Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Interplay between ligand field strength and the nephelauxetic effect in chromium(III) complexes with anionic amido ligands
P.
Yaltseva†
 a,
B.
Wittwer†
b,
D.
Leitner
b,
F. R.
Neururer
b,
F.
Tambornino
a,
B.
Wittwer†
b,
D.
Leitner
b,
F. R.
Neururer
b,
F.
Tambornino
 c,
A.
Schmidt
a,
D.
Munz
c,
A.
Schmidt
a,
D.
Munz
 *d,
O. S.
Wenger
*d,
O. S.
Wenger
 *a and
S.
Hohloch
*a and
S.
Hohloch
 *b
*b
aDepartment of Chemistry, University of Basel, St. Johanns-Ring 19, 4056 Basel, Switzerland. E-mail: oliver.wenger@unibas.ch
bDepartment of General, Inorganic and Theoretical Chemistry, University of Innsbruck, Innrain 80–82, 6020 Innsbruck, Austria. E-mail: Stephan.Hohloch@uibk.ac.at
cDepartment of Chemistry, Philipps-Universität Marburg, Hans-Meerwein-Strasse 4, 35032 Marburg, Germany
dCoordination Chemistry, Saarland University, Campus C4.1, 66123 Saarbrücken, Germany. E-mail: dominik.munz@uni-saarland.de
First published on 19th February 2026
Abstract
Incorporation of the nephelauxetic effect into ligand design enabled red-shifting of spin-flip transitions of CrIII and MnIV complexes into the near-infrared region. Using carbazolide complexes as a model, we present a strategy for tuning the ratio of ligand field strength to the Racah parameter B by combining a covalent carbazolide core with variable σ-donor ligand “side arms.” Substitution of pyridine, as in [Cr(Lpy)2]+ ([Lpy]− = 3,6-di-tert-butyl-1,8-di(pyridin-2-yl)carbazol-9-ide), with stronger σ-donors such as N-heterocyclic or mesoionic carbenes in [Cr(LNHC)2]+ or [Cr(LMIC)2]+ ([LNHC]− = 3,6-di-tert-butyl-1,8-bis(imidazolin-2-yliden-1-yl)carbazolide and [LMIC]− = 3,6-di-tert-butyl-1,8-bis(4,5,6,7-tetrahydro-2H-[1,2,3]triazolo[1,5-a]pyridin-2-yl)-carbazol-9-ide) increased the ligand field strength from 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 to 24
500 to 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1, with only a modest rise in B from 550 to 600 cm−1. This balance favors near-infrared spin-flip transitions while extending their excited-state lifetimes. Despite these advances, carbazolide-based ligands exhibit also drawbacks, including low-lying charge-transfer states and geometric distortions, which limit lifetimes and prevent emission, contrasting with other near-infrared-emissive CrIII systems. Additionally, we demonstrate an approach for estimating energies of dark, low-energy spin-flip states in CrIII complexes via photoinduced electron transfer and Rehm–Weller analysis. Our results offer guidance on balancing ligand field strength and metal–ligand bond covalency to optimize the photophysical and photochemical properties of first-row transition metal complexes.
400 cm−1, with only a modest rise in B from 550 to 600 cm−1. This balance favors near-infrared spin-flip transitions while extending their excited-state lifetimes. Despite these advances, carbazolide-based ligands exhibit also drawbacks, including low-lying charge-transfer states and geometric distortions, which limit lifetimes and prevent emission, contrasting with other near-infrared-emissive CrIII systems. Additionally, we demonstrate an approach for estimating energies of dark, low-energy spin-flip states in CrIII complexes via photoinduced electron transfer and Rehm–Weller analysis. Our results offer guidance on balancing ligand field strength and metal–ligand bond covalency to optimize the photophysical and photochemical properties of first-row transition metal complexes.
Introduction
The 3d3 electronic configuration is particularly significant in first-row transition metals, such as in CrIII and MnIV, as it gives rise to metal-centered ‘spin-flip’ doublet excited states (2MC), characterized by minimal structural distortion and prolonged lifetimes.1 Octahedral CrIII α,α′-diimine and α,α′,α″-triimine complexes, often referred to as molecular rubies, have been known for decades for their red-visible emission from these 2MC excited states (2T1/2E in octahedral geometry), with lifetimes reaching up to milliseconds.2–4 These spin-flip transitions occur within the t2g metal orbital set, leading to only weak excited state distortion relative to the ground state and consequently slow deactivation rates. This behavior stands in stark contrast to that of triplet and quintet metal-centered states in 3d6 complexes (e.g. CoIII, FeII, Cr0, MnI), where the population of an antibonding eg metal orbital results in significant geometric distortion and faster excited-state deactivation.5,6In CrIII complexes, excited-state lifetimes and emission quantum yields can be enhanced by suppressing the repopulation of Jahn–Teller distorted quartet metal-centered excited states (4MC) via back-intersystem crossing from the luminescent 2MC states.7 A key strategy to achieve this relies on increasing the ligand field strength (10 Dq), thereby widening the energy gap between the quartet and doublet metal-centered manifolds.7,8 One effective approach to enhance the ligand field strength is the optimization of the chelating ligand bite angles towards an ideal octahedral coordination environment.8,9 The effects of the bite-angle optimization have been extensively studied in RuII complexes,9,10 and were later applied to CrIII polypyridines and CoIII complexes to fine-tune their photophysical properties.3,4,11–13 These modifications have expanded their application potential in various fields, including photon upconversion,14 photoredox catalysis,15,16 circularly polarized luminescence emitters,11,17 and ratiometric pH optical sensors.12,18
According to the Tanabe–Sugano formalism (Fig. 1c),19–21 an increase in 10 Dq effectively destabilizes the 4MC states (among which 4T2 is most relevant) while having only a weak effect on the energies of the lowest 2MC states (2E, 2T1). This underscores a fundamental limitation of ligand design strategies that rely solely on the polypyridine systems.22,23 Consequently, some of the research focus in recent years has shifted toward gaining control over the 2MC excited-state energies by exploring novel ligand frameworks.22,24–27 Shifting the spin-forbidden electronic transitions further into the near-infrared-II region (NIR-II, between 1000 and 1700 nm) seems particularly appealing, as it could broaden CrIII applications further to, for example, in vivo luminescent cell imaging in biomedicine.28
 | ||
| Fig. 1 (a) Complex [Cr(Lpy)2]+ reported previously by our group,21 and complexes [Cr(LNHC)2]+ and [Cr(LMIC)2]+ investigated in this work; (b) schematic energy diagram showing the influence of the main electronic effects in the complex series; (c) Tanabe–Sugano diagram for octahedral d3 complexes. | ||
In an octahedral geometry, the energy of 2E excited state is primarily governed by d–d electronic repulsion, described by the Racah parameters B and C.19,20 This d–d electronic repulsion is influenced by the nephelauxetic effect, which arises from spatial extension of the d-orbitals facilitated by the ligand.22 This can reduce the Racah parameters compared to the free metal ion. A practical approach to modulating the nephelauxetic effect is by altering the covalency of metal–ligand bonds, as the Racah parameter B inversely correlates with it. In photoactive CrIII complexes, this concept was first explored by us via introducing anionic amido units in the axial positions of the ligand scaffold in [Cr(Lpy)2]+ ([Lpy]− = 3,6-di-tert-butyl-1,8-di(pyridin-2-yl)carbazol-9-ide, Fig. 1a).24 The presence of stronger covalent Cr–Namido bonds reduced B to 550 cm−1, compared to the 700–800 cm−1 range observed for polypyridine CrIII complexes.3,12 This, in turn, lowered the energy of the doublet excited states, resulting in emission at 1067 nm (∼1.2 eV) at 77 K in a glass matrix. Subsequent studies by other investigators reported a series of compounds, incorporating anionic amido and cyclometalated moieties, with the B values ranging from 550 to 670 cm−1, and spin-flip state emission observed between 910–980 nm (1.36–1.27 eV) in solution at room temperature.25,29–31
Introducing new anionic ligand scaffolds presents significant challenges, one of which relates to the empirical energy gap law or Marcus inverted-region behavior.32 This principle predicts that lowering the energy of a nested doublet excited state relative to the quartet ground state increases its deactivation rate. Indeed, studies on amido-based complexes [Cr(bpiR)2]+ ([bpiR]− = 1,3-bis((2-R-pyridin-2-yl)imino)isoindolin-2-ide or 1,3-bis((4-R-pyridin-2-yl)-imino)isoindolin-2-ide, R = H, Me, OMe, NMe2), with electronically diverse substituents have confirmed that CrIII excited-state dynamics follow this principle.31 The introduction of the electron-donating substituents at the ortho- or para-positions of the pyridine rings raises the energy of the photoactive doublet excited state and slows the radiative deactivation rates. Notably, in these complexes, the lowest excited state is a mixed 2MC/LMCT (MC = metal-centered, LMCT = ligand-to-metal charge transfer) state. In this specific scenario significant changes in the doublet excited-state energy can be rationalized solely by modifying substituents on the ligand backbone, without drastically changing the metal–ligand bonding situation.22
Another key challenge in optimizing photophysical properties is that reducing the Racah parameter B often comes at the expense of weakening the ligand field (10 Dq), rendering it difficult to fine-tune excited-state behavior. For instance, in the complexes [Cr(Lpy)2]+,24 Cr(o-CH2NMe2-Ph)3 (o-CH2NMe2-Ph = 4-(tert-butyl)-2-((dimethylamino)methyl)benzen-1-ide) and Cr(o-CH2P(Ph)2-Ph)3 (o-CH2P(Ph)2-Ph = 2-((diphenylphosphaneyl)methyl)-benzen-1-ide),29 the introduction of anionic π-donor ligands drastically destabilizes the t2g orbitals, leading to a reduction in 10 Dq compared to some polypyridine-based CrIII complexes.3,12 This not only results in shortening of the excited-state lifetime, but also prevents room-temperature emission in solution. To overcome this limitation, maximizing 10 Dq while keeping B values sufficiently low appears to be a reasonable strategy for optimizing 2MC state properties and minimizing non-radiative energy loss. A particularly well-suited example among known NIR-photoactive complexes is fac-Cr(ppy)3,30 which achieves a balance between these parameters. Its nearly ideal octahedral coordination and carbanionic ligand moieties yield a high 10 Dq of 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 320 cm−1 and a B parameter of 630 cm−1, resulting in a spin-flip doublet excited state with a ∼9 µs lifetime and 910 nm room-temperature emission in solution.
320 cm−1 and a B parameter of 630 cm−1, resulting in a spin-flip doublet excited state with a ∼9 µs lifetime and 910 nm room-temperature emission in solution.
In this work, we aimed to explore the interplay between B and 10 Dq parameters using tridentate carbazolide ligand systems, structurally related to the previously reported [Cr(Lpy)2]+,24 to gain deeper insight into their impact on excited-state dynamics. Pyridine in the ligand scaffold was replaced with mesoionic carbene (MIC) or N-heterocyclic carbene (NHC) moieties in [Cr(LMIC)2]+ ([LMIC]− = 3,6-di-tert-butyl-1,8-bis(4,5,6,7-tetrahydro-2H-[1,2,3]triazolo[1,5-a]pyridin-2-yl)carbazol-9-ide) and [Cr(LNHC)2]+ ([LNHC]− = 3,6-di-tert-butyl-1,8-bis(imidazolin-2-yliden-1-yl)carbazolide) respectively, keeping the central carbazolide unit intact. The strong σ-donating properties of MIC and NHC binding motifs33 facilitate the energetic destabilization of the antibonding eg metal orbitals, thereby increasing the ligand field splitting. As a result, these types of ligands are widely implemented in photoactive first row-transition metal complexes.34–36 However, to date, the photophysical properties of only two CrIII carbene complexes—[Cr(ImPyIm)2]3+ (ImPyIm = 2,6-bis(imidazole-2-ylidene)pyridine) and [Cr(ImPy)3]3+ (ImPy = 2-imidazolylpyridine)—have been fully characterized.37 Interestingly, those two complexes exhibit drastically different behaviors, while [Cr(ImPy)3]3+ displays a long-lived (∼13 µs) and emissive (room temperature, solution) excited state, [Cr(ImPyIm)2]3+ manifests a dark state with a lifetime of less than 1 ns.37 This strong difference is speculated to arise from a lower-lying 4CT/MC manifold in [Cr(ImPyIm)2]3+, effectively deactivating 2MC via back-intersystem crossing.
Both ligand systems [LNHC]− and [LMIC]− have been previously reported in the literature.38 In particular, the [LMIC]− ligand was utilized by some of us in the [Mn(LMIC)2]2+ complex,39 as the high oxidation state of MnIV requires electron-rich ligands, enabling the isolation of the complex as redox-stable species.35 Isoelectronic to CrIII, MnIV complexes feature greater ligand field splitting and metal–ligand bond covalency due to the higher effective nuclear charge of the metal center. Notably, lower-energy t2g orbitals render 4LMCT/2LLCT (LLCT = ligand-to-ligand charge transfer) states easier accessible, and in some cases the 2LMCT state becomes the lowest photoactive state.40–43 Among the known examples, [Mn(dgpy)2]4+ (dgpy = 2,6-diguanidylpyridine) and exceptionally photorobust [Mn(PhB(MeIm)3)2]2+ ([PhB(MeIm)3]− = tris(3-methylimidazolin-2-ylidene)phenylborate) have demonstrated 2LMCT states suitable for photocatalysis.40–43 We believe that the increased ligand field splitting in [Cr(LMIC)2]+ and [Cr(LNHC)2]+ complexes could ultimately align their excited-state relaxation patterns more closely with those of MnIV, and anticipate that charge-transfer states will emerge to play an important role in the future.
Results and discussion
Synthesis and characterization
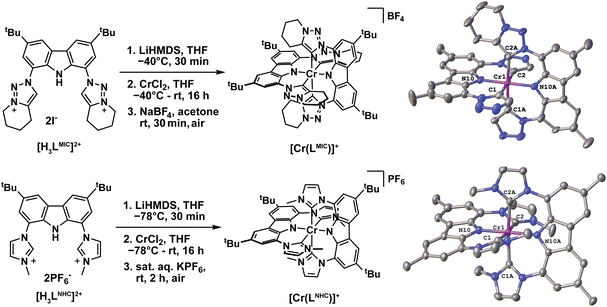
Complexes [Cr(LMIC)2]+ and [Cr(LNHC)2]+ were synthesized following a strategy similar to that used for the recently reported MnIV complex (Fig. 2).39 The pro-ligands [H3LMIC][I]2 or [H3LNHC][I]2 were deprotonated in situ in THF using lithium bis(trimethylsilyl)amide (LiHMDS), followed by addition of this solution to a suspension of chromium(II) chloride (CrCl2) in THF. Subsequent oxidation of the complexes was achieved by aqueous work-up and aerobic salt metathesis using NaBF4 or KPF6 respectively. After purification by column chromatography and/or recrystallization, the target compounds were obtained in moderate yields of 24–50% (SI). Notably, the NHC complex ([Cr(LNHC)2]+ was also recently reported by Kunz, Heinze and co-worker and its excited state dynamics were thoroughly investigated.44Single orange/red crystals of X-ray diffraction quality were obtained via vapor diffusion at room temperature from DCM/hexane ([Cr(LMIC)2]BF4) or acetone/hexane ([Cr(LNHC)2]BF4). The single crystal quality of the [BF4]− salts was found to be the best, as other counterions lead to more complicated disorder and twinning effects. Both complexes crystallize in the monoclinic system with space group P21/c with a positional disorder of the [BF4]− anions over three ([Cr(LMIC)2]BF4) or two ([Cr(LNHC)2]BF4) positions and three disordered hexane ([Cr(LMIC)2]BF4) or 1.7 disordered acetone ([Cr(LNHC)2]BF4) molecules (Fig. S53 and S54). The CrIII center is six-fold coordinated in a distorted octahedral coordination sphere by four imidazolylidene/triazolylidene units and the two amide donors. The Ccarbene–Cr–Ccarbene angles are 176.38(11)°/173.00(12)° and 175.62(12)°/172.57(12)° for C1–Cr1–C2 and C1A–Cr1–C2A, while the Namide–Cr–Namide angle N10–Cr1–N10A is found to be 179.90(13)°/178.85(11)° in [Cr(LMIC)2]BF4/[Cr(LNHC)2]BF4, respectively (Fig. 2, right). The values display that—despite the slightly higher steric bulk of the cyclohexyl ring in [Cr(LMIC)2]BF4 compared to [Cr(LNHC)2]BF4—the latter is substantially more distorted. This is also visible along the Namido–Cr–Namido axis showing the two ligands to be almost perpendicular in [Cr(LMIC)2]BF4 (88.4(1)°), while the imidazolylidene substituents are strongly tilted in [Cr(LNHC)2]BF4 hence resulting in a smaller angle between the carbazole planes (61.89(1)°). We propose that his distortion is caused by unfavorable C–HImidazole⋯C–Hcarbazole repulsions, causing the imidazolylidene moieties to rotate more strongly out of plane compared to the triazolylidene. In the latter, the potential formation of favorable C–Hcarbazole⋯Ntriazole interactions diminishes this rotation (Fig. 2, right). These unfavorable interactions are even more pronounced in [Cr(Lpy)2]+ with the six-membered pyridine donors.24
In accordance with enhanced covalency, the Cr–Namido distances of 2.026(2)/2.002(2) Å (Cr1–N10) and 2.025(2)/2.004(2) Å (Cr1–N10A) are shortened compared to the Cr–Ccarbene distances of 2.136(3)–2.144(3) Å/2.127(3)–2.160(3) Å in [Cr(LMIC)2]BF4/[Cr(LNHC)2]BF4. This observation is in line with axial compression distortion along the Namido–Cr–Namido axis, previously reported in [Cr(Lpy)2]+.24 Notably, the metal–donor distances in [Cr(LMIC)2]BF4 are substantially larger, compared to the isoelectronic MnIV triazolylidene complex [Mn(LMIC)2]2+ previously reported by some of us (M–Naverage 1.938(3); M–Caverage 2.081(3) Å).39 Additionally, the metal–carbene distances in [Cr(LMIC)2]BF4 are longer compared to previous examples of heteroleptic and homoleptic CrIII NHC complexes in the literature, e.g., by Gibson and Steed (2.087(6)–2.120(6) Å)45 or by Scattergood et al. (2.093(4)–2.106(4) Å).37 We rationalize this bond elongation by the steric pressure of the cyclohexyl groups in the homoleptic [Cr(LMIC)2]BF4 complex. Further information on the structural parameters and data of complexes [Cr(LMIC)2]BF4 and [Cr(LNHC)2]BF4 can be found in the SI (Tables S2 and S3).
Evans NMR spectroscopy of the complexes revealed strong paramagnetism (Fig. S1–S8 displaying a magnetic moment of 3.91 µB for [Cr(LMIC)2]BF4 (Fig. S3) and 3.85 µB for [Cr(LNHC)2]BF4 (Fig. S7)), consistent with the presence of three unpaired electrons (expected spin-only value = 3.87 µB) and a high-spin d3 configured CrIII center. Additionally, [Cr(LMIC)2]BF4 and [Cr(LNHC)2]X (X = [PF6]− or [BF4]−) were characterized by IR spectroscopy, high-resolution mass-spectrometry and elemental analysis (see SI for further information).
Electrochemistry
Cyclic voltammetry studies in acetonitrile at room temperature revealed the presence of three reversible redox events for both complexes (Fig. 3 and Table 1). Two reversible oxidations are observed at 0.11 V and 0.48 V vs. Fc/[Fc]+ for [Cr(LMIC)2]+, and at 0.08 V and 0.50 V![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 44vs. Fc/[Fc]+ for [Cr(LNHC)2]+. Given the proximity of the two redox processes and our previous investigations on the manganese analogue of [Mn(LMIC)2]2+,39 we propose that both oxidations are ligand-centered. Compared to [Cr(Lpy)2]+ (0.46 V and 0.78 V vs. Fc/[Fc]+, Fig. 3) the oxidations in [Cr(LMIC)2]+ and [Cr(LNHC)2]+ are anodically shifted, which is in agreement with the stronger σ-donor and weaker π-acceptor properties of NHC and MIC moieties relative to neutral N-donor ligands such as pyridines. Additionally, reversible reduction processes were recorded at −2.15 V and −1.91 V
44vs. Fc/[Fc]+ for [Cr(LNHC)2]+. Given the proximity of the two redox processes and our previous investigations on the manganese analogue of [Mn(LMIC)2]2+,39 we propose that both oxidations are ligand-centered. Compared to [Cr(Lpy)2]+ (0.46 V and 0.78 V vs. Fc/[Fc]+, Fig. 3) the oxidations in [Cr(LMIC)2]+ and [Cr(LNHC)2]+ are anodically shifted, which is in agreement with the stronger σ-donor and weaker π-acceptor properties of NHC and MIC moieties relative to neutral N-donor ligands such as pyridines. Additionally, reversible reduction processes were recorded at −2.15 V and −1.91 V![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 44vs. Fc/[Fc]+ for [Cr(LMIC)2]+ and [Cr(LNHC)2]+, respectively, while [Cr(Lpy)2]+ shows a reduction event at −1.54 V vs. Fc/[Fc]+. The strong anodic shift of these values aligns with the σ-donor capacity of the ligand series and tentatively suggests—in combination with the pronounced shift compared to the previously reported MnIV complex [Mn(LMIC)2]2+—a metal-centered reduction process.39 However, spectroelectro-EPR spectroscopic measurements (Fig. S55 and S56) and DFT calculations (vide infra) rather indicate a ligand-centered process instead of the expected metal-centered reduction. Notably, all attempts to isolate any reduced or oxidized materials failed and EPR measurements after reduction further indicate instability of resulting complex (Fig. S55 and S56). For the oxidation, a fast colour change of the solutions is observed after addition of the oxidants, however the solutions quickly convert back to their original colour, indicating photo-instability of oxidizes species. This observation is in line with the ligand centered redox-process in the corresponding MnIV complex [Mn(LMIC)2]2+ which also rapidly decomposed at room temperature.39
44vs. Fc/[Fc]+ for [Cr(LMIC)2]+ and [Cr(LNHC)2]+, respectively, while [Cr(Lpy)2]+ shows a reduction event at −1.54 V vs. Fc/[Fc]+. The strong anodic shift of these values aligns with the σ-donor capacity of the ligand series and tentatively suggests—in combination with the pronounced shift compared to the previously reported MnIV complex [Mn(LMIC)2]2+—a metal-centered reduction process.39 However, spectroelectro-EPR spectroscopic measurements (Fig. S55 and S56) and DFT calculations (vide infra) rather indicate a ligand-centered process instead of the expected metal-centered reduction. Notably, all attempts to isolate any reduced or oxidized materials failed and EPR measurements after reduction further indicate instability of resulting complex (Fig. S55 and S56). For the oxidation, a fast colour change of the solutions is observed after addition of the oxidants, however the solutions quickly convert back to their original colour, indicating photo-instability of oxidizes species. This observation is in line with the ligand centered redox-process in the corresponding MnIV complex [Mn(LMIC)2]2+ which also rapidly decomposed at room temperature.39
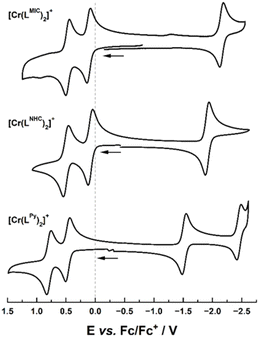 | ||
| Fig. 3 Cyclic voltammograms of 0.1 mM solutions of [Cr(LMIC)2]+ (top), [Cr(LNHC)2]+ (middle) and [Cr(LPy)2]+ (bottom) in 0.2 M NBu4PF6 in MeCN at 298 K. Scan rate: 100 mV s−1. | ||
To gain further information on the electronic structure of the native, mono-oxidized and mono-reduced complexes [Cr(LMIC)2]+, [Cr(LMIC)2]2+ and [Cr(LMIC)2]0 as well as its NHC and pyridine congeners [Cr(LNHC)2]+ and [Cr(LPy)2]+, computational investigations were performed at the density functional theory (DFT) level of theory. The DFT calculations support ligand-based oxidation as well as ligand reduction of [Cr(LMIC)2]+ (Tables S6–S7 and Fig. S57–S58).
Ground- and excited-state properties: modelling and spectroscopy
Comparative ab initio NEVPT2/CASSCF calculations were performed using the solid-state structural parameters of complexes [Cr(LMIC)2]+ (Fig. 4 and S62–S64), [Cr(LNHC)2]+ (Fig. S66), and [Cr(Lpy)2]+ (Fig. S67). Active spaces of saCASSCF(7,11) were chosen for [Cr(LMIC)2]+ (Fig. S63) and [Cr(Lpy)2]+ (Fig. S67), comprising the five 3d orbitals in combination with six ligand-based orbitals, thereof two formally occupied (carbazolide π-donor functionality) as well as four unoccupied π*-orbitals delocalized mainly across the carbazolides. For [Cr(LNHC)2]+, saCASSCF(7,10) with one π*-orbital less in the active space was required, as the considerably π-acidity of the NHCs led to otherwise difficult-to-converge wavefunctions (Fig. S66). Despite that the carbene complexes show near-ideal octahedral coordination geometries with orthogonal/coplanar ligand-π-systems (vide supra) and that the pyridine-congener is significantly distorted (e.g., dihedral angle between the two pyridine ligands ∠C–Npy–Npy–C = 34°), the electronic structures of all complexes are similar. Fig. 4 depicts the computed molecular orbital diagram for [Cr(LMIC)2]+. The computations confirm that an idealized octahedral ligand field with a 3 + 2d-orbital splitting pattern is appropriate to understand the electronic structure, and that the covalency in the bonds with the donor ligands is moderate.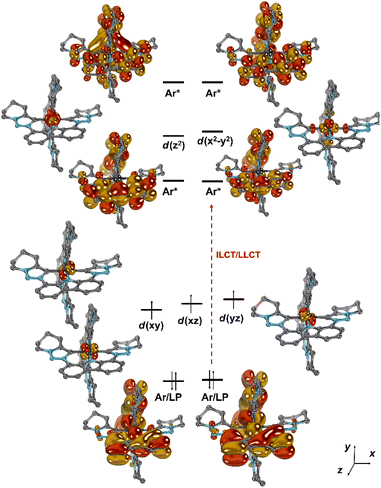 | ||
| Fig. 4 Frontier orbitals in [Cr(LMIC)2]+ according to CASSCF(7,11); the tBu groups have been truncated by H-atoms to save computation time. | ||
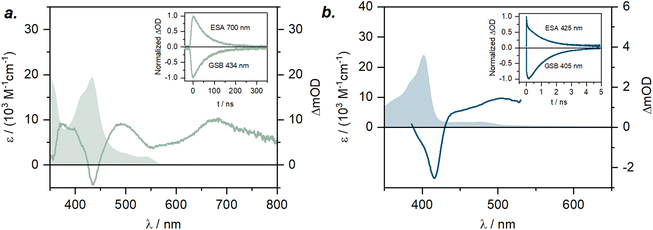
Notably, the two energetically lowest π*-orbitals of the carbazolido ligands are lower in energy than the d(x2–y2) and d(z2) orbitals. Hence, four low-intensity (starting at 525 nm, 2.36 eV, Q1–Q4; Table S10) and four high-intensity (starting at 460 nm, 2.71 eV, Q5–Q8) quartet mixed intra-ligand charge transfer (4ILCT) and ligand-to-ligand charge transfer (4LLCT) bands with predominant 4ILCT character are predicted. The weak d–d (MC) transitions (Q9–Q15) are predicted to occur in the energy range of 405–295 nm (4T2: 3.03 eV) and hence cannot be experimentally observed due to superposition by the intense charge transfer bands in the same spectral region. Corresponding vertical, that is still referring to the structural parameters of the quartet ground state, metal-centered doublet excited states are found at an energy range of 730 nm to 480 nm. The two energetically lowest D1 (1.70 eV, 730 nm) and D2 (1.75 eV, 710 nm) excited states represent the 2E and 2T1 spin-flip states (Fig. 5). Indeed, in the experimental UV-vis absorption spectrum of [Cr(LMIC)2]+ in acetonitrile, the most prominent band at 432 nm with a molar absorptivity (ε) of 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 M−1 cm−1 is assigned to 4LLCT/ILCT (vide supra). Two weaker intensity bands at 500 and 543 nm with ε of 2500 M−1 cm−1 and 1800 M−1 cm−1 similarly exhibit substantial charge transfer character.
300 M−1 cm−1 is assigned to 4LLCT/ILCT (vide supra). Two weaker intensity bands at 500 and 543 nm with ε of 2500 M−1 cm−1 and 1800 M−1 cm−1 similarly exhibit substantial charge transfer character.
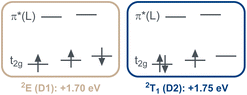 | ||
| Fig. 5 Electronic nature of the two energetically lowest doublet excited states D1 and D2 in [Cr(LMIC)2]+. | ||
Upon moving to [Cr(LNHC)2]+, the energies of the ligand as well as the metal-centered states decrease in energy (Fig. 6). The lowest quartet state Q1 excited state appears at 690 nm (1.80 eV), and is anticipated to be of CT (charge-transfer) character akin to [Cr(LMIC)2]+. The 4T2 quartet state is predicted at 420 nm (Q3, 2.94 eV), and the 2T1 and 2E doublet states at 860 nm (D1, 1.44 eV) and 685 nm (D2, 1.80 eV), respectively.
In agreement with the computational data, the ground state UV-vis electronic absorption spectrum of [Cr(LNHC)2]+ in acetonitrile (Fig. 7b) reveals an intense band at 402 nm with a molar absorptivity of 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1, which can be attributed to 4ILCT/LLCT transitions, while the bands at 450 and 477 nm (ε ∼2000 M−1 cm−1) have mainly 4LMCT character. In case of the literature-known [Cr(Lpy)2]+, the lowest-energy excited quartet state is not any more CT, but an MC state (Q1, 4T2, 2.17 eV). The Q3 state at 2.25 eV is associated with the first ILCT/LLCT transition, and the 2T1 and 2E states are found at 685 nm (1.80 eV) and 680 nm (1.82 eV), respectively. As the 4MC states are predicted at 24
000 M−1 cm−1, which can be attributed to 4ILCT/LLCT transitions, while the bands at 450 and 477 nm (ε ∼2000 M−1 cm−1) have mainly 4LMCT character. In case of the literature-known [Cr(Lpy)2]+, the lowest-energy excited quartet state is not any more CT, but an MC state (Q1, 4T2, 2.17 eV). The Q3 state at 2.25 eV is associated with the first ILCT/LLCT transition, and the 2T1 and 2E states are found at 685 nm (1.80 eV) and 680 nm (1.82 eV), respectively. As the 4MC states are predicted at 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1 (409 nm, 3.03 eV) in [Cr(LMIC)2]+ and at 23
400 cm−1 (409 nm, 3.03 eV) in [Cr(LMIC)2]+ and at 23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 cm−1 (421 nm, 2.94 eV) in [Cr(LNHC)2]+, and according to the Tanabe–Sugano formalism (Fig. 1c), these energies correspond to the ligand field strength (10 Dq). Comparing these to the pyridine analogue [Cr(Lpy)2]+, which has a 10 Dq value of 17
700 cm−1 (421 nm, 2.94 eV) in [Cr(LNHC)2]+, and according to the Tanabe–Sugano formalism (Fig. 1c), these energies correspond to the ligand field strength (10 Dq). Comparing these to the pyridine analogue [Cr(Lpy)2]+, which has a 10 Dq value of 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 cm−1 (2.17 eV), a consistent trend emerges: the ligand field splitting increases progressively going from pyridine to NHC to MIC complex, correlating with the enhanced σ-donating properties of the equatorial ligands. A larger magnitude of σ-donation leads to destabilization of the antibonding eg orbitals and subsequent increase in the 10 Dq. In the orbital picture (cf.Fig. 6 and S65), complemented by ab initio ligand field theory (AILFT; Table S14),46 we find that the energy level of the vacant eg orbital set is most elevated for the MIC-ligand, followed by the NHC and then pyridine. This observation is in line with the anticipated σ-donor strengths of these ligands MIC > NHC > py,33,47,48 namely in respect to their stereoelectronic properties, yet also their behavior in CoIII and PdII/IV complexes.6,49
500 cm−1 (2.17 eV), a consistent trend emerges: the ligand field splitting increases progressively going from pyridine to NHC to MIC complex, correlating with the enhanced σ-donating properties of the equatorial ligands. A larger magnitude of σ-donation leads to destabilization of the antibonding eg orbitals and subsequent increase in the 10 Dq. In the orbital picture (cf.Fig. 6 and S65), complemented by ab initio ligand field theory (AILFT; Table S14),46 we find that the energy level of the vacant eg orbital set is most elevated for the MIC-ligand, followed by the NHC and then pyridine. This observation is in line with the anticipated σ-donor strengths of these ligands MIC > NHC > py,33,47,48 namely in respect to their stereoelectronic properties, yet also their behavior in CoIII and PdII/IV complexes.6,49
A similar trend, yet weaker, is found for the populated t2g orbital set, with the energies decreasing in the order MIC > NHC > py. Indeed, NHCs are better π-acceptors than MICs,33,47,48,50 and hence are expected to also comparatively lower the energies of the t2g orbitals. We believe that the position of pyridine, albeit commonly considered to be less π-acidic than NHCs, is due to the distortion of the ligand framework MIC < NHC < py (vide supra), that is steric reasons. Indeed, the computations also suggest increasing D4h-character in the order MIC < NHC < py (Fig. S65 and Table S14).
Hand in hand with the computational predictions, we investigated the excited-state dynamics of the newly synthesized complexes using UV-vis transient absorption (TA) spectroscopy in acetonitrile. Following the excitation of [Cr(LMIC)2]+ at 532 nm with nanosecond pulses, the TA spectrum revealed a ground state bleach (GSB) at 434 nm, matching with the 4ILCT/LLCT band observed in the ground state absorption spectrum (Fig. 7a). Additionally, three intense excited state absorption (ESA) bands at 395, 490 and 670 nm are observed. These bands are associated with the electronic transitions originating from the 2MC state, which, based on NEVPT2/CASSCF calculations (SI, Table S10 and Fig. 4), represent the energetically lowest excited state. Given their intensity, those transitions are spin-allowed and occur within the doublet excited state manifold, leading to the population of the higher-lying 2MC or 2CT excited states.
Kinetic mono-exponential traces of ESA and GSB signals yield a 2MC excited state lifetime of 59 ns. Compared to the 4.4 ns lifetime observed in the parent complex [Cr(Lpy)2]+ (Fig. S42), this represents an increase of more than tenfold. This experimental observation aligns well with our computational results, which predict destabilization of the 4MC states and an increase in 10 Dq in the complex [Cr(LMIC)2]+ due to the strong σ-donation from the MICs (cf.Fig. 6). In addition, we anticipate minor changes in 2MC energies, as electron repulsion parameters are expected to be influenced to a relatively minor extent by the introduced ligand structural modification from pyridine to MIC. However, since the anionic carbazolide unit, primarily contributing to the Racah parameter B, remains unchanged, the destabilization of 4MC is projected to be significantly more pronounced than any variation in 2MC energy (see Table 2 and further discussion). This further leads to a larger energy gap between the lowest 4MC state and the key photoactive 2MC state, reducing the efficiency of back-intersystem crossing and ultimately contributing to a slower deactivation rate.
| Complex | [Cr(L MIC ) 2 ] + | [Cr(L NHC ) 2 ] + | [Cr(L py ) 2 ] + |
|---|---|---|---|
| a Estimated using E(2E) = 9 B + 3 C − 50(B2/10 Dq), assuming C = 3.2 B.51 b Calculated energies based on NEVPT2/CASSCF. | |||
| λ abs/nm (ε/M−1 cm−1) | 432 (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300); 500 (2500); 543 (1800) 300); 500 (2500); 543 (1800) |
402 (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900); 450 (1700); 477 (1700) 900); 450 (1700); 477 (1700) |
408 (22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300); 490 (5300); 585 (600); 695 (70) 300); 490 (5300); 585 (600); 695 (70) |
| 10 Dqb/cm−1 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
| B/cm−1 | 600a | n/a | 550![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24 24 |
| τ(2MC)/ns | 59 | 1.1 | 4.4 |
| E(2MC)/eV | 1.35 dark | n/a dark | 1.16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24 emissive (77 K) 24 emissive (77 K) |
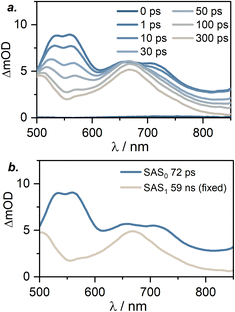
To gain further insight into excited-state dynamics on faster timescales, we analyzed the TA spectra of [Cr(LMIC)2]+ following the femtosecond-pulse excitation at 430 nm, with delay times up to 300 ps, using a global fit with a sequential excited-state population model (Fig. 8 and S24–S25). Within 1 ps after the excitation, a characteristic intense ESA double band at 550–560 nm appears, along with ESA bands at 650 and 720 nm formed. Based on spectroelectro chemical data, we attribute these spectral features to the population of 4LLCT/ILCT states. Specifically, bands at 550 and 645 nm appear in the UV-Vis differential absorption spectrum of [Cr(LMIC)2]+ upon ligand oxidation (Fig. S26c), while bands at 580 and 720 nm emerge upon ligand reduction (Fig. S26b), supporting our assignment. Subsequently, with a lifetime of 72 ps (obtained from the global fit, see Fig. 7 and S24–S25) the 550 and 720 nm bands decay, giving rise to new ESA bands at 500 and 670 nm. As previously discussed (Fig. 8 and Table 2), these new bands are attributed to the long-lived 2MC state (59 ns), which does not decay within the experiment time window considered here. Moreover, the absence of spectral features associated with ligand oxidation or/and reduction further confirms our previous 2MC state assignment. In well-investigated Cr(acac)3 (acac = acetylacetonate) and [Cr(btmp)2]3+ (btmp = 2,6-bis(4-phenyl-1,2,3-triazol-1-yl-methyl)pyridine) complexes, intersystem crossing to the doublet states is known to occur on the sub-picosecond timescale and it proceeds more rapidly than internal conversion within the quartet manifold.27,52 In our measurements, the spectral signatures of the 4CT state persist at delay times beyond 70 ps. We therefore speculate that the 70 ps time constant obtained from the global fit likely reflects a combination of intersystem crossing, internal conversion and vibrational cooling. Consistent with this interpretation, internal conversion and vibrational cooling have previously been reported to occur on the timescales up to 300 ps in polypyridine CrIII complexes.12
We anticipated that [Cr(LNHC)2]+ would exhibit photophysical properties intermediate between the parent [Cr(Lpy)2]+ and the novel [Cr(LMIC)2]+, as the σ-donation from NHC units is known to be weaker than that of MIC, bringing the 10 Dq value between the two. In the TA spectrum following excitation of [Cr(LNHC)2]+ at 355 nm with femtosecond pulses (pulse duration of ∼250 fs) at a delay time of 750 ps, a GSB at 405 nm and ESA bands at 425, 480 nm are observed (Fig. 7b). Similar to [Cr(LMIC)2]+, the GSB corresponds to the 4ILCT/LLCT band in the ground state absorption, while ESA features are attributed to the electronic transitions originating from 2MC state, predicated by DFT. The 2MC state in [Cr(LNHC)2]+ undergoes deactivation to the ground state with a lifetime of 1.1 ns. Unexpectedly, its faster excited-state relaxation compared to the parent complex [Cr(Lpy)2]+ suggests additional factors needed to be considered when rationalizing excited state dynamics. Aiming to do that, we examined the doublet and quartet excited state energies accessible upon the excitation at 355 nm (3.5 eV) (Table S13). Computational simulations predict low-lying 4LMCT states (1.8 and 1.9 eV) in close proximity to the 2MC states (1.8–1.85 eV), potentially contributing to a rapid deactivation of the latter (Fig. S65–S66 and Table S11). An analogical scenario was proposed for the homoleptic NHC complex [Cr(ImPyIm)2]3+, where excited-state decay occurred within the picosecond regime due to the low-lying charge-transfer states, populated through back-intersystem crossing.37
When discussing the excited-state properties of the parent complex [Cr(Lpy)2]+ and the novel complexes [Cr(LMIC)2]+ and [Cr(LNHC)2]+, we must address the key limitation regarding carbazolide ligands, namely unusually short excited state lifetimes relative to the other known NIR photoactive CrIII species (2MC lifetimes typically in the microsecond range) and the absence of emission in solution at room temperature. There are several plausible explanations for that excited state behavior, which are applicable for all complexes discussed herein. The most significant factor is the electronic nature of the carbazolide unit. Low-energy charge-transfer transitions from π-orbitals localized on the carbazolide moiety to the metal's d-orbitals or ligand π* orbitals (mainly localized on pyridine or carbene units) become feasible, resulting in a high density of quartet and doublet charge-transfer and metal-centered states. This complicates predictions regarding the effects of structural modifications on excited-state dynamics, as is also reflected in our experimental and computational analysis (Table S1 and Fig. S43–S44). Additionally, the greater covalency of the axial Cr–Namido bonds shortens their length compared to meridional Cr–Ccarbene or Cr–Npyridine, inducing axial compression, which is observed already in the ground state (see XRD data, Fig. S53–S54 and Table S3). This, in turn, can potentially enhance Jahn–Teller distortion in the 4MC and 4CT excited states and facilitate faster non-radiative deactivation.53
Rehm–Weller analysis
The absence of emission at room temperature in solution and technical limitations in detecting luminescence at 77 K in a glass matrix hindered the experimental determination of the 2MC excited state energies for both [Cr(LMIC)2]+ and [Cr(LNHC)2]+ complexes. Consequently, two alternative approaches were considered for estimating the doublet excited state energy. The first involves detecting spin-forbidden transitions via UV-vis ground state absorption spectroscopy, which however was not feasible in our case due to technical limitations. The second approach relies on studying a series of photoinduced electron transfer (PET) reactions, enabling estimation of the excited-state redox potential (E0(D+*/D2+)) and zero-point energy (E00) using eqn (1).54 This methodology has previously been applied to estimate the excited-state reduction potential of the photoactive 3MC state in Co(III) polypyridine complex.55 The excited state lifetime of 59 ns for [Cr(LMIC)2]+ in solution allows for diffusion-based excited-state redox reactivity, and consequently renders it suitable for this type of analysis.| E0(D+*/D2+) = E0(D+/D2+) – E00/e | (1) |
| ΔGET = [E0(D+*/D2+) – E0(A/A˙−)] × e | (2) |
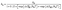 | (3) |
| ΔG‡ET = [(ΔGET/2)2 + ΔG‡ET(0)2]1/2 + ΔGET/2 | (4) |
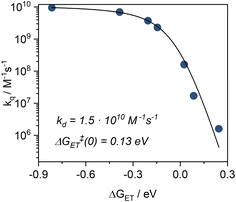
Based on the NEVPT2/CASSCF calculations, the doublet excited state energy is estimated at 1.70 eV, and the ground state oxidation potential (E0(D+/D2+)) of the complex was determined (via cyclic voltammetry) to be +0.05 V (vs. Fc/[Fc]+). Applying eqn (1), the excited state oxidation potential can be estimated to be higher than −1.65 V vs. Fc/[Fc]+. With this threshold in mind, we screened a series of electron acceptors for the oxidative quenching, including nitrobenzene and benzoquinone derivatives, with reduction potentials (E0(A/A˙−)) ranging from −1.48 V to −0.42 V vs. Fc/[Fc]+. We then performed PET experiments between the complex and selected quenchers (Table 3) in acetonitrile, using TA spectroscopy to monitor the reaction kinetics. The excited state lifetime of the 2MC state was analyzed as a function of quencher concentration using Stern–Volmer plots (Fig. S27–S33). The obtained bimolecular quenching rate constants (kq) were then evaluated as a function of the reaction driving force (ΔGET, eqn (2)) within the framework of the Rehm–Weller formalism (Fig. 9). Full Rehm–Weller plot analysis using eqn (3) and (4) with varied E0(D+*/D2+) was performed, allowing the best fit for E0(D+*/D2+) = −1.24 V. Further, a diffusion rate constant of kd = (1.49 ± 0.02)·1010 M−1 s−1 and the self-exchange activation free energy ΔGET‡(0) = 0.131 ± 0.002 eV were obtained from the fit. Those values are in line with the observations for quenching studies between [Cr(dqp)2]3+ and a series of electron donors, with kd = 1.7 × 1010 M−1 s−1 and ΔGET‡(0) = 0.14 eV, as well as with other related studies with CrIII complexes.15,56 Finally, using eqn (1), an E00 value of 1.35 eV was calculated, allowing us to construct a complete Latimer diagram for [Cr(LMIC)2]+ (Fig. 10).
| Quencher | E(A/A˙−)/V vs. Fc/[Fc]+ | k q/M−1 s−1 | ΔGET/eV |
|---|---|---|---|
| a Estimated upper limit, approximation for the quenching rate constant was made based on experiments indicating less than 10% quenching in the concentration range below 1 M. | |||
| Tetrachloro-1,4-benzoquinone | −0.42 | 9.5 × 109 | −0.82 |
| 1,4-Naphthoquinone | −0.85 | 6.9 × 109 | −0.39 |
| 1,4-Benzoquinone | −1.03 | 3.7 × 109 | −0.21 |
| 1,4-Dinitrobenzene | −1.09 | 2.3 × 109 | −0.15 |
| 4-Nitrobenzaldehyde | −1.26 | 1.6 × 108 | 0.02 |
| 4-Nitrobenzophenone | −1.32 | 1.7 × 107 | 0.08 |
| Nitrobenzene | −1.48 | 1.6 × 106a | 0.24 |
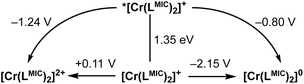 | ||
| Fig. 10 Latimer diagram of [Cr(LMIC)2]+. Ground state redox potentials obtained via cyclic voltammetry and given vs. Fc/[Fc]+. The excited state oxidation potential was determined through a series of bimolecular photoinduced electron transfer reactions involving selected electron acceptors (see Fig. 9 and Table 3). Successively, zero-point energy (E00) and excited state reduction potentials were calculated using eqn (1) and (2). | ||
Turning attention back to the NHC complex [Cr(LNHC)2]+, the excited state lifetime of 1.1 ns is too short for efficient quenching studies and the application of the same E00 estimation method used for [Cr(LMIC)2]+. According to the computational simulation, the doublet excited state energy is calculated at 1.44 eV, combining it with E0(D+/D2+) = 0.08 V vs. Fc/[Fc]+ in eqn (1), we estimate E0(D+*/D2+) to be above −1.36 V vs. Fc/[Fc]+. To probe the feasibility of the PET reaction, we tested methyl viologen (E = −1.00 V vs. Fc/[Fc]+) as an electron acceptor (Fig. S39). Less than 5% reduction in the excited state lifetime was observed for 100 mM concentration of the quencher, allowing the bimolecular quenching rate constant for this reaction to be estimated around 6.5 × 108 M−1 s−1.
When comparing the experimentally determined doublet excited state energy (E00) of 1.35 eV for [Cr(LMIC)2]+ complex and 1.16 eV for the parent [Cr(Lpy)2]+ complex (Table 2),24 we observe a minor destabilization of the 2MC excited state by 0.19 eV. This can be rationalized by the decreased covalency of the Cr–Ccarbene bond compared to the Cr–Npyridine bond. Indeed, we calculated the electron repulsion parameter B, which inversely correlates with metal-bond covalence, using the Tanabe–Sugano formalism (see equation in Fig. 1c), and the value for [Cr(LMIC)2]+ was determined to be 600 cm−1 compared to 550 cm−1 for [Cr(Lpy)2]+.24
From these observations and previous results, we can now conclude how structural modifications in our new complex affect their excited state properties. Confirming our initial hypothesis, replacing the equatorial ligand moieties from pyridines to strong σ-donating MICs leads to a significant increase in the ligand field strength while maintaining minor changes in the Racah B parameter, keeping it sufficiently low (see Table 2). Furthermore, the increased 4MC–2MC energy gap, from 1.01 eV in [Cr(Lpy)2]+ to 1.68 eV in [Cr(LMIC)2]+ complex, supports the observed reduction in the non-radiative deactivation rate of the 2MC excited state in the new complex.
Photocatalytic activity of [Cr(LMIC)2]+
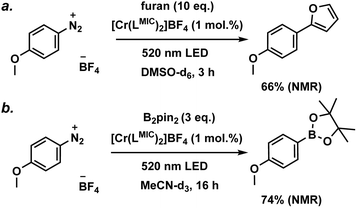
Based on these insights, we investigated the [Cr(LMIC)2]+ complex, which exhibits the longest excited state lifetime (59 ns) in the carbazolide series and an excited state oxidation potential of −1.24 V (vs. Fc/[Fc]+), for photoinduced electron transfer catalysis. Aryl diazonium salts are known to form diazo-radicals upon single electron reduction, which can further undergo C–N bond dissociation, resulting in the generation of aryl radicals and the evolution of nitrogen. The produced aryl radical can be used for a vast scope of transformations, including C–H arylations, borylations, and phosphorylations.57–59 In our study, we focused on a para-methoxy-substituted phenyldiazonium salt with a reduction potential of −1.07 V (vs. Fc/[Fc]+). Using our novel CrIII complex as a photocatalyst, we successfully demonstrated model C–H arylation and borylation reactions (Fig. 11) involving the mentioned substrate. Both photocatalytic transformations are anticipated to proceed via well-known, literature-reported mechanisms (Fig. S48 and S52).36,57–59The photocatalytic performance of [Cr(LMIC)2]+ was first evaluated for the C–H arylation of furan with a p-methoxyphenyl diazonium salt (Fig. S45–S48). Under an argon atmosphere, a catalyst loading of 1 mol%, 10 eq. of furan, and 0.11 mM of the diazonium substrate were irradiated using 520 nm LED for 3 hours. A substrate conversion of >95% and yield of 66% (NMR, relative to an internal standard, Fig. S45) were obtained. The photocatalytical borylation of p-methoxyphenyl diazonium salt with bis(pinacolato)diboron was tested as a second model transformation. Substrate conversion of >99% and 74% product yield were achieved after 16 hours of irradiation with a 520 nm LED of the reaction mixture (see Fig. S49–S51, catalyst loading of 1 mol%, 3.0 eq. of bis(pinacolato)diboron and 0.11 mM of substrate).
As indicated by the NMR yields, selected model transformations proceed efficiently under our photocatalytic conditions. For comparison, this class of reactions can be also performed using FeIII, CuI, RuII, OsII and IrIII transition metal complexes as photocatalysts, as well as metal-free systems such as eosin Y, under visible-light irradiation, typically affording moderate to high yields.36,57–59 It should be noted that the literature-reported systems display appreciable thermal reactivity, which accounts for the yields of up to 20% observed in the control experiments, both in related studies and this work (Fig. S46, S47, S50 and S51).36,57–59
Conclusions
A significant milestone in the development of CrIII and MnIV complexes was achieved when the nephelauxetic effect was strategically incorporated into ligand design, enabling red-shifting of spin-flip transitions into the NIR region.22,24,25,29,31 Building on this concept, a deeper understanding of how ligands influence the properties of these low-energy excited states is essential for their future applications in photochemistry and biomedicine.In this context, we demonstrated a feasible method for estimating the energy of dark 2MC excited states using photoinduced electron transfer and Rehm–Weller analysis. This approach has proven to be a valuable tool for probing the photophysical behavior of our new [Cr(LMIC)2]+ complex and CrIII systems in general.
Ultimately, we established a versatile strategy for tuning the ratio between ligand field strength and the Racah parameter B, by using pincer-type ligands based on covalent carbazolide core units and modifying the donor groups from pyridines, as in our previously reported [Cr(Lpy)2]+,24 to stronger σ-donors such as MICs in the novel [Cr(LMIC)2]+. This enhanced 10 Dq from 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 to 24
500 to 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1 while only modestly increasing B from 550 to 600 cm−1. This strategy achieves a precise balance between the key parameters: a low interelectronic repulsion (reflecting metal–ligand bond covalency) favorable for spin-flip transitions in the near-infrared, combined with sufficiently high ligand field strength to extend excited-state lifetimes. Together, these results provide a clear framework for designing NIR-photoactive CrIII complexes with tunable excited-state properties.
400 cm−1 while only modestly increasing B from 550 to 600 cm−1. This strategy achieves a precise balance between the key parameters: a low interelectronic repulsion (reflecting metal–ligand bond covalency) favorable for spin-flip transitions in the near-infrared, combined with sufficiently high ligand field strength to extend excited-state lifetimes. Together, these results provide a clear framework for designing NIR-photoactive CrIII complexes with tunable excited-state properties.
Author contributions
Synthesis was carried out by PY, BW and AS. PY conducted spectroscopic investigations and analysis, photocatalysis experiments. Electrochemical investigations were carried out by PY and FRN. EPR spectroscopic investigations were carried out by DL. Theoretical investigations were performed by DM. Crystal structure solution and analysis was performed by FT and SH. The idea was conceived by SH and OSW. The manuscript was written by PY, BW, DM, OSW and SH and proof read by all authors.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2304955 ([Cr(LMIC)2]BF4) and 2343870 ([Cr(LNHC)2]BF4) contain the supplementary crystallographic data for this paper.61a,bSupplementary information: 1H, Evans, UV-Vis, IR and elemental analysis data for all complexes. In addition, the SI contains supporting spectra on the photophysical measurements, EPR spectra, computational details and additional crystallographic information.60 See DOI: https://doi.org/10.1039/d5sc09069e.
Acknowledgements
Funding from the Swiss National Science Foundation through grant number 200020_207329 is acknowledged (O. S. W.). S.H. D.L. and F.R.N. thank the Austrian Science Fund Grant-DOI: https://doi.org/10.55776/P34626 and the University of Innsbruck for generous funding of this work. Roland Egger and Sylvie Mittelheisser are kindly acknowledged for measuring elemental analysis. Dr Michael Pfeffer and Dr Thomas Müller are kindly acknowledged for measuring mass spectrometry. D.M. acknowledges scientific support and HPC resources provided by the Erlangen National High Performance Computing Center (NHR@FAU) of the Friedrich-Alexander-Universität (FAU) Erlangen-Nürnberg. We thank the NHR funding provided by federal and Bavarian state authorities. The NHR@FAU hardware is partially funded by the German Research Foundation (DFG)–440719683.Notes and references
- (a) L. S. Forster, Chem. Rev., 1990, 90, 331 CrossRef CAS; (b) L. S. Forster, Coord. Chem. Rev., 2002, 227, 59 CrossRef CAS; (c) W. R. Kitzmann and K. Heinze, Angew. Chem., Int. Ed., 2023, 62, e202213207 CrossRef CAS PubMed.
- (a) N. Serpone, M. A. Jamieson, M. S. Henry, M. Z. Hoffman, F. Bolletta and M. Maestri, J. Am. Chem. Soc., 1979, 101, 2907 CrossRef CAS; (b) A. D. Kirk, Chem. Rev., 1999, 99, 1607 CrossRef CAS PubMed; (c) C. K. Ryu and J. F. Endicott, Inorg. Chem., 1988, 27, 2203 CrossRef CAS; (d) N. A. P. Kane-Maguire and C. H. Langford, J. Am. Chem. Soc., 1972, 94, 2125 CrossRef CAS; (e) M. Isaacs, A. G. Sykes and S. Ronco, Inorg. Chim. Acta, 2006, 359, 3847 CrossRef CAS.
- S. Otto, M. Grabolle, C. Förster, C. Kreitner, U. Resch-Genger and K. Heinze, Angew. Chem., Int. Ed., 2015, 54, 11572 CrossRef CAS PubMed.
- S. Treiling, C. Wang, C. Förster, F. Reichenauer, J. Kalmbach, P. Boden, J. P. Harris, L. M. Carrella, E. Rentschler, U. Resch-Genger, C. Reber, M. Seitz, M. Gerhards and K. Heinze, Angew. Chem., Int. Ed., 2019, 131, 18243 CrossRef.
- (a) J. K. McCusker, Science, 2019, 363, 484 CrossRef CAS PubMed; (b) M. Dorn, N. R. East, C. Förster, W. R. Kitzmann, J. Moll, F. Reichenauer, T. Reuter, L. Stein and K. Heinze, in Comprehensive Inorganic Chemistry III, Elsevier, 2023, pp. 707–788 Search PubMed; (c) S. Kronenberger, R. Naumann, C. Förster, N. R. East, J. Klett and K. Heinze, Nat. Commun., 2025, 16, 7850 CrossRef CAS PubMed.
- J. T. Yarranton and J. K. McCusker, J. Am. Chem. Soc., 2022, 144, 12488 CrossRef CAS PubMed.
- W. R. Kitzmann, J. Moll and K. Heinze, Photochem. Photobiol. Sci., 2022, 21, 1309 CrossRef CAS PubMed.
- G. Morselli, C. Reber and O. S. Wenger, J. Am. Chem. Soc., 2025, 147, 11608 CrossRef CAS PubMed.
- L. Hammarström and O. Johansson, Coord. Chem. Rev., 2010, 254, 2546 CrossRef.
- M. Jäger, A. Smeigh, F. Lombeck, H. Görls, J.-P. Collin, J.-P. Sauvage, L. Hammarström and O. Johansson, Inorg. Chem., 2010, 49, 374 CrossRef PubMed.
- J.-R. Jiménez, B. Doistau, C. M. Cruz, C. Besnard, J. M. Cuerva, A. G. Campaña and C. Piguet, J. Am. Chem. Soc., 2019, 141, 13244 CrossRef PubMed.
- F. Reichenauer, C. Wang, C. Förster, P. Boden, N. Ugur, R. Báez-Cruz, J. Kalmbach, L. M. Carrella, E. Rentschler, C. Ramanan, G. Niedner-Schatteburg, M. Gerhards, M. Seitz, U. Resch-Genger and K. Heinze, J. Am. Chem. Soc., 2021, 143, 11843 CrossRef CAS PubMed.
- P. Yaltseva, T. Maisuradze, A. Prescimone, S. Kupfer and O. S. Wenger, J. Am. Chem. Soc., 2025, 147, 29444 CrossRef CAS PubMed.
- (a) C. Wang, F. Reichenauer, W. R. Kitzmann, C. Kerzig, K. Heinze and U. Resch-Genger, Angew. Chem., Int. Ed., 2022, 61, e202202238 CrossRef CAS PubMed; (b) L. Aboshyan-Sorgho, M. Cantuel, S. Petoud, A. Hauser and C. Piguet, Coord. Chem. Rev., 2012, 256, 1644 CrossRef CAS; (c) S. Trippmacher, S. Demeshko, A. Prescimone, F. Meyer, O. S. Wenger and C. Wang, Chem. –Eur. J., 2024, 30, e202400856 CrossRef CAS PubMed.
- T. H. Bürgin, F. Glaser and O. S. Wenger, J. Am. Chem. Soc., 2022, 144, 14181 CrossRef PubMed.
- (a) Z. Shen and J.-L. Tu, Eur. J. Inorg. Chem., 2025, 28, e202401016 CrossRef CAS; (b) S. Sittel, A. C. Sell, K. Hofmann, C. Wiedemann, J. P. Nau, C. Kerzig, G. Manolikakes and K. Heinze, ChemCatChem, 2023, 15, e202201562 CrossRef CAS; (c) R. F. Higgins, S. M. Fatur, S. G. Shepard, S. M. Stevenson, D. J. Boston, E. M. Ferreira, N. H. Damrauer, A. K. Rappé and M. P. Shores, J. Am. Chem. Soc., 2016, 138, 5451 CrossRef CAS PubMed; (d) C. Wang, H. Li, T. H. Bürgin and O. S. Wenger, Nat. Chem., 2024, 16, 1151 CrossRef CAS PubMed.
- (a) M. Poncet, A. Benchohra, J.-R. Jiménez and C. Piguet, ChemPhotoChem, 2021, 5, 880 CrossRef CAS; (b) M. Poncet, C. Besnard, L. Guénée, J.-R. Jiménez and C. Piguet, Front. Chem., 2024, 12 DOI:10.3389/fchem.2024.1472943; (c) J.-R. Jiménez, M. Poncet, B. Doistau, C. Besnard and C. Piguet, Dalton Trans., 2020, 49, 13528 RSC; (d) C. Dee, F. Zinna, W. R. Kitzmann, G. Pescitelli, K. Heinze, L. Di Bari and M. Seitz, Chem. Comm., 2019, 55, 13078 RSC.
- S. Otto, C. Förster, C. Wang, U. Resch-Genger and K. Heinze, Chem. –Eur. J., 2018, 24, 12555 CrossRef CAS PubMed.
- Y. Tanabe and S. Sugano, J. Phys. Soc. Jpn., 1954, 9, 766 CrossRef CAS.
- S. Adachi, J. Lumin., 2024, 273, 120628 CrossRef CAS.
- C. E. Schäffer and C. Klixbüll Jørgensen, J. Inorg. Nucl. Chem., 1958, 8, 143 CrossRef.
- N. Sinha, P. Yaltseva and O. S. Wenger, Angew. Chem., Int. Ed., 2023, 62, e202303864 CrossRef CAS PubMed.
- F. Reichenauer, D. Zorn, R. Naumann, C. Förster and K. Heinze, Inorg. Chem., 2024, 63, 23487 CrossRef CAS PubMed.
- N. Sinha, J.-R. Jiménez, B. Pfund, A. Prescimone, C. Piguet and O. S. Wenger, Angew. Chem., Int. Ed., 2021, 60, 23722 CrossRef CAS PubMed.
- N. Sawicka, C. J. Craze, P. N. Horton, S. J. Coles, E. Richards and S. J. A. Pope, Chem. Comm., 2022, 58, 5733 RSC.
- (a) Y. Cheng, J. He, W. Zou, X. Chang, Q. Yang and W. Lu, Chem. Comm., 2023, 59, 1781 RSC; (b) A. Benchohra, J. Chong, C. M. Cruz, C. Besnard, L. Guénée, A. Rosspeintner and C. Piguet, Inorg. Chem., 2024, 63, 3617 CrossRef CAS PubMed.
- R. W. Jones, A. J. Auty, G. Wu, P. Persson, M. V. Appleby, D. Chekulaev, C. R. Rice, J. A. Weinstein, P. I. P. Elliott and P. A. Scattergood, J. Am. Chem. Soc., 2023, 145, 12081 CrossRef CAS PubMed.
- F. Wang, Y. Zhong, O. Bruns, Y. Liang and H. Dai, Nat. Photon., 2024, 18, 535 CrossRef CAS.
- L. Stein, C. Förster and K. Heinze, Organometallics, 2024, 43, 1766 CrossRef CAS.
- L. Stein, P. Boden, R. Naumann, C. Förster, G. Niedner-Schatteburg and K. Heinze, Chem. Comm., 2022, 58, 3701 RSC.
- Y. Cheng, Q. Yang, J. He, W. Zou, K. Liao, X. Chang, C. Zou and W. Lu, Dalton Trans., 2023, 52, 2561 RSC.
- (a) J. V. Caspar and T. J. Meyer, J. Phys. Chem., 1983, 87, 952 CrossRef CAS; (b) R. A. Marcus and N. Sutin, Biochim. Biophys. Acta Rev. Bioenerg., 1985, 811, 265 CrossRef CAS; (c) A. Ghosh, J. T. Yarranton and J. K. McCusker, Nat. Chem., 2024, 16, 1665 CrossRef CAS PubMed.
- D. Munz, Organometallics, 2018, 37, 275 CrossRef CAS.
- (a) Y. Liu, T. Harlang, S. E. Canton, P. Chábera, K. Suárez-Alcántara, A. Fleckhaus, D. A. Vithanage, E. Göransson, A. Corani, R. Lomoth, V. Sundström and K. Wärnmark, Chem. Comm., 2013, 49, 6412 RSC; (b) S. Kaufhold, N. W. Rosemann, P. Chábera, L. Lindh, I. Bolaño Losada, J. Uhlig, T. Pascher, D. Strand, K. Wärnmark, A. Yartsev and P. Persson, J. Am. Chem. Soc., 2021, 143, 1307 CrossRef CAS PubMed; (c) P. Dierks, A. Kruse, O. S. Bokareva, M. J. Al-Marri, J. Kalmbach, M. Baltrun, A. Neuba, R. Schoch, S. Hohloch, K. Heinze, M. Seitz, O. Kühn, S. Lochbrunner and M. Bauer, Chem. Commun., 2021, 57, 6640 RSC; (d) J. Steube, A. Kruse, O. S. Bokareva, T. Reuter, S. Demeshko, R. Schoch, M. A. Argüello Cordero, A. Krishna, S. Hohloch, F. Meyer, K. Heinze, O. Kühn, S. Lochbrunner and M. Bauer, Nat. Chem., 2023, 15, 468 CrossRef CAS PubMed; (e) J. Wellauer, B. Pfund, I. Becker, F. Meyer, A. Prescimone and O. S. Wenger, J. Am. Chem. Soc., 2025, 147, 8760 CrossRef CAS PubMed.
- J. P. Harris, C. Reber, H. E. Colmer, T. A. Jackson, A. P. Forshaw, J. M. Smith, R. A. Kinney and J. Telser, Can. J. Chem., 2017, 95, 547 CrossRef.
- J. Wellauer, F. Ziereisen, N. Sinha, A. Prescimone, A. Velić, F. Meyer and O. S. Wenger, J. Am. Chem. Soc., 2024, 146, 11299 CAS.
- R. W. Jones, R. A. Cowin, I. I. Ivalo, D. Chekulaev, T. M. Roseveare, C. R. Rice, J. A. Weinstein, P. I. P. Elliott and P. A. Scattergood, Inorg. Chem., 2024, 63, 8526 CrossRef CAS PubMed.
- (a) P. Pinter, C. M. Schüßlbauer, F. A. Watt, N. Dickmann, R. Herbst-Irmer, B. Morgenstern, A. Grünwald, T. Ullrich, M. Zimmer, S. Hohloch, D. M. Guldi and D. Munz, Chem. Sci., 2021, 12, 7401 RSC; (b) M. Moser, B. Wucher, D. Kunz and F. Rominger, Organometallics, 2007, 26, 1024 CrossRef CAS; (c) F. A. Watt, B. Sieland, N. Dickmann, R. Schoch, R. Herbst-Irmer, H. Ott, J. Paradies, D. Kuckling and S. Hohloch, Dalton Trans., 2021, 50, 17361 RSC.
- B. Wittwer, N. Dickmann, S. Berg, D. Leitner, L. Tesi, D. Hunger, R. Gratzl, J. van Slageren, N. I. Neuman, D. Munz and S. Hohloch, Chem. Commun., 2022, 58, 6096 RSC.
- N. R. East, C. Förster, L. M. Carrella, E. Rentschler and K. Heinze, Inorg. Chem., 2022, 61, 14616 CrossRef CAS PubMed.
- N. R. East, R. Naumann, C. Förster, C. Ramanan, G. Diezemann and K. Heinze, Nat. Chem., 2024, 16, 827 CrossRef CAS PubMed.
- N. Kaul, E. Asempa, J. A. Valdez-Moreira, J. M. Smith, E. Jakubikova and L. Hammarström, J. Am. Chem. Soc., 2024, 146, 24619 CrossRef CAS PubMed.
- T. Huang, P. Du, X. Cheng and Y.-M. Lin, J. Am. Chem. Soc., 2024, 146, 24515 CrossRef CAS PubMed.
- J. Sander, F. Gauder, S. J. Schicktanz, T. Maulbetsch, J. Kalmbach, D. Zorn, R. Naumann, C. Förster, D. Kunz, K. Heinze and Z. Allg, Anorg. Chem., 2025, 651 Search PubMed.
- D. S. McGuinness, V. C. Gibson, D. F. Wass and J. W. Steed, J. Am. Chem. Soc., 2003, 125, 12716 CrossRef CAS PubMed.
- S. K. Singh, J. Eng, M. Atanasov and F. Neese, Coord. Chem. Rev., 2017, 344, 2 CrossRef CAS.
- H. V. Huynh, Chem. Rev., 2018, 118, 9457 CrossRef CAS PubMed.
- D. J. Nelson and S. P. Nolan, Chem. Soc. Rev., 2013, 42, 6723 RSC.
- D. Munz, Chem. Sci., 2018, 9, 1155 RSC.
- (a) X. Hu, I. Castro-Rodriguez, K. Olsen and K. Meyer, Organometallics, 2004, 23, 755 CrossRef CAS; (b) K. Verlinden, H. Buhl, W. Frank and C. Ganter, Eur. J. Inorg. Chem., 2015, 2015, 2416 CrossRef CAS.
- C. K. Jørgensen, Adv. Chem. Phys., 1963, 5, 33–146 CrossRef.
- E. A. Juban, A. L. Smeigh, J. E. Monat and J. K. McCusker, Coord. Chem. Rev., 2006, 250, 1783 CrossRef CAS.
- O. S. Wenger, R. Valiente and H. U. Güdel, J. Chem. Phys., 2001, 115, 3819 CrossRef CAS.
- D. Rehm and A. Weller, Isr. J. chem., 1970, 8, 259 CrossRef CAS.
- M. M. Alowakennu, A. Ghosh and J. K. McCusker, J. Am. Chem. Soc., 2023, 145, 20786 CrossRef CAS PubMed.
- G. Morselli, T. H. Eggenweiler, M. Villa, A. Prescimone and O. S. Wenger, J. Am. Chem. Soc., 2025, 147, 28226 CrossRef CAS PubMed.
- D. P. Hari, P. Schroll and B. König, J. Am. Chem. Soc., 2012, 134, 2958 CrossRef CAS PubMed.
- J. Yu, L. Zhang and G. Yana, Adv. Synth. Catal., 2012, 354, 2625 CrossRef CAS.
- A. Ripak, S. de Kreijger, R. N. Sampaio, C. A. Vincent, É. Cauët, I. Jabin, U. K. Tambar, B. Elias and L. Troian-Gautier, Chem Catal., 2023, 3, 100490 CAS.
- (a) C. Adamo and V. Barone, J. Chem. Phys., 1999, 110, 6158 CrossRef CAS; (b) C. Angeli, R. Cimiraglia, S. Evangelisti, T. Leininger and J.-P. Malrieu, J. Chem. Phys., 2001, 114, 10252 CrossRef CAS; (c) V. Barone and M. Cossi, J. Phys. Chem. A, 1998, 102, 1995 CrossRef CAS; (d) E. Caldeweyher, S. Ehlert, A. Hansen, H. Neugebauer, S. Spicher, C. Bannwarth and S. Grimme, J. Chem. Phys., 2019, 150, 154122 CrossRef PubMed; (e) J.-D. Chai and M. Head-Gordon, Phys. Chem. Chem. Phys., 2008, 10, 6615 RSC; (f) Chemcraft - graphical software for visualization of quantum chemistry computations, Version 1.8, build 654, https://www.chemcraftprog.com Search PubMed; (g) H. E. Gottlieb, V. Kotlyar and A. Nudelman, J. Org. Chem., 1997, 62, 7512 CrossRef CAS PubMed; (h) S. Grimme, A. Hansen, S. Ehlert and J.-M. Mewes, J. Chem. Phys., 2021, 154, 64103 CrossRef CAS PubMed; (i) M. D. Hanwell, D. E. Curtis, D. C. Lonie, T. Vandermeersch, E. Zurek and G. R. Hutchison, J. Cheminform., 2012, 4, 17 CrossRef CAS PubMed; (j) G. Knizia, J. Chem. Theory Comput., 2013, 9, 4834 CrossRef CAS PubMed; (k) F. Neese, WIREs Comput. Mol. Sci., 2012, 2, 73 CrossRef CAS; (l) F. Neese, WIREs Comput. Mol. Sci., 2018, 8, e1327 CrossRef; (m) F. Neese, F. Wennmohs, A. Hansen and U. Becker, Chem. Phys., 2009, 356, 98 CrossRef CAS; (n) D. A. Pantazis, X.-Y. Chen, C. R. Landis and F. Neese, J. Chem. Theory Comput., 2008, 4, 908 CrossRef CAS PubMed; (o) V. N. Staroverov, G. E. Scuseria, J. Tao and J. P. Perdew, J. Chem. Phys., 2003, 119, 12129 CrossRef CAS; (p) C. van Wüllen, J. Chem. Phys., 1998, 109, 392 CrossRef; (q) F. Weigend, Phys. Chem. Chem. Phys., 2006, 8, 1057 RSC; (r) F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297 RSC; (s) T. Yanai, D. P. Tew and N. C. Handy, Chem. Phys. Lett., 2004, 393, 51 CrossRef CAS; (t) G. L. Stoychev, A. A. Auer and F. Neese, J. Chem. Theory Comput., 2017, 13, 554 CrossRef CAS PubMed; (u) G. M. Sheldrick, Acta Cryst. A, 2015, 71, 3 CrossRef PubMed; (v) G. M. Sheldrick, Acta Cryst. C, 2015, 71, 3 CrossRef PubMed; (w) O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339 CrossRef CAS; (x) A. L. Spek, Acta Cryst. C, 2015, 71, 9 CrossRef CAS PubMed; (y) S. Stoll and A. Schweiger, J. Mag. Reson., 2006, 178, 42 CrossRef CAS PubMed; (z) R. Izsák and F. Neese, J. Chem. Phys., 2011, 135, 144105 CrossRef PubMed.
- (a) CCDC 2304955: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2hchds; (b) CCDC 2343870: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2jnzqx.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |