Open Access Article
Open Access ArticleCrystallographic visualization of C3-hydrocarbon-induced structural transformation and guest encapsulation within a flexible coordination network
Shao-Jie
Qin†
a,
Mohana
Shivanna†
b,
Mei-Yan
Gao
b,
Cheng-Hua
Deng
 b,
Shi-Qiang
Wang
b,
Shi-Qiang
Wang
 b,
Dan
Li
a,
Shao-Dan
Fu
a,
Shuai
Qiu
a,
Bai-Qiao
Song
b,
Dan
Li
a,
Shao-Dan
Fu
a,
Shuai
Qiu
a,
Bai-Qiao
Song
 *a and
Qing-Yuan
Yang
*a and
Qing-Yuan
Yang
 *c
*c
aCollege of Materials and Chemistry & Chemical Engineering, Chengdu University of Technology, Chengdu 610059, China. E-mail: bqsong@cdut.edu.cn
bDepartment of Chemical Sciences and Bernal Institute, University of Limerick, Limerick V94 T9PX, Republic of Ireland
cSchool of Chemical Engineering and Technology, Xi'an Jiaotong University, Xi'an 710049, China. E-mail: qingyuan.yang@xjtu.edu.cn
First published on 22nd January 2026
Abstract
Flexible coordination polymers that can self-adjust, via closed (nonporous) to open (porous) switching, to match the size/shape of guest molecules are of particular interest. Herein, we report that C3 hydrocarbons can trigger a flexible SIFSIX network, [Cu(SiF6)(L)2]n (L = 1,4-bis(1-imidazolyl)benzene), SIFSIX-23-Cu, to switch from nonporous to partially open phases at 298 K with different gate opening pressures (propyne < propylene < propane). The single-crystal X-ray diffraction (SCXRD) measurements have succeeded in the crystallographic visualization of the C3-induced closed-to-open switching and the binding sites of gas molecules confined in the open phases. The results unambiguously showed that the closed phase can undergo adaptive structural expansion upon sorption of different C3 molecules, and the conformational changes of Lvia rotation and contortion under gas loading are responsible for the structural transformation. The host–gas interactions are dominated by C–H⋯F hydrogen bonds between gas molecules and SiF62− anions and the size-matched chelation of linear propyne by SiF62− pairs results in the stronger binding strength of propyne over propylene (propane), leading to the selective sorption of propyne from binary and ternary C3 gas mixtures, as confirmed by breakthrough experiments. This work highlights the vital role of host–gas interactions in governing the structural transitions and affords an efficient strategy to design high-performance flexible sorbents for challenging gas recognition and sorption.
Introduction
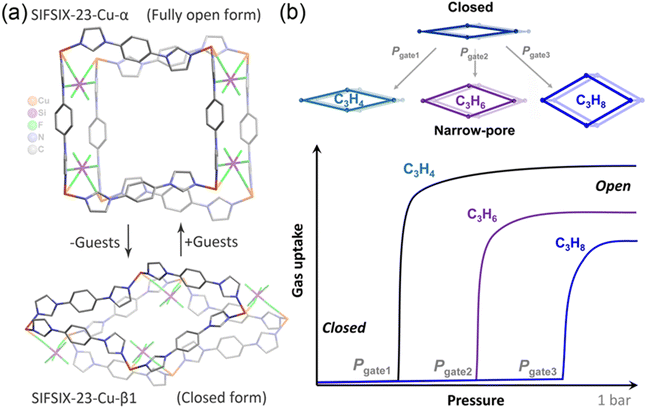
In nature, enzymes can adapt the shape and size of their binding sites to accommodate specific substrates, inspiring the development of stimuli-responsive materials (SRMs) that cover a broad scope of compositions and functional properties.1 SRMs in traditional porous solids remain scarce, presumably because most, such as activated carbons and zeolites, are rigid under ordinary conditions.2 As a new generation of porous metal–organic materials (MOMs),3,4 porous coordination polymers (PCPs)5 or metal–organic frameworks (MOFs)6 offer a high degree of diversity, modularity and tunability with respect to their architectures and functionalities.7 An emerging class of porous SRMs is exemplified by “soft”8 or flexible MOMs (FMOMs)9,10 that transform their structures, sometimes dramatically, when exposed to gases, liquids and/or vapors.11–13 These transformations usually result from coordination geometry isomerization,14–17 linker rotation/contortion,18–22 subnet displacement (in catenated networks),15 or other events.23–26 Unlike rigid MOMs usually showing characteristic type I isotherms,27 FMOMs often exhibit stepwise adsorption isotherms with visible inflections mostly associated with structural transformation.28–36 Particularly, FMOMs switching between nonporous (closed) and highly porous (open) phases under external stimuli have attracted considerable attention,14–16,37 as they show negligible gas adsorption before the gate-opening pressure but an abrupt increase in uptake once the gate is opened, i.e., a stepped type F-IV isotherm.20 Such isotherms can substantially improve the working capacity for gas storage,14,20 and are also favorable for gas separation if gate opening is differentiated by adsorbate types.35,38–40However, sorption isotherms of FMOMs offer limited information about gas–solid interactions.41,42 Insights into the structural changes associated with gas-triggered gate opening behavior and the binding sites of gas molecules trapped in the channels are essential for elucidation of the gate-opening mechanism and the design of high-performance switching sorbents for specific gases.13,43 Although different technologies including specialized IR and NMR spectroscopy,44in situ powder X-ray diffraction (PXRD)45 and molecular modelling46 have been developed, SCXRD is the most convincing approach,47–50 as it can often provide unequivocal dynamic structural information relating to gate-opening flexibility and host–guest interactions at the molecular level.18,51 However, to date, the relevant examples are limited in the literature mainly because of the added requirements of maintaining crystal singularity of FMOMs after activation and undergoing drastic structural changes and/or lack of suitable host–guest interactions to strongly bind the gas molecules onto the pore surface.52
Recently we reported a 3D switching SIFSIX network, [Cu(SiF6)(L)2]n (L = 1,4-bis(1-imidazolyl)benzene), SIFSIX-23-Cu,37 which maintained its single-crystal nature even after activation and showed reversible closed-to-open switching induced by CO2, N2 and some solvents including H2O (Fig. 1a). As part of our continuing efforts to understand host–guest interactions in flexible coordination networks, we now provide a detailed investigation of the structural changes induced by different C3 hydrocarbons in SIFSIX-23-Cu (Fig. 1b). We demonstrate C3 hydrocarbons (propyne: C3H4; propylene: C3H6; propane: C3H8) can induce the nonporous form of SIFSIX-23-Cu to switch to partially open phases, with distinctly different gate-opening pressures (C3H4 < C3H6 < C3H8), and adaptive structural expansions that are clearly visualized by single-crystal X-ray diffraction (SCXRD). SCXRD characterization also validated the origin of framework changes, and the binding sites of C3 molecules confined in the channels which confirmed stronger binding affinity of C3H4 over C3H6 (C3H8), aligning with the results of breakthrough experiments.
Results and discussion
Synthesis and crystal structures
The crystals of SIFSIX-23-Cu were obtained using our previous procedures.37 The thermogravimetric analysis (TGA) is similar to the previous result (Fig. S1) and the PXRD pattern is consistent with the calculated one (Fig. S2), validating the successful synthesis and the purity of the sample. Its structure is an analog of the reported dipyridyl-ligand-based rigid SIFSIX net with an underlying pcu topology,53 but possessing undulating square CuL2 grid layers (sql) pillared by SIFSIX anions in a cis-coordination mode.54 The anti- and syn-conformers of biimidazolyl ligand i.e., L(anti) and L(syn), coexist in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio in the sql net, providing the prerequisite for structural flexibility. The network could experience several phase contractions along with the release of guests in the channels due to conformational changes of the biimidazolyl ligand via rotation and contortion, leading to a final closed phase with negligible porosity. This nonporous phase can reversibly return to the original porous phase via the capture of different guests, including some organic solvents, H2O, CO2 and N2.
1 ratio in the sql net, providing the prerequisite for structural flexibility. The network could experience several phase contractions along with the release of guests in the channels due to conformational changes of the biimidazolyl ligand via rotation and contortion, leading to a final closed phase with negligible porosity. This nonporous phase can reversibly return to the original porous phase via the capture of different guests, including some organic solvents, H2O, CO2 and N2.
Single-component adsorption isotherms
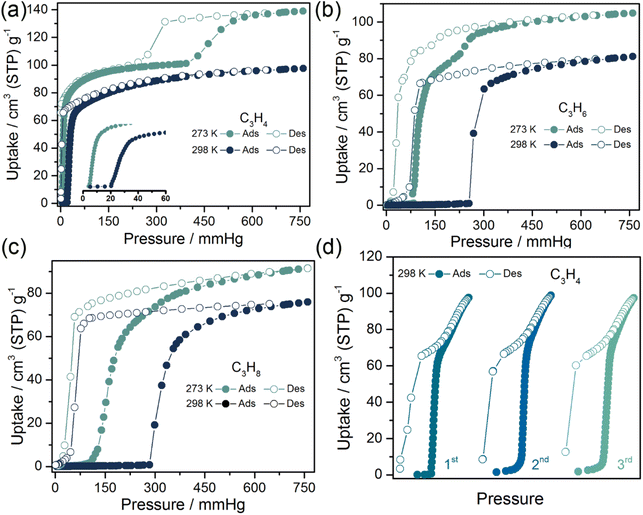
The low-pressure sorption isotherms of C3 hydrocarbons were measured at different temperatures on the anhydrous phase, SIFSIX-23-Cu-β1. As expected, the adsorption of C3 by the nonporous β1 phase exhibited desirable stepped type F-IV isotherms with distinctly different gate-opening/closing pressures, indicative of gas-induced closed-to-open switching (Fig. 2). The pre-step uptakes for all the gases are negligible which is consistent with the nonporous feature of the β1 phase. For C3H4, the sudden gate opening with a threshold pressure of 20 mmHg at 298 K leads to a steep increase in uptake up to 97 cm3 g−1 at 1 bar (Fig. 2a). The saturated adsorption capacity of C3H4 is higher than that of UTSA-200 (81.1 cm3 g−1),55 ZNU-62 (82.0 cm3 g−1),56 NKMOF-1-Ni (78.4 cm3 g−1)57 and TIFSIX-14-Cu-i (86.9 cm3 g−1)58 under similar conditions. Notably, the uptake of C3H4 at 0.1 bar (72.0 cm3 g−1) on SIFSIX-23-Cu-β1 is even higher than the uptake of SIFSIX-3-Ni (66.8 cm3 g−1),59 SIFSIX-3-Zn (50.6 cm3 g−1),59 ELM-12 (61.4 cm3 g−1),60 HOF-30a (59.8 cm3 g−1)61 and NbOFFIVE-1-Ni (42.3 cm3 g−1)59 at 1 bar, suggestive of the strong affinity for C3H4. At 273 K, the gate opening pressure was also <0.1 mmHg, but a second step was observed and the saturated uptake at 1 bar (139 cm3 g−1) was higher than that at 298 K. The second step suggests a further structural change. C3H6 and C3H8 induced gate opening of β1 with threshold pressures of 254.8 and 283.2 mmHg, and uptakes of 81.1 and 75.8 cm3 g−1 at 298 K, respectively (Fig. 2b and c). The C3H6 and C3H8 sorption isotherms at 273 K were found to exhibit lower gate-opening pressures of 84.2 and 94.1 mmHg, and increased capacities of 104.9 and 89.3 cm3 g−1, respectively. Similar to C3H4, a second step was evident in the C3H6 sorption isotherm at low temperature but not in that of C3H8. The pure gas sorption experiments revealed that the gate-opening pressures for C3 hydrocarbons increase in the following order: C3H4 < C3H6 < C3H8. Overall, the pure gas isotherms indicate that there are several open phases with various surface areas. Three cyclic C3H4 (298 K) sorption tests demonstrated the reversibility of the closed-to-open structural transition (Fig. 2d).SCXRD studies of C3-induced structural transformations
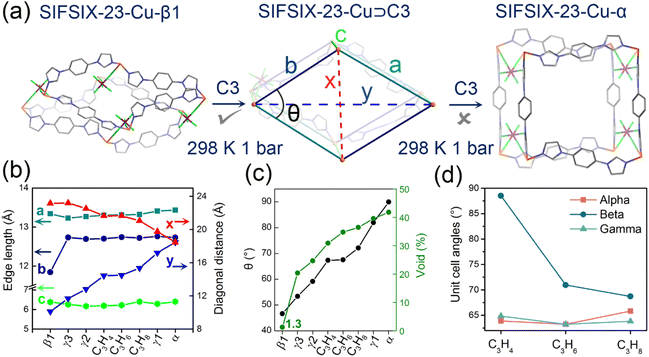
To structurally elucidate the mechanism of gas-induced nonporous-to-porous switching with different gate-opening pressures and adsorption abilities in SIFSIX-23-Cu, we determined the crystal structures of SIFSIX-23-Cu loaded with C3 gas molecules, i.e., SIFSIX-23-Cu⊃C3H4, SIFSIX-23-Cu⊃C3H6 and SIFSIX-23-Cu⊃C3H8.Single-crystal structure analysis directly revealed that the trapping of different hydrocarbons drove the gate opening of closed β1 to generate the related partially open phases, not the fully open phase α (Fig. 3a, S3 and S4). All the C3 molecule-loaded phases were crystallized in the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group (Tables S1 and S2). The observed closed-to-open transitions were fulfilled via hinge-like motions without changing the connectivity of the framework (Table S3). Although changes in Cu(II) geometry and SIFSIX configuration are small, the ∠Cu–Cu–Cu acute angle of the CuL2 parallelogram unit of the sql net significantly increased (67.4–72.2° for C3H4–C3H8vs. 46.7° in β1) (Fig. 2c), accompanied by a slight shortening of the long diagonal (21.6–21.1 Å vs. 23.2 Å) and a striking lengthening of the short diagonal (14.4–15.4 Å vs. 10.1 Å in β1) (Fig. 2b). Meanwhile, the Cu–L(anti)–Cu edge of the parallelogram shows almost no change but on the contrary the Cu–L(syn)–Cu counterpart stretches (12.7 Å vs. 11.8 Å). The latter is due to a remarkable reduction in bending distortion of L(syn) (55.2° in β1 vs. <4.1° in gas-included phases) and the Cu–imidazolyl coordination junction (35.2° in β1 vs. <9.3°) (Fig. S5–S8). Notably, the free rotation of L including the swing of the imidazolyl ring and reorientation of the phenyl group was observed from β1 to the gas-included phase (Fig. S9–S12), which drives the structure deformation (a phenomenon particularly evident in L(syn) due to its ‘angular’ characteristic), accommodates the constraints and minimizes the lattice energy. In fact, previous theoretical calculations have proven that L possesses an intrinsically low rotation energy barrier (<13 kJ mol−1) which is beneficial for the nonporous-to-porous switching observed here. The C–H⋯F hydrogen bonding between L and SIFSIX anions further stabilizes the structural changes and helps to maintain the single-crystal nature of gas-included phases (Table S4). The expansion of the framework results in an overall increase in the guest-accessible volume (1.3% (β1) vs. 30.9–36.5% (C3H4–C3H8)) as calculated by PLATON (Fig. 2c and S13).62 The pore volumes of the open phase incorporating different guests at 298 K increase in the following sequence C3H4 < C3H6 < C3H8, which substantiates that guest-dependent closed-to-open switching can adjust the open phases to adapt to guests of different molecular sizes and geometries. Notably, although the ∠Cu–Cu–Cu acute angle and the edges of the CuL2 parallelogram unit remain almost constant after loading C3H4 and C3H6, the differences in void space and unit cell parameters clearly prove the self-adaptive feature of SIFSIX-23-Cu-β1 to them (Fig. 3d and Table S2). It is noteworthy that SIFSIX-23-Cu⊃C3H4 presents the highest C3H4 uptake but the smallest void space (after removal of C3H4) among C3 induced open phases, confirming the tight binding of C3H4 in the structure. Additionally, the C3 gas loaded phases are different from those previously discovered narrow pore forms (γ1–γ3), which contain H2O and methanol as guests (Fig. S4 and Table S2), further confirming the guest-dependent structural expansion ability of β1. To our knowledge, direct SCXRD characterization of hydrocarbon-triggered structural switching from nonporous to porous phase is very rare.52 The PXRD patterns of the nonporous phase (SIFSIX-23-Cu-β1) under 1 bar of each C3 gas collected at 298 K match well with those calculated from the SCXRD-determined crystal structures, confirming the accuracy of the SCXRD measurement procedures (Fig. S14).
space group (Tables S1 and S2). The observed closed-to-open transitions were fulfilled via hinge-like motions without changing the connectivity of the framework (Table S3). Although changes in Cu(II) geometry and SIFSIX configuration are small, the ∠Cu–Cu–Cu acute angle of the CuL2 parallelogram unit of the sql net significantly increased (67.4–72.2° for C3H4–C3H8vs. 46.7° in β1) (Fig. 2c), accompanied by a slight shortening of the long diagonal (21.6–21.1 Å vs. 23.2 Å) and a striking lengthening of the short diagonal (14.4–15.4 Å vs. 10.1 Å in β1) (Fig. 2b). Meanwhile, the Cu–L(anti)–Cu edge of the parallelogram shows almost no change but on the contrary the Cu–L(syn)–Cu counterpart stretches (12.7 Å vs. 11.8 Å). The latter is due to a remarkable reduction in bending distortion of L(syn) (55.2° in β1 vs. <4.1° in gas-included phases) and the Cu–imidazolyl coordination junction (35.2° in β1 vs. <9.3°) (Fig. S5–S8). Notably, the free rotation of L including the swing of the imidazolyl ring and reorientation of the phenyl group was observed from β1 to the gas-included phase (Fig. S9–S12), which drives the structure deformation (a phenomenon particularly evident in L(syn) due to its ‘angular’ characteristic), accommodates the constraints and minimizes the lattice energy. In fact, previous theoretical calculations have proven that L possesses an intrinsically low rotation energy barrier (<13 kJ mol−1) which is beneficial for the nonporous-to-porous switching observed here. The C–H⋯F hydrogen bonding between L and SIFSIX anions further stabilizes the structural changes and helps to maintain the single-crystal nature of gas-included phases (Table S4). The expansion of the framework results in an overall increase in the guest-accessible volume (1.3% (β1) vs. 30.9–36.5% (C3H4–C3H8)) as calculated by PLATON (Fig. 2c and S13).62 The pore volumes of the open phase incorporating different guests at 298 K increase in the following sequence C3H4 < C3H6 < C3H8, which substantiates that guest-dependent closed-to-open switching can adjust the open phases to adapt to guests of different molecular sizes and geometries. Notably, although the ∠Cu–Cu–Cu acute angle and the edges of the CuL2 parallelogram unit remain almost constant after loading C3H4 and C3H6, the differences in void space and unit cell parameters clearly prove the self-adaptive feature of SIFSIX-23-Cu-β1 to them (Fig. 3d and Table S2). It is noteworthy that SIFSIX-23-Cu⊃C3H4 presents the highest C3H4 uptake but the smallest void space (after removal of C3H4) among C3 induced open phases, confirming the tight binding of C3H4 in the structure. Additionally, the C3 gas loaded phases are different from those previously discovered narrow pore forms (γ1–γ3), which contain H2O and methanol as guests (Fig. S4 and Table S2), further confirming the guest-dependent structural expansion ability of β1. To our knowledge, direct SCXRD characterization of hydrocarbon-triggered structural switching from nonporous to porous phase is very rare.52 The PXRD patterns of the nonporous phase (SIFSIX-23-Cu-β1) under 1 bar of each C3 gas collected at 298 K match well with those calculated from the SCXRD-determined crystal structures, confirming the accuracy of the SCXRD measurement procedures (Fig. S14).
SCXRD studies of guest binding
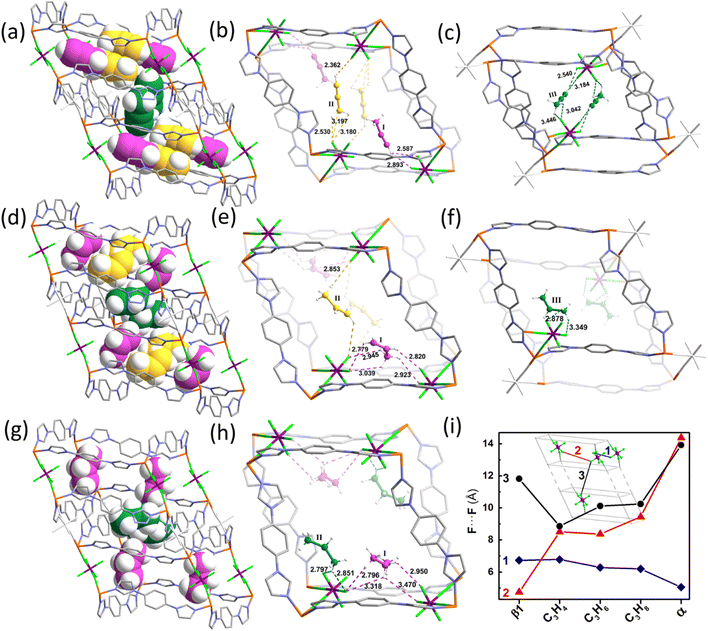
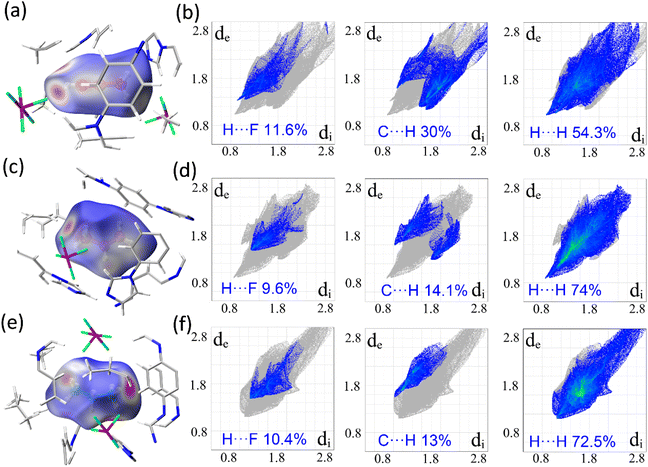
SCXRD characterization of gas-induced structural transformation can provide deep insights into the dynamic structural information at the molecular level, and additionally it can directly visualize the binding sites of gas molecules trapped in the channels, although this is challenging. Fortunately, the binding of C3 molecules in the gas-confined open phases of SIFSIX-23-Cu can be observed crystallographically (Fig. 4). For SIFSIX-23-Cu⊃C3H4, three distinct types of C3H4 molecules can be observed (Fig. 4a, S15 and S16). C3H4-I lies parallel to the CuL2 plane (Fig. 4b), with its alkynyl end oriented towards two SIFSIX anions arranged along the Cu-L(syn)-Cu edge (SIFSIX pair 1) and bound to one of them via C–H⋯F hydrogen bonds (H⋯F = 2.587/2.893 Å). The methyl group points towards the channel center, with the molecular axis inclined at 53.8° to the edge. C3H4-II lies parallel to the CuL2 plane, nestled between two SIFSIX anions (SIFSIX pair 2) along the short diagonal of the CuL2 parallelogram. The suitable F⋯F separation (8.504 Å) enables SIFSIX pair 2 to chelate C3H4-II in a head-on fashion via dual C–H⋯F hydrogen bonding interactions (H⋯F = 2.893 Å between the alkynyl end and anion, 2.530–3.197 Å between the methyl group and anion). C3H4-III is situated between two obtuse corners of adjacent cages, inclined at 30.5° to the CuL2 plane (Fig. 4c). The two ends of C3H4-III are oriented towards two SIFSIX anions (SIFSIX pair 3) from neighboring cages, and are chelated by them via C–H⋯F hydrogen bonding interactions (H⋯F = 2.540/3.184 Å between the alkynyl end and anion, 3.042–3.446 Å between the methyl group and anion) due to the suitable F⋯F distance (8.860 Å).Similarly, three different types of C3H6 can be discovered in SIFSIX-23-Cu⊃C3H6 (Fig. 4d, S17 and S18). C3H6-I is chelated by SIFSIX pair 1 in a side-on manner via various C–H⋯F hydrogen bonding interactions (H⋯F = 2.779–3.039 Å). C3H6-II is chelated by SIFSIX pair 2 in a head-on fashion via C–H⋯F hydrogen bonding interactions (H⋯F = 2.853/3.266 Å) (Fig. 4e). C3H6-III resides close to one obtuse corner of the cage and is bound to one anion of SIFSIX pair 3 via C–H⋯F hydrogen bonding interactions (H⋯F = 2.878/3.349 Å) (Fig. 4f). In SIFSIX-23-Cu⊃C3H8, two types of C3H8 molecules (C3H8-I and C3H8-II) are identified (Fig. 4g, S18 and S19), whose positions and binding manners are similar to those of C3H6-I and C3H6-III (H⋯F = 2.796–3.470 Å), respectively (Fig. 4h). Unlike C3H4-II and C3H6-II, no C3H8 molecule is chelated by SIFSIX pair 2, due to the very large SIFSIX⋯SIFSIX separation (F⋯F distance of 9.297 Å). In addition to the hydrogen bonds observed, there are numerous vdW and C–H⋯π interactions between C3 molecules and the aromatic rings of the framework (Fig. S16, S18 and S20). Based on the SCXRD results, the binding sites of C3 molecules are approximately similar, and guest-dependent non-porous-to-porous switching can generate open phases that match the size and geometry of guest molecules. It can be concluded that the binding of C3H4 is stronger than that of C3H6 and C3H8 by comparing the C–H⋯F hydrogen bonding distances and binding modes, consistent with the results of sorption experiments (Fig. S21–S29). A comparison of the interionic distances in SIFSIX pairs 1, 2, and 3 across all C3-loaded structures reveals distinct trends that vary with different adsorbates (Fig. 4i). SIFSIX pair 1 shows minimal changes for the three sorbates, whereas SIFSIX pair 2 exhibits a similar changing tendency to the diagonal distance of CuL2 (y in Fig. 3). Remarkably, SIFSIX pairs 2 and 3 change in opposite ways: for C3H4 and C3H6, pair 2 remains almost unchanged while pair 3 varies significantly; for C3H6 and C3H8, pair 2 changes substantially while pair 3 remains constant. This further confirmed that the host framework can self-adapt to accommodate different C3 molecules.
The guest–host and guest–guest interactions present in the C3-loaded structures were further analysed by means of Hirshfeld surfaces63 and their corresponding fingerprint plots were documented. The dnorm surface of each structure clearly shows a different color distribution that discloses different levels of contacts/interactions (Fig. 5a, c and e). As shown in the decomposed 2D fingerprint plots (Fig. 5b, d and f), the contribution proportions of H⋯F interactions in the Hirshfeld surface area of C3H4 (11.6%) were higher than those of C3H6 and C3H8, indicating more hydrogen bonds between C3H4 and F atoms of SiF62− anions (Fig. S30–S32). Also, the positions of spikes in the H⋯F fingerprint plots suggest the distribution of scattering points in a very short (de, di) range for C3H4, suggesting the shortest H⋯F interactions in the C3H4-loaded structure and confirming the tight binding mode of C3H4. The shortest contact between C3H4 and SiF62− anions can also be reflected by the distinct colors (red) in the dnorm surface of the Hirshfeld surface which is more apparent for C3H4 compared with C3H6 and C3H8. Given the chelating binding modes and the stronger hydrogen bonds between F atoms and the acidic alkynyl hydrogen atoms of C3H4, it can be inferred that the binding of C3H4 through H⋯F hydrogen bonds is stronger compared with C3H6 and C3H8. Additionally, the C⋯H/H⋯C short contacts are more pronounced for C3H4 (30%) compared to C3H6 and C3H8, further suggesting the tight binding fashion of C3H4 (Fig. 5b, d, and f). The H(alkyne)⋯C(host) contacts reveal the presence of C–H⋯π interactions between C3 gases and the host, especially between the methyl group of the gas molecule and the aromatic plane of ligand L (Fig. S16, S18 and S20). For all C3-loaded structures, there are numerous H⋯H short contacts, indicative of the ubiquitous vdW interactions. By carefully checking the dnorm surface for each structure, it can concluded that the guest–guest interactions also exist in all structures.
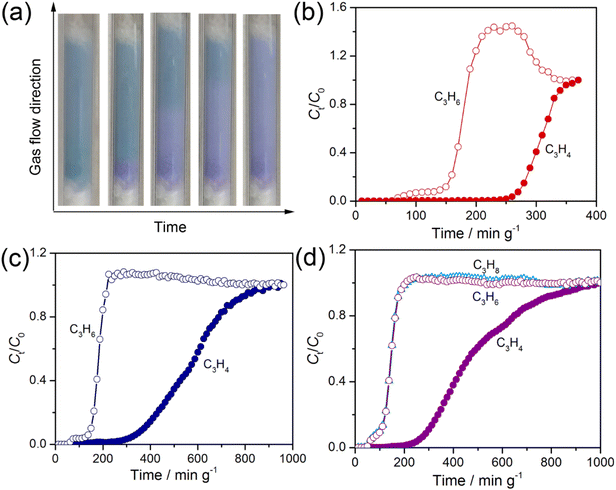
C3H4 selective sorption tests
The preferential and stronger binding affinity of C3H4 compared to C3H6 within SIFSIX-23-Cu was further investigated through quantitative fixed-bed column breakthrough experiments using binary gas mixtures of C3H4/C3H6 (40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 and 10
60 and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 v/v) at 298 K. Interestingly, the sample in the packed column underwent a color change gradually from light blue (activated β1 phase) to light purple (gas loaded phase), which evolved over time along the gas flow direction during the breakthrough experiments (Fig. 6a), showcasing the uptake of the gas molecules from the mixture. As expected, C3H6 flowed out much earlier than C3H4 did, leading to a period of purely C3H6 leaving the column (Fig. 6b and c). However, the C3H6 flow rate (Ct/C0) did not reach equilibrium directly like a non-adsorbing gas species but with a small step which is similar to that observed in adsorption isotherms, implying co-adsorption of C3H6 after gate-opening by C3H4 (Fig. S33 and S34). C3H4 were retained in the column for ca. 220 and 265 min g−1 with the adsorbed amounts of 3.93 and 1.18 mmol g−1 under 40
90 v/v) at 298 K. Interestingly, the sample in the packed column underwent a color change gradually from light blue (activated β1 phase) to light purple (gas loaded phase), which evolved over time along the gas flow direction during the breakthrough experiments (Fig. 6a), showcasing the uptake of the gas molecules from the mixture. As expected, C3H6 flowed out much earlier than C3H4 did, leading to a period of purely C3H6 leaving the column (Fig. 6b and c). However, the C3H6 flow rate (Ct/C0) did not reach equilibrium directly like a non-adsorbing gas species but with a small step which is similar to that observed in adsorption isotherms, implying co-adsorption of C3H6 after gate-opening by C3H4 (Fig. S33 and S34). C3H4 were retained in the column for ca. 220 and 265 min g−1 with the adsorbed amounts of 3.93 and 1.18 mmol g−1 under 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 and 10
60 and 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 C3H4/C3H6 gas mixtures, respectively. The average C3H6 purities in the outlet gas streams were 98.5% (40
90 C3H4/C3H6 gas mixtures, respectively. The average C3H6 purities in the outlet gas streams were 98.5% (40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60) and 99.0% (10
60) and 99.0% (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90), respectively. Three consecutive cycles of breakthrough experiments (40
90), respectively. Three consecutive cycles of breakthrough experiments (40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 C3H4/C3H6) demonstrated the stability of the sample (Fig. S35), which was further validated by PXRD tests (Fig. S36). The desorption curves after the breakthrough experiment (10
60 C3H4/C3H6) demonstrated the stability of the sample (Fig. S35), which was further validated by PXRD tests (Fig. S36). The desorption curves after the breakthrough experiment (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 C3H4/C3H6) indicated that heating was required to fully remove the adsorbed C3H4 molecules, suggestive of the stronger binding ability of C3H4 over C3H6 (Fig. S37). Notably, although under both conditions, the partial pressure for C3H6 can also trigger the gate opening behavior, SIFSIX-23-Cu still showed excellent separation performance which can probably be ascribed to the stronger binding interaction between C3H4 molecules and the host framework compared with that of C3H6, and the slow kinetics of C3H6-induced gate-opening behavior.
90 C3H4/C3H6) indicated that heating was required to fully remove the adsorbed C3H4 molecules, suggestive of the stronger binding ability of C3H4 over C3H6 (Fig. S37). Notably, although under both conditions, the partial pressure for C3H6 can also trigger the gate opening behavior, SIFSIX-23-Cu still showed excellent separation performance which can probably be ascribed to the stronger binding interaction between C3H4 molecules and the host framework compared with that of C3H6, and the slow kinetics of C3H6-induced gate-opening behavior.
To further examine the potential of SIFSIX-23-Cu for the selective adsorption of C3H4 in the presence of both C3H6 and C3H8, a breakthrough experiment with a C3H4/C3H6/C3H8 (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40
40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 v/v/v) ternary gas mixture was conducted under similar conditions (Fig. 6d). We selected this specific gas composition to ensure that the partial pressure of C3H4 exceeded the gate-opening pressure observed in the single component isotherm but those of C3H6 and C3H8 lay below their gate-opening pressures even after C3H4 in the gas mixture is completely adsorbed, which can unveil the gate opening effect of C3H6 and C3H8 on the separation performance. Surprisingly, C3H6 and C3H8 broke out of the packed column simultaneously with a negligible step, confirming that SIFSIX-23-Cu can efficiently exclude C3H6 and C3H8 (Fig. 6), and co-adsorption seems to be partly suppressed in comparison with the corresponding binary mixture. In contrast, the retention time and adsorbed amount of C3H4 in the packed column are ca. 225 min g−1 and 1.0 mmol g−1, close to those under the 10
50 v/v/v) ternary gas mixture was conducted under similar conditions (Fig. 6d). We selected this specific gas composition to ensure that the partial pressure of C3H4 exceeded the gate-opening pressure observed in the single component isotherm but those of C3H6 and C3H8 lay below their gate-opening pressures even after C3H4 in the gas mixture is completely adsorbed, which can unveil the gate opening effect of C3H6 and C3H8 on the separation performance. Surprisingly, C3H6 and C3H8 broke out of the packed column simultaneously with a negligible step, confirming that SIFSIX-23-Cu can efficiently exclude C3H6 and C3H8 (Fig. 6), and co-adsorption seems to be partly suppressed in comparison with the corresponding binary mixture. In contrast, the retention time and adsorbed amount of C3H4 in the packed column are ca. 225 min g−1 and 1.0 mmol g−1, close to those under the 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 C3H4/C3H6 mixture, which further confirmed that the gate opening effect from C3H6 does not significantly affect the selective adsorption of C3H4 from the mixture. The average concentration of C3H4 in the outlet is about 0.34%, suggestive of the effective removal of C3H4 from the ternary C3 mixture (Fig. S38). Similarly, heating was needed to completely desorb the strongly bound C3H4 molecules from the sorbent (Fig. S39). The observed good separation performance of SIFSIX-23-Cu can be attributed to the large difference in gate-opening pressures for C3H4 over C3H6 (and C3H8) and the strength of the host–guest interactions once open.
90 C3H4/C3H6 mixture, which further confirmed that the gate opening effect from C3H6 does not significantly affect the selective adsorption of C3H4 from the mixture. The average concentration of C3H4 in the outlet is about 0.34%, suggestive of the effective removal of C3H4 from the ternary C3 mixture (Fig. S38). Similarly, heating was needed to completely desorb the strongly bound C3H4 molecules from the sorbent (Fig. S39). The observed good separation performance of SIFSIX-23-Cu can be attributed to the large difference in gate-opening pressures for C3H4 over C3H6 (and C3H8) and the strength of the host–guest interactions once open.
Conclusions
In conclusion, we revealed the structural switching between nonporous and porous phases induced by C3 hydrocarbons in a flexible SIFSIX net. Sorption experiments and SCXRD experiments confirmed that the nonporous-to-porous structural switching is guest-dependent and can generate partially open phases that adapt to the size and geometry of C3 hydrocarbons. The binding sites for C3 molecules identified by SCXRD displayed stronger binding of C3H4 compared with C3H6 and C3H8, thus enhancing discrimination ability for the former. The selective adsorption of C3H4 from binary and ternary C3 gas mixtures was evaluated by experimental breakthrough tests. This work highlights the significant importance of host–guest interaction in the structural transformation and selective adsorption performance of FMOMs. Compared with the difficulty in fine-tuning the pore size and chemistry of rigid MOMs to differentiate gas species with similar properties, FMOMs switching from nonporous to guest-specific porous phases with different gate-opening pressures probably can be a potential approach to improve the recognition ability for challenging gas species.Author contributions
S. J. Qin and M. Shivanna equally contributed to this work and carried out the synthesis, characterization, sorption and X-ray diffraction measurements, and writing – original draft; D. Li, S. D. Fu and S. Qiu assisted with the characterization analysis; M. Y. Gao, C. H. Deng and S. Q. Wang assisted with the gas sorption measurements and data analysis; B. Q. Song and Q. Y. Yang carried out the methodology, supervision, and writing – review & editing; all authors contributed to preparing the manuscript.Conflicts of interest
The authors declare no competing financial interests.Data availability
CCDC 2044415, 2044416 and 2034435 contain the supplementary crystallographic data for this paper.64a–cThe data supporting this article have been included as part of supplementary information (SI). Supplementary information: the experimental sections of materials and synthesis, the methods for the SCXRD measurements, the details for breakthrough tests, and the tables and figures. See DOI: https://doi.org/10.1039/d5sc07430d.
Acknowledgements
The authors acknowledge the support of the National Natural Science Foundation of China (No. 22201025 and 22371221) and start-up funding from Chengdu University of Technology (10912 KYQD2022-09754).Notes and references
- P. Theato, B. S. Sumerlin, R. K. O’Reilly and T. H. Epps III, Chem. Soc. Rev., 2013, 42, 7055–7056 RSC.
- Y. Chai, X. Han, W. Li, S. Liu, S. Yao, C. Wang, W. Shi, I. da-Silva, P. Manuel, Y. Cheng, L. D. Daemen, A. J. Ramirez-Cuesta, C. C. Tang, L. Jiang, S. Yang, N. Guan and L. Li, Science, 2020, 368, 1002–1006 Search PubMed.
- J. J. Perry Iv, J. A. Perman and M. J. Zaworotko, Chem. Soc. Rev., 2009, 38, 1400–1417 RSC.
- Z. Chen, M. C. Wasson, R. J. Drout, L. Robison, K. B. Idrees, J. G. Knapp, F. A. Son, X. Zhang, W. Hierse, C. Kühn, S. Marx, B. Hernandez and O. K. Farha, Faraday Discuss., 2021, 225, 9–69 Search PubMed.
- S. Kitagawa, R. Kitaura and S.-i. Noro, Angew. Chem., Int. Ed., 2004, 43, 2334–2375 Search PubMed.
- H. Furukawa, K. E. Cordova, M. O'Keeffe and O. M. Yaghi, Science, 2013, 341, 1230444 Search PubMed.
- S. Mukherjee and M. J. Zaworotko, Trends Chem., 2020, 2, 506–518 Search PubMed.
- S. Horike, S. Shimomura and S. Kitagawa, Nat. Chem., 2009, 1, 695–704 Search PubMed.
- G. Férey and C. Serre, Chem. Soc. Rev., 2009, 38, 1380–1399 Search PubMed.
- A. Schneemann, V. Bon, I. Schwedler, I. Senkovska, S. Kaskel and R. A. Fischer, Chem. Soc. Rev., 2014, 43, 6062–6096 Search PubMed.
- S. Krause, N. Hosono and S. Kitagawa, Angew. Chem., Int. Ed., 2020, 59, 15325–15341 Search PubMed.
- K. Koupepidou, A. Subanbekova and M. J. Zaworotko, Chem. Commun., 2025, 61, 3109–3126 Search PubMed.
- I. Senkovska, V. Bon, L. Abylgazina, M. Mendt, J. Berger, G. Kieslich, P. Petkov, J. Luiz Fiorio, J.-O. Joswig, T. Heine, L. Schaper, C. Bachetzky, R. Schmid, R. A. Fischer, A. Pöppl, E. Brunner and S. Kaskel, Angew. Chem., Int. Ed., 2023, 62, e202218076 Search PubMed.
- J. A. Mason, J. Oktawiec, M. K. Taylor, M. R. Hudson, J. Rodriguez, J. E. Bachman, M. I. Gonzalez, A. Cervellino, A. Guagliardi, C. M. Brown, P. L. Llewellyn, N. Masciocchi and J. R. Long, Nature, 2015, 527, 357–361 Search PubMed.
- A.-X. Zhu, Q.-Y. Yang, A. Kumar, C. Crowley, S. Mukherjee, K.-J. Chen, S.-Q. Wang, D. O′Nolan, M. Shivanna and M. J. Zaworotko, J. Am. Chem. Soc., 2018, 140, 15572–15576 CrossRef PubMed.
- M.-Y. Zhou, X.-W. Zhang, H. Yi, Z.-S. Wang, D.-D. Zhou, R.-B. Lin, J.-P. Zhang and X.-M. Chen, J. Am. Chem. Soc., 2024, 146, 12969–12975 CrossRef PubMed.
- R. E. Morris and L. Brammer, Chem. Soc. Rev., 2017, 46, 5444–5462 RSC.
- D. P. van Heerden, V. J. Smith, H. Aggarwal and L. J. Barbour, Angew. Chem., Int. Ed., 2021, 60, 13430–13435 CrossRef PubMed.
- A. P. Katsoulidis, D. Antypov, G. F. S. Whitehead, E. J. Carrington, D. J. Adams, N. G. Berry, G. R. Darling, M. S. Dyer and M. J. Rosseinsky, Nature, 2019, 565, 213–217 CrossRef PubMed.
- Q.-Y. Yang, P. Lama, S. Sen, M. Lusi, K.-J. Chen, W.-Y. Gao, M. Shivanna, T. Pham, N. Hosono, S. Kusaka, J. J. Perry IV, S. Ma, B. Space, L. J. Barbour, S. Kitagawa and M. J. Zaworotko, Angew. Chem., Int. Ed., 2018, 57, 5684–5689 CrossRef PubMed.
- V. I. Nikolayenko, D. C. Castell, D. Sensharma, M. Shivanna, L. Loots, K. A. Forrest, C. J. Solanilla-Salinas, K.-i. Otake, S. Kitagawa and L. J. Barbour, Nat. Chem., 2023, 15, 542–549 CrossRef.
- H. Fang, X.-Y. Liu, H.-J. Ding, M. Mulcair, B. Space, H. Huang, X.-W. Li, S.-M. Zhang, M.-H. Yu, Z. Chang and X.-H. Bu, J. Am. Chem. Soc., 2024, 146, 14357–14367 CrossRef PubMed.
- C. Gu, N. Hosono, J.-J. Zheng, Y. Sato, S. Kusaka, S. Sakaki and S. Kitagawa, Science, 2019, 363, 387–391 CrossRef PubMed.
- H. Yang, T. X. Trieu, X. Zhao, Y. Wang, Y. Wang, P. Feng and X. Bu, Angew. Chem., Int. Ed., 2019, 58, 11757–11762 Search PubMed.
- S. K. Elsaidi, M. H. Mohamed, D. Banerjee and P. K. Thallapally, Coord. Chem. Rev., 2018, 358, 125–152 CrossRef.
- S.-H. Lo, L. Feng, K. Tan, Z. Huang, S. Yuan, K.-Y. Wang, B.-H. Li, W.-L. Liu, G. S. Day, S. Tao, C.-C. Yang, T.-T. Luo, C.-H. Lin, S.-L. Wang, S. J. L. Billinge, K.-L. Lu, Y. J. Chabal, X. Zou and H.-C. Zhou, Nat. Chem., 2020, 12, 90–97 Search PubMed.
- R.-B. Lin, S. Xiang, W. Zhou and B. Chen, Chem, 2020, 6, 337–363 Search PubMed.
- T. Kundu, M. Wahiduzzaman, B. B. Shah, G. Maurin and D. Zhao, Angew. Chem., Int. Ed., 2019, 58, 8073–8077 Search PubMed.
- H. Zeng, M. Xie, Y.-L. Huang, Y. Zhao, X.-J. Xie, J.-P. Bai, M.-Y. Wan, R. Krishna, W. Lu and D. Li, Angew. Chem., Int. Ed., 2019, 58, 8515–8519 CrossRef PubMed.
- M.-H. Yu, B. Space, D. Franz, W. Zhou, C. He, L. Li, R. Krishna, Z. Chang, W. Li, T.-L. Hu and X.-H. Bu, J. Am. Chem. Soc., 2019, 141, 17703–17712 Search PubMed.
- J. Tian, Q. Chen, F. Jiang, D. Yuan and M. Hong, Angew. Chem., Int. Ed., 2023, 62, e202215253 Search PubMed.
- T. Xu, W. Jiang, Y. Tao, M. Abdellatief, K. E. Cordova and Y.-B. Zhang, J. Am. Chem. Soc., 2024, 146, 11225–11234 Search PubMed.
- L. Zhang, B. Yu, M. Wang, Y. Chen, Y. Wang, L.-B. Sun, Y.-B. Zhang, Z. Zhang, J. Li and L. Li, Angew. Chem., Int. Ed., 2025, 64, e202418853 CrossRef PubMed.
- M. Shivanna, K.-i. Otake, S. Hiraide, T. Fujikawa, P. Wang, Y. Gu, H. Ashitani, S. Kawaguchi, Y. Kubota, M. T. Miyahara and S. Kitagawa, Angew. Chem., Int. Ed., 2023, 62, e202308438 Search PubMed.
- Z. Niu, Z. Fan, T. Pham, G. Verma, K. A. Forrest, B. Space, P. K. Thallapally, A. M. Al-Enizi and S. Ma, Angew. Chem., Int. Ed., 2022, 61, e202117807 CrossRef.
- X. Li, D. Sensharma, W. Graham, V. Bon, E. Lin, X.-J. Kong, T. He, A. A. Bezrukov, Z. Zhang, S. Kaskel, T. Thonhauser and M. J. Zaworotko, Angew. Chem., Int. Ed., 2025, 64, e202507757 CrossRef.
- B.-Q. Song, Q.-Y. Yang, S.-Q. Wang, M. Vandichel, A. Kumar, C. Crowley, N. Kumar, C.-H. Deng, V. GasconPerez, M. Lusi, H. Wu, W. Zhou and M. J. Zaworotko, J. Am. Chem. Soc., 2020, 142, 6896–6901 CrossRef.
- M. Jung, J. Park, R. Muhammad, T. Park, S.-Y. Jung, J. Yi, C. Jung, J. Ollivier, A. J. Ramirez-Cuesta, J. T. Park, J. Kim, M. Russina and H. Oh, Nat. Commun., 2025, 16, 2032 CrossRef PubMed.
- R. A. Klein, L. W. Bingel, A. Halder, M. Carter, B. A. Trump, E. D. Bloch, W. Zhou, K. S. Walton, C. M. Brown and C. M. McGuirk, J. Am. Chem. Soc., 2023, 145, 21955–21965 CrossRef.
- Q. Dong, X. Zhang, S. Liu, R.-B. Lin, Y. Guo, Y. Ma, A. Yonezu, R. Krishna, G. Liu, J. Duan, R. Matsuda, W. Jin and B. Chen, Angew. Chem., Int. Ed., 2020, 59, 22756–22762 CrossRef PubMed.
- R. M. Main, S. M. Vornholt, C. M. Rice, C. Elliott, S. E. Russell, P. J. Kerr, M. R. Warren and R. E. Morris, Commun. Chem., 2023, 6, 44 CrossRef.
- W. M. Bloch, N. R. Champness and C. J. Doonan, Angew. Chem., Int. Ed., 2015, 54, 12860–12867 CrossRef.
- T. L. Easun, F. Moreau, Y. Yan, S. Yang and M. Schröder, Chem. Soc. Rev., 2017, 46, 239–274 Search PubMed.
- M.-A. Springuel-Huet, A. Nossov, Z. Adem, F. Guenneau, C. Volkringer, T. Loiseau, G. Férey and A. Gédéon, J. Am. Chem. Soc., 2010, 132, 11599–11607 Search PubMed.
- P. L. Llewellyn, G. Maurin, T. Devic, S. Loera-Serna, N. Rosenbach, C. Serre, S. Bourrelly, P. Horcajada, Y. Filinchuk and G. Férey, J. Am. Chem. Soc., 2008, 130, 12808–12814 CrossRef PubMed.
- C. X. Bezuidenhout, V. J. Smith, P. M. Bhatt, C. Esterhuysen and L. J. Barbour, Angew. Chem., Int. Ed., 2015, 54, 2079–2083 CrossRef.
- H. Wang, M. Warren, J. Jagiello, S. Jensen, S. K. Ghose, K. Tan, L. Yu, T. J. Emge, T. Thonhauser and J. Li, J. Am. Chem. Soc., 2020, 142, 20088–20097 CrossRef.
- J.-P. Zhang, P.-Q. Liao, H.-L. Zhou, R.-B. Lin and X.-M. Chen, Chem. Soc. Rev., 2014, 43, 5789–5814 RSC.
- P. Lama, H. Aggarwal, C. X. Bezuidenhout and L. J. Barbour, Angew. Chem., Int. Ed., 2016, 55, 13271–13275 CrossRef.
- E. J. Carrington, S. F. Dodsworth, S. van Meurs, M. R. Warren and L. Brammer, Angew. Chem., Int. Ed., 2021, 60, 17920–17924 CrossRef PubMed.
- M. Đaković, M. Pisačić, M. Borovina, I. Kodrin, A. Kenđel and T. Frey, J. Am. Chem. Soc., 2025, 147, 22219–22227 CrossRef.
- P. Lama and L. J. Barbour, J. Am. Chem. Soc., 2018, 140, 2145–2150 CrossRef PubMed.
- X. Cui, K. Chen, H. Xing, Q. Yang, R. Krishna, Z. Bao, H. Wu, W. Zhou, X. Dong, Y. Han, B. Li, Q. Ren, M. J. Zaworotko and B. Chen, Science, 2016, 353, 141–144 CrossRef PubMed.
- B.-Q. Song, M.-Y. Gao, L. Mercene van Wyk, C.-H. Deng, A. C. Eaby, S.-Q. Wang, S. Darwish, D. Li, S.-J. Qin, Y.-L. Peng, Q.-Y. Yang, L. J. Barbour and M. J. Zaworotko, Chem. Sci., 2025, 16, 9010–9019 Search PubMed.
- L. Li, H.-M. Wen, C. He, R.-B. Lin, R. Krishna, H. Wu, W. Zhou, J. Li, B. Li and B. Chen, Angew. Chem., Int. Ed., 2018, 57, 15183–15188 CrossRef PubMed.
- L. Yang, X. Cui, Z. Zhang, Q. Yang, Z. Bao, Q. Ren and H. Xing, Angew. Chem., Int. Ed., 2018, 57, 13145–13149 CrossRef PubMed.
- Y.-L. Peng, C. He, T. Pham, T. Wang, P. Li, R. Krishna, K. A. Forrest, A. Hogan, S. Suepaul, B. Space, M. Fang, Y. Chen, M. J. Zaworotko, J. Li, L. Li, Z. Zhang, P. Cheng and B. Chen, Angew. Chem., Int. Ed., 2019, 58, 10209–10214 Search PubMed.
- L. Yang, X. Cui, Y. Zhang, Q. Yang and H. Xing, J. Mater. Chem. A, 2018, 6, 24452–24458 Search PubMed.
- L. Yang, X. Cui, Q. Yang, S. Qian, H. Wu, Z. Bao, Z. Zhang, Q. Ren, W. Zhou and B. Chen, Adv. Mater., 2018, 30, 1705374 CrossRef.
- L. Li, R.-B. Lin, R. Krishna, X. Wang, B. Li, H. Wu, J. Li, W. Zhou and B. Chen, J. Am. Chem. Soc., 2017, 139, 7733–7736 CrossRef.
- B. Yu, S. Geng, H. Wang, W. Zhou, Z. Zhang, B. Chen and J. Jiang, Angew. Chem., Int. Ed., 2021, 60, 25942–25948 CrossRef CAS PubMed.
- A. L. Spek, Acta Cryst. D, 2009, 65, 148–155 CrossRef CAS.
- M. A. Spackman and D. Jayatilaka, CrystEngComm, 2009, 11, 19–32 RSC.
- (a) CCDC 2044415: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc26mcw3.; (b) CCDC 2044416: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc26mcx4; (c) CCDC 2034435: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc268zyf.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |