Open Access Article
Open Access ArticleA difluorocarbene-triggered annulation/ring expansion cascade via sequential single-atom-insertions: direct assembly of 2-fluoroquinazolinones from azo compounds
Yingxian
Tian
,
Yiyan
Xiong
,
Jingyu
Wei
,
Gongming
Zhu
,
Junbiao
Chang
and
Bingxian
Liu
 *
*
Pingyuan Laboratory, State Key Laboratory of Antiviral Drugs, School of Pharmaceutical Sciences, Henan Normal University, Xinxiang, Henan 453007, China. E-mail: liubingxian@htu.edu.cn; liubx1120@163.com
First published on 3rd March 2026
Abstract
An efficient difluorocarbene-mediated tandem strategy for the synthesis of 2-fluoroquinazolin-4(3H)-ones via a sequential single-atom-insertion process has been developed. The method features a unique cascade starting from readily available azo compounds, which undergo an initial difluorocarbene-triggered annulation to form 3-halobenzopyrazole intermediates. These key intermediates are then converted into the final 2-fluoroquinazolinones through a second difluorocarbene-triggered ring expansion. This metal-free, redox-neutral protocol exhibits broad substrate scope and excellent functional group tolerance. Mechanistic investigations, including intermediate detection experiments, control experiments, isotopic labeling experiments, HRMS analysis, and Hammett studies, support the proposed reaction pathway and identify the formation of the halobenzopyrazole intermediate as a key step. The synthetic utility of the products is further demonstrated through diverse derivatizations and preliminary biological evaluation, revealing promising antitumor activity for certain fluorinated compounds compared to their defluorinated analogues, highlighting the value of the retained fluorine atom.
Introduction
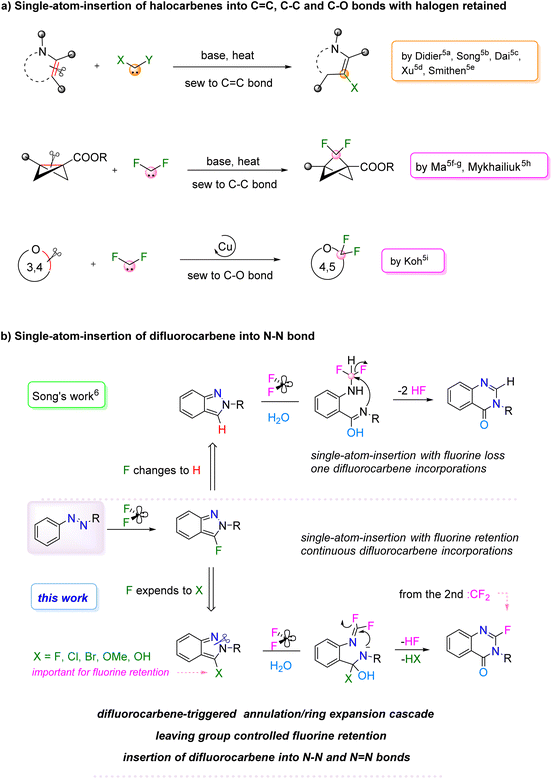
Skeletal editing has emerged as a transformative strategy in modern synthetic chemistry, enabling direct modification of molecular cores without the need for de novo synthesis.1 Among these approaches, single-atom-insertion stands out for its exceptional atom- and step-economy, offering a powerful tool for the rapid diversification of heterocyclic scaffolds.1f–i,2 Despite its potential, the development of efficient and general methods for single-atom skeletal editing remains a significant challenge, particularly those capable of preserving valuable functional groups such as fluorine.Carbene-mediated single-atom-insertion protocols have gained attention as versatile tools for precise skeletal editing, with recent advances demonstrating remarkable scope in the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond cleavage system.3–5 Notably, research groups including those of Didier,5a Song,5b Dai,5c Xu,5d and Smithen5e have successively reported the synthesis of halogenated heterocycles using halocarbene precursors (Fig. 1a). This kind of transformation retains the halogen atom, endowing the product with enhanced potential for further functionalization. However, among carbene insertions, only limited examples have achieved fluorine retention. This is notably the case for difluorocarbene (:CF2), which has been employed as a one-carbon unit in ring-expansion reactions.5a,f–i,6 For instance, the synthesis of difluorinated bicyclo[1.1.1]pentanes from bicyclo[1.1.0]butanes (BCBs) via difluorocarbene insertion into a C–C bond has been reported by Ma5f,g and Mykhailiuk.5h Koh's group also developed a copper-catalyzed approach for skeletal expansion of oxygen-containing heterocycles by incorporation of CF2 into a C–O bond.5i Beyond these limited examples, further studies revealed that fluorine retention during difluorocarbene insertion remains challenging. Specifically, Koh's group5j and Song's group6 independently realized the single-atom-insertion of difluorocarbene into C–N and N–N bonds, both resulting in complete fluorine loss. Nevertheless, in Song's work, although fluorine loss occurred, the transformation provided a new skeletal editing route to quinazolin-4(3H)-ones, a privileged class of nitrogen-containing heterocycles with broad pharmaceutical relevance.7 Given fluorine's crucial role in enhancing drug potency, metabolic stability, and target specificity,8 the development of strategies to incorporate fluorine into this pharmacophore is highly desirable. Although some methods have striven to introduce fluorinated functional groups into this skeleton,9 the direct construction of 2-fluoroquinazolin-4(3H)-ones remains unexplored. To address this gap, we herein report a novel difluorocarbene-mediated tandem process that enables the direct conversion of azo compounds into 2-fluoroquinazolin-4(3H)-ones (Fig. 1b). The transformation process involves an initial cyclization of azobenzene initiated by difluorocarbene to form a key fluorinated benzopyrazole intermediate, which subsequently undergoes a second difluorocarbene insertion to furnish the final product.10–12 This strategy not only achieves single-atom-insertion with retention of fluorine but also unveils a previously unrecognized reactivity of difluorocarbene,13–15 wherein it facilitates both annulation with azo compounds and insertion into the corresponding intermediate in a single operation. To our knowledge, reaction systems that allow continuous difluorocarbene incorporation are still limited.16
C bond cleavage system.3–5 Notably, research groups including those of Didier,5a Song,5b Dai,5c Xu,5d and Smithen5e have successively reported the synthesis of halogenated heterocycles using halocarbene precursors (Fig. 1a). This kind of transformation retains the halogen atom, endowing the product with enhanced potential for further functionalization. However, among carbene insertions, only limited examples have achieved fluorine retention. This is notably the case for difluorocarbene (:CF2), which has been employed as a one-carbon unit in ring-expansion reactions.5a,f–i,6 For instance, the synthesis of difluorinated bicyclo[1.1.1]pentanes from bicyclo[1.1.0]butanes (BCBs) via difluorocarbene insertion into a C–C bond has been reported by Ma5f,g and Mykhailiuk.5h Koh's group also developed a copper-catalyzed approach for skeletal expansion of oxygen-containing heterocycles by incorporation of CF2 into a C–O bond.5i Beyond these limited examples, further studies revealed that fluorine retention during difluorocarbene insertion remains challenging. Specifically, Koh's group5j and Song's group6 independently realized the single-atom-insertion of difluorocarbene into C–N and N–N bonds, both resulting in complete fluorine loss. Nevertheless, in Song's work, although fluorine loss occurred, the transformation provided a new skeletal editing route to quinazolin-4(3H)-ones, a privileged class of nitrogen-containing heterocycles with broad pharmaceutical relevance.7 Given fluorine's crucial role in enhancing drug potency, metabolic stability, and target specificity,8 the development of strategies to incorporate fluorine into this pharmacophore is highly desirable. Although some methods have striven to introduce fluorinated functional groups into this skeleton,9 the direct construction of 2-fluoroquinazolin-4(3H)-ones remains unexplored. To address this gap, we herein report a novel difluorocarbene-mediated tandem process that enables the direct conversion of azo compounds into 2-fluoroquinazolin-4(3H)-ones (Fig. 1b). The transformation process involves an initial cyclization of azobenzene initiated by difluorocarbene to form a key fluorinated benzopyrazole intermediate, which subsequently undergoes a second difluorocarbene insertion to furnish the final product.10–12 This strategy not only achieves single-atom-insertion with retention of fluorine but also unveils a previously unrecognized reactivity of difluorocarbene,13–15 wherein it facilitates both annulation with azo compounds and insertion into the corresponding intermediate in a single operation. To our knowledge, reaction systems that allow continuous difluorocarbene incorporation are still limited.16
Results and discussion
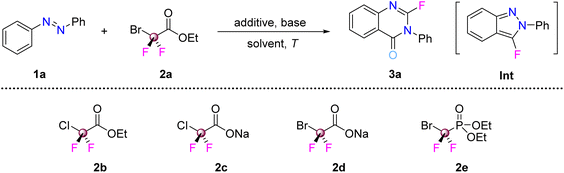
Our investigation began with the hypothesis that azo compounds might engage with difluorocarbene in a [2 + 1] cycloaddition pathway. To our surprise, treatment of azobenzene with BrCF2CO2Et under basic conditions did not yield the expected diaziridine or rearrangement-annulation products as with dichlorocarbene. Instead, unexpected 2-fluoroquinazolin-4(3H)-one 3a was isolated in good yield (Table 1, entry 1). Monitoring the reaction by TLC (thin layer chromatography) revealed the rapid formation and subsequent consumption of an intermediate, which was identified as 3-fluoro-1-phenyl-1H-benzo[d]pyrazole after careful isolation and characterization. This finding indicated that the reaction did not achieve the cyclopropanation product via a direct carbene insertion into the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond, but rather underwent a tandem sequence involving the annulation of azobenzene with difluorocarbene to a fluorinated benzopyrazole, followed by a second difluorocarbene-triggered ring expansion. The reaction conditions for the cascade were investigated then. The effects of different bases (K2CO3, Cs2CO3, NaHCO3, K3PO4, and Et3N) on the yield of 3a were studied. However, it was determined that Na2CO3 was the most promising, affording 3a in 68% yield out of all six bases tested (Table 1, entries 2–6). Other solvents (PhCl, DCM, 1,4-dioxane, and THF) proved unsuitable for this reaction, leading to either trace amounts or no formation of 3a (entries 7–10). Elevating the reaction temperature did not improve the product yield, whilst lowering it considerably reduced the reaction efficiency (entries 11 and 12). Next, the impact of difluorocarbene precursors (2b–2e) was evaluated, with BrCF2CO2Et emerging as the preferred reagent (entries 13–16). Control experiments (either no additive or no base) were also carried out (entries 17 and 18). When NaBr was omitted from the reaction, the yield of 3a was reduced slightly from 72% to 64%. We speculate that the presence of bromide ions may moderate the generation rate of difluorocarbene, avoiding the rapid accumulation of highly reactive :CF2 that tends to undergo side reactions (e.g., dimerization).17 A slower, more controlled release of difluorocarbene facilitates its sequential reaction with both the azobenzene substrate and the fluorobenzopyrazole intermediate, thereby improving the reaction yield and reducing byproduct formation. When the base was omitted from the reaction, no product was detected. Scale-up synthesis of product 3a demonstrated maintained isolated yield at a 1.0 mmol scale (entry 19). The identified intermediate was also tested under the standard conditions, giving the desired product in high yield (entry 20).
N bond, but rather underwent a tandem sequence involving the annulation of azobenzene with difluorocarbene to a fluorinated benzopyrazole, followed by a second difluorocarbene-triggered ring expansion. The reaction conditions for the cascade were investigated then. The effects of different bases (K2CO3, Cs2CO3, NaHCO3, K3PO4, and Et3N) on the yield of 3a were studied. However, it was determined that Na2CO3 was the most promising, affording 3a in 68% yield out of all six bases tested (Table 1, entries 2–6). Other solvents (PhCl, DCM, 1,4-dioxane, and THF) proved unsuitable for this reaction, leading to either trace amounts or no formation of 3a (entries 7–10). Elevating the reaction temperature did not improve the product yield, whilst lowering it considerably reduced the reaction efficiency (entries 11 and 12). Next, the impact of difluorocarbene precursors (2b–2e) was evaluated, with BrCF2CO2Et emerging as the preferred reagent (entries 13–16). Control experiments (either no additive or no base) were also carried out (entries 17 and 18). When NaBr was omitted from the reaction, the yield of 3a was reduced slightly from 72% to 64%. We speculate that the presence of bromide ions may moderate the generation rate of difluorocarbene, avoiding the rapid accumulation of highly reactive :CF2 that tends to undergo side reactions (e.g., dimerization).17 A slower, more controlled release of difluorocarbene facilitates its sequential reaction with both the azobenzene substrate and the fluorobenzopyrazole intermediate, thereby improving the reaction yield and reducing byproduct formation. When the base was omitted from the reaction, no product was detected. Scale-up synthesis of product 3a demonstrated maintained isolated yield at a 1.0 mmol scale (entry 19). The identified intermediate was also tested under the standard conditions, giving the desired product in high yield (entry 20).
| Entry | 2 | Base | Solvent | T (°C) | Yield (%) |
|---|---|---|---|---|---|
| a Reaction conditions: 1a (0.2 mmol, 1.0 equiv.), 2a (1 mmol, 5.0 equiv.), base (2.0 equiv.), and NaBr (0.1 mmol, 0.5 equiv.) in MeCN (4.0 mL) at 120 °C under an air atmosphere for 48 h; NMR yields using CH2Br2 as an internal standard; n.d. = not detected. b Isolated yield. c Et3N (0.1 mmol, 0.5 equiv.). d Without NaBr. e 1 mmol scale, 72 h. f Int instead of 1a. | |||||
| 1 | 2a | Na2CO3 | MeCN | 120 | 72 (68)b |
| 2 | 2a | K2CO3 | MeCN | 120 | 24 |
| 3 | 2a | Cs2CO3 | MeCN | 120 | n.d. |
| 4 | 2a | NaHCO3 | MeCN | 120 | 24 |
| 5 | 2a | K3PO4 | MeCN | 120 | 18 |
| 6c | 2a | Et3N | MeCN | 120 | 63 |
| 7 | 2a | Na2CO3 | PhCl | 120 | n.d. |
| 8 | 2a | Na2CO3 | DCM | 120 | Trace |
| 9 | 2a | Na2CO3 | 1,4-Dioxane | 120 | n.d. |
| 10 | 2a | Na2CO3 | THF | 120 | n.d. |
| 11 | 2a | Na2CO3 | MeCN | 130 | 68 |
| 12 | 2a | Na2CO3 | MeCN | 100 | Trace |
| 13 | 2b | Na2CO3 | MeCN | 120 | 10 |
| 14 | 2c | Na2CO3 | MeCN | 120 | n.d. |
| 15 | 2d | Na2CO3 | MeCN | 120 | n.d. |
| 16 | 2e | Na2CO3 | MeCN | 120 | n.d. |
| 17d | 2a | Na2CO3 | MeCN | 120 | 64 |
| 18 | 2a | — | MeCN | 120 | n.d. |
| 19e | 2a | Na2CO3 | MeCN | 120 | 71 |
| 20f | 2a | Na2CO3 | MeCN | 120 | 88 |
Once the optimal conditions were determined, the scope of the azobenzene substrate was investigated (Fig. 2). Various symmetrical aryl azo compounds (1a–1r) were reacted with BrCF2CO2Et under the optimized reaction conditions, affording the desired products (3a–3r) in 32–90% yields. When the azo substrate contained electron-donating groups, such as alkyl and alkoxy substituents, on the aryl ring, the reaction occurred smoothly, affording the corresponding products 3e–3h in 62–90% yield. However, electron-deficient substituents, such as trifluoromethyl, nitro, and ester groups, were not successfully transformed into the target products. Azo substrates featuring weakly electron-withdrawing groups, like chloride or bromide substituents, were transformed into their corresponding heterocyclic products in low yields (3b and 3c, 34% and 40%, respectively). An alkyl substituent at the ortho position of the phenyl ring led to the isolation of both the final product (3j, 32%) and a fluorobenzopyrazole intermediate (4j′, 46%). A methoxyl substituent only afforded the fluorobenzopyrazole intermediate (4k′, 42%). When 4j′ and 4k′ were subjected to the reaction under the standard conditions, the corresponding 3j was isolated in 6% yield, while 3k was only detected by HRMS. The distinct outcomes of ortho-substituted substrates reveal that steric hindrance (for 4j′) primarily slows the second difluorocarbene-triggered ring expansion, while electronic or coordinative interference (for 4k′) completely inhibits this step, highlighting the differential sensitivity of the two cascade steps to substituent properties. For the ortho-isopropyl substrate (4j′), the hindrance is purely steric. This slows but does not completely block the second difluorocarbene insertion, allowing a low-yield conversion to 3j. In contrast, for the ortho-methoxy substrate (4k′), the proximal oxygen atom with lone pairs likely causes electronic or coordinative interference.5j,18 This deactivates the nitrogen center, inhibiting its nucleophilic attack on the second difluorocarbene and thereby halting the reaction completely. Regio-isomers (3l & 3l′ and 3m & 3m′, 38–47%) were obtained when substrates with substituents in the meta-position on the phenyl ring were reacted. Multiple substituted phenyl azo compounds also delivered the corresponding products (3n–3r, 32–92%). Unsymmetric aryl azo compounds produced inseparable isomers in most cases (3s–3ac, 3s′–3ab′). For the majority of examples, the difference in electronic effects between the two aromatic rings was too small to achieve good regioselectivity. Nonetheless, this electronic difference did influence the ratio of the two isomers. Azo compounds featuring electron-donating groups were found to be more reactive than those without substituents or with electron-withdrawing substituents. Furthermore, the corresponding product was formed as the predominant product when the phenyl ring featured the electron-donating substituents. In particular, only a single product (3ac) was formed in 43% yield when the phenyl ring featured the strongly electron-donating N,N-dimethyl group. The results revealed that electron-rich phenyl rings might have contributed to the formation of a necessary transition state during the reaction. Finally, the tolerance of aryl–alkyl azo compounds was investigated (3ad–3ak). The desired products were isolated in 34–80% yield when alkyl, cyclohexyl, heterocyclic alkyl groups substituted azo compounds were used, which makes the reaction potentially more attractive in drug molecule synthesis.
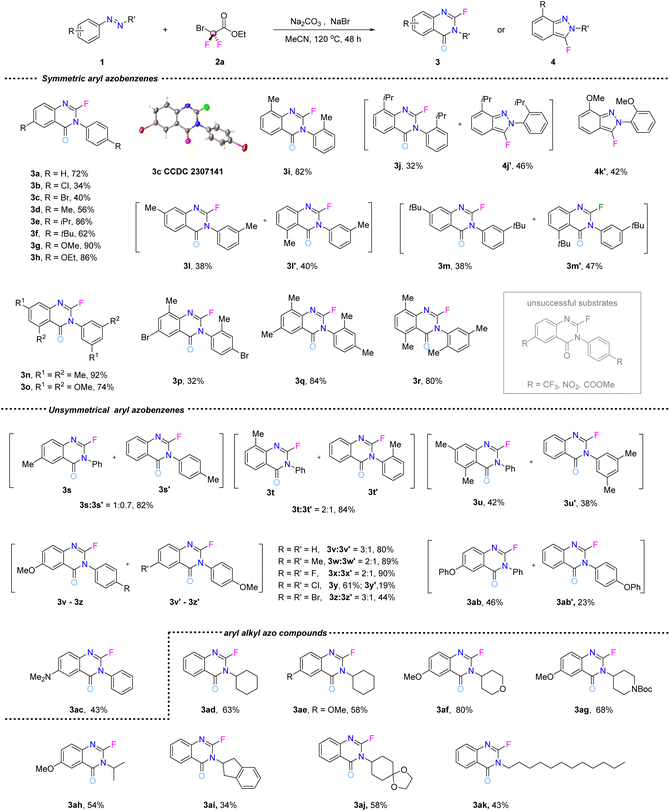 | ||
| Fig. 2 Substrate scope. All reactions unless otherwise stated were carried out with 1 (0.2 mmol), BrCF2CO2Et (1 mmol), NaBr (0.1 mmol) and Na2CO3 (0.4 mmol) in MeCN (4 mL) heated at 120 °C for 48 h. | ||
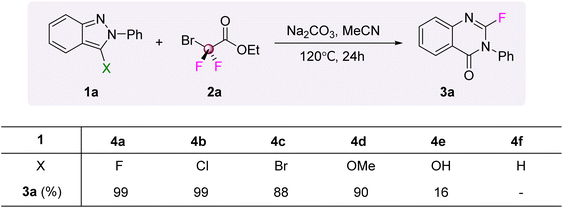
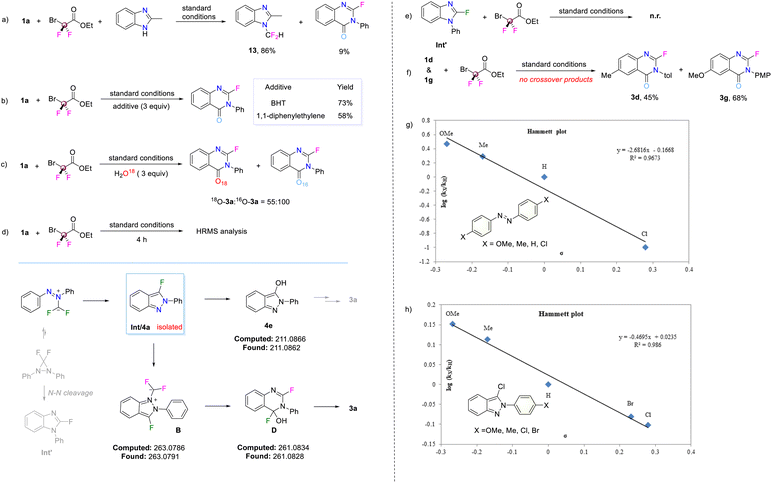
As the fluorobenzopyrazole skeleton was confirmed as the intermediate in the reaction cascade, the single-atom insertion step starting from this intermediate was optimized. The modified conditions while removing the NaBr additive and shortening the reaction time enabled efficient conversion to the corresponding product. The ring expansion process leading to a quinazolin-4(3H)-one scaffold from the fluorobenzopyrazole appears analogous to the results reported by Song for substrates unsubstituted at the 3-position. The key distinction lies in whether the fluorine atom is retained. This fluorine retention seems to be a specific outcome stemming from the use of a fluoro-substituted benzopyrazole intermediate, rather than representing a fundamentally novel reaction paradigm. However, under our conditions, the unsubstituted benzopyrazole (4f) did not yield any of the desired product, and the starting materials were recovered unchanged (Fig. 3). Besides, some different leaving groups such as Cl, Br, and OMe (4b–4d) were tested and all facilitated the formation of the target products, albeit with considerable variation in efficiency (yields ranging from 88% to 99%). The results indicate that the presence of a leaving group at the 3-position of the benzopyrazoles is of paramount importance for the reaction's success, providing a distinctive reaction pathway from the unsubstituted benzopyrazole (4f). Free hydroxyl groups only offered the product in a low yield, which may be due to the competition between the hydroxyl and nitrogen atoms when reacting with difluorocarbene (4e).
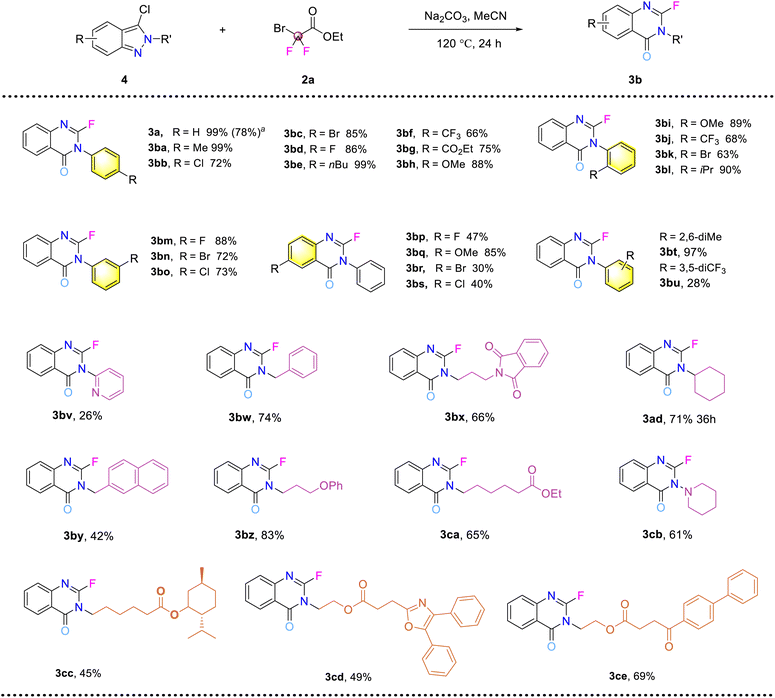
To reduce the limitations of the cascade reaction of azobenzene, the reaction using chlorobenzopyrazoles as the starting substrates was then investigated (Fig. 4). Scale-up synthesis was conducted using the easily accessed 4b, affording the desired product in 78% yield. Substituents on the N-phenyl ring all gave the corresponding products in moderate to excellent yields (3ba–3bo, 63–99%). Electron-donating groups such as alkyl groups (3ba, 3be, 3bl, 3bt) and methoxyl groups (3bh, 3bi) were favorable for the reaction, leading to efficient product synthesis. The introduction of halogens had a relatively small impact on reaction efficiency (3bb–3bd, 3bm–3bo). However, when the ortho-position was occupied by bulky Br, the yield of the corresponding product was reduced to a moderate level (3bk), potentially affected by steric hindrance which was significant in this system. Electron-withdrawing groups (CF3 and COOEt) were also tolerated by the reaction system but with reduced efficiency (3bf–3bg, 3bj). The 3,5-di-CF3 substituted substrate produced the product only in low yield (3bu, 28%). This observation of unfavorable experimental results with electron-withdrawing groups was consistent with findings in the azobenzene reaction system. An N-pyridinyl substrate featuring an electron deficient aromatic ring was tested in the reaction, affording the product in 26% yield (3bv). However, N-alkyl substrates all generated the corresponding product in moderate to good yields (3bw–3ca, 42–83%). Among these, phthalimide, cyclohexyl, naphthyl and phenol groups tethered to the chlorobenzopyrazole skeleton via an alkyl linker were all tolerated. A saturated heterocyclic ring was also tested to give a good result (3cb, 61%). Meanwhile, in order to demonstrate the potential of this reaction for modifying drug molecules, substrates from a natural fragment (menthol) and two drug molecule fragments (oxaprazine and fenbufen) were studied, yielding the corresponding products in moderate yields (3cc–3ce, 45–69%).
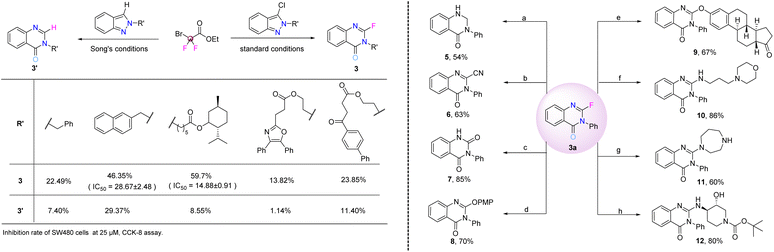
In order to show the potential impact of fluorine atom retention on the biological activity of the products, some fluorine-containing and non-fluorinated compounds (3bw, 3by, 3cc–3cevs.3bw′, 3by′, 3cc′–3ce′) were synthesized and their inhibitory effects on tumor cell proliferation were preliminarily evaluated. Notably, two fluorinated compounds displayed promising activity against SW480 cells, with IC50 values of 28.67 ± 2.48 µM for 3by and 14.88 ± 0.91 µM for 3cc. In contrast, none of the non-fluorinated counterparts showed detectable inhibition at 25 µM (Fig. 5, left, see the SI for details). These preliminary biological results are consistent with the well-recognized role of fluorine in enhancing drug potency and target specificity, confirming that the retained fluorine atom in our products is crucial for their antiproliferative activity, which highlights the practical value of our fluorine-retaining synthetic strategy. To further demonstrate the utility of this protocol, synthetic derivatizations were performed (Fig. 5, right). We demonstrated that product 3a could be subjected to a wide range of chemical modifications to synthesize compounds with biological and pharmacological relevance. Defluorination reduction of 3a using NaBH4 delivered the dihydroquinazolinone derivative 5 in 54% yield (reaction a). Nucleophilic substitution with TMSCN yielded cyanide product 6 in moderate yield (reaction b). Hydrolysis under alkaline conditions gave quinazoline-dione skeleton 7 in 85% yield (reaction c). Nucleophilic substitution reaction also proceeded smoothly with phenols and amines, affording the corresponding products in moderate to high yields (reactions d–h, 8–12, 60–86%). Notably, product 3a was successfully tethered to estrone, generating 9 in 67% yield under mild conditions. Three different pharmacologically relevant amine moieties delivered the amination products with good reaction efficiency (reactions f–h).
Mechanistic insights into the reaction were gained through several experiments. When 2-methylbenzimidazole was present, the carbene-capture byproduct (13) was formed as the dominant product instead of compound 3a (Fig. 6a). Furthermore, the reaction proceeded smoothly in the presence of radical scavengers such as BHT or 1,1-diphenylethylene, affording the desired product 3a in good yield, indicating that radical species were not involved (Fig. 6b). Isotopic labelling experiments were subsequently performed to determine the origin of the oxygen atom in the target product. Using three equivalents of H218O, 3a was isolated as a 55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 (HRMS ratio) mixture of 18O-3a and 16O-3a (Fig. 6c), suggesting that carbonyl oxygen likely originated from trace water in the solvent. In order to further identify some potential intermediates in the reaction process, high-resolution mass spectrometry (HRMS) analysis was performed during the early stages of the reaction (Fig. 6d). As noted, intermediate Int (4a) was detected and successfully isolated, while other possible intermediates (4e, B, D) were detected only by HRMS. Notably, intermediate 4e, presumably formed via hydrolysis of 4a, was identified as a side intermediate rather than a productive species, as evidenced by its low reactivity when directly used as the substrate (Fig. 3), which implies it does not participate in the main reaction pathway. In a productive pathway, 4a can further react with difluorocarbene to form the N
100 (HRMS ratio) mixture of 18O-3a and 16O-3a (Fig. 6c), suggesting that carbonyl oxygen likely originated from trace water in the solvent. In order to further identify some potential intermediates in the reaction process, high-resolution mass spectrometry (HRMS) analysis was performed during the early stages of the reaction (Fig. 6d). As noted, intermediate Int (4a) was detected and successfully isolated, while other possible intermediates (4e, B, D) were detected only by HRMS. Notably, intermediate 4e, presumably formed via hydrolysis of 4a, was identified as a side intermediate rather than a productive species, as evidenced by its low reactivity when directly used as the substrate (Fig. 3), which implies it does not participate in the main reaction pathway. In a productive pathway, 4a can further react with difluorocarbene to form the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CF2 ylide intermediate B, which is then converted to intermediate Dvia nucleophilic addition by –OH, followed by a ring-opening/ring-closing sequence. In contrast, another possible skeleton (Int′), which could be generated by defluorination-rearrangement from a [2 + 1] cycloaddition intermediate, was synthesized and found to be unreactive (Fig. 6e). A competition experiment was carried out to determine whether the reaction was intermolecular or not. A one-pot reaction of substrates 1d and 1g with BrCF2CO2Et produced only products 3d and 3g, with no crossover products detected (Fig. 6f). To assess the influence of electronic effects on the reaction efficiency, Hammett studies were performed using para-substituted azobenzenes and para-substituted phenyl chlorobenzopyrazoles (Fig. 6g and h). Good linearity was observed plotting the relative rates (lg(kX/kH)) against the substitution constant (σ). Negative ρ values (−2.68 for azobenzenes and −0.47 for chlorobenzopyrazoles) indicate positive charge accumulation in the transition states of both the difluorocarbene-triggered annulation and the difluorocarbene-triggered ring expansion steps, respectively. The substantially larger |ρ| value for the azobenzene system suggests that the initial annulation is rate-determining, whereas the smaller |ρ| value for the chlorobenzopyrazole system implies that, although a positively charged transition state is involved, the ring-expansion step is not rate-limiting. Therefore, it can be considered that the generation of intermediate 4a is the rate determining step of the reaction, and is strongly influenced by the electronic nature of the substituents, consistent with our experimental observations in the azobenzene system.
CF2 ylide intermediate B, which is then converted to intermediate Dvia nucleophilic addition by –OH, followed by a ring-opening/ring-closing sequence. In contrast, another possible skeleton (Int′), which could be generated by defluorination-rearrangement from a [2 + 1] cycloaddition intermediate, was synthesized and found to be unreactive (Fig. 6e). A competition experiment was carried out to determine whether the reaction was intermolecular or not. A one-pot reaction of substrates 1d and 1g with BrCF2CO2Et produced only products 3d and 3g, with no crossover products detected (Fig. 6f). To assess the influence of electronic effects on the reaction efficiency, Hammett studies were performed using para-substituted azobenzenes and para-substituted phenyl chlorobenzopyrazoles (Fig. 6g and h). Good linearity was observed plotting the relative rates (lg(kX/kH)) against the substitution constant (σ). Negative ρ values (−2.68 for azobenzenes and −0.47 for chlorobenzopyrazoles) indicate positive charge accumulation in the transition states of both the difluorocarbene-triggered annulation and the difluorocarbene-triggered ring expansion steps, respectively. The substantially larger |ρ| value for the azobenzene system suggests that the initial annulation is rate-determining, whereas the smaller |ρ| value for the chlorobenzopyrazole system implies that, although a positively charged transition state is involved, the ring-expansion step is not rate-limiting. Therefore, it can be considered that the generation of intermediate 4a is the rate determining step of the reaction, and is strongly influenced by the electronic nature of the substituents, consistent with our experimental observations in the azobenzene system.
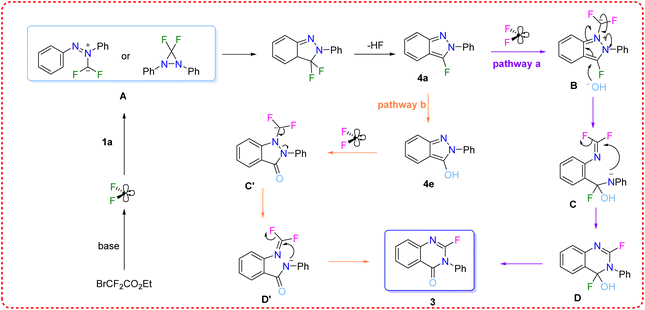
Based on the experimental results and literature precedent, Fig. 7 illustrates a proposed reaction mechanism for the reaction. Initially, BrCF2CO2Et undergoes base-mediated dehalogenation and decarboxylation to generate difluorocarbene. This species reacts with azobenzene to form either an ylide or a 3,3-difluoro-diaziridine intermediate, which rearranges with HF elimination to afford key intermediate 4a. Intermediate 4a then proceeds via two potential pathways to afford the final product. In pathway a, 4a reacts with another difluorocarbene to create ylide intermediate B. This undergoes ring-opening followed by recyclization with fluoride elimination, yielding the six-membered ring intermediate D. Subsequent HF elimination under alkaline conditions affords the final product. An alternative pathway is showcased as pathway b. In this scenario, 4a undergoes nucleophilic substitution firstly to generate intermediate 4e which can similarly yield the product through a ring-opening/recyclization cascade facilitated by another difluorocarbene (:CF2). However, given the low efficiency observed when 4e was tested experimentally, this pathway is likely less favourable.
Conclusions
In conclusion, this work presents a novel difluorocarbene-mediated tandem process that enables the direct conversion of readily available azo compounds into valuable 2-fluoroquinazolin-4(3H)-ones. The reaction firstly proceeds via a difluorocarbene triggered annulation of the azo substrate to form a key fluorinated 3-halobenzopyrazole intermediate, which subsequently undergoes a second difluorocarbene-induced ring expansion via single-atom insertion, ultimately delivering the fluorinated quinazolinone product with retention of fluorine atom. The pivotal 3-halobenzopyrazole species, which serves as the mechanistic linchpin connecting the two distinct reactivity modes of difluorocarbene in a single operation, is identified and expanded as a type of complementary substrate. The reaction has broad substrate scope (tolerating various azo compounds and pre-formed benzopyrazoles), and good functional group compatibility under metal-free and redox-neutral conditions. Comprehensive mechanistic investigations, including isotopic labeling, intermediate isolation, HRMS analyses, and Hammett analyses, provide strong support for the proposed tandem pathway and reveal that the initial annulation step is rate-determining and sensitive to electronic effects. Preliminary biological evaluation demonstrated that certain fluorinated products exhibit promising antiproliferative activity against cancer cell lines, while their defluorinated analogues are inactive, underscoring the critical role of fluorine in modulating bioactivity. Furthermore, the synthetic versatility of the 2-fluoroquinazolin-4(3H)-one scaffold was showcased through diverse derivatization reactions, including linkage with pharmacologically relevant fragments.Author contributions
B. L. conceptualized the project. Y. T. performed the experiments and analyzed the results. Y. X. and J. W. assisted in performing the experiments. G. Z. performed the experiments on anticancer activity. J. C. and B. L. supervised and directed the project. B. L. wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: further details of the experimental procedures, 1H, 13C and 19F NMR, HRMS data, X-ray crystallographic data for 3c. See DOI: https://doi.org/10.1039/d6sc00897f.CCDC 2307141 contains the supplementary crystallographic data for this paper.19
Acknowledgements
We acknowledge the financial support for this work from the National Key R&D Program of China (2021YFC0864700), the NSFC (No. 82130103), and the Central Plains Scholars and Scientists Studio Fund (2018002). We are also thankful for the financial support from “NMPA Key Laboratory for Research and Evaluation of Innovative Drug” and “Henan Key Laboratory of Organic Functional Molecules and Drug Innovation”.References
-
(a) C. Ma, C. W. Lindsley, J.-B. Chang and B. Yu, Rational Molecular Editing: A New Paradigm in Drug Discovery, J. Med. Chem., 2024, 67, 11459–11466 CrossRef CAS PubMed
; (b) B. W. Joynson and L. T. Ball, Skeletal editing: interconversion of arenes and heteroarenes, Helv. Chim. Acta, 2023, 106, e202200182 CrossRef CAS
; (c) F. P. Wu, J. L. Tyler and F. Glorius, Diversity-Generating Skeletal Editing Transformations, Acc. Chem. Res., 2025, 58, 893–906 CrossRef CAS PubMed
; (d) P. Xu and A. Studer, Skeletal Editing through Cycloaddition and Subsequent Cycloreversion Reactions, Acc. Chem. Res., 2025, 58, 647–658 CrossRef CAS PubMed
; (e) M. Peplow, ‘Almost magical’: chemists can now move single atoms in and out of a molecule's core, Nature, 2023, 618, 21–24 CrossRef CAS
; (f) J. Jurczyk, J. Woo and S. F. Kim, Single-atom logic for heterocycle editing, Nat. Synth., 2022, 1, 352–364 CrossRef CAS PubMed
; (g) T. Yuan and L. Shi, Recent advances in carbon atom addition for ring-expanding single-atom skeletal editing, Org. Chem. Front., 2024, 11, 7318–7332 RSC
; (h) L. Lv, Enantiodivergent dearomative single-atom skeletal editing: a magic methodology, Trends Chem., 2025, 7, 165–167 CrossRef CAS
; (i) C. Hui, Z. Wang, S. Wang and C. Xu, Molecular editing in natural product synthesis, Org. Chem. Front., 2022, 9, 1451–1457 RSC
.
-
(a) E. F. Plachinski and T. P. Yoon, Single-atom editing with light, Science, 2024, 386, 27 CrossRef CAS PubMed
; (b) X. Zhang, Q. Song, S. Liu, P. Sivaguru, Z. Liu, Y. Yang, Y. Ning, E. A. Anderson, G. d. Ruiter and X. Bio, Asymmetric dearomative single-atom skeletal editing of indoles and pyrroles, Nat. Chem., 2025, 17, 215–225 CrossRef CAS PubMed
; (c) Z. Siddiqi and D. Sarlah, Reimagining Dearomatization: Arenophile-Mediated Single-Atom Insertions and π-Extensions, Acc. Chem. Res., 2025, 58, 1134–1150 CrossRef CAS PubMed
; (d) N. Jiao, Q. Qin, L. Zhang, J. Wei, X. Qiu, S. Hao and X. D. An, Direct oxygen insertion into C-C bond of styrenes with air, Nat. Commun., 2024, 15, 9015 CrossRef PubMed
; (e) S. Wiesler, G. Sennari, M. V. Popescu, K. E. Gardner, K. Aida, R. S. Paton and R. Sarpong, Late-stage benzenoid-to-troponoid skeletal modification of the cephalotanes exemplified by the total synthesis of harringtonolide, Nat. Commun., 2024, 15, 4125 CrossRef CAS PubMed
; (f) H. Lu, J. Chang and H. Wei, Transition Metal-Catalyzed Nitrogen Atom Insertion into Carbocycles, Acc. Chem. Res., 2025, 58, 933–946 CrossRef CAS PubMed
; (g) B. Ghosh, P. Kafle, R. Mukherjee, R. Welles, D. Herndon, K. M. Nicholas, Y. Shao and I. Sharma, Sulfenylnitrene-mediated nitrogen-atom insertion for late-stage skeletal editing of N-heterocycles, Science, 2025, 387, 102–107 CrossRef CAS PubMed
.
-
(a) Z. Liu, P. Sivaguru, Y. Ning, Y. Wu and X. Bi, Skeletal Editing of (Hetero)Arenes Using Carbenes, Chem.–Eur. J., 2023, 29, e202301227 CrossRef CAS PubMed
; (b) Z. Liu, X. Zhang, P. Sivaguru and X. Bi, Triftosylhydrazone in Single-Atom Skeletal Editing, Acc. Chem. Res., 2025, 58, 130–149 CrossRef CAS PubMed
; (c) Q. Q. Xie and J. B. Hu, A Journey of the Development of Privileged Difluorocarbene Reagents TMSCF2X (X = Br, F, Cl) for Organic Synthesis, Acc. Chem. Res., 2024, 57, 693–713 CrossRef CAS PubMed
; (d) J. Krishnan, K. Sahib, K. G. Nair and R. R. Paul, Advances in the Synthetic Utility of Difluorocarbene Generated from TMSCF3 (Ruppert-Prakash Reagent) and Its Derivatives, Chem. Rec., 2025, 25, e202400243 Search PubMed
.
-
(a) V. G. Alfonso, K. d. l. Vega-Hernández and M. G. Suero, Single-Carbon Insertion into Single C–C Bonds with Diazirines, J. Am. Chem. Soc., 2025, 147, 57–62 CrossRef CAS PubMed
; (b) M. R. Fructos, M. Besora, A. A. C. Braga, M. M. Díaz-Requejo, F. Maseras and P. J. Pérez, Mechanistic Studies on Gold-Catalyzed Direct Arene C–H Bond Functionalization by Carbene Insertion: The Coinage-Metal Effect, Organometallics, 2017, 36, 172–179 CrossRef CAS
; (c) M. Mortén, M. Hennum and T. Bonge-Hansen, Synthesis of quinoline-3-carboxylates by a Rh (II)-catalyzed cyclopropanation-ring expansion reaction of indoles with halodiazoacetates, Beilstein J. Org. Chem., 2015, 11, 1944–1949 CrossRef PubMed
; (d) S. Peeters, L. N. Berntsen, P. Rongved and T. Bonge-Hansen, Cyclopropanation–ring expansion of 3-chloroindoles with α-halodiazoacetates: novel synthesis of 4-quinolone 3-carboxylic acid and norfloxacin, Beilstein J. Org. Chem., 2019, 15, 2156–2160 CrossRef CAS PubMed
; (e) B. D. Dherange, P. Q. Kelly, J. P. Liles, M. S. Sigman and M. D. Levin, Carbon atom insertion into pyrroles and indoles promoted by chlorodiazirines, J. Am. Chem. Soc., 2021, 143, 11337–11344 CrossRef CAS PubMed
; (f) F. P. Wu, C. C. Chintawar, R. Lalisse, P. Mukherjee, S. Dutta, J. Tyler and F. Glorius, Ring expansion of indene by photoredox-enabled functionalized carbon-atom insertion, Nat. Catal., 2024, 7, 242–251 Search PubMed
; (g) A. F. Khlebnikov, M. S. Novikov, Y. G. Gorbunova, E. E. Galenko, K. I. Mikhailov, V. V. Pakalnis and M. S. Avdontceva, Isoxazolium N-ylides and 1-oxa-5-azahexa-1,3,5-trienes on the way from isoxazoles to 2H-1,3-oxazines, Beilstein J. Org. Chem., 2014, 10, 1896–1905 Search PubMed
; (h) A. N. Koronatov, N. V. Rostovskii, A. F. Khlebnikov and M. S. Novikov, Rh (II)-Catalyzed Ring Expansion of Pyrazoles with Diazocarbonyl Compounds as a Method for the Preparation of 1,2-Dihydropyrimidines, J. Org. Chem., 2018, 83, 9210–9219 Search PubMed
; (i) J. R. Manning and H. M. Davies, Efficient route to 2H-1,3-oxazines through ring expansion of isoxazoles by rhodium carbenoids, Tetrahedron, 2008, 64, 6901–6908 Search PubMed
; (j) E. Hyland, P. Q. Kelly, A. M. McKillop, B. D. Dherange and M. D. Levin, Unified access to pyrimidines and quinazolines enabled by N–N cleaving carbon atom insertion, J. Am. Chem. Soc., 2022, 144, 19258–19264 Search PubMed
; (k) M. Tsuda, T. Morita, Y. Morita, J. Takaya and H. Nakamura, Methylene Insertion into Nitrogen-Heteroatom Single Bonds of 1,2-Azoles via a Zinc Carbenoid: An Alternative Tool for Skeletal Editing, Adv. Sci., 2024, 11, 2307563 CrossRef CAS PubMed
; (l) S. Liu, Y. Yang, Q. Song, Z. Liu, Y. Lu, Z. Wang and X. Bi, Tunable molecular editing of indoles with fluoroalkyl carbenes, Nat. Chem., 2024, 16, 988–997 Search PubMed
; (m) L. Li, H. Chen, M. Liu, Q. Zhu, H. Zhang, G. d. Ruiter and X. Bi, Silver-Catalyzed Dearomative Skeletal Editing of Indazoles by Donor Carbene Insertion, Chem.–Eur. J., 2024, 30, e202304227 Search PubMed
; (n) L. Li, Y. Ning, H. Chen, Y. Ning, P. Sivaguru, P. Liao and X. Bi, Dearomative Insertion of Fluoroalkyl Carbenes into Azoles Leading to Fluoroalkyl Heterocycles with a Quaternary Center, Angew. Chem., Int. Ed., 2024, 63, e202313807 Search PubMed
; (o) S. Liu, Y. Yang, Q. Song, Z. Liu, P. Sivaguru, Y. Zhang, G. d. Ruiter, E. A. Anderson and X. Bi, Halogencarbene-free Ciamician-Dennstedt single-atom skeletal editing, Nat. Commun., 2024, 15, 9998 CrossRef CAS PubMed
; (p) J. C. Reisenbauer, O. Green, A. Franchino, P. Finkelstein and B. Morandi, Late-stage diversification of indole skeletons through nitrogen atom insertion, Science, 2022, 377, 1104–1109 Search PubMed
.
-
(a)
R. Ghazali, E. Schick, S. Lèpre, J. Kranich and D. Didier, Regiospecific Skeletal Editing of Azetines towards Halogenated Pyrroles, ChemRxiv, 2025, preprint, DOI:10.26434/chemrxiv-2025-25grb;
(b) C. Li, L. Chen, H. Wang, Z. Yan, B. Lyu, W. Lyu and S. Song, C–F Bond Insertion into Indoles with CHBr2F: An Efficient Method to Synthesize Fluorinated Quinolines and Quinolones, Chin. J. Chem., 2024, 42, 1128–1132 CrossRef CAS
; (c) D. Ma, B. S. Martin, K. S. Gallagher, T. Saito and M. Dai, One-carbon insertion and polarity inversion enabled a pyrrole strategy to the total syntheses of pyridine-containing Lycopodium alkaloids: complanadine A and lycodine, J. Am. Chem. Soc., 2021, 143, 16383–16387 CrossRef CAS PubMed
; (d) H. Guo, S. Qiu and P. Xu, One-Carbon Ring Expansion of Indoles and Pyrroles: A Straightforward Access to 3-Fluorinated Quinolines and Pyridines, Angew. Chem., Int. Ed., 2024, 63, e202317104 CrossRef CAS PubMed
; (e) C. W. Rees and C. E. Smithen, The mechanism of heterocyclic ring expansions. Part I. The reaction of 2,3-dimethylindole with dichlorocarbene, J. Chem. Soc., 1964, 928–937 RSC
; (f) X. Ma, D. L. Sloman, Y. Han and D. J. Bennett, A selective synthesis of 2,2-difluorobicyclo [1.1. 1] pentane analogues: “BCP-F2”, Org. Lett., 2019, 21, 7199–7203 CrossRef CAS PubMed
; (g) X. Ma, W. Pinto, L. N. Pham, D. L. Sloman and Y. Han, Synthetic Studies of 2, 2-Difluorobicyclo [1.1.1] pentanes (BCP-F2): The Scope and Limitation of Useful Building Blocks for Medicinal Chemists, Eur. J. Org Chem., 2020, 29, 4581–4605 CrossRef
; (h) R. M. Bychek, V. Hutskalova, Y. P. Bas, O. A. Zaporozhets, S. Zozulya, V. V. Levterov and P. K. Mykhailiuk, Difluoro-substituted bicyclo [1.1.1] pentanes for medicinal chemistry: design, synthesis, and characterization, J. Org. Chem., 2019, 84, 15106–15117 CrossRef CAS PubMed
; (i) T. D. Tan, F. Zhou, K. P. Quirion, Y. Q. Wang, D. Z. W. Ng, X. Luo, E. C. Y. Chan, P. Liu and M. J. Koh, Catalytic difluorocarbene insertion enables access to fluorinated oxetane isosteres, Nat. Chem., 2025, 17, 719–726 CrossRef CAS PubMed
; (j) F. Zhou, T. D. Tan and M. J. Koh, Site-Selective Carbonylation of Azetidines via Copper-Catalyzed Difluorocarbene Insertion, Angew. Chem., Int. Ed., 2025, 64, e202505033 CrossRef CAS PubMed
.
- Y. Zhou, F. Chen, Z. Li, J. Dong, J. Li, B. Zhang and Q. Song, Single-atom skeletal editing of 2H-indazoles enabled by difluorocarbene, Sci. China. Chem., 2023, 66, 1975–1981 CrossRef CAS
.
-
(a) C. X. Wang, Z. L. Zhang, Q. K. Yin, J. L. Tu, J. E. Wang, Y. H. Xu, Y. Rao, T. M. Ou, S. L. Huang, D. Li, H. G. Wang, Q. J. Li, J. H. Tan, S. B. Chen and Z. S. Huang, Evaluation of New Quinazolinone Derivatives that Inhibit Bloom Syndrome Protein (BLM) Helicase, Trigger DNA Damage at the Telomere Region, and Synergize with PARP Inhibitors, J. Med. Chem., 2020, 63, 9752–9772 CrossRef CAS PubMed
; (b) R. Kaur, S. K. Manjal and R. K. Rawal, Kumar, K. Recent synthetic and medicinal perspectives of tryptanthrin, Bioorg. Med. Chem., 2017, 25, 4533–4552 CrossRef CAS PubMed
; (c) J. W. Chern, P. L. Tao, K. C. Wang, A. Gutcait, S. W. Liu, M. H. Yen, S. L. Chien and J. K. Rong, Studies on Quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides. 8.1, 2 Synthesis and Pharmacological Evaluation of Tricyclic Fused Quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides as Potential Alpha1-adrenoceptor Antagonists, J. Med. Chem., 1998, 41, 3128–3141 CrossRef CAS PubMed
; (d) J. Zhang, J. Liu, Y. Ma, D. Ren, P. Cheng, J. Zhao, F. Zhang and Y. Yao, One-pot synthesis and antifungal activity against plant pathogens of quinazolinone derivatives containing an amide moiety, Med. Chem. Lett., 2016, 26, 2273–2277 CrossRef CAS PubMed
; (e) E. P. Gillis, K. Parcella, M. Bowsher, J. H. Cook, C. Iwuagwu, B. N. Naidu, M. Patel, K. Peese and R. Fridell, et al., Potent Long-Acting Inhibitors Targeting the HIV-1 Capsid Based on a Versatile Quinazolin-4-one Scaffold, J. Med. Chem., 2023, 66, 1941–1954 CrossRef CAS PubMed
.
-
(a) S. Purser, P. R. Moore, S. Swallow and V. Gouverneur, Fluorine in medicinal chemistry, Chem. Soc. Rev., 2008, 37, 320–330 RSC
; (b) N. A. Meanwell, Fluorine and Fluorinated Motifs in the Design and Application of Bioisosteres for Drug Design, J. Med. Chem., 2018, 61, 5822–5880 CrossRef CAS PubMed
; (c) B. M. Johnson, Y. Z. Shu, X. L. Zhuo and N. A. Meanwell, Metabolic and Pharmaceutical Aspects of Fluorinated Compounds, J. Med. Chem., 2020, 63, 6315–6386 CrossRef CAS PubMed
.
-
(a) S. Almeida, R. Marti, E. Vanoli, S. Abele and S. Tortoioli, One-pot synthesis of trifluoromethylated quinazolin-4 (3H)-ones with trifluoroacetic acid as CF3 source, J. Org. Chem., 2018, 83, 5104–5113 CrossRef CAS PubMed
; (b) L. C. Wang, S. Du, Z. Chen and X. F. Wu, FeCl3-Mediated Synthesis of 2-(Trifluoromethyl) quinazolin-4 (3H)-ones from Isatins and Trifluoroacetimidoyl Chlorides, Org. Lett., 2020, 22, 5567–5571 CrossRef CAS PubMed
; (c) J. B. Lee, M. E. Kang, J. Kim, C. Y. Lee, J. M. Kee, K. Myung, J. U. Park and S. Y. Hong, Direct diversification of unmasked quinazolin-4 (3H)-ones through orthogonal reactivity modulation, Chem. Commun., 2017, 53, 10394–10397 RSC
; (d) T. Delouche, A. Gadiry-Diallo, T. Besson, S. Ogoshi and C. Fruit, Photocatalyst-Free, Visible-Light-Mediated C–H Perfluoroalkylation of Quinazolin-4 (3H)-ones with perfluoroalkyl Iodides, Synthesis, 2023, 55, 3670–3684 Search PubMed
.
- M. Shigeno, M. Imamatsu, Y. Kai, M. Kiriyama, S. Ishida, K. Nozawa-Kumada and Y. Kondo, Construction of 1, 2, 3-Benzodiazaborole by Electrophilic Borylation of Azobenzene and Nucleophilic Dialkylative Cyclization, Org. Lett., 2021, 23, 8023–8027 Search PubMed
.
-
(a) T. Fujiu, K. Izumi and S. Sekiguchi, Reaction of Azobenzene with Dichlorocarbene in Phase-transfer-catalyzed System, Bull. Chem. Soc. Jpn., 1985, 58, 1055–1056 CrossRef CAS
; (b) L. S. Hegedus and B. R. Lundmark, The course of the reaction of azobenzene with chromium carbene complexes, J. Am. Chem. Soc., 1989, 111, 9194–9198 CrossRef CAS
.
-
(a) P. Sharma, A. Kumar and M. Sharma, Synthesis of 4-[2-(2-Methylprop-1-Enylidene)-2,3-Dihydro-1H-Benzimidazole-1-Yl]-1-Napthol via Azo Group Insertion of Dimethylvinylidene Carbene under Phase Transfers Catalysis Conditions, Catal. Commun., 2006, 7, 611–617 CrossRef CAS
; (b) D. J. Tindall, C. Werlé, R. Goddard, P. Philipps, C. Farès and A. Fürstner, Structure and reactivity of half-sandwich Rh (+3) and Ir (+3) carbene complexes. Catalytic metathesis of azobenzene derivatives, J. Am. Chem. Soc., 2018, 140, 1884–1893 CrossRef CAS PubMed
.
- Selected reviews on reactions of difluorocarbene:
(a) A. D. Dilman and V. V. Levin, Difluorocarbene as a Building Block for Consecutive Bond-Forming Reactions, Acc. Chem. Res., 2018, 51, 1272–1280 CrossRef CAS PubMed
; (b) Z. Feng, Y. L. Xiao and X. G. Zhang, Transition-Metal (Cu, Pd, Ni)-Catalyzed Difluoroalkylation via Cross-Coupling with Difluoroalkyl Halides, Acc. Chem. Res., 2018, 51, 2264–2278 CrossRef CAS PubMed
; (c) J. H. Lin and J. C. Xiao, Fluorinated Ylides/Carbenes and Related Intermediates from Phosphonium/Sulfonium Salts, Acc. Chem. Res., 2020, 53, 1498–1510 CrossRef CAS PubMed
; (d) X. Y. Wang, X. Wang and J. B. Wang, Application of carbene chemistry in the synthesis of organofluorine compounds, Tetrahedron, 2019, 75, 949–964 CrossRef CAS
; (e) Y. M. Li, J. B. Luo and Y. J. Jiang, C–F bond functionalizations via fluorinated carbenes, Org. Chem. Front., 2023, 10, 5782–5804 RSC
; (f) K. Fuchibe and J. Ichikawa, Generation of difluorocarbenes and introduction of fluorinated one carbon units into carbonyl and related compounds, Chem. Commun., 2023, 59, 2532–2540 RSC
.
- Selected papers on reactions of difluorocarbene via a ylide intermediate:
(a) X. X. Ma, J. K. Su and Q. L. Song, Unconventional Transformations of Difluorocarbene with Amines and Ethers, Acc. Chem. Res., 2023, 56, 592–607 CrossRef CAS PubMed
; (b) Y. Kim, J. Heo, D. Kim, S. Chang and S. Seo, Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer, Nat. Commun., 2020, 11, 4761 CrossRef CAS PubMed
; (c) G. Zhang, Q. Shi, M. Hou, K. Yang, S. Wang, S. Wang, W. Li, C. Li, J. Qiu, H. Xu, L. Zhou, C. Wang, S. J. Li, Y. Lan and Q. l. Song, Atom Recombination of Difluorocarbene Enables 3-Fluorinated Oxindoles from 2-Aminoarylketones, CCS Chem., 2022, 4, 1671–1679 CrossRef CAS
; (d) J. K. Su, X. X. Ma, Z. L. Ou and Q. L. Song, Deconstructive Functionalizations of Unstrained Carbon-Nitrogen Cleavage Enabled by Difluorocarbene, ACS Cent. Sci., 2020, 6, 1819–1826 CrossRef CAS PubMed
; (e) J. K. Su, X. Y. Hu, H. Huang, Y. Guo and Q. L. Song, Difluorocarbene enables to access 2-fluoroindoles from ortho-vinylanilines, Nat. Commun., 2021, 12, 4986 CrossRef CAS PubMed
; (f) A. Liu, C. F. Ni, Q. Q. Xie and J. B. Hu, TMSCF2Br-Enabled Fluorination–Aminocarbonylation of Aldehydes: Modular Access to α-Fluoroamides, Angew. Chem., Int. Ed., 2022, 61, e202115467 CrossRef CAS PubMed
; (g) J. K. Su, C. B. Li, X. Y. Hu, Y. Guo and Q. L. Song, Deaminative Arylation and Alkenyaltion of Aliphatic Tertiary Amines with Aryl and Alkenylboronic Acids via Nitrogen Ylides, Angew. Chem., Int. Ed., 2022, 61, e202212740 CrossRef CAS PubMed
; (h) H. Cui, C. Ban, F. Zhu, J. Yuan, J. Du, Y. Huang, Q. Xiao, C. Huang, J. Huang and Q. Zhu, Difluorocarbene-Triggered Acyl Rearrangement Reaction: A Strategy for the Direct Introduction of the gem-Difluoromethylene Group, Org. Lett., 2023, 25, 99–103 CrossRef CAS PubMed
.
- Selected papers on [2+1] reactions of difluorocarbene:
(a) F. Wang, T. Luo, J. Hu, Y. Wang, H. Krishnan, P. Jog, S. Ganesh, G. Prakash and G. Olah, Synthesis of gem-Difluorinated Cyclopropanes and Cyclopropenes: Trifluoromethyltrimethylsilane as a Difluorocarbene Source, Angew. Chem., Int. Ed., 2011, 50, 7153–7157 CrossRef CAS PubMed
; (b) F. Wang, W. Zhang, J. M. Zhu, H. F. Li, K. W. Huang and J. B. Hu, Chloride ion-catalyzed generation of difluorocarbene for efficient preparation of gem-difluorinated cyclopropenes and cyclopropanes, Chem. Commun., 2011, 47, 2411–2413 RSC
; (c) A. Liu, X. H. Zhang, F. Zhao, C. F. Ni and J. B. Hu, Controllable Fluorocarbon Chain Elongation: TMSCF2Br-Enabled Trifluorovinylation and Pentafluorocyclopropylation of Aldehydes, J. Am. Chem. Soc., 2024, 146, 1806–1812 CrossRef CAS PubMed
; (d) R. M. Bychek, V. Hutskalova, Y. P. Bas, O. A. Zaporozhets, S. Zozulya, V. V. Levterov and P. K. Mykhailiuk, Difluoro-Substituted Bicyclo [1.1.1] pentanes for Medicinal Chemistry: Design, Synthesis, and Characterization, J. Org. Chem., 2019, 84, 15106–15117 CrossRef CAS PubMed
; (e) X. S. Ma, D. L. Sloman, Y. X. Han and D. J. Bennett, A Selective Synthesis of 2,2-Difluorobicyclo [1.1.1] pentane Analogues: “BCP-F2”, Org. Lett., 2019, 21, 7199–7203 CrossRef CAS PubMed
; (f) X. Zhang, Y. Ning, C. Tian, G. Zanoni and X. Bi, Asymmetric [2+1] cycloaddition of difluoroalkyl-substituted carbenes with alkenes under rhodium catalysis: Synthesis of chiral difluoroalkyl-substituted cyclopropanes, iScience, 2023, 26, 105896 CrossRef CAS PubMed
; (g) P. Rulliere, P. Cyr and A. B. Charette, Difluorocarbene Addition to Alkenes and Alkynes in Continuous Flow, Org. Lett., 2016, 18, 1988–1991 CrossRef CAS PubMed
.
-
(a) H. Sheng, Z. Chen and Q. Song, Palladium-Catalyzed Difluorocarbene Transfer Enabled Divergent Synthesis of γ-Butenolides and Ynones from Iodobenzene and Terminal Alkynes, J. Am. Chem. Soc., 2024, 146, 1722–1731 CrossRef CAS PubMed
; (b) A. Liu, C. Ni, Q. Xie and J. Hu, Transition-Metal-Free Controllable Single and Double Difluoromethylene Formal Insertions into C–H Bonds of Aldehydes with TMSCF2Br, Angew. Chem., Int. Ed., 2023, 62, e202217088 CrossRef CAS PubMed
; (c) H. Sheng, J. Su, X. Li, X. Li and Q. Song, Double Capture of Difluorocarbene by 2-Aminostyrenes Enables the Construction of 3-(2,2-Difluoroethyl)-2-fluoroindoles, Org. Lett., 2021, 23, 7781–7786 CrossRef CAS PubMed
; (d) A. Liu, X. Zhang, F. Zhao, C. Ni and J. Hu, Controllable Fluorocarbon Chain Elongation: TMSCF2Br-Enabled Trifluorovinylation and Pentafluorocyclopropylation of Aldehydes, J. Am. Chem. Soc., 2024, 146, 1806–1812 Search PubMed
.
- M. D. Kosobokov, V. V. Levin, M. I. Struchkova and A. D. Dilman, Nucleophilic bromo-and iododifluoromethylation of aldehydes, Org. Lett., 2014, 16, 3784 CrossRef CAS PubMed
.
-
(a) H. Sheng, J. Su, X. Li and Q. Song, Deconstrutive difunctionalizations of cyclic ethers enabled by difluorocarbene to access difluoromethyl ethers, CCS Chem., 2022, 4, 3820 Search PubMed
; (b) R. Zhang, Q. Li, Q. Xie, C. Ni and J. Hu, Difluorocarbene-Induced Ring-Opening Difluoromethylation-Halogenation of Cyclic (Thio) Ethers with TMSCF2X (X = Br, Cl), Chem.–Eur. J., 2021, 27, 17773 CrossRef CAS PubMed
; (c) D. Zuo, H. Xu, S. Tang, M. Hua, C. Song, Y. Li, J. Zhao, B. Yang and P. Li, Difluorocarbene/H2O Serving as a CO2 Equivalent for Insertion into Oxiranes, Org. Lett., 2025, 27, 5471–5475 CrossRef CAS PubMed
.
-
CCDC 2307141: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2hfrxm
.
| This journal is © The Royal Society of Chemistry 2026 |