Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Materials design for thermally improved safety in lithium-ion batteries
Songpei
Nan
,
Guoxin
Gao
 ,
Wei
Yu
,
Wei
Yu
 ,
Shujiang
Ding
,
Shujiang
Ding
 * and
Dawei
Ding
* and
Dawei
Ding
 *
*
School of Chemistry, Xi'an Jiaotong University, Engineering Research Center of Energy Storage Materials and Devices, Ministry of Education, Xi'an, 710049, China. E-mail: dingsj@mail.xjtu.edu.cn; davidding1@mail.xjtu.edu.cn
First published on 14th January 2026
Abstract
With the ever-increasing demand for high-energy-density lithium-ion batteries (LIBs) in multiscale energy storage, safety concerns have emerged as critical obstacles hindering their widespread application. The excess heat generated during the electrochemical process, if not properly managed, can accumulate and accelerate the aging of key cell components, potentially leading to catastrophic thermal runaway events such as fires and explosions. Thus far, considerable attention has been devoted to alleviating intense thermal runaway through fire-safe materials and energy-intensive thermal management technologies. However, the stabilization of the electrochemical environment through intrinsic thermal dissipation and temperature regulation governed by key material design has received comparatively little consideration. This paper aims to summarize the mechanism of thermal runway and highlight material advances for safer LIBs, with particular emphasis on the thermal-electrochemical synergy in mitigating localized overheating, stabilizing the electrochemical environment, and improving electrochemical performance. Subsequently, recent research progress in thermal management materials and strategies for dynamic temperature regulation is reviewed. Finally, current challenges are discussed, and future directions are proposed for material innovations that can be applied to high-energy-density and high-safety LIBs.
Introduction
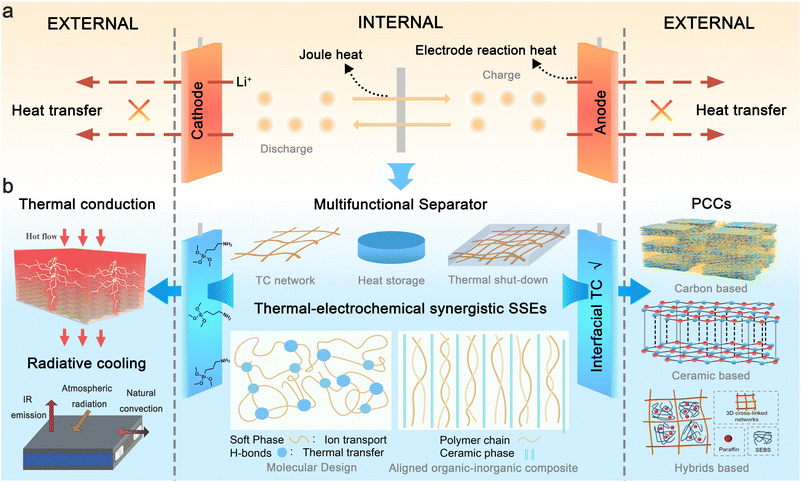
The transition from fossil fuels to renewable energy-powered industry, transportation, and human activities is widely regarded as the ultimate solution to environmental challenges caused by fossil fuel combustion. Energy storage devices play an irreplaceable role in grid-connected renewable energy systems and electric vehicle applications. Among rechargeable energy storage technologies, lithium-ion batteries (LIBs) have emerged as the predominant choice due to their high energy density, long cycle life, and the absence of memory effects. Nevertheless, safety concerns surrounding LIBs have persisted since their commercialization, primarily arising from thermal runaway-induced battery fires.1,2Thermal runaway progression in LIBs typically originates from heat accumulation dynamics, governed by the imbalance between heat generation and dissipation.3 Commercial LIBs operate through the shuttling of Li-ions in liquid electrolytes between cathodes and anodes (Fig. 1a), generating heat from joule heating and electrode reaction. Under low charging/discharging rates (<1C), generated heat can be dissipated spontaneously. However, during fast-charging (>3C) or high-power discharging scenarios (common in electric vehicle application), excessive heat generation leads to significant heat accumulation.4,5 Furthermore, anisotropic thermal conductivity (TC) within cell components and non-uniform cooling conditions induce localized temperature gradients.6 These hotspots accelerate electrochemical reaction kinetics, creating a positive feedback loop: elevated temperatures increase local current density, which further exacerbates heat generation. When cell temperature exceeds abuse thresholds, thermal runaway—characterized by uncontrolled heat release due to exothermic chemical chain reactions—is triggered, culminating in catastrophic fires or explosions.1 External abuse conditions such as overheating, overcharging, and mechanical abuse can also initiate thermal runaway through distinct failure pathways.7,8
 | ||
| Fig. 1 (a) Diagram of heat generation and accumulation during LIB operation, (b) material advances in key cell components and external thermal management for improving battery safety. Reproduced from ref. 9, copyright 2022, Elsevier. Reproduced from ref. 10, copyright 2019, Elsevier. Reproduced from ref. 11, copyright 2023, Elsevier. | ||
Substantial efforts to enhance battery safety over recent decades have focused on materials advances and thermal management systems. Materials advances mainly focus on safe electrolytes,12–15 separator engineering, thermally stable electrodes,16 and smart functional materials. Safe electrolytes include non-flammable electrolytes,17 flame retardant additives,18 and stable lithium salts.19 However, these attempts often introduce new challenges. For example, directly adding flame retardants to the electrolyte often compromises the battery's electrochemical performance.20 Recent studies demonstrate that decoupling ion transport with flame-retardant properties through the introduction of a triadic molecular synergy system can mitigate this adverse effect.21 Solid-state electrolytes with advanced safety features are considered promising candidates for next-generation batteries but may still exhibit heat generation comparable to carbonate-based electrolytes when paired with electrode materials and can ignite once temperatures reach a certain threshold.22,23 Separator thermal stability can be improved through coating polymers with inorganic materials (e.g., SiO2, Al2O3, TiO2, and ZrO2) or using materials of high melting temperatures with low shrinkage at elevated temperatures, such as polyimide, cellulose, and polyesters.24–26 Modifications of electrode materials through doping,27 surface coating,28 or an artificial cathode-electrolyte interphase/solid electrolyte interphase (SEI)29 are common strategies to enhance thermal stability, albeit often at the expense of energy density.30 Recently, an emerging class of safe functional materials (e.g., shutdown materials) has been developed to protect batteries from short-circuit-induced heat generation without side effects under normal operating conditions. These materials can be integrated into existing battery structures, such as the current collector,31 electrolyte,32–34 separator,35 or electrode.36 These shutdown materials, primarily composed of thermosensitive polymers, operate by sharply increasing internal cell resistance at high temperatures through thermal expansion or melting, thereby blocking electron conduction or Li+ pathways.37,38 Most shutdown materials primarily focus on mitigating late-stage thermal runaway phenomena, often at the expense of the battery's electrochemical performance or by relying on irreversible safety mechanisms that lead to permanent battery failure.39–42 In this regard, the introduction of reversible thermal shutdown materials is more advantageous for the recovery of electrochemical performance.43,44 In addition to the shutdown mechanism, slowing the crosstalk diffusion kinetics of lithium ions and active gases through the temperature-triggered formation of a dense crosslinked polymer network has been identified as an effective strategy to prevent the escalation of thermal runaway.45
Thermal management systems primarily involve the implementation of active cooling strategies (e.g., forced air/liquid convection,46 thermoelectric devices47) and passive thermal regulation materials (e.g., phase changing materials (PCMs),48 heat pipes,49 radiative cooling materials50). While active systems demonstrate superior cooling capacity and are particularly effective for addressing specific thermal abuse scenarios, their enormous parasitic energy consumption limits their application.51–53 Besides, conventional thermal management systems are typically applied externally to dissipate heat from the battery surface. However, this approach can induce undesired internal temperature elevation and thermal gradient due to inefficient heat transfer from the core to surface.54,55 In contrast, internal thermal management through material innovation offers enhanced safety and durability by directly regulating temperature distribution within the cell. Nevertheless, research in this aera remains in its early stage, necessitating further efforts to advance these technologies for practical implementation.
Although recent reviews have highlighted advancements in thermal management to enhance the safety of LIBs, most have primarily focused on the exploration of thermally stable battery components56–58 or external cooling technologies.59–61 There is a critical need to fundamentally prevent thermal runaway at its initial heat accumulation stage through a comprehensive design of key materials, both internally and externally. This paper aims to systematically examine advanced thermal management strategies and propose material design directions for improving the electrochemical environment to prevent thermal runaway initiation from a holistic perspective, with an emphasis on the thermal-electrochemical synergy that can significantly enhance both electrochemical performance and battery safety (Fig. 1b). Specifically, enhancing thermal homogeneity within the battery can stabilize the electrochemical environment. As a result, more stable and uniform ionic transportation and deposition can prevent hotspot generation and lithium dendrite formation. In the material design of key battery components, such as polymer-based solid electrolytes, we particularly focus on reconciling the trade-off between the thermal and electrochemical properties through materials engineering. Besides, recent research progress in thermal management materials and strategies for dynamic temperature regulation is reviewed. Finally, we summarize contemporary challenges in battery thermal management materials and propose a roadmap for safety design methodologies in next-generation LIBs.
Mechanism for thermal runaway
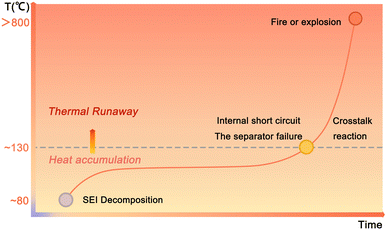
A deep understanding of the thermal runway mechanism is critical for analyzing challenges and formulating mitigation strategies. Although definitions of the thermal runaway stages vary across studies, the process can generally be divided into two stages: heat accumulation and intense thermal runaway (Fig. 2).1,62–64 The heat accumulation stage begins with the decomposition of the SEI layer on the anode surface and subsequent anode-electrolyte reactions at 80–110 °C. The relatively low cell temperature results in slow exothermic reaction rates, yielding mild heat release (∼257 J g−1). Consequently, the cell temperature rises at a moderate rate (<0.05 °C min−1), leading to a prolonged duration (minutes to days) of the heat accumulation stage.65,66If the generated heat cannot be dissipated, continuous heat accumulation may trigger the intense thermal runaway stage (130–300 °C), characterized by chemical chain reactions or crosstalk reactions.67,68 Polyolefin-based separators melt at ∼130 °C, causing internal short circuits and localized current surges.69 At temperatures above 200 °C, the cathode materials such as high-Li layered oxides decompose, releasing O2. The liberated O2 will react exothermically with carbonate solvents (e.g., ethylene carbonate, which autoignites at 140 °C in air), generating CO and CO2, and releasing intense heat (>2000 J g−1).36,70–72 Finally, the intense chemical reaction kinetics accelerate exponentially, driving temperatures to over 800 °C within seconds (heating rate >104 °C min−1), ultimately causing uncontrollable fires or explosions.73–75
In the early stage of heat accumulation, enhancing internal heat dissipation through key cell components, such as separators or electrolytes with high TC and latent heat, which are capable of buffering sharp temperature variations, can effectively mitigate the escalation of thermal runaway. However, once the temperature surpasses a critical threshold during the intense thermal runaway stage, it necessitates the use of more thermally stable components or shutdown materials to suppress chain reactions or chemical crosstalk. Thus, it is more rational to address and curb thermal runaway during the early heat accumulation stage. As previously stated, peak internal temperatures during thermal runaway can exceed 800 °C. While introducing nonflammable additives could theoretically enhance safety, these materials must satisfy two stringent criteria: chemical stability when paired with all battery components and minimal impact on electrochemical performance. However, such requirements severely restrict viable options. Furthermore, dissipating heat during intense thermal runaway is practically infeasible due to the enormous cooling power required.2 Therefore, this study focuses on the early-stage thermal management through rationally designed materials that could prevent heat accumulation. In Section 3, we systematically analyze emerging thermal management strategies, including internal heat dissipation, external thermal regulation, and design principles. These insights aim to guide the development of next-generation safe LIBs with intrinsic thermal management.
Battery materials design
Internal heat dissipation
Heat accumulation within a battery can lead to uneven temperature distribution and temperature rise if the heat is not dissipated promptly. This can accelerate battery aging, cause component failure, and ultimately trigger thermal runaway if the temperature exceeds a critical threshold.76 Therefore, internal thermal management must address heat accumulation and temperature non-uniformity by enhancing heat dissipation within the battery. This relies on the optimal design of key cell components, including separators,77 electrolytes,78 and electrodes.79Studies have shown that the application of high-TC ceramic coating materials not only enhances the thermal stability of polymer separators but also significantly reduces temperature rise during high-rate discharge, thereby improving electrochemical performance.81 Furthermore, the combination of highly heat-resistant polymer matrix materials (e.g., poly-p-phenylene terephthamide, polyimide) with thermally conductive fillers (e.g., boron nitride (BN), carbon nanotubes) optimizes electrochemical performance through synergistic mechanical-thermal-electrochemical effects.82–85 For instance, Lim et al. developed a highly thermally conductive composite separator composed of interwoven super-aligned carbon nanotubes and super-aligned BN@carbon nanotubes.86 This separator features a uniform thermal field, enabling rapid heat dissipation to prevent overheating while suppressing the polysulfide shuttle effect and lithium dendrite growth, thereby enhancing electrochemical performance.
Recently, PCMs with high enthalpy and rapid temperature response have gained significant attention for their ability to endow separators with excellent heat dissipation capabilities. Liu et al. proposed a novel phase-change functional separator inspired by the structure of a sugar gourd.87 The separator, made of melamine-encapsulated paraffin, rapidly absorbs heat generated during battery operation. By mimicking the nutrient-water transport mechanism of natural gourds, it significantly enhances the melting and heat storage efficiency of the PCM (Fig. 3a). Experimental results demonstrate that this bionic structure not only effectively slows the rate of temperature rise within the battery (Fig. 3b) but also enables rapid heat dissipation during short-circuit events, providing an innovative solution to prevent thermal runaway.
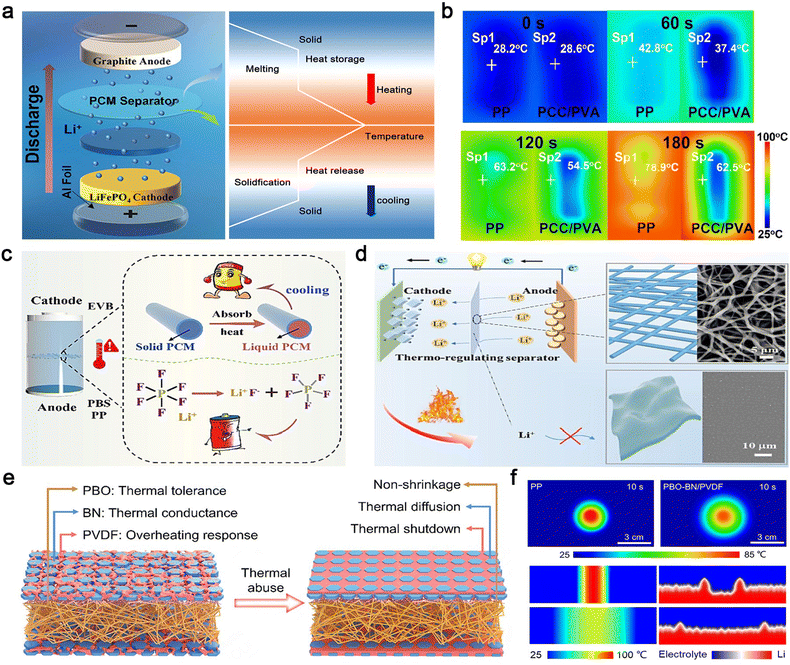 | ||
| Fig. 3 Separator materials for LIBs. (a) Battery structure and discharge diagram, (b) thermal management images of the PP separator and phase change capsules/poly(vinyl alcohol) (PCC/PVA) separator heated at 100 °C. Reproduced from ref. 87, copyright 2024, American Chemical Society. (c) High-performance LIB based on a PEG/PVDF@PBS separator, (d) battery thermal management based on the PEG/PVDF@PBS separator. Reproduced from ref. 90, copyright 2024, Elsevier. (e) Design illustration of the thermal overheating-responsive separator, (f) thermal infrared images (top-down view) of the PP and PBO–BN/PVDF separators, and the simulated Li deposition morphology on the Li metal surface (cross-sectional view) under different temperature distributions. Reproduced from ref. 91, copyright 2024, The Royal Society of Chemistry. | ||
Previous studies have demonstrated that thermosensitive polymer separators/electrolytes can serve a dual shutdown function by suppressing ionic transport and preventing crosstalk diffusion of active gases between electrodes when the temperature exceeds a critical threshold, thereby mitigating the escalation of thermal runaway.45,88,89 However, under rapid local overheating conditions caused by internal short circuits, the shutdown function may fail if the separator shrinks or melts. Unlike traditional strategies that rely on ceramic/polymer composite materials with high melting temperatures to improve separator thermal stability and safety, Wang et al. developed a polyethylene glycol/polyvinylidene fluoride (PEG/PVDF) @polybutylene succinate (PBS) core–shell separator with dual cooling and thermal shutdown functions using a one-step coaxial electrostatic spinning method (Fig. 3c).90 The cooling function of PEG in the separator effectively prevents ion conductivity degradation during aging, thereby mitigating specific capacity reduction and polarization voltage increase compared to conventional polymer separators (e.g., polypropylene (PP), polyethylene (PE), PBS). When the temperature reaches 110 °C, the PBS shell melts to block electrochemical reactions without thermal shrinkage, preventing further escalation of thermal runaway (Fig. 3d).
Guided by electro-chemo-thermal process modeling, Lu et al. proposed a separator design principle that integrates thermal tolerance, thermal conductance, and overheating response to enhance the thermal safety of high-energy-density batteries.91 They utilized a poly(p-phenylene benzobisoxazole) (PBO) membrane as the thermally tolerant matrix and composite coating layers of BN nanosheets and PVDF as the thermally conducting and overheating-responsive layers, forming a sandwich trilayer separator (denoted as PBO–BN/PVDF) (Fig. 3e). The PBO–BN/PVDF separator exhibits high mechanical strength and excellent thermal stability (almost zero shrinkage at 350 °C), preventing internal short circuits by suppressing separator shrinkage. Additionally, the thermally conducting network enhances heat dissipation, eliminating heat accumulation and large temperature gradients within the cell, which is beneficial for preventing lithium dendrite growth (Fig. 3f). Under harsh thermal conditions, the PVDF layer triggers an overheating-responsive shutdown to cut off ionic transport, ensuring safe battery operation and preventing catastrophic thermal runaway. These optimized approaches to separator design address critical challenges in battery thermal management, enhancing both performance and safety. It is worth noting that the design principles of the separator are partially applicable to the polymer-based solid electrolyte as well, since it necessitates electrolytes with high thermal conductivity, stability, and considerable mechanical strength for effective heat dissipation. Furthermore, special attention must be given to addressing ionic transport in the polymer electrolyte, which introduces significant complexity to material design. This will be discussed in detail in the following section.
The design of polymer electrolytes with dual thermal and ionic conductivity requires the incorporation of rigid chain segments and intermolecular hydrogen bonding to enhance phonon transport, as well as flexible chain segments to promote ionic migration. Additionally, mechanical strength is a critical consideration in polymer electrolyte design.112 Rigid segments (e.g., aromatic rings) and multiple intermolecular hydrogen bonds113 can improve mechanical strength. Incorporating a supramolecular structure with a hard phase for phonon transport and mechanical strength, along with a soft phase for enhanced ionic dissociation and migration in the polymer electrolyte, can achieve thermal-electrochemical synergy, thereby enhancing the stability of the electrochemical environment and improving battery safety. Zhou et al. developed a quasi-solid-state polymer electrolyte using a dynamic supramolecular structure based on multiple dynamic bonds and a phase-locking strategy (Fig. 4a).114 As shown in Fig. 4b, C–F bonds and benzene rings in the hard phase enhance thermal stability, while C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds and –CH2–O–CH2– groups in the soft phase facilitate Li+ coupling and migration, ensuring fast and uniform ion transport. Hydrogen bonds between hard phases endow the system with self-healing ability, high tensile strength, and strong stretchability. Notably, strong π–π stacking interactions between aromatic rings and multiple intermolecular hydrogen bonds also contribute to TC enhancement. When heated from room temperature to 120 °C, the polymer films exhibit stable morphology and uniform temperature distribution, indicating excellent thermal stability and efficient heat transfer properties (Fig. 4c). Notably, the well-balanced thermal, mechanical, and electrochemical properties of the polymer electrolyte significantly enhance the cycling stability of a Li–Li symmetric cell by effectively suppressing the growth of lithium dendrites.
O bonds and –CH2–O–CH2– groups in the soft phase facilitate Li+ coupling and migration, ensuring fast and uniform ion transport. Hydrogen bonds between hard phases endow the system with self-healing ability, high tensile strength, and strong stretchability. Notably, strong π–π stacking interactions between aromatic rings and multiple intermolecular hydrogen bonds also contribute to TC enhancement. When heated from room temperature to 120 °C, the polymer films exhibit stable morphology and uniform temperature distribution, indicating excellent thermal stability and efficient heat transfer properties (Fig. 4c). Notably, the well-balanced thermal, mechanical, and electrochemical properties of the polymer electrolyte significantly enhance the cycling stability of a Li–Li symmetric cell by effectively suppressing the growth of lithium dendrites.
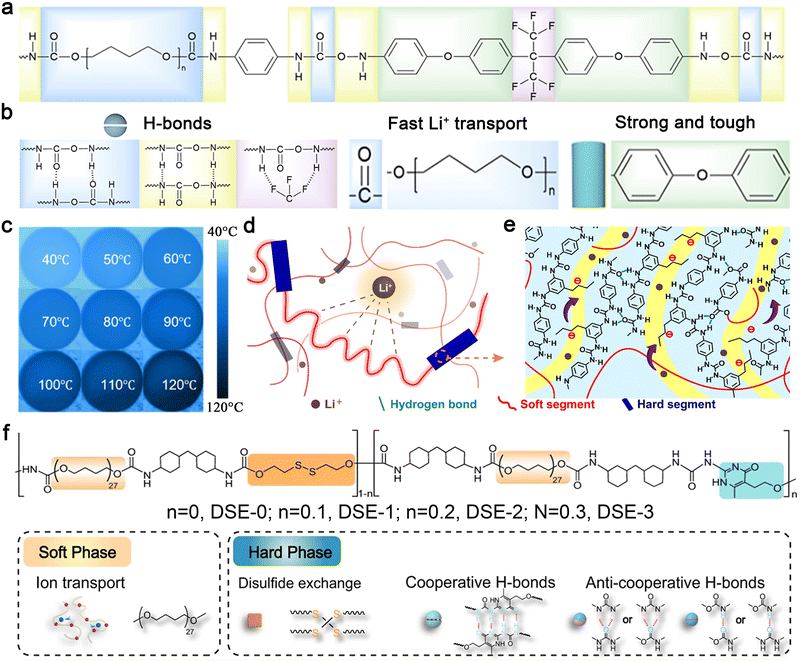 | ||
| Fig. 4 (a) Molecular formula of fluorine-containing supramolecular ionic conductive polyurethane elastomer (F-SMIU), (b) the role of each part of molecular design, (c) infrared thermogram of F-SMIU-9 (F-SMIU containing 9% filler) (40–120 °C). Reproduced from ref. 114, copyright 2025, Elsevier. Structural diagram of (d) a polyurethane-based anionic polymer with triflamide anions and (e) its hard phase. Reproduced from ref. 115, copyright 2023, American Chemical Society. (f) The chemical structure design and the proposed mechanism for the superior toughness of DSICE upon stretching. Reproduced from ref. 116, copyright 2022, Springer Nature. | ||
Wang et al. developed a polyurethane-based single-ion conducting polymer electrolyte with sulfonamide side chains to enable fast Li+ transport (Fig. 4d).115 The hard domains, composed of aromatic rings, and the soft phase, consisting of flexible poly(ethylene oxide), provide high mechanical strength and elasticity. Improved Li+ conductive pathways are achieved through covalent tethering of anionic sulfonamide side chains to the hard segments and the solvent effect of the amorphous soft phases (Fig. 4e). The intense hard segments in the backbone and strong hydrogen bonding as physical cross-links reduce phonon scattering, thereby enhancing the polymer's TC.
The dual-phase concept has also been adopted in the design of dynamic supramolecular ion-conducting elastomers (DSICE) in our group.116 This design effectively addresses the trade-off between ionic conductivity, mechanical compatibility, and thermal properties. Furthermore, the introduction of dynamic disulfide bonding and stronger supramolecular quadruple hydrogen bonding in the hard phase endows the supramolecular structure with self-healing capability and favorable recyclability (Fig. 4f). These advancements in polymer electrolyte design highlight the potential for developing high-performance, thermally stable, and mechanically robust electrolytes for next-generation solid-state batteries.
Incorporating well-dispersed inorganic fillers with high intrinsic TC and electrochemical inertness into polymer electrolytes is a straightforward method to enhance heat transfer in composite electrolytes. For example, Zheng et al. developed a novel polyethylene oxide (PEO)-based electrolyte with improved thermal response by incorporating 2D BN nanoflakes (Fig. 5a).121 The BN additive was found to enhance ionic conductivity, mechanical strength, and heat transport in the PEO-based electrolyte. As shown in Fig. 5b, the thermal diffusivity (α) and TC (κ) of BN-PEO-PVDF electrolytes were significantly higher than those of PEO-PVDF electrolytes, owing to the high TC of BN.122–124 Accordingly, the enhanced thermal response of the BN-PEO-PVDF electrolyte facilitates faster heat equalization and more homogeneous ionic transport in the composite electrolyte (Fig. 5c). The electrochemical behavior of the Li anode with the BN-PEO-PVDF electrolyte was investigated using a Li–Cu asymmetric cell. After Li deposition, a uniform stripping pattern is observed on the Li surface, and the plated Li metal on the Cu foil also appears uniform. In contrast, significant pits are observed on the Li foil surface in the cell with the PEO-PVDF electrolyte, and the deposited Li on the Cu side exhibits unevenly distributed dense and porous regions (Fig. 5d). Furthermore, both the Li metal anode and S cathode demonstrate uniform and stable transformations during electrochemical reactions in an all-solid-state Li–S cell with the BN-PEO-PVDF electrolyte, resulting in superior performance characterized by high specific capacity, as well as good cyclic and rate behaviors.
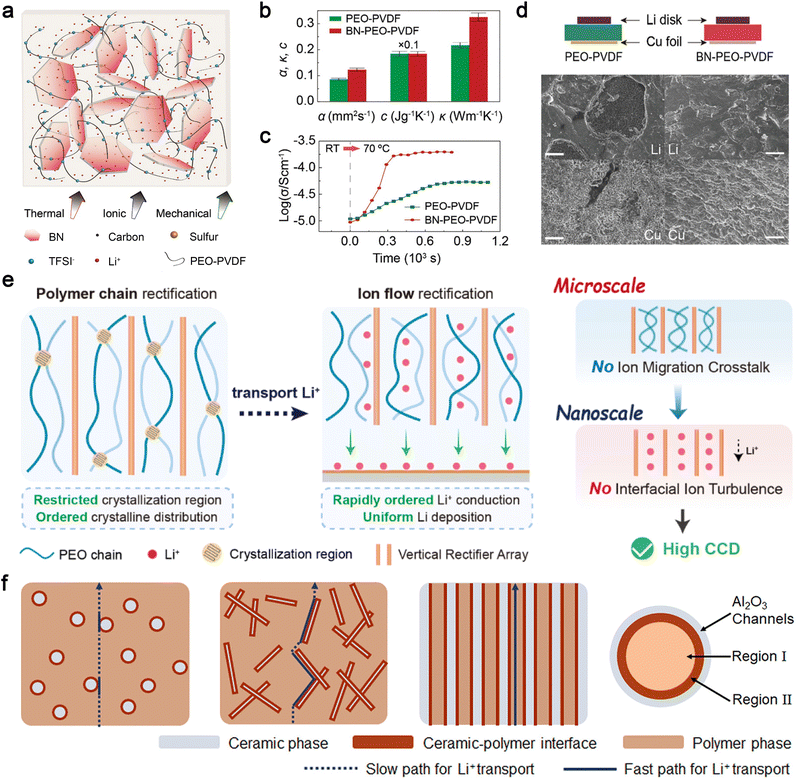 | ||
| Fig. 5 (a) Sketch of a composite electrolyte consisting of 2D BN flakes and a blended PEO-PVDF polymer with lithium bis(trifluoromethanesulfonyl)imide, (b) thermal diffusivity (α), heat capacity (c), and TC (κ) of PEO-PVDF and BN-PEO-PVDF electrolytes, (c) changes of ionic conductivities upon a temperature change, (d) sketch of Li–Cu cells and SEM images of Li metal on the surfaces of Li disks and Cu foils after Li deposition. Reproduced from ref. 121, copyright 2020, Wiley-VCH. (e) Schematic illustration of the critical current density enhancement of PEO electrolyte by a cross-scale rectification strategy. Reproduced from ref. 131, copyright 2024, Elsevier. (f) Schematics of composite solid polymer electrolytes with three types of geometrical structures of ceramic–polymer interface. Reproduced from ref. 137, copyright 2018, American Chemical Society. | ||
The overall TC of organic polymer materials is influenced by factors such as molecular chain structure, degree of crystallinity, molecular chain orientation, and interchain interactions.125,126 While high crystallinity is detrimental to ionic conductivity, improved orientation of polymer molecular chains (from nano- to microscale) benefits both thermal and ionic conductivity in polymer electrolytes.127–130 Ding et al. developed a novel ion rectifier consisting of vertical arrays of copper-ion montmorillonite (Cu-MMT) and gelatin.131 This design offers two key advantages: first, the regular micron-sized vertical array structure achieves polymer chain rectification on the microscale by suppressing crystalline growth and distribution in non-ionic conduction directions, which also enhances TC along the vertical direction. Second, the anchored anions on the Cu-MMT surface and the abundant oxygen-containing groups in gelatin weaken the binding of Li+ by the PEO chain, enabling fast and uniform Li+ diffusion on the nanoscale (Fig. 5e).
The Cui group has extensively studied the effects of nanofillers on the ionic conductivity and electrochemical stability of polymer electrolytes.132–136 Their research demonstrated that the oriented alignment of nanowire fillers significantly enhances ionic conductivity by providing prolonged fast transport pathways for Li+, as Li+ conduction at the interfaces of nanowire crossing junctions is poor (Fig. 5f).137 Additionally, the improved polymer orientation within the confined space of aligned ceramic nanotemplates synergistically enhances the TC of composite polymers. In this approach, the polymer material is melted and infiltrated into a porous template, which orders the molecular chains as they flow into the nanoscale pores, thereby increasing TC. Furthermore, the high-TC ceramic nanotemplate (e.g., Al2O3)138 provides additional TC pathways, achieving substantial heat dissipation enhancement through a “one stone, two birds” effect. These advancements highlight the potential of IPCEs to address critical challenges in battery performance by simultaneously improving thermal and ionic conductivity, mechanical strength, and electrochemical stability.
Fig. 6 illustrates the micrometer-scale structure of this solid electrolyte, which incorporates aligned poly-2,2′-disulfonyl-4,4′-benzidine terephthalamide (Li-form PBDT) grains interleaved with a nanocrystalline ionic phase. These interconnected nanocrystalline grain boundaries separate individual PBDT grains, forming an additional conductive network that facilitates fast Li+ transport. Furthermore, the aligned liquid crystal (LC) grains containing PBDT double helical rigid rods enhance the thermal properties of the molecular ionic composite (MIC), providing high thermal stability and conductivity.143 This innovative design leverages the confinement of polymer crystallinity domains to optimize both ionic and thermal transport, offering a promising solution for developing high-performance polymer electrolytes.
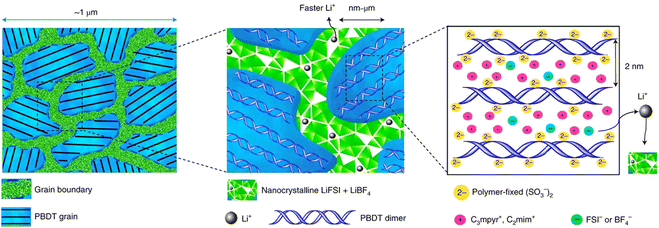 | ||
| Fig. 6 The microscale structure of a Li-loaded MIC. The grain boundaries are predominantly the condensed salt phase, which consists of nanocrystalline grains that form a conductive network supporting fast Li+ transport. The morphology of an aligned LC grain contains PBDT double helical rods filled predominantly with mobile ionic liquid cations. Reproduced from ref. 142, copyright 2021, Springer Nature. | ||
From another perspective, cathode materials in LIBs typically exhibit higher TC than polymer-based separators and electrolytes.145 Enhancing heat dissipation efficiency at the cathode/separator and cathode/collector interfaces is crucial to prevent internal heat accumulation and material fatigue.146 He et al. conducted both theoretical and experimental research on methodologies to facilitate interfacial heat transfer across material components.147 They found that small organic molecules (SOMs) with unique functional groups (e.g., –NH2) assembled at the cathode (LiCoO2, lithium cobalt oxide (LCO))-separator (PE) interface significantly enhance interfacial heat transfer. This improvement is attributed to the development of dual heat pathways through strong non-bonded interactions at the LCO-SOM and PE-SOM interfaces, high compatibility between SOMs and PE, and reduced phonon scattering.
Improving TC within the anode, the anode/collector interface, and the anode/electrolyte interface can also effectively prevent SEI decomposition by directly lowering the temperature below the critical point.154,155 To this end, incorporating high-TC materials or enhancing thermal contact through high compression are facile and effective approaches that can be adopted.156 Notably, a recently reported strategy for improving the thermal stability of zinc electrodes by enhancing heat transfer in zinc-ion batteries can also be applied to lithium metal batteries, as both systems operate under similar mechanisms. In this work, thermal transfer-enhanced layers were coated on both sides of a zinc foil.157 The top layer, composed of zinc–alginate, polyacrylamide, and BN, enables a uniform Zn2+ flux and temperature distribution, while the bottom layer, consisting of Ag/Cu coating, improves local heat diffusion and mechanical stability. This dual thermal protection effectively suppresses thermodynamically driven dendrite growth and side reactions.
External thermal regulation
External thermal management is critical for the safe operation of LIBs by directly dissipating excess heat generated during charge/discharge cycles.158 While numerous cooling technologies have been reported in recent years, this section focuses on recent advancements in the materials design of passive cooling, which offers notable advantages such as energy efficiency, compatibility, tunable functionality, and low maintenance cost. These include functional thermally conductive materials,159 PCMs,160,161 and radiative cooling materials.162In addition to the high-TC design principle, another strategy has emerged to manage both performance and safety of battery modules through rapid temperature-responsive thermal regulators.172–174 A thermal-switching material (TSM) composed of thermally expansive microspheres embedded between connected graphene layers has been designed, exhibiting a high switch ratio from thermal conduction to thermal insulation (Fig. 7a and b).173 Under normal operating conditions, the thermal regulator in its thermally conductive state (1.33 W m−1 K−1) buffers accumulated heat and reduces temperature variations between cells to less than 5 °C within 50 s, improving the electrochemical performance of the battery pack (Fig. 7c). When the temperature exceeds 100 °C, the TSM switches to a thermally insulating state to prevent thermal runaway propagation and battery explosions. It has been demonstrated that 80% of the total heat released during thermal runaway can be blocked by the responsive thermal-switching cell-to-cell TSM interlayer, successfully preventing uncontrolled chain reactions (Fig. 7d).
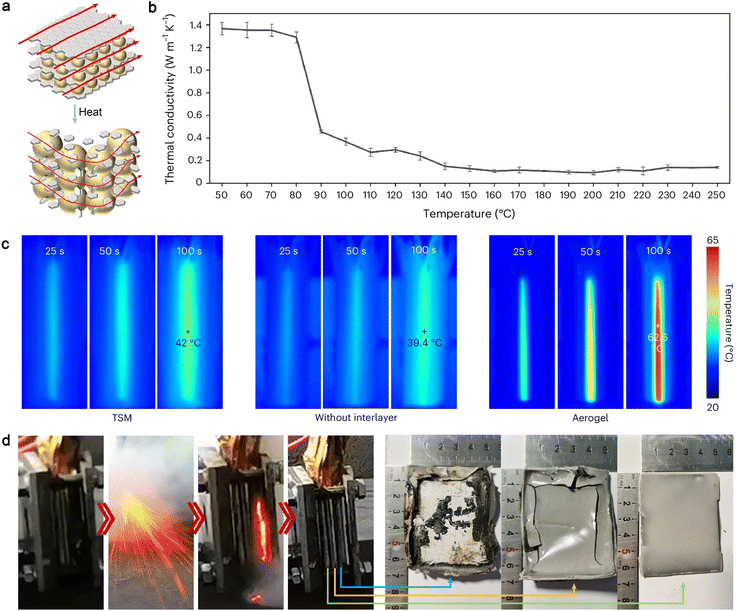 | ||
| Fig. 7 Schematic illustration of TSM design and thermal management test. (a) Thermal-switching mechanism of the TSM, (b) temperature-dependent TC of the TSM, (c) infrared images of battery modules with the TSM, with an aerogel and without an interlayer during the thermal dissipation tests, (d) the TSM remains functional as a smart thermal-protection layer throughout the thermal runaway propagation. The morphologies of the TSM change with the distance from the heat source. Reproduced from ref. 173, copyright 2024, Springer Nature. | ||
One-dimensional carbon fillers are well-suited for LIB thermal management due to their high axial TC, high strength, and high temperature resistance.187,188 Unlike disordered CNT arrangements, aligned CNTs leverage the intrinsic high axial TC of CNTs and provide continuous heat conduction pathways, significantly enhancing the thermal properties of CNT/PCM composites.189 Additionally, aligned CNTs exhibit higher paraffin loading capacity and significantly increase latent heat storage density.190 CNTs can also be combined with other 2D thermally conductive materials to create continuous pathways for heat transfer in PCCs. For example, Lin et al. introduced a novel CNT/MXene aerogel with a 3D porous structure for PCC preparation.191 The interconnected CNT pillars and MXene nanosheets, held together by hydrogen bonding and electronegativity, provide thermal transfer pathways in both horizontal and vertical directions, resulting in significant TC improvement (Fig. 8a). In battery thermal management, this composite demonstrates rapid improvement in temperature distribution, reducing the temperature rise rate from 1.85 °C to 0.92 °C under extreme high-rate (4C) charging/discharging conditions.
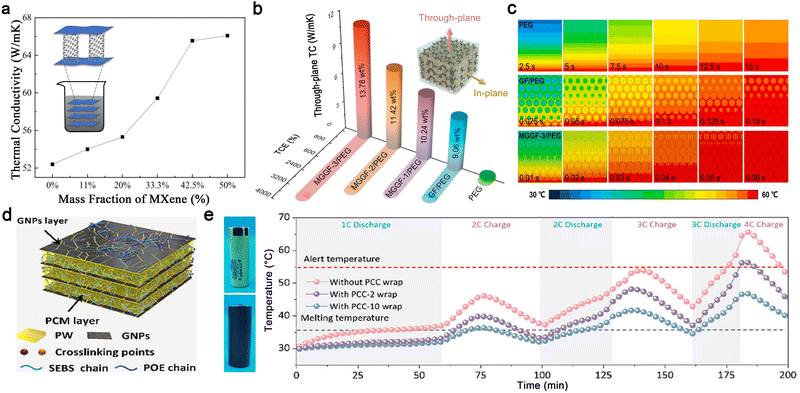 | ||
| Fig. 8 Carbon-based PCCs. (a) TC of the CNT@MXene aerogel with different mass ratios. (a) Reproduced with permission.191 copyright 2025, Elsevier. (b) Through-plane TC of PEG, GF/PEG, and MGGF/PEG, (c) simulated transient heat flux distribution. Reproduced from ref. 192, copyright 2023, Elsevier. (d) Phase change composite (PCC) with highly oriented layered structures, (e) surface temperature evolution of a battery with and without a PCC wrap during continuous charging and discharging. Reproduced from ref. 193, copyright 2025, Wiley-VCH. | ||
EG is widely used as a conductive skeleton due to its accessibility and low cost. Its hierarchical pores of micro-, meso-, and macro-sizes are particularly useful for encapsulating liquid PCMs, which are prone to leakage.194 EG/PCM composites with high latent heat and TC can effectively dissipate heat within a short time and prevent the propagation of thermal runaway processes.195,196 Notably, graphene, a well-known 2D material with high in-plane TC, can be constructed into 3D thermally conductive networks through strong intermolecular interactions or chemical bonding, further enhancing the TC of PCCs.197–199 Li et al. developed a PEG composite based on a dual-encapsulation design of vertically aligned MXene-graphene monoliths in graphene foam (MGGF).192 Impressively, the resultant MGGF/PEG composite achieved a high through-plane thermal conductivity of 11.39 W m−1 K−1 and a desirable latent heat density of up to 160.3 J g−1 (Fig. 8b). From the simulated transient heat flux distribution, it was shown that the heat flow is primarily distributed along the MGGF skeleton, which can notably promote the heat transfer rate by 2.5 times and 416.7 times compared to graphene foam (GF)/PEG and pure PEG, respectively (Fig. 8c).
Recently, incorporating functional polymers into PCCs has been shown to offer multiple advantages, such as reducing interfacial thermal resistance by enhancing contact between the composite PCMs and batteries, improving leak-proof properties through physical coating or chemical cross-linking, and providing high latent heat due to the use of multi-PCMs.11,200,201 Hu et al. designed a novel PCC using styrene–butadiene–styrene (SBS) thermoplastic elastomer as the polymer framework and CFs as thermally conductive fillers.202 The SBS framework provides intensive capillary condensation to adsorb the liquid-state paraffin PCM, thereby improving leak-proof properties. At an ambient temperature of 40 °C, the composite controlled the maximum temperature and temperature difference of the battery module to below 49.23 °C and 4.76 °C, respectively, at a high charge–discharge rate of 3C. These values are 9.71 °C and 1.89 °C lower than those achieved with natural air cooling. Moreover, a flexible, highly conductive, and recyclable PCC has been developed by employing a dual-polymer network of styrene–ethylene–butylene–styrene (SEBS) and polyolefin elastomers (POE), along with the integration of shear-induced alignment of graphene nanoplatelets (GNPs), as shown in Fig. 8d.193 The dynamic physical crosslinking network of SEBS and POE endows the PCCs with robust structural stability, tunable flexibility, and heat induced self-healing functionality. In the thermal management demonstration, the PCC-wrapped battery exhibits much lower temperature than the bare battery, particularly during high-rate charging and discharging (Fig. 8e), suggesting practical opportunities for preventing overheating and reducing the risk of thermal runaway in batteries, especially in fast charging and discharging processes.
Cheong et al. reported a vertically structured metal foam with superior heat diffusion performance compared to five other metal foam structures.206 The vertical structure achieves a faster melting rate of PCMs and uniform temperature distribution, and reduces the maximum temperature by approximately 96.8 °C. Wang et al. innovatively developed bionic antler-like microtopological copper fibers and applied them as a porous metal matrix to enhance phase change heat transfer.207 Experimental results demonstrated that the copper fiber sintered mat matrix enhances the internal heat transport efficiency in PCMs, with particularly significant TC improvement under high heat flux conditions. Additionally, studies have shown that gradient porosity and localized foam filling can further improve the heat transfer rate in PCMs, which should be considered in the design of porous metal-based PCCs.208
Recently, LMs have emerged as promising PCMs for thermal management in electronics and batteries due to their high TC and latent heat per unit volume.209 A scalable micro-encapsulated PCM and eutectic gallium–indium LM integrated composite demonstrated superior heat mitigation compared to pin-fin heat sinks, increasing full-load operation time by 4.14 times.210 This composite also shows potential for regulating transient temperature rises in fast-charging batteries under high heat flux conditions. However, gallium LM may suffer from severe supercooling, where molten gallium does not solidify due to the lack of nucleation sites. Ki et al. proposed a strategy of infusing gallium into porous copper to form intermetallic compound impurities at the interfaces, reducing the activation energy for heterogeneous nucleation.160 During repetitive heating–cooling cycles, porous-shaped gallium consistently exhibits crystallization propagation near room temperature while maintaining stable performance as a thermal buffer, making it suitable for mitigating rapid temperature increases in batteries.
Lee et al. developed a highly thermally conductive PCC using a porous ceramic skeleton of CF-crosslinked BN and a novel PCM derived from erythritol and bisphenol A grafting (denoted as ETBPA) (Fig. 9a).213 The results demonstrate that the in-plane TC of the composite exceeds 13.09 W m−1 K−1, which is more than 25 times higher than that of pure ETBPA, while maintaining a high latent heat of 98.4 J g−1 (Fig. 9b).
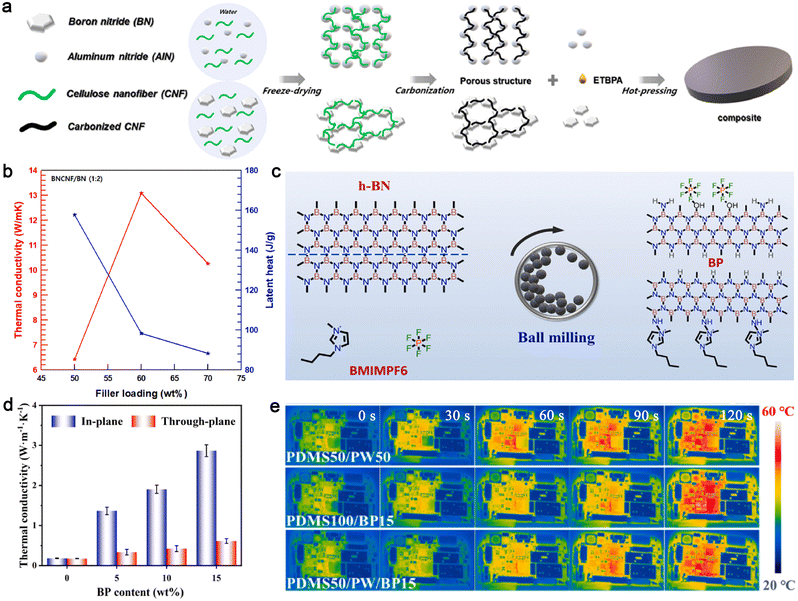 | ||
| Fig. 9 Ceramic-based PCCs. (a) Fabrication process of ETBPA composites, (b) TC and latent heat of the ETBPA/BNCNF/BN composites. Reproduced from ref. 213, copyright 2023, Elsevier. (c) Preparation process diagram of BPs, (d) K‖ and K⊥ of PDMS50/PW50/BP, (e) infrared thermal imaging picture of different composites for thermal management of mobile phones. Reproduced from ref. 214, copyright 2024, Elsevier. | ||
Additionally, the oriented alignment of BN can significantly enhance directional thermal transfer efficiency. To achieve this, Huang et al. developed a scraping coating technology to construct highly oriented BN nanosheets in a PDMS/PW matrix using strong shearing forces along the blade-casting direction.214 BN nanosheets modified with 1-butyl-3-methylimidazolium hexafluorophosphate (BMIMPF6) ionic liquid (denoted as BPs) were first prepared from bulk BN powders and BMIMPF6 ionic liquid through a one-step ball milling process (Fig. 9c). The resulting composites exhibited a high in-plane TC of 2.87 W m−1 K−1 due to the interconnected BN thermally conductive network (Fig. 9d). The high TC of the aligned BN network, combined with the thermal energy buffering of PW, enabled effective heat dissipation, reducing the working temperature of smartphones by over 11 °C (Fig. 9e).
Chen et al. reported an industry-scalable radiative cooling technology using hydromagnesite-based composites with excellent selective optical responses.222 Hydromagnesite (Mg5(CO3)4(OH)2·4H2O, Fig. 10a) has a crystal structure consisting of octahedral MgO6 centers and carbonate ions, both of which exhibit multiple mid-infrared vibrations. The manufacturing process for radiative cooling aluminum laminated foil (ALF), the commercially used outer packaging of pouch LIBs, involves a simple two-step process. Hydromagnesite particles and a high-density polyethylene (HDPE) matrix are melt-blended to form homogeneous compound particles, which are then hot-pressed onto aluminum foil to encapsulate the battery (Fig. 10b). The resulting radiative cooling ALF exhibits much higher emissivity than commercial ALF (Fig. 10c), enabling efficient thermal management of LIBs by regulating battery temperature within an optimal operating range at a high 3C discharge rate (Fig. 10d).
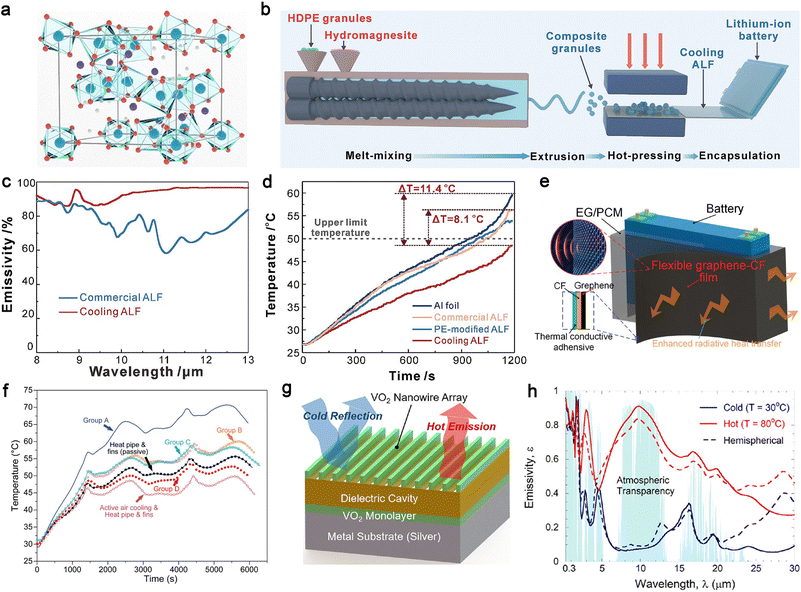 | ||
| Fig. 10 Thermal radiation materials. (a) Crystal structure of hydromagnesite in a polyhedral form. The Mg, O, C and H atoms are blue, red, purple, and white, respectively, (b) schematic diagram of the preparation process for radiative cooling ALF, (c) emissivity spectra of commercial and cooling ALFs, (d) comparison of temperature rise of LIBs encapsulated in different ALF samples at a 3C discharging rate. Reproduced from ref. 222, copyright 2025, Elsevier. (e) Schematics of the EG/PCM/graphene composite for passive battery thermal management, (f) thermal comparison among the various battery thermal management methods. Group D represents the EG/PCM/graphene composite, while Groups A, B, and C are control samples. Reproduced from ref. 223, copyright 2024, Wiley-VCH. (g) Illustration and schematic of VO2 nanowire/dielectric material/VO2 sub-monolayer coating on a Ag substrate, (h) the averaged emissivity spectrum of the optimized VO2 nanowire/BaF2/VO2 on Ag coating calculated with rigorous coupled-wave analysis. Reproduced from ref. 224, copyright 2022, Elsevier. | ||
However, addressing cooling demands with individual cooling technologies remains challenging in scenarios with substantially higher heat fluxes. To address this, Mao et al. proposed an integrated thermal management technology using an EG/PCM/graphene composite.223 The EG/PCM acts as a composite PCM with effective TC and latent heat, tightly wrapped around a battery pack. Nanostructured graphene is coated on the composite PCM with a copper foil in between to ensure efficient heat transfer. The graphene outer surface functions as a thermal radiator, efficiently dissipating the heat generated within the composite PCM (Fig. 10e). In battery cooling experiments at a 2.50C charge/discharge rate and 30 °C ambient temperature, the EG/PCM/graphene composite outperformed control samples with single cooling mechanisms (heat storage via phase change or radiative cooling). Furthermore, it demonstrated superior cooling performance compared to passive heat pipe approaches and was comparable to active air cooling and heat pipe schemes (Fig. 10f).
More recently, switchable radiative thermal management, which enables batteries to operate at optimal temperatures using emissivity-changeable materials under high and low temperatures, has gained attention for its adaptability to different application scenarios. Zhang et al. proposed a monolithic high-performance turn-down thermal emittance coating, consisting of a VO2 sub-wavelength nanowire grating top layer, an index-matched Fabry–Perot dielectric thin film middle layer, and an additional absorbing VO2 sublayer (Fig. 10g).224 Leveraging the insulator-to-metal temperature phase transition of VO2, this coating enables responsive passive radiative cooling through emissivity switching when the temperature exceeds the transition threshold (Fig. 10h). This innovation holds promising potential for self-cooling of batteries at high temperatures.
Conclusions and outlook
Conclusions
Based on a detailed analysis of the thermal runaway mechanisms in LIBs, it can be concluded that heat accumulation within LIBs leads to thermal runaway through a long-term process of chain (or crosstalk) reactions or dendrite growth-induced short circuits. Establishing efficient heat dissipation pathways both inside and outside the battery is crucial to mitigate or even prevent thermal runaway. Internal heat dissipation plays an irreplaceable role in achieving a mild and uniform temperature distribution within the cells, primarily relying on the rational design of highly thermally conductive materials without compromising electrochemical performance. Meanwhile, external thermal management is indispensable, and material-based passive cooling technologies provide a continuous, efficient, and cost-effective solution for overheating batteries. With a clear understanding of heat transfer mechanisms guiding the design of organic, inorganic, and composite materials, this review emphasizes improving the intrinsic TC of polymers through molecular structure and orientation engineering, as well as synergistically enhancing TC in composites through highly efficient thermal conduction networks. Additionally, recent advancements in multifunctional materials featuring switchable thermal and ionic conductivity, high thermal storage capacity, and responsive radiative cooling have opened new avenues for research in intelligent thermal management technologies.Outlook
The ever-growing demand for high-density and fast-charging LIBs has imposed higher requirements for safe battery materials. However, reconciling the electrochemical and thermal performance of key materials remains a significant challenge. Unfortunately, electrochemical performance has often been prioritized in application-driven material design. Therefore, more systematic studies are required to address thermal issues without compromising electrochemical properties.(1) Although solid-state LIBs are regarded as the next generation of high-density, safe, and fast-charging batteries, heat accumulation within the battery poses considerable safety risks, particularly at high charge/discharge rates, and should not be neglected. In polymer-based solid electrolytes, ionic conductivity is facilitated by flexible chain segments and polar groups, whereas phonon transport relies on rigid chain segments and multiple intermolecular hydrogen bonds. However, stronger intermolecular interactions can significantly restrict the motion of polymer chains, resulting in reduced ionic conductivity. This introduces a trade-off when integrating both soft and hard phases into the polymer structures. Decoupling the thermal and electrochemical properties of polymers through structural design remains a significant challenge.
(2) Organic–inorganic composite electrolytes, which combine the advantages of organic polymers and inorganic fillers, are regarded as the most promising solid electrolytes for high-performance solid-state LIBs. From a heat dissipation perspective, these composite electrolytes provide a versatile approach to synergistically enhance both thermal and ionic conductivity. Aligned inorganic templates with high thermal conductivity (e.g., BN, Al2O3) not only facilitate heat dissipation within the composites but also induce molecular orientation upon infiltration, thereby simultaneously improving thermal and ionic conductivity. This holds promising potential for enhancing both safety and electrochemical performance. Further mechanistic investigations into the effects of polymer/inorganic interfacial interactions on phonon scattering, ionic dissociation, and transport behavior are crucial to advancing the development of these materials.
(3) Heat accumulation can be effectively mitigated through the design of highly thermally conductive materials for both internal cell components and external thermal management systems. However, in cases of external abuse triggering instantaneous release of enormous amounts of heat during thermal runaway, existing heat dissipation mechanisms may fail to cool the overheated battery, posing a high risk of fire or explosion. Under such extreme abuse conditions, integrating switchable shutdown functions, derived from the temperature-dependent reversible expansion and contraction of polymer materials, into thermally conductive components within or between cells provides a feasible and rapid response mechanism to mitigate thermal runaway risks.
(4) This review primarily focuses on preventing thermal runaway by dissipating excess heat from the cell and maintaining a homogeneous temperature distribution under optimal working conditions. As a key aspect of external thermal management technologies, composite PCMs with high TC play a dual role: storing heat in cooling mode at high temperatures and feeding heat back at low temperatures when batteries are used in cold regions during winter. This multifunctional thermal management approach can be further enhanced by incorporating passive radiative cooling technology utilizing a VO2 metasurface with tunable thermal emittance, providing a smart, continuous, energy-efficient, and versatile thermal management solution for LIBs.
Author contributions
Songpei Nan: writing – original draft preparation, visualization, review & editing. Guoxin Gao & Wei Yu: visualization, review & editing, funding acquisition. Shujiang Ding: supervision, review & editing, funding acquisition. Dawei Ding: writing – original draft preparation, supervision, review & editing, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by the Natural Science Basic Research Program of Shaanxi (No. 2023-JC-YB-338, No. 2022TD-27), the ‘Scientist+ Engineer’ Team Construction Project funded by the Science and Technology Bureau of Xi'an City (No. 25KGYB00022), the Key projects of Shannxi Province, China (No. 2023GXLH-001) and the National Natural Science Foundation of China (No. 52573093).References
- K. Liu, Y. Liu, D. Lin, A. Pei and Y. Cui, Sci. Adv., 2018, 4, eaas9820 Search PubMed.
- Y. Wang, X. Feng, W. Huang, X. He, L. Wang and M. Ouyang, Adv. Energy Mater., 2023, 13, 2203841 CrossRef CAS.
- X. Tian, Y. Yi, B. Fang, P. Yang, T. Wang, P. Liu, L. Qu, M. Li and S. Zhang, Chem. Mat., 2020, 32, 9821–9848 CrossRef CAS.
- M. Keyser, A. Pesaran, Q. Li, S. Santhanagopalan, K. Smith, E. Wood, S. Ahmed, I. Bloom, E. Dufek, M. Shirk, A. Meintz, C. Kreuzer, C. Michelbacher, A. Burnham, T. Stephens, J. Francfort, B. Carlson, J. Zhang, R. Vijayagopal, K. Hardy, F. Dias, M. Mohanpurkar, D. Scoffield, A. N. Jansen, T. Tanim and A. Markel, J. Power Sources, 2017, 367, 228–236 Search PubMed.
- Y. Liu, Y. Zhu and Y. Cui, Nat. Energy, 2019, 4, 540–550 Search PubMed.
- M. T. F. Rodrigues, G. Babu, H. Gullapalli, K. Kalaga, F. N. Sayed, K. Kato, J. Joyner and P. M. Ajayan, Nat. Energy, 2017, 2, 17108 Search PubMed.
- S. Rana, R. Kumar and R. S. Bharj, Chem. Eng. J., 2023, 463, 142336 Search PubMed.
- X. Feng, M. Ouyang, X. Liu, L. Lu, Y. Xia and X. He, Energy Storage Mater., 2018, 10, 246–267 Search PubMed.
- D. Bao, Y. Gao, Y. Cui, F. Xu, X. Shen, H. Geng, X. Zhang, D. Lin, Y. Zhu and H. Wang, Chem. Eng. J., 2022, 433, 133519 CrossRef CAS.
- H. B. Yin, S. Y. Gao, C. C. Liao, C. M. Li, Z. D. Cai, Y. J. Xu and J. Liu, J. Cleaner Prod., 2019, 235, 359–368 CrossRef CAS.
- C. K. Zhang, X. J. Zheng, N. Xie, Y. T. Fang, Z. G. Zhang and X. N. Gao, J. Energy Storage, 2023, 58, 106364 CrossRef.
- Y. Zeng, F. Liu, Q. Zhang, D. Cheng, Y. Xu, S.-S. Chi, X. Xu, C. Wang, J. Wang, K. Xu, Y. Deng and H. Xu, Joule, 2025, 9, 102100 Search PubMed.
- M. N. Shen, Y. Wei, M. Ge, S. D. Yu, R. H. Dou, L. H. Chen, F. Wang, Y. H. Huang and H. H. Xu, Interdiscip. Mater., 2024, 3, 791–800 Search PubMed.
- M. Y. Ma, R. Z. Huang, M. Ling, Y. S. Hu and H. L. Pan, Interdiscip. Mater., 2023, 2, 833–854 Search PubMed.
- S. H. Zhang, F. Sun, X. F. Du, X. H. Zhang, L. Huang, J. Ma, S. M. Dong, A. Hilger, I. Manke, L. S. Li, B. Xie, J. D. Li, Z. W. Hu, A. C. Komarek, H. J. Lin, C. Y. Kuo, C. T. Chen, P. X. Han, G. J. Xu, Z. L. Cui and G. L. Cui, Energy Environ. Sci., 2023, 16, 2591–2602 Search PubMed.
- W. Y. Liu, Y. Z. Zhao, C. J. Yi, W. F. Hu, J. L. Xia, Y. Y. Li and J. P. Liu, Energy Environ. Mater., 2024, 7, e12719 CrossRef CAS.
- Z. Ye, C.-c. Zheng, Z.-j. Tan and Z. Li, Energy Storage Mater., 2024, 73, 103790 Search PubMed.
- X. Y. Liu, J. C. Chu, S. Xue, D. Q. Wang, Z. Y. Lu, M. Zhang, Y. Q. Liu, X. Xu, Y. L. Zhang, J. A. Long, L. J. Meng, J. Y. Yuan and M. G. He, Adv. Mater., 2025, 37, 2502086 CrossRef CAS.
- J. G. Liu, B. H. Li, J. H. Cao, X. Xing and G. Cui, J. Energy Chem., 2024, 91, 73–98 Search PubMed.
- K. Liu, W. Liu, Y. Qiu, B. Kong, Y. Sun, Z. Chen, D. Zhuo, D. Lin and Y. Cui, Sci. Adv., 2017, 3, e1601978 CrossRef PubMed.
- K. Li, A. Hu, R. Xu, W. Xu, B. Yang, T. Li, Y. Li, Z. W. Seh, J. Long and S. Chen, Adv. Energy Mater., 2025, 15, 2501236 CrossRef CAS.
- Y. Guo, S. Wu, Y.-B. He, F. Kang, L. Chen, H. Li and Q.-H. Yang, eScience, 2022, 2, 138–163 Search PubMed.
- Q. Wang, B. Mao, S. I. Stoliarov and J. Sun, Prog. Energy Combust. Sci., 2019, 73, 95–131 CrossRef.
- Z. Liu, Y. Jiang, Q. Hu, S. Guo, L. Yu, Q. Li, Q. Liu and X. Hu, Energy Environ. Mater., 2021, 4, 336–362 CrossRef CAS.
- M. Zhang, L. Wang, H. Xu, Y. Song and X. He, Nano-Micro Lett., 2023, 15, 135 Search PubMed.
- C. M. Costa, Y.-H. Lee, J.-H. Kim, S.-Y. Lee and S. Lanceros-Mendez, Energy Storage Mater., 2019, 22, 346–375 CrossRef.
- J. Q. Zhu, J. Z. Cui, S. J. Wang, Y. C. Zhang, H. Ma, Z. Liu, Y. L. Liu, P. Du, J. Y. Ma, Y. X. Zhang, J. Q. Wang and L. J. Zhang, Adv. Funct. Mater., 2025, e16197 Search PubMed.
- L. N. Liu, B. Y. Yu, L. Y. Q. Kang, W. W. Deng and X. Y. Zhao, Adv. Funct. Mater., 2023, 33, 2214781 Search PubMed.
- K. Du, A. Rudola and P. Balaya, ACS Appl. Mater. Interfaces, 2021, 13, 11732–11740 Search PubMed.
- M. Jiang, D. L. Danilov, R.-A. Eichel and P. H. L. Notten, Adv. Energy Mater., 2021, 11, 2103005 CrossRef CAS.
- I. T. Song, J. Kang, J. Koh, H. Choi, H. Yang, E. Park, J. Lee, W. Cho, Y.-m. Lee, S. Lee, N. Kim, M. Lee and K. Kim, Nat. Commun., 2024, 15, 8294 CrossRef.
- Q. Yu, W. Sun, J. Zhu, H. Li, S. Wang, L. Huang, Q. Qiu, H. Tian, H. Miao, F. Wang, C. Zhang, J. Yuan and L. Xia, J. Energy Chem., 2025, 101, 76–86 CrossRef CAS.
- J. N. Zhang, H. Wu, X. F. Du, H. Zhang, L. Huang, F. Sun, T. T. Liu, S. W. Tian, L. X. Zhou, S. J. Hu, Z. X. Yuan, B. T. Zhang, J. J. Zhang and G. L. Cui, Adv. Energy Mater., 2023, 13, 2202529 CrossRef CAS.
- H. R. Zhang, L. Huang, H. T. Xu, X. H. Zhang, Z. Chen, C. H. Gao, C. L. Lu, Z. Liu, M. F. Jiang and G. L. Cui, eScience, 2022, 2, 201–208 CrossRef CAS.
- Y. Xiao, A. Fu, Y. Zou, L. Huang, H. Wang, Y. Su and J. Zheng, Chem. Eng. J., 2022, 438, 135550 CrossRef CAS.
- J.-C. Guo, C.-Z. Chai, Y.-H. Wang, Y. Zhao, S. Xin, Y. Zhang, Y.-G. Guo and C. Bai, Proc. Natl. Acad. Sci. U. S. A., 2025, 122, e2501549122 CrossRef CAS.
- Z. Chen, P.-C. Hsu, J. Lopez, Y. Li, J. W. F. To, N. Liu, C. Wang, S. C. Andrews, J. Liu, Y. Cui and Z. Bao, Nat. Energy, 2024, 9, 623 CrossRef.
- C. R. Zhang, H. Li, S. X. Wang, Y. L. Cao, H. X. Yang, X. P. Ai and F. P. Zhong, J. Energy Chem., 2020, 44, 33–40 CrossRef.
- Z. Z. Liu, Q. Yu, N. Oli, J. F. F. Gomez, S. Qiu, H. R. Tian, Q. Qiu, W. Sun, K. H. Li, Z. S. Liu, M. M. Chen, J. L. Yuan, X. Y. Wu and L. Xia, Adv. Energy Mater., 2023, 13, 2300143 CrossRef CAS.
- Z. F. Liu, Y. T. Peng, T. Meng, L. Yu, S. Wang and X. L. Hu, Energy Storage Mater., 2022, 47, 445–452 CrossRef.
- Y. M. Deng, Z. Wang, Z. Ma and J. M. Nan, Energy Technol., 2020, 8, 1901037 CrossRef CAS.
- Q. Zhou, S. M. Dong, Z. L. Lv, G. J. Xu, L. Huang, Q. L. Wang, Z. L. Cui and G. L. Cui, Adv. Energy Mater., 2020, 10, 1903441 CrossRef CAS.
- Z. Chen, P. C. Hsu, J. Lopez, Y. Z. Li, J. W. F. To, N. Liu, C. Wang, S. C. Andrews, J. Liu, Y. Cui and Z. N. Bao, Nat. Energy, 2016, 1, 15009 CrossRef CAS.
- K. Le, C. C. Sang, Q. J. Luo, H. Li, Y. J. Fang and X. P. Ai, eTransportation, 2025, 26, 100483 CrossRef.
- T. T. Dong, G. J. Xu, B. Xie, T. Liu, T. Y. Gong, C. H. Sun, J. Z. Wang, S. Zhang, X. H. Zhang, H. R. Zhang, L. Huang and G. L. Cui, Adv. Mater., 2024, 36, 2400737 CrossRef CAS.
- D. S. Jang, S. Yun, S. H. Hong, W. Cho and Y. Kim, Energy Convers. Manage., 2022, 251, 115001 CrossRef.
- Y. S. Han, H. Tetik and M. H. Malakooti, Adv. Mater., 2024, 36, 2407073 CrossRef CAS PubMed.
- P. Ping, X. Y. Dai, D. P. Kong, Y. Zhang, H. L. Zhao, X. Z. Gao and W. Gao, Chem. Eng. J., 2023, 463, 142401 CrossRef CAS.
- Q. Liu, F. Z. Liu, S. J. Liu, Y. Wang, S. Panchal, M. Fowler, R. Fraser, J. L. Yuan and J. P. Zhao, J. Energy Storage, 2025, 109, 115234 CrossRef CAS.
- S. J. Zhu, L. Miao, J. Gao, J. L. Chen, Q. Zhou, Z. N. Pan, Z. W. Zhang, J. S. Liang, X. Y. Yang and T. Mori, Nano Energy, 2025, 144, 111328 CrossRef CAS.
- V. Tugan and U. Yardimci, J. Energy Storage, 2023, 72, 108515 CrossRef.
- Y. Ma, H. Ding, H. Y. Mou and J. W. Gao, Measurement, 2021, 186, 110115 CrossRef.
- X. Q. Sun, L. Ling, S. G. Liao, Y. H. Chu, S. Y. Fan and Y. J. Mo, Energy Convers. Manage., 2018, 155, 230–242 CrossRef.
- S. Vignesh, H. S. Che, J. Selvaraj, K. S. Tey, H. Shareef and R. Errouissi, Renewable Sustainable Energy Rev., 2025, 213, 115439 CrossRef.
- J. Xie, J. Li, C. Li, X. Huang, G. Zhang and X. Yang, eTransportation, 2025, 26, 100467 CrossRef.
- L. C. Kong, Y. Li and W. Feng, Electrochem. Energy Rev., 2021, 4, 633–679 CrossRef CAS.
- M. N. Li, J. M. Yuan, K. M. Wang, Z. Zhang, H. Z. Niu, L. L. Tan, Z. C. Miao and X. G. Han, Energy Storage Mater., 2025, 77, 104163 CrossRef.
- X. K. Hu, Y. Wang, X. N. Feng, L. Wang, M. G. Ouyang and Q. Zhang, Renewable Sustainable Energy Rev., 2025, 207, 114949 CrossRef CAS.
- F. S. Hwang, T. Confrey, C. Reidy, D. Picovici, D. Callaghan, D. Culliton and C. Nolan, Renewable Sustainable Energy Rev., 2024, 192, 114171 CrossRef CAS.
- S. A. Khan, I. Hussain, A. K. Thakur, S. Yu, K. T. Lau, S. H. He, K. J. Dong, J. T. Chen, X. R. Li, M. Ahmad and J. Y. Zhao, Energy Storage Mater., 2024, 65, 103144 CrossRef.
- Manisha, S. Tiwari, R. K. Sahdev, D. Chhabra, M. Kumari, A. Ali, R. Sehrawat and P. Tiwari, Renewable Sustainable Energy Rev., 2025, 209, 115089 CrossRef.
- Y. Zhang, J. Feng, J. Qin, Y. L. Zhong, S. Zhang, H. Wang, J. Bell, Z. Guo and P. Song, Adv. Sci., 2023, 10, 2301056 CrossRef CAS PubMed.
- S. Mallick and D. Gayen, J. Energy Storage, 2023, 62, 106894 CrossRef.
- X. Feng, D. Ren, X. He and M. Ouyang, Joule, 2020, 4, 743–770 CrossRef CAS.
- R. Chen, A. M. Nolan, J. Lu, J. Wang, X. Yu, Y. Mo, L. Chen, X. Huang and H. Li, Joule, 2020, 4, 812–821 CrossRef CAS.
- J. Liu, Z. Huang, J. Sun and Q. Wang, J. Power Sources, 2022, 526, 231136 CrossRef CAS.
- Y.-S. Duh, Y. Sun, X. Lin, J. Zheng, M. Wang, Y. Wang, X. Lin, X. Jiang, Z. Zheng, S. Zheng and G. Yu, J. Energy Storage, 2021, 41, 102888 CrossRef.
- X. Liu, D. Ren, H. Hsu, X. Feng, G.-L. Xu, M. Zhuang, H. Gao, L. Lu, X. Han, Z. Chu, J. Li, X. He, K. Amine and M. Ouyang, Joule, 2018, 2, 2047–2064 CrossRef CAS.
- D. Ren, X. Feng, L. Lu, X. He and M. Ouyang, Appl. Energy, 2019, 250, 323–332 CrossRef CAS.
- Y. Li, X. Liu, L. Wang, X. Feng, D. Ren, Y. Wu, G. Xu, L. Lu, J. Hou, W. Zhang, Y. Wang, W. Xu, Y. Ren, Z. Wang, J. Huang, X. Meng, X. Han, H. Wang, X. He, Z. Chen, K. Amine and M. Ouyang, Nano Energy, 2021, 85, 105878 CrossRef CAS.
- L. Huang, G. Xu, X. Du, J. Li, B. Xie, H. Liu, P. Han, S. Dong, G. Cui and L. Chen, Adv. Sci., 2021, 8, 2100676 CrossRef CAS.
- Z. X. Jiang, C. G. Liu, L. Huang, S. S. Zhu, X. H. Zhang, R. X. Wu, T. Y. Gong, Y. H. Wu, L. X. Guo, P. X. Han, J. Ma, G. J. Xu and G. L. Cui, Energy Environ. Sci., 2025, 18, 8232–8243 RSC.
- C. Xu, X. Feng, W. Huang, Y. Duan, T. Chen, S. Gao, L. Lu, F. Jiang and M. Ouyang, J. Energy Storage, 2020, 31, 101670 CrossRef.
- H. Wang, Z. Du, X. Rui, S. Wang, C. Jin, L. He, F. Zhang, Q. Wang and X. Feng, J. Hazard. Mater., 2020, 393, 122361 CrossRef CAS PubMed.
- L. Huang, T. Lu, G. J. Xu, X. H. Zhang, Z. X. Jiang, Z. Q. Zhang, Y. T. Wang, P. X. Han, G. L. Cui and L. Q. Chen, Joule, 2022, 6, 906–922 CrossRef CAS.
- X. J. Guo, Y. Yang, C. W. Shi, M. J. Xu, Y. F. Liu and D. Q. Zou, Energy Storage Mater., 2025, 75, 104051 CrossRef.
- L. Y. Zhou, H. W. Pan, G. J. Yin, Y. Xiang, P. P. Tan, X. Li, Y. Z. Jiang, M. W. Xu and X. Zhang, Adv. Funct. Mater., 2024, 34, 2314246 CrossRef CAS.
- M. J. Lee, J. Han, K. Lee, Y. J. Lee, B. G. Kim, K. N. Jung, B. J. Kim and S. W. Lee, Nature, 2022, 601, 217–222 CrossRef CAS PubMed.
- I. Kim, H. Kang, S. Yoon, J. B. Lee, H. W. Kim, H. K. Kim and M. Kim, Energy Storage Mater., 2024, 72, 103739 CrossRef.
- K. Fu, Y. H. Gong, J. Q. Dai, A. Gong, X. G. Han, Y. G. Yao, C. W. Wang, Y. B. Wang, Y. N. Chen, C. Y. Yan, Y. J. Li, E. D. Wachsman and L. B. Hu, Proc. Natl. Acad. Sci. U. S. A., 2016, 113, 7094–7099 CrossRef CAS.
- Y. Lu, Z. L. Wu, G. Cheng, T. A. Venkatesh, G. J. Liu, Y. Z. Bai, L. Yu and N. W. Li, Chem. Eng. J., 2023, 461, 142067 CrossRef CAS.
- X. X. Liang, Y. Yang, X. Jin, Z. H. Huang and F. Y. Kang, J. Membr. Sci., 2015, 493, 1–7 CrossRef CAS.
- H. J. Zhao, N. P. Deng, G. Wang, H. R. Ren, W. M. Kang and B. W. Cheng, Chem. Eng. J., 2021, 404, 126542 CrossRef CAS.
- Q. Xu, Q. S. Kong, Z. H. Liu, X. J. Wang, R. Z. Liu, J. J. Zhang, L. P. Yue, Y. L. Duan and G. L. Cui, ACS Sustain. Chem. Eng., 2014, 2, 194–199 CrossRef CAS.
- H. Zhang, J. Liu, M. Guan, Z. Shang, Y. W. Sun, Z. H. Lu, H. L. Li, X. Y. An and H. B. Liu, ACS Sustain. Chem. Eng., 2018, 6, 4838–4844 CrossRef CAS.
- H. Lim, H. N. Na, E. J. Jung, W. Ahn, J. B. Park, J. Hong and Y. W. Lee, Int. J. Energy Res., 2024, 2024, 3314308 CrossRef.
- S. L. Liu, B. Fan, Z. Shi, R. D. Wan, X. X. Sheng, X. L. Li, C. B. Zhu, M. N. Chen, Z. G. Xue, Y. Ding, X. Lu and J. P. Qu, ACS Appl. Mater. Interfaces, 2024, 16, 30284–30295 CrossRef CAS PubMed.
- S. S. Zhang, J. Power Sources, 2007, 164, 351–364 CrossRef CAS.
- C. J. Weber, S. Geiger, S. Falusi and M. Roth, AIP Conf. Proc., 2014, 1597, 66–81 CrossRef CAS.
- K. Wang, W. J. Wang, Y. Wang and M. Wang, Chem. Eng. J., 2024, 481, 148538 CrossRef CAS.
- H. T. Lu, A. Du, X. P. Lin, Z. Y. Zhang, S. S. Liu, Y. S. Xie, W. H. Li, J. W. Song, Y. H. Lu, W. Chen, C. P. Yang and Q. H. Yang, Energy Environ. Sci., 2024, 17, 7860–7869 RSC.
- Y. Kim, C. Li, J. Huang, Y. F. Yuan, Y. Tian and W. Zhang, Adv. Mater., 2024, 36, 2407761 CrossRef CAS.
- W. Z. Liu, X. H. Meng, Z. Y. Zhou, Q. Zheng, J. L. Shi, Y. Gong and Y. G. Guo, J. Energy Chem., 2024, 98, 123–133 CrossRef CAS.
- J. X. Hou, X. N. Feng, L. Wang, X. Liu, A. Ohma, L. G. Lu, D. S. Ren, W. S. Huang, Y. Li, M. C. Yi, Y. Wang, J. Q. Ren, Z. H. Meng, Z. Y. Chu, G. L. Xu, K. Amine, X. M. He, H. W. Wang, Y. Nitta and M. G. Ouyang, Energy Storage Mater., 2021, 39, 395–402 CrossRef.
- Y. X. Ma, J. Y. Wan, Y. F. Yang, Y. S. Ye, X. Xiao, D. T. Boyle, W. Burke, Z. J. Huang, H. Chen, Y. Cui, Z. A. Yu and S. T. Oyakhire, Adv. Energy Mater., 2022, 12, 2103720 CrossRef CAS.
- L. H. Liu, D. M. Zhang, T. R. Yang, W. H. Hu, X. L. Meng, J. S. Mo, W. Y. Hou, Q. X. Fan, K. Liu, B. Jiang, L. H. Chu and M. C. Li, J. Energy Chem., 2022, 75, 360–368 Search PubMed.
- L. G. Zhang, N. P. Deng, J. B. Kang, X. X. Wang, H. J. Gao, Y. R. Liu, H. Wang, G. Wang, B. W. Cheng and W. M. Kang, J. Energy Chem., 2023, 77, 326–337 CrossRef CAS.
- G. Su, Z. X. Zhang, M. Xiao, S. J. Wang, S. Huang, H. Guo, D. M. Han and Y. Z. Meng, Chem. Eng. J., 2024, 500, 157050 CrossRef CAS.
- L. J. Wang, H. D. Shi, Y. P. Xie and Z. S. Wu, Interdiscip. Mater., 2023, 2, 789–799 Search PubMed.
- J. Lee, Y. Kim, S. R. Joshi, M. S. Kwon and G. H. Kim, Polym. Chem., 2021, 12, 975–982 RSC.
- W. B. Lin, Y. F. Li, X. R. Liu, R. Xu, J. J. Huang, Z. Y. Jiang, Z. G. Qu, K. Xi and Y. Lin, Mater. Horiz., 2025, 12, 6765–6773 RSC.
- N. Jiang, Y. Y. Song, L. N. Wang, W. W. Liu, L. Bai, J. Yang and W. Yang, Adv. Funct. Mater., 2025, 35, 2416277 Search PubMed.
- D. X. Hu, H. Z. Huang, C. Y. Wang, Q. X. Hong, H. L. Wang, S. H. Tang, H. J. Zhang, J. S. Li, L. Y. Hu, L. Jiang, X. W. Fu, J. X. Lei, Z. M. Liu and X. He, Adv. Energy Mater., 2025, 15, 2406176 CrossRef CAS.
- Y. Meng, J. L. Hu, Q. J. Yu, M. Lei, H. L. Wu, Y. Xu, R. Qian and C. L. Li, J. Energy Chem., 2025, 110, 153–164 CrossRef CAS.
- M. H. Lin, M. G. Mohamed, C. J. Lin, Y. J. Sheng, S. W. Kuo and C. L. Liu, Adv. Funct. Mater., 2024, 34, 2406165 CrossRef CAS.
- H. Maeda, S. Wu, R. Marui, E. Yoshida, K. Hatakeyama-Sato, Y. Nabae, S. Nakagawa, M. Ryu, R. Ishige, Y. Noguchi, Y. Hayashi, M. Ishii, I. Kuwajima, F. Jiang, X. T. Vu, S. Ingebrandt, M. Tokita, J. Morikawa, R. Yoshida and T. Hayakawa, npj Comput. Mater., 2025, 11, 205 CrossRef CAS.
- X. Rodríguez-Martínez, F. Saiz, B. Dörling, S. Marina, J. L. Guo, K. Xu, H. Chen, J. Martin, I. McCulloch, R. Rurali, J. S. Reparaz and M. Campoy-Quiles, Adv. Energy Mater., 2024, 14, 2401705 CrossRef.
- H. T. Zhang, Y. Q. Guo, Y. Z. Zhao, Q. Y. Zhu, M. K. He, H. Guo, X. T. Shi, K. P. Ruan, J. Kong and J. W. Gu, Angew. Chem., Int. Ed., 2025, 64, e202500173 CrossRef CAS PubMed.
- G. Su, M. N. Geng, L. Zhong, M. Xiao, S. J. Wang, S. Huang, H. Guo, D. M. Han and Y. Z. Meng, J. Energy Chem., 2025, 103, 803–812 CrossRef CAS.
- K. Li, Z. Wang, B. R. Yang, T. Li, B. Li, J. H. Chen, Z. F. Yan, M. He, A. J. Hu and J. P. Long, Chem. Eng. J., 2024, 493, 152527 CrossRef CAS.
- C. L. Deng, B. B. Yang, Y. H. Liang, Y. Zhao, B. S. Gui, C. Y. Hou, Y. X. Shang, J. X. Zhang, T. L. Song, X. Z. Gong, N. Chen, F. Wu and R. J. Chen, Angew. Chem., Int. Ed., 2024, 63, e202400619 Search PubMed.
- H. N. Wang, J. J. Yang, X. Y. Xu, J. Geng, X. Lin, H. H. Xu and Y. H. Huang, Adv. Mater., 2025, 37, e04625 CrossRef CAS PubMed.
- T. W. Hao, Y. J. Shen, C. L. Yang, M. Q. Ma, W. H. Ruan and M. S. Lin, Chem. Eng. J., 2025, 521, 166741 CrossRef CAS.
- Y. Zhou, Y. H. Li, X. Y. Liu, J. H. Lv, Y. Q. Su and L. Weng, Energy Storage Mater., 2025, 78, 104265 CrossRef.
- N. J. Wang, X. Q. Chen, Q. Sun, Y. Song and T. Z. Xin, ACS Appl. Mater. Interfaces, 2023, 15, 39837–39846 CrossRef CAS.
- J. Chen, Y. Y. Gao, L. Shi, W. Yu, Z. J. Sun, Y. F. Zhou, S. Liu, H. Mao, D. Y. Zhang, T. Q. Lu, Q. Chen, D. M. Yu and S. J. Ding, Nat. Commun., 2022, 13, 4868 CrossRef CAS PubMed.
- J. Y. Tang, D. H. Wang, W. Qin, S. Y. Fang, T. Xu, J. M. Huang, M. Tang and Z. B. Wang, Chem. Eng. J., 2025, 513, 163082 CrossRef CAS.
- R. Sahore, B. L. Armstrong, X. M. Tang, C. H. Liu, K. Owensby, S. Kalnaus and X. C. Chen, Adv. Energy Mater., 2023, 13, 2203663 CrossRef CAS.
- Z. Wang, R. Wang, M. Luo, X. Cao, J. Wang, X. Tian and L. Li, Small, 2025, 21, 2502696 CrossRef CAS PubMed.
- Y. X. Cui, F. Xu, D. Bao, Y. Y. Gao, J. W. Peng, D. Lin, H. L. Geng, X. S. Shen, Y. J. Zhu and H. Y. Wang, J. Mater. Sci. Technol., 2023, 147, 165–175 CrossRef CAS.
- X. S. Yin, L. Wang, Y. Kim, N. Ding, J. H. Kong, D. Safanama, Y. Zheng, J. W. Xu, D. V. M. Repaka, K. Hippalgaonkar, S. W. Lee, S. Adams and G. W. Zheng, Adv. Sci., 2020, 7, 2001303 CrossRef CAS.
- A. Pakdel, Y. Bando and D. Golberg, Chem. Soc. Rev., 2014, 43, 934–959 RSC.
- H. Zhu, Y. Li, Z. Fang, J. Xu, F. Cao, J. Wan, C. Preston, B. Yang and L. Hu, ACS Nano, 2014, 8, 3606–3613 CrossRef CAS PubMed.
- W. Luo, L. Zhou, K. Fu, Z. Yang, J. Wan, M. Manno, Y. Yao, H. Zhu, B. Yang and L. Hu, Nano Lett., 2015, 15, 6149–6154 CrossRef CAS PubMed.
- Y. Liu, W. Zou, M. Yang, H. Luo, S. Yang, J. Xu and N. Zhao, Adv. Funct. Mater., 2023, 33, 2303561 CrossRef CAS.
- H. Zheng, K. Wu, Y. Zhan, K. Wang and J. Shi, J. Polym. Sci., 2023, 61, 1622–1633 CrossRef CAS.
- A. Q. Chen, Y. Y. Wu, S. X. Zhou, W. X. Xu, W. L. Jiang, Y. Lv, W. Guo, K. Y. Chi, Q. Sun, T. T. Fu, T. T. Xie, Y. Zhu and X. G. Liang, Mater. Adv., 2020, 1, 1996–2002 RSC.
- Y. J. Wang, S. Xia, H. Li and J. F. Wang, Adv. Funct. Mater., 2019, 29, 1903876 Search PubMed.
- Y. F. Xu, D. Kraemer, B. Song, Z. Jiang, J. W. Zhou, J. Loomis, J. J. Wang, M. D. Li, H. Ghasemi, X. P. Huang, X. B. Li and G. Chen, Nat. Commun., 2019, 10, 1771 CrossRef.
- X. Li, Y. Wang, K. Xi, W. Yu, J. Feng, G. Gao, H. Wu, Q. Jiang, A. Abdelkader, W. Hua, G. Zhong and S. Ding, Nano-Micro Lett., 2022, 14, 210 CrossRef CAS.
- X. Y. Li, J. Feng, Y. A. Li, N. Li, X. Jia, Y. S. Wang and S. J. Ding, Energy Storage Mater., 2024, 72, 103759 CrossRef.
- S. X. Xia, X. S. Wu, Z. C. Zhang, Y. Cui and W. Liu, Chem, 2019, 5, 753–785 CAS.
- W. Liu, S. W. Lee, D. C. Lin, F. F. Shi, S. Wang, A. D. Sendek and Y. Cui, Nat. Energy, 2017, 2, 17035 CrossRef CAS.
- W. Liu, N. Liu, J. Sun, P.-C. Hsu, Y. Li, H.-W. Lee and Y. Cui, Nano Lett., 2015, 15, 2740–2745 CrossRef CAS.
- D. Lin, W. Liu, Y. Liu, H. R. Lee, P.-C. Hsu, K. Liu and Y. Cui, Nano Lett., 2016, 16, 459–465 CrossRef CAS.
- M. Hong, Q. Dong, H. Xie, B. C. Clifford, J. Qian, X. Z. Wang, J. Luo and L. B. Hu, ACS Energy Lett., 2021, 6, 3753–3760 CrossRef CAS.
- X. Zhang, J. Xie, F. Shi, D. Lin, Y. Liu, W. Liu, A. Pei, Y. Gong, H. Wang, K. Liu, Y. Xiang and Y. Cui, Nano Lett., 2018, 18, 3829–3838 CrossRef CAS.
- V. Vishwakarma and A. Jain, J. Power Sources, 2017, 362, 219–227 CrossRef CAS.
- P. Y. Chen, S. Jin, S. F. Hong, Y. F. Qiu, Z. Y. Zhang, Y. Z. Xu, Y. L. Joo, L. A. Archer and R. Yang, J. Am. Chem. Soc., 2024, 146, 3136–3146 CrossRef CAS.
- M. Wang, H. Zhang, Y. W. Li, R. P. Liu and H. Yang, Chem. Eng. J., 2023, 476, 146658 CrossRef CAS.
- Y. L. Guo, Z. A. Li and X. Wang, Adv. Mater., 2025, 37, 2418354 CrossRef CAS.
- Y. Wang, C. J. Zanelotti, X. E. Wang, R. Kerr, L. Y. Jin, W. H. Kan, T. J. Dingemans, M. Forsyth and L. A. Madsen, Nat. Mater., 2021, 20, 1255–1263 CrossRef CAS.
- K. Ruan, Y. Guo and J. Gu, Macromolecules, 2021, 54, 4934–4944 CrossRef CAS.
- J. M. Zheng, J. Xiao, M. Gu, P. J. Zuo, C. M. Wang and J. G. Zhang, J. Power Sources, 2014, 250, 313–318 CrossRef CAS.
- J. Cho, M. D. Losego, H. G. Zhang, H. Kim, J. Zuo, I. Petrov, D. G. Cahill and P. V. Braun, Nat. Commun., 2014, 5, 4035 CrossRef CAS.
- V. Vishwakarma, C. Waghela, Z. Wei, R. Prasher, S. C. Nagpure, J. Li, F. Liu, C. Daniel and A. Jain, J. Power Sources, 2015, 300, 123–131 CrossRef CAS.
- J. L. He, W. K. Xian, L. Tao, P. Corrigan and Y. Li, ACS Appl. Mater. Interfaces, 2022, 14, 56268–56279 CrossRef CAS PubMed.
- M. A. Azam, N. E. Safie, A. S. Ahmad, N. A. Yuza and N. S. A. Zulkifli, J. Energy Storage, 2021, 33, 102096 Search PubMed.
- S. J. Deng, Y. Zhang, D. Xie, L. Yang, G. Z. Wang, X. S. Zheng, J. F. Zhu, X. L. Wang, Y. Yu, G. X. Pan, X. H. Xia and J. P. Tu, Nano Energy, 2019, 58, 355–364 CrossRef CAS.
- W. T. Zheng, X. Yu, Z. Q. Guo, G. H. Song and F. Hu, Mater. Res. Express, 2019, 6, 056410 CrossRef CAS.
- K. Liu, A. Pei, H. R. Lee, B. Kong, N. Liu, D. C. Lin, Y. Y. Liu, C. Liu, P. C. Hsu, Z. A. Bao and Y. Cui, J. Am. Chem. Soc., 2017, 139, 4815–4820 Search PubMed.
- G. Y. Zheng, S. W. Lee, Z. Liang, H. W. Lee, K. Yan, H. B. Yao, H. T. Wang, W. Y. Li, S. Chu and Y. Cui, Nat. Nanotechnol., 2014, 9, 618–623 CrossRef CAS PubMed.
- M. H. Li, S. B. Yang and B. Li, Interdiscip. Mater., 2024, 3, 805–834 Search PubMed.
- C. L. Jiang, H. T. Lu, J. L. Yang, Z. J. Sun, Y. K. Xiao, Y. X. Niu, H. F. Xu, Y. Liu, M. Wang, H. T. Yang, B. H. Cui, Y. Long, G. W. Chen, Y. Shan, Q. H. Yang and W. Chen, ACS Energy Lett., 2024, 9, 2527–2535 CrossRef CAS.
- C. Zhang, X. C. Hu, Z. W. Nie, C. Wu, N. Zheng, S. J. Chen, Y. H. Yang, R. Wei, J. M. Yu, N. Yang, Y. Yu and W. Liu, J. Adv. Ceram., 2022, 11, 1530–1541 CrossRef CAS.
- M. Steinhardt, E. I. Gillich, M. Stiegler and A. Jossen, J. Energy Storage, 2020, 32, 101680 Search PubMed.
- S. La, Y. Gao, Q. H. Cao, J. Z. Chen, A. M. Elshahawy, Y. Y. Cui, F. Bu, S. A. Makhlouf, P. S. Chee and C. Guan, Matter, 2025, 8, 102013 CrossRef CAS.
- A. R. Siddique, S. Mahmud and B. Van Heyst, J. Power Sources, 2018, 401, 224–237 Search PubMed.
- K. Zhan, Y. C. Chen, Z. Y. Xiong, Y. L. Zhang, S. Y. Ding, F. Z. Zhen, Z. S. Liu, Q. Wei, M. S. Liu, B. Sun, H. M. Cheng and L. Qiu, Nat. Commun., 2024, 15, 2905 CrossRef CAS PubMed.
- S. Ki, S. Shin, S. M. Cho, S. Bang, D. Choi and Y. Nam, Adv. Sci., 2024, 11, 2310185 CrossRef CAS PubMed.
- J. Mei, G. Q. Shi, H. Liu, Z. Wang and M. Y. Chen, J. Energy Storage, 2022, 55, 105365 CrossRef.
- W. T. Zhang, X. C. Jiang, Z. Cheng, W. X. Hu and E. S. Long, Energy Convers. Manage., 2025, 344, 120301 CrossRef.
- Y. Lin, Q. Kang, H. Wei, H. Bao, P. K. Jiang, Y. W. Mai and X. Y. Huang, Nano-Micro Lett., 2021, 13, 180 CrossRef CAS.
- Y. Cai, H. T. Yu, C. Chen, Y. Y. Feng, M. M. Qin and W. Feng, Carbon, 2022, 196, 902–912 CrossRef CAS.
- Y. Feng, Y. Sato, T. Inoue, R. Xiang, K. Suenaga and S. Maruyama, Small, 2024, 20, 2308571 CrossRef CAS PubMed.
- J. W. Xie, G. Zhou, Y. X. Sun, F. Zhang, F. Y. Kang, B. H. Li, Y. Zhao, Y. H. Zhang, W. Feng and Q. B. Zheng, Small, 2024, 20, 2305163 CrossRef CAS.
- J. Q. Ning, Z. Wang, Y. Zhang, X. Y. Xu, T. Ma, J. X. Du, H. D. Wang, J. W. Peng, D. Bao, Y. J. Zhu and H. Y. Wang, Chem. Eng. J., 2024, 502, 158110 CrossRef CAS.
- K. C. Liang, H. Zhang, Q. W. Wang and Z. L. Cheng, Carbon, 2024, 230, 119639 CrossRef CAS.
- M. Q. Wu, T. X. Li, P. F. Wang, S. Wu, R. Z. Wang and J. Lin, Small, 2022, 18, 2105647 CrossRef CAS PubMed.
- P. D. Tao, S. G. Wang, L. Chen, J. F. Ying, L. Lv, L. W. Sun, W. B. Chu, K. Nishimura, L. Fu, Y. Z. Wang, J. H. Yu, N. Jiang, W. Dai, Y. K. Lv, C. T. Lin and Q. W. Yan, Rare Met., 2023, 42, 3662–3672 CrossRef CAS.
- O. Hur, E. J. Markvicka and M. D. Bartlett, Adv. Funct. Mater., 2025, 35, 2417375 CrossRef CAS.
- L. Li, B. Fang, D. Ren, L. Fu, Y. Zhou, C. Yang, F. Zhang, X. Feng, L. Wang, X. He, P. Qi, Y. Liu, C. Jia, S. Zhao, F. Xu, X. Wei and H. Wu, ACS Nano, 2022, 16, 10729–10741 CrossRef CAS PubMed.
- J. Wang, X. Feng, Y. Yu, H. Huang, M. Zheng, Y. Xu, J. Wu, Y. Yang and J. Lu, Nat. Energy, 2024, 9, 939–946 CrossRef.
- X. L. Li, T. H. Lu, W. Z. Liu, W. H. Lv and S. Lv, Cell Rep. Phys. Sci., 2025, 6, 102617 CrossRef CAS.
- X. G. Wang, C. Y. Zhang, K. Wang, Y. Q. Huang and Z. F. Chen, J. Colloid Interface Sci., 2021, 582, 30–40 CrossRef CAS PubMed.
- E. Mohseni, W. C. Tang, K. H. Khayat and H. Z. Cui, Constr. Build. Mater., 2020, 249, 118768 CrossRef CAS.
- R. A. Mitran, D. Lincu, S. Ionita, M. Deaconu, V. V. Jerca, O. C. Mocioiu, D. Berger and C. Matei, Sol. Energy Mater. Sol. Cells, 2020, 218, 110760 CrossRef CAS.
- X. M. Yang, C. B. Li, Y. F. Ma, H. Chi, Z. J. Hu and J. F. Xie, Chem. Eng. J., 2023, 473, 145364 CrossRef CAS.
- E. Hamidi, P. B. Ganesan, R. K. Sharma and K. Yong, Renewable Sustainable Energy Rev., 2023, 176, 113196 CrossRef CAS.
- M. Clausi, M. Zahid, A. Shayganpour and I. S. Bayer, Adv. Compos. Hybrid Mater., 2022, 5, 798–812 CrossRef CAS.
- Y. Y. Chen, X. J. Guo, C. W. Shi, X. Zhou and D. Q. Zou, Compos. Pt. B-Eng., 2025, 298, 112376 CrossRef CAS.
- M. Das and U. Stachewicz, J. Energy Storage, 2024, 98, 113029 CrossRef CAS.
- X. F. Kong, R. M. Nie and J. J. Yuan, Chem. Eng. J., 2023, 462, 142168 CrossRef CAS.
- D. L. Feng, Z. H. Zhao, P. Li, Y. P. Li, J. Zha, J. K. Hu, Y. Y. Zhang and Y. H. Feng, Mater. Today, 2024, 75, 285–308 CrossRef CAS.
- G. W. Jiang, J. H. Huang, Y. S. Fu, M. Cao and M. C. Liu, Appl. Therm. Eng., 2016, 108, 1119–1125 CrossRef CAS.
- C. M. Wu, L. J. Zeng, G. J. Chang, Y. Zhou, K. Yan, L. Xie, B. Xue and Q. Zheng, Adv. Compos. Hybrid Mater., 2023, 6, 31 CrossRef CAS.
- A. Babapoor, M. Azizi and G. Karimi, Appl. Therm. Eng., 2015, 82, 281–290 CrossRef CAS.
- F. Samimi, A. Babapoor, M. Azizi and G. Karimi, Energy, 2016, 96, 355–371 CrossRef CAS.
- Q. R. Sun, N. Zhang, H. Q. Zhang, X. P. Yu, Y. L. Ding and Y. P. Yuan, Renewable Energy, 2020, 145, 2629–2636 CrossRef CAS.
- X. Zhu, L. Han, Y. F. Lu, F. Wei and X. L. Jia, Appl. Energy, 2019, 254, 113688 CrossRef CAS.
- J. Y. Lin, D. W. Liu, X. H. Liu, M. Y. Liu and Y. Y. Cui, Appl. Therm. Eng., 2025, 262, 125240 CrossRef CAS.
- B. Y. Hu, H. Guo, J. Y. Li, T. Li, M. Cao, W. Y. Qi, Z. Q. Wu, Y. Li and B. A. Li, Compos. Pt. B-Eng., 2023, 266, 110998 CrossRef CAS.
- M. Q. Wu, Y. M. Xuan, X. L. Liu, Y. G. Jing and T. X. Li, Adv. Funct. Mater., 2025, 35, 2506229 CrossRef CAS.
- Z. Y. Zeng, B. C. Zhao and R. Z. Wang, Renewable Sustainable Energy Rev., 2023, 182, 113373 CrossRef CAS.
- C. C. Li, B. Zhang and Q. X. Liu, J. Energy Storage, 2020, 29, 101339 CrossRef.
- B. Li, Z. Y. Mao, B. W. Song, X. L. Wang, W. L. Tian, Q. X. Sun, Y. F. Wang and Z. G. Jin, Appl. Therm. Eng., 2024, 242, 122477 CrossRef CAS.
- H. Badenhorst, Sol. Energy, 2019, 192, 35–68 CrossRef CAS.
- M. M. Umair, Y. Zhang, K. Iqbal, S. F. Zhang and B. T. Tang, Appl. Energy, 2019, 235, 846–873 CrossRef CAS.
- A. Allahbakhsh and M. Arjmand, Carbon, 2019, 148, 441–480 CrossRef CAS.
- W. Lee and J. Kim, J. Energy Storage, 2022, 52, 104838 CrossRef.
- W. S. Yang, Y. X. Du, Q. Deng, S. Y. Li, C. B. Li, J. X. Tian, P. H. Chen, T. Q. Fu, Y. J. Luo, Y. R. Zhang, S. W. Zhu, X. Z. Liu, Z. H. Rao and X. X. Li, Appl. Therm. Eng., 2024, 249, 123358 CrossRef CAS.
- J. Y. Hu, W. F. Huang, X. Ge, C. X. Wang, G. Q. Zhang, Y. P. Chen and C. Q. Tu, J. Energy Storage, 2024, 100, 113485 CrossRef CAS.
- Q. L. Ren, Z. X. Wang, J. J. Zhu and Z. G. Qu, Int. J. Therm. Sci., 2021, 161, 106739 CrossRef.
- L. C. Wei and J. A. Malen, Int. J. Heat Mass Transfer, 2020, 150, 119153 CrossRef.
- X. K. Yu, Y. B. Tao, Y. He and Z. C. Lv, J. Energy Storage, 2022, 46, 103930 CrossRef.
- H. J. Cheong, J. H. Yang, J. W. Hur, H. K. Gi and J. H. Shin, Appl. Therm. Eng., 2025, 270, 126210 CrossRef CAS.
- H. F. Wang, F. X. Wang, Z. T. Li, Y. Tang, B. H. Yu and W. Yuan, Appl. Energy, 2016, 176, 221–232 CrossRef CAS.
- M. Aramesh and B. Shabani, J. Energy Storage, 2022, 50, 104650 CrossRef.
- T. R. Hou, Y. M. Xing, W. Y. Zheng and Z. L. Hao, Appl. Therm. Eng., 2022, 204, 117934 CrossRef CAS.
- J. X. Wang, J. Qian, N. Wang, H. Zhang, X. Cao, F. F. Liu and G. Q. Hao, Renewable Energy, 2023, 213, 75–85 CrossRef CAS.
- W. Lee and J. Kim, Ceram. Int., 2023, 49, 18363–18370 CrossRef CAS.
- Y. Lu, R. P. Hu, X. P. Chen and Y. X. Bai, J. Mater. Sci. Technol., 2023, 160, 248–257 CrossRef CAS.
- W. Lee, J. Hong, J. Song, W. Yang and J. Kim, Compos. Sci. Technol., 2023, 240, 110092 CrossRef CAS.
- Z. J. Huang, R. Q. Wang, W. J. Jiang, Y. L. Liu, T. Y. Zhu, D. X. Sun, J. H. Yang, X. D. Qi and Y. Wang, Compos. Sci. Technol., 2024, 255, 110736 CrossRef CAS.
- S. Dey, P. S. Kirandas, D. J. Nagar and J. Mitra, ACS Appl. Energy Mater., 2025, 8, 2328–2334 CrossRef.
- H. Y. Zu, W. Dai, Y. Li, K. Li and J. T. Li, Int. J. Therm. Sci., 2021, 166, 106971 CrossRef CAS.
- Y. J. Gao, J. L. Chen, G. R. Chen, C. H. Fan and X. G. Liu, Small Methods, 2021, 5, 2100771 CrossRef CAS PubMed.
- Y. Yang, Y. H. Wei, Z. F. Guo, W. W. Hou, Y. J. Liu, H. Tian and T. L. Ren, Small Methods, 2022, 6, 2200671 Search PubMed.
- H. Zong, S. Jiang, Y. M. Sun, Y. Q. Han, S. F. Zhang and S. L. Wu, Chem. Eng. J., 2025, 523, 168894 CrossRef CAS.
- Y. Cho, H. H. Kim, S. Ahn, J. Lee, J. Lee, K. Kim, S. Kwak, H. Lee, K. Choi, K. Song and S. Lee, Adv. Funct. Mater., 2025, e10833 CrossRef CAS.
- D. Ding, X. He, S. Liang, W. Wei and S. Ding, ACS Appl. Mater. Interfaces, 2022, 14, 24690–24696 CrossRef CAS PubMed.
- L. L. Du, R. H. Li, M. W. Tan, S. Mushina, Z. G. Zhou, B. Hu, C. L. Wang, J. H. Zheng, W. Liu and W. X. Chen, Chem. Eng. J., 2025, 510, 161730 CrossRef CAS.
- J.-X. Wang, Y. Mao and N. Miljkovic, Adv. Sci., 2024, 11, 2402190 CrossRef CAS PubMed.
- K. Araki and R. Z. Zhang, Int. J. Heat Mass Transfer, 2022, 191, 122835 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |