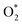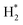Open Access Article
Open Access ArticleMolecular dynamics study of catalytic H2/O2 recombination on Pd, Pt, Cu, Ag, and Au nanoclusters using the universal neural network potential
Yusuke
Tateishi
 ab,
Louise M.
Botha
ab,
Louise M.
Botha
 b,
Alina E.
Kozhukhova
b,
Alina E.
Kozhukhova
 b,
Manabu
Sugimoto
b,
Manabu
Sugimoto
 *ac,
Ken-ichi
Aika
*ac,
Ken-ichi
Aika
 d and
Dmitri G.
Bessarabov
d and
Dmitri G.
Bessarabov
 *b
*b
aFaculty of Advanced Science and Technology, Kumamoto University, 2-39-1 Kurokami, Chuo-ku, Kumamoto 860-8555, Japan
bHySA Infrastructure, North-west University, 11 Hoffman Street, Potchefstroom, 2531, South Africa. E-mail: Dmitri.Bessarabov@nwu.ac.za
cDepartment of Pharmaceutical Sciences, Daiichi University of Pharmacy, 22-1 Tamagawa-machi, Minami-ku, Fukuoka 815-8511, Japan. E-mail: m-sugimoto@daiichi-cps.ac.jp
dNational Institute of Technology (KOSEN), Numazu College, 3600 Ooka, Numazu, Shizuoka 410-8501, Japan
First published on 23rd January 2026
Abstract
The H2/O2 catalytic recombination reaction, essential for safely utilizing hydrogen energy, was simulated rapidly and accurately using the molecular dynamics (MD) simulation with a machine-learning potential known as the ‘universal neural network potential’ (UNNP). The catalytic activities of Pd, Pt, Cu, Ag, and Au nanoclusters were systematically investigated under a 100 bar H2/O2 atmosphere at 500 K. This approach enables the investigation of the entirely dynamic catalytic reaction, rather than assembling a static configuration at 0 K. Throughout the simulation, the ability to catalyze H2O formation on the Pt surface was successfully reproduced, aligning with previous experimental findings. The simulations have revealed characteristic differences and the suitability of reactivity with H2/O2 across the studied metals, demonstrating the time evolution of gas-surface interactions and the overall detailed reaction mechanism of the H2/O2 recombination reaction. In particular, the reaction pathway O2 → OOH → H2O was revealed to occur preferentially on the Pt nanocluster, while other metals exhibited unsuitability, such as Pd's intense H trapping and Cu's surface instability due to excessive O2 dissociation. The two features of molecular O2 adsorption and the facile surface diffusion of H atoms play a crucial role as necessary conditions for catalytic activity. The accelerated MD method enables the rapid and accurate elucidation of the atomic-level mechanisms and activity differences in the H2/O2 recombination reaction, offering a fast and accurate workflow for both performance screening and mechanism discovery, thus accelerating catalyst design.
1 Introduction
Green hydrogen, produced via electrochemical water splitting through water electrolysis powered by renewable energy sources, such as wind or solar, is widely regarded as a sustainable and environmentally friendly energy carrier. Once produced, hydrogen can be compressed, liquefied, or chemically converted into, for example, ammonia or liquid organic hydrogen carriers (LOHCs) such as methylcyclohexane (MCH) and perhydro-benzyltoluene (BT-H). Each of these options offers distinct advantages for hydrogen storage and long-distance transportation.1 Additionally, green hydrogen can be combined with captured CO2 to produce e-methane—a synthetic equivalent of natural gas—and it can be directly used as a carbon-neutral fuel, provided that the released CO2 is subsequently captured and reused. Thus, green hydrogen plays a critical role in enabling the transition from fossil-based to sustainable energy systems.2–4Despite its advantages, hydrogen presents safety challenges due to its low minimum ignition energy (0.02 mJ) and wide flammability range (4–75 vol% hydrogen in air).5 Accidentally released hydrogen can rapidly move upwards, accumulate near ceilings, and mix with air in confined spaces. This, in turn, can lead to the formation of explosive mixtures, which, if ignited, will cause damage to infrastructure and loss of life. The accumulation of explosive hydrogen/air mixtures can occur in any confined space where hydrogen is used, such as garages for hydrogen-fueled cars, underground mines, fuel cell systems, and hydrogen storage facilities, including critical applications like nuclear power plants.
Various safety measures have been implemented to mitigate these risks, including the use of inert gases (e.g., nitrogen, helium, and argon),6 natural and forced ventilation,7,8 water mists,9 and the employment of safety devices such as passive autocatalytic recombiners (PARs).1,5,10,11 Among these strategies, the use of PARs stands out as one of the most robust and widely adopted solutions. A PAR mitigates the risk of explosion by catalytically converting accidentally released hydrogen into water vapor through its reaction with oxygen present in the ambient air. The term “passive” refers to the ability to initiate the recombination reaction spontaneously as soon as hydrogen concentration begins to increase. Hence, PARs do not require any external power source.
Different types (catalysts) and models (dimensions and chimney) of PARs have been developed. The PAR manufacturers include Atomic Energy Canada Ltd (AECL), Electrowatt Engineering Ltd, NIS, Siemens, AREVA, Framatome, and ISPC RET. Typical catalytic materials include Pt- and Pd-coated plates, pellets, and rods. However, these catalysts face several challenges, including high material costs, overheating risks that may lead to self-ignition, and deactivation caused by exposure to water vapor and chemical poisons such as carbon monoxide and iodine.12–14 The occurrence of hotspots on the catalyst surface can also lead to the self-ignition of the PAR due to the local initiation of combustion waves or flames that can propagate through the hydrogen/air mixture, ultimately damaging the inside components as well as the outside of the PAR.15 To solve the problems mentioned above, it is important not only to develop new advanced catalysts but also to design the catalyst structure as a functional component.15,16
Transition metals, particularly platinum group metals (PGMs), are essential catalyst materials due to their high activity and versatility, making them fundamental to the hydrogen economy. The interaction between reactants (H2 and O2) and transition metals is crucial in key chemical processes, including the oxygen evolution reaction (OER), the oxygen reduction reaction (ORR), and H2/O2 recombination.3,17,18 These reactions typically follow adsorption–desorption mechanisms, where reactants bind to the catalyst surface and detach as products. Extensive experimental research has focused on designing H2/O2 recombination catalysts to develop materials with catalytic activity comparable to or exceeding that of PGMs.11,19–28 Computational studies29–31 have also investigated electronic, thermodynamic, and structural properties of catalysts, deepening our understanding of existing catalysts and providing valuable feedback for experimental research, which is crucial for advancing catalyst design.32,33
Despite these computational advances, most previous investigations have remained essentially static, restricted to a handful of predefined elementary steps on ideal surfaces explored at 0 K. Consequently, capturing the concerted adsorption, diffusion, and recombination events has been difficult, which governs catalyst performance under realistic conditions.
To address this, we have introduced high-speed, high-precision Density Functional Theory (DFT)-level Molecular Dynamics (MD) simulations at finite temperatures, aiming to advance this research field. So far, DFT and MD simulations have been widely used in catalyst design29,30 to provide computational insights. While these methods are generally faster than experimental approaches, they can still be time-consuming, depending on the scope of the research. Recent advancements in machine learning (ML), which drive artificial intelligence (AI) technologies, have gained attention for accelerating computational research and reducing time requirements. These advancements have significantly impacted both academia and industry. ML models, such as neural network potentials (NNPs), can approximate the energies obtained from first-principles methods, like the DFT and Hartree–Fock (HF) methods, significantly faster (exceeding 105 fold) than direct calculations.34–36 The application of NNPs has proven highly effective in accelerating catalyst development and advancing understanding of catalytic processes. One notable computation platform is Matlantis, which utilizes the Universal Neural Network Potential (UNNP). Current research focuses on leveraging the UNNP within MD simulations to discover next-generation, high-performance materials and to gain deeper insights into the underlying mechanisms.37,38
In this study, we investigate the catalytic properties of Pd, Pt, Cu, Ag, and Au in the dynamical H2/O2 recombination reaction at finite temperature using universal neural network potential-driven molecular dynamics (UNNP-MD). The findings offer valuable insights into the reaction mechanisms, and contribute to improving catalytic efficiency. Among the metals analyzed, Pt is reconfirmed as the most active catalyst, offering balanced O2 adsorption and diffusivity of H atoms on the metal surface, which are key factors influencing catalytic performance. At the same time, the reliability and applicability of the UNNP-MD approach for catalyst development are examined and confirmed.
2 Computational method
The Universal Neural Network Potential (UNNP) was implemented using the Matlantis software package36,39 from Matlantis Corp., utilizing the universal PreFerred Potential (PFP).2.1 Structure modelling
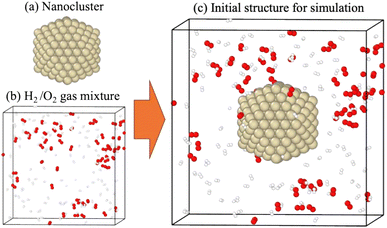
The nanoclusters were obtained from the Atomic Simulation Environment (ASE) software package.40 The nanocluster has an icosahedral morphology, preserving a high proportion of the (111)-like stable surface,41 totaling 309 atoms and a diameter of about 1 nm. Geometric optimizations of these were performed using the UNNP.To simulate the catalytic H2/O2 recombination on these nanoclusters, the nanoclusters were embedded in a cubic unit cell, surrounded by H2 and O2 molecules, as shown in Fig. 1. Using this approach and structure, we simulated the catalytic performance of the nanoclusters in an H2/O2 environment.
2.2 Molecular dynamics simulation
The MD simulations were performed under an NVT ensemble using the Nosé–Hoover thermostat,42,43 with a thermostat time constant (τ) of 10 femtoseconds (fs), retrieving force and potential energy determined by the UNNP. The temperature was fixed at 500 K, and the volume was set to 125![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 Å3. The simulations were run for 200 picoseconds (ps), with a timestep of 1 fs. For each simulation, three runs were conducted, with the initial velocities of each atom randomly altered.
000 Å3. The simulations were run for 200 picoseconds (ps), with a timestep of 1 fs. For each simulation, three runs were conducted, with the initial velocities of each atom randomly altered.
2.3 DFT calculation
The binding energies of hydrogen and oxygen on metal surfaces were investigated to validate the UNNP calculations. Spin-polarized plane-wave DFT calculations were performed using the Vienna Ab Initio Simulation Package (VASP).44 The parameters selected for the DFT calculations were kept similar to those defined for the UNNP, which is applied using the Matlantis software.3 Results and discussion
3.1 Characterization of metal surfaces in a H2/O2 gas mixture
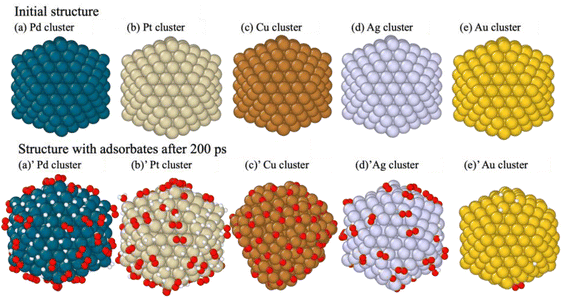
Molecular dynamics (MD) simulations were conducted over 200 ps to investigate the catalytic performance of metal nano-clusters in the H2/O2 recombination reaction. The 200 ps MD simulation, which included 484 atoms, required only about 6–7 hours. As shown in Fig. 2, structural changes in metal nanoclusters after the 200 ps simulation were clearly observed. The initial nanoclusters shown in Fig. 2(a–e) have uniform surfaces without absorbents; in contrast, the structures in Fig. 2(a–e)′ captured several types of absorbents, indicating their characteristics and potential in the H2/O2 recombination reaction. Given the difference in reactivity and adsorption characteristics, these metal nanoclusters can be classified into three classes: (I) Pd and Pt, (II) Cu, and (III) Ag and Au.Class I metals (Pd and Pt) facilitate the dissociative adsorption of hydrogen, leading to a rapid increase in H* species at the beginning of the simulation, as shown in Fig. 2(a and b). Here * denotes a species adsorbed on the metal surface. Interestingly, O2 was preferentially adsorbed rather than atomic O. On Pd and Pt, O2 molecules were primarily located at on-top/on-top sites, as shown in Fig. 2(a and b), suggesting that O2 dissociation does not easily occur on these surfaces. Hydrogen atoms on the Pd surface tend to localize on hollow sites or migrate into the nanocluster, whereas H* is found on top or bridge sites on Pt. The adsorption characteristics of these PGMs, which are required for subsequent reactions, are clearly reflected in the atomic configurations, as shown in Fig. 2(a and b)′.
In contrast, Class II metal (Cu) exhibited significantly lower reactivity and was less suitable as a recombination catalyst. The Cu nanocluster showed strong interactions with oxygen, leading to substantial O2 dissociation (Fig. 2(c)′). This resulted in a roughened surface and a lack of H and H2 adsorption, suggesting that, under the given simulation conditions, the Cu surface is unstable, making it unsuitable as an H2/O2 recombination catalyst.
Class III metal (Ag and Au) nanoclusters were nert toward H2, with only limited dissociative adsorption observed over time. Ag could adsorb molecular O2 along with a small amount of dissociated H, whereas Au displayed even lower reactivity, with only minimal surface-bonded O2 and H. These adsorption behaviors, visualized in Fig. 2(d and e)′, support the conclusion that noble metals such as Ag and Au are less effective for catalytic recombination. At the same time, Pt and Pd were emphasized as offering favorable surface characteristics, leading to better H2/O2 recombination catalysts.
The atomic diffusion dynamics of oxygen and hydrogen atoms were also analysed based on the MD simulation trajectory. The following section discusses these findings in detail.
3.2 Reaction dynamics of chemical species (reactants, intermediates, and products)
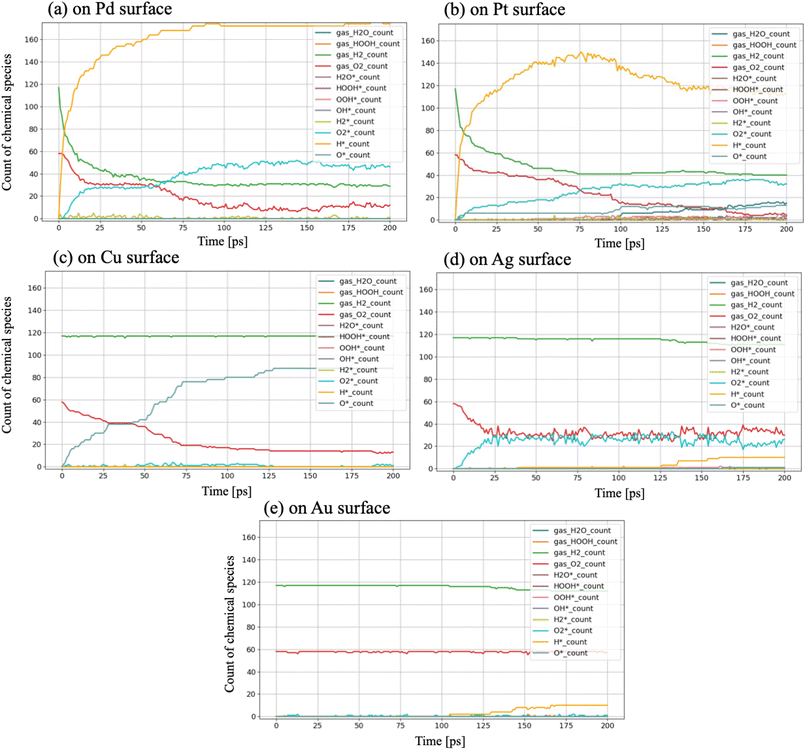
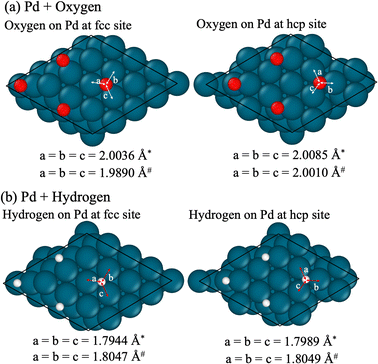
As presented in Fig. 3 and Table 1, the number of chemical species, which can be categorized as reactants, intermediates, or products, was tracked and quantified, thereby identifying the chemical bonds between these species and metal surfaces. Fig. 3 illustrates the simulated time evolution of surface adsorption and reactions. Based on these observations, the following series of elementary reactions ((R1)–(R11)) can be presumed for the H2/O2 recombination process.| H2(gas) → H* + H* | (R1) |
| O2(gas) → O* + O* | (R2) |
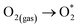 | (R3) |
| O* + H* → OH* | (R4) |
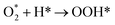 | (R5) |
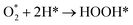 | (R6) |
| HOOH* → 2OH* | (R7) |
| OH* + H* → H2O* | (R8) |
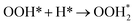 | (R9) |
| OOH*→OH* + O* | (R10) |
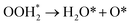 | (R11) |
| H2O* → H2O (gas) | (R12) |
| HOOH* → HOOH (gas) | (R13) |
| Metal | Reactants | Intermediates | Products | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| O* | H* | OH* | OOH* | H2O* | HOOH* | H2O(gas) | HOOH(gas) | |||
| Pd | 48 (1) | 1 (0) | 1 (1) | 171 (8) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Pt | 32 (2) | 0 (0) | 11 (2) | 113 (4) | 2 (2) | 4 (1) | 3 (2) | 1 (1) | 15 (2) | 1 (0) |
| Cu | 1 (1) | 0 (0) | 90 (2) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Ag | 23 (2) | 0 (0) | 4 (2) | 6 (3) | 3 (1) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| Au | 0 (0) | 0 (0) | 0 (0) | 5 (4) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
Chemical bonds were identified when the distance between atoms was less than the sum of the covalent bond radii of each element pair.45 Some example criteria used to classify each adsorbed species are as follows:
• H2O(gas): one oxygen atom is bonded to two hydrogen atoms, with no metal atoms within the covalent bond radii of these atoms.
• H2O2(gas): two connected oxygen atoms, each bonded to one hydrogen atom, with no metal atoms within the covalent bond radii of these atoms.
• 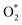 : two oxygen atoms are bonded, and at least one oxygen is bound to a metal atom within the covalent bond radii.
: two oxygen atoms are bonded, and at least one oxygen is bound to a metal atom within the covalent bond radii.
• OH*: an oxygen atom is bonded to a hydrogen atom and in contact with a metal atom within the covalent bond radii.
The quantity of all the reactants (H2, O2, 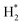 ,
, 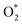 , H*, and O*), the intermediates (OH*, OOH*, H2O*, and HOOH*), and the products (H2O and HOOH), was determined using a consistent classification method. Regarding H2O and HOOH, H2O and HOOH are loosely bound to chemicals (not metals) on surfaces due to interactions such as hydrogen bonding or dispersion forces; however, those products have been counted as H2O, and not H2O*, because of any interaction with metal.
, H*, and O*), the intermediates (OH*, OOH*, H2O*, and HOOH*), and the products (H2O and HOOH), was determined using a consistent classification method. Regarding H2O and HOOH, H2O and HOOH are loosely bound to chemicals (not metals) on surfaces due to interactions such as hydrogen bonding or dispersion forces; however, those products have been counted as H2O, and not H2O*, because of any interaction with metal.
As shown in Fig. 3, adsorption in the system reaches convergence within 200 ps and reveals different features as catalysts, indicating that the chosen simulation time window is suitable to scope them, although the reaction of Pt is still ongoing towards producing H2O, as visible in Fig. S4. The temporary quantities of the chemical species, and their standard deviation after three 200 ps runs are summarized in Table 1. Based on these data, the following unique features were quantitatively identified.
As shown in Table 1, no products or intermediates were observed in this 200 ps simulation on the Pd surface. However, Pd has been recognized experimentally as an active metal in the H2/O2 recombination reaction as PdO21 or PdOx/γ-Al2O3.22 And there are also DFT studies demonstrating the H2O and H2O2 formation mechanism of Pd, depending on O2 adsorption.46,47 This discrepancy between previous studies and our result: Pd's low catalytic activity is possibly due to several conditional limitations: the system's high temperature, non-lean hydrogen, non-surface oxidization, the absence of a support, etc.22 The lack of activity in the recombination reaction should be the result of the combined effects of excessive O2 and H adsorption, affected by the above points. These findings highlight Pd's potential in H2/O2 recombination but emphasize the need for structural or alloying modifications to overcome its limitations in H2/O2 recombination reactions.
Regarding another Class I metal, 15 H2O molecules were produced during the simulation, along with surface-bound intermediates such as OOH*, OH*, and H2O*, as evidenced in Table 1. Similar to Pd, it demonstrated that O2 and O adsorption can occur on the Pt surface ((R2) and (R3)). From Fig. 3(b), on the Pt surface, H2 dissociation and H adsorption (R1) began intensely at the start of the simulation. Subsequently, a decrease in adsorbed hydrogen atoms was observed, and the formation of intermediates began at around 75 ps. Notably, as shown in Fig. 4(a), significantly larger amounts of OOH* were formed in comparison to OH*. This observation suggests that the formation of H2O does not originate primarily from OH* (R8) but rather from a reaction sequence following O2 adsorption and OOH* formation on the Pt surface (R5).
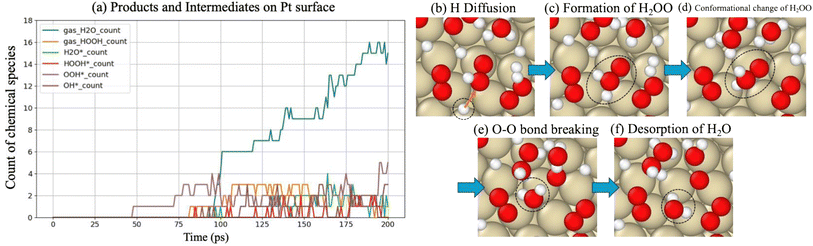 | ||
Fig. 4 Forming intermediates and products. (a) More detailed reaction time evolution (than Fig. 3(b)), specifically intermediates and products on the Pt surface. Snapshot of the simulated reaction forming H2O from OOH* and H* on the Pt surface. (b) The presence of OOH* and atomic H*. (c) 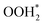 is formed through the bonding of H* with OOH*. In (d), a conformational rearrangement occurs. (e) The dissociation of is formed through the bonding of H* with OOH*. In (d), a conformational rearrangement occurs. (e) The dissociation of  into H2O and another product. Finally, H2O is desorbed from the surface into air, as shown in (f). into H2O and another product. Finally, H2O is desorbed from the surface into air, as shown in (f). | ||
Furthermore, when comparing the O2 adsorption capacities of Pd and Pt, it is evident that Pt has a slightly lower adsorption capacity (Table 1). The difference suggests that Pt's moderate Pt–OO–Pt interaction strength creates an ideal environment for forming intermediates such as OOH* and OH* ((R5) and (R6)).
As shown in Fig. 4(b–f), O2 adsorbed on the Pt surface combined with H to produce OOH*, which led to the formation of the target product, H2O. This reaction pathway is consistent with the mechanism demonstrated by Jacob's DFT study,48 in which H2O was produced via OOH*. The O2 adsorption likely serves as a critical initial state, enabling subsequent reaction steps ((R8) and (R9)), through forming key intermediates such as OH* and OOH*.
Among Class I metals, Pt exhibited optimal balance between O2 adsorption and mobility for hydrogen, making it the most effective catalyst in H2/O2 recombination. As presented in Fig. 3, Pt has adsorbed a moderate number of H* species—more than Cu or Ag but fewer than Pd—indicating Pt's moderate H* adsorption strength. As will be discussed in detail later, this adsorption property ensures sufficient surface mobility of hydrogen, which is essential for subsequent reactions leading to H2O and H2O2 formation, creating an optimal environment for catalytic activity for H2/O2 recombination. The balance of two requirements, which are O2 adsorption and mobility for hydrogen, facilitated efficient reaction progression, leading to the formation of 15H2O molecules and intermediates such as H2O*, OOH*, and OH* (Table 1).
Although a pure Cu nanocluster appeared catalytically inactive in our simulations, it is known that oxidized copper species (CuO) exhibit catalytic activity in experimental H2/O2 recombination.21 In our model and approach, partial oxidation of the Cu surface was observed; however, a fully oxidized CuO surface was not explicitly constructed or simulated due to the limited O2 gas supply and simulation time. This discrepancy highlights a limitation of our computational approach to catalytic reactions, including the surface oxidation process. To simulate the reaction for metal oxides such as PdO and CuO, computationally, prior oxidation treatment should be required.
 in our simulations supports the validity of our approach and observation. These results suggest that while Ag exhibits moderate catalytic activity, further optimization, such as alloying, is necessary for enhancing its performance in H2/O2 recombination reactions.
in our simulations supports the validity of our approach and observation. These results suggest that while Ag exhibits moderate catalytic activity, further optimization, such as alloying, is necessary for enhancing its performance in H2/O2 recombination reactions.
The Au surface exhibited minimal catalytic activity, with non-aggressive adsorption and surface reactions, except for a small amount of H adsorption. A previous study52 also demonstrated that hydrogen interacts and dissociates on Au nanoclusters. These results suggest that Au's surface characteristics are inherently unsuitable for H2/O2 recombination reactions despite its application in other catalytic processes, such as CO oxidation.53 Moreover, the low binding energies for both H and O suggest that adsorption will not readily occur on the Au surface, which aligns with the general properties of Au. Utilizing Class III metals in H2/O2 recombination reactions is challenging, but these properties possibly suppress the overly strong interactions of other elements through alloying, like the development of Pd–Au.54
Overall, Pt exhibited the highest catalytic performance, reaffirming its superiority as an ideal catalyst for the H2/O2 recombination reaction. The exceptional catalytic activity of Pt has been well-established in previous studies,5,6,24–30 and the present simulations further validate these findings. Fig. 3(b) and 4(a) highlight Pt's superior performance, which stems from its unique combination of adsorption properties. Specifically, the findings suggest that an effective H2/O2 recombination catalyst relies not on strong O2 dissociation but on efficient O2 adsorption and the facilitation of moderate-strength H adsorption, which leads to OOH* formation, as shown in Fig. 4.
In this study, we have investigated the catalytic reactivity of icosahedral nanoclusters for the five metals. They are mainly covered by the (111) surfaces. It should be noted that catalytic activity may vary in other symmetries with different types of exposed surfaces and edges. The influence of alternative morphologies, including truncated octahedra and decahedra, should also be investigated in future studies.
3.3 Diffusion of oxygen and hydrogen on metal surfaces
The previous section established that the efficiency of O2 adsorption plays a critical role in catalytic H2/O2 recombination. Therefore, this section analyzes the displacement behavior of oxygen and hydrogen on the nanoclusters to gain insights into the dynamic behavior of the adsorbates on the surfaces. At this time, the oxygen and hydrogen being analyzed are limited to those within 2.5 Å of metal atoms. This is to prevent the overestimation of displacement caused by oxygen and hydrogen in air.Fig. 5(a) illustrates the varying displacement behaviors of oxygen on each metal surface. As described above (Fig. 2(a, b and d)), on Ag, Pt, and Pd surfaces, oxygen typically exhibited O2 adsorption, while Cu formed a strong Cu–O bond. Oxygen displacement on Ag and Pd surfaces is relatively similar, at around 3.2 Å ps−1, compared to restricted oxygen displacement of Pt of around 2.9 Å ps−1. This difference indicates that O2 on the Pt surface is more stable and strongly adsorbed compared to Ag and Pd. The displacement of Cu is smaller than that of Ag and Pd, and its displacement is nearly equivalent to that of Pt, suggesting that oxygen atoms are strongly adsorbed on the Cu surface and their movement is restricted. Fig. 5(b) illustrates the difference in hydrogen diffusion properties between Pt and Pd. The average hydrogen displacement on the Pt surface is around 6.5–7.0 Å ps−1, which is significantly larger than that on Pd, at approximately 5.0 Å s−1. This suggests that hydrogen diffuses more easily on the Pt surface than on Pd, affecting the differences in accessibility to oxygen toward subsequent reactions.
The analysis of dynamic diffusion showed that Pt retains O2 on the surface with moderate strength while also facilitating the diffusion of dissociated hydrogen. In contrast, although Pd can adsorb both O2 and dissociated hydrogen, H diffusion on the Pd surface was more restricted. For the development of catalysts with performance comparable to Pt, a key strategy may involve weakening Pd's O2 adsorption ability and enhancing its H diffusion properties through alloying, thereby creating an environment similar to that of Pt.
3.4 Comparisons with DFT calculations
Finally, we briefly compared the validity of the UNNP with DFT and experimental values. The surface slab model was used to investigate the H and O adsorption structures and binding energy with the metal.The bulk structures of Pd, Pt, Cu, and Au belong to the Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group and thus adopt a face-centered cubic (FCC) crystal structure. In contrast, Ag belongs to the P63/mmc space group, corresponding to a hexagonal close-packed (HCP) crystal structure at room temperature.
m space group and thus adopt a face-centered cubic (FCC) crystal structure. In contrast, Ag belongs to the P63/mmc space group, corresponding to a hexagonal close-packed (HCP) crystal structure at room temperature.
For the crystal surfaces, the (111) surface was used for FCC structures, while the (0001) surface was used for the HCP structure. Among various adsorbates, we focus on atomic H and O on the Pt, Pd, Cu, Au, and Ag surfaces. The optimized slab structures are summarized in Fig. 6. The constructed surface slabs had a (2 × 2) surface area with four layers, totaling 16 atoms. A vacuum gap of 12 Å was added between vertically repeating slabs to ensure sufficient separation between periodic cells and minimize interactions.
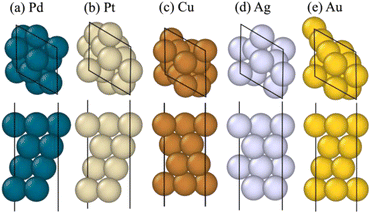 | ||
| Fig. 6 Surface slab models of (a) Pt, (b) Pd, (c) Cu, (d) Ag, and (e) Ag generated from the single crystals—upper: top view; lower: side view. | ||
| Ebind = (Eslab + Eadsorbent,isol) − E(slab+adsorbent) | (1) |
Binding energy is a key parameter for understanding the interaction between adsorbates and the surface, providing insights into adsorption strength and surface reactivity. In general, higher binding energies (more positive values) indicate a stronger interaction between the adsorbent and the surface. However, if the binding is too strong, it may hinder subsequent reaction steps—an outcome described by the Sabatier principle.59 The UNNP-based binding energies are summarized in Table 2, together with the DFT results and experimental values. When comparing the binding energies of O and H on the slab surfaces of Pd, Pt, Cu, Ag, and Au, noticeable differences are likely observed in each metal's adsorption capability and reactivity. The additional comparison results with some other DFT studies are shown in Table S1.
| X | Site | Pd | Pt | Cu | Ag | Au | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| UNNP | DFT | UNNP | DFT | UNNP | DFT | UNNP | DFT | UNNP | DFT | ||
| a Ref. 60. b Ref. 61. c Ref. 62. d Ref. 63. e Ref. 64. f Ref. 65. g Ref. 50. h Ref. 66. | |||||||||||
| O | fcc | 412.96 | 401.38 | 379.19 | 367.61 | 448.66 | 445.76 | 345.42 | 343.49 | 254.72 | 258.58 |
| hcp | 407.17 | 380.15 | 367.61 | 330.94 | 442.87 | 434.18 | 340.59 | 333.84 | 246.04 | 242.18 | |
| Exptl. | n.d. | 357.00a | 443.83b | 331.91c | 234.46d | ||||||
| H | fcc | 279.81 | 274.98 | 274.02 | 257.62 | 245.07 | 241.21 | 210.34 | 212.27 | 200.69 | 203.58 |
| hcp | 279.81 | 268.23 | 274.98 | 254.72 | 243.14 | 241.21 | 208.41 | 211.30 | 197.79 | 201.65 | |
| Exptl. | 260.00e | 268.23f | 234.46g | 176.57 to 192.97h | n.d. | ||||||
 and H* on the Pt surface (Fig. 2(a, b)′ and S8).
and H* on the Pt surface (Fig. 2(a, b)′ and S8).
In contrast, Cu showed weaker interaction with H, with binding energies of 245.07 kJ mol−1 at the fcc site and 243.14 kJ mol−1 at the hcp site, both lower than those of Pd and Pt. Similarly, Ag and Au demonstrate inactivity towards H adsorption, as reflected by their even lower binding energies: 208.41 to 210.34 kJ mol−1 for Ag and 197.79 to 200.69 kJ mol−1 for Au. Consequently, H adsorption is more challenging on these metal surfaces than on Pd and Pt surfaces. For Ag, the binding energy may have been overestimated relative to the experimental values; however, the overall accuracy of the UNNP predictions for Cu, Ag, and Au was assessed to be high.
Overall, the UNNP-MD simulations effectively estimated the catalytic activity of transition metals with both high computational efficiency and accuracy, yielding results consistent with experimental observations. The adsorption and reaction behaviors observed across the five metal surfaces offer valuable insights into their catalytic roles in the H2/O2 recombination reaction. These results suggest that designing catalysts with optimal O2 adsorption properties and moderate H adsorption strength—allowing hydrogen to diffuse on the surface—could lead to efficient and cost-effective alternatives to Pt catalysts. One possible strategy to reduce Pt usage is to dilute Pt with relatively inert metals, such as Ag or Au, while maintaining the balanced interaction with H2/O2. In the case of Pd, which binds H strongly, introducing inert metals such as Au may weaken H adsorption and thereby enhance catalytic performance. The computational workflow and resulting insights offer a strategic foundation for alloying or surface engineering aimed at improving catalytic performance in H2/O2 recombination reactions.
4 Conclusions
This study simulated the dynamic adsorption process and their subsequent reactions in H2/O2 recombination at finite temperature, which is one of the key processes for ensuring hydrogen safety. Specifically, we examined the reaction activity on Pd, Pt, Cu, Ag, and Au on metal nanocluster surfaces, using universal neural network potential (UNNP)-based molecular dynamics (MD) simulations.The UNNP approach successfully reproduced the adsorption energies of hydrogen and oxygen on each metal slab and its structure, with results showing good agreement with DFT results. Accelerated UNNP-MD simulations reaffirmed Pt as the most active catalyst surface for this reaction. These findings demonstrate that the UNNP approach is a practical and efficient tool for systematically evaluating catalytic activity in the H2/O2 recombination reaction.
Moreover, the simulations provided mechanistic insight, revealing that the catalytic activity of Pt is strongly related to the moderate-strength 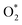 adsorption as the initial step and the ease of hydrogen diffusion across the surface. In contrast, Pd captured both of these species more strongly than Pt, limiting their catalytic activity. These results highlight the importance of achieving a balance between adsorption strength and diffusion dynamics to optimize catalytic performance. Pt's exceptional performance enhances its role as a benchmark catalyst, while the limitations observed in Cu, Pd, and Ag suggest that performance enhancements may be achieved through alloying or surface engineering. Through specific alloying, maintaining or promoting selective
adsorption as the initial step and the ease of hydrogen diffusion across the surface. In contrast, Pd captured both of these species more strongly than Pt, limiting their catalytic activity. These results highlight the importance of achieving a balance between adsorption strength and diffusion dynamics to optimize catalytic performance. Pt's exceptional performance enhances its role as a benchmark catalyst, while the limitations observed in Cu, Pd, and Ag suggest that performance enhancements may be achieved through alloying or surface engineering. Through specific alloying, maintaining or promoting selective 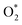 adsorption and hydrogen diffusion dynamics are expected to be important in reducing the practical cost of Pt. This exciting discovery has been obtained through dynamical H2/O2 recombination reactions at finite temperature, which is typically not achievable through DFT calculations based on single-molecule adsorption or predefined reaction models.
adsorption and hydrogen diffusion dynamics are expected to be important in reducing the practical cost of Pt. This exciting discovery has been obtained through dynamical H2/O2 recombination reactions at finite temperature, which is typically not achievable through DFT calculations based on single-molecule adsorption or predefined reaction models.
In summary, our approach has revealed the key factors required for the H2/O2 recombination reaction catalyst, referring to Pt's exceptional performance. Our in silico catalyst screening studies are ongoing, and will be reported in future publications.
Author contributions
Y. Tateishi: conceptualization, methodology, computations, formal analysis, data curation, software development, visualization, validation, writing – original draft; L. M. Botha: DFT calculations, formal analysis, data curation, validation, writing – review & editing; A. E. Kozhukhova: writing – review & editing; M. Sugimoto: conceptualization, methodology, funding acquisition, project administration, supervision, writing – review & editing; K.-i. Aika: funding acquisition, project administration, resources, supervision, writing – review & editing; D. G. Bessarabov: funding acquisition, project administration, resources, supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The detailed computational methods and supporting data are provided in the supplementary information (SI) and available from the present authors upon reasonable request. The scripts of simulation and data analysis are disclosed at [https://github.com/sugimoto-laboratory/h2o2_recombination/].Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc04712a. Trajectories in the MD simulations are available as MOV files.
Acknowledgements
This work was supported by JST/JICA, SATREPS (Science and Technology Research Partnership for Sustainable Development), “Development of new ammonia synthesis system using renewable energy and hydrogen” (JPMJSA2104) and by JST, BOOST (JPMJBS2408). In carrying out this research, we were greatly assisted by the staff of the HySA Infrastructure Center of Competence at North-West University (NWU), South Africa, and by JICA staff. We would also like to acknowledge the Centre for High-Performance Computing (CHPC) facility of South Africa in the completion of this work.References
- J. Kim, J. Yu, S. Lee, A. Tahmasebi, C. H. Jeon and J. Lucas, Advances in catalytic hydrogen combustion research: catalysts, mechanism, kinetics, and reactor designs, Int. J. Hydrogen Energy, 2021, 46, 40073–40104 Search PubMed
.
- S. Aslam, S. Rani, K. Lal, M. Fatima, T. Hardwick, B. Shirinfar and N. Ahmed, Electrochemical hydrogen production: sustainable hydrogen economy, Green Chem., 2023, 25, 9543–9573 RSC
.
- S. Karim, N. Tanwar, S. Das, R. Ranjit, A. Banerjee, N. Gulafshan, A. Gupta, A. Kumar and A. Dutta, Shaping the Future of Green Hydrogen Production: Overcoming Conventional Challenges with Molecular Catalysts, Immobilization, and Scalable Electrolyzers, ACS Catal., 2025, 15, 1073–1096 CrossRef CAS
.
- S. Safari, F. Esmaeilion, A. Rabanian, D. H. Jamali, S. Negi, S. Hoseinzadeh, F. Sayedin, S. S. Bhoglla, M. El Haj Assad, B. Das, M. A. Ehyaei, A. Ahmadi, M. Soltani and H. Afshari, Sustainable hydrogen production through water splitting: a comprehensive review, Environ. Dev. Sustain., 2025, 27, 17887–17926 CrossRef
.
- A. E. Kozhukhova, S. P. du Preez and D. G. Bessarabov, Catalytic Hydrogen Combustion for Domestic and Safety Applications: A Critical Review of Catalyst Materials and Technologies, Energies, 2021, 14, 4897 CrossRef CAS
.
- P. Yang, T. Wang, Y. Sheng, Y. Yu, R. Li, B. Su, F. Cheng, J. Qu, J. Deng and Z. Luo, Recent advances in hydrogen process safety: Deflagration behaviors and explosion mitigation strategies, Process Saf. Environ. Prot., 2024, 188, 303–316 CrossRef CAS
.
- A. A. Malakhov, A. V. Avdeenkov, M. H. Du Toit and D. G. Bessarabov, CFD simulation and experimental study of a hydrogen leak in a semi-closed space with the purpose of risk mitigation, Int. J. Hydrogen Energy, 2020, 45, 9231–9240 CrossRef CAS
.
- J. Lee, S. Cho, C. Park, H. Cho and I. Moon, Numerical analysis of hydrogen ventilation in a confined facility with various opening sizes, positions and leak quantities, Comput. Aided Process Eng., 2017, 40, 559–564 CAS
.
- Z. Wang, X. Gou, Q. Meng and H. Zhang, Effects of water sprays on hydrogen autoignition in heated air, Process Saf. Environ. Prot., 2024, 192, 915–925 CrossRef CAS
.
- L. J. Yuan, Z. C. Zhao, W. Q. Wang, Y. F. Wang and Y. J. Liu, Review of Catalysts, Substrates, and Fabrication Methods in Catalytic Hydrogen Combustion with Further Challenges and Applications, Energy Fuels, 2024, 38, 4881–4903 CrossRef CAS
.
- A. E. Kozhukhova, S. P. Du Preez and D. Bessarabov, Preparation of Highly Active and Thermally Conductive Platinum Nano-particle/Ce–Zr–Y Mixed Oxide/AAO Washcoat Catalyst for Catalytic Hydrogen Combustion Technologies, ACS Appl. Nano Mater., 2022, 5, 8161–8174 CrossRef CAS
.
- M. Klauck, E.-A. Reinecke, S. Kelm, N. Meynet, A. Bentaïb and H. J. Allelein, Passive auto-catalytic recombiners operation in the presence of hydrogen and carbon monoxide: Experimental study and model development, Nucl. Eng. Des., 2014, 266, 137–147 CrossRef CAS
.
- M. Klauck, E.-A. Reinecke and H.-J. Allelein, Effect of par deactivation by carbon monoxide in the late phase of a severe accident, Ann. Nucl. Energy, 2021, 151, 107887 CrossRef CAS
.
- K. Inagawa, D. Matsumura, M. Taniguchi, S. Uegaki, T. Nakayama, J. Urano, T. Aotani and H. Tanaka, Development of Hydrogen Oxidation Reaction Catalysts to Overcome CO Poisoning and Elucidation of Reaction Mechanism, J. Phys. Chem. C, 2023, 127, 11542–11549 CrossRef CAS
.
- I. A. Kirillov, Passive Autocatalytic Recombiners as Explosion Prevention and Mitigation Tool in Hydrogen Energy and Transport
Infrastructures, Chem. Eng. Trans., 2022, 90, 127–132 Search PubMed
.
-
P. Broeckerhoff, W. V. Lensa and E. A. Reinecke, Irregular thickness catalytic hydrogen recombination panel for water-cooled nuclear reactor minimizes the risk of fire and explosion, Ger. Pat., DE19852953C1, 2000 Search PubMed
.
- X. Gao, Y. Chen, Y. Wang, L. Zhao, X. Zhao, J. Du, H. Wu and A. Chen, Next-Generation Green Hydrogen: Progress and Perspective from Electricity, Catalyst to Electrolyte in Electrocatalytic Water Splitting, Nano–Micro Lett., 2024, 16, 237 CrossRef PubMed
.
- M. Awad, A. Said, M. H. Saad, A. Farouk, M. M. Mahmoud, M. S. Alshammari, M. L. Alghaythi, S. H. E. Abdel Aleem, A. Y. Abdelaziz and A. I. Omar, A review of water electrolysis for green hydrogen generation considering PV/wind/hybrid/hydropower/geo-thermal/tidal and wave/biogas energy systems, economic analysis, and its application, Alex. Eng. J., 2024, 87, 213–239 Search PubMed
.
- J. Kim, S. Hong, K. H. Park, J. H. Kim and J. Y. Oh, Experimental Study on Hydrogen Recombination Characteristics of a Passive Autocatalytic Recombiner during Spray Operation, Hydrogen, 2022, 3, 197–217 CrossRef CAS
.
- K. C. Sandeep, R. Bhattacharyya, C. Warghat, K. Bhanja and S. Mohan, Experimental investigation on the kinetics of catalytic recombination of hydrogen with oxygen in air, Int. J. Hydrogen Energy, 2014, 39, 17906–17912 CrossRef CAS
.
- M. Haruta and H. Sano, Catalytic combustion of hydrogen I—Its role in hydrogen utilization system and screening of catalyst materials, Int. J. Hydrogen Energy, 1981, 6, 601–608 CrossRef CAS
.
- J. F. Kramer, S.-A. S. Reihani and G. S. Jackson, Low-Temperature Combustion of Hydrogen on Supported Pd Catalysis, Proc. Combust. Inst., 2002, 29, 989–996 Search PubMed
.
- A. E. Kozhukhova, S. P. Du Preez, I. Shuro and D. G. Bessarabov, Preparation of a Cartridge-Type Pt/Al2O3 Catalyst Using a Sputter Deposition Method for Catalytic Hydrogen Combustion, Energy Fuels, 2022, 36, 13911–13923 CrossRef CAS
.
- A. E. Kozhukhova, S. P. Du Preez, A. A. Malakhov and D. G. Bessarabov, A Thermally Conductive Pt/AAO Catalyst for Hydrogen Passive Autocatalytic Recombination, Catalysts, 2021, 11, 491 CrossRef CAS
.
- A. E. Kozhukhova, S. P. Du Preez and D. G. Bessarabov, Development of Pt–Co/Al2O3 bimetallic catalyst and its evaluation in catalytic hydrogen combustion reaction, Int. J. Hydrogen Energy, 2024, 51, 1079–1096 CrossRef CAS
.
- A. E. Kozhukhova, S. P. Du Preez and D. G. Bessarabov, Preparation of Pt/Ce–Zr–Y mixed oxide/anodized aluminium oxide catalysts for hydrogen passive autocatalytic recombina-tion, Int. J. Hydrogen Energy, 2022, 47, 12726–12738 Search PubMed
.
- S. P. du Preez, D. R. Jones, M. E. A. Warwick, A. Falch, P. T. Sekoai, C. Mota das Neves Quaresma, D. G. Bessarabov and C. W. Dunnill, Thermally stable Pt/Ti mesh catalyst for catalytic hydrogen combustion, Int. J. Hydrogen Energy, 2020, 45, 16851–16864 Search PubMed
.
- S. P. du Preez, D. R. Jones, D. G. Bessarabov, A. Falch, C. Mota das Neves Quaresma and C. W. Dunnill, Development of a Pt/stainless steel mesh catalyst and its application in catalytic hydrogen combustion, Int. J. Hydrogen Energy, 2019, 44, 27094–27106 Search PubMed
.
- L. M. Botha, C. N. M. Ouma, K. O. Obodo and D. Bessarabov, Hydrogen and oxygen recombination reaction on Pt–Ni and Pt–Co based alloys using density functional theory, Surf. Sci., 2023, 736, 122354 Search PubMed
.
- L. M. Botha, K. O. Obodo, C. N. M. Ouma, D. G. Bessarabov, D. L. Sharypin, P. S. Varyushin and E. I. Plastinina, Density functional theory studies of Pd and PtxPd1-x bimetallic catalysts for the H2/O2 recombination reaction, Comput. Mater. Sci., 2025, 246, 113451 Search PubMed
.
- D. Qi, X. Luo, Y. Yao, N. Qi, X. Lu, H. Chen and H. Shi, Computational Study of H2 Catalytic Combustion on Pd38 Cluster Model and Pd(111) Slab Model, Symmetry, 2022, 14, 1544 CrossRef CAS
.
- Y. Wang, H. Shao, C. Zhang, F. Liu, J. Zhao, S. Zhu, M. K. H. Leung and J. Hu, Molecular dynamics for electrocatalysis: Mechanism explanation and performance prediction, Energy Rev., 2023, 2, 100028 CrossRef
.
- H. Hosseini, C. J. Herring, C. F. Nwaokorie, G. A. Sulley and M. M. Montemore, Computational Design of Catalysts with Experimental Validation: Recent Successes, Effective Strategies, and Pitfalls, J. Phys. Chem. C, 2024, 128, 18144–18157 CrossRef CAS PubMed
.
- I. Batatia, D. P. Kovács, G. N. C. Simm, C. Ortner and G. Csányi, Higher Order Equivariant Message Passing Neural Networks for Fast and Accurate Force Fields, arXiv, 2022, arXiv:2206.07697, DOI:10.48550/arXiv.2206.07697.
- H. Yang, C. Hu, Y. Zhou, X. Liu, Y. Shi, J. Li, G. Li, Z. Chen, S. Chen, C. Zeni, M. Horton, R. Pinsler, A. Fowler, D. Zügner, T. Xie, J. Smith, L. Sun, Q. Wang, L. Kong, C. Liu, H. Hao and Z. Lu, MatterSim: A Deep Learning Atomistic Model Across Elements, Temperatures and Pressure, arXiv, 2024, arXiv:2405.04967, DOI:10.48550/arXiv.2405.04967.
- S. Takamoto, C. Shinagawa, D. Motoki, K. Nakago, W. Li, I. Kurata, T. Watanabe, Y. Yayama, H. Iriguchi, Y. Asano, T. Onodera, T. Ishii, T. Kudo, H. Ono, R. Sawada, R. Ishitani, M. Ong, T. Yamaguchi, T. Kataoka, A. Hayashi, N. Charoenphakdee and T. Ibuka, Towards universal neural network potential for material discovery applicable to arbitrary combination of 45 elements, Nat. Commun., 2022, 13, 2991 CrossRef CAS PubMed
.
- Y. Hinuma, Neural Network Potential Molecular Dynamics Simulations of (La,Ce,Pr,Nd)0.95(Mg,Zn,Pb,Cd,Ca,Sr,Ba)0.05F2.95, Phys. Chem. B, 2024, 128, 12171–12178 CrossRef CAS PubMed
.
- A. Taborosi, K. Aoki, N. Zettsu, M. Koyama and Y. Nagao, Molecular Dynamics Simulation of Polymer Electrolyte Membrane for Understanding Structure and Proton Conductivity at Various Hydration Levels Using Neural Network Potential, Macromolecules, 2025, 58, 3720–3727 CrossRef CAS
.
- S. Takamoto, D. Okanohara, Q. J. Li and J. Li, Towards universal neural network interatomic potential, J. Mater., 2023, 9, 447–454 Search PubMed
.
- A. Hjorth Larsen, J. Jørgen Mortensen, J. Blomqvist, I. E. Castelli, R. Christensen, M. Dułak, J. Friis, M. N. Groves, B. Hammer, C. Hargus, E. D. Hermes, P. C. Jennings, P. Bjerre Jensen, J. Kermode, J. R. Kitchin, E. Leonhard Kolsbjerg, J. Kubal, K. Kaasbjerg, S. Lysgaard, J. Bergmann Maronsson, T. Maxson, T. Olsen, L. Pastewka, A. Peterson, C. Rostgaard, J. SchiØtz, O. Schütt, M. Strange, K. S. Thygesen, T. Vegge, L. Vilhelmsen, M. Walter, Z. Zeng and K. W. Jacobsen, The atomic simulation environment - A Python library for working with atoms, J. Phys.: Condens. Matter, 2017, 29, 273002 CrossRef PubMed
.
- H. Wang, S. Zhou, K. D. Gilroy, Z. Cai and Y. Xia, Icosahedral nanocrystals of noble metals: Synthesis and applications, Nano Today, 2017, 15, 121–144 CrossRef CAS
.
- S. Nosé, A unified formulation of the constant temperature molecular dynamics methods, J. Chem. Phys., 1984, 81, 511–519 CrossRef
.
- W. G. Hoover, Canonical dynamics: Equilibrium phase-space distributions, Phys. Rev. A, 1985, 31, 1965–1967 Search PubMed
.
- G. Kresse and J. Furthmiiller, Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set, Comput. Mater. Sci., 1996, 6, 15–50 Search PubMed
.
- B. Cordero, V. Gómez, A. E. Platero-Prats, M. Revés, J. Echeverría, E. Cremades, F. Barragán and S. Alvarez, Covalent radii revisited, RSC Dalton Trans., 2008, 2832–2838 RSC
.
- A. Plauck, E. E. Stangland, J. A. Dumesic and M. Mavrikakis, Active sites and mechanisms for H2O2 decomposition over Pd catalysts, Proc. Natl. Acad. Sci. U. S. A., 2016, 113, 1973–1982 Search PubMed
.
- E. M. Dietze, L. Chen and H. Grönbeck, Surface steps dominate the water formation on Pd(111) surfaces, J. Chem. Phys., 2022, 156, 064701 CrossRef CAS PubMed
.
- T. Jacob, The Mechanism of Forming H2O from H2 and O2 over a Pt Catalyst via Direct Oxygen Reduction, Fuel Cells, 2006, 6, 159–181 CrossRef CAS
.
- J. Greeley and M. Mavrikakis, Surface and Subsurface Hydrogen: Adsorption Properties on Transition Metals and Near-Surface Alloys, J. Phys. Chem. B, 2005, 109, 3460–3471 CrossRef CAS PubMed
.
- L. Xu, J. Lin, Y. Bai and M. Mavrikakis, Atomic and Molecular Adsorption on Cu(111), Top. Catal., 2018, 61, 736–750 CrossRef CAS
.
- T. Pu, A. Setiawan, A. C. Foucher, M. Guo, J.-M. Jehng, M. Zhu, M. E. Ford, E. A. Stach, S. Rangarajan and I. E. Wachs, Revealing the Nature of Active Oxygen Species and Reaction Mechanism of Ethylene Epoxidation by Supported Ag/α-Al2O3 Catalysts, ACS Catal., 2024, 14, 406–417 CrossRef CAS PubMed
.
- L. Barrio, P. Liu, J. A. Rodríguez, J. M. Campos-Martín and J. L. G. Fierro, A density functional theory study of the dissociation of H2 on gold clusters: Importance of fluxionality and ensemble effects, J. Chem. Phys., 2006, 125, 164715 Search PubMed
.
- G. J. Hutchings, Heterogeneous Gold Catalysis, ACS Cent. Sci., 2018, 4, 1095–1101 CrossRef CAS PubMed
.
- S. Seraj, P. Kunal, H. Li, G. Henkelman, S. M. Humphrey and C. J. Werth, PdAu Alloy Nanoparticle Catalysts: Effective Candidates for Nitrite Reduction in Water, ACS Catal., 2017, 7, 3268–3276 CrossRef CAS
.
- Y. Santiago-Rodríguez, J. A. Herron, M. C. Curet-Arana and M. Mavrikakis, Atomic and molecular adsorption on Au(111), Surf. Sci., 2014, 627, 57–69 Search PubMed
.
- J. A. Herron, S. Tonelli and M. Mavrikakis, Atomic and molecular adsorption on Pd(111), Surf. Sci., 2012, 606, 1670–1679 CrossRef CAS
.
- B. W. J. Chen, D. Kirvassilis, Y. Bai and M. Mavrikakis, Atomic and Molecular Adsorption on Ag(111), J. Phys. Chem. C, 2019, 123, 7551–7566 CrossRef CAS
.
- D. C. Ford, Y. Xu and M. Mavrikakis, Atomic and molecular adsorption on Pt(111), Surf. Sci., 2005, 587, 159–174 CrossRef CAS
.
-
P. Sabatier, Catalysis in Organic Chemistry, Library Press, 1922 Search PubMed
.
- J. L. Gland, B. A. Sexton and G. B. Fisher, Oxygen interactions with the Pt(111) surface, Surf. Sci., 1980, 95, 587–602 Search PubMed
.
- R. Naumann d'Alnoncourt, B. Graf, X. Xia and M. Muhler, The back-titration of chemisorbed atomic oxygen on copper by carbon monoxide investigated by microcalorimetry and transient kinetics, J. Therm. Anal. Calorim., 2008, 91, 173–179 Search PubMed
.
- C. T. Campbell, Atomic and molecular oxygen adsorption on Ag(111), Surf. Sci., 1985, 157, 43–60 CrossRef CAS
.
- N. Saliba, D. H. Parker and B. E. Koel, Adsorption of oxygen on Au(111) by exposure to ozone, Surf. Sci., 1998, 410, 270–282 Search PubMed
.
- K. Christmann, Interaction of hydrogen with solid surfaces, Surf. Sci. Rep., 1988, 9, 1–163 CrossRef
.
- B. Poelsema, G. Mechtersheimer and G. Comsa, The interaction of hydrogen with platinum(s)−9(111) × (111) studied with helium beam diffraction, Surf. Sci., 1981, 111, 519–544 CrossRef CAS
.
- G. Lee, P. T. Sprunger, M. Okada, D. B. Poker, D. M. Zehner and E. W. Plummer, Chemisorption of hydrogen on the Ag(111) surface, J. Vac. Sci. Technol., A, 1994, 12, 2119–2123 Search PubMed
.
| This journal is © The Royal Society of Chemistry 2026 |