Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Promoted hydrogen activation and spillover over Pt/Co3O4 by facet engineering of Co3O4 for enhanced catalytic hydrogenation
Hui
Yun
,
Jiao
Feng
,
Wanying
Peng
and
Mi
Xiong
 *
*
College of Materials and Chemistry & Chemical Engineering, Chengdu University of Technology, Chengdu 610059, China. E-mail: xiongmi@cdut.edu.cn
First published on 13th March 2026
Abstract
The exposed facets of supported metal catalysts play a crucial role in catalytic hydrogenation performance. However, the internal relationship between the support crystal facet and catalytic performance needs to be further explored. Herein, a series of well-defined Pt/Co3O4-x catalysts are fabricated with similar Pt nanoparticle sizes, identical metal loadings, and tailored Co3O4 crystal facets (x = o, t, c; where “o”, “t”, and “c” denote Co3O4 exposing predominantly (111), mixed (111)/(100), and (100) facets, respectively). The electronic structure of Pt nanoparticles and the hydrogen spillover capability of Pt/Co3O4 are modulated by exposing different crystal facets of Co3O4. For the 4-nitrophenol (4-NP) hydrogenation reaction with H2 as the hydrogen source, the Pt/Co3O4-o catalyst with more Pt0 species and stronger hydrogen spillover capability exhibits the best hydrogenation activity with a turnover frequency (TOF) of 164.2 h−1. Mechanistic studies indicate that, compared with Pt/Co3O4-c, the Pt/Co3O4-o exhibits weaker adsorption and activation of the nitro group, while its ability to activate H2 is stronger. The enhanced catalytic activity of Pt/Co3O4-o is attributed to promoted hydrogen activation and spillover. This work highlights support crystal facet engineering for regulating the electronic structure and hydrogen spillover effect, which provides in-depth insight into catalyst design and hydrogenation mechanism.
Introduction
Catalytic hydrogenation plays a pivotal role in environmental protection and industrial processes,1,2 particularly for the degradation of persistent organic pollutants such as 4-nitrophenol (4-NP),3 a common toxic contaminant in wastewater.4–6 Supported metal catalysts, especially platinum-based systems, have garnered significant attention due to their exceptional hydrogen activation capability.7,8 However, the catalytic performance of such systems is intricately linked not only to the active metal sites but also to the structural and electronic properties of the support material.9–14 Previous extensive research has predominantly focused on elucidating the internal relationship between metal nanoparticle characteristics (including size regulation, alloy composition, and morphological engineering) and the catalytic hydrogenation performance.15–18The microstructures of support, particularly the exposed crystal facet, have been recognized as a critical influencing factor for catalytic hydrogenation performance by modulating the geometric/electronic configurations of metal nanoparticles,19–22 facilitating support-mediated reactant activation processes,23,24 inducing interfacial charge transfer dynamics,25,26 and so on. For example, Gao et al. reported a study on tailoring the electronic states of Pd nanoparticles by modulating the exposed crystal facets of ZIF-8 supports.27 By constructing sandwich-structured ZIF-8x@Pd@ZIF-8 composites, where distinct facets were engineered, the electron density of Pd nanoparticles was precisely controlled. The (100) facet induced electron-deficient Pd species (ZIF-8c@Pd@ZIF-8) could preferentially adsorb the electron-rich nitro group of p-chloronitrobenzene, achieving excellent catalytic activity and selectivity in the hydrogenation of p-chloronitrobenzene to p-chloroaniline. Moreover, the facet engineering of nanocatalysts has proven critical for optimizing hydrogen spillover processes for selective hydrogenation. Jiang et al. reported a groundbreaking study on tailoring the catalytic performance of highly diluted Pd single-atom catalysts through facet engineering of Cu supports.28 By dispersing Pd atoms onto Cu nanosheets (exposing (111) facets) and nanocubes (exposing (100) facets), the authors revealed a striking facet-dependent behavior in the semi-hydrogenation of alkynes. While hydrogen spillover occurred on both facets, only Pd1/Cu(100) exhibited exceptional activity and selectivity even at ultralow Pd loadings (50 ppm). Despite advances in catalyst design, the role of the support facet in modulating electronic states and hydrogen spillover efficiency remains poorly understood.
Herein, we systematically investigate the facet-dependent behavior of Co3O4-supported Pt catalysts. Three distinct Co3O4 morphologies—octahedral (exposing (111) facets), cubic (exposing (100) facets), and truncated intermediates (co-exposing (111) and (100) facets)—were synthesized to anchor Pt nanoparticles with similar sizes and loadings. For the 4-nitrophenol (4-NP) hydrogenation with H2 as the hydrogen source, the Pt/Co3O4-x (x = o, t, c) catalysts present a notable dependence of catalytic activity on exposed facets of supports. And the catalytic activity follows the order: Pt/Co3O4-o > Pt/Co3O4-t > Pt/Co3O4-c. Detailed analyses indicate that, the Pt/Co3O4-o with more Pt0 species has stronger hydrogen activation and spillover capacity and thus significantly accelerates the catalytic hydrogenation performance.
Results and discussion
Catalyst characterization
A series of Co3O4 supports with different morphologies were synthesized by hydrothermal method (Fig. S1). Their scanning electron microscopy (SEM) images demonstrate that the synthesized nanoparticles exhibit good morphology (Fig. S2a–c). For different morphologies of Co3O4, the corresponding exposed crystal facets are distinct. Specifically, the octahedron Co3O4 (Co3O4-o) and cube Co3O4 (Co3O4-c) are enclosed with (111) and (100) facets, respectively, while the truncated octahedron Co3O4 (Co3O4-t) exposes mixed (111) and (100) facets (Fig. S2d–f). Subsequently, the pre-synthesized Pt nanoparticles were supported on the Co3O4-x (x = o, t, c) supports via the colloidal deposition method with ∼0.3 wt% Pt loading (determined by ICP-OES, Table S1), yielding Pt/Co3O4-o, Pt/Co3O4-t, and Pt/Co3O4-c. SEM images of Pt/Co3O4-x (x = o, t, c) show that the size and morphology of the original Co3O4-x (x = o, t, c) are almost maintained after Pt loading (Fig. 1a, e, and i). | ||
| Fig. 1 (a, e, and i) SEM images, (b, f and j) HAADF-STEM images, (c, g, and k) STEM-EDS elemental mappings, and (d, h, and l) HRTEM images of Pt/Co3O4-o, Pt/Co3O4-t, and Pt/Co3O4-c, respectively. | ||
The high-angle annular dark field scanning transmission electron microscopy (HAADF-STEM) with energy dispersive X-ray spectroscopy (EDS) elemental mapping was further employed to characterize Pt/Co3O4-x (x = o, t, c). As shown in Fig. 1b, f, and j, obvious bright spots of Pt nanoparticles are observed. Pt nanoparticles are uniformly dispersed on the Co3O4-x (x = o, t, c) supports (Fig. 1c, g, and k). And the average diameters of Pt nanoparticles are similar, about 2.2 nm. Furthermore, the lattice fringes measured in the high-resolution TEM (HRTEM) images of Pt/Co3O4-x (x = o, t, c) (Fig. 1d, h and l) are 0.23 nm, corresponding to the (111) planes of Pt. N2 adsorption–desorption experiments indicate that the Brunauer–Emmett–Teller (BET) surface areas of Pt/Co3O4-o, Pt/Co3O4-t, and Pt/Co3O4-c are 2.4, 3.4, and 3.4 m2 g−1, respectively, indicating similar surface areas (Table S1).
X-ray diffraction (XRD) was used to investigate the crystal structures of Co3O4-x and Pt/Co3O4-x (x = o, t, c), as shown in Fig. S3a. All XRD patterns show the diffraction peaks located at 19.0°, 31.3°, 36.9°, 38.6°, 44.9°, 55.7°, 59.4°, and 65.3°, which correspond to the (111), (220), (311), (222), (400), (422), (511), and (440) planes of Co3O4, respectively (PDF#43-1003). The intensity ratio of the (111) to (400) peaks for Co3O4-x (x = o, t, c) decreases in order of octahedron (0.9) > truncated octahedron (0.7) > cube (0.5) (Fig. S3b), suggesting morphology-dependent facet exposure. Compared to bare Co3O4-x (x = o, t, c) supports, the Pt-loaded counterparts (Pt/Co3O4-x) exhibit similar trends in the (111)/(400) peak intensity ratios (Fig. S3c), implying that Pt deposition does not significantly perturb the predominant facet distribution of the Co3O4 supports. A semi-quantitative analysis based on the XRD intensity ratios estimates the relative abundance of the (111) facet to be ∼100%, ∼57%, and ∼0% for Pt/Co3O4-o, -t, and -c, respectively, providing a quantitative structural descriptor for the catalyst series (Table S2). No characteristic diffraction peaks belonging to crystalline Pt nanoparticles are observed, which can be attributed to the low loading and/or small size of Pt species.
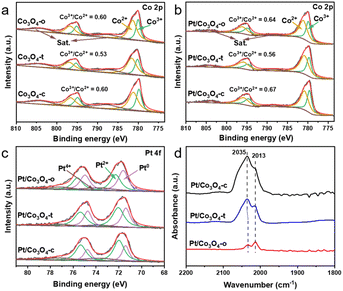
X-ray photoelectron spectroscopy (XPS) was employed to reveal the surface chemical states of Pt and Co. Fig. 2a and b show the Co 2p spectra of Co3O4-x and Pt/Co3O4-x (x = o, t, c), respectively. The peaks at ∼780.2 and 795.4 eV are assigned to Co 2p3/2 and Co 2p1/2, respectively. The peaks of Co 2p3/2 can be deconvoluted into two peaks at ∼780.0 and 781.4 eV, indicating the coexistence of Co3+ and Co2+.29 Notably, compared to the pristine Co3O4-x (x = o, t, c) supports, the Pt-loaded counterparts (Pt/Co3O4-x) with the same morphologies show an increase in the Co3+/Co2+ ratio, implying the transfer of electrons from Co to Pt.30 Moreover, the XPS spectra of Pt 4f for Pt/Co3O4-x (x = o, t, c) show two peaks located at ∼71.7 and 75.0 eV, which are attributed to Pt 4f7/2 and 4f5/2, respectively (Fig. 2c). And the Pt 4f spectra were fitted by Pt0, Pt2+, and Pt4+.31 The fitting results reveal that the Pt0 content follows the order: Pt/Co3O4-o (44.7%) > Pt/Co3O4-t (39.4%) > Pt/Co3O4-c (26.1%) (Table S3), suggesting the differentiated electronic states of Pt nanoparticles with varied Co3O4 facets. To gain insight into the state of Pt under reaction conditions, quasi-in situ XPS analysis was performed after H2 treatment at 40 °C. Intriguingly, although the Pt0 content increased for all catalysts, the facet-dependent order remained unchanged (Fig. S4, Table S3), suggesting that the intrinsic electronic modulation by the support facet is preserved under a reducing atmosphere.
 | ||
| Fig. 2 XPS spectra of Co 2p for (a) Co3O4-x and (b) Pt/Co3O4-x (x = o, t, c). (c) XPS spectra of Pt 4f and (d) CO-DRIFTS spectra for Pt/Co3O4-x (x = o, t, c). | ||
Further, diffuse-reflectance infrared Fourier transform spectroscopy (DRIFTS) of CO chemisorption was performed to evaluate the electronic states of surface Pt species in Pt/Co3O4-x (x = o, t, c), as shown in Fig. 2d. Two absorption bands observed at 2033–2035 and 2013 cm−1 are assigned to the linear absorption of CO on the terrace and step sites of Pt nanoparticles, respectively.32 Compared to the Pt/Co3O4-t and Pt/Co3O4-c, the CO adsorption peak at the terrace sites of Pt/Co3O4-o shifts to a lower wavenumber (from 2035 to 2033 cm−1). Additionally, the relative intensity of the CO signal adsorbed at the step sites follows the order: Pt/Co3O4-o > Pt/Co3O4-t > Pt/Co3O4-c. These results suggest that the Pt species on Pt/Co3O4-o surfaces are in a lower valence state,33 which aligns with the XPS result. The observed charge transfer and modified electronic state of Pt (Fig. 2) confirm that a facet-dependent metal-support interaction is effectively established through our colloidal deposition process, even in the absence of high-temperature treatment.
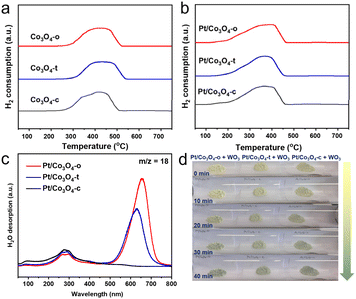
Hydrogen temperature-programmed reduction (H2-TPR) was carried out to investigate the reducibility of Co3O4-x and Pt/Co3O4-x (x = o, t, c), as shown in Fig. 3a and b. H2-TPR profiles show no reduction peak below 100 °C for any Pt/Co3O4-x catalyst, confirming the metallic state (Pt0) of the pre-synthesized nanoparticles. The prominent peaks observed at higher temperatures (∼370 °C) are therefore attributed solely to the reduction of the Co3O4 support. The shift to lower temperatures (by ∼50 °C) compared to bare Co3O4 demonstrates that metallic Pt0 promotes support reduction via hydrogen spillover.34,35
To detect spilled hydrogen, hydrogen temperature-programmed desorption coupled with mass spectrometry (H2-TPD-MS) measurements of Pt/Co3O4-x were performed. As shown in Fig. 3c and S5, the MS signals reveal that the major desorption products are H2O (m/z = 18) and OH species (m/z = 17), with no significant molecular H2 (m/z = 2) detected. This indicates that active hydrogen species generated on Pt0 sites spill over onto the Co3O4 support and react with lattice oxygen to form surface hydroxyl groups, which subsequently recombine and decompose upon heating.36,37 The profiles show two main desorption regions: a peak at ∼280 °C attributed to the removal of weakly-bound hydroxyls, and another more intense peak at ∼650 °C associated with strongly-bound hydroxyls likely formed from hydrogen that has migrated deeper into the oxide lattice. Critically, the intensity of the high-temperature peak follows the order: Pt/Co3O4-o > Pt/Co3O4-t > Pt/Co3O4-c. This provides direct evidence that the (111) facet of Co3O4 is the most effective in stabilizing and incorporating spilled hydrogen.
Further, a color change experiment was conducted to visually evaluate the hydrogen spillover effect (Fig. 3d). By mixing 1 mg of Pt/Co3O4-x catalysts and 300 mg of WO3 nanowires, the original color of the mixtures is light yellow. After being exposed to hydrogen atmosphere, the mixture of Pt/Co3O4-o and WO3 exhibits the most pronounced color change within 40 minutes, implying the strongest hydrogen spillover effect over Pt/Co3O4-o. This result aligns with the H2-TPD-MS data and is attributed to the stronger metal-support interaction in Pt/Co3O4-o, which facilitates the generation and transfer of active hydrogen species (Fig. 2c and d and S6). Further, a parallel experiment conducted in aqueous reaction conditions (Fig. S7) also yielded a distinct color change, confirming that hydrogen spillover persists in the liquid environment relevant to catalysis.
Catalytic performance of Pt/Co3O4-x (x = o, t, c) for 4-NP hydrogenation
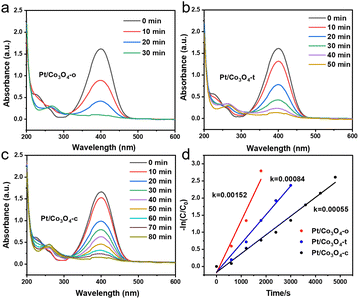
The catalytic performance of Pt/Co3O4-x (x = o, t, c) was evaluated by the 4-NP reduction. The reduction process using H2 as hydrogen source was monitored by UV-vis spectroscopy every ten minutes, and the intensity of the UV absorption peak at 400 nm was used to quantify the concentration of 4-NP. As shown in Fig. 4a–c, the peak intensity at 400 nm gradually decreased as the reaction proceeded. Simultaneously, a new peak appeared at 257 nm, which was ascribed to the formation of 4-aminobenzenol (4-AP) (Fig. S8). Among the Pt/Co3O4-o, Pt/Co3O4-t, and Pt/Co3O4-c catalysts, the Pt/Co3O4-o exhibited the shortest reaction time with nearly 100% conversion (Fig. S9). Furthermore, Fig. 4d shows a logarithmic plot of the concentration (−ln(C/C0) versus reaction time for different catalysts. The concentration (−ln(C/C0) is proportional to the reaction time, and thus the apparent kinetic rate constant (kapp) can be estimated based on slope regression of the logarithmic graph (ln(Ct/C0) = −kappt).38 Compared to the Pt/Co3O4-t and Pt/Co3O4-c catalysts (8.4 × 10−4 and 5.5 × 10−4 s−1, respectively), the Pt/Co3O4-o has the highest kapp value (1.52 × 10−3 s−1), indicating its superior catalytic activity. Under identical reaction conditions, the bare Co3O4 support exhibited negligible catalytic activity (Fig. S10).To further evaluate the intrinsic activity per Pt site, the turnover frequency (TOF) was determined based on CO chemisorption measurements. The calculated TOF values follow the same trend: Pt/Co3O4-o (164.2 h−1) > Pt/Co3O4-t (115.2 h−1) > Pt/Co3O4-c (75.6 h−1) (Table S4). Subsequently, the apparent activation energy (Ea) experiment was performed (Fig. S11). The Ea values follow the order: Pt/Co3O4-o (23.0 kJ mol−1) < Pt/Co3O4-t (25.3 kJ mol−1) < Pt/Co3O4-c (55.2 kJ mol−1). The significantly lower Ea for Pt/Co3O4-o accounts for its superior catalytic activity. More importantly, we establish a quantitative correlation between the Co3O4(111) facet exposure and the TOF (Fig. S12). This linear correlation directly demonstrates that the abundance of the (111) facet is the predominant factor governing the catalytic hydrogenation activity.
For comparison, control catalysts were also prepared via a conventional impregnation-chemical reduction method. While the same activity order (Pt/Co3O4-o > Pt/Co3O4-c) was preserved, the catalytic activities of the impregnation-prepared catalysts were substantially lower than their colloidal-deposition counterparts (Fig. S13). This lower activity is consistent with the larger and less uniform Pt nanoparticles formed by the impregnation method (Fig. S14), demonstrating the advantage of the colloidal deposition route for achieving high Pt dispersion on our low-surface-area Co3O4 supports.
The Pt/Co3O4-o was reused to test the stability for 4-NP hydrogenation. The catalytic activity was well maintained after five cycles (Fig. S15), indicating its high catalytic stability. Post-reaction characterizations (XRD, TEM, and XPS) confirm that the Pt nanoparticle size, dispersion, and chemical state, as well as the Co3O4 crystal facets, remain essentially unchanged after cycling (Fig. S16 and Table S3). Together with the minimal Pt leaching (<0.2%) confirmed by ICP-MS (Table S5), these results demonstrate the robust structural and compositional stability of the catalyst. When nitrobenzene was employed as the substrate, the Pt/Co3O4-o still exhibited the superior catalytic activity (Fig. S17 and S18).
Compared with state-of-the-art catalysts for 4-NP hydrogenation (Table S6), the optimal Pt/Co3O4-o demonstrates a competitive turnover frequency (164.2 h−1) under an exceptionally mild condition (40 °C, 1 bar H2). Many reported systems require significantly higher H2 pressures or temperatures to achieve comparable activity. This combination of high intrinsic activity, low activation barrier, robust stability, and facet-dependent performance underscores the practical promise and fundamental insight offered by our facet-engineered catalyst.
Catalytic mechanism
To elucidate the origin of the superior catalytic performance of Pt/Co3O4-o, diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) was first used to investigate the adsorption behavior of 4-NP on Pt/Co3O4-o and Pt/Co3O4-c as model catalysts, as shown in Fig. S19. The spectra of Pt/Co3O4-o and Pt/Co3O4-c show two bands at 1578 and 1316 cm−1, corresponding to asymmetric stretching and symmetric stretching vibrations of the nitro group, respectively.39 Compared with the Pt/Co3O4-c catalyst, Pt/Co3O4-o exhibits a weaker peak intensity at 1578 cm−1, suggesting a weaker adsorption capability for the nitro group. Further, the adsorption and activation capability of Pt/Co3O4-o and Pt/Co3O4-c for 4-NP and H2 was explored through kinetic experiments. Fig. 5a displays the kinetic behavior for 4-NP adsorption and activation. The reaction order with respect to 4-NP is higher for Pt/Co3O4-o (0.81) than for Pt/Co3O4-c (0.75), implying that it is more difficult for Pt/Co3O4-o to activate the nitro group. This is consistent with the result of DRIFTS. For the adsorption and activation of H2 (Fig. 5b), the reaction order of Pt/Co3O4-o (0.41) is lower than that of Pt/Co3O4-c (0.53), indicating that it is easier for Pt/Co3O4-o to activate H2.To gain theoretical insight, DFT calculations were conducted. Pt4 clusters supported on Co3O4-(111) and Co3O4-(100) (labeled as Pt4/Co3O4-(111) and Pt4/Co3O4-(100), respectively) were constructed to mimic the Pt/Co3O4-o and Pt/Co3O4-c catalysts based on the distinct exposed facets of Co3O4 supports: the octahedral Co3O4-o predominantly exposes the (111) facet, whereas the cubic Co3O4-c preferentially exposes the (100) facet (Fig. S20a and b). First, when Pt4 clusters were anchored on different Co3O4 model surfaces, the electron transfer from Co3O4 to Pt4 was about −1.65|e| for Pt4/Co3O4-(111) and −0.74|e| for Pt4/Co3O4-(100), respectively (Fig. S20c and d, Table S7). The charge density differences indicate that more electrons transfer from Co3O4 to Pt for Pt4/Co3O4-(111), leading to a lower oxidation state of Pt. This is also confirmed by the XPS results. Next, Fig. S21a–d shows the optimized adsorption structures of 4-NP on the Pt4/Co3O4-(111) and Pt4/Co3O4-(100) surfaces. The adsorption energy results presented in Fig. S21e indicate that the Pt4/Co3O4-(111) exhibits the weaker capability for facilitating the nitro group activation. Further, Fig. 5c reveals the adsorption and activation of H2 on the Pt4/Co3O4-(111) and Pt4/Co3O4-(100) surfaces, in which the energy barrier (1.17 eV) of H2 dissociation on Pt4/Co3O4-(111) is lower than that on Pt4/Co3O4-(100) (1.6 eV), implying that the activation of H2 by Pt4/Co3O4-(111) is easier. These results are consistent with the kinetics results. Furthermore, for hydrogen-atom migration from Pt to Co3O4 across the interface, the energy barrier on the Pt4/Co3O4-(111) (0.36 eV) is lower than that on the Pt4/Co3O4-(100) (0.42 eV), indicating kinetically more favourable hydrogen spillover on Co3O4-(111) (Fig. 5d and S22).
Based on the above results, the possible promotion mechanism is proposed (Fig. 5e). For the hydrogenation reaction, the activity of a catalyst is strongly related to the substrate activation ability, H2 activation ability, as well as active hydrogen spillover.40,41 With Pt/Co3O4-o and Pt/Co3O4-c as model catalysts, the kinetic experiments and DFT calculations illustrate that compared with Pt/Co3O4-c, the adsorption/activation of 4-NP on the Pt/Co3O4-o is weaker, indicating that the adsorption/activation of 4-NP is not the key factor contributing to the difference in activity in this reaction. However, the Pt/Co3O4-o has a stronger H2 dissociation ability and lower hydrogen-migration barrier. The accelerated hydrogen dissociation on electron-rich Pt sites, coupled with enhanced hydrogen spillover to the Co3O4 support, establishes a dynamic hydrogen supply chain that maximizes the utilization of active hydrogen species. The 4-NP adsorbed on Pt or near Pt-Co3O4 interfaces can continuously consume active hydrogen species, and the hydrogenation reaction can be promoted in a dynamic process.
Conclusions
In summary, a series of well-defined Pt/Co3O4-x (x = o, t, c) catalysts with tailored Co3O4 crystal facets have been successfully constructed for the hydrogenation of 4-nitrophenol. Owing to the crystal facet effect, the Pt nanoparticles in Pt/Co3O4-o are more negatively charged than those in Pt/Co3O4-c and Pt/Co3O4-t, and Pt/Co3O4-o exhibits the strongest hydrogen spillover capability. For the 4-nitrophenol (4-NP) hydrogenation reaction, the Pt/Co3O4-o has the best catalytic activity with a TOF value of 164.2 h−1. Detailed analyses reveal that the negatively charged Pt sites are favorable to the activation of hydrogen rather than the nitro group. Therefore, the enhanced catalytic activity of Pt/Co3O4-o is attributed to the promoted hydrogen activation and spillover. This work highlights crystal facet engineering of support to regulate the electronic structure and hydrogen spillover effect, which provides in-depth insight into the catalyst design and hydrogenation mechanism.Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. Hui Yun: investigation, validation, visualization, data curation, formal analysis, methodology, writing – original draft. Jiao Feng: visualization, writing – review & editing. Wanying Peng: visualization, writing – review & editing. Mi Xiong: conceptualization, funding acquisition, supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article has been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc09402j.Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (22302022), the Natural Science Foundation of Sichuan Province (24NSFSC4428), and the Cheng du University of Technology Teachers Development Research Fund (10912-KYQD2022-09565).References
- D. Wang and D. Astruc, The Golden Age of Transfer Hydrogenation, Chem. Rev., 2015, 115, 6621–6686 CrossRef CAS PubMed.
- A. V. Karim, Ł. Cichocki, C. Wang and G. Boczkaj, Advanced reduction processes (ARPs) based on catalytic hydrogenation for wastewater pollutants degradation - a special focus on process efficiency and mechanisms – a review, J. Cleaner Prod., 2025, 531, 146870 CrossRef CAS.
- S. Luo, Z. Liu, Y. Liu, E. Almatrafi, B. Shao, B. Song, C. Zhou, Y. Fu, M. He, Z. Zeng and G. Zeng, Versatile CMPs as platforms to support Ag nanocatalysts for nitrophenol hydrogenation in continuous flow-through process, Chem. Eng. J., 2022, 442, 136207 CrossRef CAS.
- B. Lai, Z. Chen, Y. Zhou, P. Yang, J. Wang and Z. Chen, Removal of high concentration p-nitrophenol in aqueous solution by zero valent iron with ultrasonic irradiation (US-ZVI), J. Hazard. Mater., 2013, 250–251, 220–228 CrossRef CAS PubMed.
- B. Lai, Y. H. Zhang, R. Li, Y. X. Zhou and J. Wang, Influence of operating temperature on the reduction of high concentration p-nitrophenol (PNP) by zero valent iron (ZVI), Chem. Eng. J., 2014, 249, 143–152 CrossRef CAS.
- Y. Ren, J. Li, L. Lai and B. Lai, Premagnetization enhancing the reactivity of Fe0/(passivated Fe0) system for high concentration p-nitrophenol removal in aqueous solution, Chemosphere, 2017, 197, 634–643 Search PubMed.
- Y. Zhang and J. Zhou, Synergistic catalysis by a hybrid nanostructure Pt catalyst for high-efficiency selective hydrogenation of nitroarenes, J. Catal., 2021, 395, 445–456 CrossRef CAS.
- Q. Zhang, J. Bu, J. Wang, C. Sun, D. Zhao, G. Sheng, X. Xie, M. Sun and L. Yu, Highly Efficient Hydrogenation of Nitrobenzene to Aniline over Pt/CeO2 Catalysts: The Shape Effect of the Support and Key Role of Additional Ce3+ Sites, ACS Catal., 2020, 10, 10350–10363 CrossRef CAS.
- Y. Wang, C. Wang, L. Wang, L. Wang and F. S. Xiao, Zeolite Fixed Metal Nanoparticles: New Perspective in Catalysis, Acc. Chem. Res., 2021, 54, 2579–2590 CrossRef CAS PubMed.
- G. Pacchioni and H. J. Freund, Controlling the charge state of supported nanoparticles in catalysis: lessons from model systems, Chem. Soc. Rev., 2018, 47, 8474–8502 RSC.
- T. W. van Deelen, C. Hernández Mejía and K. P. de Jong, Control of metal-support interactions in heterogeneous catalysts to enhance activity and selectivity, Nat. Catal., 2019, 2, 955–970 CrossRef CAS.
- M. Xiong, Y. Li, L. Sun, Q. Hu, Z. Lv, S. Xing, H. Yun, S. Zhang and Z. Gao, Magnetically recyclable bimetallic Pt-Co/CoO catalyst derived through hydrogen spillover for fast hydrogen release through ammonia borane hydrolysis, Surf. Interfaces, 2025, 62, 106242 CrossRef CAS.
- P. Zhang, T. Qin, D. Li, X. Q. Wu, Y. X. Ma, H. Q. Guo, J. Xiong, X. Liu, Z. Zhao, L. W. Chen, J. Liu and Y. C. Wei, Temperature-induced evolution of CuOx clusters in CuOx/TiO2 catalyst for boosting auto-exhaust oxidation, Appl. Catal. B Environ. Energy, 2025, 361, 124674 CrossRef CAS.
- Y. F. Li, T. Qin, Y. C. Wei, J. Xiong, P. Zhang, K. Z. Lai, H. J. Chi, X. Liu, L. W. Chen, X. L. Yu, Z. Zhao, L. N. Li and J. Liu, A single site ruthenium catalyst for robust soot oxidation without platinum or palladium, Nat. Commun., 2023, 14, 7149 CrossRef CAS PubMed.
- M. Ahmadi, H. Mistry and B. Roldan Cuenya, Tailoring the Catalytic Properties of Metal Nanoparticles via Support Interactions, J. Phys. Chem. Lett., 2016, 7, 3519–3533 CrossRef CAS PubMed.
- Z. Li, S. Ji, Y. Liu, X. Cao, S. Tian, Y. Chen, Z. Niu and Y. Li, Well-Defined Materials for Heterogeneous Catalysis: From Nanoparticles to Isolated Single-Atom Sites, Chem. Rev., 2019, 120, 623–682 CrossRef PubMed.
- L. Liu and A. Corma, Metal Catalysts for Heterogeneous Catalysis: From Single Atoms to Nanoclusters and Nanoparticles, Chem. Rev., 2018, 118, 4981–5079 CrossRef CAS PubMed.
- X. Liu, X. Wang, S. Zhen, G. Sun, C. Pei, Z. J. Zhao and J. Gong, Support stabilized PtCu single-atom alloys for propane dehydrogenation, Chem. Sci., 2022, 13, 9537–9543 RSC.
- X. Huang, C. Dang, H. Yu, H. Wang and F. Peng, Morphology Effect of Ir/La2O2CO3 Nanorods with Selectively Exposed {110} Facets in Catalytic Steam Reforming of Glycerol, ACS Catal., 2015, 5, 1155–1163 CrossRef CAS.
- Z. Hu, Z. Zou, A. Xie, C. Chen, X. Zhu, Y. Zhang, H. Zhang, H. Zhao and G. Wang, Crystal plane effect of ceria on supported copper catalyst for liquid-phase hydrogenation of unsaturated aldehyde, J. Colloid Interface Sci., 2021, 596, 34–43 CrossRef CAS PubMed.
- P. Zhang, J. Xiong, Y. C. Wei, Y. F. Li, Y. L. Zhang, J. J. Tang, W. Y. Song, Z. Zhao and J. Liu, Exposed {001} facet of anatase TiO2 nanocrystals in Ag/TiO2 catalysts for boosting catalytic soot combustion: The facet-dependent activity, J. Catal., 2021, 398, 109–122 CrossRef CAS.
- S. Wang, C. Liu, W. Hao, Y. Zhuang, J. Chen, X. Zhu, L. Wang, X. Niu, J. Mao, D. Ma and Q. Zhao, Structural evolution of metal single-atoms and clusters in catalysis: Which are the active sites under operative conditions?, Chem. Sci., 2025, 16, 6203–6218 RSC.
- M. Cargnello, V. V. T. Doan-Nguyen, T. R. Gordon, R. E. Diaz, E. A. Stach, R. J. Gorte, P. Fornasiero and C. B. Murray, Control of Metal Nanocrystal Size Reveals Metal-Support Interface Role for Ceria Catalysts, Science, 2013, 341, 771–773 CrossRef CAS PubMed.
- G. Huang, Q. H. Yang, Q. Xu, S. H. Yu and H. L. Jiang, Polydimethylsiloxane Coating for a Palladium/MOF Composite: Highly Improved Catalytic Performance by Surface Hydrophobization, Angew. Chem., Int. Ed., 2016, 55, 7379–7383 CrossRef CAS PubMed.
- Q. Fu and T. Wagner, Interaction of nanostructured metal overlayers with oxide surfaces, Surf. Sci. Rep., 2007, 62, 431–498 CrossRef CAS.
- J. F. Zhou, B. Peng, M. Ding, B. Q. Shan, Y. S. Zhu, L. Bonneviot, P. Wu and K. Zhang, The nature of crystal facet effect of TiO2-supported Pd/Pt catalysts on selective hydrogenation of cinnamaldehyde: electron transfer process promoted by interfacial oxygen species, Phys. Chem. Chem. Phys., 2024, 26, 18854–18864 RSC.
- M. L. Gao, L. Li, Z. X. Sun, J. R. Li and H. L. Jiang, Facet Engineering of a Metal-Organic Framework Support Modulates the Microenvironment of Palladium Nanoparticles for Selective Hydrogenation, Angew. Chem., Int. Ed., 2022, 61, e202211216 CrossRef CAS PubMed.
- L. Jiang, K. Liu, S. F. Hung, L. Zhou, R. Qin, Q. Zhang, P. Liu, L. Gu, H. M. Chen, G. Fu and N. Zheng, Facet engineering accelerates spillover hydrogenation on highly diluted metal nanocatalysts, Nat. Nanotechnol., 2020, 15, 848–854 CrossRef CAS PubMed.
- Z. Cai, Y. Bi, E. Hu, W. Liu, N. Dwarica, Y. Tian, X. Li, Y. Kuang, Y. Li, X. Q. Yang, H. Wang and X. Sun, Single-Crystalline Ultrathin Co3O4 Nanosheets with Massive Vacancy Defects for Enhanced Electrocatalysis, Adv. Energy Mater., 2017, 8, 1701694 CrossRef.
- Z. Li, T. He, D. Matsumura, S. Miao, A. Wu, L. Liu, G. Wu and P. Chen, Atomically Dispersed Pt on the Surface of Ni Particles: Synthesis and Catalytic Function in Hydrogen Generation from Aqueous Ammonia–Borane, ACS Catal., 2017, 7, 6762–6769 CrossRef CAS.
- H. Jeong, D. Shin, B. S. Kim, J. Bae, S. Shin, C. Choe, J. W. Han and H. Lee, Controlling the Oxidation State of Pt Single Atoms for Maximizing Catalytic Activity, Angew. Chem., Int. Ed., 2020, 59, 20691–20696 CrossRef CAS PubMed.
- J. Zhang, Y. Pan, D. Feng, L. Cui, S. Zhao, J. Hu, S. Wang and Y. Qin, Mechanistic insight into the synergy between platinum single atom and cluster dual active sites boosting photocatalytic hydrogen evolution, Adv. Mater., 2023, 35, 2300902 CrossRef CAS PubMed.
- T. Zhang, J. Jin, J. Chen, Y. Fang, X. Han, J. Chen, Y. Li, Y. Wang, J. Liu and L. Wang, Pinpointing the axial ligand effect on platinum single-atom-catalyst towards efficient alkaline hydrogen evolution reaction, Nat. Commun., 2022, 13, 6875 CrossRef CAS PubMed.
- J. Zhang, W. Chen, H. Ge, C. Chen, W. Yan, Z. Gao, J. Gan, B. Zhang, X. Duan and Y. Qin, Synergistic effects in atomic-layer-deposited PtCox/CNTs catalysts enhancing hydrolytic dehydrogenation of ammonia borane, Appl. Catal. B Environ. Energy, 2018, 235, 256–263 CrossRef CAS.
- B. Ouyang, S. Xiong, Y. Zhang, B. Liu and J. Li, The study of morphology effect of Pt/Co3O4 catalysts for higher alcohol synthesis from CO2 hydrogenation, Appl. Catal., A, 2017, 543, 189–195 CrossRef CAS.
- H. Li, M. Abdelgaid, J. R. Paudel, N. P. Holzapfel, V. Augustyn, J. R. McKone, G. Mpourmpakis and E. J. Crumlin, Operando unveiling of hydrogen spillover mechanisms on tungsten oxide surfaces, J. Am. Chem. Soc., 2025, 147, 6472–6479 CrossRef CAS PubMed.
- Y. Liu, R. Zhang, L. Lin, Y. Wang, C. Liu, R. Mu and Q. Fu, Direct observation of accelerating hydrogen spillover via surface-lattice-confinement effect, Nat. Commun., 2023, 14, 613 CrossRef CAS PubMed.
- Z. Cheng, B. He and L. Zhou, A general one-step approach for in situ decoration of MoS2 nanosheets with inorganic nanoparticles, J. Mater. Chem. A, 2015, 3, 1042–1048 RSC.
- H. Feng, Y. Yang, Y. Niu, L. Wang, P. Yin, S. Hong, B. Zhang, X. Zhang and M. Wei, Highly-efficient RuNi single-atom alloy catalysts toward chemoselective hydrogenation of nitroarenes, Nat. Commun., 2022, 13, 3188 CrossRef PubMed.
- Z. Zhao, X. Li, X. Liu, H. Gao, A. Jia, S. Xie, X. Song, X. Liu, F. Yang and Q. Yang, Pt/Fe-TiO2-Catalyzed Selective Carbonyl Hydrogenation: Fe-Promoted Hydrogen Spillover, ACS Catal., 2024, 14, 4478–4488 CrossRef CAS.
- L. Xie, J. Liang, L. Jiang and W. Huang, Effects of oxygen vacancies on hydrogenation efficiency by spillover in catalysts, Chem. Sci., 2025, 16, 3408–3429 RSC.
| This journal is © The Royal Society of Chemistry 2026 |