Open Access Article
Open Access ArticleSuperstructure-induced enhancement of solid-state triphenylacrylonitrile photochromism via a macrocycle-mediated blocking strategy
Meng-Hao
Li
,
Hui
Hui
,
Yan
Wang
and
Ying-Wei
Yang
 *
*
International Joint Research Laboratory of Nano-Micro Architecture Chemistry, College of Chemistry, Jilin University, 2699 Qianjin Street, Changchun 130012, P. R. China. E-mail: ywyang@jlu.edu.cn
First published on 22nd January 2026
Abstract
Developing materials with precisely tunable photochromic properties is crucial for innovative applications, and this requires rational design strategies to overcome photoisomerization constraints imposed by molecular stacking. Herein, we introduce a pillararene-based “blocker” strategy to mitigate π–π stacking of photoisomerizable molecules. The pillararene entity facilitates photochromic chromophore torsion by providing free volume and stabilizes photoisomerization products in conjunction with specific volatile organic compounds (VOCs) through supramolecular interactions. With this strategy, we obtain a solid-state photochromic material induced by VOCs, exhibiting reversible white-to-red color changes. This material's enhanced photochromic behavior, especially when exposed to oxygen-containing VOCs, such as CH3OH, HCOOH, and ethyl acetate, expands its potential applications in UV printing, anti-counterfeiting, and multi-dimensional information encryption. Leveraging the pillar-shaped structure and molecular recognition abilities of pillararenes, this study offers a straightforward strategy for the design of multi-stimuli-responsive photochromic molecules and functional supramolecular macrocycles.
Introduction
Photochromic compounds have attracted significant attention in scientific research and technological applications owing to their reversible color changes upon light irradiation, making them essential optical functional materials.1 This phenomenon typically stems from the light-induced interconversion between two or more metastable molecular isomers,2 resulting in altered light absorption and emission properties. Common photochromic compounds, such as spirooxazines,3 spiropyrans,4 and diarylethenes,5,6 are widely deployed in various fields, including optical information storage,7–10 specialty inks and coatings,11 and advanced smart optical devices (e.g., photochromic lenses and glasses).12 Among these, diarylethene derivatives are promising candidates, particularly for emerging applications including high-density optical data storage,13 advanced bioimaging,14 and nanoscale optoelectronics,15 due to their readily tunable chemical structures. This tunability, facilitated by multiple substitution sites, enables the fine-tuning of some crucial properties, such as high thermal stability, strong environmental resistance, low background absorption, high cycling durability, light-response wavelength, and color-change rate.10,15–17 On this basis, triphenylethylene derivatives, prepared by substituting a hydrogen atom on the diarylethene framework with an additional phenyl ring, exhibit excellent photochromic performance with high sensitivity and rapid response via reversible intramolecular ring-opening/closure reactions,18–25 enabling significant progress in applications such as photo-induced deformation and information encryption. However, due to tight π–π stacking during molecular aggregation, which impedes photocyclization, the photochromic behavior of these molecules remains significantly challenging in the solid state,2,18–23,26–28 an issue that has attracted widespread attention from researchers seeking improvement.Based on these considerations, suppressing tight π–π stacking between chromophores is a practical approach to extending and enhancing solid-state photochromic performance. Supramolecular macrocyclic compounds, including crown ethers,29,30 cyclodextrins,31 cucurbiturils,32,33 calixarenes,34 and pillararenes,35,36 offer a versatile strategy to mitigate chromophore aggregation due to their inherent cavity architectures. These macrocycles leverage non-covalent interactions (hydrogen bonding, van der Waals forces, and electrostatic interactions) and the spatial confinement of host–guest complexation to enable improved photoresponsive behavior.37–42 For instance, encapsulation in macrocyclic cavities readily stabilizes, refines, and amplifies the optical properties of guest chromophores, encompassing photoluminescence, photochromism, and photosensitization.43–52 Additionally, conjugating chromophores or other functional moieties to macrocyclic arene rims through covalent or coordination linkages has been extensively investigated to create advanced photoresponsive materials.53–58 A rationally designed macrocycle-based photochromic material should meet the following criteria: (1) judicious selection of macrocycle type, size, and rigidity is paramount to ensure optimal spatial separation of photochromic active groups. This precise spacing is essential for robust chromic behavior, especially in the solid state, synergistically mitigating activity loss from close packing and π–π stacking. (2) Flexible electronic distributions and diverse supramolecular interactions are indispensable for facilitating chromophore twisting, bond formation, or bond cleavage, thereby optimizing the photochromic response.
Considering these criteria, pillararenes, particularly pillar[5]arenes, emerge as highly advantageous for optimizing the optical properties of photoresponsive groups.57,59–62 Pillar[5]arenes possess a rigid, pillar-shaped structure with an exceptionally high symmetry and an optimal cavity size, which are crucial for acting as ordered scaffolds and effectively preventing active site aggregation.63–66 Their inherent rigidity and five-fold symmetry, along with favorable synthetic yields and readily modifiable rims,67,68 can render them ideal chromophore spacers. Moreover, the electron-rich arene units of pillar[5]arenes allow for tunable modulation of electronic distributions through delicate intermolecular interactions.69 A practical approach to achieving this involves utilizing volatile organic compound (VOC)-induced specific superstructures, which consequently redistribute the electron density of constituent units.51,58,69–71 This process occurs as VOCs bind to the macrocycles' internal cavities or external surfaces, a binding mechanism dictated by the VOCs' unique size, polarity, and specific intermolecular interactions. By leveraging charge-transfer or adsorption/desorption coupling, these materials can precisely regulate the microenvironment within the molecular framework, enabling or significantly amplifying phenomena such as vapochromism (vapor-induced color change) or vapoluminescence (vapor-induced luminescence change).
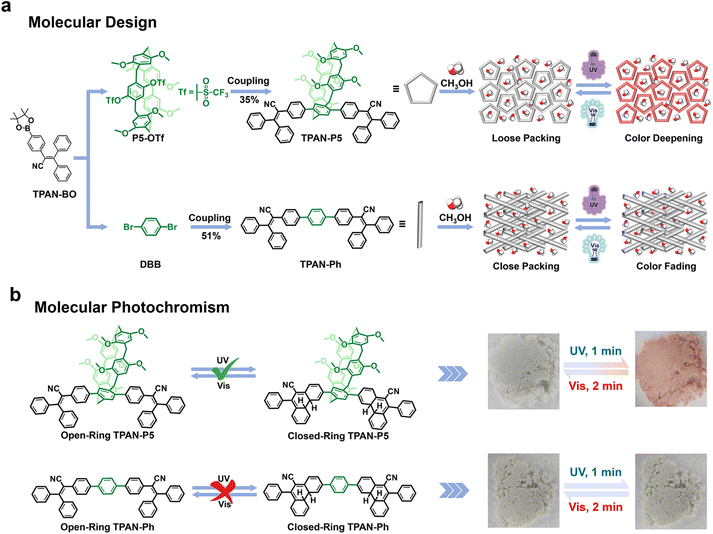
Herein, we present a strategy for optimizing photochromism by leveraging pillar[5]arenes as “blockers” to circumvent tight intermolecular packing and thereby enhance molecular spatial freedom. By modifying the pillar[5]arene rims with triphenylacrylonitrile (TPAN) units, we synthesized a TPAN-functionalized pillar[5]arene (TPAN-P5, Scheme 1a). To investigate the pillararene's crucial role in regulating photochromic activity, a control compound lacking the pillararene moiety, TPAN-Ph (Scheme 1a), was also prepared. Our findings reveal a remarkable contrast: TPAN-P5 powder, when exposed to specific VOCs such as CH3OH, HCOOH, and ethyl acetate (EA), visibly transforms into a pink hue under 365 nm light irradiation and reverts to its initial state in darkness or under white light irradiation (Scheme 1b). TPAN-Ph powder exhibits no observable photochromic properties under identical conditions. The photochromism observed in TPAN-P5 is attributed to the enhanced molecular spatial freedom conferred by the pillararene cavity. This freedom optimally separates the TPAN units, effectively preventing detrimental close packing and π–π stacking. It facilitates supramolecular interactions between the TPAN units and VOCs, which, in turn, lead to the formation of stable superstructures. These supramolecular architectures significantly reconfigure the excited-state electron distribution within the TPAN chromophore, enabling free rotation and subsequent bond formation of the TPAN units, thereby powerfully promoting the solid-state photocyclization reaction. Furthermore, the multi-stimuli-responsive TPAN-P5 powder shows excellent potential for practical applications in UV printing, anti-counterfeiting, and multi-dimensional information encryption, due to its fast response speed, anti-fatigue properties, and long color retention time.
Results and discussion
Syntheses and characterization of TPAN-P5 and TPAN-Ph
TPAN-P5 and TPAN-Ph were synthesized via a one-step Suzuki coupling reaction. The required brominated and boronated triphenylacrylonitrile derivatives (TPAN-Br and TPAN-BO), triflate-bearing pillar[5]arene (P5-OTf), and 1,4-dibromobenzene (DBB) were prepared according to literature procedures or purchased directly (Schemes S1–S3).57 Both compounds were rigorously purified by column chromatography, and their chemical structures were confirmed by 1H and 13C nuclear magnetic resonance (NMR) spectra, high-resolution mass spectrometry (HRMS), and single-crystal X-ray diffraction (SC-XRD) (Fig. S1–S7). To elucidate the influence of the pillar[5]arene moiety on the configuration and conformation of the TPAN chromophore, single crystals of TPAN-P5 and TPAN-Ph were obtained by slow solvent evaporation from mixed CH3OH/CH2Cl2 and CH3OH/CHCl3 solutions, respectively, affording transparent block-shaped crystals (Table S1).The crystal structure of TPAN-P5, determined to be monoclinic with space group P2/n, revealed that TPAN incorporation at the pillar[5]arene periphery did not substantially perturb the macrocycle's overall geometry (Fig. 1a). The cavity retained a regular pentagonal shape with a diameter of 4.7 Å, comparable to that of an unmodified pillar[5]arene.35 The TPAN unit's benzene rings, directly linked to pillar[5]arene, exhibited a 60.19° twist relative to the macrocycle plane, adopting opposing orientations (Fig. 1b and S8). This orientation, combined with the inherent void space of the pillararene cavity, promoted a staggered, non-close-packed arrangement of neighboring TPAN-P5 molecules. Consequently, the crystal packing featured one-dimensional channels along the a-axis (Fig. 1c) and a layered, staggered pattern along the b- and c-axes (Fig. S9). As expected for a photochromic TPAN derivative, the C⋯C distance between cis-oriented benzene rings was measured to be 3.378 Å (Fig. 1d), priming the molecule for photocyclization.18 Crucially, the pillararene cavity can provide ample free volume for these photoactive TPAN moieties, as evidenced by centroid-to-centroid distances of 11.524 Å and 12.396 Å between the TPAN's benzene rings directly connected to the pillararene (Fig. 1e and f). This free volume is essential for reducing steric crowding and promoting the necessary rotation and cyclization of TPAN units, processes that are otherwise hindered by intermolecular interactions. TPAN-Ph crystallized in a monoclinic lattice (space group P21/c) and exhibited an intramolecular distance of 3.189 Å between cis-oriented benzene rings, an intramolecular arrangement theoretically suitable for photocyclization (Fig. 1g). However, unlike TPAN-P5, the terphenyl moiety in TPAN-Ph exhibited a smaller dihedral angle of only 29.58° (Fig. 1h and S10), implying enhanced conjugation that can restrict torsional motion. Furthermore, TPAN-Ph displayed a close-packed arrangement with rhombus-shaped pores (Fig. 1i and S11) and significantly smaller intermolecular distances between active sites (4.911, 9.142, 11.125, 4.277, and 6.170 Å; Fig. 1j–l) compared to TPAN-P5, highlighting a more constrained packing environment.
Photochromic properties of TPAN-P5 and TPAN-Ph in solution
The photochromic properties of TPAN-P5 and TPAN-Ph were investigated in solution and solid states to assess our design strategy. Upon UV irradiation (365 nm), a TPAN-P5 solution in CH2Cl2 exhibited a new absorption band at 485 nm with increasing irradiation time, accompanied by a color change from colorless to orange-red (Fig. 2a and S12). This transformation was reversible, with the solution returning to its initial state in the dark after 27 minutes (Fig. S13). Irradiation with visible light significantly accelerated the reverse reaction, completing it in just 5 minutes (Fig. 2b and S13), highlighting the light-controlled nature of the photochromism. TPAN-Ph, when dissolved in CH2Cl2, also exhibited photochromic behavior, displaying an additional absorption peak at around 500 nm (Fig. 2c and S14). Besides, TPAN-Ph displayed spontaneous fading in the dark (4 minutes) and accelerated fading (18 seconds) under white light irradiation (Fig. 2d and S15).The photochromic performance of TPAN-P5 and TPAN-Ph was quantified (Fig. 2e), with TPAN-P5 showing a 3.6-fold higher conversion efficiency than TPAN-Ph. This enhancement is attributed to the intrinsic structural and electronic influence of pillararene. With its electron-rich, rigid cavity, pillararene optimizes the electronic landscape and molecular conformation of the TPAN unit, thereby promoting more efficient photocyclization. Moreover, the photoisomerized TPAN-P5 showed superior stability, as evidenced by its lower spontaneous and white light-induced fading rates (Fig. S16). The recovery kinetics deviated from ideal first-order behavior, necessitating direct determination of half-lives from experimental traces. TPAN-P5 showed significantly longer dark recovery (t1/2 = 82 s) compared to TPAN-Ph (t1/2 = 13 s), while white light irradiation shortened the half-lives to 36 and 7 s, respectively (Fig. 2f and S17). This has confirmed the enhanced photochromic and coloration-retention properties of TPAN-P5 compared to TPAN-Ph. Moreover, TPAN-P5 exhibited excellent cycling stability with no significant fatigue observed after six cycles, whereas TPAN-Ph retained only 73% of its initial coloration intensity after the first cycle (Fig. 2g, h and S18).
The photoinduced cyclization of the TPAN unit, responsible for the photochromism of TPAN-P5 and TPAN-Ph in solution, was confirmed by 1H NMR spectroscopy before and after irradiation at 365 nm. The TPAN unit, as a typical photochromic group capable of photoinduced cyclization, initially cyclizes upon light irradiation to form the ring-closed isomer TPAN-B (Fig. S19).20 Subsequently, due to TPAN-B's poor thermal stability, it readily transforms into the dehydrogenated form, TPAN-C, in air. Thus, prolonged UV irradiation of TPAN-P5 and TPAN-Ph in CDCl3 solution yielded the dehydrogenated species, TPAN-P5–C and TPAN-Ph–C, respectively, originating from TPAN-P5–B and TPAN-Ph–B (Fig. S20 and S21). This confirms that the photochromic mechanism of both compounds is a structural transformation caused by intramolecular cyclization. The TPAN unit endows both TPAN-P5 and TPAN-Ph with aggregation-induced emission (AIE) characteristics. While TPAN-P5 showed weak fluorescence emission at around 400 nm in tetrahydrofuran, increasing the water fraction (fw) from 0% to 95% induced a red shift of the emission peak to 502 nm, accompanied by enhanced aggregation and a maximum fluorescence intensity at fw = 95% (Fig. S22). TPAN-Ph displayed AIE, with its aggregated state exhibiting a fluorescence emission peak at 510 nm (Fig. S23). According to the CIE chromaticity diagram, TPAN-P5 transitioned from deep blue (0.16, 0.05) to blue-green (0.19, 0.40) with increasing aggregation, while the chromaticity coordinates of TPAN-Ph shifted from (0.16, 0.10) to (0.21, 0.46) (Fig. S24).
To further evaluate the influence of the pillar[5]arene cavity on the photoresponsive behavior of TPAN, we investigated the photophysical and photochemical properties of the two compounds in the aggregated state. The less densely packed TPAN within TPAN-P5 facilitated intramolecular cyclization, leading to the emergence of a new, albeit weak, absorption band at around 510 nm upon UV irradiation, a feature absent in TPAN-Ph (Fig. S25). Additionally, powder emission peaks were observed at 530 nm (TPAN-P5) and 502 nm (TPAN-Ph) (Fig. 2i), with a lower photoluminescence quantum yield (PLQY) for TPAN-P5 (18.74%) than for TPAN-Ph (27.37%) (Fig. S26). We attribute this diminished PLQY to the looser packing of TPAN in TPAN-P5, which facilitates intramolecular cyclization. This cyclization promotes absorption and restricts the AIE characteristics of TPAN, resulting in weaker fluorescence emission. To further validate this hypothesis, we calculated the free volume fraction (FVF) of TPAN-P5 and TPAN-Ph crystals. The results revealed that TPAN-P5 exhibited a higher FVF (35.01%) than TPAN-Ph (32.48%) (Fig. 3a and b). The larger FVF, correlating with a lower PLQY, suggests that the enhanced non-radiative decay pathways arising from vibrational relaxation in fluorescence emission provide more energy for photoinduced cyclization.
The electronic structure was further investigated using frontier orbital analysis based on density functional theory (DFT) calculations, aiming to elucidate how pillararenes promote the photocyclization of TPAN. In TPAN-P5, the highest occupied molecular orbital (HOMO, −5.58 eV) is predominantly localized on the pillararene framework, while the lowest unoccupied molecular orbital (LUMO, −1.97 eV) resides on the TPAN unit (Fig. 3c). This clear spatial separation establishes a distinct donor–acceptor partitioning, which supports a directional intramolecular charge-transfer (ICT) pathway upon excitation. Conversely, in TPAN-Ph, both the HOMO (−5.58 eV) and LUMO (−1.97 eV) are extensively co-localized across the entire molecular framework (Fig. 3d), indicating that its lowest-energy excitation is highly delocalized in nature. This suggests that TPAN-P5, owing to its ICT character, undergoes a substantial change in dipole moment upon photoexcitation, which, in turn, acts as a potent driving force to induce significant intramolecular charge polarization and concomitant structural rearrangements, thereby establishing optimal conditions for subsequent cyclization. Moreover, this notable dipole moment difference also renders the excited state particularly sensitive to solvent polarity. The resulting solvatochromic stabilization may drastically reduce the excited-state energy, consequently modulating both the reaction pathway and its activation barrier. In contrast, TPAN-Ph, upon delocalized excitation, shows a negligible change in dipole moment, lacks these advantageous attributes, and exhibits poor photocyclization efficiency. Additionally, owing to the ICT character induced by the pillar[5]arene acting as an electron donor, the excited-state lifetime of TPAN-P5 (7.5 ns) is considerably longer than that of TPAN-Ph (0.9 ns), thereby providing ample time for the cyclization to proceed (Fig. S27).
VOC-dependent photochromic behaviors of TPAN-P5versusTPAN-Ph in the solid state
Considering the susceptibility of TPAN-P5's large excited-state dipole moment to solvent polarity, various VOCs (such as toluene, bromoethane, 1,2-dichloroethane, CHCl3, CH2Cl2, CH3OH, CH3CH2OH, and H2O) were selected to promote TPAN cyclization, thereby further enhancing its photochromic performance in the aggregated state. Remarkably, only CH3OH vapor, under 365 nm UV irradiation, induced a pronounced pink coloration in TPAN-P5 powder, accompanied by a characteristic absorption peak at 510 nm in its UV-vis spectrum (Fig. 3e and S28). Furthermore, the 1H NMR spectrum of the CH3OH-fumigated TPAN-P5 (TPAN-P5–CH3OH) powder after 10 minutes of photoirradiation provided clear evidence of cyclization (Fig. S29). TPAN-P5–CH3OH powder exhibited reversible photochromism, reverting from pink to white upon exposure to white light (Fig. S30). In contrast, TPAN-Ph powder showed no photochromic response in analogous experiments, due to a negligible change in its excited-state dipole moment (Fig. 3f), indicating inhibited solid-state photocyclization. Similarly, exposure of the solid TPAN-Br monomer to CH3OH vapor did not induce coloration (Fig. S31). UV-vis absorption spectroscopy kinetics studies revealed the crucial influence of CH3OH on the photochromic behavior of TPAN-P5. Upon irradiation with 365 nm light, the TPAN-P5–CH3OH powder exhibited a significant and sustained enhancement of the absorption band in the 490–700 nm region within only ten minutes (Fig. 3g and S32). In stark contrast, the original TPAN-P5 powder lacked any noticeable dynamic changes in absorption under the same conditions (Fig. 3h and S32). Further kinetic analysis indicated that the TPAN-P5–CH3OH powder faded slowly in the dark, whereas visible-light irradiation significantly accelerated this fading (Fig. S33). Conversely, the original TPAN-P5 powder exhibited no measurable kinetic response under either dark or visible light conditions (Fig. S34).VOC-selective solid-state photochromism of TPAN-P5
To investigate the scope of this vapor-dependent photochromism, a broader range of VOCs was tested, such as acids, bases, aldehydes, ketones, and cycloalkanes. Among the various VOCs examined, only HCOOH, in addition to CH3OH, induced a visually discernible, amplified photochromic transformation from white to pink in the TPAN-P5 powder (Fig. S35). This transformation was also reversible upon visible light irradiation (Fig. S36). Other VOCs did not exhibit a comparable effect on TPAN-P5 photocyclization, showing only weak color changes, similar to those observed for the original TPAN-P5 powder (Fig. S37). Solid-state UV-vis absorption spectra of TPAN-P5 powders fumed with these other VOCs showed no significant peak or band at 510 nm (Fig. S38). Correspondingly, 1H NMR spectra also lacked characteristic signals for the cyclized product (Fig. S39). Furthermore, analogous to CH3OH-fumed TPAN-Ph and TPAN-Br powders, HCOOH also failed to enhance the aggregated-state photochromic ability of these compounds (Fig. S40), highlighting the necessity of the pillar-shaped cavities.To further define the characteristics of these VOCs, a range of alcohols and acids was examined. Remarkably, while CH3OH and HCOOH readily facilitated TPAN-P5 photocyclization, other alcohols and acids with larger molecular sizes (including CH3CH2OH, ethylene glycol, 1,2-propanediol, isopentanol, isobutanol, and acetic acid (HAc)) did not yield such a stable pink color change or a distinct spectroscopic signature (Fig. S41 and S42). This compelling observation strongly suggests that the VOC-induced photochromism of TPAN-P5 powder is not solely determined by interactions with VOC molecules. Instead, synergistic effects involving both the geometric size and the precise electronic influence of the VOCs are essential.
Synergy between intermolecular interactions and structural order in solid-state photochromism of TPAN-P5
Building upon the observed VOC-dependent photochromism, we hypothesize that matched molecular dimensions enable specific VOCs to efficiently intercalate into specific microenvironments within the TPAN-P5 aggregates (e.g., micropores or interfacial voids), rather than simply forming disordered aggregates. Concurrently, the intrinsic dipole moments of the VOC molecules, through the induction of local microenvironmental electric fields, fine-tune the electronic structure and energy-level distribution of TPAN-P5. This concerted effect ultimately generates an optimal molecular conformation and electronic state conducive to an efficient photocyclization reaction under 365 nm light excitation. To validate this hypothesis, we exposed TPAN-P5 powder to CH3OH vapor and monitored the resulting color change via photography at various exposure times (Fig. 4a). Simultaneously, we quantified the CH3OH content in the powder to correlate adsorption with photochromic behavior. Appreciable photochromism was observed in TPAN-P5 after only 0.5 hours of CH3OH adsorption, with no significant increase in intensity after 5 hours of continued exposure. 1H NMR spectra of these powders in CDCl3 revealed a new signal at 3.48 ppm, attributable to the –CH3 protons of CH3OH (Fig. S43). While the chemical shift of the –CH3 protons remained constant with increasing exposure time, the signal exhibited splitting, suggesting intermolecular interactions between CH3OH and TPAN-P5. Moreover, based on a CH3OH content of 0.60 mol per mol of TPAN-P5 (Fig. S44), we have further inferred that CH3OH is unlikely to be tightly encapsulated within the TPAN-P5 cavity.Additionally, powder X-ray diffraction (PXRD) measurements were performed on TPAN-P5 powders treated with various VOCs to investigate whether VOC-induced changes in the packing mode significantly influence the photochromic properties (Fig. S45). The PXRD patterns revealed distinct crystalline phases in TPAN-P5 upon exposure to CH3OH and HCOOH, both of which are known to induce photochromism. Specifically, TPAN-P5–CH3OH powder exhibited a crystalline phase corresponding to the thermodynamically stable packing of crystalline TPAN-P5. In contrast, this stable phase was not observed in the powder exposed to HCOOH. Furthermore, while cyclohexane- and triethylamine-fumigated TPAN-P5 powders showed weak reflections indicative of a packing mode similar to that of the thermodynamically stable phase, no visible pink coloration was observed. Additionally, the absence of visible pink coloration in the amorphous TPAN-P5 powder obtained by grinding underscores the importance of structural organization (Fig. S45 and S46). These results suggest that, while not dictated by a specific crystalline packing mode, photochromism of TPAN-P5 is facilitated by the order within the framework, presumably because it provides the necessary molecular environment, including sufficient spatial freedom, for photochromic transformation.
To distinguish the respective roles of CH3OH-mediated intermolecular interactions and structural order in the photochromism of TPAN-P5 powder, TPAN-P5–CH3OH powder was subjected to sequential heating at elevated temperatures (110, 120, and 130 °C) for a total of 72 hours. 1H NMR analysis revealed the presence of residual CH3OH after heating (Fig. S47), indicating strong intermolecular interactions between CH3OH and TPAN-P5. Remarkably, despite the significant loss of CH3OH and the consequent disruption of long-range order (Fig. S48), the heated powder retained detectable photochromic activity (Fig. S49). This sharply contrasts with the amorphous TPAN-P5 powder obtained by mechanical grinding, as mentioned earlier, which showed no photochromism. The critical divergence lies in the presence of residual CH3OH in the heated sample, which sustains intermolecular interactions even in a disordered state. These results demonstrate that CH3OH-derived intermolecular interactions are indispensable for initiating the photochromic response, while structural order primarily serves as a secondary enhancer, facilitating more effective CH3OH anchoring and the spatial arrangement of reactive sites. In light of the preceding analysis, the robust photochromism of TPAN-P5 is fundamentally attributed to the spatial freedom provided by its molecular pillar-shaped cavities. Structurally, the pillararene component serves as a spatial “blocker”, hindering close packing of TPAN moieties and generating voids around the reactive sites. Within these voids, CH3OH molecules may establish hydrogen bonds or other dipole–dipole interactions that promote the formation of reversible covalent bonds between adjacent benzene rings within the TPAN moiety.
Structural basis of VOC-mediated pre-reactive conformations for photocyclization
TPAN-P5–CH3OH co-crystals (Table S2) were prepared and characterized using a synchrotron radiation source to further refine the function of CH3OH. Consistent with the 1H NMR data for TPAN-P5–CH3OH powder, disordered CH3OH molecules surrounded the TPAN moiety at the end of TPAN-P5 without penetrating the cavity (Fig. 4b, the disordered CH3OH is shown with two possible orientations). C–H⋯N (2.562 Å), C–H⋯O (2.616 Å), and O–H⋯N (3.075 Å) interactions existed between CH3OH and the TPAN-P5 molecule (Fig. 4c). The terphenyl unit exhibited twist angles of 59.79° and 61.30° (Fig. 4d and S50), with distances of 3.338 and 3.408 Å between the bonding benzene rings in the TPAN-P5–CH3OH crystal, which are geometrically conducive to the photoisomerization reaction. This confirms that CH3OH mediates the formation of a pre-reactive conformation of TPAN-P5 through polar intermolecular interactions, thereby facilitating initial photoinduced cyclization.To further validate this mechanism of conformation induction, we sought to grow additional photochromic crystals with improved structural clarity. We selected ethyl acetate (EA), a suitable TPAN-P5 solvent, as a functional substitute for CH3OH. EA-fumigated TPAN-P5 (TPAN-P5–EA) powder resulted in a pink color change upon 365 nm UV irradiation (Fig. S51 and S52). The lack of chemical shift changes in TPAN-P5 and EA, coupled with an EA![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TPAN-P5 adsorption stoichiometry of 1.38
TPAN-P5 adsorption stoichiometry of 1.38![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (Fig. S53), supports an EA-induced photochromic mechanism in TPAN-P5, analogous to that of CH3OH. The complete abolition of photochromism upon thermal removal of EA at 110 °C (Fig. S54), coupled with the restoration of ordered TPAN-P5 packing (Fig. S55), further demonstrates that the photochromism in TPAN-P5–EA powder, much like that in TPAN-P5–CH3OH powder, stems from the intermolecular interactions between EA and TPAN-P5 molecules.
1 (Fig. S53), supports an EA-induced photochromic mechanism in TPAN-P5, analogous to that of CH3OH. The complete abolition of photochromism upon thermal removal of EA at 110 °C (Fig. S54), coupled with the restoration of ordered TPAN-P5 packing (Fig. S55), further demonstrates that the photochromism in TPAN-P5–EA powder, much like that in TPAN-P5–CH3OH powder, stems from the intermolecular interactions between EA and TPAN-P5 molecules.
TPAN-P5–EA co-crystals were further cultured to investigate the specific intermolecular interactions that promote aggregated-state photochromism (Table S2 and Fig. S56). EA molecules were found within the pentagonal cavity of TPAN-P5 and in the space surrounding the TPAN group (Fig. 4e). To further estimate and visualize the weak interactions between EA and TPAN-P5, we performed an independent gradient model (IGM) analysis. The resulting TPAN-P5–EA binding iso-surfaces (green and blue regions in Fig. 4f and S57) revealed weak interactions concentrated not only at the electron-deficient guest edge within the cavity, but also at reactive TPAN sites, such as the cyclizing benzene rings and the cyano group. Further structural analysis indicated that EA formed numerous weak interactions with TPAN-P5, as we had anticipated. These interactions primarily arise from C–H⋯π (3.103 Å), C–H⋯N (2.737 Å), and C–H⋯O (2.591 Å and 2.504 Å) forces between TPAN-P5 and EA (Fig. 4g and S58). Consequently, the dihedral angles between TPAN and the pillararene were measured to be 59.47° and 81.03°, with corresponding distances between the bonding benzene rings of TPAN of 3.158 and 3.294 Å (Fig. 4h). These suitable dihedral angles and short distances between the bonding benzene rings ensure photochromism. Moreover, the incorporation of EA into the crystal matrix led to discernible multiple orientations of TPAN-P5 (Fig. 4i) and triggered a space group transformation from the highly symmetric P2/n to the less symmetric P21/n (Tables S1 and S2). These structural changes, driven by specific supramolecular interactions, collectively induced an electronic rearrangement in TPAN, thereby facilitating the photocyclization reaction.
To elucidate the structural factors governing VOC-induced photochromism in TPAN-P5, we determined and compared the crystal structures of TPAN-P5 co-crystallized with non-photochromic CH3CH2OH and CH3COOH (TPAN-P5–CH3CH2OH and TPAN-P5–CH3COOH) and with photochromic CH3OH and EA. TPAN-P5 molecules in TPAN-P5–CH3CH2OH and TPAN-P5–CH3COOH co-crystals exhibited a distorted conformation. The pillararene cavity contracted, transitioning from an approximate five-fold to an asymmetric symmetry (Fig. 5a and b), a form distinct from the conformations observed in CH3OH and EA-induced crystals. Conversely, in TPAN-P5–CH3CH2OH and TPAN-P5–CH3COOH co-crystals, TPAN-P5 molecules adopted a staggered packing that lacks the continuous one-dimensional channels found in the CH3OH and EA systems (Fig. 5c–f).
Taking the TPAN-P5–CH3CH2OH co-crystal as an example, we define the TPAN moiety with the smaller terphenyl dihedral angle (53.89°) as ‘proximal TPAN’ (closer to the macrocycle) and the one with the larger dihedral angle (59.15°) as ‘distal TPAN’ (further from the macrocycle) (Fig. 5g and S59). Although the bonding distances between the reactive arene rings (3.314 and 3.283 Å) met preorganization criteria, the proximal TPAN was sterically hindered by the pillararene cavity. This hindering conformation was further stabilized by short C–H⋯N (2.656 Å) and C–H⋯O (2.585 and 2.637 Å) contacts with neighboring TPAN-P5 molecules (Fig. 5h–j and S59). Collectively, these factors impede the arene torsion and inward adjustment required for cyclization. The packing pattern and unit cell parameters of the TPAN-P5–CH3COOH co-crystal are highly similar to those of the TPAN-P5–CH3CH2OH co-crystal (Table S2, TPAN-P5–CH3COOH lattice parameters: a = 45.8868 Å, b = 13.1150 Å, c = 33.8049 Å, α = 90°, β = 132.234°, γ = 90°; TPAN-P5–CH3CH2OH lattice parameters: a = 45.8525 Å, b = 13.1186 Å, c = 33.5659 Å, α = 90°, β = 132.1990°, γ = 90°). Within this crystal packing context, TPAN-P5 molecules formed multiple intermolecular C–H⋯O (2.638 and 2.609 Å) and C–H⋯N (2.665 Å) interactions. This dense packing, mediated by these interactions, inhibits cyclization by restricting the necessary structural reorganization (Fig. S60).
We used S1-state natural transition orbital (NTO) calculations on the four VOC-induced TPAN-P5 conformers to explain how configuration affects photochromism at the electronic level. Upon excitation, all four conformers can transfer electrons from the electron-rich cavity to the flanking TPAN units (Fig. 5k–n). Increased electron density on the TPAN units is a prerequisite for bond formation, with unhindered TPAN twisting being a necessary condition. In the symmetric CH3OH- and EA-induced TPAN-P5 conformers, the excited-state electron-rich TPAN units are free from steric hindrance by the pillar-shaped cavity, enabling unimpeded twisting and bond formation and resulting in photochromism in the aggregated state. Conversely, the twisted conformations of the CH3CH2OH- and CH3COOH-exposed TPAN-P5 lead to excited-state electron localization predominantly on the proximal TPAN unit. The pillar-shaped cavity shields this TPAN unit, preventing free twisting and thus inhibiting photochromism.
Material applications
TPAN-P5's multi-stimuli-response, rapid photoresponse, and anti-fatigue properties make it suitable for UV printing, anti-counterfeiting, and information encryption. To test its use in UV printing, we created a transparent poly(methyl methacrylate) (PMMA) film with a 2 wt% TPAN-P5 doping ratio and a thickness of 0.7 µL mm−2 (Fig. S61), which provided optimal photochromic performance. The film was then used in a UV printing test: a plastic stencil was placed on it, and the exposed area was irradiated with 365 nm light for 10 seconds. The areas not covered by the stencil turned a readily visible pink color, while the covered regions remained unchanged. Furthermore, the film could be returned to its initial state within seconds upon exposure to white light, allowing for continued information input and editing using a UV light source (Fig. 6a). Utilizing the erasable nature of the TPAN-P5-containing PMMA film, pink “dog paw” and “shamrock” patterns were printed.The visually discernible, delayed fading observed under ambient daylight further enhances its practical applicability (Fig. 6b). TPAN-P5 powder exhibits a remarkable photochromic behavior that can be switched “on” and “off” by exposure to CH3OH, suggesting its applicability for anti-counterfeiting. As illustrated in Fig. 6c, we fabricated a white “dog paw” model using TPAN-P5 powder, with some areas exposed to CH3OH vapor and others left untreated. Upon UV irradiation, the regions treated with CH3OH vapor exhibited a readily discernible pink color, while the remaining areas retained white. The photochromic area can be precisely controlled by adjusting the amount of CH3OH-fumed powder applied. This method's excellent spatial resolution was demonstrated by selectively changing the color of a paw print from one “toe” to two. This suggests that TPAN-P5 powder could be used as an on-demand anti-counterfeiting marker on items such as banknotes, passports, ID cards, and packaging to enhance consumer and public safety.
Although QR codes are a popular choice for quick information access, thanks to their high storage capacity and rapid readability,28,72,73 many encryption applications require data to be concealed or delayed, a limitation of the technology. TPAN-P5 powder addresses these challenges by exhibiting a significant color change, prolonged bleaching times, and sustained color retention only when exposed to specific VOC vapors and UV light. This dual-activation mechanism, attributed to multiple layers of information encryption, enhances information security. A 3D-printed white QR code was rendered active by filling it with TPAN-P5 powder and exposing it to CH3OH fumes (Fig. 6d). Upon 365 nm UV irradiation, the QR code rapidly turned pink, revealing its embedded information—a Jilin University logo that could be scanned with a mobile phone. The QR code was non-functional without the fuming process.
Conclusions
In summary, we described a pillararene-based strategy for enhancing molecular free volume, leading to the development of TPAN-P5, a macrocyclic arene with pronounced photochromic properties. TPAN-P5 amplifies the photoisomerization characteristics of the TPAN chromophore, exhibiting superior photochromism and color retention compared to TPAN-Ph and the isolated chromophore itself. The pillararene cavity in TPAN-P5 generates a larger TPAN torsional angle, disrupting π–π stacking and creating molecular free volume that facilitates both photoisomerization and VOC intercalation. This, coupled with TPAN-P5's prominent ICT effect, resulting in a significant dipole moment difference between its excited and ground states, renders TPAN-P5 powder selectively susceptible to VOC-induced photochromism via superstructure formation. Mechanistically, polar interactions between VOCs and TPAN-P5 increase TPAN electron density, promoting TPAN's twisting and bond formation, thereby enabling solid-state photochromism. These molecular characteristics render TPAN-P5 a promising candidate for applications in UV printing, anti-counterfeiting, and multiple information encryption. This work highlights the potential of pillararenes as molecular property amplifiers, offering valuable insights for the design of photochromic molecules and the advancement of macrocyclic arene applications.Author contributions
M.-H. L. and Y.-W. Y. conceived this project and designed the experiments. M.-H. L., H. H., and Y. W. conducted the experiments. M.-H. L. drafted the work. Y.-W. Y. revised and finalised the manuscript. All authors approved the submitted version.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article are available in the SI file, which includes experimental procedures, spectroscopic and crystallographic data, photophysical characterization, and computational details. See DOI: https://doi.org/10.1039/d5sc08539j.CCDC 2379600, 2379605, 2380579, 2448876, 2488767 and 2488768 contain the supplementary crystallographic data for this paper.74a–f
Acknowledgements
The authors acknowledge the National Natural Science Foundation of China (No. 22571119) and the Key Research and Development Project of the Natural Science Foundation of Jilin Province (No. 20260203095SF) for financial support. We thank the staff of the Shanghai Synchrotron Radiation Facility (SSRF) and the User Experiment Assist System for their expert support during data acquisition.Notes and references
- Y. Kobayashi and J. Abe, Chem. Soc. Rev., 2022, 51, 2397–2415 RSC.
- T. Zhang, X.-Y. Lou, X. Li, X. Tu, J. Han, B. Zhao and Y.-W. Yang, Adv. Mater., 2023, 35, 2210551 CrossRef CAS PubMed.
- G. Berkovic, V. Krongauz and V. Weiss, Chem. Rev., 2000, 100, 1741–1754 CrossRef CAS PubMed.
- N. Tamai and H. Miyasaka, Chem. Rev., 2000, 100, 1875–1890 CrossRef CAS PubMed.
- N. F. König, D. Mutruc and S. Hecht, J. Am. Chem. Soc., 2021, 143, 9162–9168 CrossRef PubMed.
- H. Tian and S. Yang, Chem. Soc. Rev., 2004, 33, 85–97 RSC.
- C.-L. Wong, M. Ng, E. Y.-H. Hong, Y.-C. Wong, M.-Y. Chan and V. W.-W. Yam, J. Am. Chem. Soc., 2020, 142, 12193–12206 CrossRef CAS PubMed.
- D. J. Wales, Q. Cao, K. Kastner, E. Karjalainen, G. N. Newton and V. Sans, Adv. Mater., 2018, 30, 1800159 CrossRef PubMed.
- M. Estrader, J. S. Uber, L. A. Barrios, J. Garcia, P. Lloyd-Williams, O. Roubeau, S. J. Teat and G. Aromí, Angew. Chem., Int. Ed., 2017, 56, 15622–15627 CrossRef CAS PubMed.
- R. C. Shallcross, P. O. Körner, E. Maibach, A. Köhnen and K. Meerholz, Adv. Mater., 2013, 25, 4807–4813 CrossRef CAS PubMed.
- Y. Yang, J. He, Z. He and G. Jiang, Adv. Opt. Mater., 2021, 9, 2001584 CrossRef CAS.
- S. N. Corns, S. M. Partington and A. D. Towns, Color. Technol., 2009, 125, 249–261 CAS.
- J. Lott, C. Ryan, B. Valle, J. R. Johnson iii, D. A. Schiraldi, J. Shan, K. D. Singer and C. Weder, Adv. Mater., 2011, 23, 2425–2429 CrossRef CAS PubMed.
- Z. Li, X. Zeng, C. Gao, J. Song, F. He, T. He, H. Guo and J. Yin, Coord. Chem. Rev., 2023, 497, 215451 CrossRef CAS.
- C. Jia, A. Migliore, N. Xin, S. Huang, J. Wang, Q. Yang, S. Wang, H. Chen, D. Wang, B. Feng, Z. Liu, G. Zhang, D.-H. Qu, H. Tian, M. A. Ratner, H. Q. Xu, A. Nitzan and X. Guo, Science, 2016, 352, 1443–1445 CrossRef CAS PubMed.
- M. Li and W.-H. Zhu, Acc. Chem. Res., 2022, 55, 3136–3149 CrossRef CAS PubMed.
- H.-B. Cheng, S. Zhang, E. Bai, X. Cao, J. Wang, J. Qi, J. Liu, J. Zhao, L. Zhang and J. Yoon, Adv. Mater., 2022, 34, 2108289 CrossRef CAS PubMed.
- Z. Xiong, X. Zhang, L. Liu, Q. Zhu, Z. Wang, H. Feng and Z. Qian, Chem. Sci., 2021, 12, 10710–10723 RSC.
- D. Zhang, Y. Zhang, W. Gong, J. Li, S. Liu, Y. Ma and Q. Zhao, Adv. Opt. Mater., 2023, 11, 2300386 CrossRef CAS.
- D. Ou, T. Yu, Z. Yang, T. Luan, Z. Mao, Y. Zhang, S. Liu, J. Xu, Z. Chi and M. R. Bryce, Chem. Sci., 2016, 7, 5302–5306 RSC.
- Y. Fan, M. Han, A. Huang, Q. Liao, J. Tu, X. Liu, B. Huang, Q. Li and Z. Li, Mater. Horiz., 2022, 9, 368–375 RSC.
- M. Luo, J. Zhao, Q. Yan, C. Li, Y. Liu, Z. Yang, Z. Mao, S. Wang and Z. Chi, Chem. Eng. J., 2024, 499, 156214 CrossRef CAS.
- Y. Xiao, M. Shen, J. Li, H. Wang, H. Sun, Y. He, R. Huang, T. Yu and W. Huang, Adv. Funct. Mater., 2024, 34, 2312930 CrossRef CAS.
- Y. Xiao, J. Li, Z. Song, J. Liao, M. Shen, T. Yu and W. Huang, J. Am. Chem. Soc., 2025, 147, 20372–20380 CrossRef CAS PubMed.
- Z. Xie, X. Zhang, Y. Xiao, H. Wang, M. Shen, S. Zhang, H. Sun, R. Huang, T. Yu and W. Huang, Adv. Mater., 2023, 35, 2212273 Search PubMed.
- T. J. White, in Photomechanical Materials, Composites, and Systems, ed. T. J. White, John Wiley & Sons, Inc., Hoboken, NJ, 2017, pp. 393–403 Search PubMed.
- D. E. Williams, C. R. Martin, E. A. Dolgopolova, A. Swifton, D. C. Godfrey, O. A. Ejegbavwo, P. J. Pellechia, M. D. Smith and N. B. Shustova, J. Am. Chem. Soc., 2018, 140, 7611–7622 CrossRef CAS PubMed.
- G. Huang, Q. Xia, W. Huang, J. Tian, Z. He, B. S. Li and B. Z. Tang, Angew. Chem., Int. Ed., 2019, 58, 17814–17819 CrossRef CAS PubMed.
- Z. Liu, S. K. M. Nalluri and J. F. Stoddart, Chem. Soc. Rev., 2017, 46, 2459–2478 RSC.
- J. Li, D. Yim, W.-D. Jang and J. Yoon, Chem. Soc. Rev., 2017, 46, 2437–2458 RSC.
- M. T. Sikder, M. M. Rahman, M. Jakariya, T. Hosokawa, M. Kurasaki and T. Saito, Chem. Eng. J., 2019, 355, 920–941 CrossRef CAS.
- Y.-H. Liu, Y.-M. Zhang, H.-J. Yu and Y. Liu, Angew. Chem., Int. Ed., 2021, 60, 3870–3880 CrossRef CAS PubMed.
- R. D. Mukhopadhyay and K. Kim, Nat. Chem., 2023, 15, 438 CrossRef CAS PubMed.
- C. D. Gutsche, Acc. Chem. Res., 1983, 16, 161–170 CrossRef CAS.
- T. Ogoshi, S. Kanai, S. Fujinami, T.-a. Yamagishi and Y. Nakamoto, J. Am. Chem. Soc., 2008, 130, 5022–5023 CrossRef CAS PubMed.
- M. Xue, Y. Yang, X. Chi, Z. Zhang and F. Huang, Acc. Chem. Res., 2012, 45, 1294–1308 CrossRef CAS PubMed.
- T. Ogoshi, T.-a. Yamagishi and Y. Nakamoto, Chem. Rev., 2016, 116, 7937–8002 CrossRef CAS PubMed.
- H. Ju, C. N. Zhu, H. Wang, Z. A. Page, Z. L. Wu, J. L. Sessler and F. Huang, Adv. Mater., 2022, 34, 2108163 CrossRef CAS PubMed.
- T. Ogoshi, S. Takashima and T.-a. Yamagishi, J. Am. Chem. Soc., 2018, 140, 1544–1548 CrossRef CAS PubMed.
- H. Nie, Z. Wei, X.-L. Ni and Y. Liu, Chem. Rev., 2022, 122, 9032–9077 Search PubMed.
- Z. Liu and Y. Liu, Chem. Soc. Rev., 2022, 51, 4786–4827 RSC.
- X.-Y. Lou, S. Zhang, Y. Wang and Y.-W. Yang, Chem. Soc. Rev., 2023, 52, 6644–6663 RSC.
- A. Blanco-Gómez, P. Cortón, L. Barravecchia, I. Neira, E. Pazos, C. Peinador and M. D. García, Chem. Soc. Rev., 2020, 49, 3834–3862 RSC.
- G. Wu and Y.-W. Yang, Cell Rep. Phys. Sci., 2024, 5, 101873 Search PubMed.
- C.-M. Yu, X. Meng, X. Liu, Z.-Y. Zhang and C. Li, Chem. Mater., 2022, 34, 358–365 CrossRef CAS.
- S. Song, H. Zhang and Y. Liu, Acc. Mater. Res., 2024, 5, 1109–1120 CrossRef CAS.
- X.-H. Wang, N. Song, W. Hou, C.-Y. Wang, Y. Wang, J. Tang and Y.-W. Yang, Adv. Mater., 2019, 31, 1903962 Search PubMed.
- Y. Qin, X. Liu, P.-P. Jia, L. Xu and H.-B. Yang, Chem. Soc. Rev., 2020, 49, 5678–5703 Search PubMed.
- P. Spenst, R. M. Young, B. T. Phelan, M. Keller, J. Dostál, T. Brixner, M. R. Wasielewski and F. Würthner, J. Am. Chem. Soc., 2017, 139, 2014–2021 CrossRef CAS PubMed.
- Y.-D. Yang, X.-L. Chen, J. Liang, J.-W. Fang, J. L. Sessler and H.-Y. Gong, J. Am. Chem. Soc., 2023, 145, 14010–14018 Search PubMed.
- J. Huang, H. Feng, L. Zhang and K. Jie, Angew. Chem., Int. Ed., 2025, 64, e202500022 CrossRef CAS PubMed.
- K. Omoto, S. Tashiro and M. Shionoya, J. Am. Chem. Soc., 2021, 143, 5406–5412 CrossRef CAS PubMed.
- M.-H. Li, C. Xu and Y.-W. Yang, Coord. Chem. Rev., 2024, 512, 215894 Search PubMed.
- X.-Y. Lou, G. Zhang, M.-H. Li and Y.-W. Yang, Nano Lett., 2023, 23, 1961–1969 Search PubMed.
- M.-H. Li, Z. Yang, H. Hui, B. Yang, Y. Wang and Y.-W. Yang, Angew. Chem., Int. Ed., 2023, 62, e202313358 Search PubMed.
- Z. Li, Z. Yang, Y. Zhang, B. Yang and Y.-W. Yang, Angew. Chem., Int. Ed., 2022, 61, e202206144 Search PubMed.
- Q. Li, Y. Wu, J. Cao, Y. Liu, Z. Wang, H. Zhu, H. Zhang and F. Huang, Angew. Chem., Int. Ed., 2022, 61, e202202381 Search PubMed.
- Q. Li, H. Zhu and F. Huang, J. Am. Chem. Soc., 2019, 141, 13290–13294 CrossRef CAS PubMed.
- Z. Li and Y.-W. Yang, Adv. Mater., 2022, 34, 2107401 CrossRef CAS PubMed.
- H. Zhu, Q. Li, W. Zhu and F. Huang, Acc. Mater. Res., 2022, 3, 658–668 CrossRef CAS.
- X.-Y. Lou, G. Zhang, N. Song and Y.-W. Yang, Biomaterials, 2022, 286, 121595 Search PubMed.
- C.-L. Song, Z. Li, J.-R. Wu, T. Lu and Y.-W. Yang, Chem. Mater., 2022, 34, 10181–10189 CrossRef CAS.
- H. Zhang, Z. Liu, F. Xin and Y. Zhao, Coord. Chem. Rev., 2020, 420, 213425 CrossRef CAS.
- G. Zhang, B. Hua, A. Dey, M. Ghosh, B. A. Moosa and N. M. Khashab, Acc. Chem. Res., 2021, 54, 155–168 CrossRef CAS PubMed.
- Z.-Q. Wang, X. Wang and Y.-W. Yang, Adv. Mater., 2024, 36, 2301721 CrossRef CAS PubMed.
- Y. Wu, L. Shi, L. Xu, J. Ying, X. Miao, B. Hua, Z. Chen, J. L. Sessler and F. Huang, Nature, 2025, 640, 676–682 CrossRef CAS PubMed.
- H. Zhang, Z. Liu and Y. Zhao, Chem. Soc. Rev., 2018, 47, 5491–5528 RSC.
- T.-H. Shi, S. Ohtani, K. Kato, S. Fa and T. Ogoshi, Trends Chem., 2023, 5, 537–550 CrossRef CAS.
- J.-R. Wu, G. Wu, D. Li and Y.-W. Yang, Angew. Chem., Int. Ed., 2023, 62, e202218142 CrossRef CAS PubMed.
- Y.-K. Zhu, A. Liu, G. Wu, L. Yuan, Y.-H. Jin, M.-H. Li, X. Wang and Y.-W. Yang, Angew. Chem., Int. Ed., 2025, e202513299 CAS.
- D. Zou, Z. Li, D. Long, X. Dong, H. Qu, L. Yang and X. Cao, ACS Appl. Mater. Interfaces, 2023, 15, 13545–13553 Search PubMed.
- S. Dong, Q. Zang, Z.-Y. Ma, M. Tang, Z.-K. Xu, J. Nie, B. Du, J. Z. Sun and B. Z. Tang, ACS Appl. Mater. Interfaces, 2022, 14, 17794–17805 Search PubMed.
- P. Carou-Senra, L. Rodríguez-Pombo, A. Awad, A. W. Basit, C. Alvarez-Lorenzo and A. Goyanes, Adv. Mater., 2024, 36, 2309164 CrossRef CAS PubMed.
- (a) CCDC 2379600: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2kw59y; (b) CCDC 2379605: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2kw5g3; (c) CCDC 2380579: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2kx5wk; (d) CCDC 2448876: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n6805; (e) CCDC 2488767: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pjrtt; (f) CCDC 2488768: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pjrvv.
| This journal is © The Royal Society of Chemistry 2026 |