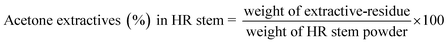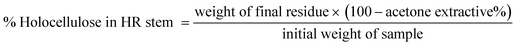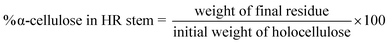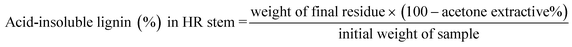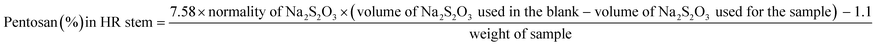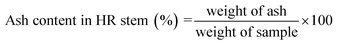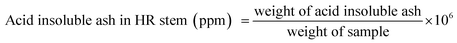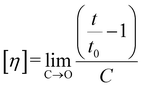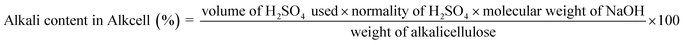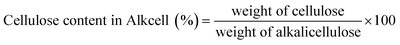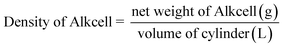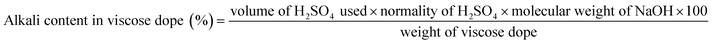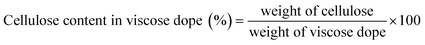Open Access Article
Open Access ArticleNon-wood dissolving pulp from Hibiscus rosasinensis for sustainable viscose fibre production
Sharad P. Pawar ab,
Suyash R. Bhoir
ab,
Suyash R. Bhoir a,
Rupesh A. Kharea and
Shashank T. Mhaske
a,
Rupesh A. Kharea and
Shashank T. Mhaske *b
*b
aPulp and Fibre Innovation Centre (A unit of Grasim Industries), Tal Panvel 410208, India
bDepartment of Polymer and Surface Engineering, Institute of Chemical Technology, Mumbai 400019, India. E-mail: st.mhaske@ictmumbai.edu.in
First published on 14th May 2026
Abstract
This study investigates the potential of Hibiscus rosasinensis stems, a widely cultivated and rapidly growing ornamental plant generating abundant lignocellulosic biomass often discarded as urban and agricultural waste, as a novel non-wood raw material for producing dissolving-grade pulp tailored for regenerated cellulose polymer applications. The bast fibres, rich in cellulose, provide a fast-growing and cost-effective alternative feedstock to conventional wood. Dissolving-grade pulp was obtained via the pre-hydrolysis kraft (PHK) process, combining water-based pre-hydrolysis for hemicellulose removal, kraft cooking for lignin elimination, and subsequent bleaching. The resulting pulp exhibited excellent quality, with α-cellulose 95.2%, hemicellulose 4.8%, residual lignin <0.1%, and intrinsic viscosity of 458 mL g−1. This high-purity pulp was successfully converted into viscose dope through conventional xanthation and regenerated into continuous cellulose polymer fibres via wet spinning. The fibres demonstrated tensile strength, elongation, dyeability, and smooth morphology, confirmed by SEM, comparable to commercial wood-derived viscose rayon, underscoring their suitability for textile applications. By valorising Hibiscus stem waste into functional polymeric fibres, this work establishes a sustainable pathway to reduce dependence on wood resources, mitigate deforestation pressures, and advance circular bioeconomy principles, while harnessing tropical biomass for greener textile industries.
1 Introduction
Hibiscus rosasinensis commonly referred to as Chinese Hibiscus or simply Hibiscus, is a perennial evergreen shrub belonging to the family Malvaceae. Native to tropical and subtropical regions of Asia, it has been widely cultivated globally as an ornamental plant due to its vibrant, showy flowers that bloom year-round in warm climates. The plant typically grows to a height of 2–5 meters, with glossy dark green leaves and a woody stem that supports extensive branching. Beyond its aesthetic appeal, Hibiscus rosasinensis has a long history in traditional medicine across cultures, where various parts including leaves, flowers, and roots are used for their anti-inflammatory, antipyretic, and antioxidant properties. However, the stem, which constitutes a significant portion of the plant's biomass, is often discarded as pruning waste or agricultural residue despite its structural richness in lignocellulosic fibers.The stem of Hibiscus rosasinensis is composed of an outer bark layer and an inner woody core, with bast fibers located in the phloem region beneath the epidermis. These bast fibers are long, flexible, and cellulosic in nature, similar to those found in other Malvaceous plants such as kenaf (Hibiscus cannabinus) and okra (Abelmoschus esculentus).1,2 Chemical composition analyses have revealed that Hibiscus rosasinensis stem contains 45–55% cellulose, 15–20% hemicellulose, 10–15% lignin, 3–5% extractives, and 1–2% ash on a dry weight basis.3,4 The cellulose fraction, particularly the α-cellulose content, has been reported in the range of 38–46%, which is slightly lower to that of softwoods (40–50%) but comparable to certain agricultural residues like corn stover (35–40%). The relatively high cellulose content, combined with lower lignin levels compared to hardwoods (20–25%), highlights its potential as feedstock.5
The global demand for cellulosic fibers continues to rise, driven by population growth, urbanization, and increasing consumption in textiles, packaging, and hygiene products. In recent years, the total fiber consumption reached approximately 118 million metric tons, of which cotton accounted for 22%, polyester 58%, and cellulosic fibers (including viscose, lyocell, and modal) approximately 6.5%. Among regenerated cellulosic fibers, viscose rayon remains the dominant type due to its cost-effectiveness, versatility, and silk-like properties such as high moisture absorbency (10–13%), good dyeability, and soft hand feel.6 The viscose process involves converting high-purity α-cellulose (>90%) into a spinnable dope via xanthation with carbon disulfide (CS2), followed by regeneration in an acid bath to form continuous filaments.7,8 Moreover, cotton, the primary natural cellulosic fiber, requires intensive irrigation (approximately 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 liters per kg of fiber) and pesticide use, exacerbating water scarcity and soil degradation in key producing countries such as India, China, and the United States.9
000 liters per kg of fiber) and pesticide use, exacerbating water scarcity and soil degradation in key producing countries such as India, China, and the United States.9
In response to these challenges, research over the past two decades has focused on alternative biomass sources for pulp and fiber production. Non-wood feedstocks, including agricultural residues (oat husks, potato pulp, rice straw, wheat straw, bagasse), industrial crops (kenaf, jute, hemp), and underutilized plants (bamboo, banana pseudo stem), have been extensively studied for their potential to yield dissolving-grade pulp.10–12 For instance, bamboo pulp has been successfully commercialized in China, with α-cellulose content reaching 92–94% after optimized pulping and bleaching.13,14 Similarly, Banana pseudo stem, a byproduct of banana cultivation, has been shown to produce pulp with 85–90% α-cellulose through soda-anthraquinone pulping, suitable for viscose production.15 Kenaf bast fiber, a close relative of Hibiscus, has been pulped using kraft and soda processes to achieve α-cellulose levels above 90%, with resulting viscose fibers exhibiting tenacity 1.60–1.80 gpd.16
Pulping is the critical first step in liberating cellulose from lignocellulosic biomass. Among various pulping methods, the prehydrolysis kraft (PHK) process has emerged as a preferred route for producing high-purity dissolving pulp due to its ability to selectively remove hemicellulose while preserving cellulose chain length.17 In the PHK process, biomass is subjected to a prehydrolysis stage to hydrolyze and solubilize hemicellulose, followed by alkaline kraft cooking with NaOH and Na2S to delignify the substrate. The resulting pulp undergoes multi-stage bleaching, commonly using chlorine dioxide (D), oxygen (O), and hydrogen peroxide (P) to achieve brightness >88% ISO and α-cellulose >90%.17,18
Despite the extensive literature on alternative feedstocks, Hibiscus rosasinensis stem remains largely unexplored for industrial fiber applications. A few preliminary studies have characterized its fiber morphology and chemical composition. Scanning electron microscopy (SEM) has revealed that Hibiscus rosasinensis bast fibers are multicellular, with individual fiber cells 1.0–2.5 mm in length and 15–25 µm in diameter, featuring a lumen and thick secondary wall typical of bast fibers. Fourier-transform infrared spectroscopy (FTIR) confirms the presence of characteristic cellulose peaks at 3300–3400 cm−1 (O–H stretching), 2900 cm−1 (C–H stretching), and 1050–1100 cm−1 (C–O–C glycosidic linkage), with minimal interference from lignin after alkali treatment. X-ray diffraction (XRD) analysis indicates a cellulose I crystalline structure with crystallinity index (CI) of 55–65%, which is lower than cotton (70–80%) but higher than softwood kraft pulp (45–50%).1
Limited attempts have been made to extract fibers from Hibiscus rosasinensis stem using simple retting or alkali treatment. For example, water retting followed by 5% NaOH treatment at 80 °C for 2 hours yielded fibers with 48% cellulose and tensile strength of 350–450 MPa.19 However, these fibers were coarse and unsuitable for fine textile applications. The stem fibers of Hibiscus rosasinensis were chemically treated with sodium hydroxide (NaOH), potassium permanganate (KMnO4), and acetic acid (CH3COOH), resulting in enhanced crystallinity (up to 65.77%), cellulose enrichment (up to 74%), and improved thermal stability (365 °C), thereby demonstrating their potential as sustainable reinforcements in composite materials. Their work established the potential of Hibiscus stem fibers for composite reinforcement applications.20 No studies to date have applied the PHK pulping process to Hibiscus rosasinensis stem to produce dissolving-grade pulp, nor have they evaluated the downstream conversion of such pulp into viscose fibers. The viscose process necessitates stringent requirements on pulp quality: α-cellulose ≥90%, hemicellulose ≤8%, lignin ≤0.1% and viscosity 400–600 mL g−1.17 Whether Hibiscus rosasinensis stem can meet these criteria remains unknown.
Furthermore, the characterization of both pulp and resulting viscose fibers is essential to establish process feasibility. Pulp properties such as degree of polymerization (DP), viscosity, and reactivity directly influence xanthation efficiency and spinning performance.8 Fiber properties- including tenacity, elongation, fineness, cross-sectional morphology, and crystallinity affect end-use performance in textiles. Comprehensive characterization using techniques such as SEM, FTIR, XRD, DSC, TGA and tensile testing is necessary to benchmark Hibiscus rosasinensis -derived viscose against commercial standards.
The absence of systematic studies on PHK pulping of Hibiscus rosasinensis stem, followed by viscose fiber production and full characterization of pulp and fibers, represents a critical knowledge gap. Without such data, the technical viability and industrial scalability of this feedstock cannot be assessed.
Critically, no investigations have applied the prehydrolysis kraft (PHK) pulping process to this biomass. As a result, essential process parameters, including optimal prehydrolysis temperature and duration, kraft cooking conditions (alkali charge, sulfidity, liquor-to-solid ratio), pulp yield, selectivity of hemicellulose removal, and achievable α-cellulose purity (>90%), remain entirely unknown.
Furthermore, the quality of any potential dissolving pulp derived from Hibiscus rosasinensis has not been evaluated against the stringent criteria required for viscose production. Key metrics such as degree of polymerization (DP) or intrinsic viscosity, residual hemicellulose and lignin content, and bleachability parameters that directly govern xanthation efficiency and spinning performance have not been determined. The downstream conversion of such pulp into viscose dope, followed by wet spinning and fiber regeneration, has likewise never been attempted. Consequently, the mechanical (tenacity, elongation at break) and structural (crystallinity, orientation, cross-sectional morphology) properties of the resulting regenerated fibers are undocumented. Most critically, no comparative assessment exists to benchmark Hibiscus rosasinensis -derived viscose against commercial standards, which demand tenacity, elongation in the required range.
To address these gaps, the present study systematically investigates Hibiscus rosasinensis stem as a novel feedstock for viscose fiber production via the PHK pulping route. The specific objectives are to produce dissolving grade pulp from Hibiscus rosasinensis by PHK pulping process, second objective is to comprehensively characterize the resulting dissolving pulp in terms of chemical composition, molecular weight distribution, viscosity, reactivity, and surface morphology; third objective is to convert the purified pulp into viscose dope and regenerate fibers through standard xanthation and wet-spinning protocols and fourth objective is to fully characterize the regenerated fibers using SEM, FTIR, XRD, DSC, TGA and mechanical testing- tenacity, elongation; by establishing a complete process–structure and property relationship, this work aims to validate Hibiscus rosasinensis stem as a sustainable, waste-derived precursor for high-performance regenerated cellulosic fibers, thereby advancing circular bioeconomy principles in textile materials.
2 Materials and methods
2.1 Materials
Hibiscus rosasinensis stem was used as the lignocellulosic raw material for the production of dissolving-grade pulp. The stems were collected from Sonambe village, Nashik district, Maharashtra, India. All chemicals used for pulping, bleaching, viscose preparation, and fibre spinning were of laboratory grade, while those used for dyeing were of analytical grade and were used without further purification. Sodium hydroxide, sodium sulfide, sulfuric acid, sodium sulfate, and zinc sulfate and bleaching chemicals were received from Grasim industries limited. Carbon disulfide procured from Labort Fine chem. Pvt. Ltd. The reactive dye, Reactofix red HE3BI used for dyeability assessment was procured from local suppliers. Deionized water was used throughout all experiments.2.2 Proximate analysis of Hibiscus rosasinensis stem
The Hibiscus rosasinensis stem chips were analysed for their chemical composition to check their potential as a source for dissolving grape pulp production. Oven-dried stem samples were milled using a Willey mill (supplied by UEC, Saharanpur) and screened through a 0.5 mm pore-sized wired screen. The resulting powder was used for quantification of the chemical composition of the Hibiscus rosasinensis stem. All analyses were performed in triplicate, and results were expressed as mean ± standard error.The extraction apparatus was assembled and heating initiated (maintaining a reflux rate of 4–6 cycles per hour throughout the extraction period). Extraction was continued for a minimum of 6 h, or until the solvent in the siphon tube became colourless, ensuring complete removal of acetone-soluble compounds.
After extraction, the flask was removed and kept in an oven at 105 ± 3 °C for 2 h to eliminate residual solvent and moisture. It was then cooled in a desiccator for 30 min and reweighed to record the weight of the residue.
The acetone extractive content was calculated as a percentage of the dry HR stem powder weight.
The extractive-free powder was then divided in two parts and processed for holocellulose and lignin content estimation.
The resulting residue was filtered by vacuum filtration, thoroughly washed with hot distilled water until free of residual chemicals, and dried at 105 °C to constant weight.
Calculation:
The dried residue, representing acid-insoluble lignin, was weighed.
A blank titration was performed under identical conditions, substituting 270 mL of 13.15% HCl diluted to 350 mL in place of the condensate plus water.
Calculation:
2.3 Preparation of Hibiscus rosasinensis stem for pulping
The collected Hibiscus rosasinensis stems were washed with water to remove dust and adhering impurities, debarked manually, followed by air drying. The dried material was cut into chips of uniform size by chipper (supplied by UEC, Saharanpur, India) to ensure homogeneous chemical penetration during pulping. The moisture content of the prepared chips was determined prior to pulping using moisture analyzer (supplied by Mettler Toledo, India).2.4 Prehydrolysis kraft (PHK) pulping
Prehydrolysis kraft (PHK) pulping was used to produce dissolving-grade pulp from Hibiscus rosasinensis stem following reported procedures for lignocellulosic biomass processing.18,28 Prehydrolysis was conducted in a rotary digester (UEC, Saharanpur, India) equipped with temperature and pressure control at 160 °C for 90 min using a liquor-to-solid ratio of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.28 After prehydrolysis, the liquor was drained and the prehydrolysed chips were collected for subsequent kraft pulping18 Kraft pulping was carried out in the same digester at 160 °C for 60 min using an active alkali charge of 16% (as Na2O), a sulfidity of 25%, and a liquor-to-wood ratio of 3.5
1.28 After prehydrolysis, the liquor was drained and the prehydrolysed chips were collected for subsequent kraft pulping18 Kraft pulping was carried out in the same digester at 160 °C for 60 min using an active alkali charge of 16% (as Na2O), a sulfidity of 25%, and a liquor-to-wood ratio of 3.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.17,28 After cooking, the pulp was washed thoroughly with water until neutral pH was achieved.
1.17,28 After cooking, the pulp was washed thoroughly with water until neutral pH was achieved.
The unbleached pulp was analyzed (see section 2.5) before proceeding for bleaching. The unbleached pulp was bleached using a multistage sequence of O-D1-E1P-Hypo-E2P-D2.17,18,29 The pulp was washed after each bleaching stage and air-dried prior to further processing.
2.5 Analysis of unbleached and bleached pulp
The chemical properties of the unbleached and bleached pulps were determined using standard analytical methods. The holocellulose, alpha cellulose, hemicellulose, viscosity and pentosans content was analyzed as per methods mentioned in section 2.2 above.Intrinsic viscosity [η] was calculated according to TAPPI T230 using the relation
2.6 Preparation of viscose dope
Viscose dope was prepared using a blend of Hibiscus rosasinensis stem dissolving-grade pulp and conventional pulp, with Hibiscus rosasinensis pulp constituting one-tenth of the total pulp content, following the conventional viscose process.6,18,32 The dried pulp blend was steeped in 17.5% (w/w) sodium hydroxide solution at 50 °C for 20 min to form alkali cellulose.6 Excess alkali was removed by pressing, and the alkali cellulose was shredded to increase surface area and analyzed for density, alkali and cellulose content (see 2.6.1 and 2.6.2).The shredded alkali cellulose was aged at 61 °C for 61 min.18,32 Xanthation was carried out by reacting the aged alkali cellulose with carbon disulfide at a dosage of 33% to form cellulose xanthate.8 The xanthated product was dissolved in 2% sodium hydroxide solution to obtain viscose dope. The viscose solution was ripened at 12 °C for 16 h, filtered to remove undissolved particles, analyzed for alkali and cellulose content (see 2.6.3) and deaerated prior to spinning.6,18,32
A 3.0000 g (±0.0001 g) portion of alkali–cellulose was dispersed in 200 mL of distilled water in a 250 mL beaker to form a suspension, which was stirred on a magnetic stirrer to dissolve the alkali for titration. The alkali content was determined by titrating the suspension with 1.0 N H2SO4 using phenolphthalein as an indicator until the color changed from pink to colorless, and the NaOH concentration was calculated from the titration volume. The neutralized cellulose was filtered through a pre-weighed G2 crucible, dried in an oven at 105 °C for 1 h, and the cellulose weight was recorded.
Calculation:
Calculation:
For alkali determination, 5.0000 g (±0.0001 g) of viscose dope was accurately weighed and transferred into a 250 mL beaker. To this, 200 mL of hot distilled water was added, and the mixture was stirred using a magnetic stirrer until complete dissolution of alkali was achieved. The resulting solution was titrated against 1.0 N H2SO4 using methyl red as the indicator. The endpoint was identified by the color change from reddish pink to pale yellow. The concentration of NaOH present in the viscose dope was calculated based on the titration volume.
For cellulose determination, 2.0000 g (±0.0001 g) of viscose dope was weighed onto a 15 × 15 cm toughened glass plate and spread into a thin film by pressing with another plate. The film was regenerated by immersing it in dilute acid, followed by thorough washing with water to remove residual alkali. The regenerated cellulose film was dried in an oven at 105 °C to constant weight, and the final cellulose content was recorded from the measured weight.
2.7 Fibre spinning and regeneration
The viscose dope was spun into fibres using a laboratory-scale wet spinning setup.6,32 Spinning was carried out through a spinneret containing 1900 holes with a diameter of 60 µm. The filaments were extruded into a coagulation bath containing sulfuric acid, sodium sulfate, and zinc sulfate, maintained at 48 °C.6 The regenerated fibres were spinned with 60% stretch, washed thoroughly to remove residual chemicals, neutralized, and dried under controlled conditions.32 The fibres were cut into staple form.2.8 Dyeing procedure
The dyeability of the regenerated viscose fibres was evaluated using reactive dye- Reactofix red HE3BI following established dyeing procedures for regenerated cellulose fibres.33 Dyeing was carried out at a dye concentration of 2.0% owf with a material-to-liquor ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20. The fibre and dye solution heated (2.5 °C min−1) to 80 °C, dyeing cycle continued at 80 °C for 45 min followed by cooling to 40 °C. After dyeing, the fibres were washed sequentially to remove unfixed dye using hot water (3 × 1
20. The fibre and dye solution heated (2.5 °C min−1) to 80 °C, dyeing cycle continued at 80 °C for 45 min followed by cooling to 40 °C. After dyeing, the fibres were washed sequentially to remove unfixed dye using hot water (3 × 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 MLR) at 70 °C, followed by cold water (1 × 1
50 MLR) at 70 °C, followed by cold water (1 × 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 MLR), a 0.1% non-ionic soap wash at 70 °C, additional hot water washing (2 × 1
50 MLR), a 0.1% non-ionic soap wash at 70 °C, additional hot water washing (2 × 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 MLR) at 70 °C, and a final cold-water rinse (1 × 1
50 MLR) at 70 °C, and a final cold-water rinse (1 × 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 MLR). The fibres were dried at 105 °C for 15 min. and further characterized for color strength.
50 MLR). The fibres were dried at 105 °C for 15 min. and further characterized for color strength.
2.9 Characterization of Hibiscus rosasinensis dissolving pulp and viscose fibre
| K/S = (1 − R)2/(2R), |
The CIELAB values (L*, a*, b*) were recorded to evaluate differences in color shades:
L* reflects the lightness or darkness (depth) of the dyed fiber. Positive a* values correspond to increasing redness, while negative values indicate greater greenness. Positive b* values indicate increasing yellowness, whereas negative values reflect greater blueness.34
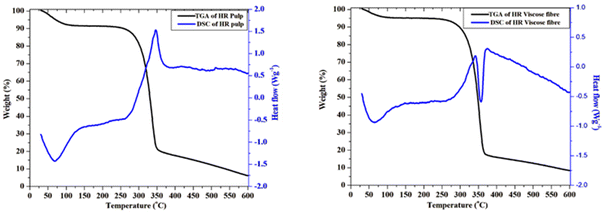
Thermogravimetric analysis (TGA) was conducted to assess the thermal stability, decomposition behaviour, and volatile content of the materials. Samples were heated from 30 °C to 600 °C at a constant heating rate (10 °C min−1) under a nitrogen purge, allowing precise monitoring of mass loss as a function of temperature. This technique provided information on onset decomposition temperatures, multi-step degradation profiles, and residual char/yield at high temperatures.
Simultaneous TGA-DSC analysis was utilized to correlate mass changes with endothermic/exothermic events, offering complementary insights into the thermal degradation mechanisms. All experiments followed standard calibration procedures.
3 Results and discussion
The present study demonstrates, for the first time, the successful utilization of Hibiscus rosasinensis stem, an underexplored lignocellulosic biomass, as a novel raw material for the production of dissolving-grade pulp, its subsequent conversion into viscose fibre, and the fibre quality confirmed through dyeability assessment. The results establish Hibiscus rosasinensis stem as a promising non-wood alternative for regenerated cellulose fibre production.3.1 Potential of Hibiscus rosasinensis stem for dissolving grade pulp production
The chemical composition of Hibiscus rosasinensis stem is presented in Table 1. Holocellulose content in HR stem found to be 76.9%, with α-cellulose at 52.4% which is a favorable level for dissolving grade pulp production. Hemicellulose (21.3%) and pentosan (11.3%) contents were moderately high, consistent with herbaceous biomass but requiring prehydrolysis or hot-water extraction to reduce hemicellulose below 5% for dissolving pulp.17 Acid-insoluble lignin (16.6%) was comparable to some hardwoods, necessitating effective delignification. Acetone extractives (2.9%) and ash (1.64%), are manageable during PHK pulping which minimize interference during bleaching, and viscose fibre production.| Sr. no. | Parameters | Hibiscus rosasinensis stem | Eucalyptus wood chips |
|---|---|---|---|
| 1 | Holocellulose (%) | 76.9 ± 0.9 | 73.9 ± 0.5 |
| 2 | Hemi cellulose (%) | 21.3 ± 1.0 | 21.8 ± 1.3 |
| 3 | Alpha cellulose (%) | 52.4 ± 1.7 | 45.7 ± 1.2 |
| 4 | Acid insoluble lignin (%) | 16.6 ± 1.0 | 27.2 ± 0.6 |
| 5 | Acetone extractives (%) | 2.9 ± 0.1 | 1.9 ± 0.1 |
| 6 | Pentosan (%) | 11.3 ± 0.4 | 16.2 ± 1.2 |
| 7 | Ash (%) | 1.64 ± 0.1 | 0.68 ± 0.1 |
| 8 | Acid insoluble (ppm) | 394.8 ± 16.2 | 100.7 ± 9.7 |
| 9 | Calcium (ppm) | 1072.5 ± 34.8 | 707.7 ± 21.2 |
| 10 | Copper (ppm) | 2.4 ± 0.1 | 1.5 ± 0.4 |
| 11 | Iron (ppm) | 11.6 ± 2.5 | 12.4 ± 2.0 |
| 12 | Magnesium (ppm) | 552.9 ± 21.00 | 210.3 ± 7.4 |
| 13 | Manganese (ppm) | 2.4 ± 0.1 | 32.4 ± 4.8 |
| 14 | Sodium (ppm) | 449.8 ± 38.3 | 62.2 ± 2.9 |
Overall, the high α-cellulose content, combined with acceptably extractives and ash, positions Hibiscus rosasinensis stem as a promising non-wood feedstock for dissolving-grade pulp. Moderate hemicellulose and lignin levels are manageable with established pre-treatment and pulping strategies used for other herbaceous sources, supporting its potential for utilization into regenerated cellulose fibres.
3.2 Dissolving-grade pulp production from Hibiscus rosasinensis stem
The Hibiscus rosasinensis stem was effectively converted into dissolving-grade pulp through prehydrolysis kraft (PHK) pulping and bleaching processes. The pulp yield obtained after PHK pulping was 34.7% and after bleaching the final with 31.2% yield.The resulting pulp exhibited high cellulose purity, indicating efficient removal of hemicellulose and lignin components. The bleached pulp exhibited an α-cellulose content of 95.2%, hemicellulose content of 4.8% and intrinsic viscosity of 458 mL g−1 (Table 2). These values fall within the typical range required for dissolving-grade pulp intended for viscose fibre production.
| Sr. no. | Parameters | Unbleached pulp | Bleached final pulp |
|---|---|---|---|
| 1 | Yield (%) | 34.7 ± 0.2 | 31.2 ± 0.2 |
| 2 | Holocellulose (%) | 97.5 ± 0.2 | 99.2 ± 0.1 |
| 3 | Hemi cellulose (%) | 8.4 ± 0.1 | 4.8 ± 0.1 |
| 4 | Alpha cellulose (%) | 91.6 ± 0.2 | 95.2 ± 0.1 |
| 5 | Pentosan (%) | 4.21 ± 0.0 | 1.98 ± 0.01 |
| 6 | Intrinsic viscosity (mL g−1) | 950 ± 6.9 | 458 ± 4.6 |
| 7 | Kappa number | 14.8 ± 0.1 | NA |
| 8 | Whiteness | −7.57 ± 0.1 | 87.7 ± 0.2 |
| 9 | Brightness | 29.52 ± 0.2 | 94.13 ± 0.2 |
| 10 | Yellowness | 40.68 ± 0.3 | 2.87 ± 0.03 |
| 11 | Ash (%) | 0.308 ± 0.0 | 0.128 ± 0.01 |
| 12 | Acid insoluble (ppm) | 138 ± 1.7 | 126 ± 1.15 |
| 13 | Calcium (ppm) | 3218.8 ± 27.7 | 32.72 ± 0.7 |
| 14 | Copper (ppm) | 13.82 ± 0.2 | 0.28 ± 0.01 |
| 15 | Iron (ppm) | 45.19 ± 0.7 | 10.64 ± 0.17 |
| 16 | Magnesium (ppm) | 75.92 ± 0.8 | 9.14 ± 0.12 |
| 17 | Manganese (ppm) | 24.08 ± 0.3 | 0.22 ± 0.01 |
| 18 | Sodium (ppm) | 732.65 ± 7.2 | 0.7 ± 0.01 |
High alpha cellulose content is a fundamental requirement for dissolving-grade pulp, as non-cellulosic constituents can adversely affect cellulose reactivity and solubility during viscose processing. The low residual lignin content of the pulp confirmed adequate delignification, which is particularly important for viscose manufacturing because residual lignin can interfere with xanthation and result in non-uniform fibre regeneration. The intrinsic viscosity of the pulp suggested a degree of polymerization suitable for viscose fibre production, balancing sufficient chain length for fibre strength with adequate solubility during dissolution.
Compared to conventional wood-based dissolving pulps, the Hibiscus rosasinensis stem pulp demonstrated comparable chemical characteristics, despite originating from a non-wood agricultural residue. This highlights the effectiveness of the applied pulping and bleaching strategy in upgrading Hibiscus rosasinensis stem biomass to dissolving-grade pulp quality. The results confirm that Hibiscus rosasinensis stem, which has received limited attention in regenerated fibre research, can be processed into high-purity cellulose suitable for industrial fibre applications.
3.3 Suitability of Hibiscus rosasinensis pulp for viscose processing
The reactivity of the dissolving-grade pulp is a critical parameter determining its suitability for viscose fibre production. The Hibiscus rosasinensis pulp blend showed good alkali accessibility during steeping, leading to uniform formation of alkali cellulose. The pressed alkali cellulose exhibited an alkali content of 15.31%, cellulose content of 32.30% and density of 176 g l−1 The viscose dope formed with the alkali cellulose exhibited acceptable properties of cellulose content of 9.68%, alkali content of 4.78% thus acceptable alkali to cellulose ratio of 0.496 for spinning and regeneration. The properties of alkali cellulose, viscose dope characteristics, are summarized in Table 3.| Parameter | Analysis stage | Results |
|---|---|---|
| Alkali (%) | Alkali cellulose | 15.31 ± 0.1 |
| Cellulose (%) | Alkali cellulose | 32.30 ± 0.23 |
| Density (gram per liter) | Alkali cellulose | 176 ± 3.46 |
| Alkali (%) | Viscose dope | 4.78 ± 0.01 |
| Cellulose (%) | Viscose dope | 9.68 ± 0.04 |
| Alkali to cellulose (ratio) | Viscose dope | 0.496 ± 0.0 |
| Viscosity (Ball fall in seconds) | Viscose dope | 59 ± 3.06 |
| Filterability | Viscose dope | 92 ± 2.65 |
Efficient steeping is indicative of effective removal of residual non-cellulosic components. During xanthation, the pulp readily reacted with carbon disulphide, forming cellulose xanthate that dissolved uniformly in dilute sodium hydroxide solution. The formation of a homogeneous viscose dope without visible undissolved particles suggests favorable pulp reactivity and accessibility of hydroxyl groups. The dope was filtered to remove any undissolved particles before deareation. This behavior is comparable to that observed for conventional dissolving pulps derived from hardwood or softwood sources. The viscose dope exhibited stable viscosity and satisfactory ripening behavior, indicating controlled depolymerization and molecular rearrangement. Proper ripening is essential to achieve optimal spinnability and fibre uniformity. The results indicate that cellulose derived from Hibiscus rosasinensis stem responds well to conventional viscose processing parameters, without requiring major process modifications.
3.4 Fibre spinning and regeneration behavior
Wet spinning of the viscose dope produced continuous regenerated fibres, demonstrating the spinnability of Hibiscus rosasinensis-based viscose solution. The absence of frequent filament breakage during spinning indicates good dope homogeneity and appropriate coagulation behavior in the spinning bath. The fibres regenerated smoothly in the coagulation bath, suggesting effective cellulose regeneration and molecular reorganization. The washing and neutralization steps successfully removed residual chemicals, resulting in clean regenerated fibres. Visual observation of the fibres indicated uniform appearance without obvious surface defects, further confirming the suitability of Hibiscus rosasinensis stem pulp for fibre formation. The successful fibre regeneration from Hibiscus rosasinensis stem pulp represents a significant outcome of this study, as it confirms that this underutilized biomass can be transformed beyond pulp production into value-added regenerated textile fibres.3.5 Physical and mechanical properties of Hibiscus rosasinensis-based viscose fibre
The mechanical properties of the regenerated viscose fibres derived from Hibiscus rosasinensis stem pulp were evaluated to assess their potential for textile applications. The fibres exhibited a tensile strength of 2.64 gpd and elongation at break of 17.92%, as summarized in Table 4.| Parameter | Results |
|---|---|
| Whiteness (Berger) | 79.73 ± 0.3 |
| Brightness (% ISO) | 83.20 ± 0.5 |
| Viscosity (mL gm−1) | 203.4 ± 1.4 |
| Denier (den) | 1.21 ± 0.0 |
| Tenacity (gpd) | 2.64 ± 0.0 |
| Elongation (%) | 17.92 ± 0.1 |
The fibres exhibited tensile strength and elongation at break values within the typical range reported for regenerated cellulose fibres. The observed tensile strength reflects the degree of polymerization of the starting pulp and the molecular orientation achieved during fibre regeneration and stretching. While regenerated cellulose fibres generally exhibit lower strength than natural fibres, the mechanical performance of the Hibiscus rosasinensis-based viscose fibres suggests adequate intermolecular bonding and chain alignment. The elongation behavior of the fibres indicates good flexibility and amorphous character, which are desirable properties for viscose fibres used in apparel and home textiles. Any minor variations in mechanical performance compared to commercial viscose fibres may be attributed to inherent differences in non-wood fibre morphology or slight variations in cellulose chain length distribution. Overall, the mechanical results confirm that viscose fibres produced from Hibiscus rosasinensis stem dissolving pulp possess sufficient structural integrity for potential textile use.
3.6 Characterization of dissolving grade pulp and viscose fibre
The dissolving grade pulp and regenerated viscose fibre were characterized using scanning electron microscopy (SEM), X-ray diffraction (XRD), differential scanning calorimetry (DSC), thermogravimetric analysis (TGA) and FTIR to elucidate the structural, morphological, chemical, and thermal transformations occurring during the viscose regeneration process. The fibres were also characterised for tenacity and elongation, dyeing performance.The longitudinal surface exhibits a smooth appearance with fine, parallel striations running along the fiber axis, which is typical of fibers regenerated via the viscose wet-spinning process (Fig. 1c). The cross-sectional images reveal a distinct multilobal (serrated or lobed) morphology with 4–6 lobes (Fig. 1d). The multilobal geometry increases the specific surface area, potentially enhancing moisture regain, dyeability, and comfort properties in textile applications.
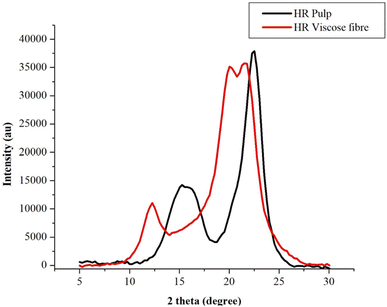 | ||
| Fig. 2 XRD image showing crystallinity profile transition from Hibiscus rosasinensis pulp to viscose fibre. | ||
The observed CrI and pure cellulose II pattern demonstrate that the Hibiscus rosasinensis stem pulp source provides cellulose with suitable molecular weight distribution and purity for effective regeneration, yielding a crystalline structure equivalent to conventional viscose fibers.
The observed decomposition behaviour arises from fundamental structural differences between HR pulp and HR viscose fibre. HR pulp, characterized by high α-cellulose content and crystallinity, decomposes predominantly via depolymerisation and levoglucosan formation. This pathway is endothermic, requiring heat absorption to disrupt the ordered hydrogen-bonded lattice of cellulose I. However, HR viscose fibre, largely amorphous due to the regeneration process, undergoes random chain scission, dehydration, and oxidative char-forming reactions. These pathways are exothermic, releasing heat during decomposition. Thus, the endothermic nature of pulp and the exothermic nature of viscose fibre directly reflect their respective supramolecular architectures and dominant degradation mechanisms.
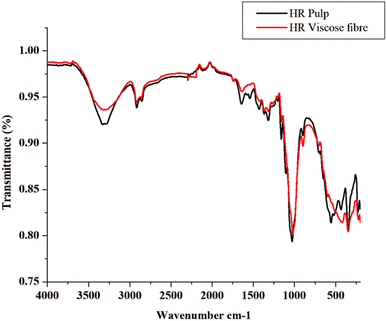
The spectra are largely similar, confirming the cellulosic composition is preserved post-regeneration. However, subtle differences are evident: the O–H band in HR viscose fibre is broader and slightly shifted to lower wavenumbers, reflecting disrupted hydrogen bonding and a transition from crystalline cellulose I (in pulp) to more amorphous cellulose II (in fibre). Intensity variations in the fingerprint region (1500–800 cm−1), including sharper peaks in the pulp and more pronounced fluctuations in the fibre, suggest changes in molecular orientation and crystallinity induced by the viscose process (dissolution in NaOH/CS2 and acid regeneration).
These findings align with reported transformations in regenerated cellulose,17 validating the efficacy of Hibiscus pulp conversion into viscose fibre. The minimal deviations underscore the material's potential for high-quality textile applications, with no unexpected impurities detected.
3.7 Dyeability and coloration performance of viscose fibre
Dyeability is a critical performance parameter for regenerated cellulose fibres intended for textile applications. The Hibiscus rosasinensis-based viscose fibres exhibited effective dye uptake when subjected to conventional dyeing processes. Dyeing was carried out using a reactive dye-Reactofix Red HE3BI at a concentration of 2%, with a material-to-liquor ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20.
20.
Uniform coloration was observed across the fibres, confirming consistent fibre morphology and efficient dye diffusion throughout the fibre structure. Colour strength (K/S values) was measured immediately after dyeing and again after 30 days to evaluate dye durability. The results indicated that the shade remained stable over time, suggesting strong dye–fibre affinity and minimal fading. These findings establish the baseline dyeability and durability of HR viscose fibres. Future work will extend this evaluation by conducting interday and intraday Lab studies, enabling a more rigorous comparison of colour performance between Hibiscus rosasinensis viscose fibres and commercially available viscose fibres. Such analysis will provide deeper insights into the durability of dye absorption and the long-term stability of coloration. The results summarized in Table 5.
| Sr. no. | Analysis interval | Viscose fibre used | Dye | L* | a* | b* | K/S |
|---|---|---|---|---|---|---|---|
| 1 | Day 1 | Control | Reactofix red HE3BI | 49.91 | 63.46 | 1.60 | 100 |
| 2 | HR viscose fibre | Reactofix red HE3BI | 48.74 | 61.52 | 0.38 | 103 | |
| 3 | Day 30 | Control | Reactofix red HE3BI | 48.73 | 61.90 | 1.61 | 100 |
| 4 | HR viscose fibre | Reactofix red HE3BI | 48.88 | 62.13 | 0.82 | 103 |
The high dye affinity of the fibres can be attributed to the hydrophilic nature of viscose and the abundance of accessible hydroxyl groups in regenerated cellulose. These functional groups facilitate strong interactions between fibre and dye molecules, resulting in good colour depth and uniformity. The dyeing results confirm that viscose fibres produced from Hibiscus rosasinensis stem pulp are compatible with commonly used dye classes for regenerated cellulose fibres. Importantly, no adverse effects on dye uptake or shade uniformity were observed due to the non-wood origin of the cellulose. This demonstrates that Hibiscus rosasinensis-based viscose fibres can be processed using standard textile dyeing protocols without additional treatment requirements.
3.8 Significance of Hibiscus rosasinensis stem as a novel and sustainable cellulose source
One of the key highlights of this work is the exploration of Hibiscus rosasinensis stem as a relatively underutilized source of dissolving-grade pulp, offering new potential for regenerated fibre production. Most existing studies focus on wood, bamboo, or commonly used agricultural residues, while Hibiscus rosasinensis stem remains largely unreported in the context of viscose fibre manufacture. The successful conversion of Hibiscus rosasinensis stem into dissolving-grade pulp and subsequently into viscose fibre demonstrates its potential to contribute to sustainable fibre production. Utilizing Hibiscus rosasinensis stem adds value to agricultural by-products, reduces dependence on forest-based resources, and supports the development of circular bioeconomy practices. The confirmation of fibre quality through dyeability further strengthens the practical relevance of this work, as it demonstrates not only fibre formation but also end-use performance relevant to the textile industry.Overall, the SEM, XRD, DSC, TGA and FTIR analyses collectively demonstrate that viscose fibers spun from the novel source exhibit morphology, crystallinity, and chemical composition comparable to standard wood-derived viscose rayon. These results support the potential of this non-wood/alternative lignocellulosic resource as a sustainable feedstock for the viscose industry, contributing to reduced reliance on forest resources and enhanced circularity in textile production.
Collectively, the results demonstrate the good quality of dissolving grade pulp produced from Hibiscus rosasinensis stem and successful conversion of dissolving pulp into regenerated viscose fibre. It exhibits morphology, crystallinity, and chemical composition comparable to standard wood-derived viscose rayon. The viscose process disrupts the hierarchical structure of native cellulose I, producing cellulose II with reduced crystallinity, modified hydrogen bonding, a distinct skin-core morphology, and slightly lower thermal stability. These structural modifications contribute to viscose's enhanced moisture regain, dyeability, and flexibility, while maintaining adequate performance for textile applications-consistent with established literature on regenerated cellulose production.
4 Conclusion
This work establishes Hibiscus rosasinensis stem as a novel and technically viable non-wood feedstock for dissolving-grade pulp and viscose fibre production. The key advance lies in demonstrating a complete, integrated pathway from raw biomass to dyed fibre under conventional viscose processing conditions, confirming compatibility with established industrial technology. Importantly, the regenerated fibres retained mechanical integrity and dyeability, showing that the non-wood origin does not compromise cellulose structure or functional group accessibility.By introducing ornamental plant stem waste into the viscose value chain, this study expands the material base for regenerated cellulose fibres and supports diversification away from wood-derived pulps. The findings contribute to sustainable polymer processing and circular bioeconomy strategies, offering both environmental and economic benefits. Future work will focus on process optimization, life-cycle assessment, structure–property correlations, and scale-up toward industrial application, thereby strengthening the case for Hibiscus stem as a renewable resource in textile fibre manufacture.
Consent to participate declaration
This research did not involve human participants or animals and was conducted in compliance with institutional and international ethical standards for research integrity.Ethics declaration
The research work outcomes mentioned in the manuscript entitled-Non-Wood Dissolving Pulp from Hibiscus rosasinensis for Sustainable Viscose Fibre Production, represents the authors' own original work and has not been previously published elsewhere. It is not currently under consideration for publication by any other journal. The paper truthfully and completely reflects the authors' research and analysis, and all meaningful contributions from co-authors have been properly acknowledged. The results are appropriately placed within the context of prior and existing research. All sources have been fully disclosed through correct citations, and any literal text reproduction has been clearly indicated using quotation marks with proper references. Furthermore, all authors have been personally and actively involved in substantial work leading to the preparation of this manuscript and collectively accept public responsibility for its content.Author contributions
SPP, RK and STM contributed to the conceptualization of the study. SPP designed the methodology, carried out the experimental investigation, and prepared the original draft of the manuscript. SB contributed to experiments related to viscose process and RK and STM were responsible for reviewing and editing the manuscript. Visualization was performed by SPP and RK. RK and STM provided supervision and overall guidance throughout the project.Conflicts of interest
The authors declare no competing interests.Data availability
All data supporting the findings of this study are available within the article.Acknowledgements
The authors duly acknowledge Mr Dattatray Yadav for spinning viscose fibre out of viscose dope, Mr Sangram Sonawane for viscose analysis and support of Pulp & Fibre Innovation Centre (Grasim industries) for providing required resources and Aditya Birla Science and Technology company Pvt. Ltd for supporting in analytical facilities. We also would like to thank the Institute of Chemical Technology (ICT) for its academic support.References
- M. T. Selvan and M. Ramesh, Unveiling the untapped potential of Hibiscus rosasinensis lignocellulosic fiber: a multifaceted characterization for advancing sustainable biocomposite applications, Int. J. Biol. Macromol., 2024, 281, 136344 CrossRef CAS PubMed.
- I. M. De Rosa, J. M. Kenny, D. Puglia, C. Santulli and F. Sarasini, Morphological, thermal and mechanical characterization of okra (Abelmoschus esculentus) fibres as potential reinforcement in polymer composites, Compos. Sci. Technol., 2010, 70, 116–122 CrossRef CAS.
- R. M. V. Saha, M. Sandhuja and C. Mittal, A comprehensive overview of Hibiscus rosasinensis and its uses: a review, Int. J. Adv. Biochem. Res., 2025, 9(5), 644–647 CrossRef.
- V. K. Varma, V. Mishra, V. K. Pal and J. Sanjog, Extraction and characterization of new Hibiscus rosasinensis stem fiber and fabrication of polymer composite, Next Res, 2025, 2(3), 100697 CrossRef.
- N. Silvy, M. S. Reza, M. N. Uddin and M. L. Akther, Comparison between different components of some available hardwood and softwood in Bangladesh, IOSR J. Biotechnol. Biochem., 2018, 4(1), 1–5 Search PubMed.
- C. Woodings, Regenerated Cellulose Fibres, CRC Press, Boca Raton, FL Woodhead Publishing, Cambridge, UK, 2001, pp. 336 Search PubMed.
- C. Felgueiras, N. G. Azoia, C. Gonçalves, M. Gama and F. Dourado, Trends on the cellulose-based textiles: raw materials and technologies, Front. Bioeng. Biotechnol., 2021, 9, 608826 CrossRef PubMed.
- S. C. Gondhalekar, P. J. Pawar, S. S. Dhumal and S. S. Thakre, Mechanism of xanthation reaction in viscose process, Cellulose, 2019, 26(3), 1595–1604 CrossRef CAS.
- A. K. Chapagain, A. Y. Hoekstra, H. H. G. Savenije and R. Gautam, The water footprint of cotton consumption: an assessment of the impact of worldwide consumption of cotton products on the water resources in the cotton producing countries, Ecol. Econ., 2006, 60, 186–203 CrossRef.
- A. M. Sarkar, J. Nayeem, M. M. Rahaman and M. S. Jahan, Dissolving pulp from non-wood plants by prehydrolysis potassium hydroxide process, Cellul. Chem. Technol., 2021, 55(1–2), 117–124 CAS.
- L. A. Worku, A. Bachheti, R. K. Bachheti, C. E. R. Reis and A. K. Chandel, Agricultural residues as raw materials for pulp and paper production: overview and applications on membrane fabrication, Membranes, 2023, 13, 228 CrossRef CAS PubMed.
- J. Wojtasz, N. Sjöstedt, B. Storm, M. M. Parayil, A. Ulefors and L. Nilsson, et al., Producing dissolving pulp from agricultural waste, RSC Sustainability, 2025, 3(5), 2210–2220 RSC.
- J. P. Shang, P. Liang, Y. Peng, D. F. Xu and Y. B. Li, One-step treatment for upgrading bleached bamboo pulp to dissolving pulp high solvency in green alkali/urea aqueous solution, Polymers, 2023, 15, 1475 CrossRef CAS PubMed.
- L. A. R. Batalha, J. L. Colodette, J. L. Gomide, L. C. A. Barbosa, C. R. A Maltha and F. J. B. Gomes, Dissolving pulp from bamboo, BioResources, 2012, 7(1), 640–651 Search PubMed.
- N. Cordeiro, M. N. Belgacem, I. C. Torres and J. C. V. P. Moura, Chemical composition and pulping of banana pseudo-stems, Ind. Crops Prod., 2004, 19(2), 147–154 CrossRef CAS.
- B. B. Mazumder, Y. Ohtani, Z. Cheng and K. Sameshima, Combination treatment of kenaf bast fiber for high viscosity pulp, Wood Sci. Technol., 2000, 46, 364–370 CrossRef CAS.
- H. Sixta, Handbook of Pulp, Wiley-VCH, Weinheim, Germany, 2006, pp. 1352 Search PubMed.
- H. Sixta, M. Iakovlev, L. Testova, A. Roselli, M. Hummel and M. Borrega, et al., Novel concepts of dissolving pulp production, Cellulose, 2013, 20(4), 1547–1561 CrossRef CAS.
- E. U. Akubueze, C. S. Ezeanyanaso, O. S. Muniru, F. C. Nwaeche, M. I. Tumbi and C. C. Igwe, et al., Extraction and characterization of bast fibres from roselle (Hibiscus sabdariffa) stem for industrial application, J. Mater. Sci. Res. Rev., 2019, 2(4), 1–7 Search PubMed.
- J. P. Supriya, R. Shetty, N. Naik, S. Maddasani and A. Hegde, Chemical characterization of Hibiscus rosasinensis plant fibers facilitated through design of experiments and artificial neural network hybrid approach, Sci. Rep., 2024, 14, 22510 CrossRef CAS PubMed.
- TAPPI T 280 pm-99, Acetone extractives of wood and pulp, Technical Association of the Pulp and Paper Industry, Atlanta, GA, USA, 1999 Search PubMed.
- A. Álvarez, S. Cachero, C. González-Sánchez, J. Montejo-Bernardo, C. Pizarro and J. L. Bueno, Novel method for holocellulose analysis of non-woody biomass wastes, Carbohydr. Polym., 2018, 189, 250–256 CrossRef PubMed.
- TAPPI T 222 om-02, Acid-insoluble Lignin in Wood and Pulp, Technical Association of the Pulp and Paper Industry, Atlanta, GA, USA, 2002 Search PubMed.
- TAPPI T 223 cm-01, Pentosans in Wood and Pulp, Technical Association of the Pulp and Paper Industry, Atlanta, GA, USA, 2001 Search PubMed.
- TAPPI T 211 om-02, Ash in Wood, Pulp, Paper, Technical Association of the Pulp and Paper Industry, Atlanta, GA, USA, 2002 Search PubMed.
- TAPPI T 244 cm-99, Acid-Insoluble Ash in Wood, Pulp, Paper, Technical Association of the Pulp and Paper Industry, Atlanta, GA, USA, 1999 Search PubMed.
- R. Martínez-Peñuñuri, A. M. García-Alegrí, P. Vázquez-Bustamante, M. Montoya-Blumenkron, D. I. Estrada-González and A. M. Avilés-Figueroa, et al., Validation and uncertainty estimation for multielement determination using ICP-OES according to the ASTM E2941-14 standard, J. Chem. Metrol., 2025, 19(1), 1–22 Search PubMed.
- M. S. Jahan, L. Ahsan, A. Noori and M. A. Quaiyyum, Dissolving pulp from Trema orientalis, BioResources, 2008, 3(3), 816–828 Search PubMed.
- W. Wei, Z. Tian, X. Ji, Q. Wang, J. Chen and G. Zhang, et al., Severity factor and dissolving pulp, BioResources, 2020, 15(2), 4323–4336 CAS.
- TAPPI T 236 om-99, Kappa Number of Pulp, Technical Association of the Pulp and Paper Industry, Atlanta, GA, USA, 1999 Search PubMed.
- TAPPI T 230 om-99, Viscosity of pulp (Capillary Viscometer Method), Technical Association of the Pulp and Paper Industry, Atlanta, GA, USA, 1999 Search PubMed.
- I. S. F. Mendes, A. Prates and D. V. Evtuguin, Production of rayon fibres from cellulosic pulps: State of the art and current developments, Carbohydr. Polym., 2021, 273, 118466 CrossRef CAS PubMed.
- G. Kumsa, G. Gebino and G. Ketema, One-bath one-step dyeing of polyester/cotton (PC) blends fabric with disperse dyes after acetylation of cotton, Discov. Mater., 2021, 1, 19 CrossRef.
- N. Waghmare, A. Nayar, S. Tandyekkal and V. Juikar, Influence of dye pickup on different functionalized regenerated cellulosic fibres, Cellulose, 2023, 30(8), 5391–5406 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |