Open Access Article
Open Access ArticleSynthesis of bacteriochlorophyll a
Duy T. M.
Chung
 ,
Khiem Chau
Nguyen†
,
Khiem Chau
Nguyen†
 ,
Yizhou
Liu
,
Yizhou
Liu
 and
Jonathan S.
Lindsey
and
Jonathan S.
Lindsey
 *
*
Department of Chemistry, North Carolina State University, Raleigh, NC 27695-8204, USA. E-mail: jlindsey@ncsu.edu
First published on 10th April 2026
Abstract
Photosynthetic tetrapyrroles absorb light to power the biosphere but have largely been neglected as targets of chemical synthesis. Bacteriochlorophyll a – a key macrocycle in the bacterial photosynthetic reaction center – contains four stereocenters at the rim of the bacteriochlorin chromophore due to the trans-dialkyl group in each pyrroline ring (B, D), and an epimerizable β-ketoester embedded in the isocyclic ring (E). Here, each pair of stereodefined vicinal substituents was introduced as a chiral 4-nitroalkanal building block, which was converted to an alkynone for subsequent coupling with an iodopyrrole (A, C), affording the AD and BC dihydrodipyrrins. The dihydrodipyrrins were equipped with reactive groups (1-formyl, AD-half; 1-(1,1-dimethoxymethyl) and 8-(3-methoxy-1,3-dioxopropyl, BC-half) suited for directed macrocycle formation. Knoevenagel condensation of AD and BC halves afforded a propenone, the nexus for constructing ring E concomitantly with the macrocycle in the subsequent one-flask, double-ring closure (Nazarov cyclization, electrophilic aromatic substitution, elimination of methanol). The aromatic bacteriopheophorbide was obtained as the 2-trimethylsilylethyl propanoate, which upon acidolysis and esterification with phytol yielded bacteriopheophytin a; subsequent magnesiation gave bacteriochlorophyll a. The modularity of the synthesis, straightforward construction of asymmetric building blocks, and convergent joining of AD and BC halves suggest that the present route may provide an entrée into diverse photosynthetic macrocycles.
Introduction
Bacteriochlorophylls, the chief pigments in anoxygenic photosynthetic bacteria, are of fundamental interest for a host of reasons. First, the long-wavelength absorption of bacteriochlorophyll a (BChl a) and its free base counterpart bacteriopheophytin a (Bpheo a) appears in the near-infrared (770 and 750 nm, respectively),1 a spectral region of immense interest in fields ranging from energy sciences to photomedicine.2 Second, anoxygenic photosynthetic bacteria3 contain only a single reaction center as opposed to the tandem (Z-scheme) reaction centers that underpin chlorophyll-based, oxygenic photosynthesis of plants and cyanobacteria.4 The comparative simplicity of the reaction center of anoxygenic photosynthetic bacteria has led to foundational use in studies of electron-transfer processes.5–8 Third, the mechanisms of energy transfer among BChl a molecules in antenna proteins continue to challenge theorists.9 Finally, while anoxygenic photosynthetic bacteria likely make only a small contribution to global photosynthetic productivity (given an estimated oceanic concentration of bacteriochlorophyll a that is ∼0.1–1% of that of chlorophyll a),10 a more substantial ecological role prevails in environments ranging from microbial mats (e.g., Fig. 1, panel A)11 to tropical rain forests.12,13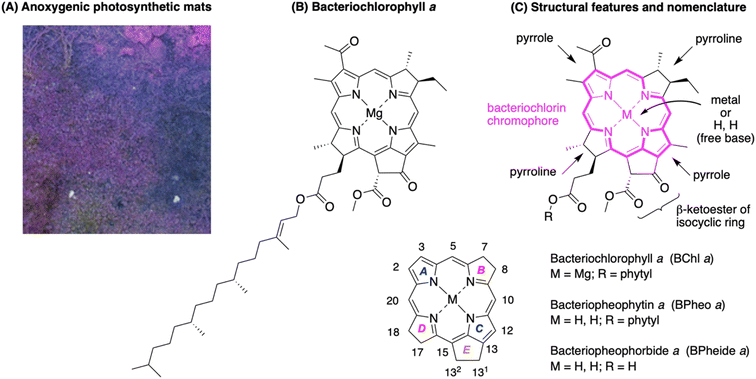 | ||
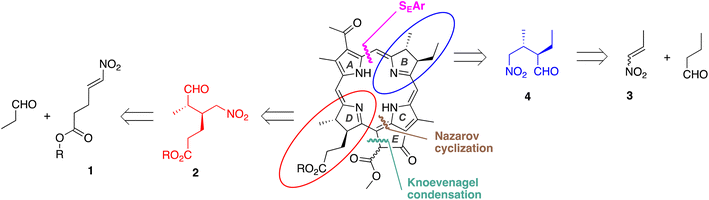
| Fig. 1 (A) Microbial mats containing anoxygenic photosynthetic bacteria.11 (B) Bacteriochlorophyll a. (C) Key motifs and nomenclature showing rings A–E. | ||
Bacteriochlorophyll a as a target of chemical synthesis presents several challenges, including (1) susceptibility to oxidative dehydrogenation to give the chlorin or porphyrin; (2) the presence of four stereocenters, two in each pyrroline ring;14 and (3) the presence of the annulated, “isocyclic” ring E. Given the absence of total synthesis, analogues have been prepared given the wide interest in photochemical processes undergirded by bacteriochlorophylls. Thus, use of native BChl a as a starting point for semisynthesis15 has afforded selected derivatives albeit in minute amounts. Isotopic feeding experiments with photosynthetic bacteria can afford exhaustive or partial,16,17 but not single-site (C, N, O), isotopic incorporation in the macrocycle, which could be pivotal in magnetic resonance experiments of photosynthetic complexes.18 Alterations to the bacterial photosynthetic reaction centers to tune electron-transfer processes have focused on modification of the protein superstructure much more than the ostensibly simpler native pigments. Model bacteriochlorins have been prepared by de novo synthesis19–21 but lack the full structural richness of the native macrocycles. The synthetic lacuna outlined for BChl a – a macrocycle first reported in the 1930s22 – is not a singularity, as the entire class of photosynthetic tetrapyrroles has fallen outside the realm of interest in modern natural products synthesis.23
In this paper, we report the synthesis of bacteriopheophorbide a (Bpheide a), methyl bacteriopheophorbide a (Me-BPheide a), bacteriopheophytin a (Bpheo a), and bacteriochlorophyll a (BChl a) starting from simple and readily available building blocks (Fig. 1, panels B and C). The terminology “bacterio” indicates the trans-tetrahydroporphyrin chromophore, “a” indicates the macrocycle family member, “pheo” indicates the free base macrocycle, and “phorbide” and “phytin” refer to the nature of the substituent (R) at the 173-carboxylic acid. The magnesium chelate of Bpheide a (bacteriochlorophyllide a) is the last biosynthetic precursor to all structural variants of BChl a,24,25 which differ by esterification with diverse hydrocarbon alcohols (ROH).26 Such a synthetic strategy should provide a general framework for gaining access to the family of ∼20 photosynthetic tetrapyrrole macrocycles.
Results and discussion
Retrosynthesis
Two key features of the synthesis entail (i) establishment of the stereochemical configuration of substituents in precursors to the pyrroline rings at an early stage of the synthesis with reliance on established asymmetric synthetic methodology, and (ii) joining of AD and BC halves via a two-step sequence: Knoevenagel condensation followed by a double-ring closure process consisting of Nazarov cyclization, electrophilic aromatic substitution, and elimination of methanol. The nascent isocyclic ring E, formed via successive Knoevenagel and Nazarov reactions, guides the appropriate joining of the AD and BC halves. The retrosynthesis is shown in Fig. 2.The synthesis of pyrroles (rings A and C) is well established.27,28 Routes to the pyrrolines (rings B and D) have relied on access to chiral hexynones29–31 that upon joining with the pyrrole afford the dihydrodipyrrin (AD or BC) bearing the trans-dialkyl substituents. The prior route to chiral hexynones used the Schreiber-modified Nicholas reaction.32,33 Here, an asymmetric Michael addition of an aliphatic aldehyde and nitroalkene (1, 3) is reported as a means to set the contiguous stereocenters in a chiral nitroaldehyde (2, 4) (Fig. 2). The new route proceeds with fewer protecting groups and enables direct introduction of the propanoic acid unit on ring D (which previously required resort to a protected propanol31).
Chiral precursors to pyrroline rings B and D
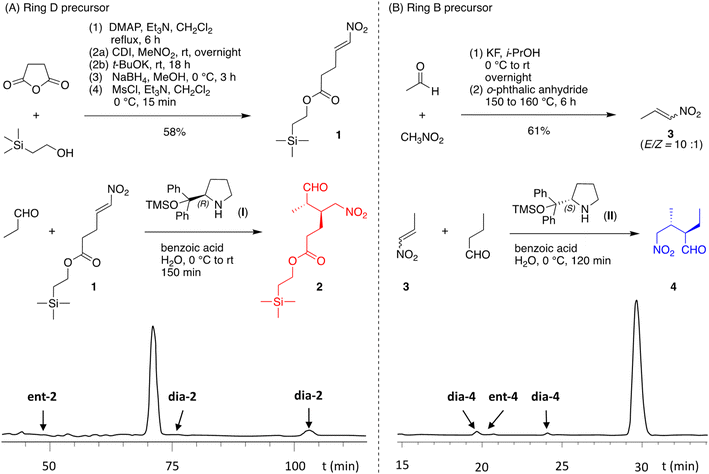
The synthesis of nitroalkenes 1 and 3 is shown in Fig. 3 panels A and B, respectively. Succinic anhydride was treated with 2-(trimethylsilyl)ethanol in the presence of triethylamine and a catalytic amount of 4-dimethylaminopyridine (DMAP)34 to give the mono-ester. A two-step one-flask procedure with 1,1′-carbonyldiimidazole followed by nitromethane gave the α-nitroketone.35 Reduction with sodium borohydride35 followed by dehydration of the resulting alcohol with methanesulfonyl chloride and triethylamine36 at low temperature provided nitroalkene 1, a stable compound isolated as an orange oil (see the absorption spectrum in Fig. S1). The nitroalkene 1 was synthesized via a streamlined manner and the above reactions were readily performed in multigram scale. The preparation of nitroalkene 3 began with the KF-catalyzed condensation of nitromethane and acetaldehyde to obtain the nitroalcohol,37 which was subjected to esterification, elimination, and distillation in one setup (Fig. S2).38 The distillate contained the desired 3 (along with water), which could be dried over Na2SO4 and stored at −20 °C under argon in the dark for several months without any significant degradation.The conditions for the Michael reaction of each nitroalkene and aldehyde were surveyed with use of chiral HPLC of reaction aliquots, with knowledge that the α-position of aliphatic aldehydes is easily epimerized39–41 upon prolonged reaction and purification by column chromatography.42–44 For these specific substrates, the best conditions were found to be those of Zhu45 – in aqueous solution containing benzoic acid at 0 °C for 2–3 hours in the presence of an O-TMS-protected diphenylprolinol (Hayashi)44 catalyst (2 mol%). Thus, nitroalkene 1 and propanal in the presence of (R)-O-TMS-protected diphenylprolinol (I) afforded predominantly one enantiomer (2, Fig. 3 panel A); the elution of the four stereoisomers was established by parallel reactions with pyrrolidine (Fig. S3–S5). Similarly, nitroalkene 3 and butanal in the presence of (S)-O-TMS-protected diphenylprolinol (II) afforded predominantly one enantiomer (4, Fig. 3 panel B); the parallel reaction with pyrrolidine also showed peaks for the four expected stereoisomers (Fig. S3–S5). In both cases, use of the enantiomer of I or II enabled identification of the enantiomer of 2 or 4, respectively, and by inference, the corresponding diastereomers (Fig. S3 and S4). Integration of the peaks gave dr = 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 53
1 and 53![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 for 2 and 4, respectively. The enantiomer of 2 was not detected whereas that for 4 gave an ee of 99%. After aqueous quenching, nitroalkanal 2 was stable at 0 °C under argon in the dark for several months without any significant epimerization, while nitroalkanal 4 underwent epimerization under the same conditions.
1 for 2 and 4, respectively. The enantiomer of 2 was not detected whereas that for 4 gave an ee of 99%. After aqueous quenching, nitroalkanal 2 was stable at 0 °C under argon in the dark for several months without any significant epimerization, while nitroalkanal 4 underwent epimerization under the same conditions.
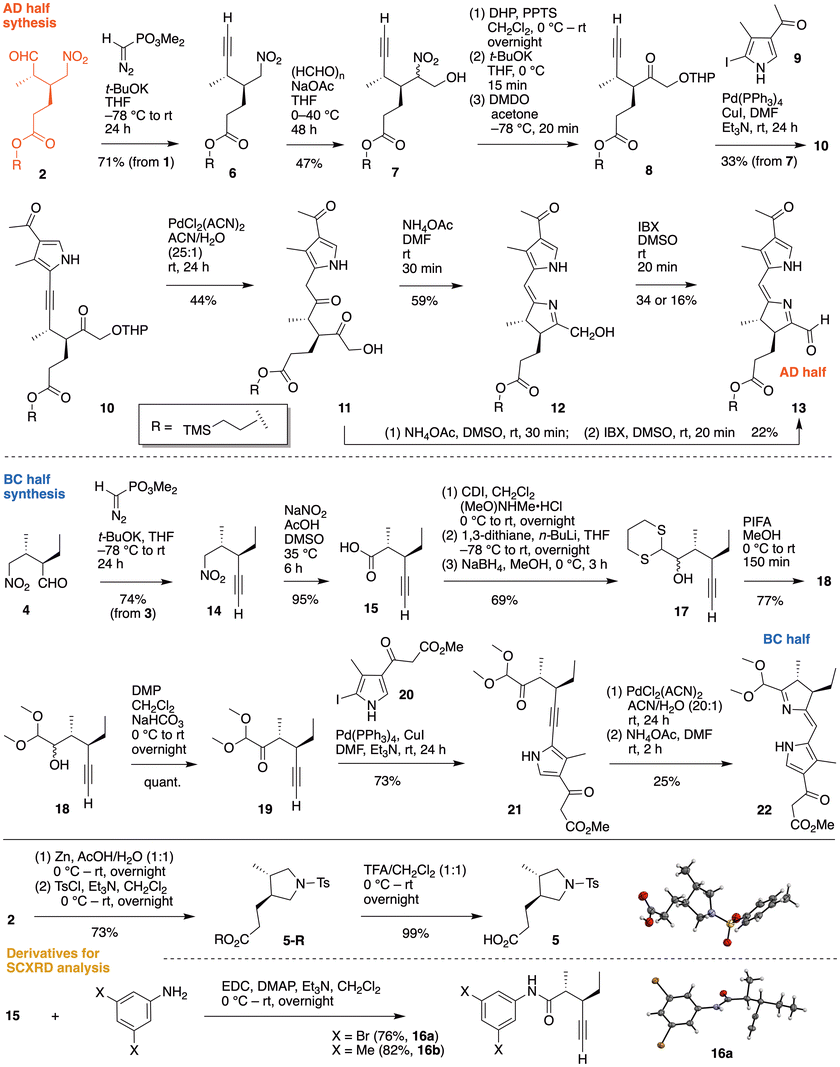
Synthesis of the AD half and the BC half
The asymmetric Michael addition of nitroalkene 1 and propanal was carried out to give chiral aldehyde 2, which was used without chromatography in subsequent reactions (Fig. 4 top panel). To confirm the stereochemistry, conversion to the pyrrolidine 5-R (by reduction of the nitro group, reductive amination, and tosylation)46 followed by removal of the 2-(trimethylsilyl)ethyl group47 gave 5 as a solid, which upon SCXRD analysis showed the desired trans-(S),(S) stereochemistry (Fig. 4 bottom panel, Fig. S6 and Table S1). The crude 2 was immediately treated with the Seyferth–Gilbert reagent (dimethyl (diazomethyl)phosphonate)48,49 to give alkyne 6 in 71% yield (from nitroalkene 1). Subsequent nitro–aldol reaction in the presence of paraformaldehyde under mild basic condition50 gave nitro-substituted chiral heptynoate 7 in 47% yield (and recovered starting material 6 in 45% yield).Attempts to transform the nitroalkyl moiety of 7 to the ketone (Nef reaction) under diverse conditions (e.g., NaOH and KMnO4,51 NaNO2,52tert-BuOK and DMDO53) afforded complex mixtures due to fragmentation, over-oxidation, and isomerization between α-ketol and α-hydroxyaldehyde species. To sidestep possible reactions of the unprotected alcohol, 7 was treated with 3,4-dihydro-2H-pyran and catalytic pyridinium p-toluenesulfonate54 to obtain the tetrahydropyranyl (THP) ether. Subsequent Nef reaction,53 entailing exposure to tert-BuOK in anhydrous THF and oxidation with DMDO, afforded upon silica pad filtration crude 8, which was used directly in the next step (Fig. 4 top panel). An analogue of compound 8 with R = phytyl was previously synthesized (via the propanol substituent)31 in exploratory studies.
The Sonogashira coupling30,55,56 of chiral heptynoate 8 and pre-A pyrrole 9 (ref. 57) gave heptynylpyrrole 10 in 33% yield (from 7). Treatment of 10 with PdCl2(MeCN)2 (ref. 58) in aqueous acetonitrile for anti-Markovnikov hydration of the internal alkynyl group gave the expected 1,4-diketone along with cleavage of the THP ether (as is known to occur59). Despite the moderate yield of the hydration (44%), attempts to characterize components other than 11 in the reaction mixture were unfruitful. The anti-Markovnikov hydration affording 11 was confirmed by nOe correlation between protons on the pyrrolic 3-methyl group and the 2-methylene group, and the absence of COSY correlation of protons on the 2-methylene group with other protons at different positions.
The Paal–Knorr type cyclization60 of 11 upon treatment with NH4OAc in anhydrous DMF at room temperature gave 1-hydroxymethyl dihydrodipyrrin 12 in 59% yield (Fig. 4 top panel). Monitoring the reaction by UV-vis absorption spectroscopy (Fig. S7, panel A) afforded spectra wherein the peak at 275 nm for 11 gave way over 30 min to a more intense peak at 325 nm for 12. Oxidation of the primary alcohol of 12 with IBX61 in anhydrous DMSO at room temperature62 gave the AD half 13 in 34% yield (0.22 mmol-scale) or 16% yield (2.6 mmol-scale). The conversion was similarly monitored by UV-vis absorption (Fig. S7, panel B). The absorbance at 325 nm (hydroxymethyl 12) decreased whereas a new peak emerged at 425 nm (aldehyde 13) after 20 min of exposure to IBX. Prolonging the reaction time for both reactions showed a decrease of the characteristic absorbance peaks (325 nm for 12, and 425 nm for 13; see Fig. S8 and S9).
The low yield of the conversion of dihydrodipyrrin 12 to 13 may be attributed to several factors. First, reactant and product can each independently give rise to a dipyrromethane (upon tautomerization) or a dipyrrin (upon oxidation). Secondly, 1-carboxaldehyde 13 can undergo self-condensation under the acidic conditions generated by IBX by-products (e.g., 2-iodobenzoic acid, 2-iodosobenzoic acid63). Third, dihydrodipyrrin 12 is a vinylogous pyrrole-α-carbinol, which as a class are known to be quite reactive toward hydroxyl elimination and polymerization. The α-carbinol 12 decomposed readily under anaerobic conditions and minimal light at −20 °C with detectable degradation (1H NMR and UV-vis analysis) within 24 h. In contrast, the oxidized product 13 remained stable under identical conditions for several months. In an effort to sidestep such pitfalls, a streamlined conversion of 11 to 13 was attempted; however, the combined yield of the final two reactions did not improve (not shown here). On the other hand, changing the solvent of the Paal–Knorr reaction from DMF to DMSO with all other parameters constant afforded an increase in overall yield of the two aforementioned reactions.
The synthesis of the BC half30 began with the homologation48,49 of the 5-nitropentanal 4 to obtain volatile nitropentyne 14 on a gram scale in 74% yield (Fig. 4 middle panel). The nitro group in 14 was converted to the carboxylic acid 15 by treatment with NaNO2 (3 equiv) in DMSO and AcOH64 (95% yield). Two anilide derivatives of 15 were isolated as solids (16a, 16b), which upon SCXRD analysis confirmed the desired trans-(R),(R) stereochemistry (Fig. 4 bottom panel, Fig. S10a, b and Tables S2a, b). Treatment of a CH2Cl2 solution of 15 with CDI and (MeO)NHMe·HCl afforded the Weinreb amide, which underwent a 2-step one-flask procedure (Corey–Seebach reaction, reduction with NaBH4) to give dithianylalcohol 17 (69% over 3 steps). Oxidative transacetalization65 of the dithiane moiety with (bis(trifluoroacetoxy)iodo)benzene (PIFA) afforded the dimethoxy group in 18 in good yield. Finally, treatment of 18 with DMP gave the chiral hexynone 19, which serves as the pre-B unit. In a similar manner as for the BC half, alkynone 19 underwent Sonogashira coupling with iodopyrrole pre-C (20)27 to obtain the hexynylpyrrole 21 in good yield. The anti-Markovnikov hydration of 21 following by Paal–Knorr type cyclization gave BC-half 22 in 25% yield. The present route provides superior access to the known BC half30 in terms of chiral precursor 4, proof of stereochemistry (16a, 16b), and streamlined procedures.
In summary, the asymmetric Michael reaction has enabled the conversion of simple starting materials to the AD and BC halves. Each compound in both routes (Fig. 4), except for crystalline derivatives 5, 16a and 16b for SCXRD analyses, was obtained as an oil or paste. The small quantities of diastereomers observed in the Michael reactions were carried forward and ensuing diastereomers could be clearly discerned by 1H NMR analysis for 6, 14, 15, and 19 (dr = 4.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 23
1, 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 33
1, 33![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 17
1, and 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively) (Fig. S11).
1, respectively) (Fig. S11).
Macrocycle formation, elaboration, and characterization
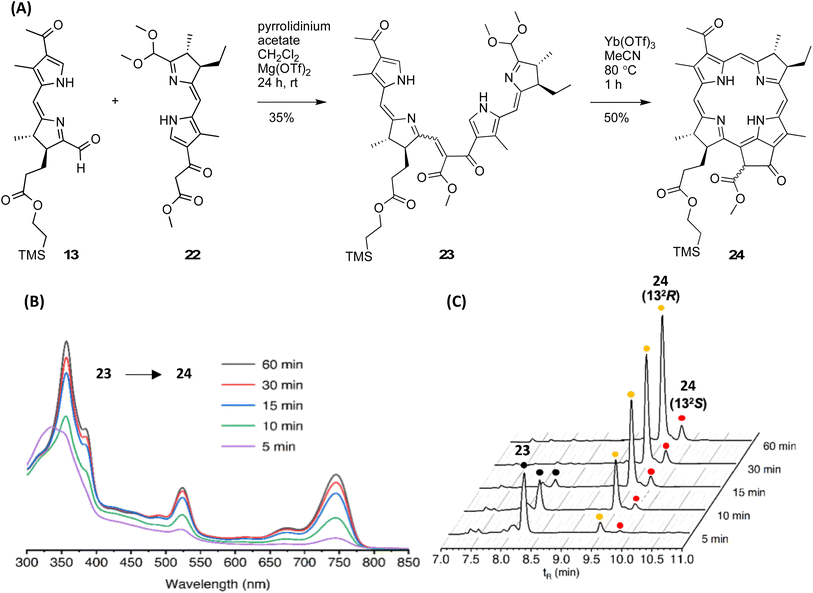
Model studies of the Knoevenagel condensation and subsequent double-ring closure have been carried out wherein one or both AD/BC halves contains gem-dimethyl substituents,21,28,29,66,67 which have identified promising conditions, albeit not with the full complement of stereodefined substituents. The Knoevenagel condensation (40 mM each half) relies on mild relatively neutral-basic conditions at room temperature, whereas the double-ring closure relies on mild acidic conditions at 80 °C.Here, the AD half (13) and the BC half (22), which collectively contain six carbonyl groups or equivalents, were treated to Knoevenagel condensation68 (Fig. 5 panel A). Application of the standard conditions (piperidine, AcOH, 3 Å MS, acetonitrile, room temperature for 20–60 h), which gave up to 73% with substrates containing simple substituents in the AD and BC halves,28 gave 11% here upon reaction of 13 and 22. Microscale screening of the conditions drawn from the literature66,69–73 of the Knoevenagel reaction with 13 and 22 in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (∼2 µmol each) at room temperature (see Table S3) gave the following observations: (1) under basic conditions (only piperidine), no product was detected, while using piperidine in conjunction with acetic acid gave low yield after prolonged reaction time. (2) Use of catalysts derived from amino acids gave low yield or no product. (3) A synergistic effect of pyrrolidinium acetate and Mg(OTf)2 was found to give a moderate yield of enone 23 as well as unreacted BC half 22. The latter conditions were thus employed in the synthesis. Reaction at 0.3 mmol each at room temperature for 24 h gave Knoevenagel enone 23 as an orange solid (see absorption spectrum in Fig. S12) in 35% yield. Examination by 1H NMR spectroscopy suggested the presence of only one enone isomer. A deeper assessment of the E/Z ratio was not attempted because (1) prior studies of similar enones have shown an E
1 ratio (∼2 µmol each) at room temperature (see Table S3) gave the following observations: (1) under basic conditions (only piperidine), no product was detected, while using piperidine in conjunction with acetic acid gave low yield after prolonged reaction time. (2) Use of catalysts derived from amino acids gave low yield or no product. (3) A synergistic effect of pyrrolidinium acetate and Mg(OTf)2 was found to give a moderate yield of enone 23 as well as unreacted BC half 22. The latter conditions were thus employed in the synthesis. Reaction at 0.3 mmol each at room temperature for 24 h gave Knoevenagel enone 23 as an orange solid (see absorption spectrum in Fig. S12) in 35% yield. Examination by 1H NMR spectroscopy suggested the presence of only one enone isomer. A deeper assessment of the E/Z ratio was not attempted because (1) prior studies of similar enones have shown an E![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Z ratio >10
Z ratio >10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1,29 (2) the two isomers interconvert during the subsequent Nazarov electrocyclization,74 and (3) use of either isomer alone gives the same yield in the Nazarov cyclization.67
1,29 (2) the two isomers interconvert during the subsequent Nazarov electrocyclization,74 and (3) use of either isomer alone gives the same yield in the Nazarov cyclization.67
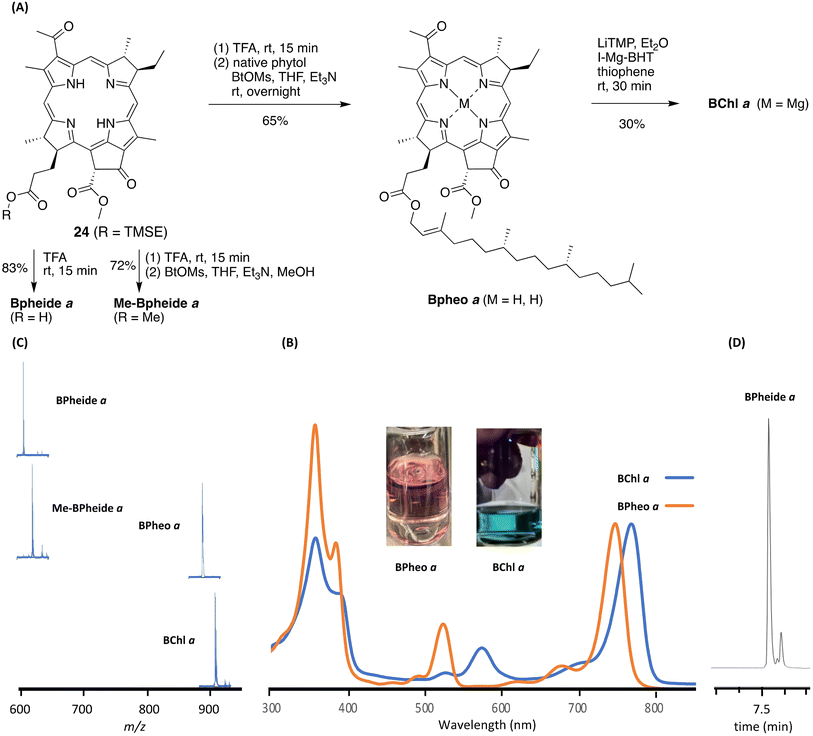
Enone 23 was treated to the acidic conditions for the double-ring closure67 (∼0.2 mM in acetonitrile containing Yb(OTf)3 at 80 °C for 1 h), which gave 2-(trimethylsilyl)ethyl bacteriopheophorbide 24 in 36% yield along with a minor amount of the 2-(trimethylsilyl)ethyl (TMSE)-cleaved product, bacteriopheophorbide a (Bpheide a, R = H). The examination of reaction samples by absorption spectroscopy (Fig. 5 panel B) showed the steady emergence of the characteristic B, Qx and Qy peaks4 of the bacteriopheophorbide. Examination by reversed-phase (RP) HPLC showed the disappearance of enone 23 within 15 min and the appearance of two peaks corresponding to bacteriopheophorbide 24 (Fig. 5 panel C). The two peaks are attributed to the two configurations of the 132-carbomethoxy group, with R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S ratio of ∼9
S ratio of ∼9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The configuration of the 132-carbomethoxy group is set upon double-ring closure.29,67 Epimerization at the 132-site in this class of macrocycles is known to occur slowly on standing,75,76 and both 132-epimers of the macrocycles are employed in native photosynthetic systems,77 although no enzyme for epimerization is known.78 The two epimers could be partially separated by preparative TLC and assessed by RP-HPLC (Fig. S13). Workup and isolation of bacteriopheophorbide 24 (combined epimers) from the reaction mixture entailed dilution with dichloromethane, washing with neutral aqueous phosphate buffer solution, concentration of the organic phase, and reversed-phase column chromatography under argon and dim lighting. Attempts to use normal phase (silica) chromatography resulted in the well-known processes of allomerization and adventitious dehydrogenation yielding multiple products (Fig. S14).
1. The configuration of the 132-carbomethoxy group is set upon double-ring closure.29,67 Epimerization at the 132-site in this class of macrocycles is known to occur slowly on standing,75,76 and both 132-epimers of the macrocycles are employed in native photosynthetic systems,77 although no enzyme for epimerization is known.78 The two epimers could be partially separated by preparative TLC and assessed by RP-HPLC (Fig. S13). Workup and isolation of bacteriopheophorbide 24 (combined epimers) from the reaction mixture entailed dilution with dichloromethane, washing with neutral aqueous phosphate buffer solution, concentration of the organic phase, and reversed-phase column chromatography under argon and dim lighting. Attempts to use normal phase (silica) chromatography resulted in the well-known processes of allomerization and adventitious dehydrogenation yielding multiple products (Fig. S14).
Removal of the 2-(trimethylsilyl)ethyl protecting group of bacteriopheophorbide 24 with TBAF or other conditions proved difficult (Fig. S15 and S16), but could be accomplished with neat trifluoroacetic acid (TFA) to give Bpheide a in 83% yield (Fig. 6 panel A). Subsequent transformations rely heavily on established procedures developed over the years for semisynthesis and reconstitution of native-origin macrocycles. Bacteriopheophorbide 24 also was treated with TFA followed by esterification (as described by Wasielewski and Svec79) of the resulting 173-carboxylic acid with an alcohol in the presence of benzotriazole-1-methanesulfonate (BtOMs)80 and triethylamine; use of methanol gave methyl bacteriopheophorbide a (Me-Bpheide a) whereas native phytol (for which syntheses are known81) gave bacteriopheophytin a (Bpheo a).
The free base macrocycle Bpheo a was treated to established conditions for magnesiation,82 which entail lithium 2,2,6,6-tetramethylpiperidide (LiTMP) and iodomagnesium 2,6-di-tert-butyl-4-methylphenolate (I-Mg-BHT) in thiophene, to afford bacteriochlorophyll a (BChl a). The long-wavelength absorption of the reaction mixture red-shifted over 30 min as expected for magnesiation. Prolonging the reaction did not give full conversion of the starting material, as also reported by Wasielewski.82 Purification by reversed-phase chromatography gave BChl a, which displayed absorption bands (Fig. 6 panel B) in the spectral regions of the near-ultraviolet (359, 390 nm, B band), green (575 nm, Qx band), and near-infrared (770 nm, Qy band). The absorption spectrum was essentially identical with an authentic (commercial) sample of BChl a (Fig. S17) – including the ratio of the intensity of the Qy band and the maximum of the B bands (IQy/IB),83 a longstanding metric – and was clearly distinct from the free base precursor Bpheo a. Indeed, dilute solutions of Bpheo a and BChl a appeared reddish purple and bluish-green, respectively (Fig. 6 panel B).
The free base macrocycles (24, BPheide a, Me-BPheide a, Bpheo a) were characterized by a variety of methods including absorption spectroscopy, 1H NMR and 13C{1H} NMR spectroscopy, MALDI-MS, and HRMS. The absorption spectra for all the macrocycles are provided in Fig. S18 with key parameters listed in Table S4 including comparison with native-origin samples.84–86 Tetrapyrrole macrocycles upon MALDI-MS analysis can ionize by electron loss to form [M˙]+ as well as by protonation to form [M+H]+.87,88 The molecular ion regions are shown for Bpheide a (C35H38N4O6), Me-BPheide a (C36H40N4O6), Bpheo a (C55H76N4O6), and BChl a (C55H74N4O6Mg) by MALDI-MS with peak maxima consistent with the target macrocycles at m/z = 610.21, 624.64, 887.25, and 910.65, respectively (Fig. 6 panel C). The MALDI-MS data for Bpheo a and BChl a were essentially identical with samples of native origin. Full mass spectra are provided in Fig. S19.
RP-HPLC analysis with detection at 740 nm of Bpheide a showed a major band and a minor band (85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 ratio) (Fig. 6 panel D), which are assigned to the well-known epimers that arise from the configuration of the 132-carbomethoxy group, as described earlier for 24. Analysis at 440 nm, where the absorption of bacteriopheophorbins is weak (ε ∼3000 M−1 cm−1, ∼1/30th of the value at 740 nm),89,90 still showed dominant peaks due to Bpheide a albeit preceded by several trace peaks (see Fig. S20 for full RP-HPLC traces at 740 nm and 440 nm). Similar RP-HPLC analysis of Bpheo a and a native-derived sample of Bpheo a exhibited nearly identical traces although the resolution was limited given the hydrophobicity of the phytyl tail (Fig. S21).
15 ratio) (Fig. 6 panel D), which are assigned to the well-known epimers that arise from the configuration of the 132-carbomethoxy group, as described earlier for 24. Analysis at 440 nm, where the absorption of bacteriopheophorbins is weak (ε ∼3000 M−1 cm−1, ∼1/30th of the value at 740 nm),89,90 still showed dominant peaks due to Bpheide a albeit preceded by several trace peaks (see Fig. S20 for full RP-HPLC traces at 740 nm and 440 nm). Similar RP-HPLC analysis of Bpheo a and a native-derived sample of Bpheo a exhibited nearly identical traces although the resolution was limited given the hydrophobicity of the phytyl tail (Fig. S21).
Attempts to use CD spectroscopy of both native-derived and synthetic samples of Bpheo a were largely uninformative (Fig. S22), given the predicted cancellation of the contributions to optical activity of the stereocenters in rings B and D, leaving only the asymmetry at the 132-position as contributive.91 While CD spectroscopy is attractive, more in-depth molecular information can be obtained by NMR spectroscopy.
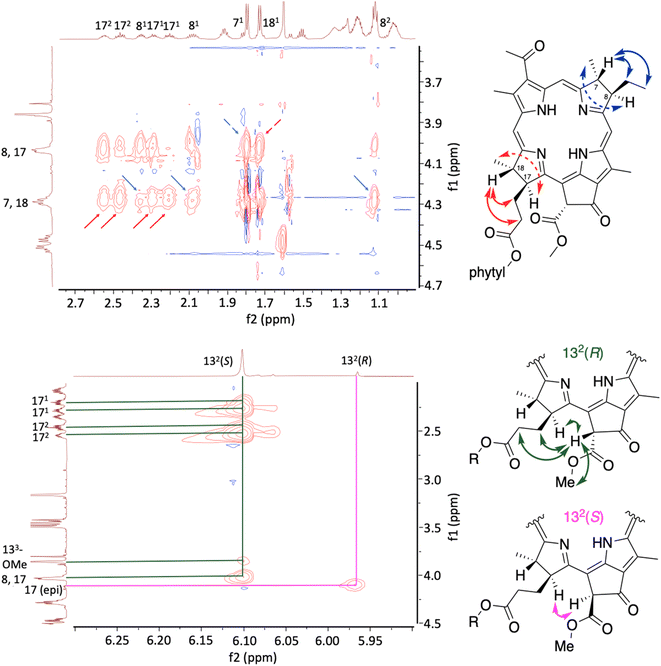
The NMR spectral features of several members of the family of native bacteriochlorophylls including BPheo a (ref. 92–95) and BChl a (ref. 96 and 97) have been described. 1H NMR spectra (in CDCl3) were collected for 24, Bpheide a, Me-Bpheide a, and Bpheo a. The spectra were generally consistent with expectations and literature values (where comparisons could be made). The 1H NMR and 13C{1H} NMR spectra of synthetic and native-derived Bpheo a samples were nearly identical (Fig. S23). The ROESY analysis for Bpheo a are shown in Fig. 7 (see Fig. S24a and b for ROESY of 24 and Bpheide a). The key observations are as follows:
(1) Dialkyl configurations in rings B and D: the methine proton at the 7-position (δ = 4.27–4.31 ppm) interacted (denoted as full arrow) with the methylene protons at the 81-position (δ = 2.05–2.57 ppm) and the methyl protons at the 82-position (δ = 1.10–1.29 ppm). Similarly, the proton at the 8-position (δ = 4.02–4.05 ppm) showed an NOE-correlation (denoted as dashed arrow) with the methyl protons at the 71-position (δ = 1.79 ppm). Corresponding interactions were observed for the methine proton at the 18-position (δ = 4.27–4.31 ppm) with the methylene protons at the 171, 172-positions (δ = 2.05–2.57 ppm); the 17-position proton (δ = 4.02–4.05 ppm) also showed a NOE-correlation with the methyl protons at the 181-position (δ = 1.72 ppm). In short, the spectral data are consistent with the trans-relationship of the vicinal alkyl substituents within pyrroline rings B and D.
(2) Epimers at the 132-position. Two distinct singlets (δ = 6.10 ppm, major; 5.96 ppm, minor, with major/minor ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.16) were assigned to the 132-methine proton. The major signal showed strong NOE-correlations with the 171, 172-protons of the propanoate group, consistent with a trans-relationship of the 132-carbomethoxy group with the 17-propanoate tail; NOE correlations also were observed with the 17-methine proton and the 133-methoxy group. The minor signal did not show NOE correlations with the 171, 172-protons, suggesting a syn-relationship of the 132-carbomethoxy group with the 17-propanoate group. In short, the epimer ratio in the 1H NMR spectrum mirrored that observed by RP-HPLC.
0.16) were assigned to the 132-methine proton. The major signal showed strong NOE-correlations with the 171, 172-protons of the propanoate group, consistent with a trans-relationship of the 132-carbomethoxy group with the 17-propanoate tail; NOE correlations also were observed with the 17-methine proton and the 133-methoxy group. The minor signal did not show NOE correlations with the 171, 172-protons, suggesting a syn-relationship of the 132-carbomethoxy group with the 17-propanoate group. In short, the epimer ratio in the 1H NMR spectrum mirrored that observed by RP-HPLC.
(3) Other stereoisomers. The congestion of the 1H NMR spectra of the macrocycles posed challenges to the identification of diastereomers arising from any cis-dialkyl configurations of rings B and D. Very weak resonances can be seen in the region between the peaks assigned to the 132(R) and 132(S) protons, which are provisionally attributed to tiny quantities of diastereomers due to cis-dialkyl configurations in rings B and D. The H5 resonance appears more cleanly diagnostic, as native-derived Bpheo a shows no such peaks (Fig. S25). Small quantities of cis-dialkyl stereoisomers are expected on the basis of the dr values (∼20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and ∼5
1 and ∼5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; Fig. S11) of the early-stage precursors, whereas enantiomers of the respective pyrroline rings are hardly expected given the high ee values. In summary, the NMR data collectively support the structure and stereochemical assignment of synthetic Bpheo a.
1; Fig. S11) of the early-stage precursors, whereas enantiomers of the respective pyrroline rings are hardly expected given the high ee values. In summary, the NMR data collectively support the structure and stereochemical assignment of synthetic Bpheo a.
Outlook
The work described herein achieves the total, fully traversed, synthesis of a photosynthetic macrocycle. Key features of the synthesis include the following: (1) use of established asymmetric methods to set the configurations of four stereocenters at a very early stage of the synthesis; (2) formation of dihydrodipyrrins by reaction of chiral alkynones (pre-B 19 and pre-D 8) with 2-iodopyrroles (pre-A 9 and pre-C 20) via Sonogashira cross-coupling reactions, Pd-mediated anti-Markovnikov hydration, and Paal–Knorr reaction; (3) exploitation of nascent ring E as a nexus for assembling the AD and BC halves via Knoevenagel reaction (mild neutral-basic conditions at high concentration) and subsequent double-ring closure (mild acidic conditions at low concentration); and (4) pre-building as much functionality into precursors thereby minimizing late-stage transformations on the intact macrocycle. The double-ring closure provides direct access to bacteriopheophorbide a as the 17-propanoate ester. The planning stages anticipated a host of well-precedented concerns: epimerization adjacent to carbonyl and imine groups,39–41 tautomerization of a dihydrodipyrrin to form the dipyrromethane,29 and oxidation98 and/or allomerization99 of bacteriochlorophyll macrocycles. A key conclusion is that intermediates and products could be handled securely, with the initial nitroalkanals 2 and 4 among all precursors proving to be most susceptible to epimerization, and the homologation products 6 and 14 as the most likely candidates for purification to enhance the dr values. To create desired patterns of substituents, streamlined transformations and use of a balance between early installation versus late-stage derivatization may be required to improve present yields such as the Knoevenagel condensation of the AD and BC halves. A key question moving forward is whether the synthetic route to bacteriochlorophyll a can be adapted to support synthetic entrée to the repertoire of native photosynthetic pigments as well as diverse pigment analogues. The latter include features not available from biosynthesis24,25 or semisynthesis15 such as single-site isotopic substitution, altered chirality, and general carbon remodeling of the macrocycle perimeter. Much synthetic work will be required to explore these possibilities.Author contributions
D. T. M. C. and K. C. N. carried out conceptualization, investigation, and methodology. Y. L. performed the initial conceptualization. J. S. L. contributed to conceptualization, funding acquisition, and supervision. D. T. M. C. and J. S. L. wrote the paper.Conflicts of interest
The authors declare no competing financial interest.Data availability
CCDC 2519170–2519172 (16b, 16a and 5) contain the supplementary crystallographic data for this paper.100a–cSupplementary information (SI): full experimental section; studies of selected reactions (asymmetric Michael addition, Knoevenagel condensation, Paal–Knorr condensation; cleavage of the TMSE group); 1H NMR and 13C{1H} NMR spectra for all new compounds; 1H NMR spectra for known pyrroles 9 and 20; macrocycle characterization data; absorption spectra; CD spectra; HPLC chromatograms; and single-crystal X-ray diffraction data. See DOI: https://doi.org/10.1039/d5sc10233b.
Acknowledgements
This work was supported by a grant from the NSF (CHE-2348052). NMR and mass spectrometry measurements were carried out in the Molecular Education, Technology, and Research Innovation Center (METRIC) at NC State University as well as at UNC Mass Spectrometry Core Laboratory. SCXRD analyses were performed by Dr Phattananawee Nalaoh at University of Tennessee at Knoxville.References
- D. Mauzerall, in The Photosynthetic Bacteria, ed. R. K. Clayton and W. R. Sistrom, Plenum Press, New York, USA, 1978, pp. 223–231 Search PubMed.
- B. Pucelik, A. Sułek and J. M. Dąbrowski, Coord. Chem. Rev., 2020, 416, 213340 CrossRef CAS.
- H. Gest and R. E. Blankenship, Photosynth. Res., 2004, 80, 59–70 CrossRef CAS PubMed.
- H. Scheer, in Chlorophylls and Bacteriochlorophylls, ed. B. Grimm, R. J. Porra, W. Rüdiger and H. Scheer, Springer Netherlands, Dordrecht, 2006, vol. 25, pp. 1–26 Search PubMed.
- J. J. Warren, J. R. Winkler and H. B. Gray, Coord. Chem. Rev., 2013, 257, 165–170 CrossRef CAS PubMed.
- S. G. Boxer, J. Phys. Chem. B, 2009, 113, 2972–2983 CrossRef CAS PubMed.
- W. Zinth and J. Wachtveitl, ChemPhysChem, 2005, 6, 871–880 CrossRef CAS PubMed.
- N. C. M. Magdaong, K. M. Faries, J. C. Buhrmaster, G. A. Tira, R. M. Wyllie, C. E. Kohout, D. K. Hanson, P. D. Laible, D. Holten and C. Kirmaier, J. Phys. Chem. B, 2022, 126, 8940–8956 CrossRef CAS PubMed.
- S. Badu, R. Melnik and S. Singh, Appl. Sci., 2020, 10, 6821 CrossRef CAS.
- R. Goericke, Limnol. Oceanogr., 2002, 47, 290–295 CrossRef CAS.
- C. Hubas, B. Jesus, M. Ruivo, T. Meziane, N. Thiney, D. Davoult, N. Spilmont, D. M. Paterson and C. Jeanthon, PLoS One, 2013, 8, e82329 CrossRef PubMed.
- A. Lehours, A. L. Jeune, J. Aguer, R. Céréghino, B. Corbara, B. Kéraval, C. Leroy, F. Perrière, C. Jeanthon and J. Carrias, Environ. Microbiol. Rep., 2016, 8, 689–698 CrossRef PubMed.
- J.-F. Carrias, C. Leroy, J.-P. Aguer, X.-T. Nguyen, J. Leflaive, B. Corbara, R. Céréghino and V. E. J. Jassey, Hydrobiologia, 2025, 852, 1613–1624 CrossRef CAS.
- M. Senge, A. Ryan, K. Letchford, S. MacGowan and T. Mielke, Symmetry, 2014, 6, 781–843 CrossRef.
- M. A. Grin and A. F. Mironov, Russ. Chem. Bull., 2016, 65, 333–349 CrossRef CAS.
- K. Czarnecki, J. R. Diers, V. Chynwat, J. P. Erickson, H. A. Frank and D. F. Bocian, J. Am. Chem. Soc., 1997, 119, 415–426 CrossRef CAS.
- S. Prakash, A. Alia, P. Gast, H. J. M. De Groot, G. Jeschke and J. Matysik, Biochemistry, 2007, 46, 8953–8960 CrossRef CAS PubMed.
- P. Bielytskyi, D. Gräsing, S. Zahn, A. Alia and J. Matysik, Appl. Magn. Reson., 2019, 50, 695–708 CrossRef CAS.
- C. Brückner, L. Samankumara and J. Ogikubo, in Handbook of Porphyrin Science, ed. K. M. Kadish, K. M. Smith and R. Guilard, World Scientific Publishing Company, Singapore, 2012, vol. 17, pp. 1–112 Search PubMed.
- S. V. Dudkin, E. A. Makarova and E. A. Lukyanets, Russ. Chem. Rev., 2016, 85, 700–730 CrossRef CAS.
- V.-P. Tran, P. Wang, N. Matsumoto, S. Liu, H. Jing, P. Nalaoh, K. Chau Nguyen, M. Taniguchi and J. S. Lindsey, J. Porphyrins Phthalocyanines, 2023, 27, 1502–1551 CrossRef CAS.
- C. B. van Neil and W. Arnold, Enzymologia, 1938, 5, 244–250 Search PubMed.
- Y. Liu, S. Zhang and J. S. Lindsey, Nat. Prod. Rep., 2018, 35, 879–901 RSC.
- D. A. Bryant, C. N. Hunter and M. J. Warren, J. Biol. Chem., 2020, 295, 6888–6925 CrossRef CAS PubMed.
- M. S. Proctor, G. A. Sutherland, D. P. Canniffe and A. Hitchcock, R. Soc. Open Sci., 2022, 9, 211903 CrossRef CAS PubMed.
- T. Mizoguchi, M. Isaji, J. Harada, Y. Tsukatani and H. Tamiaki, J. Photochem. Photobiol. B Biol., 2015, 142, 244–249 CrossRef CAS PubMed.
- P. Z. Wang, K. C. Nguyen and J. S. Lindsey, J. Org. Chem., 2019, 84, 11286–11293 CrossRef CAS PubMed.
- D. T. M. Chung, P. V. Tran, K. Chau Nguyen, P. Wang and J. S. Lindsey, New J. Chem., 2021, 45, 13302–13316 RSC.
- K. C. Nguyen, P. Wang, R. D. Sommer and J. S. Lindsey, J. Org. Chem., 2020, 85, 6605–6619 CrossRef CAS PubMed.
- K. C. Nguyen and J. S. Lindsey, J. Org. Chem., 2023, 88, 11205–11216 CrossRef CAS PubMed.
- K. Chau Nguyen, D. T. M. Chung, P. Nalaoh and J. S. Lindsey, New J. Chem., 2024, 48, 2097–2117 RSC.
- S. L. Schreiber, T. Sammakia and W. E. Crowe, J. Am. Chem. Soc., 1986, 108, 3128–3130 CrossRef CAS.
- K. M. Nicholas, Acc. Chem. Res., 1987, 20, 207–214 CrossRef CAS.
- M. T. Burger and P. A. Bartlett, J. Am. Chem. Soc., 1997, 119, 12697–12698 CrossRef CAS.
- S. T. Lima, T. R. Fallon, J. L. Cordoza, J. R. Chekan, E. Delbaje, A. R. Hopiavuori, D. O. Alvarenga, S. M. Wood, H. Luhavaya, J. T. Baumgartner, F. A. Dörr, A. Etchegaray, E. Pinto, S. M. K. McKinnie, M. F. Fiore and B. S. Moore, J. Am. Chem. Soc., 2022, 144, 9372–9379 CrossRef CAS PubMed.
- J. Melton and J. E. McMurry, J. Org. Chem., 1975, 40, 2138–2139 CrossRef CAS.
- R. H. Wollenberg and S. J. Miller, Tetrahedron Lett., 1978, 19, 3219–3222 CrossRef.
- M. Miyashita, T. Yanami and A. Yoshikoshi, Org. Synth., 1981, 60, 101 CrossRef CAS.
- S. Forsén and M. Nilsson, in The Carbonyl Group, ed. J. Zabicky, John Wiley & Sons, Ltd, Chichester, UK, 1970, vol. 2, pp. 157–240 Search PubMed.
- J. Toullec, Adv. Phys. Org. Chem., 1982, 18, 1–77 CrossRef CAS.
- J. Wirz, Adv. Phys. Org. Chem., 2010, 44, 325–356 CrossRef CAS.
- J. Burés, A. Armstrong and D. G. Blackmond, J. Am. Chem. Soc., 2011, 133, 8822–8825 CrossRef.
- K. Patora-Komisarska, M. Benohoud, H. Ishikawa, D. Seebach and Y. Hayashi, Helv. Chim. Acta, 2011, 94, 719–745 CrossRef CAS.
- Y. Hayashi, Org. Synth., 2017, 94, 252–258 CrossRef CAS.
- S. Zhu, S. Yu and D. Ma, Angew. Chem., Int. Ed., 2008, 47, 545–548 CrossRef CAS PubMed.
- B. Zheng, H. Wang, Y. Han, C. Liu and Y. Peng, Chem. Commun., 2013, 49, 4561–4563 RSC.
- L. L. Cheung, S. Marumoto, D. C. Anderson and D. S. Rychnovsky, Org. Lett., 2008, 14, 3101–3104 CrossRef PubMed.
- D. Seyferth, P. Hilbert and R. S. Marmor, J. Am. Chem. Soc., 1967, 89, 4811–4812 CrossRef CAS.
- J. C. Gilbert and U. Weerasooriya, J. Org. Chem., 1982, 47, 1837–1845 CrossRef CAS.
- F. Zhang, M. Wei, J. Dong, Y. Zhou, D. Lu, Y. Gong and X. Yang, Adv. Synth. Catal., 2010, 352, 2875–2880 CrossRef CAS.
- K. Steliou and M. A. Poupart, J. Org. Chem., 1985, 50, 4971–4973 CrossRef CAS.
- A. Gissot, S. N'Gouela, C. Matt, A. Wagner and C. Mioskowski, J. Org. Chem., 2004, 69, 8997–9001 CrossRef CAS PubMed.
- W. Adam, M. Makosza, C. R. Saha-Möller and C.-G. Zhao, Synlett, 1998, 1998, 1335–1336 CrossRef.
- M. Miyashita, A. Yoshikoshi and P. A. Grieco, J. Org. Chem., 1977, 42, 3772–3774 CrossRef CAS.
- P. A. Jacobi, J. Guo, S. Rajeswari and W. Zheng, J. Org. Chem., 1997, 62, 2907–2916 CrossRef CAS PubMed.
- R. Chinchilla and C. Nájera, Chem. Rev., 2007, 107, 874–922 CrossRef CAS PubMed.
- P. Wang and J. S. Lindsey, J. Org. Chem., 2021, 86, 11794–11811 CrossRef CAS PubMed.
- K. Imi, K. Imai and K. Utimoto, Tetrahedron Lett., 1987, 28, 3127–3130 CrossRef CAS.
- Y.-G. Wang, X.-X. Wu and Z.-Y. Jiang, Tetrahedron Lett., 2004, 45, 2973–2976 CrossRef CAS.
- W. G. O'Neal, W. P. Roberts, I. Ghosh and P. A. Jacobi, J. Org. Chem., 2005, 70, 7243–7251 CrossRef PubMed.
- M. Frigerio, M. Santagostino and S. Sputore, J. Org. Chem., 1999, 64, 4537–4538 CrossRef CAS.
- M. Frigerio and M. Santagostino, Tetrahedron Lett., 1994, 35, 8019–8022 CrossRef CAS.
- M. A. Lapitskaya, L. L. Vasiljeva and K. K. Pivnitsky, Mendeleev Commun., 2008, 18, 309–311 CrossRef CAS.
- C. Matt, A. Wagner and C. Mioskowski, J. Org. Chem., 1997, 62, 234–235 CrossRef CAS PubMed.
- G. Stork and K. Zhao, Tetrahedron Lett., 1989, 30, 287–290 CrossRef CAS.
- S. Zhang and J. S. Lindsey, J. Org. Chem., 2017, 82, 2489–2504 CrossRef CAS PubMed.
- K. C. Nguyen, P. Wang and J. S. Lindsey, New J. Chem., 2021, 45, 569–581 RSC.
- G. Jones, Org. React., 2011, 15, 204–599 CrossRef.
- M. Yamaguchi, N. Yokota and T. Minami, J. Chem. Soc., Chem. Commun., 1991, 1088–1089 RSC.
- A. Erkkilä and P. M. Pihko, J. Org. Chem., 2006, 71, 2538–2541 CrossRef PubMed.
- Y. Hu, Y.-H. He and Z. Guan, Catal. Commun., 2010, 11, 656–659 CrossRef CAS.
- R. C. M. Alves Sobrinho, P. M. de Oliveira, C. R. Montes D'Oca, D. Russowsky and M. G. Montes D'Oca, RSC Adv., 2017, 7, 3214–3221 RSC.
- A. T. Nguyen Tran, Z. Wu, D. T. M. Chung, P. Nalaoh and J. S. Lindsey, New J. Chem., 2023, 47, 13626–13637 RSC.
- J. A. Malona, J. M. Colbourne and A. J. Frontier, Org. Lett., 2006, 8, 5661–5664 CrossRef CAS PubMed.
- H. Mazaki, T. Watanabe, T. Takahashi, A. Struck and H. Scheer, Bull. Chem. Soc. Jpn., 1992, 65, 3080–3087 CrossRef CAS.
- Y. Saga and S. Nakagawa, J. Porphyrins Phthalocyanines, 2020, 24, 499–504 CrossRef CAS.
- C. J. Gisriel, C. Azai and T. Cardona, Photosynth. Res., 2021, 149, 329–343 CrossRef CAS PubMed.
- G. S. Orf, C. Gisriel and K. E. Redding, Photosynth. Res., 2018, 138, 11–37 CrossRef CAS PubMed.
- M. R. Wasielewski and W. A. Svec, J. Org. Chem., 1980, 45, 1969–1974 CrossRef CAS.
- M. Itoh, D. Hagiwara and J. Notani, Synthesis, 1975, 456–458 CrossRef CAS.
- S. Takano, T. Sugihara and K. Ogasawara, Synlett, 1991, 279–282 CrossRef CAS.
- M. R. Wasielewski, Tetrahedron Lett., 1977, 18, 1373–1376 CrossRef.
- J. H. C. Smith and A. Benitez, in Modern Methods of Plant Analysis, ed. K. Paech and M. V. Tracey, Springer Berlin Heidelberg, Berlin, Heidelberg, 1955, vol. 4, pp. 142–196 Search PubMed.
- H. Falk, G. Hoornaert, H. Isenring and A. Eschenmoser, Helv. Chim. Acta, 1975, 58, 2347–2357 CrossRef CAS.
- H. Scheer and J. J. Katz, J. Am. Chem. Soc., 1978, 100, 561–571 CrossRef CAS.
- J. Dandler, B. Wilhelm and H. Scheer, Photochem. Photobiol., 2010, 86, 182–193 CrossRef CAS PubMed.
- N. Srinivasan, C. A. Haney, J. S. Lindsey, W. Zhang and B. T. Chait, J. Porphyrins Phthalocyanines, 1999, 3, 283–291 CrossRef CAS.
- C. D. Calvano, G. Ventura, M. Trotta, G. Bianco, T. R. I. Cataldi and F. Palmisano, J. Am. Soc. Mass Spectrom., 2017, 28, 125–135 CrossRef CAS PubMed.
- E. Walter, J. Schreiber, E. Zass and A. Eschenmoser, Helv. Chim. Acta, 1979, 62, 899–920 CrossRef CAS.
- M. Meyer, H. Scheer and J. Breton, FEBS Lett., 1996, 393, 131–134 CrossRef CAS PubMed.
- C. Houssier and K. Sauer, J. Am. Chem. Soc., 1970, 92, 779–791 CrossRef CAS.
- A. N. Kozyrev, Y. Chen, L. N. Goswami, W. A. Tabaczynski and R. K. Pandey, J. Org. Chem., 2006, 71, 1949–1960 CrossRef CAS PubMed.
- G. Zheng, WO 2006/073419, 2006.
- Á. Roxin, J. Chen, A. S. Paton, T. P. Bender and G. Zheng, J. Med. Chem., 2014, 57, 223–237 CrossRef PubMed.
- Á. Roxin, T. D. MacDonald and G. Zheng, J. Porphyrins Phthalocyanines, 2014, 18, 188–199 CrossRef.
- R. G. Brereton and J. K. M. Sanders, J. Chem. Soc. Perkin Trans. I, 1983, 423–430 RSC.
- R. G. Brereton and J. K. M. Sanders, J. Chem. Soc. Perkin Trans. I, 1983, 435–437 RSC.
- L. Limantara, P. Koehler, B. Wilhelm, R. J. Porra and H. Scheer, Photochem. Photobiol., 2006, 82, 770–780 CrossRef CAS PubMed.
- K. Hyvärinen and P. H. Hynninen, Res. Adv. Org. Bioorg. Chem., 2001, 1, 1–19 Search PubMed.
- (a) CCDC 2519170: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2qkdk8; (b) CCDC 2519171: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2qkdl9; (c) CCDC 2519172: Experimental Crystal Structure Determination, 2025, DOI:10.5517/ccdc.csd.cc2qkdmb.
Footnote |
| † Deceased. |
| This journal is © The Royal Society of Chemistry 2026 |