Open Access Article
Open Access ArticlePhotocrosslinking chemistry for direct photopatterning of colloidal nanocrystals: toward pixelated light emitting diodes and beyond
Wenyue
Qing
and
Hao
Zhang
 *
*
Department of Chemistry, Center for BioAnalytical Chemistry, Key Laboratory of Bioorganic Phosphorus Chemistry & Chemical Biology (Ministry of Education), Tsinghua University, Beijing 100084, China. E-mail: hzhangchem@mail.tsinghua.edu.cn
First published on 31st March 2026
Abstract
Colloidal quantum dots (QDs) exhibit outstanding optoelectronic properties and solution processability, making them promising candidates for devices such as quantum dot light emitting diodes (QLEDs). The realization of integrated QD-based devices, particularly in QLED displays, necessitates patterning techniques that offer high resolution, high fidelity, and preserved luminescence. Direct photopatterning via photocrosslinking chemistry has emerged as a promising strategy, which achieves microscale to nanoscale patterning by using light-triggered reactions of surface ligands to modulate QD colloidal stability without compromising their optical properties. This review outlines recent advances in photocrosslinking-enabled direct photopatterning of QDs for QLED applications. We begin by introducing the underlying mechanisms and representative photochemistries for direct patterning. Subsequently, we survey various photocrosslinking chemistries and their applications in QD patterning. Drawing on case studies of representative photocrosslinking mechanisms, we discuss key molecular design principles for crosslinkers toward high-performance QLEDs. Furthermore, we extend the discussion to patterning of other functional nanomaterials, such as metal–organic frameworks, and to three-dimensional QD printing. Finally, we conclude with an outlook on future developments and the broadening applications of photocrosslinking-enabled patterning technologies.
1 Introduction
QDs are promising nanomaterials for next-generation electronic and photonic devices, owing to their outstanding optoelectronic properties and solution processability.1–7 For instance, combined attributes of wide colour gamut, narrow spectral bandwidths, and near-unity photoluminescent quantum yield (PLQY) of QDs enables the rapid developments of QLEDs and commercialization of QD televisions, among others.8–20 However, the integration of QDs, or more generally functional nanocrystals (NCs), into system-level devices requires precise deposition of QD patterns with micro- or even nanoscale precision, which necessitates high-resolution and high-fidelity patterning techniques.20,21 Over the past few decades, various QD patterning methods have been developed, including inkjet printing, transfer printing, and lithography.17,20,22–29Conventional lithography typically involves complex, multi-step processes and the use of photoresists whose solvents and chemicals could degrade the optoelectronic properties of QDs, presenting a significant bottleneck for device performance.28,29 Inkjet printing technology could achieve large-area, high-throughput, and low-cost patterning, but it is limited by nozzle size and the coffee-ring effect, typically restricting its resolution to tens to hundreds of micrometers.22,23 Transfer printing technology, by constructing micro- and nano-scale stamps, has demonstrated significant potential in achieving sub-micron-level high resolution. Recent research advances in this field have been quite encouraging, showcasing improved techniques and material compatibility.24–26 However, it is generally acknowledged that scaling these promising developments for large-scale industrial production remains a considerable challenge. In this context, the emergence of direct optical lithography of functional inorganic nanomaterials (DOLFIN), proposed by Talapin group in 2017, offers a ground breaking photoresist-free strategy for QD patterning and their integrated device applications.31 The core mechanism of this approach is altering the QD dispersibility through surface photochemical reactions of ligands via UV light stimulus. Compared to traditional lithography, DOLFIN, or more generally direct photopatterning process, is greatly simplified by consisting of only film deposition, UV exposure, and developing steps. This straightforward procedure, along with the elimination of harsh irradiation, unfavourable chemicals, and other detrimental processes, has established direct photopatterning as a highly promising method for QD patterning. Over the past years, a variety of photochemical mechanisms and designs for direct photopatterning have been developed by researchers across the world, utilizing ligand decomposition, desorption, crosslinking, and stripping mechanisms.31–67 These advancements have led to direct photopatterning techniques that preserve the optoelectronic properties of QDs and enabled the fabrication of high-performance QLED arrays.
Among these photochemistries, the ligand crosslinking approach is particularly noteworthy for making patterned QDs with high photoluminescent (PL) and electroluminescent (EL) performance.32–40 First, these ligand crosslinking chemistries involve QDs with well-passivated surface by their original or photocrosslinkable ligands, representing a nondestructive approach to preserve the luminescent properties of the QDs. Second, many groups including us have developed various ligand crosslinking methods for direct photopatterning of QDs and relevant pixelated devices. These studies include the use of crosslinkable ligands, dual ligands, and additive, nonspecific or specific crosslinkers. Data from these studies and rich chemical designs are critical for providing an in-depth summary of representative advances and key design principles of the photochemistries for direct photopatterning of QDs. Last but not least, rationally designed photocrosslinking chemistries have enabled full-colour, high-resolution (over 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 875 ppi),34 and precise patterning of luminescent QDs and NCs (II–VI, III–V, and lead halide based perovskites) with fully preserved PLQYs.33,34,36–38 The EL characteristics of patterned QLEDs made with these chemistries have also reached the stage of being comparable with their nonpatterned counterparts, with encouraging demos in passive- or active-matrix-driven QLED displays.34,36,38
875 ppi),34 and precise patterning of luminescent QDs and NCs (II–VI, III–V, and lead halide based perovskites) with fully preserved PLQYs.33,34,36–38 The EL characteristics of patterned QLEDs made with these chemistries have also reached the stage of being comparable with their nonpatterned counterparts, with encouraging demos in passive- or active-matrix-driven QLED displays.34,36,38
In this review, we highlight the remarkable progress made during the past five years in photocrosslinking-enabled direct photopatterning of QDs and their applications in QLEDs. Recently, several representative reviews and perspectives have been dedicated to summarizing these developments and elaborating on the relationship between patterning chemistry and device performance.41–48 Distinguished from previous work, this review offers a focused analysis in three key aspects: (1) a focused photocrosslinking pathway analysis. While covering representative types of direct photopatterning chemistries, we place particular emphasis on photocrosslinking pathways, which have demonstrated strong compatibility with passive- and active-matrix backplanes and enabled the fabrication of high-performance pixelated QLEDs. (2) From mechanism to patterned QLEDs. We systematically summarize design principles for photocrosslinkers toward highly luminescent QD patterns, review recent progress in directly photopatterned QLED performance, and provide guidelines for further development of patterning chemistries. (3) Extended applications and outlook. Beyond conventional 2D patterning, we also discuss the emerging role of photochemistry in 3D printing and offer perspectives on next-generation photocrosslinkers with functionalities that extend beyond patterning. In this review, we start by briefly introducing the mechanisms of photopatterning and representative types of photochemistries involved. Second, we provide a comprehensive survey of different types of photocrosslinking chemistries and their applications in QD patterning. Third, we propose key molecular design principles for photocrosslinkers toward high-performance QLEDs based on the data from studies on representative crosslinkers. Fourth, we extend the discussion to more recent advances on the crosslinking-based patterning of other functional nanomaterials (e.g. MOFs) and QD printing in three dimensions. Finally, we conclude with an outlook on future developments and the expanding applications of photocrosslinking-enabled patterning technique.
2 Fundamentals of direct photopatterning of colloidal NCs
2.1 Comparison of traditional and resist-free direct photolithography of NCs
Direct photopatterning relies on the interplay of surface photochemistry and colloidal stability of QDs. Photochemical reactions taken place on QD surface upon light or other stimuli change the QD dispersibility. The irradiation-induced solubility changes, as with traditional photolithography, set the basis of direct photopatterning. However, direct photopatterning does not involve photoresists, leading to much fewer steps than traditional photolithography, as shown in Scheme 1. Direct photolithography effectively preserves the intrinsic physicochemical and luminescent properties of patterned QDs by fundamentally avoiding the detrimental chemical exposures inherent in conventional photolithography—such as those from photoresists, developers, and etching agents.30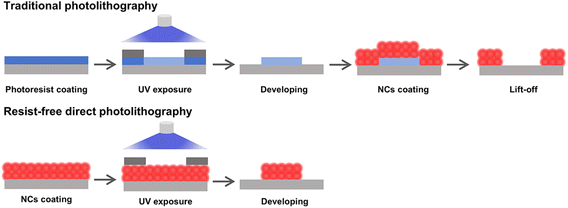 | ||
| Scheme 1 Comparison of patterning steps between traditional and resist-free direct photolithography. Both cases show the negative-tone patterning. | ||
2.2 Various photochemistries for NC patterning
The advancement of direct photopatterning for NCs is fundamentally rooted in the exploitation of specific photochemical reactions that modulate colloidal stability. These methods can be broadly categorized into four representative mechanistic classes (Scheme 2): ligand decomposition, ligand desorption, ligand crosslinking, and ligand binding, each employing a distinct pathway to cause solubility contrast of NCs between exposed and unexposed regions.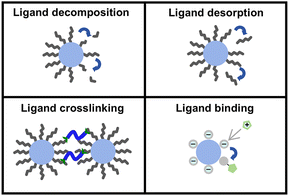 | ||
| Scheme 2 Four representative photochemistries for direct patterning, including ligand decomposition, ligand desorption, ligand crosslinking, and ligand binding. | ||
3 Photocrosslinking chemistries for direct photopatterning of NCs
Photocrosslinking-enabled direct patterning has emerged as an effective strategy to achieve nondestructive, high-definition, and high-throughput NC patterns. The core mechanism is to form covalent bonds between the ligands of neighbouring QDs upon photochemical reactions of crosslinkable ligands or additive crosslinkers. Here, three categories of representative photocrosslinking chemistries will be discussed and compared, including crosslinkable ligands, dual ligands, and additive crosslinkers.3.1 Rationale of photocrosslinking chemistry as an adaptive strategy
Ligand surface passivation is a critical determinant for both the high PLQY and the stability of QDs, serving as a key factor for their optoelectronic performance in device applications. Several patterning methods involve ligand exchange or ligand removal, processes that inevitably introduce surface defect states and trap sites, leading to pronounced PLQY degradation through nonradiative recombination. In contrast, photocrosslinking-enabled patterning, typically implemented via additive crosslinkers or photosensitive long-chain ligands, maintains high surface ligand coverage and effective passivation. More specifically, additive-crosslinker-based approach can retain the native ligands, which are already optimized for effective surface passivation during synthesis, thereby preserving the high luminescent properties of the as-prepared QDs.Beyond optical performance, photocrosslinking also offers distinct practical advantages for scalable manufacturing: (1) process compatibility and ease of operation. When using additive crosslinkers, the method requires no ligand exchange—only the direct addition of crosslinker to the original QD solution. This seamlessly integrates with existing commercial QD inks and established QLED fabrication processes. (2) Material stability and scalability. In crosslinking approaches, QDs remain protected either by their original organic ligands or by designed long-chain photocrosslinkable ligands, ensuring excellent colloidal stability and extended shelf lifetime—key attributes for reproducible large-scale production. (3) Solvent versatility and environmental adaptability. Through rational design of dispersing ligands or photocrosslinkable ligands, QDs can be dispersed in solvents spanning a wide polarity range, including environmentally friendly options.34,61 This tuneable solubility enhances compatibility with industrial printing and coating processes, further supporting the transition toward scalable manufacturing.
3.2 Different classes of photocrosslinking chemistry
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds in native ligands (e.g., oleylamine or oleic acid) on QDs, thereby promoting the formation of new C
C bonds in native ligands (e.g., oleylamine or oleic acid) on QDs, thereby promoting the formation of new C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C or C–C bonds between adjacent ligands and ultimately leading to QD crosslinking.52–54 However, the reliance on high-energy sources limits its application in large-scale, low-cost patterning, and the irradiation can severely quench the luminescence of QDs. In comparison, photochemically initiated ligand crosslinking operates under much gentler exposure conditions and demonstrates superior compatibility with standard photolithographic methods. Bang and co-workers developed a random semiconducting polymer ligand, PTPA-N3-SH, which integrates a triphenylamine-based hole-transporting moiety with a UV-crosslinkable azide group (Fig. 1a).59 The patterning capability of the method enables the fabrication of features as small as 5 µm. QLED devices based on the resulting patterned QDs achieved a maximum external quantum efficiency (EQEmax) of 6% and a maximum luminance (Lmax) exceeding 11
C or C–C bonds between adjacent ligands and ultimately leading to QD crosslinking.52–54 However, the reliance on high-energy sources limits its application in large-scale, low-cost patterning, and the irradiation can severely quench the luminescence of QDs. In comparison, photochemically initiated ligand crosslinking operates under much gentler exposure conditions and demonstrates superior compatibility with standard photolithographic methods. Bang and co-workers developed a random semiconducting polymer ligand, PTPA-N3-SH, which integrates a triphenylamine-based hole-transporting moiety with a UV-crosslinkable azide group (Fig. 1a).59 The patterning capability of the method enables the fabrication of features as small as 5 µm. QLED devices based on the resulting patterned QDs achieved a maximum external quantum efficiency (EQEmax) of 6% and a maximum luminance (Lmax) exceeding 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cd m−2, demonstrating both high resolution and promising electroluminescent performance. They further designed a poly(2-cinnamoyloxyethyl) methacrylate crosslinkable ligand which enables perovskite NC patterning by utilizing [2 + 2] cycloaddition reaction between adjacent olefin groups upon 365 nm UV irradiation.60 This photocyclization mechanism directly connects individual NCs into a crosslinked network structure. This dual-functional ligand enables direct photopatterning of lead halide-based perovskite NC films under UV light irradiation, achieving microscale feature sizes (<10 µm) while fully preserving their high luminescence.
000 cd m−2, demonstrating both high resolution and promising electroluminescent performance. They further designed a poly(2-cinnamoyloxyethyl) methacrylate crosslinkable ligand which enables perovskite NC patterning by utilizing [2 + 2] cycloaddition reaction between adjacent olefin groups upon 365 nm UV irradiation.60 This photocyclization mechanism directly connects individual NCs into a crosslinked network structure. This dual-functional ligand enables direct photopatterning of lead halide-based perovskite NC films under UV light irradiation, achieving microscale feature sizes (<10 µm) while fully preserving their high luminescence.
 | ||
| Fig. 1 Representative photocrosslinking chemistries for QD direct photopatterning. (a) Crosslinkable ligands.59 Adapted with permission from ref. 59. Copyright 2020 American Chemical Society. (b) Dual ligands, including crosslinking ligands and dispersing ligands.34 Adapted with permission from ref. 34. Copyright 2022 Springer Nature. (c) Nonspecific nitrene-based crosslinker additives.35,55 Adapted with permission from ref. 35 and 55. Copyright 2022 American Association for the Advancement of Science and 2020 Springer Nature. (d) Nonspecific carbene-based crosslinker additives.37 Adapted with permission from ref. 37. Copyright 2024 American Chemical Society. (e) Specific thiol-based crosslinker additives.56 Adapted with permission from ref. 56. Copyright 2023 American Association for the Advancement of Science. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ppi) under mild UV doses and with minimal photocrosslinker content, seamlessly integrating into industrial-standard microfabrication processes. The patterned QDs exhibit virtually no loss in intrinsic optical properties, while the fabricated QLEDs achieve high device performance, with EQE approaching 20% for red-emitting CdSe QLED devices. The approach further demonstrates excellent compatibility with scalable, large-area processing techniques, as exemplified by the uniform patterning of multicolour QDs across six-inch silicon wafers, underscoring its effectiveness for advanced photonic and display applications.
000 ppi) under mild UV doses and with minimal photocrosslinker content, seamlessly integrating into industrial-standard microfabrication processes. The patterned QDs exhibit virtually no loss in intrinsic optical properties, while the fabricated QLEDs achieve high device performance, with EQE approaching 20% for red-emitting CdSe QLED devices. The approach further demonstrates excellent compatibility with scalable, large-area processing techniques, as exemplified by the uniform patterning of multicolour QDs across six-inch silicon wafers, underscoring its effectiveness for advanced photonic and display applications.
Lately, Lee et al. introduces a bifunctional ligand/crosslinker, 4-(3-trifluoromethyl)-3H-diazirin-3-yl) benzoic acid (TDBA), which integrates both a carboxylate anchoring group and a photoactive diazirine moiety.63 By enabling a surface-anchored, direct crosslinking mechanism, this strategy not only circumvents the optical deterioration associated with ligand exchange but also significantly enhances crosslinking density and pattern fidelity. Moreover, Wang group recently developed a class of photosensitive ligands incorporating methacrylate and carboxyl groups that enable direct photopatterning of QDs in industrial compatible solvents such as PGMEA.61 Through ligand exchange, these dual-functional molecules anchor onto QD surfaces via their carboxyl groups, conferring colloidal stability in PGMEA while rendering the QDs photosensitive. Upon exposure to 365 or 405 nm light, the methacrylate units undergo crosslinking, enabling high-resolution patterning without the need for additional photo-polymerizable additives.
Replacing the original ligands on the QD surface with functional new ligands requires repeated solvent/anti-solvent washing steps and exposes the QDs to air, likely leading to a decline in colloidal stability and a reduction in luminescent performance. Thus Kwak et al. designed a bifunctional FPA-S ligand that integrates both photo-induced ligand exchange and photo-crosslinking capabilities.64 With a dihydrolipoic acid (DHLA) moiety at one end for robust anchoring onto QD surfaces via photoligation, and an azide group at the other end for photocrosslinking, this ligand enables rapid and stable in situ ligand exchange for InP-based QDs, realizing high-resolution QD patterning and enhanced performance in QLEDs.
Nonspecific crosslinkers offer broad compatibility with native ligand shells, enabling patterning across diverse QD compositions without requiring surface pre-modification. Certain nonspecific crosslinking strategies—such as those based on benzophenone or carbene chemistry—have been shown to be nondestructive, preserving QD luminescence without compromising efficiency. However, this generality often comes at the cost of lower reaction selectivity, which can lead to side reactions and potential quenching of QD luminescence, as noted in Section 4.1.4. In contrast, specific crosslinkers (e.g., thiol-ene click chemistry) provide high reaction selectivity and efficiency, which minimizes side reactions and byproducts. Nevertheless, their application usually necessitates QD surfaces to be pre-functionalized with complementary reactive groups, adding complexity to material synthesis and processing.
Common nonspecific crosslinker additives include nitrene- and carbene-based crosslinkers. Kang group reported a nitrene-based crosslinker, ethane-1,2-diyl bis(4-azido-2,3,5,6-tetrafluorobenzoate) (abbreviated as EBT), to achieve crosslinking QD patterns (Fig. 1c, left).55 EBT crosslinkers generate reactive nitrene intermediates upon 254 nm irradiation, which then undergo nonspecific C–H insertion reactions with the native long-chain ligands (such as oleylamines and oleic acids), thereby connecting adjacent QDs. This crosslinking mechanism enables photopatterning at ultralow exposure doses of only 2–3 mJ cm−2 while reaching a fine resolution of 2 µm. Importantly, the patterned QDs retain their original PLQY, and red-emitting devices fabricated with the crosslinked CdSe/CdZnS QD layers exhibit EQE as high as 14.6%, matching the performance of devices prepared from pristine QDs.
EBT crosslinkers can also enable direct photopatterning of perovskite NCs. Liu et al. developed a method named DOPPLCER, which achieved high-resolution patterning of perovskite NCs down to 5 µm (Fig. 1c, right).35 Upon exposure to 254 nm light at 60 mJ cm−2, the patterned perovskite NC film retained 63% of its original PLQY. Importantly, a post-treatment step further enhanced the relative PLQY retention to approximately 125%. When incorporated into light-emitting devices, the crosslinked FAPbBr3 perovskite LEDs realized a record EQE of 6.8% and luminance over 2 × 104 cd m−2, establishing new performance benchmarks for patterned perovskite LEDs at the time.
In parallel, our group developed a photosensitive carbene-based crosslinker 3,3′-(4,4′-(perfluorobutane-1,4-diyl) bis(4,1-phenylene)) bis(3-(trifluoromethyl)-3H-diazirine, which effectively undergo C–H insertion with neighbouring long-chain ligands under 365 nm exposure in nitrogen atmosphere (Fig. 1d).37 Compared to nitrene-based crosslinker, this carbene-based crosslinker is much milder and thus suitable for more fragile NCs such as perovskites. Thus this approach based on carbene-based crosslinker could retain 90% PLQY of pristine CsPbBr3 films. Crosslinked FAPbBr3 LEDs achieved a Lmax exceeding 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cd m−2 and an EQEmax of 16%. This represents a significant improvement of PL and EL performance over the DOPPLCER method using nitrene-based crosslinkers.
000 cd m−2 and an EQEmax of 16%. This represents a significant improvement of PL and EL performance over the DOPPLCER method using nitrene-based crosslinkers.
Specific crosslinkers react exclusively with particular functional groups (e.g., C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds) of surface ligands on QDs. A common strategy employs thiol-terminated crosslinkers that, under UV light, undergo a thiol–ene reaction with C
C bonds) of surface ligands on QDs. A common strategy employs thiol-terminated crosslinkers that, under UV light, undergo a thiol–ene reaction with C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C groups of the native ligands to form C–S covalent bonds. Cho group employed pentaerythritol tetrakis(3-mercaptopropionate) as a dual-role crosslinker, whose four terminal thiol groups generate radicals upon UV irradiation (Fig. 1e).56 These thiol radicals rapidly undergo photo-induced thiol-ene click reactions with the C
C groups of the native ligands to form C–S covalent bonds. Cho group employed pentaerythritol tetrakis(3-mercaptopropionate) as a dual-role crosslinker, whose four terminal thiol groups generate radicals upon UV irradiation (Fig. 1e).56 These thiol radicals rapidly undergo photo-induced thiol-ene click reactions with the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of native ligands on the perovskite NC surface, forming stable C–S linkages. By leveraging the inherent photocatalytic activity of the perovskite material itself, this system significantly enhances the reaction efficiency and lowers the required exposure dose to only ≈30 mJ cm−2 under 275 nm irradiation. Consequently, high-resolution multicolour patterning (<1 µm) is achieved with complete preservation of the luminescent properties. Moreover, the crosslinkers concurrently serve as a surface-passivating agent, further improving the PLQY and photo-stability of the patterned perovskite NCs. These thiol additives are also demonstrated to be applied to nondestructive patterning of InP QDs as well.62
C bonds of native ligands on the perovskite NC surface, forming stable C–S linkages. By leveraging the inherent photocatalytic activity of the perovskite material itself, this system significantly enhances the reaction efficiency and lowers the required exposure dose to only ≈30 mJ cm−2 under 275 nm irradiation. Consequently, high-resolution multicolour patterning (<1 µm) is achieved with complete preservation of the luminescent properties. Moreover, the crosslinkers concurrently serve as a surface-passivating agent, further improving the PLQY and photo-stability of the patterned perovskite NCs. These thiol additives are also demonstrated to be applied to nondestructive patterning of InP QDs as well.62
More recently, Lee group systematically compared dithiol crosslinkers TBBT and BPDT for patterning Cd-based QDs.65 Although both maintained high PLQY, BPDT-based devices achieved higher EQE (9.73% vs. 6.83%). DFT calculations revealed that BPDT's biphenyl structure enhances π-orbital overlap and charge injection, providing molecular-level insights for crosslinker design. Maeng et al. screened dithiol crosslinkers for perovskite NC patterning, finding that longer carbon chains (C8–C12) reduced the required concentrations of crosslinkers by two orders of magnitude.66 1,8-Octanedithiol and 1,10-decanedithiol emerged as optimal due to their solvent compatibility and stability. Combined with post-patterning exchanges, efficient perovskite NC LEDs were fabricated. The CsPbBr3 patterned devices reached a EQEmax of 14.7% and luminescence over 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cd m−2, while CsPbBrxI3−x patterned devices achieved a EQEmax of 13.1% and Lmax of 637 cd m−2.
400 cd m−2, while CsPbBrxI3−x patterned devices achieved a EQEmax of 13.1% and Lmax of 637 cd m−2.
 | ||
| Fig. 2 Representative patterns achieved via different photocrosslinking mechanisms, showing the patterning resolution, scalability, and fidelity. (a) Patterned via crosslinkable ligands containing azide or cinnamoyl groups.59,60 Adapted with permission from ref. 59 and 60. Copyright 2020 American Chemical Society and 2021 American Chemical Society. (b) Patterned via dual ligands, with crosslinking ligands containing benzophenone or methacrylate groups.34,38 Adapted with permission from ref. 34 and 38. Copyright 2022 Springer Nature and 2025 Springer Nature. (c) Patterned via nonspecific nitrene- or carbene-based crosslinkers.33,36,37,55,57 Adapted with permission from ref. 33, 36, 37, 55 and 57. Copyright 2022 Wiley-VCH, 2024 American Chemical Society, 2024 American Chemical Society, 2020 Springer Nature, and 2024 Springer Nature. (d) Patterned via specific thiol-based crosslinkers.56 Adapted with permission from ref. 56. Copyright 2023 American Association for the Advancement of Science. | ||
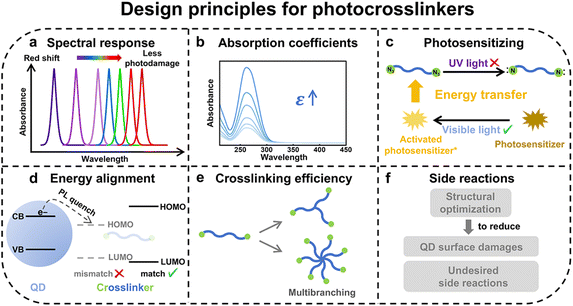
4 Design principles for photocrosslinkers toward high-performance pixelated QLEDs
The pursuit of high-performance patterned QLEDs requires photolithographic strategies that are effective in patterning while nondestructive toward the intrinsic optoelectronic properties of QDs. A central challenge is that the introduced photosensitive crosslinkers could trigger nonradiative recombination pathways at the QD surface and induce undesirable side reactions, compromising luminescent efficiency and charge transport properties. This section focuses on key chemical design principles for photocrosslinkers that minimize damage to QDs, preserve or even enhance QD properties, and enable the fabrication of high-performance, patterned QLEDs (Scheme 3). In the following, we take the widely studied nitrene- and carbene-based crosslinkers as examples to explain each of the design principles in detail and illustrate how chemical design addresses associated challenges and requirements with case studies.4.1 Design principles for photocrosslinkers
4.1.1.1 Conjugated group functionalization. In our earlier work, structural functionalization was attempted on nitrene-based crosslinkers by introducing conjugated groups to red-shift their absorption wavelength (Fig. 3a).32 Although this approach successfully shifted the absorption maximum from 254 nm to 365 nm, it concurrently lowered the electronic energy levels of the crosslinkers, which facilitated electron transfer from the QDs, thereby inducing luminescence quenching.
 | ||
| Fig. 3 Comparison of nitrene- and carbene-based crosslinkers. (a) Molecular structures of nitrene- and carbene-based crosslinkers designed for spectral shifting.32 (b) Different photochemitries of nitrene- and carbene-based crosslinkers.32 (c) Efficacy of nitrene- and carbene-based crosslinkers in maintaining the PL properties of QDs.32 Adapted with permission from ref. 32. Copyright 2022 Wiley-VCH. | ||
4.1.1.2 Photocrosslinkers with extended absorption. Beyond functionalizing deep UV-sensitive crosslinkers towards longer-wavelength absorption, an alternative approach involves exploring other classes of photocrosslinkers. For instance, our group developed carbene-based crosslinkers for nondestructive QD patterning and systematically investigated their photochemical process and impacts on QD properties (Fig. 3a and b).32 Upon light irradiation, carbene-based crosslinkers generate reactive carbene intermediates, which undergo nonspecific C–H insertion with the native ligands of QDs to achieve crosslinked patterns. Compared to nitrene-based crosslinkers, carbene-based crosslinkers possess longer-wavelength spectral response. The extended absorption spectrum of carbene-based crosslinkers (200−400 nm) enables QD patterning at longer wavelengths (e.g., 365 nm, i-line), thereby reducing UV-induced photodamage to QDs. This also aligns with optoelectronic characterization results. Carbene-based patterning of Cd-based QD films achieves ≈90% PLQY retention, whereas azide-based patterning only retains ≈55% PLQY (Fig. 3c).
4.1.1.3 Photosensitizing. We proposed another photosensitizer-assisted approach for direct QD photopatterning, which employs Ir(tBu-ppy)3 to absorb h-line (405 nm) light and activate nitrene-based crosslinkers via Dexter energy transfer, generating nitrene intermediates for ligand crosslinking (Scheme 3c and Fig. 4a, right).57 This approach successfully red-shifted the required wavelength of nitrene-based crosslinkers from deep ultraviolet (254 nm) to the h-line (405 nm), enabling high-efficiency and high-resolution patterning with a minimum pixel size of 2 µm. The patterned QD film retained approximately 90% of its pristine PLQY with an improvement of over 30% in PLQY retention compared to samples patterned upon deep UV irradiation.
 | ||
| Fig. 4 Optimization of nitrene- and carbene-based crosslinkers to address the proposed design principles. (a and b) Optimization for (a) nitrene- and (b) carbene-based crosslinkers for minimizing damages to QDs.33,36,57,70 Adapted with permission from ref. 33, 36, 57 and 70. Copyright 2022 Wiley-VCH, 2024 American Chemical Society, 2024 Springer Nature, and 2025 Springer Nature. | ||
In contrast, carbene intermediates, due to high energy barriers, are less prone to such side reactions. Carbene-based ligands have been reported to effectively passivate the QD surfaces, thereby maintaining the optical properties of QD films throughout the patterning process.74,75 Although carbene-based crosslinkers do not produce harmful byproducts, their propensity for side reactions with oxygen substantially limits their practical applicability in QLED manufacturing. As shown in Fig. 3b, singlet carbene intermediates (1PC) are generated upon UV irradiation and further undergo C–H insertion between surface ligands to achieve patterning of QDs. Nevertheless, due to the small singlet–triplet energy gap, 1PC undergoes rapid intersystem crossing to the triplet state (3PC). The resulting 3PC is readily quenched by oxygen (3O2). In ambient air, this side reaction becomes the dominant pathway, which depletes the 1PC population and leads to an unacceptably low efficiency of the C–H insertion reaction. The results of DFT calculations, Hammett substituent parameter analysis, and electron paramagnetic resonance (EPR) experiments revealed that introducing electron-donating groups (EDGs) at the para-position of the diazirine moiety effectively lowers the energy of 1PC and induces an inversion of the singlet-triplet energy (Fig. 4b).36 Consequently, 3PC becomes the excited state, which means the photogenerated 1PC no longer readily converts to the oxygen-sensitive 3PC. Instead, it preferentially undergoes the desired C–H insertion reaction for QD patterning. Based on this mechanism, carbene-based crosslinkers functionalized with electron-donating alkoxy substituents were designed. This enabled the nondestructive patterning of both Cd-based and Cd-free QD films under 365 nm irradiation in an ambient atmosphere, without compromising their optoelectronic performance.
The discussions of above principles primarily use nitrene- and carbene-based crosslinkers as examples. However, these outlined principles and chemical modification strategies are also applicable to other types of crosslinkers. Based on DFT-guided design, Kang and co-workers structurally engineered benzophenone-based crosslinkers by substituting EDGs (–N(CH2)4 and –S–) at the para-positions. This modification resulted in a red-shifted absorption into the UV-A region and a significantly enhanced extinction coefficient from 60 M−1 cm−1 to 2.02 × 104 M−1 cm−1 at 365 nm.34 As for design of thiol-based crosslinkers, guided by DFT calculations, Lee and co-workers revealed that conjugation extension could enhance orbital overlap and reduce the bandgap of the crosslinker, leading to improved charge transport within the QD film. Meanwhile, frontier energy-level alignment lowers the injection barrier between the crosslinker and the QDs by matching their HOMO/LUMO levels, thereby facilitating efficient carrier injection. Together, these two design strategies synergistically preserve the PLQY while substantially boosting the EL efficiency of the resulting devices, as evidenced by the higher EQE achieved with BPDT-based crosslinkers compared to TBBT-based ones.65 Moreover, as demonstrated in the work of Cho's group, systematic molecular engineering—including the selection of dithiol functionalities, optimization of alkyl chain length, and modulation of polarity—led to the design of ODT and DDT as efficient thiol crosslinkers. Their tailored structure ensures high colloidal stability in formulated inks, enables effective liquid-crystal-assisted patterning, and promotes photocatalytic crosslinking under low UV doses.66
4.2 State-of-the-art of direct photopatterned QLEDs using photocrosslinkers
Based on the systematic understanding of molecular design principles and optimization strategies for crosslinkers, research has progressively advanced toward developing designs of crosslinkers that are better aligned with luminescent properties of QDs and their practical application requirements. This progress has enabled photocrosslinking-based patterned QLEDs to achieve significant performance enhancements, gradually approaching state-of-the-art non-patterned device performance. The latest advancements have further applied these photocrosslinking patterning techniques to QLED fabrication, successfully realizing high-performance patterned devices (Table 1).| Photochemistries | QDs (color) | L max (cd m−2) | EQEmax | CEmax (cd A−1) | Lifetime (h) | Ref. |
|---|---|---|---|---|---|---|
| a Contents marked with * represents data measured from pixelated QLEDs. Device performance is co-determined by materials chemistry (QDs, ligands, crosslinkers) and device engineering (charge transport layers, interfaces, device architecture) and thus emphasizing performance improvement within the same method is more meaningful than making absolute comparisons across different methods and device systems. | ||||||
| Patterned II−VI QLEDs | ||||||
| PTPA-N3-SH crosslinkable ligands | CdSe/ZnCdS (R) | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 720 720 |
6.3% | 5.7 | N/A | 59 |
| MMPE/MEMA/MMES crosslinkable ligands | CdSe/ZnS (R) | 104−105 | 22.0% | N/A | N/A | 61 |
| CdSe/ZnS (G) | 104−105 | 14.4% | ||||
| CdSe/ZnS (B) | 103−104 | 11.4% | ||||
| Dual ligands (benzophenone) | CdSe/CdZnSe/ZnSeS (R) | ≈105 | ≈20% | N/A | N/A | 34 |
| CdSe/CdZnSe/ZnSeS (G) | ≈105 | ≈15% | ||||
| Dual ligands (TPP) | CdSe/ZnS (R) | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 103 103 |
20.2% | 25.1 | T95@1 k = 9280 | 38 |
| CdSe/ZnS (G) | 57![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 326 326 |
25.6% | 94.7 | T95@1 k = 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 261 261 |
||
| CdSe/ZnS (B) | 7472 | 21.6% | 12.5 | T95@1 k = 135 | ||
| Dual ligands (diazirines) | CdZnSe (R) | 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 369 369 |
10.3% | N/A | N/A | 63 |
| Dual ligands (azobenzene) | CdZnSe/CdZnS/ZnS (R) | ≈3 × 103 | 8.2% | N/A | T95@1 k = 970 | 102 |
| Nitrene-based crosslinkers | CdSe/CdZnSe/ZnSeS (R) | 105−106 | 14.6% | N/A | T95@10 k = 10 | 55 |
| CdSe/CdZnSeS (G) | ≈105 | ≈6% | N/A | |||
| CdZnS/ZnS (B) | 103−104 | ≈3% | N/A | |||
| Nitrene-based crosslinkers | CdZnSe/CdZnS/ZnS (R) | 6693 | 12.6% | N/A | T95@1 k = 1823 | 32 |
| Rigid nitrene-based crosslinkers | CdSe QDs (R) | 179![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 872 872 |
21.1% | 23.3 | T95@10 k = 3.1 | 70 |
| CdSe QDs (G) | 244![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 884 884 |
9.6% | 40.9 | N/A | ||
| CdSe QDs (B) | 4939 | ≈2% | 1.47 | N/A | ||
| Carbene-based crosslinkers | CdZnSe/CdZnS/ZnS (R) | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
11.7% | N/A | T95@1 k = 4852 | 32 |
| Benzonphenone-based crosslinkers | CdSe/ZnS (R) | 109![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 879 879 |
16.5% | N/A | T95@1 k = 2258 | 76 |
| Azide-alkyne click chemistry | CdZnSe/ZnSe (R) | 166![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 612 612 |
20.1% | N/A | T95@1 k = 582 | 40 |
| CdSe/ZnSe/ZnS (G) | 105−106 | 15.4% | N/A | |||
| CdZnSe/ZnSe/ZnS (B) | 104−105 | 8.5% | N/A | |||
| Azide-alkyne click chemistry* | CdZnSe/ZnSe (R) | 104−105 | ≈9% | N/A | N/A | 40 |
| CdSe/ZnSe/ZnS (G) | ≈105 | ≈6% | ||||
| CdZnSe/ZnSe/ZnS (B) | ≈104 | ≈3% | ||||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Patterned III−V QLEDs | ||||||
| Dual ligands (FPA-S) | InP/ZnSe/ZnS (R) | 105−106 | N/A | 4.3 | T95@150 = 950 | 64 |
| Nitrene-based crosslinkers | InP/ZnSeS (G) | ≈4000 | 8.3% | N/A | T90@1k = 156 | 33 |
| EDG-modified carbene-based crosslinkers | InP/ZnSe/ZnS (R) | 39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 590 590 |
15.3% | 20.2 | N/A | 36 |
| EDG-modified carbene-based crosslinkers* | InP/ZnSe/ZnS (R) | ≈4000 | 13.3% | 20.8 | N/A | 36 |
| Thiol-ene click chemistry | InP/ZnS (G) | 8463 | N/A | 23.0 | N/A | 62 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||||
| Patterned perovskite NC LEDs | ||||||
| Dual ligands (methacrylate) | CsPbBr3 (G) | 51![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 181 181 |
18.6% | N/A | T50 = 1.95 | 77 |
| Dual ligands (acrylate) | CsPbBr3 (G) | <100 | 1% | N/A | N/A | 86 |
| Nitrene-based crosslinkers | FAPbBr3 (G) | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
6.8% | 28.5 | N/A | 35 |
| CsPbBr3 (G) | 1929 | 1.8% | 5.6 | |||
| Carbene-based crosslinkers | FAPbBr3 (G) | 64![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 726 726 |
16% | 60 | T50@100 = 26 | 37 |
| Thiol-ene click chemistry | CsPbBr3 (G) | ≈100 | ≈2.5% | N/A | N/A | 56 |
| Thiol-ene click chemistry* | CsPbBr3 (G) | 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 968 968 |
13.1% | N/A | N/A | 78 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cd m−2.70
000 cd m−2.70
 | ||
| Fig. 5 Representative examples of patterned QLEDs achieved by nitrene-based crosslinkers. (a) Cd-based patterned QLED via nitrene-based crosslinkers.55 Adapted with permission from ref. 55. Copyright 2020 Springer Nature. (b) CsPbBr3 LED via nitrene-based crosslinkers.35 Adapted with permission from ref. 35. Copyright 2022 American Association for the Advancement of Science. (c) InP-based patterned QLED via structurally optimized nitrene-based crosslinkers.33 Adapted with permission from ref. 33. Copyright 2022 Wiley-VCH. | ||
 | ||
| Fig. 6 Representative examples of patterned QLEDs achieved by carbene-based crosslinkers. (a) Cd-based QLED via carbene-based crosslinkers under inert atmosphere.32 Adapted with permission from ref. 32. Copyright 2022 Wiley-VCH. (b) FAPbBr3 LED via carbene-based crosslinkers under inert atmosphere.37 Adapted with permission from ref. 37. Copyright 2024 American Chemical Society. (c) InP-based QLED via electronically optimized carbene-based crosslinkers under ambient atmosphere.36 Adapted with permission from ref. 36. Copyright 2024 American Chemical Society. | ||
As for specific crosslinkers, Lee's group employed BPDT based on thiol-ene click chemistry to fabricate high-performance patterned CdSe-based QLEDs.65 The resulting devices retained the intrinsic optoelectronic properties of the QDs while achieving an EQEmax of 9.73% and a high luminance of 10, 000 cd m−2. Notably, the current efficiency and power efficiency of the BPDT-patterned QLEDs were nearly identical to those of the non-patterned reference devices, demonstrating that this crosslinking strategy enables high-resolution patterning without compromising device performance. Besides, Zhang and colleagues developed red, green, and blue monochromatic patterned QLEDs based on the light-triggered azide-alkyne cycloaddition reaction.40 In this system, 10-undecynoic acid was used as the alkyne-functionalized photosensitive ligand to replace native oleic acid, while 4,4′-bis(azidomethyl)biphenyl served as the photocrosslinkers. The resulting patterned devices outperformed their non-patterned counterparts, achieving a EQEmax of 20.05% (red, CdZnSe/ZnSe), 15.37% (green, CdSe/ZnSe/ZnS), and 8.49% (blue, CdZnSe/ZnSe/ZnS), compared to 18.15%, 14.15%, and 7.02% for the corresponding prototype devices, respectively.
Similarly, dual-ligand strategy has also enabled high-performance patterned QLEDs. Hahm et al. constructed patterned QLEDs using dual-ligand CdSe/CdxZn1−xSe/ZnSeySy−1 QDs, achieving device performance comparable to that of prototype devices with an EQEmax of approximately 20% (Fig. 7c).34 Furthermore, they fabricated a 10 × 10 passive matrix full-colour RGB display prototype (PM-QLED), demonstrating the practical application potential of this patterning method in the field of optoelectronic displays (Fig. 8a). In recent work, Lee and colleagues employed TDBA as PXLs.63 Using this bifunctional molecule, high-resolution (≈2 µm) CdZnSe-based patterned QLEDs were successfully fabricated, achieving a Lmax of 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 369 cd m−2 and a peak EQE of 10.3%. These results demonstrate the effectiveness of TDBA-based ligand design for producing high-performance patterned QLEDs. The Wang group developed red-emitting patterned QLEDs using methacrylate-anchored CdSe/ZnS QDs that are processable in the industry-standard solvent PGMEA, achieving a peak EQE of 22% and a Lmax exceeding 40
369 cd m−2 and a peak EQE of 10.3%. These results demonstrate the effectiveness of TDBA-based ligand design for producing high-performance patterned QLEDs. The Wang group developed red-emitting patterned QLEDs using methacrylate-anchored CdSe/ZnS QDs that are processable in the industry-standard solvent PGMEA, achieving a peak EQE of 22% and a Lmax exceeding 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cd m−2 (Fig. 7d).61 More recently, they reported another strategy using TPP ligand as photoinitiator to initiate a chain-propagation reaction, leading to the crosslinking of the QDs. The resulted red-, green-, and blue-emitting CdSe/ZnS patterned QLED achieves EQEmax of 20.2%, 25.6%, and 21.6% respectively.38 As shown in Fig. 8c, this method was further demonstrated in fabrication of an active-matrix QD display prototype (AM-QLED).
000 cd m−2 (Fig. 7d).61 More recently, they reported another strategy using TPP ligand as photoinitiator to initiate a chain-propagation reaction, leading to the crosslinking of the QDs. The resulted red-, green-, and blue-emitting CdSe/ZnS patterned QLED achieves EQEmax of 20.2%, 25.6%, and 21.6% respectively.38 As shown in Fig. 8c, this method was further demonstrated in fabrication of an active-matrix QD display prototype (AM-QLED).
 | ||
| Fig. 7 Representative examples of patterned QLEDs achieved by thiol-based crosslinkers and dual ligand strategy. (a) InP-based QLED via thiol-based crosslinkers.62 Adapted with permission from ref. 62. Copyright 2023 Wiley-VCH. (b) CsPbBr3 LED via thiol-based crosslinkers.56 Adapted with permission from ref. 56. Copyright 2023 American Association for the Advancement of Science. (c and d) Cd-based QLEDs via dual ligand strategy.34,61 Adapted with permission from ref. 34 and 61. Copyright 2022 Springer Nature and 2025 American Chemical Society. | ||
 | ||
| Fig. 8 Active- and passive-matrix displays of photocrosslinked QD patterns. (a) PM-QLED of CdSe-based QDs via dual ligands.34 Adapted with permission from ref. 34. Copyright 2022 Springer Nature. (b) AM-QLED of InP-based QDs via carbene-based crosslinkers.36 Adapted with permission from ref. 36. Copyright 2024 American Chemical Society. (c) AM-QLED of RGB QDs via TPP ligands.38 Adapted with permission from ref. 38. Copyright 2025 Springer Nature. | ||
Photocrosslinking approach is also a compatible patterning method for charge-transport layers in devices. For instance, Li et al. fabricated full-color pixelated CdSe-based QLEDs with an EQE of 11% by combining a region-selective assembly process with photocrosslinking lithography, using the nitrene-based crosslinker 4,4′-dithiobis(phenylazide).26 Instead of patterning QD films, Zhang also presented an alternative strategy of creating high-quality photocrosslinked TFB films with ordered molecular arrangement using a nitrene-based crosslinker 2,6-bis(4-azidobenzylidene)-4-methylcyclohexanone (AMBK).19 Red-emitting CdSe/ZnS QLED devices incorporating the photocrosslinked TFB layers demonstrated significantly enhanced performance, achieving an EQEmax of 24.6% and a current efficiency (CE) of 24.3 cd A−1. The device lifetime (T95) was markedly improved from 460 to 1256 hours. Furthermore, the feasibility of this approach was also demonstrated by the successful fabrication of pixelated QLED devices using patterned TFB layers.
Kang group developed structurally optimized nitrene-based non-specific crosslinkers IP-6-Lixer to construct green-emitting InP/ZnSeS patterned devices, with an EQEmax of 8.3% and a luminance over 1000 cd m−2 (Fig. 5c).33 Oh group utilized thiol-based specific crosslinkers PTMP to build InP/ZnSe/ZnS patterned devices via a thiol-ene reaction (Fig. 7a).62 The resulted devices achieved a Lmax of 8463 cd m−2 and a peak current efficiency of 23 cd A−1. Besides, Seo et al. has utilized FPA-S as PXLs to successfully fabricate red-emitting InP/ZnSe/ZnS patterned QLED featuring a CEmax of 4.3 and a Lmax over 105−106 cd m−2.64
Recently, our group developed electronically optimized carbene-based crosslinkers, Tris-TAD, which are activated by i-line UV irradiation to generate air-stable singlet carbenes that directly link adjacent QDs via concerted C–H insertion into the surface ligands. The fabricated InP/ZnSe/ZnS patterned QLEDs achieved a EQEmax of 15.3% and Lmax over 39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 590 cd m−2, establishing a record performance for patterned InP-based QLEDs (Fig. 6c).36 Based on this method, we further fabricated an AM-QLED using Tris-TAD (Fig. 8b). The InP-based QD film was integrated with a pixelated thin-film transistor (TFT) array (backplane: 16 × 16 TFT array), where each patterned QD film was aligned precisely with its underlying TFT. This architecture enabled precise selective control of charge injection into individual pixels (size: 380 × 650 µm) via Bluetooth from a mobile device, without interference from adjacent pixels or leakage current. Importantly, the performance of individual pixels of AM-QLED was also characterized, exhibiting a CE of 20.8 cd A−1 and an estimated peak EQE of 13.3%. These values closely match those obtained from QLED devices based on full crosslinked film. This consistency of pixel-level device has validated the technical feasibility of a transition path for QD-integrated devices to industrial applications.
590 cd m−2, establishing a record performance for patterned InP-based QLEDs (Fig. 6c).36 Based on this method, we further fabricated an AM-QLED using Tris-TAD (Fig. 8b). The InP-based QD film was integrated with a pixelated thin-film transistor (TFT) array (backplane: 16 × 16 TFT array), where each patterned QD film was aligned precisely with its underlying TFT. This architecture enabled precise selective control of charge injection into individual pixels (size: 380 × 650 µm) via Bluetooth from a mobile device, without interference from adjacent pixels or leakage current. Importantly, the performance of individual pixels of AM-QLED was also characterized, exhibiting a CE of 20.8 cd A−1 and an estimated peak EQE of 13.3%. These values closely match those obtained from QLED devices based on full crosslinked film. This consistency of pixel-level device has validated the technical feasibility of a transition path for QD-integrated devices to industrial applications.
In 2022, our group has developed DOPPLCER method using nitrene-based crosslinkers for perovskite NCs patterning and further demonstrated patterned PeLEDs employing CsPbBr3 which established then-record performance metrics with an EQEmax of 6.8% and Lmax over 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cd m−2 (Fig. 5b).35 To further enhance the performance of PeLEDs, we employed carbene-based crosslinkers to fabricate patterned FAPbBr3 PeLEDs (Fig. 6b).37 These devices achieved an EQEmax of 16% and a Lmax over 60
000 cd m−2 (Fig. 5b).35 To further enhance the performance of PeLEDs, we employed carbene-based crosslinkers to fabricate patterned FAPbBr3 PeLEDs (Fig. 6b).37 These devices achieved an EQEmax of 16% and a Lmax over 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cd m−2, representing a significant improvement over counterparts patterned using nitrene-based crosslinkers. The results underscore the milder reactivity and enhanced compatibility of carbene-based crosslinking with fragile perovskite NCs.
000 cd m−2, representing a significant improvement over counterparts patterned using nitrene-based crosslinkers. The results underscore the milder reactivity and enhanced compatibility of carbene-based crosslinking with fragile perovskite NCs.
Maeng et al. presented a photocatalytic patterning method based on thiol-ene click reaction to fabricate CsPbBr3 patterned LED, achieving EQEmax of ≈2.5% and Lmax≈100 cd m−2 (Fig. 7b).56 More recently, they reported a dual strategy combining ligand re-engineering and anion exchange to achieve high-performance patterned PeLEDs.66 High-resolution, nondestructive photocatalytic patterning was first realized using optimized thiol-based crosslinkers (ODT/DDT), followed by a film-state ligand-exchange process that further enhanced the optoelectronic properties of the patterned films. As a result, green CsPbBr3 patterned devices reached a peak EQE of 14.7% with a luminance of ≈25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cd m−2, while red CsPbBrxI3−x patterned devices achieved an EQE exceeding 13.1%, marking the first demonstration of directly photopatterned red perovskite LEDs.
400 cd m−2, while red CsPbBrxI3−x patterned devices achieved an EQE exceeding 13.1%, marking the first demonstration of directly photopatterned red perovskite LEDs.
5 Extension of photocrosslinking to other types of nanomaterials and 3D printing
Beyond QDs, photocrosslinking-enabled patterning also provides an effective pathway for the patterning of other types of nanomaterials, such as MOFs, and offers viable solutions for 3D printing of inorganic nanomaterials.5.1 Direct photopatterning of MOFs
MOFs are a class of porous crystalline materials formed by the self-assembly of metal ions/clusters and organic linkers. They possess exceptional surface areas, tuneable pore architectures, and compositional diversity.87 These properties have spurred their recent adoption in microelectronics and nanophotonics.88–90 Integrating photocrosslinking chemistry with colloidal MOFs processing, we developed a universal methodology for patterning MOFs (CLIP-MOF).91 This technique enables large-area (e.g., 4-inch wafer) patterning of diverse MOFs (including ZIF-8, ZIF-7, HKUST-1, and UiO-66) with micro-to nanoscale resolution (≈5 µm with UV; ≈70 nm with electron-beam lithography) on rigid and flexible substrates, in both single- and multi-component configurations (Fig. 9a). | ||
| Fig. 9 Photocrosslinking chemistries in patterning of colloidal MOFs and 3D nanoprinting of QDs. (a) Direct patterning of MOFs via nitrene-based crosslinkers.91 Adapted with permission from ref. 91. Copyright 2024 Springer Nature. (b) Direct patterning of ZIFs via photoinduced fluorination.93 Adapted with permission from ref. 93. Copyright 2025 Wiley-VCH. (c and d) 3D printing of QDs via PEB (c) and 3D Pin (d).99,100 Adapted with permission from ref. 99 and 100. Copyright 2022 American Association for the Advancement of Science and 2023 American Association for the Advancement of Science. | ||
The mechanism of CLIP-MOF is similar with direct photopatterning of QDs: both exploit photo-induced changes in colloidal stability. Alternatively, the inherent chemical lability of MOFs towards stimuli like acids, bases, or water can also be harnessed for direct lithography. Leveraging this, Tu group introduced a method using photoacid generators (PAGs).92 UV exposure liberates protons from PAGs, which subsequently decompose the MOFs in irradiated areas—functioning as a positive-tone photoresist—to yield patterns with a resolution of ≈2 µm while preserving the material's intrinsic properties. Our group devised a complementary, negative-tone strategy based on photo-induced fluorination of zeolitic imidazolate frameworks (ZIFs) using methyl 4-azido-2,3,5,6-tetrafluorobenzoate (ATFB).93 Rather than destructively decomposing MOFs with high-dose radiation, this method uses an ultra-low dose (254 nm, 10 mJ cm−2) to covalently graft ATFB onto the ZIF ligands. This fluorination enhances the water stability of the exposed regions, allowing the unexposed, pristine ZIFs to be developed away with water/ammonia solution (Fig. 9b). This technique is generalizable across several water-labile ZIFs (ZIF-8, ZIF-7, ZIF-67, ZIF-L), achieving feature sizes of ≈2 µm.
Critically, these direct lithographic methods maintain the structural integrity, chemical composition, and porosity of the original MOFs. The resulting patterned MOF films show promise for transformative applications in diffraction-based gas sensing, pixelated electrochromic devices, fluorescent sensor arrays, information encryption, and intelligent, multiplexed sensing platforms.
5.2 3D nanoprinting of QDs
3D printing is a transformative fabrication technology that enables the creation of complex three-dimensional structures with customizable material compositions, thereby unlocking new functionalities across advanced fields such as aerospace, energy materials, bioengineering, and personalized manufacturing.94–97 Particularly, the introduction of femtosecond laser-based techniques has pushed the resolution into the nanoscale, allowing the fabrication of intricate 3D architectures with nanoscale precision and unprecedented functional integration.Semiconductor inorganic materials are indispensable in integrated circuits and photonic chips due to their superior optoelectronic properties. The ability to 3D print semiconductors and other inorganics could offer a viable pathway for out-of-plane processing of integrated chips. However, current 3D printing is largely confined to polymers and metals, lacking effective methods for micro/nano-scale printing of semiconductors and many other inorganic materials.98 To address this challenge, Liu et al. presented a strategy termed photoexcitation-induced chemical bonding (PEB), which utilizes electron–hole pairs generated by photoexcitation to modify QD surface chemistry and further trigger interparticle chemical bonding (Fig. 9c).99 In this process, mercaptopropionic acid (MPA) ligands are attached to CdSe/ZnS core/shell QDs via thiol coordination, leaving their carboxylic acid groups exposed to the solvent. When irradiated with a femtosecond laser at 780 nm through two-photon absorption, the excited QDs produce charge carriers whose energy levels promote the oxidation and removal of the bound MPA ligands. The newly created coordination-deficient sites on the QD surface then selectively bind to carboxylate terminals from MPA ligands anchored on adjacent QDs, forming robust inter-dot bridges that lock the assembly into a precise 3D network. As a result, high-accuracy, high-resolution 3D structures (feature sizes down to 80 nm) of QDs with photonic properties maintained were achieved using this method.
Furthermore, Li et al. has leveraged photo-crosslinking chemistry to develop a universal 3D printing method for inorganic nanomaterials (3D Pin).100 The 3D Pin process employs NC solutions as inks, into which an azide-based crosslinker is incorporated. Under femtosecond laser irradiation, the decomposition of the crosslinker generates nonspecific C–H insertion reactions, leading to covalent crosslinking between the organic ligands capping the NCs (Fig. 9d). The nonspecificity of the nitrene intermediate insertion renders this method universally applicable to diverse materials, including semiconductors (e.g., II–VI, III–V, metal halide perovskites), metals (e.g., Au), and metal oxides (e.g., In2O3, TiO2). Furthermore, by blending different NC solutions, hybrid tetrahedral structures comprising CdSe/ZnS, TiO2, PbS/CdS, and In2O3 have been successfully printed. The 3D Pin technique achieves nanoscale resolution (≈150 nm), enabling the fabrication of arbitrarily complex 3D architectures. The printed structures exhibit a high inorganic mass fraction of approximately 90%, paving a promising fabrication route for applications in 3D holographic displays and integrated 3D optoelectronic devices.
6 Conclusion and outlook
6.1 Conclusion
This review surveys the progress of photocrosslinking chemistry for directly patterning colloidal QDs. We focus on the key design principles driving this field toward a critical application of high-performance, pixelated QLEDs. The evolution from simple proof-of-concept to advanced patterning methods is rooted in a deeper understanding of the underlying photochemistry. The field now recognizes that the molecular design of ligands and crosslinkers must achieve dual objectives: ensuring high-resolution patterning while preserving the optoelectronic properties of the QDs. This dedicated focus on “nondestructive” photocrosslinking chemistry has contributed to the successful fabrication of patterned QLEDs with continuously improving optoelectronic performance. These encouraging advances suggest the great promise of photocrosslinking-based approaches for incorporating QDs into integrated optoelectronic platforms, such as high-resolution displays and advanced photonics.6.2 Outlook
Author contributions
W. Q. wrote the draft and prepared figures of this review article. H. Z. supervised the preparation of the draft, edited and co-revised the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was financially supported by the Beijing Natural Science Foundation (JQ24003) (H. Z.), the Natural Science Foundation of China (No. 22274087) (H. Z.), and Tsinghua University Dushi Program (H. Z.).Notes and references
- A. P. Alivisatos, Semiconductor Clusters, Nanocrystals, and Quantum Dots, Science, 1996, 271, 933–937 CrossRef CAS.
- D. V. Talapin, J.-S. Lee, M. V. Kovalenko and E. V. Shevchenko, Prospects of Colloidal Nanocrystals for Electronic and Optoelectronic Applications, Chem. Rev., 2010, 110, 389–458 CrossRef CAS PubMed.
- M. V. Kovalenko, L. Manna, A. Cabot, Z. Hens, D. V. Talapin, C. R. Kagan, V. I. Klimov, A. L. Rogach, P. Reiss, D. J. Milliron, P. Guyot-Sionnnest, G. Konstantatos, W. J. Parak, T. Hyeon, B. A. Korgel, C. B. Murray and W. Heiss, Prospects of Nanoscience with Nanocrystals, ACS Nano, 2015, 9, 1012–1057 CrossRef CAS PubMed.
- C. R. Kagan, E. Lifshitz, E. H. Sargent and D. V. Talapin, Building Devices from Colloidal Quantum Dots, Science, 2016, 353, aac5523 CrossRef PubMed.
- J. M. Pietryga, Y.-S. Park, J. Lim, A. F. Fidler, W. K. Bae, S. Brovelli and V. I. Klimov, Spectroscopic and Device Aspects of Nanocrystal Quantum Dots, Chem. Rev., 2016, 116, 10513–10622 CrossRef CAS PubMed.
- F. P. García de Arquer, D. V. Talapin, V. I. Klimov, Y. Arakawa, M. Bayer and E. H. Sargent, Semiconductor Quantum Dots: Technological Progress and Future Challenges, Science, 2021, 373, eaaz8541 CrossRef PubMed.
- A. L. Efros and L. E. Brus, Nanocrystal Quantum Dots: From Discovery to Modern Development, ACS Nano, 2021, 15, 6192–6210 CrossRef CAS PubMed.
- S. Coe, W.-K. Woo, M. Bawendi and V. Bulović, Electroluminescence from Single Monolayers of Nanocrystals in Molecular Organic Devices, Nature, 2002, 420, 800–803 CrossRef CAS.
- Y. Shirasaki, G. J. Supran, M. G. Bawendi and V. Bulovic, ́ Emergence of Colloidal Quantum-Dot Light-Emitting Technologies, Nat. Photonics, 2013, 7, 13–23 CrossRef CAS.
- X. Dai, Z. Zhang, Y. Jin, Y. Niu, H. Cao, X. Liang, L. Chen, J. Wang and X. Peng, Solution-Processed, High-Performance Light Emitting Diodes Based on Quantum Dots, Nature, 2014, 515, 96–99 CrossRef CAS PubMed.
- X. Dai, Y. Deng, X. Peng and Y. Jin, Quantum-Dot Light-Emitting Diodes for Large-Area Displays: Towards The Dawn of Commercialization, Adv. Mater., 2017, 29, 1607022 CrossRef PubMed.
- D. S. Dolzhnikov, H. Zhang, J. Jang, J. S. Son, M. G. Panthani, S. Chattopadhyay, T. Shibata and D. V. Talapin, Composition-Matched Molecular “Solders” for Semiconductors, Science, 2015, 347, 425–428 CrossRef CAS PubMed.
- C. R. Kagan and C. B. Murray, Charge Transport in Strongly Coupled Quantum Dot Solids, Nat. Nanotechnol., 2015, 10, 1013–1026 CrossRef CAS PubMed.
- M. Yuan, M. Liu and E. Sargent, Colloidal Quantum Dot Solids for Solution-Processed Solar Cells, Nat. Energy, 2016, 1, 16016 CrossRef CAS.
- X. Tang, M. M. Ackerman, M. Chen and P. Guyot-Sionnest, Dual-Band Infrared Imaging Using Stacked Colloidal Quantum Dot Photodiodes, Nat. Photonics, 2019, 13, 277–282 CrossRef CAS.
- J. Kim, S.-M. Kwon, Y. K. Kang, Y.-H. Kim, M.-J. Lee, K. Han, A. Facchetti, M.-G. Kim and S. K. Park, A Skin-Like Two-Dimensionally Pixelized Full-Color Quantum Dot Photodetector, Sci. Adv., 2019, 5, eaax8801 CrossRef CAS PubMed.
- J. Yoo, K. Lee, U. J. Yang, H. H. Song, J. H. Jang, G. H. Lee, M. S. Bootharaju, J. H. Kim, K. Kim, S. I. Park, J. D. Seo, S. Li, W. S. Yu, J. I. Kwon, M. H. Song, T. Hyeon, J. Yang and M. K. Choi, Highly Efficient Printed Quantum Dot Light-Emitting Diodes through Ultrahigh-Definition Double-Layer Transfer Printing, Nat. Photonics, 2024, 18, 1105–1112 CrossRef CAS.
- Q. Zhang, K. Yang, C. Luo, Z. Lin, W. Chen, Y. Yu, H. Hu and F. Li, Nanosecond Response Perovskite Quantum Dot Light-Emitting Diodes with Ultra-High Resolution for Active Display Application, Light Sci. Appl., 2025, 14, 285 CrossRef CAS PubMed.
- R. Xu, J. Fan, Z. Yao, C. Gu, Q. Nie, R. Li, J. Wang, R. J. Smith, L. Qian, C. Xiang and T. Zhang, Enhanced Molecular Stacking Enabled by Photo-Induced Crosslinking of Hole Transport Materials for High-Performance QLED, Adv. Funct. Mater., 2025, e10728 Search PubMed.
- X. Zhang, S. Ding, Z. Tang, Z. Yao, T. Zhang, C. Xiang and L. Qian, Nanostructured Materials for Next-Generation Display Technology, Nat. Rev. Electr. Eng., 2025, 2, 263–276 CrossRef.
- E.-L. Hsiang, Z. Yang, Q. Yang, P.-C. Lai, C.-L. Lin and S.-T. Wu, AR/VR Light Engines: Perspectives and Challenges, Adv. Opt. Photonics, 2022, 14, 783–861 CrossRef.
- C. Xiang, L. Wu, Z. Lu, M. Li, Y. Wen, Y. Yang, W. Liu, T. Zhang, W. Cao, S. W. Tsang, B. Shan, X. Yan and L. Qian, High Efficiency and Stability of Ink-Jet Printed Quantum Dot Light Emitting Diodes, Nat. Commun., 2020, 11, 1646 CrossRef CAS PubMed.
- P. Yang, L. Zhang, D. J. Kang, R. Strahl and T. Kraus, High-Rosolution Inkjet Printing of Quantum Dot Light-Emitting Microdiode Arrays, Adv. Optical Mater., 2019, 8, 1901429 CrossRef.
- T.-H. Kim, K.-S. Cho, E. K. Lee, S. J. Lee, J. Chae, J. W. Kim, D. H. Kim, J.-Y. Kwon, G. Amaratunga, S. Y. Lee, B. L. Choi, Y. Kuk, J. M. Kim and K. Kim, Full-Color Quantum Dot Displays Fabricated by Transfer Printing, Nat. Photonics, 2011, 5, 176–182 CrossRef CAS.
- M. K. Choi, J. Yang, K. Kang, D. C. Kim, C. Choi, C. Park, S. J. Kim, S. I. Chae, T.-H. Kim, J. H. Kim, T. Hyeon and D. H. Kim, Wearable Red-Green-Blue Quantum Dot Light-Emitting Diode Array Using High-Resolution Intaglio Transfer Printing, Nat. Commun., 2015, 6, 7149 CrossRef CAS PubMed.
- C. Zhong, K. Yu, Y. Qie, Y. Yu, Y. Lu, G. Deng, T. Guo, H. Hu and F. Li, Ultrahigh-Resolution Full-Color Quantum Dot LEDs Based on Region-Selective Interfacial Self-Assembly, Adv. Funct. Mater., 2025, 35, 2510076 CrossRef CAS.
- C. Hu, T. Aubert, Y. Justo, S. Flamee, M. Cirillo, A. Gassenq, O. Drobchak, F. Beunis, G. Roelkens and Z. Hens, The Micropatterning of Layers of Colloidal Quantum Dots with Inorganic Ligands Using Selective Wet Etching, Nanotechnology, 2014, 25, 175302 CrossRef PubMed.
- M. Yuan, J. Feng, H. Li, H. Gao, Y. Qiu, L. Jiang and Y. Wu, Remote Epitaxial Crystalline Perovskites for Ultrahigh-Resolution Micro-LED Displays, Nat. Nanotechnol., 2025, 20, 381–387 CrossRef CAS PubMed.
- P. Zhang, G. Yang, F. Li, J. Shi and H. Zhong, Direct in Situ Photolithography of Perovskite Quantum Dots Based on Photocatalysis of Lead Bromide Complexes, Nat. Commun., 2022, 13, 6713 CrossRef CAS PubMed.
- Z. Gao, J. Shi and G. Yang, Quantum Dots Photoresist for Direct Photolithography Patterning, Adv. Optical Mater., 2024, 12, 2401106 CrossRef CAS.
- Y. Wang, I. Fedin, H. Zhang and D. V. Talapin, Direct Optical Lithography of Functional Inorganic Nanomaterials, Science, 2017, 357, 385–388 CrossRef CAS PubMed.
- S. Lu, Z. Fu, F. Li, K. Weng, L. Zhou, L. Zhang, Y. Yang, H. Qiu, D. Liu, W. Qing, H. Ding, X. Sheng, M. Chen, X. Tang, L. Duan, W. Liu, L. Wu, Y. Yang, H. Zhang and J. Li, Beyond a Linker: The Role of Photochemistry of Crosslinkers in the Direct Optical Patterning of Colloidal Nanocrystals, Angew. Chem., Int. Ed., 2022, 61, e202202633 CrossRef CAS PubMed.
- J. Yang, M. Lee, S. Y. Park, M. Park, J. Kim, N. Sitapure, D. Hahm, S. Rhee, D. Lee, H. Jo, Y. H. Jo, J. Lim, J. Kim, T. J. Shin, D. C. Lee, K. Kwak, J. S. Kwon, B. Kim, W. K. Bae and M. S. Kang, Nondestructive Photopatterning of Heavy-Metal-Free Quantum Dots, Adv. Mater., 2022, 34, 2205504 CrossRef CAS PubMed.
- D. Hahm, J. Lim, H. Kim, J.-W. Shin, S. Hwang, S. Rhee, J. H. Chang, J. Yang, C. H. Lim, H. Jo, B. Choi, N. S. Cho, Y.-S. Park, D. C. Lee, E. Hwang, S. Chung, C.-m. Kang, M. S. Kang and W. K. Bae, Direct Patterning of Colloidal Quantum Dots with Adaptable Dual-Ligand Surface, Nat. Nanotechnol., 2022, 17, 952–958 CrossRef CAS PubMed.
- D. Liu, K. Weng, S. Lu, F. Li, H. Abudukeremu, L. Zhang, Y. Yang, J. Hou, H. Qiu, Z. Fu, X. Luo, L. Duan, Y. Zhang, H. Zhang and J. Li, Direct Optical Patterning of Perovskite Nanocrystals with Ligand Cross-Linkers, Sci. Adv., 2022, 8, eabm8433 CrossRef CAS PubMed.
- Z. Fu, S. F. Musolino, W. Qing, H. Li, F. J. de Zwart, Z. Zheng, M. Cai, Y. Gao, B. de Bruin, X. Dai, J. E. Wulff and H. Zhang, Direct Photopatterning of Colloidal Quantum Dots with Electronically Optimized Diazirine Cross-Linkers, J. Am. Chem. Soc., 2024, 146, 28895–28905 CrossRef CAS PubMed.
- D. Liu, K. Weng, H. Zhao, S. Wang, H. Qiu, X. Luo, S. Lu, L. Duan, S. Bai, H. Zhang and J. Li, Nondestructive Direct Optical Patterning of Perovskite Nanocrystals with Carbene-Based Ligand Cross-Linkers, ACS Nano, 2024, 18, 6896–6907 CrossRef CAS PubMed.
- J. Guan, J. Ma, L. Zhou, Y. Zou, L. Ye, W. Hou, P.-k. Zhang, X. Dai, H. Zhang, D. V. Talapin and Y. Wang, Direct Ambient Photopatterning of RGB Quantum Dots for Light-Emitting Diodes with EQE Exceeding 20, Nat. Commun., 2025, 16, 9689 CrossRef PubMed.
- Z. Fu, L. Zhou, Y. Yin, K. Weng, F. Li, S. Lu, D. Liu, W. Liu, L. Wu, Y. Yang, H. Li, L. Duan, H. Xiao, H. Zhang and J. Li, Direct Photo-Patterning of Efficient and Stable Quantum Dot Light-Emitting Diodes via Light-Triggered, Carbocation-Enabled Ligand Stripping, Nano Lett., 2023, 23, 2000–2008 CrossRef CAS PubMed.
- Q. Nie, J. Fan, R. Xu, Z. Yao, Y. Xiao, C. Xiang, L. Qian and T. Zhang, Direct Optical Patterning of Quantum Dot Light-Emitting Diodes Based on Ultrafast, Low-Energy, Site Controlled Click Chemistry Reaction, Adv. Funct. Mater., 2025, 35, 2420829 CrossRef CAS.
- J.-A. Pan, H. Cho, I. Coropceanu, H. Wu and D. V. Talapin, Stimuli-Responsive Surface Ligands for Direct Lithography of Functional Inorganic Nanomaterials, Acc. Chem. Res., 2023, 56, 2286–2297 CrossRef CAS PubMed.
- S. Y. Park, S. Lee, J. Yang and M. S. Kang, Patterning Quantum Dots via Photolithography: A Review, Adv. Mater., 2023, 35, 2300546 CrossRef CAS PubMed.
- S. Baek and J. S. Son, Recent Advances in Direct Optical Patterning of Inorganic Materials and Devices, Adv. Phys. Res., 2024, 3, 2300069 CrossRef.
- Y. Yang, J. Guan, N. Zhang, L. Ru, Y. Zou and Y. Wang, Advances in Direct Optical Lithography of Nanomaterials, J. Mater. Chem. A, 2024, 12, 32505–32525 RSC.
- Y. Qiu, Y. Yu, S. Wang and M. Chen, Advances in Quantum Dot Direct Photolithographic Patterning, ACS Mater. Lett., 2024, 6, 3176–3189 CrossRef CAS.
- M. Ibáñez, S. C. Boehme, R. Buonsanti, J. De Roo, D. J. Milliron, S. Ithurria, A. L. Rogach, A. Cabot, M. Yarema, B. M. Cossairt, P. Reiss, D. V. Talapin, L. Protesescu, Z. Hens, I. Infante, M. I. Bodnarchuk, X. Ye, Y. Wang, H. Zhang, E. Lhuillier, V. I. Klimov, H. Utzat, G. Rainò, C. R. Kagan, M. Cargnello, J. S. Son and M. V. Kovalenko, Prospects of Nanoscience with Nanocrystals: 2025 Edition, ACS Nano, 2025, 9, 1012–1057 Search PubMed.
- S. Maeng and H. Cho, Direct Optical Lithography: Toward Nondestructive Patterning of Nanocrystal Emitters, Acc. Mater. Res., 2025, 6, 393 CrossRef CAS.
- N. Lee, D. A. Taylor, D. Choi, D. Kwak and J.-S. Lee, Advances in Photopatterning of Quantum Dots: Mechanisms, Materials, and Device Applications, ACS Energy Lett., 2026, 11, 1495–1513 CrossRef CAS.
- Y. Wang, J.-A. Pan, H. Wu and D. V. Talapin, Direct Wavelength-Selective Optical and Electron-Beam Lithography of Functional Inorganic Nanomaterials, ACS Nano, 2019, 13, 13917–13931 CrossRef CAS PubMed.
- H. Cho, J. A. Pan, H. Wu, X. Lan, I. Coropceanu, Y. Wang, W. Cho, E. A. Hill, J. S. Anderson and D. V. Talapin, Direct Optical Patterning of Quantum Dot Light-Emitting Diodes via in Situ Ligand Exchange, Adv. Mater., 2020, 32, 2003805 CrossRef CAS PubMed.
- L. Li, S. Chakrabarty, K. Spyrou, C. K. Ober and E. P. Giannelis, Studying the Mechanism of Hybrid Nanoparticle Photoresists: Effect of Particle Size on Photopatterning, Chem. Mater., 2015, 27, 5027–5031 CrossRef CAS.
- M. H. V. Werts, M. Lambert, J.-P. Bourgoin and M. Brust, Nanometer Scale Patterning of Langmuir−Blodgett Films of Gold Nanoparticles by Electron Beam Lithography, Nano Lett., 2002, 2, 43–47 CrossRef CAS.
- F. Palazon, M. Prato and L. Manna, Writing on Nanocrystals: Patterning Colloidal Inorganic Nanocrystal Films through Irradiation-Induced Chemical Transformations of Surface Ligands, J. Am. Chem. Soc., 2017, 139, 13250–13259 CrossRef CAS PubMed.
- D. B. Dement, M. K. Quan and V. E. Ferry, Nanoscale Patterning of Colloidal Nanocrystal Films for Nanophotonic Applications Using Direct Write Electron Beam Lithography, ACS Appl. Mater. Interfaces, 2019, 11, 14970–14979 CrossRef CAS PubMed.
- J. Yang, D. Hahm, K. Kim, S. Rhee, M. Lee, S. Kim, J. H. Chang, H. W. Park, J. Lim, M. Lee, H. Kim, J. Bang, H. Ahn, J. H. Cho, J. Kwak, B. Kim, C. Lee, W. K. Bae and M. S. Kang, High-Resolution Patterning of Colloidal Quantum Dots via Non-Destructive, Light-Driven Ligand Crosslinking, Nat. Commun., 2020, 11, 2874 CrossRef CAS PubMed.
- S. Maeng, S. J. Park, J. Lee, H. Lee, J. Choi, J. K. Kang and H. Cho, Direct Photocatalytic Patterning of Colloidal Emissive Nanomaterials, Sci. Adv., 2023, 9, eadi6950 CrossRef CAS PubMed.
- W. Qing, Y. Si, M. Cai, L. Zhou, L. Wu, Z. Hou, D. Liu, X. Tian, W. Liu, L. Lin and H. Zhang, Photosensitizer-Assisted Direct 2D Patterning and 3D Printing of Colloidal Quantum Dots, Nano Res., 2024, 17, 10460–10466 CrossRef CAS.
- J.-A. Pan, Z. Rong, Y. Wang, H. Cho, I. Coropceanu, H. Wu and D. V. Talapin, Direct Optical Lithography of Colloidal Metal Oxide Nanomaterials for Diffractive Optical Elements with 2π Phase Control, J. Am. Chem. Soc., 2021, 143, 2372–2383 CrossRef CAS PubMed.
- J. Ko, J. H. Chang, B. G. Jeong, H. J. Kim, J. F. Joung, S. Park, D. H. Choi, W. K. Bae and J. Bang, Direct Photolithographic Patterning of Colloidal Quantum Dots Enabled by UV Crosslinkable and Hole-Transporting Polymer Ligands, ACS Appl. Mater. Interfaces, 2020, 12, 42153–42160 CrossRef PubMed.
- J. Ko, K. Ma, J. F. Joung, S. Park and J. Bang, Ligand-Assisted Direct Photolithography of Perovskite Nanocrystals Encapsulated with Multifunctional Polymer Ligands for Stable, Full-Colored, High-Resolution Displays, Nano Lett., 2021, 21, 2288–2295 CrossRef CAS PubMed.
- P. Xiao, J. Ma, Z. Zhang, Y. Zou, H. Luo, J. Guan, J. Zhang, L. Zhou, W. Hou, P. Zhang, D. V. Talapin and Y. Wang, Ligand-Engineered Direct Optical Lithography of Nanocrystals with Industrially Compatible Solvents, ACS Nano, 2025, 19, 14509–14520 CrossRef CAS PubMed.
- S. Shin, K. Kang, H. Jang, N. Gwak, S. Kim, T. A. Kim and N. Oh, Ligand-Crosslinking Strategy for Efficient Quantum Dot Light-Emitting Diodes via Thiol-Ene Click Chemistry, Small Methods, 2023, 7, 2300206 CrossRef CAS PubMed.
- N. Lee, D. Choi, K.-J. Ko, H.-S. Kim, J. Yu and J.-S. Lee, Direct Optical Lithography Using Diazirine Cross-Linker for Quantum Dot Light Emitting Diodes and Enhancing Photoluminescence Quantum Yield through Post-Treatment, ACS Nano, 2025, 19, 22253–22261 CrossRef CAS PubMed.
- H. Seo, S. Z. Hassan, Y. J. Kim, T. Lee, G. W. Baek, C. So, D. S. Chung and J. Kwak, Direct Photolithography of Colloidal Inp-Based Quantum Dots Utilizing the Photoligation Method, Chem. Mater., 2025, 37, 1424–1431 CrossRef CAS.
- J.-M. Kim, D. Choi, N. Lee, W.-S. Chae, H.-S. Kim, J. Yu and J.-S. Lee, Role of Conjugated Structure of Cross-linkers in Patterned QLEDs, Nano Lett., 2025, 25, 10435–10441 CrossRef CAS PubMed.
- S. Maeng, J. Kim, T. Kim, S. Lee, S. Han, S. Park, C. Kim, J. Kim, J.-Y. Lee and H. Cho, Dual-Strategy Direct Photocatalytic Patterning for Efficient Perovskite Nanocrystal LED Displays, Adv. Mater., 2025, 37, e08217 CrossRef CAS PubMed.
- Y. Lee, J. Shin, S. Shin, E. A. Kim, J. Y. Lee, N. Gwak, S. Kim, J. Seo, H. Kong, D. Yeo, J. Na, S. Kim, J. Lee, S.-Y. Cho, J. Lee, T. A. Kim and N. Oh, Adv. Mater., 2025, 37, 2415436 CrossRef CAS PubMed.
- J.-A. Pan, H. Wu, A. Gomez, J. C. Ondry, J. Portner, W. Cho, A. Hinkle, D. Wang and D. V. Talapin, Ligand-Free Direct Optical Lithography of Bare Colloidal Nanocrystals Via Photo-Oxidation of Surface Ions with Porosity Control, ACS Nano, 2022, 16, 16067–16076 CrossRef CAS PubMed.
- H. Baek, S. Kang, J. Heo, S. Choi, R. Kim, K. Kim, N. Ahn, Y.-G. Yoon, T. Lee, J. B. Chang, K. S. Lee, Y.-G. Park and J. Park, Insights into Structural Defect Formation in Individual InP/ZnSe/ZnS Quantum Dots under Uv Oxidation, Nat. Commun., 2024, 15, 1671 CrossRef CAS PubMed.
- Z. Chen, Z. Man, S. Rao, J. Zhao, S. Wang, R. Zhang, F. Teng and A. Tang, Rigid Crosslinker-Assisted Nondestructive Direct Photolithograph for Patterned QLED Displays, Light Sci. Appl., 2025, 14, 251 CrossRef CAS PubMed.
- K. A. Schnapp, R. Poe, E. Leyva, N. Soundararajan and M. S. Platz, Exploratory Photochemistry of Fluorinated Aryl Azides. Implications for The Design of Photoaffinity Labeling Reagents, Bioconjug. Chem., 1993, 4, 172–177 CrossRef CAS PubMed.
- W. T. Borden, N. P. Gritsan, C. M. Hadad, W. L. Karney, C. R. Kemnitz and M. S. Platz, The Interplay of Theory and Experiment in The Study of Phenylnitrene, Acc. Chem. Res., 2000, 33, 765–771 CrossRef CAS PubMed.
- N. P. Gritsan, A. D. Gudmundsdóttir, D. Tigelaar, Z. Zhu, W. L. Karney, C. M. Hadad and M. S. Platz, A Laser Flash Photolysis and Quantum Chemical Study of The Fluorinated Derivatives of Singlet Phenylnitrene, J. Am. Chem. Soc., 2001, 123, 1951–1962 CrossRef CAS PubMed.
- D. E. Westmoreland, R. López-Arteaga and E. A. Weiss, N-Heterocyclic Carbenes as Reversible Exciton-Delocalizing Ligands for Photoluminescent Quantum Dots, J. Am. Chem. Soc., 2020, 142, 2690–2696 CrossRef CAS PubMed.
- L. Du, N. A. Nosratabad, Z. Jin, C. Zhang, S. Wang, B. Chen and H. Mattoussi, Luminescent Quantum Dots Stabilized by N-Heterocyclic Carbene Polymer Ligands, J. Am. Chem. Soc., 2021, 143, 1873–1884 CrossRef CAS PubMed.
- Z. Zhai, C. Gu, W. Wang, H. Tan, C. Han, T. Zhang and C. Xiang, Nondestructive Direct Photolithography of Colloidal Quantum Dots Enabled by Benzophenone-Based Crosslinkers, Nano Res., 2025, 18, 94907980 CrossRef CAS.
- J. Ren, B. Dong, Y. Liao, L. Liu, C. Zhang, X. Zhou, H. Liu, F. Liu, L. Chen and Y. Wang, Highly Luminescent Photo-Crosslinkable Perovskite Nanocrystals: Advancing Direct Optical Patterning, 3D Painting, and Backlight Displays, Adv. Funct. Mater., 2026, e29700 CrossRef.
- Y. Huo, C. Luo, C. Wu, Z. Ren, H. Wang, D. Zhu, L. Wang, X. Zhou, Z. Zheng, X. Wang and Y. Chen, Ambient Direct Lithography Patterning of Ultra-Stable Perovskite Quantum Dots for High-Resolution Light-Emitting Diodes, Adv. Funct. Mater., 2025, 35, 2504261 CrossRef CAS.
- M. D. Tessier, D. Dupont, K. De Nolf, J. De Roo and Z. Hens, Economic and Size-Tunable Synthesis of InP/ZnE (E = S, Se) Colloidal Quantum Dots, Chem. Mater., 2015, 27, 4893–4898 CrossRef CAS.
- J. L. Stein, W. M. Holden, A. Venkatesh, M. E. Mundy, A. J. Rossini, G. T. Seidler and B. M. Cossairt, Probing Surface Defects of Inp Quantum Dots Using Phosphorus Kα and Kβ X-Ray Emission Spectroscopy, Chem. Mater., 2018, 30, 6377–6388 CrossRef CAS.
- J. Shamsi, A. S. Urban, M. Imran, L. De Trizio and L. Manna, Metal Halide Perovskite Nanocrystals: Synthesis, Post-Synthesis Modifications, and Their Optical Properties, Chem. Rev., 2019, 119, 3296–3348 CrossRef CAS PubMed.
- Y. Jiang, C. Sun, J. Xu, S. Li, M. Cui, X. Fu, Y. Liu, Y. Liu, H. Wan, K. Wei, T. Zhou, W. Zhang, Y. Yang, J. Yang, C. Qin, S. Gao, J. Pan, Y. Liu, S. Hoogland, E. H. Sargent, J. Chen and M. Yuan, Synthesis-on-Substrate of Quantum Dot Solids, Nature, 2022, 612, 679–684 CrossRef CAS PubMed.
- A. Fakharuddin, M. K. Gangishetty, M. Abdi-Jalebi, S.-H. Chin, A. R. Yusoff, D. N. Congreve, W. Tress, F. Deschler, M. Vasilopoulou and H. J. Bolink, Perovskite Light-Emitting Diodes, Nat. Electron., 2022, 5, 203–216 CrossRef CAS.
- S. Lim, S. Han, D. Kim, J. Min, J. Choi and T. Park, Key Factors Affecting the Stability of CsPbI3 Perovskite Quantum Dot Solar Cells: A Comprehensive Review, Adv. Mater., 2023, 35, e2203430 CrossRef PubMed.
- D. Kim, T. Yun, S. An and C. L. Lee, How to Improve the Structural Stabilities of Halide Perovskite Quantum Dots: Review of Various Strategies to Enhance the Structural Stabilities of Halide Perovskite Quantum Dots, Nano Converg., 2024, 11, 4 CrossRef CAS PubMed.
- W. Y. E. Ong, Y. Z. D. Tan, L. J. Lim, T. G. Hoang and Z.-K. Tan, Crosslinkable Ligands for High-Density Photo-Patterning of Perovskite Nanocrystals, Adv. Mater., 2024, 37, 2409564 CrossRef PubMed.
- H. Furukawa, K. E. Cordova, M. O'Keeffe and O. M. Yaghi, The Chemistry and Applications of Metal-Organic Frameworks, Science, 2013, 341, 1230444 CrossRef PubMed.
- A. Bétard and R. A. Fischer, Metal-Organic Framework Thin Films: From Fundamentals to Applications, Chem. Rev., 2012, 112, 1055–1083 CrossRef PubMed.
- M. D. Allendorf, R. Dong, X. Feng, S. Kaskel, D. Matoga and V. Stavila, Electronic Devices Using Open Framework Materials, Chem. Rev., 2020, 120, 8581–8640 CrossRef CAS PubMed.
- I. Stassen, N. Burtch, A. Talin, P. Falcaro, M. Allendorf and R. Ameloot, An Updated Roadmap for The Integration of Metal-Organic Frameworks with Electronic Devices and Chemical Sensors, Chem. Soc. Rev., 2017, 46, 3185–3241 RSC.
- X. Tian, F. Li, Z. Tang, S. Wang, K. Weng, D. Liu, S. Lu, W. Liu, Z. Fu, W. Li, H. Qiu, M. Tu, H. Zhang and J. Li, Crosslinking-Induced Patterning of Mofs by Direct Photo- and Electron-Beam Lithography, Nat. Commun., 2024, 15, 2920 CrossRef CAS PubMed.
- Z. Zhu, F. Li, J. Li, Q. Chen, W. Li, Z. Tang, W. Xu, W. Shen, T. H. Tao, L. Sun, Y. Fu and M. Tu, Direct Optical Patterning of Metal–Organic Frameworks via Photoacid–Induced Etching, ACS Nano, 2024, 18, 19628–19635 CAS.
- X. Tian, W. Li, F. Li, M. Cai, Y. Si, H. Tang, H. Li and H. Zhang, Direct Photopatterning of Zeolitic Imidazolate Frameworks via Photoinduced Fluorination, Angew. Chem., Int. Ed., 2025, 64, 202500476 CrossRef PubMed.
- S. V. Murphy and A. Atala, 3D Bioprinting of Tissues and Organs, Nat. Biotechnol., 2014, 32, 773–785 CrossRef CAS PubMed.
- J. M. Pearce, Building Research Equipment with Free, Open-Source Hardware, Science, 2012, 337, 1303–1304 CrossRef CAS PubMed.
- K. Sun, T.-S. Wei, B. Y. Ahn, J. Y. Seo, S. J. Dillon and J. A. Lewis, 3D Printing of Interdigitated Li-Ion Microbattery Architectures, Adv. Mater., 2013, 25, 4539–4543 CrossRef CAS PubMed.
- A. T. Polonsky and T. M. Pollock, Closing The Science Gap in 3D Metal Printing, Science, 2020, 368, 583–584 CrossRef CAS PubMed.
- E. MacDonald and R. Wicker, Multiprocess 3D Printing for Increasing Component Functionality, Science, 2016, 353, aaf2093 CrossRef CAS PubMed.
- S. Liu, Z. Hou, L. Lin, F. Li, Y. Zhao, X. Li, H. Zhang, H. Fang, Z. Li and H. Sun, 3D Nanoprinting of Semiconductor Quantum Dots by Photoexcitation-Induced Chemical Bonding, Science, 2022, 377, 1112–1116 CrossRef CAS PubMed.
- F. Li, S. Liu, W. Liu, Z. Hou, J. Jiang, Z. Fu, S. Wang, Y. Si, S. Lu, H. Zhou, D. Liu, X. Tian, H. Qiu, Y. Yang, Z. Li, X. Li, L. Lin, H. Sun, H. Zhang and J. Li, 3D Printing of Inorganic Nanomaterials by Photochemically Bonding Colloidal Nanocrystals, Science, 2023, 381, 1468–1474 CrossRef CAS PubMed.
- C. Mao, S. Ju, J. Zheng, Y. Zheng, Z. Xu, L. Lin, H. Hu, K. Yang, T. Guo and F. Li, Ultra-High-Resolution Perovskite Quantum Dot Light-Emitting Diodes, Adv. Optical Mater., 2023, 11, 2202058 CrossRef CAS.
- C. Luo, Z. Zheng, Y. Ding, Z. Ren, H. Shi, H. Ji, X. Zhou and Y. Chen, High-Resolution, Highly Transparent, and Efficient Quantum Dot Light-Emitting Diodes, Adv. Mater., 2023, 35, 2303329 CrossRef CAS PubMed.
- H. Li, J. Zhang, W. Wen, Y. Zhao, H. Gao, B. J, Y. Wang, L. Jiang and Y. Wu, Highly Efficient Light-Emitting Diodes via Self-Assembled InP Quantum Dots, Nat. Commun., 2025, 16, 4257 CrossRef CAS PubMed.
- Y. Lian, Y. Wang, Y. Yuan, Z. Ren, W. Tang, Z. Liu, S. Xing, K. Ji, B. Yuan, Y. Yang, Y. Gao, S. Zhang, K. Zhou, G. Zhang, S. D. Stranks, B. Zhao and D. Di, Downscaling Micro- and Nano-Perovskite LEDs, Nature, 2025, 640, 62–68 CrossRef CAS PubMed.
- D. Hahm, J. Park, I. Jeong, S. Rhee, T. Lee, C. Lee, S. Chung, W. K. Bae and S. Lee, Surface Engineered Colloidal Quantum Dots for Complete Green Process, ACS Appl. Mater. Interfaces, 2020, 12, 10563–10570 CrossRef CAS PubMed.
- Z. Fu, Z. Zheng, Y. Xie, H. Abudukeremu, L. Zhou, S. Zang, K. Tian, Q. Liu, Y. Li and H. Zhang, Direct Photopatterning and Erasing of Quantum Dots via Ligand Photoisomerization, Chem. Commun., 2026, 62, 4089–4093 RSC.
| This journal is © The Royal Society of Chemistry 2026 |