Open Access Article
Open Access ArticleDynamic-structural-distortion of spheroidene activates a hidden 3Ag− state mediating carotenoid-to-bacteriochlorophyll energy transfer in a light-harvesting 2 complex
Bo
Peng†
a,
Mingqing
Chen†
b,
Tengfei
Ma†
a,
Yifan
Huang
b,
Peng
Wang
 *b and
Weimin
Liu
*b and
Weimin
Liu
 *a
*a
aSchool of Physical Science and Technology, ShanghaiTech University, Shanghai 201210, China. E-mail: liuwm@shanghaitech.edu.cn
bKey Laboratory of Advanced Light Conversion Materials and Biophotonics, School of Chemistry and Life Resources, Renmin University of China, Beijing 100872, P. R. China. E-mail: wpeng_chem@ruc.edu.cn
First published on 3rd February 2026
Abstract
Carotenoids extend the absorption range of photosynthesis and transfer excitation energy to (bacterio-)chlorophylls with remarkable efficiency, yet the microscopic mechanism of this process, especially the role of the SX intermediate, remains unresolved. Here, we use femtosecond stimulated Raman spectroscopy, whose high vibrational frequency and temporal resolutions enable direct tracking of excited-state intermediates and their symmetry characteristics. By probing spheroidene in both solution and the light-harvesting 2 complex of Rhodobacter sphaeroides, we reveal structural change in the S2 (1Bu+) state that forms distorted SX and S1 (2Ag−) intermediates. The SX state is assigned to optically forbidden 3Ag− configuration rather than the earlier 1Bu− or Ag+ proposals and is identified as an efficient pathway for energy transfer to bacteriochlorophylls. The spheroidene-to-bacteriochlorophyll energy transfer efficiencies are quantified as 32% via the SX state, combined with 50% from the S2 state and 12% from the S1 state, yielding an overall transfer efficiency of 94%, in excellent agreement with previous reports. We propose that the observed structural distortions of spheroidene dynamically enhance coulombic coupling with surrounding bacteriochlorophylls, which may underlie the remarkably high efficiency of excitation energy transfer.
Introduction
Carotenoids are essential for photosynthesis, where they harvest solar energy and transfer it to (bacterio-)chlorophylls with remarkable efficiency.1 Despite decades of study, the microscopic mechanisms remain elusive, particularly the role of intermediate optically forbidden states and their role in facilitating ultrafast energy transfer. The bacterial light-harvesting 2 (LH2) complex provides an ideal system to address this challenge.2,3 In the Rhodobacter sphaeroides LH2 complex, ultrafast spectroscopy has shown that the spheroidene transfers excitation energy to neighboring BChls with efficiencies exceeding >90%.4–7 However, the contribution of the SX intermediate and the precise nature of its electronic symmetry remain controversial.7–16 Previous experiments assigned an optically forbidden 1Bu− symmetry to this SX state;9–12 the involvement of the optically forbidden 1Bu− state reconciled with the theoretical calculation, which positions it between optically allowed S2 (1Bu+) and optically forbidden S1 (2Ag−) states for the conjugation length N = 10.13–15 By contrast, other theoretical studies proposed an optically forbidden Ag+ assignment,16 leaving both the nature and the functional role of the SX state unresolved. This controversy arises largely because experimentally determining the precise symmetry of the intermediate state between S2 and S1 remains highly challenging.In this work, we address this challenge by employing femtosecond stimulated Raman spectroscopy (FSRS)17,18 to investigate the real-time structural evolution of spheroidene in both solution and the intact LH2 complex extracted from Rhodobacter sphaeroides 2.4.1. FSRS offers distinct advantages over conventional electronic spectroscopy by simultaneously providing high temporal and spectral resolution, enabling direct observation of individual excited-state intermediates.19 Moreover, the symmetry properties of carotenoid excited states can be disentangled via characteristic vibrational modes. For instance, Raman signals near 1800 cm−1 have been linked to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibronic coupling mode involving Ag− symmetry,17,18,20–25 while additional features reveal C
C stretching vibronic coupling mode involving Ag− symmetry,17,18,20–25 while additional features reveal C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching diabatic mixing between Bu+ and Ag− (or Bu−) states.13,17,26–28 These capabilities establish FSRS as a powerful tool for probing the structural and symmetry evolution of photoexcited carotenoids.
C stretching diabatic mixing between Bu+ and Ag− (or Bu−) states.13,17,26–28 These capabilities establish FSRS as a powerful tool for probing the structural and symmetry evolution of photoexcited carotenoids.
Leveraging these strengths, we discovered a functional SX state between the S2 (1Bu+) and S1 (2Ag−) states that does not conform to the previously proposed 1Bu− or Ag+ assignments, but instead corresponds to an optically forbidden 3Ag− state. This state arises from photoinduced structural distortions in the S2 (1Bu+) state, leading to formation of twisted SX (3Ag−) and S1 (2Ag−) states. Importantly, we show that the SX state contributes 32% to excitation energy transfer to BChls, together with 50% from S2 and 12% from S1, yielding a total transfer efficiency of 94% in excellent agreement with previous reports.4–7
Methods
Sample preparation
Tris(hydroxymethyl)aminomethane (Tris) (>99%) and diethylaminoethyl cellulose (DEAE-52) were purchased from Solarbio Life Science (Beijing), inorganic salts (both A. R.) were purchased from Sinopharm Chemical Reagent Beijing Co., and lauryl dimethylamine oxide (LDAO, 30%) and organic solvent were purchased from Sigma-Aldrich.The bacterial cultivation and protein purification processes were similar to those described in a previous report.29Rhodobacter (Rba.) sphaeroides 2.4.1 cells were grown in M22+ medium at 30 °C under light for 3 days anaerobically and were harvested by centrifugation.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond number 10 (N = 10) was extracted and purified from Rba. sphaeroides 2.4.1 cells following a previous protocol.31 A methanol-acetone mixture (2
C bond number 10 (N = 10) was extracted and purified from Rba. sphaeroides 2.4.1 cells following a previous protocol.31 A methanol-acetone mixture (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7, v/v) was used to extract the pigment from wet cells several times. The obtained dry pigment mixture was dissolved in n-hexane and purified by Al2O3 column chromatography using a gradient n-hexane-diethyl ether mixture as the eluent. All-trans-spheroidene was recrystallized before further use in spectroscopic measurements.
7, v/v) was used to extract the pigment from wet cells several times. The obtained dry pigment mixture was dissolved in n-hexane and purified by Al2O3 column chromatography using a gradient n-hexane-diethyl ether mixture as the eluent. All-trans-spheroidene was recrystallized before further use in spectroscopic measurements.
Transient absorption spectroscopy
A commercial spectrometer (Helios Fire, Ultrafast System) is used to measure the femtosecond transient absorption spectra. A Ti:sapphire laser amplifier system (Astrella, Coherent, Inc., 35 fs, 7 W, 1 kHz) is used to produce the 800 nm fundamental beam. An actinic pump, which is at 480 nm for the experiment of spheroidene in n-hexane solution, 500 nm for the experiment of spheroidene in DMSO solution and 510 nm for the experiment of the LH2 complex, is generated using an optical parametric amplifier (OPerA Solo, Coherent, Inc.). The supercontinuum white light generated by focusing the 800 nm fundamental beam on a sapphire crystal plate worked as the probe pulse with a range from 470 to 640 nm. The instrument response function (IRF) of the system is ∼120 fs.Femtosecond stimulated Raman spectroscopy
We obtained a tunable narrowband picosecond (ps) Raman pump, a broadband femtosecond (fs) Raman probe, and an fs actinic pump by splitting the fundamental laser pulses (Coherent, Astrella, 35 fs, 7 mJ per pulse, 1 kHz repetition rate) into three. The actinic pump centered at 480/500/510 nm was generated using an optical parametric amplifier (OperA Solo, Coherent, Inc.). About 3 W of fundamental pulses were introduced into a second harmonic bandwidth compressor (SHBC, Coherent, Inc.) to produce ps 400 nm pulses and subsequently generate a ps Raman pump pulse through a ps-OPA system (TOPAS-400, Coherent, Inc.). The Raman pump wavelengths for Stokes excited-state Raman experiments for spheroidene in n-hexane solution are chosen as 530 nm, for spheroidene in DMSO solution and the LH2 complex are chosen as 550 nm, and for anti-Stokes experiments for spheroidene in n-hexane solution and the LH2 complex are chosen as 580 nm and 620 nm. Per pulse energy is about 150 nJ. About 15 mW of the fundamental laser output was focused onto a 2 mm thick single-crystal sapphire plate to generate the supercontinuum white Raman probe pulse. The spectral resolution of the FSRS setup was determined to be ∼14 cm−1, as estimated from the full width at half maximum (FWHM) of the narrowest ground-state Raman band (802 cm−1) of cyclohexane measured under identical experimental conditions (see Fig. S1). The instrument response time was measured to be about 150 fs by cross correlation between the Raman probe pulses and fs actinic pump.Results and discussion
The steady-state absorption spectra of the free spheroidene in n-hexane and DMSO and the LH2 complex are illustrated in Fig. 1a. The free spheroidene in n-hexane solution exhibits a characteristic S0 → S2 (0–0) transition peak at 485 nm. Upon dissolution in DMSO solvent with higher polarizability, this absorption maximum undergoes a bathochromic shift to 503 nm.32–35 A comparable redshift is observed in the LH2-bound spheroidene, where the 0–0 transition appears at 510 nm, indicating polarizability environment perturbation of the carotenoid's electronic structure.35 | ||
| Fig. 1 (a) Steady-state absorption spectra of the LH2 complex and spheroidene (SPH) in DMSO and n-hexane solution; the inset shows the LH2 complex structure from Rhodobacter sphaeroides 2.4.1. (PDB:7PBW).30 The ground state FSRS spectra of spheroidene in n-hexane (b), LH2 complex (c), and DMSO (d). | ||
Fig. 1b–d compares the ground state FSRS spectra of the free spheroidene and LH2 complex. Three characteristic vibrational regions are identified: (1) The Raman modes at ∼1520 cm−1 are assigned to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching mode in the polyene chain. (2) The 1155 cm−1 feature arises from coupled C–C stretching and in-plane C–H bending modes. (3) Multiple peaks between 850 and 1000 cm−1 are diagnostic of C–H out-of-plane (HOOP) wagging vibrations. The detection of the HOOP modes in spheroidene indicates that there is distortion in the carotenoid backbone in its ground state.36–39
C stretching mode in the polyene chain. (2) The 1155 cm−1 feature arises from coupled C–C stretching and in-plane C–H bending modes. (3) Multiple peaks between 850 and 1000 cm−1 are diagnostic of C–H out-of-plane (HOOP) wagging vibrations. The detection of the HOOP modes in spheroidene indicates that there is distortion in the carotenoid backbone in its ground state.36–39
Excited-state structural dynamics are probed using FSRS with an actinic pulse energy of 150 nJ per pulse (Fig. 2–4, S5 and S6). To achieve the excited state Raman signal with a high signal to noise ratio, we employed Stokes-side FSRS (s-FSRS) using a 530 nm actinic pump for free spheroidene in n-hexane and a 550 nm for the LH2 complex and free spheroidene in DMSO (raw data are shown in Fig. S7). A Raman pump was used to obtain a high frequency (1000–2000 cm−1) excited state Raman signal (green dashed lines labeled in Fig. S2). A strong transient absorption (TA) induced nonlinear background in the 100–1000 cm−1 range obscures the low-frequency Raman signal in s-FSRS. To overcome this, 580 nm (for free spheroidene in n-hexane) and 620 nm (for the LH2 complex and free spheroidene in DMSO) Raman pump wavelengths were selected for anti-Stokes-side FSRS (a-FSRS) to achieve pre-resonant enhancement of the low frequency range (100–1000 cm−1) excited-state Raman signal (red dashed lines labeled in Fig. S2). These Raman pump wavelengths were selected to align with the excited-state absorption (ESA) bands and the stimulated emission (STE) band of the electronic excited states, based on the TA spectra shown in Fig. S2.
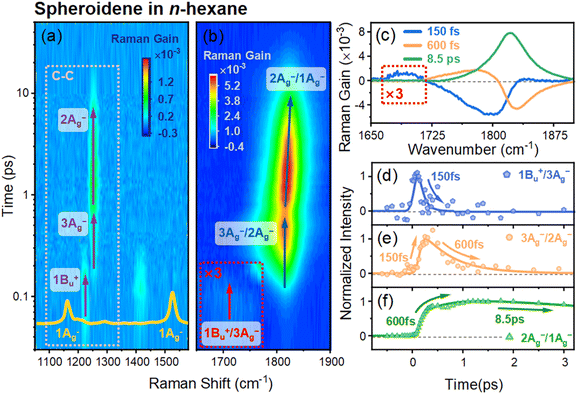
Fig. 2b shows the contour plot of 2D s-FSRS spectra of free spheroidene in the frequency range of 1650–1900 cm−1 in n-hexane, revealing a weak Raman mode at 1705 cm−1 and intense, broad Raman modes at ∼1817 cm−1 that exhibit intricate rise and decay dynamics. The decay-associated difference spectra (DADS) extracted from global analysis is used to analyze the FSRS spectra, as shown in Fig. 2c. The DADS of the Raman modes in the frequency range of 1650–1900 cm−1 reveal complex dynamics; the three-time components (τ1 = 150 fs, τ2 = 600 fs, and τ3 = 8.5 ps) from global fitting for these modes are consistent with the TA data obtained in the visible range (see Fig. S2). The Raman mode at 1705 cm−1 (red arrow in Fig. 2b) exhibits a single decay lifetime of τ1 = 150 fs, as shown in Fig. 2d, which signifies an ultrafast decay originating from the S2 (1Bu+) state. The detailed origin and assignment of this mode will be discussed later.
As depicted by dark blue arrows in Fig. 2b and the DADS in Fig. 2c, the s-FSRS reveals a two-stage evolution of Raman modes at ∼1810 cm−1: stage 1, the emergence of a Raman band at 1816 cm−1 within 150 fs (τ1) that subsequently decays with a time constant of 600 fs (τ2) and stage 2, the appearance of a new Raman mode at 1817 cm−1, exhibiting an increase in intensity governed by τ2 (600 fs), followed by a subsequent decrease with a time constant of τ3 = 8.5 ps.
As shown in Fig. S2b, the Raman pump wavelength was chosen to overlap with the ESA band of components τ2 and τ3 in TA spectra. Under these resonance conditions, the s-FSRS signals are dominated by the Raman gain signal with a Lorentzian line shape without noticeable dispersive distortion.40,41 Consequently, both the transient Raman frequencies and amplitudes extracted from the two-stage Raman modes reliably reflect the underlying vibrational dynamics, allowing comparison of peak positions and their temporal evolution.
The transient Raman mode (1817 cm−1) in stage 2, previously attributed to the in-phase C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching mode of the optically forbidden S1 (2Ag−) state, arises from the vibronic coupling between S1 (2Ag−) and S0 (1Ag−) states.17,24,25 According to the C2h point group symmetry of all-trans polyenes, this coupling results in a frequency upshift to ∼1817 cm−1 for the in-phase C
C stretching mode of the optically forbidden S1 (2Ag−) state, arises from the vibronic coupling between S1 (2Ag−) and S0 (1Ag−) states.17,24,25 According to the C2h point group symmetry of all-trans polyenes, this coupling results in a frequency upshift to ∼1817 cm−1 for the in-phase C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching mode in the 2Ag− excited state while simultaneously downshifting the corresponding mode frequency (1525 cm−1) in the 1Ag− ground state (Fig. 1b).25,42,43 As shown in Fig. 2f, analysis of the transient amplitude of the Raman signal reveals a biphasic kinetic trace of this vibronic coupling mode: a slow rise dynamics (600 fs) followed by a decay with a lifetime of 8.5 ps. The 600 fs rise component is attributed to the population in a higher vibrational state in S1 (2Ag−) (hot S1) from the upper excited state, accompanied by a vibrational cooling process. This population ultimately relaxes to S0 (1Ag−) states within 8.5 ps.44 Notably, the vibrational mode undergoes a progressive blue shift during the S1 → S0 internal conversion process (Fig. S3), a spectral shift directly correlating with the vibrational cooling dynamics in the S1 (2Ag−) state.17
C stretching mode in the 2Ag− excited state while simultaneously downshifting the corresponding mode frequency (1525 cm−1) in the 1Ag− ground state (Fig. 1b).25,42,43 As shown in Fig. 2f, analysis of the transient amplitude of the Raman signal reveals a biphasic kinetic trace of this vibronic coupling mode: a slow rise dynamics (600 fs) followed by a decay with a lifetime of 8.5 ps. The 600 fs rise component is attributed to the population in a higher vibrational state in S1 (2Ag−) (hot S1) from the upper excited state, accompanied by a vibrational cooling process. This population ultimately relaxes to S0 (1Ag−) states within 8.5 ps.44 Notably, the vibrational mode undergoes a progressive blue shift during the S1 → S0 internal conversion process (Fig. S3), a spectral shift directly correlating with the vibrational cooling dynamics in the S1 (2Ag−) state.17
In contrast to the S1 dynamics, the temporal evolution of the Raman signal in stage 1 exhibits an initial ∼150 fs rising dynamics followed by ∼600 fs decay, as shown in Fig. 2e. The 150 fs rise coincides with the ultrafast depopulation of the S2 (1Bu+) state (Fig. 2d), indicating that the formation of the stage 1 Raman signal is directly correlated with the rapid S2 relaxation process. The subsequent ∼600 fs decay occurs on the same timescale as the ∼600 fs rise observed for the S1 (2Ag−) state in stage 2 (Fig. 2f), suggesting a kinetic correspondence between the decay of the stage 1 signal and the build-up of the S1 population in stage 2. This temporal correlation provides conclusive evidence for the existence of an intermediate state (SX) mediating the S2 (1Bu+)-to-S1 (2Ag−) state transition, in comparison with the transient Raman signals of the S2 and S1 states in Fig. 2d and f. Notably, analogous to the 2Ag−/1Ag− vibronic coupling observed in stage 2, the detection of the unusually high frequency mode in stage 1 is indicative of vibronic coupling. Prior experimental and theoretical studies have characterized this intermediate (SX) state as a discrete single excited state, with its symmetry assignment to 1Bu−9–12 or Ag+16 configuration. However, our observation of a vibronic coupling mode rules out the Ag+ assignment. According to the pseudo-parity selection rule, states with “−” Pariser labels cannot couple with “+” states via C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations, and no vibronic coupling is expected between the 2Ag− and 1Bu+ states or between the 2Ag− and Ag+9,35 (unless the energy gap between “+” and “−” type states is very close to each other, which can induce a diabatic mixing17,45). Although the 1Bu− state lies energetically between S2 (1Bu+) and S1 (2Ag−), fluorescence excitation spectroscopy studies have shown that internal conversion from 1Bu− to 2Ag− states is markedly inefficient,46 suggesting that the 1Bu−/2Ag− vibronic coupling is symmetry-forbidden. In contrast, efficient internal conversion from 3Ag− to 2Ag− has been obtained, consistent with strong C
C stretching vibrations, and no vibronic coupling is expected between the 2Ag− and 1Bu+ states or between the 2Ag− and Ag+9,35 (unless the energy gap between “+” and “−” type states is very close to each other, which can induce a diabatic mixing17,45). Although the 1Bu− state lies energetically between S2 (1Bu+) and S1 (2Ag−), fluorescence excitation spectroscopy studies have shown that internal conversion from 1Bu− to 2Ag− states is markedly inefficient,46 suggesting that the 1Bu−/2Ag− vibronic coupling is symmetry-forbidden. In contrast, efficient internal conversion from 3Ag− to 2Ag− has been obtained, consistent with strong C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C vibronic coupling between these two states.35 This agrees with our experimental results and supports assigning the stage 1 Raman mode (1816 cm−1) to C
C vibronic coupling between these two states.35 This agrees with our experimental results and supports assigning the stage 1 Raman mode (1816 cm−1) to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C vibronic coupling between 3Ag− and 2Ag− states.
C vibronic coupling between 3Ag− and 2Ag− states.
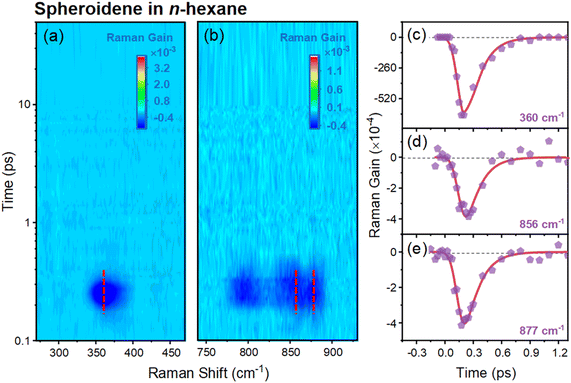
In the ground state geometry of spheroidene, the 3Ag− state lies above S2 (1Bu+) states.13–15,28,44 Upon the vertical excitation to the S2 (1Bu+) state, however, energetic inversion may occur along the bond length alteration (BLA) coordinate16,45 and polyene chain backbone distortion coordinate17 near the S2 potential energy minimum, where the 3Ag− state acts as an intermediate state mediating the S2 (1Bu+)-to-S1 (2Ag−) state transition. This inversion highlights the critical role of geometric changes in modulating excited-state energy. A pronounced BLA decrease in excited states induces an increase in conjugated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond length and a concomitant decrease in C–C bond lengths.47 This structural reorganization directly impacts the vibrational spectrum, as evidenced by shifts in Raman-active modes.47 In our experiment, as shown in Fig. 2a, an excited state Raman mode observed at 1223 cm−1 with a single decay lifetime of ∼150 fs corresponds to the in-phase C–C stretching Raman mode, exhibiting a 66 cm−1 upshift in the S2 (1Bu+) state related to the S0 (1Ag−) state (1157 cm−1).48 This blueshift aligns with the BLA induced stiffening of the conjugated C–C bonds.45,47 A further frequency upshift to 1252 cm−1 in SX (3Ag−) and S1 (2Ag−) states suggests additional C–C bond shortening (Fig. 2a), probably indicating a twisting dynamics in the S2 (1Bu+) state, leading to the formation of further structural distortion in the subsequent two dark (3Ag− and 2Ag−) excited states. In addition, a weak transient Raman feature at around ∼1400 cm−1 is observed with a single decay lifetime of ∼150 fs. This mode is assigned to CH3 symmetric deformation of the carotenoid methyl groups38 and may be sensitive to initial photoinduced structural rearrangements of the polyene backbone or side groups. Support for this interpretation is provided by the appearance of enhanced low-frequency excited state Raman modes at less than 1000 cm−1 observed in a-FSRS by using a redder Raman pump at 550 nm. The a-FSRS spectrum reveals low-frequency Raman loss signals at 360 cm−1 (Fig. 3a and c), 780 cm−1, 856 cm−1, and 877 cm−1 (Fig. 3b, d and e), all exhibiting ∼180 fs decay lifetimes, consistent with S2 state dynamics. As demonstrated in prior anti-Stokes side FSRS studies, the Raman loss signal in the excited state is observed only when the wavelengths of the Raman pump and Raman probe are pre-resonant with the electronic transition of the STE band in the TA spectrum,21,49 confirming selective probing of the optically bright S2 (1Bu+) state.
C bond length and a concomitant decrease in C–C bond lengths.47 This structural reorganization directly impacts the vibrational spectrum, as evidenced by shifts in Raman-active modes.47 In our experiment, as shown in Fig. 2a, an excited state Raman mode observed at 1223 cm−1 with a single decay lifetime of ∼150 fs corresponds to the in-phase C–C stretching Raman mode, exhibiting a 66 cm−1 upshift in the S2 (1Bu+) state related to the S0 (1Ag−) state (1157 cm−1).48 This blueshift aligns with the BLA induced stiffening of the conjugated C–C bonds.45,47 A further frequency upshift to 1252 cm−1 in SX (3Ag−) and S1 (2Ag−) states suggests additional C–C bond shortening (Fig. 2a), probably indicating a twisting dynamics in the S2 (1Bu+) state, leading to the formation of further structural distortion in the subsequent two dark (3Ag− and 2Ag−) excited states. In addition, a weak transient Raman feature at around ∼1400 cm−1 is observed with a single decay lifetime of ∼150 fs. This mode is assigned to CH3 symmetric deformation of the carotenoid methyl groups38 and may be sensitive to initial photoinduced structural rearrangements of the polyene backbone or side groups. Support for this interpretation is provided by the appearance of enhanced low-frequency excited state Raman modes at less than 1000 cm−1 observed in a-FSRS by using a redder Raman pump at 550 nm. The a-FSRS spectrum reveals low-frequency Raman loss signals at 360 cm−1 (Fig. 3a and c), 780 cm−1, 856 cm−1, and 877 cm−1 (Fig. 3b, d and e), all exhibiting ∼180 fs decay lifetimes, consistent with S2 state dynamics. As demonstrated in prior anti-Stokes side FSRS studies, the Raman loss signal in the excited state is observed only when the wavelengths of the Raman pump and Raman probe are pre-resonant with the electronic transition of the STE band in the TA spectrum,21,49 confirming selective probing of the optically bright S2 (1Bu+) state.
DFT calculations assign these modes to specific structural motions. The 360 cm−1 mode corresponds to polyene chain in-plane skeletal bending (Fig. S4a), while 856 cm−1 and 877 cm−1 modes are assigned to HOOP vibrations (Fig. S4b and c). Notably, carotenoid HOOP modes in 800–1000 cm−1 regimes exhibit exceptional sensitivity to restoring forces during chromophore twisting,17,20 a phenomenon validated across biological and chemical systems via FSRS.21,50,51 Collectively, these observations provide explicit evidence for spheroidene twisting during S2 state evolution. This conformational change, combined with the BLA reducing 3Ag− state energy, facilitates its emergence as an intermediate SX state between S2 (1Bu+) and S1 (2Ag−) states.
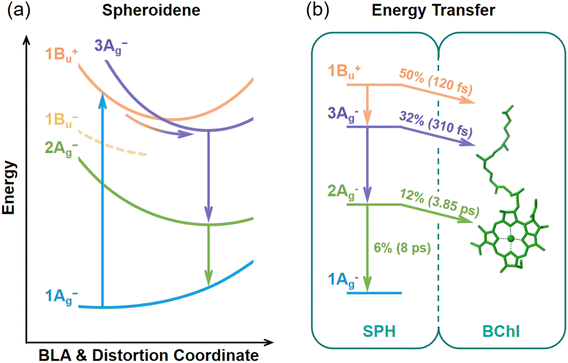
To summarize our findings thus far, DADS of the FSRS for spheroidene demonstrate three decay components, indicating a four-state relaxation scheme  . As proposed in the schematic diagram (Fig. 5a), the potential energy (PE) curves illustrate the photoreaction pathway driven by displacements along the BLA and twisting coordinates of C–C/C
. As proposed in the schematic diagram (Fig. 5a), the potential energy (PE) curves illustrate the photoreaction pathway driven by displacements along the BLA and twisting coordinates of C–C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds. Following vertical excitation, the wavepacket on the S2 (1Bu+) state undergoes ultrafast relaxation and subsequently decays along these coordinates to populate a structurally distorted SX (3Ag−) state. Notably, the emergence of the 1705 cm−1 mode (red arrow in Fig. 2b) arises from concurrent BLA reduction and backbone distortion. These collective distortions induce energetic inversion between S2 (1Bu+) and SX (3Ag−) states, causing them to approach each other and form an exceptionally narrow energy gap during the inversion.26,45 Consequently, this facilitates diabatic mixing between the S2 (1Bu+) and SX (3Ag−) states, leading to the observable modes at 1705 cm−1.17,26,45 The emergence of this mode corresponds to the instantaneous formation of the S2 state, followed by 150 fs decay. This decay is synchronized with the population dynamics of the SX (3Ag−) state on the same time scale and is then followed by intramolecular conversion to the S1 (2Ag−) state within 600 fs. Finally, the S1 population relaxes into a vibrationally hot, distorted S0 ground state, which ultimately cools to regain the equilibrium all-trans S0 configuration.17
C bonds. Following vertical excitation, the wavepacket on the S2 (1Bu+) state undergoes ultrafast relaxation and subsequently decays along these coordinates to populate a structurally distorted SX (3Ag−) state. Notably, the emergence of the 1705 cm−1 mode (red arrow in Fig. 2b) arises from concurrent BLA reduction and backbone distortion. These collective distortions induce energetic inversion between S2 (1Bu+) and SX (3Ag−) states, causing them to approach each other and form an exceptionally narrow energy gap during the inversion.26,45 Consequently, this facilitates diabatic mixing between the S2 (1Bu+) and SX (3Ag−) states, leading to the observable modes at 1705 cm−1.17,26,45 The emergence of this mode corresponds to the instantaneous formation of the S2 state, followed by 150 fs decay. This decay is synchronized with the population dynamics of the SX (3Ag−) state on the same time scale and is then followed by intramolecular conversion to the S1 (2Ag−) state within 600 fs. Finally, the S1 population relaxes into a vibrationally hot, distorted S0 ground state, which ultimately cools to regain the equilibrium all-trans S0 configuration.17
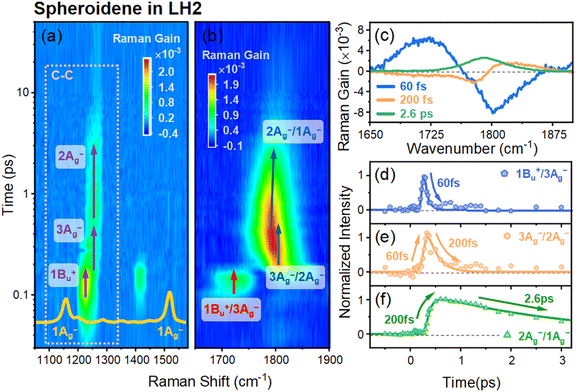
To further elucidate how the protein environment modulates the structural dynamics of spheroidene, we performed FSRS measurements on the LH2 complex. Fig. 4a and b display the 2D s-FSRS spectra of the LH2 complex in the frequency range of 1000–1900 cm−1. The protein-bound spheroidene pigment demonstrates analogous structural dynamics to that obtained from free spheroidene, as schematically illustrated in the PE diagram (Fig. 5a). Notably, as shown in Fig. 4b and c, the LH2 complex exhibits enhanced Raman-active mode at 1721 cm−1, which is attributed to S2 (1Bu+)/SX (3Ag−) diabatic mixing. This mode originates from photon-induced structural distortions of spheroidene in the LH2 complex that reduce the energy gap between S2 (1Bu+) and SX (3Ag−) during their inversion process. Supporting evidence includes (i) a blueshift excited-state C–C stretching mode at 1228 cm−1versus its ground-state counterpart at 1156 cm−1 (Fig. 4a), reflecting BLA reduction and (ii) the activation of skeletal motion and HOOP modes at 358 and 885 cm−1 (Fig. S5), indicating backbone distortions. Importantly, the diabatic mixing signal of spheroidene is much more pronounced in LH2 than in n-hexane solution, highlighting the role of the protein environment in modulating the spheroidene's energy levels.35,46
As shown in Table 1, a comparative analysis of the 1Bu+/3Ag− C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C diabatic mixing mode undergoes a 16 cm−1 blue shift, while the 3Ag−/2Ag−and 2Ag−/1Ag− vibronic coupling C
C diabatic mixing mode undergoes a 16 cm−1 blue shift, while the 3Ag−/2Ag−and 2Ag−/1Ag− vibronic coupling C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching modes exhibit systematic redshifts of 6 cm−1 and 27 cm−1, respectively, in the LH2 complex compared to free spheroidene in n-hexane. A similar downshifting trend is observed for ground-state 1Ag− C
C stretching modes exhibit systematic redshifts of 6 cm−1 and 27 cm−1, respectively, in the LH2 complex compared to free spheroidene in n-hexane. A similar downshifting trend is observed for ground-state 1Ag− C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching mode (Fig. 1b and c) and correlates with the redshift of the S0 (1Ag−) → S2 (1Bu+) electronic transition in steady absorption spectra (Fig. 1a). Together, these observations point to an enhanced environment polarizability within the protein binding pocket.35,37,52
C stretching mode (Fig. 1b and c) and correlates with the redshift of the S0 (1Ag−) → S2 (1Bu+) electronic transition in steady absorption spectra (Fig. 1a). Together, these observations point to an enhanced environment polarizability within the protein binding pocket.35,37,52
| Spheroidene in n-hexane | Spheroidene in DMSO | LH2 complex | |
|---|---|---|---|
| 1Bu+/3Ag− | 1705 cm−1 | 1711 cm−1 | 1721 cm−1 |
| 3Ag−/2Ag− | 1816 cm−1 | 1804 cm−1 | 1811 cm−1 |
| 2Ag−/1Ag− | 1817 cm−1 | 1788 cm−1 | 1790 cm−1 |
| 1Ag− | 1525 cm−1 | 1524 cm−1 | 1522 cm−1 |
Quantitatively, the average polarizability of spheroidene in the LH2 complex (R = 0.334) exceeds that in n-hexane (R = 0.23).35,37 To further substantiate the role of environmental polarizability, spheroidene dissolved in DMSO (R = 0.28)35 exhibits comparable frequency shifts (see Table 1). Relative to n-hexane, the 1Bu+/3Ag− C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C diabatic mixing mode blueshifts to 1711 cm−1 and 3Ag−/2Ag−, 2Ag−/1Ag−, and 1Ag− related C
C diabatic mixing mode blueshifts to 1711 cm−1 and 3Ag−/2Ag−, 2Ag−/1Ag−, and 1Ag− related C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C vibronic coupling modes redshift to 1804 cm−1, 1788 cm−1, and 1524 cm−1. In addition, an intensified 1Bu+/3Ag− diabatic mixing signature was observed at 1711 cm−1 (Fig. S6a and b). These results confirm that increased environmental polarizability plays a critical role in modulating the excited-state energy landscape of spheroidene.37,46
C vibronic coupling modes redshift to 1804 cm−1, 1788 cm−1, and 1524 cm−1. In addition, an intensified 1Bu+/3Ag− diabatic mixing signature was observed at 1711 cm−1 (Fig. S6a and b). These results confirm that increased environmental polarizability plays a critical role in modulating the excited-state energy landscape of spheroidene.37,46
Comparative analysis of the DADS reveals distinct excited-state decay dynamics for spheroidene in different environments. However, the spheroidene demonstrates comparable tri-exponential decay components in both DMSO (τ1 = 120 fs, τ2 = 550 fs, and τ3 = 8 ps) (Fig. S6b) and n-hexane solvents (τ1 = 150 fs, τ2 = 600 fs, and τ3 = 8.5 ps) (Fig. 2c), indicating that solvent polarizability exerts only minor effects on the intrinsic excited-state relaxation pathways of spheroidene.34,53 In sharp contrast, as shown in Fig. 4c, a remarkable acceleration of excited-state relaxation emerges within the LH2 complex, exhibiting significantly shortened lifetimes (τ1 = 60 fs, τ2 = 200 fs, and τ3 = 2.6 ps), which are consistent with the TA results (Fig. S2c and d).54 This significant decrease in spheroidene's S2, SX, and S1 lifetimes, compared with their lifetimes in solutions, provides clear evidence for efficient spheroidene → BChl excitation energy transfer (EET).
To quantitatively evaluate these transfer processes, we calculate spheroidene → BChl EET lifetimes of S2 (τET-1 = 120 fs), SX (τET-2 = 310 fs), and S1 (τET-3 = 3.85 ps) states, which are given by 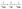 , where τLH2 and τSPH are experimentally measured lifetimes of spheroidene in the LH2 complex (τLH2-1 = 60 fs, τLH2-2 = 200 fs, and τLH2-3 = 2.6 ps) and in DMSO solution (τSPH-1 = 120 fs, τSPH-2 = 550 fs, and τSPH-3 = 8 ps),55,56 respectively. The energy transfer efficiencies (ΦET) are then determined from
, where τLH2 and τSPH are experimentally measured lifetimes of spheroidene in the LH2 complex (τLH2-1 = 60 fs, τLH2-2 = 200 fs, and τLH2-3 = 2.6 ps) and in DMSO solution (τSPH-1 = 120 fs, τSPH-2 = 550 fs, and τSPH-3 = 8 ps),55,56 respectively. The energy transfer efficiencies (ΦET) are then determined from 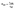 , yielding values of 50% for S2, 64% for SX, and 68% for S1 states.
, yielding values of 50% for S2, 64% for SX, and 68% for S1 states.
In addition to overall EET quantum yield of spheroidene to BChl, internal conversion between excited states needs to be considered. Specifically, 50% of the initial S2 population undergoes internal conversion to the SX state. The SX state then transfers ∼32% (50 × 64%) of the total energy to BChl, while the remaining ∼18% (50 × 36%) is internally converted to the S1 state. The S1 state subsequently transfers ∼12% (18 × 68%) of the total energy to BChl, with the remaining ∼6% (18 × 32%) relaxing to the ground state S0. Therefore, the total contributions of spheroidene to BChl energy transfer are quantified as 50% from S2, 32% from SX, and 12% from S1, yielding an overall quantum yield (ΦALL) of 94% (Fig. 5b), in excellent agreement with previous experimental measurements.4–7
Theoretical studies suggest that structural distortion can break the C2h symmetry of carotenoids, thereby enhancing coulombic coupling between the carotenoid S1 (2Ag−) and BChl (Qy) states.57 Consistent with this prediction, our FSRS results identify the dynamic structural distortion of spheroidene in its S2 (1Bu+) state, giving rise to distorted intermediate SX (3Ag−) and S1 (2Ag−) states. The involvement of such structurally distorted intermediates has been widely recognized in natural light-harvesting systems.17,58–60 Photoexcitation of carotenoids induces subtle backbone twisting, which enhances coulombic coupling to nearby chlorophylls and facilitates carotenoid-to-chlorophyll energy transfer.17 In our case, the structurally distorted intermediates of spheroidene may act as effective channels for ultrafast excitation energy delivery to BChls.
Conclusions
In this work, we employed FSRS to directly observe photoinduced structural change of spheroidene in its S2 (1Bu+) state, leading to the formation of subsequent distorted SX and S1 (2Ag−) intermediates. These results resolve the long-standing controversy regarding the SX state: we show that BLA reduction and backbone twisting drive an energetic inversion between the 3Ag− and S2 (1Bu+) states, thereby establishing a distorted SX (3Ag−) intermediate that mediates the S2 (1Bu+) → S1 (2Ag−) relaxation pathway. Quantitative analysis reveals spheroidene-to-BChl energy transfer efficiencies of 50% from S2, 32% from SX, and 12% from S1 states, yielding an overall efficiency of 94% in excellent agreement with previous reports. Photoinduced structural change not only activates the dark 3Ag− state but may also enhance coulombic coupling between spheroidene and neighboring BChl molecules, thereby enabling ultrafast and highly efficient carotenoid-to-BChl energy transfer within the LH2 complex. By linking structural distortion with dark-state mediation, this study provides new insight into how photosynthetic light-harvesting systems exploit optically forbidden intermediate-state pathways to achieve remarkable transfer efficiencies.Author contributions
B. P., M. C., and T. M. conceived, designed and performed the experiments. M. C. prepared the samples. B. P. and T. M. performed the experiments and analysed the experimental results. B. P., M. C., and T. M. co-wrote the manuscript. Y. H. did the DFT calculations. All authors participated in discussions and edited the manuscript. W. L. and P. W. directed the project.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): cyclohexane ground-state FSRS; 2D TA spectra of spheroidene in n-hexane, DMSO, and LH2 complex; transient Raman frequency shift of the 3Ag− mode of spheroidene in n-hexane; DFT calculation of spheroidene; 2D a-FSRS of spheroidene in LH2 complex; 2D s-FSRS of free spheroidene in DMSO. See DOI: https://doi.org/10.1039/d5sc08508j.Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 22373068 and 22073112); the Double First-Class Initiative Fund of ShanghaiTech University; ShanghaiTech Start-up Funding (F-0201-16-006).References
- A. Telfer, A. Pascal and A. Gall, in Carotenoids, ed. G. Britton, S. Liaaen-Jensen and H. Pfander, Birkhäuser Basel, Basel, 2008, ch. 4, pp. 265–308 Search PubMed.
- Y.-Q. Li, Y.-H. Yan, R.-Y. Gao, J.-W. Zou, Y.-L. Wu, X.-Y. Yue, Y. Lu, X.-P. Wang, M.-Q. Chen, Q.-W. Li, H.-Y. Wang, P. Wang, L.-J. Yu, J. Zheng and J.-P. Zhang, Triplet excitation dynamics of photosynthetic light-harvesting antennae: mechanistic insights into the conjugation regulated carotenoid functionality, Phys. Chem. Chem. Phys., 2025, 27, 12462–12473 Search PubMed.
- J.-F. Hao, N. Yamano, C.-H. Qi, Y. Zhang, F. Ma, P. Wang, L.-J. Yu and J.-P. Zhang, Carotenoid-Mediated Long-Range Energy Transfer in the Light Harvesting–Reaction Center Complex from Photosynthetic Bacterium Roseiflexus castenholzii, J. Phys. Chem. B, 2023, 127, 10360–10369 Search PubMed.
- P. J. Walla, P. A. Linden, C.-P. Hsu, G. D. Scholes and G. R. Fleming, Femtosecond dynamics of the forbidden carotenoid S1 state in light-harvesting complexes of purple bacteria observed after two-photon excitation, Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 10808–10813 Search PubMed.
- H. Cong, D. M. Niedzwiedzki, G. N. Gibson, A. M. LaFountain, R. M. Kelsh, A. T. Gardiner, R. J. Cogdell and H. A. Frank, Ultrafast Time-Resolved Carotenoid to-Bacteriochlorophyll Energy Transfer in LH2 Complexes from Photosynthetic Bacteria, J. Phys. Chem. B, 2008, 112, 10689–10703 Search PubMed.
- S. C. Chi, D. J. Mothersole, P. Dilbeck, D. M. Niedzwiedzki, H. Zhang, P. Qian, C. Vasilev, K. J. Grayson, P. J. Jackson, E. C. Martin, Y. Li, D. Holten and C. Neil Hunter, Assembly of functional photosystem complexes in Rhodobacter sphaeroides incorporating carotenoids from the spirilloxanthin pathway, Biochim. Biophys. Acta Bioenerg., 2015, 1847, 189–201 CrossRef CAS PubMed.
- J.-P. Zhang, T. Inaba, Y. Watanabe and Y. Koyama, Partition of carotenoid-to-bacteriochlorophyll singlet-energy transfer through two channels in the LH2 complex from Rhodobacter sphaeroides G1C, Chem. Phys. Lett., 2001, 340, 484–492 Search PubMed.
- E. Papagiannakis, J. T. M. Kennis, I. H. M. van Stokkum, R. J. Cogdell and R. van Grondelle, An alternative carotenoid-to-bacteriochlorophyll energy transfer pathway in photosynthetic light harvesting, Proc. Natl. Acad. Sci. U. S. A., 2002, 99, 6017–6022 CrossRef CAS PubMed.
- T. Sashima, H. Nagae, M. Kuki and Y. Koyama, A new singlet-excited state of all-trans-spheroidene as detected by resonance-Raman excitation profiles, Chem. Phys. Lett., 1999, 299, 187–194 Search PubMed.
- F. S. Rondonuwu, K. Yokoyama, R. Fujii, Y. Koyama, R. J. Cogdell and Y. Watanabe, The role of the 11Bu− state in carotenoid-to-bacteriochlorophyll singlet-energy transfer in the LH2 antenna complexes from Rhodobacter sphaeroides G1C, Rhodobacter sphaeroides 2.4.1, Rhodospirillum molischianum and Rhodopseudomonas acidophila, Chem. Phys. Lett., 2004, 390, 314–322 Search PubMed.
- E. E. Ostroumov, R. M. Mulvaney, R. J. Cogdell and G. D. Scholes, Broadband 2D Electronic Spectroscopy Reveals a Carotenoid Dark State in Purple Bacteria, Science, 2013, 340, 52–56 CrossRef CAS PubMed.
- E. Ostroumov, M. G. Müller, C. M. Marian, M. Kleinschmidt and A. R. Holzwarth, Electronic Coherence Provides a Direct Proof for Energy-Level Crossing in Photoexcited Lutein and β-Carotene, Phys. Rev. Lett., 2009, 103, 108302 Search PubMed.
- Y. Koyama, Y. Kakitani, T. Miki, R. Christiana and H. Nagae, Excited-State Dynamics of Overlapped Optically-Allowed 1Bu+ and Optically-Forbidden 1Bu- or 3Ag- Vibronic Levels of Carotenoids: Possible Roles in the Light-Harvesting Function, Int. J. Mol. Sci., 2010, 11, 1888–1929 CrossRef CAS PubMed.
- K. Furuichi, T. Sashima and Y. Koyama, The first detection of the 3Ag− state in carotenoids using resonance-Raman excitation profiles, Chem. Phys. Lett., 2002, 356, 547–555 Search PubMed.
- Y. Koyama, F. S. Rondonuwu, R. Fujii and Y. Watanabe, Light-harvesting function of carotenoids in photo-synthesis: The roles of the newly found 11Bu− state, Biopolymers, 2004, 74, 2–18 Search PubMed.
- J. Feng, C. W. Tseng, T. Chen, X. Leng, H. Yin, Y. C. Cheng, M. Rohlfing and Y. Ma, A new energy transfer channel from carotenoids to chlorophylls in purple bacteria, Nat. Commun., 2017, 8, 71 CrossRef PubMed.
- T. Ma, M. Ruan, R. Zhao, Z. Wang, Y. Wang, Y. Huang, Y. Weng and W. Liu, Distorted Intermediate SX (1Bu–) State in Xanthophylls Drives Efficient Energy Transfer in Light-Harvesting Complex II, J. Phys. Chem. Lett., 2025, 16, 6711–6718 Search PubMed.
- Z. Wang, Y. Chen, J. Jiang, X. Zhao and W. Liu, Mapping photoisomerization dynamics on a three-state model potential energy surface in bacteriorhodopsin using femtosecond stimulated Raman spectroscopy, Chem. Sci., 2025, 16, 3713–3719 RSC.
- P. G. Lynch, A. Das, S. Alam, C. C. Rich and R. R. Frontiera, Mastering Femtosecond Stimulated Raman Spectroscopy: A Practical Guide, ACS Phys. Chem. Au, 2024, 4, 1–18 CrossRef CAS PubMed.
- B. Peng, Z. Wang, J. Jiang, Y. Huang and W. Liu, Investigation of ultrafast intermediate states during singlet fission in lycopene H-aggregate using femtosecond stimulated Raman spectroscopy, J. Chem. Phys., 2024, 160, 194304 Search PubMed.
- Y. Chen, Z. Wang, J. Jiang and W. Liu, Resolving Dual Photoreaction Channels of All-Trans-Retinal Using Femtosecond Stimulated Raman Spectroscopy, J. Phys. Chem. B, 2025, 129, 7550–7556 CrossRef CAS PubMed.
- P. Kukura, D. W. McCamant and R. A. Mathies, Femtosecond Time-Resolved Stimulated Raman Spectroscopy of the S2 (1Bu+) Excited State of β-Carotene, J. Phys. Chem. A, 2004, 108, 5921–5925 Search PubMed.
- S. Shim and R. A. Mathies, Development of a Tunable Femtosecond Stimulated Raman Apparatus and Its Application to β-Carotene, J. Phys. Chem. B, 2008, 112, 4826–4832 Search PubMed.
- H. Hashimoto and Y. Koyama, The C=C stretching Raman lines of [β-carotene isomers in the S1 state as detected by pump-probe resonance Raman spectroscopy, Chem. Phys. Lett., 1989, 154, 321–325 Search PubMed.
- H. Nagae, M. Kuki, J.-P. Zhang, T. Sashima, Y. Mukai and Y. Koyama, Vibronic Coupling through the In-Phase, C=C Stretching Mode Plays a Major Role in the 2Ag- to 1Ag- Internal Conversion of all-trans-β-Carotene, J. Phys. Chem. A, 2000, 104, 4155–4166 CrossRef CAS.
- M. S. Marek, T. Buckup, J. Southall, R. J. Cogdell and M. Motzkus, Highlighting short-lived excited electronic states with pump-degenerate-four-wave-mixing, J. Chem. Phys., 2013, 139 Search PubMed.
- T. Miki, T. Buckup, M. S. Krause, J. Southall, R. J. Cogdell and M. Motzkus, Vibronic coupling in the excited-states of carotenoids, Phys. Chem. Chem. Phys., 2016, 18, 11443–11453 RSC.
- Y. Kakitani, T. Miki, Y. Koyama, H. Nagae, R. Nakamura and Y. Kanematsu, Vibrational relaxation and internal conversion in the overlapped optically-allowed 1Bu+ and optically-forbidden 1Bu- or 3Ag- vibronic levels of carotenoids: Effects of diabatic mixing as determined by Kerr-gate fluorescence spectroscopy, Chem. Phys. Lett., 2009, 477, 194–201 CrossRef CAS.
- D. M. Niedzwiedzki, C. N. Hunter and R. E. Blankenship, Evaluating the Nature of S0-Called S*-State Feature in Transient Absorption of Carotenoids in Light-Harvesting Complex 2 (LH2) from Purple Photosynthetic Bacteria, J. Phys. Chem. B, 2016, 120, 11123–11131 CrossRef CAS PubMed.
- P. Qian, D. J. K. Swainsbury, T. I. Croll, P. Castro-Hartmann, G. Divitini, K. Sader and C. N. Hunter, Cryo-EM Structure of the Rhodobacter sphaeroides Light-Harvesting 2 Complex at 2.1 Å, Biochemistry, 2021, 60, 3302–3314 CrossRef CAS PubMed.
- T. Sashima, M. Shiba, H. Hashimoto, H. Nagae and Y. Koyama, The 2Ag− energy of crystalline all-trans-spheroidene as determined by resonance-Raman excitation profiles, Chem. Phys. Lett., 1998, 290, 36–42 CrossRef CAS.
- M. Maiuri, D. Polli, D. Brida, L. Lüer, A. M. LaFountain, M. Fuciman, R. J. Cogdell, H. A. Frank and G. Cerullo, Solvent-dependent activation of intermediate excited states in the energy relaxation pathways of spheroidene, Phys. Chem. Chem. Phys., 2012, 14, 6312–6319 RSC.
- P. O. Andersson, T. Gillbro, L. Ferguson and R. J. Cogdell, Absorption spectral shifts of carotenoids related to medium polarizability, Photochem. Photobiol., 1991, 54, 353–360 CrossRef CAS.
- A. N. Macpherson and T. Gillbro, Solvent Dependence of the Ultrafast S2−S1 Internal Conversion Rate of β-Carotene, J. Phys. Chem. A, 1998, 102, 5049–5058 CrossRef CAS.
- M. Kuici, H. Nagae, R. J. Cogdell, K. Shimada and Y. Koyama, Solvent effect on spheroidene in nonpolar and polar solutions and the environment of spheroidene in the light-harvesting complexes of rhodobacter sphaeroides 2.4.1 as revealed by the energy of the 1Ag−→1Bu+ absorption and the frequencies of the vibronically coupled C=C stretching raman lines in the 1Ag− and 1Bu− states, Photochem. Photobiol., 1994, 59, 116–124 CrossRef.
- A. A. Pascal, Z. Liu, K. Broess, B. van Oort, H. van Amerongen, C. Wang, P. Horton, B. Robert, W. Chang and A. Ruban, Molecular basis of photoprotection and control of photosynthetic light-harvesting, Nature, 2005, 436, 134–137 CrossRef CAS PubMed.
- M. J. Llansola-Portoles, A. A. Pascal and B. Robert, in Methods in Enzymology, ed. E. T. Wurtzel, Academic Press, 2022, vol. 674, ch. 4, pp. 113–135 Search PubMed.
- S. Saito and M. Tasumi, Normal-coordinate analysis of β-carotene isomers and assignments of the Raman and infrared bands, J. Raman Spectrosc., 1983, 14, 310–321 CrossRef CAS.
- J. Yu, L.-M. Fu, L.-J. Yu, Y. Shi, P. Wang, Z.-Y. Wang-Otomo and J.-P. Zhang, Carotenoid Singlet Fission Reactions in Bacterial Light Harvesting Complexes As Revealed by Triplet Excitation Profiles, J. Am. Chem. Soc., 2017, 139, 15984–15993 CrossRef CAS PubMed.
- S.-Y. Lee, D. Zhang, D. W. McCamant, P. Kukura and R. A. Mathies, Theory of femtosecond stimulated Raman spectroscopy, J. Chem. Phys., 2004, 121, 3632–3642 CrossRef CAS PubMed.
- K. Niu, B. Zhao, Z. Sun and S.-Y. Lee, Analysis of femtosecond stimulated Raman spectroscopy of excited-state evolution in bacteriorhodopsin, J. Chem. Phys., 2010, 132, 084510 CrossRef PubMed.
- G. Orlandi, F. Zerbetto and M. Z. Zgierski, Theoretical analysis of spectra of short polyenes, Chem. Rev., 1991, 91, 867–891 CrossRef CAS.
- T. Noguchi, H. Hayashi, M. Tasumi and G. H. Atkinson, Solvent effects on the ag carbon-carbon double bond stretching mode in the 21Ag- excited state of beta.-carotene and two derivatives: picosecond time-resolved resonance Raman spectroscopy, J. Phys. Chem., 1991, 95, 3167–3172 CrossRef CAS.
- F. S. Rondonuwu, Y. Watanabe, R. Fujii and Y. Koyama, A first detection of singlet to triplet conversion from the 11Bu− to the 13Ag state and triplet internal conversion from the 13Ag to the 13Bu state in carotenoids: dependence on the conjugation length, Chem. Phys. Lett., 2003, 376, 292–301 CrossRef CAS.
- D. Accomasso, S. Arslancan, L. Cupellini, G. Granucci and B. Mennucci, Ultrafast Excited-State Dynamics of Carotenoids and the Role of the SX State, J. Phys. Chem. Lett., 2022, 13, 6762–6769 CrossRef CAS PubMed.
- Y. Koyama, Y. Miki, T. Kameyama, R. J. Cogdell and Y. Watanabe, Low-lying electronic levels of spheroidene bound to the light-harvesting (LH2) complex of Rhodobacter sphaeroides 2.4.1 as determined by fluorescence and fluorescence—excitation spectroscopy at 170 K, Chem. Phys. Lett., 1993, 208, 479–485 CrossRef CAS.
- M. Jakučionis, I. Gaiži
![[u with combining overline]](https://www.rsc.org/images/entities/char_0075_0305.gif) nas, J. Šulskus and D. Abramavičius, Simulation of Ab Initio Optical Absorption Spectrum of β-Carotene with Fully Resolved S0 and S2 Vibrational Normal Modes, J. Phys. Chem. A, 2022, 126, 180–189 CrossRef PubMed.
nas, J. Šulskus and D. Abramavičius, Simulation of Ab Initio Optical Absorption Spectrum of β-Carotene with Fully Resolved S0 and S2 Vibrational Normal Modes, J. Phys. Chem. A, 2022, 126, 180–189 CrossRef PubMed. - T. Noguchi, S. Kolaczkowski, C. Arbour, S. Aramaki, G. H. Atkinson, H. Hayashi and M. Tasumi, Resonance Raman spectrum of the excited 2Ag state of β-carotene, Photochem. Photobiol., 1989, 50, 603–609 CrossRef CAS.
- J. Wei, Y. Wu, R. Pu, L. Shi, J. Jiang, J. Du, Z. Guo, Y. Huang and W. Liu, Tracking Ultrafast Structural Dynamics in a Dual-Emission Anti-Kasha-Active Fluorophore Using Femtosecond Stimulated Raman Spectroscopy, J. Phys. Chem. Lett., 2021, 12, 4466–4473 CrossRef CAS PubMed.
- P. Kukura, D. W. McCamant, S. Yoon, D. B. Wandschneider and R. A. Mathies, Structural Observation of the Primary Isomerization in Vision with Femtosecond-Stimulated Raman, Science, 2005, 310, 1006–1009 CrossRef CAS PubMed.
- C. Fang, R. R. Frontiera, R. Tran and R. A. Mathies, Mapping GFP structure evolution during proton transfer with femtosecond Raman spectroscopy, Nature, 2009, 462, 200–204 CrossRef CAS PubMed.
- M. Macernis, J. Sulskus, S. Malickaja, B. Robert and L. Valkunas, Resonance Raman Spectra and Electronic Transitions in Carotenoids: A Density Functional Theory Study, J. Phys. Chem. A, 2014, 118, 1817–1825 CrossRef CAS PubMed.
- H. A. Frank, J. A. Bautista, J. Josue, Z. Pendon, R. G. Hiller, F. P. Sharples, D. Gosztola and M. R. Wasielewski, Effect of the Solvent Environment on the Spectroscopic Properties and Dynamics of the Lowest Excited States of Carotenoids, J. Phys. Chem. B, 2000, 104, 4569–4577 CrossRef CAS.
- M. Ricci, S. E. Bradforth, R. Jimenez and G. R. Fleming, Internal conversion and energy transfer dynamics of spheroidene in solution and in the LH-1 and LH-2 light-harvesting complexes, Chem. Phys. Lett., 1996, 259, 381–390 CrossRef CAS.
- E. E. Ostroumov, R. M. Mulvaney, J. M. Anna, R. J. Cogdell and G. D. Scholes, Energy transfer pathways in light-harvesting complexes of purple bacteria as revealed by global kinetic analysis of two-dimensional transient spectra, J. Phys. Chem. B, 2013, 117, 11349–11362 CrossRef CAS PubMed.
- B. P. Krueger, G. D. Scholes and G. R. Fleming, Calculation of Couplings and Energy-Transfer Pathways between the Pigments of LH2 by the ab Initio Transition Density Cube Method, J. Phys. Chem. B, 1998, 102, 5378–5386 CrossRef CAS.
- C.-P. Hsu, P. J. Walla, M. Head-Gordon and G. R. Fleming, The Role of the S1 State of Carotenoids in Photosynthetic Energy Transfer: The Light-Harvesting Complex II of Purple Bacteria, J. Phys. Chem. B, 2001, 105, 11016–11025 CrossRef CAS.
- T. Wei, V. Balevičius, T. Polívka, A. V. Ruban and C. D. P. Duffy, How carotenoid distortions may determine optical properties: lessons from the Orange Carotenoid Protein, Phys. Chem. Chem. Phys., 2019, 21, 23187–23197 Search PubMed.
- K. F. Fox, W. P. Bricker, C. Lo and C. D. P. Duffy, Distortions of the Xanthophylls Caused by Interactions with Neighboring Pigments and the LHCII Protein Are Crucial for Studying Energy Transfer Pathways within the Complex, J. Phys. Chem. B, 2015, 119, 15550–15560 Search PubMed.
- S. Ghosh, J. D. Roscioli, M. M. Bishop, J. K. Gurchiek, A. M. LaFountain, H. A. Frank and W. F. Beck, Torsional Dynamics and Intramolecular Charge Transfer in the S2 (11Bu+) Excited State of Peridinin: A Mechanism for Enhanced Mid-Visible Light Harvesting, J. Phys. Chem. Lett., 2016, 7, 3621–3626 Search PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |