solids can lead to h-type or j-type aggregation or mixed forms, depending on the intermolecular geometries. While this is generally dictated by the crystal structure, dynamic COFs are capable of substantial geometry changes. Indeed, the switchable geometry of 2D PDI COFs with molecular displacements of up to 10 Å upon toggling between the respective cp to op phases gives rise to geometry-dependent absorption and emission spectra.81 While the op COFs are strongly h-aggregated due to their closely-packed PDI columns, this effect is compensated by interactions between these columns in the cp COFs due to the shortened distances. As a result, the cp phase COFs approximate “null-aggregates” with monomer-like absorption and emission spectra. These findings highlight the potential impact of dynamic frameworks across multiple research fields.
Fang et al. developed a piezochromic 3D COF with donor–acceptor moieties composed of a twisted electron-rich triphenylamine rotor linked to an electron-deficient benzimidazole.202 Due to constrained vibrations, this material showed aggregation-induced emission (AIE) under mild pressure below 1 GPa, followed by aggregation-caused quenching (ACQ) at higher pressures. This was accompanied by a continuous and reversible red-shift of the emission maximum from 500 nm to 687 nm. Similar piezochromic fluorescence behaviour has also been observed for bicarbazole-based 3D COFs.45
Hydroxy-modified dynamic 2D and 3D COFs have been found to exhibit fully reversible colour changes upon water adsorption.77,95,181 This behaviour has been attributed to an enolimine–ketoenamine tautomerism that is facilitated by the structural contraction of the COFs. As this process is selective to water and distinct from the solvatochromic colour changes induced by adsorption of other solvent vapours,16,181 these COFs can act as colorimetric water sensors.
6.4. Mechanochemical actuation
Dynamic COFs could serve as mechanochemical actuators that translate the geometry changes of individual crystallites into macroscopic displacements. This concept was first demonstrated using particles of the flexible FCOF-5 dispersed in a polyvinylidene fluoride (PVDF) matrix.80 The as-prepared film was rolled up due to the contraction of the guest-free COF, but straightened reversibly and repeatably when exposed to THF vapour.This concept was further extended to light-operated actuation by embedding a photoresponsive acylhydrazone-linked COF into a polyethyleneglycol (PEG) matrix.203 Illumination of the COF/PEG film with UV light triggered the E → Z isomerisation of the photoswitches, resulting in a contraction of the COF. Since most light was absorbed close to the illuminated surface, the contraction was stronger on that side, causing the bending of the composite film. This process was fully reversible upon illumination with blue light or treatment at 100 °C.
7. Conclusion and outlook
Dynamic COFs are a fascinating family of soft porous crystals that have emerged from being a chance find to a class of COFs with carefully engineered flexibility and designed structural, catalytic, and electronic properties. While most dynamic COFs to date are based on diamond-like topologies, the design space for soft porous COFs is rapidly expanding to a multitude of 3D and 2D topologies. The discovery of new dynamic COFs so far has predominantly been driven by experimental studies, but future theoretical models could allow for identifying the most promising framework candidates in silico, thus significantly accelerating the materials development. In particular, quantitative metrics such as the energy barriers for expansion/contraction and a detailed understanding of the interactions between framework and guest molecules could provide important guidelines for designing new dynamic COFs. With a rapidly expanding materials basis and the recent insights into their atomic structure and origins of the framework dynamics in hand, new materials can be tailored for diverse applications. Potential impact of the new material ranges from economically important uses in gas separation and monomer purification to future technologies including the topochemical control of chemical reactions, adaptive sensing, spin-optoelectronics, and soft robotics.Author contributions
LZ and FA performed the literature review and data analysis, and wrote the manuscript. FA supervised the project.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results have been included and no new data were generated or analysed as part of this review.Acknowledgements
The authors acknowledge funding from the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – Project No. 525243720.References
- C. S. Diercks and O. M. Yaghi, The atom, the molecule, and the covalent organic framework, Science, 2017, 355, eaal1585 CrossRef.
- J. W. Colson and W. R. Dichtel, Rationally synthesized two-dimensional polymers, Nat. Chem., 2013, 5, 453 CrossRef CAS.
- P. J. Waller, F. Gándara and O. M. Yaghi, Chemistry of Covalent Organic Frameworks, Acc. Chem. Res., 2015, 48, 3053 CrossRef CAS PubMed.
- M. S. Lohse and T. Bein, Covalent Organic Frameworks: Structures, Synthesis, and Applications, Adv. Funct. Mater., 2018, 28, 1705553 CrossRef.
- Y. Song, Q. Sun, B. Aguila and S. Ma, Opportunities of Covalent Organic Frameworks for Advanced Applications, Adv. Sci., 2019, 6, 1801410, DOI:10.1002/advs.201801410.
- A. P. Côte, A. I. Benin, N. W. Ockwig, M. O'Keeffe, A. J. Matzger and O. M. Yaghi, Porous, Crystalline, Covalent Organic Frameworks, Science, 2005, 310, 1166, DOI:10.1126/science.1120411.
- A. P. Côte, H. M. El-Kaderi, H. Furukawa, J. R. Hunt and O. M. Yaghi, Reticular Synthesis of Microporous and Mesoporous 2D Covalent Organic Frameworks, J. Am. Chem. Soc., 2007, 129, 12914 CrossRef.
- H. M. El-Kaderi, J. R. Hunt, J. L. Mendoza-Cortés, A. P. Côté, R. E. Taylor, M. O'Keeffe and O. M. Yaghi, Designed Synthesis of 3D Covalent Organic Frameworks, Science, 2007, 316, 268 CrossRef CAS PubMed.
- C. J. Doonan, D. J. Tranchemontagne, T. G. Glover, J. R. Hunt and O. M. Yaghi, Exceptional ammonia uptake by a covalent organic framework, Nat. Chem., 2010, 2, 235, DOI:10.1038/NCHEM.548.
- E. L. Spitler and W. R. Dichtel, Lewis acid-catalysed formation of two-dimensional phthalocyanine covalent organic frameworks, Nat. Chem., 2010, 2, 672 CrossRef CAS.
- X. Feng, Y. Honsho, O. Saengsawang, L. Liu, L. Wang, A. Saeki, S. Irle, S. Seki, Y. Dong and D. Jiang, An Ambipolar Conducting Covalent Organic Framework with Self-Sorted and Periodic Electron Donor-Acceptor Ordering, Adv. Mater., 2012, 24, 3026 CrossRef CAS PubMed.
- D. N. Bunck and W. R. Dichtel, Internal Functionalization of Three-Dimensional Covalent Organic Frameworks, Angew. Chem., Int. Ed., 2012, 51, 1885 CrossRef CAS PubMed.
- M. Dogru, M. Handloser, F. Auras, T. Kunz, D. Medina, A. Hartschuh, P. Knochel and T. Bein, A Photoconductive Thienothiophene-Based Covalent Organic Framework Showing Charge Transfer Towards Included Fullerene, Angew. Chem., Int. Ed., 2013, 52, 2920, DOI:10.1002/anie.201208514.
- M. Calik, F. Auras, L. M. Salonen, K. Bader, I. Grill, M. Handloser, D. D. Medina, M. Dogru, F. Löbermann, D. Trauner, A. Hartschuh and T. Bein, Extraction of Photogenerated Electrons and Holes from a Covalent Organic Framework Integrated Heterojunction, J. Am. Chem. Soc., 2014, 136, 17802, DOI:10.1021/ja509551m.
- S. Lin, C. S. Diercks, Y.-B. Zhang, N. Kornienko, E. M. Nichols, Y. Zhao, A. R. Paris, D. Kim, P. Yang, O. M. Yaghi and C. J. Chang, Covalent organic frameworks comprising cobalt porphyrins for catalytic CO2 reduction in water, Science, 2015, 349, 1208, DOI:10.1126/science.aac8343.
- L. Ascherl, E. W. Evans, M. Hennemann, D. Di Nuzzo, A. G. Hufnagel, M. Beetz, R. H. Friend, T. Clark, T. Bein and F. Auras, Solvatochromic covalent organic frameworks, Nat. Commun., 2018, 9, 3802, DOI:10.1038/s41467-018-06161-w.
- X. Wang, L. Chen, S. Y. Chong, M. A. Little, Y. Wu, W.-H. Zhu, R. Clowes, Y. Yan, M. A. Zwijnenburg, R. S. Sprick and A. I. Cooper, Sulfone-containing covalent organic frameworks for photocatalytic hydrogen evolution from water, Nat. Chem., 2018, 10, 1180, DOI:10.1038/s41557-018-0141-5.
- Q. Hao, Z.-J. Li, C. Lu, B. Sun, Y.-W. Zhong, L.-J. Wan and D. Wang, Oriented Two-Dimensional Covalent Organic Framework Films for Near-Infrared Electrochromic Application, J. Am. Chem. Soc., 2019, 141, 19831, DOI:10.1021/jacs.9b09956.
- X. Yan, S. Lyu, X.-Q. Xu, W. Chen, P. Shang, Z. Yang, G. Zhang, W. Chen, Y. Wang and L. Chen, Superhydrophilic 2D Covalent Organic Frameworks as Broadband Absorbers for Efficient Solar Steam Generation, Angew. Chem., Int. Ed., 2022, 61, e202201900 CrossRef CAS PubMed.
- F. J. Uribe-Romo, J. R. Hunt, H. Furukawa, C. Klöck, M. O'Keeffe and O. M. Yaghi, A Crystalline Imine-Linked 3-D Porous Covalent Organic Framework, J. Am. Chem. Soc., 2009, 131, 4570, DOI:10.1021/ja8096256.
- Y.-B. Zhang, J. Su, H. Furukawa, Y. Yun, F. Gándara, A. Duong, X. Zou and O. M. Yaghi, Single-Crystal Structure of a Covalent Organic Framework, J. Am. Chem. Soc., 2013, 135, 16336, DOI:10.1021/ja409033p.
- T. Ma, E. A. Kapustin, S. X. Yin, L. Liang, Z. Zhou, J. Niu, L.-H. Li, Y. Wang, J. Su, J. Li, X. Wang, W. D. Wang, W. Wang, J. Sun and O. M. Yaghi, Single-crystal x-ray diffraction structures of covalent organic frameworks, Science, 2018, 361, 48, DOI:10.1126/science.aat7679.
- Y. Liu, Y. Ma, Y. Zhao, X. Sun, F. Gándara, H. Furukawa, Z. Liu, H. Zhu, C. Zhu, K. Suenaga, P. Oleynikov, A. S. Alshammari, X. Zhang, O. Terasaki and O. M. Yaghi, Weaving of organic threads into a crystalline covalent organic framework, Science, 2016, 351, 365, DOI:10.1126/science.aad4011.
- L. Liang, Y. Qiu, W. D. Wang, J. Han, Y. Luo, W. Yu, G.-L. Yin, Z.-P. Wang, L. Zhang, J. Ni, J. Niu, J. Sun, T. Ma and W. Wang, Non-Interpenetrated Single-Crystal Covalent Organic Frameworks, Angew. Chem., Int. Ed., 2020, 59, 17991 CrossRef CAS PubMed.
- Q. Zhu, X. Wang, R. Clowes, P. Cui, L. Chen, M. A. Little and A. I. Cooper, 3D Cage COFs: A Dynamic Three-Dimensional Covalent Organic Framework with High-Connectivity Organic Cage Nodes, J. Am. Chem. Soc., 2020, 142, 16842, DOI:10.1021/jacs.0c07732.
- S. Wan, F. Gándara, A. Asano, H. Furukawa, A. Saeki, S. K. Dey, L. Liao, M. W. Ambrogio, Y. Y. Botros, X. Duan, S. Seki, J. F. Stoddart and O. M. Yaghi, Covalent Organic Frameworks with High Charge Carrier Mobility, Chem. Mater., 2011, 23, 4094 CrossRef CAS.
- X. Chen, N. Huang, J. Gao, H. Xu, F. Xu and D. Jiang, Towards covalent organic frameworks with predesignable and aligned open docking sites, Chem. Commun., 2014, 50, 6161 RSC.
- T.-Y. Zhou, S.-Q. Xu, Q. Wen, Z.-F. Pang and X. Zhao, One-Step Construction of Two Different Kinds of Pores in a 2D Covalent Organic Framework, J. Am. Chem. Soc., 2014, 136, 15885 CrossRef CAS PubMed.
- A. Natraj, W. Ji, J. Xin, I. Castano, D. W. Burke, A. M. Evans, M. J. Strauss, M. Ateia, L. S. Hamachi, N. C. Gianneschi, Z. A. ALOthman, J. Sun, K. Yusuf and W. R. Dichtel, Single-Crystalline Imine-Linked Two-Dimensional Covalent Organic Frameworks Separate Benzene and Cyclohexane Efficiently, J. Am. Chem. Soc., 2022, 144, 19813, DOI:10.1021/jacs.2c07166.
- D. Bessinger, L. Ascherl, F. Auras and T. Bein, Spectrally Switchable Photodetection with Near-Infrared-Absorbing Covalent Organic Frameworks, J. Am. Chem. Soc., 2017, 139, 12035, DOI:10.1021/jacs.7b06599.
- L. Ascherl, E. W. Evans, J. Gorman, S. Orsborne, D. Bessinger, T. Bein, R. H. Friend and F. Auras, Perylene-Based Covalent Organic Frameworks for Acid Vapor Sensing, J. Am. Chem. Soc., 2019, 141, 15693, DOI:10.1021/jacs.9b08079.
- K. Muggli, L. Spies, D. Bessinger, F. Auras and T. Bein, Electrically Conductive Carbazole and Thienoisoindigo-Based COFs Showing Fast and Stable Electrochromism, ACS Nanosci. Au, 2023, 3, 153 CrossRef CAS PubMed.
- H. L. Nguyen, C. Gropp and O. M. Yaghi, Reticulating 1D Ribbons into 2D Covalent Organic Frameworks by Imine and Imide Linkages, J. Am. Chem. Soc., 2020, 142, 2771, DOI:10.1021/jacs.9b13971.
- Y. Hu, S. J. Teat, W. Gong, Z. Zhou, Y. Jin, H. Chen, J. Wu, Y. Cui, T. Jiang, X. Cheng and W. Zhang, Single crystals of mechanically entwined helical covalent polymers, Nat. Chem., 2021, 13, 660, DOI:10.1038/s41557-021-00686-2.
- Z. Chen, K. Wang, Y. Tang, L. Li, X. Hu, M. Han, Z. Guo, H. Zhan and B. Chen, Reticular Synthesis of One-Dimensional Covalent Organic Frameworks with 4-c sql Topology for Enhanced Fluorescence Emission, Angew. Chem., Int. Ed., 2023, 62, e202213268, DOI:10.1002/anie.202213268.
- C. Yang, J. Le, Y. Kuang, Z. Meng, H. Dai, Y. Zhou and H. Qiu, Quasi-One-Dimensional Zigzag Covalent Organic Frameworks for Photocatalytic Hydrogen Evolution from Water, J. Am. Chem. Soc., 2024, 146, 26198, DOI:10.1021/jacs.4c07780.
- E. L. Spitler, J. W. Colson, F. J. Uribe-Romo, A. R. Woll, M. R. Giovino, A. Saldivar and W. R. Dichtel, Lattice Expansion of Highly Oriented 2D Phthalocyanine Covalent Organic Framework Films, Angew. Chem., Int. Ed., 2012, 51, 2623 CrossRef CAS PubMed.
- S. Dalapati, M. Addicoat, S. Jin, T. Sakurai, J. Gao, H. Xu, S. Irle, S. Seki and D. Jiang, Rational design of crystalline supermicroporous covalent organic frameworks with triangular topologies, Nat. Commun., 2015, 6, 7786 CrossRef CAS PubMed.
- H. Li, F. Chen, X. Guan, J. Li, C. Li, B. Tang, V. Valtchev, Y. Yan, S. Qiu and Q. Fang, Three-Dimensional Triptycene-Based Covalent Organic Frameworks with ceq or acs Topology, J. Am. Chem. Soc., 2021, 143, 2654 CrossRef CAS PubMed.
- C. E. Pelkowski, A. Natraj, C. D. Malliakas, D. W. Burke, M. I. Bardot, Z. Wang, H. Li and W. R. Dichtel, Tuning Crystallinity and Stacking of Two-Dimensional Covalent Organic Frameworks through Side-Chain Interactions, J. Am. Chem. Soc., 2023, 145, 21798, DOI:10.1021/jacs.3c03868.
- H. Zheng, J. Ji, Y. Yusran, J. Chang, F. Chen, Z. Wang, J. Suo, J. Zhang, H. Li, D. Zhao, V. Valtchev, S. Qiu and Q. Fang, Three-Dimensional Covalent Organic Frameworks with jcg Topology Based on a Trinodal Strategy, J. Am. Chem. Soc., 2025, 147, 13667, DOI:10.1021/jacs.5c00961.
- Y. Li, L. Guo, Y. Lv, Z. Zhao, Y. Ma, W. Chen, G. Xing, D. Jiang and L. Chen, Polymorphism of 2D Imine Covalent Organic Frameworks, Angew. Chem., Int. Ed., 2021, 60, 5363 CrossRef CAS PubMed.
- B. Gui, J. Xin, Y. Cheng, Y. Zhang, G. Lin, P. Chen, J.-X. Ma, X. Zhou, J. Sun and C. Wang, Crystallization of Dimensional Isomers in Covalent Organic Frameworks, J. Am. Chem. Soc., 2023, 145, 11276, DOI:10.1021/jacs.3c01729.
- Y. Liu, J. Li, J. Lv, Z. Wang, J. Suo, J. Ren, J. Liu, D. Liu, Y. Wang, V. Valtchev, S. Qiu, D. Zhang and Q. Fang, Topological Isomerism in Three-Dimensional Covalent Organic Frameworks, J. Am. Chem. Soc., 2023, 145, 9679, DOI:10.1021/jacs.3c01070.
- J. Fang, X. Yu, Y. Liu, Y. Yusran, Y. Wang, V. Valtchev, S. Qiu, B. Zou and Q. Fang, Piezofluorochromism in Covalent Organic Frameworks: Pressure-Induced Emission Enhancement and Blue-Shifted Emission, Angew. Chem., Int. Ed., 2024, 63, e202409099 CrossRef CAS PubMed.
- X. Wang, Y. Wada, T. Shimada, A. Kosaka, K. Adachi, D. Hashizume, K. Yazawa, H. Uekusa, Y. Shoji, T. Fukushima, M. Kawano and Y. Murakami, Triple Isomerism in 3D Covalent Organic Frameworks, J. Am. Chem. Soc., 2024, 146, 1832, DOI:10.1021/jacs.3c13863.
- Y. Li, Q. Chen, T. Xu, Z. Xie, J. Liu, X. Yu, S. Ma, T. Qin and L. Chen, De Novo Design and Facile Synthesis of 2D Covalent Organic Frameworks: A Two-in-One Strategy, J. Am. Chem. Soc., 2019, 141, 13822, DOI:10.1021/jacs.9b03463.
- W. Hao, D. Chen, Y. Li, Z. Yang, G. Xing, J. Li and L. Chen, Facile Synthesis of Porphyrin Based Covalent Organic Frameworks via an A2B2 Monomer for Highly Efficient Heterogeneous Catalysis, Chem. Mater., 2019, 31, 8100 CrossRef CAS.
- G. Xing, W. Zheng, L. Gao, T. Zhang, X. Wu, S. Fu, X. Song, Z. Zhao, S. Osella, M. Martínez-Abadía, H. I. Wang, J. Cai, A. Mateo-Alonso and L. Chen, Nonplanar Rhombus and Kagome 2D Covalent Organic Frameworks from Distorted Aromatics for Electrical Conduction, J. Am. Chem. Soc., 2022, 144, 5042 CrossRef CAS PubMed.
- Y. Zhao, S. Das, T. Sekine, H. Mabuchi, T. Irie, J. Sakai, D. Wen, W. Zhu, T. Ben and Y. Negishi, Record Ultralarge-Pores, Low Density Three-Dimensional Covalent Organic Framework for Controlled Drug Delivery, Angew. Chem., Int. Ed., 2023, 62, e202300172, DOI:10.1002/anie.202300172.
- Y. Yin, Y. Zhang, X. Zhou, B. Gui, G. Cai, J. Sun and C. Wang, Single-Crystal Three-Dimensional Covalent Organic Framework Constructed from 6-Connected Triangular Prism Node, J. Am. Chem. Soc., 2023, 145, 22329 CrossRef CAS PubMed.
- D. Beaudoin, T. Maris and J. D. Wuest, Constructing monocrystalline covalent organic networks by polymerization, Nat. Chem., 2013, 5, 830 CrossRef CAS PubMed.
- B. J. Smith and W. R. Dichtel, Mechanistic Studies of Two-Dimensional Covalent Organic Frameworks Rapidly Polymerized from Initially Homogenous Conditions, J. Am. Chem. Soc., 2014, 136, 8783, DOI:10.1021/ja5037868.
- E. Jin, M. Asada, Q. Xu, S. Dalapati, M. A. Addicoat, M. A. Brady, H. Xu, T. Nakamura, T. Heine, Q. Chen and D. Jiang, Two-dimensional sp2 carbon–conjugated covalent organic frameworks, Science, 2017, 357, 673, DOI:10.1126/science.aan0202.
- J. Han, J. Feng, J. Kang, J.-M. Chen, X.-Y. Du, S.-Y. Ding, L. Liang and W. Wang, Fast growth of single-crystal covalent organic frameworks for laboratory x-ray diffraction, Science, 2024, 383, 1014, DOI:10.1126/science.adk8680.
- S. Li, S. Xu, E. Lin, T. Wang, H. Yang, J. Han, Y. Zhao, Q. Xue, P. Samorì, Z. Zhang and T. Zhang, Synthesis of single-crystalline sp2-carbon-linked covalent organic frameworks through imine-to-olefin transformation, Nat. Chem., 2025, 17, 226–232, DOI:10.1038/s41557-024-01690-y.
- L. Ascherl, T. Sick, J. T. Margraf, S. H. Lapidus, M. Calik, C. Hettstedt, K. Karaghiosoff, M. Döblinger, T. Clark, K. W. Chapman, F. Auras and T. Bein, Molecular docking sites designed for the generation of highly crystalline covalent organic frameworks, Nat. Chem., 2016, 8, 310, DOI:10.1038/nchem.2444.
- F. Auras, L. Ascherl, A. H. Hakimioun, J. T. Margraf, F. C. Hanusch, S. Reuter, D. Bessinger, M. Döblinger, C. Hettstedt, K. Karaghiosoff, S. Herbert, P. Knochel, T. Clark and T. Bein, Synchronized Offset Stacking: A Concept for Growing Large-Domain and Highly Crystalline 2D Covalent Organic Frameworks, J. Am. Chem. Soc., 2016, 138, 16703, DOI:10.1021/jacs.6b09787.
- Z. Xie, B. Wang, Z. Yang, X. Yang, X. Yu, G. Xing, Y. Zhang and L. Chen, Stable 2D Heteroporous Covalent Organic Frameworks for Efficient Ionic Conduction, Angew. Chem., Int. Ed., 2019, 58, 15742 CrossRef CAS PubMed.
- N. Keller, T. Sick, N. N. Bach, A. Koszalkowski, J. M. Rotter, D. D. Medina and T. Bein, Dibenzochrysene enables tightly controlled docking and stabilizes photoexcited states in dual-pore covalent organic frameworks, Nanoscale, 2019, 11, 23338, 10.1039/C9NR08007D.
- M. Matsumoto, R. R. Dasari, W. Ji, C. H. Feriante, T. C. Parker, S. R. Marder and W. R. Dichtel, Rapid, Low Temperature Formation of Imine-Linked Covalent Organic Frameworks Catalyzed by Metal Triflates, J. Am. Chem. Soc., 2017, 139, 4999, DOI:10.1021/jacs.7b01240.
- R. L. Li, N. C. Flanders, A. M. Evans, W. Ji, I. Castano, L. X. Chen, N. C. Gianneschi and W. R. Dichtel, Controlled growth of imine-linked twodimensional covalent organic framework nanoparticles, Chem. Sci., 2019, 10, 3796 RSC.
- L. Yi, Y. Gao, S. Luo, T. Wang and H. Deng, Structure Evolution of 2D Covalent Organic Frameworks Unveiled by Single-Crystal X-ray Diffraction, J. Am. Chem. Soc., 2024, 146, 19643, DOI:10.1021/jacs.4c05705.
- H. Furukawa and O. M. Yaghi, Storage of Hydrogen, Methane, and Carbon Dioxide in Highly Porous Covalent Organic Frameworks for Clean Energy Applications, J. Am. Chem. Soc., 2009, 131, 8875, DOI:10.1021/ja9015765.
- Z. Zhou, T. Ma, H. Zhang, S. Chheda, H. Li, K. Wang, S. Ehrling, R. Giovine, C. Li, A. H. Alawadhi, M. M. Abduljawad, M. O. Alawad, L. Gagliardi, J. Sauer and O. M. Yaghi, Carbon dioxide capture from open air using covalent organic frameworks, Nature, 2024, 635, 96, DOI:10.1038/s41586-024-08080-x.
- D. B. Shinde, S. Kandambeth, P. Pachfule, R. R. Kumar and R. Banerjee, Bifunctional Covalent Organic Frameworks with Two Dimensional Organocatalytic Micropores, Chem. Commun., 2015, 51, 310, 10.1039/C4CC07104B.
- S.-Y. Ding, J. Gao, Q. Wang, Y. Zhang, W.-G. Song, C.-Y. Su and W. Wang, Construction of Covalent Organic Framework for Catalysis: Pd/COF-LZU1 in Suzuki Miyaura Coupling Reaction, J. Am. Chem. Soc., 2011, 133, 19816 CrossRef CAS PubMed.
- Y. Wu, H. Xu, X. Chen, J. Gao and D. Jiang, A π-electronic covalent organic framework catalyst: π-walls as catalytic beds for Diels–Alder reactions under ambient conditions, Chem. Commun., 2015, 51, 10096, 10.1039/C5CC03457D.
- C. S. Diercks, S. Lin, N. Kornienko, E. A. Kapustin, E. M. Nichols, C. Zhu, Y. Zhao, C. J. Chang and O. M. Yaghi, Reticular Electronic Tuning of Porphyrin Active Sites in Covalent Organic Frameworks for Electrocatalytic Carbon Dioxide Reduction, J. Am. Chem. Soc., 2018, 140, 1116, DOI:10.1021/jacs.7b11940.
- B. Hou, S. Yang, K. Yang, X. Han, X. Tang, Y. Liu, J. Jiang and Y. Cui, Confinement-Driven Enantioselectivity in 3D Porous Chiral Covalent Organic Frameworks, Angew. Chem., Int. Ed., 2021, 60, 6086, DOI:10.1002/anie.202013926.
- W. Liu, L. Gong, Z. Liu, Y. Jin, H. Pan, X. Yang, B. Yu, N. Li, D. Qi, K. Wang, H. Wang and J. Jiang, Conjugated Three-Dimensional High-Connected Covalent Organic Frameworks for Lithium–Sulfur Batteries, J. Am. Chem. Soc., 2022, 144, 17209, DOI:10.1021/jacs.2c07596.
- B. Yu, R.-B. Lin, G. Xu, Z.-H. Fu, H. Wu, W. Zhou, S. Lu, Q.-W. Li, Y. Jin, J.-H. Li, Z. Zhang, H. Wang, Z. Yan, X. Liu, K. Wang, B. Chen and J. Jiang, Linkage conversions in single-crystalline covalent organic frameworks, Nat. Chem., 2024, 16, 114, DOI:10.1038/s41557-023-01334-7.
- D. Bessinger, K. Muggli, M. Beetz, F. Auras and T. Bein, Fast-Switching Vis-IR Electrochromic Covalent Organic Frameworks, J. Am. Chem. Soc., 2021, 143, 7351, DOI:10.1021/jacs.0c12392.
- N. Keller, M. Calik, D. Sharapa, H. R. Soni, P. M. Zehetmaier, S. Rager, F. Auras, A. C. Jakowetz, A. Görling, T. Clark and T. Bein, Enforcing Extended Porphyrin J-Aggregate Stacking in Covalent Organic Frameworks, J. Am. Chem. Soc., 2018, 140, 16544, DOI:10.1021/jacs.8b08088.
- L. Gilmanova, V. Bon, L. Shupletsov, D. Pohl, M. Rauche, E. Brunner and S. Kaskel, Chemically Stable Carbazole-Based Imine Covalent Organic Frameworks with Acidochromic Response for Humidity Control Applications, J. Am. Chem. Soc., 2021, 143, 18368 CrossRef CAS PubMed.
- M. R. Rao, Y. Fang, S. De Feyter and D. F. Perepichka, Conjugated Covalent Organic Frameworks via Michael Addition-Elimination, J. Am. Chem. Soc., 2017, 139, 2421 CrossRef CAS PubMed.
- X. Yao, Y. Zhang, Y. Qiu, W. Jiang, H. Chen, T. Zeng, L. Wei, S. Jiang, Y. Zhao, Y. Ma and Y.-B. Zhang, A Phototautomeric 3D Covalent Organic Framework for Ratiometric Fluorescence Humidity Sensing, J. Am. Chem. Soc., 2025, 147, 9665, DOI:10.1021/jacs.4c17776.
- L. Wei, T. Sun, Z. Shi, Z. Xu, W. Wen, S. Jiang, Y. Zhao, Y. Ma and Y.-B. Zhang, Guest-adaptive molecular sensing in a dynamic 3D covalent organic framework, Nat. Commun., 2022, 13, 7936, DOI:10.1038/s41467-022-35674-8.
- Y.-X. Ma, Z.-J. Li, L. Wei, S.-Y. Ding, Y.-B. Zhang and W. Wang, A Dynamic Three-Dimensional Covalent Organic Framework, J. Am. Chem. Soc., 2017, 139, 4995, DOI:10.1021/jacs.7b01097.
- X. Liu, J. Li, B. Gui, G. Lin, Q. Fu, S. Yin, X. Liu, J. Sun and C. Wang, A Crystalline Three-Dimensional Covalent Organic Framework with Flexible Building Blocks, J. Am. Chem. Soc., 2021, 143, 2123, DOI:10.1021/jacs.0c12505.
- F. Auras, L. Ascherl, V. Bon, S. M. Vornholt, S. Krause, M. Döblinger, D. Bessinger, S. Reuter, K. W. Chapman, S. Kaskel, R. H. Friend and T. Bein, Dynamic two-dimensional covalent organic frameworks, Nat. Chem., 2024, 16, 1373, DOI:10.1038/s41557-024-01527-8.
- S. Horike, S. Shimomura and S. Kitagawa, Soft porous crystals, Nat. Chem., 2009, 1, 695, DOI:10.1038/nchem.444.
- S. Krause, N. Hosono and S. Kitagawa, Chemistry of Soft Porous Crystals: Structural Dynamics and Gas Adsorption Properties, Angew. Chem., Int. Ed., 2020, 59, 15325, DOI:10.1002/anie.202004535.
- J. D. Evans, V. Bon, I. Senkovska, H.-C. Lee and S. Kaskel, Four-dimensional metal-organic frameworks, Nat. Commun., 2020, 11, 2690, DOI:10.1038/s41467-020-16527-8.
- A. Schneemann, V. Bon, I. Schwedler, I. Senkovska, S. Kaskel and R. A. Fischer, Flexible metal–organic frameworks, Chem. Soc. Rev., 2014, 43, 6062, 10.1039/C4CS00101J.
- S. Liu, L. Wei, T. Zeng, W. Jiang, Y. Qiu, X. Yao, Q. Wang, Y. Zhao and Y.-B. Zhang, Single-Crystal Dynamic Covalent Organic Frameworks for Adaptive Guest Alignments, J. Am. Chem. Soc., 2024, 146, 34053, DOI:10.1021/jacs.4c13377.
- A. Yao, H. Xu, K. Shao, C. Sun, C. Qin, X. Wang and Z. Su, Guest-induced structural transformation of single-crystal 3D covalent organic framework at room and high temperatures, Nat. Commun., 2025, 16, 1385, DOI:10.1038/s41467-025-56750-9.
- S. Krause, V. Bon, I. Senkovska, U. Stoeck, D. Wallacher, D. M. Többens, S. Zander, R. S. Pillai, G. Maurin, F. O.-X. Coudert and S. Kaskel, A pressure-amplifying framework material with negative gas adsorption transitions, Nature, 2016, 532, 348, DOI:10.1038/nature17430.
- A. Ghoufi, K. Benhamed, L. Boukli-Hacene and G. Maurin, Electrically Induced Breathing of the MIL-53(Cr) Metal–Organic Framework, ACS Cent. Sci., 2017, 3, 394 CrossRef CAS PubMed.
- A. Knebel, B. Geppert, K. Volgmann, D. I. Kolokolov, A. G. Stepanov, J. Twiefel, P. Heitjans, D. Volkmer and J. Caro, Defibrillation of soft porous metal-organic frameworks with electric fields, Science, 2017, 358, 347, DOI:10.1126/science.aal2456.
- A. Ghoufi, A. Subercaze, Q. Ma, P. G. Yot, Y. Ke, I. Puente-Orench, T. Devic, V. Guillerm, C. Zhong, C. Serre, G. Férey and G. Maurin, Comparative Guest, Thermal, and Mechanical Breathing of the Porous Metal Organic Framework MIL-53(Cr): A Computational Exploration Supported by Experiments, J. Phys. Chem. C, 2012, 116, 13289 CrossRef CAS.
- S. Krause, J. D. Evans, V. Bon, S. Crespi, W. Danowski, W. R. Browne, S. Ehrling, F. Walenszus, D. Wallacher, N. Grimm, D. M. Többens, M. S. Weiss, S. Kaskel and B. L. Feringa, Cooperative light-induced breathing of soft porous crystals via azobenzene buckling, Nat. Commun., 2022, 13, 1951, DOI:10.1038/s41467-022-29149-z.
- J. A. Mason, J. Oktawiec, M. K. Taylor, M. R. Hudson, J. Rodriguez, J. E. Bachman, M. I. Gonzalez, A. Cervellino, A. Guagliardi, C. M. Brown, P. L. Llewellyn, N. Masciocchi and J. R. Long, Methane storage in flexible metal–organic frameworks with intrinsic thermal management, Nature, 2015, 527, 357, DOI:10.1038/nature15732.
- X. Liu, Z. Wang, Y. Zhang, N. Yang, B. Gui, J. Sun and C. Wang, Gas-Triggered Gate-Opening in a Flexible Three-Dimensional Covalent Organic Framework, J. Am. Chem. Soc., 2024, 146, 11411, DOI:10.1021/jacs.4c01331.
- Y. Xu, T. Sun, T. Zeng, X. Zhang, X. Yao, S. Liu, Z. Shi, W. Wen, Y. Zhao, S. Jiang, Y. Ma and Y.-B. Zhang, Symmetry-breaking dynamics in a tautomeric 3D covalent organic framework, Nat. Commun., 2023, 14, 4215, DOI:10.1038/s41467-023-39998-x.
- G. J. Halder, C. J. Kepert, B. Moubaraki, K. S. Murray and J. D. Cashion, Guest-Dependent Spin Crossover in a Nanoporous Molecular Framework Material, Science, 2002, 298, 1762, DOI:10.1126/science.1075948.
- S. B. Alahakoon, S. D. Diwakara, C. M. Thompson and R. A. Smaldone, Supramolecular design in 2D covalent organic frameworks, Chem. Soc. Rev., 2020, 49, 1344, 10.1039/C9CS00884E.
- K. Geng, T. He, R. Liu, S. Dalapati, K. T. Tan, Z. Li, S. Tao, Y. Gong, Q. Jiang and D. Jiang, Covalent Organic Frameworks: Design, Synthesis, and Functions, Chem. Rev., 2020, 120, 8814, DOI:10.1021/acs.chemrev.9b00550.
- X. Guan, F. Chen, Q. Fang and S. Qiu, Design and applications of three dimensional covalent organic frameworks, Chem. Soc. Rev., 2020, 49, 1357, 10.1039/C9CS00911F.
- W. K. Haug, E. M. Moscarello, E. R. Wolfson and P. L. McGrier, The luminescent and photophysical properties of covalent organic frameworks, Chem. Soc. Rev., 2020, 49, 839, 10.1039/C9CS00807A.
- Y. Li, W. Chen, G. Xing, D. Jiang and L. Chen, New synthetic strategies toward covalent organic frameworks, Chem. Soc. Rev., 2020, 49, 2852, 10.1039/D0CS00199F.
- Z. Li, T. He, Y. Gong and D. Jiang, Covalent Organic Frameworks: Pore Design and Interface Engineering, Acc. Chem. Res., 2020, 53, 1672, DOI:10.1021/acs.accounts.0c00386.
- R.-R. Liang, S.-Y. Jiang, R.-H. A and X. Zhao, Two-dimensional covalent organic frameworks with hierarchical porosity, Chem. Soc. Rev., 2020, 49, 3920, 10.1039/D0CS00049C.
- H. Wang, H. Wang, Z. Wang, L. Tang, G. Zeng, P. Xu, M. Chen, T. Xiong, C. Zhou, X. Li, D. Huang, Y. Zhu, Z. Wang and J. Tang, Covalent organic framework photocatalysts: structures and applications, Chem. Soc. Rev., 2020, 49, 4135, 10.1039/D0CS00278J.
- Z. Wang, S. Zhang, Y. Chen, Z. Zhang and S. Ma, Covalent organic frameworks for separation applications, Chem. Soc. Rev., 2020, 49, 708, 10.1039/C9CS00827F.
- N. Keller and T. Bein, Optoelectronic processes in covalent organic frameworks, Chem. Soc. Rev., 2021, 50, 1813, 10.1039/D0CS00793E.
- R. Liu, K. T. Tan, Y. Gong, Y. Chen, Z. Li, S. Xie, T. He, Z. Lu, H. Yang and D. Jiang, Covalent organic frameworks: an ideal platform for designing ordered materials and advanced applications, Chem. Soc. Rev., 2021, 50, 120, 10.1039/D0CS00620C.
- X. Zhao, P. Pachfule and A. Thomas, Covalent organic frameworks (COFs) for electrochemical applications, Chem. Soc. Rev., 2021, 50, 6871, 10.1039/D0CS01569E.
- D. Zhu, G. Xu, M. Barnes, Y. Li, C.-P. Tseng, Z. Zhang, J.-J. Zhang, Y. Zhu, S. Khalil, M. M. Rahman, R. Verduzco and P. M. Ajayan, Covalent Organic Frameworks for Batteries, Adv. Funct. Mater., 2021, 31, 2100505, DOI:10.1002/adfm.202100505.
- H. Chen, H. S. Jena, X. Feng, K. Leus and P. Van Der Voort, Engineering Covalent Organic Frameworks as Heterogeneous Photocatalysts for Organic Transformations, Angew. Chem., Int. Ed., 2022, 61, e202204938, DOI:10.1002/anie.202204938.
- X. Guan, Q. Fang, Y. Yan and S. Qiu, Functional Regulation and Stability Engineering of Three-Dimensional Covalent Organic Frameworks, Acc. Chem. Res., 2022, 55, 1912, DOI:10.1021/acs.accounts.2c00200.
- C. Qian, L. Feng, W. L. Teo, J. Liu, W. Zhou, D. Wang and Y. Zhao, Imine and imine-derived linkages in two-dimensional covalent organic frameworks, Nat. Rev. Chem., 2022, 6, 881, DOI:10.1038/s41570-022-00437-y.
- P. She, Y. Qin, X. Wang and Q. Zhang, Recent Progress in External-Stimulus-Responsive 2D Covalent Organic Frameworks, Adv. Mater., 2022, 34, 2101175, DOI:10.1002/adma.202101175.
- C. Wang, Z. Zhang, Y. Zhu, C. Yang, J. Wu and W. Hu, 2D Covalent Organic Frameworks: From Synthetic Strategies to Advanced Optical-Electrical-Magnetic Functionalities, Adv. Mater., 2022, 34, 2102290, DOI:10.1002/adma.202102290.
- S. Haldar, A. Schneemann and S. Kaskel, Covalent Organic Frameworks as Model Materials for Fundamental and Mechanistic Understanding of Organic Battery Design Principles, J. Am. Chem. Soc., 2023, 145, 13494, DOI:10.1021/jacs.3c01131.
- K. T. Tan, S. Ghosh, Z. Wang, F. Wen, D. Rodríguez-San-Miguel, J. Feng, N. Huang, W. Wang, F. Zamora, X. Feng, A. Thomas and D. Jiang, Covalent organic frameworks, Nat. Rev. Methods Primers, 2023, 3, 1, DOI:10.1038/s43586-022-00181-z.
- D. Blätte, F. Ortmann and T. Bein, Photons, Excitons, and Electrons in Covalent Organic Frameworks, J. Am. Chem. Soc., 2024, 146, 32161 CrossRef PubMed.
- Z. a. Guo, Z. Zhang and J. Sun, Topological Analysis and Structural Determination of 3D Covalent Organic Frameworks, Adv. Mater., 2024, 36, 2312889, DOI:10.1002/adma.202312889.
- W. Zhao, Q. Zhu, X. Wu and D. Zhao, The development of catalysts and auxiliaries for the synthesis of covalent organic frameworks, Chem. Soc. Rev., 2024, 53, 7531, 10.1039/D3CS00908D.
- D. Jiang, V. G. W. Tan, Y. Gong, H. Shao, X. Mu, Z. Luo and S. He, Semiconducting Covalent Organic Frameworks, Chem. Rev., 2025, 125, 6203–6308, DOI:10.1021/acs.chemrev.4c00950.
- H. Liu, S.-S. Zhu, Y. Zhi, H. Yue and X. Liu, Donor-acceptor type covalent organic frameworks: design, optimization strategies, and applications, Chem. Sci., 2025, 16, 12768–12803, 10.1039/D5SC01267H.
- T. Y. Lee, J. Yoon, E. Jo, S. Woo, K. Lee, S. Koo and D. W. Kang, Design and Synthesis of Single-Crystalline Covalent Organic Frameworks: The Use of Modulator for Reversible Bond Control, Chem.–Eur. J., 2025, e01731, DOI:10.1002/chem.202501731.
- Y.-F. Xie, S.-Y. Ding, J.-M. Liu, W. Wang and Q.-Y. Zheng, Triazatruxene based covalent organic framework and its quick-response fluorescence-on nature towards electron rich arenes, J. Mater. Chem. C, 2015, 3, 10066, 10.1039/C5TC02256H.
- F. Haase, K. Gottschling, L. Stegbauer, L. S. Germann, R. Gutzler, V. Duppel, V. S. Vyas, K. Kern, R. E. Dinnebiera and B. V. Lotsch, Tuning the stacking behaviour of a 2D covalent organic framework through non-covalent interactions, Mater. Chem. Front., 2017, 1, 1354 RSC.
- L. Frey, J. F. Pöhls, M. Hennemann, A. Mähringer, S. Reuter, T. Clark, R. T. Weitz and D. D. Medina, Oriented Thiophene-Extended Benzotrithiophene Covalent Organic Framework Thin Films: Directional Electrical Conductivity, Adv. Funct. Mater., 2022, 32, 2205949, DOI:10.1002/adfm.202205949.
- S. Feng, H. Xu, C. Zhang, Y. Chen, J. Zeng, D. Jiang and J.-X. Jiang, Bicarbazole-based redox-active covalent organic frameworks for ultrahigh-performance energy storage, Chem. Commun., 2017, 53, 11334, 10.1039/C7CC07024A.
- J. M. Rotter, R. Guntermann, M. Auth, A. Mähringer, A. Sperlich, V. Dyakonov, D. D. Medina and T. Bein, Highly conducting Wurster-type twisted covalent organic frameworks, Chem. Sci., 2020, 11, 12843, 10.1039/D0SC03909H.
- S.-L. Cai, Y.-B. Zhang, A. B. Pun, B. He, J. Yang, F. M. Toma, I. D. Sharp, O. M. Yaghi, J. Fan, S.-R. Zheng, W.-G. Zhang and Y. Liu, Tunable Electrical Conductivity in Oriented Thin Films of Tetrathiafulvalene-Based Covalent Organic Framework, Chem. Sci., 2014, 5, 4693, 10.1039/C4SC02593H.
- H. Li, P. Shao, S. Chen, G. Li, X. Feng, X. Chen, H.-J. Zhang, J. Lin and Y.-B. Jiang, Supramolecular Alternating Donor–Acceptor Assembly toward Intercalated Covalent Organic Frameworks, J. Am. Chem. Soc., 2020, 142, 3712, DOI:10.1021/jacs.9b13559.
- M. Wang, S. Fu and P. Petkov, et al., Exceptionally high charge mobility in phthalocyanine-based poly(benzimidazobenzophenanthroline)-ladder-type two-dimensional conjugated polymers, Nat. Mater., 2023, 22, 880, DOI:10.1038/s41563-023-01581-6.
- Y. Zhao, L. Guo, F. Gándara, Y. Ma, Z. Liu, C. Zhu, H. Lyu, C. A. Trickett, E. A. Kapustin, O. Terasaki and O. M. Yaghi, A Synthetic Route for Crystals of Woven Structures, Uniform Nanocrystals, and Thin Films of Imine Covalent Organic Frameworks, J. Am. Chem. Soc., 2017, 139, 13166 CrossRef CAS PubMed.
- X. Yan, H. Li, P. Shang, H. Liu, J. Liu, T. Zhang, G. Xing, Q. Fang and L. Chen, Facile synthesis of 3D covalent organic frameworks via a two-in-one strategy, Chem. Commun., 2021, 57, 2136–2139, 10.1039/D0CC08129A.
- D. Reinhard, W.-S. Zhang, Y. Vaynzof, F. Rominger, R. R. Schröder and M. Mastalerz, Triptycene-Based Porous Metal-Assisted Salphen Organic Frameworks: Influence of the Metal Ions on Formation and Gas Sorption, Chem. Mater., 2018, 30, 2781 CrossRef CAS.
- F. Jin, E. Lin, T. Wang, S. Geng, T. Wang, W. Liu, F. Xiong, Z. Wang, Y. Chen, P. Cheng and Z. Zhang, Bottom-Up Synthesis of 8-Connected Three-Dimensional Covalent Organic Frameworks for Highly Efficient Ethylene/Ethane Separation, J. Am. Chem. Soc., 2022, 144, 5643, DOI:10.1021/jacs.2c01058.
- S. Das, T. Sekine, H. Mabuchi, T. Irie, J. Sakai, Y. Zhao, Q. Fang and Y. Negishi, Three-Dimensional Covalent Organic Framework with scu-c Topology for Drug Delivery, ACS Appl. Mater. Interfaces, 2022, 14, 48045, DOI:10.1021/acsami.2c15152.
- Y. Zhao, T. Irie, D. Wen, H. Mabuchi, K. Sasaki, M. Nozaki, R. Tomioka, W. Zhu, S. Das, T. Ben and Y. Negishi, Highly Selective Separation of Benzene/Cyclohexane by Three-Dimensional Covalent Organic Framework with 8,8-Connected bcu Net Topology, ACS Mater. Lett., 2024, 6, 3063, DOI:10.1021/acsmaterialslett.4c00756.
- M. Lu, S.-B. Zhang, R.-H. Li, L.-Z. Dong, M.-Y. Yang, P. Huang, Y.-F. Liu, Z.-H. Li, H. Zhang, M. Zhang, S.-L. Li and Y.-Q. Lan, 3D Covalent Organic Frameworks with 16-Connectivity for Photocatalytic C(sp3)–C(sp2) Cross-Coupling, J. Am. Chem. Soc., 2024, 146, 25832 CrossRef CAS PubMed.
- G. Lin, H. Ding, D. Yuan, B. Wang and C. Wang, A Pyrene-Based, Fluorescent Three-Dimensional Covalent Organic Framework, J. Am. Chem. Soc., 2016, 138, 3302 CrossRef CAS PubMed.
- C. A. Hunter and J. K. M. Sanders, The nature of pi–pi interactions, J. Am. Chem. Soc., 1990, 112, 5525, DOI:10.1021/ja00170a016.
- C. R. Martinez and B. L. Iverson, Rethinking the term “pi-stacking”, Chem. Sci., 2012, 3, 2191 RSC.
- K. Wang, B. Hou, J. Dong, H. Niu, Y. Liu and Y. Cui, Controlling the Degree of Interpenetration in Chiral Three-Dimensional Covalent Organic Frameworks via Steric Tuning, J. Am. Chem. Soc., 2024, 146, 21466, DOI:10.1021/jacs.4c04183.
- J. Ding, X. Guan, J. Lv, X. Chen, Y. Zhang, H. Li, D. Zhang, S. Qiu, H.-L. Jiang and Q. Fang, Three-Dimensional Covalent Organic Frameworks with Ultra-Large Pores for Highly Efficient Photocatalysis, J. Am. Chem. Soc., 2023, 145, 3248, DOI:10.1021/jacs.2c13817.
- Y. Wang, Z. Qiao, H. Li, R. Zhang, Z. Xiang, D. Cao and S. Wang, Molecular Engineering for Modulating Photocatalytic Hydrogen Evolution of Fully Conjugated 3D Covalent Organic Frameworks, Angew. Chem., Int. Ed., 2024, 63, e202404726, DOI:10.1002/anie.202404726.
- B. J. Smith, A. C. Overholts, N. Hwang and W. R. Dichtel, Insight Into the Crystallization of Amorphous Imine-Linked Polymer Networks to 2D Covalent Organic Frameworks, Chem. Commun., 2016, 52, 3690 RSC.
- Z. Wang, Z. Zhang, H. Qi, A. Ortega-Guerrero, L. Wang, K. Xu, M. Wang, S. Park, F. Hennersdorf, A. Dianat, A. Croy, H. Komber, G. Cuniberti, J. J. Weigand, U. Kaiser, R. Dong and X. Feng, On-water surface synthesis of charged two-dimensional polymer single crystals via the irreversible Katritzky reaction, Nat. Synth., 2022, 1, 69, DOI:10.1038/s44160-021-00001-4.
- Z. Zhou, L. Zhang and Y. Yang, et al., Growth of single-crystal imine-linked covalent organic frameworks using amphiphilic amino-acid derivatives in water, Nat. Chem., 2023, 15, 841, DOI:10.1038/s41557-023-01181-6.
- J. Guo, Y. Xu, S. Jin, L. Chen, T. Kaji, Y. Honsho, M. A. Addicoat, J. Kim, A. Saeki, H. Ihee, S. Seki, S. Irle, M. Hiramoto and D. Jiang, Conjugated organic framework with three-dimensionally ordered stable structure and delocalized π clouds, Nat. Commun., 2013, 4, 2736 CrossRef PubMed.
- M. Wang, M. Ballabio, M. Wang, H.-H. Lin, B. P. Biswal, X. Han, S. Paasch, E. Brunner, P. Liu, M. Chen, M. Bonn, T. Heine, S. Zhou, E. Cánovas, R. Dong and X. Feng, Unveiling Electronic Properties in Metal–Phthalocyanine-Based Pyrazine-Linked Conjugated Two-Dimensional Covalent Organic Frameworks, J. Am. Chem. Soc., 2019, 141, 16810, DOI:10.1021/jacs.9b07644.
- Z. Meng, R. M. Stolz and K. A. Mirica, Two-Dimensional Chemiresistive Covalent Organic Framework with High Intrinsic Conductivity, J. Am. Chem. Soc., 2019, 141, 11929, DOI:10.1021/jacs.9b03441.
- Q. Fang, Z. Zhuang, S. Gu, R. B. Kaspar, J. Zheng, J. Wang, S. Qiu and Y. Yan, Designed synthesis of large-pore crystalline polyimide covalent organic frameworks, Nat. Commun., 2014, 5, 4503, DOI:10.1038/ncomms5503.
- Q. Fang, J. Wang, S. Gu, R. B. Kaspar, Z. Zhuang, J. Zheng, H. Guo, S. Qiu and Y. Yushan, 3D Porous Crystalline Polyimide Covalent Organic Frameworks for Drug Delivery, J. Am. Chem. Soc., 2015, 137, 8352 CrossRef CAS PubMed.
- K. Liu, H. Qi, R. Dong, R. Shivhare, M. Addicoat, T. Zhang, H. Sahabudeen, T. Heine, S. Mannsfeld, U. Kaiser, Z. Zheng and X. Feng, On-water surface synthesis of crystalline, few-layer two-dimensional polymers assisted by surfactant monolayers, Nat. Chem., 2019, 11, 994, DOI:10.1038/s41557-019-0327-5.
- K. C. Ranjeesh, R. Illathvalappil, S. D. Veer, J. Peter, V. C. Wakchaure, Goudappagouda, K. V. Raj, S. Kurungot and S. S. Babu, Imidazole-Linked Crystalline Two-Dimensional Polymer with Ultrahigh Proton-Conductivity, J. Am. Chem. Soc., 2019, 141, 14950 CrossRef CAS PubMed.
- F. Haase, E. Troschke, G. Savasci, T. Banerjee, V. Duppel, S. Dörfler, M. M. J. Grundei, A. M. Burow, C. Ochsenfeld, S. Kaskel and B. V. Lotsch, Topochemical conversion of an imine- into a thiazole-linked covalent organic framework enabling real structure analysis, Nat. Commun., 2018, 9, 2600 CrossRef PubMed.
- P. J. Waller, Y. S. AlFaraj, C. S. Diercks, N. N. Jarenwattananon and O. M. Yaghi, Conversion of Imine to Oxazole and Thiazole Linkages in Covalent Organic Frameworks, J. Am. Chem. Soc., 2018, 140, 9099 CrossRef CAS PubMed.
- S. Kandambeth, A. Mallick, B. Lukose, M. V. Mane, T. Heine and R. Banerjee, Construction of Crystalline 2D Covalent Organic Frameworks with Remarkable Chemical (Acid/Base) Stability via a Combined Reversible and Irreversible Route, J. Am. Chem. Soc., 2012, 134, 19524 CrossRef CAS PubMed.
- C. R. DeBlase, K. E. Silberstein, T.-T. Truong, H. C. D. Abruña and W. R. Dichtel, β-Ketoenamine-Linked Covalent Organic Frameworks Capable of Pseudocapacitive Energy Storage, J. Am. Chem. Soc., 2013, 135, 16821 CrossRef CAS PubMed.
- A. Acharjya, P. Pachfule, J. Roeser, F.-J. Schmitt and A. Thomas, Vinylene-Linked Covalent Organic Frameworks by Base-Catalyzed Aldol Condensation, Angew. Chem., Int. Ed., 2019, 58, 14865, DOI:10.1002/anie.201905886.
- S. Bi, C. Yang, Z. Wenbei, J. Xu, L. Liu, D. Wu, X. Wang, Y. Han, Q. Liang and F. Zhang, Two-dimensional semiconducting covalent organic frameworks via condensation at arylmethyl carbon atoms, Nat. Commun., 2019, 10, 2467 CrossRef PubMed.
- X. Zhuang, W. Zhao, F. Zhang, Y. Cao, F. Liu, S. Bia and X. Feng, A two-dimensional conjugated polymer framework with fully sp2-bonded carbon skeleton, Polym. Chem., 2016, 7, 4176 RSC.
- S. Ghouse, Z. Guo, S. Gámez-Valenzuela, D. Mücke, B. Zhang, L. Gao, S. Paasch, Y. Fu, C. Huang, C. Naisa, E. Brunner, M. Bonn, M. C. Ruiz Delgado, J. Sun, R. Zou, U. Kaiser, M. Wang and X. Feng, Towards single-crystalline two-dimensional poly(arylene vinylene) covalent organic frameworks, Nat. Chem., 2026 DOI:10.1038/s41557-025-02048-8.
- E. J. Carrington, C. A. McAnally, A. J. Fletcher, S. P. Thompson, M. Warren and L. Brammer, Solvent-switchable continuous-breathing behaviour in a diamondoid metal–organic framework and its influence on CO2 versus CH4 selectivity, Nat. Chem., 2017, 9, 882 CrossRef CAS PubMed.
- A. Pratasouskaya, V. Bon, A. Müller, S. Kaskel and F. Auras, Dynamic Two-Dimensional Covalent Organic Frameworks with Large Solvent-Induced Lattice Expansion, J. Am. Chem. Soc., 2024, 146, 29491, DOI:10.1021/jacs.4c08918.
- Y. Chen, Z.-L. Shi, L. Wei, B. Zhou, J. Tan, H.-L. Zhou and Y.-B. Zhang, Guest-Dependent Dynamics in a 3D Covalent Organic Framework, J. Am. Chem. Soc., 2019, 141, 3298, DOI:10.1021/jacs.8b13691.
- S. Cai, B. Sun, X. Li, Y. Yan, A. Navarro, A. Garzón-Ruiz, H. Mao, R. Chatterjee, J. Yano, C. Zhu, J. A. Reimer, S. Zheng, J. Fan, W. Zhang and Y. Liu, Reversible Interlayer Sliding and Conductivity Changes in Adaptive Tetrathiafulvalene-Based Covalent Organic Frameworks, ACS Appl. Mater. Interfaces, 2020, 12, 19054, DOI:10.1021/acsami.0c03280.
- Y. Liu, Y. Ma, J. Yang, C. S. Diercks, N. Tamura, F. Jin and O. M. Yaghi, Molecular Weaving of Covalent Organic Frameworks for Adaptive Guest Inclusion, J. Am. Chem. Soc., 2018, 140, 16015, DOI:10.1021/jacs.8b08949.
- F. Yu, W. Liu, B. Li, D. Tian, J.-L. Zuo and Q. Zhang, Photostimulus-Responsive Large-Area Two-Dimensional Covalent Organic Framework Films, Angew. Chem., Int. Ed., 2019, 58, 16101, DOI:10.1002/anie.201909613.
- T. Ma, J. Li, J. Niu, L. Zhang, A. S. Etman, C. Lin, D. Shi, P. Chen, L.-H. Li, X. Du, J. Sun and W. Wang, Observation of Interpenetration Isomerism in Covalent Organic Frameworks, J. Am. Chem. Soc., 2018, 140, 6763, DOI:10.1021/jacs.8b03169.
- T. Sun, L. Wei, Y. Chen, Y. Ma and Y.-B. Zhang, Atomic-Level Characterization of Dynamics of a 3D Covalent Organic Framework by Cryo-Electron Diffraction Tomography, J. Am. Chem. Soc., 2019, 141, 10962, DOI:10.1021/jacs.9b04895.
- H. Chi, Y. Liu, Z. Li, W. Chen and Y. He, Direct visual observation of pedal motion-dependent flexibility of single covalent organic frameworks, Nat. Commun., 2023, 14, 5061, DOI:10.1038/s41467-023-40831-8.
- C. Kang, Z. Zhang, S. Kusaka, K. Negita, A. K. Usadi, D. C. Calabro, L. Saunders Baugh, Y. Wang, X. Zou, Z. Huang, R. Matsuda and D. Zhao, Covalent organic framework atropisomers with multiple gas-triggered structural flexibilities, Nat. Mater., 2023, 22, 636, DOI:10.1038/s41563-023-01523-2.
- T. Zeng, Y. Ling, W. Jiang, X. Yao, Y. Tao, S. Liu, H. Liu, T. Yang, W. Wen, S. Jiang, Y. Zhao, Y. Ma and Y.-B. Zhang, Atomic observation and structural evolution of covalent organic framework rotamers, Proc. Natl. Acad. Sci. U. S. A., 2024, 121, e2320237121, DOI:10.1073/pnas.2320237121.
- M. Wang, T. Zeng, Y. Yu, X. Wang, Y. Zhao, H. Xi and Y.-B. Zhang, Flexibility On-Demand: Multivariate 3D Covalent Organic Frameworks, J. Am. Chem. Soc., 2024, 146, 1035, DOI:10.1021/jacs.3c11944.
- Y. Wang, Y. Liu, H. Li, X. Guan, M. Xue, Y. Yan, V. Valtchev, S. Qiu and Q. Fang, Three-Dimensional Mesoporous Covalent Organic Frameworks through Steric Hindrance Engineering, J. Am. Chem. Soc., 2020, 142, 3736, DOI:10.1021/jacs.0c00560.
- X. Li, T. Zhao, F. Wang, W. Wu, Y. Sun, H. Ren and F. Sun, Isoreticular 3D covalent organic frameworks with non-interpenetrated pcu-derived dia topology: pore regulation from micropores to mesopores, Chem. Sci., 2025, 16, 7339, 10.1039/D5SC01227A.
- Z. Tang, J. Chen, L. Sheng, Z. Li, Y. Yang, J. Wang, Y. Tang, X. He and H. Xu, Enhancing Gas Adsorption in Three-Dimensional Covalent Organic Frameworks via Conformational Effects, Chem. Mater., 2025, 37, 5217–5225, DOI:10.1021/acs.chemmater.5c00942.
- Y. Cheng, H. Du, Y. Wang, J. Xin, Y. Dong, X. Wang, X. Zhou, B. Gui, J. Sun and C. Wang, A Dynamic Covalent Organic Framework with Entangled 2D Layers, J. Am. Chem. Soc., 2025, 147, 6355, DOI:10.1021/jacs.4c17962.
- P. Albacete, J. I. Martínez, X. Li, A. Lopez-Moreno, S. Mena-Hernando, A. E. Platero-Prats, C. Montoro, K. P. Loh, E. M. Pérez and F. l. Zamora, Layer-Stacking-Driven Fluorescence in a Two-Dimensional Imine-Linked Covalent Organic Framework, J. Am. Chem. Soc., 2018, 140, 12922, DOI:10.1021/jacs.8b07450.
- C. Zhao, C. S. Diercks, C. Zhu, N. Hanikel, X. Pei and O. M. Yaghi, Urea-Linked Covalent Organic Frameworks, J. Am. Chem. Soc., 2018, 140, 16438, DOI:10.1021/jacs.8b10612.
- L. D. Tran, K. F. Presley, J. K. Streit, J. Carpena-Núñez, L. K. Beagle, T. A. Grusenmeyer, M. J. Dalton, R. A. Vaia, L. F. Drummy, N. R. Glavin and L. A. Baldwin, Divergent Properties in Structural Isomers of Triphenylamine-Based Covalent Organic Frameworks, Chem. Mater., 2022, 34, 529, DOI:10.1021/acs.chemmater.1c02565.
- S. Jhulki, A. M. Evans, X.-L. Hao, M. W. Cooper, C. H. Feriante, J. Leisen, H. Li, D. Lam, M. C. Hersam, S. Barlow, J.-L. Brédas, W. R. Dichtel and S. R. Marder, Humidity Sensing through Reversible Isomerization of a Covalent Organic Framework, J. Am. Chem. Soc., 2020, 142, 783, DOI:10.1021/jacs.9b08628.
- Z.-B. Zhou, H.-H. Sun, Q.-Y. Qi and X. Zhao, Gradually Tuning the Flexibility of Two-Dimensional Covalent Organic Frameworks via Stepwise Structural Transformation and Their Flexibility-Dependent Properties, Angew. Chem., Int. Ed., 2023, 62, e202305131, DOI:10.1002/anie.202305131.
- X. Shi, Q. Liu, H. Shao, C. Kang, H. Li, W.-H. Li, A. Mukherjee, J. Ren, N. Zhu, W. Zhao, D. Shi, Z. Zhang, Y. Wang, M. Lin and D. Zhao, Polyhedral Crystal Films of Covalent Organic Frameworks, J. Am. Chem. Soc., 2025, 147, 29130–29141, DOI:10.1021/jacs.5c07737.
- T. Sick, J. M. Rotter, S. Reuter, S. Kandambeth, N. N. Bach, M. Döblinger, J. Merz, T. Clark, T. B. Marder, T. Bein and D. D. Medina, Switching on and off Interlayer Correlations and Porosity in 2D Covalent Organic Frameworks, J. Am. Chem. Soc., 2019, 141, 12570, DOI:10.1021/jacs.9b02800.
- C. Kang, Z. Zhang, V. Wee, A. K. Usadi, D. C. Calabro, L. S. Baugh, S. Wang, Y. Wang and D. Zhao, Interlayer Shifting in Two-Dimensional Covalent Organic Frameworks, J. Am. Chem. Soc., 2020, 142, 12995, DOI:10.1021/jacs.0c03691.
- C. Kang, Z. Zhang, A. K. Usadi, D. C. Calabro, L. S. Baugh, K. Chai, Y. Wang and D. Zhao, Tunable Interlayer Shifting in Two-Dimensional Covalent Organic Frameworks Triggered by CO2 Sorption, J. Am. Chem. Soc., 2022, 144, 20363, DOI:10.1021/jacs.2c08214.
- Y. Zhu and W. Zhang, Reversible tuning of pore size and CO2 adsorption in azobenzene functionalized porous organic polymers, Chem. Sci., 2014, 5, 4957, 10.1039/C4SC02305F.
- G. Das, T. Prakasam, M. A. Addicoat, S. K. Sharma, F. Ravaux, R. Mathew, M. Baias, R. Jagannathan, M. A. Olson and A. Trabolsi, Azobenzene-Equipped Covalent Organic Framework: Light-Operated Reservoir, J. Am. Chem. Soc., 2019, 141, 19078, DOI:10.1021/jacs.9b09643.
- Y. Zhao, X. Tao, B. Xu, W. Liu and S. Lin, Robust Thiazole-Linked Covalent Organic Frameworks with Post-Modified Azobenzene Groups: Photo-Regulated Dye Adsorption and Separation, Adv. Funct. Mater., 2024, 34, 2401895, DOI:10.1002/adfm.202401895.
- D. Laliberté, T. Maris, E. Demers, F. Helzy, M. Arseneault and J. D. Wuest, Molecular Tectonics. Hydrogen-Bonded Networks Built from Tetra- and Hexaanilines, Cryst. Growth Des., 2005, 5, 1451, DOI:10.1021/cg049566f.
- L. Wei, X. Hai, T. Xu, Z. Wang, W. Jiang, S. Jiang, Q. Wang, Y.-B. Zhang and Y. Zhao, Encoding ordered structural complexity to covalent organic frameworks, Nat. Commun., 2024, 15, 2411, DOI:10.1038/s41467-024-46849-w.
- L. Grunenberg, G. Savasci, M. W. Terban, V. Duppel, I. Moudrakovski, M. Etter, R. E. Dinnebier, C. Ochsenfeld and B. V. Lotsch, Amine-Linked Covalent Organic Frameworks as a Platform for Postsynthetic Structure Interconversion and Pore-Wall Modification, J. Am. Chem. Soc., 2021, 143, 3430, DOI:10.1021/jacs.0c12249.
- M. Zhang, Y. Li, W. Yuan, X. Guo, C. Bai, Y. Zou, H. Long, Y. Qi, S. Li, G. Tao, C. Xia and L. Ma, Construction of Flexible Amine-linked Covalent Organic Frameworks by Catalysis and Reduction of Formic Acid via the Eschweiler–Clarke Reaction, Angew. Chem., Int. Ed., 2021, 60, 12396, DOI:10.1002/anie.202102373.
- Y. Li, Y. Wan, X. Deng, M. Zhao, B. Zhang and Z. Wang, Postsynthetic Construction of Single-Crystal Formamide-Linked Covalent Organic Frameworks, J. Am. Chem. Soc., 2025, 147, 29526–29533, DOI:10.1021/jacs.5c10341.
- H. Wang, L. Yi, G. Hu, X. Zhou and H. Deng, Rigidity Reinforcement of 3D Covalent Organic Frameworks by Controlling Interpenetration, J. Am. Chem. Soc., 2025, 147, 9067, DOI:10.1021/jacs.4c18749.
- Y. Li, Y. Han, M. Chen, Y. Feng and B. Zhang, Construction of a flexible covalent organic framework based on triazine units with interesting photoluminescent properties for sensitive and selective detection of picric acid, RSC Adv., 2019, 9, 30937, 10.1039/C9RA06583K.
- X. Han, T. Ma, B. L. Nannenga, X. Yao, S. E. Neumann, P. Kumar, J. Kwon, Z. Rong, K. Wang, Y. Zhang, J. A. R. Navarro, R. O. Ritchie, Y. Cui and O. M. Yaghi, Molecular weaving of chicken-wire covalent organic frameworks, Chem, 2023, 9, 2509, DOI:10.1016/j.chempr.2023.07.015.
- Y. Liu, C. S. Diercks, Y. Ma, H. Lyu, C. Zhu, S. A. Alshmimri, S. Alshihri and O. M. Yaghi, 3D Covalent Organic Frameworks of Interlocking 1D Square Ribbons, J. Am. Chem. Soc., 2019, 141, 677, DOI:10.1021/jacs.8b12177.
- C. Wang, Y. Wang, R. Ge, X. Song, X. Xing, Q. Jiang, H. Lu, C. Hao, X. Guo, Y. Gao and D. Jiang, A 3D Covalent Organic Framework with Exceptionally High Iodine Capture Capability, Chem.–Eur. J., 2018, 24, 585, DOI:10.1002/chem.201705405.
- J. Zou, D. Wen and Y. Zhao, Flexible three-dimensional diacetylene functionalized covalent organic frameworks for efficient iodine capture, Dalton Trans., 2023, 52, 731, 10.1039/D2DT03362C.
- Y. Liu, H. Chi, Y. Tan, Z. Chen and Y. He, Guest-Induced Structural Torsion in Single Covalent Organic Frameworks for Enhanced Photocatalysis, Nano Lett., 2025, 25, 11365–11373, DOI:10.1021/acs.nanolett.5c02501.
- J. Fang, Z. Fu, X. Chen, Y. Liu, F. Chen, Y. Wang, H. Li, Y. Yusran, K. Wang, V. Valtchev, S. Qiu, B. Zou and Q. Fang, Piezochromism in Dynamic Three-Dimensional Covalent Organic Frameworks, Angew. Chem., Int. Ed., 2023, 62, e202304234, DOI:10.1002/anie.202304234.
- X. Guo, T. Mao, Z. Wang, P. Cheng, Y. Chen, S. Ma and Z. Zhang, Fabrication of Photoresponsive Crystalline Artificial Muscles Based on PEGylated Covalent Organic Framework Membranes, ACS Cent. Sci., 2020, 6, 787, DOI:10.1021/acscentsci.0c00260.
| This journal is © The Royal Society of Chemistry 2026 |