 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Homogeneous catalysis in liquid organic hydrogen carriers: advances, challenges, and future directions
Juliana Mana
Edor
 *a,
Gershon
Amenuvor
b,
Phillimon
Modisha
*a,
Gershon
Amenuvor
b,
Phillimon
Modisha
 a and
Dmitri
Bessarabov
a
a and
Dmitri
Bessarabov
a
aHySA Infrastructure Centre of Competence, Faculty of Engineering, North-West University, Private Bag X6001, Potchefstroom, 2531, South Africa. E-mail: 36967432@mynwu.ac.za
bDepartment of Chemistry, Kwame Nkrumah University of Science and Technology, Kumasi, Ghana
First published on 16th February 2026
Abstract
Liquid organic hydrogen carriers (LOHCs) offer a promising solution for safe, efficient, and reversible hydrogen storage and transport. While heterogeneous catalysts have traditionally dominated LOHC hydrogenation and dehydrogenation, homogeneous catalysts present distinct advantages, including high selectivity, tunable active sites, and operation under milder conditions. This review systematically examines recent advances in the design and application of homogeneous catalysts for the hydrogenation and dehydrogenation of both homocyclic LOHCs, such as benzene, toluene, and naphthalene derivatives, and N-heterocyclic LOHCs, including quinolines, carbazoles, and indoles. Emphasis is placed on structure–activity relationships, mechanistic insights, and emerging trends, with a focus on catalysts based on both noble and earth-abundant metals. Beyond catalyst performance, we address key challenges in homogeneous catalyst recovery, recycling, and process integration, all of which are critical for industrial scalability. By identifying current gaps in the literature and outlining future research priorities, this work aims to guide the development of efficient, reversible, and economically viable homogeneous catalytic systems for next-generation hydrogen storage technologies.
1. Introduction
Hydrogen is valued for its high gravimetric energy density and is a promising candidate for clean and sustainable energy. It can be generated from water using renewable energy sources such as solar or wind power in regions with abundant energy and then transported to areas lacking sufficient energy generation. However, hydrogen's low energy density by volume in the gaseous state remains a major challenge regarding its storage and transportation.1Researchers have been investigating various hydrogen storage options to overcome these challenges.2–7 Options include the use of inorganic chemical hydrides, metal–organic frameworks, and metal hydrides to develop efficient and safe storage solutions that facilitate the transportation of hydrogen from energy-rich areas to places where it can be used effectively, thereby promoting a sustainable hydrogen economy.8
Due to their numerous advantages, liquid organic hydrogen carriers (LOHCs) have gained attention as a highly promising solution for hydrogen storage/release and transportation technology.9–12 LOHC systems consist of organic compounds that ideally allow for the bidirectional storage/release of hydrogen through catalytic (de)hydrogenation approaches while retaining their molecular integrity across numerous storage cycles. To be technically viable, LOHCs should also possess a viable range of chemical and physical characteristics, which include low melting points, thermal stability, advantageous (de)hydrogenation kinetics and thermodynamics, broad large-scale accessibility, and low toxicity.13
Since their conceptual introduction in the 1970s, with systems such as the toluene–methylcyclohexane pair, LOHCs have demonstrated practical applications, including large-scale demonstrations like Chiyoda Corporation's SPERA Hydrogen™ technology.14 Benzyltoluene (BT), developed through collaboration between FAU Erlangen–Nürnberg and adopted by Hydrogenious Technologies, also exemplifies a commercially viable LOHC owing to its high hydrogen storage capacity (HSC) and stability.15 Such successes highlight their potential for facilitating hydrogen transport over long distances.1,14,15 However, challenges remain, including concerns about toxicity, high energy demand for dehydrogenation, and the need for further advancements in catalyst efficiency.16 This review provides an overview of recent progress in homogeneous catalysis for the (de)hydrogenation of LOHCs, with particular emphasis on homocyclic and N-heterocyclic systems. Homogeneous catalysts, particularly those based on platinum group metals (PGMs) like Ru, Ir, and Rh, and earth-abundant metals like Co and Ni, offer high selectivity and activity under mild conditions for the (de)hydrogenation of certain LOHCs. Even though challenges such as catalyst separation, high costs, and limited scalability hinder industrial adoption, their high selectivity and low temperature requirements make them suitable for systems that operate at low temperatures. Homogeneous catalysts often remain dissolved in the LOHC, requiring energy-intensive separation methods like distillation. However, advanced techniques such as biphasic systems and nanofiltration, discussed in section 6, provide less energy-demanding alternatives. While several reviews have covered heterogeneous catalysts in LOHC systems,17,18 fewer have explored the nuances of homogeneous catalysis.19–21 This review uniquely examines the role of ligand design, metal–ligand cooperativity, and precious and non-precious metal catalysts in improving catalytic efficiency for LOHC applications. Rather than presenting a full techno-economic analysis, this review also discusses key techno-economic descriptors that govern the practical deployment of aromatic and heteroaromatic LOHC systems, with emphasis on catalyst performance, operating conditions, and system-level constraints. In addition, some major industrial applications of homogeneous catalysis and various strategies for homogeneous catalyst recovery and reuse are discussed briefly. By analyzing the advances in catalyst design, mechanistic insights, and the sustainability of LOHC systems, we aim to provide an overview that is comprehensive in the field while identifying challenges and future directions for research.
2. Overview of LOHC systems
While LOHCs have proven effective for hydrogen storage, understanding the differences between the various systems is critical for optimizing their performance. Several organic compounds have the potential to be used as LOHCs. These include simple compounds such as formic acid (FA), methanol, liquefied ammonia, and amino alcohols.22 In addition, LOHC media based on functional groups such as amides and imides, often operating via metal–ligand cooperative pincer catalysts, have been reported and are discussed extensively in the literature.23,24 These systems generally operate under comparatively mild temperature conditions.Other LOHC candidates are based on homocyclic and heterocyclic aromatic compounds, such as methylcyclohexane, decalin, perhydrocarbazole, and indoline, which typically require higher operating temperatures for hydrogen release. For LOHC-based hydrogen storage systems intended for mobile or vehicle-integrated applications, the operating temperature of the hydrogen release step represents a critical design constraint. Many vehicular platforms cannot accommodate the high temperatures (>250–300 °C) required for the dehydrogenation of several established aromatic LOHCs due to limitations associated with heat management, system complexity, start-up time, and catalyst durability. Examples include perhydrobenzyltoluene, employed by Hydrogenious Technologies,15 which operates under high dehydrogenation temperatures. Consequently, non-aromatic LOHC media that enable hydrogen release at lower temperatures (<200 °C) have attracted increasing attention for temperature-sensitive applications.25,26 These considerations highlight the importance of matching LOHC thermodynamics and kinetics with application-specific temperature constraints, particularly for mobile systems where high-temperature operation is impractical.
However, the present review focuses specifically on aromatic homocyclic and heterocyclic LOHC systems, which constitute a distinct class of hydrogen carriers with unique hydrogenation–dehydrogenation mechanisms, thermodynamic profiles, and catalyst requirements. The following sections, therefore, examines these aromatic systems and their key properties.
2.1. Homocyclic LOHCS
Homocyclic hydrocarbons are considered the simplest prospects for LOHCs due to the smooth transformation between hydrogenated alicyclic and dehydrogenated aromatic compounds.27,28 Systems such as cyclohexane–benzene,29–31 methylcyclohexane–toluene,32–34 and decalin–naphthalene35–38 pairs have been studied as examples because of their wide industrial prevalence, commercial accessibility, and relatively low toxicity. In recent times, systems utilizing diphenylmethane,39,40 BT,41–43 dibenzyltoluene (DBT),10,11,44,45 and biphenyl46–48 have been examined as well. The primary benefit of using cyclic hydrocarbons as LOHCs is their ability to supply COx-free hydrogen. Another notable feature is their high hydrogen weight-specific energy density, ranging from 6–8 wt%. Additionally, due to their low melting and high boiling points, they are well-suited for the storage and transport of hydrogen from centralized production sites to refuelling facilities. A low melting point and a high boiling point are essential for a potential LOHC to ensure the substance retains its liquid state across a broad temperature range. This facilitates convenient hydrogen storage and transport by preventing solidification at low temperatures and excessive vaporization at high temperatures, ensuring both practicality and safety.49Table 1 illustrates the properties of various homocyclic compounds as potential LOHCs, while Fig. 1 shows the structures of selected homocyclic LOHCs.| Properties of selected homocyclic LOHC pairs | ||||||
|---|---|---|---|---|---|---|
| Homocyclic aromatic LOHC system | State | Melting point (°C) | Boiling point (°C) | Enthalpy (kJ mol−1 H2) | Hydrogen storage capacity (wt%) | References |
| Benzene/cyclohexane | L/L | 5.5/ | 80 | 54.0 | 7.2 | 16, 41, 52 |
| Toluene/methylcyclohexane | L/L | −95/−127 | 111/101 | 57.5 | 6.2 | 16, 31, 41, 52 |
| Naphthalene/decalin | S/L | 80 | 218 | 61.9 | 7.3 | 16, 41, 20 |
| Dibenzyltoluene/perhydrodibenzyltoluene | L/L | −39/−34 | 390 | 61.5 | 6.2 | 20, 53 |
| Benzyltoluene/perhydrobenzyltoluene | L/L | −30 | 280 | 64 | 6.25 | 20 |
| Diphenylmethane/dicyclohexylmethane | L/L | 26/−19 | 265/248–250 | — | 6.7 | 31 |
| Biphenyl/bicyclohexyl | S/L | 69–71/3–4 | 254–255/238 | — | 7.3 | 31, 52 |
2.2. Heterocyclic LOHCs
The dehydrogenation of cyclic hydrocarbons to form homocyclic aromatics is an attractive process for hydrogen release but faces limitations due to its inherent endothermic nature. This reaction typically requires high dehydrogenation enthalpies (∼64–69 kJ mol−1 of H2) compared to the exothermic and readily achievable hydrogenation of aromatic hydrocarbons.50 This difference in energy requirements significantly impacts the feasibility of using homogeneous catalysts, as they can operate at relatively low temperatures while overcoming the high energy barrier.28 Meanwhile, introducing heteroatoms into the aromatic ring system offers a potential avenue for overcoming the limitations of dehydrogenation.Studies by Pez and coworkers51 suggest that incorporating heteroatoms results in a considerable loss of aromaticity upon dehydrogenation, thereby lowering the enthalpy of dehydrogenation. This opens doors for exploring dehydrogenation reactions with heterocyclic substrates using homogeneous catalysts. Following this, research interest in the catalyzed (de)hydrogenation of N-heterocycles was focused on molecules such as carbazoles, quinolines, and indoles.20 Their advantages include the ease of reversibility, easier H2 release than from alicyclics, favorable kinetic and thermodynamic properties, and low vapor pressure. Furthermore, they contain only the widely available elements C, H, and N. Fig. 2 shows the structures of selected heterocyclic LOHCs, and Table 2 presents the properties of various heterocyclic compounds useful as potential LOHCs.
| Properties | ||||||
|---|---|---|---|---|---|---|
| N-Heterocyclic LOHC system | State | Melting point (°C) | Boiling point (°C) | Enthalpy (kJ mol−1 H2) | Hydrogen storage capacity (wt%) | References |
| Perhydro-N-ethylcarbazole/N-ethylcarbazole | L/S | −85/69 | 261/355 | 54.0 | 5.80 | 16, 52, 54 |
| N-Phenylcarbazole | 91–93 | 57.5 | 6.94 | 20 | ||
| Decahydroquinoline/quinoline | L/L | 37/−15 | 214/252 | 61.9 | 7.18 | 54 |
| Tetrahydroquinoline/quinoline | L/L | 9–14/−15 | 249/252 | 61.9 | 3 | 18 |
| Decahydroquinaldine/quinaldine | L/L | −23/−2 | 232/271 | 64 | 6.58 | 54 |
| 1-Methylperhydroindole/1-methylindole | L/S | −25/−20 | 230/239 | 51.9 | 5.76 | 54, 61 |
| 2,6-Dimethyldecahydro-1,5-naphthyridine/2,6-Dimethyl-1,5-naphthyridine | S/S | — | — | — | 5.94 | 52 |
| Pyrazine/piperazine | S/S | 52/106 | 115/146 | — | 5.3 | 144 |
Both homocyclic and heterocyclic LOHC systems rely heavily on efficient catalysis to facilitate reversible hydrogen storage. Homogeneous catalysis is an emerging and powerful approach for enhancing the performance of these systems, as discussed in the sections that follow.
3. Advances in homogenously catalyzed de(hydrogenation) of homocyclic LOHCs
Aromatic hydrogenation typically requires more stringent conditions than in the case of nonaromatic unsaturated bonds. While heterogeneous catalysts have proven effective for LOHC-relevant aromatic hydrogenation systems, homogeneous metal complex catalysts remain relatively less studied, specifically in the field of LOHC technology for hydrogen storage. Heterogeneous catalysts benefit from increased surface area and support properties, such as Lewis acidity or Brønsted basicity. Additionally, heterogeneous catalysis is advantageous due to its ease of separation, which reduces operating costs and makes it widely used in industry. However, its drawbacks include limited activity and selectivity.55 In contrast, homogeneous catalysts can be optimized through careful ligand design to enhance their catalytic activity.21,30 Homogeneous catalysis, though less common in industrial applications due to the challenging and costly catalyst separation, offers superior selectivity and higher activity and eliminates mass transfer limitations, potentially enabling lower operating temperatures.553.1. Hydrogenation of homocyclic aromatics with catalysts based on platinum group metals
Boxwell et al.29 in 2002 designed a system using catalyst 1 ([Ru(p-cymene)(η2-TRIPHOS)Cl][PF6]), where TRIPHOS = 1,1,1-tris(diphenylphosphinomethyl)ethane illustrated in Fig. 3, for hydrogenating arenes in dichloromethane (DCM) and ionic liquid. The system is effective in catalyzing the hydrogenation of toluene and benzene (90 °C, 61 bar H2 pressure, 1 h) to afford the corresponding cyclohexane, with turnover frequency (TOF) values of 205 h−1 and 476 h−1, respectively. The catalyst showed high activity in both ionic liquids and DCM, with significantly higher activity in ionic liquids. Unlike in DCM, where it decomposes, no considerable activity loss was observed after five runs in ionic liquids. Several allylbenzenes were also hydrogenated with this system. Particularly, the availability of a free phosphine is relevant for selectivity towards arene hydrogenation; otherwise, the hydrogenation does not occur.Rojas' group56 devised the hydrogenation of various aromatics with a homogeneous Ru-based catalyst. The catalyst [Ru(η5-C5H5)Cl(TPPDS)2] (2) (TPPDS: P(C6H5)(C6H4SO3)2) reduced toluene, benzene, and m-xylene in a biphasic (n-heptane/water) medium to the corresponding cyclohexane analogues. Toluene, m-xylene, and benzene, generated products in yields of 80%, 25%, and 43%, respectively, after 4 h reaction time (105 °C, 96 bar H2, and substrate/catalyst ratio 600![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The activity increased with H2 pressure, temperature, pH, and ionic strength. Furthermore, the aqueous catalytic system can undergo several cycles with little reduction in activity.56 Water-soluble Ru complexes 3–5 were analyzed in the hydrogenation of arenes. Complexes 3 and 5 exhibited similar TOF values, with 3 being more active than 5 while complex 4 was significantly more active than complexes 3 and 5. The presence of mercury had little impact on the activity of 3 and 5. However, adding mercury to the reaction medium with 4 resulted in a considerable reduction in turnover number, indicating the decomposition of 4 to form catalytically active colloidal species.57 Arun et al.30 used 3-hydroxyquinoline-2-carboxalidene-4-aminoantipyrine (L)-coordinated Ru complex (6) in the hydrogenation of benzene to produce a mixture of cyclohexadiene and cyclohexane in 18% and 82% yields, respectively (Scheme 1). A TOF of 5372 h−1 was recorded in reducing benzene (80 °C, 50 bar H2 pressure, 3.64 × 10−6 mol catalyst, and 0.34 mol benzene). Mercury drop tests confirmed a homogeneously catalyzed process. Under identical reaction conditions, the Ru complex (6) gave a superior conversion with a TOF of 5372 h−1, while the nickel catalyst (7) gave a lower conversion with a TOF of 1718 h−1.
1). The activity increased with H2 pressure, temperature, pH, and ionic strength. Furthermore, the aqueous catalytic system can undergo several cycles with little reduction in activity.56 Water-soluble Ru complexes 3–5 were analyzed in the hydrogenation of arenes. Complexes 3 and 5 exhibited similar TOF values, with 3 being more active than 5 while complex 4 was significantly more active than complexes 3 and 5. The presence of mercury had little impact on the activity of 3 and 5. However, adding mercury to the reaction medium with 4 resulted in a considerable reduction in turnover number, indicating the decomposition of 4 to form catalytically active colloidal species.57 Arun et al.30 used 3-hydroxyquinoline-2-carboxalidene-4-aminoantipyrine (L)-coordinated Ru complex (6) in the hydrogenation of benzene to produce a mixture of cyclohexadiene and cyclohexane in 18% and 82% yields, respectively (Scheme 1). A TOF of 5372 h−1 was recorded in reducing benzene (80 °C, 50 bar H2 pressure, 3.64 × 10−6 mol catalyst, and 0.34 mol benzene). Mercury drop tests confirmed a homogeneously catalyzed process. Under identical reaction conditions, the Ru complex (6) gave a superior conversion with a TOF of 5372 h−1, while the nickel catalyst (7) gave a lower conversion with a TOF of 1718 h−1.
 | ||
| Scheme 1 Benzene hydrogenation catalyzed by Schiff base-coordinated Ru and Ni complexes.21,30 | ||
In 2010, Vangelis et al.31 showed that the Rh/TPPTS-catalyzed (TPPTS: triphenylphosphine trisulfonate) hydrogenation of benzene exhibits remarkably high catalytic performance (TOF > 204![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h−1), producing cyclohexane quantitatively in an organic/aqueous biphasic system (130 °C, 80 bar H2 pressure, 5 min). The catalyst was readily recycled by employing phase separation techniques. Recycling experiments showed consistently high catalytic activities over five consecutive runs. Borowski et al.58 successfully partially hydrogenated anthracene under mild conditions (3 bar H2 pressure, 80 °C) using the homogeneous bis(dihydrogen) Ru complex 8. The reaction selectively hydrogenated the terminal aromatic rings, yielding a mixture of 4H-anthracene and 8H-anthracene. Hydrogenation of the first ring was significantly faster than the second. However, prolonged reaction times resulted in catalyst decomposition and metal precipitation, preventing complete conversion to the 8H-anthracene. Chatterjee et al.59 utilized a dinuclear monohydrido-bridged ligand complexed to Ru[{(η6-p-cymene)RuCl}2(μ-H–μ-Cl)] (9), demonstrating exceptional efficiency and selectivity in the hydrogenation of diverse arenes and heteroarenes. The complete hydrogenation of various arenes (including benzene, toluene, naphthalene, and anthracene) to the generated cycloalkanes was achieved in high yields using 1 mol% catalyst loading (90 °C, 50 bar H2, 40 h). However, further analysis revealed that the arenes are hydrogenated by in situ-generated Ru NPs.
000 h−1), producing cyclohexane quantitatively in an organic/aqueous biphasic system (130 °C, 80 bar H2 pressure, 5 min). The catalyst was readily recycled by employing phase separation techniques. Recycling experiments showed consistently high catalytic activities over five consecutive runs. Borowski et al.58 successfully partially hydrogenated anthracene under mild conditions (3 bar H2 pressure, 80 °C) using the homogeneous bis(dihydrogen) Ru complex 8. The reaction selectively hydrogenated the terminal aromatic rings, yielding a mixture of 4H-anthracene and 8H-anthracene. Hydrogenation of the first ring was significantly faster than the second. However, prolonged reaction times resulted in catalyst decomposition and metal precipitation, preventing complete conversion to the 8H-anthracene. Chatterjee et al.59 utilized a dinuclear monohydrido-bridged ligand complexed to Ru[{(η6-p-cymene)RuCl}2(μ-H–μ-Cl)] (9), demonstrating exceptional efficiency and selectivity in the hydrogenation of diverse arenes and heteroarenes. The complete hydrogenation of various arenes (including benzene, toluene, naphthalene, and anthracene) to the generated cycloalkanes was achieved in high yields using 1 mol% catalyst loading (90 °C, 50 bar H2, 40 h). However, further analysis revealed that the arenes are hydrogenated by in situ-generated Ru NPs.
3.2. Catalysts based on earth-abundant metals
Muetterties and Hirsekorn60 in 1974, developed the tris(phosphite) cobalt allyl complex 11 (Fig. 4), employed in homogeneous arene hydrogenation. Under mild conditions, hydrogenation of arenes was observed, tolerating mesitylene, xylene, and naphthalene, and resulting in cis-cycloalkanes in all instances. At 25 °C, benzene hydrogenation to cyclohexane occurred at a slow rate, with approximately 20 catalyst cycles completed in 48 h. A comprehensive mechanistic analysis suggested that the cis selectivity stemmed from consecutive syn additions of a Co-allyl hydride over the arene's C–C bonds, followed by the reduction of the diene and olefin products, thus indicating homogeneous catalysis.61 Rothwell62 has shown that Nb and Ta complexes with sterically shielded alkoxide ligands are efficient precatalysts in the all-cis arene hydrogenations.The tantalum trihydride complex 12 could be separated and utilized directly, whereas the phosphine-free niobium counterpart was generally produced in situ through hydrogenolysis of the tribenzyl-ligated precursor.62
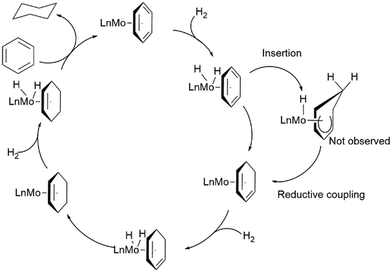
A Mo-catalyzed arene hydrogenation method was developed by Joannou et al.63 in 2018. The pyridine(diimine) Mo catalyst [(4-tBu-iPrPDI)Mo(CH2SiMe3)2] (iPrPDI: 2,6-(2,6-(C(CH3)2H)2C6H3N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CMe)2C5H3N) 13 was able to hydrogenate benzene and several substituted arenes, employing a 5 mol% catalyst loading at 60 °C, 4 bar of H2, and 24 h, demonstrating that the barrier to hydrogenating arenes can be greatly reduced by transition metal complexes. Then Hierlmeier et al.64 investigated Mo cyclohexadienyl complexes [(PIP)Mo(COD)] (PIP: phosphino(imino)pyridine, COD: 1,5-cyclooctadiene) 14–16 in the hydrogenation of benzene. Cyclohexane was produced using 0.18 mol% catalyst loading (at 23 °C, and 6.89 bar H2 pressure). Scheme 2 illustrates the suggested catalytic cycle. When using the iso-propyl-substituted variant 16, a turnover number (TON) of 56 was obtained. This was a significant improvement over the previously reported system [(4-tBu-iPrPDI)Mo(CH2SiMe3)2],63 which had a TON of 19 at 23 °C. The system also catalyzed the hydrogenation of toluene to form methylcyclohexane at 4 bar H2 pressure. The development of the above Mo catalysts demonstrates that, in homogeneous arene hydrogenation, an arene incorporation into the metal–hydride bond proceeds with a relatively minimal kinetic barrier when using Mo phosphino(imino)pyridine complexes.64 The first step begins with the η6-binding of the arene, after which the dihydrogen undergoes oxidative addition to the metal. Then, inserting the arene's C(sp2)–C(sp2) bond into a metal hydride bond results in the formation of a cyclohexadiene complex, followed by reductive elimination. Subsequent hydrogenations proceed similarly through oxidative addition, insertion, and reductive elimination steps to afford the cycloalkane. Furthermore, a similar approach, commencing with the dihydride Mo complex, is conceivable.64 Viereck et al.65 also demonstrated the hydrogenation of naphthalene derivatives using enantioenriched Mo complex 17, [4-tBu-(tBuOIP)Mo(COD)] (OIP: oxazoline imino(pyridine)). Results indicated that hydrogenating the various substrates (eight examples) produced the corresponding decalin in yields ranging between 45 and 95% with enantioselectivities up to 95% ee. Klabunde et al.66 reported that the η6-nickel(II) catalyst 18 reduces toluene to methylcyclohexane under high H2 pressure (101 bar) at 25 °C. However, the complex showed signs of decomposition, forming C6F5H and inactive Ni(0) species; hence, the observed TONs were limited to ≤10. A Ti(IV)–guanidine complex (19) was used in the hydrogenation of various poly and monocyclic aromatics. The substrates (toluene-d8, benzene-d6, anthracene, and naphthalene) were reduced to afford the corresponding cyclohexanes at 80 °C under very low H2 pressure. NMR data (1H, 2H, and 13C NMR) suggested that the Ti catalyst 19a was converted Ti catalyst 19b, which is the active species when H2 is introduced, as illustrated in Scheme 3.67
CMe)2C5H3N) 13 was able to hydrogenate benzene and several substituted arenes, employing a 5 mol% catalyst loading at 60 °C, 4 bar of H2, and 24 h, demonstrating that the barrier to hydrogenating arenes can be greatly reduced by transition metal complexes. Then Hierlmeier et al.64 investigated Mo cyclohexadienyl complexes [(PIP)Mo(COD)] (PIP: phosphino(imino)pyridine, COD: 1,5-cyclooctadiene) 14–16 in the hydrogenation of benzene. Cyclohexane was produced using 0.18 mol% catalyst loading (at 23 °C, and 6.89 bar H2 pressure). Scheme 2 illustrates the suggested catalytic cycle. When using the iso-propyl-substituted variant 16, a turnover number (TON) of 56 was obtained. This was a significant improvement over the previously reported system [(4-tBu-iPrPDI)Mo(CH2SiMe3)2],63 which had a TON of 19 at 23 °C. The system also catalyzed the hydrogenation of toluene to form methylcyclohexane at 4 bar H2 pressure. The development of the above Mo catalysts demonstrates that, in homogeneous arene hydrogenation, an arene incorporation into the metal–hydride bond proceeds with a relatively minimal kinetic barrier when using Mo phosphino(imino)pyridine complexes.64 The first step begins with the η6-binding of the arene, after which the dihydrogen undergoes oxidative addition to the metal. Then, inserting the arene's C(sp2)–C(sp2) bond into a metal hydride bond results in the formation of a cyclohexadiene complex, followed by reductive elimination. Subsequent hydrogenations proceed similarly through oxidative addition, insertion, and reductive elimination steps to afford the cycloalkane. Furthermore, a similar approach, commencing with the dihydride Mo complex, is conceivable.64 Viereck et al.65 also demonstrated the hydrogenation of naphthalene derivatives using enantioenriched Mo complex 17, [4-tBu-(tBuOIP)Mo(COD)] (OIP: oxazoline imino(pyridine)). Results indicated that hydrogenating the various substrates (eight examples) produced the corresponding decalin in yields ranging between 45 and 95% with enantioselectivities up to 95% ee. Klabunde et al.66 reported that the η6-nickel(II) catalyst 18 reduces toluene to methylcyclohexane under high H2 pressure (101 bar) at 25 °C. However, the complex showed signs of decomposition, forming C6F5H and inactive Ni(0) species; hence, the observed TONs were limited to ≤10. A Ti(IV)–guanidine complex (19) was used in the hydrogenation of various poly and monocyclic aromatics. The substrates (toluene-d8, benzene-d6, anthracene, and naphthalene) were reduced to afford the corresponding cyclohexanes at 80 °C under very low H2 pressure. NMR data (1H, 2H, and 13C NMR) suggested that the Ti catalyst 19a was converted Ti catalyst 19b, which is the active species when H2 is introduced, as illustrated in Scheme 3.67
 | ||
| Scheme 2 Proposed mechanism for the Mo-catalysed arene hydrogenation.64 | ||
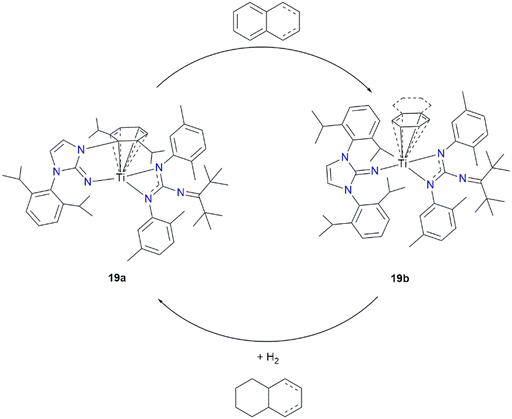 | ||
| Scheme 3 Interconversion between titanium catalysts 19a and 19b.67 | ||
In 2019, Zeng and coworkers68 reported the regiocontrolled polycyclic hydrocarbons hydrogenation with Cr and Co catalysts at ambient temperature for the first time. Using CrCl3 and MeMgBr with diamino ligands in THF, only one terminal benzene ring of anthracene was selectively hydrogenated at 50 bar H2 pressure. In contrast, when Co(acac)3 was used, both terminal benzene rings in the anthracene framework were hydrogenated when using isopropanol and MeMgBr at ambient temperature (RT) in THF, under 80 bar H2. The proposed mechanism involves low-valent metal species as active catalysts that undergo migratory insertion into the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond of anthracene, forming an intermediate that is subsequently hydrogenated, and the active species is regenerated.
C bond of anthracene, forming an intermediate that is subsequently hydrogenated, and the active species is regenerated.
Johnson et al.69 in 2025 reported benzene hydrogenation utilizing early transition metal catalyst precursors. Diverse precursors, including transition metal complexes of groups 4 and 5, were investigated for benzene hydrogenation. For group 4, homoleptic –CH2tBu complexes, [M(CH2tBu)4] (M = Ti, Zr, Hf), were selected, while group 5 catalysts were based on –CH2SiMe3 ligands, specifically [V(CH2SiMe3)3(THF)] and [M2(μ2-CSiMe3)2(CH2SiMe3)4] (M = Nb, Ta). In all cases, 1H NMR spectroscopy confirmed cyclohexane as the sole product after catalysis. The results illustrated that employing milder reaction conditions (120 °C, ca. 27 bar H2), groups 4 and 5 transition metal complexes are active catalysts in hydrogenating benzene. The most active catalysts were Nb- and Hf-based, achieving initial TOF values of 1055 and 1155 mol C6H6 mol M−1 h−1, respectively. Remarkably, under identical reaction conditions, these TOFs exceed those achieved with conventional heterogeneous catalysts, such as RANEY® Ni and 5 wt% Pd/C, thus highlighting the potential of group 4 and 5 metal complexes as effective alternatives for use in benzene hydrogenation.
3.3. Reversible hydrogenation of aromatic LOHCs using homogeneous catalysts
Ir pincer complex 10 was demonstrated in homogeneous catalysis by Jensen for hydrogen storage/release in aromatic hydrocarbons.70,71 Complex 10 showed activity for the reversible hydrogenation of the methylcyclohexane/toluene pair (Scheme 4). The oxidation reaction proceeded inside a reaction chamber fitted with a tubular membrane enabling selective H2 gas permeation at 190 °C. The hydrogenation also proceeded in the same flask at 10 bar H2 pressure and >100 °C. Table 3 provides a summary of catalysts discussed for the homogeneous hydrogenation of homocyclic LOHCs.| Catalyst | Substrate/product | Conv. (%) | Selectivity (%) | TON | TOF (h−1) |
|---|---|---|---|---|---|
| [Ru(p-cymene)(η2-TRIPHOS)Cl][PF6] | Benzene/cyclohexane | 476 | |||
| Toluene/methylcyclohexane | 205 | ||||
| [Ru(η5-C5H5)Cl(TPPDS)2] | Benzene/cyclohexane | 43 | |||
| Toluene/methylcyclohexane | 80 | ||||
| [Ru(η6-C10H14)(pta)Cl2] | Benzene/cyclohexane | 170 | |||
| Toluene/methylcyclohexane | 130 | ||||
| [Ru(η6-C10H14)(TPPTS)Cl2] | Benzene/cyclohexane | 488 | |||
| Toluene/methylcyclohexane | 365 | ||||
| [Ru(η6-C10H14)(pta)2Cl]+ | Benzene/cyclohexane | 150 | |||
| Toluene/methylcyclohexane | 129 | ||||
| [RuII(3-hydroxyquinoxaline-2-carboxalidene-4-aminoantipyrine)(Cl)(H2O)2]·H2O | Benzene/cyclohexane | 20.7 | 82 | — | 5372 |
| [RuH2(η2-H2)2{P(C6H11)3}2] | Anthracene | 100 | 4H anth = 99.3 | 0.3 | |
| 8H anth = 0.7 | 50 | ||||
| [{(η6-p-Cymene)RuCl}2(μ-H–μ-Cl)] | Benzene/cyclohexane | 90 | 150 | ||
| Toluene/methylcyclohexane | 90 | ||||
| Naphthalene/decalin | >99 | ||||
| Anthracene/tetradecahydroanthracene | 40 | ||||
| RhCl3·3H2O/TPPTS | Benzene/cyclohexane | 57 | 100 | 204![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
|
| [NiII(3-hydroxyquinoxaline-2-carboxalidene-4-aminoantipyrine)(Cl)(H2O)2]·H2O | Benzene/cyclohexane | 5.5 | 31 | 1718 | |
| (4-tBu-iPrPDI)Mo(CH2SiMe3)2 | Benzene/cyclohexane | 98 | 19 | ||
| [(PIP)Mo(COD)] | Benzene/cyclohexane | 56 | |||
| 4-tBu-(tBuOIP)Mo(COD) | Naphthalenes/decalins | 45–95% | |||
| [η6-Toluenebis-(pentafluorophenyl)nickel(II)] | Toluene/methylcyclohexane | ≤10 | |||
| [(η6-ImDippN)(xyketguan)Ti] | Benzene-d6/cyclohexane-d6 | 1 | 0.05 | ||
| Toluene-d8/methylcyclohexane-d8 | 1.8 | 0.09 | |||
| Naphthalene/tetralin | 4.4 | 0.22 | |||
| Anthracene/octahydroanthacene | 4.6 | 0.23 | |||
| [Ti(CH2tBu)4] | Benzene | 23 | 173 | ||
| [Zr(CH2tBu)4] | Benzene | 97 | 691 | ||
| [Hf(CH2tBu)4] | Benzene | 96 | 1155 | ||
| [V(CH2SiMe3)3(THF)] | Benzene | 0 | 0 | ||
| [Nb2(μ2-CSiMe3)2(CH2SiMe3)4] | Benzene | 98 | 1055 | ||
| [Ta2(μ2-CSiMe3)2(CH2SiMe3)4] | Benzene | 44 | 177 |
4. Advances in homogenously catalyzed (de)hydrogenation of N-heterocyclic compounds
4.1. Quinolines
The quinoline moiety (Fig. 5) is an attractive candidate for LOHCs. Quinolines are liquid at ambient conditions, with high boiling and melting points as outlined in Table 4.20,72 Quinolines are primarily derived from fossil oil processing, wood preservation, or can be synthetically produced from anilines.20 Quinoline derivatives are also found in many natural products, particularly quinoline-based alkaloids.73 Although the HSC of quinolines is very high, the high dehydrogenation enthalpy of 61.9 kJ mol−1,74 moderate toxicity (LD50 = 460 mg kg−1), and potential adverse effects on organisms across various trophic levels and food chains75 limit their widespread application in LOHC technology.20| Quinoline derivative | State | Melting point (°C) | Boiling point (°C) | HSC (wt%) |
|---|---|---|---|---|
| Quinoline | Liquid | −40 | 238 | 7.2 |
| 2-Methylquinoline | Liquid | −9 | 247.6 | 6.6 |
| Isoquinoline | Liquid | 27 | 243.3 | 7.2 |
4.1.1.1. Direct hydrogenation. Pressurized H2 gas is required for direct hydrogenation, necessitating complex high-pressure equipment. Despite this, direct hydrogenation offers benefits such as high efficiency, yielding products of high purity, and easy scalability for industrial use.75 Various earth-abundant metal complexes have been employed to hydrogenate quinolines and other N-heterocycles directly (Fig. 6). For instance, an efficient cobalt catalyst for hydrogenating a diverse group of compounds was designed by Duan et al.76 utilizing the moisture and air-stable, scalable complex (20), which is easily activated by potassium hydroxide. Quinolines bearing different groups were quantitatively hydrogenated effortlessly using 0.1 mol% catalyst loading and 10 mol% KOH in iPrOH under 30 bar H2 at 100 °C for 48 h. The half-sandwich [WCl(η5-Cp)(CO)3] complex (21) was explored for the homogeneous reduction of quinolines using pressurized H2 gas.77 Applying 2 mol% catalyst loading in iPrOH, under high H2 pressure (50 bar), afforded 94% tetrahydroquinoline (THQ) in 14 h. Bench-stable nickel complexes with metallated tripodal ligands (22) efficiently hydrogenated quinoline quantitatively to THQ with a catalyst loading of 2.5 mol% in toluene under 10 bar H2 at 30 °C for 15 h.78 Papa et al.79 reported room temperature N-heterocycle hydrogenation catalysis using NNP–Mn(I) pincer complex (23).
At 20 bar H2 pressure, catalyst loading of 2.5 mol%, and 10 mol% of KOtBu in THF, hydrogenating various quinolines resulted in excellent conversions. Relative to the earlier results reported by employing MnCO5Br as a catalyst, it was revealed that complex 23 was superior in hydrogenating quinolines with substituents at positions 5 and 8.79 Kumar et al.80 designed a bis-NHC-armed CNHC–Mn pincer catalyst (24), hydrogenating heteroarenes efficiently. In particular, quinoline was hydrogenated to 1,2,3,4-THQ in 97% yield using 2 mol% catalyst loading, 20 mol% KOtBu under 60 bar H2 pressure, and 120 °C for 12 h. The group also reported a mesoionic carbene–Mn(I) complex (25) in N-heteroarene hydrogenation. However, only 20% product yield was achieved when quinoline was reduced under the optimum reaction conditions.81
4.1.1.2. Mechanistic studies. In efforts to further understand the mechanism, Michaliszyn et al.78 employed DFT methods (B3LYP/6-31g*) with a focus on the heterolytic activation route of H2 as illustrated in Scheme 5. The initial intermediate, Ni-quinoline adduct (1Q), is 15.1 kcal·mol−1 more stable than the decoordinated complex (1□). Coordination of H2 to 1□ forms 1H2 (ΔG = 19.1 kcal mol−1 from 1Q) through a low-energy transition state, yielding a highly labile η2-H2 complex. Notably, η2-H2 coordination elongates the trans Ni–C bond by 0.037 Å, similar to the effect of quinoline coordination (0.045 Å). The oxidative addition of H2 to Ni(II) was ruled out due to a high activation barrier (ΔG‡ = 33.4 kcal mol−1). Instead, heterolytic H2 activation is facilitated by quinoline. The formation of the 1H2–4a adduct stabilizes 1H2 (ΔG = −3.1 kcal mol−1), followed by a low-energy transition state (ΔG‡ = 0.7 kcal mol−1), resembling frustrated Lewis pair behavior, where Ni acts as a Lewis acid and quinoline as a base. This results in exergonic H–H bond cleavage, weakening the Ni–C bond via trans effect (elongation 0.076 Å). Molecular orbital analysis reveals that H2 destabilizes the Ni–C bond in 1H2, with its LUMO (Ni dZ2) resembling that of 1NTf2, where NTf2 is weakly coordinating. The 1H2–4a adduct stabilizes the Ni–C bond through C–Ni–H interaction, showcasing the electronic flexibility of Ni. Following heterolytic cleavage, the protonated quinoline (4a(NH)) undergoes nucleophilic attack by Ni(II)–H at position 4 (ΔG‡ = 5.6 kcal mol−1; overall barrier = 19.6 kcal mol−1), yielding 4a(NH,4H). A rapid isomerization equilibrium with 4a(3H,4H) (ΔG‡ = 5.5 kcal·mol−1) aligns with previous reports and deuteration experiments.82 Finally, 4a(3H,4H) undergoes heterolytic H2 activation to form 4a(NH,3H,4H), followed by a barrierless Ni(II)–H attack at position 2, yielding the final product (5a).78
 | ||
| Scheme 5 Proposed catalytic cycle. Red values show initial quinoline hydrogenation yielding 4a(NH,4H) and isomerizing to 4a(3H,4H). Green values relate to the reduction of 4a(3H,4H) to 5a78 (image reproduced with permission from John Wiley & Sons, Inc., all rights reserved). | ||
4.1.1.3. Transfer hydrogenation. Transfer hydrogenation (TH) refers to the process of adding hydrogen to a molecule using an alternative source of hydrogen instead of molecular hydrogen, as it offers a practical and efficient way to produce various hydrogenated substances. This method serves as a useful alternative to DH. TH has recently attracted significant attention in hydrogenation research. The advantages of TH include (i) the absence of hazardous pressurized H2 gas and complex experimental setups, (ii) its availability and low cost, and the ease of handling the hydrogen donors, (iii) recyclability of the main byproduct, and (iv) ease of access to and the stability of the catalysts used.83 TH offers a straightforward and viable substitute for traditional hydrogenation methods and eliminates the need for high-pressure apparatus or gas handling.84
4.1.1.3.1. Using silanes as a hydrogen source. Bi and coworkers85 reported an effective silver catalyst in reducing electron-deficient N-heteroarenes and quinolines in aqueous medium with hydrosilanes, as illustrated in Scheme 6. They reported that in just 10 min, 2-methylquinoline was reduced to the 1,2,3,4-THQ derivative in 98% yield at RT. The system enhances quinoline reduction in the absence of both a base and a ligand and performs under modest conditions. Similarly, in 2022, the [Ir(COD)Cl]2-catalyzed TH of various quinolines was performed using hydrosilane as the hydrogen source in methanol without any external ligand or base. The system allowed for the scalable synthesis of the 1,2,3,4-THQ core moiety under mild conditions.86
4.1.1.3.2. Using FA as a source of hydrogen. In 1984, Watanabe et al.87 reported the TH of quinolines using dichlorotris(triphenylphosphine) Ru complex with FA acting as a hydrogen transfer agent. The reactions were achieved using stoichiometric amounts of FA at 180 °C. Quinoline and 2-methylquinoline were transformed to the respective THQ derivatives in yields of 95% and 93%, respectively. In 2022, Sun's group used in situ-generated Ir catalysts with chiral 2-pyridyl imidazoline ligands in an asymmetric TH of quinolines and other N-heteroaryl compounds. When a catalyst loading of 0.2 mol% with 2.5 equiv FA was utilized in water at RT, the hydrogenation of 2-methylquinoline resulted in 92% yield with 62% ee in 6 h.88 Maji and Choudhury,89 achieved the TH of quinolines with a Cp*Ir half-sandwich complex bearing an uracil-based bifunctional abnormal N-heterocyclic carbene (NHC) ligand soluble in water. Employing a catalyst loading of 0.06 mol% and the hydrogen source as HCOOH/HCOONa buffer solution, they achieved 99% product yield (45 °C, 1 h). Cabrero-Antonino et al.90 reported the first homogeneous earth-abundant metal catalyst for the TH of quinolines in 2017. The active species was generated in situ by combining (CoBF4)2·6H2O with tris(2-(-(diphenylphosphino)phenyl) phosphine ligand to hydrogenate quinolines, giving up to 99% conversion. Similar results were obtained with the analogous well-defined complex of Co under the same reaction conditions.
4.1.1.3.3. Using ammonia borane as a hydrogen source. Vermaak et al.91 developed the first Ni(II)-catalyzed TH of quinolines, where ammonia borane (AB) was utilized as the source of hydrogen. They discovered that the precatalysts, an in situ-generated Ni(II)-bis(pyrazolyl)pyridine complex, effectively hydrogenated quinoline derivatives, achieving yields as high as 90% after 30 min at 25 °C. NMR analysis indicated that the presence of a hydride Ni(II) complex accounts for the TH of quinoline to 1,2,3,4-THQ, proceeding through the 1,4-dihydroquinoline intermediate. Jia et al.92 described the efficient TH of quinolines to form 1,2,3,4-THQ with complexes based on Pd(II) containing NHC and bridged phenylene bis(thione) ligands, where AB was employed as the source of hydrogen under gentle reaction conditions. Notably, higher activities were observed with binuclear Pd(NHC) complexes relative to the Pd(NHC) mononuclear complexes under identical reaction conditions. Wu's group also reported TH of indoles and quinolines catalyzed by zirconium hydride, with AB being the source of hydrogen.93 The yields of the hydrogenated products reached up to 94%, with good tolerance toward diverse functionalities. Preliminary mechanistic investigations suggest a concerted activation pathway involving both N–H and B–H bonds.93
Maji et al.94 reported the hemilabile Mn complex with amine, benzimidazole, and sulfur-containing ligands for the TH of various quinoline derivatives, sourcing hydrogen from AB. The well-defined complex could hydrogenate quinolines to the corresponding THQ in up to 85% yield at 35 °C in THF. Bhatt and Natte95 developed a no-ligand TH of various heteroarenes employing a commercial homogeneous RuCl3·xH2O precatalyst with AB as a hydrogen source. This method is simple, user-friendly, and accommodates a diverse range of functionalities, enabling even challenging heterocycle substrates to be reduced. Under optimal reaction conditions of 120 °C, 5–10 mol% catalyst loading, and 24 h reaction time, quinoline and 2-methylquinoline were hydrogenated to their THQ derivatives in isopropanol in yields of 91% and 87%, respectively. Mao et al.96 demonstrated Mn-catalyzed asymmetric TH of quinolines using AB hydrogen source in aqueous media. They reported the reduction of an array of quinoline substituted at position 2, to their THQ derivatives, yielding up to 97% and ee's as high as 99%. Furthermore, Chu et al.97 also demonstrated the catalytic TH of quinolines to 1,2,3,4-THQs using Mn(I) PNP pincer complex 26 (Fig. 7) with AB in the presence of KOtBu.
4.1.1.3.4. Other hydrogenating agents. Fujita et al.98 in 2004 developed an efficient approach to the TH of quinolines using a Cp*Ir complex as the catalyst. This approach enabled the chemo- and regioselective TH of quinolines with isopropanol serving as the source of hydrogen, yielding a range of 1,2,3,4-THQs. They discovered that adding perchloric or triflic acid or other Brønsted acids was necessary to achieve protonation and activate the substrate for effective hydrogenation (Scheme 7). In 2017, He et al.99 developed a highly efficient TH of quinolines employing Hantzsch ester as the hydrogen transfer agent using mild reaction conditions. Utilizing 2.5 equiv Hantzsch ester and 1 mol% load of Fe(OTf)2 at 40 °C, quinoline was converted to THQ in 96% yield. Various methyl-substituted quinolines were also dehydrogenated with yields ranging 86–96%. Recently, methyl formate was employed as a hydrogen source in the TH of quinoline derivatives with an Ir(III) complex. Using 0.2 mol% catalyst loading and 10 equiv. methyl formate, quinoline was converted to THQ in 92% yield.100 Gong et al.101 also reported the Mn-catalyzed TH of quinolines and other N-heteroarenes. The molecular Mn complex catalyst system they developed employed 2-propanol or ethanol as the hydrogen source. This straightforward transformation demonstrates an efficient catalyst system and high tolerance for various functional groups. Overall, this advancement presents an effective and practical synthetic approach for producing useful heterocycles. Employing DFT studies revealed an outer-sphere mechanism, where the 3,4-dihydroquinoline pathway is preferred to either the 1,2- or 1,4-dihydroquinoline routes in the TH of quinoline to THQ. Additionally, the rate-limiting step involves transporting a hydride to the Mn center from propan-2-olate, which regenerates the catalyst.101
 | ||
| Scheme 7 TH of quinolines with isopropanol as the hydrogen source.98 | ||
4.1.1.3.5. Mechanistic considerations of TH reactions. Maji et al.94 proposed the mechanism illustrated in Scheme 8 for the Mn-catalyzed TH of quinolines with AB as the hydrogen donor. Initially, the reaction of Mn complex with AB afforded the pentacoordinate intermediate (Int-A) when HBr and CO are eliminated via dehydrohalogenation promoted by AB.57,58 Next, Int-B (Mn(I) hydride) was produced via dehydrogenating AB by Int-A, releasing H2N·BH2. Subsequently, the thiomethoxy side arm of the ligand is dissociated, and quinoline is bonded to the metal center, resulting in Int-C. Interestingly, adding an external source of ligand dramatically decreased the yield of 1,2,3,4-THQ, and the Mn(I) complex with a lightly bound hemilabile sulfur side arm displayed better activity relative to the other complexes with strongly bound ligands. Thereafter, the quinoline's C–N bond is incorporated into the Mn–H bond, affording an intermediate (Int-D). Afterwards, 1,2-dihydroquinoline is produced through proton migration from the N–H ligand backbone, regenerating Int-A, the active catalyst.
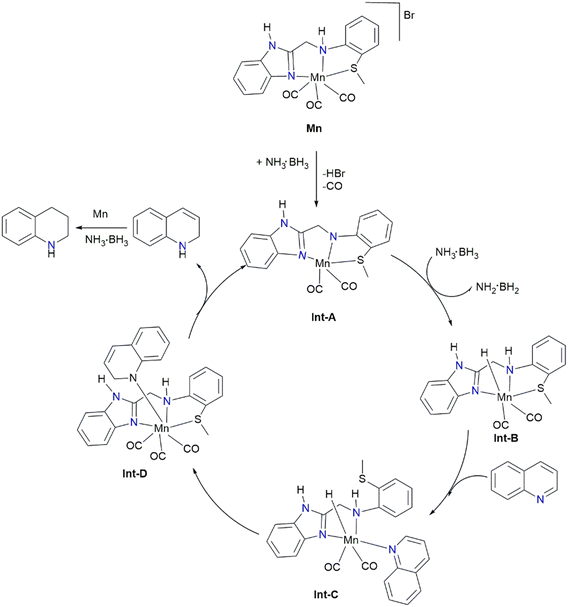 | ||
| Scheme 8 Mechanism for the TH of quinoline using AB as hydrogen source.94 | ||
4.1.1.4. Regioselective hydrogenation of carbocycles in quinolines. In compounds with several aromatic components, the ring with the minimal resonance energy preferentially undergoes hydrogenation. For instance, it is well established that the hydrogenation of quinolines and related compounds occurs at the heteroaromatic ring, being the least aromatic.102 However, Borowski et al.103 hydrogenated certain polynuclear N-heteroaromatic compounds with bis(dihydrogen) Ru complex [RuH2(PCy3)2(H2)2], 27 (Fig. 8), and found the conversion of isoquinoline and quinoline to their corresponding 5,6,7,8-tetrahydro carbocycle-hydrogenated products under gentle conditions (Scheme 9). Their findings indicated that RuH2(PCy3)2 complexes engage in η4-coordination with the carbocycle, leading to regioselective reduction under mild conditions.
Later, Urban et al.104 in 2011 reportedly employed a chiral Ru complex with NHC ligands (28) in quinoxaline asymmetric hydrogenation, affording the corresponding carbocycle-hydrogenated products with good enantioselectivity. Notably, the NHC ligand greatly influences the regioselectivity, highlighting the impact of electronic tuning in catalyst design. Kuwano et al.105 also researched the hydrogenation of isoquinolines and quinolines utilizing the Ru/PhTRAP (29) in asymmetric catalysis. Again, hydrogenating the carbocycle was preferred to the heterocycle in isoquinolines and quinolines. In a different analysis by Kuwano and colleagues, where the selective hydrogenation of isoquinoline carbocycles was accomplished using Ru(η-methallyl)2(COD) together with the trans-chelate chiral PhTRAP (30) ligand, it was suggested that the diphosphine ligand's huge bite angle could be essential for the remarkable chemoselectivity.106Scheme 10 illustrates the plausible pathways suggested for the hydrogenation of quinoline carbocycles using Ru-based catalysts.105,106
 | ||
| Scheme 10 Suggested routes for hydrogenating isoquinoline carbocycles.105,106 | ||
Recently, Luo et al.107 reported a highly selective homogeneous Ru-catalyzed hydrogenation of the carbocyclic ring in fused heteroarenes such as quinolines, isoquinolines, and quinoxalines. The catalyst system consists of a Ru complex with a chiral spiroketal-based diphosphine (SKP) ligand 31. The method gives excellent chemoselectivity, favoring 5,6,7,8-tetrahydro products over the conventional 1,2,3,4-tetrahydro counterparts. They also investigated various ligands in Ru-catalyzed quinoline hydrogenation using similar conditions as the SKP system.
The ligands (32–36 and 38, Fig. 9) gave inferior reactivity and selectivity, while 37 and 39 resulted in poor conversion. On the contrary, 40, an N-heterocyclic carbene, was highly reactive but lacked selectivity. Interestingly, the 2,2′-bipyridine ligand 41 favored the heterocycle-reduced product, indicating that the competing carbo/heterocycle hydrogenation pathways are influenced by ligand electronics and steric effects. Using [Ru(p-cymene)Cl2]2 instead of Ru(methallyl)2(COD) with 31 gave almost identical results, suggesting a common active Ru species in both cases. The mechanism involves an inner-sphere hydrogen transfer pathway, where an initial η4-coordination of the carbocycle to Ru facilitates metal-to-ligand hydride transfer, followed by sequential oxidative addition of H2 and reductive elimination of C–H.
 | ||
| Fig. 9 Scope of ligands screened for the Ru-catalyzed hydrogenation of the carbocyclic ring in quinoline using the Ru(methallyl)2(COD) precursor. | ||
4.1.1.5. Full hydrogenation of quinoline to decahydroquinoline. Homogeneously catalyzed hydrogenation of quinoline typically produces the THQ derivatives, whereas the decahydroquinoline (DHQ) derivatives require more stringent reaction conditions with heterogeneous catalyst systems.20 In 2020, Chatterjee et al.59 reported a one-pot dual catalysis for hydrogenating arenes and heteroarenes using the Ru complex [{(η6-p-cymene)RuCl}2(μ-H–μ-Cl)]. They demonstrated that adding mercury or phenanthroline had little or no impact on reducing quinoline to THQ. Indications were that a molecular complex facilitates this transformation. On the contrary, fully hydrogenating THQ or quinoline was effectively halted when these poisons were present, implying that Ru nanoparticles account for the carbocyclic ring reduction. Considering their findings, they proposed that the full reduction of quinoline is achieved via “dual catalysis”, where an organometallic complex aids in the heterocycle hydrogenation, and NPs produced in situ facilitate the homocycle conversion.
Viereck et al.65 recently introduced an innovative catalyst system for the diastereoselective and enantioselective complete reduction of naphthalenes and quinolines. Utilizing the earth-abundant Mo as a more sustainable transition metal, they strategically identified an oxazoline imino(pyridine) ligand framework. Using a large tert-butyl substituent on the pyridine ring proved essential for achieving high enantioselectivity, even though it is positioned further from the metal center. This extra steric hindrance likely prevents hydrogenation of the pyridine, thereby avoiding catalyst degradation and minimizing racemic background reactions that could lower the enantiomeric excess. A series of quinolines with 2,6-disubstitution is exposed to standard hydrogenation conditions. Remarkably, every 2,6-disubstituted quinoline was converted to a DHQ single diastereomeric product, establishing four stereocenters in one process.
Hydrogenating 2,6-dimethylquinoline gave a 40% yield with DHQ having over 98% ee. Furthermore, the hydrogenation of 2-methylquinoline led to a 17% yield, with its DHQ counterpart having 91% ee. As the position 6's substituent surged in size, specifically in 6-isopropyl-2-methylquinoline, the DHQ was obtained in 8% yield with a 77% ee. Altering the substituent at position 2 to a phenyl group substantially slowed the reaction, resulting in a mix of phenyl, heterocycle, and carbocycle reduction products, likely resulting from the greater steric hindrance. The varied product mix underscores the Mo catalyst's distinctive reactivity, as it facilitates arene ring hydrogenation while promoting enantioselective heterocycle hydrogenation. The full reduction of the two rings in quinoline was peculiar to the 2,6-substitution and moving away to different substitution sites resulted in mixed products. Changing the heteroarene substitution hindered full reduction yielding a mixture with 18% carbocycle-reduced and 81% heterocycle-reduced products (98% ee). This suggests that the full hydrogenation of quinolines depends on their steric environment. Furthermore, quinolines without substitution next to a heteroatom did not get fully hydrogenated to DHQ; instead, a mix of heterocycle and carbocycle-reduced derivatives was detected.
Manas et al.112 in 2015 reported on two Ir catalysts 43 and 44 for the reversible hydrogenation of quinaldine. Although neither system surpassed 42 in terms of dehydrogenation efficiency, both 43 and 44 are much more efficient for the hydrogenation of quinaldine. Vivancos et al.113 developed an effective approach for reversibly (de)hydrogenating derivatives of quinoline with a triazolylidene-based Ir complex, operating in water without additives under mild conditions. Complexes 45 and 46 were effective catalysts in both reactions, with the reaction greatly influenced by the partial pressure of hydrogen in the medium. Impressively, when using 0.5 mol% 47 at 90 °C, the hydrogenation of quinoline in water resulted in 100% conversion. Meanwhile, the reverse dehydrogenation at 100 °C with 2 mol% catalyst achieved 90% conversion.
These reversible catalytic transformations are highly efficient in aqueous media, with the shift between hydrogenation and dehydrogenation governed solely by the presence or absence of hydrogen gas.
Similarly, Wang et al.114 in 2019 successfully performed both hydrogenation and dehydrogenation of the quinoline/THQ pair using the electron-rich Ir catalyst 48 in aqueous solution under mild conditions. However, the catalyst was not recycled in multiple hydrogenation–dehydrogenation cycles.
Choudhury's group successfully developed reusable, bifunctional Ir-based catalysts for (de)hydrogenation of N-heteroarenes in aqueous solution.115,116 These catalysts, when used in aqueous media, were effective for (de)hydrogenation reactions. The key design features included the incorporation of an abnormal/normal N-heterocyclic carbene (a/nNHC)−Ir(III) to enhance the stability and robustness of the complex while facilitating efficient hydride transfer due to the strong sigma-donor properties of the a/nNHC ligand. In addition, a bifunctional metal–ligand unit to promote cleavage of H–H (or N–H) bonds through cooperative mechanisms was employed, and the incorporation of uracil nucleobase in the ligand structure provided solubility in water through hydrogen bonding in the complex's second coordination sphere. This design led to the development of Ir catalyst 48 with long-term durability; furthermore, it was capable of operating under mild conditions, and high product yields could be achieved. Ru complexes bearing multimodal proton-responsive CNN(H) pincer ligands were also found to demonstrate considerable activity in both quinoline dehydrogenation and hydrogenation.117 Besides the platinum group metals (PGMs), a few earth-abundant metal complexes have been employed for the reversible hydrogenation of quinolines. In 2014, Chakraborty et al.118 used a molecular Fe complex bearing a pincer bis(phosphino) amine ligand for the hydrogenation and acceptorless dehydrogenation of N-heterocycles. Iron is appealing, owing to its low toxicity, cost, abundance, and having similar properties to Ir catalysts. The full conversion of 1,2,3,4-tetrahydroquinaldine requires a reaction in xylene at 140 °C for 30 h, using a catalyst loading of 3 mol%, whereas the reverse hydrogenation involves a reaction for 24 h in THF at 80 °C, at high pressure (5–10 bar H2), using 10 mol% of KOtBu. Furthermore, the acceptorless, reversible hydrogenation reactions involving quinolines have been reported to proceed with a molecular Co complex and pincer aminobis(phosphine) [PN(H)P] ligand.119 Dahiya et al.120 reported a stable molecular Cp*Co(III)-catalyst for the TH of quinoline derivatives and cyclic amines oxidative dehydrogenation in aqueous media. They reported that the hydrogenation is facilitated by FA as a hydrogen donor, whereas the dehydrogenation utilizes O2 gas as the oxidant. These methods open new opportunities to explore air-stable Co catalysts for eco-friendly (de)hydrogenation processes.
NCP-type pincer Ir complex (52) bearing a robust benzoquinoline framework demonstrated efficient quinoline dehydrogenation. Quantitative product yields were obtained when THQ and tetrahydroisoquinoline were dehydrogenated at 150 °C with a catalyst load of 1 mol%.124
Muthaiah and Hong125 reported the Ru-catalyzed dehydrogenation of various THQ derivatives with commercially available Ru–H complexes. Using 2.5 mol% catalyst loading, Shvo's catalyst quantitatively dehydrogenated tetrahydroisoquinoline at 165 °C in mesitylene (24 h). The 1,10-phenanthroline-5,6-dione Ru complex with Co(salophen) cocatalyst system was utilized in dehydrogenating several THQs. The system exhibited efficient activities, with various substrates, when 2.5 mol% [Ru(phd)3]2+ catalyst and 5 mol% Co(salophen) cocatalyst loadings were used under ambient conditions.126 Their initial studies compared the previously optimized phd/ZnI2 catalyst with simple octahedral [Fe(phd)3]2+ and [Ru(phd)3]2+ complexes for THQ dehydrogenation. The phd/ZnI2 system exhibited low activity, and the activity was lost after ∼6–7 h, resulting in ≤20% conversion. The [Fe(phd)3]2+ had a comparable rate on the onset but improved stability, while [Ru(phd)3]2+ exhibited significantly higher activity with 93% quinoline yield achieved within 24 h. Adding Bu4NI (1 mol%) confirmed that the redox couple (I−/I3−) facilitates aerobic oxidation of the hydrogenated catalyst. Noteworthy is that THQ was dehydrogenated under optimized conditions to afford 89% isolated product yield.126 Wang et al.127 dehydrogenated THQ derivatives utilizing pyrazoyl-indolylpyridine Ru(II) organometallics in good yields (up to 93%). With a monohydrido-bridged dinuclear Ru catalyst in the acceptorless dehydrogenation of heterocycles, THQ was converted to quinoline in 85% yield with 1 mol% catalyst loading at 135 °C in toluene.128
Homogeneous Pd catalysts have also been utilized in the dehydrogenation of THQs. For example, Wang et al.129 achieved 92% yield when THQ was dehydrogenated using Pd(MeCN)2Cl2 in dichloroethane at 120 °C in the presence of O2. Similarly, when using 5 mol% Pd(OAc)2 with a neocuproine catalyst system in DMF, THQ afforded quinoline in 83% isolated yield in 1 h.130 An inexpensive CuI catalyst was used by Jung and coworkers131 for the dehydrogenation of 1,2,3,4-THQ at RT under mild conditions. Notably, using 10 mol% each of CuI and di-tert-butyl azodiformate and 20 mol% 4-dimethylaminopyridine additives enhanced the dehydrogenation to afford up to 92% quinoline yield.131 Shen et al.132 used 2 mol% Cu(I) complex 53 (Fig. 12) to dehydrogenate THQs in utilizing 1,2-dichlorobenzene and O2 (120 °C, 3 h) to afford a high yield. This method is advantageous as it is carried out in the absence of additives. Zumbrägel et al.133 used oxovanadium(V) complex with gentle reaction conditions under O2 to dehydrogenate THQs.
When using 10 mol% of the complex in water (60 °C, 1 bar O2, 48 h), 91% quinaldine was obtained from tetrahydroquinaldine. In 2020, Bera et al.134 reported dehydrogenating N-heterocycles using a Ni catalyst and molecular oxygen. Impressively, the system converted THQ to quinoline in excellent yield.
4.2. Carbazoles
Carbazole is an alternative LOHC with a high HSC (6.7 wt%) and stability under favourable conditions, hence ideal for prolonged storage. Nonetheless, with a 68–70 °C melting point, it becomes solid at RT following oxidation.135 The structures of selected carbazoles used as LOHCs are illustrated in Fig. 13.Wang et al.6 studied dehydrogenating N-ethylperhydrocarbazole using Ir pincer complexes 54–56 (Fig. 14). They tested the three catalysts at 200 °C and found that they produced varied, partially dehydrogenated products: N-ethyloctahydrocarbazole, N-ethyltetrahydrocarbazole, and N-ethylcarbazole (NEC). Catalyst 56 showed the highest activity, where NEC was converted within 9 h to yield 70% N-ethyloctahydrocarbazole and 30% N-ethyltetrahydrocarbazole. Extending the reaction time led to further dehydrogenation, resulting in 82% N-ethyltetrahydrocarbazole and 18% NEC, with a TON that reached 436. Lowering the temperature to 150 °C, the efficiency dropped significantly, producing only 23% N-ethyloctahydrocarbazole and 2% N-ethyltetrahydrocarbazole.7 No evidence of dehydrogenation was observed in experiments performed at 100 °C. This likely indicates that the high enthalpy of the substrate imposes thermodynamic limitations at lower temperatures.7 According to later reports, the selective dehydrogenations of perhydroindoles and perhydrocarbazoles specifically occur at the five-membered rings.136
4.3. Indoles
Indoles (Fig. 15), structurally akin to carbazoles, have attracted interest as potential LOHC systems. These compounds are readily accessible and exhibit favorable LOHC characteristics, including positive kinetic and thermodynamic profiles for hydrogen release from perhydroindoles using relatively mild temperatures.137,138Table 5 details the properties of some commonly studied indole derivatives as LOHCs.| Indole/carbazole derivative | mp (°C) | bp (°C) | HSC (wt%) | ΔH (kJ mol−1 H2) | Ref. |
|---|---|---|---|---|---|
| 1-Methylindole (1-MID), 2-methylindole (2-MID), N-ethylindole (NEID), 7-ethylindole (7-EID), 8-ethylindole (8-EID). | |||||
| Indole/8H-indole | 6.4 | 53.6 | 20 | ||
| 1-MID/8H-1-MID | −20/−25 | 239/230 | 5.76 | 51.9 | 20 |
| 2-MID/8H-2-MID | 57 | 273 | 5.76 | 51.7 | 20 |
| NEID/8H-NEID | −17.8 | 253.5 | 5.23 | ||
| 7-EID/8H-7-EID | −14/−20 | 230/230 | 5.23 | ||
| H-Carbazole | 246.3 | 6.7 | 48.1 | 54 | |
| N-Methylcarbazole | 89 | 6.2 | 49.1 | 54 | |
| N-Ethylcarbazole | 70 | 363 | 5.7 | 47.8 | 53, 54 |
The (de)hydrogenation of indoles and their derivatives, such as 1-methylindole (1-MID), 2-methylindole (2-MID), N-ethylindole (NEID), and 7-ethylindole (7-EID), among others, has been primarily explored using heterogeneous catalyst systems.20,139 For example, Li et al.140 demonstrated the hydrogenation of 2-MID using a catalyst composed of 5 wt% Ru/Al2O3 at temperatures between 120 and 170 °C, with a rapid conversion rate. The subsequent dehydrogenation of its hydrogenated form, 8H-2-methylindole, was achieved with a 5 wt% Pd/Al2O3 catalyst at temperatures ranging from 160 to 200 °C, resulting in complete dehydrogenation within 4 h at 190 °C, without any side reactions. Compared to heterogeneous catalysts, the use of homogeneous catalysts in these transformations for applications in LOHCs is rather limited. The homogeneously catalyzed (de)hydrogenation of indole derivatives is discussed in the sections that follow.
Wang et al.141 in 2010, reported the first example of a highly enantioselective hydrogenation of simple indoles using Pd(OCOCF3)2/(R)-H8-BINAP (BINAP = 2,2′-bis(diphenylphosphino)-1,1′-binaphthyl, 57), activated using a Brønsted acid (Scheme 11). Notably, 2-methyl-1H-indole was readily hydrogenated. Up to 95% conversion and 91% ee were achieved when using 2.4 mol% (R)-H8-BINAP and 2 mol% Pd(OCOCF3)2 for 24 h at room temperature. The researchers proposed that the reaction's enantioselectivity results from the reversible protonation of the substrate, which allows selective hydrogenation of only the targeted enantiomer of the activated indole.
 | ||
| Scheme 11 Enantioselective reduction of unprotected indoles.141 | ||
Núñez-Rico et al.146 subsequently demonstrated the asymmetric hydrogenation of unprotected indoles employing in situ-generated Ir complexes obtained as a single enantiomer from phosphine–phosphite (P–OP) ligand 58 (Fig. 16). The hydrogenation of 2-methyl-1H-indole with 1 mol% Ir precatalyst in THF (rt, 80 bar H2), using stoichiometric quantities of sulfonic acid additives, resulted in 2-methylindoline. Interestingly, the conversion was dependent on the acid strength, where organic acids gave conversions of up to 20% while the stronger sulfonic acid derivatives elevated the conversions up to ∼80%. Several reaction conditions were investigated, encompassing the reaction time, solvent type, and catalyst loading. Using the optimal reaction conditions, various indole derivatives were successfully hydrogenated to achieve high conversions (up to 92%) and 91% ee.146
Wen et al.147 demonstrated that an effective catalyst system using a Rh/ZhaoPhos complex performs remarkably in acidic media. Various substituted indoles were reduced, resulting in appreciable enantioselectivities. Impressively, 2-methylindole was hydrogenated to afford 2-methylindoline in 98% yield and 98% ee with a substrate/[Rh(COD)Cl]2/ligand ratio = 100/0.50/1.0 (rt, 40.53 bar H2, 48 h).147 In another study, the conversion of indole to indoline was accomplished in quantitative yield when using a catalytic system derived from [{(η6-p-cymene)RuCl}2(μ-H–μ-Cl)] (90 °C, 50 bar H2, 40 h). After combing various approaches comprising kinetic studies, in situ reaction monitoring, electron microscopy, and poisoning evaluations, the suggestion was made that the initial Ru complex functions as a precatalyst, and the heteroarene reductions are rather facilitated by a monomeric Ru complex.59Table 6 provides a summary of the catalysts for homogeneous hydrogenation of the various N-heterocyclic LOHCs discussed above.
| Catalyst | Substrate/product | Conv./yield(%) | Selectivity (%) | Ref. |
|---|---|---|---|---|
| Co-Tetraphosphine | Quinoline/1,2,3,4-1,2,3,4-THQ | >99 | — | 76 |
| [WCl(η5-Cp)(CO)3] | Quinoline/1,2,3,4-THQ | 94 | — | 77 |
| [Ni(TIM(PAr)2)3](NTf2) | Quinoline/1,2,3,4-THQ | 99 | — | 78 |
| NNP–Mn(I) | Quinoline/1,2,3,4-THQ | >99 | — | 79 |
| CNHC–Mn | Quinoline/1,2,3,4-THQ | 97 | — | 80 |
| Imidazolylidene–Mn(I) | Quinoline/1,2,3,4-THQ | 20 | — | 81 |
| AgOTf | 2-Methylquinoline/1,2,3,4-THmQ | 98 | — | 85 |
| [Ir(cod)Cl]2/hydrosilanes | Quinoline/1,2,3,4-THQ | 94 | — | 86 |
| Ru(PPh3)3Cl2 | Quinoline/1,2,3,4-THQ | 95 | 80 | 87 |
| 2-Methylquinoline/1,2,3,4-THmQ | 93 | 100 | ||
| Ir/imidazoline | 2-Methylquinoline/1,2,3,4-THmQ | 92 | 62 (ee) | 88 |
| Cp*Ir/uracil–NHC | Quinoline/1,2,3,4-THQ | 99 | — | 89 |
| (CoBF4)2·6H2O/phosphine | Quinoline/1,2,3,4-THQ | 99 | — | 90 |
| Ni(II)-bis(pyrazolyl)pyridine | Quinoline/1,2,3,4-THQ | 97 | — | 91 |
| NHC–Pd(II)-SCS | Quinoline/1,2,3,4-THQ | 95 | — | 92 |
| Cp2ZrH2 | Quinoline/1,2,3,4-THQ | 94 | — | 93 |
| Mn–benzimidazole | Quinoline/1,2,3,4-THQ | 85 | — | 94 |
| RuCl3·xH2O | Quinoline/1,2,3,4-THQ | 91 | — | 95 |
| 2-Methylquinoline/1,2,3,4-THmQ | 87 | |||
| Mn(I) PNP | Quinoline/1,2,3,4-THQ | 97 | 99 (ee) | 96, 97 |
| Cp*Ir | Quinoline/1,2,3,4-THQ | 93 | — | 98 |
| Fe(OTf)2 | Quinoline/1,2,3,4-THQ | 96 | — | 99 |
| Ir(III) N^N | Quinoline/1,2,3,4-THQ | 92 | — | 100 |
| Mn-PNP | Quinoline/1,2,3,4-THQ | 99 | — | 101 |
| Mn(I) PNP | Quinoline/1,2,3,4-THQ | 94 | — | 102 |
| [RuH2(PCy3)2(H2)2] | Quinoline/5,6,7,8-THQ | 100 | 99 | 103 |
| Isoquinoline/5,6,7,8-THiQ | 88.5 | 100 | ||
| Ru/PhTRAP | 2-Phenylquinoline/5,6,7,8-THpQ | 97 | 97 | 104 |
| Ru-Ph-SKP | Quinoline/5,6,7,8-THQ | >99 | >99 | 105 |
| [{(η6-p-Cymene)RuCl}2(μ-H–μ-Cl)] | Quinoline/DHQ | >99 | 98 (ee) | 65 |
| Mo/oxazoline imino(pyridine) | 2,6-Dimethylquinoline/2,6-dimethylDHQ | 100 | 98 (ee) | 64 |
| 2-Methylquinoline/2-methylDHQ | 100 | 91 (ee) | ||
| Pd(OCOCF3)2/(R)-H8-BINAP | 2-MID/2-Methylindoline | 95 | 91 (ee) | 141 |
| Ir/P–OP | 2-MID/2-Methylindoline | 80 | 91 (ee) | 146 |
| Rh/ZhaoPhos | 2-MID/2-Methylindoline | 99 | 98 (ee) | 147 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 equilibrium between N-methyltetrahydroindole and N-methylindole, eliminating over 4 wt% hydrogen. Thus, the kinetic barrier to dehydrogenation is overcome at higher temperatures, leading to thermodynamic control.148
1 equilibrium between N-methyltetrahydroindole and N-methylindole, eliminating over 4 wt% hydrogen. Thus, the kinetic barrier to dehydrogenation is overcome at higher temperatures, leading to thermodynamic control.148
Later, Brayton and Jensen,136 explored the solvent-free dehydrogenation of various perhydroindolic substrates, including N-methylperhydroindole (MePHI), using Ir complex 56. The dehydrogenation of the heterocyclic ring in all indolic molecules commenced at 170 °C, while the cyclohexane ring remained unaffected until 200 °C. Consequently, the reactions were conducted at 195 °C to achieve an optimal dehydrogenation rate, without affecting the cyclohexane ring. To recover the products, filtration through an alumina plug or distillation was employed. MePHI gave a quantitative yield with 1 mol% of complex 56 after 48 h. However, benzoyl perhydroindole, perhydro-2-methylindole, perhydroindole, and perhydroskatole exhibited low dehydrogenation yields of 6%, 1.5%, 3%, and 2.5%, respectively, with only slight improvements achieved when both catalyst loading and reaction time were increased.
Wang et al.114 achieved a 97% conversion when indole was hydrogenated at 120 °C with an electron-rich Ir complex bearing a dipyridyl amine ligand, using just 0.2 mol% catalyst loading. Additionally, the rare-earth praseodymium catalyst was developed to efficiently dehydrogenate indoline in high yield. Utilizing 7.5 mol% catalyst loading and 15 mol% 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) additive, indoline was dehydrogenated to 98% (1 bar O2, 12 h). This approach showed excellent compatibility with various functional groups, such as quinoline, under mild conditions (Scheme 12).149
 | ||
| Scheme 12 Aerobic dehydrogenation of N-heterocycles using rare-earth metal catalysts.147 | ||
In 2020, Kawauchi et al.150 used Grubb's catalyst to perform the aerobic dehydrogenation of N-heterocycles. Using 0.1 mol% catalyst in EtOAc for 46 h, indoline was dehydrogenated in 90% yield at 70 °C.150 Ru hydride complexes with a pyrazolyl-(2-indol-1-yl)-pyridine ligand effectively catalyzed the acceptorless dehydrogenation of N-heterocycles, showcasing high catalytic efficiency across a wide range of substrates. Notably, indoline was converted into 82% yield of indole. Substrates of indoline featuring a 2-Ph, 2-Me, or 3-Me groups were also dehydrogenated efficiently, resulting in indoles with yields ranging from 78% to 91%.127 Osmium polyhydride was also identified as an efficient catalyst precursor for dehydrogenating cyclic amines to aromatic heterocycles. With 10 mol% catalyst loading and a temperature of 140 °C, excellent conversion of 2-methylindoline to 2-methylindole was achieved.151 Liu's group also developed a technique involving 2 mol% of copper complex 46, with 10 mol% of TEMPO additive and oxygen as an oxidant, at RT in THF, to afford high yields of indoles.132
Zubar et al.152 reported an efficient method for the chemoselective hydrogenation and acceptorless dehydrogenation of N-heterocycles using a molecular Mn complex (Scheme 13). Indole was hydrogenated using 2 mol% of the Mn complex at 100 °C and 50 bar H2 for 36 h and 5 mol% of the base, affording 92% indoline yield. This catalytic system is compatible with various functional groups, producing the reduced heterocycle derivatives with high yields. Further analysis indicated a mechanism involving metal–ligand cooperativity. Various substituted indoles were also analyzed under the established reaction conditions, resulting in the respective indoline derivatives in appreciable yields. The dehydrogenation of indoline under oxidant-free conditions using 1 mol% Mn complex (120 °C, 24 h) led to 89% indole. Notably, hydrogenation commences with the activation of the complex by KOtBu, forming active species A, which then reacts with H2 to generate H-N–Mn–H species B, facilitating indole conversion to indoline. In the dehydrogenation, species A converts indoline to indole, forming species B, which subsequently releases H2.
 | ||
| Scheme 13 Mechanism for the Mn-complex-mediated reversible hydrogenation of indole.152 | ||
4.4. Other potential LOHC candidates based on N-heterocycles
The naphthyridine structure, with two distanced nitrogen atoms, prevents bidentate coordination to the metal center. Using 5 mol% catalyst loading, Cp*Ir complex (59) bearing a bipyridonate ligand selectively dehydrogenated 2,6-dimethyldecahydro-1,5-naphthyridine to 2,6-dimethyl-1,5-naphthyridine in quantitative yield at 138 °C in p-xylene. Similarly, the reverse hydrogenation reaction afforded a stereoisomeric mixture in 92% yield; however, the catalyst showed possible deactivation at 100 °C under H2 (61 bar). Shifting to a Cp*Ir complex (60) bearing a robust phenanthroline-2,9-dione ligand led to a nearly quantitative hydrogenation yield and a 98% dehydrogenation yield, confirming high catalytic activity. Successive hydrogenation and dehydrogenation reactions gave yields that reached 100% and 92%, respectively.
2,5-Dimethylpiperazine was obtained quantitatively when using 0.50 mol% 61 (Fig. 20) and 1.0 mol% 6,6′-dihydroxy-2,2′-bipyridine, and a hydrogen/solvent (H/S) ratio of 60 (30 mmol hydrogen uptake in 0.50 mL solvent) was achieved.
Under optimal catalytic conditions, reversible hydrogenation was achieved in one vessel, in which the reaction was repeated four times without catalytic loss, as illustrated in Scheme 14. This system demonstrates an efficient hydrogen storage method with a high H/S ratio of 30 (30 mmol hydrogen in 1.0 mL solvent).
 | ||
| Scheme 14 Method for interconverting compounds a and b through reversible hydrogenation catalyzed by 61.154 | ||
Solvent-free interconversion between a and b was also investigated with 78% hydrogenation efficiency recorded. Employing this approach resulted in the storage of 1144 mL (46.8 mmol) of hydrogen using 2.18 mL of 2,5-dimethylpyrazine.
5. Techno-economic considerations for aromatic and heteroaromatic LOHCs
The techno-economic viability of aromatic and heteroaromatic LOHC systems is governed by a set of interrelated descriptors that link molecular structure and catalytic performance to system-level feasibility. Key parameters include operating temperature and heat demand, hydrogen release kinetics, catalyst cost and lifetime, hydrogen purity, and long-term carrier stability. Together, these factors determine capital expenditure (CAPEX), operating costs (OPEX), and application suitability.A dominant techno-economic constraint for both aromatic and heteroaromatic LOHCs is the high endothermicity of the dehydrogenation step, which typically requires operating temperatures in the range of 200–350 °C. Such elevated temperatures impose significant penalties in terms of heat management, reactor materials, start-up energy demand, and system integration, particularly for mobile or on-board applications. While heteroaromatic LOHCs may benefit from heteroatom-induced electronic effects that lower activation barriers and improve kinetics, these systems generally remain within a temperature regime best suited to stationary or hybrid energy storage concepts, where waste heat integration can partially offset the thermal demand.155–157
Beyond thermodynamics, hydrogen release kinetics directly influence reactor size and hydrogen throughput. Slow dehydrogenation rates necessitate larger reactors and longer residence times, increasing system footprint and capital cost. Aromatic LOHCs often exhibit sluggish kinetics in the absence of high temperatures or highly active catalysts, whereas heteroaromatic LOHCs can show enhanced rates due to polar functionalities or metal–ligand cooperative activation pathways. However, these kinetic advantages must be balanced against catalyst stability and selectivity under prolonged operation, underscoring the importance of catalyst robustness from a techno-economic perspective.156,158
Catalyst cost, lifetime, and recyclability represent another critical techno-economic descriptor, particularly for systems relying on homogeneous catalysis. Both aromatic and heteroaromatic LOHCs frequently employ precious-metal catalysts (e.g., Ru, Ir, Pd), making high turnover numbers, long operational lifetimes, and efficient catalyst recovery essential for economic viability. Catalyst degradation or difficult separation can substantially increase operating costs and limit scalability.158,159 In parallel, the purity of the released hydrogen plays a decisive role in downstream processing costs; even trace contaminants can necessitate additional purification steps, particularly for fuel-cell applications.160
Finally, the availability, synthesis cost, and long-term chemical stability of the LOHC medium itself significantly impact system lifetime and operating expenditure. Carriers that exhibit high robustness over repeated hydrogenation–dehydrogenation cycles offer clear economic advantages, whereas degradation or side-product formation reduces effective hydrogen capacity and necessitates carrier replacement.155,157 Overall, these considerations highlight that the techno-economic performance of aromatic and heteroaromatic LOHCs is dictated by trade-offs between hydrogen capacity, operating conditions, catalytic efficiency, and carrier durability, rather than by any single parameter in isolation.
6. Gaps in the literature
We have established that the following appear to be current gaps in the literature pertaining to the homogeneously catalyzed (de)hydrogenation of LOHCs (addressed further below).• Limited literature on the dehydrogenation of homocyclic compounds using homogeneous catalysis (as far as we are aware, only one homogeneous catalytic system has been described for reversible hydrogen storage/release, and it utilizes an Ir pincer complex).
• A paucity of literature on homogeneous catalysts for the (de)hydrogenation of benzyltoluene (BT) and dibenzyltoluene (DBT).
• A paucity of literature on the complete/full hydrogenation of N-heterocyclic LOHCs.
• Apparently, no information is available on large-scale demonstrations with homogeneous systems.
In addressing the homogeneous (de)hydrogenation of potential homocyclic LOHCs, efforts to date have focused on the hydrogenation of toluene and benzene to produce the corresponding cyclohexane, with limited literature on other potential homocyclic LOHCs. Furthermore, to our knowledge, only one homogeneous catalyst system has been described for the reversible hydrogen storage employing an Ir pincer complex for homocyclic LOHCs. The paucity of studies on the (de)hydrogenation of homocyclic systems, such as DBT and BT, also represents a significant gap in the field. With their potential for high hydrogen storage capacities and stability, these systems warrant further investigation, particularly using homogeneous catalysts with tunable ligand environments. Furthermore, there is very little information on the complete/full (de)hydrogenation of critical heterocyclic LOHC candidates such as quinolines, indoles, and carbazoles. It appears that most of the catalytic systems reported to date for the (de)hydrogenation often result in partially (de)hydrogenated products. For instance, the quinoline/DHQ pair with HSC 7.2 is a promising LOHC candidate. However, research on hydrogenating quinoline completely and the corresponding dehydrogenation of DHQ remains limited. Most studies have instead concentrated on the quinoline/THQ pair, which has a lower HSC of 2.3 wt%.
It is recommended that future research be focused on the development of efficient homogeneous catalyst systems for the complete (de)hydrogenation of LOHC pairs that have shown promise to date. Studies should also be directed at developing dual-functional catalysts capable of both hydrogenation and dehydrogenation with high efficiency.
7. The role of homogeneous catalysts in industrial processes
For many years, homogeneous catalysts have been important components of the chemical and material industries. It is impossible to overstate their importance in the efficient production of fine chemicals and materials with high levels of product selectivity, low temperature requirements, and other moderate reaction conditions. In addition to contributing to minimizing the costs to businesses, homogeneous catalysts can be fine-tuned to enhance their reactivity, selectivity, and robustness.161–165 Homogeneous catalysts are relatively easily studied during their reactions because of their molecular nature. Unquestionably, these are the reasons why many of them are still used by industries decades after they first appeared.For example, the Cativa process used to produce acetic acid efficiently via methanol carbonylation, using an Ir catalyst [Ir(CO)2I2]−, has been in use for almost three decades now after its development by BP Chemicals.166,167 The Cativa process is an improved version of the Monsanto process, which makes use of Rh instead of Ir, and requires the presence of excess water and several drying columns for product drying.168 Although the Cativa process is promoted by the presence of Ru as a cocatalyst, the formation of propanoic acid as a byproduct is reduced.166
It is considered relevant to include mention here of the synthesis of pharmaceuticals and other important complex molecules on commercial scales via the widely known Suzuki–Miyaura carbon–carbon bond cross-coupling reactions.169–171 The Pd-based catalysts reported since 1979 by Suzuki and coworkers consist of molecular complexes of Pd(II) with acetates and phosphine as ligands. The precatalyst initially undergoes reductive elimination to generate the active catalyst in the form of Pd(0) and neutral ligands.170,172 Despite the numerous challenges associated with the utilization of the Suzuki–Miyaura cross-coupling catalyst, such as potential deactivation by certain reagents and limited reactivity with a few aryl halides, it has paved the way for further research into related catalysts.173,174
Yet another industrially important homogeneous catalysis process that has retained its significance for almost 90 years is the hydroformylation reaction, popularly known as the Oxo process.175,176 The Oxo process, discovered in 1938 by Otto Roelen, is the main route to long-chain aldehydes and alcohols from terminal alkenes.175 The original catalyst used is the monohydridotetracarbonyl cobalt(I) complex, HCo(CO)4.177 Later complexes bearing other ligand systems, such as phosphines anchored on Co and Rh metals, have been found to offer excellent performance, achieving high selectivity.174–177 The track records of these industrially important homogeneous catalysts that turned the fortunes of many industries have presented hope for the current generation to search for new homogeneous catalysts that would also address energy and climate sustainability issues.
8. Recovery and recycling of homogeneous catalysts
The main challenge associated with the use of homogeneous catalysts to date has been the recovery of the catalyst from the final reaction mixture. Generally, the catalyst, the reactant, and the product are all present in one phase. Obviously, industries would want to recover high-value catalysts in each cycle of production and reuse them to contain costs. Homogeneous catalyst recovery is not entirely impossible; a few recovery strategies do exist, while others are emerging, depending on the nature of the catalyst, the properties of the product, and the solvent systems used. Recovery approaches include distillation, extraction, chromatography, heterogenization, membrane separation, and biphasic system technology. These methods are situation-specific; some are applicable in industry, while others are potential remedies to a challenge. Three different catalyst recovery approaches are now described in sections 7.1–7.3.8.1. Catalyst recovery by distillation
Catalyst recovery via distillation is suitable for volatile products and thermally stable catalysts.178–182 This method is efficient and is employed in industry for the removal of certain products, leaving the catalyst behind for reuse. A typical example of an industrial process that uses the distillation approach to recover catalysts successfully is the Takasago International Corporation's (−)-menthol synthesis from myrcene.183 The homogeneous Ru-BINAP catalyst used by Takasago International Corporation (Japan) is usually recovered as a precipitate after the distillation of (−)-menthol from the final reaction mixture.183 Another example of product distillation from the catalyst is in the Cativa process, where the product and the residual gases are distilled from the catalyst, and the residual gases are recycled back into the reactor.184 On a laboratory scale, several instances of catalyst recovery by distillation have been reported.184–186 As mentioned earlier, a challenge sometimes encountered is the volatility of the product. High-boiling-point products require high-temperature conditions, which must be balanced by the thermal stability of the catalyst. Horváth's group demonstrated the complete distillation of γ-valerolactone from Shvo's catalyst, then enabling the recycling of the catalyst four times.185 γ-Valerolactone has a high boiling point of approximately 207 °C.186 The practicality of catalyst recovery from γ-valerolactone was further studied by Amenuvor et al.187 who successfully distilled the product completely from a Ru^Zn cluster catalyst six times, after each cycle, without any significant loss of catalyst activity. These works have demonstrated the industrial applicability of the recovery and reuse of robust catalysts in the biorefinery industry.To improve energy efficiency and minimize waste, reactive distillation (RD) technology is currently employed in some industries for catalyst recovery.188,189 In this case, the catalytic reaction and distillation processes are designed as a single unit, and as the product is formed, it is quickly removed by distillation. This technique also enhances product yield and selectivity by decreasing the concentration of the product in the reaction mixture. Thus, RD is very useful here for those reactions that require certain reactants in excess or in rate-limiting reactions.188,190 Although the RD technology requires improvement in terms of its relatively intricate design and operation,190 it has been successfully used in several processes, including the production of methyl acetate, methyl tert-butyl ether, and tert-amyl methyl ether, which are used as additives.189
8.2. Catalyst recovery using biphasic systems
The biphasic system offers another ingenious approach to homogeneous catalyst recovery from the final reaction mixture. This technique takes advantage of the distribution of the catalyst and product between two immiscible solvents (generally water and an organic solvent). Biphasic catalysis has found industrial application over the past three decades, with the first appearance being the hydroformylation of propylene to n-butyraldehyde by Ruhrchemie AG, Germany.178,191 One of the basic requirements that makes the biphasic system efficient is the ability of the catalyst to be water-soluble, achieved by making use of water-soluble ligands. The ligand used by Ruhrchemie AG was TPPTS in the form of a Rh catalyst [HRh(CO)(TPPTS)3], developed by Rhône–Poulenc, France.178 TPPTS was further extended to catalyzing the telomerization of dienes and hydrocyanation of olefins using Pd and Ni, respectively.178 Following this was the introduction of a series of sulfonated phosphine ligands such as BINAS, NAPHOS, NORBOS, and BISBIS, as shown in Fig. 21.192,193 | ||
| Fig. 21 Examples of ligands used in biphasic catalysis.162 | ||
The main challenge encountered with water-soluble phosphine ligands is the complexity of their syntheses and the required purification. For instance, attempts to synthesize trisulfonated phosphine ligands usually result in byproducts such as monosulfonated and disulfonated analogues. Some of these impurities remain in the desired ligand even after purification.178 These earlier works stimulated the expansion of research in the area, focusing on other water-soluble ligands and a special group of ionic liquids that are immiscible with some organic solvents.194–197 Because of the difficulties in obtaining water-soluble metal catalysts, researchers have been considering immiscible organic solvents that discriminate the solubilities of both the catalyst and the product.198–200 This eliminates the special design of catalysts for the sake of solubility, since most metal catalysts dissolve in polar organic solvents. The catalyst preferably remains in the polar organic phase while the product moves into the nonpolar organic phase. The Shell higher olefin process (SHOP) for the commercial production of high α-olefins (C6–C20) is the oldest successful biphasic catalysis carried out using a transition metal complex [Ni(COD)Ph2PCH2COO].182,195,200 The catalyst, generated in situ from NiCl2, Ph2PCH2COOH, COD, and sodium borohydride as a reducing agent, is soluble in 1,4-butanediol, while the olefin products are not.195,200 This creates a perfect organic–organic biphasic system where the product serves as one phase and 1,4-butanediol as the other phase.
8.3. Catalyst recovery by membrane filtration
A new technique for the separation of homogeneous catalysts and products is organic solvent nanofiltration (OSN). In OSN, use is made of organic solvent-stable membranes that allow separation based on molecular weight differences and branching or bulkiness of side groups.201–204 Generally, the membrane allows small molecular products to pass through while retaining the catalyst, which is recycled back into the reactor.This favors catalysts with large molecular weight ligands, such as triphenylphosphine and its derivatives.201,203 Ideally, one would prefer to make use of OSN rather than traditional methods of distillation because OSN is more selective and the energy demand is lower, leading to cost savings and the preservation of catalyst stability.205,206 However, research is still ongoing in efforts to overcome its complexity, including the prediction of membrane–solvent behavior, modulation of membrane pore size relative to the sizes of components in mixtures to be separated, feed concentration, temperature considerations, and transmembrane pressure.202,207–211
Notwithstanding these challenges, the OSN technique still has the potential to improve homogeneous catalysis. OSN has been used industrially by Exxon Mobil Corporation (USA) and W.R. Grace (USA) in solvent lube dewaxing with an energy cost saving of ∼20%.212
Interesting work has been carried out by Janssen et al.203 to develop an in-house engineered continuous flow nanofiltration reactor for the hydroformylation of 1-octene. Using a robust Rh catalyst based on a sterically bulky and rigid phosphine ligand, the system was able to retain >99% of the Rh catalyst after 13 days of operation, whilst maintaining very high conversions. There are several more reports of other laboratory-scale investigations involving the use of the OSN strategy for the separation of products from homogeneous catalysts.207,213–215 Looking ahead, OSN could become the future most preferred method for homogeneous catalyst recovery once the main challenges associated with it are resolved.
9. Conclusion – remaining challenges and recommendations
The homogeneously catalyzed (de)hydrogenation of LOHCs, particularly homocyclic and heterocyclic systems, has seen remarkable advancements in recent years. The development of transition metal-based catalysts with tailored ligands has enhanced the efficiency, selectivity, and sustainability of LOHC (de)hydrogenation processes. Moreover, the exploration of heteroaromatic LOHCs has expanded the potential of LOHC technologies, offering improved hydrogen storage capacity and favorable thermodynamics.This review highlights the recent advances in the homogeneously catalyzed (de)hydrogenation of LOHCs, as well as challenges related to the industrial application of homogeneous catalysis, including catalyst recovery and reuse. LOHCs present a promising pathway for efficient energy storage and safe hydrogen transportation, which is very critical in the transition to a sustainable hydrogen economy. However, despite rapid progress, several key challenges must be addressed to facilitate the practical deployment of the LOHC technology.
One of the primary challenges lies in the compatibility of heteroaromatic LOHCs with existing energy infrastructure. Many heteroaromatic LOHCs are solid at ambient conditions, posing logistical challenges for storage and transportation. To meet the ideal LOHC criteria, both hydrogen-rich and hydrogen-lean states should exist in the liquid phase. Among the examined N-heterocyclic compounds, some quinoline and indole derivatives meet this requirement, coupled with a high hydrogen storage capacity ranging between 5.2 and 7.2 wt%. However, their widespread adoption is hindered by high dehydrogenation enthalpies and toxicity concerns, which limit their practicality in the LOHC technology.
Another major limitation is the efficiency of homogeneous catalytic systems for industrial applications, which is constrained by catalyst stability, cost, and recyclability. Homogeneous hydrogenation processes frequently yield partially hydrogenated products, and when full hydrogenation is achieved, the product selectivity often remains suboptimal, reducing the overall hydrogen storage capacity. Furthermore, the recovery and reuse of homogeneous catalysts in large-scale industrial applications remain challenging, adding to operational costs and sustainability concerns.
Furthermore, in homogeneous catalysis LOHCs, the hydrogen–to–substrate (H/S) ratio is a fundamental metric that integrates both the theoretical storage capacity of the carrier, and the practical demands placed on the catalytic cycle. The H/S ratio describes the number of hydrogen equivalents that can be chemically bound per molecule of the substrate during complete hydrogenation; this directly defines the gravimetric hydrogen storage capacity (wt% H2), a primary target for LOHC technologies. For example, N-ethylcarbazole (NEC), one of the most studied LOHC candidates, can be fully hydrogenated to dodecahydro-NEC by uptake of six equivalents of H2, corresponding to an ideal capacity of ∼5.8 wt% H2, a level that approaches practical benchmarks for hydrogen carriers. Achieving and sustaining a high effective H/S ratio in practice hinges on catalyst performance across multiple sequential hydrogenation steps. Homogeneous catalysts, with their tunable ligand environments and capability to mediate H–H activation under milder conditions than many heterogeneous systems, offer a route to drive hydrogenation efficiently toward fully saturated LOHC states. However, this same requirement underscores a key challenge![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) catalysts must maintain activity and selectivity through the entire sequence of hydrogen additions, not only in early steps—otherwise the system stalls at partial hydrogenation and the realized H/S ratio falls below theoretical values. Moreover, the H/S ratio is tightly coupled to operational and system considerations. High H/S ratios necessitate catalysts capable of handling elevated hydrogen pressures and multiple turnovers without degradation, and they influence reactor design, mass transfer, and thermodynamics; incomplete attainment of the target H/S ratio decreases the effective hydrogen density and compromises cyclic reversibility. In this context, homogeneous catalyst designs that can sustain high conversion and selectivity across full hydrogenation–dehydrogenation cycles are critical for translating LOHC chemistry into practical hydrogen storage media.
catalysts must maintain activity and selectivity through the entire sequence of hydrogen additions, not only in early steps—otherwise the system stalls at partial hydrogenation and the realized H/S ratio falls below theoretical values. Moreover, the H/S ratio is tightly coupled to operational and system considerations. High H/S ratios necessitate catalysts capable of handling elevated hydrogen pressures and multiple turnovers without degradation, and they influence reactor design, mass transfer, and thermodynamics; incomplete attainment of the target H/S ratio decreases the effective hydrogen density and compromises cyclic reversibility. In this context, homogeneous catalyst designs that can sustain high conversion and selectivity across full hydrogenation–dehydrogenation cycles are critical for translating LOHC chemistry into practical hydrogen storage media.
Additionally, rational catalyst design has yet to fully integrate computational methodologies. Advanced computational modelling and mechanistic studies could significantly enhance catalyst development, enabling the targeted design of more stable, efficient, and selective catalytic systems. The synergy between experimental and theoretical approaches can accelerate breakthroughs in LOHC technology.
In conclusion, recommendations for future research should include the following. Looking ahead, research should focus on developing new LOHC systems that can meet the US DOE's requirements. Additionally, priority should be placed on rational catalyst design, incorporating computational methods, and bridging the gap between laboratory-scale innovations and large-scale implementations. Further, advancing the understanding of structure–activity relationships in catalysis, along with innovations in green chemistry, can also promote the sustainable and widespread adoption of LOHC technologies.
Research advancements have revealed that in some homogeneous (de)hydrogenation reactions, the true active species are in situ-generated soluble metal nanoclusters or colloidal nanoparticles (NPs). These species often exhibit high catalytic activity, even for challenging substrates. For instance, the full hydrogenation of quinoline to DHQ has been challenging with homogeneous catalysts alone. However, recent studies indicate that dual catalysis, where a molecular complex facilitates heteroarene hydrogenation while in situ-generated Ru NPs promote arene hydrogenation, can achieve this transformation. The stabilization of such NPs and clusters by ligands, additives, and/or solvents leads to hybrid catalytic systems that integrate features of both homogeneous and heterogeneous catalysis. The rational design of these hybrid catalysts represents a promising avenue for enhancing LOHC technology and improving its industrial viability.
By addressing these challenges, the future of LOHC systems holds immense promise for advancing the hydrogen economy and achieving sustainable energy solutions. Continued research efforts in catalyst innovation, process optimization, and computational modelling will be critical in realizing the full potential of LOHCs as a cornerstone of next-generation energy storage and hydrogen transport technologies.
Author contributions
Juliana Mana Edor: writing – original draft, review & editing. Gershon Amenuvor: writing – original draft, review & editing. Phillimon Modisha: writing – review & editing, and supervision. Dmitri Bessarabov: conceptualization and resources.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analysed as part of this review.Acknowledgements
We are grateful to the North-West University, Hydrogen South Africa (HySA Infrastructure CoC), and the Kwame Nkrumah University of Science and Technology for their support.References
- T.-H. Le, N. Tran and H.-J. Lee, Development of Liquid Organic Hydrogen Carriers for Hydrogen Storage and Transport, Int. J. Mol. Sci., 2024, 25, 1359 CrossRef CAS PubMed.
- V. Tietze, S. Luhr and D. Stolten, Bulk Storage Vessels for Compressed and Liquid Hydrogen, In Hydrogen Science and Engineering: Materials, Processes, Systems and Technology, ed. D. Stolten and B. Emonts, Wiley, 2016, pp. 659–690 Search PubMed.
- A. Alekseev, Hydrogen Liquefaction, In Hydrogen Science and Engineering: Materials, Processes, Systems and Technology, ed. D. Stolten and B. Emonts, Wiley, 2016, pp. 733–762 Search PubMed.
- S. J. Wang, Z. Y. Zhang, Y. Tan, K. X. Liang and S. H. Zhang, Review on the characteristics of existing hydrogen energy storage technologies, Energy Sources, Part A, 2023, 45, 985 CrossRef CAS.
- S. Satyapal, J. Petrovic, C. Read, G. Thomas and G. Ordaz, The U.S. Department of Energy's National Hydrogen Storage Project: Progress towards Meeting Hydrogen-Powered Vehicle Requirements, Catal. Today, 2007, 120, 246 CrossRef CAS.
- Z. Wang, I. Tonks, J. Belli and C. M. Jensen, Dehydrogenation of N-Ethyl Perhydrocarbazole Catalyzed by PCP Pincer Iridium Complexes: Evaluation of a Homogeneous Hydrogen Storage System, J. Organomet. Chem., 2009, 694, 2854 CrossRef CAS.
- A. Ali, M. Usman, H. M. Ali, U. Sajjad, Md. A. Aziz and M. N. Shaikh, Bayesian surrogate assisted neural network model to predict the hydrogen storage in 9-ethylcarbazole, Int. J. Thermofluids, 2025, 27, 101270 Search PubMed.
- J.-Y. Cho, H. Kim, J.-E. Oh and B. Y. Park, Recent Advances in Homogeneous/Heterogeneous Catalytic Hydrogenation and Dehydrogenation for Potential Liquid Organic Hydrogen Carrier (LOHC) Systems, Catalysts, 2021, 11, 1497 Search PubMed.
- P. M. Modisha, C. N. M. Ouma, R. Garidzirai, P. Wasserscheid and D. Bessarabov, The Prospect of Hydrogen Storage Using Liquid Organic Hydrogen Carriers, Energy Fuels, 2019, 33, 2778 CrossRef CAS.
- P. Modisha, P. Gqogqa, R. Garidzirai, C. N. M. Ouma and D. Bessarabov, Evaluation of Catalyst Activity for Release of Hydrogen from Liquid Organic Hydrogen Carriers, Int. J. Hydrogen Energy, 2019, 44, 21926 Search PubMed.
- P. Modisha, R. Garidzirai, H. Güneş, S. E. Bozbag, S. Rommel, E. Uzunlar, M. Aindow, C. Erkey and D. Bessarabov, A Promising Catalyst for the Dehydrogenation of Perhydro-Dibenzyltoluene: Pt/Al2O3 Prepared by Supercritical CO2 Deposition, Catalysts, 2022, 12, 489 Search PubMed.
- J. M. Edor, M. C. Joseph, J. H. L. Jordaan, H. C. M. Vosloo and A. J. Swarts, Formic Acid Dehydrogenation Catalysis Using Novel Pyridyl-Formamidine Half-Sandwich Ruthenium(II) Complexes, Appl. Organomet. Chem., 2025, 39, e70016 Search PubMed.
- P. Bachmann, J. Steinhauer, F. Späth, F. Düll, U. Bauer, R. Eschenbacher, F. Hemauer, M. Scheuermeyer, A. Bösmann, M. Büttner, C. Neiß, A. Görling, P. Wasserscheid, H.-P. Steinrück and C. Papp, Dehydrogenation of the Liquid Organic Hydrogen Carrier System 2-Methylindole/2-Methylindoline/2-Methyloctahydroindole on Pt(111), J. Chem. Phys., 2019, 151, 144711 CrossRef PubMed.
- K. C. Tan, Y. S. Chua, T. He and P. Chen, Strategies of Thermodynamic Alternation on Organic Hydrogen Carriers for Hydrogen Storage Application: A Review, Green Energy Resour., 2023, 1, 100020 CrossRef.
- Q. Du, L. Liu and T. Zhou, General and Efficient Copper-Catalyzed Oxazaborolidine Complex in Transfer Hydrogenation of Isoquinolines under Mild Conditions, ACS Omega, 2020, 5, 21219 CrossRef CAS PubMed.
- N. Brückner, K. Obesser, A. Bösmann, D. Teichmann, W. Arlt, J. Dungs and P. Wasserscheid, Evaluation of Industrially Applied Heat-Transfer Fluids as Liquid Organic Hydrogen Carrier Systems, ChemSusChem, 2014, 7, 229 CrossRef PubMed.
- H. S. Alghamdi, A. Ali, A. M. Ajeebi, A. Jedidi, M. Sanhoob, M. Aktary, A. H. Shabi, M. Usman, W. Alghamdi, S. Alzahrani, Md. A. Aziz and M. N. Shaikh, Catalysts for Liquid Organic Hydrogen Carriers (LOHCs): Efficient Storage and Transport for Renewable Energy, Chem. Rec., 2024, 24, e202400082 CrossRef CAS PubMed.
- D. Wei, X. Shi, R. Qu, K. Junge, H. Junge and M. Beller, Toward a Hydrogen Economy: Development of Heterogeneous Catalysts for Chemical Hydrogen Storage and Release Reactions, ACS Energy Lett., 2022, 7, 3734 Search PubMed.
- Y. Zhang, J. Wang, F. Zhou and J. Liu, An effective strategy for hydrogen supply: catalytic acceptorless dehydrogenation of N-heterocycles, Catal. Sci. Technol., 2021, 11, 3990 Search PubMed.
- Y. Li, X. Guo, S. Zhang and Y. A. He, Perspective Review on N-Heterocycles as Liquid Organic Hydrogen Carriers and Their Hydrogenation/Dehydrogenation Catalysts, Energy Fuels, 2024, 38, 12447 CrossRef CAS.
- T. Yamada, K. Park and H. Sajiki, Homogeneously Catalyzed Aromatic Reduction, In Industrial Arene Chemistry: Markets, Technologies, Sustainable Processes and Case Studies of Aromatic Commodities, ed. J. Mortier and M. Maas-Brunner, Wiley-VCH, Weinheim, 2023, pp. 849–881 Search PubMed.
- A. Lin and G. Bagnato, Revolutionising energy storage: The Latest Breakthrough in liquid organic hydrogen carriers, Int. J. Hydrogen Energy, 2024, 63, 315 CrossRef CAS.
- A. Kumar, T. Janes, N. A. Espinosa-Jalapa and D. Milstein, Selective Hydrogenation of Cyclic Imides to Diols and Amines and Its Application in the Development of a Liquid Organic Hydrogen Carrier, J. Am. Chem. Soc., 2018, 140, 7453 CrossRef CAS PubMed.
- Y. Xie, P. Hu, Y. Ben-David and D. Milstein, A Reversible Liquid Organic Hydrogen Carrier System Based on Methanol-Ethylenediamine and Ethylene Urea, Angew. Chem., Int. Ed., 2019, 58, 5105 CrossRef CAS PubMed.
- B. Loges, A. Boddien, H. Junge and M. Beller, Controlled Generation of Hydrogen from Formic Acid Amine Adducts at Room Temperature and Application in H2/O2 Fuel Cells, Angew. Chem., Int. Ed., 2008, 47, 3962 CrossRef CAS PubMed.
- D. R. Palo, R. A. Dagle and J. D. Holladay, Methanol Steam Reforming for Hydrogen Production, Chem. Rev., 2007, 107, 3992 CrossRef CAS PubMed.
- T. He, Q. Pei and P. Chen, Liquid Organic Hydrogen Carriers, J. Energy Chem., 2015, 24, 587 CrossRef.
- T. Shimbayashi and K. Fujita, Metal-Catalyzed Hydrogenation and Dehydrogenation Reactions for Efficient Hydrogen Storage, Tetrahedron, 2020, 76, 130946 CrossRef CAS.
- C. J. Boxwell, P. J. Dyson, D. J. Ellis and T. Welton, A Highly Selective Arene Hydrogenation Catalyst That Operates in Ionic Liquid, J. Am. Chem. Soc., 2002, 124, 9334 CrossRef CAS PubMed.
- V. Arun, N. Sridevi, P. P. Robinson, S. Manju and K. K. M. Yusuff, Ni(II) and Ru(II) Schiff Base Complexes as Catalysts for the Reduction of Benzene, J. Mol. Catal. A:Chem., 2009, 304, 191 CrossRef CAS.
- C. Vangelis, A. Bouriazos, S. Sotiriou, M. Samorski, B. Gutsche and G. Papadogianakis, Catalytic Conversions in Green Aqueous Media: Highly Efficient Biphasic Hydrogenation of Benzene to Cyclohexane Catalyzed by Rh/TPPTS Complexes, J. Catal., 2010, 274, 21 CrossRef CAS.
- J. Meng, F. Zhou, H. Ma, X. Yuan, Y. Wang and J. A. Zhang, Review of Catalysts for Methylcyclohexane Dehydrogenation, Top. Catal., 2021, 64, 509 CrossRef CAS.
- H.-L. Ye, S.-X. Liu, C. Zhang, Y.-Q. Cai and Y.-F. Shi, Dehydrogenation of Methylcyclohexane over Pt-Based Catalysts Supported on Functional Granular Activated Carbon, RSC Adv., 2021, 11, 29287 RSC.
- S. Yolcular and Ö. Olgun, Hydrogen Storage in the Form of Methylcyclohexane, Energy Sources, Part A, 2007, 30, 149 CrossRef.
- P. Gupta, G. Hierlmeier, C. Baete, M. V. Pecoraro, P. Tosatti, K. Puentener and P. J. Chirik, Asymmetric Hydrogenation of Naphthalenes with Molybdenum Catalysts: Ligand Design Improves Chemoselectivity, ACS Catal., 2024, 14, 15545 CrossRef CAS.
- S. Hodoshima, H. Arai, S. Takaiwa and Y. Saito, Catalytic decalin Dehydrogenation/Naphthalene Hydrogenation Pair as a Hydrogen Source for Fuel-Cell Vehicle, Int. J. Hydrogen Energy, 2003, 28, 1255 CrossRef CAS.
- Y. Tuo, L. Yang, H. Cheng, M. Yang, Y.-A. Zhu and P. Li, Density Functional Theory Study of Decalin Dehydrogenation for Hydrogen Release on Pt(111) and Pt(211), Int. J. Hydrogen Energy, 2018, 43, 19575 CrossRef CAS.
- L. M. Kustov, A. L. Tarasov and B. P. Tarasov, Intermetallide Catalysts for Hydrogen Storage on the Basis of Reversible Aromatics Hydrogenation/Dehydrogenation Reactions, Int. J. Hydrogen Energy, 2013, 38, 5713 CrossRef CAS.
- M. Kerscher, T. Klein, P. S. Schulz, E. Veroutis, S. Dürr, P. Preuster, T. M. Koller, M. H. Rausch, I. G. Economou, P. Wasserscheid and A. P. Fröba, Thermophysical Properties of Diphenylmethane and Dicyclohexylmethane as a Reference Liquid Organic Hydrogen Carrier System from Experiments and Molecular Simulations, Int. J. Hydrogen Energy, 2020, 45, 28903 Search PubMed.
- D. J. Han, Y. S. Jo, B. S. Shin, M. Jang, J. W. Kang, J. H. Han, S. W. Nam and C. W. Yoon, A Novel Eutectic Mixture of Biphenyl and Diphenylmethane as a Potential Liquid Organic Hydrogen Carrier: Catalytic Hydrogenation, Energy Technol., 2019, 7, 113 CrossRef CAS.
- B. Bong, C. Mebrahtu, D. Jurado, A. Bösmann, P. Wasserscheid and R. Palkovits, Hydrogen Loading and Release Potential of the LOHC System Benzyltoluene/Perhydro Benzyltoluene over S–Pt/TiO2 Catalyst, ACS Eng. Au, 2024, 4, 359 CrossRef CAS.
- D. Strauch, P. Weiner, B. B. Sarma, A. Körner, E. Herzinger, P. Wolf, A. Zimina, A. Hutzler, D. E. Doronkin, J.-D. Grunwaldt, P. Wasserscheid and M. Wolf, Bimetallic Platinum Rhenium Catalyst for Efficient Low Temperature Dehydrogenation of Perhydro Benzyltoluene, Catal. Sci. Technol., 2024, 14, 1775 Search PubMed.
- A. Leinweber and K. Müller, Hydrogenation of the Liquid Organic Hydrogen Carrier Compound Monobenzyl Toluene: Reaction Pathway and Kinetic Effects, Energy Technol., 2018, 6, 513 Search PubMed.
- R. Garidzirai, P. Modisha, I. Shuro, J. Visagie, P. Van Helden and D. Bessarabov, The Effect of Mg and Zn Dopants on Pt/Al2O3 for the Dehydrogenation of Perhydrodibenzyltoluene, Catalysts, 2021, 11, 490 Search PubMed.
- P. Modisha and D. Bessarabov, Stress Tolerance Assessment of Dibenzyltoluene-Based Liquid Organic Hydrogen Carriers, Sustainable Energy Fuels, 2020, 4, 4662 RSC.
- A. N. Kalenchuk and L. M. Kustov, Activity of Mono-, Bi-, and Trimetallic Catalysts Pt-Ni-Cr/C in the Bicyclohexyl Dehydrogenation Reaction, Molecules, 2022, 27, 8416 CrossRef CAS PubMed.
- L. M. Kustov and A. N. Kalenchuk, Effect of Cr on a Ni-Catalyst Supported on Sibunite in Bicyclohexyl Dehydrogenation in Hydrogen Storage Application, Catalysts, 2022, 12, 1506 Search PubMed.
- A. N. Kalenchuk, V. I. Bogdan, S. F. Dunaev and L. M. Kustov, Effect of Surface Hydrophilization on Pt/Sibunit Catalytic Activity in Bicyclohexyl Dehydrogenation in Hydrogen Storage Application, Int. J. Hydrogen Energy, 2018, 43, 6191 CrossRef CAS.
- C. Chu, K. Wu, B. Luo, Q. Cao and H. Zhang, Hydrogen Storage by Liquid Organic Hydrogen Carriers: Catalyst, Renewable Carrier, and Technology – A Review, Carbon Resour. Convers., 2023, 6, 334 Search PubMed.
- R. Biniwale, S. Rayalu, S. Devotta and M. Ichikawa, Chemical Hydrides: A Solution to High Capacity Hydrogen Storage and Supply, Int. J. Hydrogen Energy, 2008, 33, 360 Search PubMed.
- G. P. Pez, A. R. Scott, A. C. Cooper and H. Cheng, Hydrogen storage by reversible hydrogenation of pi-conjugated substrates, US7101530B2, September 5, 2006, https://patents.google.com/patent/US7101530B2/en, (Accessed 2025-03-10).
- Q.-L. Zhu and Q. Xu, Liquid Organic and Inorganic Chemical Hydrides for High-Capacity Hydrogen Storage, Energy Environ. Sci., 2015, 8, 478 Search PubMed.
- E. Díaz, P. Rapado-Gallego and S. Ordonez, Systematic evaluation of physicochemical properties for the selection of alternative liquid organic hydrogen carriers, J. Energy Storage, 2023, 59, 106511 Search PubMed.
- B. S. Shin, C. W. Yoon, S. K. Kwak and J. W. Kang, Thermodynamic assessment of carbazole-based organic polycyclic compounds for hydrogen storage applications via a computational approach, Int. J. Hydrogen Energy, 2018, 43, 12158 Search PubMed.
- E. Farnetti, R. D. Monte and J. Kašpar, Homogeneous and Heterogeneous Catalysis, Eolss Publishers, Oxford, UK, 2009, vol. 2, p. 502 Search PubMed.
- T. Suárez, A. Guzmán, B. Fontal, M. Reyes, F. Bellandi, R. R. Contreras, P. Cancines, G. León and L. Rojas, Hydrogenation of Aromatics with [Ru(η5-C5H5)Cl(TPPDS)2] in Biphasic Medium, Transition Met. Chem., 2006, 31, 176 CrossRef.
- P. J. Dyson, D. J. Ellis and G. Laurenczy, Minor Modifications to the Ligands Surrounding a Ruthenium Complex Lead to Major Differences in the Way in Which They Catalyse the Hydrogenation of Arenes, Adv. Synth. Catal., 2003, 345, 211 CrossRef CAS.
- A. F. Borowski, L. Vendier, S. Sabo-Etienne, E. Rozycka-Sokolowska and A. V. Gaudyn, Catalyzed Hydrogenation of Condensed Three-Ring Arenes and Their N-Heteroaromatic Analogues by a Bis(Dihydrogen) Ruthenium Complex, Dalton Trans., 2012, 41, 14117 RSC.
- B. Chatterjee, D. Kalsi, A. Kaithal, A. Bordet, W. Leitner and C. Gunanathan, One-Pot Dual Catalysis for the Hydrogenation of Heteroarenes and Arenes, Catal. Sci. Technol., 2020, 10, 5163 RSC.
- E. L. Muetterties and F. J. Hirsekorn, Homogeneous Catalysis of Aromatic Hydrocarbon Hydrogenation Reactions, J. Am. Chem. Soc., 1974, 96, 4063 Search PubMed.
- P. J. Dyson, Arene Hydrogenation by Homogeneous Catalysts: Fact or Fiction?, Dalton Trans., 2003, 2964 Search PubMed.
- I. P. A. Rothwell, New Generation of Homogeneous Arene Hydrogenation Catalysts, Chem. Commun., 1997, 1331 RSC.
- M. V. Joannou, M. J. Bezdek and P. J. Chirik, Pyridine(Diimine) Molybdenum-Catalyzed Hydrogenation of Arenes and Hindered Olefins: Insights into Precatalyst Activation and Deactivation Pathways, ACS Catal., 2018, 8, 5276 CrossRef CAS.
- G. Hierlmeier, P. Tosatti, K. Puentener and P. J. Chirik, Identification of Cyclohexadienyl Hydrides as Intermediates in Molybdenum-Catalyzed Arene Hydrogenation, Angew. Chem., 2023, 135, e202216026 CrossRef.
- P. Viereck, G. Hierlmeier, P. Tosatti, T. P. Pabst, K. Puentener and P. J. Chirik, Molybdenum-Catalyzed Asymmetric Hydrogenation of Fused Arenes and Heteroarenes, J. Am. Chem. Soc., 2022, 144, 11203 Search PubMed.
- K. J. Klabunde, B. B. Anderson, M. Bader and L. J. Radonovich, Π-Arene Complexes of Nickel(II) Stabilized by Sigma-Bonded pentafluorophenyl Ligands. Homogeneous Arene Hydrogenation Catalysis and Unusually Labile Π-Arene Ligands, J. Am. Chem. Soc., 1978, 100, 1313 Search PubMed.
- A. Gómez-Torres, J. R. Aguilar-Calderón, A. M. Encerrado-Manriquez, M. Pink, A. J. Metta-Magaña, W. Lee and S. Fortier, Titanium-Mediated Catalytic Hydrogenation of Monocyclic and Polycyclic Arenes, Chem. – Eur. J., 2020, 26, 2803 CrossRef PubMed.
- B. Han, P. Ma, X. Cong, H. Chen and X. Zeng, Chromium- and Cobalt-Catalyzed, Regiocontrolled Hydrogenation of Polycyclic Aromatic Hydrocarbons: A Combined Experimental and Theoretical Study, J. Am. Chem. Soc., 2019, 141, 9018 CrossRef CAS PubMed.
- R. Johnson, P. Hu, J. Pugh, R. K. Haridasan and K. Searles, Benzene Hydrogenation Utilizing Organometallic Early Transition Metal Precursors, Catal. Sci. Technol., 2025, 15, 41 RSC.
- C. M. Jensen, Iridium PCP Pincer Complexes: Highly Active and Robust Catalysts for Novel Homogeneous Aliphatic Dehydrogenations, Chem. Commun., 1999, 24, 2443 Search PubMed.
- C. M. Jensen, Hydrogen storage, US6074447A, June 13, 2000, https://patents.google.com/patent/US6074447A/en, (Accessed on 2025-03-10).
- J. Zheng, H. Zhou, C.-G. Wang, E. Ye, J. W. Xu, X. J. Loh and Z. Li, Current Research Progress and Perspectives on Liquid Hydrogen Rich Molecules in Sustainable Hydrogen Storage, Energy Storage Mater., 2021, 35, 695 CrossRef.
- S. P. Verevkin, S. P. Safronov, A. A. Samarov and S. V. Vostrikov, Hydrogen Storage: Thermodynamic Analysis of Alkyl-Quinolines and Alkyl-Pyridines as Potential Liquid Organic Hydrogen Carriers (LOHC), Appl. Sci., 2021, 11, 11758 Search PubMed.
- M. Zhou, Y. Miao, Y. Gu and Y. Xie, Recent Advances in Reversible Liquid Organic Hydrogen Carrier Systems: From Hydrogen Carriers to Catalysts, Adv. Mater., 2024, 36, 2311355 Search PubMed.
- Y. Luo, X. Yue, P. Wei, A. Zhou, X. Kong and S. Alimzhanova, A State-of-the-Art Review of Quinoline Degradation and Technical Bottlenecks, Sci. Total Environ., 2020, 747, 141136 Search PubMed.
- Y.-N. Duan, X. Du, Z. Cui, Y. Zeng, Y. Liu, T. Yang, J. Wen and X. Zhang, Homogeneous Hydrogenation with a Cobalt/Tetraphosphine Catalyst: A Superior Hydride Donor for Polar Double Bonds and N-Heteroarenes, J. Am. Chem. Soc., 2019, 141, 20424 Search PubMed.
- T. Vielhaber, C. Heizinger and C. Topf, Homogeneous Pressure Hydrogenation of Quinolines Effected by a Bench-Stable Tungsten-Based Pre-Catalyst, J. Catal., 2021, 404, 451 Search PubMed.
- K. Michaliszyn, E. S. Smirnova, A. Bucci, V. Martin-Diaconescu and J. Lloret Fillol, Well-defined Nickel P3C Complexes as Hydrogenation Catalysts of N-Heteroarenes Under Mild Conditions, ChemCatChem, 2022, 14, e202200039 Search PubMed.
- V. Papa, J. Fessler, F. Zaccaria, J. Hervochon, P. Dam, C. Kubis, A. Spannenberg, Z. Wei, H. Jiao, C. Zuccaccia, A. Macchioni, K. Junge and M. Beller, Efficient Hydrogenation of N-Heterocycles Catalyzed by NNP–Manganese(I) Pincer Complexes at Ambient Temperature, Chem. – Eur. J., 2023, 29, e202202774 Search PubMed.
- R. Kumar, M. K. Pandey, A. Bhandari and J. Choudhury, Balancing the Seesaw in Mn-Catalyzed N-Heteroarene Hydrogenation: Mechanism-Inspired Catalyst Design for Simultaneous Taming of Activation and Transfer of H2, ACS Catal., 2023, 13, 4824 CrossRef CAS.
- R. Kumar, M. K. Pandey, I. K. Pandey, A. Kumar and J. Choudhury, An Imidazolylidene-Based Mesoionic Carbene–Mn(I) Complex and Its Catalytic Potential in N-Heteroarene Hydrogenation, Eur. J. Inorg. Chem., 2023, 26, e202300411 CrossRef CAS.
- G. E. Dobereiner, A. Nova, N. D. Schley, N. Hazari, S. J. Miller, O. Eisenstein and R. H. Crabtree, Iridium-Catalyzed Hydrogenation of N-Heterocyclic Compounds under Mild Conditions by an Outer-Sphere Pathway, J. Am. Chem. Soc., 2011, 133, 7547 CrossRef CAS PubMed.
- D. Wang and D. Astruc, The Golden Age of Transfer Hydrogenation, Chem. Rev., 2015, 115, 6621 CrossRef CAS PubMed.
- S. Werkmeister, J. Neumann, K. Junge and M. Beller, Pincer-Type Complexes for Catalytic (De)Hydrogenation and Transfer (De)Hydrogenation Reactions: Recent Progress, Chem. – Eur. J., 2015, 21, 12226 CrossRef CAS PubMed.
- Y. Wang, B. Dong, Z. Wang, X. Cong and X. Bi, Silver-Catalyzed Reduction of Quinolines in Water, Org. Lett., 2019, 21, 3631 Search PubMed.
- M. Zhang, B. Han, H. Ma, L. Zhao, J. Wang and Y. Zhang, Hydrosilanes as Hydrogen Source: Iridium-Catalyzed Hydrogenation of N-Heteroarenes, Chin. J. Org. Chem., 2022, 42, 1170 CrossRef CAS.
- Y. Watanabe, T. Ohta, Y. Tsuji, T. Hiyoshi and Y. Tsuji, Ruthenium Catalyzed Reduction of Nitroarenes and Azaaromatic Compounds Using Formic Acid, Bull. Chem. Soc. Jpn., 1984, 57, 2440 CrossRef CAS.
- H. Qi, L. Wang, Q. Sun and W. Sun, Asymmetric Transfer Hydrogenation of Quinoline Derivatives Catalyzed by Chiral Iridium-Imidazoline Complex in Water, Mol. Catal., 2022, 532, 112715 CAS.
- B. Maji and J. Choudhury, Reusable Water-soluble Homogeneous Catalyst in Aqueous-phase Transfer Hydrogenation of N-heteroarenes with Formic Acid: Uracil-based Bifunctional Ir-NHC Catalyst Is the Key, Appl. Organomet. Chem., 2024, 38, e6720 CrossRef CAS.
- J. R. Cabrero-Antonino, R. Adam, K. Junge, R. Jackstell and M. Beller, Cobalt-Catalysed Transfer Hydrogenation of Quinolines and Related Heterocycles Using Formic Acid under Mild Conditions, Catal. Sci. Technol., 2017, 7, 1981 RSC.
- V. Vermaak, H. C. M. Vosloo and A. J. Swarts, Fast and Efficient Nickel(II)-catalysed Transfer Hydrogenation of Quinolines with Ammonia Borane, Adv. Synth. Catal., 2020, 362, 5788 CrossRef CAS.
- W.-G. Jia, L.-L. Gao, Z.-B. Wang, J.-J. Wang, E.-H. Sheng and Y.-F. Han, NHC-Palladium(II) Mononuclear and Binuclear Complexes Containing Phenylene-Bridged Bis(Thione) Ligands: Synthesis, Characterization, and Catalytic Activities, Organometallics, 2020, 39, 1790 CrossRef CAS.
- X. Cui, W. Huang and L. Wu, Zirconium-Hydride-Catalyzed Transfer Hydrogenation of Quinolines and Indoles with Ammonia Borane, Org. Chem. Front., 2021, 8, 5002 Search PubMed.
- A. Maji, S. Gupta, D. Panja, S. Sutradhar and S. Kundu, Hydrogenation of N-Heterocycles by a Well-Defined Phosphine-Free Manganese Catalyst Using Ammonia Borane, Organometallics, 2023, 42, 3385 CrossRef CAS.
- T. Bhatt and K. Natte, Transfer Hydrogenation of N- and O-Containing Heterocycles Including Pyridines with H3N–BH3 Under the Catalysis of the Homogeneous Ruthenium Precatalyst, Org. Lett., 2024, 26, 866 CrossRef CAS PubMed.
- W. Mao, D. Song, J. Guo, K. Zhang, C. Zheng, J. Lin, L. Huang, L. Zheng, W. Zhong and F. Ling, Manganese-Catalyzed Asymmetric Transfer Hydrogenation of Quinolines in Water Using Ammonia Borane as a Hydrogen Source, Green Chem., 2024, 26, 5933 RSC.
- X. Chu, G. Zhou, M. Pang and H. Zhang, Manganese-Catalysed Transfer Hydrogenation of Quinolines under Mild Conditions, Eur. J. Org. Chem., 2023, 26, e202300597 CrossRef CAS.
- K. Fujita, C. Kitatsuji, S. Furukawa and R. Yamaguchi, Regio- and Chemoselective Transfer Hydrogenation of Quinolines Catalyzed by a Cp*Ir Complex, Tetrahedron Lett., 2004, 45, 3215 CrossRef CAS.
- R. He, P. Cui, D. Pi, Y. Sun and H. Zhou, High Efficient Iron-Catalyzed Transfer Hydrogenation of Quinolines with Hantzsch Ester as Hydrogen Source under Mild Conditions, Tetrahedron Lett., 2017, 58, 3571 Search PubMed.
- A. Sau, D. Mahapatra, A. Maji, S. Dey, A. Roy and S. Kundu, Methyl Formate, an Alternative Transfer Hydrogenating Agent for Chemoselective Reduction of N-Heteroarenes and Azoarenes, Org. Lett., 2024, 26, 4486 Search PubMed.
- Y. Gong, J. He, X. Wen, H. Xi, Z. Wei and W. Liu, Transfer Hydrogenation of N-Heteroarenes with 2-Propanol and Ethanol Enabled by Manganese Catalysis, Org. Chem. Front., 2021, 8, 6901 Search PubMed.
- M. P. Wiesenfeldt, Z. Nairoukh, T. Dalton and F. Glorius, Selective Arene Hydrogenation for Direct Access to Saturated Carbo- and Heterocycles, Angew. Chem., Int. Ed., 2019, 58, 10460 CrossRef CAS PubMed.
- A. F. Borowski, S. Sabo-Etienne, B. Donnadieu and B. Chaudret, Reactivity of the Bis(Dihydrogen) Complex [RuH2(η2-H2)2(PCy3)2] toward N-Heteroaromatic Compounds. Regioselective Hydrogenation of Acridine to 1,2,3,4,5,6,7,8-Octahydroacridine, Organometallics, 2003, 22, 1630 CrossRef CAS.
- S. Urban, N. Ortega and F. Glorius, Ligand-Controlled Highly Regioselective and Asymmetric Hydrogenation of Quinoxalines Catalyzed by Ruthenium N-Heterocyclic Carbene Complexes, Angew. Chem., Int. Ed., 2011, 50, 3803 CrossRef CAS PubMed.
- R. Kuwano, R. Ikeda and K. Hirasada, Catalytic Asymmetric Hydrogenation of Quinoline Carbocycles: Unusual Chemoselectivity in the Hydrogenation of Quinolines, Chem. Commun., 2015, 51, 7558 RSC.
- Y. Jin, Y. Makida, T. Uchida and R. Kuwano, Ruthenium-Catalyzed Chemo- and Enantioselective Hydrogenation of Isoquinoline Carbocycles, J. Org. Chem., 2018, 83, 3829 CrossRef CAS PubMed.
- C. Luo, C. Wu, X. Wang, Z. Han, Z. Wang and K. Ding, Ruthenium-Catalyzed Carbocycle-Selective Hydrogenation of Fused Heteroarenes, J. Am. Chem. Soc., 2024, 146, 35043 CrossRef CAS PubMed.
- A. P. Koskin, J. Zhang, O. B. Belskaya, O. A. Bulavchenko, D. A. Konovalova, S. A. Stepanenko, A. V. Ishchenko, I. G. Danilova, V. L. Yurpalov, Y. V. Larichev, R. Kukushkin and P. M. Yeletsky, Efficiency of High-Loaded Nickel Catalysts Modified by Mg in Hydrogen Storage/Extraction Using Quinoline/Decahydroquinoline Pair as LOHC Substrates, J. Magnesium Alloys, 2024, 12, 3245 CrossRef CAS.
- R. Yamaguchi, C. Ikeda, Y. Takahashi and K. Fujita, Homogeneous Catalytic System for Reversible Dehydrogenation−Hydrogenation Reactions of Nitrogen Heterocycles with Reversible Interconversion of Catalytic Species, J. Am. Chem. Soc., 2009, 131, 8410 Search PubMed.
- X.-B. Zhang and Z. A. Xi, Theoretical Study of the Mechanism for the Homogeneous Catalytic Reversible Dehydrogenation−Hydrogenation of Nitrogen Heterocycles, Phys. Chem. Chem. Phys., 2011, 13, 3997 RSC.
- H. Li, J. Jiang, G. Lu, F. Huang and Z.-X. Wang, On the “Reverse Gear” Mechanism of the Reversible Dehydrogenation/Hydrogenation of a Nitrogen Heterocycle Catalyzed by a Cp*Ir Complex: A Computational Study, Organometallics, 2011, 30, 3131 CrossRef CAS.
- M. G. Manas, L. S. Sharninghausen, E. Lin and R. H. Crabtree, Iridium Catalyzed Reversible Dehydrogenation–Hydrogenation of Quinoline Derivatives under Mild Conditions, J. Organomet. Chem., 2015, 792, 184 CrossRef CAS.
- Á. Vivancos, M. Beller and M. Albrecht, NHC-Based Iridium Catalysts for Hydrogenation and Dehydrogenation of N-Heteroarenes in Water under Mild Conditions, ACS Catal., 2018, 8, 17 CrossRef.
- S. Wang, H. Huang, C. Bruneau and C. Fischmeister, Iridium-Catalyzed Hydrogenation and Dehydrogenation of N-Heterocycles in Water under Mild Conditions, ChemSusChem, 2019, 12, 2350 CrossRef CAS PubMed.
- B. Maji, A. Bhandari, D. Bhattacharya and J. Choudhury, Reusable Single Homogeneous Ir(III)–NHC Catalysts for Bidirectional Hydrogenation–Dehydrogenation of N-Heteroarenes in Water, Organometallics, 2022, 41, 1609 CrossRef CAS.
- B. Maji and J. Choudhury, Switchable Hydrogenation with a Betaine-Derived Bifunctional Ir–NHC Catalyst, Chem. Commun., 2019, 55, 4574 RSC.
- P. Sánchez, M. Hernández-Juárez, N. Rendón, J. López-Serrano, L. L. Santos, E. Álvarez, M. Paneque and A. Suárez, Hydrogenation/Dehydrogenation of N-Heterocycles Catalyzed by Ruthenium Complexes Based on Multimodal Proton-Responsive CNN(H) Pincer Ligands, Dalton Trans., 2020, 49, 9583 RSC.
- S. Chakraborty, W. W. Brennessel and W. D. Jones, A Molecular Iron Catalyst for the Acceptorless Dehydrogenation and Hydrogenation of N-Heterocycles, J. Am. Chem. Soc., 2014, 136, 8564 CrossRef CAS PubMed.
- R. Xu, S. Chakraborty, H. Yuan and W. D. Jones, Acceptorless, Reversible Dehydrogenation and Hydrogenation of N-Heterocycles with a Cobalt Pincer Catalyst, ACS Catal., 2015, 5, 6350 CrossRef CAS.
- P. Dahiya, N. Garg, R. Poli and B. Sundararaju, Hydrogenation and Dehydrogenation of N-Heterocycles under Cp*Co(III) Catalysis, Dalton Trans., 2023, 52, 14752 RSC.
- J. Wu, D. Talwar, S. Johnston, M. Yan and J. Xiao, Acceptorless Dehydrogenation of Nitrogen Heterocycles with a Versatile Iridium Catalyst, Angew. Chem., 2013, 125, 7121 CrossRef.
- W. Yao, Y. Zhang, X. Jia and Z. Huang, Selective Catalytic Transfer Dehydrogenation of Alkanes and Heterocycles by an Iridium Pincer Complex, Angew. Chem., Int. Ed., 2014, 53, 1390 CrossRef CAS PubMed.
- J. Jeong, T. Shimbayashi and K. Fujita, Effect of a Substituent in Cyclopentadienyl Ligand on Iridium-Catalyzed Acceptorless Dehydrogenation of Alcohols and 2-Methyl-1,2,3,4- Tetrahydroquinoline, Catalysts, 2019, 9, 846 CrossRef CAS.
- Y. Wang, L. Qian, Z. Huang, G. Liu and Z. Huang, NCP-type Pincer Iridium Complexes Catalyzed Transfer-Dehydrogenation of Alkanes and Heterocycles, Chin. J. Chem., 2020, 38, 837 CrossRef CAS.
- S. Muthaiah and S. H. Hong, Acceptorless and Base-Free Dehydrogenation of Alcohols and Amines Using Ruthenium-Hydride Complexes, Adv. Synth. Catal., 2012, 354, 3045 CrossRef CAS.
- A. E. Wendlandt and S. S. Stahl, Modular o-Quinone Catalyst System for Dehydrogenation of Tetrahydroquinolines under Ambient Conditions, J. Am. Chem. Soc., 2014, 136, 11910 CrossRef CAS PubMed.
- Q. Wang, H. Chai and Z. Yu, Acceptorless Dehydrogenation of N-Heterocycles and Secondary Alcohols by Ru(II)-NNC Complexes Bearing a Pyrazoyl-Indolyl-Pyridine Ligand, Organometallics, 2018, 37, 584 CrossRef CAS.
- D. Manikpuri, D. R. Pradhan, B. Chatterjee and C. Gunanathan, Ruthenium-Catalyzed Acceptorless Dehydrogenation of Heterocycles, J. Chem. Sci., 2022, 134, 112 CrossRef CAS.
- Y. Wang, C. Li and J. Huang, External-Ligand-Free Aerobic Oxidation of N- and C-Containing Cyclic Systems under Pd-Catalyzed Conditions, Asian J. Org. Chem., 2017, 6, 44 CrossRef CAS.
- H. Yoo, Y. Yang, S. L. Kim, S. H. Son, Y. H. Jang, J. Shin and N. Kim, Syntheses of 1-H-Indoles, Quinolines, and 6-Membered Aromatic N-Heterocycle-Fused Scaffolds via Palladium(II)-Catalyzed Aerobic Dehydrogenation under Alkoxide-Free Conditions, Chem. – Asian J., 2021, 16, 3469 CrossRef CAS PubMed.
- D. Jung, M. H. Kim and J. Kim, Cu-Catalyzed Aerobic Oxidation of Di-Tert-Butyl Hydrazodicarboxylate to Di-Tert-Butyl Azodicarboxylate and Its Application on Dehydrogenation of 1,2,3,4-Tetrahydroquinolines under Mild Conditions, Org. Lett., 2016, 18, 6300 Search PubMed.
- Y. Shen, F. Chen, Z. Du, H. Zhang, J. Liu and N. Liu, Cu(I) Complexes Catalyzed the Dehydrogenation of N-Heterocycles, J. Org. Chem., 2024, 89, 4530 CrossRef CAS PubMed.
- N. Zumbrägel, M. Sako, S. Takizawa, H. Sasai and H. Gröger, Vanadium-Catalyzed Dehydrogenation of N-Heterocycles in Water, Org. Lett., 2018, 20, 4723 CrossRef PubMed.
- S. Bera, A. Bera and D. Banerjee, Nickel-Catalyzed Dehydrogenation of N-Heterocycles Using Molecular Oxygen, Org. Lett., 2020, 22, 6458 CrossRef CAS PubMed.
- A. Ali and M. N. Shaikh, Recent Developments in Catalyst Design for Liquid Organic Hydrogen Carriers: Bridging the Gap to Affordable Hydrogen Storage, Int. J. Hydrogen Energy, 2024, 78, 1 CrossRef CAS.
- D. F. Brayton and C. M. Jensen, Solvent Free Selective Dehydrogenation of Indolic and Carbazolic Molecules with an Iridium Pincer Catalyst, Chem. Commun., 2014, 50, 5987 Search PubMed.
- P. Rao and M. Yoon, Potential Liquid-Organic Hydrogen Carrier (LOHC) Systems: A Review on Recent Progress, Energies, 2020, 13, 6040 CrossRef CAS.
- S. A. Stepanenko, D. M. Shivtsov, A. P. Koskin, I. P. Koskin, R. G. Kukushkin, P. M. Yeletsky and V. A. Yakovlev, N-Heterocyclic Molecules as Potential Liquid Organic Hydrogen Carriers: Reaction Routes and Dehydrogenation Efficacy, Catalysts, 2022, 12, 1260 Search PubMed.
- A. Søgaard, M. Scheuermeyer, A. Bösmann, P. Wasserscheid and A. Riisager, Homogeneously-Catalysed Hydrogen Release/Storage Using the 2-Methylindole/2- Methylindoline LOHC System in Molten Salt-Organic Biphasic Reaction Systems, Chem. Commun., 2019, 55, 2046 RSC.
- L. Li, M. Yang, Y. Dong, P. Mei and H. Cheng, Hydrogen Storage and Release from a New Promising Liquid Organic Hydrogen Storage Carrier (LOHC): 2-Methylindole, Int. J. Hydrogen Energy, 2016, 41, 16129 CrossRef CAS.
- D.-S. Wang, Q.-A. Chen, W. Li, C.-B. Yu, Y.-G. Zhou and X. Zhang, Pd-Catalyzed Asymmetric Hydrogenation of Unprotected Indoles Activated by Brønsted Acids, J. Am. Chem. Soc., 2010, 132, 8909 CrossRef CAS PubMed.
- R. Kuwano, K. Sato, T. Kurokawa, D. Karube and Y. Ito, Catalytic Asymmetric Hydrogenation of Heteroaromatic Compounds, Indoles, J. Am. Chem. Soc., 2000, 122, 7614 CrossRef CAS.
- R. Kuwano, K. Kaneda, T. Ito, K. Sato, T. Kurokawa and Y. Ito, Highly Enantioselective Synthesis of Chiral 3-Substituted Indolines by Catalytic Asymmetric Hydrogenation of Indoles, Org. Lett., 2004, 6, 2213 CrossRef CAS PubMed.
- R. Kuwano, M. Kashiwabara, K. Sato, T. Ito, K. Kaneda and Y. Ito, Catalytic Asymmetric Hydrogenation of Indoles Using a Rhodium Complex with a Chiral Bisphosphine Ligand PhTRAP, Tetrahedron: Asymmetry, 2006, 17, 521 Search PubMed.
- A. Baeza and A. Pfaltz, Iridium-Catalyzed Asymmetric Hydrogenation of N-Protected Indoles, Chem. – Eur. J., 2010, 16, 2036 CrossRef CAS PubMed.
- J. L. Núñez-Rico, H. Fernández-Pérez and A. Vidal-Ferran, Asymmetric Hydrogenation of Unprotected Indoles Using Iridium Complexes Derived from P–OP Ligands and (Reusable) Brønsted Acids, Green Chem., 2014, 16, 1153 RSC.
- J. Wen, X. Fan, R. Tan, H.-C. Chien, Q. Zhou, L. W. Chung and X. Zhang, Brønsted-Acid-Promoted Rh-Catalyzed Asymmetric Hydrogenation of N-Unprotected Indoles: A Cocatalysis of Transition Metal and Anion Binding, Org. Lett., 2018, 20, 2143 Search PubMed.
- Z. Wang, J. Belli and C. M. Jensen, Homogeneous Dehydrogenation of Liquid Organic Hydrogen Carriers Catalyzed by an Iridium PCP Complex, Faraday Discuss., 2011, 151, 297 Search PubMed.
- T. Zhang, Y. Lv, Z. Zhang, Z. Jia and T.-P. Loh, A Rare Earth Metal Catalyzed Aerobic Dehydrogenation of N-Heterocycles, Org. Lett., 2023, 25, 4468 Search PubMed.
- D. Kawauchi, K. Noda, Y. Komatsu, K. Yoshida, H. Ueda and H. Tokuyama, Aerobic Dehydrogenation of N-Heterocycles with Grubbs Catalyst: Its Application to Assisted-Tandem Catalysis to Construct N-Containing Fused Heteroarenes, Chem. – Eur. J., 2020, 26, 15793 Search PubMed.
- M. A. Esteruelas, V. Lezáun, A. Martínez, M. Oliván and E. Oñate, Osmium Hydride Acetylacetonate Complexes and Their Application in Acceptorless Dehydrogenative Coupling of Alcohols and Amines and for the Dehydrogenation of Cyclic Amines, Organometallics, 2017, 36, 2996 CrossRef CAS.
- V. Zubar, J. C. Borghs and M. Rueping, Hydrogenation or Dehydrogenation of N-Containing Heterocycles Catalyzed by a Single Manganese Complex, Org. Lett., 2020, 22, 3974 CrossRef CAS PubMed.
- K. Fujita, Y. Tanaka, M. Kobayashi and R. Yamaguchi, Homogeneous Perdehydrogenation and Perhydrogenation of Fused Bicyclic N-Heterocycles Catalyzed by Iridium Complexes Bearing a Functional Bipyridonate Ligand, J. Am. Chem. Soc., 2014, 136, 4829 CrossRef CAS PubMed.
- K. Fujita, T. Wada and T. Shiraishi, Reversible Interconversion between 2,5-Dimethylpyrazine and 2,5-Dimethylpiperazine by Iridium-Catalyzed Hydrogenation/Dehydrogenation for Efficient Hydrogen Storage, Angew. Chem., Int. Ed., 2017, 56, 10886 CrossRef CAS PubMed.
- D. Teichmann, K. Stark, K. Müller, G. Zöttl, P. Wasserscheid and W. Arlt, Energy storage in residential and commercial buildings via Liquid Organic Hydrogen Carriers (LOHC), Energy Environ. Sci., 2012, 5, 9044 RSC.
- W. Peters, M. Eypasch, T. Frank, J. Schwerdtfeger, C. Körner, A. Bösmanna and P. Wasserscheid, Efficient hydrogen release from perhydro-N-ethylcarbazole using catalyst-coated metallic structures produced by selective electron beam melting, Energy Environ. Sci., 2015, 8, 641 RSC.
- P. Preuster, C. Papp and P. Wasserscheid, Liquid Organic Hydrogen Carriers (LOHCs): Toward a Hydrogen-free Hydrogen Economy, Acc. Chem. Res., 2017, 50, 74 CrossRef CAS PubMed.
- R. H. Morris, Brønsted–Lowry Acid Strength of Metal Hydride and Dihydrogen Complexes, Chem. Rev., 2016, 116, 8588 CrossRef CAS PubMed.
- R. H. Crabtree, Homogeneous Transition Metal Catalysis of Acceptorless Dehydrogenative Alcohol Oxidation: Applications in Hydrogen Storage and to Heterocycle Synthesis, Chem. Rev., 2017, 117, 9228 CrossRef CAS PubMed.
- M. Grasemann and G. Laurenczy, Formic acid as a hydrogen source – recent developments and future trends, Energy Environ. Sci., 2012, 5, 8171 RSC.
- D. Jankovič, M. Mihelač, Ž. Testen, B. Likozar, M. Huš and M. Gazvoda, Mechanistically Guided Development of Homogenous Nickel Catalysis through Rapid Computational Catalyst Screening, J. Catal., 2024, 429, 115265 CrossRef.
- K. Wu and A. G. Doyle, Parameterization of Phosphine Ligands Demonstrates Enhancement of Nickel Catalysis via Remote Steric Effects, Nat. Chem., 2017, 9, 779 CrossRef CAS PubMed.
- B. Cornils and W. A. Herrmann, Concepts in Homogeneous Catalysis: The Industrial View, J. Catal., 2003, 216, 23 Search PubMed.
- A. Bösmann, G. Franciò, E. Janssen, M. Solinas, W. Leitner and P. Wasserscheid, Activation, Tuning, and Immobilization of Homogeneous Catalysts in an Ionic Liquid/Compressed CO2 Continuous-Flow System, Angew. Chem., Int. Ed., 2001, 40, 2697 Search PubMed.
- W. Yang, G. A. Filonenko and E. A. Pidko, Performance of Homogeneous Catalysts Viewed in Dynamics, Chem. Commun., 2023, 59, 1757 RSC.
- G. J. Sunley and D. J. Watson, High Productivity Methanol Carbonylation Catalysis Using Iridium. The CativaTM Process for the Manufacture of Acetic Acid, Catal. Today, 2000, 58, 293 CrossRef CAS.
- M. J. Howard, G. J. Sunley, A. D. Poole, R. J. Watt and B. K. Sharma, New Acetyls Technologies from BP Chemicals, In Studies in Surface Science and Catalysis, Elsevier, 1999, vol. 121, pp. 61–68 Search PubMed.
- M. J. Howard, M. D. Jones, M. S. Roberts and S. A. Taylor, C1 to Acetyls: Catalysis and Process, Catal. Today, 1993, 18, 325 Search PubMed.
- A. E. Lubaev, C. C. Marvin, A. W. Dombrowski and Z. Qureshi, Suzuki–Miyaura Cross-Coupling of Unprotected Ortho-Bromoanilines with Benzyl, Alkyl, Aryl, Alkenyl and Heteroaromatic Boronic Esters, RSC Adv., 2024, 14, 29184 RSC.
- N. Miyaura and K. Yamada, A New Stereospecific Cross-Coupling by the Palladium-Catalyzed Reaction of 1-Alkenylboranes with 1-Alkenyl or L-Alkynyl Halides, Tetrahedron Lett., 1979, 36, 3437 CrossRef.
- S. E. Hooshmand, B. Heidari, R. Sedghi and R. S. Varma, Recent Advances in the Suzuki–Miyaura Cross-Coupling Reaction Using Efficient Catalysts in Eco-Friendly Media, Green Chem., 2019, 21, 381 RSC.
- T. E. Barder and S. L. Buchwald, Insights into Amine Binding to Biaryl Phosphine Palladium Oxidative Addition Complexes and Reductive Elimination from Biaryl Phosphine Aryl palladium Amido Complexes via Density Functional Theory, J. Am. Chem. Soc., 2007, 129, 12003 CrossRef CAS PubMed.
- A. K. King, A. Brar, G. Li and M. Findlater, Homogeneous and Recyclable Palladium Catalysts: Application in Suzuki–Miyaura Cross-Coupling Reactions, Organometallics, 2023, 42, 2353 CrossRef CAS.
- Y.-K. Jeon, J.-Y. Lee, S.-E. Kim and W.-S. Kim, Highly Selective Room-Temperature Suzuki–Miyaura Coupling of Bromo-2-Sulfonyloxypyridines for Unsymmetrical Diarylpyridines, J. Org. Chem., 2020, 85, 7399 CrossRef CAS PubMed.
- M. J. H. Russell, Water Soluble Rhodium Catalysts. A Hydroformylation System for the Manufacture of Aldehydes for the Fine Chemicals Market, Platinum Met. Rev., 1988, 32, 179 CrossRef CAS.
- G. D. Frey, 75 Years of Oxo Synthesis – The Success Story of a Discovery at the OXEA Site Ruhrchemie, J. Organomet. Chem., 2014, 754, 5 CrossRef CAS.
- G. G. Stanley, Hydroformylation (OXO) catalysis, Kirk-Othmer Encyclopedia of Chemical Technology, 2000, pp. 1–19 Search PubMed.
- B. Cornils and E. G. Kuntz, Introducing TPPTS and Related Ligands for Industrial Biphasic Processes, J. Organomet. Chem., 1995, 502, 177 CrossRef CAS.
- C. Crause, L. Bennie, L. Damoense, C. L. Dwyer, C. Grove, N. Grimmer, W. J. van Rensburg, M. M. Kirk, K. M. Mokheseng, S. Otto and P. J. Steynberg, Bicyclic Phosphines as Ligands for Cobalt-Catalysed Hydroformylation, Dalton Trans., 2003, 10, 2036 RSC.
- D. M. Hood, R. A. Johnson, A. E. Carpenter, J. M. Younker, D. J. Vinyard and G. G. Stanley, Highly Active Cationic Cobalt(II) Hydroformylation Catalysts, Science, 2020, 548, 542 CrossRef PubMed.
- D. M. Hood, R. A. Johnson, D. J. Vinyard, F. R. Fronczek and G. G. Stanley, Cationic Cobalt(II) Bisphosphine Hydroformylation Catalysis: In Situ Spectroscopic and Reaction Studies, J. Am. Chem. Soc., 2023, 145, 19715 CrossRef CAS PubMed.
- T. A. Fassbach, J.-M. Ji, A. J. Vorholt and W. Leitner, Recycling of Homogeneous Catalysts–Basic Principles, Industrial Practice, and Guidelines for Experiments and Evaluation, ACS Catal., 2024, 14, 7289 CrossRef CAS.
- M. Emura and H. A. Matsuda, Green and Sustainable Approach: Celebrating the 30th Anniversary of the Asymmetric l-Menthol Process, Chem. Biodiversity, 2014, 11, 1688 CrossRef CAS PubMed.
- J. H. Jones, The Cativa™ process for the manufacture of acetic acid, Platinum Met. Rev., 2000, 44, 94 CrossRef CAS.
- V. Fábos, L. T. Mika and I. T. Horváth, Selective Conversion of Levulinic and Formic Acids to γ-Valerolactone with the Shvo Catalyst, Organometallics, 2014, 33, 181 CrossRef.
- F. Valentini, G. Brufani, B. Di Erasmo and L. Vaccaro, γ-Valerolactone (GVL) as a Green and Efficient Dipolar Aprotic Reaction Medium, Curr. Opin. Green Sustainable Chem., 2022, 36, 100634 CrossRef CAS.
- G. Amenuvor, J. Darkwa and B. C. Makhubela, Homogeneous Polymetallic Ruthenium(II)^Zinc(II) Complexes: Robust Catalysts for the Efficient Hydrogenation of Levulinic Acid to γ-valerolactone, Catal. Sci. Technol., 2018, 8, 2370 RSC.
- A. A. Kiss, Novel Catalytic Reactive Distillation Processes for a Sustainable Chemical Industry, Top. Catal., 2019, 62, 1132 CrossRef CAS.
- G. J. Harmsen, Reactive Distillation: The Front-Runner of Industrial Process Intensification, Chem. Eng. Process., 2007, 46, 774 CrossRef CAS.
- R. Muthia, A. G. T. Reijneveld, A. G. J. Van Der Ham, A. J. B. Ten Kate, G. Bargeman, S. R. A. Kersten and A. A. Kiss, Novel Method for Mapping the Applicability of Reactive Distillation, Chem. Eng. Process., 2018, 128, 263 CrossRef CAS.
- B. Cornils, W. A. Herrmann and R. W. Eckl, Industrial Aspects of Aqueous Catalysis, J. Mol. Catal. A:Chem., 1997, 116, 27 CrossRef CAS.
- B. Miehlich, A. Savin, H. Stoll and H. Preuss, Results Obtained with the Correlation Energy Density Functionals of Becke and Lee, Yang and Parr, Chem. Phys. Lett., 1989, 157, 200 CrossRef CAS.
- H. Bahrmann, K. Bergrath, H.-J. Kleiner, P. Lappe, C. Naumann, D. Peters and D. Regnat, BINAS-Synthesis and Use of a New Ligand for Propylene Hydroformylation, J. Organomet. Chem., 1996, 520, 97 CrossRef.
- H. Mao, H. Yu, J. Chen and X. Liao, Biphasic Catalysis Using Amphiphilic Polyphenols-Chelated Noble Metals as Highly Active and Selective Catalysts, Sci. Rep., 2013, 3, 2226 Search PubMed.
- F. Joó, Biphasic Catalysis−Homogeneous, In Encyclopedia of Catalysis, ed. I. Horváth, Wiley, 2010 Search PubMed.
- H. Olivier-Bourbigou and F. Hugues, Applications of Ionic Liquids to Biphasic Catalysis, In Green Industrial Applications of Ionic Liquids, ed. R. D. Rogers, K. R. Seddon and S. Volkov, Springer Netherlands, Dordrecht, 2003, pp. 67–84 Search PubMed.
- T. J. Geldbach and P. J. A. Dyson, Versatile Ruthenium Precursor for Biphasic Catalysis and Its Application in Ionic Liquid Biphasic Transfer Hydrogenation: Conventional vs Task-Specific Catalysts, J. Am. Chem. Soc., 2004, 126, 8114 CrossRef CAS PubMed.
- Q. Chu, M. S. Yu and D. P. Curran, New Fluorous/Organic Biphasic Systems Achieved by Solvent Tuning, Tetrahedron, 2007, 63, 9890 Search PubMed.
- A. P. Dobbs and M. R. Kimberley, Fluorous Phase Chemistry: A New Industrial Technology, J. Fluorine Chem., 2002, 118, 3 Search PubMed.
- W. Keim, Oligomerization of Ethylene to α-Olefins: Discovery and Development of the Shell Higher Olefin Process (SHOP), Angew. Chem., Int. Ed., 2013, 52, 12492 Search PubMed.
- J. Dreimann, A. Vorholt, M. Skiborowski and A. Behr, Removal of Homogeneous Precious Metal Catalysts via Organic Solvent Nanofiltration, Chem. Eng. Trans., 2016, 47, 343 Search PubMed.
- J. Scarpello, The Separation of Homogeneous Organometallic Catalysts Using Solvent-Resistant Nanofiltration, J. Membr. Sci., 2002, 203, 71 CrossRef CAS.
- M. Janssen, J. Wilting, C. Müller and D. Vogt, Continuous Rhodium-Catalyzed Hydroformylation of 1-Octene with Polyhedral Oligomeric Silsesquioxanes (POSS) Enlarged Triphenylphosphine, Angew. Chem., 2010, 122, 7904 Search PubMed.
- A. V. Volkov, G. A. Korneeva and G. F. Tereshchenko, Organic Solvent Nanofiltration: Prospects and Application, Russ. Chem. Rev., 2008, 77, 983 Search PubMed.
- S. K. Lim, K. Goh, T.-H. Bae and R. Wang, Polymer-Based Membranes for Solvent-Resistant Nanofiltration: A Review, Chin. J. Chem. Eng., 2017, 25, 1653 Search PubMed.
- F. Xu, S. Zhao, J. Song, Y. Peng and B. Su, Organic Solvent Nanofiltration Membrane with In Situ Constructed Covalent Organic Frameworks as Separation Layer, Membranes, 2024, 14, 234 CrossRef CAS PubMed.
- J.-K. Schnoor, M. Fuchs, A. Böcking, M. Wessling and M. A. Liauw, Homogeneous Catalyst Recycling and Separation of a Multicomponent Mixture Using Organic Solvent Nanofiltration, Chem. Eng. Technol., 2019, 42, 2187 CrossRef CAS.
- R. P. Lively and D. S. Sholl, From Water to Organics in Membrane Separations, Nat. Mater., 2017, 16, 276 CrossRef CAS PubMed.
- J. Geens, A. Hillen, B. Bettens, B. Van Der Bruggen and C. Vandecasteele, Solute Transport in Non-aqueous Nanofiltration: Effect of Membrane Material, J. Chem. Technol. Biotechnol., 2005, 80, 1371 CrossRef CAS.
- J. A. Whu, B. C. Baltzis and K. K. Sirkar, Nanofiltration Studies of Larger Organic Microsolutes in Methanol Solutions, J. Membr. Sci., 2000, 170, 159 Search PubMed.
- P. Schmidt and P. Lutze, Characterisation of Organic Solvent Nanofiltration Membranes in Multi-Component Mixtures: Phenomena-Based Modelling and Membrane Modelling Maps, J. Membr. Sci., 2013, 445, 183 CrossRef CAS.
- R. M. Gould, L. S. White and C. R. Wildemuth, Membrane Separation in Solvent Lube Dewaxing, Environ. Prog., 2001, 20, 12 CrossRef CAS.
- J.-K. Schnoor, J. Bettmer, J. Kamp, M. Wessling and M. A. Liauw, Recycling and Separation of Homogeneous Catalyst from Aqueous Multicomponent Mixture by Organic Solvent Nanofiltration, Membranes, 2021, 11, 423 CrossRef CAS PubMed.
- Z. Wen, D. Pintossi, M. Nuño and T. Noël, Membrane-Based TBADT Recovery as a Strategy to Increase the Sustainability of Continuous-Flow Photocatalytic HAT Transformations, Nat. Commun., 2022, 13, 6147 Search PubMed.
- T. Aca Bayrakdar, F. Nahra, D. Ormerod and S. P. Nolan, Integrating Membrane Separation with Gold-catalyzed Carboxylative Cyclization of Propargylamine and Catalyst Recovery via Organic Solvent Nanofiltration, J. Chem. Technol. Biotechnol., 2021, 96, 3371 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |


























