 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Outer-surface charge modulation of photothermal diffusion voltage enables ultrasensitive sensing in nanofluidic membranes
Yihan
Ma†
a,
Xinyi
Yang†
a,
Bingquan
Qi
b,
Xiaoping
Yang
a,
Zhengxu
He
b,
Ning
Feng
a,
Li
Dai
b,
Aiqing
Zhang
a,
Yu
Huang
 *b and
Fan
Xia
*b and
Fan
Xia
 b
b
aKey Laboratory of Catalysis and Energy Materials Chemistry of Ministry of Education, Hubei Key Laboratory of Catalysis and Materials Science, South-Central Minzu University, Wuhan 430074, China
bState Key Laboratory of Geomicrobiology and Environmental Changes, Engineering Research Center of Nano-Geomaterials of Ministry of Education, Faculty of Materials Science and Chemistry, China University of Geosciences, Wuhan 430074, China. E-mail: yuhuang@cug.edu.cn
First published on 25th February 2026
Abstract
Precise ionic transport regulation is central to nanofluidic sensing, yet quantitative readout at ultratrace analyte levels remains challenging because conventional externally biased measurements primarily transduce target binding through resistance changes. When trace analytes induce negligible steric variation, the system resistance is essentially unchanged, yielding insufficient ionic current contrast. Here we develop an outer-surface charge-modulated, photothermal diffusion voltage-driven strategy in an MXene nanofluidic membrane. Under 808 nm illumination, the strong photothermal conversion of Ti3C2Tx establishes a stable transmembrane temperature gradient across K+-permselective lamellar nanochannels, generating a tunable photothermal diffusion voltage (Vdiff). Trace-level binding events markedly modulate the outer-surface charge density, thereby altering the K+ transference number and amplifying minute charge variations into pronounced changes in Vdiff and the zero-bias ionic current, even when steric hindrance and resistance remain nearly constant. Using microcystin-LR (MC-LR) as a model toxin, this strategy enables ultratrace detection down to 1 × 10−7 µg L−1, delivering a 105-fold sensitivity enhancement over conventional external voltage-driven readout while retaining high selectivity against structural analogues, and reliable quantification in real water matrices. This work establishes a light-addressable route to actively regulate nanofluidic voltages via outer-surface charge, opening opportunities for photoresponsive nanofluidic sensors and iontronic circuitry.
Introduction
In biological systems, protein nanopores precisely regulate transmembrane ionic currents, enabling selective transport of molecules and maintaining essential physiological functions such as nerve signal transmission, muscle contraction, and osmotic homeostasis.1–3 These natural channels represent highly optimized systems that couple structural dynamics with functional specificity, utilizing subtle variations in charge distribution, steric hindrance, and membrane composition to achieve selective and efficient ion transport.4–6 The remarkable performance of biological nanopores has inspired extensive efforts to develop artificial solid-state nanochannels that mimic their selective transport behavior.7–10 Using conventional external voltage-driven sensing strategies, target-specific recognition probes are integrated onto the surfaces of solid-state nanochannels so that molecular binding events are transduced into measurable ionic current changes through alterations in interfacial properties such as wettability, steric hindrance, and surface charge.11–14 This biomimetic approach has enabled the detection of a wide range of targets, from small ions and biomolecules to pathogens,15–18 as demonstrated by advances in electrochemical aptamer-based biosensors,19,20 responsive nanochannels,21,22 and bio-inspired smart nanochannels.23,24 In these systems, ionic currents are typically recorded at a fixed external voltage (Vext) and therefore follow Ohm's law (I =![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) V/R), so the sensing signal mainly arises from target-induced changes in the effective channel resistance R. However, at ultratrace analyte concentrations, the variations in wettability, steric hindrance, and surface charge induced within the nanochannels are extremely subtle, leading to negligible changes in R and insufficient ionic-current contrast. As a result, the detection sensitivity of current solid-state nanochannel sensors still lags far behind that of biological protein nanopores, which can reach single-molecule precision.25–27 To enhance these modest resistance changes within the fixed-voltage framework, most external voltage-driven nanochannel sensors exploit steric hindrance, where target binding triggers conformational changes in recognition probes that slightly alter the effective pore size and thus modulate ionic transport.28–30 A variety of signal-amplification schemes, such as super-sandwich assays,31,32 rolling circle amplification,33,34 and cyclic detection strategies,35,36 have been integrated to magnify this steric effect. Nevertheless, the extent of steric modulation is intrinsically limited by the structures of recognition probes, many of which cannot be readily engineered to generate sufficiently large geometric changes, thereby constraining further improvements in sensitivity and generality within the conventional external voltage-driven strategy.
V/R), so the sensing signal mainly arises from target-induced changes in the effective channel resistance R. However, at ultratrace analyte concentrations, the variations in wettability, steric hindrance, and surface charge induced within the nanochannels are extremely subtle, leading to negligible changes in R and insufficient ionic-current contrast. As a result, the detection sensitivity of current solid-state nanochannel sensors still lags far behind that of biological protein nanopores, which can reach single-molecule precision.25–27 To enhance these modest resistance changes within the fixed-voltage framework, most external voltage-driven nanochannel sensors exploit steric hindrance, where target binding triggers conformational changes in recognition probes that slightly alter the effective pore size and thus modulate ionic transport.28–30 A variety of signal-amplification schemes, such as super-sandwich assays,31,32 rolling circle amplification,33,34 and cyclic detection strategies,35,36 have been integrated to magnify this steric effect. Nevertheless, the extent of steric modulation is intrinsically limited by the structures of recognition probes, many of which cannot be readily engineered to generate sufficiently large geometric changes, thereby constraining further improvements in sensitivity and generality within the conventional external voltage-driven strategy.
This situation motivates re-examination of biological protein nanopores, where many systems, such as the bacterial porin OmpF, possess charged vestibules that act as electrostatic gates to preconcentrate counter-ions or repel co-ions, thereby strongly influencing ionic transport.37,38 These observations suggest that modulation of surface charge, rather than steric hindrance alone, plays a decisive role in regulating ion transport and provides a promising direction for constructing highly sensitive, universal nanochannel sensors. Inspired by the structures of biological protein nanopores, we have previously demonstrated that selective functionalization of the outer surface alone is sufficient to modulate transmembrane ion transport, achieved by precisely constructing nanochannels with spatially resolved inner and outer surface chemistries.39 Compared with the geometrically confined inner surface, the outer surface lies in an open and accessible environment, enabling efficient probe immobilization, direct physicochemical characterization, and highly effective interaction with target analytes.40–42 Subsequent studies further established that tuning outer-surface charge provides a powerful means of regulating ion transport and amplifying sensing responsiveness in nanochannel systems.43–50 Building on this concept, we recently introduced partitioned-interface nanochannels, revealing that variations in outer-surface charge density dominate ion transport behavior, outweighing steric hindrance effects.51 However, translating these subtle changes in outer-surface charge density into robust and quantitative readout signals for trace-level analyte detection remains a formidable challenge.
Here, we develop a photothermal diffusion voltage-driven sensing strategy as an alternative to conventional external voltage-driven approaches to enable ultratrace-level detection in nanochannels. A stacked MXene nanofluidic membrane is employed as the device platform, in which outer-surface charge is transduced through a photothermal diffusion voltage (Vdiff) rather than relying solely on resistance changes, as illustrated in Scheme 1. By exploiting the near-infrared (NIR) photothermal response of MXene, a stable transmembrane temperature gradient (ΔT) is established across K+-permselective lamellar interlayer nanochannels within the membrane, creating a non-isothermal mode in which the ionic response is governed by outer-surface charge-dependent ion permselectivity. Direct comparison with the conventional external voltage-driven mode under a fixed external bias (Vext) on the same membrane device decouples the contributions of steric hindrance and outer-surface charge, revealing that the latter dominates the ionic response to ultratrace microcystin-LR (MC-LR). Combined experiments and numerical simulations further clarify how aptamer-mediated modulation of outer-surface charge reshapes ion transport characteristics and yields substantial signal gain. Consequently, this strategy enables quantitative detection of MC-LR down to 1 × 10−7 µg L−1 with sensitivity enhanced by more than 105-fold compared with conventional external voltage-driven measurements, establishing outer-surface-charge-modulated photothermal diffusion voltage as a general route for actively regulating ionic transport in solid-state nanochannels.
Results and discussion
Fabrication and characterization of an outer-surface functionalized MXene nanofluidic membrane
The Ti3C2Tx MXene nanosheets were initially obtained by in situ etching of the MAX phase (Ti3AlC2) with an etchant composed of HCl and LiF, as previously reported.52 This process selectively removed the Al layer while simultaneously increasing the interlayer spacing (Fig. S1). As illustrated in Fig. 1A, the resulting colloidal suspension of exfoliated MXene nanosheets displayed a distinct Tyndall effect, signifying high dispersibility. Transmission electron microscopy (TEM) images revealed nearly transparent MXene nanosheets with pristine surfaces and large dimensions, which are suitable for constructing lamellar interlayer nanochannels in a nanofluidic membrane (Fig. 1C). Atomic force microscopy (AFM) analyses further determined the lateral dimensions of the MXene nanosheets that ranged from 200 to 500 nm, with an average thickness of approximately 2 nm (Fig. 1D and E). Subsequently, the nanosheets were restacked into a flexible, black-gray membrane through vacuum-assisted filtration (Fig. S2 and 1B). Despite its hydrophilic nature, the free-standing MXene membrane remained stable in water for up to one week under dark conditions, demonstrating mechanical robustness for biosensor applications (Fig. S3A). In addition, XRD characterization after immersion for 1, 2, and 7 days showed a well-defined (002) reflection without a noticeable shift toward lower 2θ, suggesting no evident irreversible interlayer expansion or structural degradation (Fig. S3B). Consistently, the ionic current responses under both external voltage-driven and photothermal diffusion voltage-driven operation remained stable over 7 days, supporting operational integrity under the sensing conditions (Fig. S3C and D). Scanning electron microscopy (SEM) observations confirmed that the MXene membrane consisted of stacked nanosheet layers, resulting in a characteristic lamellar microstructure (Fig. 1F and G). The slit-like gaps formed between nanosheets serve as subnanometer-scale fluidic nanochannels. Additionally, a distinct (002) diffraction peak at 6.22° was observed in the X-ray diffraction (XRD) pattern, corresponding to an interlayer spacing d(002) of 14.22 Å according to Bragg's law (Fig. 1H). Considering the theoretical thickness of a single MXene layer (∼9.8 Å), the effective interlayer spacing (ion diffusion space) within the MXene nanochannels was calculated to be approximately 4.42 Å, supporting efficient ion transport under confinement. To selectively modify the aptamer on the outer surface of the nanochannels (APT@MXene), a gold layer was deposited onto one side of the membrane using physical vapor deposition, as described in our previous work.39 The aptamer for MC-LR, functionalized with sulfhydryl groups, serves as a probe and is subsequently immobilized on the outer surface of the MXene membrane via Au–S bonding. Time of flight secondary ion mass spectrometry (ToF-SIMS) was employed to determine the distribution of the aptamer along APT@MXene. A threshold of 5% peak normalized intensity (indicated by the black dashed line) was applied to define the presence of elements. The Au region was measured at approximately 10 nm, consistent with the deposition thickness (Fig. 1I). In contrast, ToF-SIMS analysis revealed negligible aptamer penetration (effectively 0 nm) from the outermost membrane, confirming its predominant localization on the Au-deposited outer surface. X-ray photoelectron spectroscopy (XPS), coupled with argon ion sputtering, was employed to confirm the successful fabrication of APT@MXene. Spectra were acquired before and after a single Ar-ion sputtering step to a target depth of ∼30 nm. As depicted in Fig. 1J, the inherent O and C elements corresponded to the original MXene structure and its terminal functional groups. Following aptamer grafting, newly observed N 1s (399.8 eV) and P 2p (133.9 eV) peaks were ascribed to the incorporation of the aptamer. After argon ion sputtering, the N 1s and P 2p signals disappeared, while Ti 2p and F 1s signals appeared, further confirming that the aptamer was immobilized on the outer surface of the MXene and subsequently removed by sputtering. Detailed changes in the N 1s and P 2p signals under different conditions are also clearly illustrated in Fig. 1K and L. Taken together, the ToF-SIMS depth profile and the XPS sputtering results consistently indicate that the aptamer is predominantly localized on the Au-deposited outer surface. Additionally, LSCM analysis confirmed successful surface functionalization using the DCFH-labeled aptamer. Strong green fluorescence was observed only after immobilization (Fig. S4). The limited fluorescence penetration depth (∼2.8 µm), compared to the membrane thickness (∼20 µm), further verified that the aptamer was primarily localized on the outer surface. Furthermore, confocal fluorescence images collected from four randomly selected locations on the same membrane showed spatially homogeneous signals, and RuHex chronocoulometry yielded consistent aptamer surface coverage across the membrane (Fig. S5). Together, these results establish an outer-surface-functionalized nanochannel platform (APT@MXene) for subsequent comparisons between conventional external voltage-driven and photothermal diffusion voltage-driven modes.Constraints of the conventional external voltage-driven mode for outer-surface charge modulation
To establish a baseline and delineate the limits of conventional external voltage-driven transduction based on outer-surface functionalization, APT@MXene was first evaluated in the conventional external voltage-driven mode within the trace-concentration regime of MC-LR (Fig. 2A), where both steric hindrance and outer-surface-charge variations are expected to be subtle. Interlayer spacing (XRD, Fig. S6) and surface wettability (static contact angle) were quantified over a broad concentration range encompassing this regime and remained invariant within experimental uncertainty (Fig. 2B and C), indicating that nanochannel geometry and wetting do not appreciably affect ionic transport under these conditions. Molecular docking predicted that MC-LR binding induced a conformational transition of the aptamer from an extended chain to a hairpin-like structure, stabilized by groove-binding noncovalent interactions (Fig. 2D). Consistent with this prediction, CD spectra at high concentrations (≥1 µg L−1) showed decreased ellipticity at 209 nm and a red shift near 272 nm (Fig. 2E), and UV absorption exhibited hypochromicity at 260 nm (Fig. 2F), both hallmark signatures of DNA-aptamer folding. Importantly, these spectral signatures were absent within the trace-concentration window, suggesting that target-induced steric modulation was negligible in the regime of interest. Surface ionic accessibility was further probed by electrochemical impedance spectroscopy (EIS) using the [Fe(CN)6]3−/[Fe(CN)6]4− couple.53 Under these conditions, the charge-transfer resistance (Rct) served as a proxy for the ease with which the anionic probe reached electron-active sites on the MXene surface, integrating steric accessibility and local electrostatics. A discernible decrease in Rct emerged only at high concentrations, consistent with substantial aptamer conformational rearrangements that reduced steric hindrance, while no meaningful change was detected in the trace-concentration window (Fig. 2G). In contrast, chronocoulometry demonstrated that the outer-surface charge density became progressively more negative with increasing MC-LR concentration, with values of −1.38, −1.95, and −2.34 µC cm−2, and the effect was detectable even at 10−4 µg L−1 (Fig. 2H). Despite this measurable increase in outer-surface charge density, the I–V characteristics remained essentially indistinguishable across conditions (Fig. 2I), and at a fixed bias of −2 V the ionic current readout (I = Vext/R) showed no statistically significant difference (Fig. 2J). Taken together, these observations indicated that, in the trace-concentration regime, ionic readout in the conventional external voltage-driven mode was constrained by an effectively unchanged steric-hindrance-dominated resistance baseline, such that small variations in outer-surface charge were insufficient to produce a measurable change in current.Amplification of outer-surface charge modulation in the photothermal diffusion voltage-driven mode
Building on the observation that ionic readout in the conventional external voltage-driven mode was insensitive to minor variations in outer-surface charge due to the invariant channel resistance, we implemented a photothermal diffusion voltage-driven strategy to investigate whether the ionic signal could be actively regulated in the trace-concentration regime. The MXene nanofluidic membrane bridged a two-compartment electrochemical cell filled with identical KCl solutions and Ag/AgCl electrodes, and no external bias was applied. Ionic current was generated and recorded by asymmetrically irradiating one side of the membrane with 808 nm NIR light, thereby creating a temperature gradient (ΔT) across the lamellar nanochannels and establishing the photothermal diffusion voltage-driven mode. Throughout this work, the irradiated side is defined as the hot side and the opposite side is defined as the cold side, with ΔT = Thot − Tcold. Upon NIR illumination, the membrane produced a rapid photocurrent strictly confined to the light-on periods. Repeated on/off cycling yielded stable and reproducible switching between high- and low-current states (Fig. 3B). Owing to the intrinsic photothermal response of MXene, both the ionic current and the nanochannel temperature gradient ΔT scaled approximately linearly with the incident irradiance (Fig. 3C). In contrast to the conventional external voltage-driven case, where the ionic current was invariant, the photothermal diffusion voltage-driven ionic current, measured at zero bias under fixed 808 nm illumination, increased with MC-LR concentration, rising from 15.4 nA (0 µg L−1) to 25.3 nA at 10−4 µg L−1 and 27.9 nA at 10−3 µg L−1 (Fig. 3D and E). These results indicated that photothermal diffusion voltage-driven mode effectively amplifies subtle outer-surface charge variations into measurable ionic current changes on the same APT@MXene device.Mechanistically, previous studies have established that the asymmetric light stimulation of photoresponsive MXene-based nanofluidic system creates a temperature gradient across the membrane, which facilitates selective cation transport.54,55 This permselectivity is governed by surface charge, particularly at low electrolyte concentrations, due to the formation of electric double layers (EDLs) at the charged nanochannel interfaces. The resulting selective cation flux gives rise to a directional photothermal diffusion voltage (Vdiff) under the imposed temperature gradient ΔT. When the nanochannels are connected to symmetric Ag/AgCl electrodes, this diffusion voltage, together with the temperature-dependent redox voltages of the electrodes, manifests experimentally as an open-circuit photovoltage (Voc).56,57 Under non-isothermal conditions, the magnitudes of both Vdiff and Voc increase with enhanced cationic permselectivity. Therefore, we hypothesize that MC-LR binding modulates outer-surface charge, thereby altering cationic permselectivity and tuning Vdiff to induce a detectable change in ionic current.
To substantiate this framework in our system, we measured the transmembrane ionic conductance of the MXene nanofluidic membrane across a wide range of KCl concentrations. As shown in Fig. S8, conductance increased linearly at higher concentrations (10−2 to 1 M) but deviated from bulk scaling and approached saturation at lower concentrations (10−3 to 10−6 M). This behavior reflected competition between bulk conduction and surface charge governed transport, influenced by the Debye length (λD), which is inversely correlated with electrolyte concentration and determines ion selectivity according to Poisson–Boltzmann theory.58–60 Within this regime, we examined the photothermal diffusion voltage-driven ionic-current response of APT@MXene to 10−3 µg per L MC-LR across a range of KCl concentrations (10−1 M to 10−5 M). The largest ionic current change was observed at 10−4 M KCl (Fig. S10), whereas the conventional external voltage-driven system remained essentially unchanged across concentrations (Fig. S11). Having identified this optimal condition, we conducted a quantitative analysis to link the observed current modulation to changes in ionic selectivity. According to the non-isothermal ion-transport theory, the diffusion voltage Vdiff(T) and the experimentally measured open-circuit photovoltage Voc(T) can be expressed as57
 | (1) |
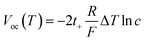 | (2) |
To gain deeper insight into the mechanism underlying ionic signal regulation, we performed a theoretical analysis of ion transport in the photothermal diffusion voltage-driven mode based on the Poisson–Nernst–Planck (PNP) framework and the Einstein–Stokes equation. The model explicitly distinguishes the negatively charged inner walls of the lamellar nanochannels (MXene nanosheets) from the outer surface region associated with aptamer immobilization and subsequent MC-LR treatment. The geometric design and corresponding boundary conditions are detailed in Fig. S13 and Table S5. From a thermodynamic perspective, directional ion transport in a light-responsive MXene nanofluidic membrane under asymmetric light stimulation can be rationalized by the dependence of Gibbs free energy on temperature, expressed as follows:57
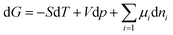 | (3) |
Ultratrace detection in the photothermal diffusion voltage-driven mode
Having established the mechanistic basis of photothermal diffusion voltage-driven amplification of outer-surface charge modulation, we investigated its potential for ultrasensitive detection of trace-level MC-LR. To quantify sensing performance, the target-induced ionic current increase ratio was defined as f = (IAPT@MXene+MC-LR − IAPT@MXene)/IAPT@MXene, where IAPT@MXene+MC-LR and IAPT@MXene represent the ionic current in the presence and absence of MC-LR with specific concentrations, respectively. A threshold of f > 20% was established as a reliable indicator of target recognition, whereas lower ratios were attributed to experimental noise or nonspecific adsorption. Upon exposure to increasing MC-LR concentrations, APT@MXene exhibited a progressive enhancement in ionic current in the photothermal diffusion voltage-driven mode (Fig. 5A). As illustrated in Fig. 5B, f increased markedly from 21.25% at 1 × 10−7 µg L−1 to 81.13% at 1 × 10−3 µg L−1, following a linear relationship with the logarithm of concentration: f = (0.15 × log[MC-LR] + 1.24) × 100% (R2 = 0.996). Based on the 20% threshold, the limit of detection (LOD) was determined to be 1 × 10−7 µg L−1, seven orders of magnitude below the World Health Organization (WHO) guideline value, thereby demonstrating the capacity for early-stage detection of trace MC-LR contamination. Compared with the conventional external voltage-driven mode, the photothermal diffusion voltage-driven strategy afforded a 105-fold improvement in sensitivity. Additionally, Table S4 compares the performance of our photothermal diffusion voltage-driven strategy with that of other advanced techniques, highlighting its superior detection limit across a broad dynamic range. To evaluate the nanochannels' selectivity for MC-LR, we analyzed the current increase ratios induced by major microcystin analogs. The three predominant MC variants found in surface and drinking water sources, namely MC-RR, MC-YR, and MC-LY, were selected as potential interference factors. As shown in Fig. 5C, exposure to 1 × 10−4 µg L−1 of these four analogues yielded corresponding current increase ratios of 63.33%, 3.00%, 10.67%, and 4.67%, respectively. Statistical analysis confirmed significant discrimination (p < 0.001), underscoring the specificity of the photothermal diffusion voltage-driven mode, in stark contrast to the negligible selectivity observed in conventional external voltage-driven measurements (Fig. 5D). Finally, the applicability of the platform was validated in tap, lake, and mineral water samples using standard addition. Across spiking concentrations from 1 × 10−6 to 1 × 10−3 µg L−1, recoveries ranged from 96.76% to 109.30% with relative standard deviations (RSD) below 4.5% (Table 1), indicating negligible matrix interference and robust quantification accuracy. Collectively, these results demonstrate that photothermal diffusion voltage-driven amplification of outer-surface charge modulation enables precise and ultrasensitive control of ionic transport in the MXene nanofluidic membrane, establishing a reliable platform for early-stage environmental monitoring and food safety assessment of MC-LR.| Samples | Spiked (µg L−1) | Detected (µg L−1) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| Tap water | 1 × 10−3 | 1.045 × 10−3 | 104.50 | 1.59 |
| 1 × 10−4 | 1.042 × 10−4 | 104.20 | 0.84 | |
| 1 × 10−5 | 1.093 × 10−5 | 109.30 | 1.16 | |
| 1 × 10−6 | 1.042 × 10−6 | 104.20 | 4.50 | |
| Lake water | 1 × 10−3 | 9.676 × 10−4 | 96.76 | 3.08 |
| 1 × 10−4 | 1.091 × 10−4 | 109.10 | 3.52 | |
| 1 × 10−5 | 1.066 × 10−5 | 106.60 | 3.68 | |
| 1 × 10−6 | 9.939 × 10−7 | 99.39 | 2.99 | |
| Mineral water | 1 × 10−3 | 1.064 × 10−3 | 106.40 | 1.36 |
| 1 × 10−4 | 9.947 × 10−5 | 99.47 | 0.58 | |
| 1 × 10−5 | 1.061 × 10−5 | 106.10 | 2.27 | |
| 1 × 10−6 | 1.019 × 10−6 | 101.90 | 2.11 |
Conclusions
In this work, we have established an outer-surface charge-modulated photothermal diffusion voltage-driven sensing strategy in an MXene nanofluidic membrane for ultratrace detection of MC-LR. Unlike conventional external voltage-driven readout that mainly converts target binding into ionic-current changes through resistance variation, our approach leverages a light-induced transmembrane temperature gradient to generate a diffusion voltage (Vdiff) whose magnitude is governed by K+ permselectivity and thus by outer-surface charge. Consequently, trace-level charge modulations, which produce little change in steric hindrance or the resistance baseline, are transduced into pronounced variations in Vdiff and the zero-bias ionic current. This mechanism enables MC-LR quantification down to 1 × 10−7 µg L−1 with high selectivity against structural analogues and robust performance in real water samples. More broadly, these results demonstrate that outer-surface charge, rather than steric hindrance, can serve as a dominant and addressable control parameter for ionic transport in lamellar nanochannels, providing a general framework for light-regulated nanofluidic sensors and iontronic devices.Author contributions
Yihan Ma: conceptualization, formal analysis, investigation, resources, data curation and writing. Xinyi Yang: methodology, formal analysis, investigation and resources. Bingquan Qi: methodology and investigation. Xiaoping Yang: methodology and investigation. Zhengxu He: methodology and investigation. Ning Feng: methodology and investigation. Li Dai: methodology and investigation. Aiqing Zhang: conceptualization and supervision. Yu Huang: conceptualization, supervision, review & editing and project administration. Fan Xia: supervision and project administration.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings can be found in the article and supplementary information (SI) and are available from the authors upon reasonable request. Supplementary information: experimental procedures, characterization data, and I–V curves. See DOI: https://doi.org/10.1039/d5sc10110g.Acknowledgements
This work was supported by the National Natural Science Foundation of China (22090050, 22176180, U24A20502, and 22125602), the National Key R&D Program of China (2021YFA1200403), the Natural Science Foundation of Hubei Province (2024AFA001), and the Fundamental Research Funds for the Central Universities (CZQ23039).References
- J. Huang, X. Pan and N. Yan, Structural biology and molecular pharmacology of voltage-gated ion channels, Nat. Rev. Mol. Cell Biol., 2024, 25, 904–925 CrossRef CAS PubMed.
- Y. Li, T. Yuan, B. Huang, F. Zhou, C. Peng, X. Li, Y. Qiu, B. Yang, Y. Zhao, Z. Huang and D. Jiang, Structure of human NaV1.6 channel reveals Na+ selectivity and pore blockade by 4,9-anhydro-tetrodotoxin, Nat. Commun., 2023, 14, 1030 CrossRef CAS PubMed.
- Z. He, Y.-C. Tu, C.-W. Tsai, J. Mount, J. Zhang, M.-F. Tsai and P. Yuan, Structure and function of the human mitochondrial MRS2 channel, Nat. Struct. Mol. Biol., 2025, 32, 459–468 CrossRef CAS PubMed.
- W. Xin, J. Fu, Y. Qian, L. Fu, X.-Y. Kong, T. Ben, L. Jiang and L. Wen, Biomimetic KcsA channels with ultra-selective K+ transport for monovalent ion sieving, Nat. Commun., 2022, 13, 1701 CrossRef CAS PubMed.
- L. Leisle, K. Lam, S. Dehghani-Ghahnaviyeh, E. Fortea, J. D. Galpin, C. A. Ahern, E. Tajkhorshid and A. Accardi, Backbone amides are determinants of Cl− selectivity in CLC ion channels, Nat. Commun., 2022, 13, 7508 CrossRef CAS PubMed.
- T. S. Turney, V. Li and S. G. Brohawn, Structural Basis for pH-gating of the K+ channel TWIK1 at the selectivity filter, Nat. Commun., 2022, 13, 3232 CrossRef PubMed.
- Z. Shang, J. Zhao, M. Yang, Y. Xiao, W. Chu, S. Xu, X. Zhang, X. Yi, M. Lin and F. Xia, Precise control of transmembrane current via regulating bionic lipid membrane composition, Sci. Adv., 2024, 10, eadq0118 CrossRef CAS PubMed.
- E. Hoenig, Y. Han, K. Xu, J. Li, M. Wang and C. Liu, In situ generation of (sub) nanometer pores in MoS2 membranes for ion-selective transport, Nat. Commun., 2024, 15, 7911 CrossRef CAS PubMed.
- K. Zou, H. Ling, Q. Wang, C. Zhu, Z. Zhang, D. Huang, K. Li, Y. Wu, W. Xin, X.-Y. Kong, L. Jiang and L. Wen, Turing-type nanochannel membranes with extrinsic ion transport pathways for high-efficiency osmotic energy harvesting, Nat. Commun., 2024, 15, 10231 CrossRef CAS PubMed.
- M. Tsutsui, W.-L. Hsu, C. Hsu, D. Garoli, S. Weng, H. Daiguji and T. Kawai, Transmembrane voltage-gated nanopores controlled by electrically tunable in-pore chemistry, Nat. Commun., 2025, 16, 1089 CrossRef CAS PubMed.
- A. Stuber, A. Douaki, J. Hengsteler, D. Buckingham, D. Momotenko, D. Garoli and N. Nakatsuka, Aptamer Conformational Dynamics Modulate Neurotransmitter Sensing in Nanopores, ACS Nano, 2023, 17, 19168–19179 CrossRef CAS PubMed.
- X. Zhang, Y. Dai, J. Sun, J. Shen, M. Lin and F. Xia, Solid-State Nanopore/Nanochannel Sensors with Enhanced Selectivity through Pore-in Modification, Anal. Chem., 2024, 96, 2277–2285 CrossRef CAS PubMed.
- Z.-Q. Li, L.-Q. Huang, K. Wang and X.-H. Xia, Developing Solid-State Single-, Arrayed-, and Composite-Nanopore Sensors for Biochemical Sensing Applications, Acc. Mater. Res., 2024, 5, 761–771 CrossRef CAS.
- X. Wang, Y. Liu, R. Ren, J. B. Edel and A. P. Ivanov, Single-Molecule Protein Profiling Using Nanopores and Dimeric Aptamer-Modified DNA Carriers, Angew. Chem., Int. Ed., 2025, 64, e202505902 CrossRef CAS PubMed.
- R. T. Arwani, S. C. L. Tan, A. Sundarapandi, W. P. Goh, Y. Liu, F. Y. Leong, W. Yang, X. T. Zheng, Y. Yu, C. Jiang, Y. C. Ang, L. Kong, S. L. Teo, P. Chen, X. Su, H. Li, Z. Liu, X. Chen, L. Yang and Y. Liu, Stretchable ionic–electronic bilayer hydrogel electronics enable in situ detection of solid-state epidermal biomarkers, Nat. Mater., 2024, 23, 1115–1122 CrossRef CAS PubMed.
- C. Wang, J. Li, X. Li, W. Li, Y. Li, Y. Huang, C. Wang, Z. Liu, M. Wang, N. Chen, M. Chen, L. Pan, F. Zhang, J. Bi, L. Li, W. Hu and X. Chen, Bio-inspired organic electrosense transistor for impalpable perception, Sci. Adv., 2025, 11, eads7457 CrossRef CAS PubMed.
- W. Cheng, X. Wang, Z. Xiong, J. Liu, Z. Liu, Y. Jin, H. Yao, T.-S. Wong, J. S. Ho and B. C. K. Tee, Frictionless multiphasic interface for near-ideal aero-elastic pressure sensing, Nat. Mater., 2023, 22, 1352–1360 CrossRef CAS PubMed.
- Y. Huang, L. Liu, C. Luo, W. Liu, X. Lou, L. Jiang and F. Xia, Solid-state nanochannels for bio-marker analysis, Chem. Soc. Rev., 2023, 52, 6270–6293 RSC.
- P. Dauphin-Ducharme, N. Arroyo-Currás and K. W. Plaxco, Correction to “High-Precision Electrochemical Measurements of the Guanine-, Mismatch-, and Length-Dependence of Electron Transfer from Electrode-Bound DNA Are Consistent with a Contact-Mediated Mechanism”, J. Am. Chem. Soc., 2019, 141, 8000 CrossRef CAS PubMed.
- K. K. Leung, J. Gerson, N. Emmons, J. M. Heemstra, T. E. Kippin and K. W. Plaxco, The Use of Xenonucleic Acids Significantly Reduces the In Vivo Drift of Electrochemical Aptamer-Based Sensors, Angew. Chem., Int. Ed., 2024, 63, e202316678 CrossRef CAS PubMed.
- C. Zhao, J. Hou, M. Hill, B. Freeman, H. Wang and H. Zhang, Enhanced Gating Effects in Responsive Sub-nanofluidic Ion Channels, Acc. Mater. Res., 2023, 4, 786–797 CrossRef CAS.
- X. Li, G. Jiang, M. Jian, C. Zhao, J. Hou, A. W. Thornton, X. Zhang, J. Z. Liu, B. D. Freeman, H. Wang, L. Jiang and H. Zhang, Construction of angstrom-scale ion channels with versatile pore configurations and sizes by metal-organic frameworks, Nat. Commun., 2023, 14, 286 CrossRef CAS PubMed.
- P. Liu, X.-Y. Kong, L. Jiang and L. Wen, Ion transport in nanofluidics under external fields, Chem. Soc. Rev., 2024, 53, 2972–3001 RSC.
- C. Zhu, Y. Wu, X. Li, X. Li, X. Liang, L. Jiang and L. Wen, Engineered Nanofluidics for Molecular Recognition and Physical Perception, Angew. Chem., Int. Ed., 2025, 64, e202506689 CrossRef CAS PubMed.
- X. Zhang, H. Cai, T. Hu, M. Lin, Y. Dai and F. Xia, DNA-Functionalized Solid-State Nanochannels with Enhanced Sensing, Acc. Mater. Res., 2025, 6, 285–293 CrossRef CAS.
- I. C. Nova, J. Ritmejeris, H. Brinkerhoff, T. J. R. Koenig, J. H. Gundlach and C. Dekker, Detection of phosphorylation post-translational modifications along single peptides with nanopores, Nat. Biotechnol., 2024, 42, 710–714 CrossRef CAS PubMed.
- R. Wang, Y. Zhang, Q. D. Y. Ma and L. Wu, Recent advances of small molecule detection in nanopore sensing, Talanta, 2024, 277, 126323 CrossRef CAS PubMed.
- X. Li, T. Zhai, P. Gao, H. Cheng, R. Hou, X. Lou and F. Xia, Role of outer surface probes for regulating ion gating of nanochannels, Nat. Commun., 2018, 9, 40 CrossRef PubMed.
- Z. Zhu, D. Wang, Y. Tian and L. Jiang, Ion/Molecule Transportation in Nanopores and Nanochannels: From Critical Principles to Diverse Functions, J. Am. Chem. Soc., 2019, 141, 8658–8669 CrossRef CAS PubMed.
- L. Liang, F. Qin, S. Wang, J. Wu, R. Li, Z. Wang, M. Ren, D. Liu, D. Wang and D. Astruc, Overview of the materials design and sensing strategies of nanopore devices, Coord. Chem. Rev., 2023, 478, 214998 CrossRef CAS.
- T. Sun, Z. Zhao, W. Liu, Z. Xu, H. He, B. Ning, Y. Jiang and Z. Gao, Development of sandwich chemiluminescent immunoassay based on an anti-staphylococcal enterotoxin B Nanobody–Alkaline phosphatase fusion protein for detection of staphylococcal enterotoxin B, Anal. Chim. Acta, 2020, 1108, 28–36 CrossRef CAS PubMed.
- W.-Q. Zhang, Y.-D. Tu, H. Liu, R. Liu, X.-J. Zhang, L. Jiang, Y. Huang and F. Xia, A Single Set of Well-Designed Aptamer Probes for Reliable On-site Qualitative and Ultra-Sensitive Quantitative Detection, Angew. Chem., Int. Ed., 2024, 63, e202316434 CrossRef CAS PubMed.
- M. Lin, M. Yang, Y. Xiao, J. Zhao, Z. Shang, X. Liu, L. Wang, J. Pan, X. Yi, X. Zhang and F. Xia, Graphene Oxide Nanofluidic Ion Channels with Two-Gene Rolling Circle Amplification for Ultrasensitive and Specific Detection of SARS-CoV-2, Anal. Chem., 2025, 97, 22153–22163 CrossRef CAS PubMed.
- K. Loha, T. Boonkoom, H. Pitakjakpipop, I. Alam, A. Treetong, P. Boonbanjong, I. Chatnuntawech, S. Teerapittayanon, U. F. Keyser, A. Schulte and D. Japrung, Structural and Kinetic Profiling of Rolling Circle Amplification via Solid-State Nanopore Sensing Using miR-21 as a Model, ACS Sens., 2025, 10, 7014–7024 CrossRef CAS PubMed.
- N. S. Shrikrishna and S. Gandhi, Nanopore-based sensing for biomarker detection: from fundamental principles to translational diagnostics, J. Nanobiotechnol., 2025, 23, 663 CrossRef CAS PubMed.
- P. Li, J. Zhao, D. Liang, C. Peng, J. Zhu, B. Yeom, Z. Wang, Y. Zhao and W. Ma, Construction of Biomimetic Nanochannel, Property Regulation, and Biomarker Detection, Small, 2025, 21, 2501740 CrossRef CAS PubMed.
- E. M. Nestorovich, T. K. Rostovtseva and S. M. Bezrukov, Residue Ionization and Ion Transport through OmpF Channels, Biophys. J., 2003, 85, 3718–3729 CrossRef CAS PubMed.
- H. Bajaj, S. Acosta Gutierrez, I. Bodrenko, G. Malloci, M. A. Scorciapino, M. Winterhalter and M. Ceccarelli, Bacterial Outer Membrane Porins as Electrostatic Nanosieves: Exploring Transport Rules of Small Polar Molecules, ACS Nano, 2017, 11, 5465–5473 CrossRef CAS PubMed.
- Q. Ma, Y. Li, R. Wang, H. Xu, Q. Du, P. Gao and F. Xia, Towards explicit regulating-ion-transport: nanochannels with only function-elements at outer-surface, Nat. Commun., 2021, 12, 1573 CrossRef CAS PubMed.
- Z. Si, H. Xu, M. Lin, Y. Jiang, Q. Du, H. Ma, H. Liang, P. Gao and F. Xia, Polydopamine-Induced Modification on the Highly Charged Surface of Asymmetric Nanofluidics: A Strategy for Adjustable Ion Current Rectification Properties, Anal. Chem., 2022, 94, 2493–2501 CrossRef CAS PubMed.
- W. Zhang, M. Chen, Q. Ma, Z. Si, S. Jin, Q. Du, L. Zhang, Y. Huang and F. Xia, Role of Outer Surface Probes on Bullet-Shaped Asymmetric Solid-State Nanochannels for Lysozyme Protein Sensing, Anal. Chem., 2024, 96, 2445–2454 CrossRef CAS PubMed.
- L. Dai, W.-Q. Zhang, D. Ding, C. Luo, L. Jiang, Y. Huang and F. Xia, Outer-Surface Functionalized Solid-State Nanochannels for Enhanced Sensing Properties: Progress and Perspective, ACS Nano, 2024, 18, 7677–7687 CrossRef CAS PubMed.
- L. Ma, Z. Liu, B. Ai, J. Man, J. Li, K. Wu and Y. Qiu, Ion transport through short nanopores modulated by charged exterior surfaces, J. Chem. Phys., 2024, 160, 074701 CrossRef CAS PubMed.
- Y. Qiao, J.-J. Hu, Y. Hu, C. Duan, W. Jiang, Q. Ma, Y. Hong, W. H. Huang, F. Xia and X. Lou, Detection of Unfolded Cellular Proteins Using Nanochannel Arrays with Probe-Functionalized Outer Surfaces, Angew. Chem., Int. Ed., 2023, 62, e202309671 CrossRef CAS PubMed.
- J.-J. Hu, W. Jiang, Y. Qiao, Q. Ma, Q. Du, J.-H. Jiang, X. Lou and F. Xia, Enzyme Regulating the Wettability of the Outer Surface of Nanochannels, ACS Nano, 2023, 17, 11935–11945 Search PubMed.
- C. Zhang, X. Zhang, H. Zhang, Z. Gong, X. Ren, M. Guo and Y. Qiu, Modulation of electroosmotic flow through short nanopores by charged exterior surfaces, Electrochim. Acta, 2025, 543, 147623 CrossRef CAS.
- L. Liu, D. Ding, C. Luo, X. Wang, Y. Tian, S.-C. Liu, X. Lou, Y. Huang, L. Mao and F. Xia, Nanochannels with Varied Outer Surface Charges for Protein Discrimination, Anal. Chem., 2025, 97, 10792–10801 CrossRef CAS PubMed.
- Y. Chen, X. Li, X. Yue, W. Yu, Y. Shi, Z. He, Y. Wang, Y. Huang, F. Xia and F. Li, Sub-femtomolar drug monitoring via co-calibration mechanism with nanoconfined DNA probes, Nat. Commun., 2025, 16, 1863 Search PubMed.
- B. Ai, Z. Gong, H. Zhang, Z. Lu, T. Sui and Y. Qiu, Modulation of Ion Distributions and Electroneutrality inside Nanopores by Ion Transport under Electric Fields, ACS Appl. Mater. Interfaces, 2026, 18, 4457–4470 Search PubMed.
- H. Zhang, B. Ai, Z. Gong, T. Sui, Z. S. Siwy and Y. Qiu, Ion Transport through Differently Charged Nanoporous Membranes: From a Single Nanopore to Multinanopores, Anal. Chem., 2025, 97, 19218–19231 Search PubMed.
- M. Lin, J. Zhao, X. Yi, Y. Xiao, Z. Shang, L. Xu, X. Lei, J. Pan, Y. Huang, X. Zhang and F. Xia, Role of the real first interface in regulating ionic signal of nanochannels, Nat. Commun., 2025, 16, 8360 CrossRef CAS PubMed.
- L. Ding, D. Xiao, Z. Lu, J. Deng, Y. Wei, J. Caro and H. Wang, Oppositely Charged Ti3C2T MXene Membranes with 2D Nanofluidic Channels for Osmotic Energy Harvesting, Angew. Chem., Int. Ed., 2020, 59, 8720–8726 CrossRef CAS PubMed.
- N. Nakatsuka, A. Faillétaz, D. Eggemann, C. Forró, J. Vörös and D. Momotenko, Aptamer Conformational Change Enables Serotonin Biosensing with Nanopipettes, Anal. Chem., 2021, 93, 4033–4041 CrossRef CAS PubMed.
- J. Lao, S. Wu, J. Gao, A. Dong, G. Li and J. Luo, Electricity generation based on a photothermally driven Ti3C2Tx MXene nanofluidic water pump, Nano Energy, 2020, 70, 104481 CrossRef CAS.
- J. Yeom, A. Choe, J. Lee, J. Kim, J. Kim, S. H. Oh, C. Park, S. Na, Y.-E. Shin, Y. Lee, Y. G. Ro, S. K. Kwak and H. Ko, Photosensitive ion channels in layered MXene membranes modified with plasmonic gold nanostars and cellulose nanofibers, Nat. Commun., 2023, 14, 359 CrossRef CAS PubMed.
- S. Hong, G. Zou, H. Kim, D. Huang, P. Wang and H. N. Alshareef, Photothermoelectric Response of Ti3C2Tx MXene Confined Ion Channels, ACS Nano, 2020, 14, 9042–9049 CrossRef CAS PubMed.
- K. Chen, L. Yao and B. Su, Bionic Thermoelectric Response with Nanochannels, J. Am. Chem. Soc., 2019, 141, 8608–8615 CrossRef CAS PubMed.
- Q. Ma, W. Chu, X. Nong, J. Zhao, H. Liu, Q. Du, J. Sun, J. Shen, S.-M. Lu, M. Lin, Y. Huang and F. Xia, Local Electric Potential-Driven Nanofluidic Ion Transport for Ultrasensitive Biochemical Sensing, ACS Nano, 2024, 18, 6570–6578 CrossRef CAS PubMed.
- L. Ma, X. An, F. Song and Y. Qiu, Effective Charged Exterior Surfaces for Enhanced Ionic Diffusion through Nanopores under Salt Gradients, J. Phys. Chem. Lett., 2022, 13, 5669–5676 CrossRef CAS PubMed.
- L. Ma, Z. Liu, J. Man, J. Li, Z. S. Siwy and Y. Qiu, Modulation mechanism of ionic transport through short nanopores by charged exterior surfaces, Nanoscale, 2023, 15, 18696–18706 RSC.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |






