Open Access Article
Open Access ArticleThe critical role of isolated Ti3+ sites in MIL-125 for photocatalytic nitrate reduction: performance enhancement and deactivation mechanism
Lijun
Liao
 *ac,
Guangquan
Zhao
b,
Ruiwen
Shu
*ad,
Xuepeng
Wang
*ac,
Guangquan
Zhao
b,
Ruiwen
Shu
*ad,
Xuepeng
Wang
 b,
Jinxin
Zhang
a and
Ruting
Yuan
*b
b,
Jinxin
Zhang
a and
Ruting
Yuan
*b
aSchool of Chemical and Blasting Engineering, School of Earth and Environment, Anhui University of Science and Technology, Huainan, 232001, China. E-mail: lijun.liao@aust.edu.cn; rwshu@aust.edu.cn
bSchool of Chemistry and Chemical Engineering, Qilu University of Technology (Shandong Academy of Sciences), Jinan, Shandong 250353, China. E-mail: rutingyuan@qlu.edu.cn
cHubei Key Laboratory of Novel Reactor and Green Chemical Technology, Wuhan Institute of Technology, Wuhan 430072, China
dState Key Laboratory of New Textile Materials and Advanced Processing, Wuhan Textile University, Wuhan 430200, China
First published on 23rd February 2026
Abstract
Photocatalytic reduction of nitrate (NO3−) to ammonia (NH3) represents a sustainable route for wastewater treatment and chemical production. Metal–organic frameworks (MOFs) such as MIL-125 are promising catalysts, yet their performance often decays during reaction. This study reveals that the aggregation state of Ti3+ sites in MIL-125 is a critical factor governing its photocatalytic efficiency. We demonstrate that the as-synthesized MIL-125, featuring isolated Ti3+ sites, achieves a high NH3 production rate of 738.32 µmol g−1 h−1 under full-spectrum light without sacrificial agents. Comprehensive characterization and theoretical calculations indicate that these isolated Ti3+ sites elevate the material's conduction band and enhance NO3− adsorption, thereby lowering the energy barrier for the rate-determining step. However, prolonged photoreaction leads to linker deficiency, resulting in agglomeration of Ti3+ sites. This structural evolution causes a negative shift in the conduction band potential, impedes charge separation, and increases the reaction barrier, leading to a noticeable loss in activity over cycles. This work underscores the pivotal role of maintaining isolated metal sites for designing stable and efficient MOF-based photocatalysts for nitrogen cycling.
1 Introduction
Ammonia (NH3) is an indispensable chemical widely used in agricultural fertilizers and is considered a promising carbon-free energy carrier due to its high hydrogen density.1,2 Currently, NH3 is predominantly synthesized via the Haber–Bosch process, which operates under severe conditions of high temperature (400–500 °C) and pressure (150–300 atm), consuming 1.0–2.0% of the world's energy supply and accounting for approximately 1.6% of global carbon emissions.3,4 The extreme energy demands are primarily due to the high dissociation energy (941 kJ mol−1) of the inert N![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N bond in dinitrogen (N2) and its low solubility in aqueous systems, thereby posing significant kinetic challenges.5,6 Photocatalytic nitrate (NO3−) reduction for ammonia synthesis under ambient conditions may serve as a complementary approach rather than a direct replacement for the Haber–Bosch process in large-scale industrial settings. This route is advantageous because the N
N bond in dinitrogen (N2) and its low solubility in aqueous systems, thereby posing significant kinetic challenges.5,6 Photocatalytic nitrate (NO3−) reduction for ammonia synthesis under ambient conditions may serve as a complementary approach rather than a direct replacement for the Haber–Bosch process in large-scale industrial settings. This route is advantageous because the N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond in NO3− has a lower dissociation energy (204 kJ mol−1), and nitrate is highly soluble in water and abundantly present as a pollutant in wastewater.7,8 The overall reaction (NO3− + 9H+ + 8e− → NH3 + 3H2O) offers a pathway for simultaneous wastewater treatment and value-added ammonia synthesis under ambient conditions.9
O bond in NO3− has a lower dissociation energy (204 kJ mol−1), and nitrate is highly soluble in water and abundantly present as a pollutant in wastewater.7,8 The overall reaction (NO3− + 9H+ + 8e− → NH3 + 3H2O) offers a pathway for simultaneous wastewater treatment and value-added ammonia synthesis under ambient conditions.9
Photocatalytic NH3 production from NO3− reduction is a promising strategy for simultaneous wastewater treatment and sustainable NH3 synthesis. The efficiency of this process is primarily governed by the catalyst's ability to adsorb NO3−, multi-electron transfer efficiency, and reaction energy barriers.10 Recent advancements focus on sophisticated material design to optimize these steps. Defect engineering, such as introducing boron dopants and nitrogen vacancies into g-C3N4, can modulate the electronic structure to strengthen nitrate adsorption and inhibit hydrogen evolution, thereby achieving high photocatalytic performance for NH3 generation.11 The construction of Ta3N5/CdIn2S4 S-scheme heterojunctions can promote efficient charge separation while preserving strong redox potentials, which is crucial for the demanding 8-electron reduction process.12 In addition, the incorporation of single-atom catalysts, such as Ni on defective WO3 or Pt on Ti-deficient TiO2 nanosheets, provides highly active sites that not only lower the energy barriers for key reaction steps but also enhance selectivity by minimizing undesirable N2 formation.13,14 Moreover, strategies like coupling the reduction reaction with the oxidation of sacrificial agents such as formic acid can generate additional reducing radicals such as CO2˙−, which synergistically enhance the overall reduction efficiency.15
Metal–organic frameworks (MOFs) are promising photocatalysts for NH3 production via NO3− reduction, owing to their structural tunability, which enhances catalytic activity.16,17 Doping Ru into a Ni-MOF can shift the d-band center of adjacent Ni sites, thereby optimizing the adsorption strength of nitrogen intermediates and boosting catalytic performance.18 Similarly, incorporating Cu2+ into NH2-MIL-125 can induces p–d orbital hybridization between Cu 3d and nitrate O 2p orbitals, thereby stabilizing bidentate nitrate adsorption and lowering the activation energy barrier for a high NH3 yield of 32.8 mg g−1 h−1.19 Another prominent strategy involves constructing nanoarchitectured heterojunctions, such as the S-scheme system in NH2-MIL-125@Co(OH)2 confined within a ZIF-8 “nanohouse”, which improves electron–hole separation and utilizes the ZIF-8 shell's positive charge to enrich nitrate reactants and suppress competing hydrogen evolution, thus achieving a high NH3 yield of 2454.9 µmol g−1 h−1.20 These studies demonstrate that rational design of MOF-based photocatalysts is a highly effective route for efficient photocatalytic NH3 synthesis. Structural defects in MOFs, whether intrinsically formed or deliberately engineered to enhance catalytic properties, are frequently implicated in performance decay under practical operating conditions. Defect sites, such as missing linkers or clusters, can act as preferential adsorption centers for poisons, leading to site blocking and reduced catalytic activity, as demonstrated in NH3-SCR systems for NOx abatement where poisoning accelerates deactivation.21 Moreover, defects can lower the thermal and chemical stability of MOFs, promoting sintering of metal nodes, phase transformations, or framework collapse, which irreversibly diminishes catalytic efficiency over time.22 For instance, in thermally defect-engineered precious group MOFs such as Ru/Rh-HKUST-1, while the initial activity may be enhanced, uncontrolled defect generation can introduce unstable, reduced metal sites that facilitate unwanted side reactions or structural degradation, ultimately leading to catalyst deactivation.23 A critical balance in defect density and characterization is essential to harness benefits while mitigating performance decay. However, to our knowledge, the influence of the change in the Ti3+ site induced by surface reconstruction and defect generation during the reaction on NH3 photosynthesis efficiency has never been reported.
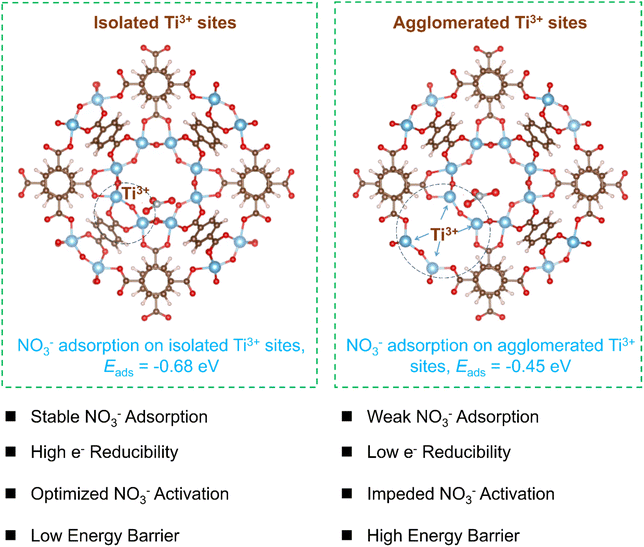
In this study, we demonstrate the presence of isolated Ti3+ sites in as-prepared MIL-125 to enhance NO3− adsorption and generate high-energy electrons, lowering reaction energy barriers and promoting NH3 photosynthesis efficiency (Fig. 1 left). However, agglomerated Ti3+ sites can form during photocatalytic nitrate reduction due to increased missing linkers in MIL-125 (Fig. 1, right), thereby decreasing catalytic performance. Theoretical and experimental results show that isolated Ti3+ sites in MIL-125 increase the electron density and the conduction band minimum, thereby enhancing NO3− adsorption and photo-induced electron reduction capacity. The highest NH3 photosynthesis rate of 738.32 µmol gcat−1 h−1 can be achieved in the absence of a sacrificial agent under full-spectrum illumination.
2 Materials and methods
Commercial reagents were utilized in this work, including terephthalic acid (TPA), N,N-dimethylformamide (DMF), tetra-n-butyl titanate (Ti(OC4H9)4), and methanol (CH3OH), which were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Terephthalic acid (3 g, 18 mmol) was dissolved in 54 mL of N,N-dimethylformamide (DMF) and stirred for 30 min until complete dissolution. Meanwhile, tetra-n-butyl titanate Ti(OC4H9)4 (1.56 mL, 4.5 mmol) was added dropwise to 6 mL of methanol and stirred for 30 min to form a transparent solution. Then, the latter solution containing the titanate was slowly added to the former solution containing the organic linker. The color of the mixture slowly changed from pale yellow to milky white under continuous stirring for 30 min. The mixture was afterwards heated in an oil bath at 100 °C for 24 h under reflux. After cooling to room temperature, the suspension was centrifuged at 6000 rpm for 3 min and then washed with 25 mL DMF to remove the unreacted organic ligands. Afterwards, the collected white MIL-125 powder was washed with 25 mL methanol three times and dried at 80 °C in an oven overnight. Characterization and photocatalytic nitrate reduction reaction with specific details are described in the SI.3 Results and discussion
3.1 NO3− to NH4+ photosynthesis performance
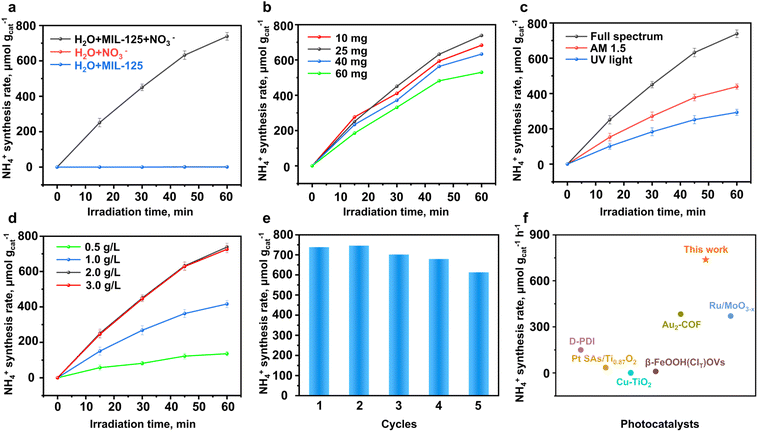
The evaluation of NO3− reduction for NH4+ photosynthesis was first performed in a 50 mL NaNO3 solution (2 g L−1 of NO3−) without any sacrificial agents under full-spectrum illumination. Generally, 25 mg of as-prepared MIL-125 was employed as the photocatalyst in the reaction system, above which light illumination was applied. As depicted in Fig. 2a, the as-made MIL-125 exhibits excellent photocatalytic performance for NO3− reduction to NH4+, achieving a NH4+ photosynthesis rate of 738.32 µmol gcat−1 h−1. The control experiments were conducted by omitting NO3− or MIL-125 from the reaction system. The reaction mixture was illuminated for 1 h and sampled every 15 min. Ultrapure water (18.2 MΩ cm) and high-purity NaNO3 (>99%) were utilized during the photocatalytic reactions to exclude potential ammonia contamination. Direct analysis of NaNO3 solution by Nessler's reagent method was conducted, thereby confirming the absence of ammonia in the prepared solutions. It is observed that no NH4+ was formed under light illumination without NO3− or MIL-125 (Fig. 2a). In addition, no NH4+ was detected in the catalysis system of MIL-125 in the absence of light illumination, indicating that the NO3− reduction to NH4+ process was driven by photocatalysis. Since the photocatalytic efficiency is directly influenced by reaction parameters, the reaction parameters, including photocatalyst dosage (Fig. 2b), light source (Fig. 2c), and nitrate concentration (Fig. 2d) are thoroughly investigated to further enhance photocatalytic activity for ammonia synthesis from nitrate reduction. As presented in Fig. 2b, the optimized photocatalyst dosage was 25 mg to achieve the highest photocatalytic activity for NO3− reduction to NH4+. In addition, due to the low UV content in solar energy, various light sources were used to evaluate photocatalytic performance, as shown in Fig. 2c. The optimal NH4+ formation efficiency with MIL-125 is achieved under full-spectrum light illumination.The nitrate concentration was optimized to enhance photocatalytic performance, as demonstrated in Fig. 2d. It is observed that the relative increase in nitrate concentration improves the reaction kinetics of the photocatalytic nitrate reduction reaction, thereby enhancing photocatalytic activity. Notably, during all conducted experiments, MIL-125 demonstrated slightly decreased photocatalytic activity for the NH4+ synthesis rate. Therefore, long-term recyclability measurements were further investigated for NH4+ production via NO3− reduction (Fig. 2e). Similarly, the MIL-125 nanoparticles exhibit a significant decrease in photocatalytic performance after 5 consecutive cycles. As shown in Fig. 2e, the NH4+ photosynthesis rate decreased from 738.32 to 612.82 µmol gcat−1 h−1 after recyclability measurements probably due to structural defects created during the reaction process, which will be discussed in subsequent sections. Although MIL-125 presented decreased activity for NH4+ photosynthesis, comparative analyses with other photocatalysts suggest that the as-prepared MIL-125 achieved a decent photocatalytic activity for nitrated reduction without a sacrificial agent and cocatalyst, as shown in Fig. 2f and Table S1 (SI).
3.2 Characterization and electronic properties of MIL-125 before and after the reaction
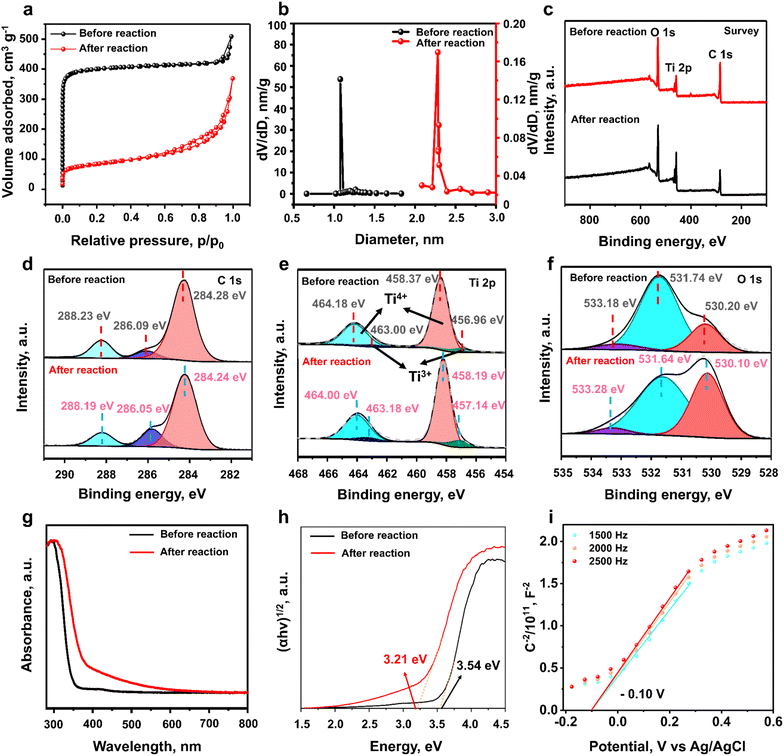
Since the photocatalytic performance of MIL-125 in the nitrate reduction reaction decreased with reaction time, various characterization studies of the sample before and after the reaction were conducted to unravel the mystery of this phenomenon. The crystal structures of the freshly prepared MIL-125 and the sample after a 1 h photocatalytic NO3− reduction reaction were examined by XRD. As revealed in Fig. 3a, the XRD patterns of MIL-125 after the 1 h reaction did not demonstrate compelling differences compared to the pristine MIL-125 before the reaction, suggesting that the main phase structure of MIL-125 was retained after the photocatalytic nitrate reduction reaction.24 The SEM image of MIL-125 (Fig. 3b) reveals a cake-like morphology with random pores. Notably, after 1 h of photocatalytic nitrate reduction, the sample demonstrated a similar morphology with a rough surface, as shown in Fig. 3c. This result indicates that certain surface reconstruction occurred on MIL-125 during the nitrate photo–reduction reaction, although the crystal structure of the sample was maintained after the reaction. A closer observation of the TEM images (Fig. 3d–f) shows that the diameter of the MIL-125 nanoparticles is around 400 nm. Fig. 3g–i shows that the MIL-125 sample after the reaction demonstrated similar particle sizes and morphology. Compared with the pristine MIL-125, the sample after the reaction also shows surface roughness, as observed in the TEM images (Fig. 3g–i). In addition, more pores can be observed in the samples after photocatalytic nitrate reduction, suggesting that possible structural defects may have been generated during the reaction.To investigate the porosity of MIL-125 before and after the reaction, the BET surface area and pore size distribution were analyzed from the N2 sorption isotherm. As shown in Fig. 4a, both samples exhibit type IV N2 sorption isotherm curves with an untypical hysteresis loop,25 suggesting the existence of hierarchical porous structures.26 The N2 isotherm curve of the pristine MIL-125 shows significant adsorption at a low relative pressure (P/P0 below 0.01) before reaching a plateau, confirming the presence of large amounts of micropores in the sample.27 In contrast, after 1 h of nitrate reduction reaction, only a slight increase in the adsorption isotherm at a relatively low pressure can be observed, indicating that most micropores in the sample disappeared after the photocatalytic reaction. Notably, compared to the pristine MIL-125, the hysteresis loop is much more pronounced in the N2 sorption isotherm of the sample after the photocatalytic nitrate reduction reaction, indicating that more mesopores were produced after the reaction. In addition, the BET surface area of MIL-125 decreased sharply from 1195 to 278 m2 g−1 after the reaction, probably due to partial removal of the organic linkers, consistent with the N2 adsorption isotherm results. The decreased surface area could provide fewer active sites and reduced mass transfer efficiency, thereby decreasing the photocatalytic activity. The pore size distributions also verify the generation of mesopores in the sample after the photocatalytic reaction, as presented in Fig. 4b.
To unravel the surface electronic structure and chemical state of the samples before and after the reaction, XPS measurements were performed (Fig. 4c–f). In the survey spectra, three dominant signals at 284, 458, and 531 eV can be ascribed to C 1s, Ti 2p, and O 1s, respectively, as shown in Fig. 4c. The C 1s XPS spectra in Fig. 4d confirm the presence of terephthalic acid in both samples. The XPS peaks at 284.28, 286.09, and 288.23 eV can be ascribed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C–O, and C
C, C–O, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds, respectively, in the organic ligands in MIL-125.28 The binding energies at 458.37 and 464.18 eV in the Ti 2p XPS spectra can be attributed to Ti 2p3/2 and Ti 2p1/2 of the tetra-valent Ti4+ in the Ti-oxo clusters, respectively.29 The XPS signals of tri-valence Ti3+ at the binding energies of 456.96 and 463.00 eV are observed in both pristine MIL-125 and the sample after the reaction, suggesting the presence of organic ligand defects in both samples.30 A small amount (ca. 10%) of missing linker defects are typically present in MOFs synthesized by a regular process31 Notably, the ratios of Ti3+ in the as-prepared MIL-125 obviously increased after the photocatalytic nitrate reduction reaction due to the increased missing ligands, thereby resulting in the agglomeration of the active Ti3+ sites. In addition, the binding energy of Ti3+ in the sample after the reaction shifted to a higher energy by 0.18 eV, which may be due to electron delocalization among agglomerated Ti3+ sites, resulting in a decrease in the electron cloud density around Ti3+.29 This result could lead to a decreased reduction capacity of the Ti3+ sites, thereby undermining the photocatalytic activity for nitrate reduction. In the O 1s XPS spectra (Fig. 4f), three binding energies centred at 530.20, 531.74, and 533.18 eV can be attributed to Ti–O, C
O bonds, respectively, in the organic ligands in MIL-125.28 The binding energies at 458.37 and 464.18 eV in the Ti 2p XPS spectra can be attributed to Ti 2p3/2 and Ti 2p1/2 of the tetra-valent Ti4+ in the Ti-oxo clusters, respectively.29 The XPS signals of tri-valence Ti3+ at the binding energies of 456.96 and 463.00 eV are observed in both pristine MIL-125 and the sample after the reaction, suggesting the presence of organic ligand defects in both samples.30 A small amount (ca. 10%) of missing linker defects are typically present in MOFs synthesized by a regular process31 Notably, the ratios of Ti3+ in the as-prepared MIL-125 obviously increased after the photocatalytic nitrate reduction reaction due to the increased missing ligands, thereby resulting in the agglomeration of the active Ti3+ sites. In addition, the binding energy of Ti3+ in the sample after the reaction shifted to a higher energy by 0.18 eV, which may be due to electron delocalization among agglomerated Ti3+ sites, resulting in a decrease in the electron cloud density around Ti3+.29 This result could lead to a decreased reduction capacity of the Ti3+ sites, thereby undermining the photocatalytic activity for nitrate reduction. In the O 1s XPS spectra (Fig. 4f), three binding energies centred at 530.20, 531.74, and 533.18 eV can be attributed to Ti–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and hydroxyl groups, respectively.29 It can be observed that the ratio of C
O, and hydroxyl groups, respectively.29 It can be observed that the ratio of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O significantly decreased in the used sample due to the removal of organic ligands in MIL-125.
O significantly decreased in the used sample due to the removal of organic ligands in MIL-125.
3.3 Investigation of the reaction mechanism
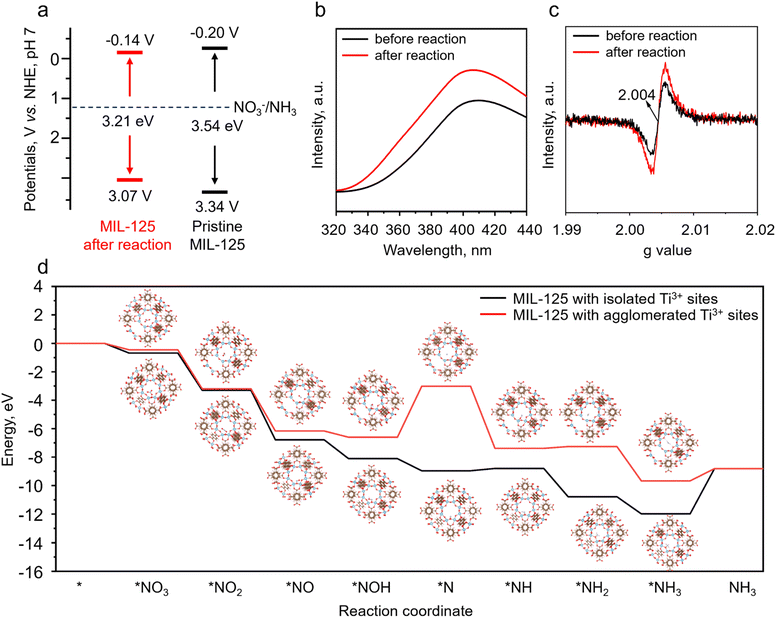
The optical properties of the samples were analyzed using solid-state UV-vis DRS. A redshift of the light absorption edge of MIL-125 was observed in Fig. 4g due to the formation of structural defects. The bandgap energy of the samples before and after the reaction was determined by the Tauc plot to be 3.54 and 3.21 eV, respectively (Fig. 4h). Although the light-utilizing ability of the sample after the reaction was improved, the photocatalytic performance for nitrate reduction gradually decreased (Fig. 2a–e). This phenomenon can be explained by the fact that photocatalytic activity is not only determined by light utilization but also by the reducibility of photo-generated electrons, reactant adsorption, and the reaction energy barrier at the rate-determining step. As mentioned before, the increased linker defects induced aggregated Ti3+ sites, thereby changing the band structures, lowering the reduction potential, and weakening nitrate adsorption possibly due to the downward d-band center shift.32 Mott–Schottky plots of the MIL-125 samples (Fig. 4i and S1 in the SI) before and after the reaction were utilized to determine the corresponding band structures. Positive slopes were identified in the Mott–Schottky plots, suggesting the n-type nature of the MIL-125 photocatalyst.33 The flat band potentials of pristine MIL-125 and the one after the reaction were determined to be −0.10 and −0.04 V, respectively. In n-type semiconductors, the conduction band typically showed a more negative potential, around −0.10 V, relative to the corresponding flat band potential.34 Therefore, the conduction band minima of pristine MIL-125 and the one after the reaction can be calculated to be −0.20 and −0.14 V, respectively.Based on the above results and the obtained bandgap energies, the band structures of the samples are proposed in Fig. 5a. Both band alignments are suitable for nitrate reduction to ammonia (NO3−/NH3, 1.20 V vs. NHE),3 as presented in Fig. 5a. The conduction band edge (−0.20 V) of pristine MIL-125 with isolated Ti3+ sites is more negative than that (−0.14 V) of the sample after the reaction with agglomerated Ti3+ sites, indicating that the reducibility of photo-generated electrons decreased as the reaction proceeded due to the change of band alignment. To investigate the charge-separation efficiency of the samples, photoluminescence (PL) measurements were conducted with an excitation wavelength of 240 nm. As shown in Fig. 5b, the sample after the reaction exhibited much higher PL intensity than the original MIL-125, suggesting decreased charge separation and transfer efficiency.35 The oxygen vacancies of MIL-125 before and after the reaction induced by the missing linkers were studied by EPR. As presented in Fig. 5c, the pristine MIL-125 exhibits an EPR signal at g = 2.004 in the spectrum, indicating that certain amounts of structural defects were present in the as-prepared original sample.36 The sample after the photocatalytic nitrate reduction reaction presents higher EPR signal intensity than the original MIL-125, confirming that more oxygen vacancies were created because of the removal of organic ligands during the reaction process. This result is consistent with XPS and N2 sorption isotherm results. Therefore, the XPS, EPR, and BET trends are consistent with the above-mentioned hypothesis that certain amounts of organic ligands were lost during the photocatalytic nitrate reduction process. Thermogravimetric analysis (TGA) was utilized to provide direct confirmation of ligand loss. As shown in Fig. S2 (SI), approximately 25% of the organic linkers lost during the photocatalytic nitrate reduction process,37 which is consistent with the XPS, EPR, and BET results, providing direct evidence of the linker loss.
Based on the above investigations, fewer structural defects in pristine MIL-125 could induce the formation of isolated Ti3+ sites, as previously proposed in Fig. 1. Many more ligand defects formed during the photocatalytic nitrate reduction process could lead to the generation of agglomerated Ti3+ sites. Density functional theory (DFT) calculations were applied to further investigate the photocatalytic mechanism and rate-determining step in the NO3− reduction to NH3 process. The energy barriers for elemental steps in the nitrate reduction to ammonia process in the presence of MIL-125 with isolated Ti3+ sites or agglomerated Ti3+ sites are presented in Fig. 5d. The adsorption energy of NO3− on MIL-125 with isolated Ti3+ sites (−0.68 eV) is obviously higher than that of MIL-125 with agglomerated Ti3+ sites (−0.45 eV), which could accelerate the nitrate reduction process. The rate-determining step in nitrate reduction in the presence of MIL-125 with agglomerated Ti3+ sites is the formation of *N from *NOH with a reaction energy barrier of 3.59 eV (Fig. 5d). With the presence of isolated Ti3+ sites in MIL-125, the rate-determining step in nitrate reduction to ammonia is the desorption of NH3 with a lower reaction energy barrier of 3.17 eV. Although this barrier is reduced compared to the agglomerated case, it is by no means negligible and represents a significant kinetic limitation even for the optimized catalyst. The high desorption energy suggests that NH3 release may inherently constrain the maximum achievable activity, underscoring that the initial high performance relies on the balanced enhancement of nitrate adsorption and charge separation to partially offset this limitation. Future study should be conducted to decrease the large barrier for NH3 desorption to further improve NH3 photosynthesis efficiency. Nevertheless, the existence of isolated Ti3+ sites in MIL-125 could promote the nitrate adsorption and charge separation and transfer efficiency, and lower the reaction energy barrier for NH3 photosynthesis. However, the increased amounts of missing ligands in MIL-125 during the nitrate reduction process induced agglomerated Ti3+ sites, thereby lowering the reducibility of photo-generated electrons, increasing the reaction energy barrier, and decreasing photocatalytic activity for NH3 photosynthesis. The DFT calculations provide a mechanistic foundation for the experimental performance decay of MIL-125 in photocatalytic nitrate reduction, explicitly linking the increased energy barrier for the rate-determining step to the observed kinetic slowdown. For isolated Ti3+ sites, the rate-determining step is NH3 desorption with a barrier of 3.17 eV, whereas agglomerated sites shift the bottleneck to *N formation from *NOH with a higher barrier of 3.59 eV, as depicted in Fig. 5d. This computational barrier increase directly correlates with the experimental decline in NH4+ production rate from 738.32 to 612.82 µmol g−1 h−1 over recycling cycles, shown in Fig. 2e, where Ti3+ site aggregation impedes charge separation and elevates the kinetic hurdle. Thus, the synergy between DFT and experimental data solidifies that preserving isolated Ti3+ sites is crucial to maintain low energy barriers and high efficiency.
4 Conclusions
In conclusion, we demonstrate that the photocatalytic efficiency of MIL-125 for nitrate reduction to ammonia is critically governed by the atomic dispersion of Ti3+ sites within its structure. The as-synthesized catalyst, featuring isolated Ti3+ sites, achieves a high NH3 production rate of 738.32 µmol g−1 h−1 by facilitating strong nitrate adsorption and providing photo-generated electrons with high reduction potential. However, prolonged photoreaction induces increased loss of organic linkers, leading to the formation of agglomerated Ti3+ sites. This transformation detrimentally alters the catalyst's properties, causing a positive shift in the conduction band that reduces electron reducibility, impairing charge separation efficiency, and increasing the energy barrier of the rate-determining step. Consequently, the agglomerated Ti3+ sites result in a significant decline in photocatalytic activity. These findings reveal that the initial high performance is intrinsically linked to the presence of isolated Ti3+ sites, while the subsequent deactivation is a direct result of their aggregation under operating conditions. Therefore, the long-term stability and efficiency of MIL-125-based photocatalysts depend on developing strategies to mitigate linker deficiency and preserve the atomic dispersion of active metal sites, highlighting a crucial design principle for durable MOF photocatalysts in sustainable nitrogen cycling applications.Author contributions
L. L., R. S. and R. Y. contributed to the conception and design of the experiments. L. L. directed the project and analyzed the data. L. L. and G. Z. performed the experiments and analyzed the data. L. L. wrote the manuscript. X. W. and J. Z. provided valuable suggestions during the project. All authors discussed the results and commented on the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI).Supplementary information: characterization and experimental details, DFT calculations details, comparison of the photocatalytic ammonia synthesis efficiency between MIL-125 in this work and recently reported photocatalysts, and Mott–Schottky curves of MIL-125 after a 1 h photocatalytic nitrate reduction reaction. See DOI: https://doi.org/10.1039/d5sc09647b.
Acknowledgements
We gratefully acknowledge the support of the Scientific Research Foundation for High-level Talents of Anhui University of Science and Technology (No. 2025yjrc0125), the State Key Laboratory of Heavy Oil Processing, China University of Petroleum (No. SKLHOP202402009), the Shandong Provincial Natural Science Foundation (No. ZR2023QD036), the Hubei Key Laboratory of Novel Reactor and Green Chemical Technology (No. NRG202506), and the State Key Laboratory of New Textile Materials and Advanced Processing, Wuhan Textile University (No. FZ2025028).Notes and references
- G. Youn, I. Hong, B. Song, D. Lee, Y. Yun, H. S. Moon and K. Yong, Sol. RRL, 2024, 8, 2300880 Search PubMed.
- X. He, J. Li, R. Li, D. Zhao, L. Zhang, X. Ji, X. Fan, J. Chen, Y. Wang, Y. Luo, D. Zheng, L. Xie, S. Sun, Z. Cai, Q. Liu, K. Ma and X. Sun, Inorg. Chem., 2023, 62, 25–29 CrossRef CAS PubMed.
- H. S. Moon, B. Song, J. Jeon, T.-H. Lai, Y.-P. Chang, Y.-D. Lin, J. K. Park, Y.-G. Lin, Y.-J. Hsu, H. Shin, Y. Yun and K. Yong, Appl. Catal., B, 2023, 339, 123185 CrossRef CAS.
- K. Fan, W. Xie, J. Li, Y. Sun, P. Xu, Y. Tang, Z. Li and M. Shao, Nat. Commun., 2022, 13, 7958 CrossRef CAS PubMed.
- J. Li, R. Chen, J. Wang, Y. Zhou, G. Yang and F. Dong, Nat. Commun., 2022, 13, 1098 Search PubMed.
- H. Hirakawa, M. Hashimoto, Y. Shiraishi and T. Hirai, ACS Catal., 2017, 7, 3713–3720 CrossRef CAS.
- S. Yin, Z. Guan, Y. Zhu, D. Guo, X. a. Chen and S. Wang, ACS Nano, 2024, 18, 27833–27852 CrossRef CAS PubMed.
- L. Wei, Y. Zhang, C. Zhang, C. Yao, C. Ni and X. Li, ACS Appl. Nano Mater., 2023, 6, 13127–13136 Search PubMed.
- S. Han, H. Li, T. Li, F. Chen, R. Yang, Y. Yu and B. Zhang, Nat. Catal., 2023, 6, 402–414 Search PubMed.
- A. P. Varghese and S. K. Lakhera, Renewable Sustainable Energy Rev., 2024, 200, 114537 CrossRef CAS.
- I. Hong, H. S. Moon, B. J. Park, Y.-A. Chen, Y.-P. Chang, B. Song, D. Lee, Y. Yun, Y.-J. Hsu, J. W. Han and K. Yong, Chem. Eng. J., 2024, 484, 149506 Search PubMed.
- Z. Liu, S. Fan, X. Li, Z. Niu, J. Wang, C. Bai, J. Duan, M. O. Tadé and S. Liu, Appl. Catal., B, 2023, 327, 122416 Search PubMed.
- Y. Wang, H. Yin, X. Zhao, Y. Qu, A. Zheng, H. Zhou, W. Fang and J. Li, Appl. Catal., B, 2024, 341, 123266 Search PubMed.
- H. Jung, G. Cha, H. Kim, J. Will, X. Zhou, E. Spiecker, J. Breu and P. Schmuki, J. Am. Chem. Soc., 2025, 147, 9049–9055 Search PubMed.
- A. P. Varghese, R. T. P, B. Neppolian, A. S. AlArifi and S. K. Lakhera, ACS Appl. Energy Mater., 2024, 7, 6527–6536 Search PubMed.
- C. Zhang, Y. Wu, D. Li and H.-L. Jiang, Chem. Sci., 2025, 16, 13149–13172 Search PubMed.
- T. Wang, Y. Wang, M. Sun, A. Hanif, H. Wu, Q. Gu, Y. S. Ok, D. C. W. Tsang, J. Li, J. Yu and J. Shang, Chem. Sci., 2020, 11, 6670–6681 Search PubMed.
- Y. Yao, X. Wei, H. Zhou, K. Wei, B. Kui, F. Wu, L. Chen, W. Wang, F. Dai, P. Gao, N. Wang and W. Ye, ACS Catal., 2024, 14, 16205–16213 Search PubMed.
- Y. Zhao, J. Shen, J. Yuan, H. Mao, X. Cheng, Z. Xu and Z. Bian, Nano Energy, 2024, 109499 Search PubMed.
- Y. Xi, Y. Xiang, T. Bao, Z. Li, C. Zhang, L. Yuan, J. Li, Y. Bi, C. Yu and C. Liu, Angew. Chem., Int. Ed., 2024, 63, e202409163 CrossRef CAS PubMed.
- J. Deng, X. Hu, A. J. Klaver, J. Liu, B. Liu, L. Bai, M. Xie and D. Zhang, Chem. Rev., 2025, 125, 11260–11357 CrossRef CAS PubMed.
- S. C. Moore, M. R. Smith, J. L. Trettin, R. A. Yang and M. L. Sarazen, ACS Energy Lett., 2023, 8, 1397–1407 CrossRef CAS.
- W. R. Heinz, I. Agirrezabal-Telleria, R. Junk, J. Berger, J. Wang, D. I. Sharapa, M. Gil-Calvo, I. Luz, M. Soukri, F. Studt, Y. Wang, C. Wöll, H. Bunzen, M. Drees and R. A. Fischer, ACS Appl. Mater. Interfaces, 2020, 12, 40635–40647 CrossRef CAS PubMed.
- C.-D. Hao, Z.-Y. Zhang, A.-X. Yu, J.-J. Li, Q. Liu, X.-J. Bo, D.-Y. Du, S. Yuan and Z.-M. Su, Inorg. Chem., 2025, 64, 10001–10011 Search PubMed.
- C. Fan, V. Nguyen, Y. Zeng, P. Phadungbut, T. Horikawa, D. D. Do and D. Nicholson, Microporous Mesoporous Mater., 2015, 209, 79–89 Search PubMed.
- Y. Sun, H. Ji, Y. Sun, G. Zhang, H. Zhou, S. Cao, S. Liu, L. Zhang, W. Li, X. Zhu and H. Pang, Angew. Chem., Int. Ed., 2024, 63, e202316973 Search PubMed.
- L. Hu, W. Wu, L. Gong, H. Zhu, L. Jiang, M. Hu, D. Lin and K. Yang, Angew. Chem., Int. Ed., 2023, 62, e202215296 Search PubMed.
- Q. Zhang, Y. Chen, X. Yu, Y. Yin, Y. Ru and G. Tian, Catal. Sci. Technol., 2023, 13, 4525–4533 RSC.
- X. Song, Y. Wang, T. Zhu, J. Liu and S. Zhang, Chem. Eng. J., 2021, 416, 129126 Search PubMed.
- C. Liu, Q. Chen, Y. Chen, J. C. Yu, J. Wu and L. Wu, J. Colloid Interface Sci., 2025, 678, 616–626 Search PubMed.
- L. Liu, Z. Chen, J. Wang, D. Zhang, Y. Zhu, S. Ling, K.-W. Huang, Y. Belmabkhout, K. Adil, Y. Zhang, B. Slater, M. Eddaoudi and Y. Han, Nat. Chem., 2019, 11, 622–628 CrossRef CAS PubMed.
- X. Sun, L. Sun, G. Li, Y. Tuo, C. Ye, J. Yang, J. Low, X. Yu, J. H. Bitter, Y. Lei, D. Wang and Y. Li, Angew. Chem., Int. Ed., 2022, 61, e202207677 Search PubMed.
- M. Shi, Z. Liu, S. Zhang, S. Liang, Y. Jiang, H. Bai, Z. Jiang, J. Chang, J. Feng, W. Chen, H. Yu, S. Liu, T. Wei and Z. Fan, Adv. Energy Mater., 2022, 12, 2103657 Search PubMed.
- C. An, A. Sikandaier, X. Guo, Y. Zhu, H. Tang and D. Yang, Acta Phys.-Chim. Sin., 2024, 40, 2405019 Search PubMed.
- L. Ni, Y. Xiao, X. Zhou, Y. Jiang, Y. Liu, W. Zhang, J. Zhang and Z. Liu, Inorg. Chem., 2022, 61, 19552–19566 Search PubMed.
- P. H. M. Andrade, H. Ahouari, C. Volkringer, T. Loiseau, H. Vezin, M. Hureau and A. Moissette, ACS Appl. Mater. Interfaces, 2023, 15, 31032–31048 CrossRef CAS PubMed.
- G.-Y. Han, Y. Ji, X.-Y. Li, Y. Gai, G.-Z. Hou, Q.-Y. Cheng, Z. Zhang, Y. Liu, J. Wang, P. Van Der Voort, G. Hou, X.-P. Wu, G. He and X. Feng, Adv. Sci., 2025, 12, e10578 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |