Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Alumanyl silanides as multifunctional reagents for olefin cycloaddition, CO hydrosilylation, and reductive CO coupling
Moritz
Ludwig†
 a,
Johannes
Voigtland†
a,
Johannes
Voigtland†
 a,
Petra
Vasko
a,
Petra
Vasko
 b,
Sebastian
Stigler
b,
Sebastian
Stigler
 a and
Shigeyoshi
Inoue
a and
Shigeyoshi
Inoue
 *a
*a
aTUM School of Natural Sciences, Department of Chemistry, Catalysis Research Center and Institute for Silicon Chemistry, Technical University of Munich, Lichtenbergstraße 4, 85748 Garching bei München, Germany. E-mail: s.inoue@tum.de
bDepartment of Chemistry, P.O. Box 55 (A.I. Virtasen Aukio 1), 00014 University of Helsinki, Finland
First published on 15th January 2026
Abstract
Alumanyl silanides represent a rare class of main group complexes, characterized by an anionic Al–Si bond, stabilized through an intimate ion pair. Computations revealed pronounced multiple-bond character in these alumanyl silanides, which is further enhanced upon counter ion sequestration. Despite these electronic features, such bonding motifs remain largely unexplored in experimental chemistry. In this work, we investigate the reactivity of a sodium alumanyl silanide, stabilized by bulky silyl groups and an N-heterocyclic imine (NHI), towards alkenes, alkynes, aldehydes, CO2, and CO. The addition of ethylene, styrene, or mesitylaldehyde to said alumanyl silanide affords the corresponding [2+2]-cycloaddition products, characterized by polarized Al–Si–C–C and Al–Si–C–O heterocycles, respectively. Furthermore, the investigated alumanyl silanide captures two equivalents of CO2. One molecule is inserted between the Al center and its adjacent NHI ligand, whereas a second molecule of CO2 adds across the central Al–Si bond. Moreover, the title compound selectively adds one equivalent of CO to the Al–Si core. This is followed by 1,2-hydrogen migration from the silicon center to the carbon in the formed Al–Si–C–O cycle, showcasing a rare main group-mediated hydrosilylation of CO. The mechanism for this formation is examined using DFT calculations, which reveal the generation of a cyclic carbene intermediate as the key step. At low temperatures, the intermediate is successfully trapped in the presence of additional CO and an N-heterocyclic carbene (NHC), yielding silanyl ethynolates via reductive coupling of two CO molecules.
Introduction
The generation of C–C bonds from carbon monoxide as a C1 feedstock is of fundamental importance for large-scale industrial processes, such as the Fischer–Tropsch, Monsanto, or Cativa processes.1 To efficiently break the stable C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) O bond, these processes rely on transition-metal-based catalysts that enable low-energy reaction pathways but are often associated with toxicity and high procurement cost.2 In recent years, more earth-abundant and environmentally friendly low-valent s- and p-block elements have shown transition-metal-like reactivity in the activation and transformation of carbon monoxide.3
O bond, these processes rely on transition-metal-based catalysts that enable low-energy reaction pathways but are often associated with toxicity and high procurement cost.2 In recent years, more earth-abundant and environmentally friendly low-valent s- and p-block elements have shown transition-metal-like reactivity in the activation and transformation of carbon monoxide.3
The first report using molten potassium for CO coupling dates back to the 1830s.4 Since then, group 1 metals (Li, Na, K)5 have been investigated for CO homologation via C–C coupling. Modern approaches are based on related organometallic reducing agents, such as benzyl potassium,6 dilithiomethane,7 alkali metal amides8,9 and phosphides.10,11 Potassium and lithium silylamides have even enabled complete cleavage of the CO bond, resulting in the formation of cyanides and hexamethyldisiloxane (Scheme 1, I).12 According to calculations, key step for this reaction was the formation of a transient carbene after nucleophilic attack at CO and subsequent 1,3-silyl migration. Since then, further systems, relying on the synergistic effect of alkali metals and p-block elements such as boryl lithium13,14 or simple silanides,15,16 have been reported to insert CO in the highly polarized B–Li or Si–Li bond (Scheme 1, II).17 These findings complement the well-established reactivity of classical organolithium compounds.18 Furthermore, systems based on a cooperative effect of multiple reactive centers, including low-valent dimeric3,19–26 or multiple-bonded main-group compounds27–30 and frustrated Lewis pairs (FLPs),31 enable CO activation.32 Noteworthy is the reductive coupling of CO by a disilenide, characterized by an anionic [Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si]− center and a lithium counter ion, yielding an alkynyl silane (Scheme 1, III).33 Said silane is assumed to be formed via a ketenyl intermediate. Elsewhere, the selective formation of ketenes (C
Si]− center and a lithium counter ion, yielding an alkynyl silane (Scheme 1, III).33 Said silane is assumed to be formed via a ketenyl intermediate. Elsewhere, the selective formation of ketenes (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) from two CO molecules using bis-N-heterocyclic silylenes (NHSi) was reported (Scheme 1, IV).34 Here, the NHSi units were bridged via a xanthene or ferrocene spacer ligand to control the distance between the active Si(II) centers and to incorporate two carbonyl moieties in between.
O) from two CO molecules using bis-N-heterocyclic silylenes (NHSi) was reported (Scheme 1, IV).34 Here, the NHSi units were bridged via a xanthene or ferrocene spacer ligand to control the distance between the active Si(II) centers and to incorporate two carbonyl moieties in between.
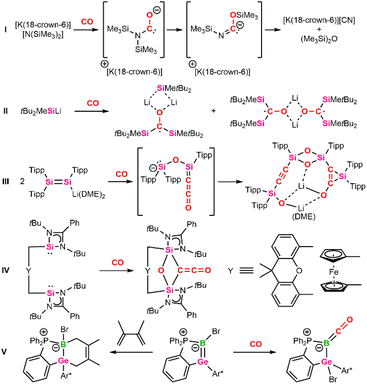 | ||
| Scheme 1 Selected examples of main-group mediated CO activation (Tipp = 2,4,6-iPr3C6H3, Ar* = 2,6-Tipp2C6H3, Dipp = 2,6-iPr3C6H3, DME = dimethoxyethane). | ||
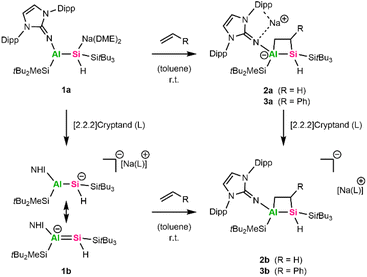
In 2023, we isolated an alumanyl silanide 1a (Scheme 2) featuring an Al–Si multiple bond, stabilized by bulky silyl groups, an NHI, and a Si–Na contact ion pair.35 The Al–Si bond in 1a is highly polarized toward silicon, as shown by NMR and bonding analyses. Correspondingly, 1a reacts as a silicon-centered nucleophile in the presence of TMSI or phenylacetylene and enables salt metathesis with NHC-stabilized coinage metal halides to form alumanyl silyl coinage metal complexes.36 When the sodium counter ion of 1a was sequestered, the multiple bond character was enhanced, emphasizing its aluminata-silene resonance structure 1b marked by an anionic [Al![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si]− core (Scheme 2).35 Apart from a proposed transient intermediate with multiple-bonding between aluminum and silicon,37 there is only one other isolated aluminata-silene reported. This species exhibits a very short Al
Si]− core (Scheme 2).35 Apart from a proposed transient intermediate with multiple-bonding between aluminum and silicon,37 there is only one other isolated aluminata-silene reported. This species exhibits a very short Al![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si bond and displays classic double bond reactivity, including the full oxidation of the Al
Si bond and displays classic double bond reactivity, including the full oxidation of the Al![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si bond via double sulfur insertion and intramolecular C–H addition across the Al
Si bond via double sulfur insertion and intramolecular C–H addition across the Al![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si bond at elevated temperatures.38 In contrast, we were not able to exploit the multiple-bond character in 1a/1b experimentally so far.
Si bond at elevated temperatures.38 In contrast, we were not able to exploit the multiple-bond character in 1a/1b experimentally so far.
To date, only a few heterodiatomic complexes formed from heavier group 13 and group 14 elements are known, and their reactivity has hardly been investigated.28,39,40 Despite the rich chemistry of boraalkenes41 and borata-alkenes,42 which feature neutral or anionic B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C/[B
C/[B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C]− double bonds, the list of examples is limited to borasilenes (B
C]− double bonds, the list of examples is limited to borasilenes (B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si)43,44 and borata-silenes ([B
Si)43,44 and borata-silenes ([B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si]−),45 neutral four-membered aromatic B2Si2 heterocycles,46 boragermenes (B
Si]−),45 neutral four-membered aromatic B2Si2 heterocycles,46 boragermenes (B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge),43,47,48 a zwitterionic stannaborene (B
Ge),43,47,48 a zwitterionic stannaborene (B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Sn),49 heavier allenic anions of the type [>E14
Sn),49 heavier allenic anions of the type [>E14![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) E13
E13![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) E14<]− (E13 = Ga, In; E14 = Si, Ge)50 and an aluminate complex characterized by an anionic [Al
E14<]− (E13 = Ga, In; E14 = Si, Ge)50 and an aluminate complex characterized by an anionic [Al![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C]− core.51 Very recently, our group published the first alumene, which is characterized by a neutral [Al
C]− core.51 Very recently, our group published the first alumene, which is characterized by a neutral [Al![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C] motif.19 The difficulty for the formation of multiple bonding, particularly between heavier group 13 and 14 elements, arises due to polarization of the E13–E14 (E = p-block element) bond and the intrinsic Lewis acidity of the triels in addition to weaker π-bonding due to poor orbital overlap when going down the group.39,52
C] motif.19 The difficulty for the formation of multiple bonding, particularly between heavier group 13 and 14 elements, arises due to polarization of the E13–E14 (E = p-block element) bond and the intrinsic Lewis acidity of the triels in addition to weaker π-bonding due to poor orbital overlap when going down the group.39,52
It should be noted that the only in-depth reactivity study has been reported for a zwitterionic boragermene.48 Interestingly, said boragermene shows a versatile chemistry, including the addition of carbon-based Lewis acids (NHC, AdNC, CO) at the nucleophilic boron center to form B–C bonds (Scheme 1, V). In addition, the B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ge double bond was accessed by [2+2]- and [2+4]-cycloaddition reactions with CO2 and dimethylbutadiene or partial bond oxidation with selenium to give the respective three-, four-, and six-membered heterocycles.48
Ge double bond was accessed by [2+2]- and [2+4]-cycloaddition reactions with CO2 and dimethylbutadiene or partial bond oxidation with selenium to give the respective three-, four-, and six-membered heterocycles.48
Inspired by the reports on silicon-centered CO activation, we were interested in the capacity of 1a to activate CO based on its predominant silanide character.15,27,33,34 To further complement our earlier reactivity studies and highlight the reactivity dualism between π-bonding-type and zwitterionic reactivity (nucleophilic Si and electrophilic Al center), the partial multiple bond character of the central Al–Si bond is investigated. Here we report on the ability of 1a/1b to undergo [2+2]-cycloaddition reactions in the presence of olefins and the facile activation of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O/C
O/C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) O bonds in mesitylaldehyde, CO2, and CO by 1a.
O bonds in mesitylaldehyde, CO2, and CO by 1a.
Results and discussion
[2+2]-Cycloaddition with alkenes and alkynes
A typical reactivity observed not only for alkenes but also for their heavier analogues is the cycloaddition reaction across unsaturated carbon–carbon bonds. As previously discussed, [2+2]- and [2+4]-cycloaddition reactions have been well-documented for highly polarized heteroatomic double bonds.48 To further investigate the multiple-bonding character of alumanyl silanide 1a and aluminata-silene 1b, we were interested in their behavior towards unsaturated substrates. It should be noted that 1a possesses a sterically highly crowded aluminum center, limiting the choice of suitable substrates. As such, no reaction was observed for sterically encumbered substrates diphenylacetylene, cis-stilbene, cis- and trans-3-hexene, 1,5-cyclooctadiene, or norbornadiene, but rather a decomposition of the starting material 1a above 90 °C.We, therefore, probed the reactivity of 1a with terminal unsaturated compounds, including neohexene, styrene, ethylene, and acetylene. While neohexene did not react with 1a even at elevated temperatures up to 90 °C, 1a was completely consumed in the presence of 1 bar acetylene, yet no selective products could be identified in the 1H NMR spectrum. Exposure of a C6D6 solution of 1a to 1 bar ethylene at room temperature results in complete discoloration of the orange color of 1a. The formal [2+2]-cycloaddition product 2a (Scheme 2) was obtained as an off-white solid in 72% yield after workup.
The 1H NMR spectrum of 2a shows four broad resonances around 2.72, 1.78, 0.96, and 0.43 ppm, corresponding to the diastereotopic protons from the two CH2-groups in the newly formed Al–Si–C–C ring. Furthermore, the 1H–13C HSQC NMR spectrum matches these proton resonances to carbon shifts at 7.3 and 18.4 ppm in the 13C NMR spectra.
The resonance of the Si-bonded proton overlaps with the signal of the SitBu3 group at 1.27 ppm (C6D6) in the 1H NMR spectrum and was assigned via1H–29Si HMBC experiments. The Si–H bond stretching was observed at 1973 cm−1 in the infrared spectrum, which aligns with the Si–H band of 1a (1920 cm−1).3529Si NMR spectroscopy reveals a significantly low-field shift to −82.3 ppm for the central Si–H group in comparison to 1a (−163.0 ppm), resulting from the change in the silicon coordination. For the substituted silyl groups, silicon shifts similar to the starting material (1a: 37.3/0.8 ppm SitBu3/SitBu2Me; 2a: 17.0/−0.2 ppm SitBu3/SitBu2Me) were observed.
Single crystals of 2a were obtained by cooling a concentrated solution of a pentane/toluene mixture at −35 °C. SC-XRD clearly confirmed the connectivity of 2a (Fig. 1); however, bonding parameters are not discussed due to the limited data quality.
Thus far, [2+2]-cycloaddition reactions with ethylene are predominantly known for heavier group 13 and 14 compounds featuring homodiatomic multiple bonds, with only a few examples being reported for highly polarized heteroatomic multiple bonds.53 In the case of phosphaalumenes, complete cleavage of the Al![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) P double bond via the insertion of 2 equivalents of ethylene is reported, with the formation of an Al–P–C–C heterocyclic ring proposed as an intermediate, whereas reactions with sterically more hindering alkenes and alkynes result in the formation of typical [2+2]-cycloaddition products.54
P double bond via the insertion of 2 equivalents of ethylene is reported, with the formation of an Al–P–C–C heterocyclic ring proposed as an intermediate, whereas reactions with sterically more hindering alkenes and alkynes result in the formation of typical [2+2]-cycloaddition products.54
In terms of molecular heterocycles containing Al–Si bonds, a few examples are known: besides the aforementioned aluminata-silene and its reaction products, Al2Si2 and AlSiC3N heterocycles were obtained from silylene-stabilized aluminylene derivatives either by dimerization or the addition of 2-methylquinoline.37,38,55 Furthermore, a six-membered Al2Si6 cycle was obtained by the reaction of a bis-silylene and an aluminum hydride.56 Adding to this, alumanyl silanide 1a allows for facile access to aluminum silicon heterocycles in moderate to good yields. Such four-membered heterocycles are prone to further functionalization based on the ring strain, adding to a polarized and weak Al–C single bond.54,57 Unexpectedly, 2a remains stable up to 120 °C in solution (C6D6 in a sealed NMR tube) and does not react with another ethylene molecule before unselective decomposition occurs.
The analogous [2+2]-cycloaddition product 3a (Scheme 2) was obtained in 62% yield when 1a was reacted with styrene at room temperature overnight, as apparent from multinuclear NMR spectroscopy. A doublet of doublets (dd) at 3.03 ppm (2JSi–H = 10 Hz; 3JH–H = 5 Hz) can be assigned to the Si–H group (1JSi–H = 116 Hz) in the 1H- and 1H–29Si HMBC spectra. The CH group's proton of the newly formed Al–Si–C–C heterocycle overlaps with resonances of the Dipp group protons in the range of 1.52–1.43 ppm, but it can be assigned via a 1H–29Si-HMBC experiment. The endocyclic CH2 group, featuring two diastereotopic protons, gives rise to a broad singlet at 1.81 ppm and a doublet of doublets (dd) at 2.45 ppm (3JHA–H = 5 Hz; 3JHB–H = 15 Hz). The central Si atom in 3a resonates at −91.6 ppm in the 29Si NMR spectra and is in a similar range to the Si center in 2a (−82.3 ppm). To date, we have not been able to grow crystals of 3a suitable for SC-XRD.
For comparison and to investigate the influence of the sodium counter ion on the observed [2+2]-cycloaddition reactions, we repeated the experiments with aluminata-silene 1b, which has no contact counter cation and exhibits a more pronounced multiple bond character compared to 1a.35 Upon addition of ethylene or styrene to a solution of 1b in C6D6 at room temperature, an immediate discoloration is observed. Multinuclear NMR spectroscopy confirms the selective formation of the [2+2]-cycloaddition products 2b and 3b, respectively. The characteristic Si–H group can be observed around 3.09 ppm (1JSi–H = 116 Hz) for 2b and 3.16 ppm (1JSi–H = 123 Hz) for 3b in the 1H NMR spectrum and produces similar shifts in the 29Si spectra (−81.7 ppm/−91.2 ppm 2b/3b), respectively. Alternatively, 2b and 3b can be synthesized by sequestering the sodium counter cation in 2a and 3a by adding stoichiometric amounts of [2.2.2]-cryptand. Based on this, we can conclude that the role of the Na counter cation in the formation of the [2+2]-cycloaddition products is negligible. Attempts to reverse the discussed [2+2]-cycloadditions at elevated temperatures up to 120 °C and under vacuum were unsuccessful.
[2+2]-Cycloaddition of mesitylaldehyde and CO2
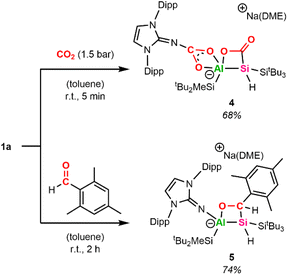
In recent years, various bimetallic systems featuring an electron-rich Lewis base adjacent to a Lewis acidic center have been utilized for CO2 activation.58 Due to the strongly polarized Al–Si bond present in alumanyl silanide 1a, we estimated that the activation of other polarized bonds, such as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds found in ketones, aldehydes, and CO2, would be feasible. Discoloration is observed when pressurizing a J-Young tube of 1a in C6D6 with 1 bar CO2 at room temperature, with a white precipitate forming within 30 min. The precipitate can be redissolved by adding small amounts of acetonitrile. Multinuclear NMR spectroscopy analysis of the resulting C6D6/MeCN-d3 mixture confirms the uptake of two CO2 molecules by 1a.
O double bonds found in ketones, aldehydes, and CO2, would be feasible. Discoloration is observed when pressurizing a J-Young tube of 1a in C6D6 with 1 bar CO2 at room temperature, with a white precipitate forming within 30 min. The precipitate can be redissolved by adding small amounts of acetonitrile. Multinuclear NMR spectroscopy analysis of the resulting C6D6/MeCN-d3 mixture confirms the uptake of two CO2 molecules by 1a.
One molecule is inserted between the aluminum center and the NHI, while a second molecule adds across the central Al–Si bond, yielding the [2+2]-cycloaddition product 4 (Scheme 3). The proton bonded to the central Si gives a single resonance at 4.22 ppm in the 1H NMR spectrum, which lies within the typical region for supersilyl-substituted tetra-coordinated silanes.591H–13C HMBC spectroscopy reveals a cross-peak of said proton with the carbon in the Al–Si–C–O heterocycle at δC = 188.4 ppm. Similar shifts in 13C NMR spectra have been observed for analogous cycloaddition products of disilene (185.9 ppm)60 and germaborene (184.2 ppm).48 In contrast to the germaborene, which is polarized towards the boron center, the carbon of CO2 adds to the more electron-rich silicon center in 1a. As a result, the Si center in 4 resonates significantly more downfield in the 29Si NMR spectrum at −75.8 ppm compared to the starting material (−163.0 ppm); however, it is upfield shifted in comparison to the disilene congener (11.8 ppm).60 This can be attributed to the presence of a proton bonded to the central silicon in 4. A resonance of weak intensity at 162.2 ppm in the 13C NMR spectrum of 4 was assigned to the CO2 moiety inserted in the N–Al bond. Similar characteristic signals in the 13C NMR spectrum around 166.0–168.9 ppm have been observed after the addition of CO2 to coinage metal derivatives of 1a.36 To verify our assignment, we repeated the experiment with 13C-labeled CO2, yielding the isotopomer 413C (for more details, see SI). Both carbon signals (188.4 and 162.2 ppm) are strongly intensified in the 13C NMR spectra of 413C compared to 4. Moreover, the resonance corresponding to the central silicon atom appears as a triplet at −75.8 ppm (1JC–Si = 70.5 Hz) in the 29Si NMR spectra of 413C due to coupling of the Si center with the neighboring 13C carbon in the formed Al–Si–C–O ring.61
The straightforward capturing of CO2 by our system prompted us to investigate the reactivity of 1a towards ketones and aldehydes as well. [2+2]-Cycloaddition reactions of disilenes in this manner are well documented.27,60,62 A similar reactivity has recently been described for a phosphagallene, which among others is able to add CO2 and benzaldehyde, forming a P–Ga–O–C cycle in the process.63 After unsuccessful attempts with benzaldehyde, we chose mesitylaldehyde as a more electron-rich aldehyde derivative to react with 1a. Compound 5 (Scheme 3), characterized by an Al–Si–C–O heterocyclic ring, was isolated as a colorless powder in 74% yield after workup. The SiH group resonates at δH = 4.44 ppm (1JSi–H = 127 Hz), which is in a similar low-field area to 4 and gives a doublet signal (3JH–H = 8 Hz) due to proton–proton coupling with the proton of the neighboring CH group. The mesityl-bound CH resonates at δH = 5.65 ppm and matches the 13C carbon signal at δC = 65.8 ppm determined via1H–13C HSQC analysis. The central silicon resonates at −59.9 ppm in the 29Si NMR spectrum, which is in accordance with the central Si shifts of analogous cycloaddition products obtained from disilene and benzaldehyde (δSi = −54.6/−52.5 ppm).60
Single crystals suitable for SC-XRD analysis were obtained by slow evaporation from a concentrated C6D6 solution at r.t. and confirm the structure of the [2+2]-cycloaddition product 5 (Fig. 1). Compound 5 is characterized by a non-planar Al–Si–C–O heterocycle (∑ = 353.61°). The distance between the tetrahedral Si and Al center in 5 amounts to 2.5502(5) Å and is significantly elongated with regard to alumanyl silanides in general (2.4179(10) Å, 2.4113(6) Å, 2.3878(10) Å).35 The sodium counter ion in 5 is encapsulated by the NHI (contact with the exocyclic nitrogen and one aromatic carbon of the Dipp substituent), one DME molecule, and the oxygen of the formed 4-membered heterocycle. The distance between the sodium cation and the anionic aluminum center amounts to 3.1101(6) Å.
CO activation
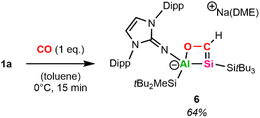
Lastly, we were interested in the ability of alumanyl silanides to activate the stable triple bond in CO.Pressurizing a toluene-d8 solution of 1a with 1 bar CO at 0 °C results in a color change from orange to green. 1H NMR spectroscopy reveals the formation of a room-temperature unstable species that decomposes completely within minutes but remains stable for several weeks at −35 °C in toluene. Performing the reaction not with excess, but with 1 equivalent CO at 0 °C in toluene (for more details, see SI) enabled the isolation of the cyclic silacarbene 6 (Scheme 4) as orange powder in 64% yield after workup.
In the 1H NMR spectrum of 6, the vinylic proton is observed at 8.73 ppm (1JC–H = 155 Hz), which is significantly low-field shifted in comparison to previously reported silyl-substituted silacarbenes (5.53–7.43 ppm, 1JC–H = 133–153 Hz).64 This strong deshielding might be caused by the endocyclic oxygen and aluminum in addition to the electropositive silyl-substituents at the four-membered Al–Si–C–O ring. 13C and 29Si NMR spectra reveal that the Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond resonates at 199.9 ppm (13C) and 43.5 ppm (29Si). Repeating the experiment employing 13C-labeled CO (for more details, see SI) results in the central silicon signal appearing as a doublet (1JC–Si = 33 Hz) in 29Si NMR. To the best of our knowledge, there are no complexes featuring Al–Si–C–O cycles reported in the literature. However, cyclic silacarbenes made from disilenide and carboxylic acid chlorides, which possess a four-membered Si–Si–C–O core formally isovalent to 6, have been isolated.65 For said complexes, low-field shifted carbon (213.4–231.6 ppm) and high-field shifted silicon (17.5–34.4 ppm) signals have been observed, pointing to a less polarized Si
C bond resonates at 199.9 ppm (13C) and 43.5 ppm (29Si). Repeating the experiment employing 13C-labeled CO (for more details, see SI) results in the central silicon signal appearing as a doublet (1JC–Si = 33 Hz) in 29Si NMR. To the best of our knowledge, there are no complexes featuring Al–Si–C–O cycles reported in the literature. However, cyclic silacarbenes made from disilenide and carboxylic acid chlorides, which possess a four-membered Si–Si–C–O core formally isovalent to 6, have been isolated.65 For said complexes, low-field shifted carbon (213.4–231.6 ppm) and high-field shifted silicon (17.5–34.4 ppm) signals have been observed, pointing to a less polarized Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond in 6.
C double bond in 6.
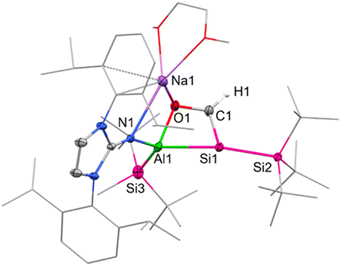
Single crystals suitable for SC-XRD were obtained from cooling a concentrated solution of 6 in hexane at −35 °C. The central Al–Si bond in 6 (Fig. 2) measures 2.5596(12) Å, which is slightly longer than the peripheral Al–Si bond (2.5358(14) Å) in 6 and in range with the central Al–Si bond in 5 (2.5502(5) Å). The endocyclic silicon carbon distance amounts to 1.781(4) Å, which is in accordance with previously reported Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond lengths (1.702(5)–1.778(3) Å).64 The C–O distance in 6 amounts to 1.394(4) Å, in line with a carbon–oxygen single bond.66 The 4-membered Al–Si–C–O heterocycle is slightly distorted (∑ = 357.39°), with the 3-coordinated silicon center occupying a pyramidalized geometry (∑ = 349.14°). Sodium is coordinated by the endocyclic oxygen, the exocyclic nitrogen of the NHI, two aromatic carbon atoms of the Dipp substituent, as well as one molecule of DME, like the coordination found in 5. Based on the structural analysis, 6 is formed via [2+2]-cycloaddition of CO across the Al–Si bond, followed by migration of the silicon-bonded proton to the newly introduced carbon of CO.
C bond lengths (1.702(5)–1.778(3) Å).64 The C–O distance in 6 amounts to 1.394(4) Å, in line with a carbon–oxygen single bond.66 The 4-membered Al–Si–C–O heterocycle is slightly distorted (∑ = 357.39°), with the 3-coordinated silicon center occupying a pyramidalized geometry (∑ = 349.14°). Sodium is coordinated by the endocyclic oxygen, the exocyclic nitrogen of the NHI, two aromatic carbon atoms of the Dipp substituent, as well as one molecule of DME, like the coordination found in 5. Based on the structural analysis, 6 is formed via [2+2]-cycloaddition of CO across the Al–Si bond, followed by migration of the silicon-bonded proton to the newly introduced carbon of CO.
Computational study
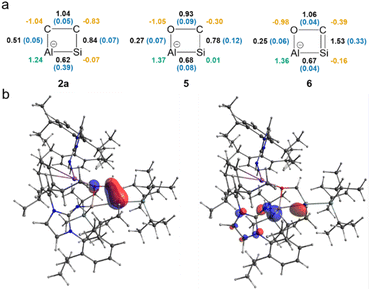
To elucidate the bonding situation in the newly formed heterocycles, both QTAIM and NBO analyses were conducted on the optimized structures of 2a, 5, and 6 (for more details, see SI). Upon reaction of 1a with ethylene, mesitylaldehyde, or CO, both the Si and Al centers are oxidized, and the central Al–Si bond order is reduced, resulting in Wiberg bond indices around 0.62–0.68 in 2a, 5, and 6 (Fig. 3). Interestingly, 2a displays a remarkably high ellipticity in the Al–Si Bond Critical Point (BCP), implying a stronger partial π-character found for the Al–Si core in 2a compared to 5 and 6.67 Furthermore, the BCPs indicate that the interactions between the Al–Si unit and the substrates are non-covalent in all three cases, indicated by low electron densities and a positive Laplacian, typical features of closed-shell interactions.68 The calculations confirm an endocyclic Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond found in 6 with a WBI of 1.529 and high ellipticity at the BCP (ε = 0.3269 a.u.). Following the natural difference in electronegativity between carbon and silicon, NBO analysis shows a σ(Si–C) bond polarized towards carbon (Si/C: 27%/72%; 1.96 electrons occupancy). In contrast, both atoms contribute almost equally (Si/C: 48%/52%) to the purely p-type π-bond with an occupancy of 1.90 electrons, thus indicating an authentic Si
C double bond found in 6 with a WBI of 1.529 and high ellipticity at the BCP (ε = 0.3269 a.u.). Following the natural difference in electronegativity between carbon and silicon, NBO analysis shows a σ(Si–C) bond polarized towards carbon (Si/C: 27%/72%; 1.96 electrons occupancy). In contrast, both atoms contribute almost equally (Si/C: 48%/52%) to the purely p-type π-bond with an occupancy of 1.90 electrons, thus indicating an authentic Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond with significant π-character.
C double bond with significant π-character.
For the polar-covalent endocyclic C–O bond of 6, the WBI value is slightly above 1, suggesting conjugation within the four-membered ring. Moreover, based on NBO analysis, the Al–O interaction in 6 is mainly dative in nature. This is evident from the second-order perturbation analysis, where the donation from the lone pair of O to the empty p-orbital of Al is 42.7 kcal mol−1. The QTAIM-derived delocalization index for the Al–O bond is 0.22, which is significantly lower than 1, the expected value for a typical single bond. The highest occupied molecular orbital (HOMO) in 6 corresponds to the Si–C π-bond, which is clearly depicted by the frontier orbitals, whereas the HOMO-1 is delocalized across the Al–Si moiety and to the aluminum center adjacent NHI and silyl group (Fig. 3). The LUMO is delocalized over the aryl substituents at the NHI. This leads to a moderate HOMO–LUMO gap of 3.49 eV. Natural charges (derived from NBO) and QTAIM charges describe an overall polarized heterocycle found in 2a, 5, and 6 (Fig. 3) with a positive and strongly Lewis acidic aluminum center in all cases, despite its fourfold coordination.
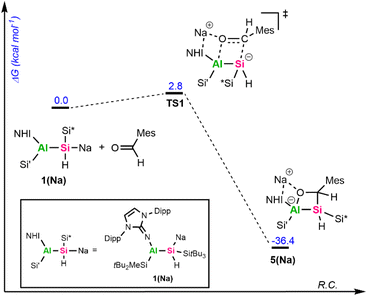
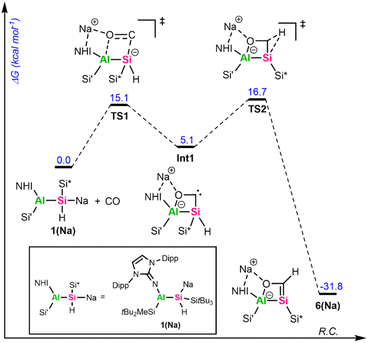
To get further insights into the mechanism of the formation of 5 and 6, we investigated these reactions computationally using alumanyl silanide 1(Na) with an intimate ion pair with Si–Na contact as model compound (Fig. 4 and 5).
The addition of mesitylaldehyde to 1(Na) does not happen stepwise but proceeds via a concerted cyclic transition state (TS) with a small activation energy of 2.8 kcal mol−1 (Fig. 4). The addition of CO to 1(Na) begins with an analogous TS1 (Fig. 5), where the Si–C and Al–O bonds are formed simultaneously. Although the reaction barrier here is higher than for the addition of MesCHO, it remains relatively low at 15.1 kcal mol−1, leading to the formation of the cyclic singlet carbene Int1. This reactive intermediate features a small HOMO–LUMO gap of 3.04 eV, with the frontier orbitals mainly localized on the lone pair (HOMO) and the empty p-orbital (LUMO) of the carbenic carbon (Fig. S72). Through another low-barrier TS (TS2), 1,2-hydrogen migration from the endocyclic silicon center to the neighboring carbon atom proceeds to yield the silacarbene 6(Na). Both cycloaddition reactions are strongly exergonic (ΔG = −36.4 kcal mol−1 for 5(Na) and −31.8 kcal mol−1 for 6(Na)). The silicon-centered activation of CO was already verified in mechanistic studies for forming sila-ketenyl anions from bis-silyl-substituted silicon anion radicals in our group.69 The formation of a transient carbene as an initial step has furthermore been calculated for CO activation by alkali metal amides9,70 and phosphides10 and was proposed for the reaction of 1,2,4,3-triazaborol-3-yl-lithium with CO.13 The group of Apeloig calculated the mechanism for the reduction of CO with Me3SiLi.15 In their study, a second Me3SiLi molecule interacts with the initially formed carbene intermediate, yielding a bis(silyl)-substituted ketyl radical. In contrast, the central silicon in 1a is sterically shielded and possesses a proton in α-position, enabling 1,2-hydrogen migration. For synthesizing cyanides and isocyanides from CO, an analogue of 1,3-silyl migration subsequent to carbene formation has been proposed.70 The aforementioned reactions typically proceed via initial coordination of the CO oxygen atom to an electropositive metal center, followed by nucleophilic attack at the CO carbon atom to generate a new carbon-based nucleophile that reacts further. Consequently, main-group-mediated CO activation generally affords formal 1,1-carbon-addition products.3 In contrast, [2+2]-cycloadditions of CO, as observed with alumanyl silanide 1a, are exceedingly rare in main-group chemistry. For zinc diazo alkyl complexes, a formal [2+3]-cycloaddition of CO has been reported.71 However, this activation of CO proceeds via initial nucleophilic attack by the diazo nitrogen at the electrophilic CO carbon, facilitated by coordination of the carbon center to zinc and subsequent isomerization. Therefore, this activation is akin to classical silanide-mediated CO insertion. In contrast, the calculations for 1(Na) indicate a truly concerted [2+2]-cycloaddition pathway, which differs from the previously reported reaction pathways and relies on the intrinsic polarity of the central Si–Al bond, with the counter cation playing only a minor role. To the best of our knowledge, a silanide-based hydrosilylation of CO has not been reported to date, despite recent progress in the field of main group-mediated CO activation.3
Attempts to trap the proposed cyclic carbene Int1 by addition of transition metal halides ([Rh(COD)Cl]2, CuCl), oxidation agents like S8 or Woollins' reagent as Se source, or H2 at low temperature (−60 °C) to a toluene solution of 1a under CO atmosphere (1 bar) have been unsuccessful. Instead, we were able to grow crystals suitable for SC-XRD by pressurizing a concentrated toluene solution of 1a with 2 bar CO at −35 °C. It is worth mentioning that these crystals are extremely temperature sensitive and decompose above −35 °C by gas evolution, as observed several times under the microscope during the picking process.
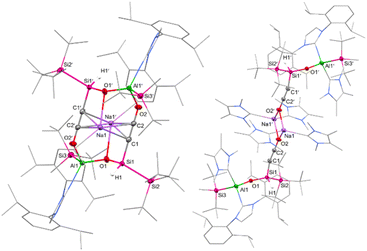
Structure analysis revealed the formation of 7 (Scheme 5) from the reaction of 1a with two equivalents of CO, involving C–O bond cleavage and C–C coupling. This results in a silanyl/silyl-substituted ethynolate, with one oxygen inserted into the central Al–Si bond. Two ethynolates then dimerize via Al–O bond formation to give the anionic product 7 with two sodium counter ions.
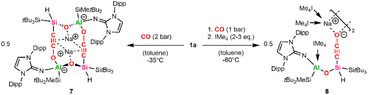 | ||
| Scheme 5 Synthesis of the silanyl ethynolate dimers 7 and 8 (IMe4 = 1,3,4,5-tetramethylimidazol-2-ylidene). | ||
Compound 7 crystallized in the space group P21/n and shows a dimeric structure, where the second half is symmetry-generated (Fig. 6, symmetry-generated atoms are marked with a prime). The dimer is characterized by a distorted 12-atom heterocycle, with the two sodium counter ions hovering below and above the non-planar ring. The silicon and the aluminum center are found in a tetrahedral coordination environment with the Al1–O1 bond (1.744(3) Å) being significantly shorter than the Al1–O2′ bond (1.867(2) Å), indicating a weaker interaction between the ethynolate (–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C–O−) moiety of one half and the corresponding aluminum center of the second half. The Si–C–C–O fragment is almost linear, with measured Si–C–C and C–C–O angles of 178.0(3)° and 179.4(4)°, respectively. Within said fragment, the C1–C2 and C2–O2 bond lengths amount to 1.219(4) Å and 1.253(4) Å, respectively. These values lie between the bond lengths reported for C–C triple bonds and C–O single bonds found in silanyl ethanolates (dC–C = 1.18 Å, dC–O = 1.31–1.34 Å),72,73 as well as C
C–O−) moiety of one half and the corresponding aluminum center of the second half. The Si–C–C–O fragment is almost linear, with measured Si–C–C and C–C–O angles of 178.0(3)° and 179.4(4)°, respectively. Within said fragment, the C1–C2 and C2–O2 bond lengths amount to 1.219(4) Å and 1.253(4) Å, respectively. These values lie between the bond lengths reported for C–C triple bonds and C–O single bonds found in silanyl ethanolates (dC–C = 1.18 Å, dC–O = 1.31–1.34 Å),72,73 as well as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and C
C and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds of bis(silyl)-substituted ketenes (dC–C = 1.26–1.28 Å, dC–O = 1.18–1.21 Å).33,34 Consequently, 7 might be best described by two resonance structures: an ethynolate structure with a C1–C2 triple bond and C2–O2 and O2–Al1 single bonds, and a ketene with a C
O double bonds of bis(silyl)-substituted ketenes (dC–C = 1.26–1.28 Å, dC–O = 1.18–1.21 Å).33,34 Consequently, 7 might be best described by two resonance structures: an ethynolate structure with a C1–C2 triple bond and C2–O2 and O2–Al1 single bonds, and a ketene with a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O fragment stabilizing the aluminum center via donor–acceptor interaction between the oxygen and aluminum. An analogous ketene intermediate, formed during the reductive cleavage of CO using disilenide, has been confirmed via trapping using transition metal hexacarbonyls (M(CO)6, M = Cr, Mo, W).33 Despite numerous attempts, we were not able to isolate a room-temperature stable derivative of 7.
O fragment stabilizing the aluminum center via donor–acceptor interaction between the oxygen and aluminum. An analogous ketene intermediate, formed during the reductive cleavage of CO using disilenide, has been confirmed via trapping using transition metal hexacarbonyls (M(CO)6, M = Cr, Mo, W).33 Despite numerous attempts, we were not able to isolate a room-temperature stable derivative of 7.
The formation of 7 presents a rare example of main-group mediated reductive homologation of CO.3,32 This is enabled by the cooperative interaction of the Lewis acidic aluminum center and the neighboring nucleophilic silicon center, stabilized by a sodium counter cation. Similar reactions have been reported for main group complexes involving B–Li,13,14 Mg–Mg,20–26 Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si33 and B
Si33 and B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) B29 bonds as active centers. Additionally, CO homologations mediated by acyclic silylenes,74 bis(silylenes),34,75 aluminum(I) compounds76 and dialanes19 are known.
B29 bonds as active centers. Additionally, CO homologations mediated by acyclic silylenes,74 bis(silylenes),34,75 aluminum(I) compounds76 and dialanes19 are known.
The CO activation through alumanyl silanide 1a is proposed to proceed in an analogous fashion to that reported for lithium disilenide.33 For both systems, one key step is the formation of a heterocyclic carbene. The carbene is stabilized by a second CO molecule acting as a Lewis base, yielding a ketene, a step that is corroborated by preliminary DFT calculations (see SI). In the case of the disilenide, calculations on a simplified model showed a rearrangement after ketene formation, resulting in a silanyl/silyl-substituted ethynolate. In our case, we assume the heterocyclic Al–Si–C–O ring to rearrange after ketene formation in a similar manner with cleavage of the highly polarized central Al–Si bond and the endocyclic C–O bond and formation of a thermodynamically favored Si–O bond. The resulting silanyl ethynolate then dimerizes through reaction of the terminal oxygen with the aluminum center of a second silanyl ethynolate to give 7.
We assumed the addition of NHC as a stronger Lewis base would better stabilize the cyclic carbene intermediate than an additional molecule of CO. Since 1a reacts with 1 equivalent of NHC via exchange of the DME donor, we added 2–3 equivalents of IMe4 (1,3,4,5-tetramethylimidazol-2-ylidene) to a cooled (−60 °C) toluene solution of 1a under CO (1 bar) atmosphere. This approach yielded the NHC-stabilized silanyl ethynolate 8 (Scheme 5). Like 7, compound 8 is not stable at ambient temperature and could not be isolated. However, the solid-state structure of 8 (Fig. 6) was unambiguously determined using SC-XRD after obtaining colorless cuboid-shaped crystals from a concentrated toluene solution at −35 °C under CO atmosphere. Instead of the anticipated NHC-stabilized carbene, the structure of 8 presents a silanyl ethynolate dimer analogous to 7, with the second half of the molecule being symmetry-generated. The two fragments in 8 are bridged by the two sodium counter ions, which each coordinate to the terminal oxygen of the ethynolate moiety and the carbenic carbon of two IMe4 molecules, respectively. Thus, the addition of IMe4 inhibits the dimerization via Al–O bond formation as observed for 7. Another equivalent of IMe4 coordinates to the aluminum center, resulting in a tetrahedral geometry. The Al–C distance (2.090(2) Å) lies in the upper range of reported Al–CIMe4 bond lengths (1.99–2.09 Å).77 Notably, the distance between the exocyclic nitrogen of the NHI and the Al center measures 1.819(7) Å and is significantly elongated in comparison to the Al1–N1 bond length found in 7 (1.751(2) Å) or reported alumanyl silanides (1.77–1.78 Å).35 This marks a strong σ-interaction between the NHC and the aluminum center and a weakened bonding towards the NHI. The C1–C2 and C2–O2 bond distances in 8 measure 1.219(3) Å and 1.247(2) Å and match the respective bond lengths in 7. Dissentingly, the Si–C–C–O fragment in 8 is less linear with Si–C–C and C–C–O bond angles of 167.02(17)° and 178.9(2)°, respectively, owing to steric repulsion exerted by the coordinating NHC ligands. Another difference is a considerably shorter Si1–C1 bond found in 8 with respect to 7 (7: dSi1–C1 = 1.864(3) Å, 8: dSi1–C1 = 1.7891(19) Å) or literature known silyl ethynolates (1.83–1.84 Å).72,73 Most recently, a silyl ethynolate dimer bridged by two Eu(II) centers was reported, exhibiting almost linear geometry (Si–C–C/C–C–O angles = 176.4(5)°/179.4(7)°) and comparable bond lengths (dC–O = 1.25 Å, dC–C = 1.20 Å, dSi–C = 1.79 Å) to those in 8.78 NRT analysis for this silyl ethynolate indicates a major ethynolate resonance contribution (74.1%) alongside a substantial ketenyl component (22.9%) despite its linear geometry and four-coordinate silicon center. Consistent with these findings, a predominant ethynolate character for 7 and 8, with minor contributions from a possible ketene/ketenyl resonance structure, is assumed.
Conclusions
In summary, we report the isolation and characterization of previously unknown Al–Si–C–C (2a, 2b, 3a, 3b) and Al–Si–C–O heterocycles (4, 5). These were obtained by the formal [2+2]-cycloaddition of ethylene, styrene, mesitylaldehyde, and CO2 to the polarized Al–Si bond found in alumanyl silanide/aluminata-silene 1a/1b. In the presence of 1 equivalent CO, an Al–Si–C–O heterocycle containing a Si–C double bond is formed via CO addition to the Al–Si core of 1a, followed by proton migration. The formal hydrosilylation of the stable C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) O bond in carbon monoxide once more attests the potential of heavier heteroatomic low-valent main group compounds for oxidative addition and further functionalization of small molecules, both critical steps in catalytic cycles. The formation of 6 is proposed to proceed via a heterocyclic carbene intermediate, which could be trapped in the presence of additional CO and NHC at low temperature, yielding the silanyl ethynolates 7 and 8. Thus, the reductive coupling of CO was observed for the first time for a low-valent compound with an Al–Si bond as the active site. All things considered, this in-depth reactivity study of alumanyl silanides has experimentally confirmed the multiple bond character found for the anionic Al–Si core of the title compound. Reactions that have been usually observed for heavier homodiatomic main-group compounds like disilenes have now been attested with heterodiatomic congeners, which formally possess the [Al–Si−] core isovalent to [Si
O bond in carbon monoxide once more attests the potential of heavier heteroatomic low-valent main group compounds for oxidative addition and further functionalization of small molecules, both critical steps in catalytic cycles. The formation of 6 is proposed to proceed via a heterocyclic carbene intermediate, which could be trapped in the presence of additional CO and NHC at low temperature, yielding the silanyl ethynolates 7 and 8. Thus, the reductive coupling of CO was observed for the first time for a low-valent compound with an Al–Si bond as the active site. All things considered, this in-depth reactivity study of alumanyl silanides has experimentally confirmed the multiple bond character found for the anionic Al–Si core of the title compound. Reactions that have been usually observed for heavier homodiatomic main-group compounds like disilenes have now been attested with heterodiatomic congeners, which formally possess the [Al–Si−] core isovalent to [Si![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Si]. We hope that these results will fuel the search for heavier heterodiatomic low-valent main group species, for which examples are exceedingly rare, and many combinations across the periodic table are still missing.
Si]. We hope that these results will fuel the search for heavier heterodiatomic low-valent main group species, for which examples are exceedingly rare, and many combinations across the periodic table are still missing.
Author contributions
M. L. and J. V. conceived and performed the synthetic experiments and analysed the data. P. V. designed and performed the theoretical analyses. S. S. measured, solved, and refined the XRD data. S. I. conceived and supervised the project. M. L., J. V. and S. I. wrote the manuscript with input and critical revision from all authors.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2499949 (2a), 2499950 (5), 2499951 (6), 2499952 (7), 2499953 (8) and 2499954 (6′) contain the supplementary crystallographic data for this paper.79a–fAll data supporting this article have been included in the main text or the supplementary information (SI). Supplementary information: full experimental procedures, NMR spectra, SC-XRD structures, FTIR spectra, computational details, and optimized xyz-coordinates. See DOI: https://doi.org/10.1039/d5sc09910b.
Acknowledgements
The authors thank F. Wiesmann for assistance with initial reactivity studies. P. V. would like to thank The Research Council of Finland for funding (project no. 359912 and 338271) and the CSC – IT Center for Science, Finland, for computational resources. The authors are grateful to the European Research Council (ALLOWE101001591) for financial support.Notes and references
- J.-B. Peng, H.-Q. Geng and X.-F. Wu, Chem, 2019, 5, 526–552 CAS.
- (a) A. Y. Khodakov, W. Chu and P. Fongarland, Chem. Rev., 2007, 107, 1692–1744 CrossRef CAS PubMed; (b) A. W. Budiman, J. S. Nam, J. H. Park, R. I. Mukti, T. S. Chang, J. W. Bae and M. J. Choi, Catal. Surv. Asia, 2016, 20, 173–193 CrossRef CAS.
- S. Fujimori and S. Inoue, J. Am. Chem. Soc., 2022, 144, 2034–2050 CrossRef CAS PubMed.
- (a) E. Weiss and W. Büchner, Helv. Chim. Acta, 1963, 46, 1121–1127 CrossRef CAS; (b) W. Büchner and E. Weiss, Helv. Chim. Acta, 1964, 47, 1415–1423 CrossRef; (c) J. Liebig, Ann. Phys., 1834, 109, 90–97 Search PubMed; (d) U. Rosenthal, Chem.–Eur. J., 2020, 26, 14507–14511 CrossRef CAS PubMed.
- (a) P. W. Lednor and P. C. Versloot, J. Chem. Soc., Chem. Commun., 1983, 284 RSC; (b) W. Büchner, Helv. Chim. Acta, 1963, 46, 2111–2120 CrossRef.
- T. Wang, M. Xu, A. R. Jupp, Z.-W. Qu, S. Grimme and D. W. Stephan, Chem.–Asian J., 2021, 16, 3640–3644 CrossRef CAS PubMed.
- M. Xu, T. Wang, Z.-W. Qu, S. Grimme and D. W. Stephan, Angew. Chem., 2021, 133, 25485–25489 CrossRef.
- U. Wannagat and H. Seyffert, Angew Chem. Int. Ed. Engl., 1965, 4, 438–439 CrossRef.
- M. Xu, Z.-W. Qu, S. Grimme and D. W. Stephan, J. Am. Chem. Soc., 2021, 143, 634–638 CrossRef CAS PubMed.
- M. Xu, A. R. Jupp and D. W. Stephan, Angew. Chem., 2019, 131, 3586–3590 CrossRef.
- (a) Z.-J. Quan and X.-C. Wang, Org. Chem. Front., 2014, 1, 1128–1131 RSC; (b) A. R. Jupp and J. M. Goicoechea, Angew. Chem., Int. Ed., 2013, 52, 10064–10067 CrossRef CAS PubMed; (c) F. F. Puschmann, D. Stein, D. Heift, C. Hendriksen, Z. A. Gal, H.-F. Grützmacher and H. Grützmacher, Angew. Chem., Int. Ed., 2011, 50, 8420–8423 CrossRef CAS PubMed.
- M. Xu, B. Kooij, T. Wang, J. H. Lin, Z.-W. Qu, S. Grimme and D. W. Stephan, Angew. Chem., 2021, 133, 17102–17106 CrossRef.
- W. Lu, H. Hu, Y. Li, R. Ganguly and R. Kinjo, J. Am. Chem. Soc., 2016, 138, 6650–6661 CrossRef CAS PubMed.
- B. Wang, G. Luo, M. Nishiura, Y. Luo and Z. Hou, J. Am. Chem. Soc., 2017, 139, 16967–16973 CrossRef CAS.
- Y. Kratish, D. Pinchuk, A. Kaushansky, V. Molev, B. Tumanskii, D. Bravo-Zhivotovskii and Y. Apeloig, Angew. Chem., Int. Ed., 2019, 58, 18849–18853 CrossRef CAS.
- P. Jutzi and F.-W. Schröder, J. Organomet. Chem., 1970, 24, C43–C44 CrossRef CAS.
- J. M. Gil-Negrete and E. Hevia, Chem. Sci., 2020, 12, 1982–1992 Search PubMed.
- (a) T. Fukuyama, T. Totoki and I. Ryu, Org. Lett., 2014, 16, 5632–5635 Search PubMed; (b) Z. Rappoport and I. Marek, The Chemistry of Organolithium Compounds, John Wiley & Sons, 2004 Search PubMed; (c) G. E. G. Liñares and N. S. Nudelman, J. Phys. Org. Chem., 2003, 16, 569–576 Search PubMed; (d) Q. Song, J. Chen, X. Jin and Z. Xi, J. Am. Chem. Soc., 2001, 123, 10419–10420 Search PubMed; (e) C. Narayana and M. Periasamy, Synthesis, 1985, 1985, 253–268 CrossRef.
- J. A. Kelly, A. Kostenko and S. Inoue, Nat. Synth., 2025, 4, 1577–1586 Search PubMed.
- H.-Y. Liu, R. J. Schwamm, S. E. Neale, M. S. Hill, C. L. McMullin and M. F. Mahon, J. Am. Chem. Soc., 2021, 143, 17851–17856 Search PubMed.
- A. Paparo, K. Yuvaraj, A. J. R. Matthews, I. Douair, L. Maron and C. Jones, Angew. Chem., Int. Ed., 2021, 60, 630–634 Search PubMed.
- K. Yuvaraj, I. Douair, D. D. L. Jones, L. Maron and C. Jones, Chem. Sci., 2020, 11, 3516–3522 Search PubMed.
- K. Yuvaraj, I. Douair, A. Paparo, L. Maron and C. Jones, J. Am. Chem. Soc., 2019, 141, 8764–8768 CrossRef CAS.
- K. Yuvaraj, J. C. Mullins, T. Rajeshkumar, I. Douair, L. Maron and C. Jones, Chem. Sci., 2023, 14, 5188–5195 Search PubMed.
- R. Mondal, M. J. Evans, D. T. Nguyen, T. Rajeshkumar, L. Maron and C. Jones, Chem. Commun., 2024, 60, 1016–1019 Search PubMed.
- A. O'Reilly, M. D. Haynes, Z. R. Turner, C. L. McMullin, S. Harder, D. O'Hare, J. R. Fulton and M. P. Coles, Chem. Commun., 2024, 60, 7204–7207 RSC.
- F. J. Kiefer, A. Kostenko, R. Holzner and S. Inoue, J. Am. Chem. Soc., 2025, 147, 26663–26673 CrossRef CAS PubMed.
- C. Weetman, Chem.–Eur. J., 2021, 27, 1941–1954 CrossRef CAS PubMed.
- H. Braunschweig, T. Dellermann, R. D. Dewhurst, W. C. Ewing, K. Hammond, J. O. C. Jimenez-Halla, T. Kramer, I. Krummenacher, J. Mies, A. K. Phukan and A. Vargas, Nat. Chem., 2013, 5, 1025–1028 CrossRef CAS PubMed.
- (a) J. Böhnke, H. Braunschweig, T. Dellermann, W. C. Ewing, K. Hammond, J. O. C. Jimenez-Halla, T. Kramer and J. Mies, Angew. Chem., Int. Ed., 2015, 54, 13801–13805 CrossRef; (b) M. Arrowsmith, J. Böhnke, H. Braunschweig and M. A. Celik, Angew. Chem., Int. Ed., 2017, 56, 14287–14292 CrossRef CAS PubMed.
- (a) D. W. Stephan, Science, 2016, 354, 6317 CrossRef PubMed; (b) R. Dobrovetsky and D. W. Stephan, J. Am. Chem. Soc., 2013, 135, 4974–4977 CrossRef CAS PubMed; (c) M. Devillard, B. de Bruin, M. A. Siegler and J. I. van der Vlugt, Chem.–Eur. J., 2017, 23, 13628–13632 CrossRef CAS PubMed; (d) D. W. Stephan, Chem. Soc. Rev., 2023, 52, 4632–4643 RSC.
- R. Y. Kong and M. R. Crimmin, Dalton Trans., 2020, 49, 16587–16597 RSC.
- M. Majumdar, I. Omlor, C. B. Yildiz, A. Azizoglu, V. Huch and D. Scheschkewitz, Angew. Chem., Int. Ed., 2015, 54, 8746–8750 CrossRef CAS PubMed.
- Y. Wang, A. Kostenko, T. J. Hadlington, M.-P. Luecke, S. Yao and M. Driess, J. Am. Chem. Soc., 2019, 141, 626–634 CrossRef CAS PubMed.
- M. Ludwig, D. Franz, A. Espinosa Ferao, M. Bolte, F. Hanusch and S. Inoue, Nat. Chem., 2023, 15, 1452–1460 CrossRef CAS PubMed.
- J. Voigtland, M. Ludwig, S. Stigler and S. Inoue, 00, ChemistryEurope, 2025, e202500216 Search PubMed.
- A. Saddington, S. Dong, S. Yao, J. Zhu and M. Driess, Angew. Chem., Int. Ed., 2024, 63, e202410790 CAS.
- N. Parvin, Ankur, B. Morgenstern and D. Scheschkewitz, Angew. Chem., Int. Ed., 2025, 64, e202422007 CrossRef CAS.
- P. P. Power, Organometallics, 2020, 39, 4127–4138 CrossRef CAS.
- A. Agarwal and S. K. Bose, Chem.–Asian J., 2020, 15, 3784–3806 CrossRef CAS PubMed.
- (a) R. Boese, P. Paetzold, A. Tapper and R. Ziembinski, Chem. Ber., 1989, 122, 1057–1060 CrossRef CAS; (b) A. Tapper, T. Schmitz and P. Paetzold, Chem. Ber., 1989, 122, 595–601 CrossRef CAS; (c) B. Glaser, E. Hanecker, H. Nöth and H. Wagner, Chem. Ber., 1987, 120, 659–667 CrossRef CAS.
- (a) A. Pelter, B. Singaram, L. Warren and J. W. Wilson, Tetrahedron, 1993, 49, 2965–2978 CrossRef CAS; (b) R. J. Maza, J. J. Carbó and E. Fernández, Adv. Synth. Catal., 2021, 363, 2274–2289 CrossRef CAS; (c) E. Fernández, Chem. Rec., 2024, 24, e202300349 CrossRef PubMed.
- N. Nakata, R. Izumi, V. Y. Lee, M. Ichinohe and A. Sekiguchi, Chem. Lett., 2005, 34, 582–583 CrossRef CAS.
- (a) N. Nakata and A. Sekiguchi, J. Am. Chem. Soc., 2006, 128, 422–423 CrossRef CAS PubMed; (b) N. Nakata and A. Sekiguchi, Chem. Lett., 2007, 36, 662–663 CrossRef CAS.
- (a) Y. Suzuki, S. Ishida, S. Sato, H. Isobe and T. Iwamoto, Angew. Chem., Int. Ed., 2017, 56, 4593–4597 CrossRef CAS; (b) D. Franz, T. Szilvási, A. Pöthig and S. Inoue, Chem.–Eur. J., 2019, 25, 11036–11041 CrossRef CAS.
- (a) S. K. Kushvaha, P. Kallenbach, S. S. Rohman, M. K. Pandey, Z. Hendi, F. Rüttger, R. Herbst-Irmer, D. Stalke, P. Parameswaran and H. W. Roesky, J. Am. Chem. Soc., 2023, 145, 25523–25527 CrossRef CAS PubMed; (b) J. Fan, L. Yue, C. Liu, B. Rao, G. Zhou, A. Li and B. Su, J. Am. Chem. Soc., 2024, 146, 39–44 CrossRef CAS PubMed.
- (a) P. Tholen, Z. Dong, M. Schmidtmann, L. Albers and T. Müller, Angew. Chem., Int. Ed., 2018, 57, 13319–13324 CrossRef CAS PubMed; (b) D. Raiser, C. P. Sindlinger, H. Schubert and L. Wesemann, Angew. Chem., 2020, 132, 3175–3180 CrossRef.
- C. Reik, L. W. Jenner, H. Schubert, K. Eichele and L. Wesemann, Chem. Sci., 2024, 15, 11358–11366 RSC.
- M. Zweigart, K. Eichele, H. Schubert, C. P. Sindlinger and L. Wesemann, J. Am. Chem. Soc., 2023, 145, 12452–12458 CrossRef CAS PubMed.
- (a) N. Nakata, R. Izumi, V. Y. Lee, M. Ichinohe and A. Sekiguchi, J. Am. Chem. Soc., 2004, 126, 5058–5059 CrossRef CAS PubMed; (b) N. Nakata, R. Izumi, V. Y. Lee, M. Ichinohe and A. Sekiguchi, Chem. Lett., 2008, 37, 1146–1147 Search PubMed.
- C. Yan and R. Kinjo, Angew. Chem., Int. Ed., 2022, 61, e202211800 CrossRef CAS PubMed.
- (a) P. v. R. Schleyer and D. Kost, J. Am. Chem. Soc., 1988, 110, 2105–2109 CrossRef CAS; (b) P. Bag, C. Weetman and S. Inoue, Angew. Chem., Int. Ed., 2018, 57, 14394–14413 CrossRef CAS PubMed; (c) H. Braunschweig and R. D. Dewhurst, Organometallics, 2014, 33, 6271–6277 CrossRef CAS; (d) V. Y. Lee, A. Sekiguchi, J. Escudié and H. Ranaivonjatovo, Chem. Lett., 2010, 39, 312–318 CrossRef CAS; (e) R. Borthakur and V. Chandrasekhar, Coord. Chem. Rev., 2021, 429, 213647 Search PubMed.
- (a) C. Weetman, A. Porzelt, P. Bag, F. Hanusch and S. Inoue, Chem. Sci., 2020, 11, 4817–4827 Search PubMed; (b) P. Bag, A. Porzelt, P. J. Altmann and S. Inoue, J. Am. Chem. Soc., 2017, 139, 14384–14387 CrossRef CAS PubMed; (c) Y. Peng, B. D. Ellis, X. Wang, J. C. Fettinger and P. P. Power, Science, 2009, 325, 1668–1670 CrossRef CAS PubMed; (d) T. Sasamori, T. Sugahara, T. Agou, K. Sugamata, J.-D. Guo, S. Nagase and N. Tokitoh, Chem. Sci., 2015, 6, 5526–5530 RSC; (e) D. Wendel, D. Reiter, A. Porzelt, P. J. Altmann, S. Inoue and B. Rieger, J. Am. Chem. Soc., 2017, 139, 17193–17198 Search PubMed.
- S. Nees, T. Wellnitz, F. Dankert, M. Härterich, S. Dotzauer, M. Feldt, H. Braunschweig and C. Hering-Junghans, Angew. Chem., Int. Ed., 2023, 62, e202215838 CrossRef CAS PubMed.
- X. Chen, D. Yang, F. Cao and Z. Mo, J. Am. Chem. Soc., 2024, 146, 29278–29284 CrossRef CAS PubMed.
- J. Li, M. Zhong, H. Keil, H. Zhu, R. Herbst-Irmer, D. Stalke, S. De, D. Koley and H. W. Roesky, Chem. Commun., 2019, 55, 2360–2363 RSC.
- (a) T. Kawashima, Bull. Chem. Soc. Jpn., 2003, 76, 471–483 Search PubMed; (b) M. J. Evans, S. E. Neale, M. D. Anker, C. L. McMullin and M. P. Coles, Angew. Chem., Int. Ed., 2022, 61, e202117396 CrossRef CAS PubMed.
- M. Pérez-Jiménez, H. Corona, F. de La Cruz-Martínez and J. Campos, Chem.–Eur. J., 2023, 29, e202301428 Search PubMed.
- N. Wiberg and W. Niedermayer, J. Organomet. Chem., 2001, 628, 57–64 CrossRef CAS.
- N. Wiberg, W. Niedermayer, K. Polborn and P. Mayer, Chem.–Eur. J., 2002, 8, 2730 CrossRef CAS PubMed.
- A. G. Brook, F. Abdesaken, G. Gutekunst and N. Plavac, Organometallics, 1982, 1, 994–998 CrossRef CAS.
- (a) N. Wiberg, J. Organomet. Chem., 2001, 619, 110–131 CrossRef CAS; (b) V. Y. Lee, M. Ichinohe and A. Sekiguchi, Chem. Lett., 2001, 30, 728–729 CrossRef; (c) A. Kostenko and M. Driess, J. Am. Chem. Soc., 2018, 140, 16962–16966 CrossRef CAS PubMed; (d) A. Schäfer, M. Weidenbruch and S. Pohl, J. Organomet. Chem., 1985, 282, 305–313 CrossRef.
- L. S. Szych, J. Bresien, L. Fischer, M. J. Ernst and J. M. Goicoechea, Chem. Sci., 2025, 16, 7397–7410 RSC.
- S. Honda, Y. Kato, S. Ishida and T. Iwamoto, Chem. Lett., 2021, 50, 866–869 Search PubMed.
- I. Bejan, D. Güclü, S. Inoue, M. Ichinohe, A. Sekiguchi and D. Scheschkewitz, Angew. Chem., Int. Ed. Engl., 2007, 46, 3349–3352 Search PubMed.
- F. H. Allen and A. J. Kirby, J. Am. Chem. Soc., 1984, 106, 6197–6200 Search PubMed.
- (a) C. S. López, O. Nieto Faza, F. P. Cossío, D. M. York and A. R. de Lera, Chem.–Eur. J., 2005, 11, 1734–1738 Search PubMed; (b) J. A. Cabeza, J. F. van der Maelen and S. García-Granda, Organometallics, 2009, 28, 3666–3672 CrossRef CAS.
- F. Cortés-Guzmán and R. Bader, Coord. Chem. Rev., 2005, 249, 633–662 Search PubMed.
- S. Fujimori, A. Kostenko, R. Scopelliti and S. Inoue, Nat. Synth., 2023, 2, 688–694 Search PubMed.
- M. Xu, B. Kooij, T. Wang, J. H. Lin, Z.-W. Qu, S. Grimme and D. W. Stephan, Angew. Chem., Int. Ed., 2021, 60, 16965–16969 CrossRef CAS PubMed.
- S. Jiang, Y. Cai, T. Rajeshkumar, I. Del Rosal, L. Maron and X. Xu, Angew. Chem., Int. Ed., 2023, 62, e202307244 CrossRef CAS PubMed.
- S. Companys, P. A. Peixoto, C. Bosset, S. Chassaing, K. Miqueu, J.-M. Sotiropoulos, L. Pouységu and S. Quideau, Chem.–Eur. J., 2017, 23, 13309–13313 Search PubMed.
- Y. Minami, T. Anami and T. Hiyama, Chem. Lett., 2014, 43, 1791–1793 CrossRef.
- A. V. Protchenko, P. Vasko, D. C. H. Do, J. Hicks, M. Á. Fuentes, C. Jones and S. Aldridge, Angew. Chem., Int. Ed., 2019, 58, 1808–1812 CrossRef CAS PubMed.
- Y. Xiong, S. Yao, T. Szilvási, A. Ruzicka and M. Driess, Chem. Commun., 2020, 56, 747–750 Search PubMed.
- (a) M. J. Evans, M. G. Gardiner, M. D. Anker and M. P. Coles, Chem. Commun., 2022, 58, 5833–5836 Search PubMed; (b) R. Y. Kong and M. R. Crimmin, J. Am. Chem. Soc., 2018, 140, 13614–13617 Search PubMed; (c) R. Y. Kong, M. Batuecas and M. R. Crimmin, Chem. Sci., 2021, 12, 14845–14854 RSC; (d) A. Heilmann, J. Hicks, P. Vasko, J. M. Goicoechea and S. Aldridge, Angew. Chem., Int. Ed., 2020, 59, 4897–4901 Search PubMed; (e) A. Heilmann, M. M. D. Roy, A. E. Crumpton, L. P. Griffin, J. Hicks, J. M. Goicoechea and S. Aldridge, J. Am. Chem. Soc., 2022, 144, 12942–12953 Search PubMed.
- (a) L. Werner, J. Hagn and U. Radius, Chem.–Eur. J., 2023, 29, e202303111 Search PubMed; (b) L. Werner, S. Mann and U. Radius, Eur. J. Inorg. Chem., 2023, 26, e202300398 CrossRef CAS; (c) D. Dhara, F. Fantuzzi, M. Härterich, R. D. Dewhurst, I. Krummenacher, M. Arrowsmith, C. Pranckevicius and H. Braunschweig, Chem. Sci., 2022, 13, 9693–9700 Search PubMed; (d) D. Dhara, A. Jayaraman, M. Härterich, R. D. Dewhurst and H. Braunschweig, Chem. Sci., 2022, 13, 5631–5638 RSC.
- Q. Wen, R. Li, X. Liu, Q. Peng and Y. Chen, J. Am. Chem. Soc., 2025, 147, 36862–36869 Search PubMed.
- (a) CCDC 2499949: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pxdjk; (b) CCDC 2499950: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pxdkl; (c) CCDC 2499951: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pxdlm; (d) CCDC 2499952: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pxdmn; (e) CCDC 2499953: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pxdnp; (f) CCDC 2499954: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pxdpq.
Footnote |
| † M. L. and J. V. contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |