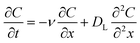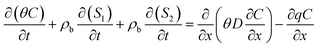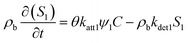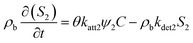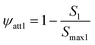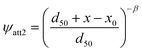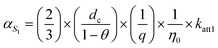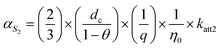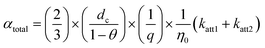Open Access Article
Open Access ArticleInfluence of solution chemical properties and porous medium surface coatings on the transport and retention behavior of polystyrene microplastics
Abliz Abdurahman *a,
Xiaoli Maa and
Rui Gaob
*a,
Xiaoli Maa and
Rui Gaob
aChemistry Department, College of Pharmacy, Xinjiang Medical University, Urumqi, 830017, China. E-mail: abliz@xjmu.edu.cn; 1065158755@qq.com
bInstitute for Advanced Study, Chengdu University, Chengdu, 610106, China. E-mail: gaorui@cdu.edu.cn
First published on 14th May 2026
Abstract
The transport and retention behavior of microplastics (MPs) in saturated porous media is a hot issue in the fields of ecological security and public health in contemporary society. In this study, the transport and retention behaviors of polystyrene microplastics (PS-MPs) in saturated porous media were systematically investigated under various environmental conditions, including humic acid (HA) concentration, ionic strength (IS), mass fraction of iron oxyhydroxide coated quartz sand (ω), and pH in the presence of HA. A series of column experiments were conducted, and all breakthrough curves and retention profiles were successfully simulated using a two kinetic sites attachment-detachment model (TKSADM) that distinguishes between chemical adsorption (Site 1) and physical retention (Site 2). Results demonstrated that increasing HA concentration from 1 to 10 mg L−1 enhanced PS-MP transport, which was attributed to HA adsorption creating steric hindrance and enhancing electrostatic repulsion. In contrast, HA conditions, increasing IS from 0.1 to 100 mM progressively suppressed MP transport, with model parameters revealing a dramatic 30-fold increase in physical retention rate (katt2) and a 2.8-fold increase in chemical adsorption capacity (Smax1/C0), reflecting the dual role of IS in compressing the electrical double layer and inducing HA conformational collapse. Increasing the mass fraction of iron oxyhydroxide coated quartz sand (ω from 0.15 to 0.75) substantially inhibited PS-MP transferability, with both chemical adsorption rate (katt1) and physical retention rate (katt2) increasing by 57% and 83%, respectively, accompanied by a 79% increase in Smax1/C0. This balanced enhancement reflects the dual role of iron oxyhydroxide coatings: increased surface roughness creating physical straining sites and ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Fe–OH groups providing abundant reactive sites for specific adsorption. Increasing pH from 6.0 to 10.0 substantially enhanced MP mobility, with all retention – related parameters decreasing monotonically: katt1 decreased by 34%, katt2 decreased by 52%, and Smax1/C0 exhibited the most dramatic decline (71%), revealing a “pH-switching effect” on physical retention sites – activated at low pH and deactivated at high pH due to enhanced electrostatic repulsion and HA conformational extension. This study provides quantitative mechanistic insights into MP fate in subsurface environments and underscores the need to consider coupled effects of multiple environmental factors when predicting MP transport behavior.
Fe–OH groups providing abundant reactive sites for specific adsorption. Increasing pH from 6.0 to 10.0 substantially enhanced MP mobility, with all retention – related parameters decreasing monotonically: katt1 decreased by 34%, katt2 decreased by 52%, and Smax1/C0 exhibited the most dramatic decline (71%), revealing a “pH-switching effect” on physical retention sites – activated at low pH and deactivated at high pH due to enhanced electrostatic repulsion and HA conformational extension. This study provides quantitative mechanistic insights into MP fate in subsurface environments and underscores the need to consider coupled effects of multiple environmental factors when predicting MP transport behavior.
1. Introduction
With the extensive use and improper disposal of plastic products, microplastics (MPs) have been widely distributed in terrestrial and aquatic environments around the world,1,2 and have entered porous medium systems through various pathways such as surface runoff,3,4 irrigation,5,6 and atmospheric deposition.7–9 MPs have characteristics such as persistence,10,11 bioaccumulation,12,13 and potential toxicity.10,14 They can adsorb coexisting contaminants such as heavy metals,15 organic pollutants (e.g., PFOA),16 and pathogenic microorganisms,17,18 forming “composite pollution carriers” that further amplify their ecological hazards.19The transport and retention behaviors of MPs in porous media directly determine their environmental dispersion range, fate, and potential ecological exposure risks.20–23 These processes are not a simple physical filtration process, but rather the result of the coupled effect of multiple factors, including the inherent characteristics of MPs (such as particle size, shape, density, surface charge, and functional groups), the chemical properties of the solution (pH, ionic strength (IS), dissolved organic matter (DOM) content), and the properties of the medium (pore structure, mineral composition, and the presence of biofilms).3,6 Among these, the surface charge of MPs and porous medium particles exerts a decisive influence on the transport behavior of MPs within porous media. This effect primarily operates by regulating electrostatic interactions between particles. When MPs carry the same charge as the surfaces of porous media (such as sand grains, clay minerals, etc.), the electrostatic repulsion between them reduces the possibility of MPs being captured by the medium, thereby enhancing their transport capacity.24,25 On the contrary, when the surface charge of the medium is opposite to that of the MPs, a strong electrostatic attraction promotes the adhesion and retention of MPs, greatly restricting their transport within the pores.26,27
In recent years, preliminary progress has been made in studying the transport of MPs within porous media.28,29 IS and DOM further modulate the influence of surface charge. High IS can compress the double layer on the surface of MPs, weakening electrostatic repulsion, leading to the aggregation and retention of MPs that are otherwise prone to transport due to charge repulsion.27,30 For example, in an environment containing calcium ions (Ca2+), divalent cations can act as a “bridge” to connect negatively charged MPs and negatively charged medium surfaces, significantly enhancing retention.31 Meanwhile, after DOM molecules (such as humic acid HA) are adsorbed onto the surface of MPs, they may increase their negative charge or provide steric hindrance effect, thereby promoting the transport of MPs under certain conditions.30,32 The pH of the solution regulates the zeta potential by affecting the protonation state of functional groups on the surface of particles, thereby affecting particle transport behavior.33,34 Under low pH conditions, the surface of the medium may partially protonate, reducing its negative or even positive charge, which is beneficial for the transport of negatively charged MPs. Under high pH conditions, the degree of deprotonation on the surface of the medium increases, the surface negativity increases, and the electrostatic repulsion between the negatively charged MPs increases, improving their transport performances.25
Systematically elucidating the transport mechanisms of MPs under the coupled effects of multiple factors is a scientific prerequisite for accurately predicting their environmental behavior and conducting risk assessments.35,36 Nevertheless, most studies examine only the independent effects of a single factor or a few factors (such as IS, pH, and HA), overlooking the potential synergistic or antagonistic effects that may exist between different factors – a core characteristic of real-world complex environments. In addition, regarding the influence of chemical modification on the surface of the medium, especially the microscopic mechanism of how variable charge coatings such as iron oxyhydroxide can form a ternary interaction with MPs and HAs, it remains unclear. More importantly, most existing MPs transport studies employ single kinetic site models or equilibrium approaches,37 which assumed that there is a specific type of active site in the reaction system, and all these sites are energetically homogeneous, with the same adsorption energy and reaction activation energy. Therefore, the structures and fitting processes of these models are relatively simple. However, they have the following fundamental limitations in describing the transport behavior of particulate matter, especially when conducting systematic parameter analysis for multiple environmental factors. (1) It is impossible to distinguish between these two fundamentally different processes: physical retention and chemical adsorption; (2) unable to fit the hyper-exponential decay retention profiles observed in experiments; (3) the differential effects of environmental factors on different sites cannot be explained; (4) they lack comparability with the widely applied two kinetic sites model studies in the field. These makes them difficult to accurately describe the potential differences in kinetic processes of MPs on heterogeneous surfaces. This gap hinders mechanistic understanding of how different environmental factors regulate MPs fate through distinct pathways.
To address the aforementioned research gap, this study systematically investigates the influence of key environmental factors – including HA concentration, IS, pH, and surface charge heterogeneity of quartz sand particles, on the transport and retention behavior of polystyrene model microplastics (PS-MPs) within saturated porous media systems. The research process employs two kinetic sites model to quantitatively analyze and deeply elucidate the relevant interaction processes and mechanisms, revealing the core control mechanisms under multi-factor coupling effects. This contributes to refining the theoretical framework for MPs environmental behavior within groundwater and soil porous media systems. It also provides crucial theoretical foundations and data support for scientifically assessing groundwater pollution risks from MPs and formulating targeted prevention, control, and remediation strategies for MPs contamination in soil and groundwater.
2. Materials and methods
2.1. Preparation of PS-MPs suspension
PS-MPs purchased from Sigma-Aldrich, Inc., USA. To prepare the PS-MPs stock solutions, 100 mg of the original PS-MPs powder was mixed into 1 L of deionized (DI) water and dispersed by an ultrasonic cleaner (KQ – 300VDE, Supmile, China) for 30 min to ensure complete dispersion of the particles. Prior to each column test, the PS-MPs stock solutions were diluted with the desired background solutions (HA, NaCl or both) to a target concentration 40 mg L−1. The pH of each suspension of PS-MPs and their respective background solutions was set between 6.0 and 10.0 (6.0, 8.0 and 10.0) using solutions of 1.0 mM HCl or NaOH to achieve the desired adjustment. The concentration of PS-MPs suspension was determined using total organic carbon analyzer (TOC – L CPH, SHIMADZU, Japan). And the zeta potentials of PS-MPs in solutions varying in HA concentration, IS, and pH were measured using zeta potential analyzer (Zhongcheng Digital Technology model JS94G, China) (see SI).2.2. Porous media
Quartz sand (Shaanxi Zhouzhi County Quartz Sand Co, Ltd China) was selected for the column tests as the porous medium, with a particle size range of 10–20 mesh. Before the experiment, the quartz sand was treated in 10% (V![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V) HNO3 solution for 24 h to remove the impurity, and then cleaned with tap water and DI water until the pH of the aqueous solution reached a neutral level.28 Then dry overnight at 105 °C and store in a sealed container for later use.
V) HNO3 solution for 24 h to remove the impurity, and then cleaned with tap water and DI water until the pH of the aqueous solution reached a neutral level.28 Then dry overnight at 105 °C and store in a sealed container for later use.
Quartz sand coated with iron oxyhydroxides are prepared according to the method reported by Stahl et al.38,39 500 g of quartz sand was added to an evaporating dish and then 0.17 mol L−1 of Fe (NO3)3 387.5 mL and 0.52 mol L−1 of NaOH 90 mL were held to precipitate onto the quartz sand. The mixture was placed in a drying oven at 105 °C for 72 hours with periodic stirring to prevent salt crusting on the surface of the quartz sand. The quartz sand was washed with 1.0 mM HCl and 1.0 mM NaOH to remove the poorly adsorbed iron on the surface of the quartz sand. Scanning electron microscopy–energy dispersive X-ray analysis (SEM–EDX) (SSX – 550, Shimadzu, Japan) results showed that 75 ± 3% of the quartz sand surface were coated with iron oxyhydroxides.38,40 Finally, the zeta potentials of bare and iron oxyhydroxides coated quartz sand were measured by zeta potential meter (BI – EKA, Brookhaven Instruments Corp., Holtsville, NY) under the same conditions as in the subsequent column experiments (see SI).
2.3. Column experiments
PS-MPs transport experiments were conducted using glass chromatography columns (with diameter of 2.6 cm and a length of 12 cm) packed with wet quartz sand (Fig. 1). The porosity of the sand columns determined gravimetrically according to the method outlined by Jaisi et al.41 and it varied between 0.37 to 0.40. In the experiment on the effect of iron oxyhydroxide surface coatings on the transport and retention of PS-MPs, the porous medium was prepared by uniformly mixing surface coated quartz sand and bare quartz sand in different mass ratios.38The column experiments were completed with the assistance of a peristaltic pump (BT100L, Baoding Fluid Technology Co., Ltd, China) in the down-flow mode at a flow rate of 1 mL min−1 (Darcy velocity of 3.13 × 10−6 cm s−1) to maintain the stability of the experimental conditions, and various conditions as detailed in Table 1. Initially, the quartz sand column was rinsed with DI water until no visible impurities were present in the effluent. Subsequently, the chosen background solution was infused into the column for 3 h to create stable chemical conditions. Then, PS-MPs suspension in 4 PV of the same background solution was passed through the column. Finally, several PVs background solutions introduced to the column to flush out the unattached PS-MPs. During the experiment, the effluent was manually collected every 4 minutes, and the concentration of PS-MPs determined by the total organic carbon analyzer (TOC – L CPH, Tsushima, Japan). Following the completion of each transport test, each column was dissected into 12 layers of 1 cm segments, and the quartz sand grains in each fraction were extracted with DI water to determine spatial distribution profile of retained PS-MPs, as described earlier studies.38,40 All column tests were performed at least twice.
| HAa (mg L−1) | ISb (mM) | ωc | pH | Zeta potentials (mV) | Φf | |
|---|---|---|---|---|---|---|
| PS-MPsd | Quartz sande | (×104 kT) | ||||
| a Humic acid (pH 6.0).b Ionic strength (NaCl as background electrolyte).c Fraction of the quartz sand grain surfaces coated by iron oxyhydroxide.d Zeta potential of PS-MPs.e Zeta potential of quartz sand or average value of uncoated/coated quartz sand grain.f Energy barriers between PS-MPs and quartz sand calculated by DLVO theory.g Electrostatic attraction predominates between polystyrene PS-MPs and quartz sand particles, and there is no energy barrier that hinders particle contact. | ||||||
| 0 | 0.1 | 0 | 6.0 | −57.36 ± 0.54 | −26.38 ± 0.63 | 346 |
| 1 | 0.1 | 0 | 6.0 | −59.45 ± 0.69 | −29.15 ± 1.35 | 418 |
| 5 | 0.1 | 0 | 6.0 | −61.79 ± 0.31 | −29.87 ± 0.36 | 455 |
| 10 | 0.1 | 0 | 6.0 | −61.84 ± 1.40 | −30.85 ± 1.62 | 492 |
| 10 | 1 | 0 | 6.0 | −61.43 ± 1.00 | −28.82 ± 0.57 | 28.5 |
| 10 | 10 | 0 | 6.0 | −50.82 ± 3.45 | −28.06 ± 2.01 | 11.1 |
| 10 | 100 | 0 | 6.0 | −31.09 ± 2.75 | −22.64 ± 1.14 | 0.624 |
| 10 | 0.1 | 0.15 | 6.0 | −61.84 ± 1.40 | −24.43 ± 0.24 | 34.8 |
| 10 | 0.1 | 0.30 | 6.0 | −61.84 ± 1.40 | −17.96 ± 0.31 | 21.2 |
| 10 | 0.1 | 0.45 | 6.0 | −61.84 ± 1.40 | −11.63 ± 0.08 | 9.42 |
| 10 | 0.1 | 0.60 | 6.0 | −61.84 ± 1.40 | −5.590 ± 0.38 | 0.496 |
| 10 | 0.1 | 0.75 | 6.0 | −61.84 ± 1.40 | 1.410 ± 0.11 | —g |
| 10 | 0.1 | 0.15 | 8.0 | −65.03 ± 1.13 | −29.82 ± 0.25 | 46.8 |
| 10 | 0.1 | 0.15 | 10.0 | −66.21 ± 5.52 | −33.61 ± 0.41 | 58.6 |
The PS-MPs breakthrough curves (BTCs) was plotted as a function of the pore volume (PV) as the horizontal coordinate and the standardized concentration of PS-MPs in the effluent, C/C0 (dimensionless), as the vertical coordinate. The retention profiles (RPs) were plotted based on the functional relationship between the standardized concentration S/C0 [M−1] of PS-MPs in each gram of quartz sand and the column depth, to explore the retention distribution of PS-MPs in the sand column. Finally, the sum of the masses of PS-MPs in the effluent and retained in the quartz sand column will be compared with the mass of the PS-MPs injected into the column to calculate the mass balance. In addition, the tracer experiment was conducted under similar experimental conditions using a tracer solution (0.2 mM KNO3, with NO3− as the tracer), and the concentrations of NO3− were quantified by UV-Vis Spectrophotometer at 220 nm.37
2.4. Mathematical model
Derjaguin–Landau–Verwey–Overbeek (DLVO) theory was used to evaluate the interaction energy between PS-MPs and quartz sand across various experimental scenarios. The total interaction energy (Φtotal) between particles is the sum of van der Waals forces (Φvdw) and electrostatic interactions (Φelec).42 The PS-MPs–quartz sand system is considered as a ball–plate interaction system43 to estimate the interaction energy (see SI).The tracer transport was described by the one-dimensional (1D) advection and dispersion model (ADE):
The transport of MPs in porous media is a highly complex process involving transport, attachment, detachment, blocking, straining, release, aggregation, and sedimentation.26,32,44,45 Therefore, its transport and retention are governed by a combination of various mechanisms.46,47 In this study, the transport of PS-MPs are described by coupling the advection–dispersion equation with a two kinetic sites attachment-detachment mode (TKSADM).48,49 Mathematical expression of this model is as follows:28,48–51
In the equation, the first term on the left-hand side represents the rate of change of the liquid-phase concentration of PS-MPs over time. The second and third items on the – hand side represents the rates of change in PS-MPs retention in the solid phase at site 1 (S1) and site 2 (S2) over time. The first item on the right-hand side is the diffusion term, which describes the diffusion driven by the concentration gradient of PS-MPs. The second item is the convection term, which describes the transport of PS-MPs carried by water flow.
In this study, two types of kinetic sites S1 and S2 are representing chemical adsorption sites and physical straining sites, respectively.50,51 Among them, S1 typically corresponds to an adsorption process dominated by surface chemical forces (DLVO forces).50,51 PS-MPs attach to the surface of sand grains through electrostatic attraction, van der Waals forces, etc. The capacity of such sites is limited;50,51 as deposited PS-MPs occupied the surface of porous medium particles, subsequent adsorption is inhibited. And S2 typically corresponds to a physical retention mechanism. It is a purely geometric mechanism by which PS-MPs are trapped in a porous medium. When a PS-MPs size is greater than or close to the size of a pore throat, the particles cannot pass through and is physically retained. A key characteristic of this process is significant spatial heterogeneity: near the injection end (the column inlet), the pore throats become blocked first, and the straining efficiency decreases more rapidly the deeper one goes into the medium.50,51 The kinetic equations for each of the two sites are:38,40,50,51
where katt1 [T−1] and katt2 [T−1] are the first-order rate constants for the attachment of PS-MPs to S1 and S2, kdet1 [T−1] and kdet2 [T−1] are the first-order rate constant for the detachment of PS-MPs from S1 and S2 back into the aqueous phase. ψ1 and ψ2 are respectively representing the retention functions of S1 and S2. The classic Langmuir blocking function is the retention function for the S1, which is mainly controlled by solution chemistry and surface chemistry. Its mathematical expression is:51–53
where S1 is the current solid-phase retention concentration of PS-MPs at site 1, Smax1 is the maximum retention capacity, which represents the maximum solid-phase concentration of PS-MPs that can attach to S1. As for S2, the depth-dependent straining function is adopted:50,51
where d50 [L] is the median particle size, x0 [L] is the reference distance (typically taken as the coordinates at the column inlet), x [L] is the distance of PS-MPs from the column inlet, and β [–] is the empirical factor that controls the shape of the PS-MPs RPs, which is set as 0.432.49
The transport and retention parameters (D, katt1, katt2, kdet1,kdet2 Smax1) of the BTCs and RPs obtained from the column experiments of the tracer (NO3−) and PS-MPs were fitted using the hydrodynamic software HYDRAUS-1D54 and optimized by least square method.55
In studies of MPs transport within saturated porous media, attachment efficiency (α) serves as a core parameter for quantifying the probability of actual attachment occurring after MPs collide with porous media surfaces. It directly links theoretical collision frequency to actual retention behavior, making it crucial for predicting the environmental fate of MPs.22 In the Physicochemical Filtration Theory (PCPT), the attachment efficiency α is central to evaluating the trapping mechanism of particles within porous media, particularly their transport behavior in water-saturated porous media.42 In our research, we assumed that α is not a single fitting parameter but is instead “allocated” across two attachment pathways:
| αtotal ≈ αS1 + αS2 |
The two terms on the right-hand side of equation correspond to the “effective” attachment efficiencies at S1 and S2, respectively, and can be explicitly expressed in terms of rate constants:56
where η0 [–] is the single-collector contact efficiency of quartz sand, and it is determined by three physical capture mechanisms: Brownian diffusion ηD, direct interception ηI, and gravitational sedimentation ηG.57 Its specific mathematical expression is (see SI 5):
| η0 = ηD + ηI + ηG |
Ultimately, the value of α can be calculated with column transport experimental data based on the following expression:
3. Result and discussion
3.1. Characterization of PS-MPs and quartz sand
According to the scanning electron microscope (SEM) (SU8010, Hitachi, Japan) test results, the PS-MPs are spherical in shape (Fig. 2A). Based on Dynamic Light Scattering (DLS) analysis (MasterSizer 2000, Malvern Instruments, UK), the size of PS-MPs in ultrapure water (18.2 MΩ cm, prepared by the Milli-Q system) at pH 6.0 ranged from 3.31 to 138 µm, and the median diameter (D50) is about 10.6 ± 0.2 µm (Fig. 2B), this is exactly the same as the average particle size of 10 µm provided by the supplier. The specific surface area (SSA) of PS-MPs is 0.98 ± 0.32 m2 g−1 determined by Brunauer–Emmett–Teller (BET) method (ASAP 2460, Micromeritics, US).The particle size test results show that the average particle size of bare quartz sand is approximately 1.76 ± 0.35 mm (n = 1000). The surface morphology and elemental composition of quartz sand was measured by scanning electron microscope (SU8010, Hitachi, Japan) combined with energy dispersive X-ray spectrometer (EDS), and the results are shown in Fig. 3. As shown in Fig. 3A, quartz sand particles exhibit an irregular blocky morphology with well-defined individual particle outlines. The particle surfaces are rough, featuring naturally fractured textures with uneven patterns and irregular edges. No obvious signs of artificial modification or chemically eroded surface features were observed, reflecting their primary clastic morphology. The EDS spectrum (Fig. 3B) exhibits strong characteristic peaks for Si (energy ≈ 1.7 keV) and O (energy ≈ 0.5 keV), which are the dominant elemental signals in the sample. Their relative signal intensities align with the elemental composition ratio of SiO2, the primary component of quartz sand. Additionally, the weak C peak (≈0.2 keV) in the spectrum originates from environmental carbon-based contaminants adsorbed on the sample surface. The Pt peak (≈2.0 keV) corresponds to the conductive platinum layer sprayed onto the sample surface prior to SEM testing (to enhance the conductivity of insulating quartz sand for testing). While the Fe peak (≈6.4 keV) represents a characteristic peak for trace impurities, indicating the presence of minimal iron–based impurity components in the sample.
3.2. Effect of HA concentration on transport and retention of PS-MPs
To explore the effect of DOM in pore solution on the transport of MPs in porous media, HA is employed to conduct the column experiments, and the BTCs and RPs of PS-MPs under different HA concentrations are shown in Fig. 4. The results revealed that the presence of HA clearly facilitated the transport of PS-MPs under all examined conditions. As the HA concentration increased, the transportability of PS-MPs was enhanced, and the promoting effect of HA exhibited strong dependence on HA concentration. For example, when HA concentration increased from 0 to 10 mg L−1, the C/C0 of PS-MPs rose from 0.442 to 0.799, and the calculated α values of PS-MPs decreased from 0.052 to 0.014 (Table 2). Correspondingly, the effluent rates increased from 49.80% to 84.51%, and the retention rates of PS-MPs were decreased from 48.90% to 17.82% (Table 2). Additionally, under HA conditions, the PS-MPs recovery rates are relatively high (97.01 to 102.33%) and almost all of them are recovered. Similar promotion effects of HA have been observed on the transport of other MPs and engineering inorganic nanoparticles (EINs).31,58 | ||
| Fig. 4 Measured and fitted breakthrough curves (A) and retention profiles (B) for PS-MPs at different HA concentration at pH 6.0 in uncoated quartz sand. | ||
| HA mg L−1 | IS mM | ω | pH | (C/C0)max | α | Meffa% | Mretb% | Mret-inletc% | Mtotd% |
|---|---|---|---|---|---|---|---|---|---|
| a Refers to the effluent percentage of PS-MPs recovered from column tests.b Refers to the retained percentage of PS-MPs recovered from column tests.c Refers to the PS-MPs retained near the column inlet.d Refers to the total percentage of PS-MPs recovered from column tests. | |||||||||
| 0 | 0.1 | 0 | 6.0 | 0.442 | 0.052 | 49.80 | 48.90 | 24.50 | 98.70 |
| 1 | 0.1 | 0 | 6.0 | 0.466 | 0.049 | 52.67 | 46.72 | 23.40 | 99.39 |
| 5 | 0.1 | 0 | 6.0 | 0.526 | 0.041 | 54.40 | 41.64 | 20.67 | 97.01 |
| 10 | 0.1 | 0 | 6.0 | 0.799 | 0.014 | 84.51 | 17.82 | 9.78 | 102.3 |
| 10 | 1 | 0 | 6.0 | 0.743 | 0.019 | 84.14 | 22.55 | 12.44 | 106.7 |
| 10 | 10 | 0 | 6.0 | 0.483 | 0.046 | 50.81 | 45.57 | 22.39 | 96.38 |
| 10 | 100 | 0 | 6.0 | 0.369 | 0.063 | 38.70 | 54.54 | 28.07 | 93.24 |
| 10 | 0.1 | 0.15 | 6.0 | 0.615 | 0.031 | 65.36 | 35.04 | 15.57 | 100.4 |
| 10 | 0.1 | 0.30 | 6.0 | 0.528 | 0.041 | 55.83 | 41.68 | 20.14 | 97.51 |
| 10 | 0.1 | 0.45 | 6.0 | 0.464 | 0.049 | 48.88 | 46.83 | 23.30 | 95.71 |
| 10 | 0.1 | 0.60 | 6.0 | 0.441 | 0.052 | 44.42 | 51.31 | 24.80 | 95.73 |
| 10 | 0.1 | 0.75 | 6.0 | 0.391 | 0.060 | 40.99 | 52.49 | 27.17 | 93.49 |
| 10 | 0.1 | 0.15 | 8.0 | 0.725 | 0.020 | 76.37 | 26.01 | 11.68 | 102.4 |
| 10 | 0.1 | 0.15 | 10.0 | 0.756 | 0.018 | 79.57 | 23.36 | 10.61 | 102.9 |
The facilitated transport of PS-MPs attributed to HA involves electrostatic interaction, steric repulsion, and competition for available adsorption sites on quartz sand surfaces. Initially, as listed in Table 1, all the studied PS-MPs and quartz sand grains carry more negative charges with presence of HA in the solution. And, as the HA concentration increased, the absolute value of their zeta potential increased. This is mainly because HA regulates the negative charge density on the particle surface through surface adsorption.59–61 Thus, electrostatic repulsion between PS-MPs and quartz sand grains is improved. The relatively higher DLVO energy barriers (Φmax) between PS-MPs and quartz sand grains are observed (Fig. 5) with the presence of HA in solution, causing PS-MPs to be difficult to capture by quartz sand particles, leading to enhanced transport of PS-MPs in sandy column. Then, at higher concentrations (such as 10 mg L−1), Steric hindrance caused by HA prevented PS-MPs from agglomerating,42,62 as confirmed by inverted microscope images (Fig. 5), and further enhanced the transport of PS-MPs in sandy column. Previous studies have also reported that the improvement in particle transport performance under HA conditions is attributed to changes in particle surface charge due to HA adsorption and the decomposition of previously aggregated particles.37,60,63,64 Lastly, HA may compete with PS-MPs for adsorption on quartz sand surfaces,60,65 reducing the number of active adsorption sites available for PS-MPs. This could also weaken the retention of PS-MPs within the column.
RPs of PS-MPs under different concentrations of HA conditions are presented in Fig. 4B. Under all tested conditions, the RPs exhibited a hyper-exponential decay pattern, characterized by substantial retention near the column inlet (0–4 cm) followed by a sharp decline and relatively low retention in deeper sections, and the hyper-exponential RPs were more pronounced at low HA concentrations. For instance, at 1 mg L−1 HA, approximately 50.08% of the total retained PS-MPs were found in the first 4 cm of the column, while only 49.91% was retained beyond 8 cm (Table 2). This is similar to the results of some previous studies.38
The observed hyper-exponential retention profiles can be attributed to the combined effects of physical straining, pore throat trapping and chemical heterogeneity.66 The greater retention near the column inlet compared to the outlet is not simply due to katt1 > katt2, but rather reflects the spatial distribution of S2 retention mechanisms.44,45,67 Physical straining and pore throat trapping occur preferentially near the inlet because: (i) the probability of encountering pore constrictions is highest at the column entrance, and (ii) once particles are captured, they are removed from the mobile phase and cannot travel further. This spatial bias, combined with the high katt1 values, produces the characteristic hyper-exponential retention profiles. In contrast, chemical adsorption (S2) can occur throughout the column wherever reactive surface sites are accessible, contributing to the low but non-zero retention observed in deeper sections.68
3.3. Effect of IS on transport and retention of PS-MPs
Under HA conditions, the effects of IS on the transport and retention behaviors of PS-MPs in bare sandy column were illustrated in Fig. 6. The transportability of PS-MPs in the saturated sandy column decreased as the IS increased. Specifically, as shown in the Fig. 7A, the transport velocities of PS-MPs decreased with increased IS, resulting in delayed and broadened BTCs. As lowest IS (0.1 mM) presence in HA solution, C/C0 and outflow rates of PS-MPs were as high as 0.799 and 84.51%, respectively, while they decreased to 0.369 and 38.70% in solution with 100 mM (Table 2). An increase in the α values of PS-MPs are observed as the solution IS increased, especially at high solution IS (Table 2). This indicated that increased IS not only slows transport rates but also reduces the transport capacity of PS-MPs. This is consistent with the transport trend of other nanoparticles (such as nHAP) under different IS with HA conditions.38 | ||
| Fig. 7 Measured and fitted breakthrough curve (A) and retention profiles (B) for PS-MPs under different IS at pH 6.0 in the presence of 10 mg L−1 HA. | ||
Under the tested IS conditions, the zeta potentials of PS-MPs, and quartz sand were negative (Table 1). And the zeta potentials of them became less negative as the IS increased, mainly due to the compressed double layer and the shielding charge effect of Na+.69 For example, when IS increased from 0.01 mM to 100 mM, the zeta potential of PS-MPs and quartz sand changed from −61.84 mV and −30.85 mV to −31.09 mV and −22.64 mV, separately. Therefore, the increase in IS resulted in reduced electrostatic repulsion forces between PS-MPs with quartz sand (Fig. 4), leading to increased deposition in the sandy columns.64,70–72 Additionally, the weakening of electrostatic repulsion between PS-MPs leads to their self-agglomeration, resulting in an increase in particle size64,73 (Fig. 6). These large particles are prone to physical trapping in the pore structure of quartz sand.45,74 However, HA enhanced the transport ability of MPs through steric hindrance and electrostatic repulsion,63,75 but this effects weakened by the ion shielding effect under high IS conditions.76,77 High concentrations of ions compress the molecular structure of HA, reducing its steric hindrance ability, while also promoting the co-agglomeration of HA and PS-MPs, further exacerbating particle deposition.78,79
Under the presence of HA, the RPs of PS-MPs in all IS treatment groups exhibited hyper-exponential decay characteristics, meaning that the retention concentration decreased rapidly and non-linearly with distance. Retention levels at the inlet end (0–4 cm) were significantly higher than in the latter half of the column (Fig. 6B). Notably, the hyper-exponential decay characteristic intensified with increased IS. Under low IS conditions (0.1 mM NaCl), the retention curve is relatively flat, with retention levels as high as 9.78% at the inlet end (0–4 cm). When the IS increased to 100 mM, the hyper-exponential decay characteristic was most pronounced, with retention levels as high as 28.07% at the inlet end (0–4 cm) (Table 2).
Under HA conditions, TKSAD model provided excellent fits for experimental data across all IS treatment groups (R2 > 0.997, Table 3). Model parameters revealed a significant regulatory effect of IS on the retention mechanism of PS-MPs. As the IS increased from 0.1 mM to 100 mM, the attachment rate katt1 at S1 monotonically increased from 0.361 min−1 to 0.546 min−1, representing an approximate 51% increase. Concurrently, the maximum retention capacity Smax1/C0 at S1 substantially increased from 0.74 to 2.78 g−1, representing a 2.8-fold increase (Table 3). This indicates that high IS not only accelerates the kinetic rate of chemical adsorption, more importantly, creates additional physical retention sites. This may be attributed to MPs agglomeration caused by HA conformation collapse and the formation of uneven organic coatings on the surface of the medium by HA. Notably, the adsorption rate constant katt2 at S2 exhibits a more pronounced response to IS: it increases sharply from 0.00328 min−1 at 0.1 mM to 0.102 min−1 at 100 mM, representing a 30-fold increase (Table 3). This indicates that high IS significantly promotes physical straining and pore throat trapping of PS-MPs by compressing the double electric layer and shielding electrostatic repulsion. The desorption rates kdet1 and kdet2 at both sites approached zero under all conditions (kdet1 ≈ 10−6 to 10−7 min−1, kdet2 ≈ 0), confirming that the retention of PS-MPs in the presence of HA is essentially irreversible. Neither physical trapping nor chemical adsorption facilitates desorption.
| HA (mg L−1) | IS (mM) | ω | pH | katt1a (min−1) | kdet1b (min−1) | katt2c (min−1) | kdet2d (min−1) | Smax1/C0e (S/C0) (g−1) | R2f |
|---|---|---|---|---|---|---|---|---|---|
| a First-order retention coefficient on S1.b First-order detachment coefficient on S1.c First-order retention coefficient on S2.d First-order detachment coefficient on S2.e Maximum solid phase concentration of PS-MPs on S1 normalized by the input PS-MPs concentration.f Squared Person's correlation coefficient. | |||||||||
| 0 | 0.1 | 0 | 6.0 | 4.98 × 10−1 | 1.08 × 10−5 | 7.99 × 10−2 | 1.54 × 10−8 | 2.09 | 0.999 |
| 1 | 0.1 | 0 | 6.0 | 4.88 × 10−1 | 1.66 × 10−6 | 7.18 × 10−2 | 5.07 × 10−8 | 2.05 | 0.997 |
| 5 | 0.1 | 0 | 6.0 | 4.62 × 10−1 | 9.25 × 10−7 | 5.50 × 10−2 | 6.27 × 10−8 | 1.68 | 0.995 |
| 10 | 0.1 | 0 | 6.0 | 3.61 × 10−1 | 5.32 × 10−7 | 3.28 × 10−3 | 6.51 × 10−8 | 0.74 | 0.998 |
| 10 | 1 | 0 | 6.0 | 4.59 × 10−1 | 8.10 × 10−7 | 5.61 × 10−3 | 6.46 × 10−8 | 0.98 | 0.997 |
| 10 | 10 | 0 | 6.0 | 4.93 × 10−1 | 8.16 × 10−7 | 7.11 × 10−2 | 1.93 × 10−8 | 1.92 | 0.997 |
| 10 | 100 | 0 | 6.0 | 5.46 × 10−1 | 1.01 × 10−6 | 1.02 × 10−1 | 1.36 × 10−8 | 2.78 | 1 |
| 10 | 0.1 | 0.15 | 6.0 | 3.72 × 10−1 | 1.90 × 10−6 | 5.45 × 10−2 | 9.08 × 10−9 | 1.42 | 1 |
| 10 | 0.1 | 0.30 | 6.0 | 3.88 × 10−1 | 1.95 × 10−6 | 6.10 × 10−2 | 8.54 × 10−9 | 1.83 | 1 |
| 10 | 0.1 | 0.45 | 6.0 | 4.65 × 10−1 | 6.40 × 10−6 | 7.36 × 10−2 | 7.78 × 10−9 | 2.25 | 1 |
| 10 | 0.1 | 0.60 | 6.0 | 5.59 × 10−1 | 9.36 × 10−6 | 9.94 × 10−2 | 7.40 × 10−9 | 2.50 | 1 |
| 10 | 0.1 | 0.75 | 6.0 | 5.85 × 10−1 | 1.07 × 10−5 | 9.98 × 10−2 | 6.66 × 10−9 | 2.54 | 0.999 |
| 10 | 0.1 | 0.15 | 8.0 | 2.44 × 10−1 | 1.01 × 10−6 | 3.33 × 10−2 | 9.46 × 10−8 | 0.50 | 0.999 |
| 10 | 0.1 | 0.15 | 10.0 | 2.40 × 10−1 | 8.14 × 10−7 | 2.61 × 10−2 | 9.56 × 10−8 | 0.41 | 0.994 |
3.4. Effect of iron oxyhydroxides coatings of quartz sand on transport and retention of PS-MPs
The BTCs and RPs of PS-MPs in the presence of HA under different λ conditions are shown in the Fig. 8. The transportability of PS-MPs in the saturated porous media decreased as the λ increased. For example, as λ increased, the time required for PS-MPs to exit the column increased, and the C/C0 decreases. When λ is 0 (There is no iron oxyhydroxides coated quartz sand inside the column), the C/C0 reached 0.796, while when λ reaches 0.75, the C/C0 decreased to 0.391. An increased in the α values of PS-MPs were observed as the λ increased, especially at high λ (Table 2). Correspondingly, the mass recoveries of PS-MPs in the effluent decreased from 84.51 to 40.99% (Table 2), this trend aligns with observed behaviors in the transport of MPs within saturated sandy columns.60 This may be attributed to the formation of nano- to micrometer scale iron oxide particle aggregates on the quartz sand surface by the hydroxy iron oxide coating, which significantly increases the micro-roughness and surface area of the medium.80,81 The direct consequences of this coarse structure are: (1) at the pore scale, hydroxy iron oxide aggregates may partially block pore throats or create more tortuous flow paths, enhancing physical straining of PS-MPs; (2) at the particle scale, the rough surface increases contact points and friction coefficients between PS-MPs and the medium, making it harder for particles that come into contact to be resuspended by water flow. This explains why katt2 increase synchronously with increasing ω (Table 3), more hydroxy iron oxides mean more “physical barrier points,” and these barrier points exhibit higher capture efficiency. Contrast to the untreated sandy column, the transport behaviors of PS-MPs were weakened in hydroxy iron oxide coated quartz sand, regardless of the presence of HA (Table 2). This might be due to the fact that the transport of PS-MPs in sand with iron oxyhydroxides was not sensitive to HA.60 In addition, The![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Fe–OH groups provided by the hydroxy iron oxide coatings serve as excellent chemical adsorption sites. In the presence of HA, these sites may interact with PS-MPs through multiple pathways: Firstly, cations (Na+) in the solution form bridges between negatively charged PS-MPs and negatively charged
Fe–OH groups provided by the hydroxy iron oxide coatings serve as excellent chemical adsorption sites. In the presence of HA, these sites may interact with PS-MPs through multiple pathways: Firstly, cations (Na+) in the solution form bridges between negatively charged PS-MPs and negatively charged ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Fe–O−, causing the PS-MPs to become immobilized within the column.82 Secondly, certain areas of the iron oxide coating may provide hydrophobic sites.37 More importantly, the iron oxyhydroxides coated quartz sand surface exhibits a strong positive charge (12.79 mV) at pH 6.0 (Table 1), and the isoelectric point (pHPZC) of iron oxyhydroxide is 8.4.38 This may lead to PS-MPs strongly and irreversibly adsorb to the surface of the iron oxyhydroxides coated quartz sand through electrostatic adsorption. And the more quartz sands were coated with iron oxyhydroxides, the more PS-MPs (or PS-MPs-HA) will be adsorbed on its surface. In other words, the attachment is directly proportional to λ. Similarly, the time required for PS-MPs to occupy the most favorable attachment sites also increases with the increase of λ. Only after the sites related to iron oxyhydroxides are occupied can substantial penetration begin, which is consistent with the results found in previous related studies.38 The strength of these chemical interactions far exceeds that of pure DLVO electrostatic forces, leading to a significant increase in katt1 and rendering adsorption irreversible once it occurs. For instance, compared with the experimental group where only HA concentration was varied, the total mass balance of PS-MPs gradually deteriorated with the increase of λ (102.33 to 93.49%). When λ is relatively large, the RPs cannot fully reflect the quality of PS-MPs in the sand. This might be because the PS-MPs retained on the iron oxyhydroxide coated quartz sand were not completely removed during the extraction process.
Fe–O−, causing the PS-MPs to become immobilized within the column.82 Secondly, certain areas of the iron oxide coating may provide hydrophobic sites.37 More importantly, the iron oxyhydroxides coated quartz sand surface exhibits a strong positive charge (12.79 mV) at pH 6.0 (Table 1), and the isoelectric point (pHPZC) of iron oxyhydroxide is 8.4.38 This may lead to PS-MPs strongly and irreversibly adsorb to the surface of the iron oxyhydroxides coated quartz sand through electrostatic adsorption. And the more quartz sands were coated with iron oxyhydroxides, the more PS-MPs (or PS-MPs-HA) will be adsorbed on its surface. In other words, the attachment is directly proportional to λ. Similarly, the time required for PS-MPs to occupy the most favorable attachment sites also increases with the increase of λ. Only after the sites related to iron oxyhydroxides are occupied can substantial penetration begin, which is consistent with the results found in previous related studies.38 The strength of these chemical interactions far exceeds that of pure DLVO electrostatic forces, leading to a significant increase in katt1 and rendering adsorption irreversible once it occurs. For instance, compared with the experimental group where only HA concentration was varied, the total mass balance of PS-MPs gradually deteriorated with the increase of λ (102.33 to 93.49%). When λ is relatively large, the RPs cannot fully reflect the quality of PS-MPs in the sand. This might be because the PS-MPs retained on the iron oxyhydroxide coated quartz sand were not completely removed during the extraction process.
 | ||
| Fig. 8 Measured and fitted breakthrough curve (A) and retention profile (B) for PS-MPs under different λ at pH 6.0 in the presence of 10 mg L−1 HA. | ||
However, the retention of PS-MPs increased with the increases of λ, and the hyper-exponential shape of the RPs (Fig. 8B) is also more obvious. When λ increased from 0 to 0.75, the retention rates of PS-MPs increased about three times (from 17.82 to 52.49%), and these results indicate that RPs provide useful information on the quantitative aspects of PS-MPs.
3.5. Effect of pH on transport and retention of PS-MPs
The regulatory effect of pH on PS-MPs transport and retention behavior under specific conditions of HA concentration (10 mg L−1), IS (0.1 mM), and hydroxy iron oxide coating content (ω = 0.15) is shown in the Fig. 9. As shown in the figure, the transport and retention behaviors of PS-MPs were significantly affected by the solution pH presence of HA and IS. When the solution pH increases from 6.0 to 10.0, the BTCs shifts to the left, indicating faster transport rates of PS-MPs in the sandy column. At the same time, the C/C0 of BTCs increases as the solution pH increases, indicating that the weakly alkaline solution is favorable for the transport of PS-MPs in porous media in this study. | ||
| Fig. 9 Measured and fitted breakthrough curve (A) and retention profile (B) for PS-MPs under different pH condition at λ = 0.15 in the presence of 10 mg L−1 HA. | ||
The significant regulatory effect of pH on PS-MPs retention can be thoroughly explained from the perspectives of DLVO theory and surface chemistry. The isoelectric point of PS-MPs typically falls between pH 2.0 and 4.0.37,82 Within the pH range of this study (6.0–10.0), the MPs surface carries a strong negative charge.37,82 The iron oxyhydroxide coatings exhibits a higher isoelectric point (approximately pH 7–9).83 At pH 6.0, it carries a positive or weakly negative charge. At pH 8.0–10.0, it becomes negatively charged due to deprotonation of the ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Fe–OH group (
Fe–OH group (![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Fe–OH →
Fe–OH → ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Fe–O− + H+). Therefore, as the pH increases from 6.0 to 10.0, the electrostatic interactions between PS-MPs and the surface of the medium undergo a fundamental shift. At pH 6.0, electrostatic attraction or weak repulsion exists between PS-MPs (negatively charged) and iron oxyhydroxide coatings (positively or weakly negatively charged), resulting in a low DLVO energy barrier. Particles readily approach the surface and become retained. At pH 8.0–10.0, strong electrostatic repulsion exists between PS-MPs (strongly negatively charged) and iron oxyhydroxide coatings (negatively charged), significantly increasing the DLVO energy barrier. Particles struggle to overcome this barrier and approach the surface.84 This increase in electrostatic repulsion directly leads to a significant decrease in katt1 – chemical adsorption requires particles to overcome an energy barrier to contact active sites on the surface, and an elevated barrier reduces the adsorption rate. In addition, the hydrodynamic sizes of PS-MPs shrink with increasing pH.64 Both the deposition and size exclusion straining are weakened during the transport of PS-MPs under this condition, and it is reasonable for MPs to transport easily through saturated porous media.
Fe–O− + H+). Therefore, as the pH increases from 6.0 to 10.0, the electrostatic interactions between PS-MPs and the surface of the medium undergo a fundamental shift. At pH 6.0, electrostatic attraction or weak repulsion exists between PS-MPs (negatively charged) and iron oxyhydroxide coatings (positively or weakly negatively charged), resulting in a low DLVO energy barrier. Particles readily approach the surface and become retained. At pH 8.0–10.0, strong electrostatic repulsion exists between PS-MPs (strongly negatively charged) and iron oxyhydroxide coatings (negatively charged), significantly increasing the DLVO energy barrier. Particles struggle to overcome this barrier and approach the surface.84 This increase in electrostatic repulsion directly leads to a significant decrease in katt1 – chemical adsorption requires particles to overcome an energy barrier to contact active sites on the surface, and an elevated barrier reduces the adsorption rate. In addition, the hydrodynamic sizes of PS-MPs shrink with increasing pH.64 Both the deposition and size exclusion straining are weakened during the transport of PS-MPs under this condition, and it is reasonable for MPs to transport easily through saturated porous media.
Increased pH also affects the charge state and conformation of HA. HA contains numerous carboxyl and phenolic hydroxyl groups with pKa values around 3–5 (primarily involving the dissociation of carboxyl groups),85,86 which are fully deprotonated within the pH range of this study, carrying a strong negative charge. As pH rises, electrostatic repulsion within HA molecules intensifies, causing the molecular conformation to shift from coiled to extended. At pH 6.0, HA molecules adopt a relatively curled conformation, potentially partially masking the PS-MPs surface. At pH 8.0–10.0, HA molecules extend, forming a thicker steric hindrance layer that further enhances isolation between PS-MPs and the medium surface. This likely explains the significant decrease in Smax1 – the steric hindrance layer formed by the extended HA conformation “shields” many available retention sites.
It is worth noting that when the pH value of the solution changes from 6.0 to 10.0, the value of katt2 decreases from 0.00545 to 0.00261 min−1, with a change range of 52.11%. This disparity reveals the primary pathway through which pH affects physical retention not by reducing the efficiency of physical capture, but by diminishing the number of available sites for physical capture.87 The steric barrier layer formed by the extended HA conformation may cover physical capture sites on the originally rough surface (such as pore throats and shadow zones), rendering these sites “inactive.”88 Simultaneously, enhanced electrostatic repulsion makes it difficult for PS-MPs to enter certain narrow pores, further reducing opportunities for effective physical capture.
Consistent with the trend in model parameters, the RPs exhibits a weakening hyper-exponential decay characteristic as pH increases (Fig. 9B). At pH 6.0, the RPs displays a typical hyper-exponential decay pattern: a high retention ratio at the inlet end with rapid decay. As pH rises to 8.0 and 10.0, the entire retention curve shifts significantly downward, the retention ratio at the inlet end decreases substantially, and the curve shape becomes flatter.
This morphological evolution can be perfectly explained by the model parameters: at pH 6.0, Smax1/C0 reaches 1.42 g−1, indicating a large number of chemical adsorption sites can efficiently capture PS-MPs; katt1 is 0.0545 min−1, suggesting a significant contribution from chemical adsorption along the flow path. The combined effect of both factors produces a hyper-exponential curve characterized by high retention at the inlet and sustained capture along the flow path. When pH increased to 8.0 and 10.0, Smax1/C0 sharply decreased to 0.50 and 0.41 g−1, indicating a significant reduction in the number of chemical adsorption sites. Concurrently, katt2 decreased to 0.0333 and 0.0261 min−1, diminishing the contribution of physical retention and a steep decline in capture efficiency at the inlet. Consequently, the entire retention curve shifted markedly downward, and the hyper-exponential characteristics weakened.
4. Conclusion
This study addresses the identified research gap by demonstrating that: (i) PS-MPs retention in saturated porous media involves two mechanistically distinct processes – chemical adsorption and physical retention, that respond differently to environmental factors; (ii) the TKSAD model, even with S2 exhibiting negligible desorption, is necessary to capture these distinct mechanisms and their differential environmental responses; and (iii) the model parameters provide quantitative mechanistic insights that cannot be obtained from one kinetic site models or from breakthrough curves alone. These findings advance beyond previous studies that treated MPs retention as a single process or employed one kinetic site models that cannot distinguish between physical and chemical mechanisms.5. Environmental implications
These findings have important implications for predicting the fate of MPs in natural aquatic and soil environments. In freshwater systems with low IS and circumneutral pH, the presence of NOM may facilitate MPs transport over long distances. In contrast, in seawater – intruded coastal aquifers or saline – alkali soils characterized by high IS, MPs are likely to be retained near the source zone, particularly in the presence of metal oxide minerals. The strong pH dependence suggests that acid rain events or alkaline pollution incidents could dramatically alter MPs mobility. Furthermore, the irreversible nature of retention (kdet ≈ 0) implies that once deposited, MPs may persist in the subsurface even under changing geochemical conditions, posing long-term ecological risks. Future studies should consider the coupled effects of multiple environmental factors and extend these findings to more complex natural porous media.Author contributions
Abliz Abdurahman and Xiaoli Ma are the two co-first authors of this paper. Abliz Abdurahman: investigation, methodology, software, data curation, formal analysis, writing – original draft. Rui Gao: visualization, investigation, data curation. Xiaoli Ma: conceptualization, methodology, writing – review & editing. All authors have given approval to the final version of the manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting the findings of this study are included within the article and its supplementary information (SI) files hosted by the Royal Society of Chemistry. Supplementary information: experimental methods, additional supporting figures, tables, raw data summaries, and statistical analyses that further support the main findings of this study. See DOI: https://doi.org/10.1039/d5ra08025h.Acknowledgements
This study was supported by the Research Fund for High-Level Talent of Xinjiang Medical University.References
- Y. Seo, V. Chevali, Y. Lai, Z. Zhou, G. Chen, P. Burey, S. Wang and P. Song, Environ. Manage., 2025, 377, 124556–124567 CAS.
- I. M. Jaikumar, M. Tomson, A. Meyyazhagan, B. Balamuralikrishnan, R. Baskaran, M. Pappuswamy, H. Kamyab, E. Khalili and M. Farajnezhad, Green Anal. Chem., 2025, 12, 100202 CrossRef.
- C. Geng, Y. Gao, H. Zhang, D. Xue, S. He, B. Wang, X. Wang and J. Zhao, Environ. Chem. Lett., 2024, 22, 691–713 CrossRef CAS.
- W. Li, G. Brunetti, A. Bolshakova and C. Stumpp, Sci. Total Environ., 2024, 935, 11 Search PubMed.
- Z. Jia, W. Wei, Y. Wang, Y. Chang, R. Lei and Y. Che, Rev. Environ. Contam. Toxicol., 2022, 260, 1–18 Search PubMed.
- H. Wang, J. Zhang, Y. Chen, Y. Xia, P. Jian and H. Liang, Front. Mar. Sci., 2024, 11, 1346275 CrossRef.
- T. Mutshekwa, F. Mulaudzi, V. P. Maiyana, L. Mofu, L. F. Munyai and F. M. Murungweni, PLoS One, 2025, 20, 313840–313854 CrossRef.
- Y. Ankit, K. Ajay, S. Nischal, S. Kaushal, V. Kataria, E. Dietze and A. Anoop, Environ. Pollut., 2024, 361, 124629 CrossRef CAS PubMed.
- Y. Dahal and S. Babel, Air Qual. Atmos. Health, 2025, 18, 425–445 CrossRef CAS.
- M.-P. Albert and J. Joaquim, Toxics, 2020, 8, 40 CrossRef PubMed.
- T. Wang, D. Liu, R. Liu, F. Yuan, Y. Ding, J. Tao, Y. Wang, W. Yu, Y. Fang and B. Li, Environ. Sci. Technol., 2025, 59, 7667–7677 CrossRef CAS.
- S. Kühn, J. A. v. Franeker, A. M. O'Donoghue, A. Swiers, M. Starkenburg, B. v. Werven, E. Foekema, E. Hermsen, M. Egelkraut-Holtus and H. Lindeboom, Environ. Pollut., 2019, 257, 113569 CrossRef PubMed.
- S. Paul, S. Nath, S. Bhattacharjee and S. Mukherjee, Blue Biotechnol., 2024, 1, 1–8 Search PubMed.
- L. G. A. Barboza, L. R. Vieira, V. Branco, C. Carvalho and L. Guilhermino, Sci. Rep., 2018, 8, 15655 CrossRef.
- Y. Zhang, D. Li, K. Hui, H. Wang, Y. Yuan, F. Fang, Y. Jiang, B. Xi and W. Tan, J. Environ. Chem. Eng., 2025, 13, 115806 CrossRef CAS.
- A. Huvet, L. Frère, C. Lacroix, E. Rinnert, C. Lambert and I. Paul-Pont, Reg. Stud. Mar. Sci., 2025, 89, 104279 Search PubMed.
- S. Emami, S. Bagheri and A. Gholamhosseini, Environ. Sci. Pollut. Res., 2025, 32, 26641–26651 CrossRef CAS PubMed.
- S. Oberbeckmann and M. Labrenz, Ann. Rev. Mar. Sci, 2020, 12, 209–232 CrossRef.
- N. Zhu, Z. Li, Y. Yu, Z. Liu, X. Liang, W. Wang and J. Zhao, Ecotoxicol. Environ. Saf., 2025, 295, 118138 CrossRef CAS PubMed.
- Y. Wang, L. Xu, H. Chen and M. Zhang, Sci. Total Environ., 2022, 808, 152154 CrossRef CAS.
- L. Zeng, C. Yuan, T. Xiang, X. Guan, L. Dai, D. Xu, D. Yang, L. Li and C. Tian, Nanomaterials, 2024, 14, 1060 CrossRef CAS PubMed.
- Y. Wei, Y. Chen, X. Cao, T.-C. J. Yeh, J. Zhang, Z. Zhan, Y. Cui and H. Li, Environ. Sci. Technol., 2024, 58, 12 Search PubMed.
- F. Li, D. Huang, G. Wang, M. Cheng, H. Chen, W. Zhou, R. Xiao, R. Li, L. Du and W. Xu, Sci. Total Environ., 2024, 926, 171658 CrossRef CAS PubMed.
- M. Li, L. He, X. Zhang, H. Rong and M. Tong, Environ. Pollut., 2020, 267, 115534 CrossRef CAS PubMed.
- G. Hul, H. Okutan, P. L. Coustumer, S. R. Gentile, S. Zimmermann, P. Ramaciotti, P. Perdaems and S. Stoll, Nanomaterials, 2024, 16, 529 CrossRef PubMed.
- Y. Jiang, S. Zhou, J. Fei, Z. Qin, X. Yin, H. Sun and Y. Sun, Water Res., 2022, 215, 118262 CrossRef CAS PubMed.
- M. N. Singer, B. Mohsen, V. E. Katzourakis, M. R. A. Shehhi and C. V. Chrysikopoulos, J. Environ. Chem. Eng., 2025, 13, 118002 CrossRef CAS.
- X. Zhang, L. Wu, X. Han, Y. Shi, J. Huang, B. Ding, Y. Zhang, Z. Zhang, Y. Shi and F. Li, Chemosphere, 2025, 370, 143942–143951 CrossRef CAS PubMed.
- D. Zhou, Y. Cai and Z. Yang, Environ. Pollut., 2022, 293, 118503 CrossRef CAS PubMed.
- M. Tan, L. Feng, S. Bian, J. Duan, X. Li, X. Sun, Y. Sun, S. Wang and X. Yuan, ACS ES&T Eng., 2024, 4, 1230–1239 Search PubMed.
- X. Wang, Y. Diao, Y. Dan, F. Liu, H. Wang, W. Sang and Y. Zhang, Chemosphere, 2022, 309, 136658 CrossRef CAS PubMed.
- X. Wang, Y. Dan, Y. Diao, F. Liu, H. Wang, W. Sang and Y. Zhang, Sci. Total Environ., 2022, 847, 157576 CrossRef CAS PubMed.
- Y. Hao, F. Zhu, H. Cheng, S. Komarneni and J. Ma, J. Phys. Chem. Solid., 2024, 185, 111787 CrossRef CAS.
- S. Wang, R. Walker-Gibbons, B. Watkins, M. Flynn and M. Krishnan, Nat. Nanotechnol., 2024, 19, 485–493 CrossRef CAS PubMed.
- F. Feng, W. Ye, S. Xiang, X. Fan, X. Liu, H. Liu and W. Zhang, Front. Environ. Sci. Eng., 2025, 19, 142 CrossRef.
- H. Luo, L. Chang, T. Ju and Y. Li, ACS Omega, 2024, 9, 50064–50077 CrossRef CAS PubMed.
- M. Tan, L. Liu, M. Zhang, Y. Liu and C. Li, J. Hazard Mater., 2021, 413, 125410 CrossRef CAS PubMed.
- D. Wang, S. A. Bradford, R. W. Harvey, B. Gao, L. Cang and D. Zhou, Environ. Sci. Technol., 2012, 46, 2738–2745 CrossRef CAS PubMed.
- R. S. Stahl and B. R. James, Soil Sci. Soc. Am. J., 1991, 55, 1291–1294 CrossRef CAS.
- D. Wang, S. A. Bradford, M. Paradelo, W. J. G. M. Peijnenburg and D. Zhou, Soil Sci. Soc. Am. J., 2012, 76, 375–388 CrossRef CAS.
- D. P. Jaisi, N. B. Saleh, R. E. Blake and M. Elimelech, Environ. Sci. Technol., 2008, 42, 8317 CrossRef CAS PubMed.
- Y. Wang, L. Xu, H. Chen and M. Zhang, Environ. Sci. Technol., 2022, 808, 152154 CAS.
- Q. Ma, Y. Sun, S. Zhou, X. Yin and H. Sun, Environ. Manag., 2025, 377, 124696 CAS.
- H. Xu, Y. Zhang, W. Zhang, Y. Tang, Y. Zhou, P. Tang and T. Zhang, Phys. Fluids, 2024, 36, 053301 CrossRef.
- X. Chu, T. Li, Z. Li, A. Yan and C. Shen, Water, 2019, 11, 2474 CrossRef CAS.
- L. Zeng, C. Yuan, T. Xiang, X. Guan, L. Dai, D. Xu, D. Yang, L. Li and C. Tian, Nanomaterials, 2024, 4, 1060 CrossRef.
- M. N. Singer, B. Mohsen, V. E. Katzourakis, M. R. A. Shehhi and C. V. Chrysikopoulos, J. Environ. Chem. Eng., 2025, 13, 118002 CrossRef CAS.
- J. F. Schijven and J. Simunek, J. Contam. Hydrol., 2002, 55, 113–135 CrossRef CAS PubMed.
- S. A. Bradford, J. Simunek, M. Bettahar, M. T. V. Genuchten and S. R. Yates, Environ. Sci. Technol., 2003, 37, 2242 CrossRef CAS.
- P. Babakhani, J. Bridge, R.-A. Doong and T. Phenrat, Adv. Colloid Interface Sci., 2017, 246, 75–104 CrossRef CAS PubMed.
- F. J. Leij, S. A. Bradford and A. Sciortino, J. Contam. Hydrol., 2016, 195, 40–51 CrossRef CAS PubMed.
- N. H. Pham and D. V. Papavassiliou, Comput. Part. Mech., 2017, 4, 87–100 CrossRef.
- Y. F. Adrian, U. Schneidewind, S. A. Bradford, J. Simunek, T. M. Fernandez-Steeger and R. Azzam, Environ. Pollut., 2018, 236, 195–207 CrossRef CAS PubMed.
- J. Šimůnek, M. Th, v. Genuchten and M. Šejna, Vadose Zone J., 2016, 15, 1–25 CrossRef.
- D. W. Marquardt, SIAM J. Appl. Math., 1963, 11, 431 CrossRef.
- K. K. Norrfors, V. Micić, O. Borovinskaya, F. v. d. Kammer, T. Hofmann and G. Cornelis, Environ. Sci. Nano, 2021, 8, 1801 RSC.
- N. Tufenkji and M. Elimelech, Environ. Sci. Technol., 2004, 38, 529 CrossRef CAS PubMed.
- M. Wang, T. Lu, W. Chen, H. Zhang, W. Qi, Y. Song and Z. Qi, Colloids Surf., A, 2020, 607, 125486 CrossRef CAS.
- Z. Zhang, C. Wang, L. You, H. Chen, J. Dong, Q. Chen, Z. Wei, L. Zhang, M. Zhang and Z. Liu, J. Hazard Mater., 2025, 499, 140260 CrossRef CAS PubMed.
- D. Zhou, Y. Cai and Z. Yang, Sci. Total Environ., 2024, 946, 174270–174278 CrossRef CAS PubMed.
- I. Ioannidis, E. Antoniou and I. Pashalidis, J. Environ. Chem. Eng., 2025, 13, 115064 CrossRef CAS.
- L. Cai, L. Hu, H. Shi, J. Ye, Y. Zhang and H. Kim, Chemosphere, 2018, 197, 142–151 CrossRef CAS PubMed.
- S. Dong, W. Cai, J. Xia, L. Sheng, W. Wang and H. Liu, Environ. Pollut., 2021, 268, 115828 CrossRef CAS PubMed.
- S. Li, H. Liu, R. Gao, A. Abdurahman, J. Dai and F. Zeng, Environ. Pollut., 2018, 237, 126–132 CrossRef CAS PubMed.
- X. Lu, L. Wang, J. Li, W. Li, R. Yan, X. Duan and Y. Tang, Water Res., 2025, 281, 123661 CrossRef CAS PubMed.
- A. Elrahmani, R. I. Al-Raoush, J. A. Hannun, M. T. Albaba and T. D. Seers, Sci. Total Environ., 2025, 974, 179238 CrossRef CAS PubMed.
- J. Wang, Y. Gao, L. Ren, H. Zhao, T. Long, Y. Li, C. Tsakiroglou, O. Alfarisi and X. Wang, Chem. Eng. J., 2025, 520, 166099 CrossRef CAS.
- X. Y. Xu, Y. Ji, D. Hu and B. J. Li, Adv. Mater. Res., 2013, 781–784, 2344–2348 CAS.
- W.-K. Ho and K. S.-Y. Leung, Water Res., 2021, 201, 117317 CrossRef CAS PubMed.
- L. Cai, L. L. Hu, H. H. Shi, J. W. Ye, Y. F. Zhang and H. J. Kim, Chemosphere, 2018, 197, 142–151 CrossRef CAS PubMed.
- O. S. Alimi, J. F. Budarz, L. M. Hernandez and N. Tufenkji, Environ. Sci. Technol., 2018, 52, 1704–1724 CrossRef CAS PubMed.
- O. Olena and S. Serge, Environ. Sci. Nano, 2018, 5, 792–799 RSC.
- W. Zhao, Z. Su, T. Geng, Y. Zhao, Y. Tian and P. Zhao, Chemosphere, 2022, 309, 136593 CrossRef CAS PubMed.
- X. Wu, X. Lyu, Z. Li, B. Gao, X. Zeng, J. Wu and Y. Sun, Sci. Total Environ., 2020, 707, 136065 CrossRef CAS PubMed.
- S. Dong, C. Zhuo, Z. Wangwei, D. Zhiyue, L. Chuanbin and J. HanXia, J. Nanopart. Res., 2019, 21, 233 CrossRef CAS.
- H. Zhang, J. Sun and L.-H. Guo, NanoImpact, 2016, 3–4, 75–80 CrossRef.
- C. Peng, C. Shen, S. Zheng, W. Yang, H. Hu, J. Liu and J. Shi, Nanomaterials, 2017, 7, 326 CrossRef PubMed.
- S. A. Mason, D. Garneau, R. Sutton, Y. Chu, K. Ehmann, J. Barnes, P. Fink, D. Papazissimos and D. L. Rogers, Environ. Pollut., 2016, 218, 1045–1054 CrossRef CAS PubMed.
- A. M. Mahon, B. O'Connell, M. G. Healy, O. Connor, R. Officer, R. Nash and L. Morrison, Environ. Sci. Technol., 2017, 51, 810–818 CrossRef CAS PubMed.
- T. Wang, W. Cao, Y. Wang, C. Qu, Y. Xu and H. Li, Ecotoxicol. Environ. Saf., 2023, 262, 115179 CrossRef CAS PubMed.
- G. Li, J. Phys. Conf., 2025, 2941, 012010 CrossRef.
- B. Chang, B. He, G. Cao, Z. Zhou, X. Liu, Y. Yang, C. Xu, F. Hu, J. Lv and W. Du, Sci. Total Environ., 2023, 884, 163832 CrossRef CAS PubMed.
- M. Kosmulski, E. Maczka, E. Jartych and J. B. Rosenholm, Adv. Colloid Interface Sci., 2003, 103, 57–76 CrossRef CAS PubMed.
- J. N. Ryan, M. Elimelech, R. A. Ard, R. W. Harvey and P. R. Johnson, Environ. Sci. Technol., 1999, 33, 63–73 CrossRef CAS.
- G. R. Choppin and L. Kullberg, J. Inorg. Nucl. Chem., 1978, 40, 651–654 CrossRef CAS.
- M. Fukushima, S. Tanaka, H. Nakamura and S. Ito, Talanta, 1996, 43, 383–390 CrossRef CAS PubMed.
- J.-H. Choi, S.-O. Kim, E. Linardy, E. C. Dreaden, V. P. Zhdanov, P. T. Hammond and N.-J. Cho, J. Phys. Chem. B, 2015, 119, 10554 CrossRef CAS PubMed.
- H. Fakour, Y.-F. Pan and T.-F. Lin, Water Air Soil Pollut., 2015, 226, 14 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |